CME
Key Studies in Breast Cancer: Independent Conference Coverage of the 2025 ESMO Congress
Physicians: Maximum of 1.50 AMA PRA Category 1 Credits™
European Learners: 1.50 EBAC® CE Credit
Released: December 15, 2025
Expiration: June 14, 2026
Activity
DESTINY-Breast11: Neoadjuvant T-DXd With or Without THP vs ddAC-THP in High-Risk HER2-Positive EBC
Paolo Tarantino, MD, PhD:
The phase III DESTINY-Breast11 trial addressed 2 key questions.1,2 The first question, which informs the study’s primary endpoint, is whether the anti-HER2 ADC T-DXd as neoadjuvant therapy followed by chemotherapy and dual HER2 blockade increases tumor shrinkage and complete disappearance of the tumor at surgery, defined as a pathologic complete response (pCR), compared to standard neoadjuvant chemotherapy plus dual HER2 blockade. To address this question, the investigators randomized patients with high-risk stage II or III HER2-positive breast cancer into 3 arms to receive either standard treatment with 4 doses of dose-dense doxorubicin/cyclophosphamide (ddAC) followed by taxane/trastuzumab/pertuzumab (THP), 4 cycles of T-DXd and 4 cycles of THP, or 8 cycles of T-DXd. The second question asked about the safety of substituting all or part of the standard regimen with T-DXd.
Of importance, the trial was designed with 3 treatment arms, but the T-DXd alone arm was stopped early. The reasons for closing this arm were multifactorial and included a low pCR rate unlikely to be an improvement vs the control arm and a delay in the timing of surgery. Overall, the expected practice-changing results of this trial come from the sequential therapy arms with THP.
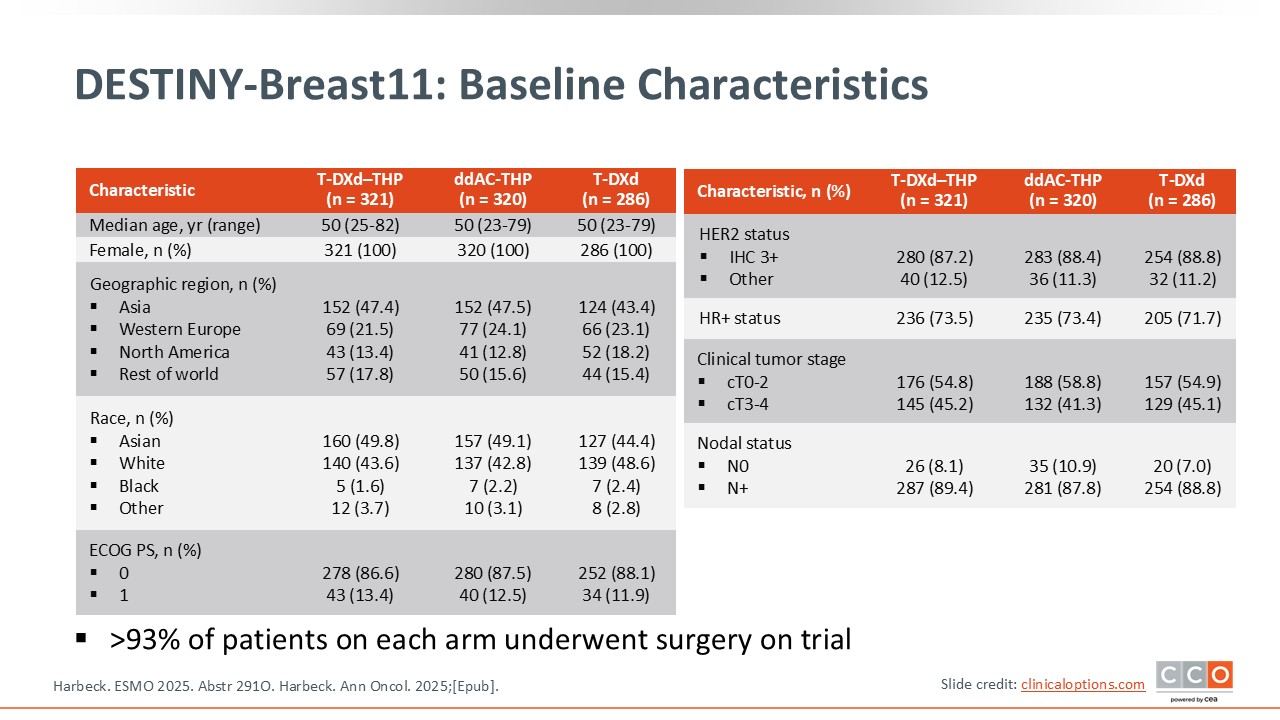
DESTINY-Breast11: Baseline Characteristics
Paolo Tarantino, MD, PhD:
I believe there are 2 key points that should be considered when interpreting the data from DESTINY-Breast11.1,2 First, this is a relatively high-risk population, meaning patients had either tumors that were clinical tumor stage ≥cT3 with any nodal status, or tumors that were node positive, and patients with inflammatory breast cancer were included. The rates of pCR in this high-risk population, with high tumor burden, tend to be lower compared to patients with lower-risk disease.
The second important thing to note is that this trial included mostly patients with HR-positive disease. Approximately three quarters of the patients enrolled had HR-positive, HER2-positive disease. Across trials it has been observed that HR-positive tumors are less likely to achieve a pCR.3,4
Combining the high-risk status of this population together with high levels of HR-positive status, we can anticipate that this population is expected to have a relatively low pCR rate compared to other trials, and we do observe this in the control arm. However, the incorporation of T-DXd in the treatment regimen enabled a relevant improvement in pCR rate.
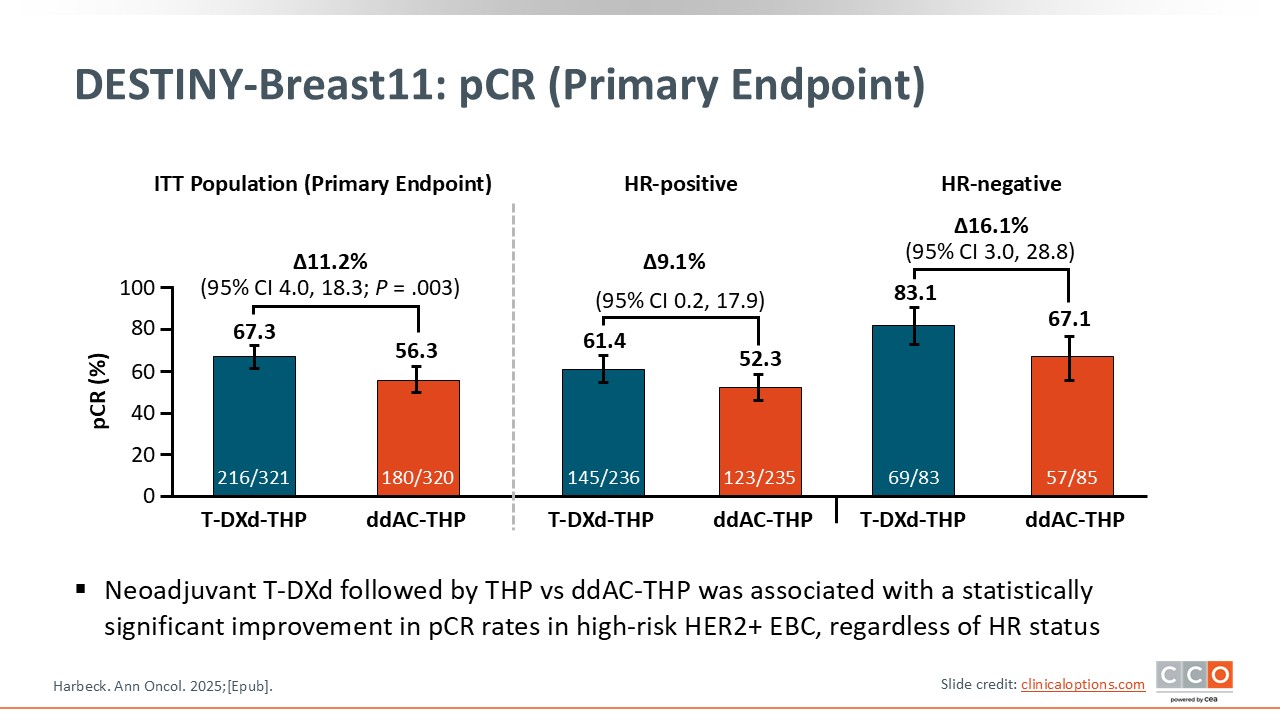
DESTINY-Breast11: pCR (Primary Endpoint)
Paolo Tarantino, MD, PhD:
The primary endpoint of the trial was pCR.1,2 At the time of surgery, 4 cycles of T-DXd followed by 4 cycles of THP increased the pCR rate compared to the control arm by 11.2%, which was statistically significant (P = .003). The pCR rate with T-DXd was 67.3% vs 56.3% with ddAC. The improvement in pCR was observed irrespective of HR status with a delta of 9% in patients with HR-positive disease and a larger delta of 16% in patients with HR-negative disease, where pCR rate was 83% with the T-DXd–THP regimen. Overall, the trial showed strong results in terms of the primary endpoint of pCR.
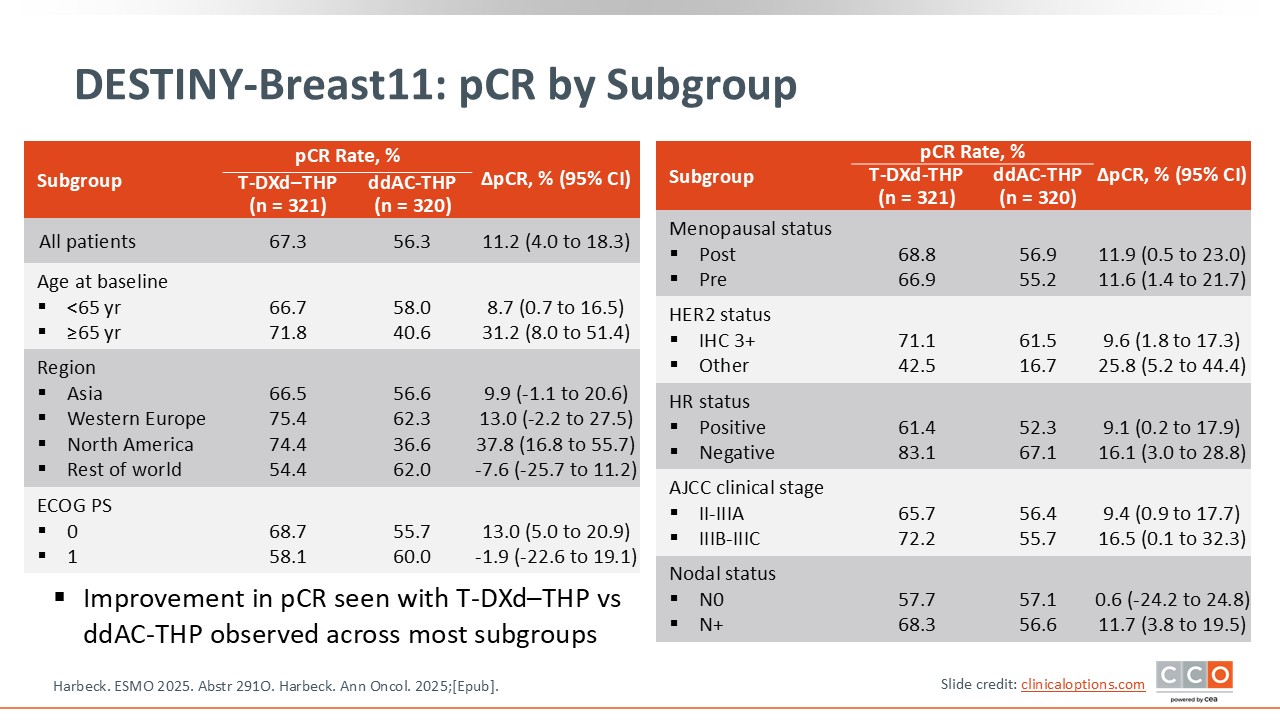
DESTINY-Breast11: pCR by Subgroup
Paolo Tarantino, MD, PhD:
In terms of subgroups, every patient seemed to benefit from T-DXd followed by THP vs ddAC followed by THP.1,2 Looking specifically at nodal status, patients with positive nodes had a greater benefit than those who were node negative. However, 90% of the population was node positive and patients with node-negative disease were a very small fraction of the study, so I would not overinterpret this finding.
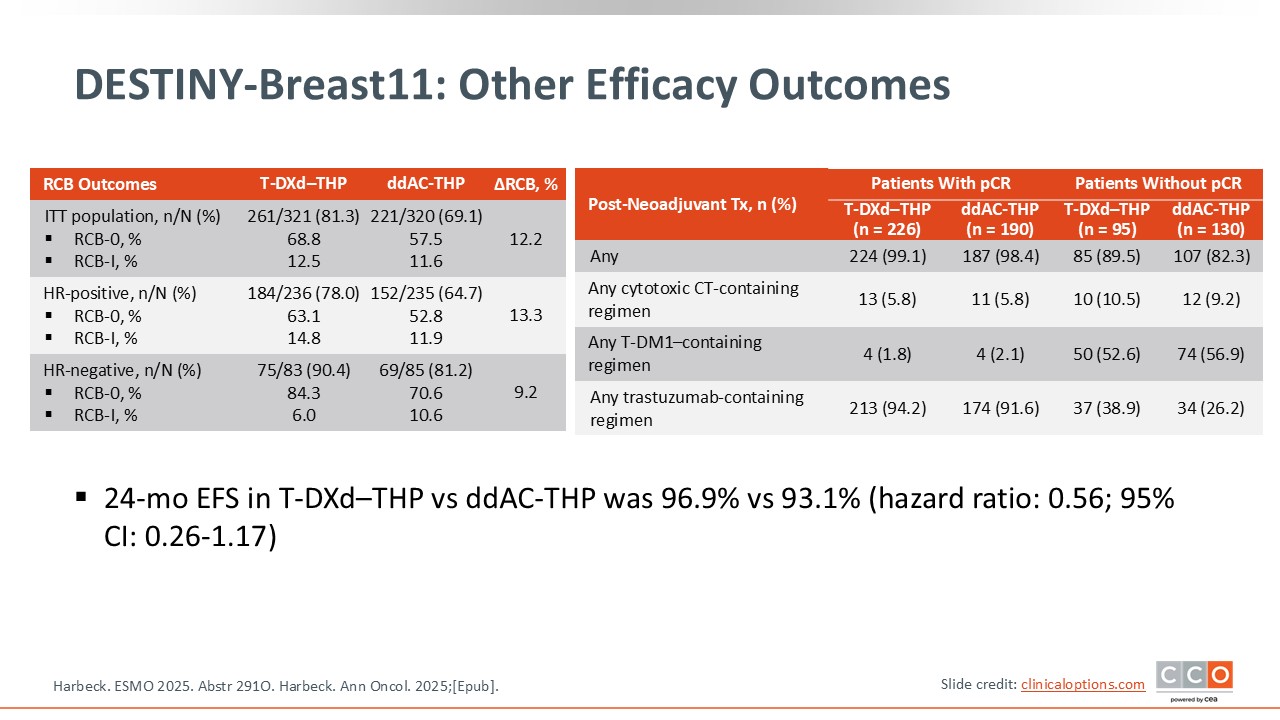
DESTINY-Breast11: Other Efficacy Outcomes
Paolo Tarantino, MD, PhD:
Examining event-free survival (EFS), a secondary endpoint, there was a positive trend with a hazard ratio of 0.56 although the data are still very immature.1,2 The curves diverge relatively early and at 2 years after surgery, approximately 97% of the patients are free from recurrence events or death with T-DXd–THP, compared to 93% in the control arm. It is still too early to interpret but it is promising to see that EFS is trending in favor of T-DXd–THP.
The post-neoadjuvant treatment that patients received is of importance to interpret this study. Approximately half of the patients who did not achieve pCR received post-neoadjuvant T-DM1. However, more than 40% of patients did not receive T-DM1 if they had residual disease. This may impact EFS and OS interpretation as we continue to follow the patients in this trial.
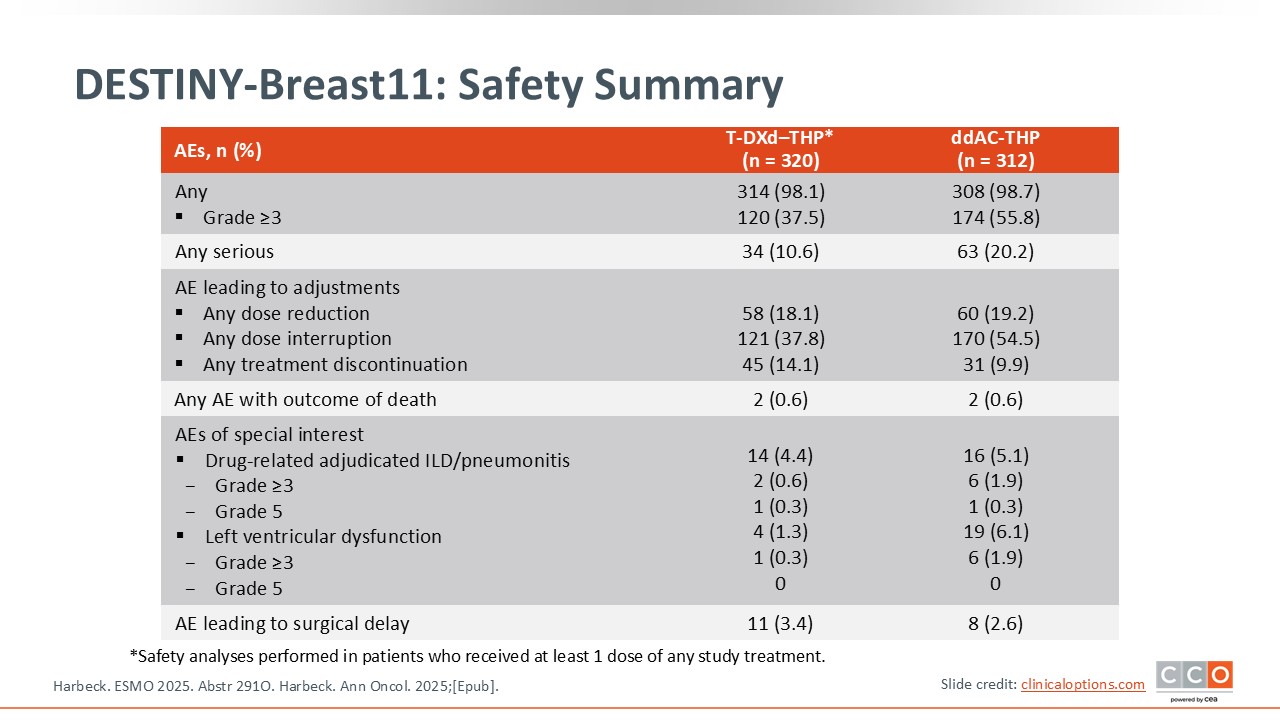
DESTINY-Breast11: Safety Summary
Paolo Tarantino, MD, PhD:
In terms of safety, it is important to note that there was less cardiotoxicity with T-DXd compared to the standard regimen.1,2 The rate of left ventricular dysfunction was 6.1% with the control arm, compared to 1.3% with the T-DXd–THP arm. The rate of interstitial lung disease (ILD) was low and comparable in both arms at 5.1% in the control arm and 4.4% in the T-DXd–THP arm, with 1 grade 5 event in each arm. Overall, the safety profile of T-DXd–THP was favorable with fewer grade 3 adverse events (AEs) and a lower rate of treatment interruptions vs ddAC-THP.
As a reminder, the T-DXd monotherapy arm was closed early. In this arm investigators found a pCR rate of 43%, which was lower than the other arms, while EFS was comparable to the control arm. In terms of safety, we know that T-DXd alone is usually associated with less peripheral neuropathy compared to taxanes and less cardiotoxicity compared to anthracyclines. Overall, T-DXd alone showed strong results, but not better than standard sequential treatment.
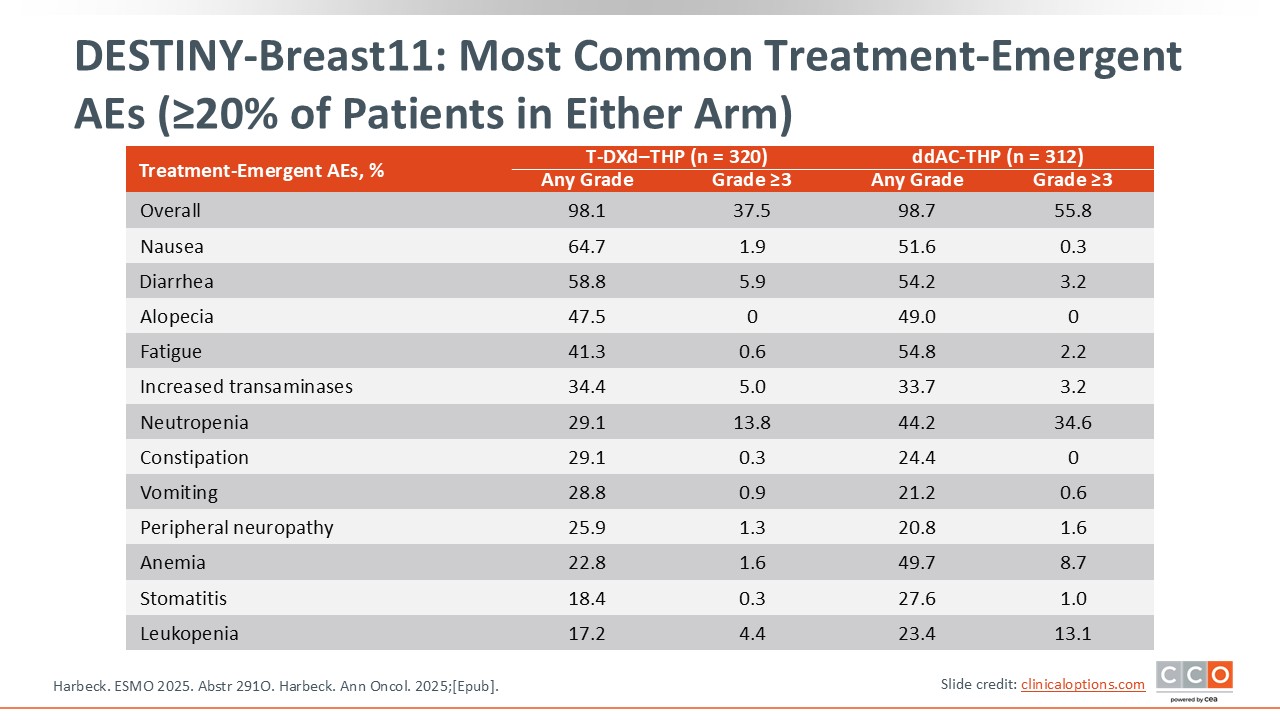
DESTINY-Breast11: Most Common Treatment-Emergent AEs (≥20% of Patients in Either Arm)
Paolo Tarantino, MD, PhD:
There were fewer grade ≥3 AEs with T-DXd–THP vs ddAC-THP, including hematologic AEs.1,2 Gastrointestinal toxicity was similar except for a slightly higher rate of grade ≥3 nausea with T-DXd.
DESTINY-Breast11: Conclusions
Paolo Tarantino, MD, PhD:
Ideally, we would have wanted the trial to be powered based on EFS as well as pCR. However, examining these data in the context of the DESTINY-Breast05 trial of adjuvant T-DXd that found a benefit in terms of IDFS, one can expect that T-DXd in the early-stage setting for HER2-positive disease has an impact on reducing the rate of recurrence and in DESTINY-Breast11 in improving pCR rates.
I do feel that these results are practice changing and introduce T-DXd in the neoadjuvant setting for moderate- to high-risk HER2-positive early-stage breast cancer. That said, we will need to determine if all patients meeting the criteria of this trial actually need T-DXd, or if we could have a selective approach to screen patients with lower-risk disease with THP and then decide which patients require escalation with T-DXd. Overall I do think that these results are practice changing and bring T-DXd sequenced with THP as a new option in the neoadjuvant setting for high-risk HER2-positive breast cancer.
Cristina Saura, MD, PhD:
I think that it is difficult to see how we will change our practice based on the results of the DESTINY-Breast11 trial without integrating the results of the DESTINY-Breast05 trial. DESTINY-Breast11 found that neoadjuvant T-DXd followed by THP increased pCR, but this was a very specific population with high-risk HER2-positive disease. As noted, the trial included a higher than expected number of patients with HR-positive disease, more than 70%. This trial offers a new option, but I agree that we are still to define which patients are best treated upfront with T-DXd, beyond the clinical inclusion criteria of DESTINY-Breast11.
Considering the results of the DESTINY-Breast05 trial, some patients could receive the SoC neoadjuvant therapy and if they do not achieve a pCR, then receive T-DXd in the adjuvant setting. I am eager to see biomarkers from this trial to understand which patients derive more benefit from 4 cycles of neoadjuvant T-DXd vs using the SoC regimen and then receiving 14 cycles of T-DXd in the adjuvant setting if they do not achieve a pCR.
DESTINY-Breast05: Adjuvant T-DXd vs T-DM1 in High-Risk HER2+ EBC With Residual Invasive Disease After Neoadjuvant Therapy
Paolo Tarantino, MD, PhD:
The phase III DESTINY-Breast05 trial is examining T-DXd as adjuvant therapy vs SoC T-DM1 as defined by the KATHERINE trial.5,6 The trial enrolled patients with HER2-positive breast cancer and residual disease after neoadjuvant treatment. To be enrolled, patients were required to have inoperable disease at baseline and/or positive axillary lymph nodes after neoadjuvant chemotherapy. DESTINY-Breast05 is asking if adjuvant T-DXd for 14 cycles in this high-risk population can reduce the risk of recurrence compared to adjuvant T-DM1 for 14 cycles with a primary endpoint of IDFS. The hope is that the results would mirror those of DESTINY-Breast03, which showed that T-DXd outperformed T-DM1 in the metastatic setting.7
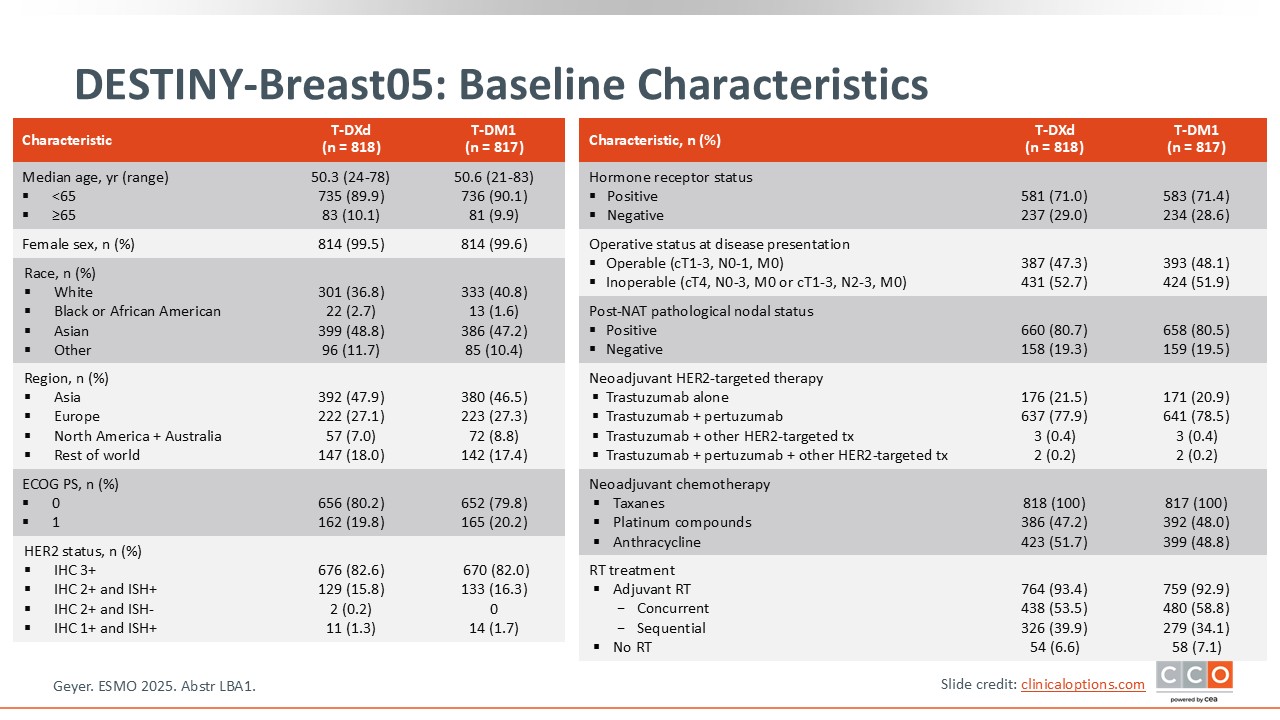
DESTINY-Breast05: Baseline Characteristics
Paolo Tarantino, MD, PhD:
A key factor of note is that DESTINY-Breast05 included a very–high risk population that was much higher risk compared to population of the KATHERINE trial.5,6 The KATHERINE trial enrolled patients with any residual disease at surgery.6 By contrast, DESTINY-Breast05 enrolled patients who had inoperable disease prior to neoadjuvant therapy—those with cT4 and any nodal status or cT1-3 with N2 or N3 disease, or if the disease was operable before starting neoadjuvant treatment, but at surgery there was node-positive residual disease. This is a very–high risk population, meaning that the results of DESTINY-Breast05 apply to a selected population and not to the broader population from the KATHERINE trial.
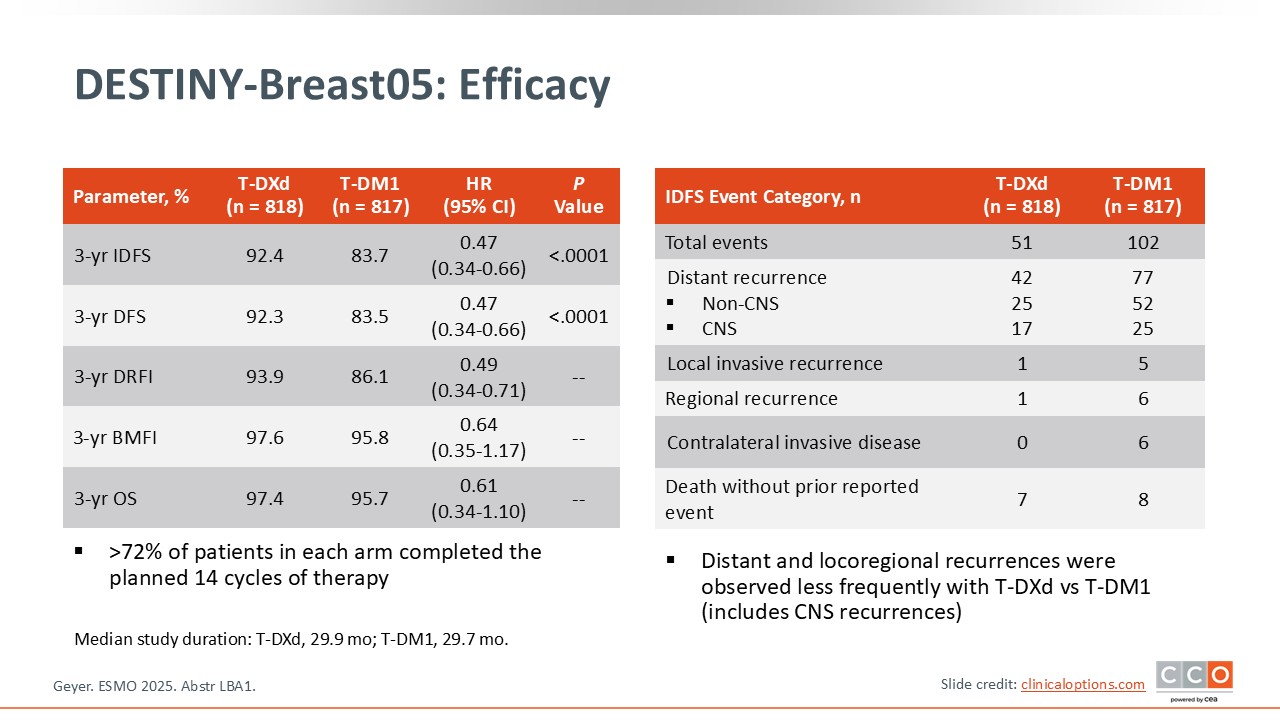
DESTINY-Breast05: Efficacy
Paolo Tarantino, MD, PhD:
The 3-year IDFS with T-DXd was 92.4% vs 83.7% with T-DM1 with a hazard ratio of 0.47, which was highly statistically significant with a delta of 8.7%.5 This was better than what was expected, according to the statistical design. A key secondary endpoint, disease-free survival (DFS), was also significantly higher with T-DXd. It is outstanding to see such favorable IDFS and DFS in such a high-risk population.
In addition, there was a numerical decrease in central nervous system metastases with T-DXd. There is hope that T-DXd may have prevented or delayed brain metastases observed in this patient population. Other secondary endpoints, including distant recurrence–free interval were improved and most of the recurrences prevented were distant recurrences. OS results are immature but are trending in favor of T-DXd with a hazard ratio of 0.61.
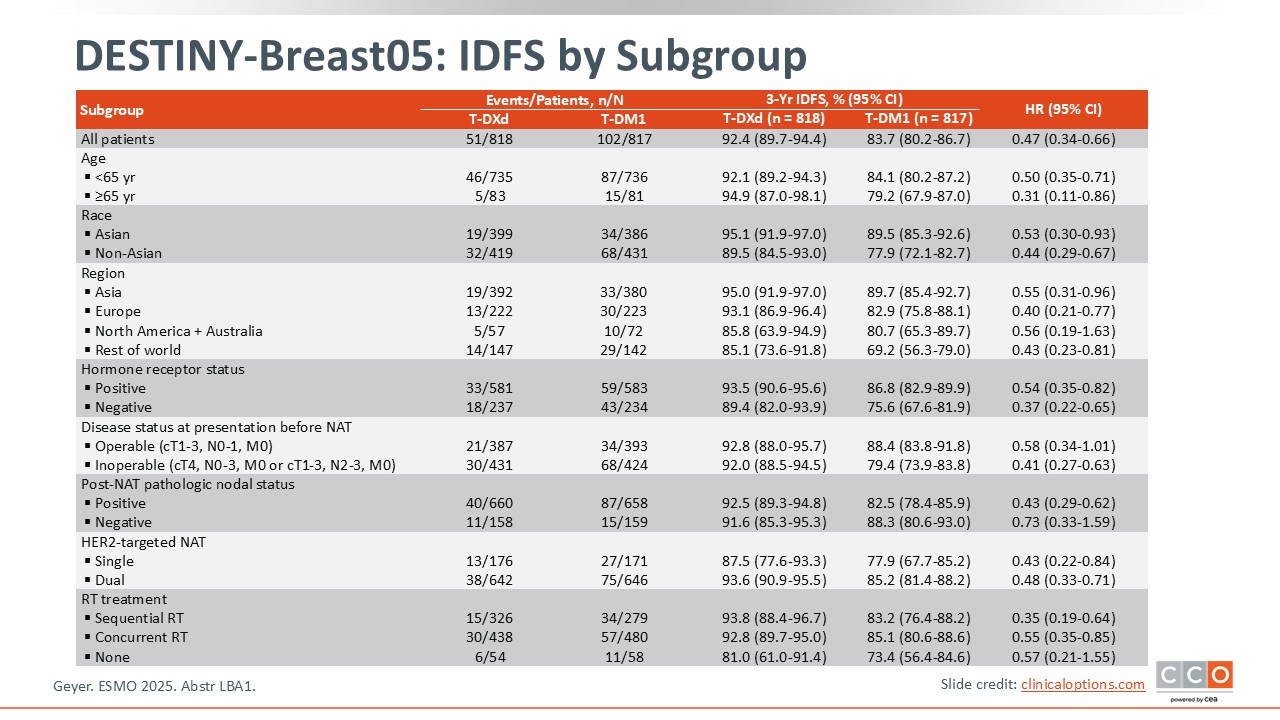
DESTINY-Breast05: IDFS by Subgroup
Paolo Tarantino, MD, PhD:
IDFS favored T-DXd in all subgroups analyzed.
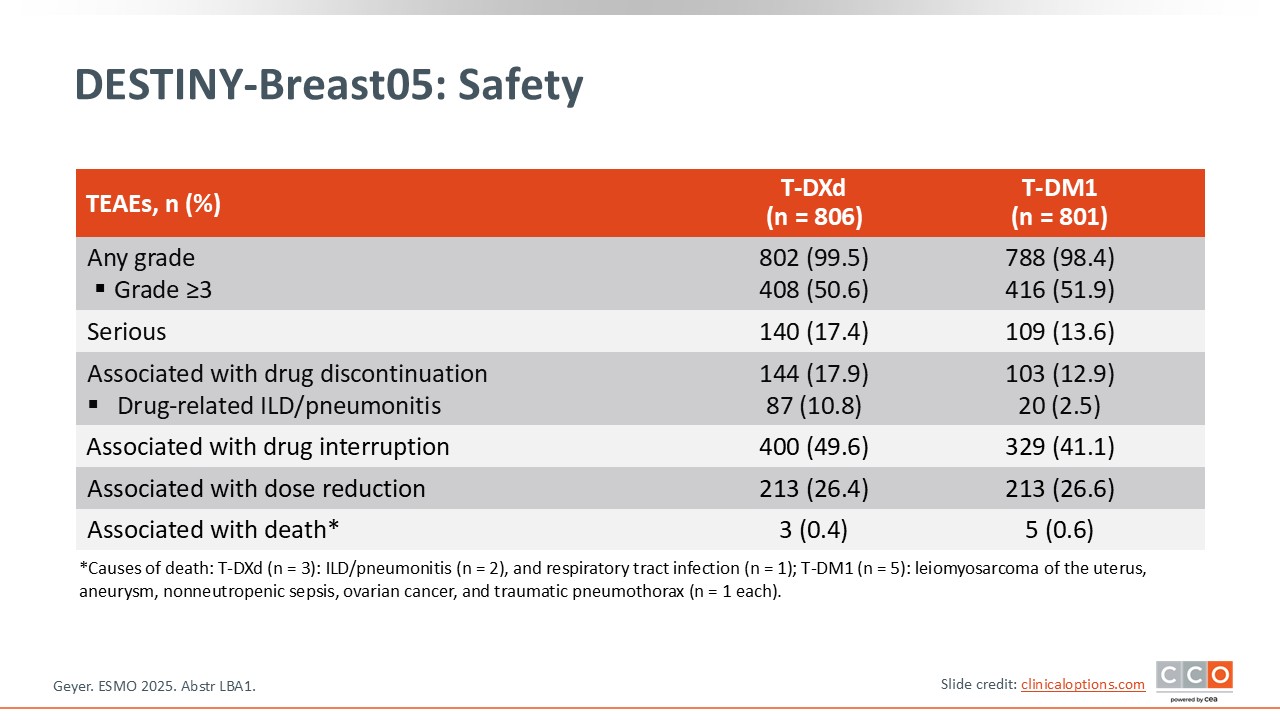
DESTINY-Breast05: Safety
Paolo Tarantino, MD, PhD:
In terms of safety, the majority of patients in both arms completed the 14-cycle treatment course, but approximately 25% in the T-DM1 arm and 30% in the T-DXd arm did not.5 The rate of ILD with T-DXd was approximately 11%, and there were 2 deaths related to ILD with T-DXd, which is a reminder of how important it is to monitor for lung toxicity when patients are receiving this drug.
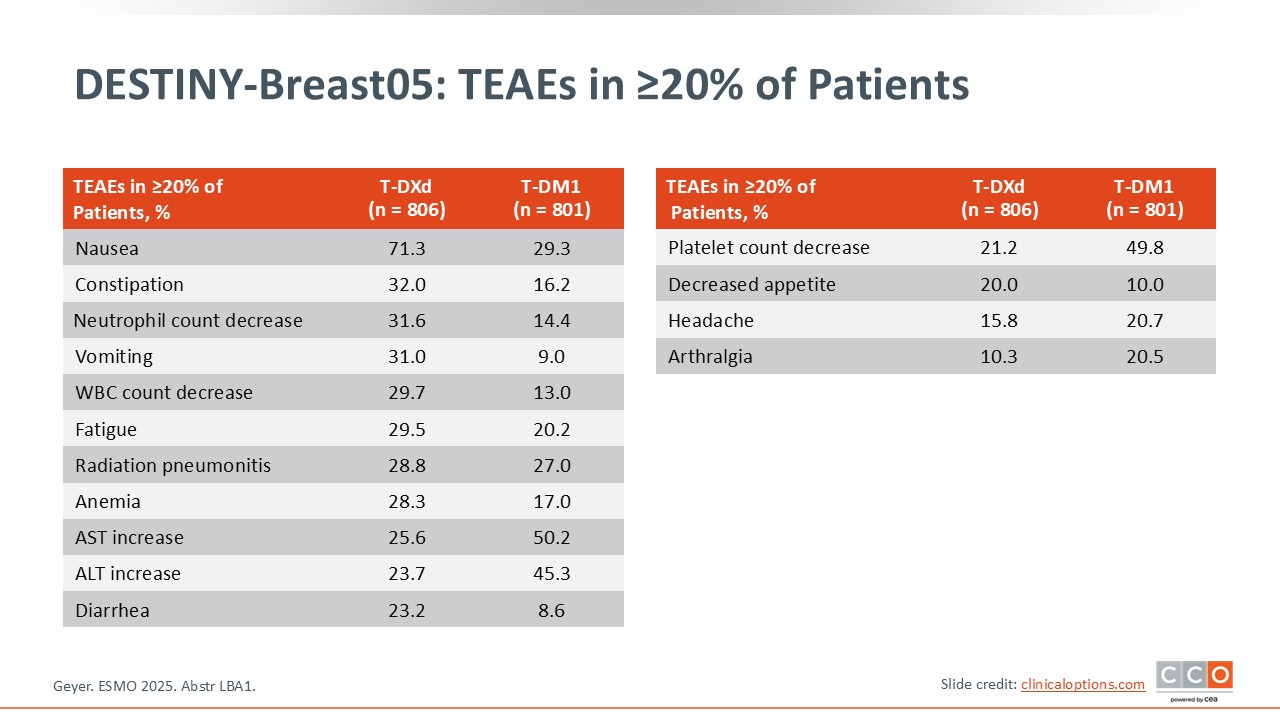
DESTINY-Breast05: TEAEs in ≥20% of Patients
Paolo Tarantino, MD, PhD:
The safety profile was similar to that previously established for T-DXd with no new safety signals.5
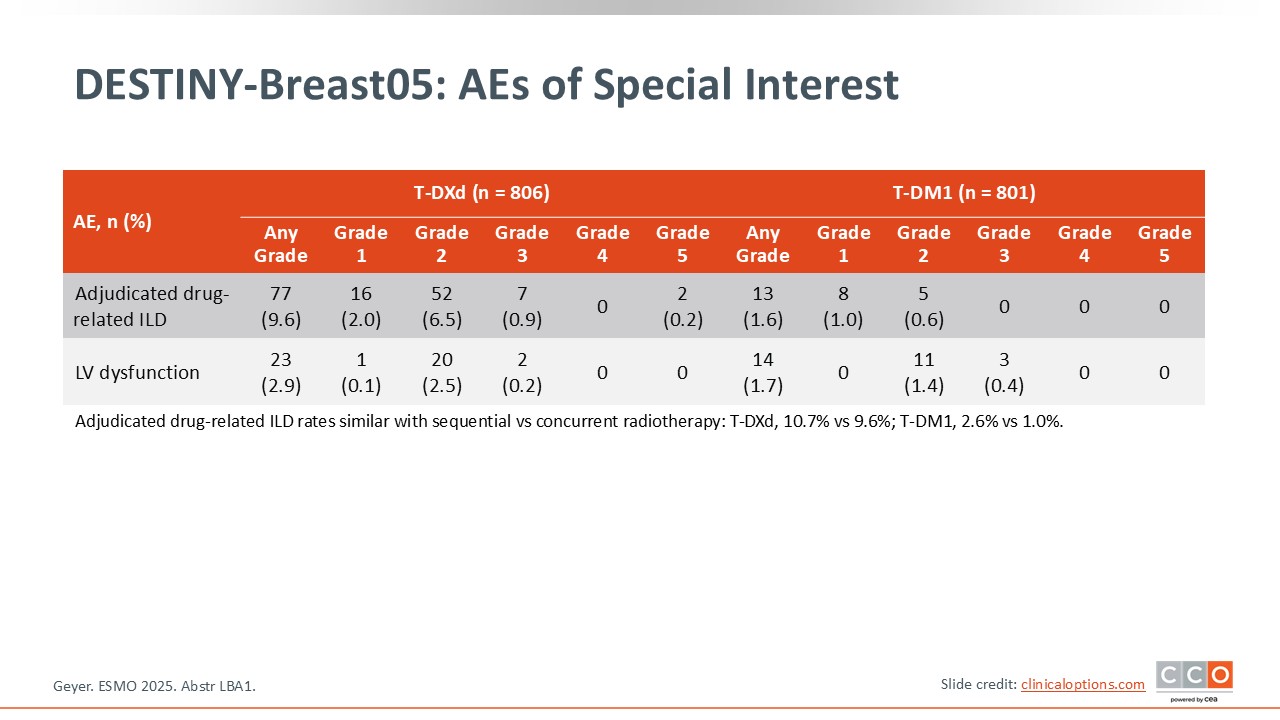
DESTINY-Breast05: AEs of Special Interest
Paolo Tarantino, MD, PhD:
The administration of radiation either concomitantly or sequentially with adjuvant therapy did not significantly affect the rate of pneumonitis. Any-grade left ventricular dysfunction was uncommon in both arms. Overall, the safety of T-DXd seems to be reasonable, despite some degree of ILD, and the results in terms of IDFS are outstanding.
DESTINY-Breast05: Conclusions
Paolo Tarantino, MD, PhD:
The results of DESTINY-Breast05 are immediately practice changing. They bring T-DXd to the clinic for patients with high-risk HER2-positive breast cancer and residual disease at surgery that is node positive or for patients with inoperable disease at baseline. This means that now we are going to have 2 ADCs that can be used in the post-neoadjuvant setting:
- T-DXd for high-risk cases, with a high disease burden at baseline or residual nodal disease post neoadjuvant therapy; or
- T-DM1 if there is a lower burden of residual disease at surgery, according to the KATHERINE trial population.
Overall, I do hope that these results mean that we are going to be able to cure many more patients in the future, despite having residual disease at surgery.
Cristina Saura, MD, PhD:
For those patients who meet the inclusion and exclusion criteria of DESTINY-Breast05, I would be eager to treat them with T-DXd if they had residual disease. Of importance is the need to focus on the population included in the DESTINY-Breast05 trial who are patients with high-risk disease, because not all patients with residual disease were included on the trial. I would be happy to use T-DXd tomorrow for those patients with high risk.
Although T-DXd is not yet approved in this setting, the IDFS benefit is very clear with a delta of approximately 9%—for this population that is very relevant—coupled with the decrease in central nervous system relapses and in distant relapse free survival. It makes me very confident about a future OS benefit for this specific population.
One more important thing that I want to stress from a practical perspective is that there was no impact on ILD events using either sequential or concomitant radiotherapy. This was a concern but does not appear to be the case.