CME
Decera Clinical Education Independent Conference Highlights of the San Antonio Breast Cancer Symposium 2025: Early Breast Cancer
Physicians: Maximum of 0.50 AMA PRA Category 1 Credit™
Released: March 02, 2026
Expiration: September 01, 2026
Activity
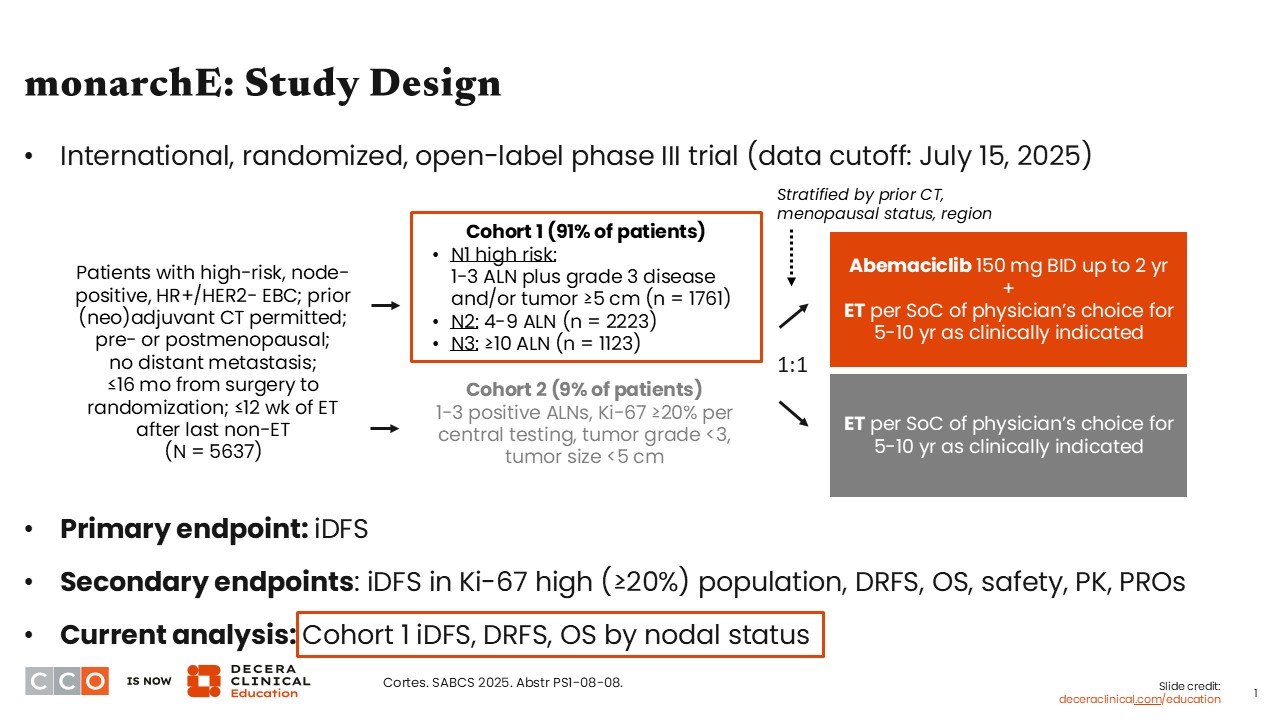
monarchE: Study Design
Virginia Kaklamani, MD, DSc:
I will start by discussing results presented from a subgroup analysis of the phase III monarchE trial evaluating adjuvant abemaciclib with endocrine therapy (ET) vs ET alone in patients with high-risk, node-positive, HR-positive/ HER2-negative EBC who may have previously received (neo)adjuvant treatment with chemotherapy but did not have distant metastases (N = 5637).1
In a previous report presented at the European Society of Medical Oncology (ESMO) Annual Congress 2025, investigators for the monarchE study showed an improved overall survival (OS), a key secondary endpoint, in favor of adjuvant abemaciclib with ET vs ET alone in the intention-to-treat (ITT) population (hazard ratio: 0.842; 95% CI: 0.722-0.981; P = .0273),2 which is quite remarkable in the adjuvant setting.
The update presented at SABCS 2025 reported outcomes from this trial by nodal status. More specifically, it evaluated these outcomes in cohort 1, where all patients had at least 1 positive lymph node, but there were different categories of risk, including N1 patients with 1-3 axillary lymph nodes (ALNs) with grade 3 disease and/or tumor size ≥5 cm (n = 1761);
N2, with 4-9 ALNs (n = 2223); and N3, with ≥10 ALNs (n = 1123). The objective of this analysis was to look at iDFS, distant relapse-free survival (DRFS), and OS outcomes by nodal status.
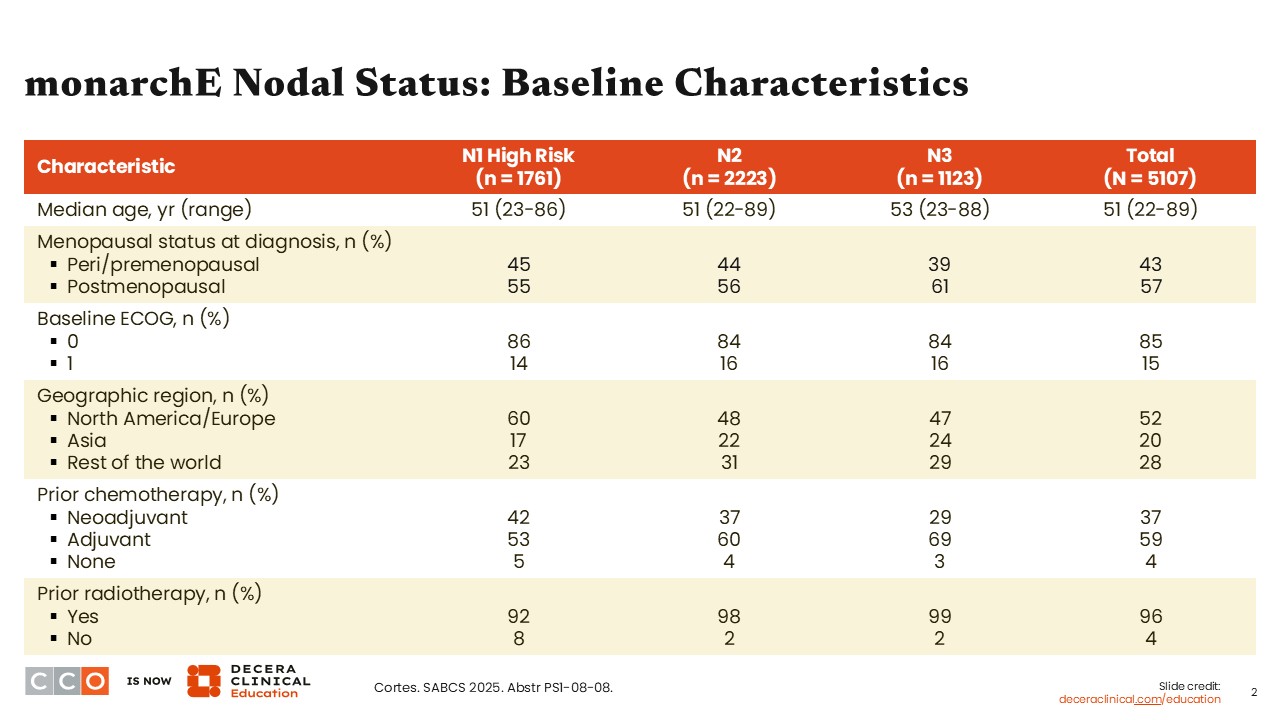
monarchE Nodal Status: Baseline Characteristics
Virginia Kaklamani, MD, DSc:
For this analysis, baseline characteristics were stratified into patient subgroups according to nodal status (ie, N1 high risk, N2, and N3). Because those in the N1 group must have had high-risk features, this cohort does not represent all patients with N1 disease who participated in the overall study.1
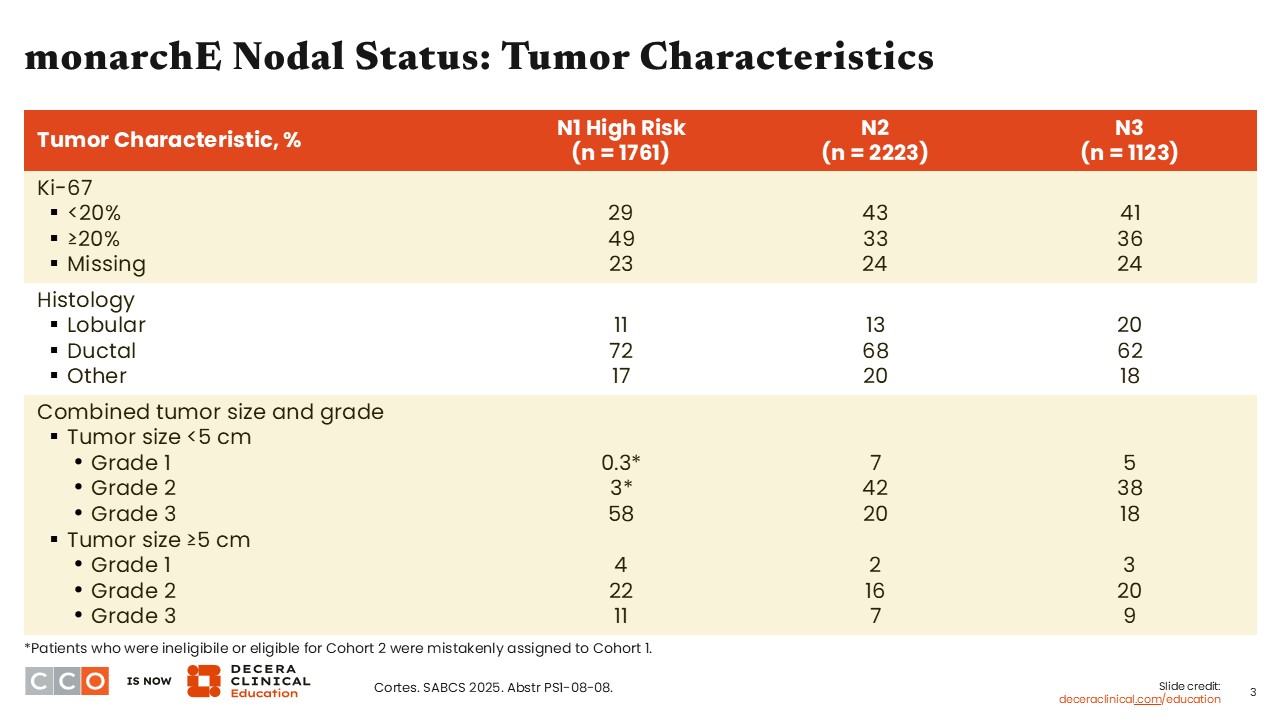
monarchE Nodal Status: Tumor Characteristics
Virginia Kaklamani, MD, DSc:
Study investigators also looked at tumor characteristics for the 3 nodal subgroups within cohort 1. The proportion of patients with a Ki-67 status of <20% was slightly higher in the N2 and N3 groups vs the N1 high-risk group (43%, 41%, and 23%, respectively). By contrast, the proportion of patients with Ki-67 ≥20% was slightly higher in the N1 high-risk group vs the N2 and N3 groups (49%, 33%, and 36%, respectively).
Regarding histology, the proportion of tumors with lobular histology was highest in the N3 group vs the N2 and N1 high-risk groups (20% vs 13% vs 11%, respectively).
Overall, there were more patients with tumor size <5 cm and grade 3 disease in the N1 group vs the N2 and N3 groups. There were more grade 3 tumors in the N1 high-risk group vs the N2 and N3 groups (58% vs 20% vs 18%, respectively). However, the proportion of tumors ≥5 cm that were either grade 1, 2, or 3 was similar regardless of nodal status.
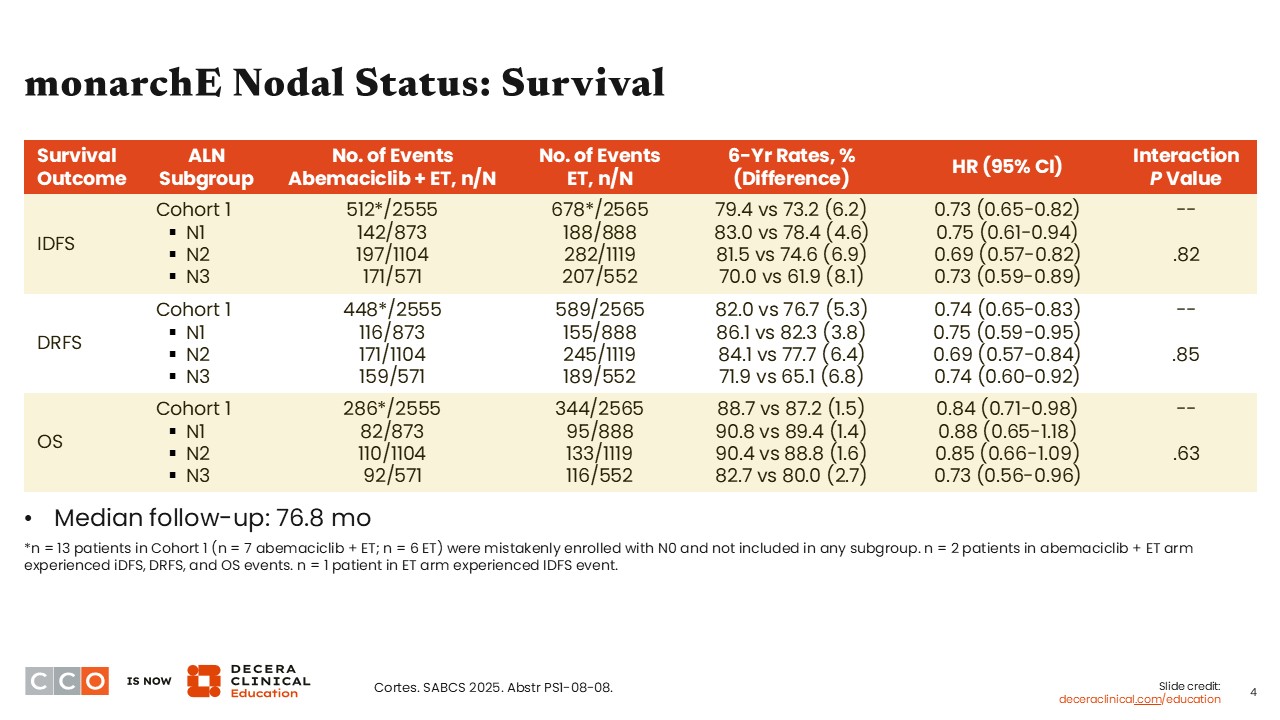
monarchE Nodal Status: Survival
Virginia Kaklamani, MD, DSc:
After a median follow-up of 76.8 months, we can see a consistent benefit for the combination of abemaciclib with ET vs ET alone regarding iDFS (hazard ratio: 0.69-0.75) and DRFS (hazard ratio: 0.69-0.75) across all nodal subgroups evaluated. Regarding OS, the magnitude of benefit was not substantially different across nodal subgroups (hazard ratio: 0.73-0.88), with some of the 95% confidence intervals for the N1 high-risk and the N2 nodal subgroups crossing 1.0. Furthermore, those with greater disease burden or N3 nodal status saw higher absolute benefit with the addition of abemaciclib to ET vs ET alone.1
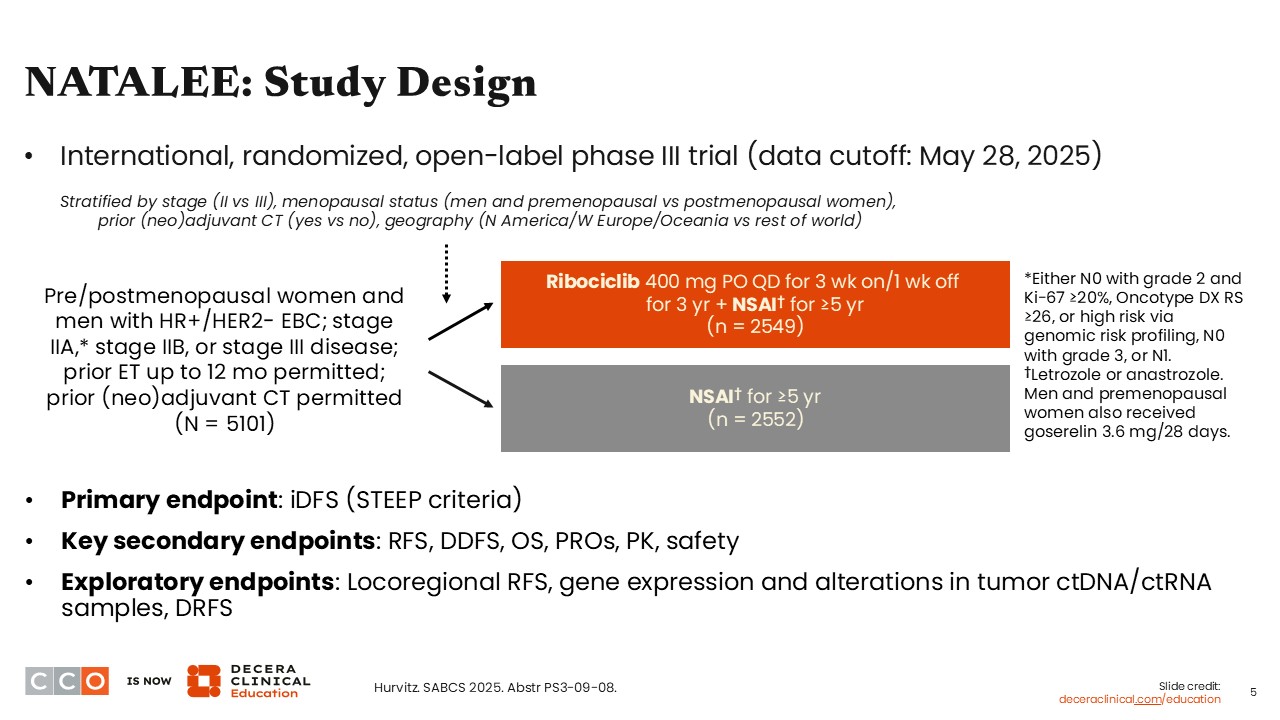
NATALEE: Study Design
Virginia Kaklamani, MD, DSc:
The phase III NATALEE trial was a similar design to that of monarchE. However, NATALEE evaluated ribociclib with an NSAI vs NSAI alone in pre/postmenopausal patients with HR-positive/HER2-negative EBC (stage IIA [either N0 with grade 2 and Ki-67 ≥20%, Oncotype DX risk score of ≥26, or high risk via genomic risk profiling, N0 with grade 3, or N1]; stage IIB, or stage III disease). It is important to note that in NATALEE, ribociclib was administered at a lower dose—400 mg daily for 3 weeks on and 1 week off for 3 years—compared with what is usually given in the metastatic setting (600 mg PO).3 Prior ET within the last 12 months was permitted; no prior (neo)adjuvant chemotherapy was allowed. Another difference that may be important to note here is that ribociclib cannot be given in combination with tamoxifen due to the risk of increased QT prolongation.3
An update for this trial was also presented at the ESMO Congress 2025, reporting on 5-year outcomes and showing a continued and durable iDFS benefit with ribociclib plus NSAI vs NSAI alone (hazard ratio: 0.716; 95% CI: 0.618-0.829; P <.0001).4,5
At SABCS 2025, NATALEE study investigators reported 5-year outcomes of DDFS across several patient subgroups.6 Part of that analysis looked specifically at the patient population with node-negative disease who are also at a higher risk of recurrence (ie, with grade 2 and Ki-67 ≥20%, Oncotype DX risk score of ≥26, or high risk via genomic risk profiling, N0 with grade 3), or N1. Here, I believe, they attempted to answer the question of whether these patients, too, should receive ribociclib with NSAI.
NATALEE: DDFS and DRFS in ITT Population
Virginia Kaklamani, MD, DSc:
In the ITT population, there was a consistent benefit in DDFS (hazard ratio: 0.709; 95% CI: 0.608-0.827) and DRFS (hazard ratio: 0.699; 95% CI: 0.594-0.824) in favor of the arm receiving ribociclib with an NSAI vs an NSAI alone. The median follow-up for DDFS and DRFS was 55.5 months and 55.7 months, respectively. I think these data illustrate the increased benefit seen with giving ribociclib to patients with HR-positive, HER2-negative EBC for 3 years.
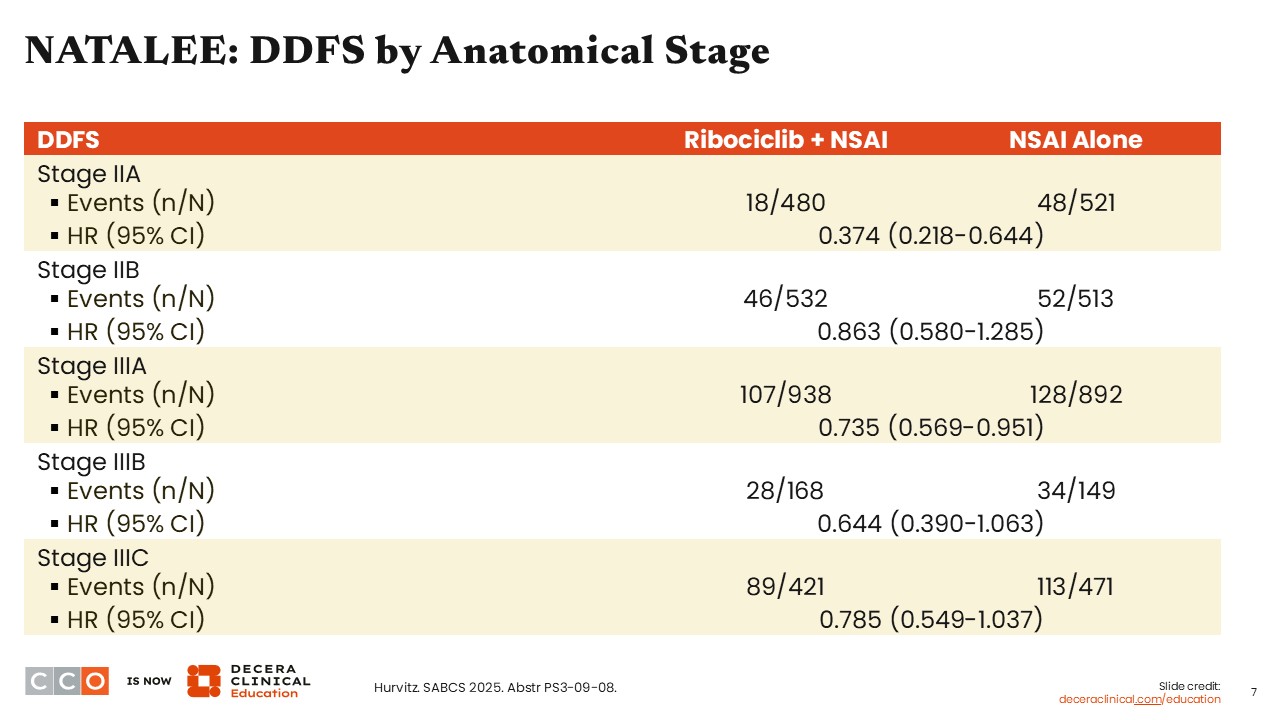
NATALEE: DDFS by Anatomical Stage
Virginia Kaklamani, MD, DSc:
Looking at DDFS by anatomical stage (eg, stage IIA to stage IIIC), we can appreciate a consistency in DDFS benefit with the combination of ribociclib and NSAI vs NSAI alone. The magnitude of benefit was remarkable among patients with lower stage/risk disease (IIA), with a hazard ratio of 0.374 (95% CI: 0.218-0.644). We also observe that as the stage of disease increases (from IIB to IIIA, IIIB, and IIIC), the absolute benefit also appears to increase (hazard ratio: 0.863 vs 0.735, 0.644, and 0.785, respectively).
I think that given the benefit seen in patients with node-negative disease who are also at a higher risk of recurrence (ie, with grade 2 and Ki-67 ≥20%, Oncotype DX risk score of ≥26, or high risk via genomic risk profiling, N0 with grade 3), or N1, these data suggest that we should consider recommending ribociclib with NSAI to these patients.
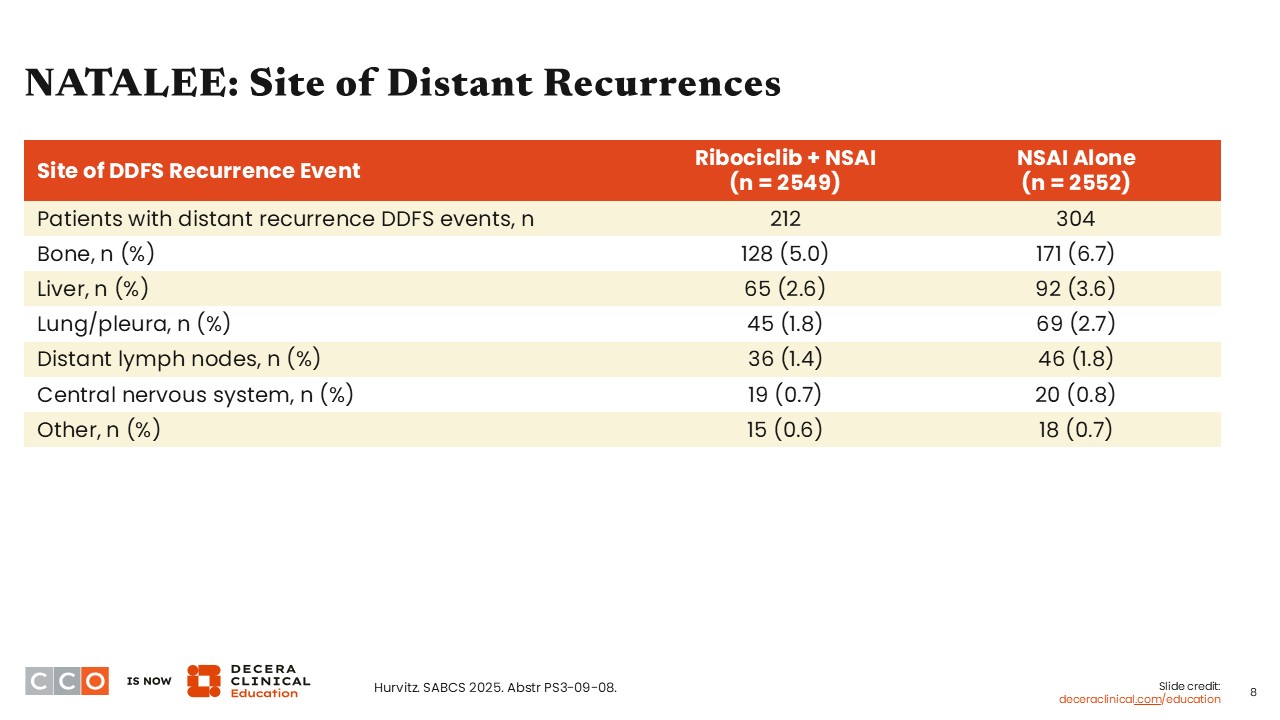
NATALEE: Site of Distant Recurrences
Virginia Kaklamani, MD, DSc:
Patients who develop metastatic disease after receiving adjuvant CDK4/6 inhibitor therapy have often been poorly characterized and represent an area of research with limited data.
In NATALEE, we have some data on the sites of distant disease recurrence. Most commonly, patients treated with either ribociclib with NSAI and NSAI alone developed metastatic disease to bone (5.0% and 6.7%), liver (2.6% and 3.6%), and lung/pleura (1.8% and 2.7%). Based on the data presented, we can estimate that it was less likely for patients in the ribociclib with NSAI arm to develop metastases to the bone compared with patients receiving NSAI alone. The proportion of patients who developed distant lymph nodes, central nervous system, or other sites of metastases was comparable between the treatment arms.
I think that what we saw in NATALEE regarding sites of metastatic disease development is similar to what we often see in patients with HR-positive breast cancer. Further, there appear to be no substantial differences compared with what we have seen in patients who had not received a CDK4/6 inhibitor.
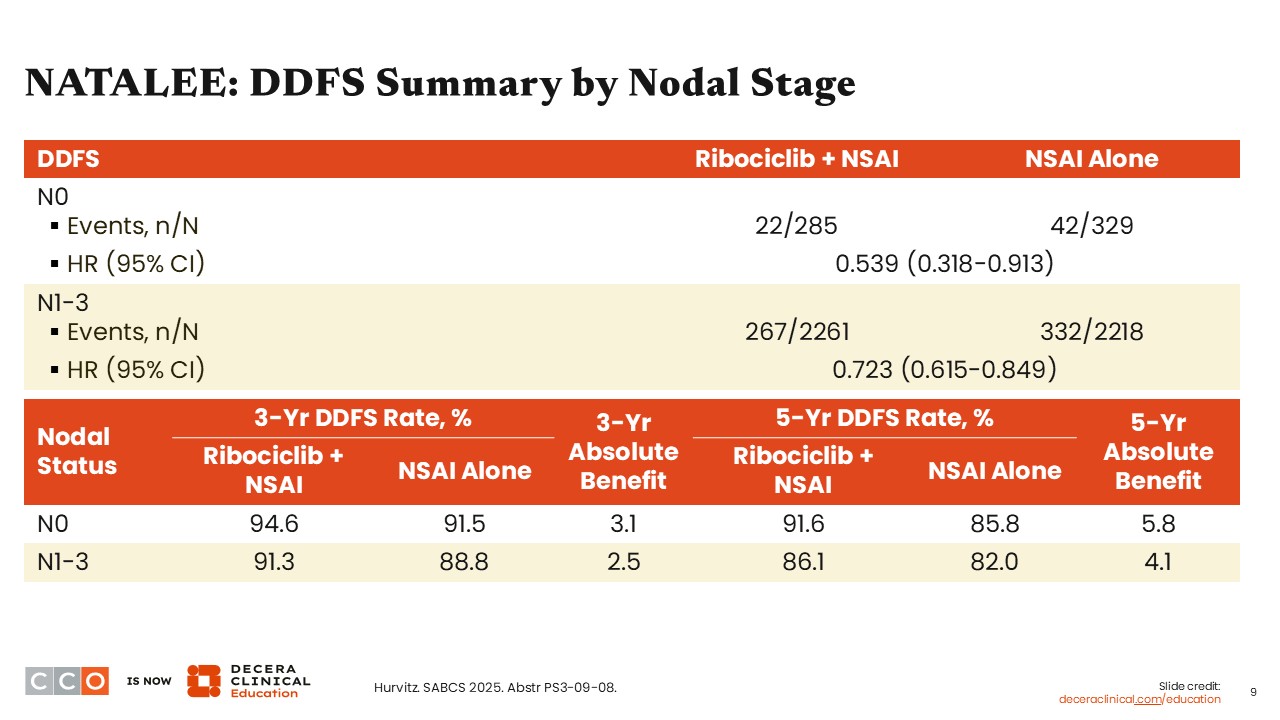
NATALEE: DDFS Summary by Nodal Stage
Virginia Kaklamani, MD, DSc:
Looking at DDFS outcomes by nodal stage, patients with N0 at high risk of recurrence saw a DDFS benefit with ribociclib with an NSAI vs an NSAI alone (hazard ratio: 0.539; 95% CI: 0.318-0.913), and the benefit was sustained based on reported absolute 3-year DDFS of 3.1 months (3-year DDFS rate: 94.6% vs 91.5%) and 5-year DDFS of 5.8 months (5-year DDFS rate: 91.6% vs 85.8%).
Similarly, patients with N1-3 disease experienced a DDFS benefit with ribociclib with NSAI vs NSAI alone (hazard ratio: 0.723; 95% CI: 0.615-0.849), and this trend was also sustained at 3 and 5 years (3-year absolute DDFS benefit: 2.5 months; 5-year absolute DDFS benefit: 4.1 months).
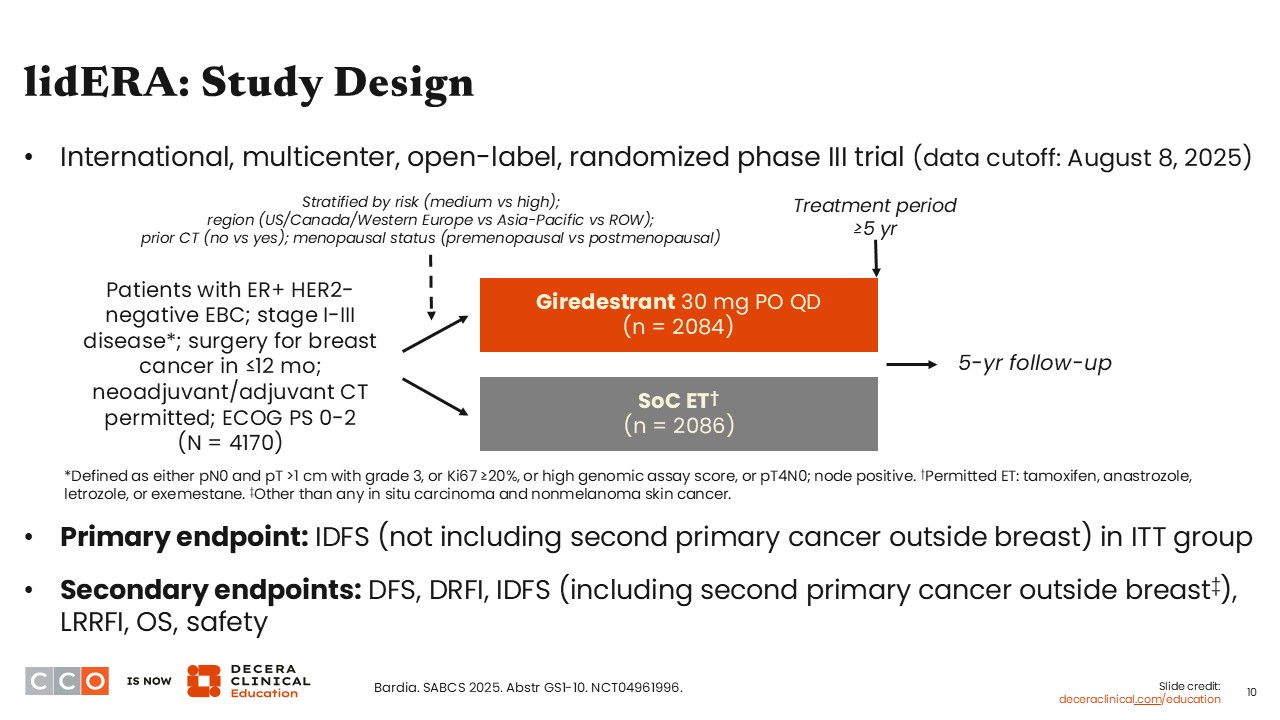
lidERA: Study Design
Virginia Kaklamani, MD, DSc:
One of the pivotal presentations at SABCS 2025 was the report from the phase III lidERA Breast Cancer trial. This brings us into a new era where EBC trials are evaluating oral selective estrogen receptor degraders (SERDs) in the adjuvant setting. lidERA is the first to report positive data in this setting.
The lidERA trial evaluated the use of adjuvant giredestrant compared with SoC ET given for ≥5 years to patients with stage I to III estrogen receptor (ER)-positive, HER2-negative EBC who had undergone surgery within the previous 12 months. Previous (neo)adjuvant chemotherapy treatment was allowed.
The primary endpoint of the study was iDFS, and key secondary endpoints included disease-free survival (DFS), distant recurrence-free interval (DRFI), iDFS (including second primary cancer other than breast cancer), locoregional recurrence-free interval, OS, and safety.
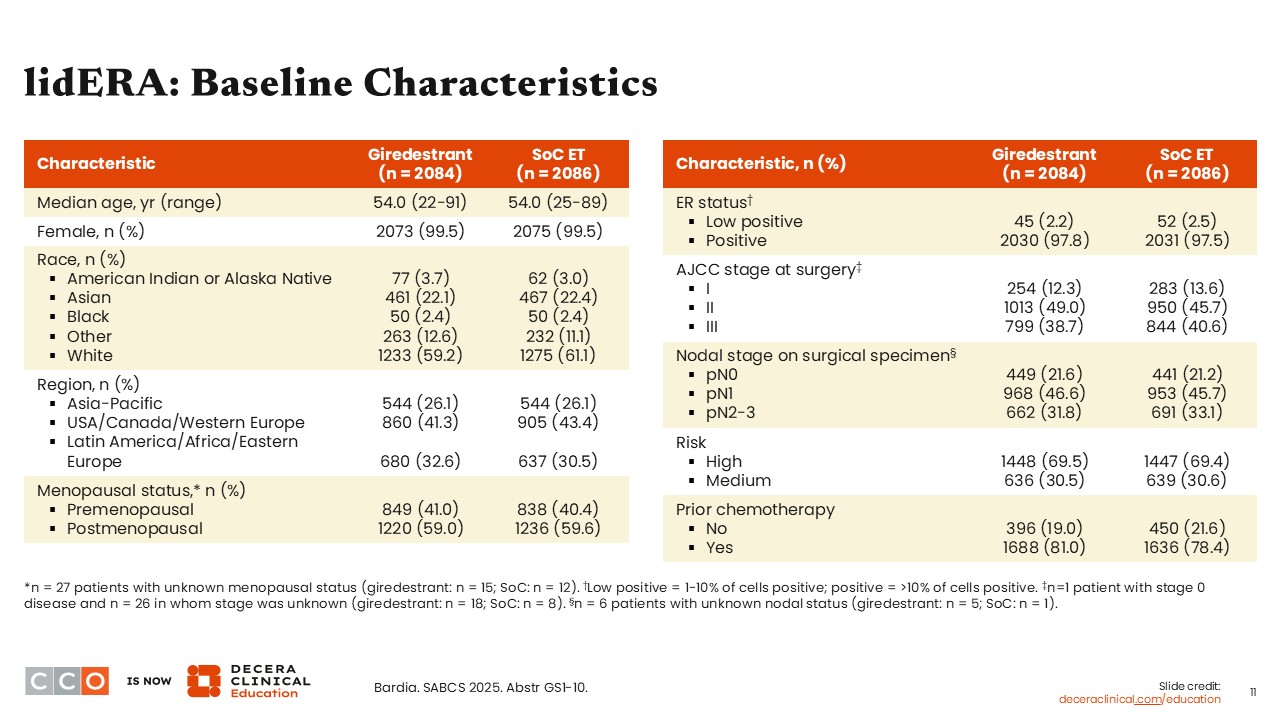
lidERA: Baseline Characteristics
Virginia Kaklamani, MD, DSc:
Looking at the baseline characteristics, both treatment arms were well balanced. Most patients in either arm had stage II (49.0% and 45.7%) or stage III (38.7% and 40.6%) disease, with approximately 12% to 14% of patients overall having stage I breast cancer at the time of surgery.7 This last point is important because these patients with stage I disease were generally not included in clinical trials evaluating CDK4/6 inhibitors in the adjuvant setting. Assuming giredestrant gets approved in this setting, we are going to be faced with treatment decisions on determining whether a patient should receive giredestrant or a CDK4/6 inhibitor.
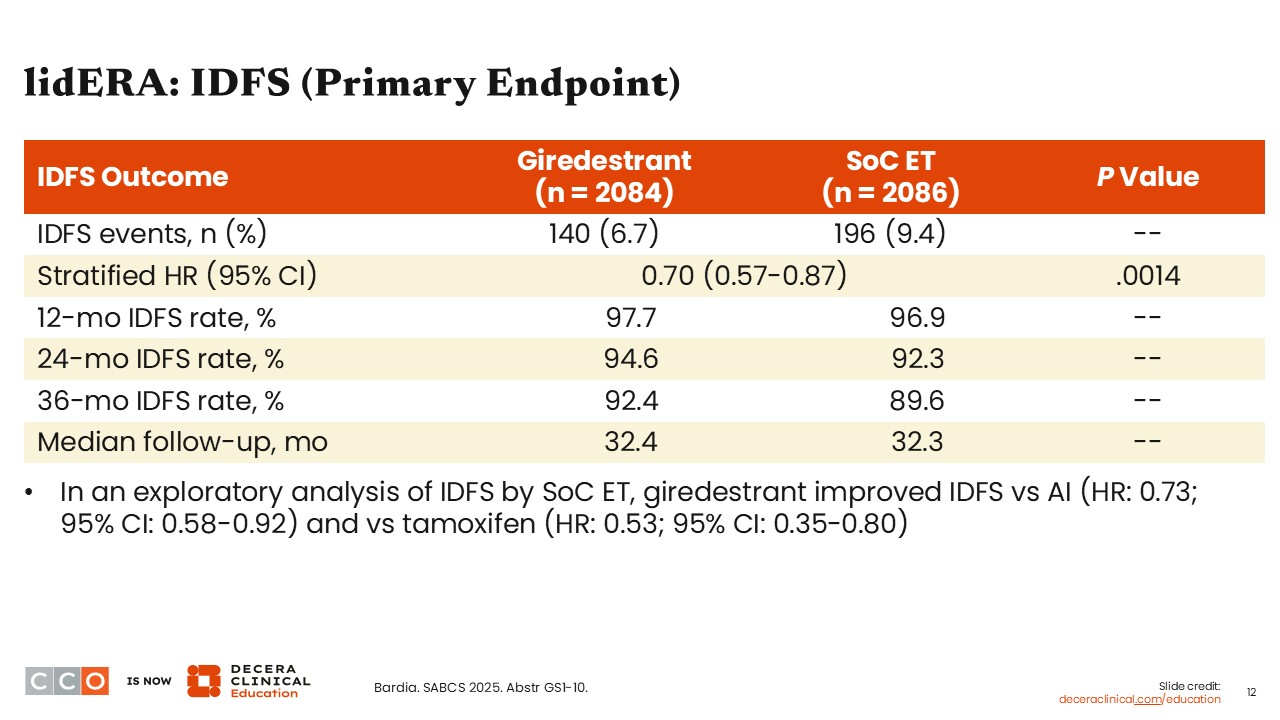
lidERA: IDFS (Primary Endpoint)
Virginia Kaklamani, MD, DSc:
In lidERA, we saw fewer iDFS events and a statistical improvement in favor of the giredestrant arm vs the SoC ET arm (hazard ratio: 0.70; 95% CI: 0.57-0.87; P = .0014). A trend for numerical benefit for iDFS with giredestrant vs the SoC ET arm was also seen at 12 months (97.7% vs 96.9%), 24 months (94.6% vs 92.3%), and at 36 months (92.4% vs 89.6%).7
Data from an exploratory analysis also showed improved iDFS with giredestrant independent of whether the SoC arm had received an AI (hazard ratio: 0.73; 95% CI: 0.58-0.92) or tamoxifen (hazard ratio: 0.53; 95% CI: 0.35-0.80).
In my opinion, despite being early results, they are quite remarkable—in favor of the potential use/recommendation of the oral SERD giredestrant as opposed to any of the options in the SoC ET arm.
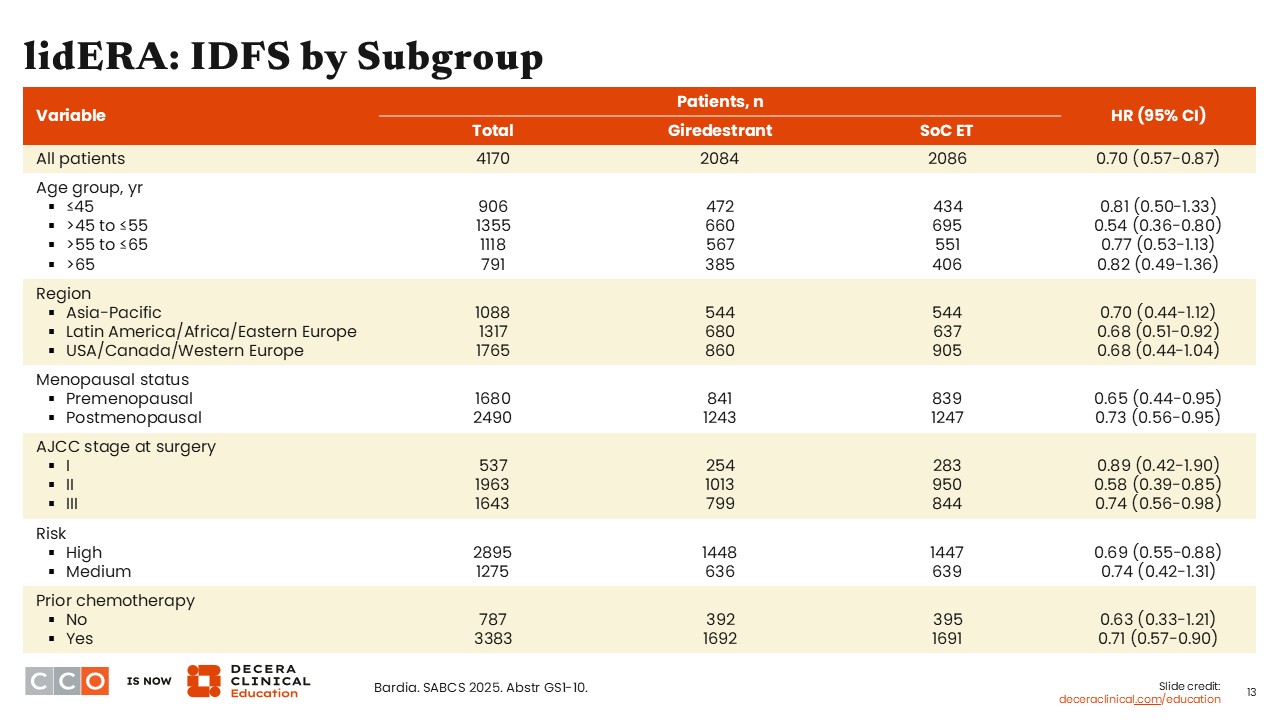
lidERA: IDFS by Subgroup
Virginia Kaklamani, MD, DSc:
IDFS was improved across patient subgroups receiving giredestrant vs SoC ET including in patients with stage II (hazard ratio: 0.58; 95% CI: 0.39-0.85) and stage III disease (hazard ratio: 0.74; 95% CI: 0.56-0.98), those with high-risk disease (hazard ratio: 0.69; 95% CI: 0.55-0.88), and those who had previously received chemotherapy (hazard ratio: 0.71; 95% CI: 0.57-0.90). By contrast, although the hazard ratio was 0.89 for patients with stage I breast cancer, the 95% confidence interval crossed 1.0 (95% CI: 0.42-1.90), indicating no statistically significant benefit for these patients.
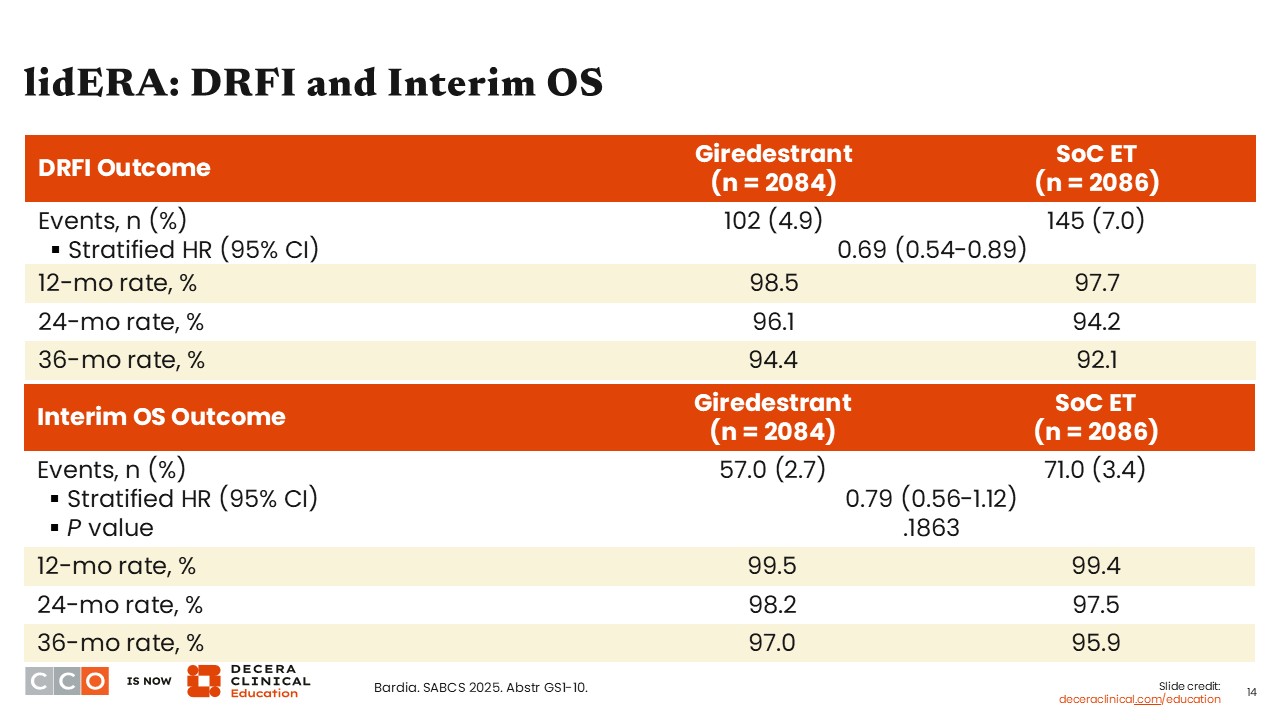
lidERA: DRFI and Interim OS
Virginia Kaklamani, MD, DSc:
The DRFI outcomes also showed a benefit in favor of the giredestrant arm compared with the SoC ET arm (stratified hazard ratio: 0.69; 95% CI: 0.54-0.89). OS data were immature at this presentation. However, interim OS results show a numerical trend in favor of the giredestrant arm vs SoC ET (stratified hazard ratio: 0.79; 95% CI: 0.56-1.12; P = .1863).
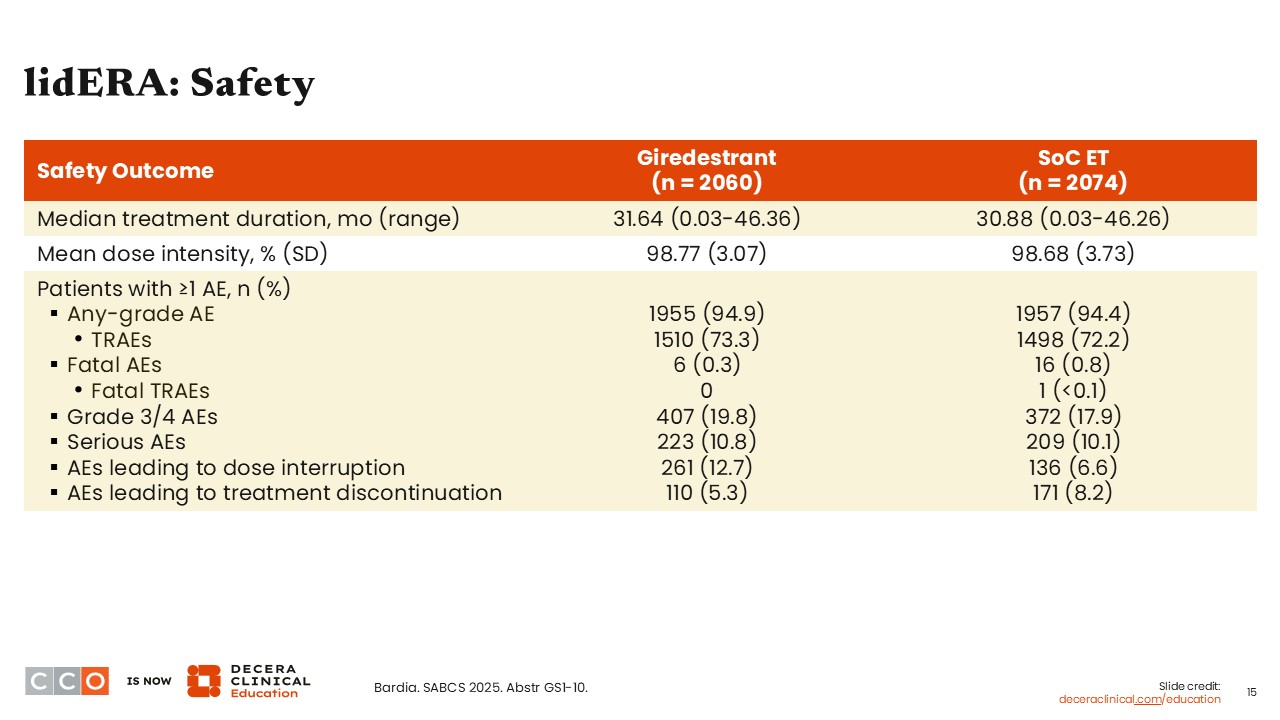
lidERA: Safety
Virginia Kaklamani, MD, DSc:
Regarding the safety of oral SERDs in the adjuvant setting, it recapitulates what we have seen for oral SERDs in the metastatic setting.
In my opinion, safety outcomes from the lidERA trial did not look different than what has been reported previously. Moreover, giredestrant is a well-tolerated agent with a favorable safety profile. The rate of any grade treatment-related adverse events (AEs) was similar between the giredestrant arm and the SoC ET arm (73.3% and 72.2%), as was the rate of grade 3/4 AEs (19.8% vs 17.9%), and serious AEs (10.8% vs 10.1%). Of note, 5% of patients in the giredestrant arm compared with 8% in the SoC ET arm discontinued treatment due to AEs.
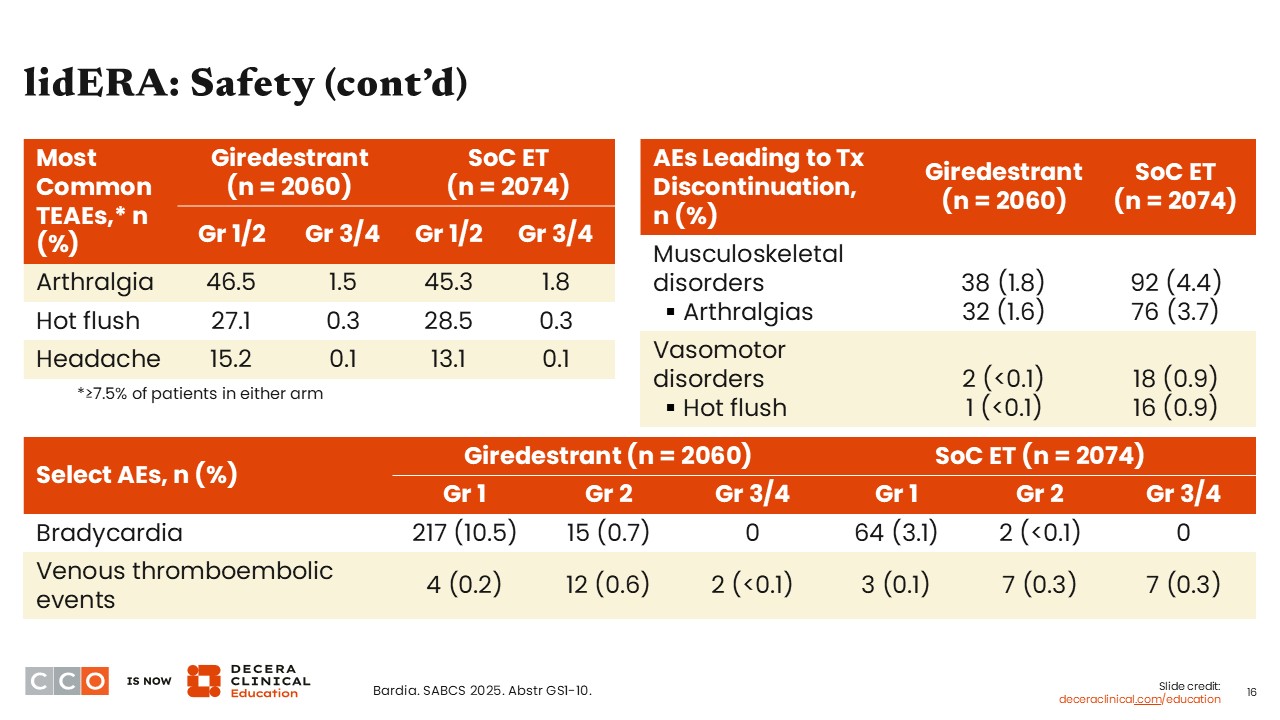
lidERA: Safety (cont’d)
Virginia Kaklamani, MD, DSc:
The most common grade 1/2 treatment-emergent AEs with both giredestrant and SoC ET were arthralgia (46.5% vs 45.3%), hot flush (27.1% vs 28.5%), and headache (15.2% vs 13.1%). Rates of grade 3/4 arthralgia, hot flush, and headache were under 2% for both arms. Musculoskeletal disorders leading to treatment discontinuation were less common with giredestrant vs SoC ET (1.8% vs 4.4%). Arthralgias led to giredestrant discontinuation in 1.6% of patients compared with 3.7% in the SoC ET arm. Hot flush also led to treatment discontinuation in both arms, but it was less than 0.1% with giredestrant and 0.9% with SoC ET.
Of note, bradycardia is a specific AE that is seen with oral SERDs. In lidERA, most bradycardia AEs were grade 1 for both study arms, but this is something to keep in mind for patients receiving giredestrant. Overall rates of venous thromboembolic events were low in both treatment arms and were reported in fewer than 1% of patients.
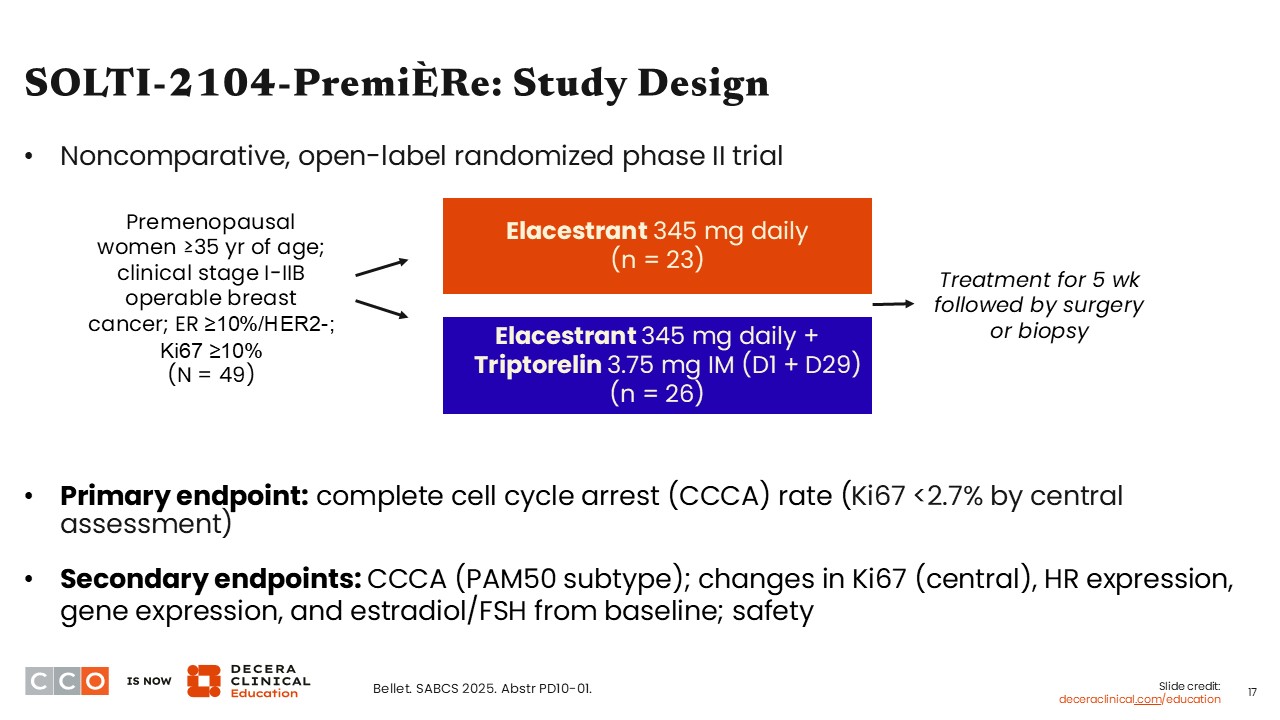
SOLTI-2104-PremiÈRe: Study Design
Virginia Kaklamani, MD, DSc:
Fulvestrant is an injectable SERD with multiple indications for postmenopausal women with advanced HR-positive breast cancer.8 The oral SERD elacestrant is indicated for the treatment of postmenopausal women or adult men with ER-positive, HER2-negative, ESR1-mutated advanced or metastatic breast cancer with disease progression following at least 1 line of ET.9 The question remains: Why do we not use these agents in premenopausal patients, particularly because the mechanism of action is different from that of AIs? A concern with oral SERD use, especially among premenopausal patients, is the development of ovarian cysts due to increases in estradiol levels.
This brings us to the phase II SOLTI-2104-PriemiÈRe trial, which was a noncomparative study evaluating elacestrant monotherapy and elacestrant with triptorelin in premenopausal women 35 years of age or older with stage I-IIB, ER-positive/HER2-negative EBC.10 Triptorelin is a synthetic gonadotropin-releasing hormone agonist that, with continuous administration, suppresses follicle-stimulating hormone and luteinizing hormone secretion, ultimately lowering testosterone and estrogen to castrate levels in premenopausal women. Elacestrant alone or elacestrant with triptorelin was administered for 5 weeks, then patients proceeded to surgery or biopsy.
The trial set out to determine if elacestrant is safe and efficacious to use in this premenopausal patient population. The primary endpoint of the study was the rate of complete cell cycle arrest (CCCA), defined as a Ki-67 of less than 2.7% per central assessment.
SOLTI-2104-PremiÈRe: CCCA Rate, Ki67 Change, ER/PgR Level Change
Virginia Kaklamani, MD, DSc:
Five weeks of treatment may potentially not be enough time to evaluate the risk for ovarian cysts, but it is plenty of time to look at preliminary efficacy results.
The primary endpoint of CCCA rate based on Ki-67 <2.7% was 29% with elacestrant alone and 27% with elacestrant and triptorelin. In other words, the addition of triptorelin did not appear to impact CCCA rate. The geometric mean change in Ki-67 was -62% (95% CI: -74.7% to -43.0%) in the elacestrant arm and -71.9% (95% CI: -82.7% to -54.2%) in the elacestrant with triptorelin arm. These were not intended to be compared but to show preliminary data. Of importance, the ER levels decreased by -42.6% in the elacestrant alone arm and -32.8% in the elacestrant with triptorelin arm.
SOLTI-2104-PremiÈRe: Other Study Outcomes
Virginia Kaklamani, MD, DSc:
Other outcomes measured by the investigators included gene expression. They reported consistent downregulation of 41 genes with elacestrant, and that 53% of 193 genes analyzed were differentially expressed following elacestrant exposure compared with 62% after exposure to elacestrant with triptorelin. Of importance, and although data were not shown, investigators indicated there also was a decrease in the PAM50 recurrence scores across both study arms, which suggests there is a benefit in using elacestrant in premenopausal patients.