CME
Key Studies in Breast Cancer: Independent Conference Coverage of the 2025 ESMO Congress
Physicians: Maximum of 1.50 AMA PRA Category 1 Credits™
European Learners: 1.50 EBAC® CE Credit
Released: December 15, 2025
Expiration: June 14, 2026
Activity
evERA BC: Giredestrant + Everolimus in Advanced ER+/HER2- Breast Cancer Previously Treated With a CDK4/6 Inhibitor
Paolo Tarantino, MD, PhD:
The phase III evERA BC trial compares the oral selective estrogen receptor degrader (SERD) giredestrant plus everolimus to SoC ET plus everolimus in patients with previously treated ER-positive/HER2-negative advanced breast cancer.8 This trial builds on previous trials, in particular BOLERO-2, that demonstrated that everolimus combined with ET can be beneficial in patients previously exposed to ET, leading to the approval of exemestane plus everolimus.9 Patients on the evERA BC trial must have experienced progression on ET and CDK4/6 inhibitors but received no prior chemotherapy. The primary endpoint of the trial was investigator-assessed PFS in patients with tumors harboring ESR1 mutations with a coprimary endpoint of investigator-assessed PFS in the intention-to-treat (ITT) population.
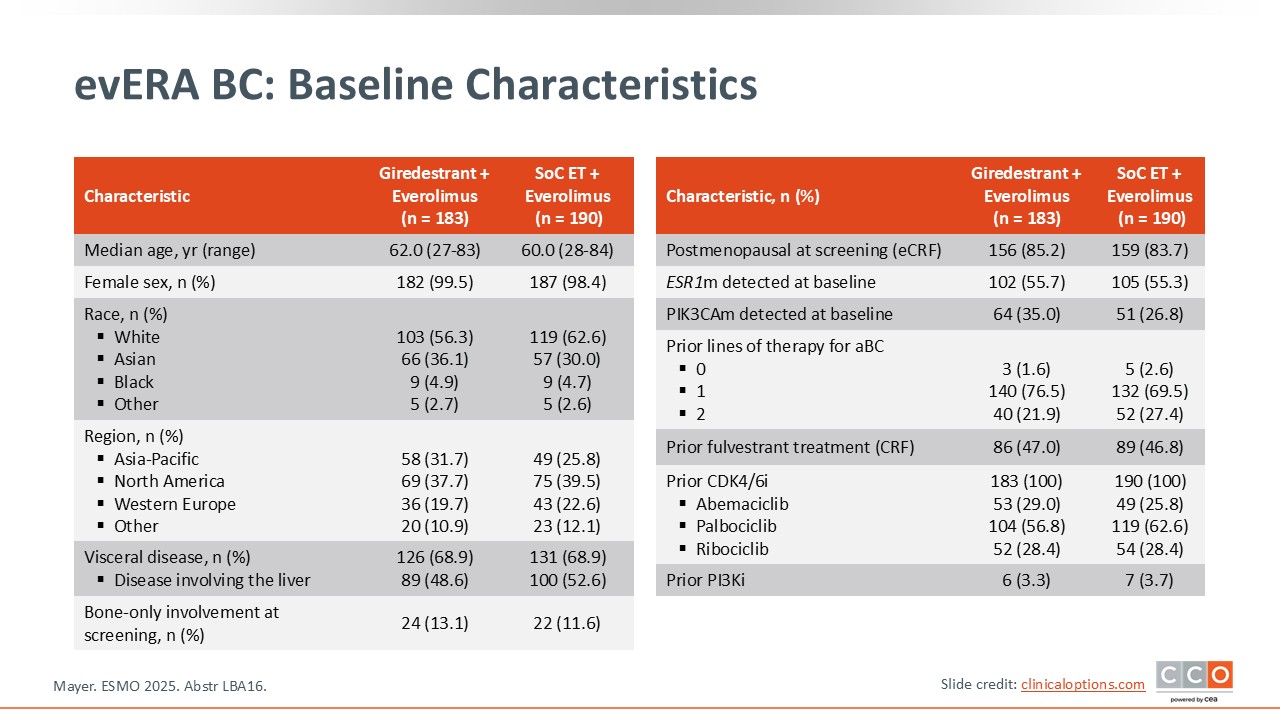
evERA BC: Baseline Characteristics
Paolo Tarantino, MD, PhD:
Of importance, the population enrolled in evERA BC was a lower-risk population compared to similar trials, because it did not allow for patients who received prior chemotherapy.8 ET was limited to 2 or fewer lines of therapy and it allowed patients with measurable disease or evaluable disease. The majority of patients had received only 1 previous line of therapy. Patients with or without ESR1 mutations were enrolled in this trial. As we review the evERA BC trial results, we should remember that in previous trials, patients with ESR1 mutations benefit more from oral SERDs.10
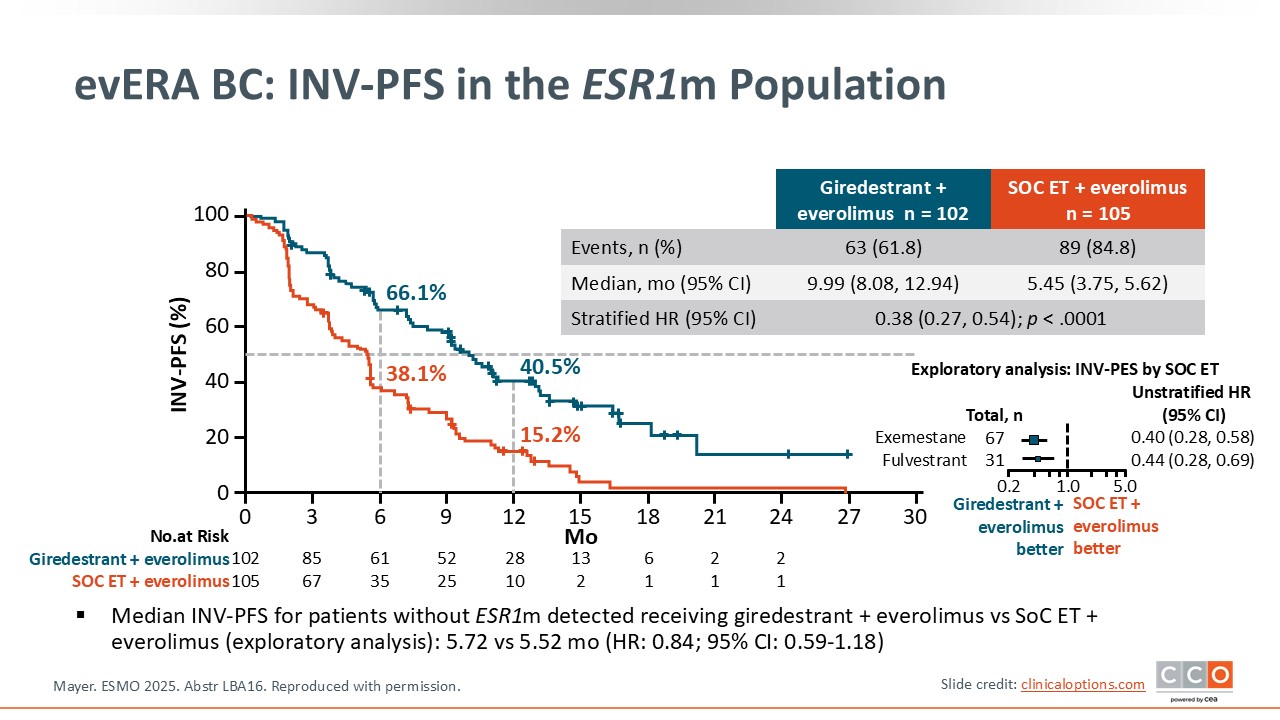
evERA BC: Investigator-Assessed PFS in the ESR1m Population
Paolo Tarantino, MD, PhD:
The PFS results show that in the ESR1-mutant population, which was approximately 55% of the study population, replacing SoC ET with giredestrant was highly beneficial.8 In the giredestrant/everolimus arm, the median PFS was 9.99 months compared to 5.45 months with SoC ET plus everolimus with a hazard ratio of 0.38, which was statistically significant. The PFS curves diverge very early and remain separate.
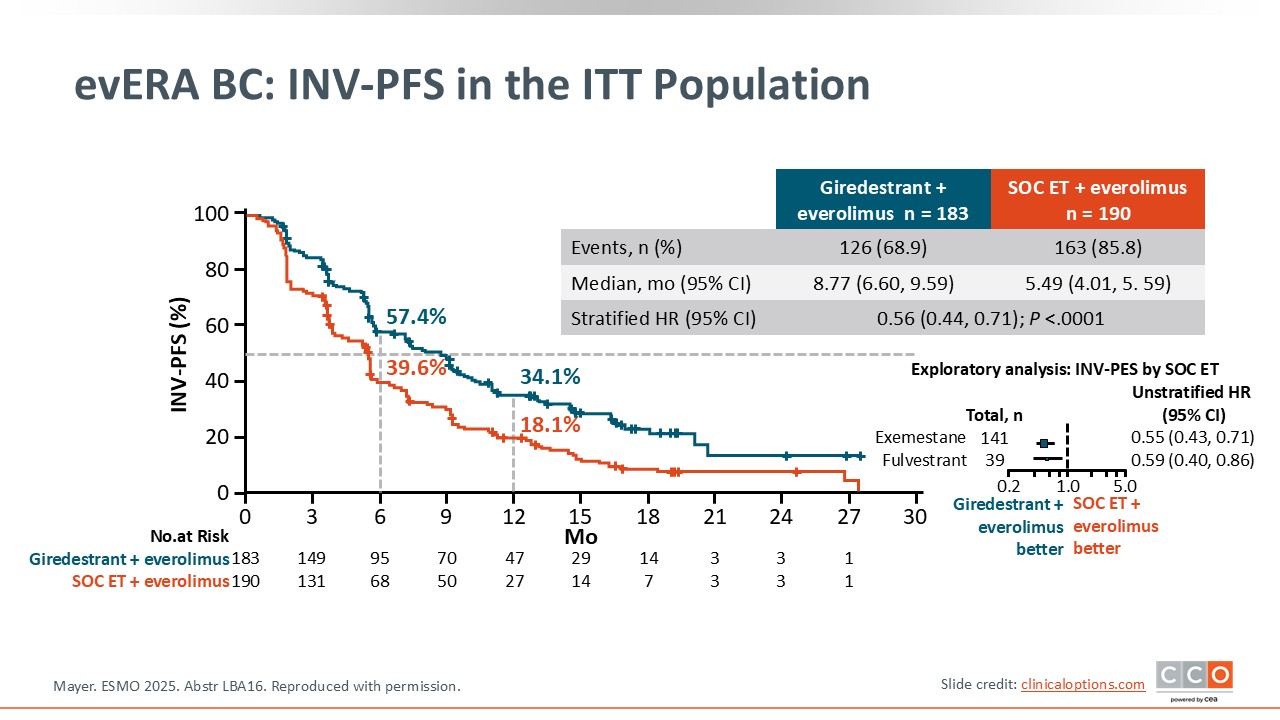
evERA BC: Investigator-Assessed PFS in the ITT Population
Paolo Tarantino, MD, PhD:
Investigator-assessed PFS in the ITT population, which was a coprimary endpoint, also shows an improvement in PFS that was statistically significant although with a smaller difference between the arms.8 The median PFS for giredestrant/everolimus is 8.8 months compared to 5.5 months in the control arm with a statistically significant hazard ratio of 0.56.
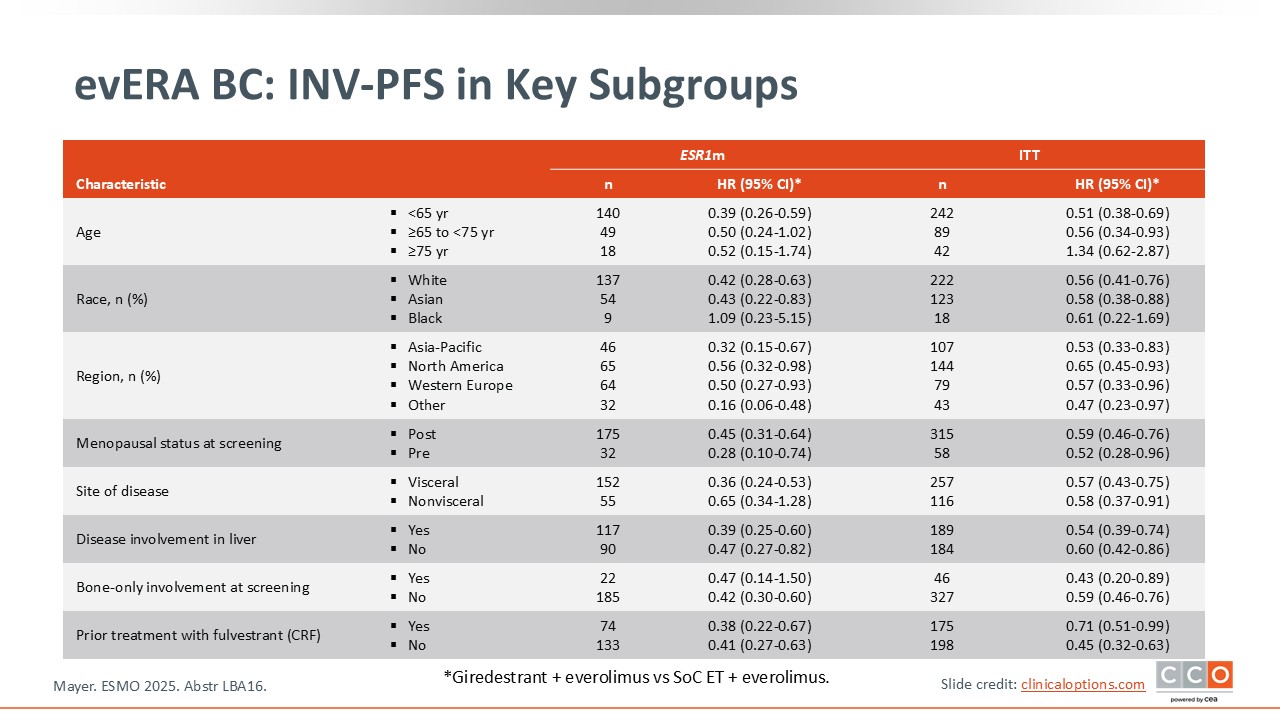
evERA BC: Investigator-Assessed PFS in Key Subgroups
Paolo Tarantino, MD, PhD:
The median PFS favored giredestrant in all subgroups analyzed.8
evERA BC: Interim OS
Paolo Tarantino, MD, PhD:
OS data are immature but favor giredestrant in the ITT population and regardless of ESR1 mutation status at this interim analysis.8 Of note, in an exploratory analysis of the population with no ESR1 mutation, which comprised approximately 45% of the patient population, no improvement in PFS was observed with a hazard ratio of 0.84. So PFS did not seem to be prolonged by giredestrant in the ESR1 wild-type cohort, although other endpoints were improved.
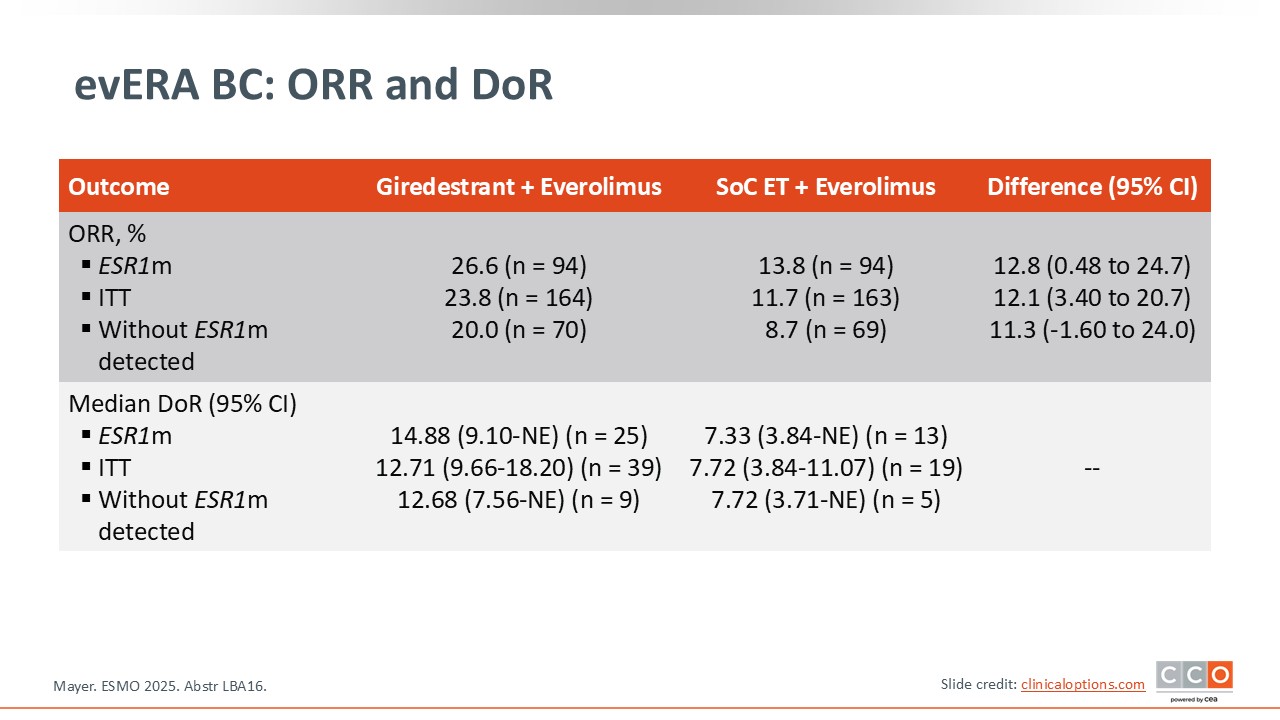
evERA BC: ORR and DoR
Paolo Tarantino, MD, PhD:
There was an improvement in overall response rate with giredestrant in patients with ESR1 mutations (from 14% to 27%) and in patients without ESR1 mutations (from 9% to 20%).8 The median duration of response is also longer with giredestrant irrespective of ESR1 status.
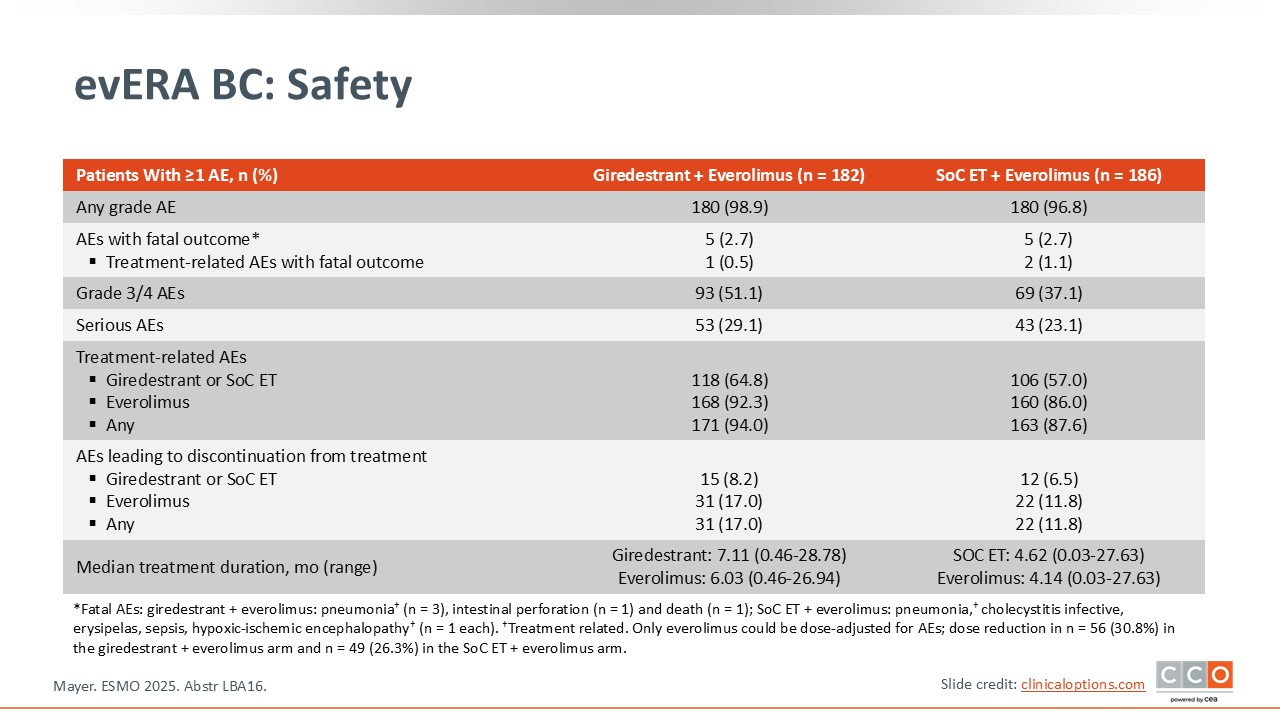
evERA BC: Safety
Paolo Tarantino, MD, PhD:
In terms of safety, giredestrant did not seem to add much toxicity and no new safety signals were detected.8
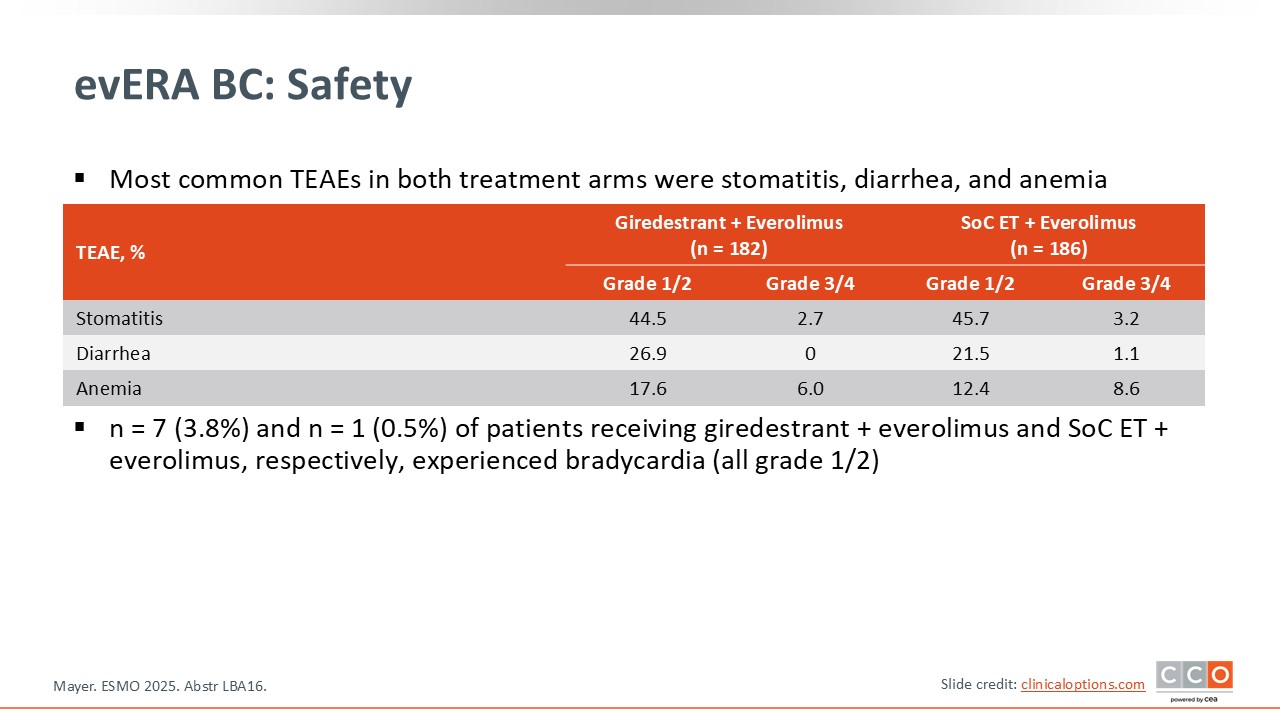
evERA BC: Safety
Paolo Tarantino, MD, PhD:
Most of the toxicities observed are associated with everolimus.8 Stomatitis was seen in approximately half of the population in both arms, although it was mostly low grade. Diarrhea, anemia, nausea, and fatigue were also observed at a similar incidence in the 2 treatment arms and are all common side effects with everolimus.
evERA BC: Conclusions
Paolo Tarantino, MD, PhD:
I think these are some of the most promising data we have in the second-line setting post CDK4/6 plus ET therapy. They clearly show that combination treatment can be beneficial even in the control arm with ET plus everolimus. The combination resulted in a median PFS of more than 5 months, but the PFS was even higher with giredestrant plus everolimus, demonstrating this is a very active combination.
These data are clearly practice changing for patients with ESR1 mutations where there was a doubling in PFS. But if the drug approval includes patients with ESR1 wild-type disease, it would not be unreasonable to also consider this combination for ESR1 wild-type, given the convenient oral formulation of both drugs, as compared to the injective route of administration of fulvestrant.
Overall, I think this combination may be more reasonable than single-agent SERD for a large percentage of patients, mostly because with combination treatment we see less evidence of rapid disease progression after the first scan. We know that with single-agent ET there are a large percentage of patients, up to 40% to 50%, who progress by the time of the first scan. In this trial with giredestrant plus everolimus, we did not observe this immediate drop. I think these are practice-changing results, particularly for patients with ESR1-mutant disease where the combination of giredestrant/everolimus will be one of the preferred treatment approaches in the future once approved.
Cristina Saura, MD, PhD:
I think the practical implications of the evERA BC trial are quite clear. Once available, I would use giredestrant plus everolimus as second-line therapy post CDK4/6 plus ET because the benefit is clear for the ITT population as well as for patients with ESR1 mutations. Outcomes were also improved for patients without ESR1 mutations. This is a very clear, elegant trial, resulting in a therapeutic combination that is useful for patients without performing a biomarker test. In addition, we have been using everolimus for many years and understand its toxicity profile quite well. Thus, with an improved PFS with giredestrant, this combination should be easy to use.
There are other SERDs under development and although some have regulatory approval for patients with ESR1 mutations, this is the first trial using an oral SERD in combination with everolimus. In the EMBER-3 trial, a combination of imlunestrant plus abemaciclib improved PFS vs standard ET regardless of ESR1 mutation status.11 Recently, imlunestrant was approved by the FDA as monotherapy for the treatment of adults with ER-positive, HER2-negative, ESR1-mutated advanced or metastatic breast cancer with disease progression following at least 1 line of ET.12 If this combination is approved based on the ITT data, this is another combination that we could use without biomarker selection. Of note, the patient populations in evERA BC and EMBER-3 were quite different, but in each trial the median PFS improvement was significant for those particular populations.
VIKTORIA-1: Gedatolisib + Fulvestrant ± Palbociclib vs Fulvestrant in PIK3CA Wild-Type HR+/HER2- Advanced Breast Cancer
Paolo Tarantino, MD, PhD:
The phase III VIKTORIA-1 trial asked if the addition of gedatolisib, a pan-PI3k/mTOR/AKT inhibitor, improved PFS when added to second-line fulvestrant or fulvestrant plus palbociclib.13 VIKTORIA-1 had 2 cohorts, 1 for patients with PIK3CA wild-type disease and another for patients with PIK3CA-mutant disease. The results presented at ESMO were for the cohort with PIK3CA wild-type disease. Patients were randomized to either fulvestrant, fulvestrant plus gedatolisib, or triplet therapy with fulvestrant, palbociclib, and gedatolisib.
Of importance, gedatolisib is administered IV once per week for 3 weeks followed by 1 week off. By contrast the evERA BC regimen was fully oral,8 whereas the regimen for VIKTORIA-1 included fulvestrant by intramuscular injection, oral palbociclib, and IV gedatolisib.
The coprimary endpoints of the trial were PFS by blinded independent central review (BICR) for the triplet vs fulvestrant and fulvestrant plus gedatolisib vs fulvestrant.
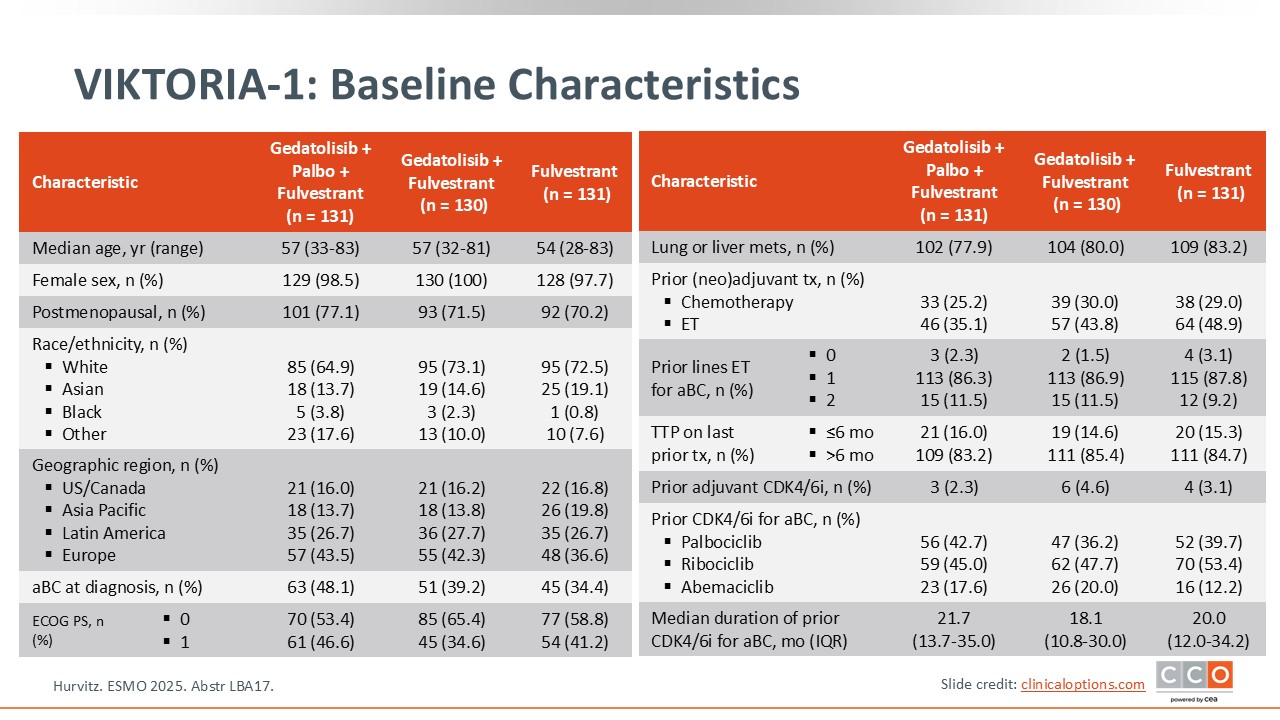
VIKTORIA-1: Baseline Characteristics
Paolo Tarantino, MD, PhD:
It is important to point out some similarities and differences between the patient populations in the previous trial, evERA BC, and VIKTORIA-1.8,13 Both trials included patients pretreated with CDK4/6 inhibitors with a maximum of 2 lines of prior ET and no previous chemotherapy. However, since gedatolisib can lead to hyperglycemia, VIKTORIA-1 did not enroll patients with type 1 diabetes or with type 2 diabetes and a hemoglobin A1C greater than 6.4%. Patients also had to have measurable disease by RECIST 1.1, which means a higher-risk population in VIKTORIA-1 compared to evERA.
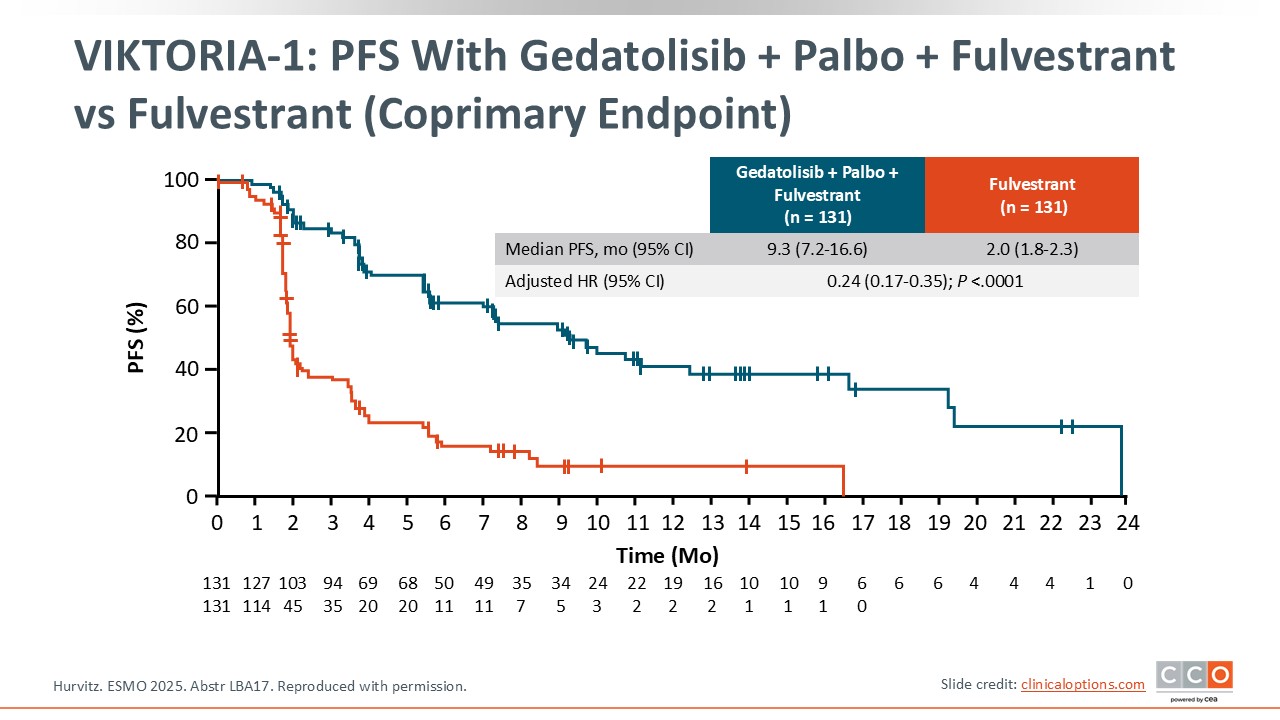
VIKTORIA-1: PFS With Gedatolisib + Palbo + Fulvestrant vs Fulvestrant (Coprimary Endpoint)
Paolo Tarantino, MD, PhD:
Triplet therapy with gedatolisib, fulvestrant, and palbociclib significantly improved PFS vs fulvestrant alone with a median PFS of 9.3 months vs 2.0 months with fulvestrant monotherapy, with a hazard ratio of 0.24, which was highly statistically significant.13 The curves separate very early. There was clear evidence of rapid progression at the first scans with fulvestrant but this was prevented with the triplet.
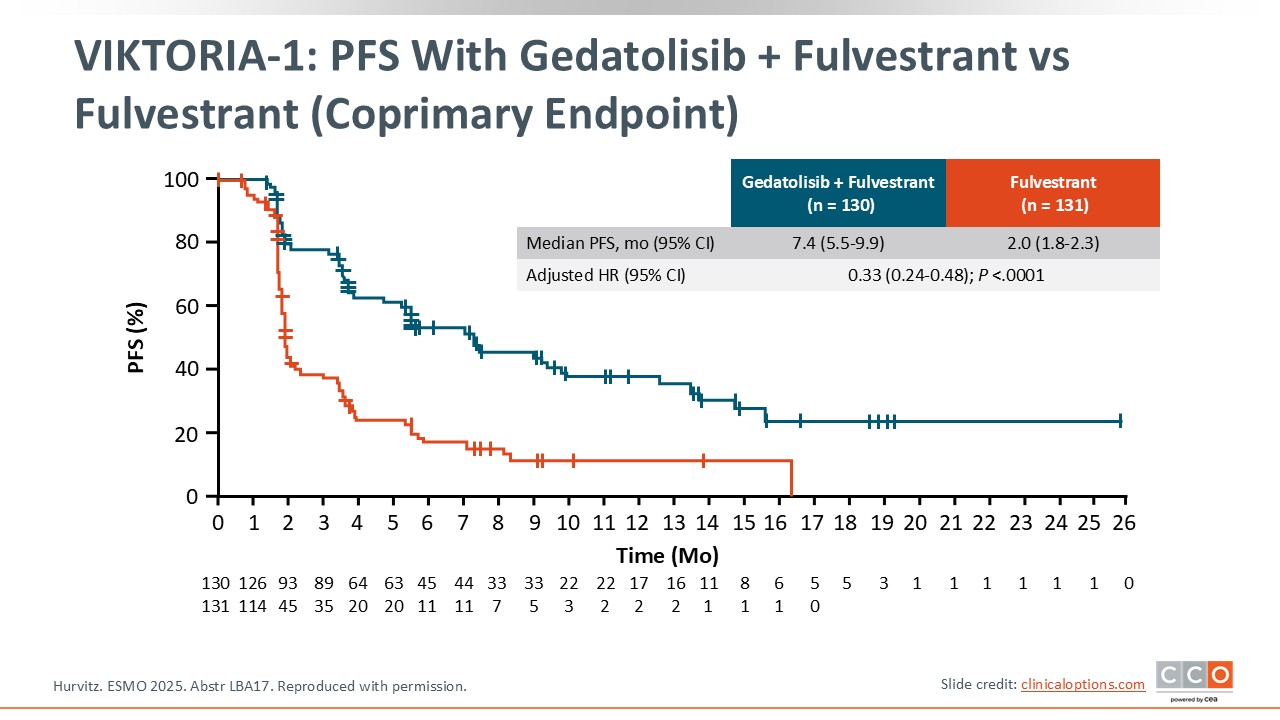
VIKTORIA-1: PFS With Gedatolisib + Fulvestrant vs Fulvestrant (Coprimary Endpoint)
Paolo Tarantino, MD, PhD:
Median PFS with gedatolisib plus fulvestrant was significantly higher at 7.4 months vs 2.0 months with fulvestrant, with a hazard ratio of 0.33.13
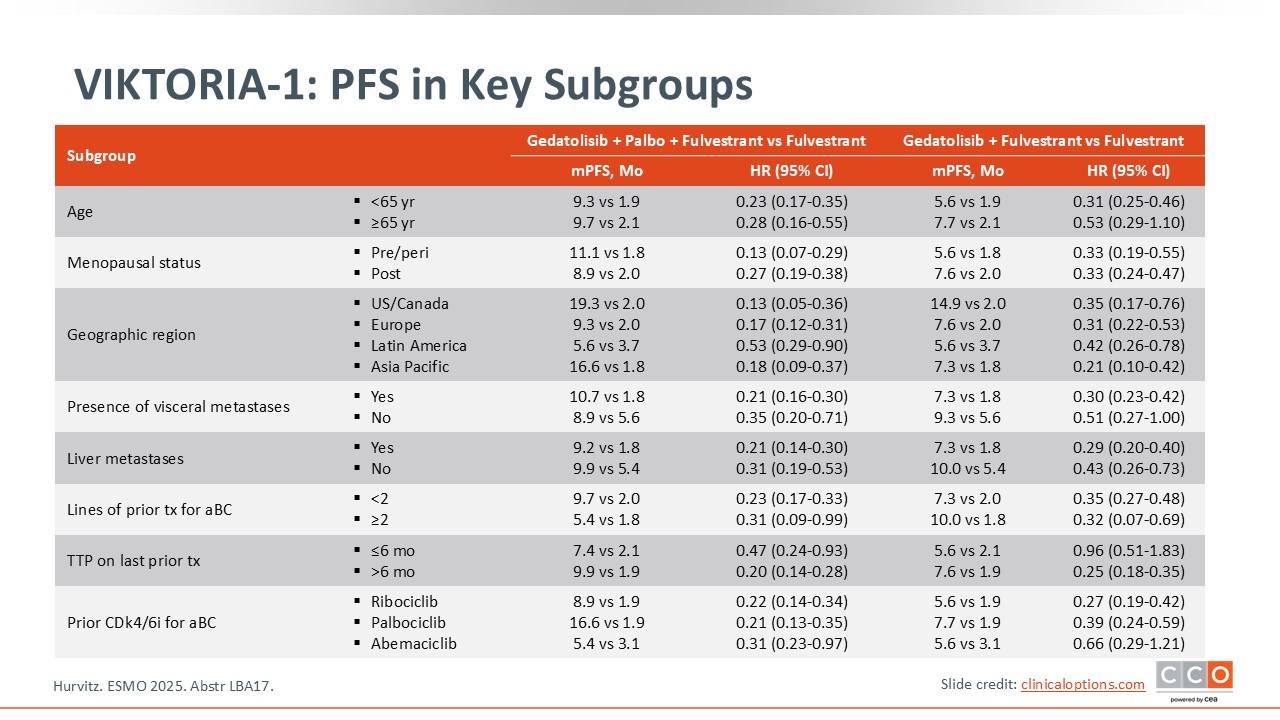
VIKTORIA-1: PFS in Key Subgroups
Paolo Tarantino, MD, PhD:
In the subgroup analysis, patients with visceral metastases had a median PFS of more than 10 months.13 I think combinations with gedatolisib could be appealing for patients who have higher-risk disease and/or liver metastasis, whereas for patients with lower-risk disease other options may be preferred.
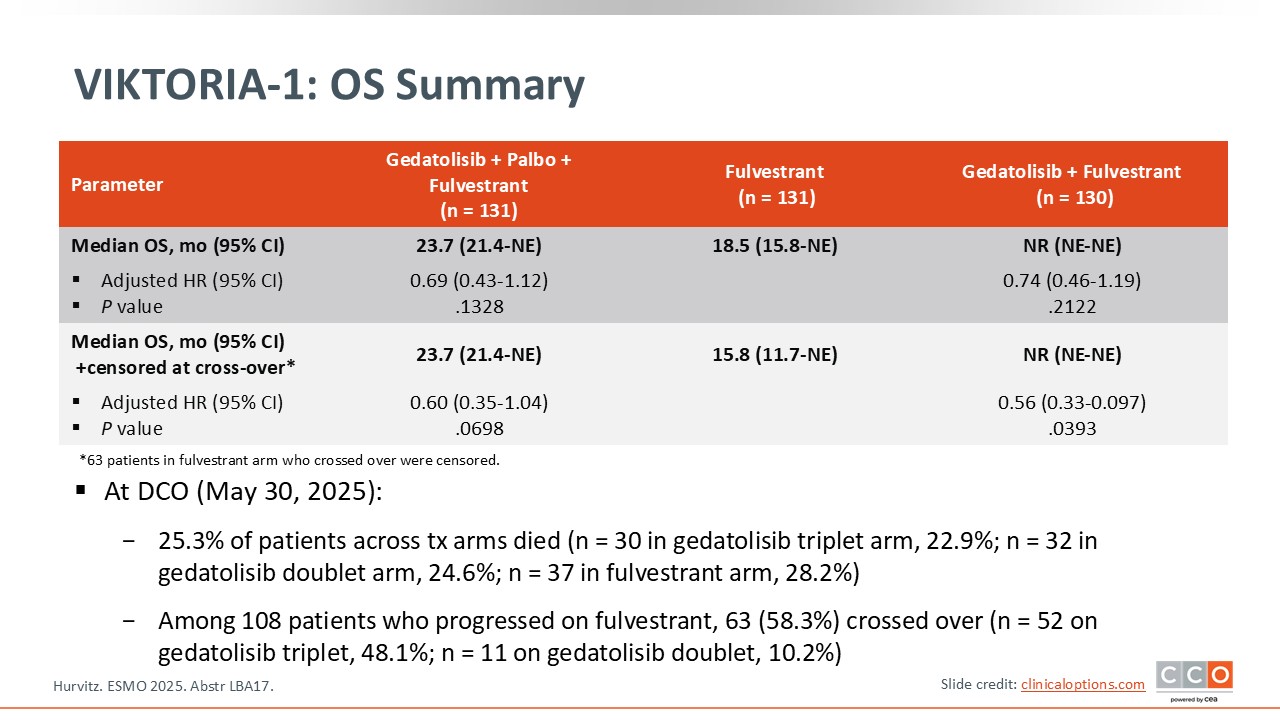
VIKTORIA-1: OS Summary
Paolo Tarantino, MD, PhD:
There was a favorable trend in OS in both gedatolisib treatment arms.13 Of note, this trial had a built-in crossover option allowing patients who progressed on fulvestrant to receive 1 of the 2 gedatolisib regimens.
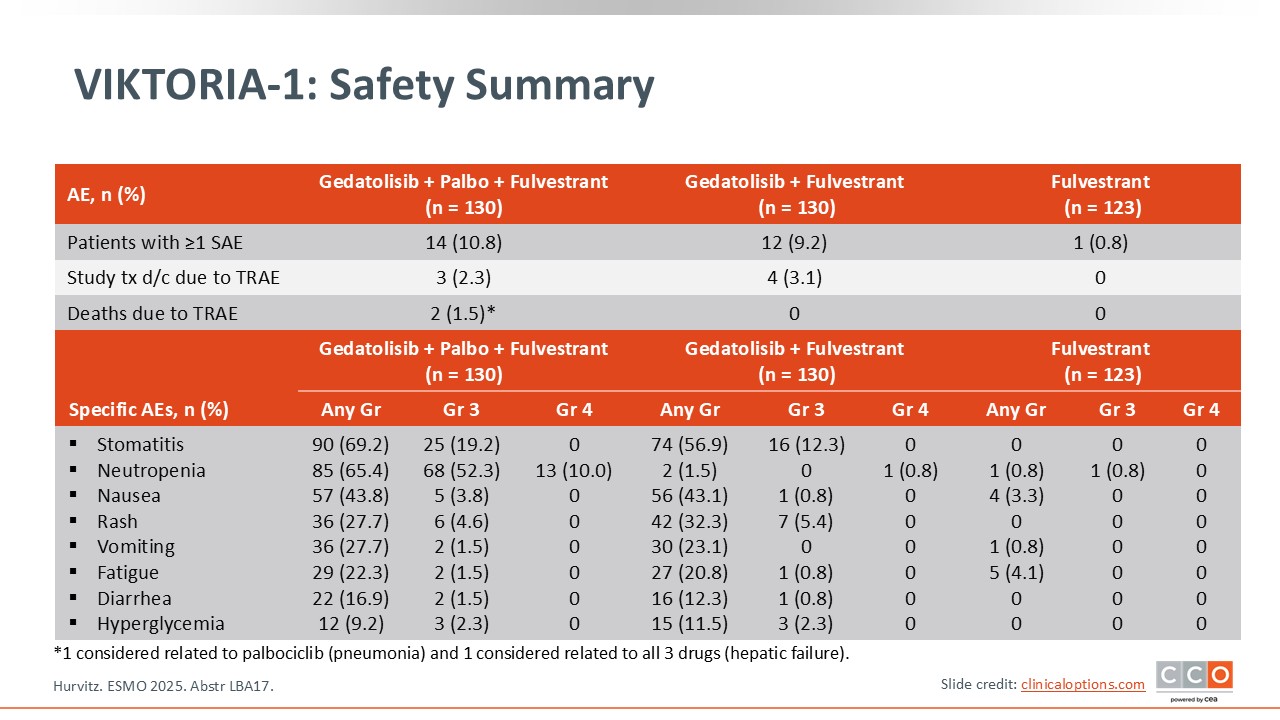
VIKTORIA-1: Safety Summary
Paolo Tarantino, MD, PhD:
In terms of toxicity, gedatolisib is associated with qualitative similar toxicity to what we see with everolimus.13 Stomatitis is a key side effect with approximately 70% any-grade stomatitis and almost 20% grade 3 with the triplet. There was also some degree of neutropenia in the arm that included palbociclib. Nausea, rash, and diarrhea were observed in the gedatolisib treatment arms and were predominantly lower grade. Hyperglycemia was low at 9.2% with the triplet and 11.5% for doublet therapy. There were 2 grade 5 events in the triplet arm: 1 due to pneumonia attributed to palbociclib and 1 due to hepatic failure in a patient with liver metastases deemed related to all 3 drugs. There were no grade 5 events in the doublet arm or the fulvestrant alone arm.
VIKTORIA-1: Conclusions
Paolo Tarantino, MD, PhD:
VIKTORIA-1 will bring a highly active doublet or triplet, depending on approval, to the clinic for patients with pretreated HR-positive metastatic breast cancer but there are some caveats.13 First, it is still not clear whether you really need the palbociclib, because both the triplet and doublet combinations appear quite active. Second, the IV infusion administration of gedatolisib could raise some challenges in this population that is usually treated without infusions. We know that having an oral regimen is advantageous in terms of logistics and patient preference. There seems to be more toxicity, especially with the triplet compared to that observed in evERA BC8 and with other options available in this setting. I believe that this combination will be helpful in the clinic and there may be patients who benefit from it, but I do think it comes with some challenges for use in clinical practice.
Cristina Saura, MD, PhD:
The VICTORIA-1 trial is quite difficult for me to translate into clinical practice because the data presented are only for the PI3K wild-type population, which is less interesting. Although PFS was increased with gedatolisib in both combinations compared with fulvestrant, I would note that monotherapy is not a standard anymore in this specific scenario. Although gedatolisib demonstrates good efficacy and safety, I think the main limitation of gedatolisib is that it is an IV drug given weekly every 3 out of 4 weeks. One of the main advantages of treating patients in the endocrine sensitive–setting is the ability to prescribe oral treatments that prevent the patients from having to come to the hospital frequently without a lot of blood tests and renal function tests. For patients in this setting who may be treated with gedatolisib, you need to use the IV formulation of the drug. In my opinion, it is difficult to envision a future for this drug given that other agents in this setting such as PI3K/AKT/mTOR pathway inhibitors can be administered orally.
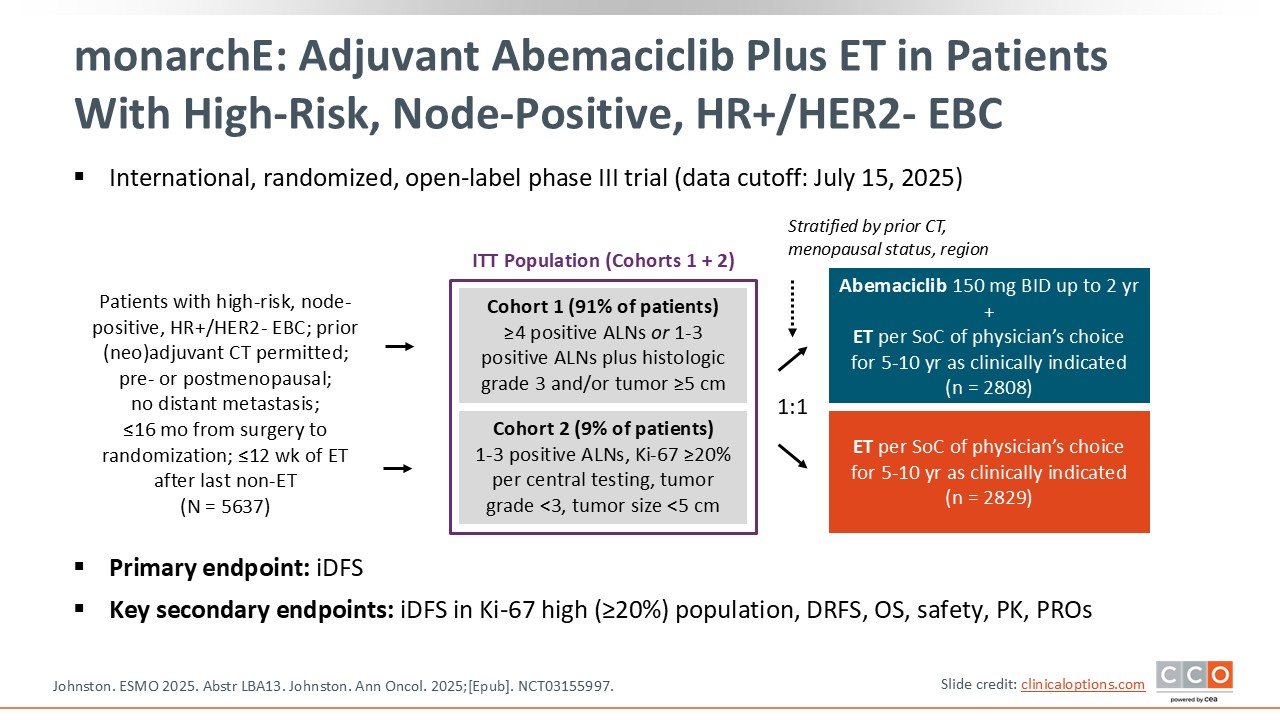
monarchE: Adjuvant Abemaciclib Plus ET in Patients With High-Risk, Node-Positive, HR+/HER2- EBC
Cristina Saura, MD, PhD:
Shifting to early-stage HR-positive/HER2-negative breast cancer, there is an update to the phase III monarchE trial.14,15 This trial examined the addition of the CDK4/6 inhibitor abemaciclib to ET in the adjuvant setting for a high-risk patient population with HR-positive/HER2-negative early breast cancer.16,17
We know that this is an important population, because currently more than 75% of the patients diagnosed with breast cancer have HR-positive disease. Most of these patients, at least those in developed countries, are diagnosed with a localized breast tumor. So this is the most common situation that we face when a patient is diagnosed with breast cancer. We know that we can cure more than 85% of these patients, but there are some patients with high-risk characteristics that need improved outcomes. This is the question that the monarchE trial addresses: How can we improve the outcomes of those patients with HR-positive, HER2-negative, node positive, and high-risk characteristics?
Of importance are the characteristics of the patients eligible to enroll on the monarchE trial. This included patients with at least 4 positive nodes or 1-3 positive nodes and grade 3 or greater pathology findings or large tumors of 5 cm or more. There were a small number of patients included in cohort 2 with 1-3 axillary lymph nodes and a Ki-67 score of more than 20%. Unlike the NATALEE trial of adjuvant ribociclib, no patients with node-negative disease were included in this trial.18 Also of note is that abemaciclib is given twice daily for 2 years. Physician’s choice of ET included tamoxifen, aromatase inhibitors, and GnRH analogues but by contrast NATALEE excluded tamoxifen. This is a benefit of the monarchE trial because we know that some patients cannot tolerate the AEs associated with aromatase inhibitors.
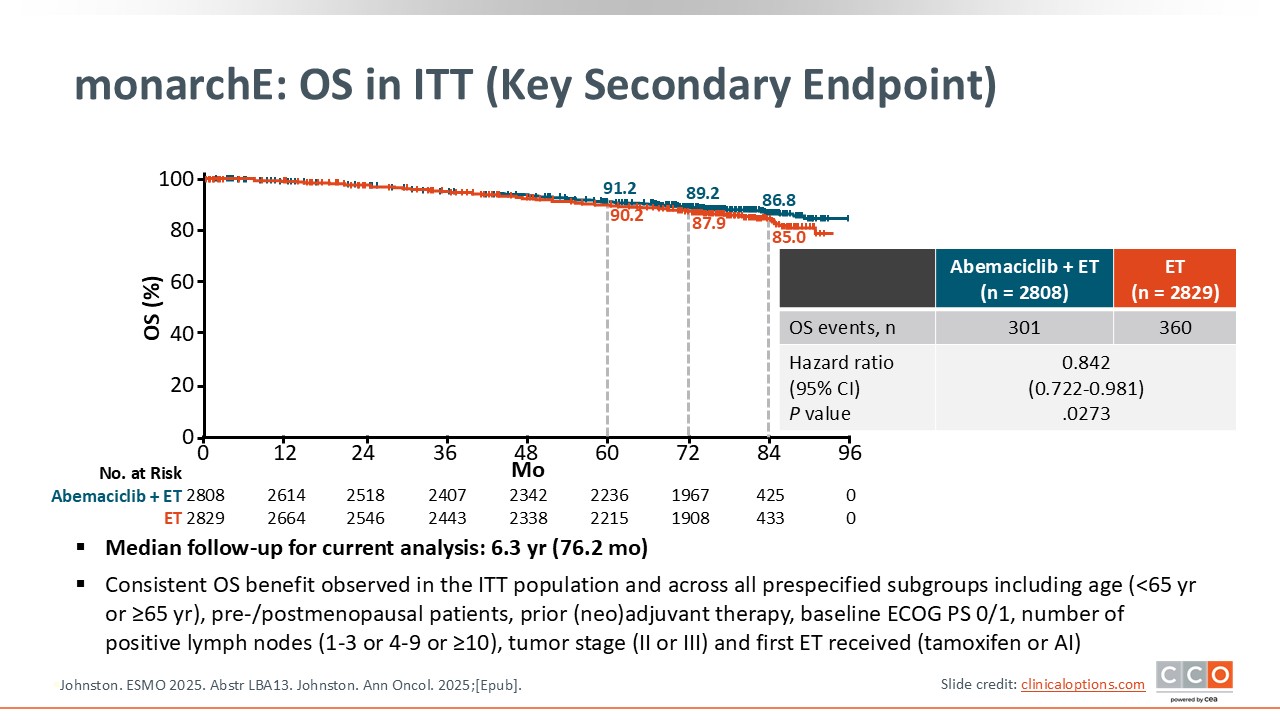
monarchE: OS in ITT (Key Secondary Endpoint)
Cristina Saura, MD, PhD:
With a median follow-up of 6.3 years, for the first time we see an OS benefit with adjuvant abemaciclib that is statistically significant with a hazard ratio of 0.842.14,15 I think that this is clinically meaningful and it is what we may expect from a clinical trial in the adjuvant setting. I think that we will see that this benefit could be even higher with a longer follow-up. Adjuvant abemaciclib plus ET is definitely something that we have to discuss and offer the patients who fulfill the criteria of the trial given the statistically significant OS benefit for this specific population.
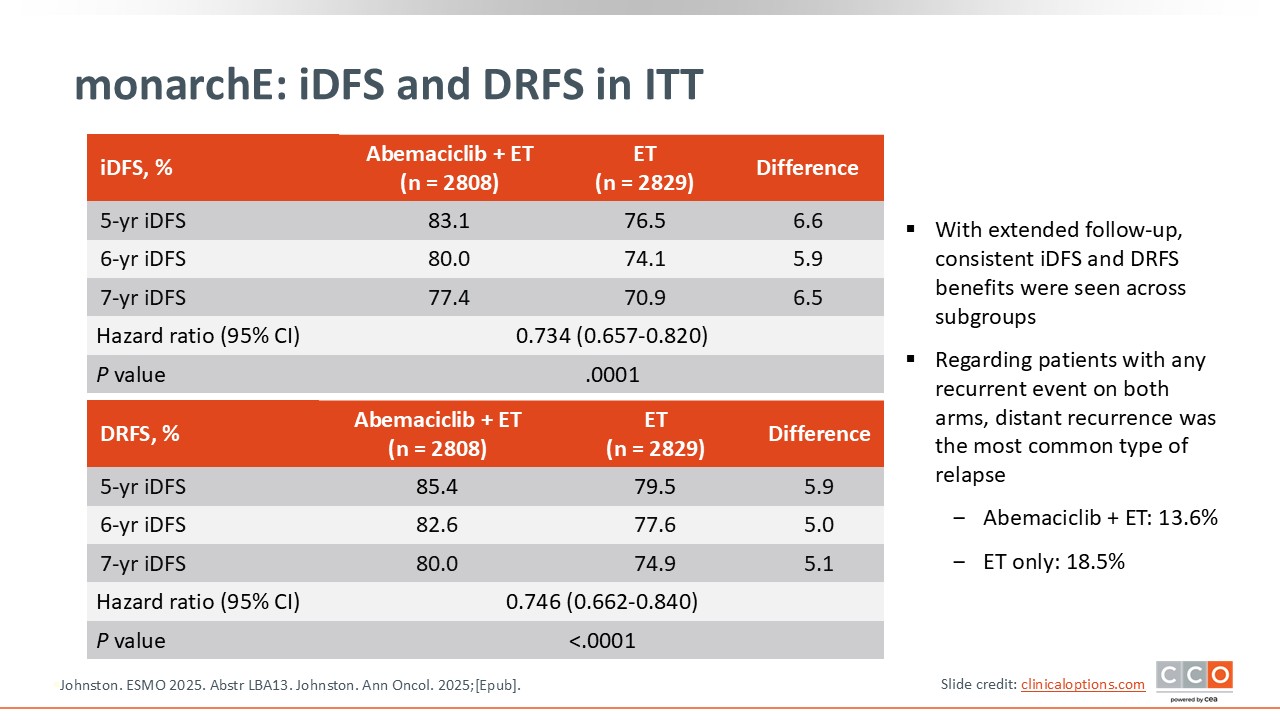
monarchE: iDFS and DRFS in ITT
Cristina Saura, MD, PhD:
The IDFS benefit continues to be sustained over longer follow-up with abemaciclib.14,15
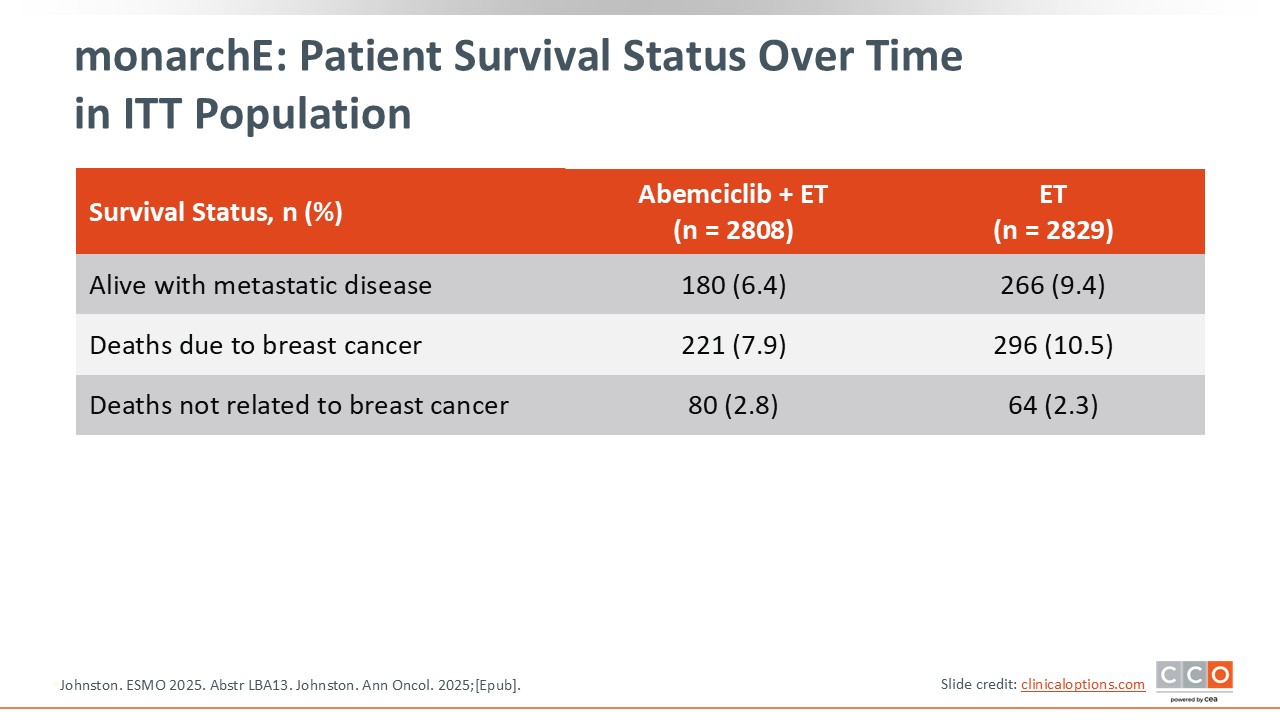
monarchE: Patient Survival Status Over Time in ITT Population
Cristina Saura, MD, PhD:
Of note, fewer patients in the abemaciclib arm are currently living with metastatic disease.14,15 It is anticipated that this will result in a larger OS benefit over time.
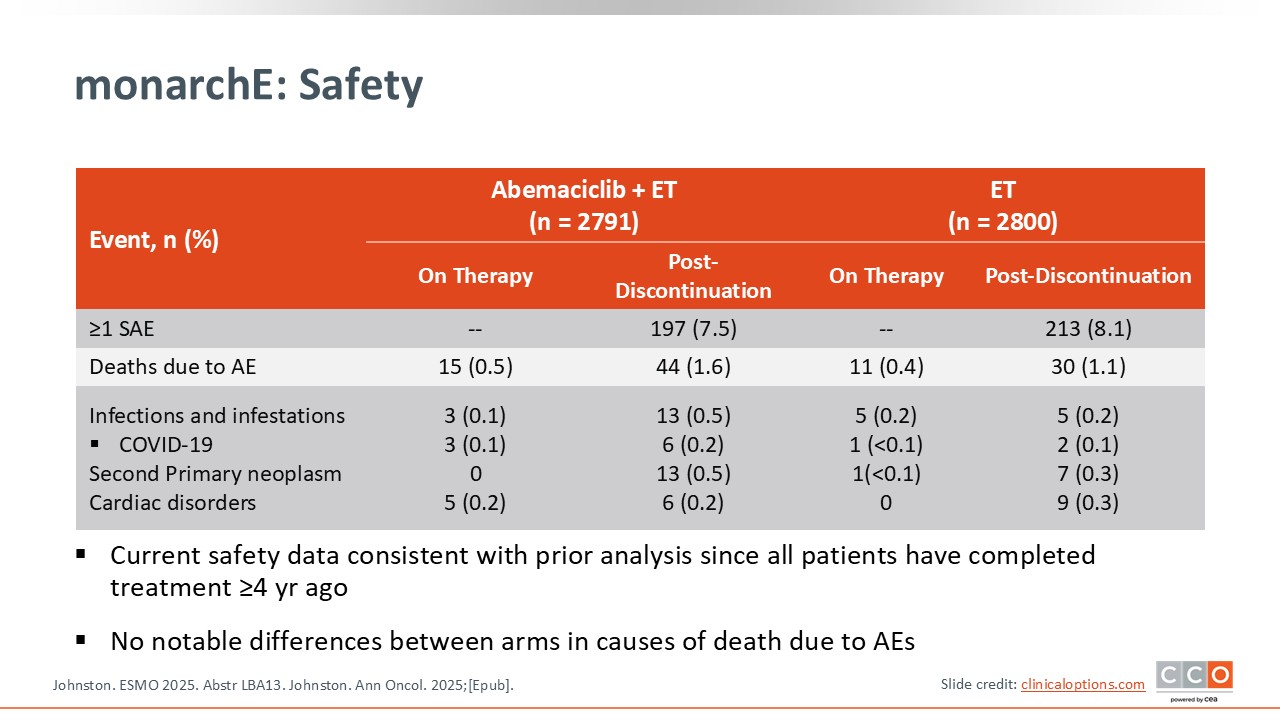
monarchE: Safety
Cristina Saura, MD, PhD:
The safety profile is consistent with earlier analyses.14,15
monarchE: Conclusions
Cristina Saura, MD, PhD:
This demonstration of an OS benefit is important, but I hope that in the future we can refine the population that benefits from 2 years of abemaciclib because 85% of the patients in the control arm without abemaciclib are still alive at this median 6.3 years of follow-up. In the future we need to identify additional biomarkers like ctDNA beyond the selection criteria for this trial to better select patients likely to benefit. Ki-67 is a prognostic factor but not predictive of abemaciclib benefit.19
An update on the NATALEE trial of 3 years of adjuvant ribociclib plus ET was also presented at ESMO 2025.20 It is important to note that the trial population in NATALEE is a lower-risk population than that included in monarchE. They included patients with node-negative disease but with high-risk features, a high Ki-67, or high grade and they were able to select patients within the node-negative population who derive a benefit from receiving 3 years of ribociclib.
Paolo Tarantino, MD, PhD:
The monarchE trial changed clinical practice several years ago by introducing adjuvant abemaciclib for 2 years and demonstrating improvements in IDFS for patients with high-risk HR-positive/HER2-negative breast cancer.16 Now we finally see a significant benefit in OS, albeit small, with a delta of 1.8% and a hazard ratio of 0.84. It is challenging to demonstrate an OS advantage in this population with early breast cancer. Even a small delta of 1.8% is clinically relevant and confirms current practice because we are already using abemaciclib in this population. I think it will lead to a stronger preference for abemaciclib over ribociclib in this very–high risk population of patients with more than 4 positive nodes or 1-3 positive nodes and other risk factors. I think I will prefer abemaciclib acknowledging that the toxicity profile of the 2 drugs is different and needs to be considered when making a treatment decision.
The 5-year update from NATALEE showed a maintained benefit in terms of IDFS and trend towards an OS benefit.20 Combined with the significant OS benefit with abemaciclib, this confirms that both abemaciclib and ribociclib are active in the adjuvant space. We need to see how the data keep maturing and whether the OS curves are going to remain separated in monarchE and if the positive OS trend in NATALEE continues with additional follow-up.