CE / CME
Conference to Clinic Highlights From the 2025 ASH Annual Meeting: Key Studies in Leukemias
Physician Assistants/Physician Associates: 1.00 AAPA Category 1 CME credit
Pharmacists: 1.00 contact hour (0.1 CEUs)
Physicians: maximum of 1.00 AMA PRA Category 1 Credit™
Nurse Practitioners/Nurses: 1.00 Nursing contact hour
Released: February 27, 2026
Expiration: August 26, 2026
Activity
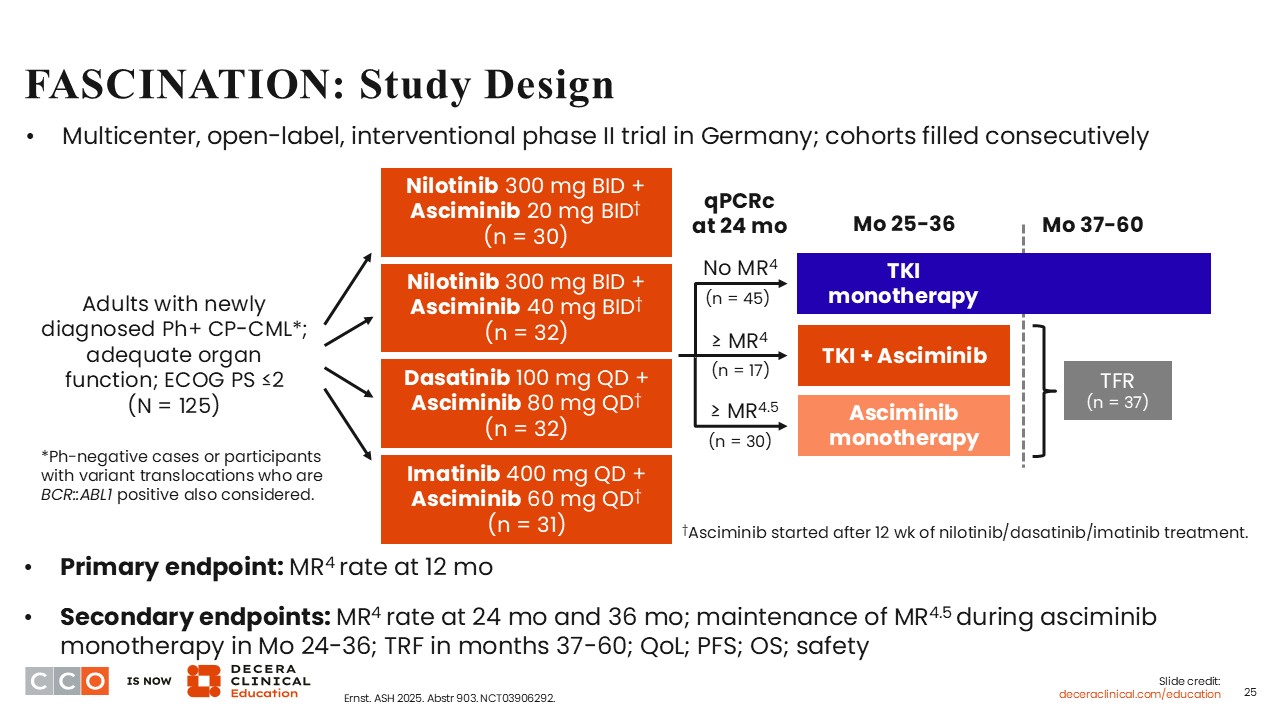
FASCINATION: Study Design
Amir T. Fathi, MD:
There were several interesting studies in CML presented at ASH 2025. The first is the phase II FASCINATION study that evaluated asciminib, an allosteric inhibitor of BCR-ABL1 that targets a myristoyl site.26 Asciminib is FDA approved for the treatment of adults with newly diagnosed or previously treated Philadelphia-positive (Ph+) chronic phase CML (CP-CML).27 At ASH 2025, long-term results after 3 years of follow-up on the FASCINATION trial of asciminib and conventional ATP-competing BCR-ABL1 inhibitors for patients with newly diagnosed CML were presented.28
FASCINATION is a multicenter, multicohort phase II study of first-line asciminib in combination with conventional ATP-competing BCR-ABL1 inhibitors for patients with CP-CML.28 Patients with newly diagnosed CP-CML with adequate organ function were eligible to participate in the study, which was performed in Germany. Patients (N = 125) received treatment in 4 cohorts: (1) nilotinib (300 mg BID) + asciminib (20 mg BID); (2) nilotinib (300 mg BID) + asciminib (40 mg BID); (3) dasatinib (100 mg QD) + asciminib (80 mg QD); and (4) imatinib (400 mg QD) + asciminib (60 mg QD).
Patients who did not achieve 4-log molecular response (MR4) at Month 24 discontinued the asciminib combination and received an ATP-competitive tyrosine kinase inhibitor (TKI) as monotherapy based on investigator’s discretion. Patients who achieved MR4 continued to receive the asciminib combination therapy for 1 year. Those who achieved MR4.5 continued treatment with asciminib monotherapy (80 mg QD) for 1 year. At Month 36, treatment with asciminib was discontinued for all patients. Patients who maintained MR4 or better for ≥1 year proceeded to enter a treatment-free remission phase.
The primary endpoint was the rate of MR4 at Month 12, and the secondary endpoints included MR4 at Months 24 and 36, maintenance of MR4.5 during asciminib monotherapy in Months 24-36, progression-free survival, OS, quality of life, and safety.
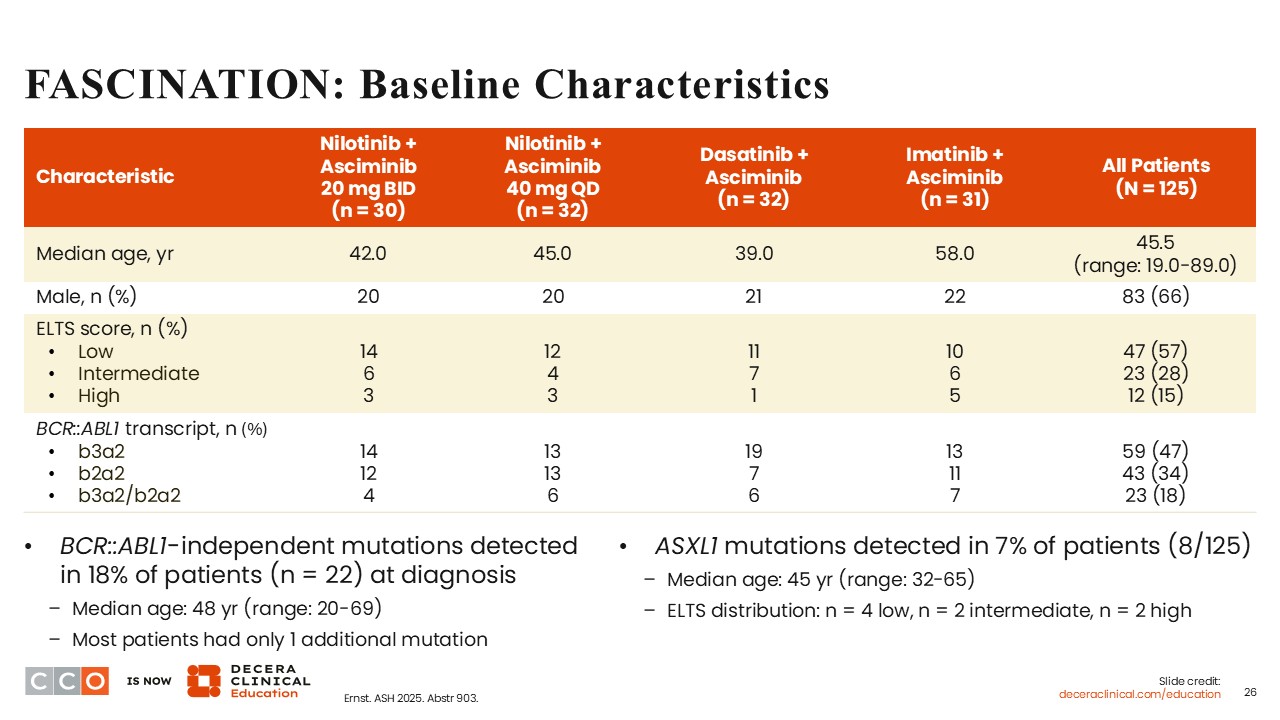
FASCINATION: Baseline Characteristics
Amir T. Fathi, MD:
Overall, the median age was 45.5 years (range: 19-89). The majority of patients were male (66%), as expected with CML. BCR-ABL1–independent mutations were detected in 18% of patients at diagnosis. ASXL1 mutations, which are sometimes seen in CML, were detected in 7% of patients.
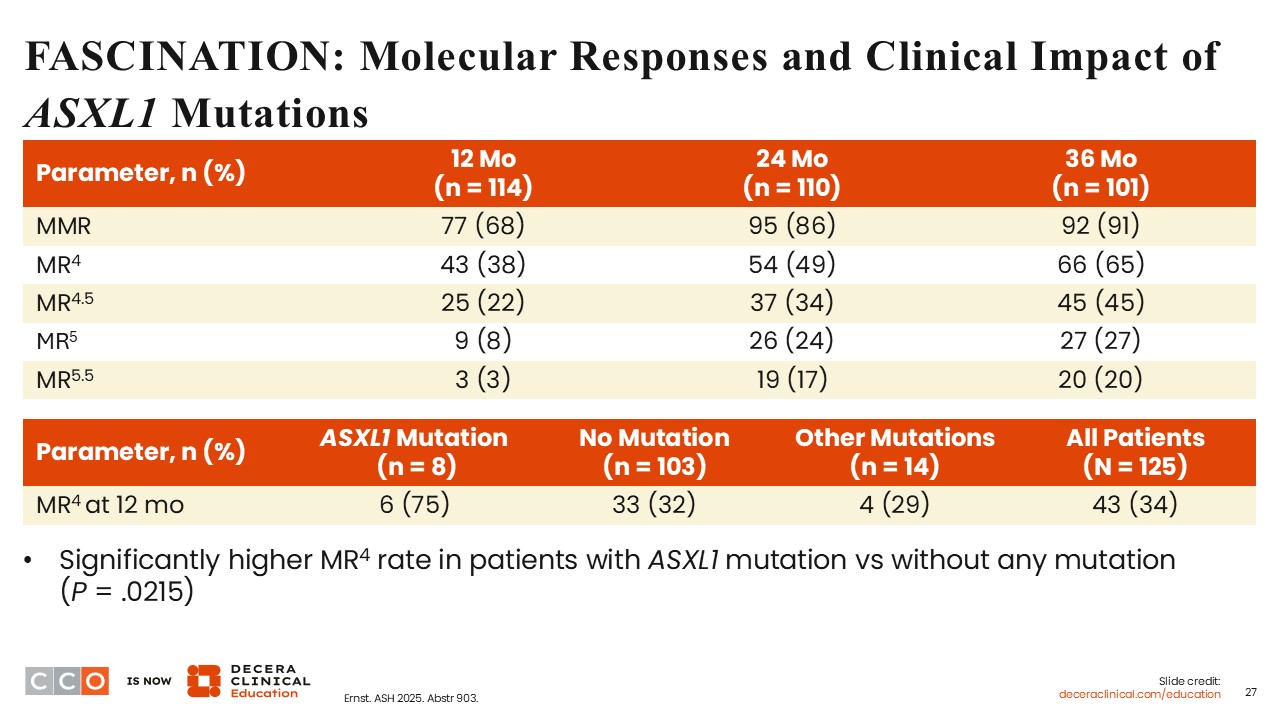
FASCINATION: Molecular Responses and Clinical Impact of ASXL1Mutations
Amir T. Fathi, MD:
The MMR rate at Months 12, 24, and 36 were 68%, 86%, and 91%, respectively. MR4 rate also increased over time, and at Months 12, 24, and 36, it was 38%, 49%, and 65%, respectively. Similarly, the deeper responses, MR4.5, MR5, and MR5.5 rates increased over time. At Month 12, there was a significantly higher rate of MR4 among patients with ASXL1 mutations compared with those without any mutation (P = .0215). These were interesting findings.
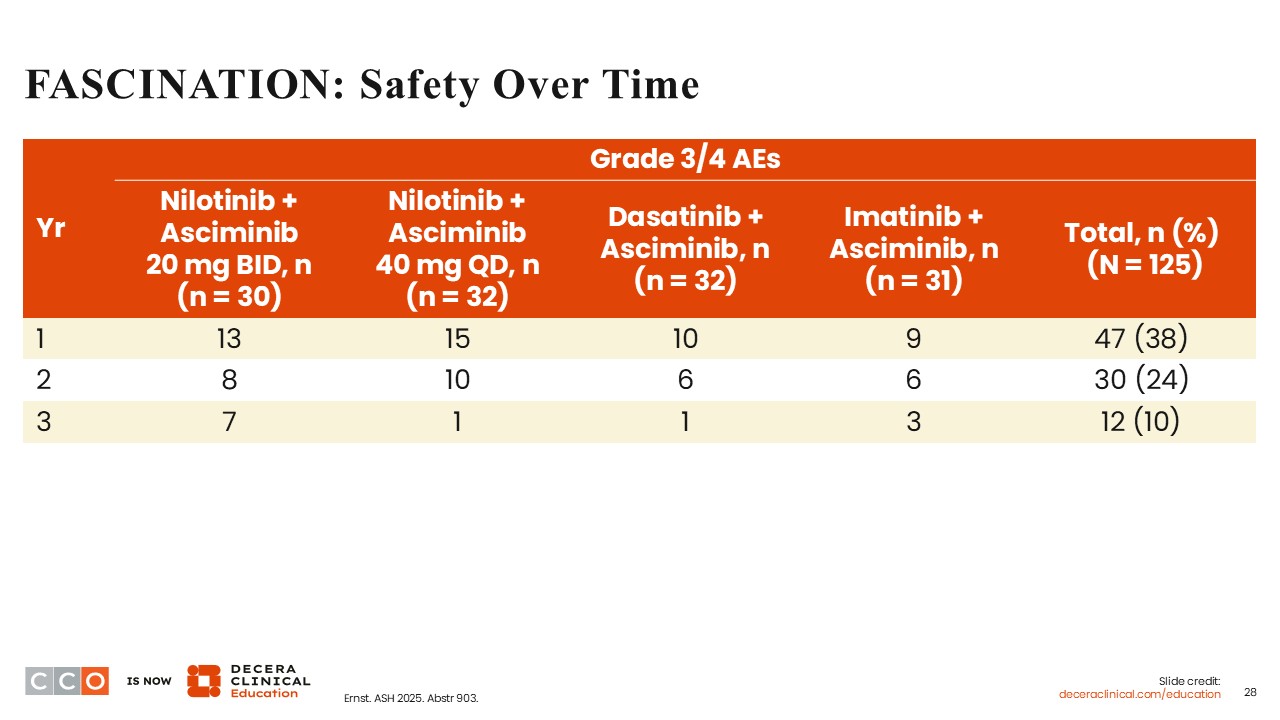
FASCINATION: Safety Over Time
Amir T. Fathi, MD:
The incidence of grade 3/4 AEs reduced over time.
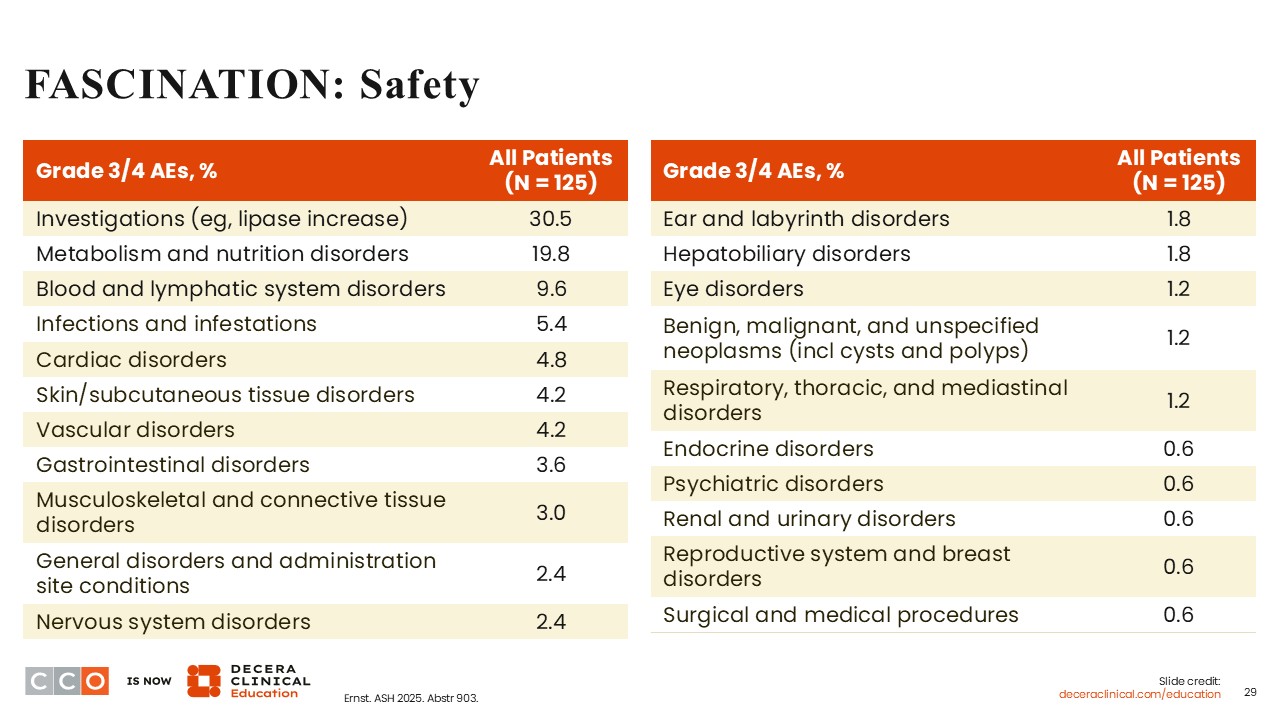
FASCINATION: Safety
Amir T. Fathi, MD:
The incidence of grade 3 or 4 AEs reported, such as lipase increases and metabolism and nutrition disorders, is likely related to nilotinib. The incidence of grade 3 or 4 cardiac disorders, skin or subcutaneous tissue disorders, musculoskeletal and connective tissue disorders, and nervous system disorders was fairly uncommon. Also, blood and lymphatic system disorders were reported in only 9.6% of patients.
FASCINATION: Failures and Progressions
Amir T. Fathi, MD:
Five patients experienced MMR loss, and in 1 of the patients, this was due to an emergent BCR-ABL1 E275G mutation. Two patients experienced disease progression to blast phase, and 1 patient died from myocardial infarction at 24 months.
Fascination: Conclusions and Clinical Implications
Amir T. Fathi, MD:
After 3 years of follow-up, the molecular responses across treatment cohorts were promising and seemed to deepen over time. There was no evidence of patients developing clinically relevant BCR-ABL mutations. The tolerability of the treatments improved over time, and there were no unexpected safety signals.
Courtney D. Dinardo, MD:
Asciminib in combination with other TKIs is an interesting strategy in the treatment of patients with CP-CML. This approach has the potential to lead to more responses, deeper responses, and the development of fewer resistance mechanisms.
Of special interest are the data showing a significantly higher MR4 rate in patients with ASXL1 mutation compared with those without any mutation. In my experience at my institution, patients with ASXL1 mutations tend to have more cytopenias and trouble tolerating regimens. Even though only 8 patients were studied, the finding that more patients with this mutation respond to treatment without significant toxicities is very interesting.
Amir T. Fathi, MD:
Of note, asciminib monotherapy in CML is very effective, and there are ongoing efforts to further investigate its utility as monotherapy in various subgroups of patients with Ph+ CP-CML (NCT07354074).
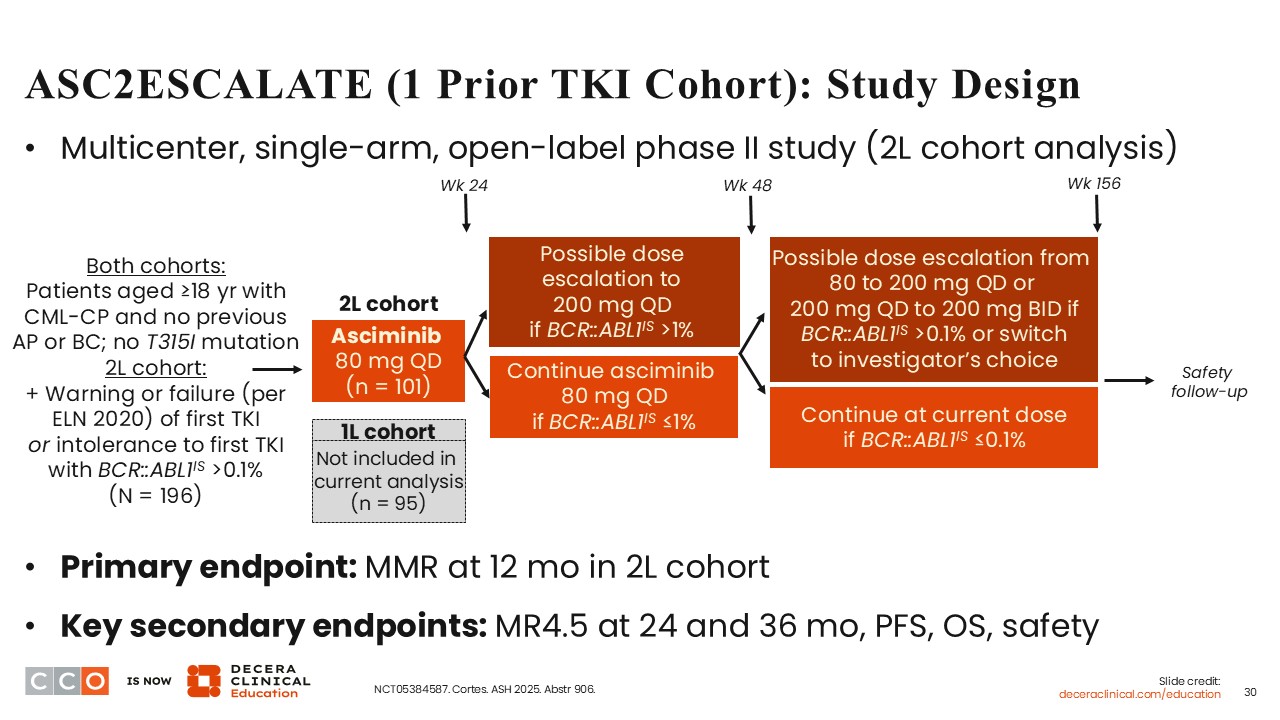
ASC2ESCALATE (1 Prior TKI Cohort): Study Design
Courtney D. Dinardo, MD:
ASC2ESCALATE is a multicenter, multicohort, single-arm, open-label phase II study of asciminib monotherapy as first- and second-line therapy for patients with CP-CML (NCT05384587). The eligibility criteria included patients 18 years of age or older with CP-CML without previous accelerated phase or blast crisis and without a BCR T315I mutation. At ASH 2025, data from the cohort of patients who received asciminib monotherapy as second-line therapy after failure of a first-line TKI were presented.29
A total of 101 patients with CP-CML received second-line asciminib at 80 mg QD. At Week 24, there was the potential to escalate the asciminib dose to 200 mg QD if BCR-ABL1IS was >1%. As designed, it will be possible to determine the effectiveness of second-line asciminib as well as to determine whether dose escalation can be an effective strategy.
The primary endpoint was MMR at 12 months in the second-line cohort, and the secondary endpoints were MR4.5 at 24 and 36 months, progression-free survival, OS, and safety.
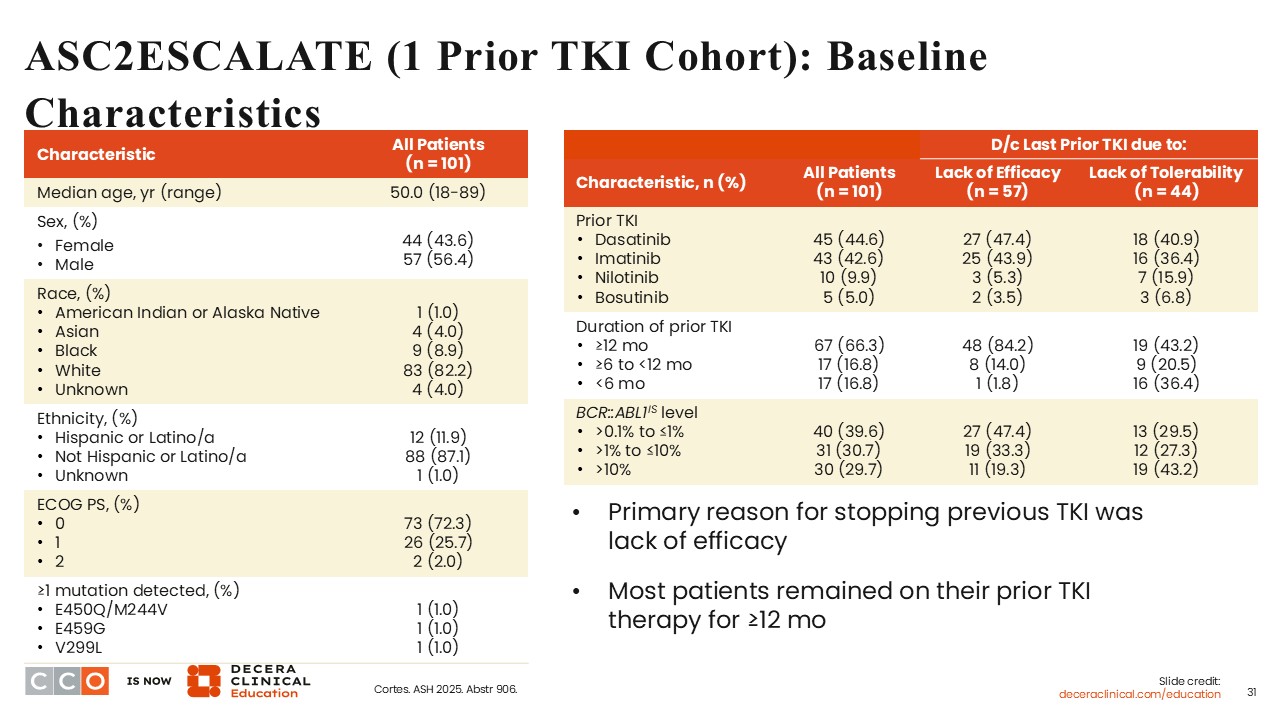
ASC2ESCALATE (1 Prior TKI Cohort): Baseline Characteristics
Courtney D. Dinardo, MD:
The median age was 50 years (range: 18-89), 56.4% were males, and 72.3% of the patients had an ECOG PS of 0. Of interest, only 3 patients had ≥1 BCR-ABL1 mutation. Of 101 patients, 44.6% had previously received dasatinib, 42.6% had previously received imatinib, 9.9% had previously received nilotinib, and 5% had previously received bosutinib. The majority of patients (66.3%) had previously received a TKI for ≥12 months.
This suggests that these patients received a TKI for ≥1 year, with the primary reason for stopping treatment based on the lack of efficacy of reaching the set BCR-ABL milestones. Only 39.6% of the patients achieved BCR-ABL1IS level >0.1% to ≤1%. Approximately 30% of the patients achieved BCR-ABL1IS level >10%.
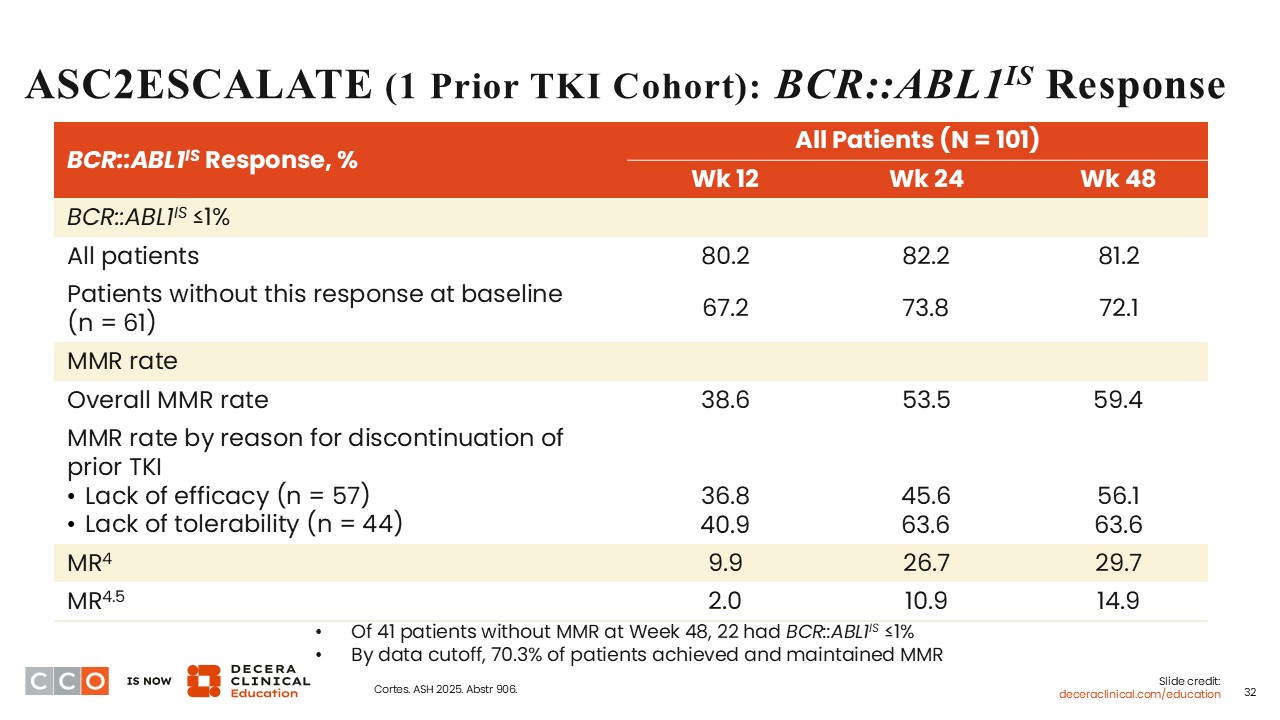
ASC2ESCALATE (1 Prior TKI Cohort): BCR-ABL1IS Response
Courtney D. Dinardo, MD:
At Weeks 12, 24, and 48, the overall MMR rates were 38.6%, 53.5%, and 59.4%, respectively. Regardless of whether the previous TKI was discontinued due to lack of efficacy or lack of tolerability, the MMR rates increased over time. Of particular interest, patients who discontinued the previous TKI due to lack of tolerability seemed to tolerate asciminib monotherapy well with MMR rates of 40.9%, 63.6%, and 63.6% at Weeks 12, 24, and 48, respectively.
ASC2ESCALATE (1 Prior TKI Cohort): Additional Outcomes
Courtney D. Dinardo, MD:
For 18 patients (17.8%), the asciminib dose was escalated from 80 mg to 200 mg QD based on molecular response at Week 24 (n = 4) and Week 48 (n = 14). At the time of data cutoff, 17 were still receiving asciminib. BCR-ABL1IS levels generally declined further after escalation, with most patients showing reductions from >1% or >0.1% toward molecular response targets by Week 48. All 3 patients with baseline mutations achieved MMR by Week 48 on asciminib, and some reached deeper responses. None of these patients experienced disease progression or death.
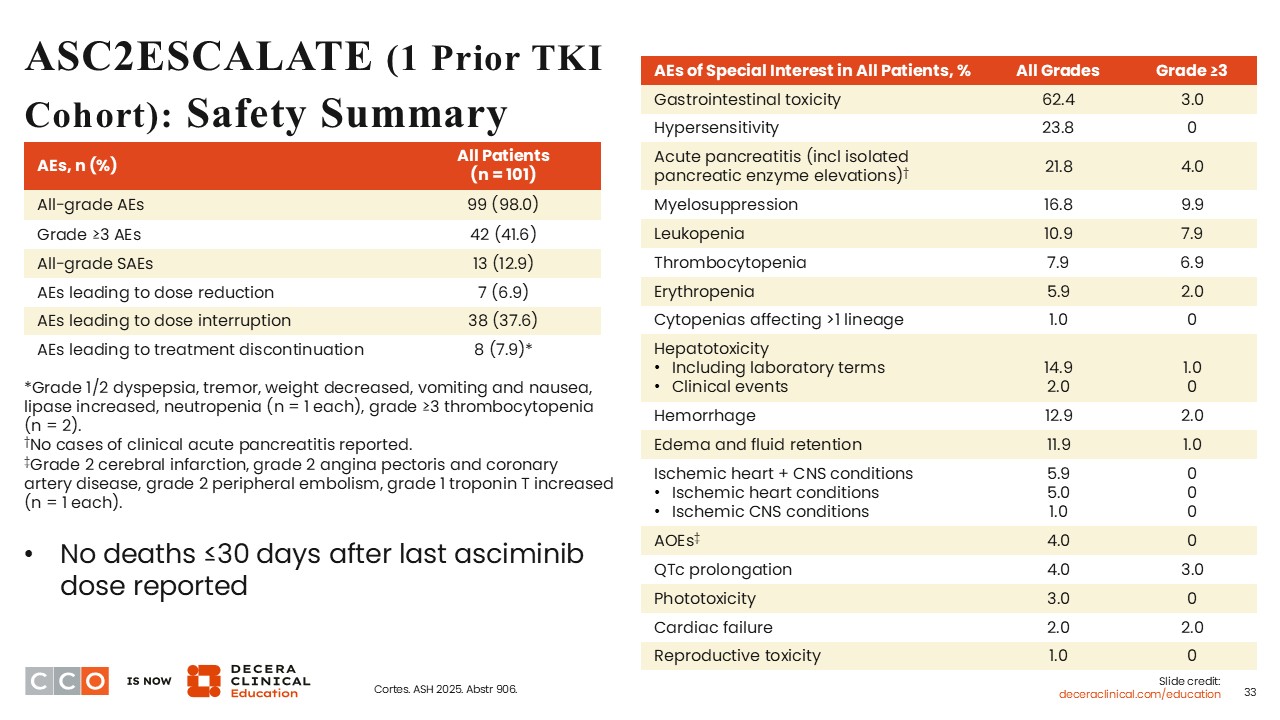
ASC2ESCALATE (1 Prior TKI Cohort): Safety Summary
Courtney D. Dinardo, MD:
Only a few patients (6.9%) required a dose reduction due to AEs. AEs leading to dose interruption occurred in 37.6% of patients, and AEs leading to treatment discontinuation occurred in 7.9%. The AEs of special interest included myelosuppression (16.8%), and of these, 9.9% were grade ≥3. Also, grade ≥3 leukopenia and thrombocytopenia occurred in 7.9% and 6.9%, respectively. This is important because in patients with CP-CML, sustained blood count recovery is important. Other AEs of special interest of any grade reported included GI toxicity (62.4%) and pancreatitis (21.8%).
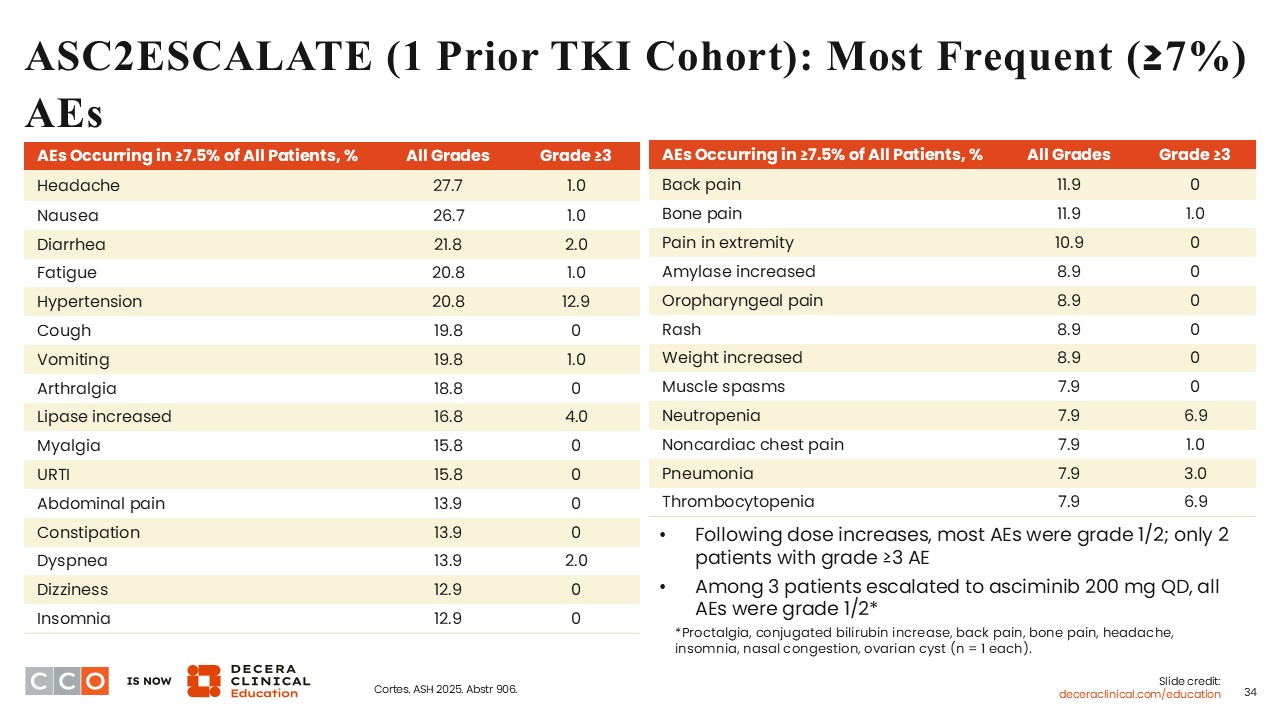
ASC2ESCALATE (1 Prior TKI Cohort): Most Frequent (≥7%) AEs
Courtney D. Dinardo, MD:
The most frequent AEs occurring in ≥7% were primarily grade 1/2, and these included headache, nausea, diarrhea, fatigue, and hypertension.
ASC2ESCALATE: Conclusions and Clinical Implications
Courtney D. Dinardo, MD:
The phase II ASC2ESCALATE met its primary endpoint of achieving MMR at 12 months in patients receiving asciminib monotherapy as second-line treatment after not achieving an optimal response according to ELN 2020 (at 6 and 12 months). Per protocol dose escalation led to an asciminib dose increase in 17.8% of patients. Also, asciminib monotherapy was well tolerated with few discontinuations due to AEs.
Amir T. Fathi, MD:
The goal of this study is to enhance the benefits of the use of asciminib in the second-line setting. Of note, asciminib is already known to be well tolerated and effective in patients with CP-CML. An intriguing part of the study design is that the dose of asciminib was aggressively escalated in an attempt to deepen response when used as second-line therapy after failure of a prior TKI, and the study met its primary endpoint. Hopefully, this allows healthcare professionals to feel more comfortable about potentially increasing the dose of asciminib and achieving deeper responses.
Courtney D. Dinardo, MD:
I have used asciminib in a handful of patients, and I have found it to be overall well tolerated in patients who had trouble tolerating other TKIs. My anecdotal clinical experience mirrors some of the data highlighted in this study.