CE / CME
Expanded Options for Diverse Patients: Individualizing Antibiotics in Acute Bacterial Skin and Skin Structure Infections
Pharmacists: 1.00 contact hour (0.1 CEUs)
Physicians: maximum of 1.00 AMA PRA Category 1 Credit™
Nurse Practitioners/Nurses: 1.00 Nursing contact hour
Released: March 12, 2026
Expiration: September 11, 2026
Activity
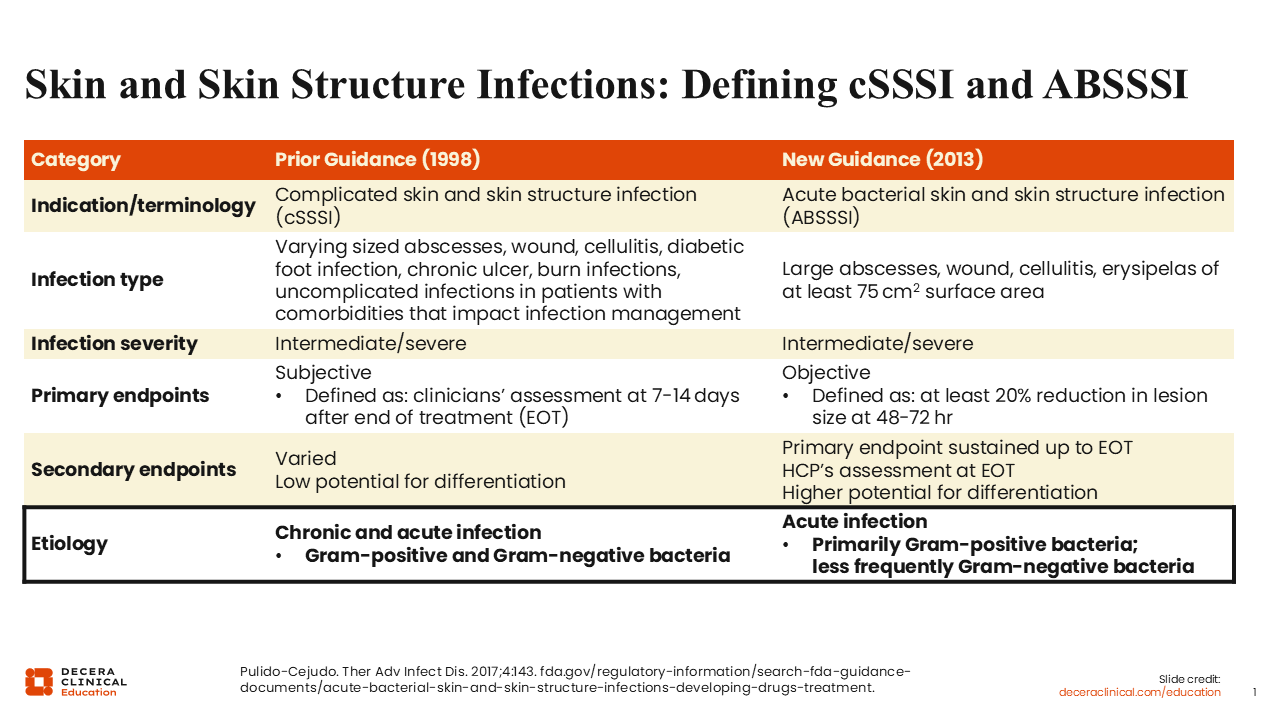
Skin and Skin Structure Infections: Defining cSSSI and ABSSSI
Before we delve into this topic, I want to provide some clarification about the evolving regulatory definitions of skin and skin structure infections that are currently used within the literature. In particular, I want to define complicated skin and skin structure infections (cSSSIs) and ABSSSIs.
These definitions are primarily driven by FDA guidance. Previous guidance focused on cSSSIs, but in 2013, there was a shift in the FDA guidance to a greater focus on ABSSSIs. The definition for cSSSIs previously included a wider variety of infection types, such as abscesses, wounds, cellulitis, diabetic foot, and uncomplicated infections with certain comorbidities that affect infection management.1,2
The new guidance streamlines the types of skin infection included and focuses on abscesses, wounds, cellulitis, and erysipelas. A major distinguishing factor from the previous guidance is that the surface area of the skin infection must be ≥75 cm².1,2
In terms of infection severity, the definition remains the same between the 1998 and 2013 guidelines. Although less important for today's discussion, the 2013 guidelines shifted the primary endpoints used to grant FDA approval for drugs for the treatment of skin infections. With the previous guidance, primary endpoints were based on subjective clinical assessment of response 7-14 days after the end of treatment, whereas secondary endpoints were varied. By contrast, with the new guidance centering on those with ABSSSIs, primary endpoints focus on a more objective acute response, defined as a ≥20% reduction in lesion size within 48-72 hours after initiation of therapy.1,2
In this module, discussion of cSSSIs will encompass acute and chronic presentations caused by both gram-positive and gram-negative infections because of the definition including a wider variety of infections. By contrast, ABSSSIs are acute infections, predominantly caused by gram-positive pathogens, and less frequently gram-negative organisms.1,2
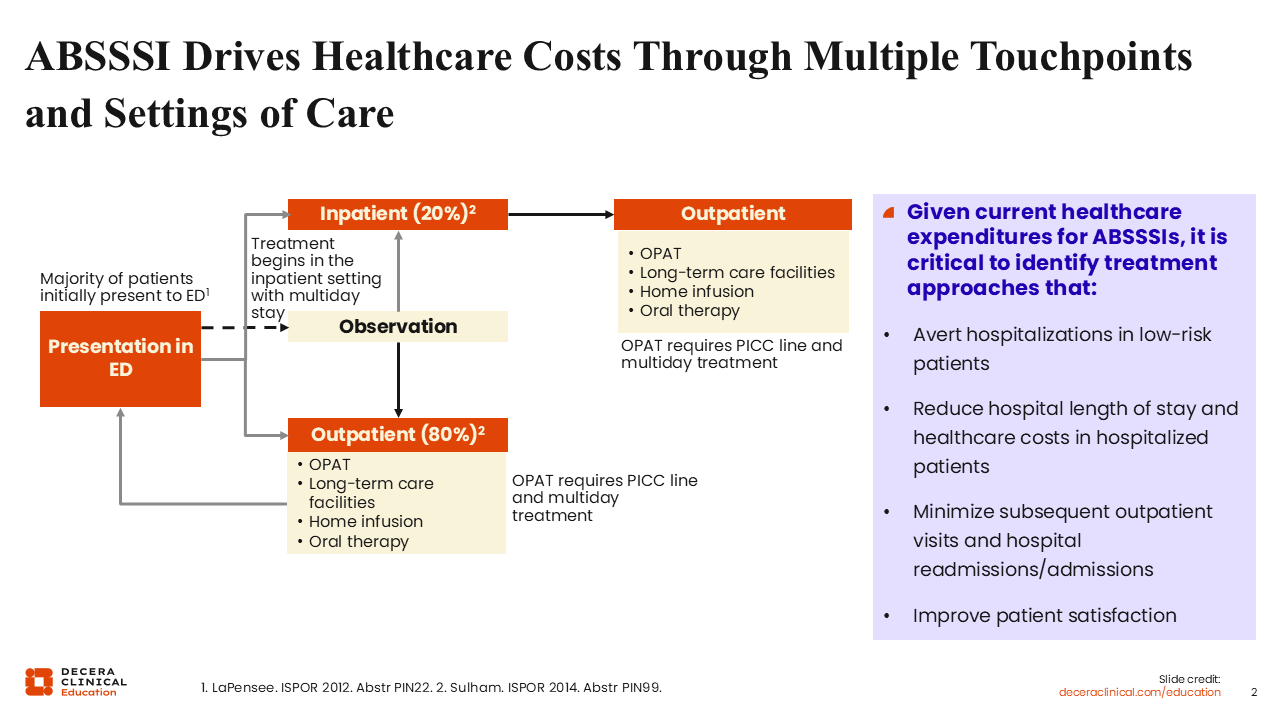
ABSSSI Drives Healthcare Costs Through Multiple Touchpoints and Settings of Care
Most patients with ABSSSI initially present to the emergency department (ED). It is estimated that 3-4 million patients in the United States present annually to the ED with a skin infection.3,4 Most of these cases are appropriately triaged and designated to the outpatient setting for treatment. However, approximately 20% require hospital admission.4,6
At first glance, an 80% outpatient treatment rate may suggest we are managing these infections efficiently. However, if 4 million patients present to the ED with ABSSSI, that still translates to nearly 1 million hospitalizations annually for skin infections in the United States. Furthermore, many patients initially treated in the inpatient setting require additional follow-up care after discharge, adding to overall healthcare utilization.3-6
Given the substantial healthcare expenditures associated with ABSSSI, it is critical to identify strategies that safely avert hospitalization in low-risk patients, reduce length of stay, and minimize downstream outpatient visits and readmissions. Of most importance, healthcare professionals (HCPs) should focus on optimizing clinical outcomes while preserving antimicrobial stewardship and improving patient-centered outcomes, including return to normal activities.
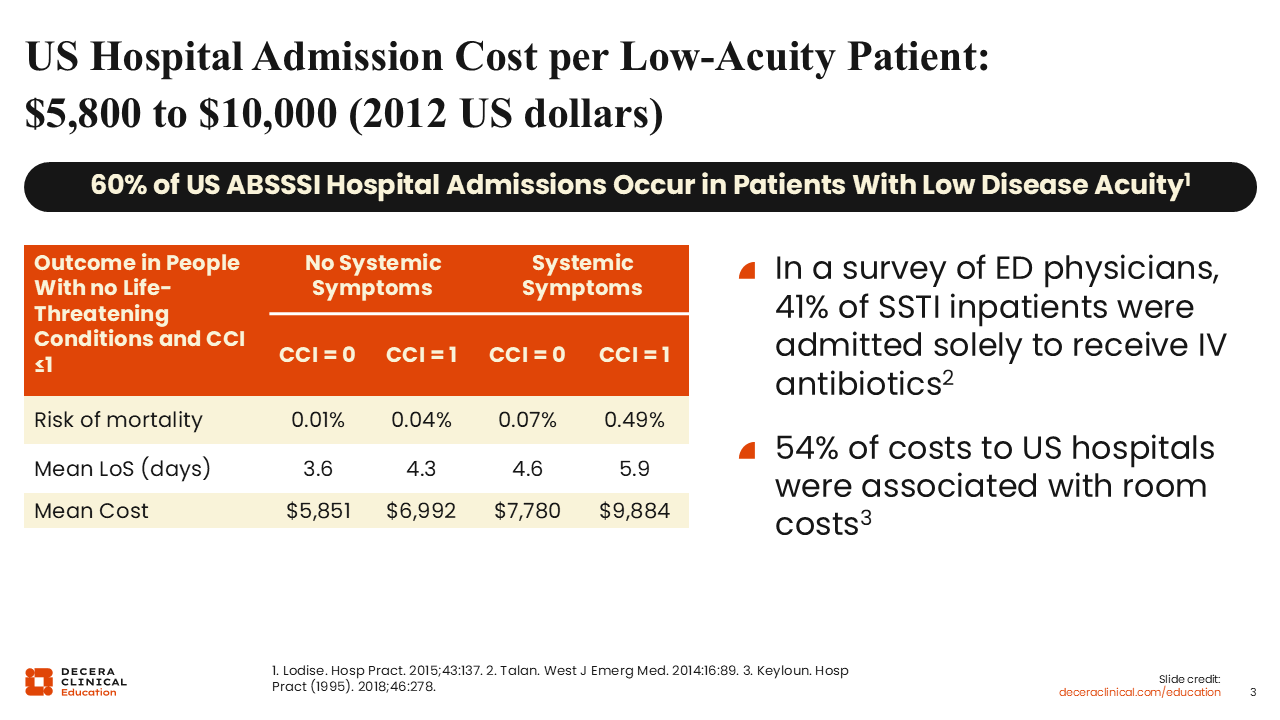
US Hospital Admission Cost per Low Acuity Patient: $5,800-$10,000 (2012 US dollars)
One reason it is so important to avoid unnecessary hospitalization is the substantial economic burden associated with admission. A study I was involved in reported that each US hospital admission in 2012 for ABSSSI costs between $5,800 and $10,000.6
This study also found that most ABSSSI admissions occurred in patients with low disease severity. Most of these patients had no systemic signs or symptoms of infection and 0-1 comorbidity as defined by the Charlson Comorbidity Index. The risk of mortality was extremely low in this population, but the average hospital length of stay was approximately 4 days.6
When we examine what is driving the majority of these hospitalization costs, it is largely room and board. These patients often do not require intensive acute care services. In many cases, the primary reason for hospital admission is to receive intravenous (IV) antibiotics.7,8
This highlights an important opportunity: Identifying patients who can safely receive effective outpatient therapy may substantially reduce healthcare expenditures without compromising clinical outcomes.
Risk Stratification in the ED
How can we improve the quality and efficiency of care for patients who present to the ED with skin infections?
Risk stratification is of paramount importance. First, address the fundamental clinical issues9:
- Assess for hemodynamic instability
- Evaluate for a potential deep-seated infection, including necrotizing fasciitis
- Determine whether surgical intervention is required
- Assess for unstable comorbidities or social factors that may interfere with successful outpatient management
If the answer to all of these is no, HCPs can begin considering appropriate outpatient treatment strategies9:
- Use of oral antibiotics
- Once-daily parental antibiotics given in the ED with follow-up for additional dosing
- Outpatient IV antibiotics
- Single-dose or extended half-life IV agents that may facilitate discharge without the need for prolonged outpatient parenteral antimicrobial therapy (OPAT)
The key principle is aligning treatment intensity with illness severity and patient-specific risk factors.
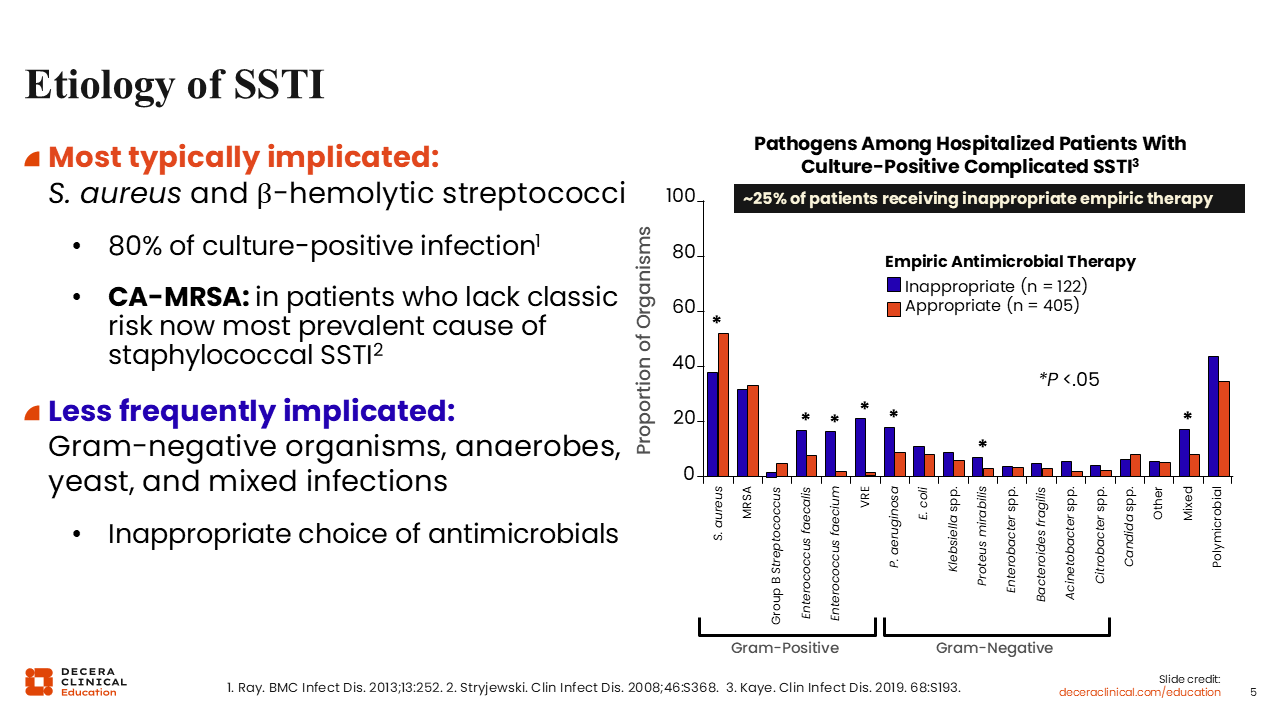
Etiology of SSTI
Before we get into different treatment options, I think it is important to understand what is driving these infections. What is the most common etiology? Despite shifts in healthcare delivery and resistance patterns, the predominant pathogens in SSTIs remain Staphylococcus aureus and β-hemolytic streptococci, based on culture-positive data.10
HCPs must recognize that, particularly in patients presenting with purulent SSTIs, community-acquired methicillin-resistant S. aureus (MRSA) has become the most prevalent cause of staphylococcal infection, even among patients who lack traditional MRSA risk factors.11 Gram-negative organisms and anaerobes are less frequently implicated but may be relevant in specific clinical contexts.
Empiric therapy remains critically important. Among hospitalized patients with culture-positive cSSTIs, approximately 1 in 4 patients receive inappropriate empiric therapy, despite HCPs’ best efforts to identify likely pathogens.
When treatment appropriateness is stratified by pathogen, infections caused by Staphylococcus and Streptococcus species are more often treated appropriately. However, higher rates of inappropriate therapy are observed in infections caused by MRSA, Enterococcus species, and certain gram-negative organisms, particularly in the setting of mixed or polymicrobial infections.12,13
Microbiology of Common SSTI Pathogens
When considering common skin and soft tissue infection (SSTI) pathogens, the 2 that consistently predominate are β-hemolytic streptococci and S. aureus.14
If a patient presents with cellulitis, erysipelas, or a nonpurulent SSTI, the most likely etiology is β-hemolytic streptococci, most commonly Streptococcus pyogenes.10,12,14
By contrast, purulent infections are most commonly caused by S. aureus, and source control through incision and drainage is often required.10,12,14
Common Risk Factors for Initial and Recurrent ABSSSI
Common risk factors for initial and recurrent infections include immunocompromising conditions and significant comorbidities, particularly in patients at the extremes of age (very young or old individuals). Persons who inject drugs are also a vulnerable population. In addition, otherwise healthy individuals who participate in contact sports or reside in crowded living conditions are at increased risk for ABSSSI.
At the individual level, important considerations include:
- History of MRSA colonization
- Prior SSTI
- Recent or frequent hospitalization
- Prior antibiotic exposure
Patients are also at increased risk when there is compromise of skin integrity, which serves as a portal of bacterial entry and subsequent infection.15-17
Recognition of these risk factors should inform empiric antibiotic selection and decisions regarding MRSA coverage.
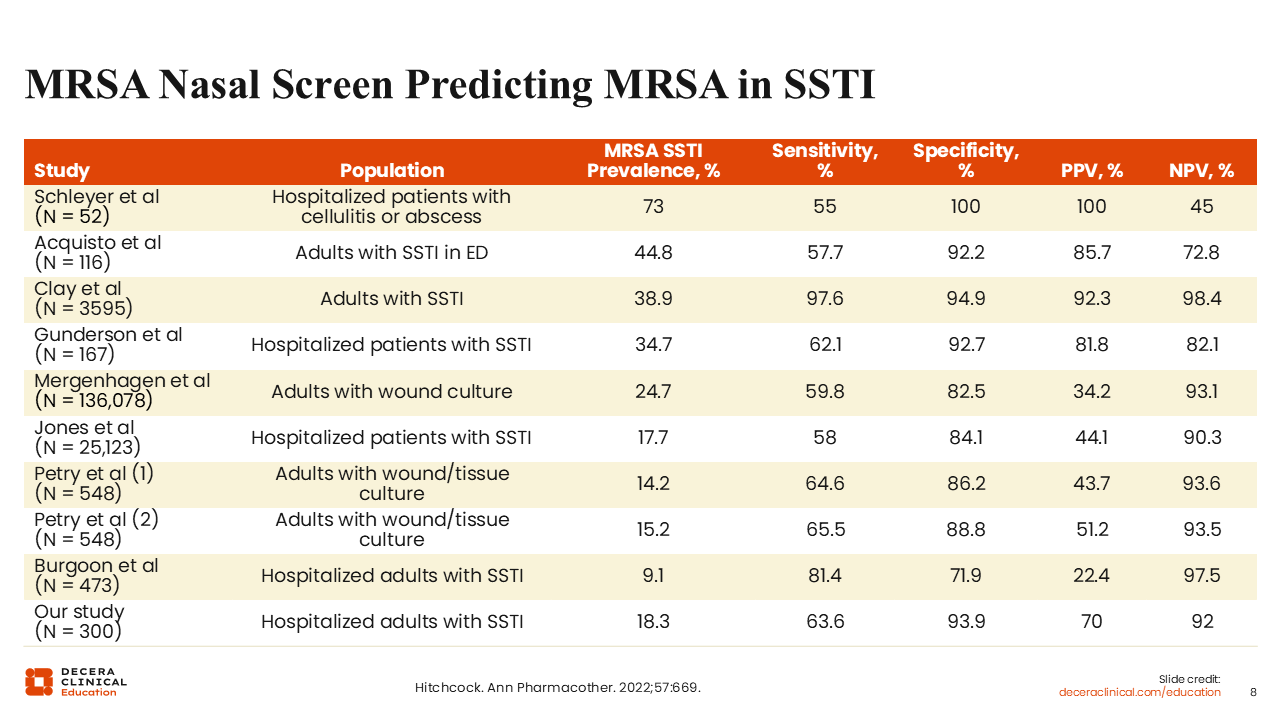
MRSA Nasal Screen Predicting MRSA in SSTI
There a few things that HCPs can do right at the outset for someone who presents with an SSTI. I find that an easy thing to do is an MRSA nasal screening test. This test has been evaluated across many studies in the literature, and although its sensitivity is not very high, it is very specific, with rates >80% in most cases.18
Patients Hospitalized With Serious Skin Infections: Some Involve More Than 1 Pathogen
In certain cases, particularly among hospitalized patients with more severe skin infections, polymicrobial involvement may occur.
Mixed infections often include S. aureus or β-hemolytic streptococci in combination with gram-negative organisms. Common additional pathogens include Enterobacterales and nonlactose-fermenting gram-negative bacilli.19
Recognition of patient-specific risk factors, infection severity, and anatomic location is essential when considering the possibility of polymicrobial infection and determining whether broader empiric coverage is warranted.
Gram-Positive Pathogens: Not the Only Considerations in ABSSSI
Predisposing factors for ABSSSI, particularly for polymicrobial infections or infections involving gram-negative pathogens, can be grouped into 3 broad categories:
- Host-related and clinical factors, including significant comorbid conditions, chronic wounds, immobility, or debilitation.
- Healthcare exposure and prior antibiotic use: Recent hospitalization or prior antimicrobial therapy increases the risk of recurrent infection and raises the likelihood of polymicrobial or gram-negative involvement.
- Anatomic considerations: Infections below the waist, in the perineal region, or those associated with trauma or bite wounds warrant heightened awareness for gram-negative pathogens and anaerobes.12,20-22
Recognizing these factors helps guide empiric therapy beyond routine gram-positive coverage when clinically appropriate.
Diagnostic Challenges in ABSSSI
A key consideration when evaluating patients with ABSSSI is identifying the most likely pathogen based on clinical presentation and patient-specific risk factors.
However, diagnostic uncertainty is common. In many cases, a causative pathogen is not identified.
Superficial wound cultures are generally discouraged, as they frequently reflect colonizing organisms rather than the true pathogen. Blood cultures are often negative, and even cultures obtained from purulent material may not reliably represent deeper tissue involvement.16,23
More invasive diagnostic methods, such as needle aspiration or punch biopsy cultures, have limited yield. At best, these techniques may identify a pathogen in up to 40% and 30% of cases, respectively.24
As a result, management of ABSSSI frequently relies on clinical judgment, local epidemiology, and individualized risk assessment rather than microbiologic confirmation alone.