CME
Decera Clinical Education Independent Conference Highlights of the San Antonio Breast Cancer Symposium 2025: Early Breast Cancer
Physicians: Maximum of 0.50 AMA PRA Category 1 Credit™
Released: March 02, 2026
Expiration: September 01, 2026
Activity
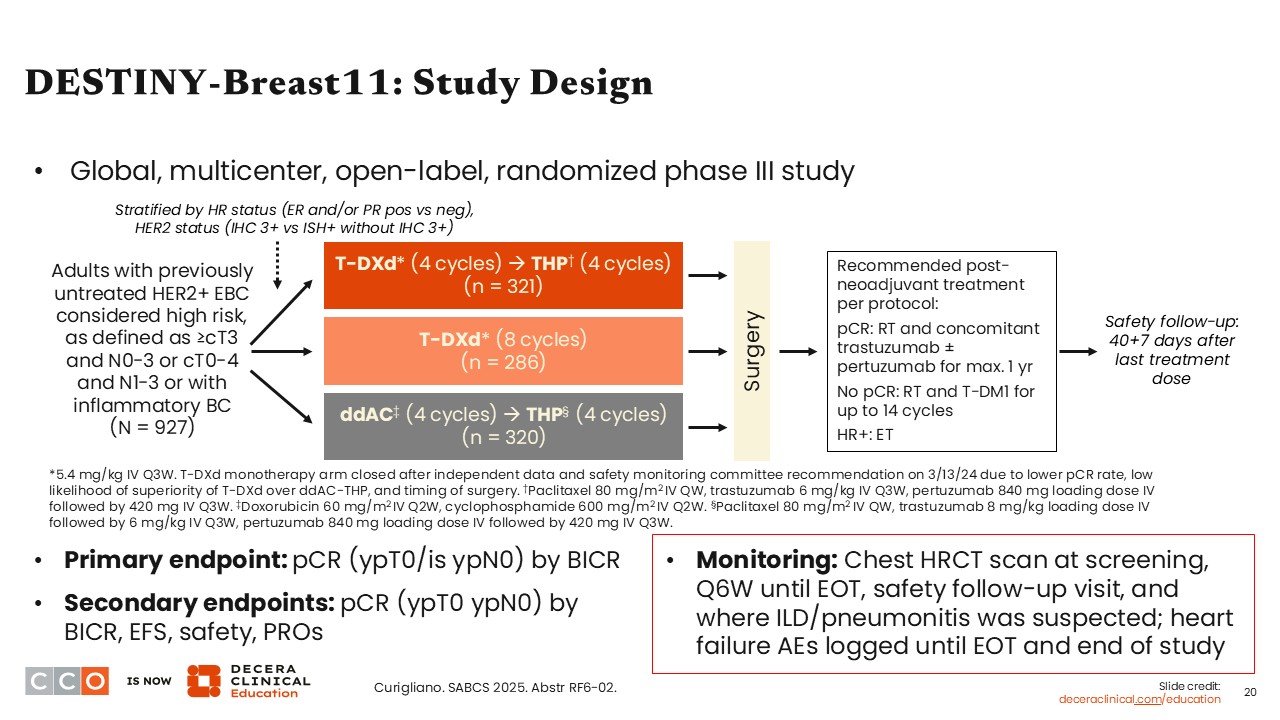
DESTINY-Breast11: Study Design
Virginia Kaklamani, MD, DSc:
Moving on to another trial that had initial results presented at ESMO Congress 2025,11 DESTINY-Breast11 is an interesting and complex clinical trial that I feel will likely change the current SoC.
The DESTINY-Breast11 trial enrolled patients with previously untreated, high-risk, HER2-positive EBC. High risk was defined as cT3 or higher and N0-N3, cT0-cT4 and N1-N3, or with inflammatory breast cancer. Patients were randomized into 3 treatment arms: T-DXd monotherapy for 8 cycles; T-DXd for 4 cycles followed by THP (ie, paclitaxel with trastuzumab and pertuzumab) for 4 cycles; or ddAC for 4 cycles followed by THP for 4 cycles, which was considered the SoC arm. Patients received treatment neoadjuvantly and then proceeded to surgery.
Of note, the T-DXd monotherapy arm was discontinued in March 2024 according to the independent data monitoring committee’s recommendation. There were multiple reasons for this recommendation, such as timing of surgery, low likelihood to show superior outcomes vs the SoC arm, and a decreased pathologic complete response (pCR) rate. Therefore, the results focused on the T-DXd/THP arm vs the ddAC/THP arm.
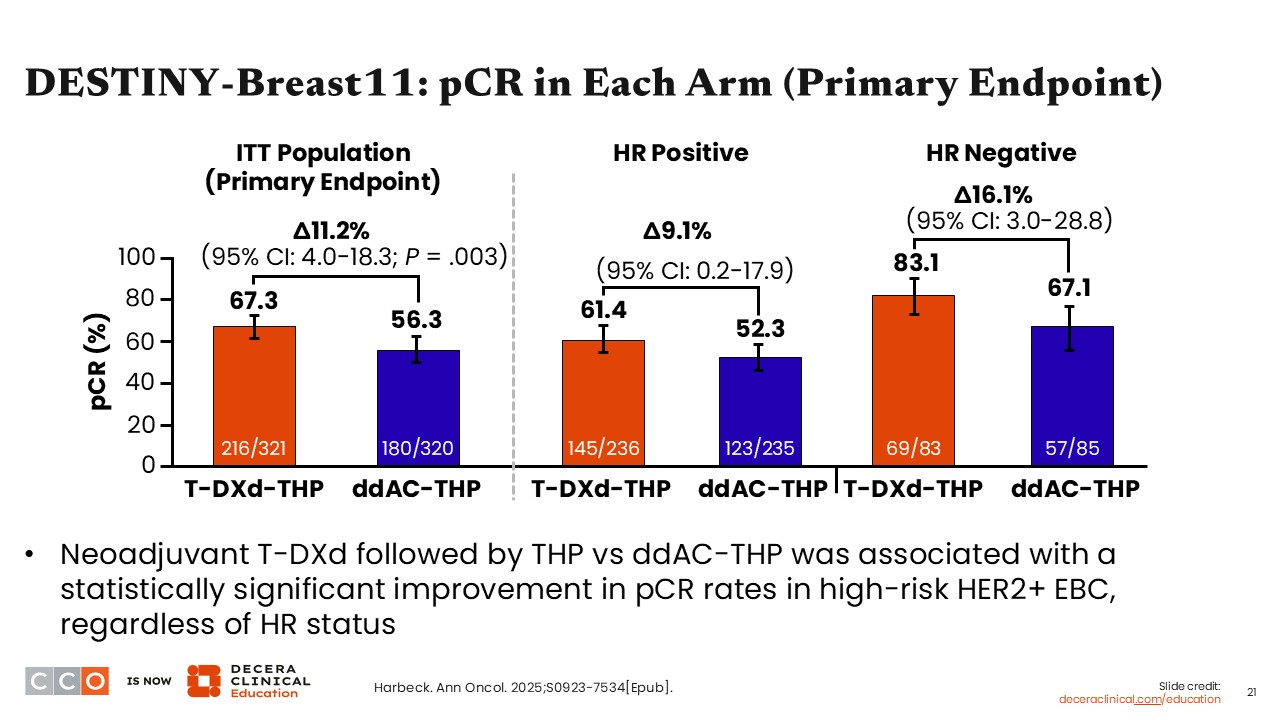
DESTINY-Breast11: pCR in Each Arm (Primary Endpoint)
Virginia Kaklamani, MD, DSc:
There was a statistically significant improvement in pCR rates in the ITT population that favored T-DXd/THP compared with ddAC/THP. The pCR rate was 67.3% in the T-DXd/THP arm compared with 56.3% in the ddAC/THP arm (difference: 11.2%; 95% CI: 4.0-18.3; P = .003).
Furthermore, pCR rates were higher among those with HR-negative disease vs HR-positive disease, which is a finding other clinical trials have reported. Taken together, the data show that 4 cycles of T-DXd followed by 4 cycles of THP can improve presurgery outcomes compared with SoC ddAC/THP therapy.
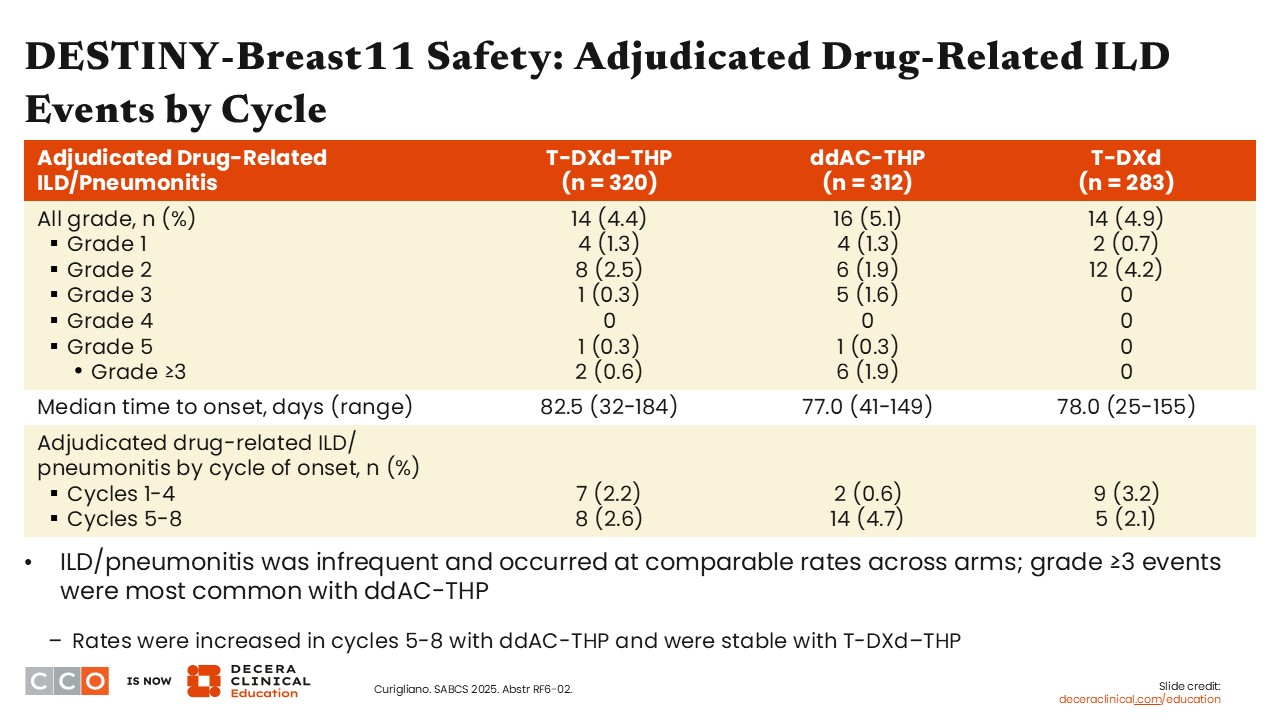
DESTINY-Breast11 Safety: Adjudicated Drug-Related ILD Events by Cycle
Virginia Kaklamani, MD, DSc:
The safety update from DESTINY-Breast11, presented at SABCS 2025, looked at treatment-related ILD/pneumonitis. This is an AE of interest because T-DXd is associated with an incidence rate of 10% to 14% for ILD as seen across the DESTINY-Breast clinical trial program.12 In the adjuvant/curative setting, ILD/pneumonitis becomes an even more important toxicity to address.
In DESTINY-Breast11, the reported rates of ILD/pneumonitis were relatively low, affecting 4.4% of patients in the T-DXd/THP arm and 5.1% of patients in the ddAC/THP arm. Most ILD/pneumonitis events were grade 1 or 2, and there were few grade 3 events in the T-DXd/THP arm (0.6%) and the ddAC/THP arm (1.9%). Of note, 1 patient in each study arm experienced grade 5 ILD/pneumonitis.
The median time to ILD/pneumonitis onset was 82.5 days (range: 32-184 days) with T-DXd/THP vs 77.0 days (range: 41-149 days) with ddAC/THP. That is similar to what I have seen in the metastatic setting as well.13
Based on this analysis presented at SABCS 2025, it appears that 4 cycles of T-DXd followed by THP results in a much lower rate of ILD/pneumonitis compared with what I have seen in the metastatic setting. That could be because patients with metastatic disease are exposed to T-DXd for much longer.
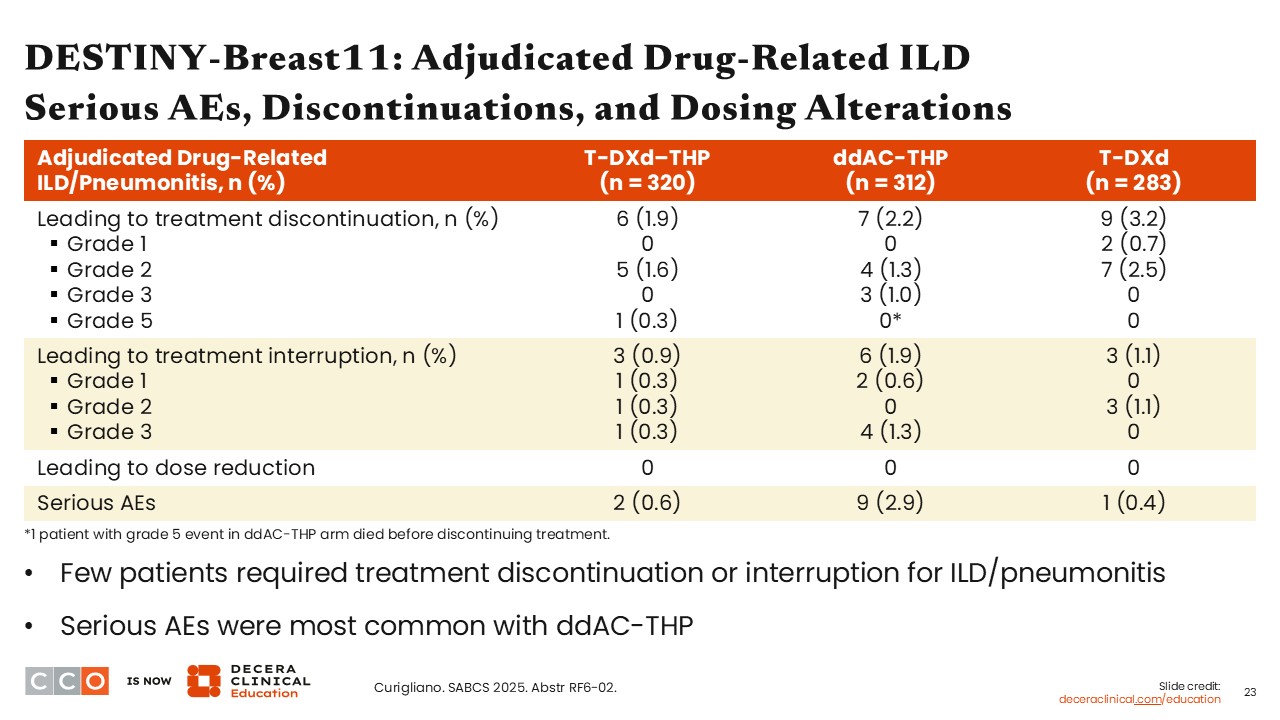
DESTINY-Breast11: Adjudicated Drug-Related ILD Serious AEs, Discontinuations, and Dosing Alterations
Virginia Kaklamani, MD, DSc:
Regarding treatment discontinuation, ILD/pneumonitis resulted in treatment discontinuation in approximately 2% of patients in both study arms. Treatment interruptions were low in both arms: fewer than 1% with T-DXd/THP and approximately 2% with ddAC/THP.
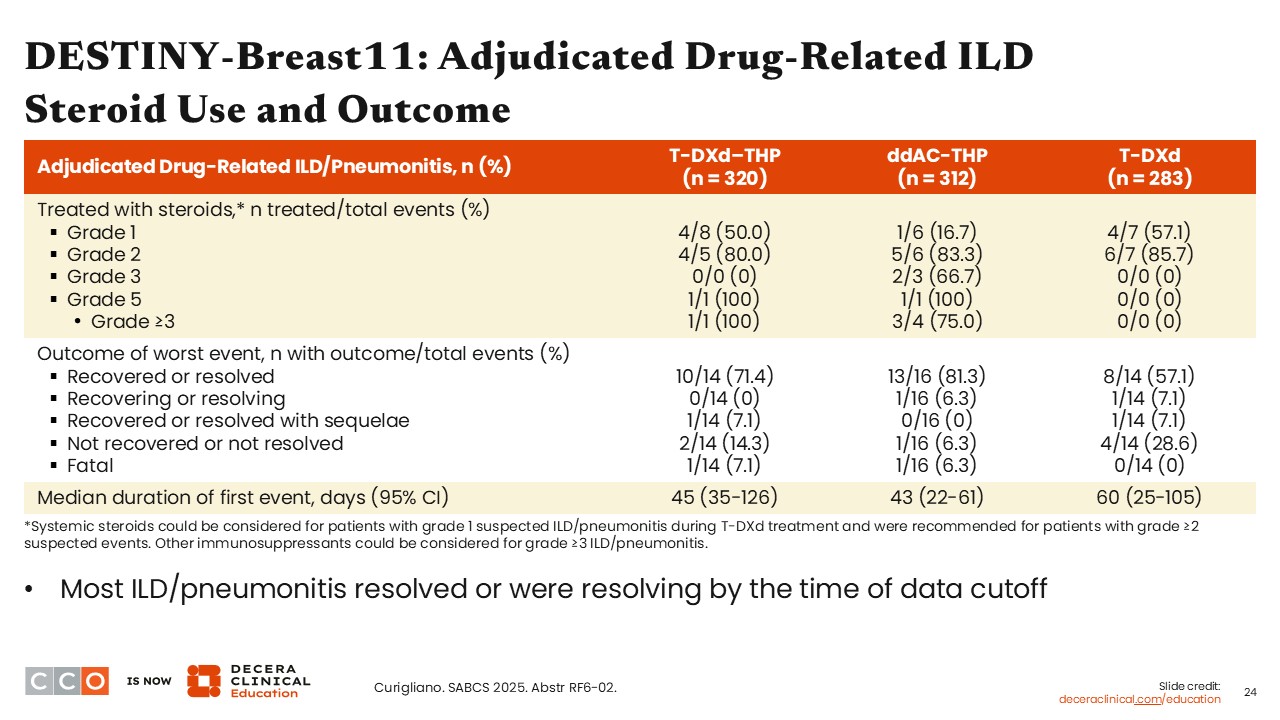
DESTINY-Breast11: Adjudicated Drug-Related ILD Steroid Use and Outcomes
Virginia Kaklamani, MD, DSc:
Overall, a lower number of patients in the T-DXd/THP vs ddAC/THP arms were treated with corticosteroids for ILD/pneumonitis. Fifty percent of patients with grade 1 ILD/pneumonitis received treatment with corticosteroids in the T-DXd/THP arm vs 16.7% in the ddAC/THP arm.
DESTINY-Breast11: Other AEs and Management
Virginia Kaklamani, MD, DSc:
Regarding other AEs, incident rates of left ventricular dysfunction were higher in the ddAC/THP arm (1.9%) compared with the T-DXd/THP arm (0.3%). That is to be expected. Further, more patients treated with T-DXd/THP reported nausea and vomiting compared with those treated with ddAC/THP, but those receiving ddAC/THP were more likely to use 3 or more antiemetics vs those receiving T-DXd/THP. This last part is important because this was a global trial, and the standards we follow in the United States regarding the use of antiemetics are not the same standards that the rest of the world follows. This is important to note because T-DXd is considered a highly emetogenic agent, and the preference, at least in the United States, is to use 4 antiemetics, including olanzapine. But that may not be the SoC in many other parts of the world.
Regarding peripheral neuropathy, this AE was more common in the T-DXd/THP arm vs the ddAC/THP arm, but events were generally low grade.
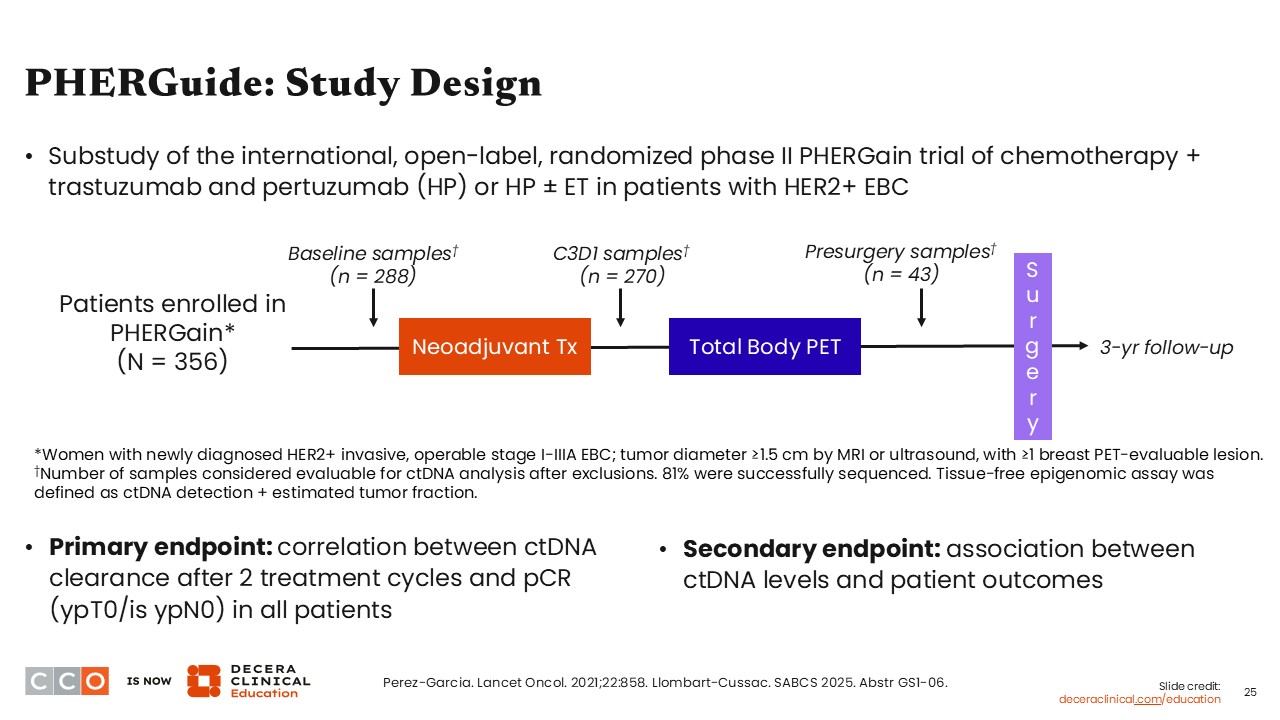
PHERGuide: Study Design
Virginia Kaklamani, MD, DSc:
Looking at another interesting study presented at SABCS 2025, the phase II PHERGuide trial examined a tumor epigenomic-based circulating tumor DNA (ctDNA) platform to detect minimal residual disease and strengthen prediction of pCR and long-term outcomes in patients with HER2-positive EBC who received neoadjuvant dual HER2-blockade treatment with trastuzumab and pertuzumab with or without ET.14,15
In PHERGain, an early FDG-PET–based assessment combined with pCR guided the omission of chemotherapy in patients receiving neoadjuvant trastuzumab with pertuzumab. This adaptive PET- and pCR-guided approach resulted in chemotherapy omission in 37.9% of patients while maintaining a 3-year iDFS rate of 94.8% in the pCR-adapted cohort. This adaptive strategy produced remarkable pCR rates and should be studied more in the future, as it might allow us to de-escalate therapy for our patients who have a good response with a nonchemotherapy-based regimen.
The presentation at SABCS 2025 was for the substudy known as PHERGuide, and it looked at ctDNA and how this impacted outcomes.15
PHERGuide: Correlation Between ctDNA at Baseline and Disease Stage, Nodal Status, and pCR
Virginia Kaklamani, MD, DSc:
The ctDNA samples were collected at several points throughout the trial. Then, this substudy used the data to define the correlation between outcomes and ctDNA. First, the likelihood of finding ctDNA was high. Approximately 33%, 71%, and 93% of those with stage I, II, and III EBC had positive ctDNA detected at baseline. Similar trends were seen by nodal status, with 87.8% of those with node-positive disease having positive ctDNA detected compared with 55.0% of those with node-negative disease.
Investigators reported there appeared to be no correlation between the ctDNA detected at baseline and a patient’s pCR status (P = .583).
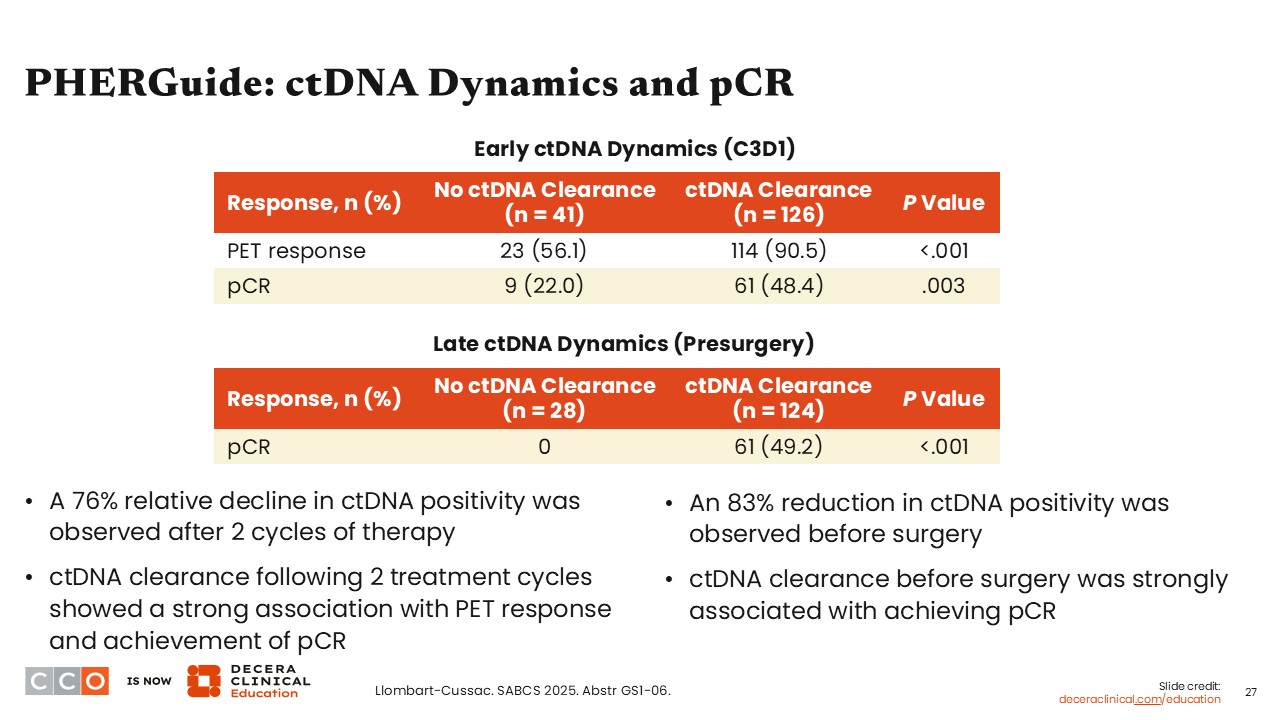
PHERGuide: ctDNA Dynamics and pCR
Virginia Kaklamani, MD, DSc:
Taking a closer look at ctDNA dynamics and pCR in the PHERGuide study, most patients with ctDNA-positive status at baseline (76%) converted to ctDNA-negative status after 2 cycles of treatment. Of importance, the majority (97%) of those with ctDNA-negative status at baseline remained as such at cycle 3, and patients without ctDNA clearance had a significantly lower rate of response measured by PET scan compared with those with ctDNA clearance (P <.001).
I think this study showed that ctDNA had a strong association with treatment response.
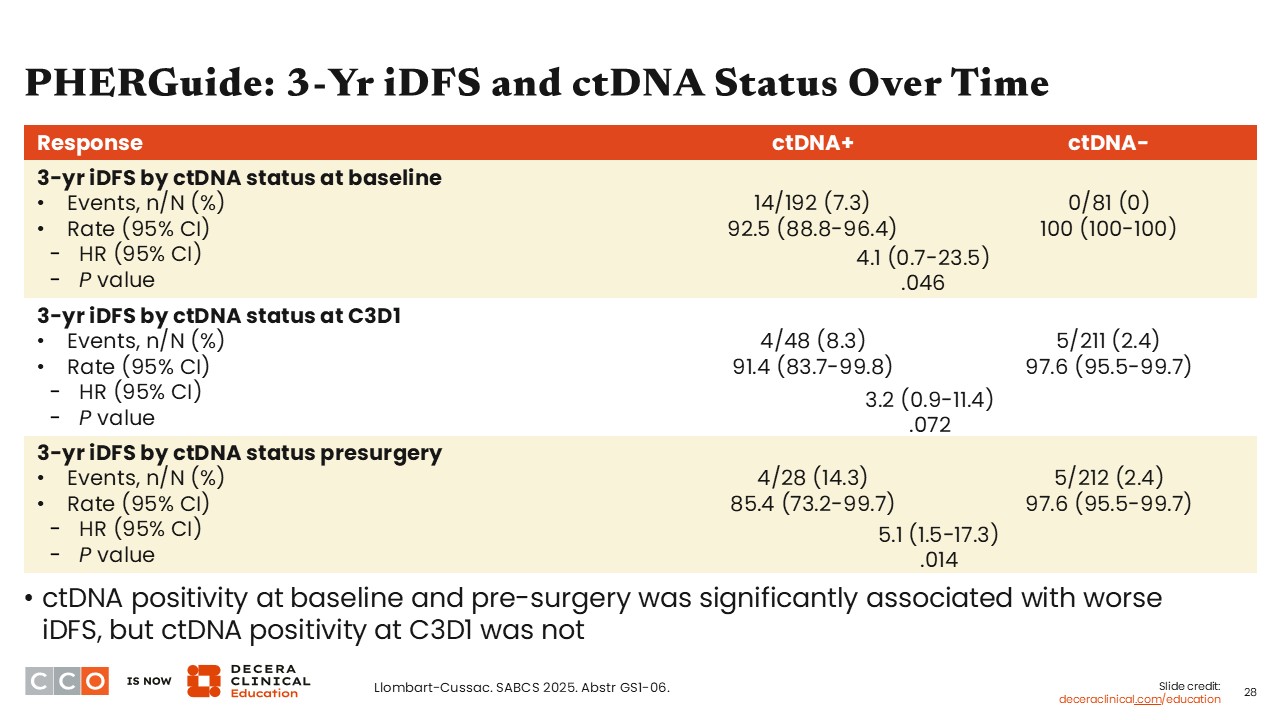
PHERGuide: 3-Yr iDFS and ctDNA Status Over Time
Virginia Kaklamani, MD, DSc:
Patients with no ctDNA at baseline had a better 3-year iDFS rate compared with those who had positive ctDNA status (100% vs 92.5%, respectively; hazard ratio: 4.1; 95% CI: 0.7-23.5; P = .046). This observation was consistent when looking at the ctDNA status at cycle 3 Day 1 and based on ctDNA status before surgery.
What future studies now need to determine is whether ctDNA assessments can help us stratify patients into different lines of therapy. This is something that is currently missing from this study.
PHERGuide: 3-Yr iDFS and ctDNA Dynamics
Virginia Kaklamani, MD, DSc:
Looking at early vs late ctDNA dynamics, patients without ctDNA at baseline and those who experienced early ctDNA clearance had a better 3-year iDFS rate vs those who did not achieve ctDNA clearance (100% vs 95.8% vs 92.2%, respectively; hazard ratio: 2.0; 95% CI: 0.5-8.5; P = .31).
There was a similar trend regarding late ctDNA dynamics before surgery where patients without ctDNA at baseline and those who experienced ctDNA clearance had a better 3-year iDFS rate vs those who did not achieve ctDNA clearance, and this was statistically significant (100% vs 95.8% vs 83.6%, respectively; hazard ratio: 4.5; 95% CI: 1.2-16.6; P = .027).
ALTTO ET Analysis: Study Design
Virginia Kaklamani, MD, DSc:
At SABCS 2025, we saw data presented from an exploratory analysis of the phase III ALTTO (BIG 2-06) trial examining the incorporation of lapatinib in anti-HER2 therapy for patients with HER2-positive EBC. Patients with HER2-positive disease were divided into several anti-HER2 treatment cohorts: trastuzumab monotherapy, lapatinib monotherapy, trastuzumab followed by lapatinib, or trastuzumab with lapatinib combination. Then, 3 different chemotherapy modalities (ie, chemotherapy, anthracycline followed by taxane, or docetaxel with carboplatin) were administered per physician’s choice, and patients with HR-positive disease received adjuvant ET after completion of chemotherapy.16,17
The exploratory analysis presented at SABCS 2025 evaluated the efficacy of ET therapy with AI compared with a selective estrogen receptor modulator (SERM; tamoxifen), both with ovarian function suppression treatment (if premenopausal), in patients with HR-positive/HER2-positive EBC. Of note, this is an area in which we are lacking data to inform personalized care. In the phase III studies like SOFT and TEXT, most patients (77.2%-94.1%) had HER2-negative disease.18 That is why we do not have much data to inform ET choices (ie, AI or tamoxifen) when looking to help patients with HR-positive/HER2-positive EBC to improve outcomes.
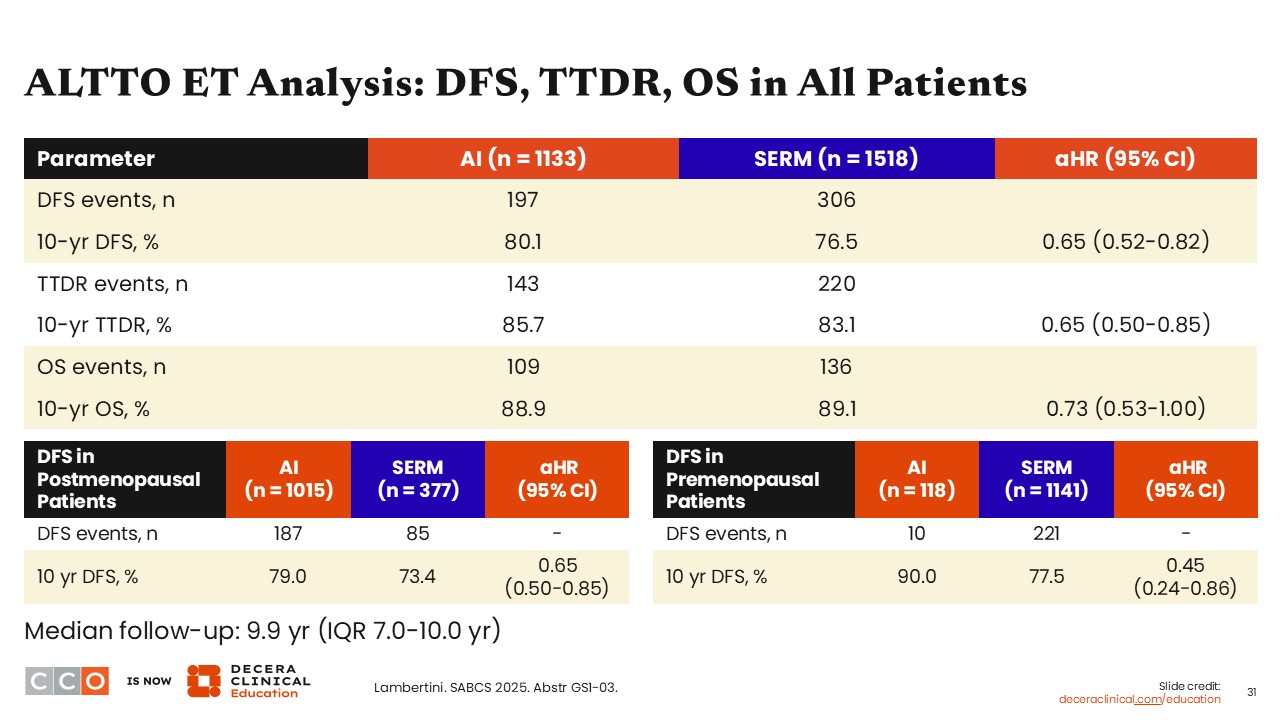
ALTTO ET Analysis: DFS, TTDR, OS in All Patients
Virginia Kaklamani, MD, DSc:
After a median follow-up of 9.9 years, the patients receiving AI (n = 1133) compared with SERM (n =1518) had improved 10-year DFS rates of 80.1% vs 76.5% (hazard ratio: 0.65; 95% CI: 0.52-0.82) and 10-year rates for time to distant relapse (TTDR rates: 85.7% 83.1%; hazard ratio: 0.65; 95% CI: 0.50-0.85).
When looking at DFS according to menopausal status, both postmenopausal patients (hazard ratio: 0.65; 95% CI: 0.50-0.85) and premenopausal patients (hazard ratio: 0.45; 95% CI: 0.24-0.86) experienced more benefit with AI compared with SERM.
By contrast, the 10-year OS rates were comparable between the 2 arms, and the confidence interval reached 1 (hazard ratio: 0.73; 95% CI: 0.53-1.00).
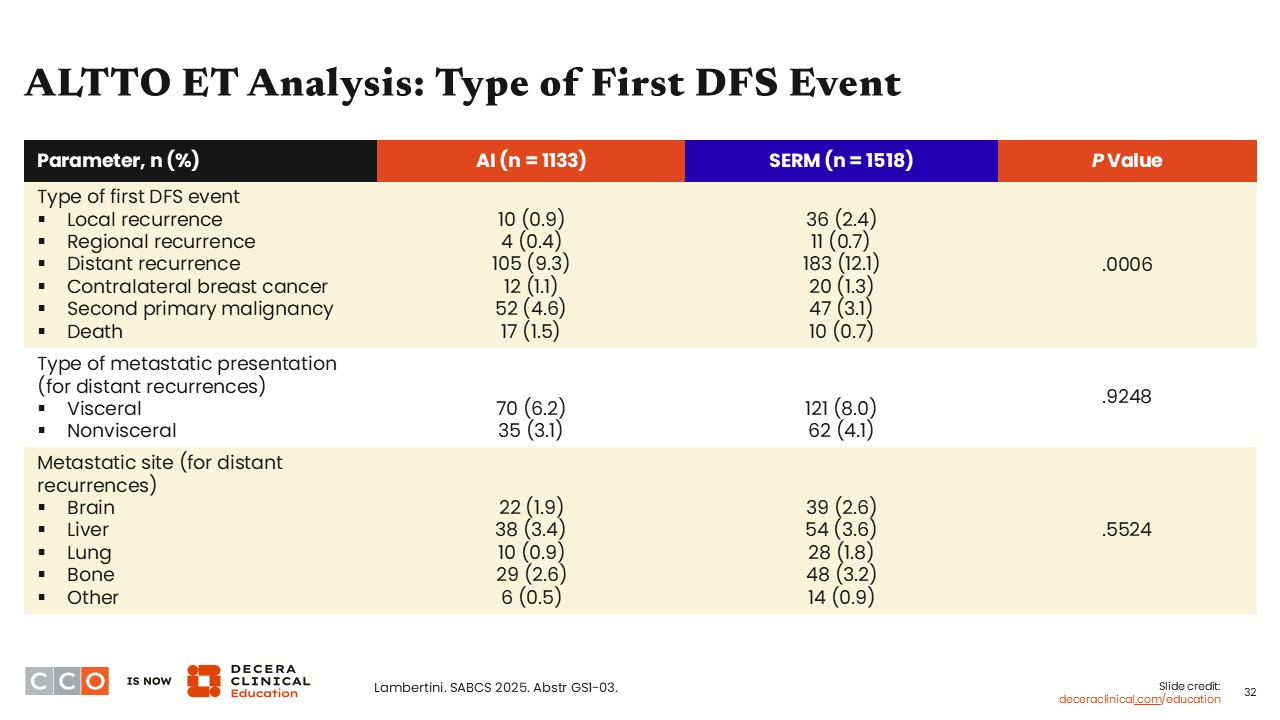
ALTTO ET Analysis: Type of First DFS Event
Virginia Kaklamani, MD, DSc:
Looking at the type of recurrence, there were fewer DFS events for local (0.9% vs 2.4%), regional (0.4% vs 0.7%), and distant recurrence (9.3% vs 12.1%) for those receiving AI vs SERM (P = .0006). Yet there was not a huge difference between the 2 study arms in terms of metastatic sites for distant recurrences (P = .5524). Type of metastatic presentation (for distant recurrences) and metastatic site (for distant recurrences) were comparable between the 2 arms, with no statistical differences noted (P >.05).
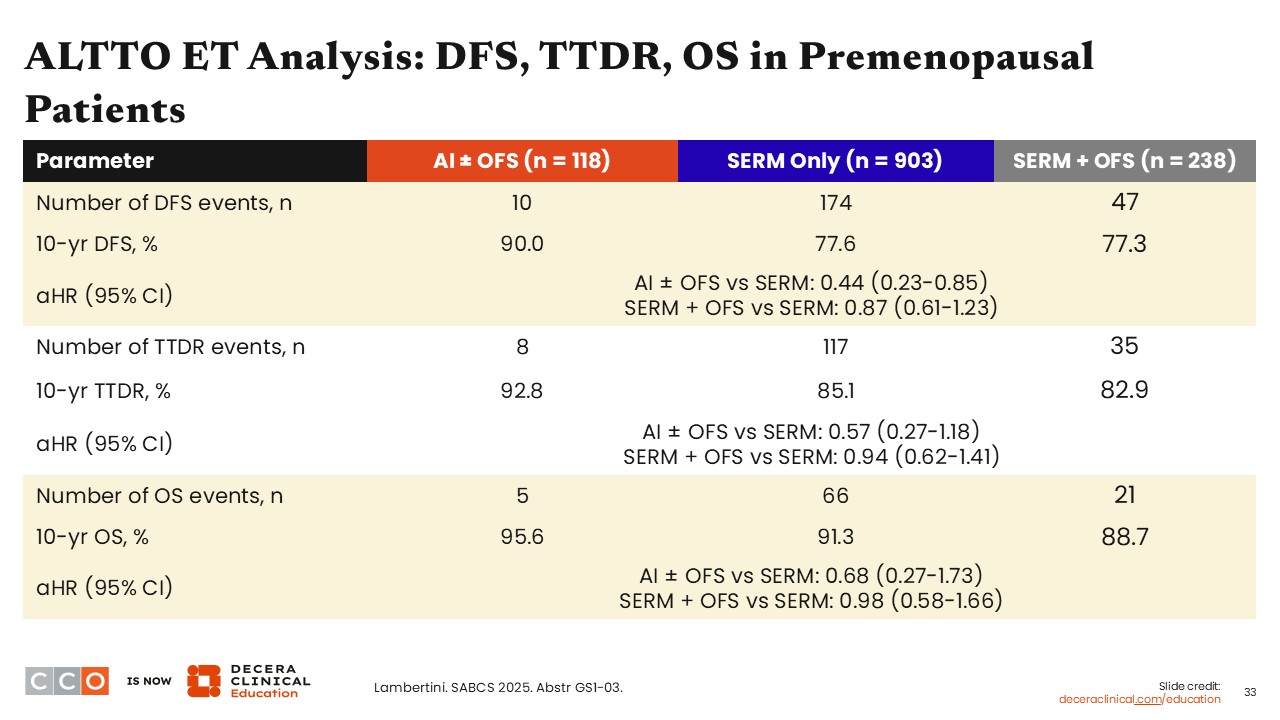
ALTTO ET Analysis: DFS, TTDR, OS in Premenopausal Patients
Virginia Kaklamani, MD, DSc:
Investigators also performed DFS, TTDR, and OS analyses in patients with premenopausal status and based on receipt of OFS vs not. Results show data were similar to what we have seen before. In other words, patients who received AI with OFS compared with SERM only or SERM plus OFS benefited more with regard to 10-year DFS rate (90.0% vs 77.6% vs 77.3%), 10-year TTDR rates (92.8% vs 85.1% vs 82.9%), and 10-year OS rates (95.6% vs 91.3% vs 88.7%).