CE / CME
Key Studies in Lymphomas/CLL: Independent Conference Coverage of the 2025 ASH Annual Meeting
Physician Assistants/Physician Associates: 1.00 AAPA Category 1 CME credit
Pharmacists: 1.00 contact hour (0.1 CEUs)
Physicians: maximum of 1.00 AMA PRA Category 1 Credit™
Nurse Practitioners/Nurses: 1.00 Nursing contact hour
Released: February 26, 2026
Expiration: August 25, 2026
Activity
CLL17: Continuous Ibrutinib vs Fixed-Duration Venetoclax Plus Obinutuzumab or Venetoclax Plus Ibrutinib for Untreated CLL
Brad Kahl, MD
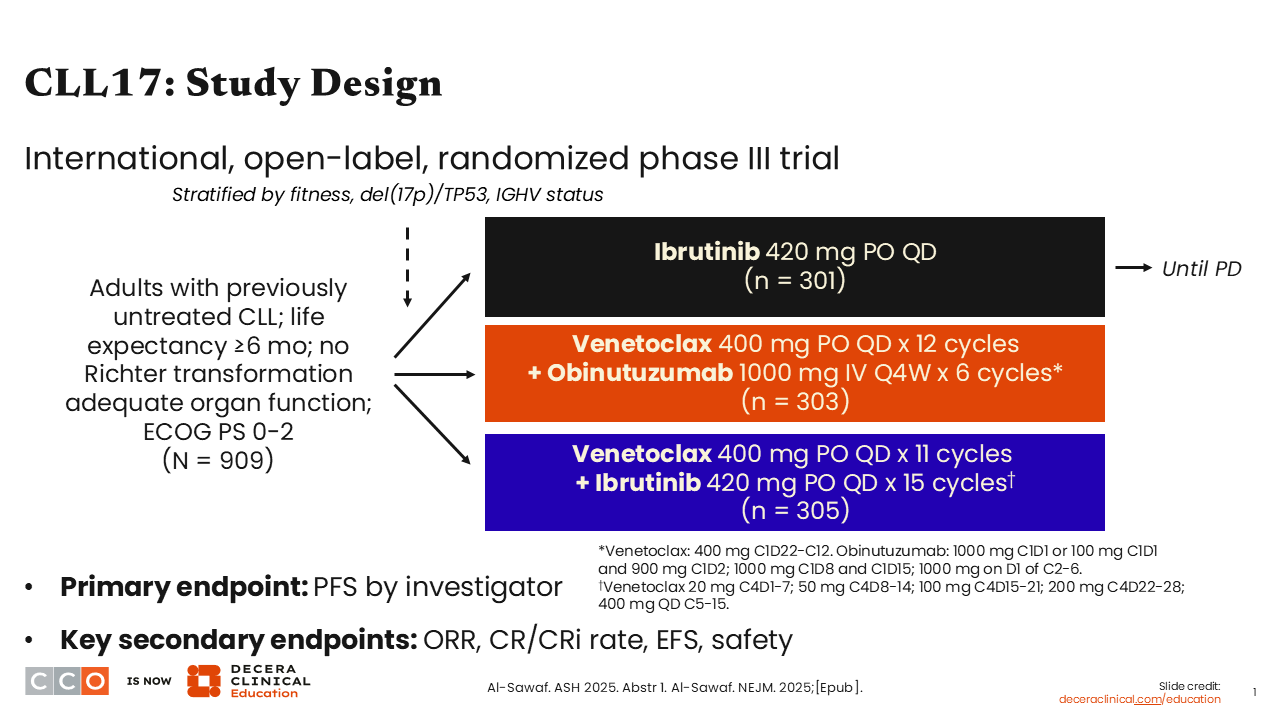
Let’s begin with a discussion of the phase III CLL17 trial, which was one of the more impactful presentations at the 2025 ASH meeting.1,2
The paradigm for frontline CLL therapy has been to offer patients either indefinite covalent BTK inhibitor therapy or a fixed-duration regimen.3 Historically, venetoclax plus obinutuzumab has been the fixed-duration regimen most commonly used in the US, although with positive data from the phase III AMPLIFY study last year, fixed-duration acalabrutinib plus venetoclax may also be considered.4 CLL17 evaluated combinations of fixed-duration venetoclax plus obinutuzumab or venetoclax plus ibrutinib.1,2
Patients with untreated CLL were randomized 1:1:1 to either indefinite ibrutinib, fixed-duration venetoclax plus obinutuzumab, or venetoclax plus ibrutinib. With this last combination, ibrutinib was given for 3 months, with venetoclax added in cycle 4, followed by another year of this all-oral combination.
The study enrolled over 900 patients in 2021 and 2022. The median observation time was 34 months. The primary endpoint was noninferiority for PFS with the fixed-duration regimens.
CLL17: Baseline Characteristics
Brad Kahl, MD
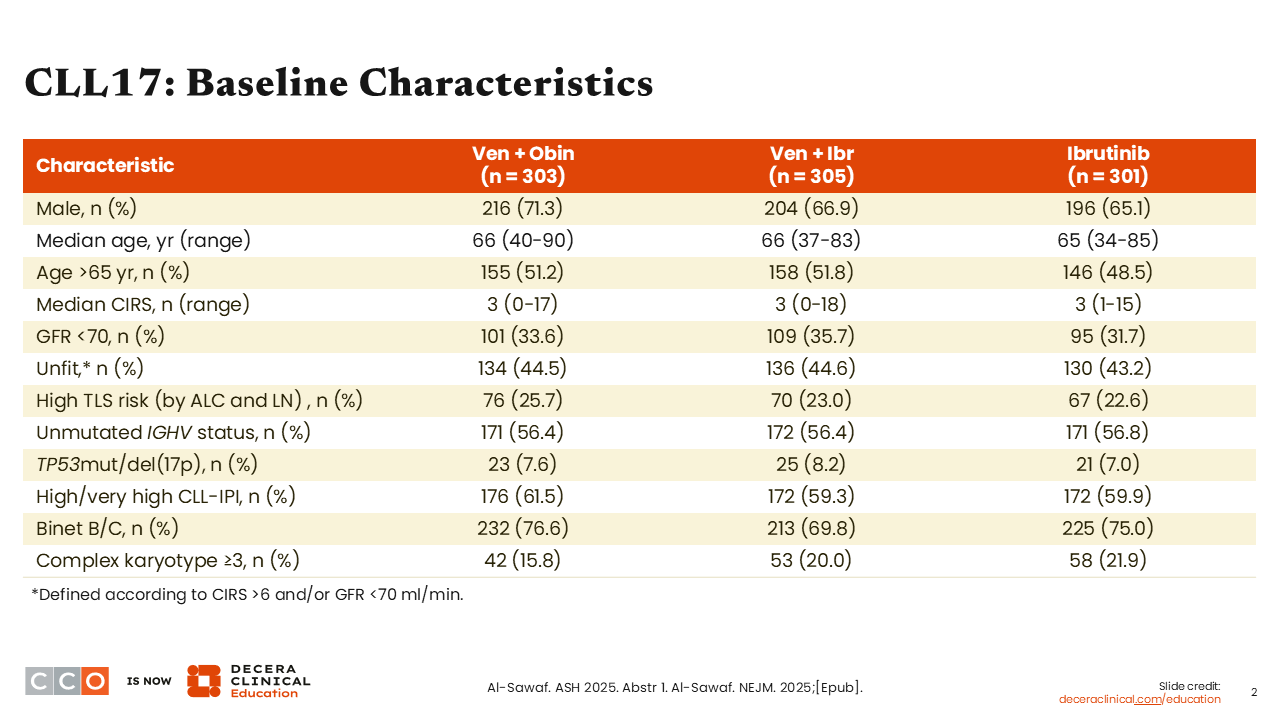
Patients were well-balanced between arms. The median age was 66 years, so maybe a slightly younger population than what we typically see in our practices, but a fairly characteristic breakdown with a mix of mutated and unmutated IGHV, and approximately 7% to 8% of patients with CLL with a TP53 aberrancy.
CLL17: Progression-Free Survival
Brad Kahl, MD
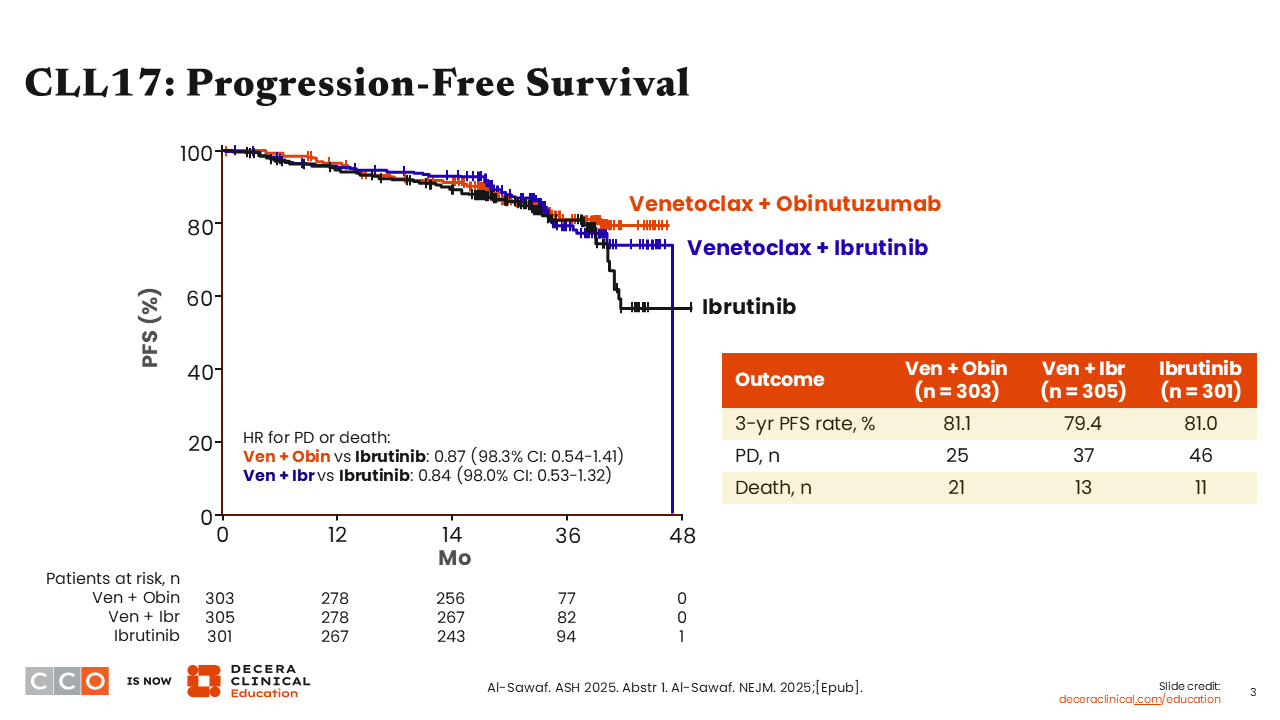
The bottom line from this trial is that PFS was similar between the 3 regimens, with 3-year PFS rates of 81.1%, 79.4%, and 81.0% in the venetoclax plus obinutuzumab, venetoclax plus ibrutinib, and ibrutinib arms, respectively. As such, both fixed-duration targeted therapies achieved noninferior PFS vs continuous ibrutinib.
CLL17: PFS by Subgroup
Brad Kahl, MD
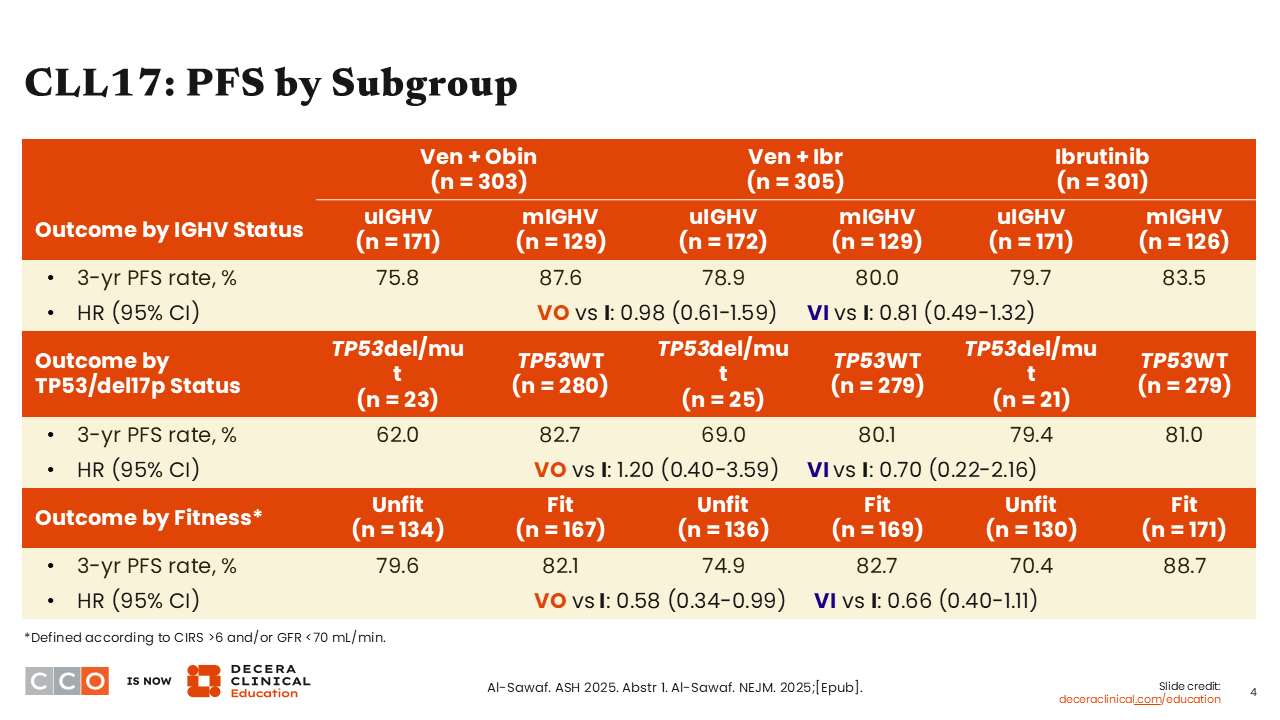
Looking at the PFS subgroup analysis, I do not think we can say that the regimens are performing differently in CLL with mutated vs unmutated IGHV. There may be the beginning of a signal that venetoclax plus obinutuzumab is performing better in CLL with mutated IGHV vs unmutated IGHV, but this is something we had seen in previous trials with venetoclax plus obinutuzumab.
Differences might emerge with longer follow-up, so I do not think the story on the durability of remission is quite settled on these 3 regimens. We probably need 4-6 years of follow-up before we have the final answer there.
There were not many patients in this trial who had CLL with TP53 mutations, but as expected, the time-limited therapies did not appear to work as well in TP53-aberrant disease.
CLL17: Overall Survival and Causes of Death
Brad Kahl, MD
There were no differences in overall survival (OS) between regimens. Numerically, 3-year OS rates were slightly lower in the venetoclax plus obinutuzumab arm. Although not statistically significant, there were a few more infectious deaths observed with venetoclax plus obinutuzumab than with the other regimens.
The numbers are small, but this is something that has caught my attention, and it is starting to inform my practice, whereby I might favor venetoclax plus obinutuzumab for my younger patients with IGHV-mutated disease, but in my older, frail patients, I might start to shy away from that regimen because of the comparative infection risk.
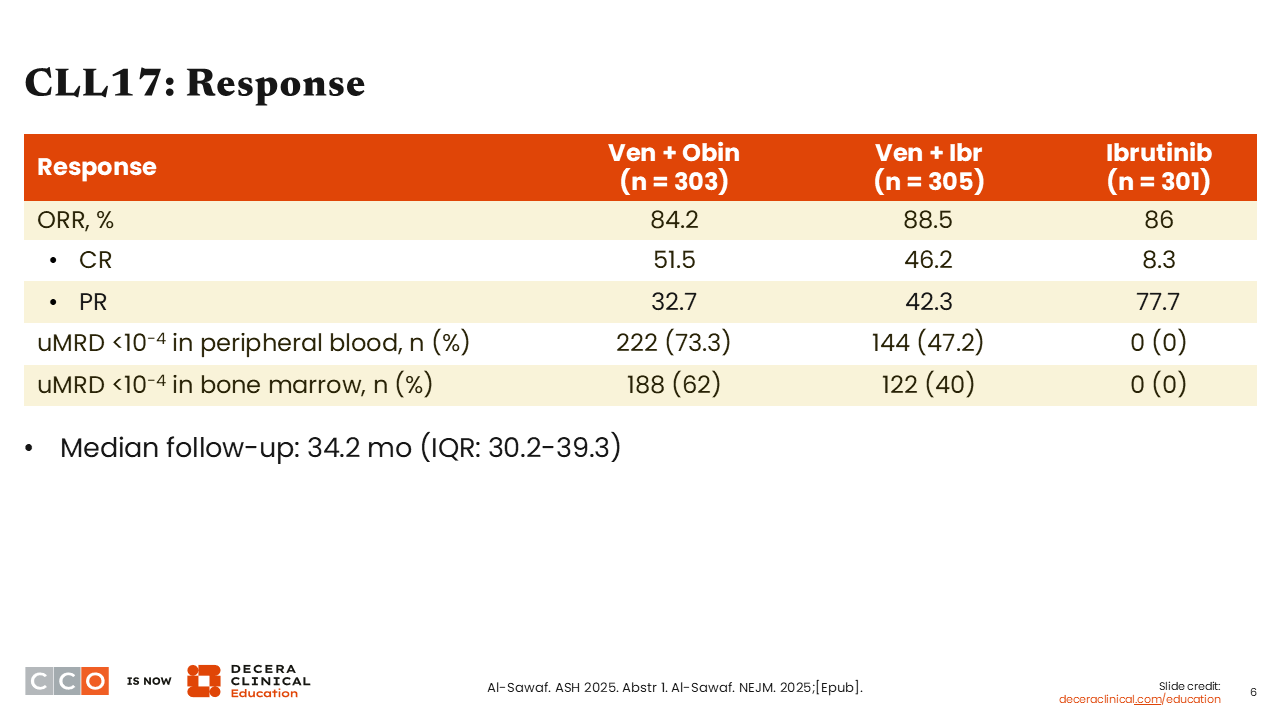
CLL17: Response
Brad Kahl, MD
In terms of response, the ORRs were above 80% in all 3 arms. As expected, single-agent ibrutinib rarely produced complete remissions. Among patients treated with venetoclax plus obinutuzumab, 51.5% achieved complete remission; with venetoclax plus ibrutinib, 46.2% of patients achieved complete remission.
The probability of reaching a measurable residual disease (MRD) undetectable (10-4) state in the peripheral blood was highest in the venetoclax plus obinutuzumab arm (73%), with 47% of patients treated with venetoclax plus ibrutinib becoming MRD undetectable. Patients treated with single-agent ibrutinib rarely reach MRD undetectable status.
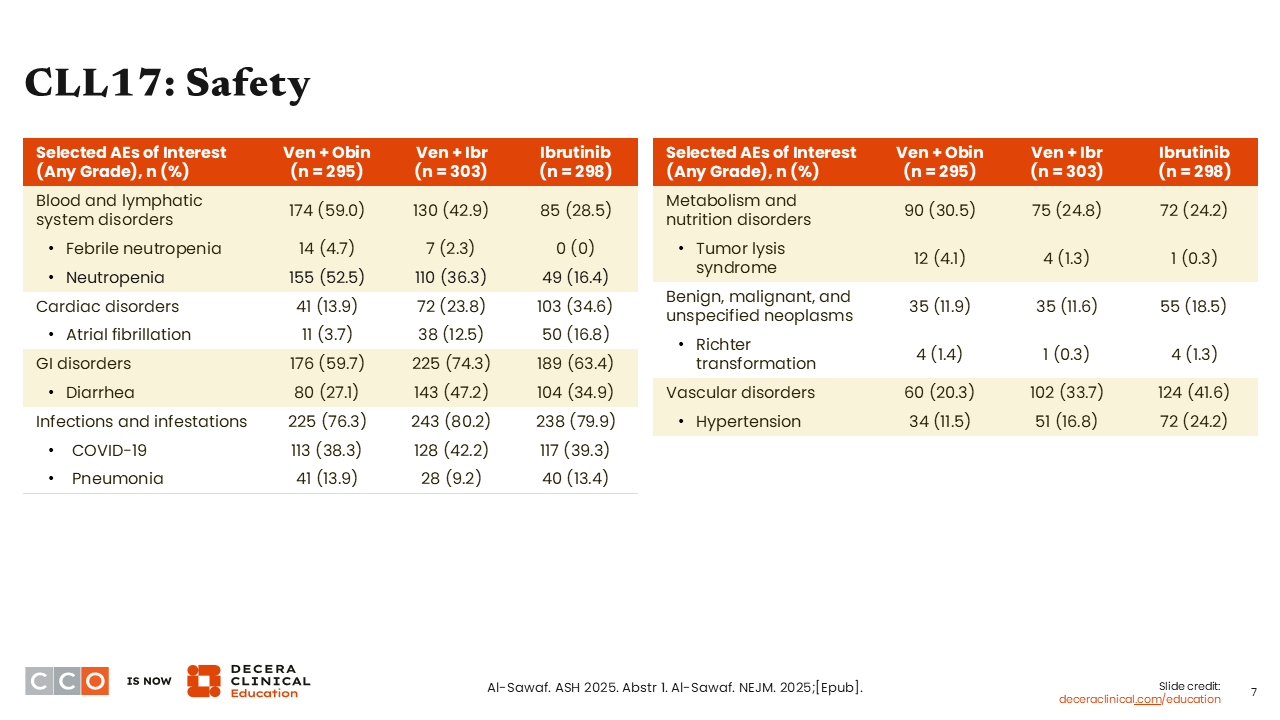
CLL17: Safety
Brad Kahl, MD
There were really no new safety signals that came out of this trial. Each of these regimens is well-established. As in the past, the biggest risk in any CLL trial is the risk of infection.
Conclusions
Brad Kahl, MD
The bottom line from this study was that the 2 fixed-duration regimens appeared to perform similarly to indefinite ibrutinib, and it makes the 2 time-limited options good options to offer patients. I do not think there were any big new safety signals that were observed, and so we now have 3 very good options to offer our patients: time-limited venetoclax plus obinutuzumab, time-limited venetoclax plus a BTK inhibitor, or indefinite therapy with a BTK inhibitor, and I think it just takes some individualization.
Jeremy Abramson, MD
I agree completely. I think this is a very important study. It is the first study that we have directly comparing a time-limited approach vs a continuous BTK inhibitor–based approach.
What is nice about this study is that we see noninferiority with time-limited therapy. In general, I prefer time-limited therapy where possible, as it offers less exposure to drugs and less exposure to potential toxicities over time. This study validates the fact that there are 2 different time-limited strategies that effectively give us very similar PFS results vs a continuous therapy.
I view this study as a comparison of paradigms with a BTK inhibitor only, a BTK inhibitor with venetoclax, and venetoclax plus obinutuzumab. I no longer give ibrutinib as initial therapy in CLL or in any other disease. This study helps show us why: the ibrutinib-containing arms have a 12% to 15% incidence of atrial fibrillation, along with the other potential side effects of BTK inhibition.
But I think in the context of the AMPLIFY trial, the CLL17 trial validates that a BTK inhibitor plus venetoclax-based approach is highly effective. As Dr Kahl pointed out, these studies have shown that obinutuzumab-containing arms have higher rates of infection, including serious infections. This study adds to my shift to considering acalabrutinib plus venetoclax as a highly effective time-limited therapy option for most patients.
Venetoclax plus obinutuzumab is validated here as highly effective and has the highest rate of MRD undetectability; this does not appear to impact PFS, at least at the 3-year time point. So, both are appropriate time-limited strategies, and I think this continues the shift away from single-agent BTK inhibition.
BRUIN CLL-314: Pirtobrutinib vs Ibrutinib in Treatment-Naive and BTKi-Naive R/R CLL/SLL
Jeremy Abramson, MD
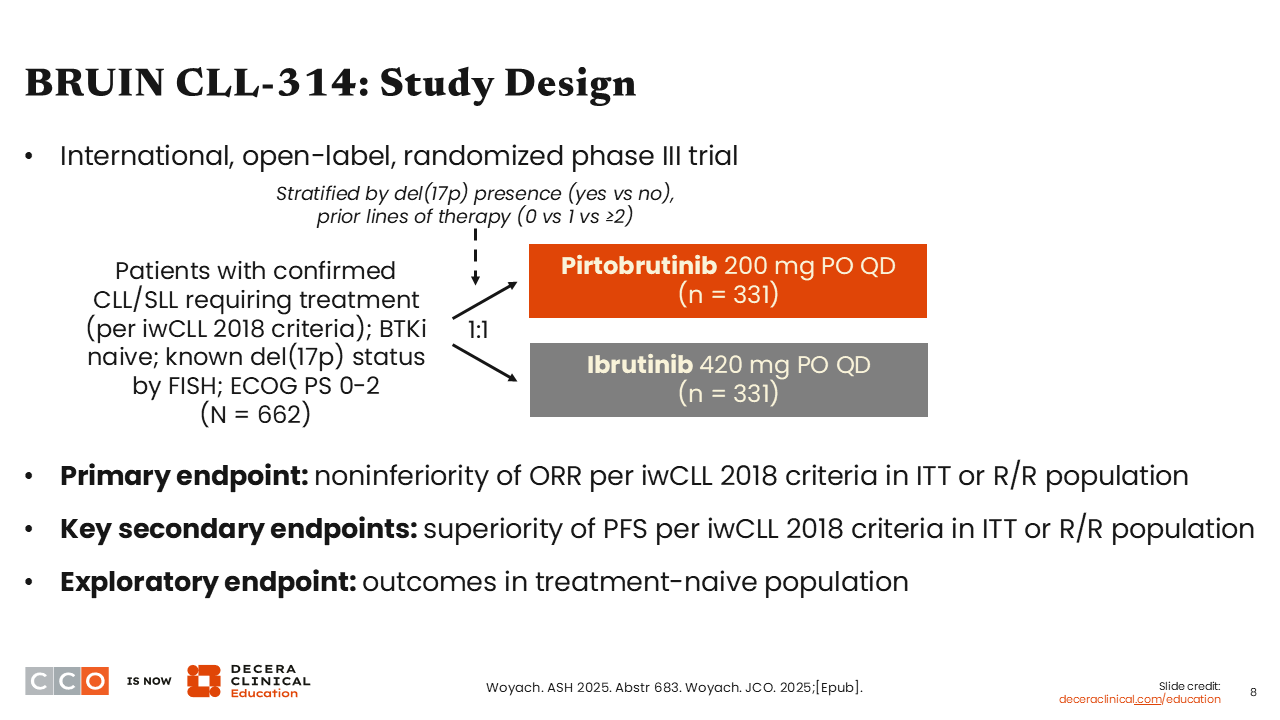
BRUIN CLL-314 was another very important study with data presented at ASH 2025. This phase III study compared pirtobrutinib vs ibrutinib in treatment-naive or R/R CLL.5 This is the very first study we have comparing covalent vs noncovalent BTK inhibitors in a randomized, controlled trial.
Our current standard of care in CLL includes single-agent BTK inhibition as a standard option, typically favoring a next-generation covalent BTK inhibitor (acalabrutinib or zanubrutinib) based on improved tolerability with these agents compared with ibrutinib.6,7 In the R/R setting, we also now have the noncovalent BTK inhibitor pirtobrutinib available.8,9
The BRUIN CLL-314 study assessed whether the noncovalent BTK inhibitor pirtobrutinib is noninferior to the covalent BTK inhibitor ibrutinib for BTK inhibitor-naive CLL.5
Just over 600 patients were randomized 1:1 to receive pirtobrutinib 200 mg per day or ibrutinib 420 mg per day. As is typical, these drugs were continued until progression or intolerance. Patients in this study were treatment-naive or had previously treated CLL, but of importance, they had to be BTK inhibitor–naive to go on this study. The primary endpoint was noninferiority by response rate, and superiority for PFS was a secondary endpoint.
BRUIN CLL-314: Baseline Characteristics
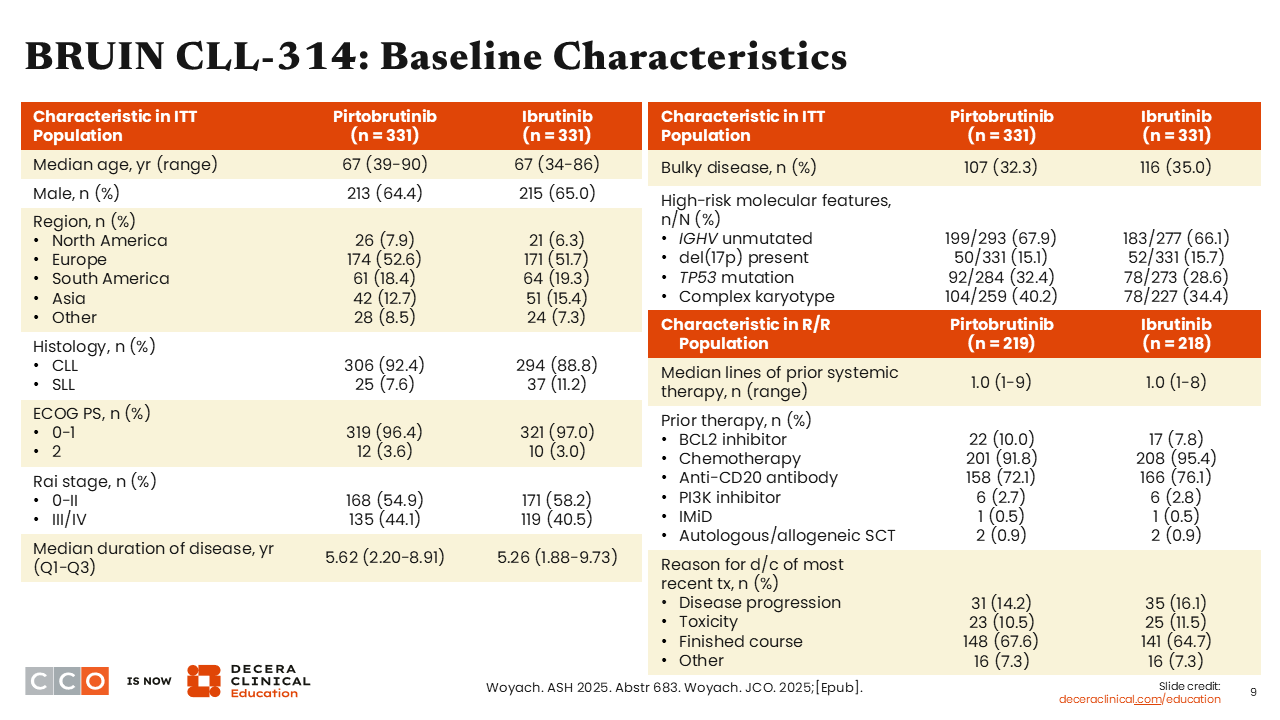
Jeremy Abramson, MD
Patient characteristics were well-balanced across both arms. The median patient age was 67 years. Patients had received a median of 1 prior line of therapy. Only approximately one third of patients had a CLL with a TP53 mutation, approximately 15% had del(17p), and two thirds had IGHV unmutated disease.
BRUIN CLL-314: ORR
Jeremy Abramson, MD
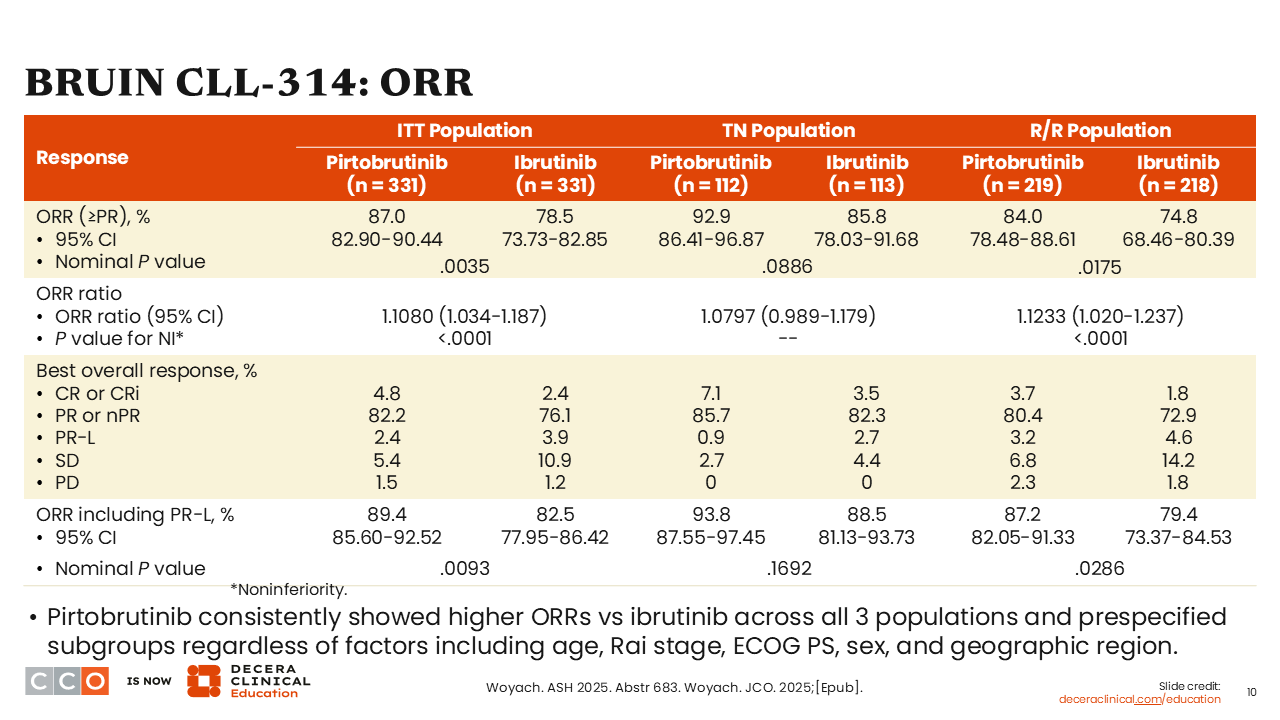
Looking at the primary endpoint of ORR, although outcomes were favorable in both arms, a higher ORR was observed with pirtobrutinib at 87.0% compared with ibrutinib at 78.5% in the ITT population (P = .0035). ORRs were higher with pirtobrutinib in both the treatment-naive and R/R populations.
Remember, this study was powered as a noninferiority study, and it is clear that pirtobrutinib is noninferior to ibrutinib in terms of overall response.
BRUIN CLL-314: PFS by INV in ITT and R/R Populations
Jeremy Abramson, MD
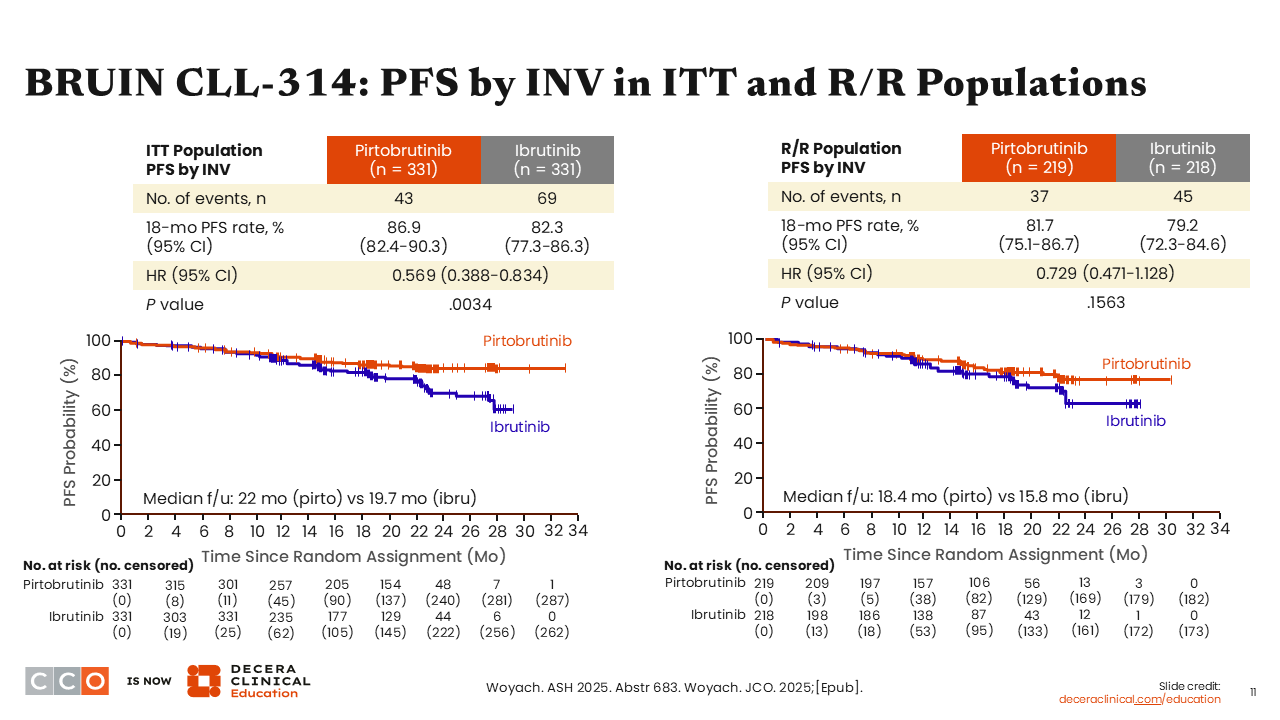
Turning to the secondary endpoint of PFS, after a median follow-up of 22 months in the pirtobrutinib arm and 19.7 months in the ibrutinib arm, PFS favoring pirtobrutinib over ibrutinib was observed, with a 43% reduction in the risk of progression or death (HR: 0.569; P = .0034) in the ITT population. The 18-month PFS rates were 87% and 82%, respectively.
In the R/R population, there was a small numerical difference in PFS with pirtobrutinib vs ibrutinib, which did not meet statistical significance (HR: 0.729; P = .1563).
BRUIN CLL-314: PFS by IRC in ITT and PFS by INV in TN Population
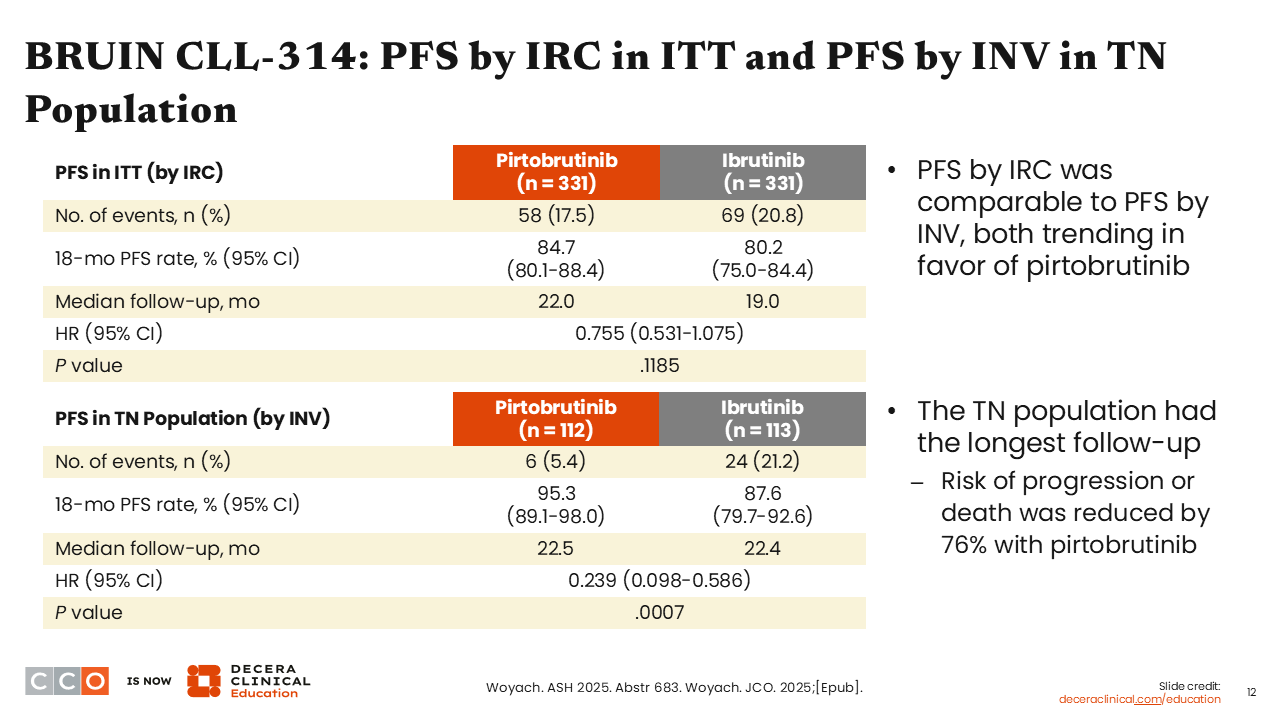
Jeremy Abramson, MD
We see that the majority of PFS benefit was garnered in the treatment-naive population, with 18-month PFS rates of 95.3% with pirtobrutinib vs 87.6% with ibrutinib (HR: 0.239, P = .0007).
BRUIN CLL-314: Select Treatment-Emergent AEs
Jeremy Abramson, MD
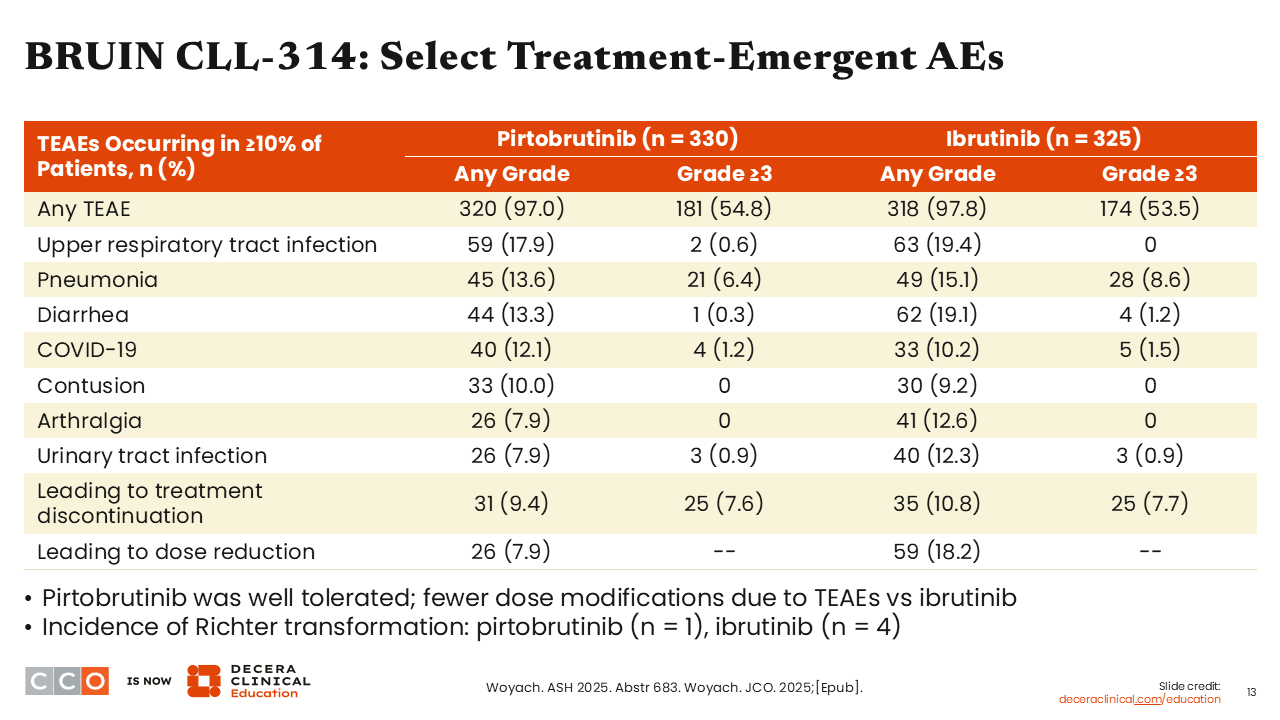
Turning our attention to safety, we see, not surprisingly, that pirtobrutinib has a more favorable safety profile. There is a risk of diarrhea in both arms, though this is a bit lower with pirtobrutinib.
The rate of TEAEs leading to dose reductions was also lower with pirtobrutinib compared with ibrutinib (7.9% vs 18.2%), reflecting its more favorable tolerability.
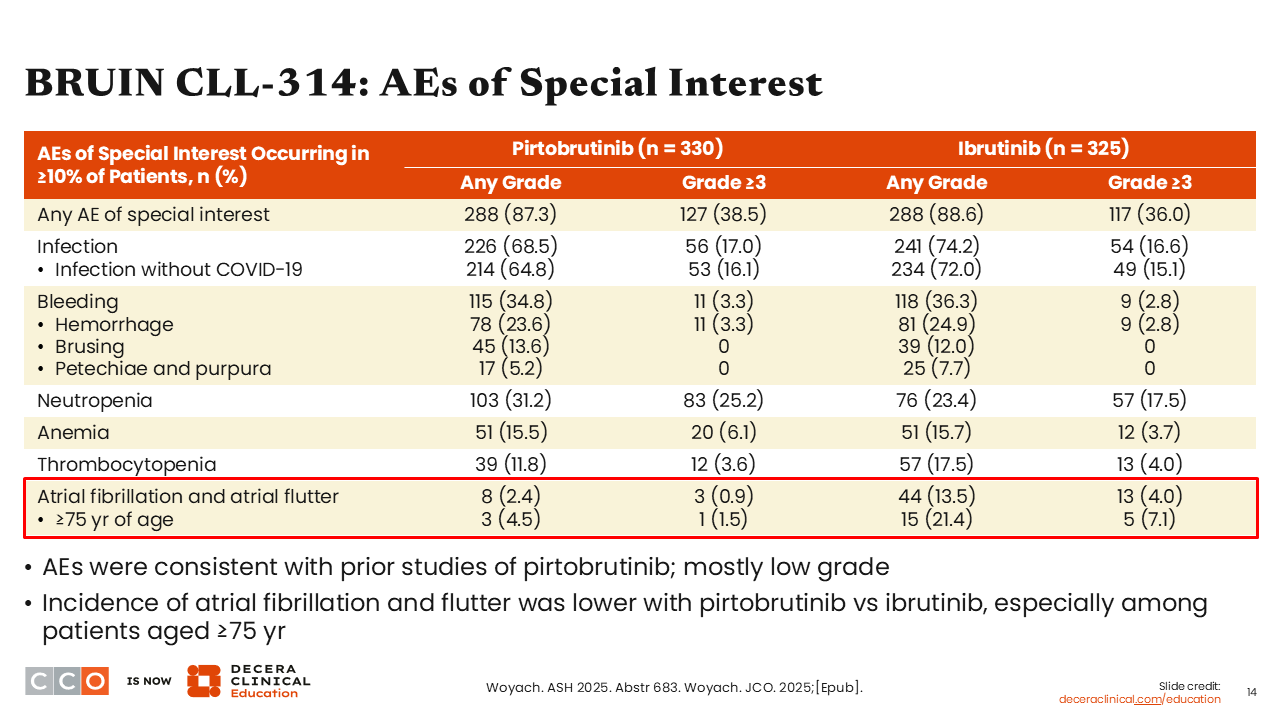
BRUIN CLL-314: AEs of Special Interest
Jeremy Abramson, MD
In particular, the rate of atrial fibrillation was lower with pirtobrutinib. Whereas we see an incidence of 13.5% in patients treated with ibrutinib, the incidence was only 2.4% in patients receiving pirtobrutinib. There is evidence of a class effect of bruising, which we see in both arms.
BRUIN CLL-314: Conclusions
Jeremy Abramson, MD
My take-home message here is that pirtobrutinib is a highly effective noncovalent BTK inhibitor. In this first head-to-head trial of a covalent BTK inhibitor (ibrutinib) and a noncovalent BTK inhibitor (pirtobrutinib), we clearly see evidence of noninferiority.
I would argue we are actually seeing evidence of superiority, suggesting a benefit with pirtobrutinib over ibrutinib in terms of response as well as PFS, particularly in treatment-naive CLL. We are also seeing a more favorable safety profile over ibrutinib, particularly in terms of reduced incidence of atrial fibrillation and reduced need for dose reduction, favoring pirtobrutinib.
These data continue to highlight pirtobrutinib as a highly effective noncovalent BTK inhibitor in CLL with a more favorable safety profile compared with ibrutinib.
Brad Kahl, MD:
I agree. This is an important study. However, its impact in the United States may be modest because most people are not using ibrutinib anymore. The natural question, then, is how would pirtobrutinib stack up against the BTK inhibitors we are more likely to use, like zanubrutinib or acalabrutinib? Obviously, we cannot answer those questions from this trial.
Of note, data from the randomized phase III BRUIN CLL-313 trial were also presented at ASH 2025. In this study, pirtobrutinib demonstrated significant efficacy improvements over bendamustine plus rituximab in patients with treatment-naive CLL/SLL (24-month PFS: 93.4% vs 70.7%; P <.0001; ORR: 94.3% vs 80.9%). Pirtobrutinib was again associated with low rates of atrial fibrillation/flutter (1.4%).
In December 2025, we saw the pirtobrutinib label expanded.10 Whereas pirtobrutinib previously had accelerated approval for use after failure of a BTK inhibitor and a BCL2 inhibitor, it now has a full approval and can be used after just a covalent BTK inhibitor. This means pirtobrutinib could be used in the second line ahead of a BCL2 inhibitor.
The BRUIN CLL-314 data do not tell you whether to do that or not, but the bottom line is that we have another very good drug in CLL, and exactly where to position it in lines of therapy is a story in progress right now, and it is not totally clarified by this particular study.
Jeremy Abramson, MD:
I think that is very true. In the ALPINE trial in R/R CLL, zanubrutinib was associated with a superior PFS over ibrutinib, so not all covalent BTK inhibitors are created equal.7 However, it is nice to have multiple options. I am probably still sticking with a next-generation covalent BTK inhibitor ahead of the noncovalent, ahead of definitive data that there is superiority in that context, but a rising tide lifts all boats, and the more effective drugs we have, the better for our patients.
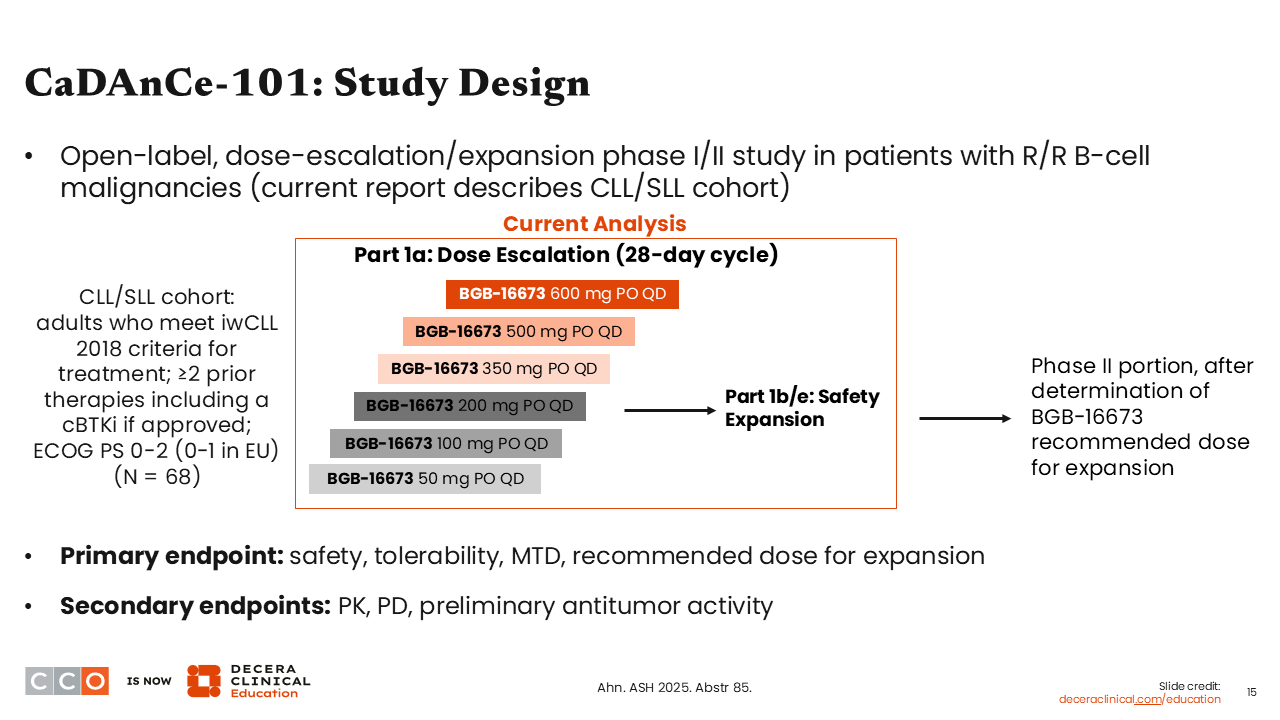
CaDAnCe-101: Phase I Study Evaluating BTK Degrader BGB-16673 in R/R CLL/SLL
Brad Kahl, MD:
Next, let’s discuss the updated analysis of the phase I CaDAnCe-101 study of the BTK degrader BGB-16673 in R/R CLL.11
BTK degraders are an interesting class of drugs that disable the BTK enzyme in a completely different way than BTK kinase inhibitors. By targeting the BTK enzyme for proteasomal degradation, they are relatively unaffected by the traditional mutations that can make BTK inhibitors like acalabrutinib, zanubrutinib, and pirtobrutinib ineffective.12,13 This is a whole new way to attack BTK in CLL and has shown significant promise in clinical trials.
CaDAnCe-101: Baseline Characteristics
This was a heavily pretreated, high-risk group.11 Almost all patients had received a prior covalent BTK inhibitor. Many patients had received a prior noncovalent BTK inhibitor, and most patients had a prior BCL2 inhibitor. TP53 mutations and/or del(17p) were present in 67.6% of patients.
CaDAnCe-101: Overall Safety
Brad Kahl, MD:
BGB-16673 appeared to be relatively safe. There were very few serious treatment-related adverse events (AEs), no serious treatment-related AEs leading to death, and very few AEs leading to treatment discontinuation.
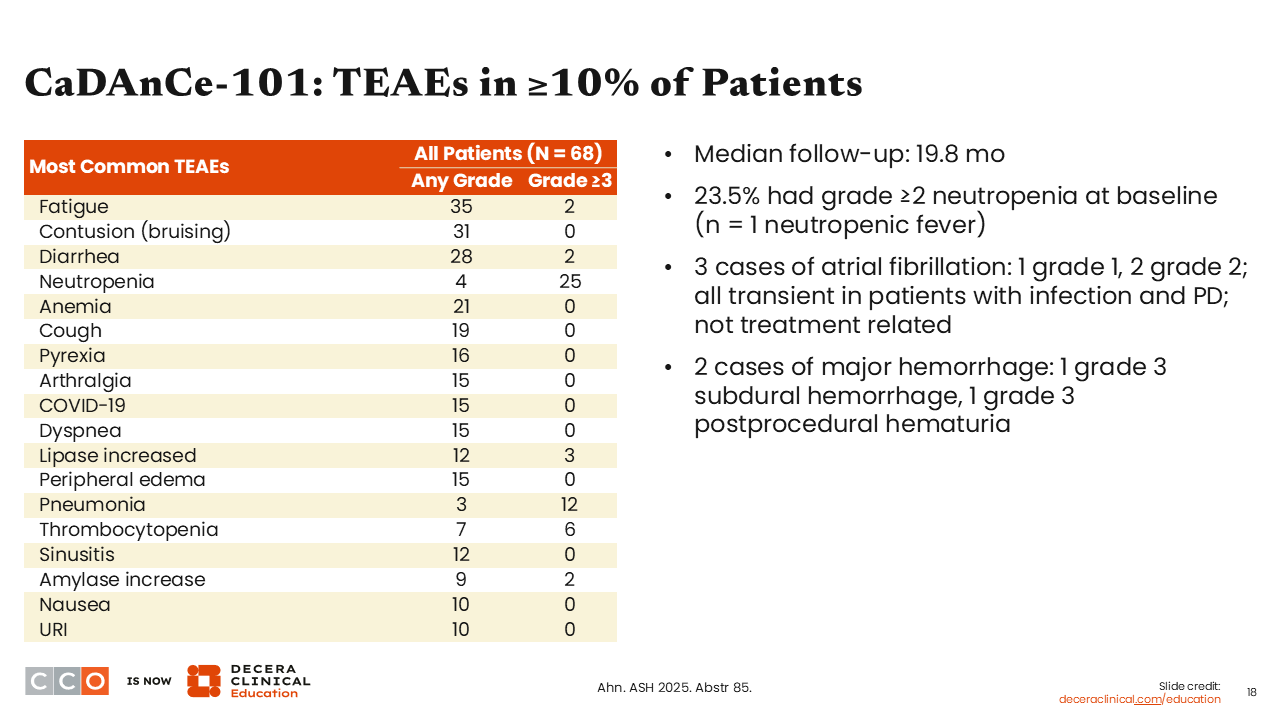
CaDAnCe-101: TEAEs in ≥10% of Patients
Brad Kahl, MD:
AEs with BGB-16673 were similar to what we see with other agents that target BTK. Bruising was reported in 31% of patients; rates of diarrhea were low, there was some neutropenia (25% grade ≥3), which might be unique to this drug, and, as in any CLL study, there is a certain risk for infection, which is a combination of the underlying disease and the therapy that is being administered.
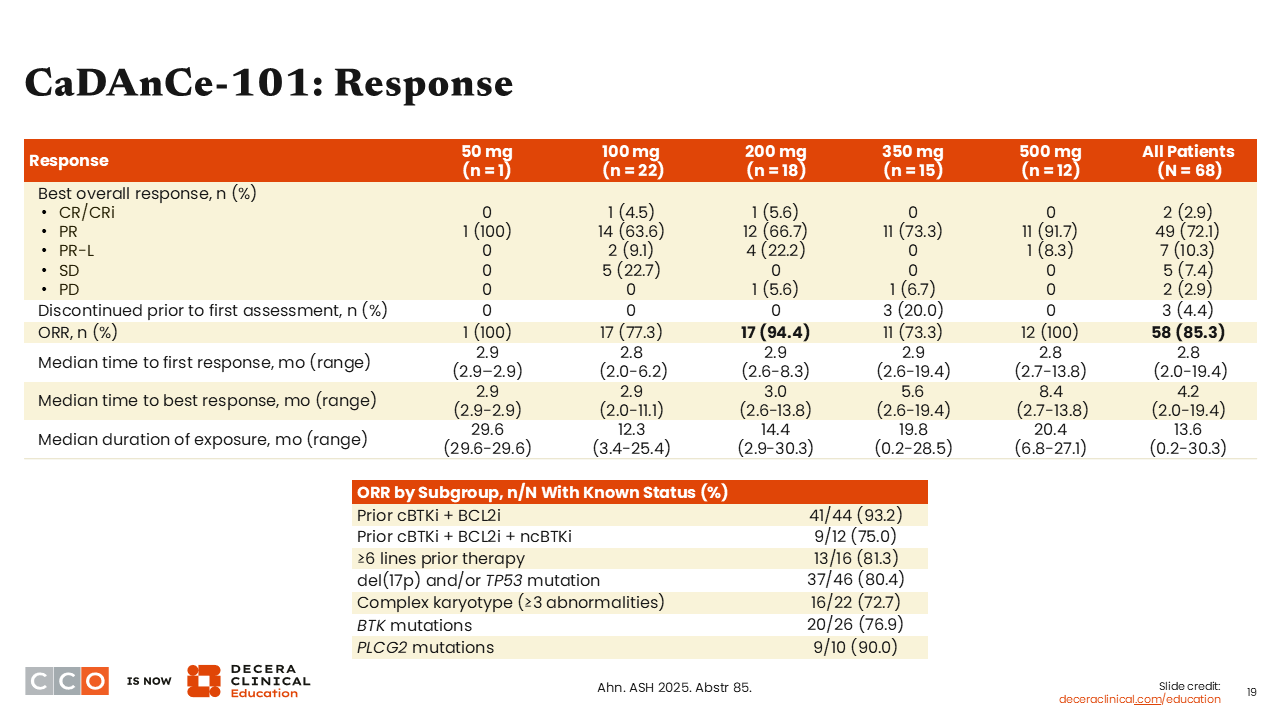
CaDAnCe-101: Response
Brad Kahl, MD:
At BGG-16673 dose levels of 200 mg daily or higher, impressive response rates exceeding 80% to 90% were observed. In the overall population of 68 patients, the ORR was 85%, which is a very impressive response rate in this patient population.
Looking at response rates across subgroups, high ORRs were observed, including patients with BTK mutations, patients previously treated with a BTK inhibitor (covalent and noncovalent) and BCL2 inhibitor, and patients with numerous prior lines of therapy. The bottom line was that it did not matter what prior therapy had been received; all patients responded to this BTK degrader therapy.
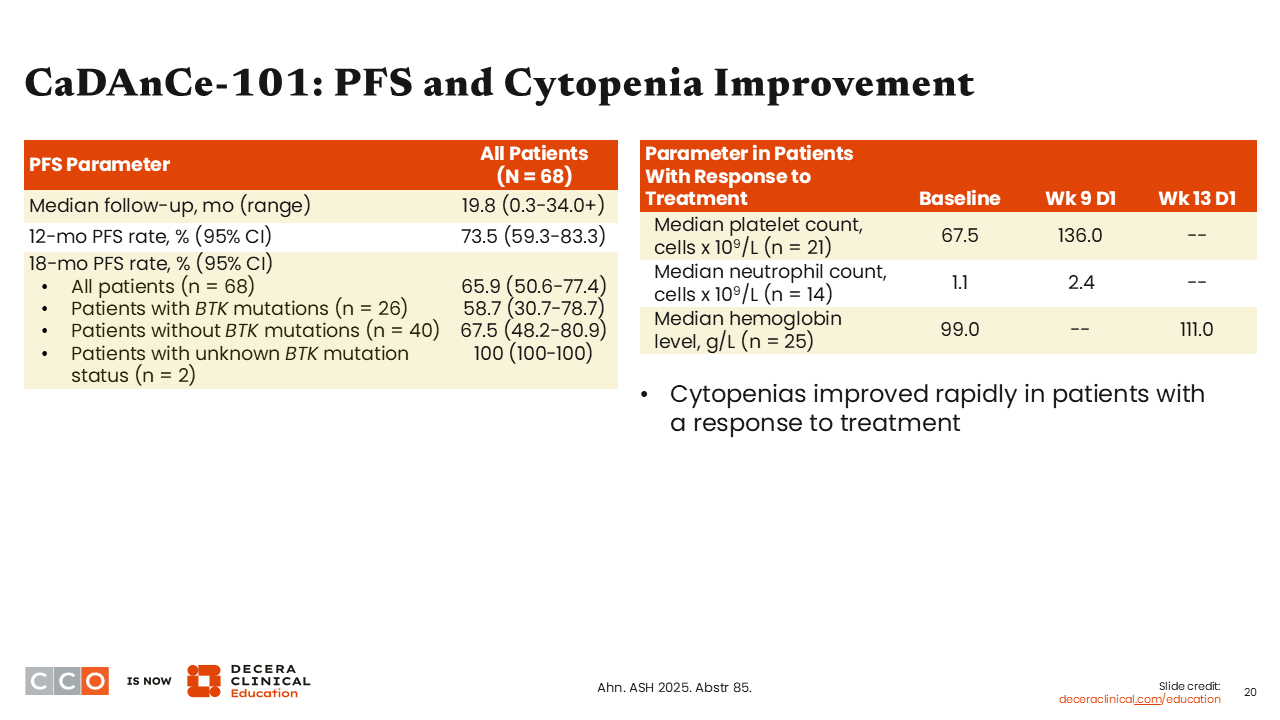
CaDAnCe-101: PFS and Cytopenia Improvement
Brad Kahl, MD:
After a relatively short median follow-up of 19.8 months, the 18-month PFS rate was 65.9%.11 I think that is very respectable durability for this patient population.
Conclusions
Brad Kahl, MD:
This drug, and the BTK degrader class of drugs, are one of the more exciting approaches to treatment for CLL in development right now. This study showed why there is reason to be excited with this agent when you get to therapeutic dose levels. We have ORRs exceeding 85% in patients with heavily pretreated CLL with failure of a covalent BTK inhibitor and BCL2 inhibitor and an ORR of 75% in patients after a prior noncovalent BTK inhibitor. I predict that this class of drugs will be approved in relapsed CLL and probably in other diseases and will become a mainstay in our treatment as the development of these drugs matures.
Jeremy Abramson, MD:
I will join you on that very sturdy limb in suggesting that these drugs have a future. I think what is so impressive is the efficacy of BGB-16673 across high-risk subgroups, whether they had received prior covalent and/or noncovalent BTK inhibitors. Responses were also seen in the majority of patients with resistance mutations to covalent BTK inhibitors.
The noncovalent BTK inhibitors such as pirtobrutinib do not overcome all mechanisms of BTK inhibitor resistance, and they overcome some resistance mutations better than others. As you point out, this mechanism of action is independent of specific mutations, so BTK degradation may be more broadly applicable to our patients progressing on a traditional covalent or noncovalent BTK inhibitor. I think this approach has a bright future.
EPCORE FL-1: Epcoritamab With Rituximab and Lenalidomide (R2) vs R2 for R/R FL
Jeremy Abramson, MD:
EPCORE FL-1 was an important randomized trial in R/R FL that evaluated the addition of epcoritamab to rituximab and lenalidomide (R2).14 I think this is a practice-changing trial, so much so that it led to an FDA approval about a week before the data from this study were presented at the ASH meeting.15
Patients with FL with high tumor burden typically still receive chemoimmunotherapy in the frontline setting. In the second-line setting, the most common regimen used in my practice for quite some time has been R2, a chemotherapy-sparing approach with excellent efficacy.16
Randomized data were presented about a year ago showing that one could add to the R² backbone in relapsed FL, with a positive trial adding tafasitamab to R².17 EPCORE FL-1 assessed another potential way to improve upon R2 in relapsed FL.
In this phase III trial, patients with R/R FL with at least 1 prior line of therapy were randomized 1:1 to receive standard R2 as time-limited therapy for 1 year either alone or with epcoritamab, which was administered through step-up dosing in cycle 1, after which it was given weekly in cycles 2-3 and then once a month through cycle 12, when all treatment was discontinued.14 The trial had dual primary endpoints of ORR and PFS.
The median number of prior therapies for patients in this trial was 1; the majority of patients were treated in the second-line setting. There was a population of high-risk patients; approximately 40% of patients had progression of disease within 2 years of starting frontline therapy (POD24), and approximately a third of patients were refractory to their immediate prior line of treatment.
EPCORE FL-1: Efficacy
Jeremy Abramson, MD:
Looking at the first coprimary endpoint, a dramatic improvement in PFS was noted with the addition of epcoritamab to R2, with a 79% reduction in the risk of progression or death (HR: 0.21; P <.0001). The estimated 16-month PFS rate was 85.5% with epcoritamab plus R2 vs 40.2% with R2 alone.
The PFS benefit observed with the addition of epcoritamab to R2 was seen regardless of high-risk subset, including patients with prior POD24, those who were refractory to their immediate prior line of therapy, and those refractory to both a CD20 antibody and an alkylating agent.
Looking at the second coprimary endpoint, ORR was also improved with epcoritamab plus R2 over R2 alone (95% vs 79%, respectively). More striking was the CR rate, which was 83% with epcoritamab plus R2 compared with 50% with R2 alone. Thus, the trial showed improved efficacy across both primary and secondary endpoints.
OS was not a primary endpoint of this study; however, the 16-month OS was 95.8% with epcoritamab plus R2 compared with 88.8% with R2 alone (HR: 0.38; P = .0039). There was an interesting trend with separation of the OS curves favoring epcoritamab plus R2 over R2 alone. Although the follow-up are short for this analysis, I think these are intriguing data that warrant further follow-up with ongoing evaluation over time.
EPCORE FL-1: Safety
Jeremy Abramson, MD:
Turning to safety, I would say there were no surprises. Cytopenias were the most common toxicity in both arms, largely reflecting the lenalidomide component. More infections and neutropenia were observed when adding epcoritamab to R2.
Cytokine release syndrome (CRS) was also noted with the addition of the BsAb. CRS occurred in approximately a third of patients treated with epcoritamab plus R2, but it was entirely low grade and occurred almost entirely during the very first cycle of step-up dosing. I would say there is some extra toxicity when adding the BsAb, but it is the toxicity that one would expect from adding epcoritamab to R2—a slight accentuation of cytopenias, CRS, and risk of infection.
EPCORE FL-1: Conclusions
Jeremy Abramson, MD:
Overall, these data validated epcoritamab plus R2 as a time-limited 1-year therapy for patients with R/R FL, inducing high rates of CR and durable PFS, at least with approximately 15 months of follow-up. Epcoritamab plus R2, which is a current standard of care for second-line and later FL, was clearly superior to the R2 control arm in terms of PFS and response.
Data from this study led to the FDA approval of epcoritamab in combination with lenalidomide and rituximab for the treatment of patients with R/R FL.15 The additional efficacy is indeed impressive. In my practice, for any patient for whom I am considering R2 for relapsed FL, I would consider the addition of epcoritamab to amplify the efficacy. I would do so knowing that this does come with increased side effects, including cytopenias, infection, and CRS. In counseling our patients, that does need to be balanced against efficacy. However, this is clearly a highly effective second-line and later regimen for FL, allowing us for the very first time to bring a BsAb into the second-line treatment paradigm for FL.
Brad Kahl, MD:
I am excited to have the option of moving a bispecific into second-line treatment for R/R FL. I like the fact that this is time-limited therapy. It is currently a bit of a whirlwind in second-line therapy for FL with the tafasitamab plus R2 approval just 7 months ago.18 I have barely had a chance to use that regimen, and now this one comes along.
I will admit, I spent a few hours in the last week just staring at the data sets from both trials. Although cross-trial comparisons can be problematic, the patient populations in EPCORE FL-1 and inMIND were similar, and the control arm of R2 performed essentially identically in the 2 trials.14,17 Comparing the 2 trials, epcoritamab plus R2 may provide additional efficacy over tafasitamab plus R2, so I think that will become my go‑to in second line for most patients. I might consider a slightly different paradigm for an older, frailer patient where I may not want to take on the AE risks that we mentioned, including infections (33% grade ≥3 with the triplet in this trial). For these patients, I might consider second-line tafasitamab plus R2 and save the bispecific for third-line treatment.
For the majority of my patients, however, epcoritamab plus R2 will become my new preferred second-line treatment option.
Jeremy Abramson, MD:
That is my feeling as well. I think the efficacy is really pronounced, but on a patient-by-patient basis, it is important to recognize that patients did very well on both arms of EPCORE FL-1. The risk–benefit ratio should be considered on a patient-by-patient basis. I tend to like the idea of keeping the targeting of CD19 in reserve to target in later lines of therapy. We are doubling down on the CD20 strategy with this approach, which means that CD19 targeting remains naive for the future.
Surovatamig (AZD0486; CD19 x CD3 BsAb) in R/R FL
Brad Kahl, MD:
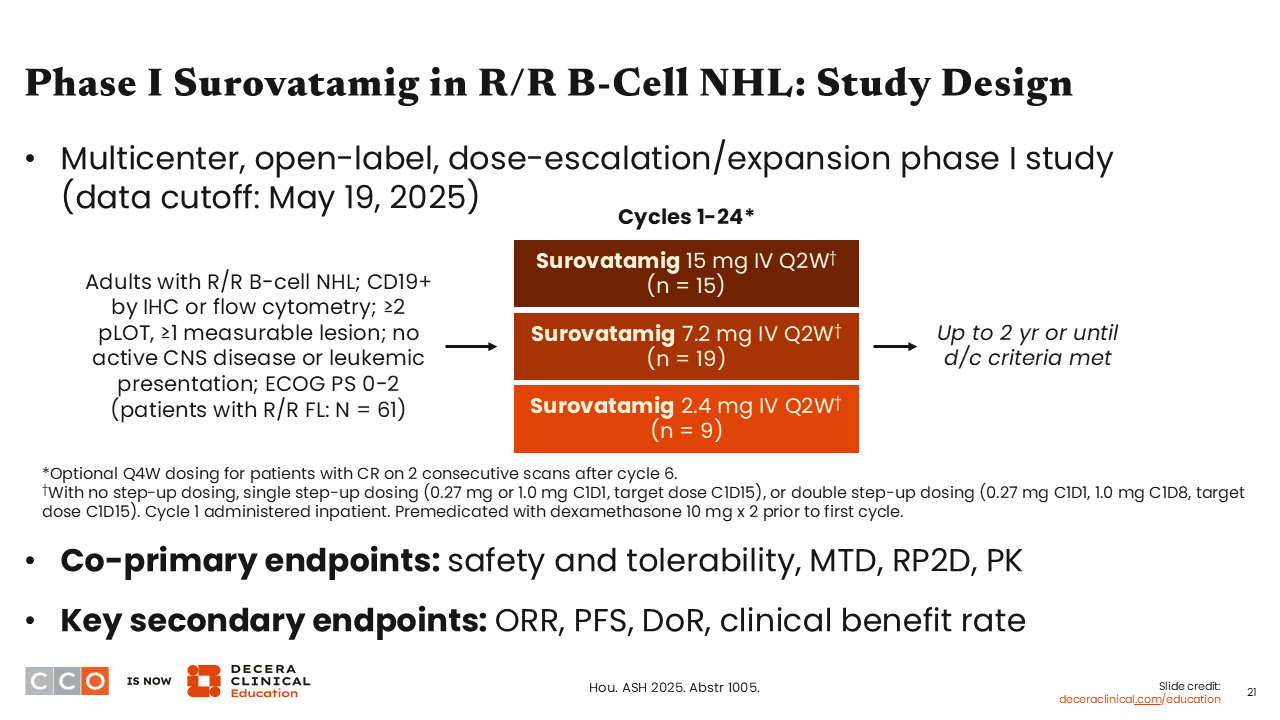
Another interesting presentation from ASH 2025 included data with a novel BsAb T‑cell engager, surovatamig, which targets CD19 and CD3.19
This open-label, dose-escalation/expansion phase I study included adults with CD19-positive R/R FL with at least 2 prior lines of therapy. As with most BsAbs, there was a step-up dosing strategy to get to the target dose. The point of this trial was to identify a target dose. Three different doses were assessed: 2.4 mg, 7.2 mg, and 15 mg, given every 2 weeks for a total of a 2-year duration. The coprimary endpoints were safety and tolerability, maximum tolerated dose, recommended phase 2 dose, and pharmacokinetics.
The trial enrolled 61 patients who were heavily pretreated with a median of 3 prior lines of therapy.
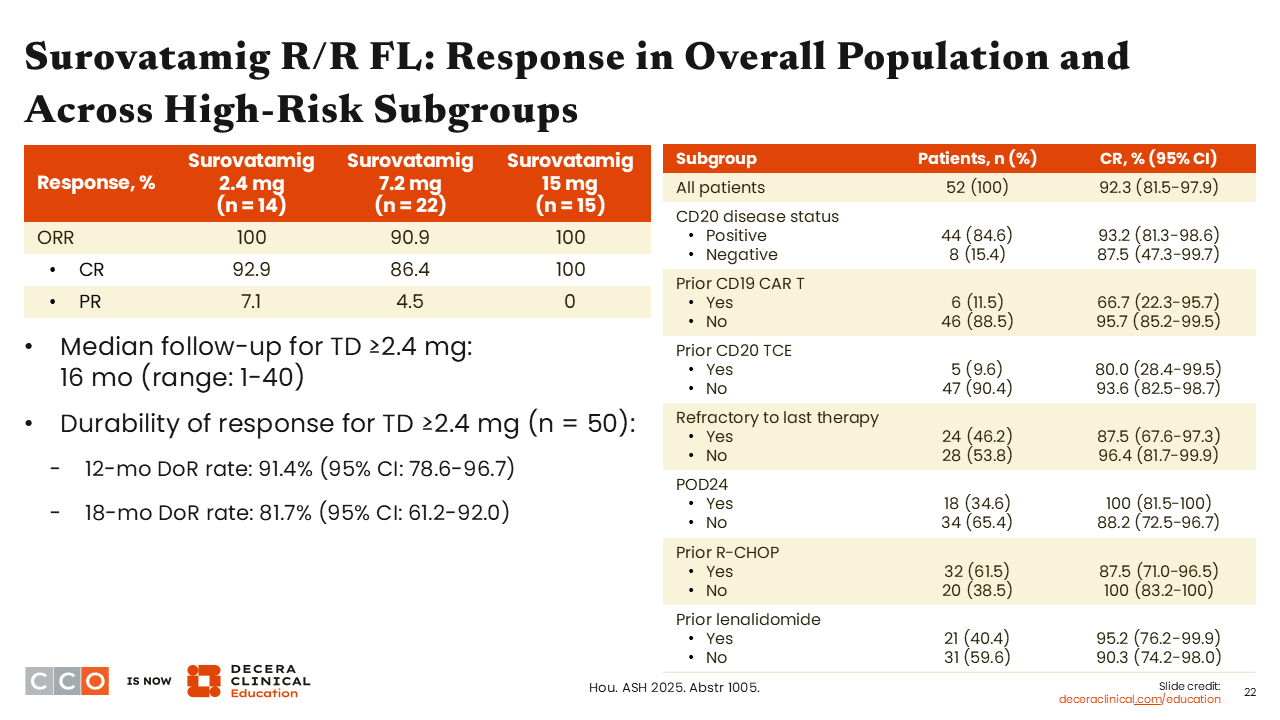
Surovatamig R/R FL: Response
Brad Kahl, MD:
The response rates with surovatamig were impressive. Among the 14 patients in the 2.4-mg cohort, the ORR was 100%, with the majority being CRs (92.9%). At 7.2 mg, the ORR was 90.9%, with 86.4% CRs, and at the 15-mg dose, the ORR was 100%, all CRs. This is not something we see very often in a phase I clinical trial.
Surovatamig R/R FL: PFS
Brad Kahl, MD:
At 12 months, almost 90% of patients were still in remission, and at 18 months, 83.5% were still in remission. Follow-up was short, but this was a very impressive remission duration in this phase I study.
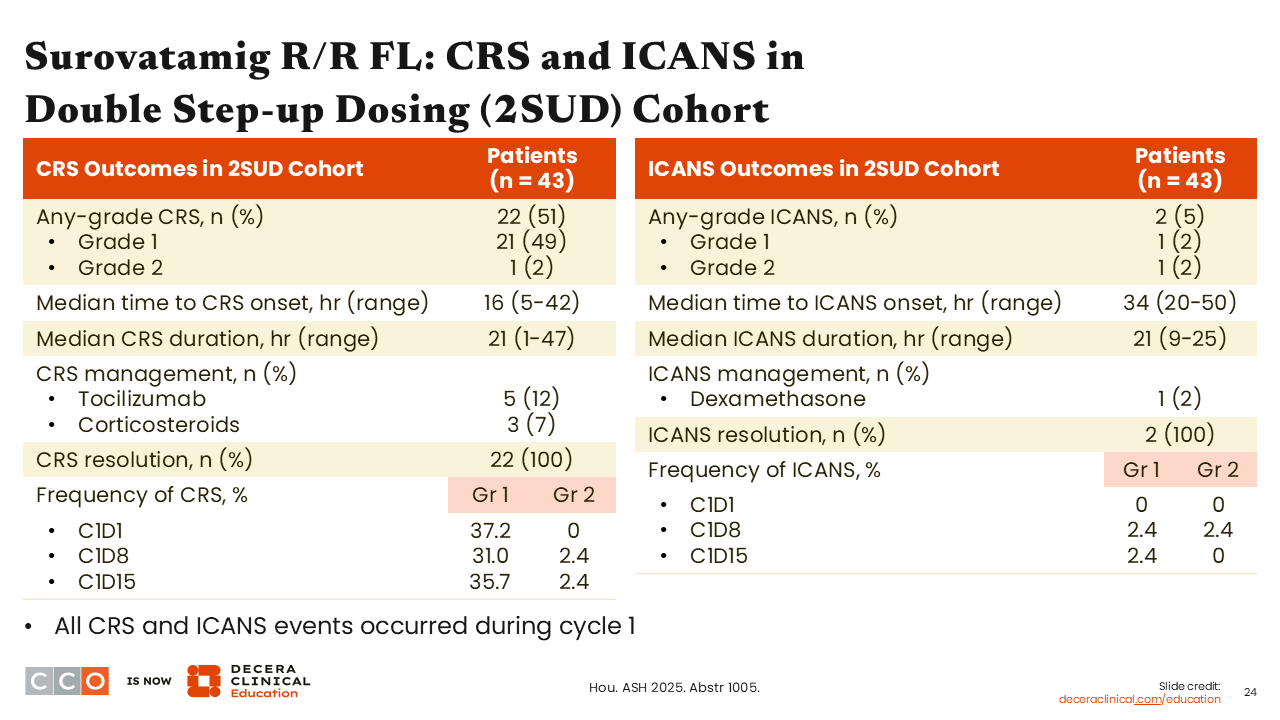
Surovatamig R/R FL: CRS and ICANS in Double Step-up Dosing (2SUD) Cohort
Brad Kahl, MD:
There was some CRS during the ramp-up, but it was typically grade 1. Overall, grade 1 CRS occurred in 49% of patients in the double step-up dosing cohort, and grade 2 CRS occurred in 2%.
There were a few instances of immune effector cell–associated neurotoxicity syndrome, which is something we do not typically see with the anti-CD20 BsAbs, but this was quite low, at 5% incidence (2 of 43 patients).
Surovatamig R/R FL: Conclusions
Brad Kahl, MD:
Surovatamig is a novel BsAb targeting CD19 and CD3 that showed very impressive early response data in R/R FL with an acceptable safety profile. I think we are going to see a lot of development of this drug now, given this very promising phase I signal. This agent is now being assessed in phase II and III clinical trials in patients with FL.
Jeremy Abramson, MD:
This is a very exciting BsAb. The CR rates are really quite phenomenal for a monotherapy BsAb in a heavily pretreated population. Of note, this agent targets a different antigen than all of the other BsAbs currently used to treat B-cell lymphomas, which target CD20.
Patients with FL get an anti-CD20 antibody in every line of therapy. We do see patients with R/R FL in which CD20 expression is downregulated or lost. Having a CD19-targeted BsAb opens those patients to a new class of drugs, along with CAR T-cell therapy in the multiple R/R setting. However, BsAbs are off-the-shelf, easy to administer, and it is hard to beat the CR rates we are seeing, at least in this smaller study. I am excited to see further development going forward. I think this drug could have an exciting future in FL and other B-cell lymphoma histologies.
Sonrotoclax (BGB-11417) in R/R Mantle Cell Lymphoma After Prior BTK Inhibitor Therapy
Jeremy Abramson, MD:
Our next study of interest assessed sonrotoclax, a next-generation BCL2 inhibitor, as monotherapy in patients with R/R mantle cell lymphoma (MCL) who had previously received a BTK inhibitor.20
BTK inhibitors are a standard therapy in MCL. They have long been standard therapy in the R/R setting; more recently, they have entered our upfront treatment armamentarium for both younger and older patients alike, in combination with things like the TRIANGLE regimen, or with bendamustine plus rituximab, per the ECHO trial.21,22
However, treatment of patients relapsing after BTK inhibitors has generally proved challenging. The noncovalent BTK inhibitor pirtobrutinib is approved by the FDA for patients with R/R MCL after at least 2 lines of systemic therapy, including a BTK inhibitor.23 CAR T-cell therapies are now available for R/R MCL with excellent efficacy.24 Of course, CAR T-cell therapy can be a logistically complicated regimen. Moreover, some patients with refractory MCL have fairly aggressive disease after progression on a BTK inhibitor. So, we need novel strategies for patients progressing on covalent BTK inhibition.
Venetoclax does have modest single-agent activity in MCL, and it has been combined with multiple regimens, including the AVO (acalabrutinib, venetoclax, and obinutuzumab) and the BOVen (obinutuzumab, zanubrutinib, and venetoclax) regimens, all of which have shown some activity.25-27
The current study focuses on sonrotoclax, a next-generation BCL2 inhibitor that has higher potency and more selectivity against BCL2 than venetoclax, with less BCL-xL inhibition.28 BCL-xL inhibition leads to some of the toxicities of venetoclax, including cytopenias.28
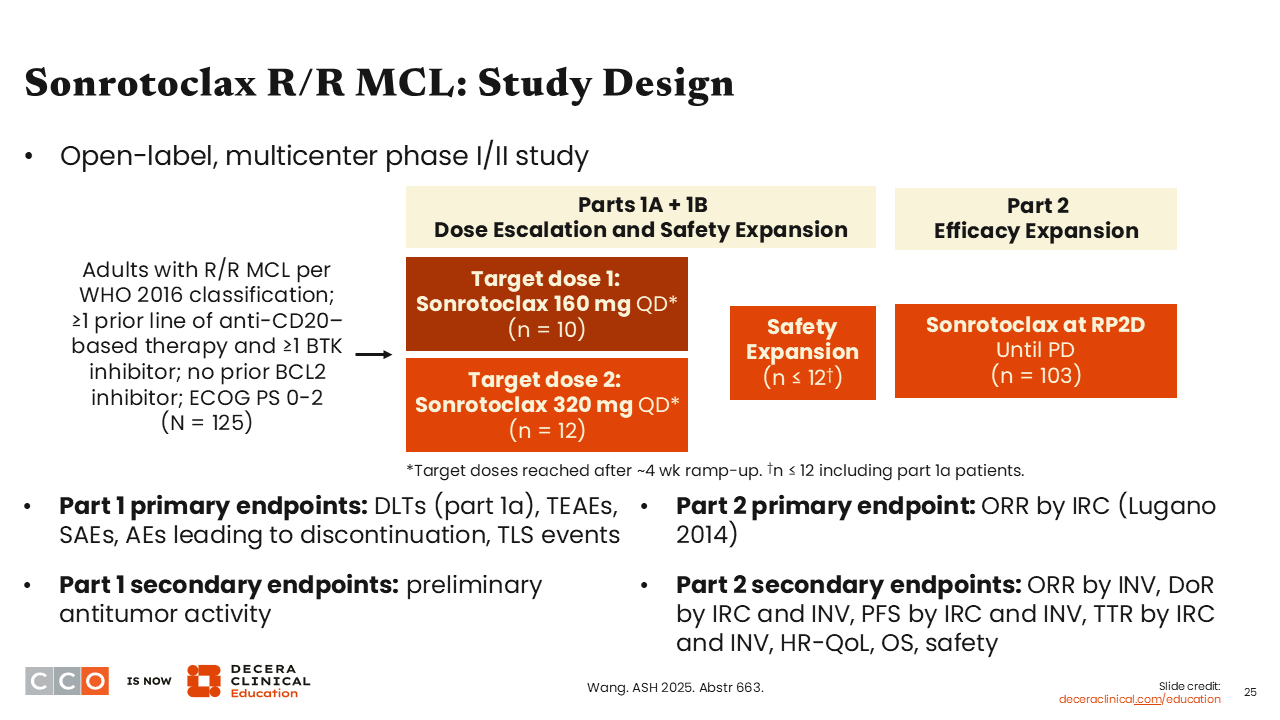
Sonrotoclax was assessed in a phase I/Ib study followed by a phase II dose expansion in patients with R/R MCL with ≥1 prior line of anti-CD20–based therapy and ≥1 BTK inhibitor. Two dose levels of sonrotoclax were studied: 160 mg daily and 320 mg daily.20 Dosing was continued until intolerance or progression.
This was a large study, with 125 patients enrolled. The 320-mg dose was selected in the early dose finding.
Sonrotoclax R/R MCL: Baseline Characteristics
Jeremy Abramson, MD:
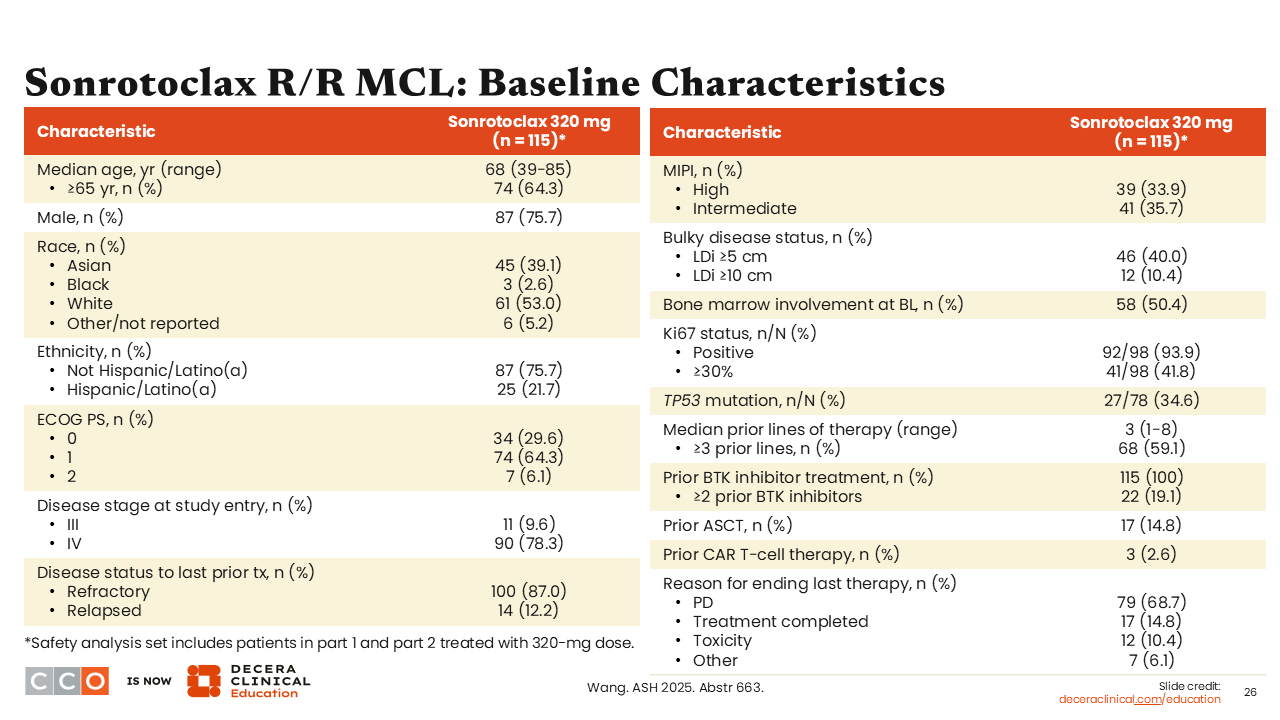
A total of 115 patients were treated with sonrotoclax 320 mg once daily.20 The median age of patients was 68 years. Of importance, 87% of these patients were refractory to their prior line of therapy, and the vast majority of these patients were refractory to BTK inhibitors. All patients had received a prior BTK inhibitor.
Sonrotoclax R/R MCL: Safety
Jeremy Abramson, MD:
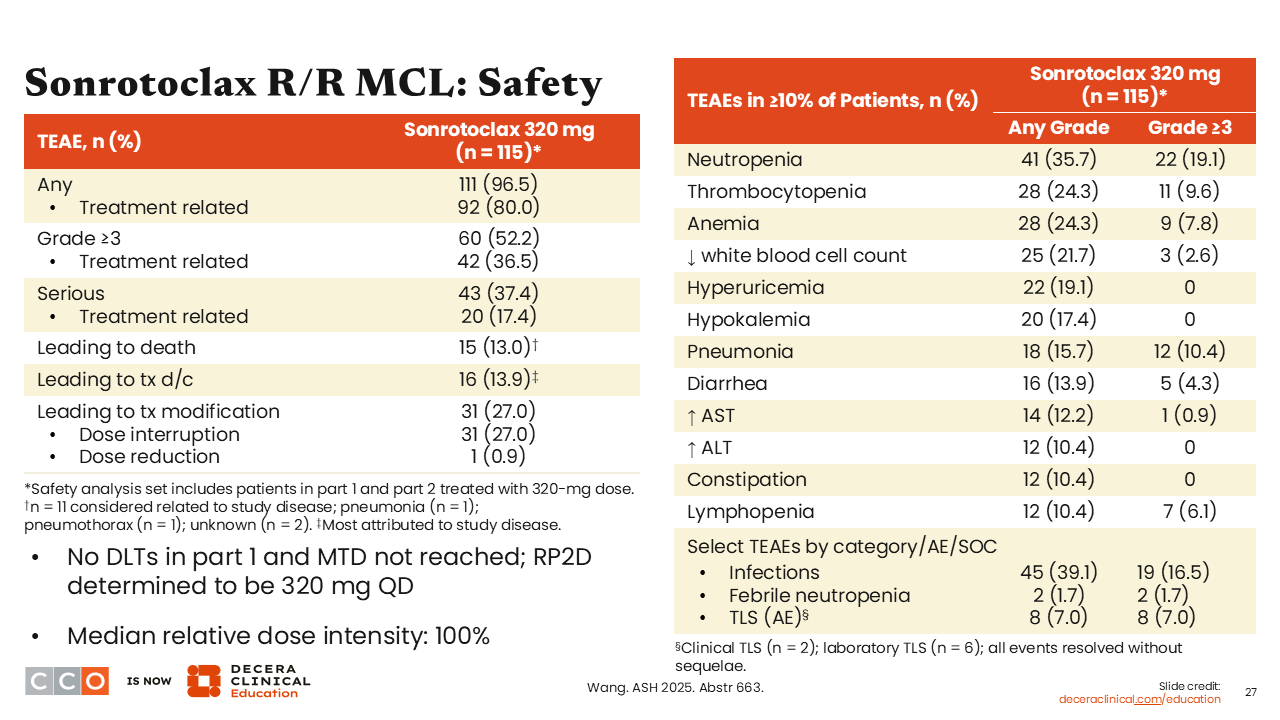
This was a phase I study, so safety is first and foremost. Sonrotoclax was a well-tolerated drug; there were few treatment interruptions or treatment discontinuations due to side effects.
The most common side effects, as with the first-generation BCL2 inhibitor, were cytopenias, with neutropenia, thrombocytopenia, and anemia occurring frequently. Approximately a third of patients (35.7%) developed neutropenia of any grade, and approximately a quarter of patients developed anemia (24.3%) or thrombocytopenia (24.3%).
We do consider tumor lysis syndrome (TLS) as a toxicity of note in patients receiving a BCL2 inhibitor. In this study, there was a low incidence, with 8 patients (7%) developing TLS. This included 2 patients developing clinical TLS and the remaining 6 developing laboratory-only TLS. All events resolved without sequelae and did not require any treatment discontinuation, so this was mild and manageable.
Sonrotoclax R/R MCL: Efficacy
Jeremy Abramson, MD:
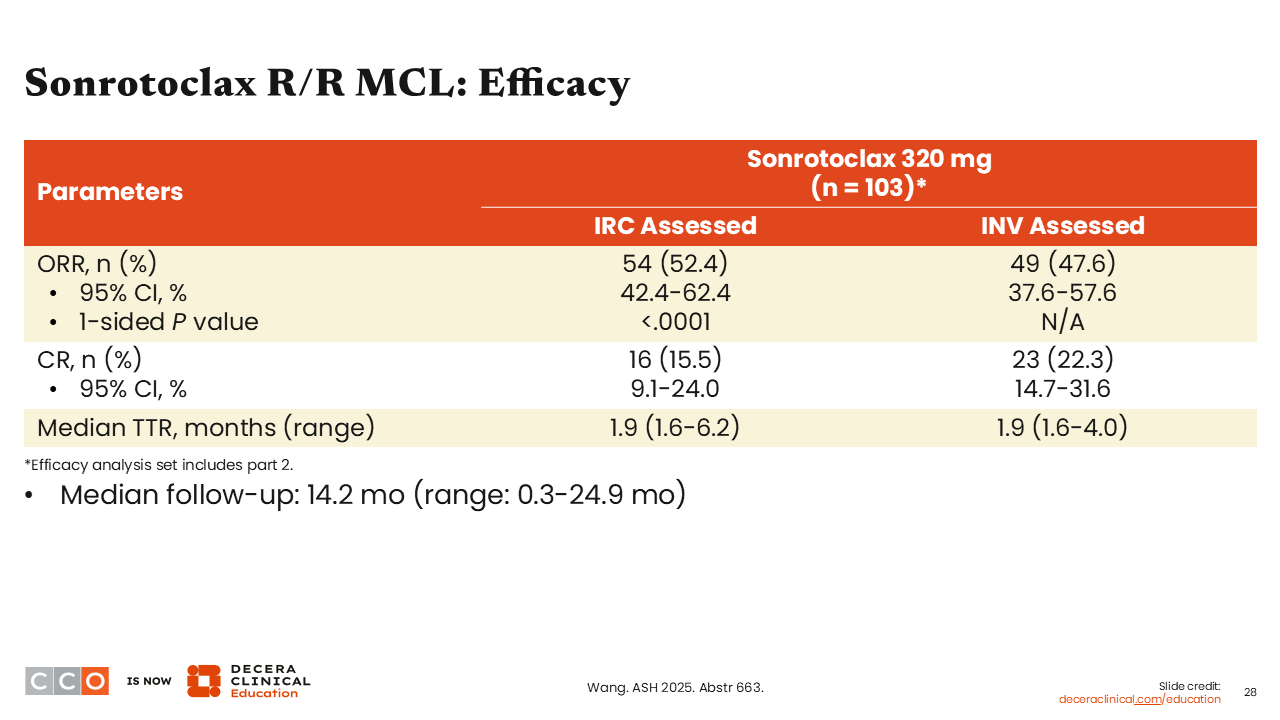
Turning to efficacy, remembering this was a heavily pretreated, uniformly BTK inhibitor-treated population, the independent review committee–assessed ORR was 52%, with 15.5% CRs. The study thus met its predefined primary endpoint.
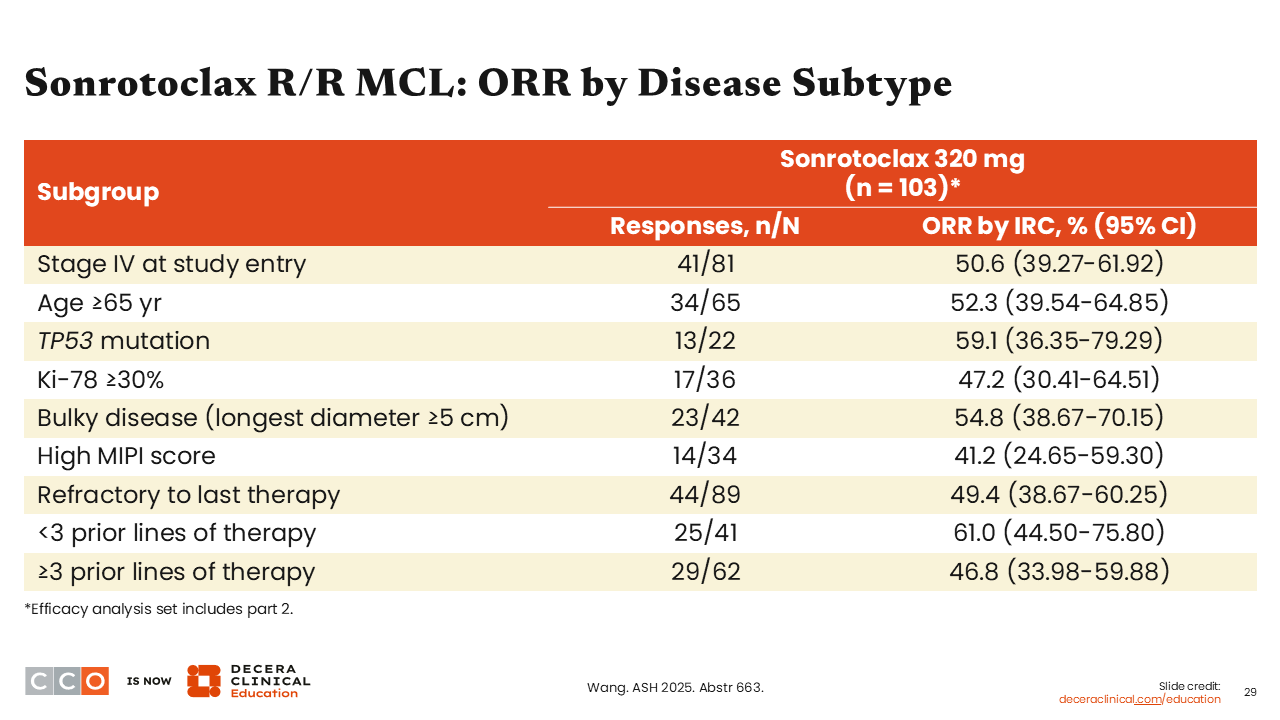
Sonrotoclax R/R MCL: ORR by Disease Subtype
Jeremy Abramson, MD:
Responses were seen across high-risk subsets, including those with a TP53 mutation, high Ki67, ≥3 prior lines of therapy, or refractoriness to their immediate prior line of treatment.
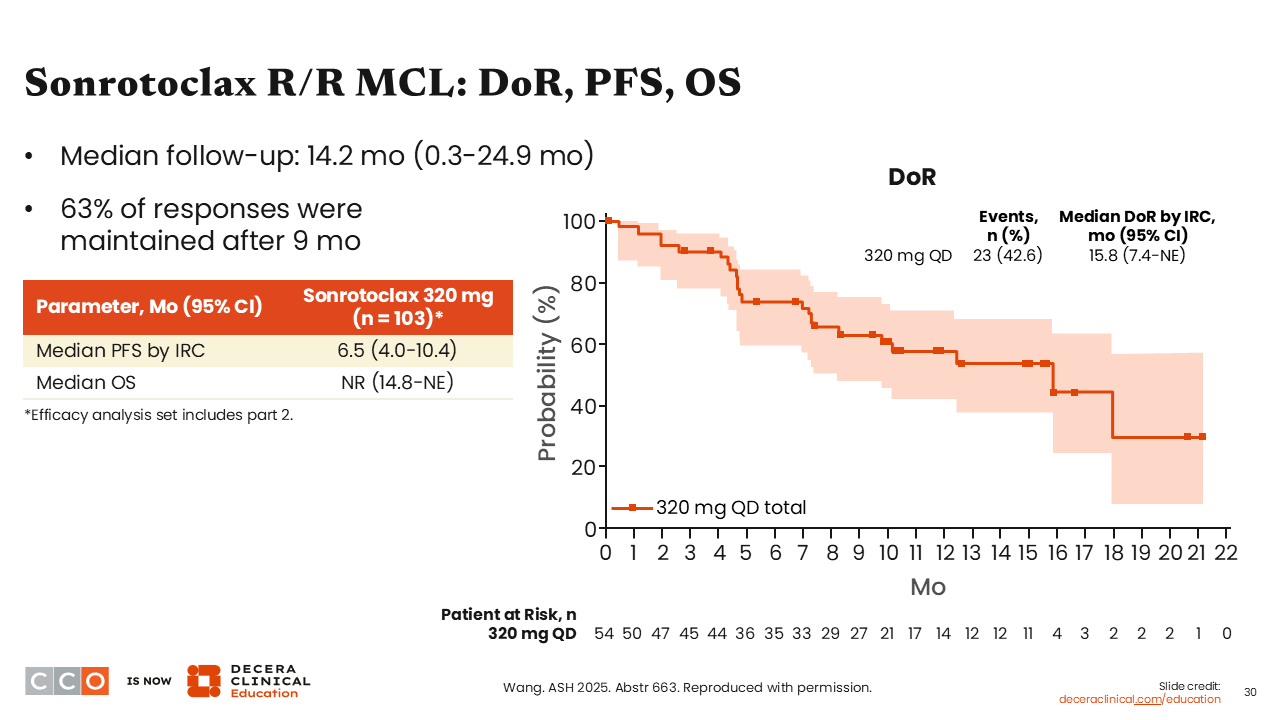
Sonrotoclax R/R MCL: DoR, PFS, OS
Jeremy Abramson, MD:
In this population of patients with MCL refractory to BTK inhibitors, the duration of response was encouraging, with a median duration of response of 15.8 months, and the median PFS for the entire population was 6.5 months.
Conclusions
Jeremy Abramson, MD:
These are very encouraging findings and suggest that sonrotoclax is a very effective drug in this population. I would be particularly eager to see it in combination strategies going forward.
Brad Kahl, MD:
I agree. An important study is ongoing in which patients with relapsed MCL are being randomized to single-agent zanubrutinib or the combination of zanubrutinib and sonrotoclax (NCT0731652). This drug might find its home in a combination strategy, but even as a single agent, it is proving to be a useful drug. As a monotherapy, it could be used as a bridge to CAR T or after CAR T-cell therapy failure. It would be great to have another option for this setting of relapsed MCL where we do not have a really robust toolbox.
Let’s revisit our questions from earlier in the activity.