CME
Key Studies in Breast Cancer: Independent Conference Coverage of the 2025 ESMO Congress
Physicians: Maximum of 1.50 AMA PRA Category 1 Credits™
European Learners: 1.50 EBAC® CE Credit
Released: December 15, 2025
Expiration: June 14, 2026
Activity
Therapy for Patients With TNBC Not Eligible for PD-L1 Therapy
Cristina Saura, MD, PhD:
The addition of pembrolizumab to chemotherapy is SoC for patients with metastatic TNBC and a tumor PD-L1 expression level of combined positive score ≥10.21,22 However, approximately 60% to 70% of patients with metastatic TNBC are not candidates for PD-L1 therapy either because their tumors are PD-L1 negative or if PD-L1 positive, they are not eligible to receive this type of therapy due to comorbidities.21,23 Of note the median PFS expected in the first-line setting with standard chemotherapy is approximately 6 months, with a 5-year OS of approximately 15% for patients with metastatic TNBC.21,24
The other important point here is the attrition rate. Approximately 50% of the patients who received first-line therapy are not candidates for a second line of therapy either because of death or they progress with deterioration of performance status that prevents continuation with active treatment.23 This is a large percentage of the TNBC population that needs better therapies and this is the primary issue that both the ASCENT-03 and TROPION-Breast02 trials propose to address.25-27
ASCENT-03: First-line Sacituzumab Govitecan vs CT for Patients With Advanced TNBC Ineligible for PD-1/PD-L1 Inhibitors
Cristina Saura, MD, PhD:
The phase III ASCENT-03 trial examined the TROP-2 ADC sacituzumab govitecan vs chemotherapy in patients with metastatic TNBC ineligible for PD-L1 inhibitors.25,26 Sacituzumab govitecan is administered intravenously on Day 1 and 8 every 21 days. The chemotherapy schedules allowed in the SoC arm included paclitaxel or nab-paclitaxel, but also combination chemotherapy. In my opinion, the chemotherapy arm is optimal because they allow a combination of chemotherapies, and this is important to note when interpreting the results. Another issue of importance is that it must have been 6 months or longer since the last treatment in the curative setting, patients who relapse in less than 6 months were not eligible for this trial. Of note, the trial included the option to allow crossover from the chemotherapy arm to second-line sacituzumab govitecan upon progression with standard chemotherapy. These features are key to interpreting the results of this trial and to compare with the TROPION-Breast02 trial we will discuss next.
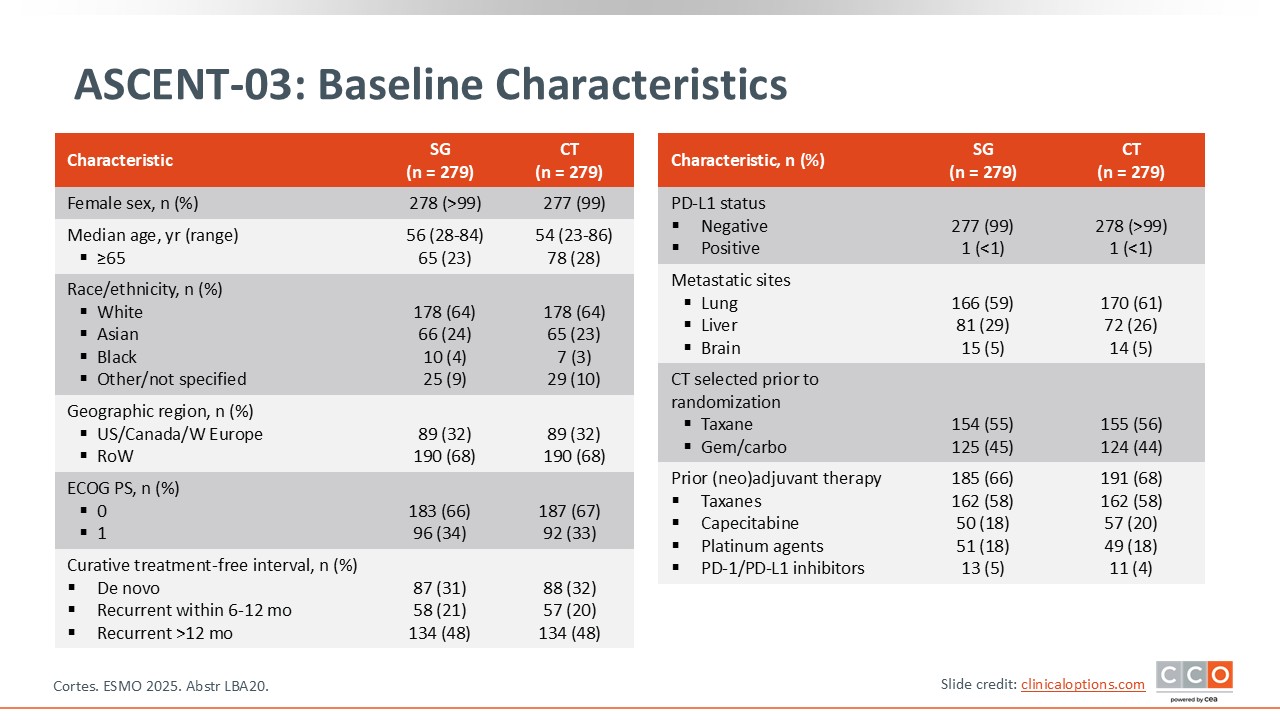
ASCENT-03: Baseline Characteristics
Cristina Saura, MD, PhD:
Patients with de novo disease comprised 31% to 32% of the population.25,26 Approximately 20% of patients had a recurrence between 6 and 12 months and 48% had recurrent disease after 12 months. With respect to selection of chemotherapy prior to randomization, approximately 55% selected a taxane vs approximately 45% selecting the combination of gemcitabine/carboplatin.
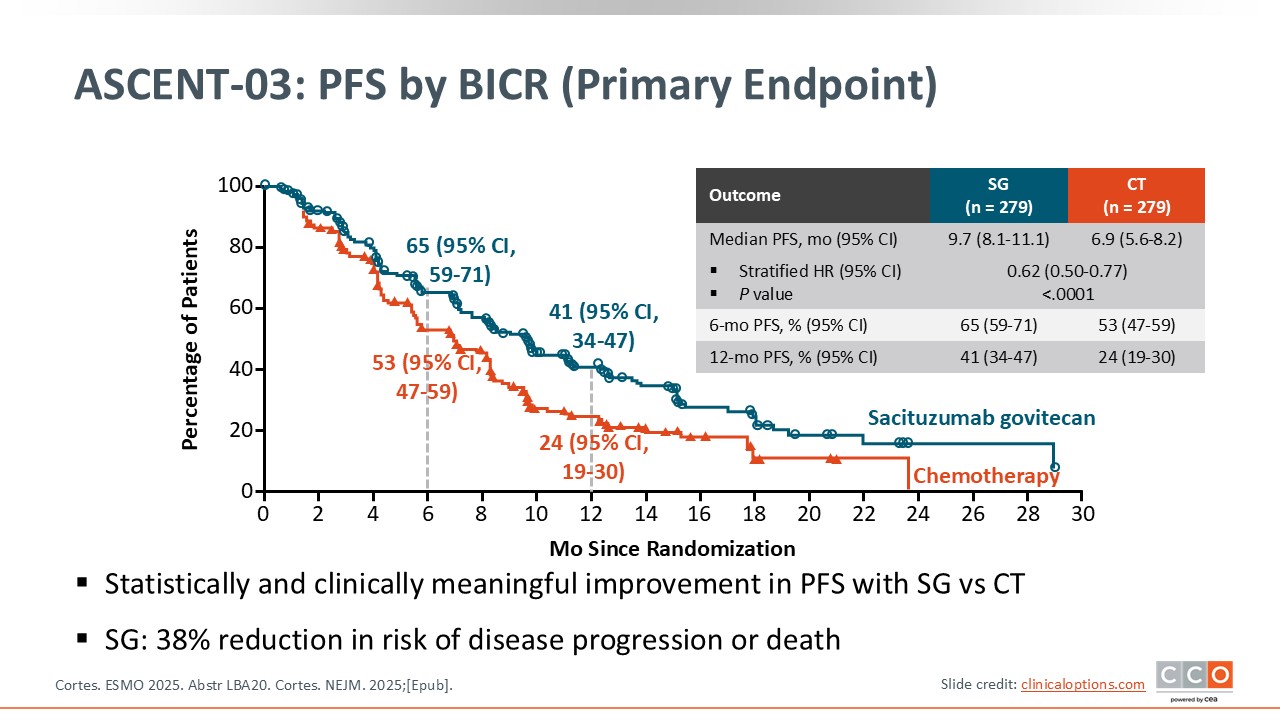
ASCENT-03: PFS by BICR (Primary Endpoint)
Cristina Saura, MD, PhD:
The primary endpoint of ASCENT-03 is PFS by BICR.25,26 The results are very clear with a statistically significant improvement in median PFS from 6.9 months with chemotherapy to 9.7 months with sacituzumab govitecan. The hazard ratio was 0.62. I agree with the authors that this is clinically meaningful for this specific patient population.
ASCENT-03: Response and Survival
Cristina Saura, MD, PhD:
The overall response rate was similar between the 2 arms, but the duration of response was longer with sacituzumab govitecan.25,26 Data for OS are immature at the time of this presentation.
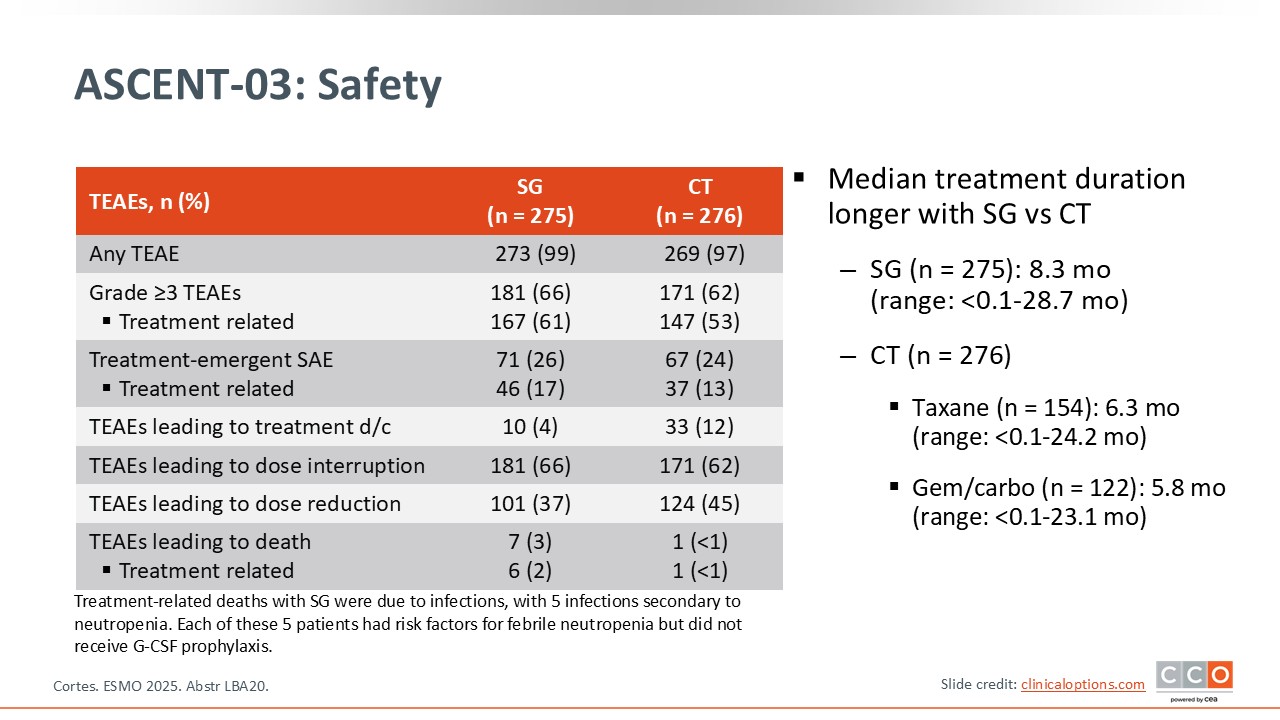
ASCENT-03: Safety
Cristina Saura, MD, PhD:
It is important to note that even with a longer duration of treatment with sacituzumab govitecan, the grade 3 or higher treatment-related AEs are very similar between both arms.25,26 There were more discontinuations and dose reductions with chemotherapy.
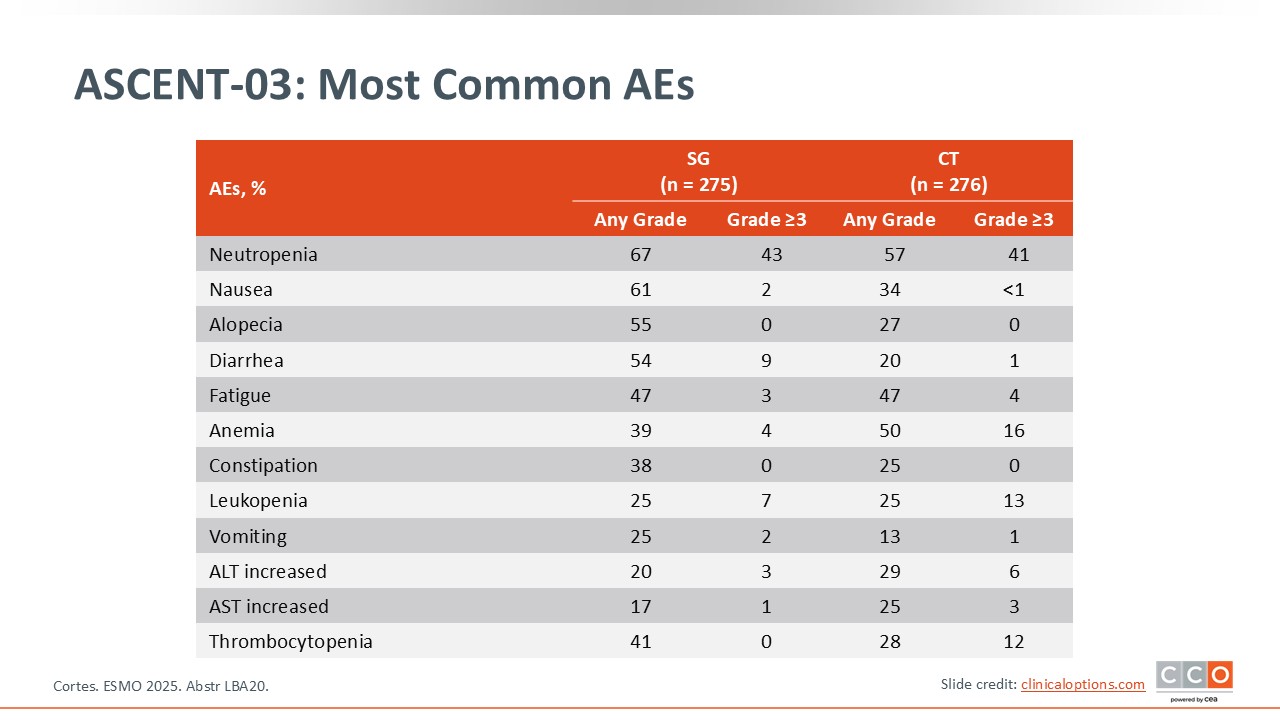
ASCENT-03: Most Common AEs
Cristina Saura, MD, PhD:
The safety profile of sacituzumab govitecan is very well known, mainly neutropenia and diarrhea, and that was observed in this trial.25,26 There are no new safety concerns with sacituzumab govitecan.
TROPION-Breast02: Dato-DXd vs Investigator’s Choice of CT as First-line Treatment for TNBC Not Eligible for Immune Checkpoint Inhibitors
Cristina Saura, MD, PhD:
We will now discuss the phase III TROPION-Breast02 trial of the TROP-2 ADC, Dato-Dxd.27 This trial compared the efficacy and safety of Dato-DXd to investigator’s choice of chemotherapy in the frontline setting for patients with advanced TNBC. Similar to the ASCENT-03 trial, patients had to be ineligible for immunotherapy.25,26 Dato-DXd is administered intravenously every 3 weeks. Only single-agent chemotherapy was allowed in the comparator arm. Unlike ASCENT-03, there was no minimum disease-free interval for patients to enroll on the TROPION-Breast02 trial, thus some of the patients included had a poor prognosis due to relapse less than 6 months after treatment in the early disease setting. Dual primary endpoints were OS and PFS by BICR.
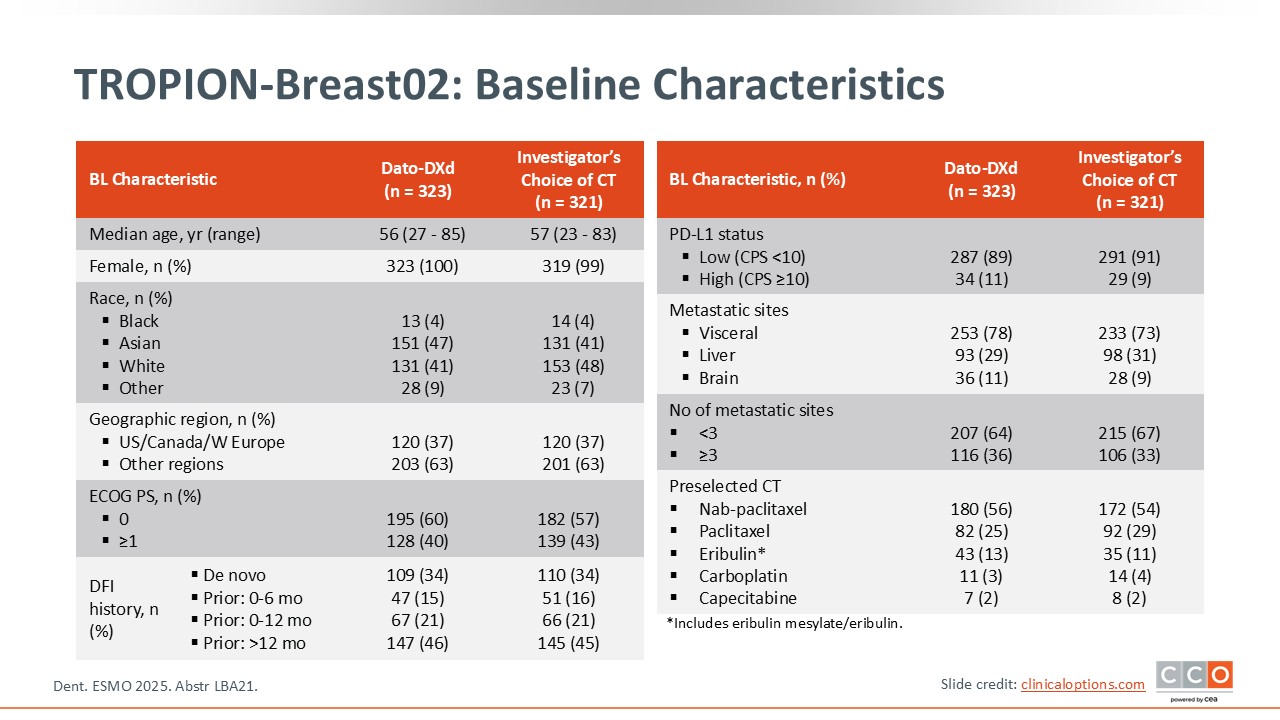
TROPION-Breast02: Baseline Characteristics
Cristina Saura, MD, PhD:
Among the patients randomized to the Dato-DXd arm, 34% had de novo disease, 15% had experienced a relapse between 0 and 6 months, 21% experienced a relapse from 0-12 months, and 46% had a disease-free interval of more than 12 months.27 Patients on the chemotherapy arm had similar characteristics.
With respect to preselection of chemotherapy in the comparator arm, 54% selected nab-paclitaxel, 29% received paclitaxel, and 11% received eribulin.
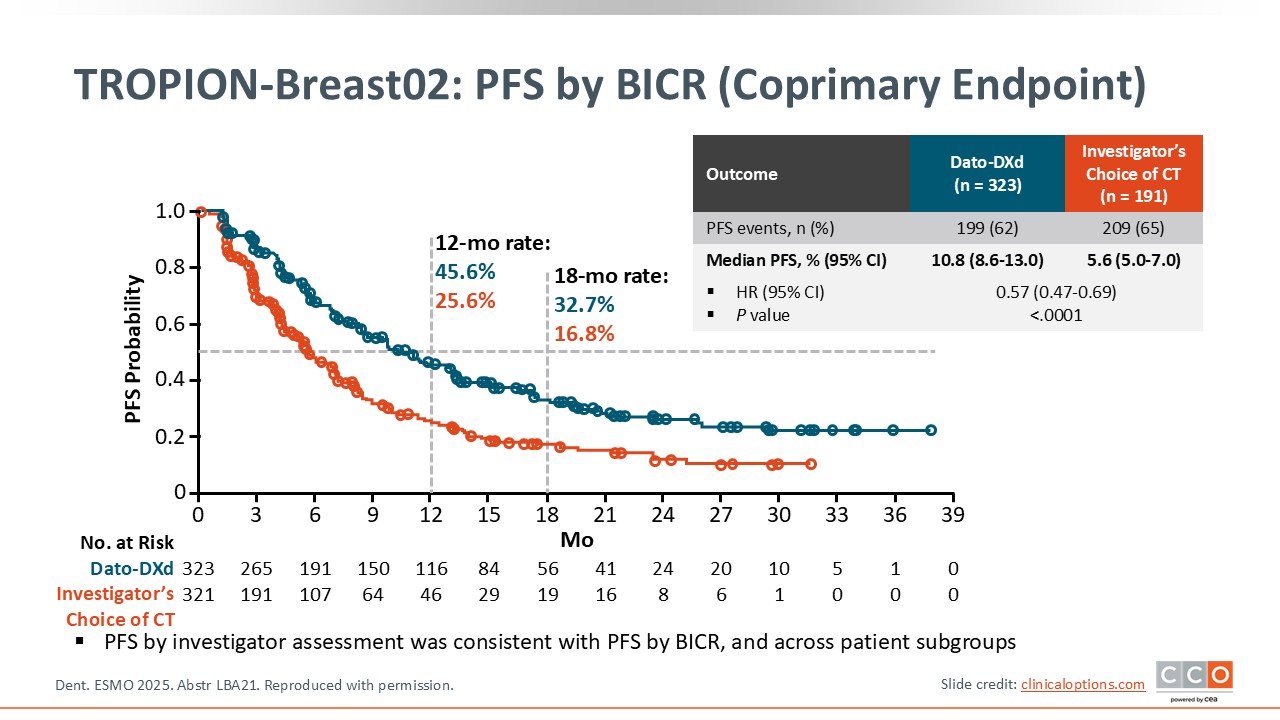
TROPION-Breast02: PFS by BICR (Coprimary Endpoint)
Cristina Saura, MD, PhD:
The coprimary endpoint of PFS was positive with a median PFS of 10.8 months with Dato-DXd vs 5.6 months with chemotherapy.27 The hazard ratio was 0.57 and was statistically significant. This is a clinically meaningful result.
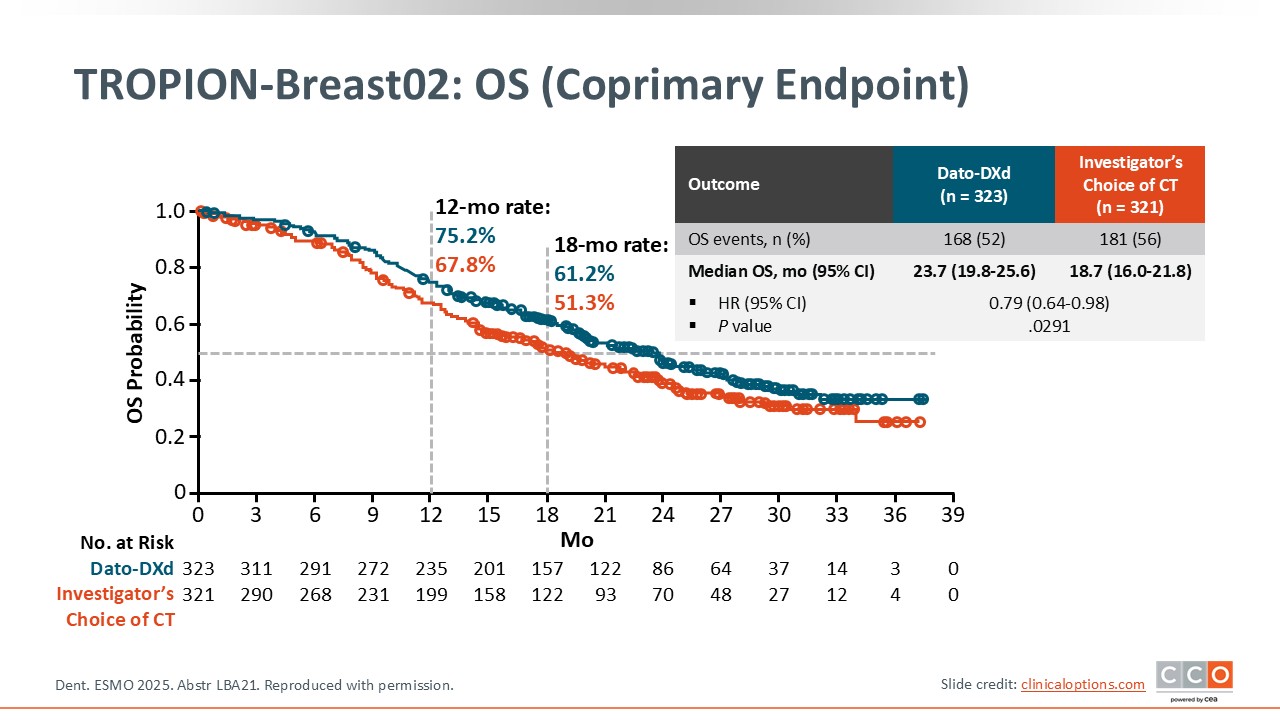
TROPION-Breast02: OS (Coprimary Endpoint)
Cristina Saura, MD, PhD:
The other coprimary endpoint of OS was also statistically significant with a median OS of 23.7 months with Dato-DXd vs 18.7 months with chemotherapy.27 The hazard ratio was 0.79. Again, this is clinically significant for our patients.
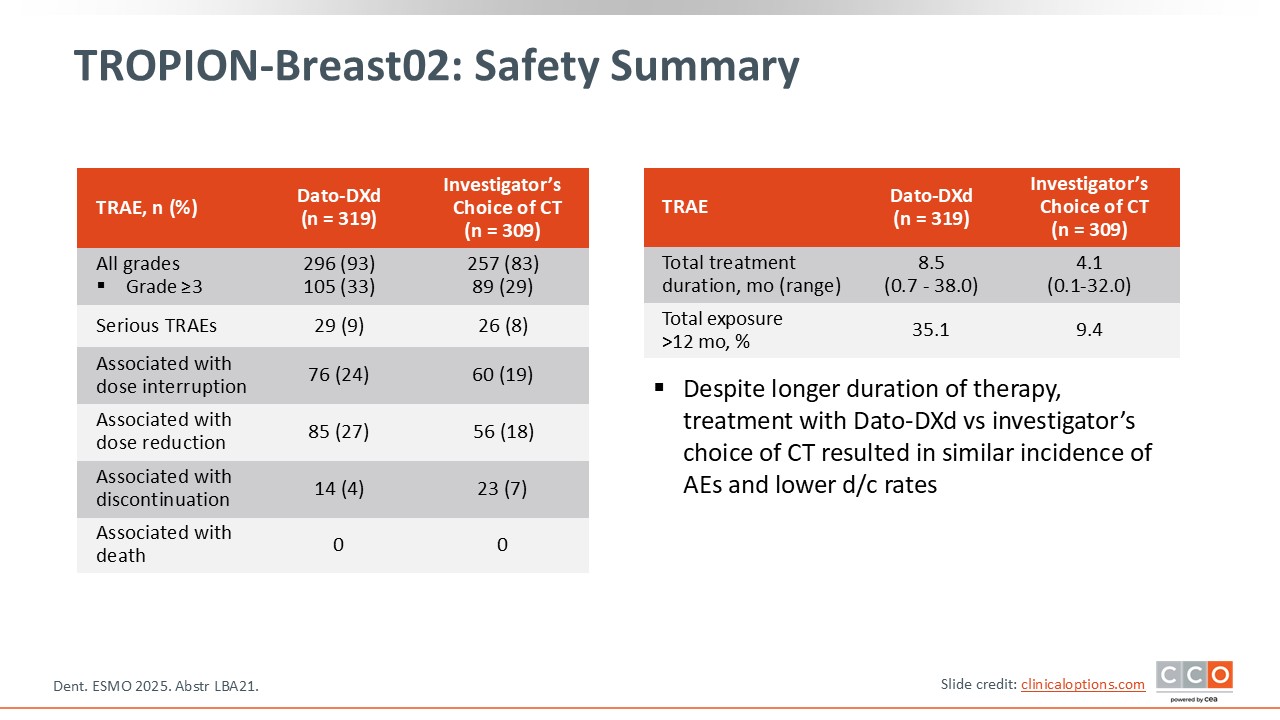
TROPION-Breast02: Safety Summary
Cristina Saura, MD, PhD:
The rate of grade 3 or higher AEs is similar in both arms with no increase in toxicity with a longer treatment duration for Dato-DXd.27 Dose interruptions and reductions were higher with Dato-DXd but the rate of treatment discontinuation is similar.
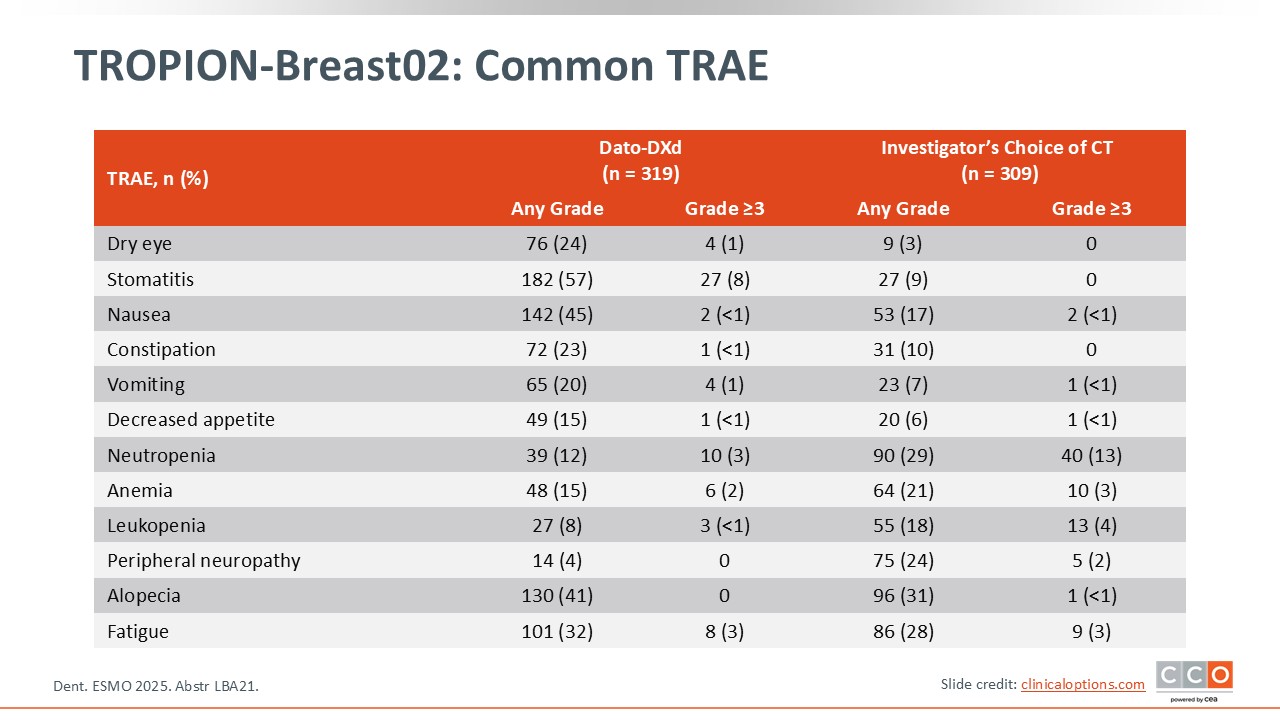
TROPION-Breast02: Common TRAE
Cristina Saura, MD, PhD:
The majority of patients (57%) receiving Dato-DXd experienced any-grade stomatitis.27 Grade 3 or greater events occurred in 8% of patients. This is an important safety concern that should be addressed with the use of primary prophylaxis and monitoring. Additional AEs of note with Dato-DXd included nausea (45% any grade) and dry eye (24% any grade).
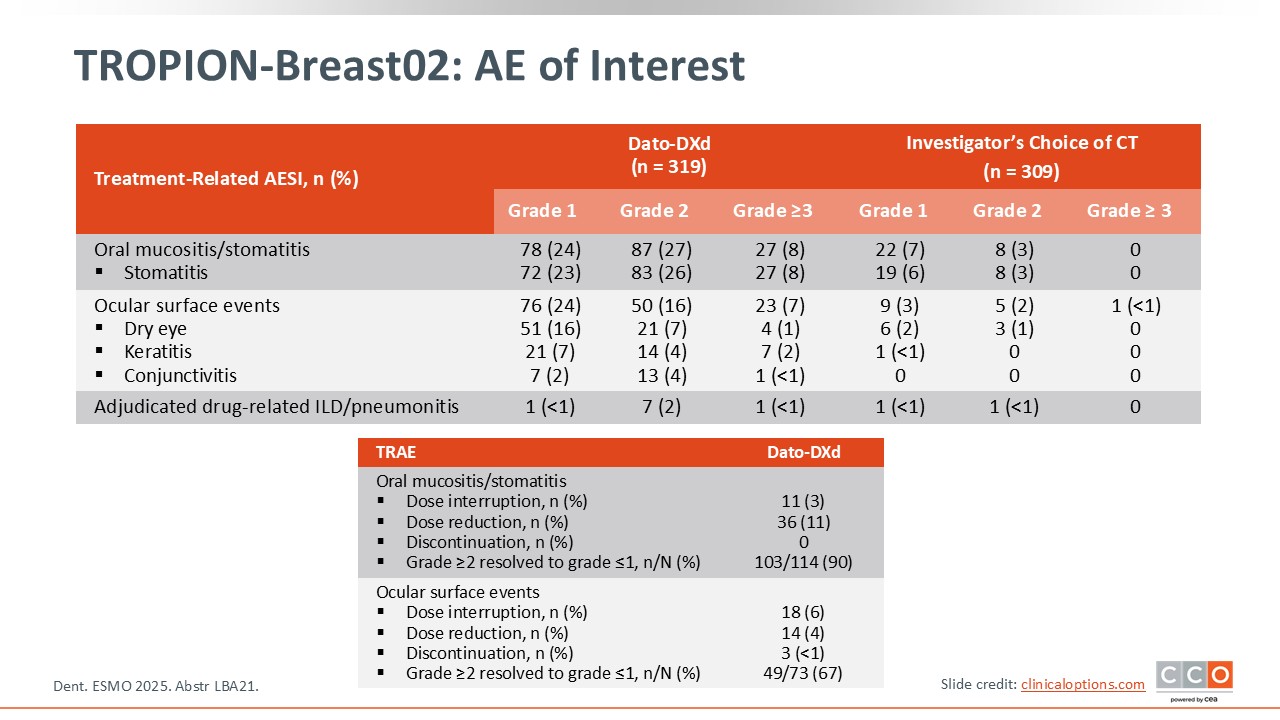
TROPION-Breast02: AE of Interest
Cristina Saura, MD, PhD:
This slide offers further details about the rate of AEs of interest including stomatitis, ocular events, and ILD.27 Oral mucositis/stomatitis led to both dose interruptions and reductions but no discontinuations. Ocular AEs led to dose interruptions, reductions, and treatment discontinuation. These data reinforce the need to identify and treat these toxicities promptly.
ASCENT-03 and TROPION-Breast02: Conclusions
Cristina Saura, MD, PhD:
I think that the overall implication for both trials is that we have in front of us a new SoC for patients with advanced TNBC ineligible for immunotherapy. We have seen the revolution of immunotherapy for those patients with metastatic TNBC and PD-L1–positive disease. But for those patients with PD-L1–negative tumors or who are unable to receive PD-L1 therapy, the SoC for first-line therapy will change with the results of these 2 trials. I expect to have these treatments approved soon to improve the outcomes of patients in the clinic. I want to reinforce that we know the safety profile of both drugs very well and it will be very important to be aware of that and treat or prevent the toxicities to maximize the benefit of either therapy.
With respect to choosing between the 2 ADCs, I think that we will have to consider patient comorbidities and other practical issues such as dosing schedule since Dato-DXd is given every 3 weeks and sacituzumab govitecan is given on Days 1 and 8 every 3 weeks. IN addition, the OS data for the ASCENT-03 trial are not yet mature. I would say that there is no single issue that will guide the decision on selecting Dato-DXd vs sacituzumab govitecan. We will integrate the OS data with sacituzumab govitecan when available, patient comorbidities, and the safety profile of each drug to decide which is the best for each patient.
Paolo Tarantino, MD, PhD:
I think that the results from both ASCENT-03 and TROPION-Breast02 are immediately practice changing. The choice between sacituzumab govitecan and Dato-DXd will likely be based on the different toxicity profiles of these agents and the differences in treatment schedule.
Compared to sacituzumab govitecan there are advantages and disadvantages with Dato-DXd. The key advantages are the positive OS results, administration only once every 3 weeks, and less neutropenia with no requirement for growth factor support. Dato-DXd does come with a different toxicity profile including stomatitis, ocular toxicity, and ILD, albeit infrequent.
Overall, examining these results from the 2 trials, I do feel that the TROPION-Breast02 data were slightly stronger compared to the ASCENT-03 data in terms of improvement of PFS, objective response rate, OS, and reduced life-threatening toxicities. I will have a slight preference for Dato-DXd in this setting, but I acknowledge that the main choice will be based on toxicities and patient preferences.
In terms of second-line treatment options once TROP2 ADCs are established in the first line, there have been some real-world studies conducted in this field. A recent retrospective study found that sequencing an ADC with a topoisomerase-1 payload after a prior topoisomerase-1 ADC, in this case it was sacituzumab govitecan after T-DXd, leads to cross-resistance.28 Whereas if you switch mechanism of action and use standard chemotherapy after T-DXd, this seems to retain meaningful activity.
Considering these results, I do not think that sequencing ADCs back-to-back is a good approach. This is why among patients who progress on first-line sacituzumab govitecan or Dato-DXd, I would favor utilizing chemotherapy, either a microtubule inhibitor like a taxane or eribulin, or even a platinum-based treatment. Then I would consider another topoisomerase-1 ADC later, either T-DXd if the disease is HER2-low or the other TROP2 ADC not used in the first line.
ASCENT-04/KEYNOTE-D19: First-line Sacituzumab Govitecan + Pembrolizumab vs CT + Pembrolizumab in PD-L1–Positive Advanced TNBC
Cristina Saura, MD, PhD:
The phase III ASCENT-04/KEYNOTE-D19 trial compared the efficacy of sacituzumab govitecan plus pembrolizumab to chemotherapy plus pembrolizumab in patients with treatment-naive, PD-L1–positive, advanced TNBC.29 The primary endpoint of the trial was PFS by BICR and as reported previously, it was positive. The median PFS with sacituzumab plus pembrolizumab was 11.2 months vs 7.8 months with chemotherapy plus pembrolizumab. The hazard ratio was 0.65 (95% CI: 0.51-0.84, P <.001). The OS data were immature. The secondary endpoint of patient-reported outcomes was presented at ESMO 2025.30
ASCENT-04/KEYNOTE-D19 PROs: Time to Deterioration in Physical Functioning
Cristina Saura, MD, PhD:
The time to first deterioration in physical functioning was similar in the 2 treatment arms.30 A sensitivity analysis of time to first deterioration showed a numerically longer time to deterioration with sacituzumab govitecan vs chemotherapy suggesting a potential for a delay in the onset of deterioration of physical functioning using sacituzumab govitecan.
ASCENT-04/KEYNOTE-D19 PROs: Time to Deterioration Across EORTC QLQ-C30 Domains
Cristina Saura, MD, PhD:
When the authors examined the time to deterioration by EORTC QLQ-C30 domains, several scores favor the use of sacituzumab govitecan plus pembrolizumab, specifically emotional functioning, pain, and insomnia.30 I think that it is not only important to see an improvement in median PFS, but also this improvement in quality of life for the patients treated with frontline sacituzumab govitecan plus pembrolizumab vs chemotherapy plus pembrolizumab.
ASCENT-04/KEYNOTE-D19: Conclusions
Paolo Tarantino, MD, PhD:
The efficacy data from ASCENT-04 already changed practice by bringing the combination of sacituzumab govitecan and pembrolizumab into the first-line setting for PD-L1–positive metastatic TNBC.29
Patient-reported outcomes and quality of life were mostly preserved with sacituzumab govitecan plus pembrolizumab vs chemotherapy plus pembrolizumab, with the exception of a few points. As expected based its toxicity profile, there was more deterioration in terms of gastrointestinal domains for the sacituzumab govitecan arm with more nausea, vomiting, and diarrhea. But it was interesting to see better emotional functioning and pain in the sacituzumab govitecan arm, suggesting that when the drug is efficacious, you can reduce the pain compared to chemotherapy and improve emotional functioning. These data reinforce that gastrointestinal AEs and neutropenia—although that is not captured here—are more common with sacituzumab govitecan compared to traditional chemotherapy. However, sacituzumab govitecan can be beneficial if it is able to control the disease for longer, both in terms of pain and in terms of the patient’s emotional state. For other data points related to quality of life, there is not much difference with chemotherapy.
Whenever there is an aggressive disease like TNBC, using a highly potent drug can be beneficial. However, it is important to remember and to remind the patients that every drug we use comes with some expected costs in terms of toxicity. In this case, these costs are mostly related to gastrointestinal side effects, which are usually manageable with supportive treatment. Managing the neutropenia associated with sacituzumab govitecan is a little more challenging. It requires identifying patients who are more prone to developing neutropenia, which is extremely important to prevent febrile neutropenia and sepsis. I will utilize these results to explain to patients what to expect from this new treatment option.
Key Takeaways in Breast Cancer From ESMO 2025
Cristina Saura, MD, PhD:
ESMO 2025 was a fantastic congress and I think there are 3 main takeaways. The first is from the ASCENT-03 and TROPION-Breast02 trials in TNBC for patients not eligible to receive PD-L1 therapy. The results with sacituzumab govitecan and Dato-DXd in the first-line setting are ready to be used in the clinic for these patients when approved by the regulatory agencies.
The second takeaway of this congress is the arrival of T-DXd to the early stage disease setting for HER2-positive breast cancer, mainly as adjuvant therapy in the residual disease indication. I think that this is ready for use in clinical practice for patients meeting the DESTINY-Breast05 trial criteria. I find it more challenging to understand when to use T-DXd in the neoadjuvant setting based on the DESTINY-Breast11 data, but for some patients with very–high risk disease neoadjuvant T-DXd could be an option.
The third takeaway is the update from the monarchE trial with an OS benefit in HR-positive/HER2-negative breast cancer reinforcing the value of adjuvant abemaciclib for patients with high-risk disease. In addition, the updated NATALEE results continued to show an increase in IDFS in the overall population and particularly in the node-negative population. This is a very important point that definitely reinforces my management of patients with high-risk node-negative disease using adjuvant ribociclib.