CE / CME
New Data and Clinical Implications in MPNs and MDS: ASH 2025 Highlights
Physician Assistants/Physician Associates: 1.00 AAPA Category 1 CME credit
Pharmacists: 1.00 contact hour (0.1 CEUs)
Physicians: maximum of 1.00 AMA PRA Category 1 Credit™
Nurse Practitioners/Nurses: 1.00 Nursing contact hour
Released: February 26, 2026
Expiration: August 25, 2026
Activity
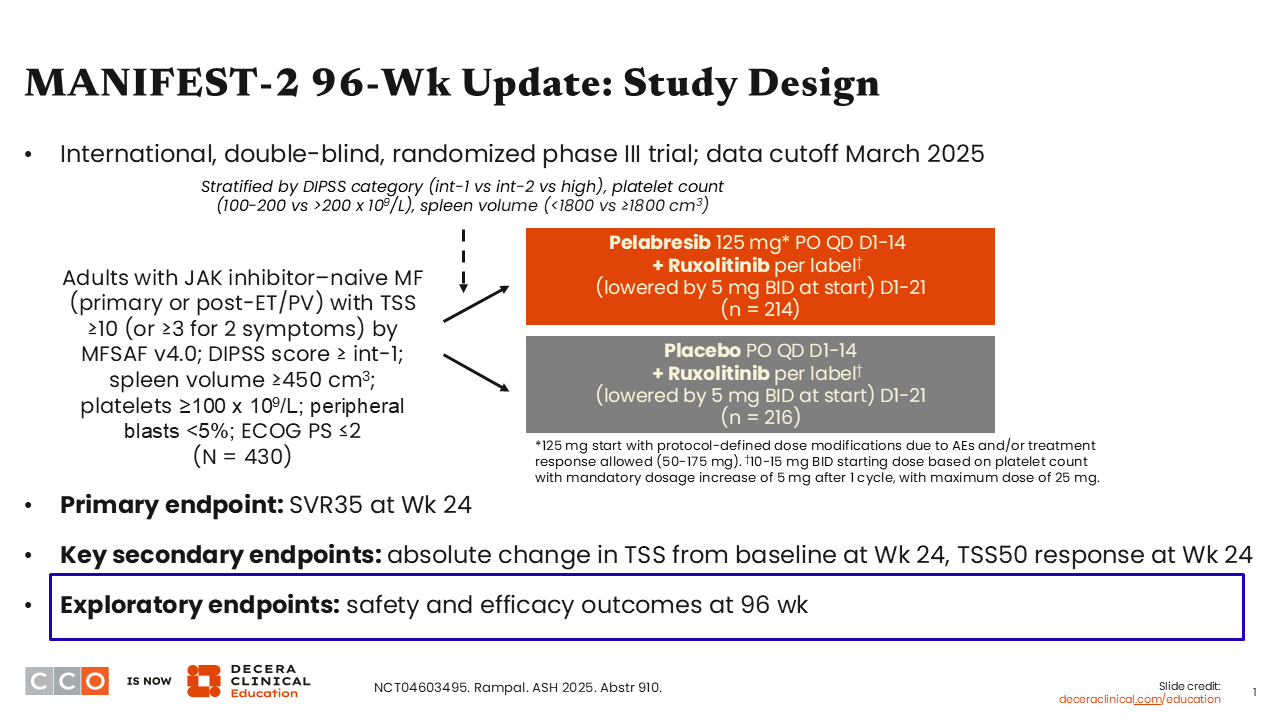
MANIFEST-2 96-Wk Update: Study Design
Rami Komrokji, MD:
The first abstract to highlight is the MANIFEST-2 96-week update. This phase III study enrolled adults with JAK2 inhibitor–naive MF. Patients were randomized to either ruxolitinib alone or ruxolitinib plus the BET inhibitor pelabresib in an effort to improve the outcomes of frontline ruxolitinib therapy.1
Participants were required to have a TSS of ≥10 (or ≥2 symptoms with an average score ≥3), a prognostic risk factor score of intermediate-1 or higher per the Dynamic International Prognostic Scoring System, and splenomegaly (spleen volume ≥450 cm3). Patients were treated with pelabresib for 14 days plus ruxolitinib per label or ruxolitinib per label plus placebo for 14 days. The primary endpoint was SVR35 at Week 24, and key secondary endpoints included the absolute change in TSS from baseline and TSS50.
It is important to note that this is 1 of the largest trials conducted in MF, where more than 400 patients were randomized, and the 2 arms were well balanced and reflective of this population.2
MANIFEST-2 96-Wk Update: Spleen Volume Reduction
Rami Komrokji, MD:
In terms of the primary endpoint, SVR35, as previously reported, at Week 24, we saw superiority, with almost a doubling of the response in the intent-to-treat population: 66% with pelabresib plus ruxolitinib vs 35% with ruxolitinib alone. The response was well maintained at Week 96, with rates of 45% vs 30%, respectively.2
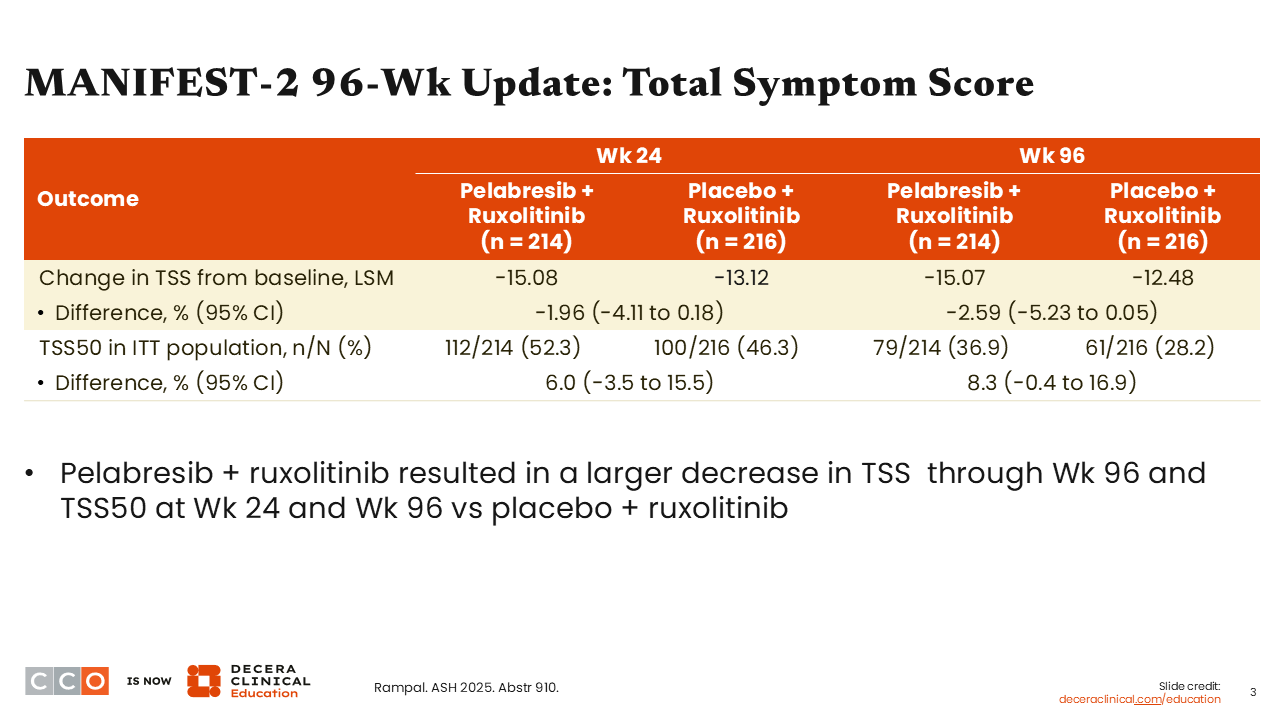
MANIFEST-2 96-Wk Update: Total Symptom Score
Rami Komrokji, MD:
The TSS was improved at Week 24 and not only maintained but was even better at Week 96, decreasing by 15.07 from baseline with pelabresib plus ruxolitinib vs 12.48 with ruxolitinib alone. Of importance, TSS improvement was not statistically significant, likely in part because ruxolitinib already provides strong symptom control, leaving limited room for additional measurable improvement (a ceiling effect) when pelabresib was added. In addition, at Week 96, TSS50 was achieved by approximately 37% and 28% of patients in the pelabresib plus ruxolitinib and ruxolitinib alone arms, respectively.2
MANIFEST-2 96-Wk Update: Dual SVR35 and TSS50 Responders
Rami Komrokji, MD:
When we look at both SVR35 and TSS50, the combination of pelabresib and ruxolitinib resulted in a higher frequency of this dual endpoint At Week 96, twice as many patients achieved this dual endpoint vs those treated with ruxolitinib alone (31.8% vs 15.7%).2
MANIFEST-2 96-Wk Update: Additional Outcomes
Rami Komrokji, MD:
Additional outcomes from the 96-week update, including hemoglobin response, RBC transfusion, and leukemic transformation, are shown here. Pelabresib plus ruxolitinib was associated with a larger decrease in reticulin fiber and megakaryocyte densities as well as consistent reductions in the VAFs of phenotype driver mutations.2
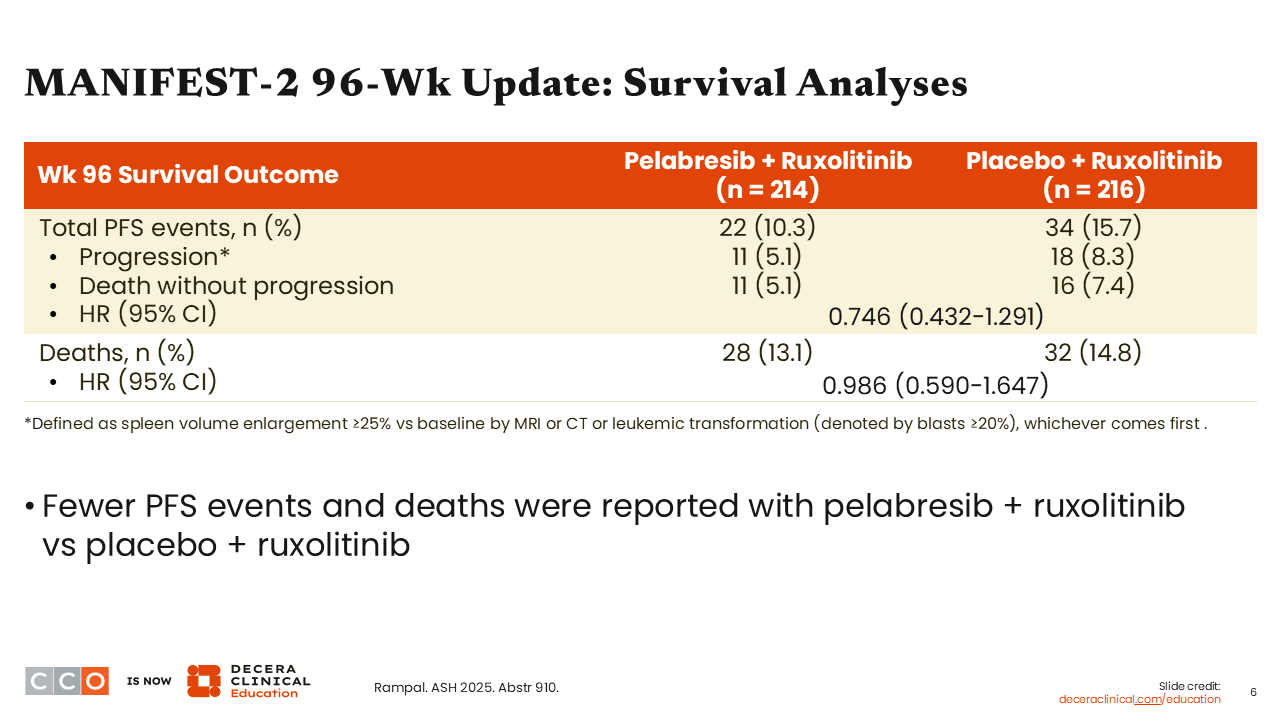
MANIFEST-2 96-Wk Update: Survival Analyses
Rami Komrokji, MD:
When we look at progression-free survival and death, there were fewer events reported with pelabresib plus ruxolitinib vs ruxolitinib alone. Total progression-free survival events were reported in 10.3% of patients in the pelabresib plus ruxolitinib arm vs 15.7% in the ruxolitinib alone arm. At the 96-week update, 13.1% of patients treated with pelabresib plus ruxolitinib had died vs 14.8% treated with ruxolitinib alone, with a hazard ratio of 0.986.2
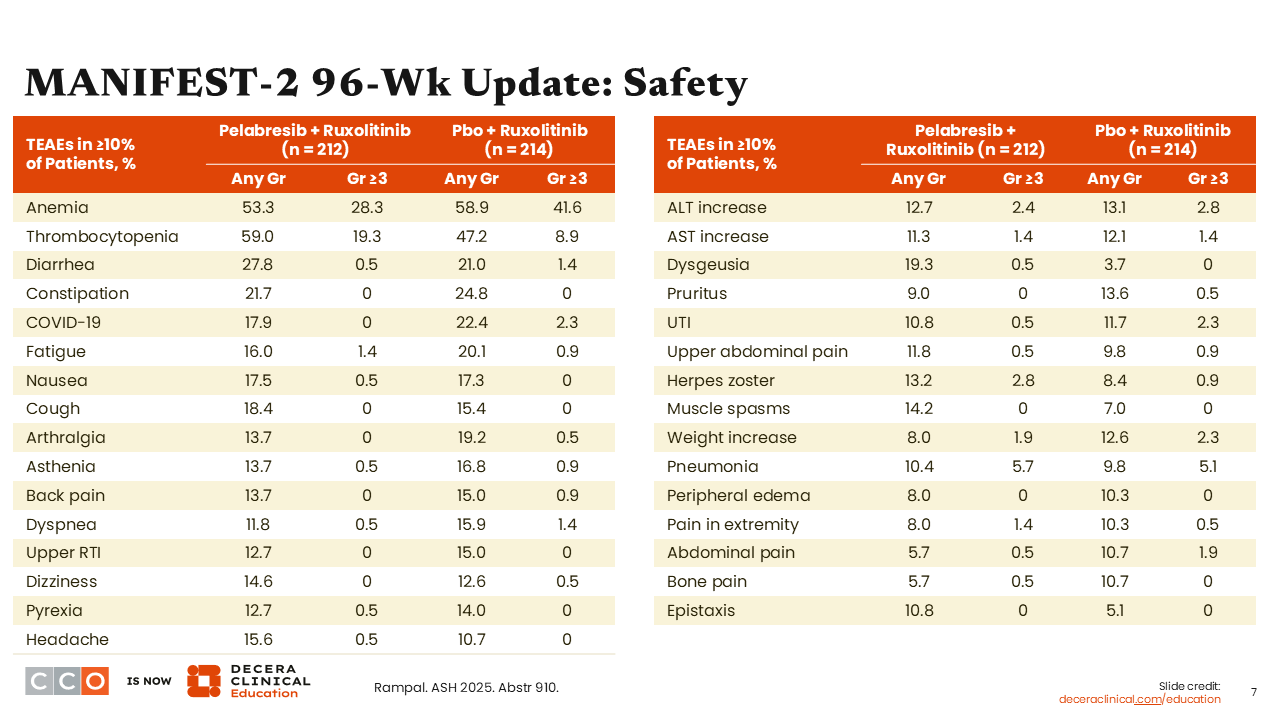
MANIFEST-2 96-Wk Update: Safety
Rami Komrokji, MD:
Treatment-emergent adverse events occurring in ≥10% of patients are shown in these tables. There were no additional major toxicities associated with the addition of pelabresib to ruxolitinib. The treatment-emergent adverse events were expected and consistent with the known toxicity profile of ruxolitinib.2
MANIFEST-2 96-Wk Update: Clinical Implications
Rami Komrokji, MD:
The investigators concluded that addition of pelabresib to ruxolitinib led to better clinical outcomes, namely, almost doubling the spleen response that was maintained at Week 96, symptom improvement, and dual responses, with some suggestion of potential disease modification. Another phase III trial with a similar design but stricter eligibility criteria is currently planned, with enrollment expected to focus on patients who have greater symptom burden at diagnosis. An important open question this study aims to address is which patients are most likely to benefit from upfront combination therapy.
Amer Zeidan, MBBS, MHS:
The data certainly look promising, but unfortunately, they are not sufficient for approval, at least in the US. Traditionally, it has been difficult to add another drug to ruxolitinib in patients with high-risk MDS without causing additional toxicities that compromise the outcome.
I'm optimistic that the redesigned trial that is being planned will be able to better discriminate symptom improvement, because we have already observed a significant reduction in spleen size, and hopefully this combination will become available.
INCA033989 in ET: Study Design
Rami Komrokji, MD:
The next trial to highlight is one looking at the monoclonal antibody INCA033989 in ET. This first-in-class antibody targets mutant calreticulin, and this study is examining its safety and early efficacy in patients with ET.
In this open-label phase I study, patients were required to have a diagnosis of high-risk ET, defined by age, prior thrombotic events, or bleeding tendency, with a pathogenic calreticulin mutation in exon 9. Participants also had to be resistant or intolerant to ≥1 prior therapy and have a platelet count above 450 x 109/L. In the dose-escalation phase, patients were treated with INCA033989 24 mg to 2500 mg intravenously every 2 weeks. The primary endpoint was safety, and the secondary endpoints included response (defined per European LeukemiaNet response criteria) as well as some disease modification endpoints.
Of the 55 patients enrolled, almost 90% had received prior hydroxyurea treatment, and nearly one third had received prior anagrelide or interferon-based therapy (NCT05936359).3
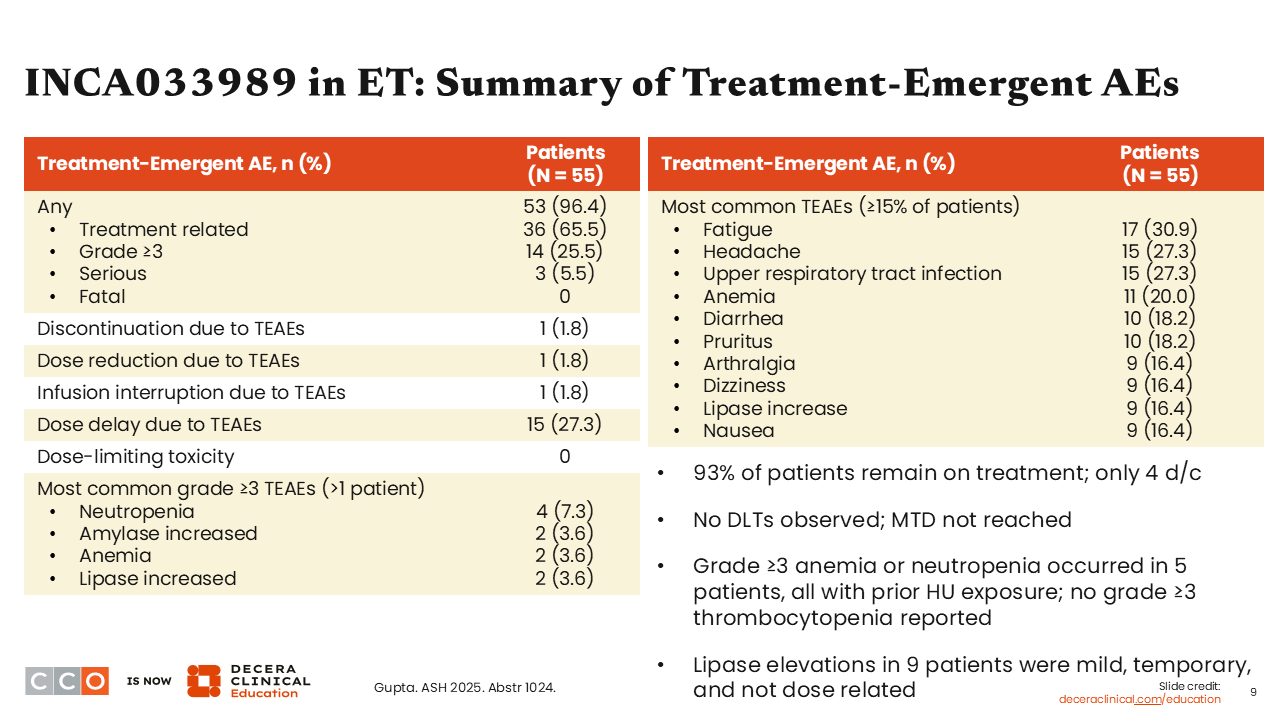
INCA033989 in ET: Summary of Treatment-Emergent AEs
Rami Komrokji, MD:
In general, INCA033989 was well tolerated. At the time of this presentation, 93% of patients remained on treatment. No DLTs were observed, and the MTD was not reached.
The toxicity profile seems to consist mostly of myelosuppression, where grade 3+ anemia or neutropenia occurred in 5 patients, all with prior hydroxyurea exposure. There was no grade 3+ thrombocytopenia reported, and elevated lipase levels in 9 patients were mild, temporary, and not dose related.3
INCA033989 in ET: Hematologic Responses
Rami Komrokji, MD:
From an efficacy standpoint, rapid and durable normalization of platelet counts was noted early on, as were durable hematologic and complete hematologic responses, with durable being defined as ≥12 weeks.
At doses ranging from 24-250 mg, 56.0% of patients achieved a durable hematologic response, and at doses ranging from 400-2500 mg, 64.3% of patients achieved a durable hematologic response. Furthermore, 44.0% and 46.4% of patients achieved a durable complete hematologic response at doses ranging from 24-250 mg and from 400-2500 mg, respectively.3
INCA033989 in ET: Molecular Responses
Rami Komrokji, MD:
Perhaps the most important consideration of this approach is its disease-modifying ability. A reduction in the VAF of mutant CALR was noted in 96% of patients, where more than half (52%) achieved ≥25% reduction in the VAF, and nearly one third (31%) achieved a ≥50% reduction. The responses were seen early, typically within 3-6 months, and in the bone marrow, investigators noted a marked reduction in the number of mutant CALR-positive megakaryocytes, which would suggest, together with the reduced allele burden, that INCA033989 could be a disease-modifying treatment.3
INCA033989 in ET: Clinical Implications
Rami Komrokji, MD:
The investigators concluded that INCA033989 was well tolerated and led to a rapid decrease in platelet counts, durable hematologic responses, and changes in megakaryocytes. I think these data are really interesting and open the gate for immune therapy in this subset of patients who comprise almost one third of those with ET. However, in ET, normalization of platelets or achievement of a hematologic response is not considered a gold standard endpoint.
The path to approval will involve showing that INCA033989 is associated with disease modification. If INCA033989 can change the natural history of the disease, it will have a path forward. Other efforts to target mutant CALR include bispecific antibodies and vaccine-based approaches. This is just the beginning of trials in this field.
Amer Zeidan, MBBS, MHS:
I agree. Scientifically, these results are very interesting. Historically, disease-modifying treatments like hydroxyurea or anagrelide have not been used, so it is exciting to see antibodies directed at a mutation that drives the disease being tested, and we are seeing molecular responses and platelet normalization.
INCA033989 ± Ruxolitinib in MF: Study Design
Rami Komrokji, MD:
The final abstract related to MPN that I want to discuss outlines 2 phase I trials, NCT05936359 and NCT06034002, of INCA033989 with or without ruxolitinib in patients with MF. Again, nearly one third of patients with MF will harbor a calreticulin mutation that can be targeted, so this study enrolled such patients. There were 2 study arms: a monotherapy arm and a combination arm. Patients with primary or post-ET MF with mutant CALR who were either intolerant or resistant to ≥12 weeks of JAK inhibition as monotherapy or ineligible for a JAK inhibitor were treated with monotherapy, and patients who had prior ruxolitinib for ≥12 weeks with a suboptimal response were treated with the combination therapy. The coprimary endpoints were DLTs and safety, and key secondary endpoints included spleen volume reduction by ≥25% (SVR25) and SVR35 at Weeks 12 and 24, as well as symptom improvement. The study enrolled 52 patients in the monotherapy arm and 20 patients in the combination arm, and the baseline characteristics were very consistent with what is known for this disease subtype.4
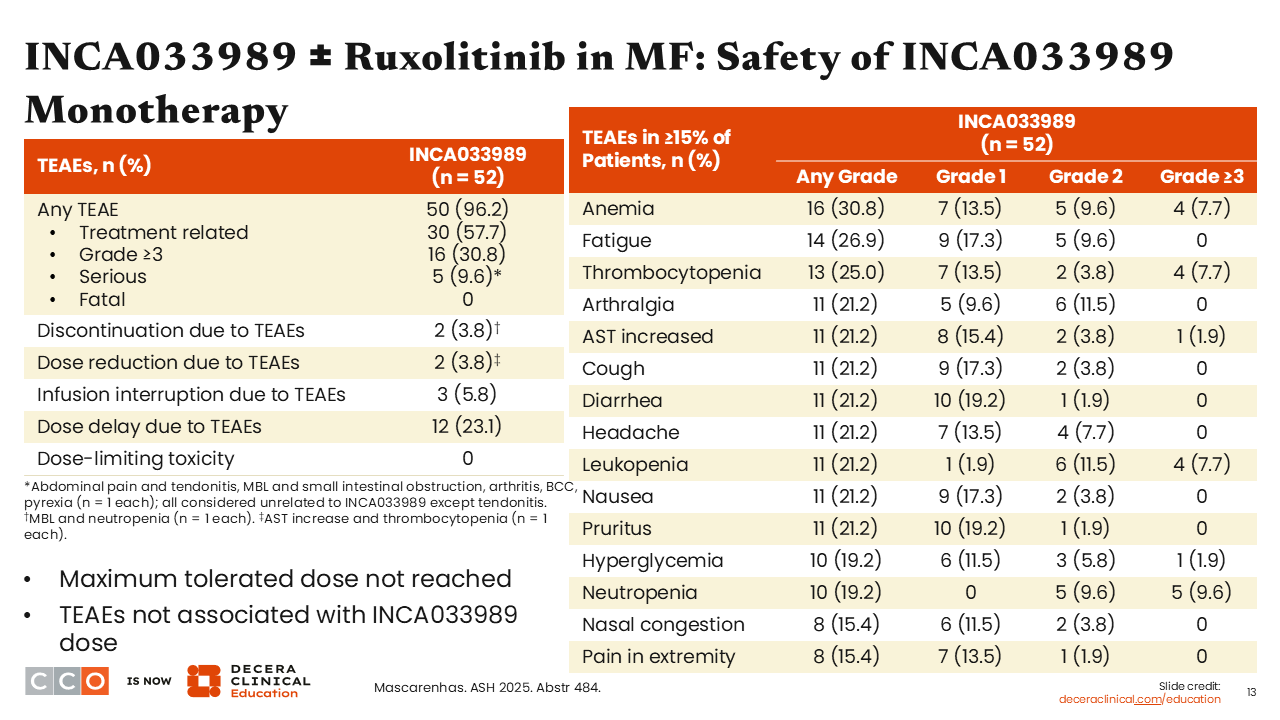
INCA033989 ± Ruxolitinib in MF: Safety of INCA033989 Monotherapy
Rami Komrokji, MD:
In terms of safety, as a monotherapy, the MTD was not reached, and nearly one third (30.8%) of patients had grade 3+ toxicity—mostly anemia, thrombocytopenia, and leukopenia (7.7% each). So, grade 3+ myelosuppression was the key adverse event observed.4
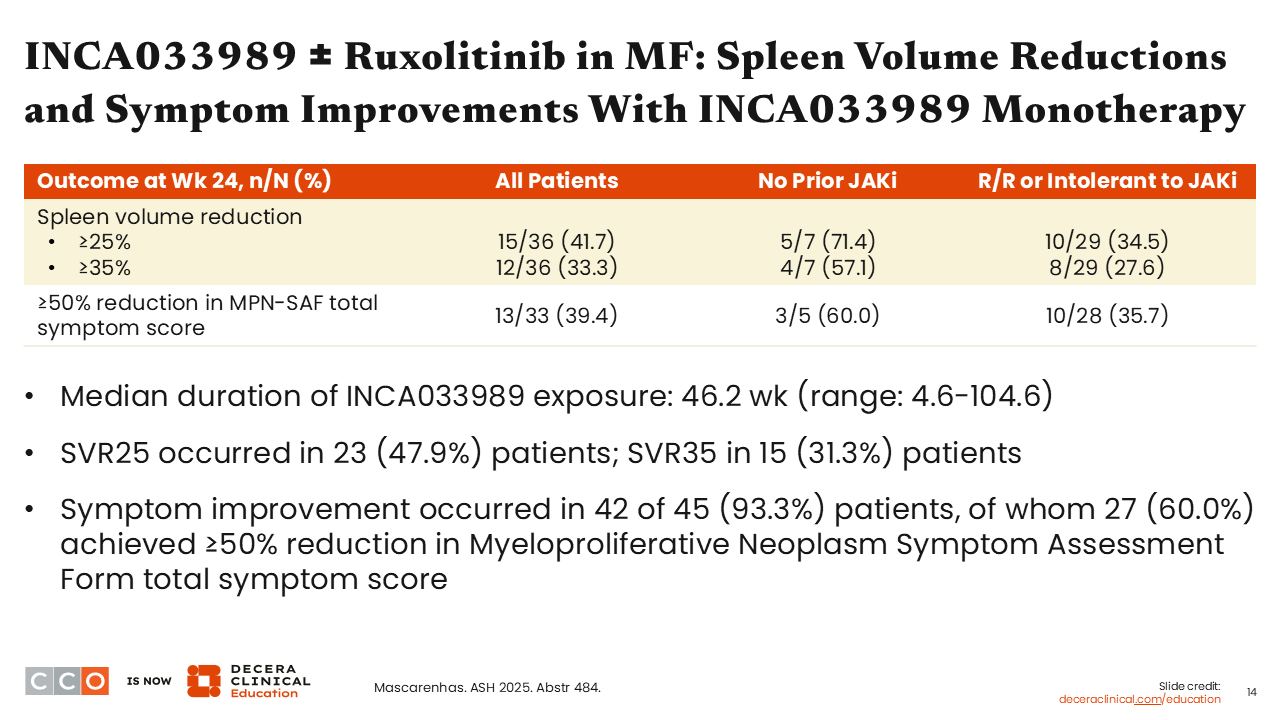
INCA033989 ± Ruxolitinib in MF: Spleen Volume Reductions and Symptom Improvements With INCA033989 Monotherapy
Rami Komrokji, MD:
When we look at the clinical efficacy of INCA033989 as a monotherapy, again, in patients resistant or intolerant to or not eligible for a JAK inhibitor, almost one third (33.3%) experienced spleen volume reduction of ≥35% at Week 24.
Symptom improvement was noted in more than 90% of patients, where 60% had a more than 50% reduction in TSS. Benefit was observed across subgroups.4
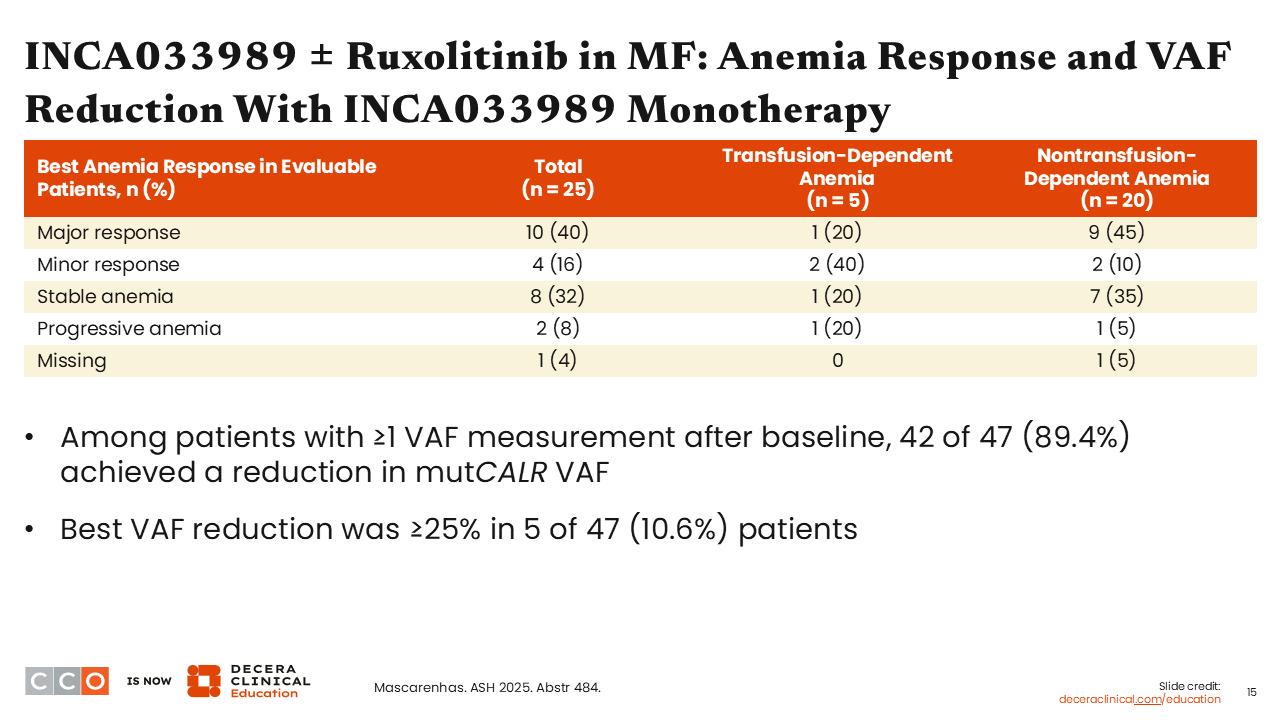
INCA033989 ± Ruxolitinib in MF: Anemia Response and VAF Reduction With INCA033989 Monotherapy
Rami Komrokji, MD:
There were some anemia responses noted with the monotherapy and some indication of disease modification. For example, among all patients, the best anemia response was a major response in 40%, and the best anemia response was a minor response in 16%. Also, the VAF was reduced by ≥25% in 5 of 47 patients.4
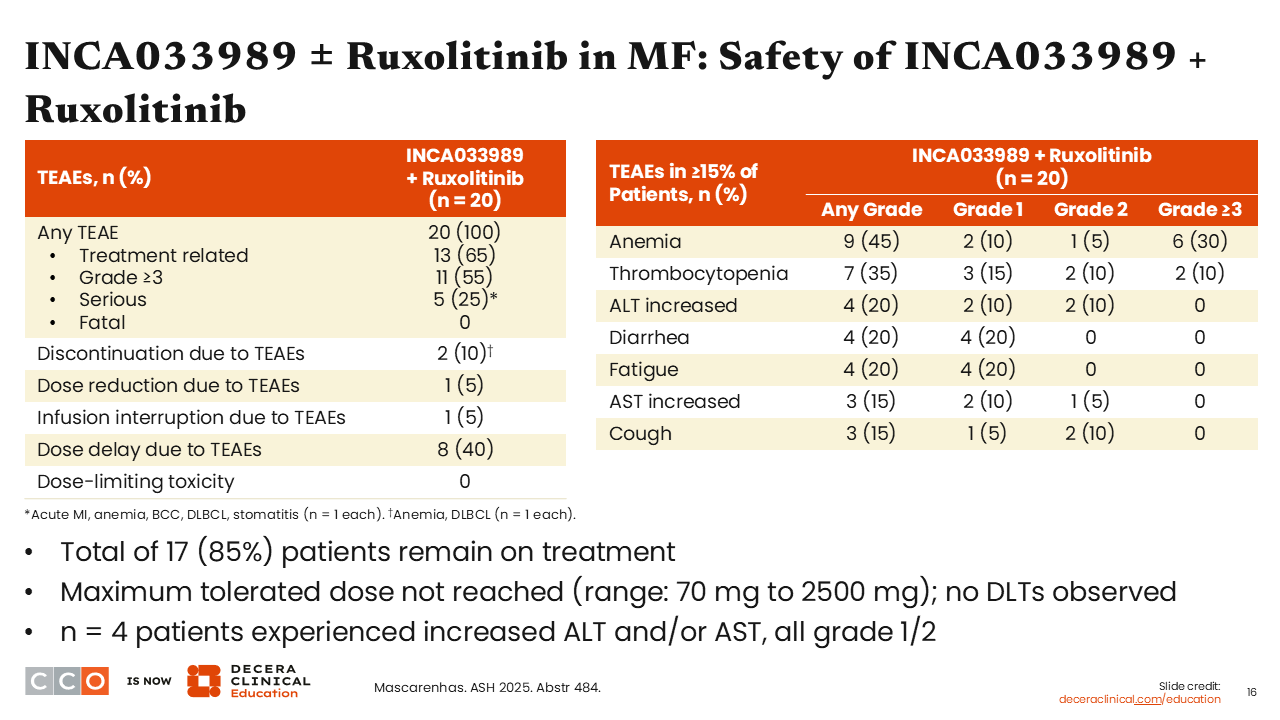
INCA033989 ± Ruxolitinib in MF: Safety of INCA033989 + Ruxolitinib
Rami Komrokji, MD:
In the combination arm, the safety profile was similar. The MTD was not reached. Four patients experienced an increase in ALT and/or AST levels, and the toxicity profile was, again, mostly grade 3+ myelosuppression.4
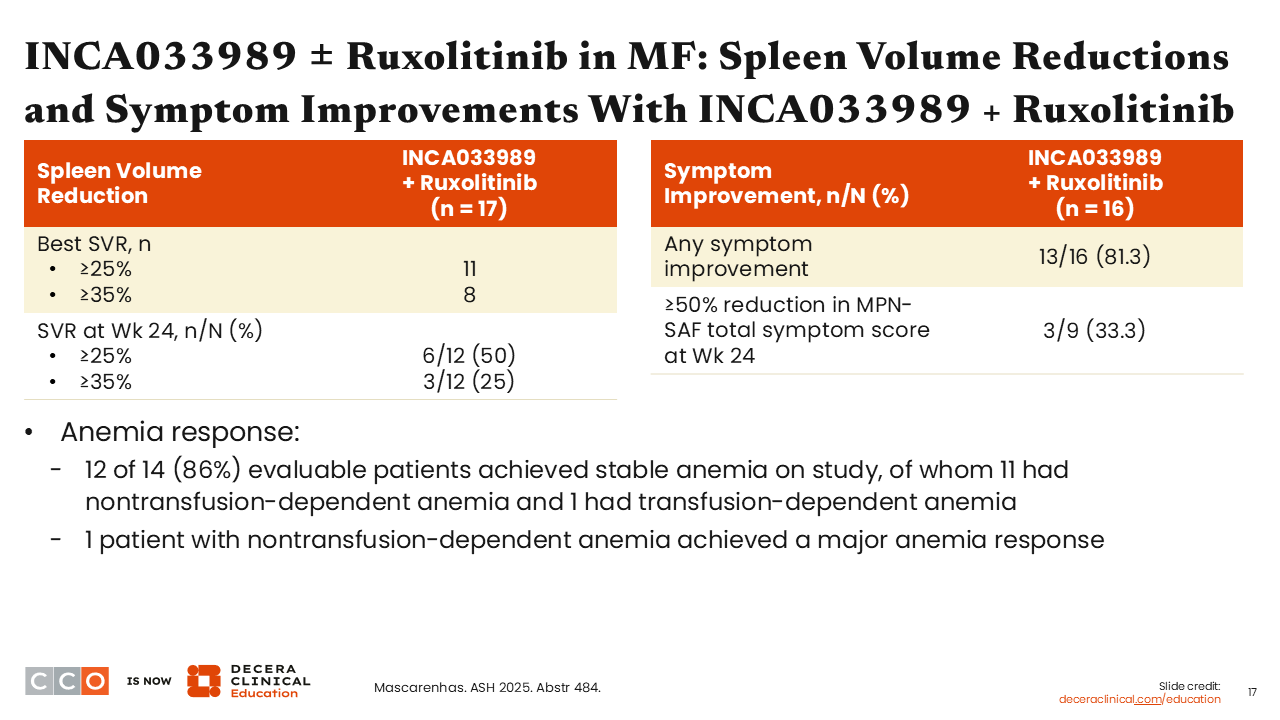
INCA033989 ± Ruxolitinib in MF: Spleen Volume Reductions and Symptom Improvements With INCA033989 + Ruxolitinib
Rami Komrokji, MD:
In the combination arm, 25% of patients achieved SVR35, and one third had a ≥50% reduction in TSS. The majority of patients (86%) achieved stable hemoglobin levels with no further worsening of anemia, which has been a limitation observed with ruxolitinib as it is known to induce anemia and render some patients to be transfusion dependent.4
INCA033989 ± Ruxolitinib in MF: Improved Bone Marrow Pathology With Monotherapy
Rami Komrokji, MD:
The investigators reported reductions in the numbers of mutant calreticulin-positive megakaryocytes, suggesting that this combination could target the underlying driver mutation and be disease modifying.4
INCA033989 ± Ruxolitinib in MF: Clinical Implications
Rami Komrokji, MD:
In conclusion, INCA033989 appears to be a promising therapy. Scientifically, this is a novel approach that harnesses the immune system to target calreticulin-mutant MPNs. In MF, monotherapy for patients who are resistant to or intolerant of JAK inhibitors is effective. Patients are achieving spleen responses and symptom improvement, but the dosing in the combination regimen, which is beneficial for patients with a suboptimal response, still needs to be defined. The message is to encourage community healthcare professionals to refer patients with a calreticulin mutation to ongoing clinical trials.
Amer Zeidan, MBBS, MHS:
As in the previous study, I think we are seeing similar activity in terms of clinical improvement in the spleen, but of importance, in addition to symptoms, the results suggest disease modification. I think this is where we are heading with myeloid malignancies, including MF. However, we need to break the ceiling with ruxolitinib monotherapy because it may provide a chance to demonstrate survival improvement in a more conclusive way than what has been seen so far with ruxolitinib monotherapy. I look forward to seeing additional data with this drug.
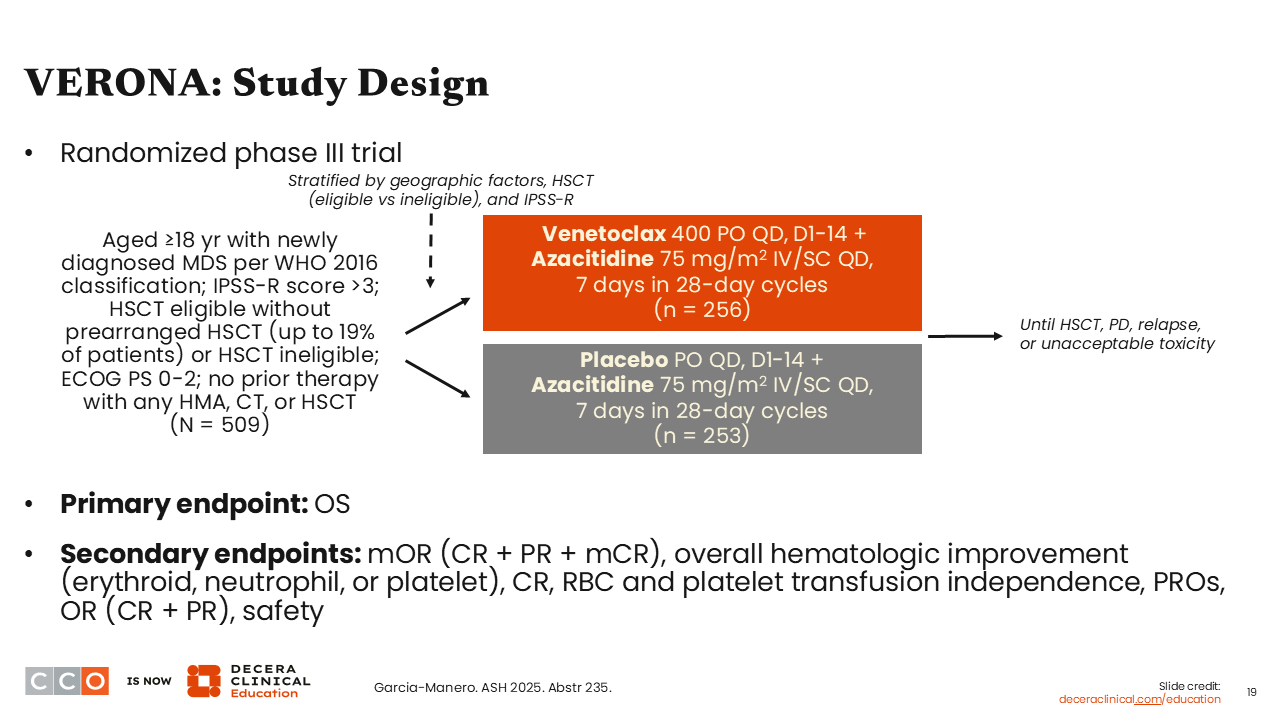
VERONA: Study Design
Amer Zeidan, MBBS, MHS:
Next, I am going to discuss a subgroup analysis from the phase III VERONA trial looking at venetoclax in patients with higher-risk MDS. This trial enrolled adults with newly diagnosed higher-risk MDS. However, it is important to note that it capped the number of patients who were determined to be transplant eligible to 19% and randomized patients to receive venetoclax plus azacitidine or placebo plus azacitidine. The primary endpoint was overall survival (OS), which was reported previously.5 This analysis aimed to identify subgroups of patients who might benefit more from treatment with the combination of venetoclax plus azacitidine.
Baseline characteristics were generally well balanced, although TP53 mutations were detected in 24.5% of patients in the venetoclax plus azacitidine arm vs 18.7% in the azacitidine alone arm. Median age was approximately 72 years, and more than 40% of patients in each arm had ≥10% blasts in the bone marrow (NCT04401748).6
VERONA: Efficacy
Amer Zeidan, MBBS, MHS:
When we look at efficacy, there was no difference in OS between the 2 arms among the overall population. However, there was a trend toward improved OS with the combination in patients <75 years of age and in patients who had a high to very high IPSS risk, as well as those with a high blast count or with RUNX1 or EZH2 mutations at baseline.6
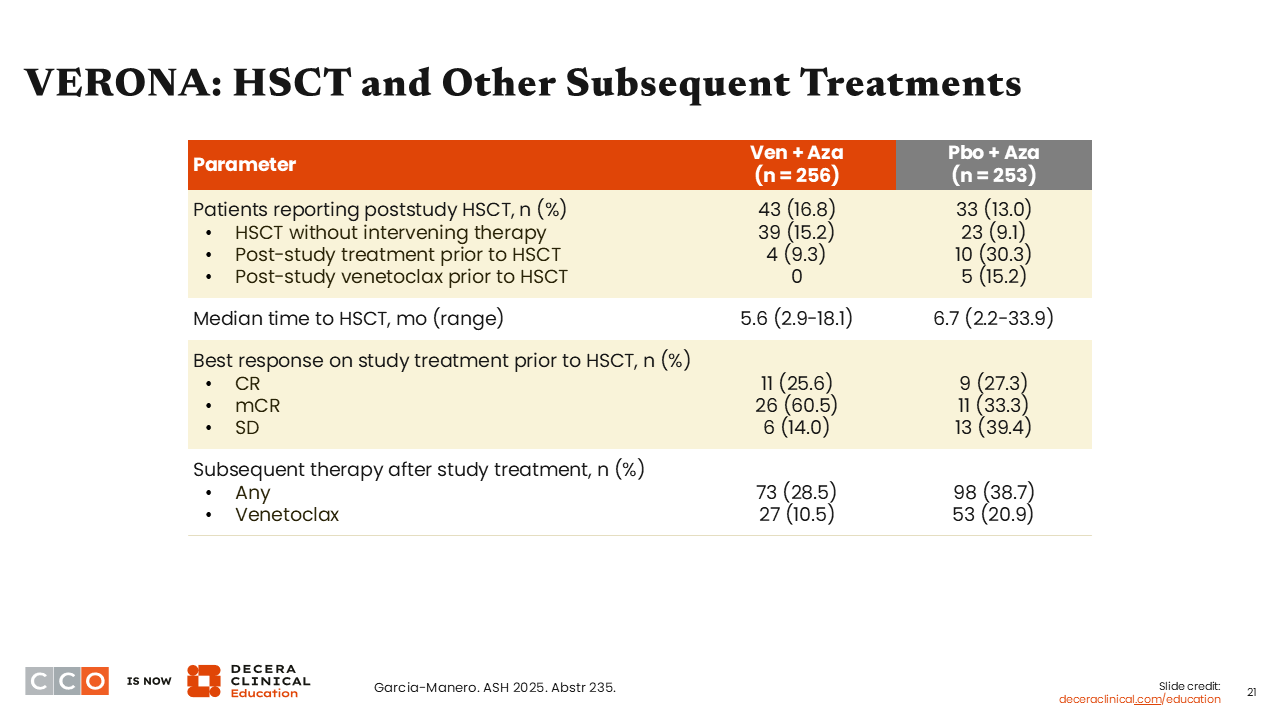
VERONA: HSCT and Other Subsequent Treatments
Amer Zeidan, MBBS, MHS:
The transplant outcomes are interesting. We would expect more patients on venetoclax plus azacitidine to undergo transplant, but the percentage of patients receiving poststudy transplant was approximately 17% vs 13% with azacitidine plus placebo, so only slightly higher.
However, the complete response (CR) rate for those on study treatment before transplant was highly similar, and the median time to transplant was approximately 1 month sooner with venetoclax plus azacitidine vs azacitidine plus placebo. Of importance, more patients treated with azacitidine plus placebo received subsequent therapy after study treatment (38.7%) vs venetoclax plus azacitidine (28.5%).6
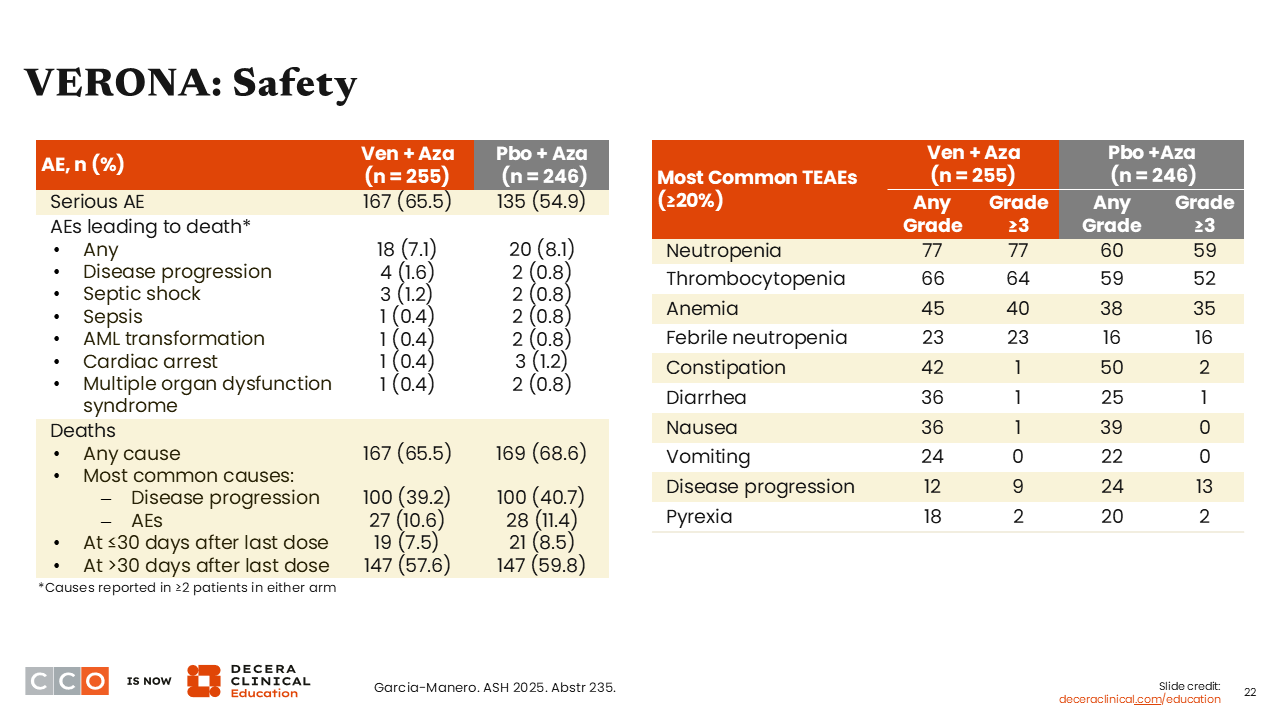
VERONA: Safety
Amer Zeidan, MBBS, MHS:
In terms of safety, there were no new signals. There was more myelosuppression with the addition of venetoclax, as expected, and it was encouraging to see no significant increase in early deaths, which suggests appropriate management of myelosuppression.6
VERONA: Clinical Implications
Amer Zeidan, MBBS, MHS:
In conclusion, primary analysis of VERONA did not show an OS benefit with venetoclax plus azacitidine vs azacitidine and placebo in patients with previously untreated, higher-risk MDS, but subgroup analyses suggested improvements in certain populations, including those <75 years of age, those with high risk by IPSS-R, and those with high blast counts at baseline.
This study was largely disappointing to the field because there was much hope for breaking through azacitidine monotherapy as a standard of care in patients with MDS. I believe there were some design issues, such as capping the number of patients going to transplant at eligibility and allowing patients with <5% blasts to go on trial. However, I don't think the door is completely closed on the use of BCL-2 inhibition in higher-risk MDS. Indeed, other BCL-2 inhibitors are currently being studied, so hopefully we will see more data with these agents.
Rami Komrokji, MD:
I totally agree. It was unfortunate that the study was negative for the primary endpoint, but there is no doubt that experts in the field believe there is value to venetoclax, as was suggested in real world data presented at ASH showing benefit. It is clear from this subgroup analysis that some populations, perhaps those with a higher blast percentage, increased risk of disease, bridge to transplant, and certain molecular subtypes, like ASXL-1, may benefit.
IMerge Post Hoc Analysis: Study Design
Amer Zeidan, MBBS, MHS:
The next abstract to discuss is a post hoc analysis from the phase III IMerge trial (NCT02598661) that led to the approval of imetelstat, a first-in-class telomerase inhibitor, for adults with low- to intermediate-1–risk MDS who are transfusion dependent (requiring ≥4 RBC units over 8 weeks) and are not responsive to or eligible for erythropoiesis-stimulating agents.7 However, two thirds of patients treated with imetelstat will experience grade 3 or higher neutropenia or thrombocytopenia early in treatment, usually in the first 2-3 cycles, but these events are reversible.
Investigators conducted this post hoc analysis to understand whether early treatment-emergent neutropenia and thrombocytopenia within the first 2 cycles correlate with subsequent clinical responses. In total, 226 patients who were enrolled in the phase II section, randomized phase III section, and phase III QTc substudy were evaluated.8
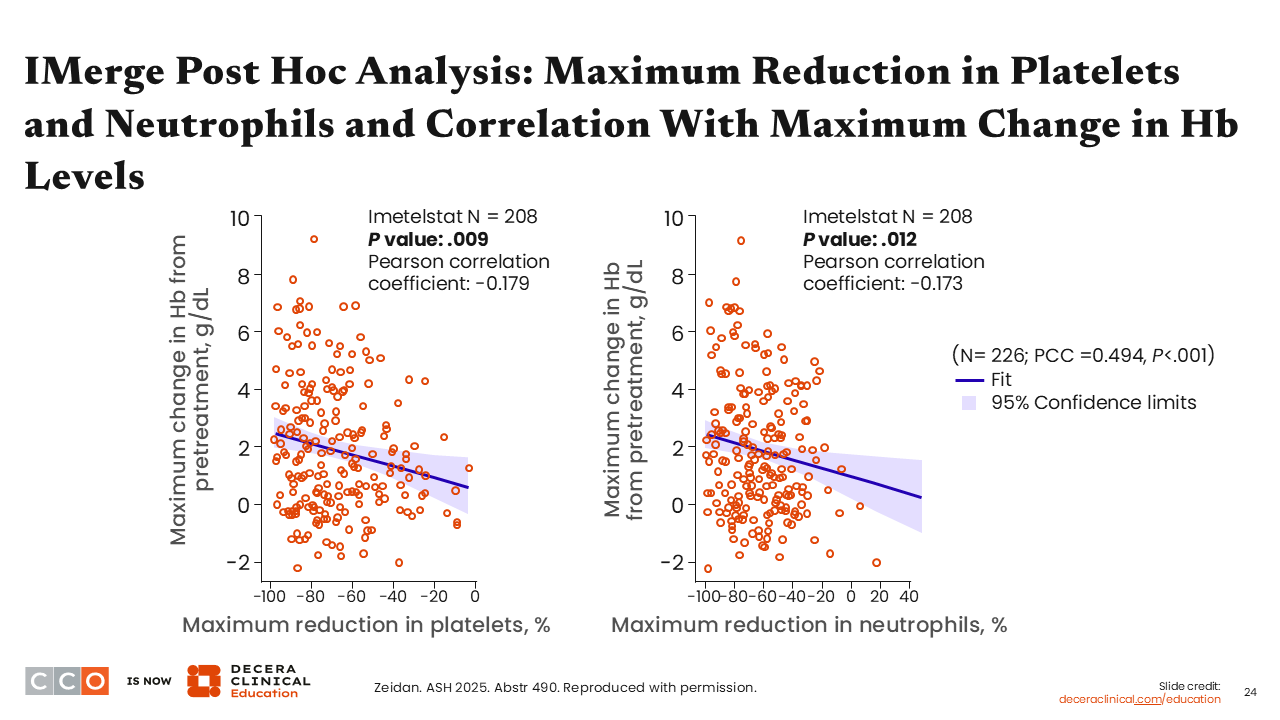
IMerge Post Hoc Analysis: Maximum Reduction in Platelets and Neutrophils and Correlation With Maximum Change in Hb Levels
Amer Zeidan, MBBS, MHS:
At baseline, the median platelet count was 236 x 109/L and the median neutrophil count was 2.6 x 109/L. The investigators first aimed to correlate the maximum reduction in platelet and neutrophil counts as percentages from baseline with the maximum increase in hemoglobin for each patient. The linear analysis showed a statistically significant correlation between these reductions in platelet and neutrophil counts with maximum increase in hemoglobin.8
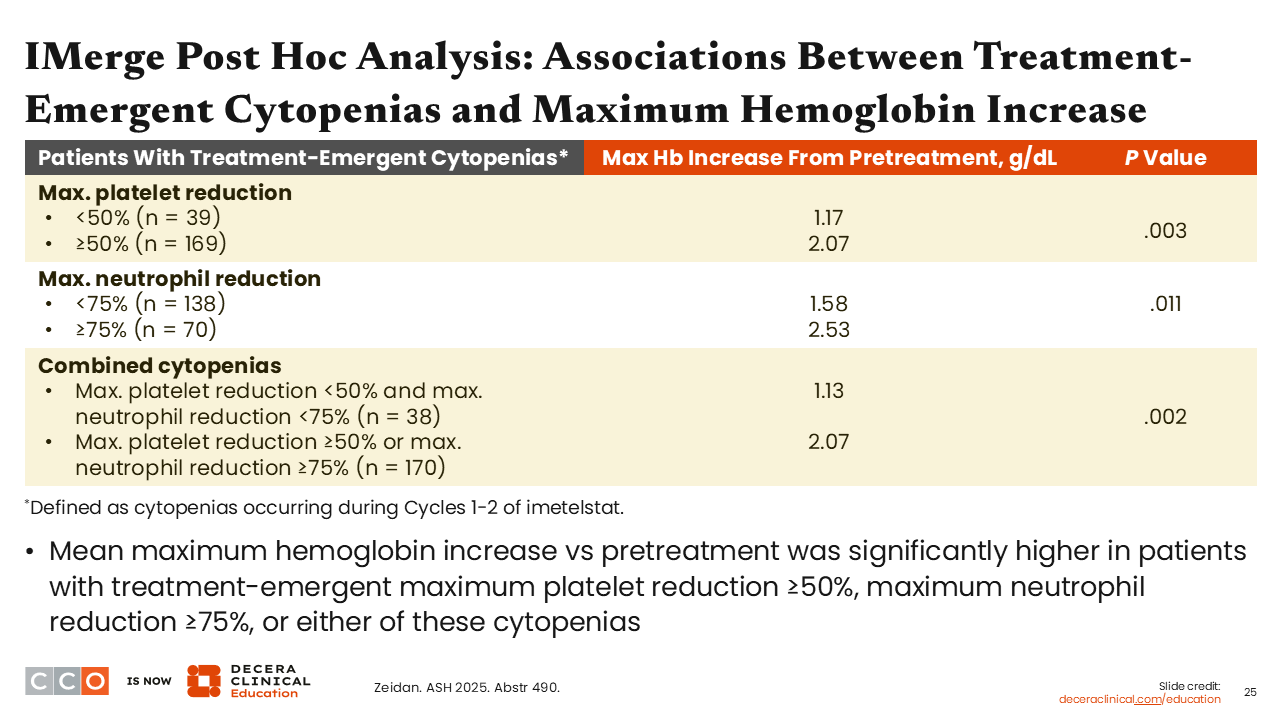
IMerge Post Hoc Analysis: Associations Between Treatment-Emergent Cytopenias and Maximum Hemoglobin Increase
Amer Zeidan, MBBS, MHS:
The maximum hemoglobin increase was approximately 2 g/dL for participants with ≥50% maximum reduction in platelets vs approximately 1 g/dL for participants with less than 50% maximum reduction in platelets.8
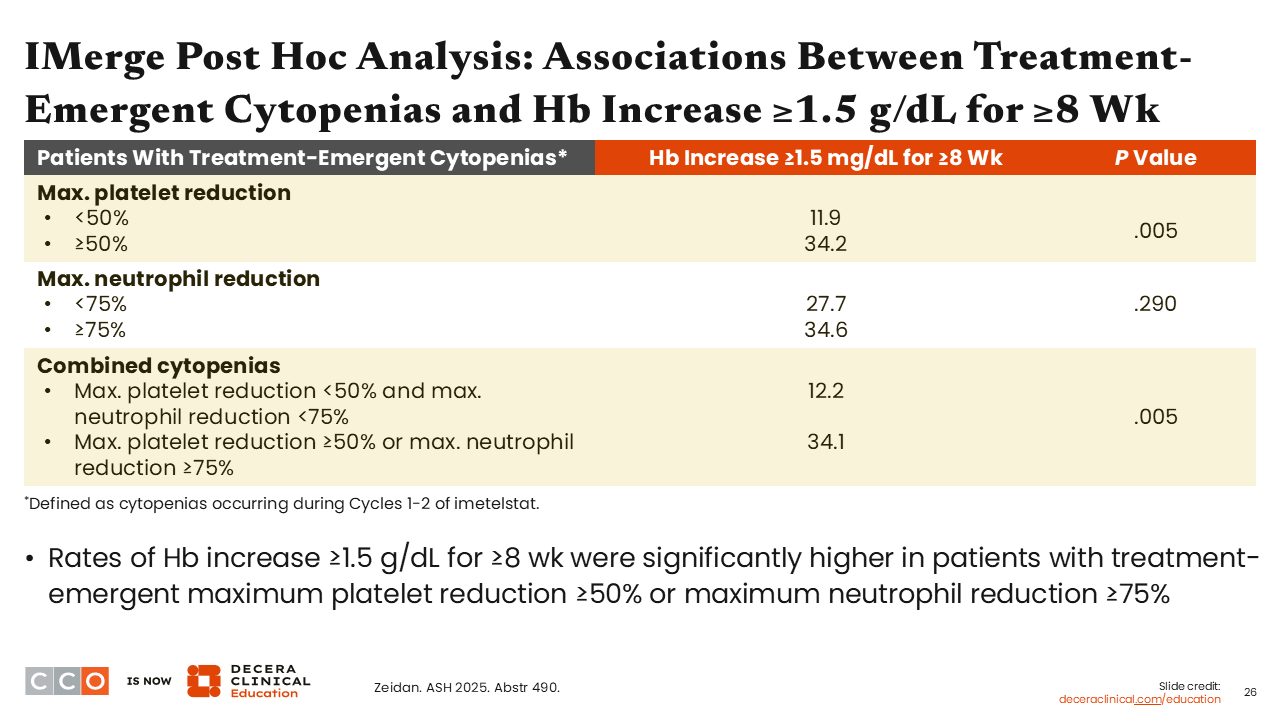
IMerge Post Hoc Analysis: Associations Between Treatment-Emergent Cytopenias and Hb Increase ≥1.5 g/dL for ≥8 Wk
Amer Zeidan, MBBS, MHS:
Next, investigators correlated hemoglobin increases of ≥1.5 g/dL lasting ≥8 weeks with treatment-emergent cytopenias and obtained a similar correlation, although the degree of maximum neutrophil reduction was not statistically significant.8
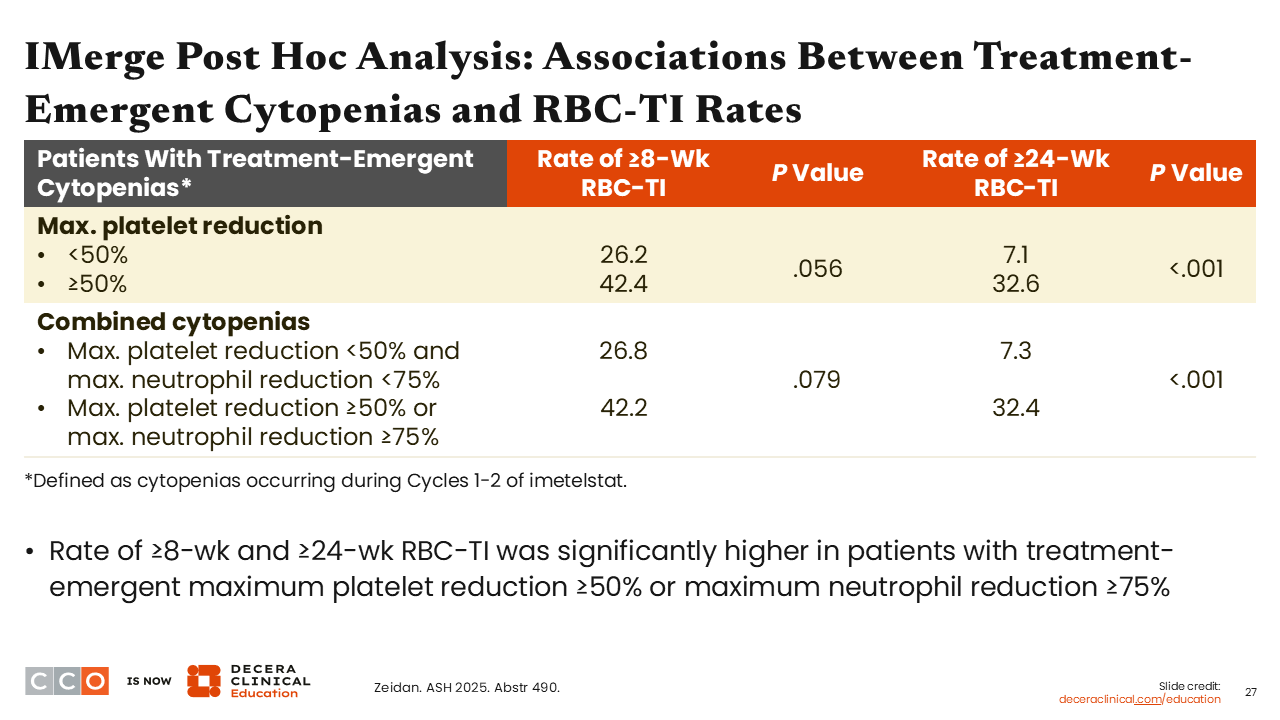
IMerge Post Hoc Analysis: Associations Between Treatment-Emergent Cytopenias and RBC-TI Rates
Amer Zeidan, MBBS, MHS:
When looking at transfusion independence, investigators found that patients with a maximum platelet reduction of ≥50% had a higher chance of achieving transfusion independence lasting ≥8 weeks and a significantly higher chance of achieving transfusion independence lasting ≥24 weeks.
Together, these values support that early reductions in platelet and neutrophil counts are predictors of subsequent responses in terms of transfusion independence and likely suggest an on-target effect of imetelstat on the MDS clone, where clearance of this clone might be the reason for early thrombocytopenia and neutropenia, with subsequent recovery of the erythroid lineage.8
IMerge Post Hoc Analysis: Clinical Implications
Amer Zeidan, MBBS, MHS:
To conclude, there is a good correlation between a maximum neutrophil reduction of ≥75% and a maximum platelet reduction of ≥50% in the first 2 cycles with achieving a durable maximum hemoglobin increase, as well as transfusion independence, suggesting that these factors could be used as predictive biomarkers and suggesting that imetelstat could be disease modifying and should be investigated further.
Rami Komrokji, MD:
I agree. It is well known that grade 3 and 4 thrombocytopenia and neutropenia are common in the first 2 rounds of imetelstat therapy, but cytopenia on treatment now predicts response, and the message is not to abandon treatment because these patients are going to respond. However, this does not mean that treatment will not be withheld and dose adjusted according to those cytopenias.
The main message for community oncologists is that cytopenia observed on treatment is an early sign of response. These findings also potentially address a correlation to the drug’s mechanism of action.
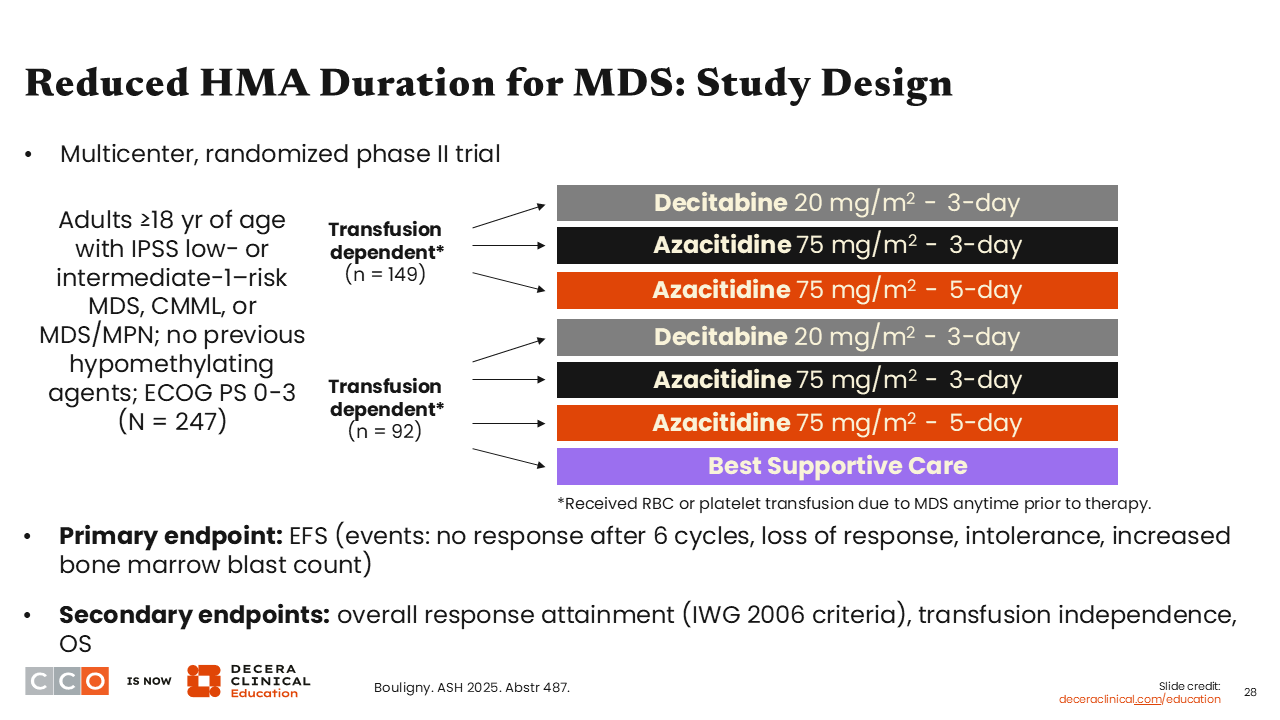
Reduced HMA Duration for MDS: Study Design
Amer Zeidan, MBBS, MHS:
The last abstract to review was a multicenter, randomized study looking at using shorter courses of hypomethylating agents in the management of lower-risk MDS. Hypomethylating agents continue to be widely used for MDS—generally high-risk MDS but also lower-risk MDS—but since they are also associated with increased risks of neutropenia and thrombocytopenia, there have been efforts to reduce the days of administration to improve the side effect profile while achieving good transfusion independency rates.
This trial was designed to compare shorter courses of decitabine and azacitidine (3 days) to a 5-day schedule of azacitidine, which is commonly used in the community. There were 2 groups of patients: those who were transfusion independent and those who were transfusion dependent. All study participants had anemia. In the transfusion-dependent cohort, some patients were also randomized to supportive care. The primary endpoint was event-free survival (EFS), defined as no response after 6 cycles, loss of response, intolerance, or increased bone marrow blast counts suggesting progression. Of interest, many patients did not have prior exposure to erythropoiesis-stimulating agents, and approximately 50% to 70% of patients were transfusion dependent.9
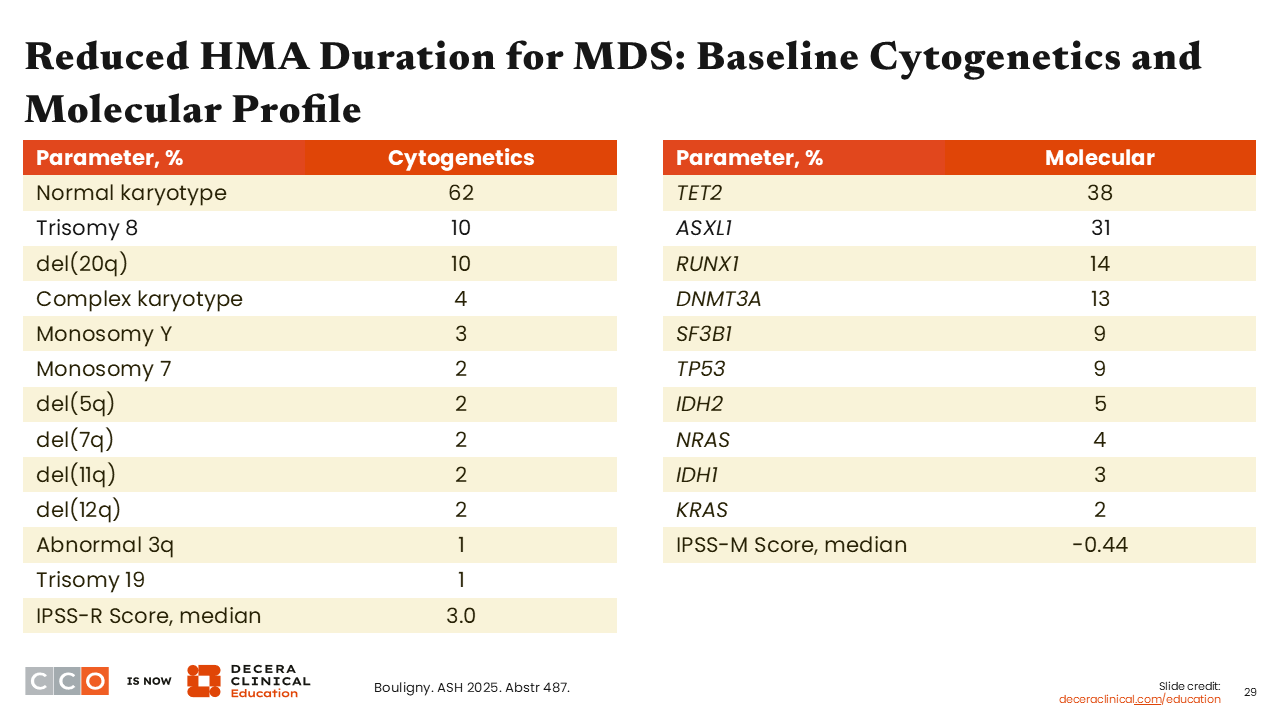
Reduced HMA Duration for MDS: Baseline Cytogenetics and Molecular Profile
Amer Zeidan, MBBS, MHS:
In terms of cytogenetics, many patients had a normal karyotype, as expected, but molecular mutations were also common, as is known in MDS. Only 9% of patients had a TP53 mutation, and the most common mutation, found in 38% of patients, was in TET2.9
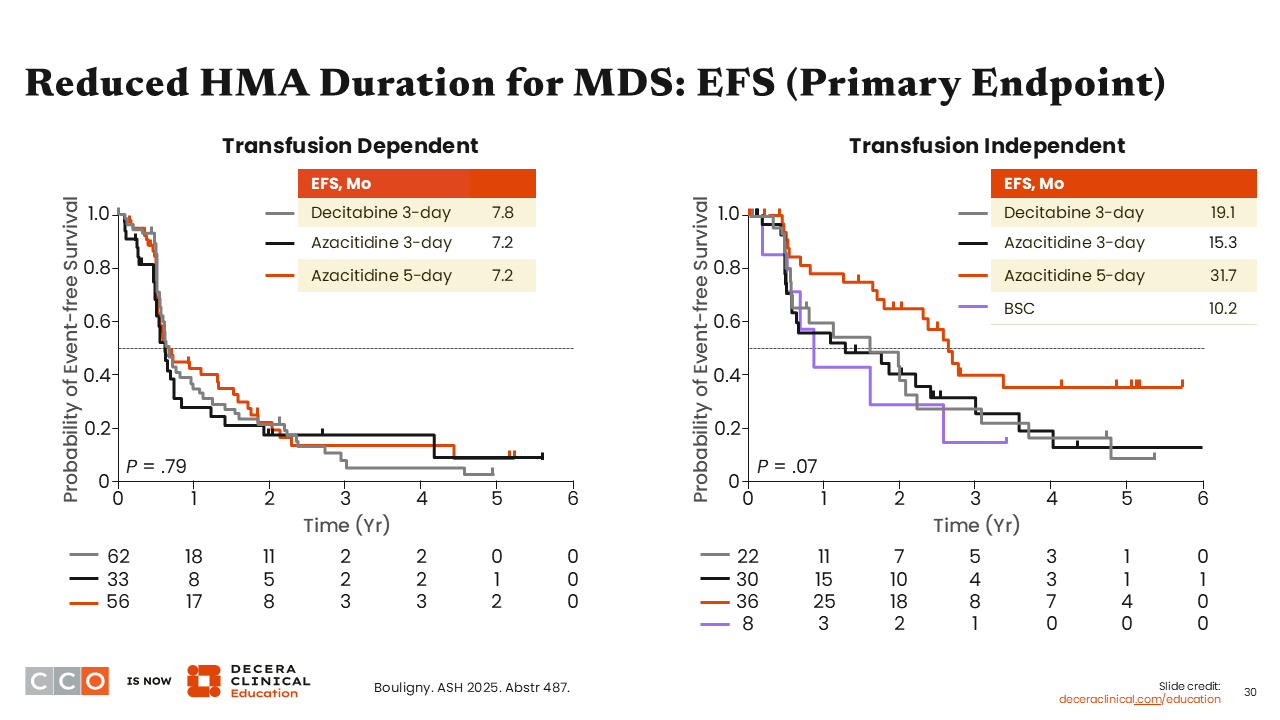
Reduced HMA Duration for MDS: EFS (Primary Endpoint)
Amer Zeidan, MBBS, MHS:
In the transfusion-dependent cohort, there was no significant difference in EFS between the 3 courses (ie, decitabine for 3 days, azacitidine for 3 days, or azacitidine for 5 days). EFS was approximately 7-8 months in all cohorts. However, in the transfusion-independent cohort, EFS favored the use of azacitidine for 5 days, where EFS was 31.7 months vs approximately 15-19 months for azacitidine for 3 days and decitabine for 3 days, respectively.
The authors performed a multivariable analysis in both the transfusion-dependent and transfusion-independent cohorts that confirmed the value of 5-day azacitidine, even after adjusting for other prognostic variables that can affect EFS.9
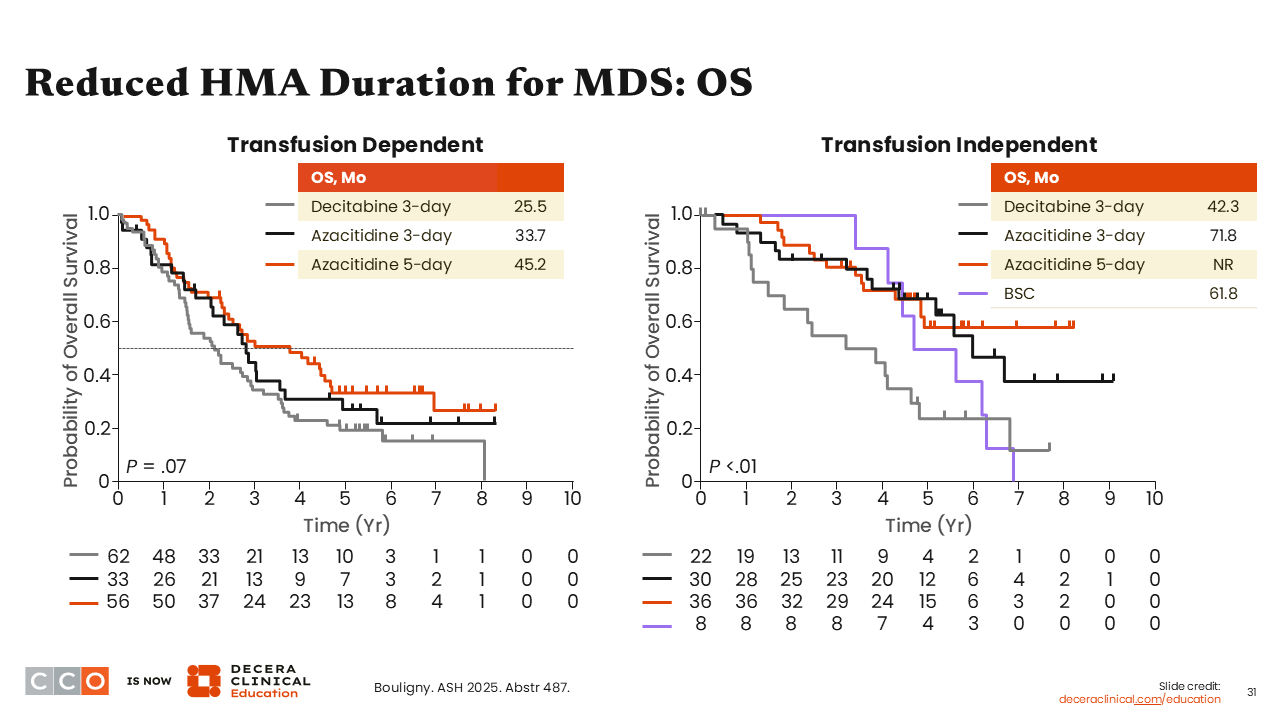
Reduced HMA Duration for MDS: OS
Amer Zeidan, MBBS, MHS:
In the transfusion-dependent cohort, OS was longest in the 5-day azacitidine cohort, at 45.2 months, which was not statistically significant. However, in the transfusion-independent cohort, OS was significantly prolonged in favor of 5-day azacitidine, where OS was not reached.9
Similarly, multivariable analysis of OS in both cohorts showed favor for 5-day azacitidine.9
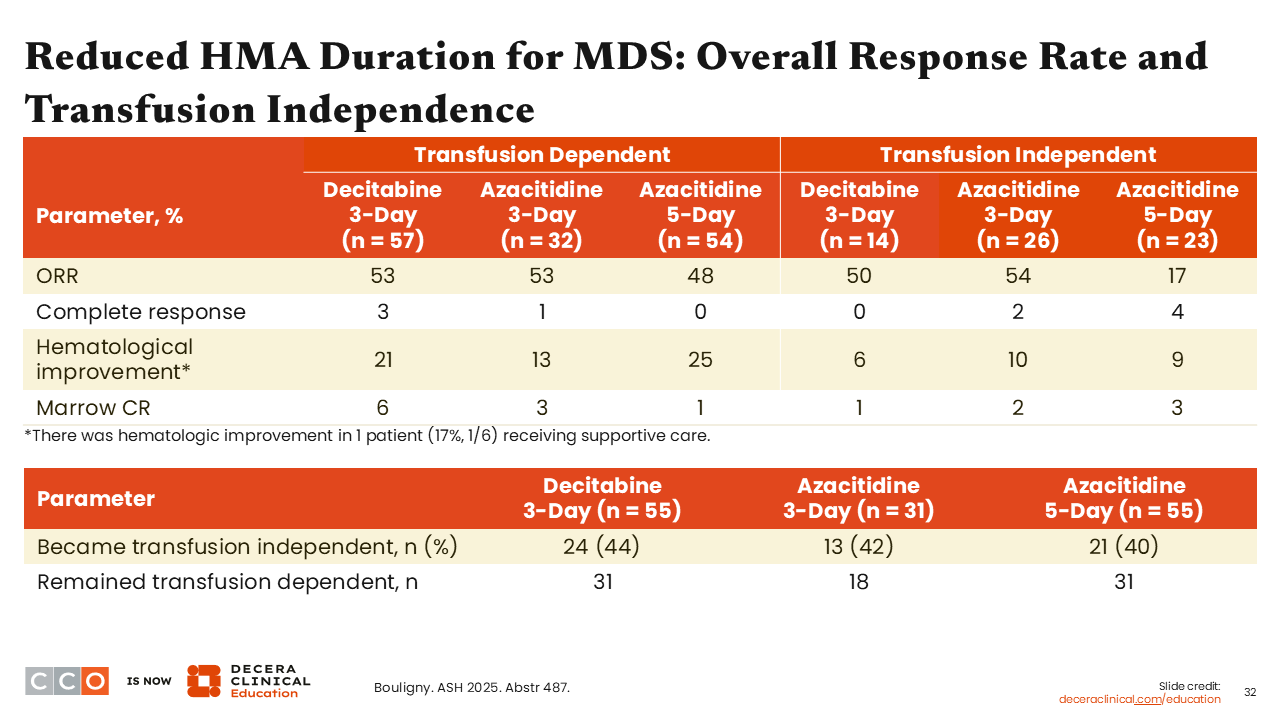
Reduced HMA Duration for MDS: Overall Response Rate and Transfusion Independence
Amer Zeidan, MBBS, MHS:
The authors did not see a significant difference in terms of the overall response rate or transfusion independency between the 5 treatment arms.9
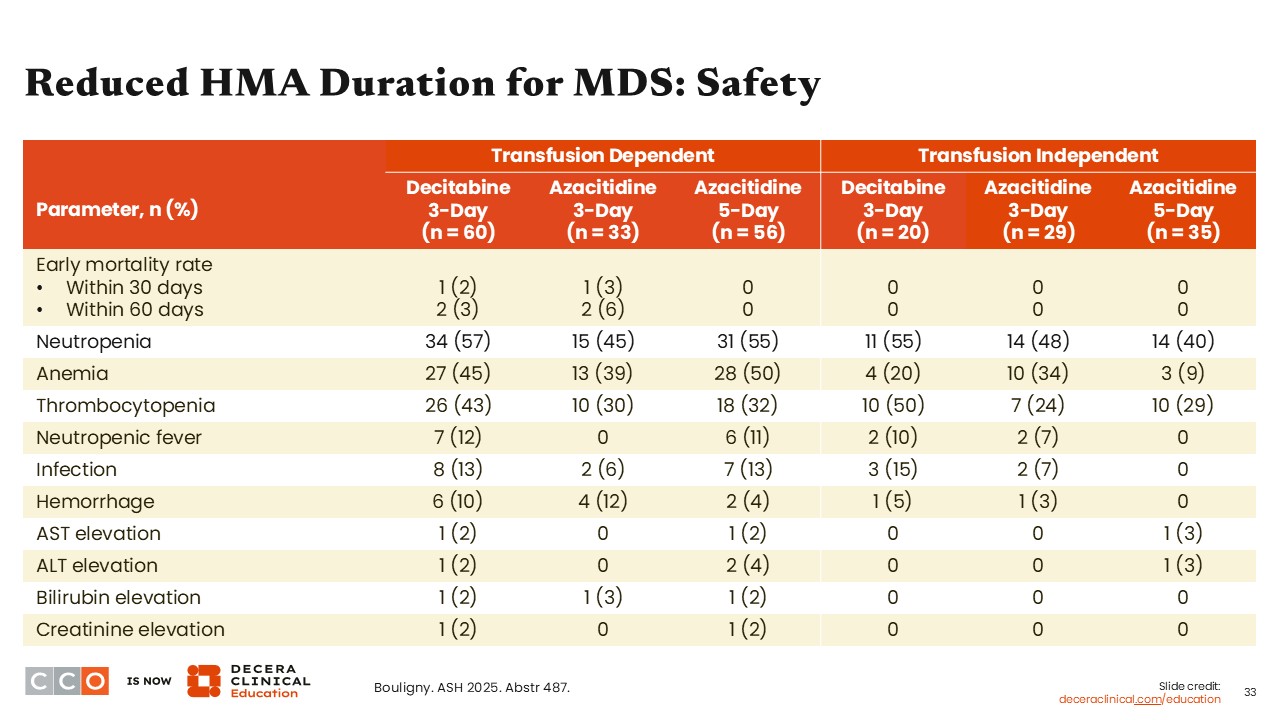
Reduced HMA Duration for MDS: Safety
Amer Zeidan, MBBS, MHS:
This analysis confirmed that the use of decitabine and azacitidine is very safe, where the common side effects were primarily reductions in blood counts, which are common, but the rates of nonhematologic side effects, including infection and liver enzyme elevations, were generally low. Of importance, there was very little early mortality with any of these regimens, especially with 5-day azacitidine, suggesting that its use is very safe in patients with lower-risk MDS.9
Reduced HMA Duration for MDS: Clinical Implications
Amer Zeidan, MBBS, MHS:
The authors concluded that shorter durations of hypomethylating agents for lower-risk MDS are clinically feasible, with low risks of complications and clinical benefits in the form of EFS. It seems that the 5-day azacitidine course is a better approach than 3-day decitabine and 3-day azacitidine. However, there was no direct comparison between 5-day azacitidine and 5-day decitabine, and there was no comparison against the oral version of decitabine, which is now approved for adults with MDS in combination with cedazuridine.10
I think these results provide reassurance that shorter courses of hypomethylating agents are a reasonable option for patients with lower-risk MDS, especially those who are refractory to other agents and for patients with significant neutropenia and thrombocytopenia, in addition to anemia.
Rami Komrokji, MD:
This study was designed several years ago in an era where the landscape was different. To your point, we did not have luspatercept or imetelstat. The EFS and OS advantages were seen mostly in patients who were transfusion independent. It is likely that those patients tended to have thrombocytopenia, neutropenia, or another indication to treat, whereas in patients who were transfusion dependent, anemia was probably the main indication.
Overall, it is reassuring to see that decitabine and azacitidine are safe in lower-risk MDS, yielding reasonable transfusion independency. However, I don’t believe these should move to frontline treatment. They should be reserved for after failure of other current therapies or when cytopenias dictate their use.