CE / CME
Expanded Options for Diverse Patients: Individualizing Antibiotics in Acute Bacterial Skin and Skin Structure Infections
Pharmacists: 1.00 contact hour (0.1 CEUs)
Physicians: maximum of 1.00 AMA PRA Category 1 Credit™
Nurse Practitioners/Nurses: 1.00 Nursing contact hour
Released: March 12, 2026
Expiration: September 11, 2026
Activity
Long-Acting Lipoglycopeptides
The long-acting lipoglycopeptides, dalbavancin and oritavancin, disrupt peptidoglycan, similar to vancomycin’s method of action. Oritavancin also has some transpeptidation activity and membrane disruption. It is more bactericidal, at least in the laboratory.55,56
These agents are administered as a single infusion, which can potentially streamline outpatient therapy by eliminating the need for OPAT and avoid hospitalization in patients with soft tissue infections.55,56
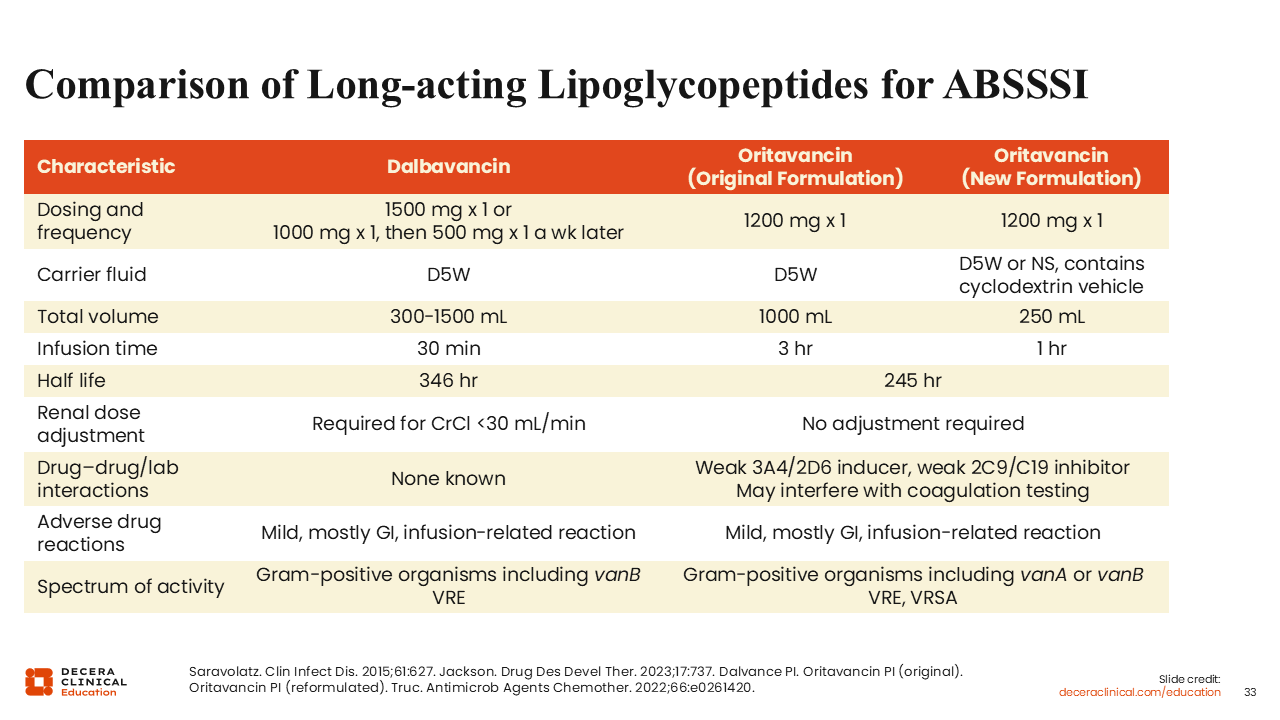
Comparison of Long-Acting Lipoglycopeptides for ABSSSI
Dalbavancin and oritavancin obtained FDA approval approximately at the same time. The original indication for dalbavancin was a 1000-mg loading dose, followed by 500 mg 1 week later. That has been largely replaced because of a follow-up trial and indication. Now, dalbavancin is administered in a dose of 1500 mg at once over a 30-minute infusion.55-58
The original formulation of oritavancin was a 3-hour infusion, which was restrictive for a lot of infusion centers. When both of these came to market at the same time, I think there was a predominance toward dalbavancin because the logistics of a 30-minute infusion were more feasible than a 3-hour infusion.55-58
Oritavancin has since come out with a new formulation. It is the same dose but over a 1-hour infusion, using a cyclodextrin vehicle.55-58
These agents are advantageous because they are one and done, with comparable efficacy to 7-10 days of a glycopeptide in the hospital.55-58
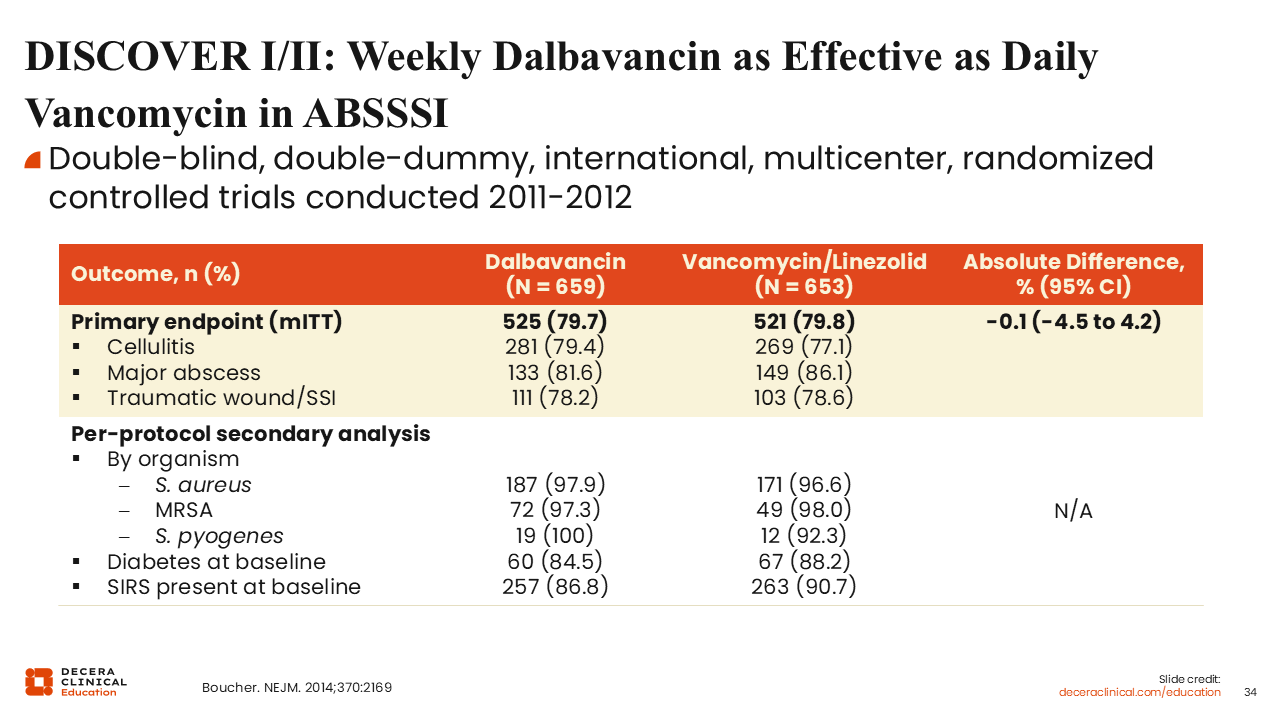
DISCOVER I/II: Weekly Dalbavancin as Effective as Daily Vancomycin in ABSSSI
The DISCOVER-I and II trials compared dalbavancin with vancomycin followed by linezolid therapy. Based on the types of infections—cellulitis, major abscesses, and wound infections—and broken down by pathogen, dalbavancin proved to be noninferior to vancomycin.59
DISCOVER I/II and DUR001-303: Dalbavancin Safety vs Vancomycin in ABSSSI
A post hoc safety analysis revealed that treatment-related adverse events, including nephrotoxicity, were significantly lower with dalbavancin. The frequency of nephrotoxicity was less than one half with dalbavancin compared with vancomycin.60
SOLO I/II: Oritavancin as Effective as Vancomycin for ABSSSI
Similarly, the SOLO trials examined oritavancin vs vancomycin for the treatment of ABSSSI and resulted in FDA approval for oritavancin for ABSSSI in 2014.56,61,62
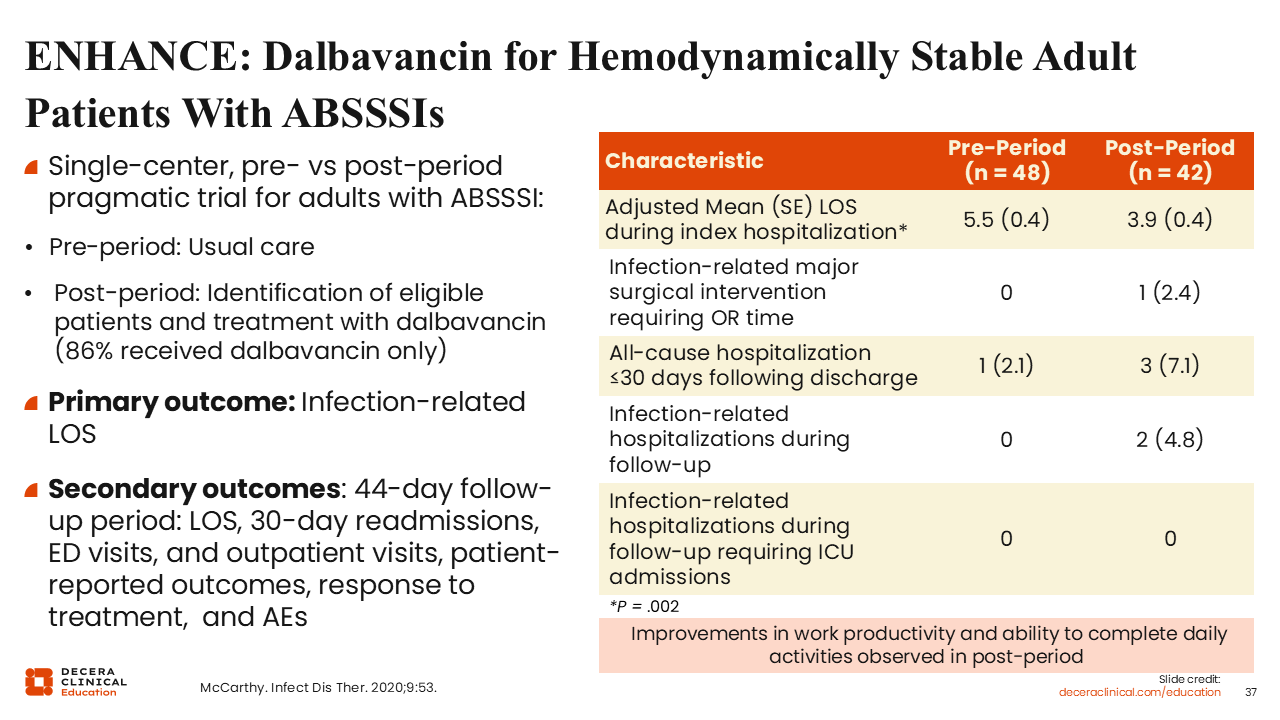
ENHANCE: Dalbavancin for Hemodynamically Stable Adult Patients With ABSSSIs
Although randomized trials demonstrate strong efficacy, an important question is whether these results translate into meaningful real-world clinical outcomes.
To assess the real-world effectiveness of dalbavancin, the ENHANCE study evaluated its use in hemodynamically stable hospitalized adults with ABSSSIs to determine whether it could facilitate earlier discharge. These were patients who otherwise would have required several additional days of inpatient IV therapy but were clinically stable and appropriate for transition of care.63
The ENHANCE study was a single-center, preimplementation and postimplementation pragmatic trial. The primary outcome was hospital length of stay, and secondary outcomes evaluated whether use of a single-dose long-acting agent was associated with an increased risk of adverse clinical outcomes after discharge.63
Implementation of dalbavancin was associated with a significant reduction in mean hospital length of stay compared with standard-of-care therapy. Of importance, there were no statistically significant differences in infection-related readmissions or other adverse outcomes, although there was a numerically higher rate of all-cause readmissions that did not reach statistical significance in the dalbavancin group.63
These findings suggest that, in carefully selected, clinically stable patients, dalbavancin may support earlier discharge without evidence of increased short-term adverse clinical outcome.
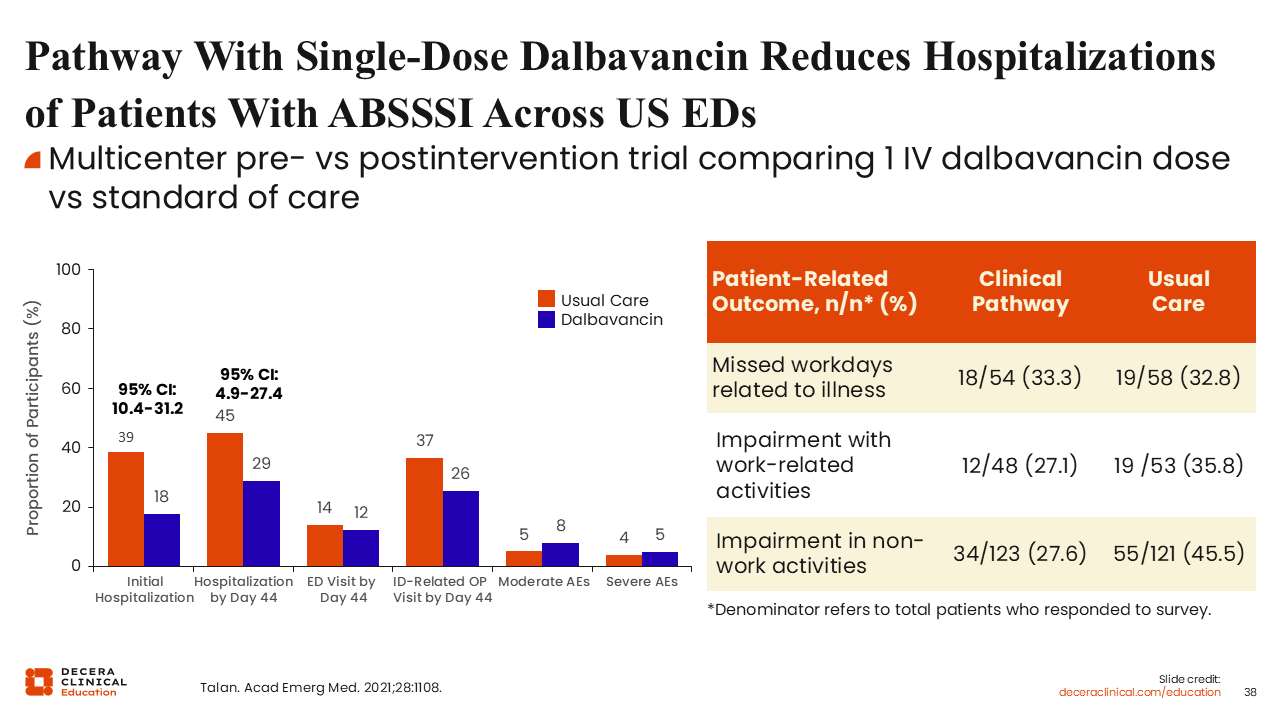
Pathway With Single-Dose Dalbavancin Reduces Hospitalizations of ABSSSI Patients Across US EDs
Another question that often arises with long-acting agents is whether use of a single-dose antibiotic, when administered in the ED to facilitate discharge and avoid hospitalization, places patients at increased risk for subsequent healthcare utilization or recurrent infection.
This study sought to answer that question. The investigators evaluated implementation of a clinical pathway incorporating single-dose dalbavancin for hemodynamically stable patients presenting to the ED with ABSSSI who would otherwise have required hospital admission for IV therapy.64 The study was a multicenter, pragmatic evaluation conducted across multiple US EDs. Eligible patients were clinically stable, did not require surgical intervention, and were appropriate for outpatient management with reliable follow-up.
Investigators reported a reduction in initial hospitalizations from 39% with standard-of-care therapy to 18% with dalbavancin. Of importance, there was no increase in subsequent hospitalizations through Day 44, ED visits through Day 44, or infection-related outpatient visits, despite administration of a single-dose regimen.64
Another concern has been whether adverse events associated with a long-acting agent might be prolonged because of its extended half-life. The study demonstrated similar rates of moderate and severe adverse events between dalbavancin and standard-of-care therapy. Consistent with prior clinical trials, adverse events observed with dalbavancin were not associated with prolonged duration compared with standard-of-care therapy.64
The investigators also evaluated patient-reported outcomes. Although patients reported similar numbers of missed workdays in both groups, those treated with dalbavancin reported less impairment in work-related activities, suggesting potential quality-of-life benefits in appropriately selected patients.64
Early Directed Oritavancin in the ED
There are similar real-world data evaluating early directed oritavancin use in the ED. This is one of several studies assessing the implementation of oritavancin in this setting.65
Consistent with findings from other pathway-based studies, this analysis demonstrated a substantial reduction in hospital length of stay with oritavancin compared with standard-of-care therapy, with a mean reduction of approximately 60 hours. Investigators also evaluated whether discharge after a single dose was associated with worse downstream outcomes. The data showed similar rates of 30-day readmissions and recurrent abscesses between the oritavancin and standard-of-care groups.65
Even in a small community hospital, this pilot program of 122 patients demonstrated measurable operational and financial impact. Although oritavancin has a higher drug acquisition cost relative to some standard-of-care agents, the study reported a reduction of more than 200 hospital bed days, corresponding to approximately $310,000 in institutional cost avoidance at the time of analysis.65
In capitated reimbursement environments, hospitalizations for SSTIs may exceed reimbursement. Therefore, reducing the length of stay when clinically appropriate can be financially relevant to institutions, and early use of long-acting IV therapy may represent one strategy to support safe discharge in carefully selected patients.
Future Directions
In optimizing care of skin infections, a useful rule of thumb is that nonpurulent infections are most commonly caused by β-hemolytic streptococci, whereas purulent infections are most often because of S. aureus, including MRSA, depending on risk factors.
Gram-negative or polymicrobial infections are typically a concern in specific populations and anatomic locations, particularly infections below the waist, in the perineal region, or in patients with significant chronic comorbidities.
Historically, many patients with ABSSSI have been treated with IV antibiotics, but IV therapy carries risk. Beyond adherence challenges with OPAT, catheter-related complications and thrombotic events may occur.
Fortunately, multiple oral agents are now available that can facilitate early discharge or, in appropriate patients, avoid hospitalization altogether.
Agent selection depends on the likely pathogens, risk of MRSA, potential for polymicrobial or gram-negative involvement, and local resistance patterns. Of note, some agents with strong MRSA activity have limited activity against β-hemolytic streptococci, which is important given the diagnostic uncertainty that often accompanies SSTIs. Treatment decisions should also incorporate patient comorbidities, tolerability, and cost.
With an estimated 4-5 million ED visits annually for skin infections in the United States, data-driven institutional protocols are essential. A critical decision point is whether hospitalization is necessary. Although most patients are managed as outpatients, approximately 1 million hospital admissions annually are attributed to skin infections.
Strong antimicrobial stewardship programs, therefore, are essential to ensure appropriate agent selection and duration. In select cases, agents with higher acquisition costs may be justified if they improve efficiency of care, reduce length of stay, and provide appropriate spectrum coverage.