CE / CME
Conference to Clinic Highlights From the 2025 ASH Annual Meeting: Key Studies in Leukemias
Physician Assistants/Physician Associates: 1.00 AAPA Category 1 CME credit
Pharmacists: 1.00 contact hour (0.1 CEUs)
Physicians: maximum of 1.00 AMA PRA Category 1 Credit™
Nurse Practitioners/Nurses: 1.00 Nursing contact hour
Released: February 27, 2026
Expiration: August 26, 2026
Activity
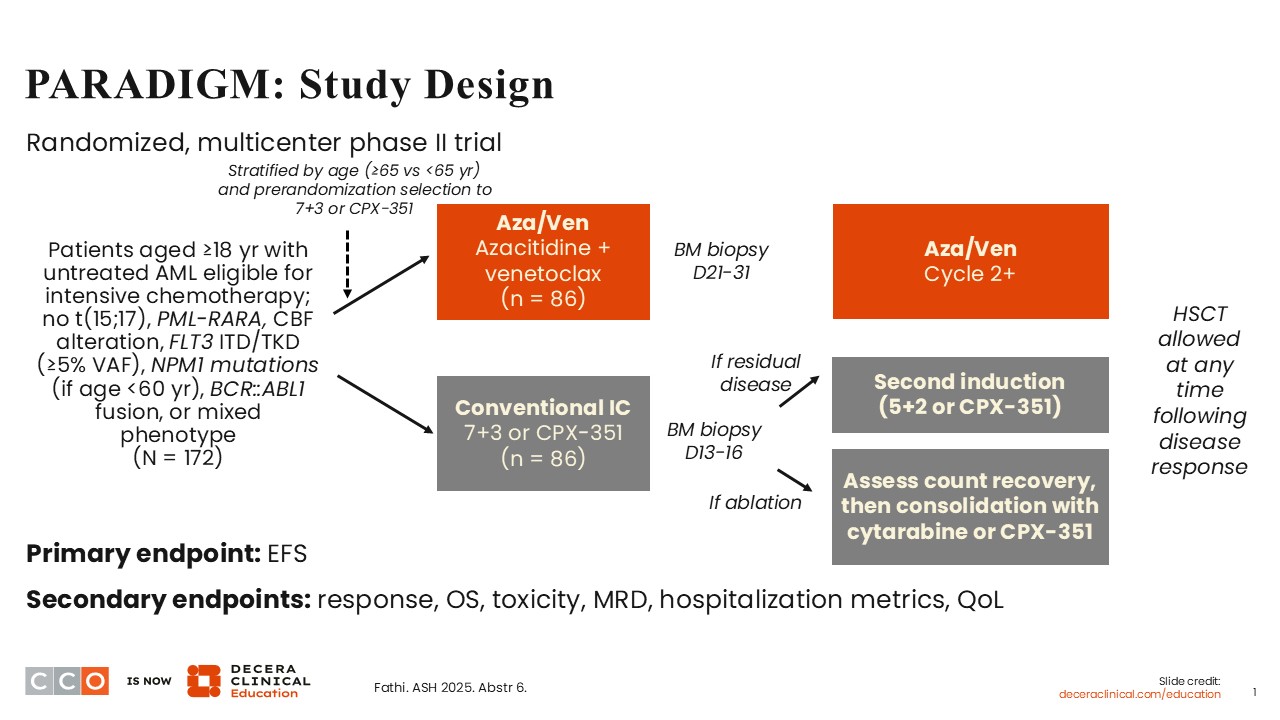
PARADIGM: Study Design
Amir T. Fathi, MD:
For several decades, intensive induction chemotherapy with cytarabine and anthracycline (7+3) has been the standard of care for the management of fit patients with newly diagnosed AML.1 However, substantial toxicity has been associated with this regimen, and treatment outcomes have remained suboptimal. For patients who are not eligible for intensive chemotherapy, the combination of venetoclax, a potent BCL2 inhibitor, with a hypomethylating agent, azacitidine, has become the standard of care based on the overall survival (OS) advantage over azacitidine alone demonstrated in the phase III VIALE-A study.2 In fit patients with AML, multiple retrospective studies have suggested that the combination of a hypomethylating agent, such as azacitidine or decitabine, with venetoclax, may result in superior efficacy and better tolerability when compared with intensive chemotherapy.3,4 Hence, this hypothesis was tested prospectively in the randomized phase II PARADIGM study.5
PARADIGM is a multicenter, randomized phase II trial that investigated azacitidine plus venetoclax vs conventional intensive induction chemotherapy for fit patients 18 years of age or older with previously untreated AML.5 Of importance, several subsets of patients with AML were excluded from participation in this trial, including patients with t(15;17), promyelocytic leukemia (PML)–retinoic acid receptor alpha fusion (otherwise known as the alteration that causes acute PML), patients with core binding factor (CBF) AML, and those with FLT3 ITD/TKD mutations (≥5% variant allele frequency). Patients younger than 60 years of age with disease harboring NPM1 mutations, those with mixed phenotype, and those harboring BCR/ABL1 fusions were also excluded from participation in this study.
Eligible patients were randomly assigned to receive azacitidine plus venetoclax (n = 86) or conventional induction chemotherapy, either 7+3 or CPX-351. Of note, the choice to receive either 7+3 or CPX-351 was established prior to randomization. All patients were stratified by age (≥65 vs <65) prior to randomization.
Following midtreatment bone marrow assessment on Days 13-16, patients on the conventional induction chemotherapy arm with residual disease had the option to receive a second induction treatment with 5+2 or CPX-351 followed by consolidation therapy with cytarabine or CPX-351 if response is achieved. On both treatment arms, patients were allowed to undergo HSCT if response was achieved. This is an important endpoint in terms of assessments in this study.
The primary endpoint was EFS, and the secondary endpoints included response, OS, measurable residual disease (MRD), hospitalization metrics, toxicity and quality of life.
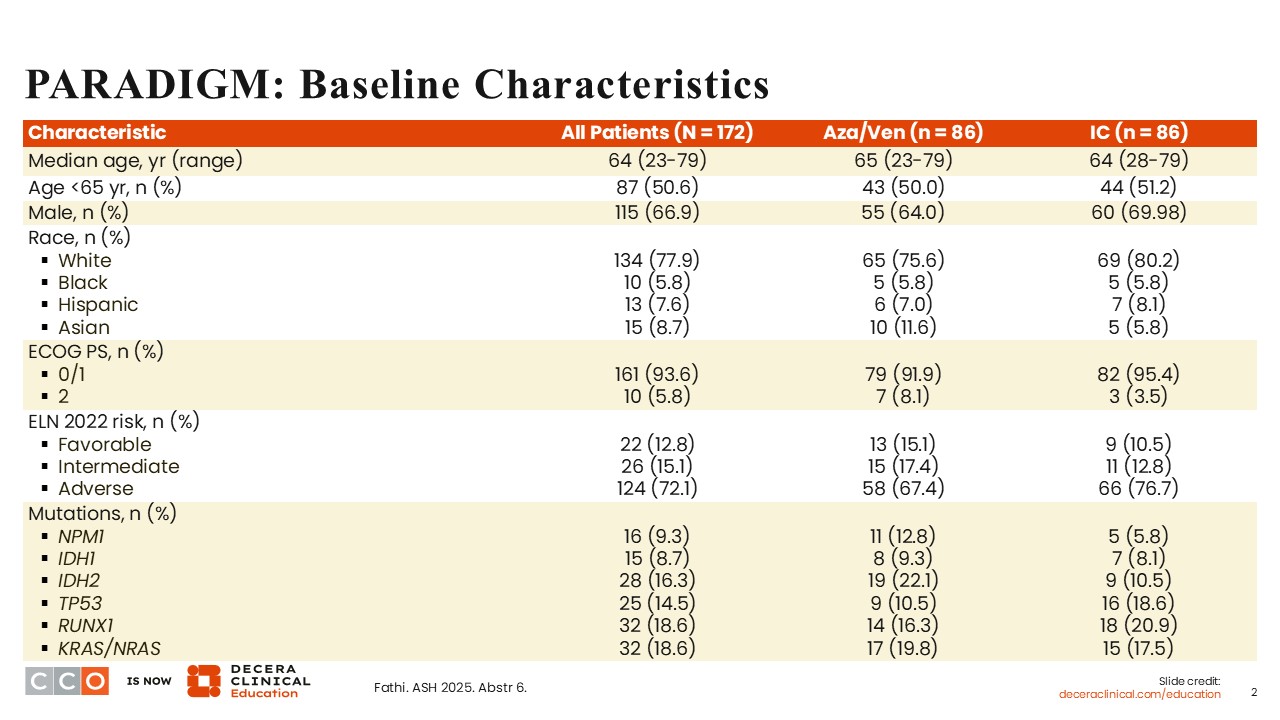
PARADIGM: Baseline Characteristics
Amir T. Fathi, MD:
The baseline characteristics were fairly well balanced between arms. The median age was 65 years (range: 23-79) on the azacitidine/venetoclax arm and 64 years (range: 28-79) on the induction chemotherapy arm. On both arms, the majority of the patients were White (approximately 80%). Approximately 6% were Black, approximately 8% were Hispanic and approximately 9% were Asian. At least 50% of the patients were ≥65 years of age on either arm. More than 90% of the patients had an Eastern Cooperative Oncology Group (ECOG) performance status (PS) of 0 or 1, demonstrating that most of the patients on the study were fit. Approximately 70% of patients had adverse risk by European LeukemiaNet (ELN) 2022 classification, with only one third having favorable or intermediate risk. This can be explained by the exclusion criteria, which precluded the participation of patients with CBF AML, FLT3 ITD/TKD mutations, and those younger than 60 years of age with NPM1 mutations.
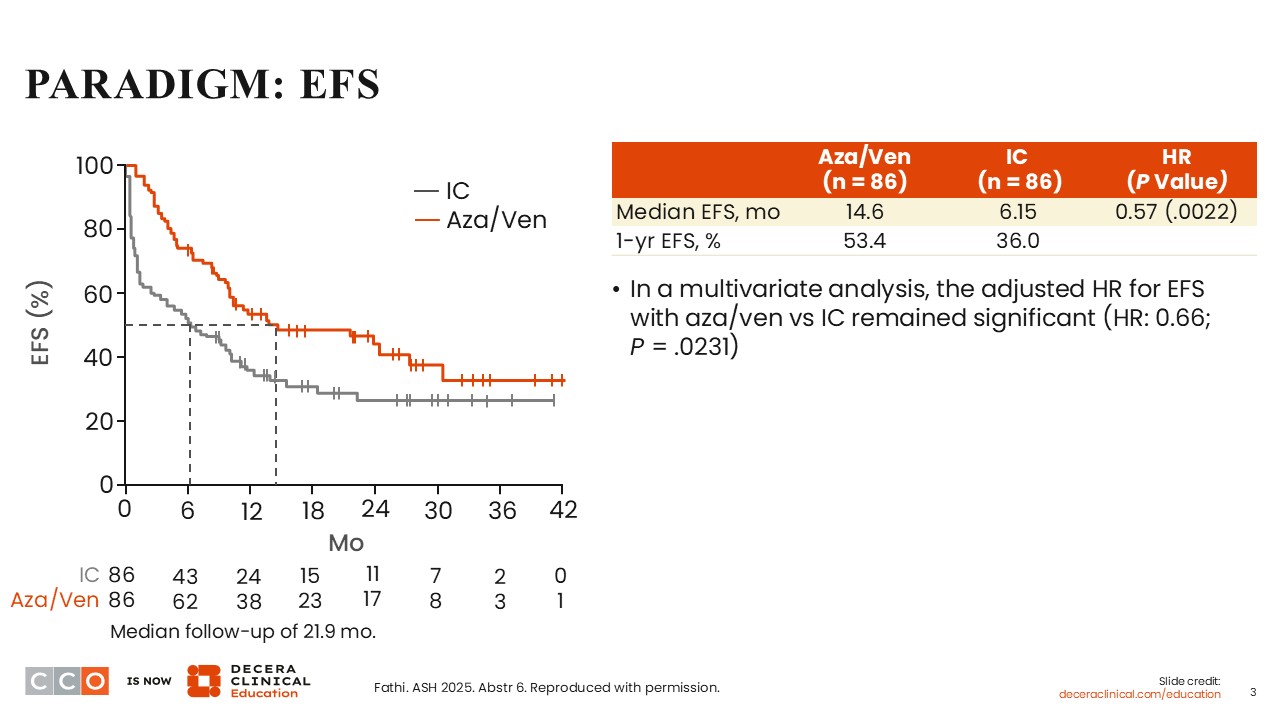
PARADIGM: EFS
Amir T. Fathi, MD:
After a median follow-up of 21.9 months, the primary endpoint of EFS was met with a superior EFS of 14.6 months demonstrated on the azacitidine/venetoclax arm vs 6.15 months on the induction chemotherapy arm (HR: 0.57; P = .0022). The 1-year EFS rate was 53.4% for azacitidine/venetoclax vs 36% for intensive chemotherapy. In a multivariate analysis, the adjusted HR for EFS with azacitidine/venetoclax vs induction chemotherapy was significant despite adjustments for factors such as age, ELN 2022 risk, and certain mutations (HR: 0.66; P = .0231).
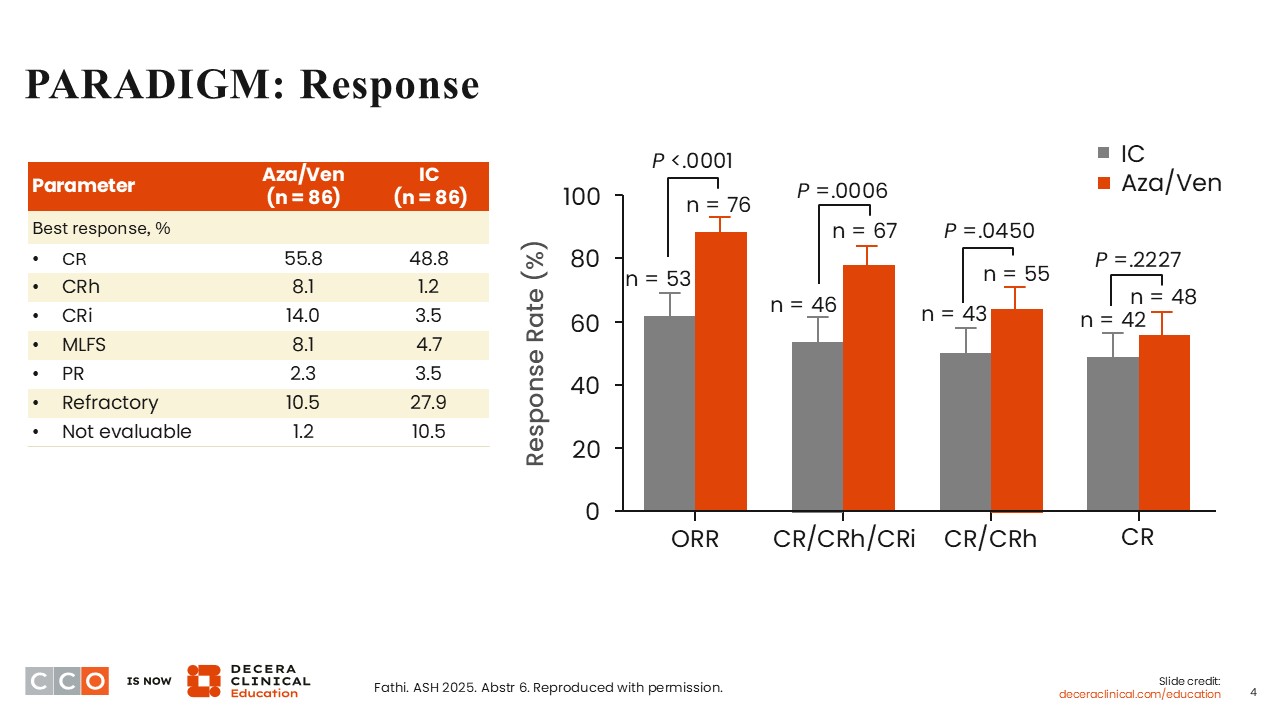
PARADIGM: Response
Amir T. Fathi, MD:
The ORRs were improved in the azacitidine/venetoclax arm vs the induction chemotherapy arm (P <.0001). Although the complete remission (CR) rate was numerically higher with azacitidine/venetoclax (55.8%) compared with induction chemotherapy (48.8%), there was no statistically significant difference between arms (P = .2227). There was a statistically significant improvement on the azacitidine/venetoclax arm (77.9%) vs the induction chemotherapy arm (53.5%), when the rates of CR, CR with partial hematologic recovery (CRh), and CR with incomplete blood count recovery (CRi) were combined (P = .0006).
PARADIGM: Additional Efficacy Outcomes
Amir T. Fathi, MD:
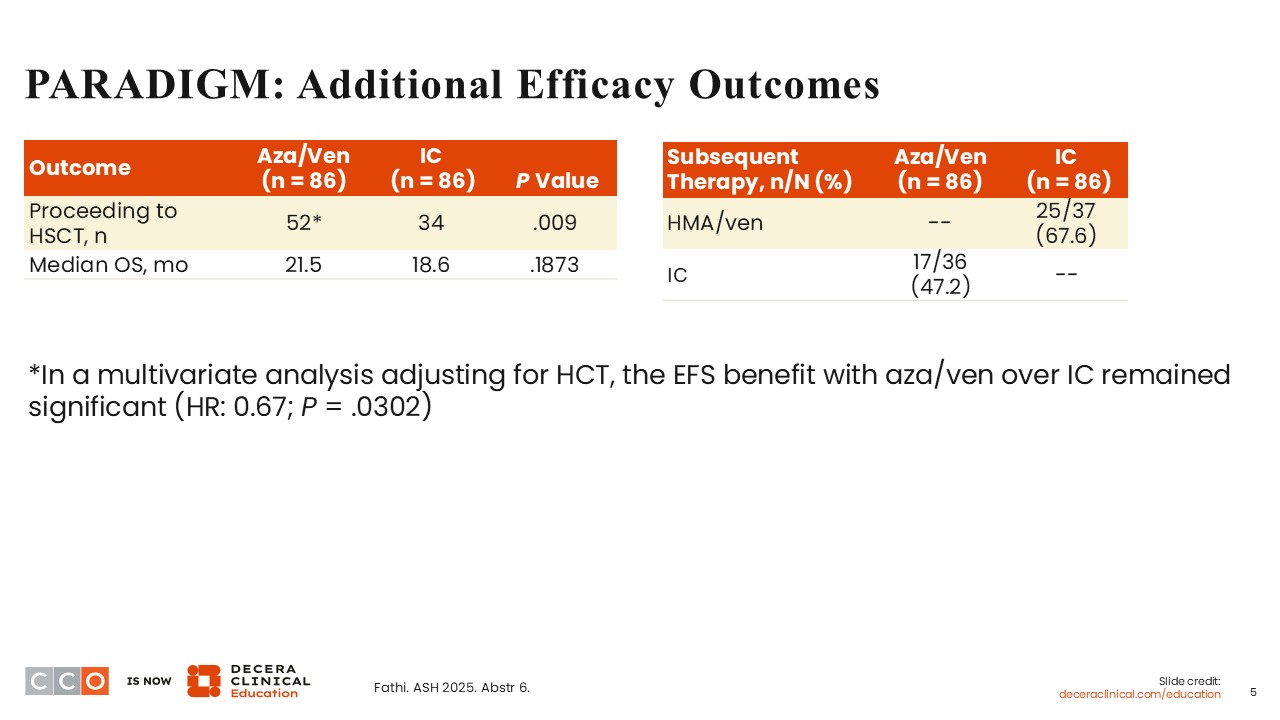
A significantly higher proportion of patients proceeded to HSCT after receiving azacitidine plus venetoclax compared with intensive chemotherapy (n = 52 vs n = 34; P = .009). There was no statistically significant difference in OS between arms (21.5 months vs 18.6 months; P = .1873). Of note, proceeding to HSCT was an important factor that contributed to improvements in outcomes in both arms. In a multivariate analysis after adjustments for HSCT, the EFS improvement with azacitidine/venetoclax over induction chemotherapy remained significant (HR: 0.67; P = .0302).
PARADIGM: PROs and Healthcare Utilization
Amir T. Fathi, MD:
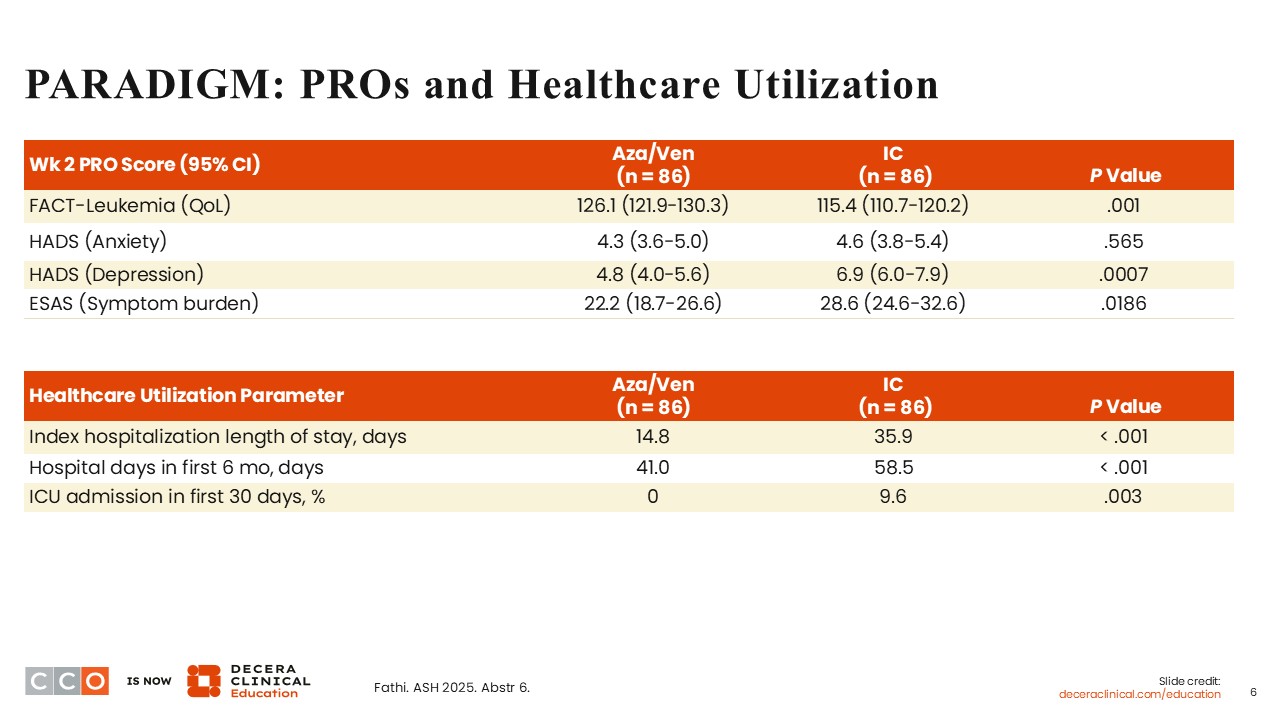
At Week 2, it is not surprising that patients who received azacitidine/venetoclax reported significantly better quality of life (P = .001), depression symptoms (P = .0007), and overall symptom burden (P = .0186) compared with those who received induction chemotherapy. In the first 30 days of treatment, patients who received azacitidine/venetoclax were less likely to require care in the intensive care unit (P = .003), and the index hospitalization duration was greater among patients who received induction chemotherapy (14.8 days vs 35.9 days; P <.001). During the first 6 months of treatment, the length of hospital stay was significantly higher among patients who received induction chemotherapy compared with those who received azacitidine/venetoclax (58.5 days vs 41 days; P <.001).
PARADIGM: Safety
Amir T. Fathi, MD:
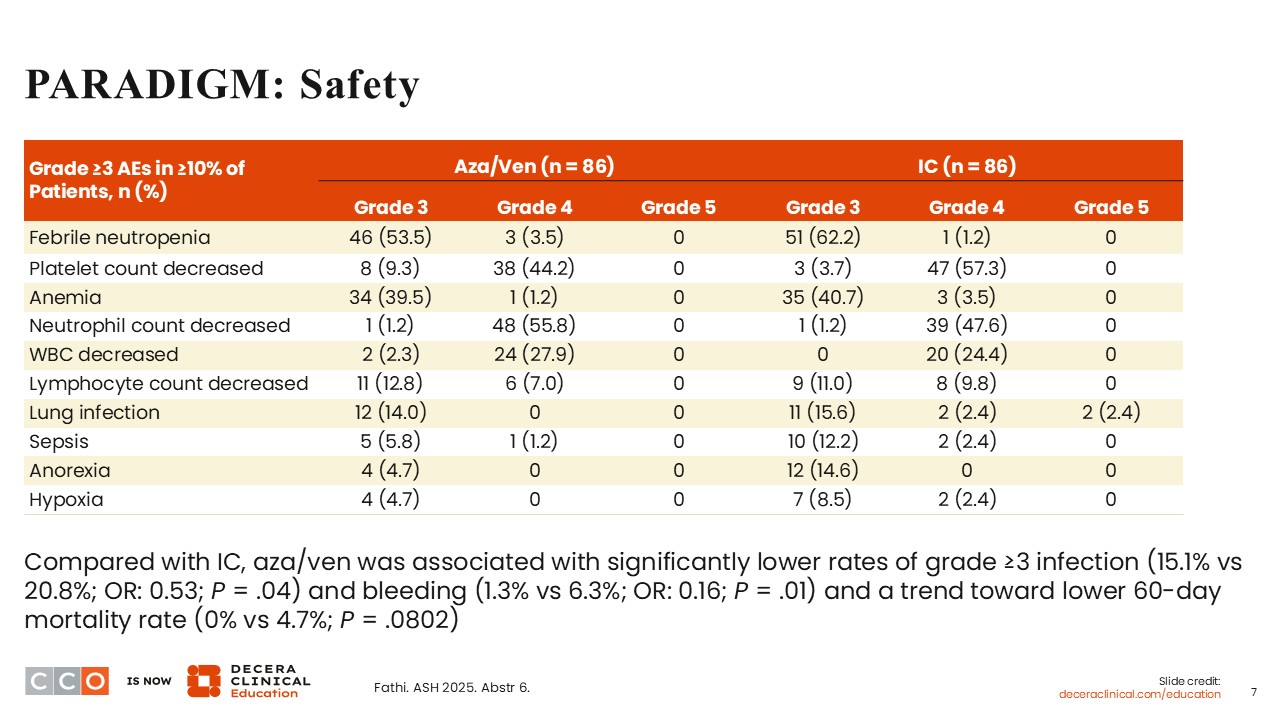
On either of the arms, treatment led to myelosuppression, including grade ≥3 febrile neutropenia, decreased platelet count, anemia, decreased neutrophil count, and decreased white blood cell and lymphocyte counts. The incidence of cytopenias on both arms was fairly similar. However, azacitidine/venetoclax was associated with lower rates of grade ≥3 infection (15.1% vs 20.8%; OR: 0.53; P = .04) and bleeding complications (1.3% vs 6.3%; OR: 0.16; P = .01) compared with induction chemotherapy. On the azacitidine/venetoclax arm, a lower 60-day mortality rate was also noted (0 vs 4.7%; P = .0802).
PARADIGM: Conclusions and Clinical Implications
Amir T. Fathi, MD:
In summary, the randomized phase II PARADIGM trial met its primary endpoint, with an improvement in EFS in favor of azacitidine/venetoclax compared with intensive induction chemotherapy. The combination of azacitidine with venetoclax resulted in deeper and more frequent responses compared with induction chemotherapy and allowed a significantly higher proportion of patients to successfully bridge to allogeneic HSCT.
These data support the use of azacitidine/venetoclax for fit patients with intermediate- or adverse-risk, FLT3 wild-type AML who are eligible to receive intensive chemotherapy with a plan to undergo HSCT. PARADIGM demonstrated that there is the potential for these patients to receive bridging therapy with a nonintensive regimen before transplant without the need to receive intensive chemotherapy and without experiencing the challenges and toxicities associated with intensive chemotherapy.
Courtney D. Dinardo, MD:
The PARADIGM trial is really important because it is the first to definitively show that azacitidine/venetoclax is appropriate for younger and fit patients who can tolerate intensive induction chemotherapy. This has been suspected for several years, and the PARADIGM trial has proven the suspicions to be true. A very important caution and warning, however, is that the use of azacitidine/venetoclax in this setting is not for every patient with AML because several subsets of patients, such as those with CBF AML and those with FLT3 mutations, were excluded from participating in this study. PARADIGM has shown that many younger and fit patients with AML and ELN 2022 adverse risk and complex cytogenetics, who are eligible to receive intensive induction chemotherapy can potentially fare better with azacitidine/venetoclax and proceed to undergo the needed HSCT.
Another really important and interesting finding is that undergoing HSCT is an essential part of the PARADIGM treatment pathway. It is not merely receiving azacitidine/venetoclax alone, but azacitidine/venetoclax followed by HSCT. In essence, the bridging therapy is to get patients to achieve a remission without increased toxicity and then be able to undergo HSCT.
Amir T. Fathi, MD:
I completely agree. It is particularly important to note that patients with FLT3-mutated AML, those younger than 60 years of age with NPM1 mutations, and those with CBF AML were not included in the study. Based on these exclusion criteria in the PARADIGM trial, these subgroups of patients should not yet receive bridging therapy with azacitidine/venetoclax. It is likely that in the future, a nonintensive hypomethylating agent–based regimen may become appropriate for patients in these categories. For instance, different combinations including menin inhibitors such as revumenib and ziftomenib are currently being investigated in AML. I hope that the results from the PARADIGM trial will lead to additional studies that will help to move the field forward, particularly among subgroups of patients excluded from participation in the PARADIGM trial.
SAVE (Newly Diagnosed AML Cohort): Study Design
Courtney D. Dinardo, MD, MSCE:
The interaction between menin and KMT2A is a key dependency in acute leukemias resulting from KMT2A rearrangements, NPM1 mutations, or NUP98 rearrangements. The inhibition of this interaction is known to promote leukemic cell differentiation and apoptosis.6-8 Of note, NUP98 fusions are associated with upregulated HOXA9 and susceptible to menin inhibition.8 Therefore, the SAVE trial investigated a nonintensive combination including a hypomethylating agent, venetoclax, and a menin inhibitor for patients with AML harboring NPM1 mutations, KMT2A rearrangements, or NUP98 rearrangements who are not eligible to receive intensive chemotherapy.9
The phase I/II trial investigated the efficacy and safety of the all oral combination of revumenib with decitabine/cedazuridine and venetoclax for patients 12 years of age or older with newly diagnosed or relapsed/refractory AML or myeloid mixed phenotype acute leukemia (MPAL) harboring NPM1 mutations, KMT2A rearrangement, or NUP98 rearrangements.9 To be eligible for participation, patients with newly diagnosed disease had to be ineligible to receive high-intensity chemotherapy. At ASH 2025, results from the cohort of patients with newly diagnosed AML were presented. It is important to note the uniqueness of the patient cohort in that it included children and young adolescents. Revumenib was dosed at 270 mg when administered without a strong CYP3A inhibitor, and at 160 mg when administered with a strong CYP3A inhibitor. All patients had to undergo bone marrow assessment on Day 14 of cycle 1, in addition to the end of cycle assessment. If bone marrow blasts were <5%, revumenib treatment was held after Day 21. This design allowed for count recovery and tolerability during that first cycle of treatment. Another interesting design of the study is that patients who proceeded to receive stem cell transplant (SCT) received maintenance revumenib for 1 year post SCT.
The primary endpoints in the phase I portion of the study were safety, maximum tolerated dose, and determination of the recommended phase II dose. For the phase II portion of the study, the primary endpoint was efficacy. Secondary endpoints included OS, recurrence-free-survival, and MRD.
SAVE (Newly Diagnosed AML Cohort): Baseline Characteristics
Courtney D. Dinardo, MD:
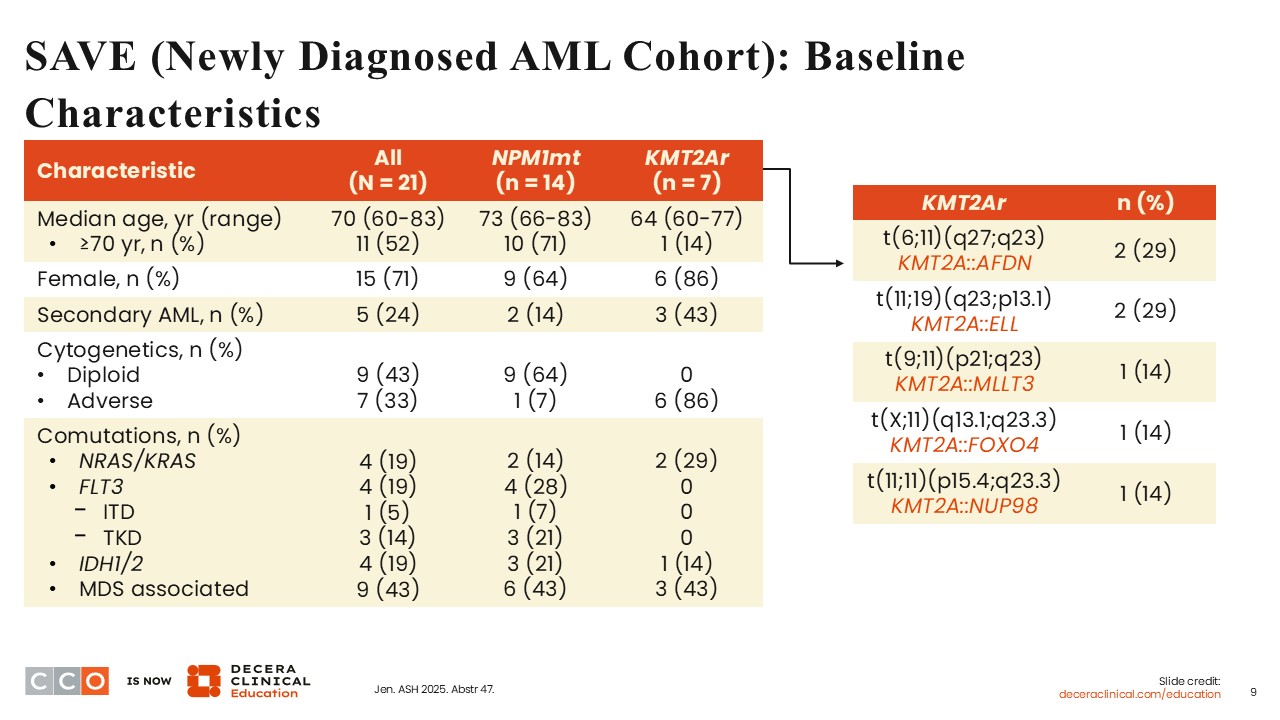
At ASH 2025, data from 21 patients with newly diagnosed AML were presented. Out of the 21 patients, 14 had NPM1-mutant AML, and 7 had KMT2A-rearranged AML harboring 5 different KMT2A rearrangements. The median age was 70 years (range: 60-83), demonstrating an older, chemotherapy-ineligible population of patients. In the cohort of 14 patients with NPM1-mutant disease, comutations with NRAS/KRAS, FLT3, or IDH1/2 were reported. Of interest, 4 of the patients with NPM1-mutant AML also had FLT3 mutations, including 3 patients harboring FLT3 TKD mutations and 1 harboring FLT3 ITD mutations.
In the cohort of 7 patients with KMT2A-rearranged AML, 2 patients harbored comutations with NRAS/KRAS, and 1 patient harbored comutations with IDH1/2.
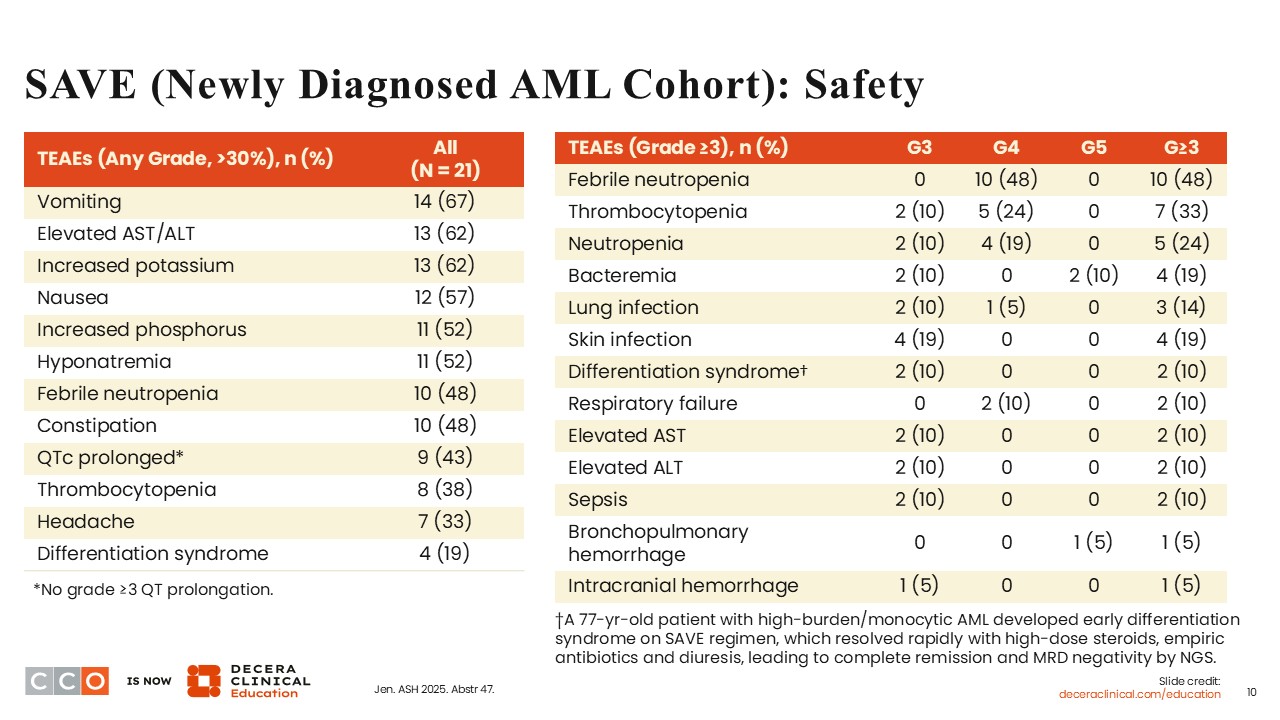
SAVE (Newly Diagnosed AML Cohort): Safety
Courtney D. Dinardo, MD:
It is already recognized that the use of azacitidine plus venetoclax results in a 30% to 40% rate of cytopenias and cytopenia-related toxicities. As expected, the SAVE trial showed that 48% of the patients experienced febrile neutropenia (grade 4: 48%) and 38% experienced thrombocytopenia (grade ≥3: 33%). These safety data are at par with what is expected with the use of a hypomethylating agent in combination with venetoclax. Also, 43% of patients experienced QTc prolongation, all of which were grade 1/2. Differentiation syndrome, a known adverse event (AE) of menin inhibitors, was also reported in 19% of patients (grade ≥3: 10%).
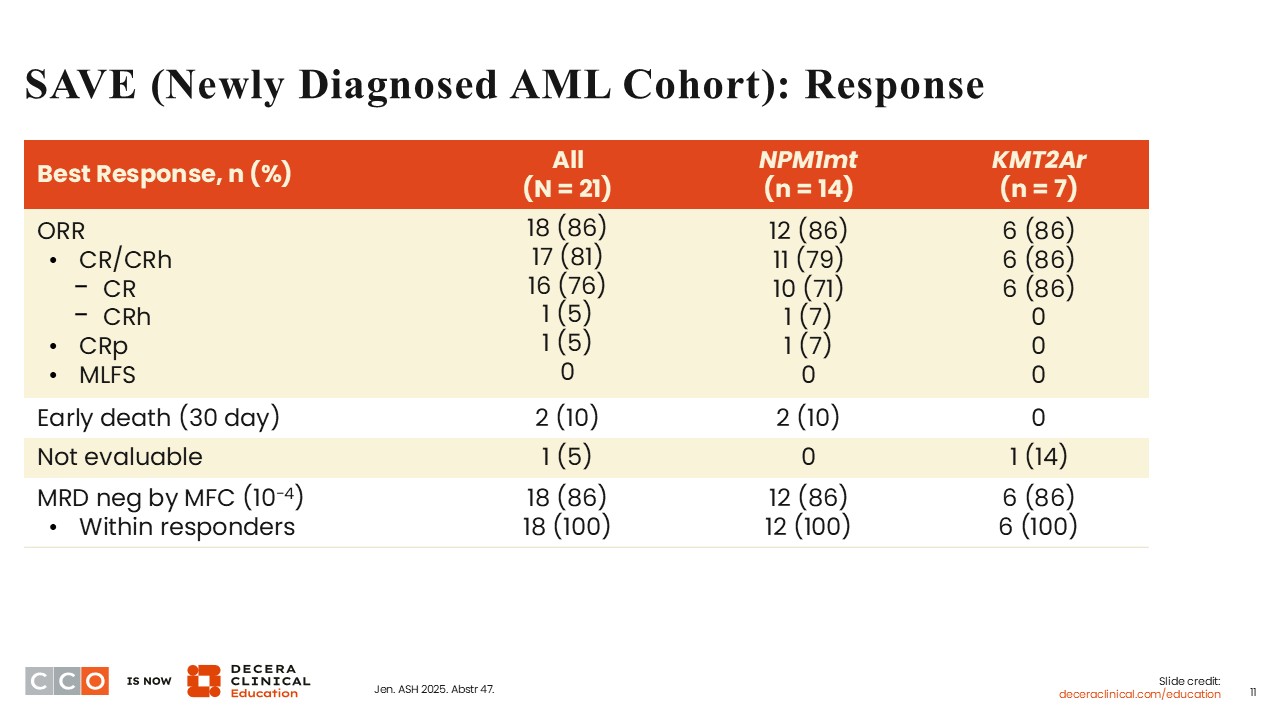
SAVE (Newly Diagnosed AML Cohort): Response
Courtney D. Dinardo, MD:
Even though a sample size of 21 patients is small, an ORR of 86% demonstrated with the all-oral regimen studied in the SAVE trial was impressive. The CR/CRh rate was 81% in the overall population. Among patients with KMT2A-rearranged AML, a particularly adverse risk group of patients, the ORR was 86% with a CR/CRh rate of 86%. Also, the MRD negativity rate by flow cytometry (10-4) was 86%.
These response rates are encouraging and deeper than what is normally seen in this patient population with the use of a hypomethylating agent and venetoclax alone without the addition of a third agent. Although the SAVE trial is not a randomized controlled trial and the sample size is small, these results are still impressive and encouraging.
There were 2 deaths within 30 days of treatment, and both occurred in patients with NPM1-mutant AML, indicating that even a nonintensive regimen of a hypomethylating agent and venetoclax plus revumenib can have early toxicity in older patients who mostly have preexisting comorbidities. This underscores the need for careful monitoring of patients when they are receiving this regimen.
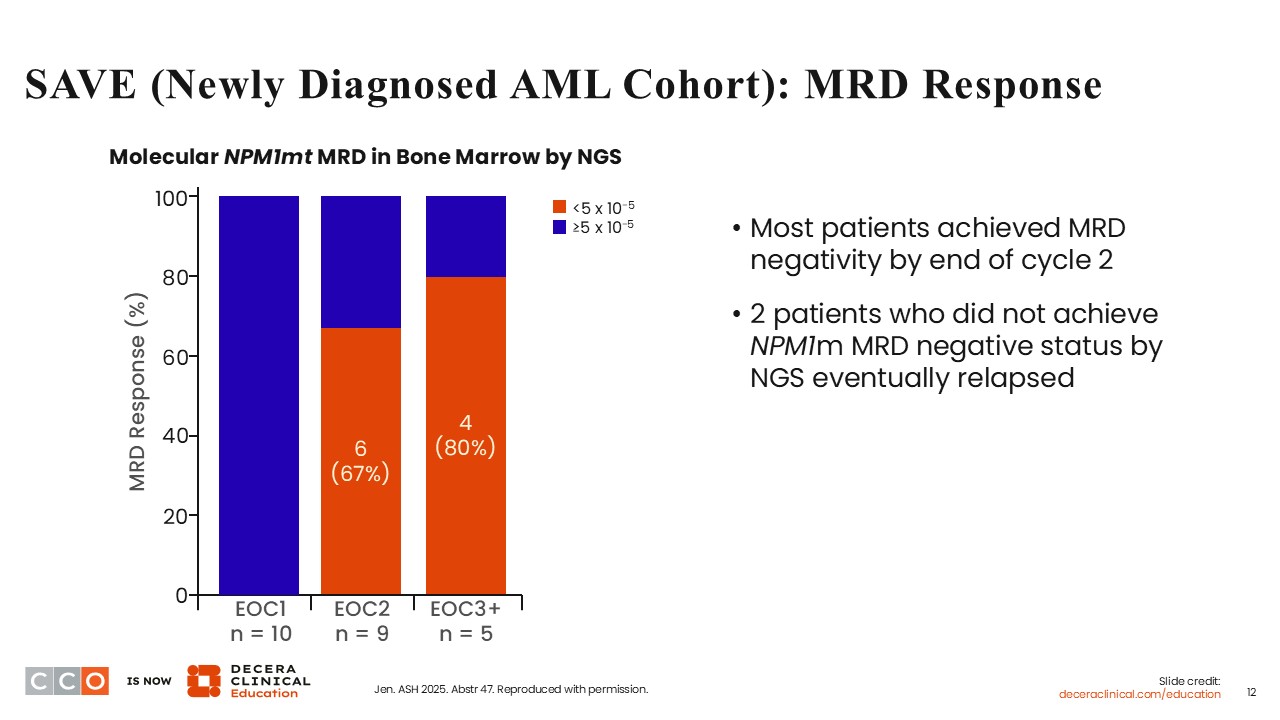
SAVE (Newly Diagnosed AML Cohort): MRD Response
Courtney D. Dinardo, MD:
In addition to MRD determination by flow cytometry, MRD was also determined by next-generation sequencing (NGS), a more sensitive tool. Among patients with NPM1-mutant AML, 6 out of 9 achieved MRD <5 x 10-5 at the end of cycle 2, and 4 out of 5 achieved MRD <5 x 10-5 at the end of cycle 3. Two patients who did not achieve MRD negativity by the end of cycle 2 subsequently experienced disease progression. Overall, it appears that the determination of MRD status by NGS is an appropriate tool to determine the durability of response achieved with the SAVE regimen.
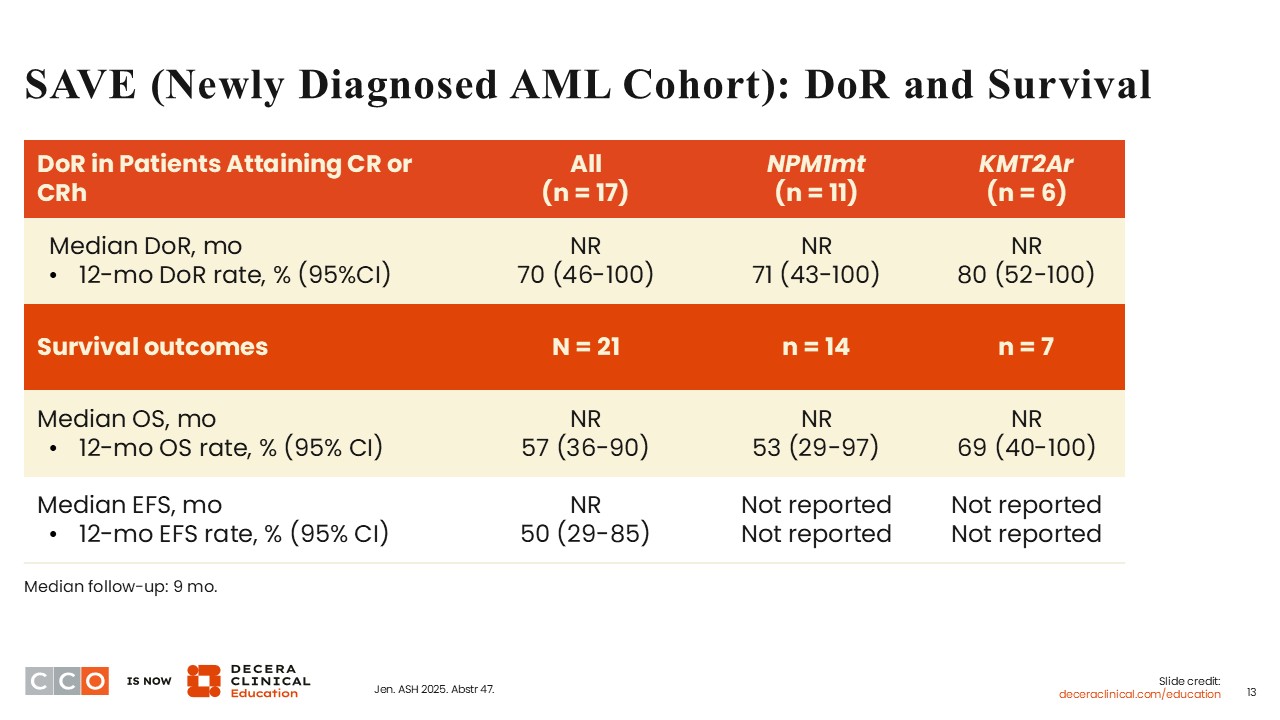
SAVE (Newly Diagnosed AML Cohort): DoR and Survival
Courtney D. Dinardo, MD:
After a relatively short median follow-up of 9 months, the median duration of response (DoR), OS, and EFS had not been reached. However, in the evaluable population of patients, the rates of DoR, OS, and EFS at 1 year were 70%, 57%, and 50%, respectively.
SAVE (Newly Diagnosed AML Cohort): Patient Outcomes and Detection of MEN1 Mutations
Courtney D. Dinardo, MD:
One third of the patients achieved a CR and proceeded to undergo SCT, and as of the time of data cutoff, none of these patients had experienced relapse. This is encouraging. One of the patients who received treatment on the SAVE trial transitioned into hospice care. In total, 4 deaths occurred, with 2 occurring in the induction phase and 2 occurring in the continuation phase. In total, 3 patients experienced disease relapse, 1 of whom had an emergent MEN1 M327V mutation.
In patients with AML receiving a menin inhibitor, acquisition of MEN1 mutations is a primary mechanism of resistance leading to relapse. After a short median follow-up of 9 months, the MEN1 M327V mutation was identified in 1 patient with AML who initially achieved a CR and MRD negativity by flow cytometry but unfortunately experienced relapse after cycle 3.
That only 1 patient acquired the MEN1 mutation in the SAVE trial after a median follow-up of 9 months suggests that the all-oral combination of revumenib, decitabine/cedazuridine, and venetoclax has a promising potential to prevent the early development of on-target resistance mechanisms.
SAVE: Conclusions and Clinical Implications
Courtney D. Dinardo, MD:
The SAVE regimen demonstrated high response rates and impressive MRD negativity. However, these results need to be confirmed in larger patient cohorts with longer follow-up. The detection of just 1 on-target acquired resistance mutation (MEN1 M327V) in only 1 patient with relapsed disease is encouraging. Of note, differentiation syndrome associated with the use of menin inhibitors should be identified early and promptly treated with corticosteroids. Myelosuppression continues to remain a concern with the use of regimens containing a hypomethylating agent and venetoclax.
Amir T. Fathi, MD:
These promising results are impressive and provide hope that the treatment landscape for patients with AML will gradually move away from intensive chemotherapy to nonintensive combinations such as the all-oral SAVE regimen. Of note, the SAVE trial and the PARADIGM trial have shown that these nonintensive regimens are feasible and effective. However, in older patients, there is a concern about a higher incidence of bone marrow suppression and early deaths. We still need to find a way to better align the approach to treatment when we get into triplets so that older patients can better tolerate the regimen and experience benefit from it. To address this concern, there are already efforts underway to reduce the duration of venetoclax when used in a triplet regimen.
As an all-oral regimen, it is potentially convenient for patients. Even though it was associated with 2 early deaths, with longer-term follow-up, the results can be confirmed and the treatment regimen optimized.
Courtney D. Dinardo, MD:
The use of an all-oral triplet regimen including a targeted therapy with a hypomethylating agent and venetoclax backbone seems to be very effective and was generally well tolerated. The results obtained from the use of nonintensive regimens studied in PARADIGM and SAVE will open up more avenues for the evaluation of such regimens in patients with AML who are likely to be able to tolerate these regimens well. With longer follow-up, we will be able to get a better sense of the durability of response and OS, especially among patients with adverse risk factors such as those with AML harboring NPM1 mutations and KMT2A rearrangements.
KOMET-007: Study Design
Amir T. Fathi, MD:
Similar to revumenib, ziftomenib is a menin inhibitor. Ziftomenib acts by disrupting the formation of the menin-KMT2A complex and blocking the oncogenic activity of mutant NPM1, resulting in terminal differentiation of leukemia blasts. By so doing, it elicits response and remission in leukemia cells.10 Recently, ziftomenib received FDA approval as monotherapy for patients with relapsed/refractory AML with a susceptible NPM1 mutation who have no satisfactory alternative treatment options.11 Ziftomenib has also demonstrated activity in combination with other regimens in NPM1-mutant AML.12-14 Therefore, KOMET-007 is investigating ziftomenib-containing triplet combination in patients with AML.15
KOMET-007 is a multicenter, open-label, phase I dose-escalation and dose-expansion study for patients 18 years of age or older with newly diagnosed or relapsed/refractory NPM1-mutant AML (NCT05735184). KOMET-007 is investigating venetoclax/azacitidine or venetoclax in combination with ziftomenib or standard induction cytarabine/daunorubicin (7+3) chemotherapy in combination with ziftomenib.
At ASH 2025, data from a cohort of 40 patients with newly diagnosed NPM1-mutant AML treated with ziftomenib in combination with venetoclax and azacitidine were presented.15 Ziftomenib at a dose of 600 mg per day was initiated on Day 8 of cycle 1 following the completion of the 7 days of azacitidine and ongoing venetoclax dosing in 28-day cycles. The coprimary endpoints were CR rate and safety, and the secondary endpoints were composite CR (CRc) rate, DoR, ORR, and MRD.
KOMET-007: Baseline Characteristics
Amir T. Fathi, MD:
The median age was 75 years (range: 53-93). Out of 40 patients, 53% were females, 78% were White, 3% had an ECOG PS of 0, 40% had an ECOG PS of 1, and 58% had an ECOG PS of 2. Comutations with NPM1 mutations included FLT3 mutations (35%) and IDH1/2 mutations (23%).
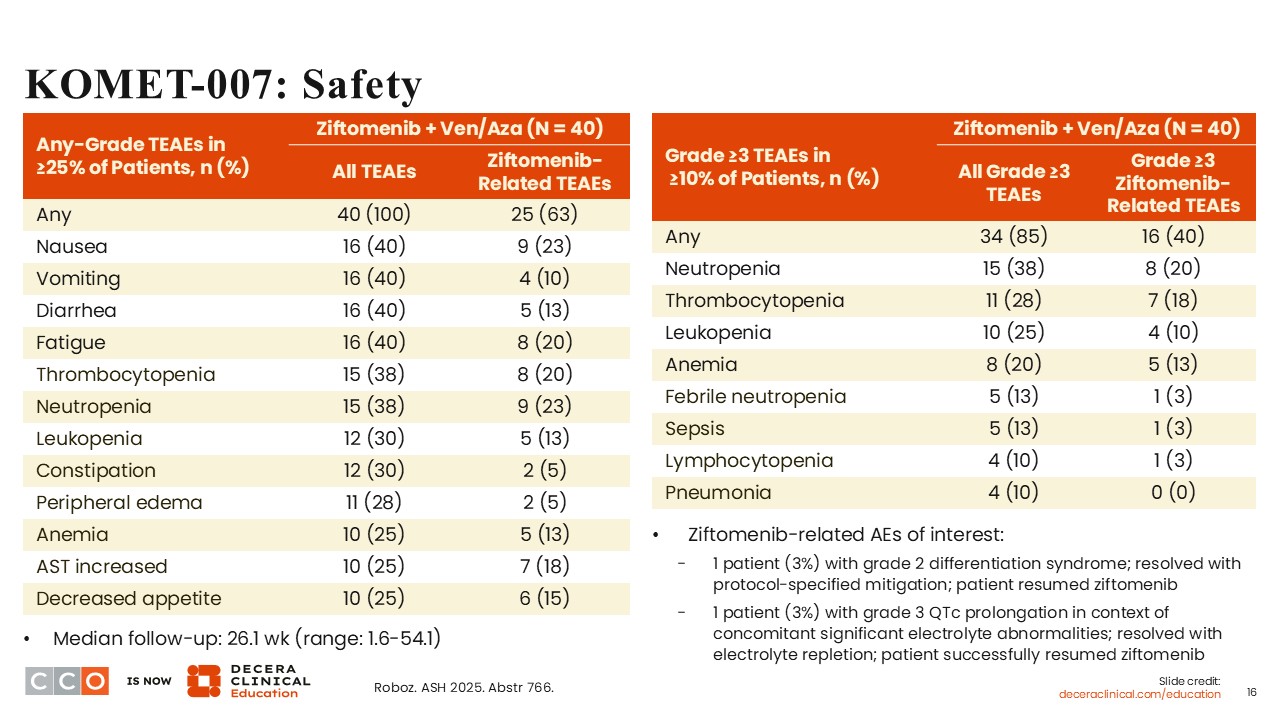
KOMET-007: Safety
Amir T. Fathi, MD:
Ziftomenib-related treatment-emergent adverse events (TEAEs) of any grade occurring in ≥25% of the patients were reported in 63%. The majority of the TEAEs were cytopenias, including thrombocytopenia (20%), neutropenia (23%), and leukopenia (13%). Grade ≥3 ziftomenib-related TEAEs occurring in ≥10% of patients included neutropenia (20%), thrombocytopenia (18%), anemia (13%), leukopenia (10%), and lymphocytopenia (3%).
Differentiation syndrome related to ziftomenib (grade 2) was reported in 1 patient; however, symptoms resolved with protocol-specified mitigation, and the patient was able to resume treatment with ziftomenib. Grade 3 QTc prolongation was reported in 1 patient, but this AE involved concomitant significant electrolyte abnormalities. The patient was able to resume ziftomenib therapy following symptom resolution. Of note, unlike revumenib, ziftomenib is not as associated with QTc prolongation.
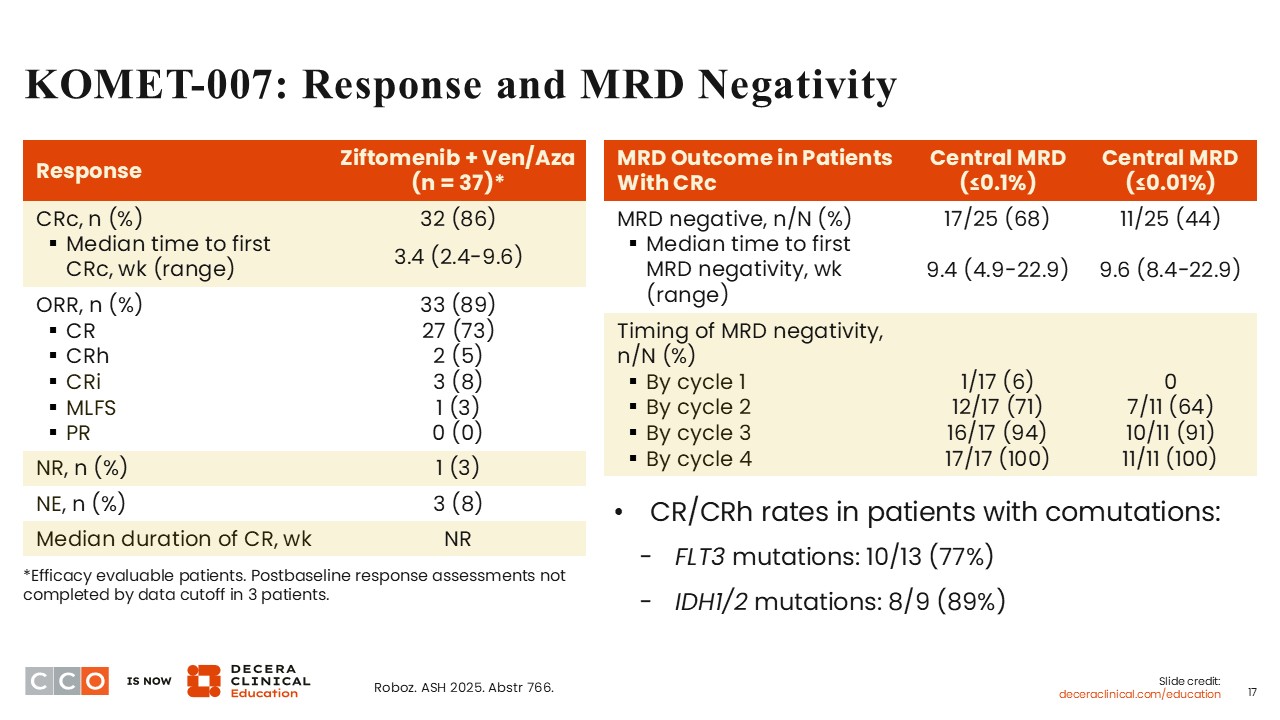
KOMET-007: Response and MRD Negativity
Amir T. Fathi, MD:
The CRc rate was promising at 86%, including 73% of the patients who achieved a CR. Of note, the ORR was 89%. Among evaluable patients with comutations, the CR/CRh rates were 77% in those with FLT3 mutations and 89% in those with IDH1/2 mutations.
Assessment of MRD in 25 evaluable patients demonstrated MRD negativity by central NGS at a threshold of ≤0.1% in 68%. The MRD negativity rate was 44% when determined at a threshold of ≤0.01%. By the end of cycle 2, the rate of MRD negativity was approximately 64% to 71%. All patients achieved MRD negativity by cycle 4 of treatment
KOMET-007: Additional Clinical Outcomes
Amir T. Fathi, MD:
The median time to absolute neutrophil count (ANC) recovery of ≥0.5-1.0 x 109/L was approximately 36-37 days (range: 1-69). This is akin to what is expected with azacitidine and venetoclax. The median time to platelet recovery ≥50 x 109/L was 24 days (range: 0-84), and the median time to platelet recovery to ≥100 x 109/L was 30 days (range: 20-77). After treatment, 5 patients proceeded to undergo HSCT, after which 3 received ziftomenib maintenance. At the time of data analysis, 27 out of 40 patients (68%) who received ziftomenib plus venetoclax and azacitidine were still alive and on study.
KOMET-007: Conclusions and Clinical Implications
Amir T. Fathi, MD:
The regimen of ziftomenib plus venetoclax and azacitidine for patients with newly diagnosed NPM1-mutant AML who are ineligible to receive intensive chemotherapy was active and well tolerated. The CRc rate of 86% is promising and more than expected with the use of azacitidine and venetoclax alone. These results support the continued evaluation of this regimen. The ongoing randomized phase III KOMET-017 trial is investigating ziftomenib in combination with either venetoclax plus azacitidine or 7+3 therapy for patients with untreated NPM1-mutant or KMT2A-rearranged AML (NCT07007312).
Courtney D. Dinardo, MD:
The results obtained with the triplet regimen investigated in the KOMET-007 study highlight previous observations in other trials that the use of a hypomethylating agent and venetoclax in combination with targeted therapy for patients with newly diagnosed AML is well-tolerated and leads to potentially deeper remissions than those observed with a hypomethylating agent and venetoclax alone. The patient population in KOMET-007 is almost twice that reported in SAVE; yet the ORRs are similar at approximately 90% in both trials. Also, the rate of MRD negativity as determined by NGS by the end of cycle 2 was similar in both trials at approximately 70%. These results support the need for further evaluation of the KOMET-007 trial regimen in a larger study.
Amir T. Fathi, MD:
I agree. Hopefully, we will soon see more data from KOMET-007 with longer follow-up. With the evaluation of these nonintensive triplet regimens in randomized controlled studies, we will be better able to determine the efficacy and safety of these regimens in the future.
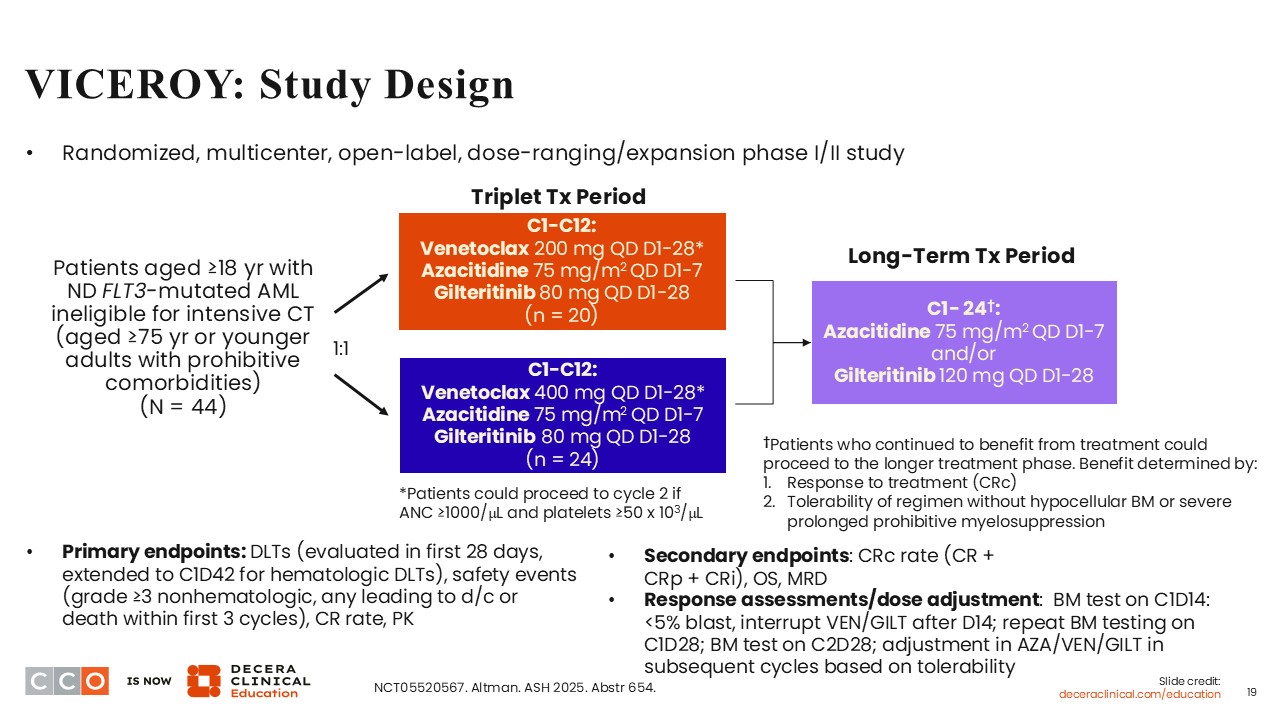
VICEROY: Study Design
Courtney D. Dinardo, MD:
Survival outcomes with azacitidine and venetoclax in patients with FLT3-mutated AML are dismal.16 Therefore, a single-center, proof-of-principle study evaluated triplet therapy with azacitidine and venetoclax in combination with the FLT3 inhibitor, gilteritinib. This trial demonstrated the efficacy of the triplet combination with a CR rate of 90% and a 1-year OS rate of 83% in patients with newly diagnosed FLT3-mutant AML who are ineligible for intensive induction chemotherapy.17 It is important to further determine if outcomes can be significantly improved with the addition of gilteritinib to azacitidine plus venetoclax in a larger cohort of patients with FLT3-mutant AML. The efficacy and safety of azacitidine plus venetoclax and gilteritinib in the VICEROY trial for patients with FLT3-mutant AML were presented at ASH 2025.18
VICEROY is a randomized, dose-ranging/dose-expansion phase I/II study investigating the combination of gilteritinib with azacitidine and venetoclax dosed at either 200 mg or 400 mg daily.18 It is very important to point out that this is a multicenter trial involving more than 9 different institutions.
Patients aged 18 years of age or older with newly diagnosed FLT3-mutant AML who are not candidates for intensive chemotherapy were eligible to participate in this trial. Also, patients 75 years of age or older and younger patients with prohibitive comorbidities were eligible to participate. In total, 44 patients were randomly assigned to receive the standard dose of azacitidine plus gilteritinib dosed at 80 mg daily in combination with 200 mg or 400 mg of venetoclax. Of note, the use of gilteritinib at 80 mg is lower than the FDA-approved daily dose of gilteritinib (120 mg) when used as a single agent.19 Being a dose-finding study, 2 different doses of venetoclax were investigated in VICEROY. Bone marrow tests were performed on Day 14 of cycle 1. If <5% blasts or hypocellular bone marrow is detected, the administration of venetoclax and gilteritinib is interrupted after Day 14.
The primary endpoints were dose-limiting toxicities (DLTs), safety, CR rate, and pharmacokinetics. The secondary endpoints were CRc rate, OS, and MRD.
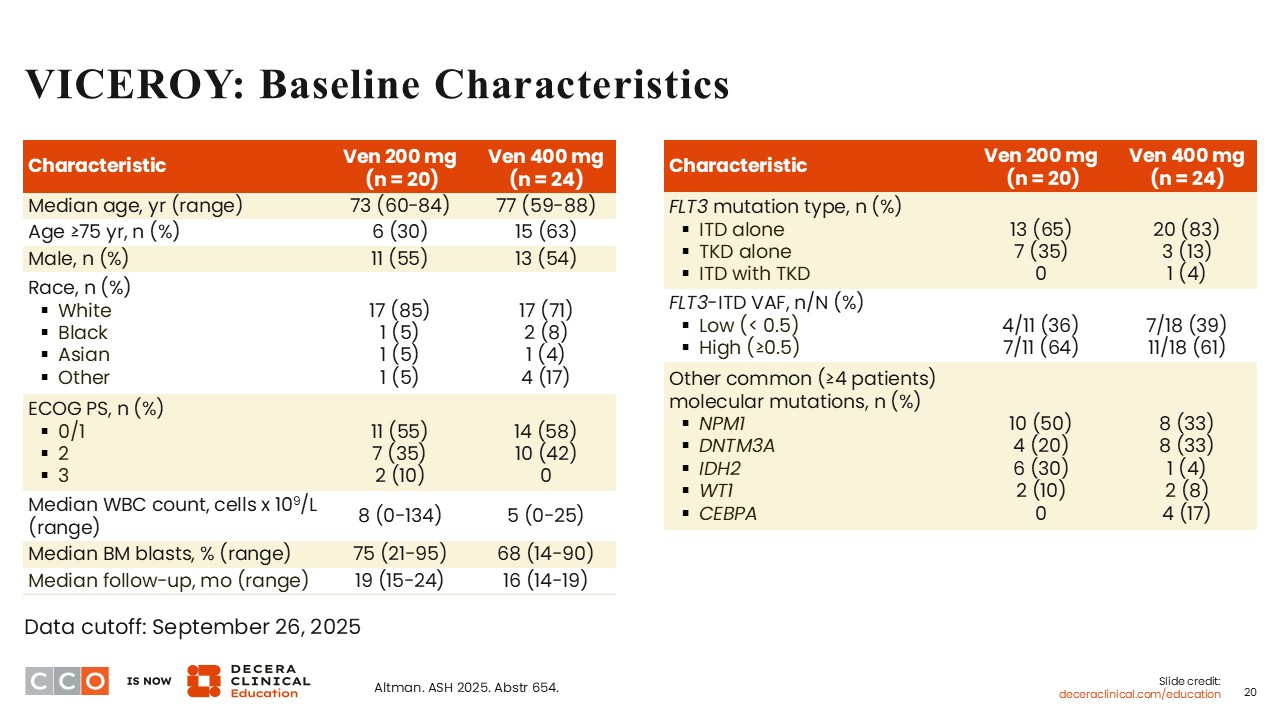
VICEROY: Baseline Characteristics
Courtney D. Dinardo, MD:
The baseline characteristics were fairly well balanced between the 2 treatment arms. The median age was 73 years (range: 60-84) in the 200 mg arm, and 77 years (range: 59-88) in the 400 mg arm. This confirms that the patient population was older and chemotherapy ineligible.
In both arms, approximately 60% of patients had an ECOG PS of 0-1, and approximately 40% had an ECOG PS of 2. The majority of patients had AML harboring FLT3 ITD mutations, detected in 65% of patients on the 200 mg arm and in 83% of those on the 400 mg arm. Co-occurrence of FLT3 mutations and NPM1 mutations was identified in 50% of patients on the 200 mg arm vs 33% on the 400 mg arm. On either arm, mutations in DNMT3A, IDH2 and WT1 were also detected. Four patients had AML harboring CEBPA mutations, all of whom were on the 400 mg.
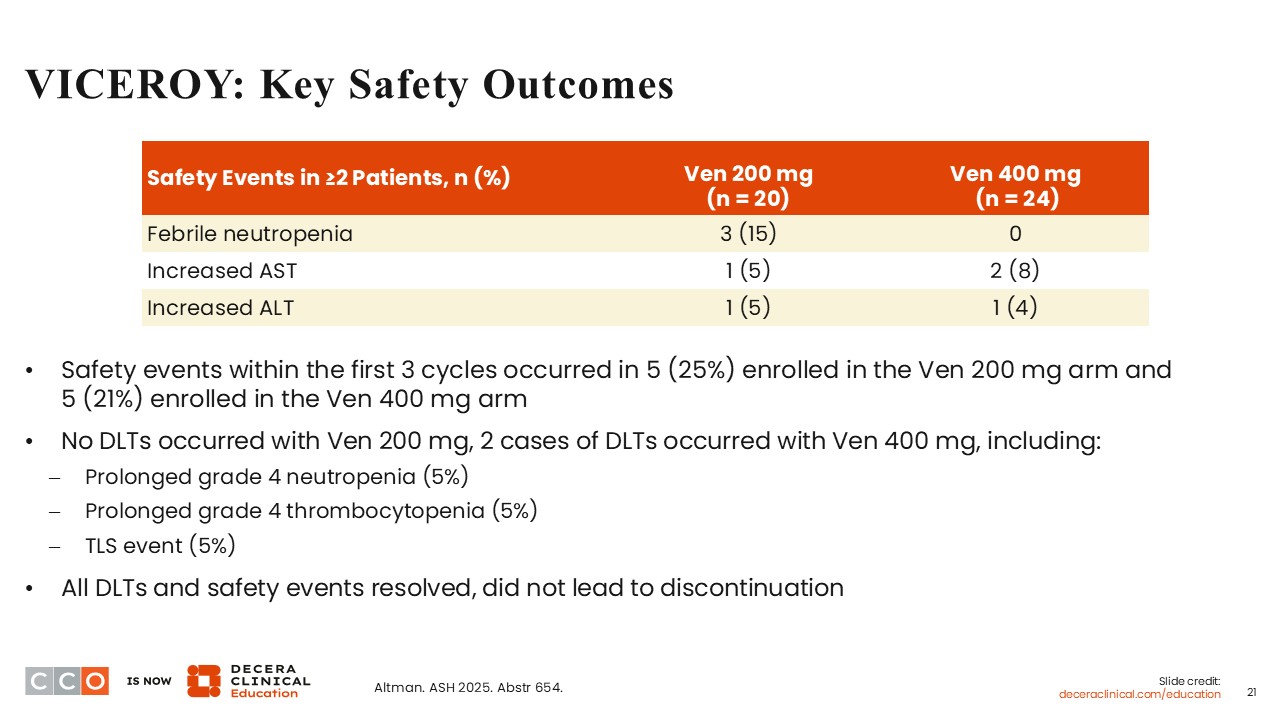
VICEROY: Key Safety Outcomes
Courtney D. Dinardo, MD:
The reported safety events were febrile neutropenia and increased aspartate transaminase/alanine transaminase. There were no DLTs reported on the 200 mg arm. However, 2 cases of DLTs occurred with 400 mg of venetoclax, including prolonged grade 4 neutropenia (5%), prolonged grade 4 thrombocytopenia (5%), and tumor lysis syndrome (5%). All DLTs and safety events resolved, and none led to treatment discontinuation.
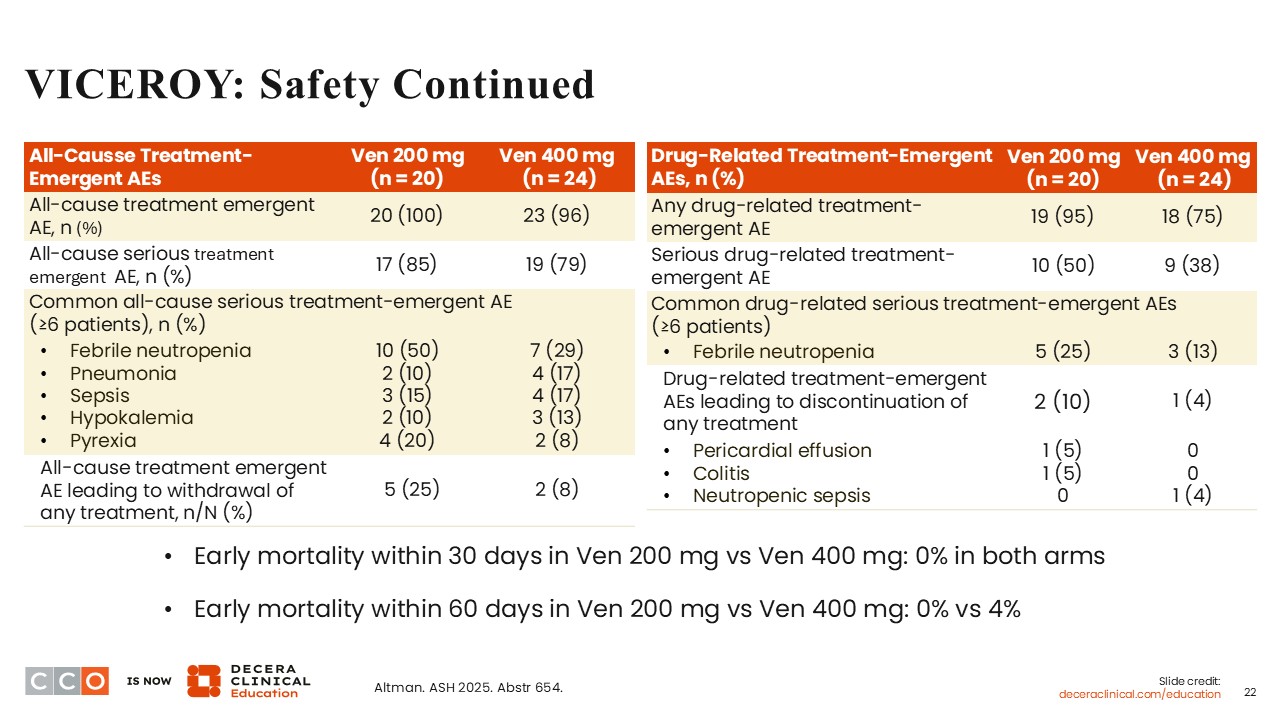
VICEROY: Safety Continued
Courtney D. Dinardo, MD:
Common all-cause TEAEs on both arms included febrile neutropenia, pneumonia, sepsis, hypokalemia, and pyrexia. Drug-related TEAEs leading to treatment discontinuation on the 200 mg arm were pericardial effusion (5%) and colitis (5%). On the 400 mg arm, neutropenic sepsis led to treatment discontinuation in 1 patient. There were no deaths within 30 days of treatment on either arm. However, within 60 days of treatment, the mortality rate on the 400 mg arm was 4%.
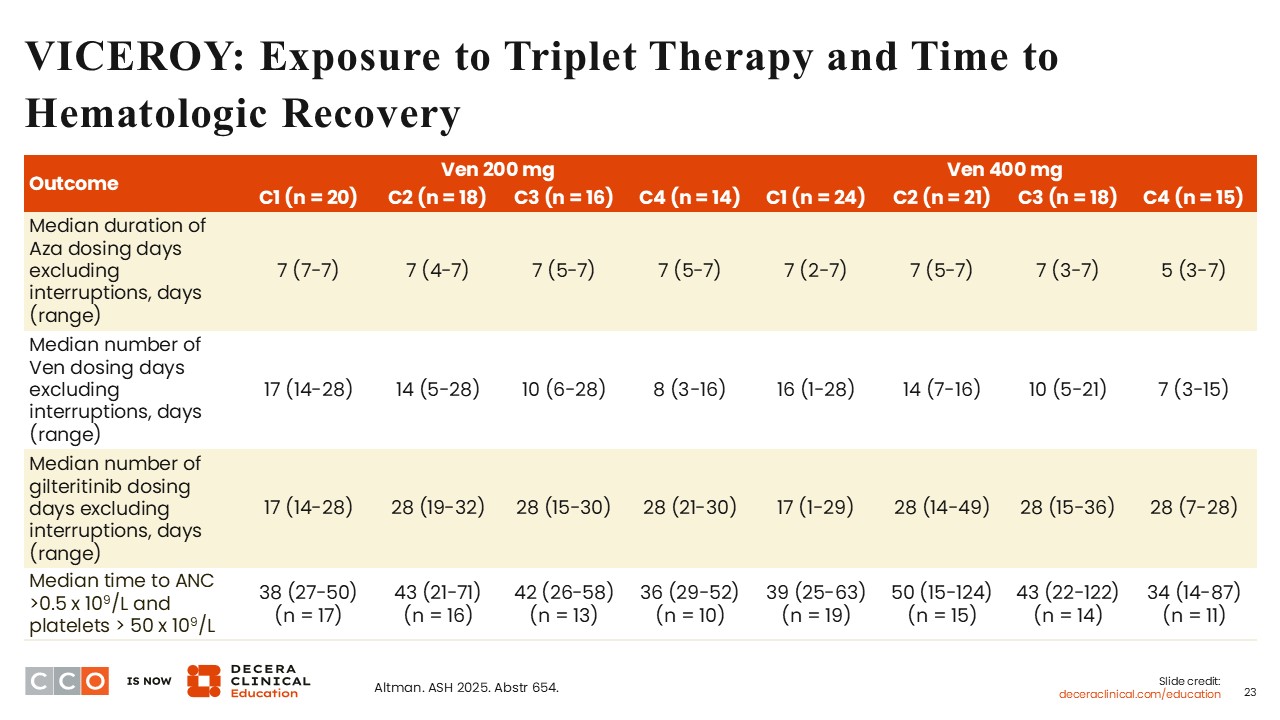
VICEROY: Exposure to Triplet Therapy and Time to Hematologic Recovery
Courtney D. Dinardo, MD:
It is important to note that the median duration of azacitidine dosing was 7 days in cycles 1-4, especially on the venetoclax 200 mg arm. On both treatment arms, the median number of gilteritinib dosing days was 17 in cycle 1, and this returned to a continuous 28 in cycles 2-4. This highlights the fact that most patients were clearing bone marrow blasts to <5% on Day 14 of cycle 1. On average, the median number of venetoclax dosing days in all patients in cycle 2 was 14, and this reduced in cycles 3 and 4. The median time to ANC recovery >0.5 x 109/L and platelet recovery >50 x 109/L ranged from 34-50 days in cycles 1 to 4 on both arms.
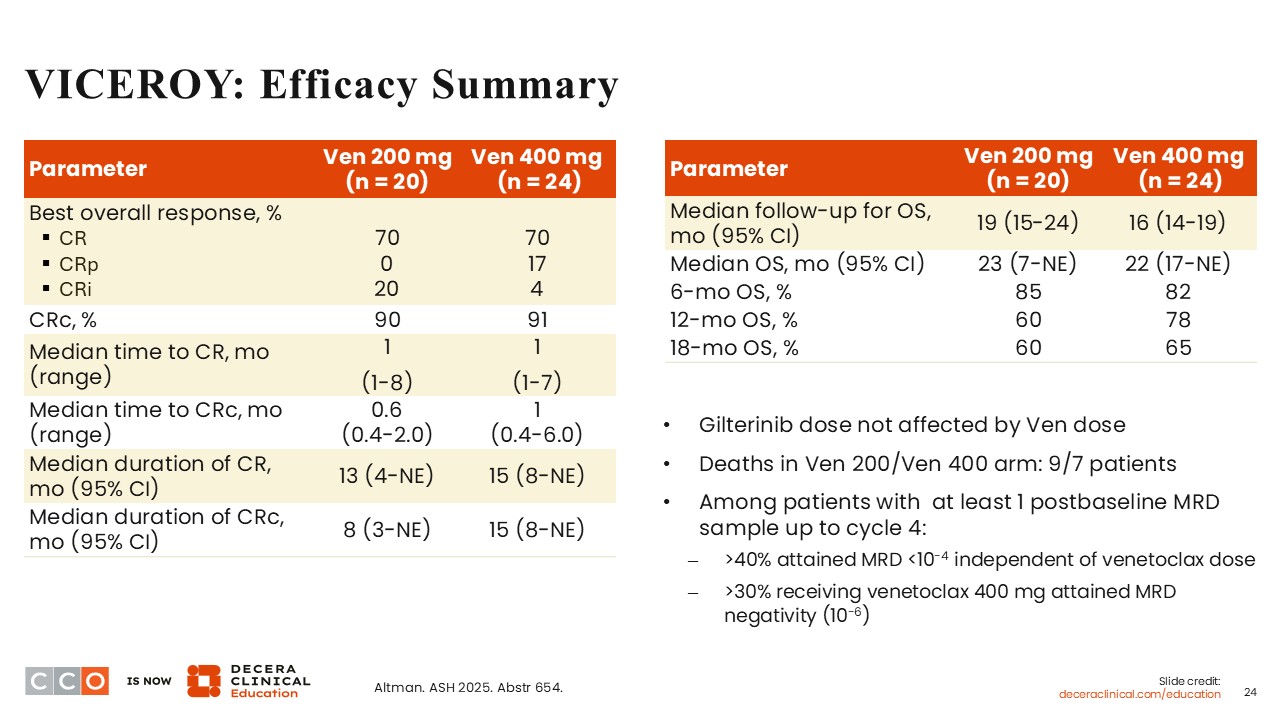
VICEROY: Efficacy Summary
Courtney D. Dinardo, MD:
The CR rate was 70% in both arms, whereas the CRc rate was 90% in the 200 mg of venetoclax arm vs 91% in the 400 mg of venetoclax arm. The median time to CR was 1 month on both arms, and the median time to CRc was 0.6 months in the 200 mg arm vs 1 month in the 400 mg arm. These results are impressive considering that the patient population being studied had FLT3-mutant AML, with the majority harboring FLT3 ITD mutations.
After a median follow-up of 19 months, the median OS was 23 months on the 200 mg arm. After a median follow-up of 16 months, the median OS was 22 months on the 400 mg arm. These results are promising. Of note, gilteritinib dose was not affected by the dose of venetoclax studied in each arm. There were 9 deaths in the 200 mg arm vs 7 deaths in the 400 mg arm.
VICEROY: Conclusions and Clinical Implications
Courtney D. Dinardo, MD:
This triplet regimen of azacitidine plus venetoclax and gilteritinib led to promising safety and efficacy in the population of patients with FLT3-mutant AML. The incidence of DLTs was low, and so was the 60-day mortality rate, which was 4% on the 400 mg of venetoclax arm. The investigators stated that patient enrollment on the phase II portion of the study incorporating the 400 mg dose has been completed and dose adjustment recommendations are ongoing. This combination is encouraging and needs to be studied in a larger patient population.
Amir T. Fathi, MD:
This is another triplet combination for newly diagnosed older patients with AML. Whereas the KOMET-007 trial investigated a triplet combination for patients with NPM1-mutant AML, VICEROY was for patients with FLT3 mutations. Although the results are for only 40 patients with FLT3-mutant AML, the response rates in VICEROY are promising, and the median OS seems better than what was reported for the cohort of 42 patients with FLT3 mutations in the randomized phase III VIALE-A trial that investigated the addition of venetoclax to azacitidine.16
In the VIALE-A trial, the CR rate with azacitidine plus venetoclax (400 mg daily) in patients with FLT3-mutant AML was 38.1%, and the median OS was 12.5 months. If the results demonstrated in the VICEROY trial are replicated in a randomized trial in a larger patient population with FLT3-mutant AML, the triplet regimen will be transformative, particularly if the patients, who are older with preexisting comorbidities, are able to tolerate the regimen well.18
One of the bigger challenges with the use of triplet combinations including a novel agent such as a FLT3 inhibitor is that they can induce bone marrow suppression. There are ongoing efforts to optimize treatment duration using various approaches to incorporate the 3 agents in a way that will reduce the associated myelosuppressive effects. VICEROY also attempts to do this by studying 2 different doses of venetoclax.
Also, the trial design to perform an early bone marrow biopsy to guide decision-making about how to approach venetoclax and gilteritinib dosing was very useful because it can decrease the potential bone marrow suppression and allowed the regimen to be better tolerated in an older patient population. This may have translated into improved OS, and the higher and deeper response rates reported. Also, the early mortality rate was promising, and there is potentially a subset of these patients that may even be able to proceed to bone marrow transplantation with better and improved outcomes.
I have collaborated with investigators at MD Anderson in studies of 2 different triplet regimens including FLT3 inhibitors for patients with AML, and we have seen very similar outcomes in terms of response and safety. That the VICEROY regimen was able to move forward as a multicenter study and that the study reported similar outcomes is really encouraging and speaks to the fact that these regimens can be safely utilized if the optimal dosing strategy and treatment duration can be confirmed in the future. There are plans to look at these regimens in a more randomized fashion in the future. Overall, these are exciting times for patients with FLT3-mutant AML.
Courtney D. Dinardo, MD:
I agree. This is another study of a triplet regimen in a targeted patient population, highlighting that these regimens elicit impressive response rates, deep remissions, and hopefully more durable remissions with the ability to transition to transplant in a timely and effective manner. The potential to move these triplet regimens into the management of younger and fit patients with AML is promising, and this is an important question for the future.
In fact, the ongoing randomized phase II/III FIT-AML trial is investigating gilteritinib in combination with azacitidine and venetoclax vs induction chemotherapy (7+3) in combination with a FLT3 inhibitor for fit patients 18-75 years of age with newly diagnosed FLT3-ITD–mutant AML (NCT07425808). This is an important randomized trial that will provide clarity in the years to come.
PVEK + Azacitidine + Venetoclax in AML: Study Design
Amir T. Fathi, MD:
CD123 is a cell surface protein with low expression on normal hematopoietic stem cells but overexpressed in 93% to 97% of AML cases.20-22 Pivekimab sunirine (PVEK) is a first-in-class antibody–drug conjugate (ADC) with a high affinity for CD123.23 A phase Ib/II study is investigating PVEK as monotherapy or in combination with venetoclax and/or azacitidine for patients with CD123-positive AML (NCT04086264). In an interim analysis of this phase Ib/II study, PVEK plus venetoclax and azacitidine demonstrated encouraging response rates, and the safety profile was manageable.24 At ASH 2025, results from the phase Ib/II trial with a focus on the subgroup of unfit patients with newly diagnosed CD123-positive AML who received the triplet combination of PVEK, venetoclax, and azacitidine were presented.25
This phase Ib/II trial is a multicohort, dose-escalation/dose-expansion trial for patients aged 18 years of age or older with AML. Of note, the age cutoff of ≥18 years was a broad age category because the study is also investigating this triplet combination in the relapsed/refractory setting, a patient population that is not included in the ASH 2025 report.
The eligibility criteria included a confirmed diagnosis of CD123-positive AML by local flow cytometry or immunohistochemistry.25 Fit patients had an ECOG PS of ≤1. For the expansion phase of the study, unfit patients were classified as those 75 years of age or older, those younger than 75 years of age with an ECOG PS of 2-3, or those with ≥1 preexisting comorbid condition.
To reiterate, the focus of the ASH 2025 presentation was on unfit patients with newly diagnosed CD123-positive AML. These patients received azacitidine plus either 14 days of venetoclax plus PVEK or 28 days of venetoclax plus PVEK. The dose of venetoclax studied was 400 mg daily. Bone marrow evaluations were performed on Days 12-14 in the cohort of patients who received 14 days of venetoclax, and on Days 18-21 in the cohort of patients who received 28 days of venetoclax. This was done to determine the duration of venetoclax in cycle 1, end of cycle 2, and thereafter, every other cycle to determine responses and bone marrow suppression. It is important to note that in this trial, the effort to adjust the duration of venetoclax was to improve tolerability to the regimen, especially regarding myelosuppression frequently associated with the use of triplet combinations in AML, and to optimize treatment dose and duration.
In total, 76 patients with previously untreated AML were enrolled. Of these patients, 49 met the criteria for designation as unfit. The primary endpoint was CR rates, and the secondary endpoints included safety and MRD, which was defined as <0.1% using flow cytometry by central testing.
PVEK + Azacitidine + Venetoclax in AML (Untreated Unfit Cohorts): Baseline Characteristics and Treatment Exposure
Amir T. Fathi, MD:
The median age was 77 years (range: 58-85), 29% had an ECOG PS of 0, 57% had an ECOG PS of 1, 12% had an ECOG PS of 2, and 2% had an ECOG PS of 3. The majority of the patients had an ECOG PS of ≤1. The majority of the patients had adverse-risk disease (51%), 20% had intermediate-risk disease, and 18% had favorable-risk disease. Of important note, 35% had disease harboring a TP53 mutation. Other mutations detected included NPM1 (22%), FLT3 (20%), and IDH1/2 (8%).
In general, the median duration of azacitidine exposure was 5.7 months (range: 0.9-23.4). The median duration of venetoclax exposure was 5.1 months (range: 0.9-22.9), and the median duration of PVEK exposure was 5.5 months (range: 0.7-22.7). For all patients in Cohort 1, the median duration of venetoclax exposure was 4.5 months (2.1-22.9), and in Cohort 2, the median duration of venetoclax exposure was 5.7 months (range: 0.9-15.2).
PVEK + Azacitidine + Venetoclax in AML (Untreated Unfit Cohorts): Response and OS
Courtney D. Dinardo, MD:
Among 49 evaluable patients who were classified as fit with newly diagnosed CD123-positive AML, the CR rate was 63%, the CR/CRi rate was 80%, and the CR/CRh rate was 74%. These response rates are impressive. Of particular interest is the median duration of CR or CR/CRi, which was 16.1 months with a median time to CR or CR/CRi of 1.2 months. MRD response among 26 evaluable patients who achieved CR was 92%, and MRD response among 32 evaluable patients who achieved CR/CRi was 91%.
Analysis according to cohort showed a CR rate of 50% in Cohort 1 and a CR rate of 67% in Cohort 2. The CR/CRi rates were 70% and 82% in Cohorts 1 and 2, respectively. MRD negativity rates in patients who achieved a CR were 80% and 95% in Cohorts 1 and 2, respectively. Among those who achieved CR/CRi, the MRD negativity rates were 83% and 92%, respectively. Overall, this combination elicited remarkably high and deep responses.
Among the 49 patients included in the study, 17 had TP53-mutant AML and 32 had TP53 wild-type disease. In the TP53 mutation–positive subgroup, the CR rate was 35%, the CR/CRi rate was 53%, and the CR/CRh rate was 47%. In the subgroup of patients with TP53–wild-type disease, the CR rate was 78%, the CR/CRi rate was 94%, and the CR/CRh rate was 88%. The median duration of CR or CR/CRi in the TP53 mutation–positive and TP53–wild-type subgroups was 7.8 months and 20.7 months, respectively.
After a median follow-up of 16.1-17.6 months, the median OS in all patients was 12.4 months compared with 8.5 months among patients with TP53 mutation–positive AML. The median OS was not reached in the subgroup of patients with TP53–wild-type AML. The 1-year OS rates for all patients, and among patients with and without TP53 mutation–positive AML were 51%, 6%, and 75%, respectively. These results demonstrate abysmal efficacy with the combination of PVEK plus azacitidine and venetoclax in patients with TP53 mutation–positive AML.
PVEK + Azacitidine + Venetoclax in AML (Untreated Unfit Cohorts): Safety
Courtney D. Dinardo, MD:
Any grade hematologic TEAEs associated with treatment included anemia (45%), neutropenia (74%), febrile neutropenia (35%), thrombocytopenia (67%), decreased white blood cell count (49%), and decreased lymphocyte count (10%). Grade ≥3 hematologic TEAEs included anemia (39%), neutropenia (67%), febrile neutropenia (35%), thrombocytopenia (63%), decreased white blood cell count (49%), and decreased lymphocyte count (10%). Hematologic TEAEs are expected with the use of azacitidine/venetoclax-based therapies.
Grade ≥3 nonhematologic TEAEs included pneumonia (16%), hypertension (14%), sepsis (12%), hypokalemia (10%), and bone marrow failure, fatigue, hypophosphatemia, and weight loss (6%, each). A theoretical concern in patients receiving ADCs targeting CD123 or anti-CD33 ADCs like gemtuzumab ozogamicin is the occurrence of veno-occlusive disease. Of note, there was no incidence of veno-occlusive disease with PVEK in this trial. Overall, serious TEAEs occurred in 78% of patients. There were 2 deaths in Cohort 2 and none reported in Cohort 1.
PVEK + Azacitidine + Venetoclax in AML (Untreated Unfit Cohorts): Conclusions and Clinical Implications
Courtney D. Dinardo, MD:
The use of azacitidine and venetoclax plus PVEK for unfit patients with newly diagnosed CD123-positive AML demonstrated high CR rates and CR/CRi with durable responses, primarily in patients with TP53–wild-type AML. This triplet combination was well tolerated, and no new safety signals were reported. These results need to be evaluated in a larger, randomized trial to confirm the results obtained.
Amir T. Fathi, MD:
This trial is interesting and unique because it does not specifically target a subtype of AML, per se. The regimen is more broadly applicable to AML. The response rates are impressive, and the regimen was well tolerated. That peripheral edema occurred in 4% of patients is an interesting finding that is probably related to targeting CD123. This potential toxicity needs to be carefully monitored in patients receiving PVEK.
It is disappointing that the treatment outcomes in the subgroup of patients with TP53 mutation–positive disease were suboptimal. Of note, this subtype of AML is highly resistant and tough to treat. Hopefully, this clinical need will be better addressed in the future. Overall, the combination of PVEK with azacitidine and venetoclax is another potential triplet combination for older and unfit patients with newly diagnosed CD123-positive AML.
Courtney D. Dinardo, MD:
I agree. This is yet another exciting study that investigated a triplet combination. Of particular note, it is not targeting any specific genetic alteration, and as such, all patients with CD123-positive AML, except those with TP53 mutation–positive disease, will be candidates for treatment with the regimen.