CE / CME
Expanded Options for Diverse Patients: Individualizing Antibiotics in Acute Bacterial Skin and Skin Structure Infections
Pharmacists: 1.00 contact hour (0.1 CEUs)
Physicians: maximum of 1.00 AMA PRA Category 1 Credit™
Nurse Practitioners/Nurses: 1.00 Nursing contact hour
Released: March 12, 2026
Expiration: September 11, 2026
Activity
Choice of Therapy for ABSSSI
Because culture data are often unavailable or insufficient for diagnosis, empiric treatment decisions should be guided by the most likely pathogens based on clinical presentation and regional resistance patterns.23,24
Often, this means making a best guess as to what the causative pathogens are. Gram-positive organisms, Staphylococci and Streptococci, predominate, but HCPs must understand the probability of other organisms appearing as well.23,24
As an HCP, I have seen 2 patients in the past 48 hours with skin infections caused by different pathogens. Case one was a 40-year-old patient with no comorbidities in the ED. The patient was diagnosed with a Staphylococci infection. The second patient presented with red, inflamed skin after a vascular surgical procedure, and the culture grew Pseudomonas. Therefore, depending on the circumstance and the setting, HCPs need to make empiric treatment decisions based on probability, and not necessarily think Staphylococci and Streptococci all the time.
I like to think of choosing an antibiotic or treating any infection as similar to stopping a car, in that there are many factors to consider when trying to come to a complete halt. How fast the car is going is similar to illness severity, and the road surface, whether you are on smooth pavement or an icy downhill road, is a metaphor for comorbidities. As a driver must consider all these factors when applying brakes, HCPs also have to factor in comorbidities, risk factors, and how sick the patient is when making treatment decisions.
IDSA Treatment Recommendations: Purulent and Nonpurulent SSTIs
The Infectious Diseases Society of America (IDSA) practice guidelines for the management of SSTIs break them down in 2 major categories: nonpurulent or purulent.23
Nonpurulent SSTIs are predominantly caused by the β‑hemolytic Streptococci whereas purulent infections are predominantly S. aureus. Treatment selection is based on those key features.23
The second decision-making component according to the guidelines is how sick the patient is. That will determine whether the patient will be seen in the ED and sent home, or admitted for observation, then sent home, or fully admitted for inpatient treatment.23
With a purulent infection, source control predominates. All of these patients generally need an incision and drainage procedure or, at least, a drainage of the abscess, packing of the abscess, and subsequent antibiotic therapy. With a nonpurulent infection, however, the guidelines generally recommend going straight to antibiotic therapy.23
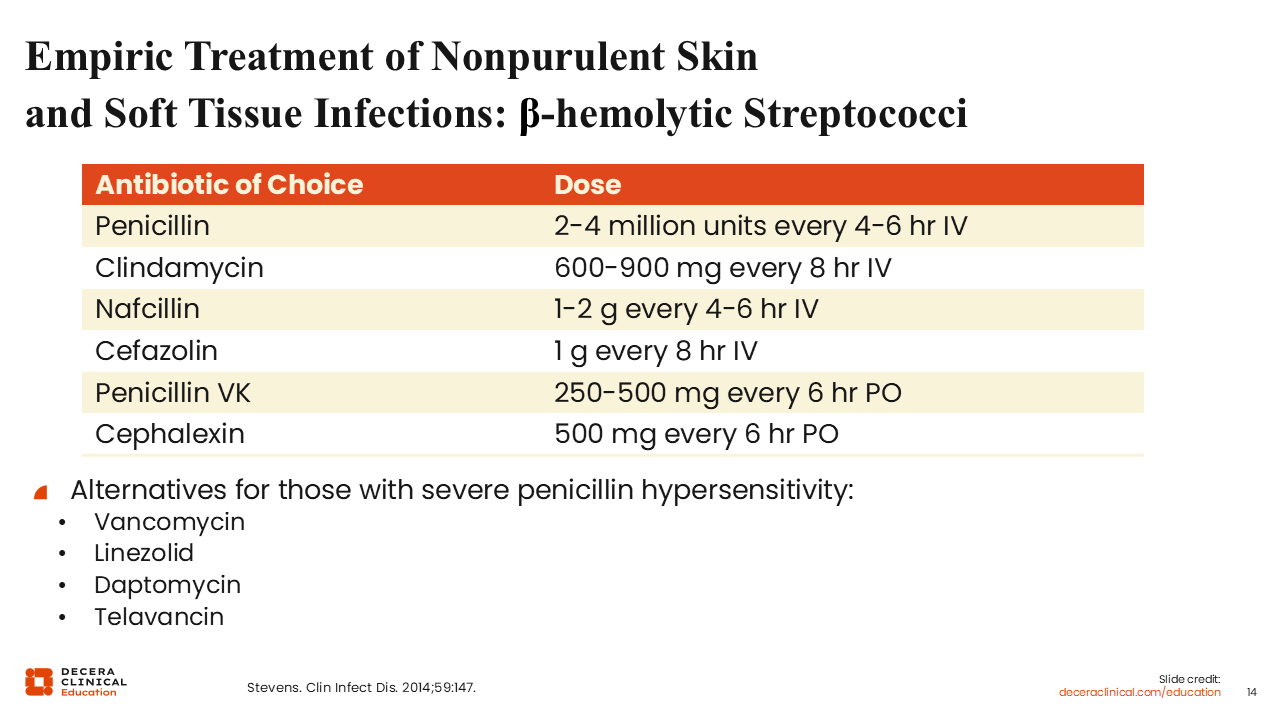
Empiric Treatment of Nonpurulent Skin and Soft Tissue Infections: β-Hemolytic Streptococci
Treatment for nonpurulent infections should antibiotics for β-hemolytic Streptococci. The first treatments that come to mind may be penicillin, nafcillin, cefazolin, cephalosporins, oral penicillin V potassium, or cephalexin.23
In the real world, penicillin is not generally used, although I was more cavalier in my early practice days. For example, if someone had a lymphedema from a previous lymph node dissection and developed shaking chills and then a few hours later presented with a red, inflamed arm, I would be very confident that was a classic group-based Strep infection. So, I might use penicillin in that instance. The downside is that penicillin would not cover any potential Staphylococci if that happens to be present. For patients with severe penicillin allergies, clindamycin and linezolid or IV vancomycin and daptomycin are good alternatives.23
Empiric Treatment of Purulent SSTIs: MSSA
For purulent infections, we must shift our concerns over to Staphylococci. When treating empirically, whether in the inpatient or outpatient setting, it is increasingly difficult to justify immediate use of β-lactam alone, without culture data. Agents like nafcillin and cefazolin do not provide coverage for MRSA. Although MRSA rates have dropped dramatically in the past 10 years, it is still challenging to feel confident prescribing cephalosporin or penicillin empirically for a purulent infection without confirmed susceptibility data.23
That said, there are options. Clindamycin is an interesting choice there, but its utility depends on local susceptibility. For example, where I practice in San Diego, California, methicillin-susceptible S. aureus (MSSAs) and MRSAs are largely clindamycin susceptible. Therefore, I would feel pretty comfortable giving clindamycin empirically to a patient with a purulent infection. However, this susceptibility profile is not consistent nationwide, so HCPs must be familiar with their local antibiogram before selecting it.23
Other options include the tetracyclines, such as doxycycline and minocycline. On paper, TMP/SMX is also a reasonable option, although in the real world, patients treated with TMP/SMX have higher relapse rates compared with other agents.23
Empiric Treatment of Purulent SSTIs: MRSA
When MRSA is a concern, TMP/SMX and doxycycline remain appropriate oral treatment options in many cases. However, rather than β-lactams and cephalosporins, other treatment options include vancomycin, linezolid, daptomycin, and clindamycin.23
Then there are the anti-MRSA cephalosporins. In the United States, ceftaroline was the only cephalosporin with MRSA activity from approximately 2011-2012 onward. In 2024, ceftobiprole became available as a second option. These 2 options are similar in terms of their anti-MRSA activity, but differences in dosage and infusion times must be considered when choosing between them.23,25
The Yin and Yang of Antibiotics
Next, I delve into the nuances of antibiotic prescribing. When prescribing antibiotics, HCPs often think of it in black and white terms: susceptible or resistant. However, there is more at play.
Antibiotics are complicated compounds. Different antibiotic classes interact with our immune system in different ways, antagonizing or synergizing with distinct components of our immune system. Different antibiotics also exert disparate effects on bacterial physiology that are hard to capture in clinical trials but can profoundly affect patient outcomes.23
For example, HCPs tend to reach for bactericidal therapy, the β-lactam antibiotics, in the presence of high-inoculum infections. However, in most soft tissue infections, the bacterial inoculum of infection tends to be quite low; it often falls below the level of detection in nonpurulent infections.23
The only infections in the skin with high bacterial burden are abscesses, where management is primarily a source control issue. Incision and drainage reduce the inoculum far more than antibiotics alone. In this context, antibiotics serve as adjunctive therapy rather than primary intervention. This highlights an important distinction: Treating a soft tissue infection is very different from treating a high-inoculum infection. Although β-lactams are appealing because they are “cidal” and act rapidly, their role in many soft tissue infections may be less central than traditionally assumed. Furthermore, their bactericidal effect can induce toxin production and cause further inflammation, which could be disadvantageous for certain patients.23
In contrast to the “cidal” antibiotics, there are drugs that are more bacteriostatic that inhibit bacterial growth and reduce toxin production. These are ribosomal agents, such as clindamycin, the oxazolidinones, tetracyclines, and macrolides. Although some HCPs hesitate to use them because they are not as potent for bacterial killing, I find them quite favorable and effective for certain patients, particularly those for whom reduction of bacterial toxins and inflammation is a high priority.23
Where does vancomycin fall on this spectrum? It does not have rapid bactericidal activity, nor does it reduce bacterial toxin production. I think, mechanistically, vancomycin looks like a pretty inefficient agent. In general, I think vancomycin and other glycopeptide agents are most useful in outpatient settings where convenience and adherence are priorities. In hospitalized patients, however, where rapid clinical response is often desired, vancomycin’s slower bactericidal activity and lack of toxin suppression may limit its efficiency.23
In short, antibiotic selection is not simply a matter of susceptibility. It requires consideration of bacterial burden, toxin production, immune interactions, pharmacodynamics, and the clinical context in which the drug is being used.
Adverse Drug Events: Common With Outpatient Parenteral Antibiotics
In my experience, HCPs tend to favor IV antibiotic therapy, particularly for the advantage of increased treatment adherence and compliance. However, there can be consequences when committing patients to OPAT.
Adverse drug events are not uncommon with OPAT, and it is accompanied with risks of thromboembolic events and central line–associated bloodstream infections. Although line infections are not particularly common, they still occur. There is also a fairly high rate of ED visits while on OPAT. The rate of readmission on OPAT is also considerable. In my experience, risk factors for readmission while on OPAT include obesity and polymicrobial infection, although higher readmission rates with OPAT may also reflect greater disease severity at initiation and a higher overall burden of comorbidities compared with patients who initiate oral therapy. Regardless, OPAT is not without challenges and is not always a seamless option for many patients.26
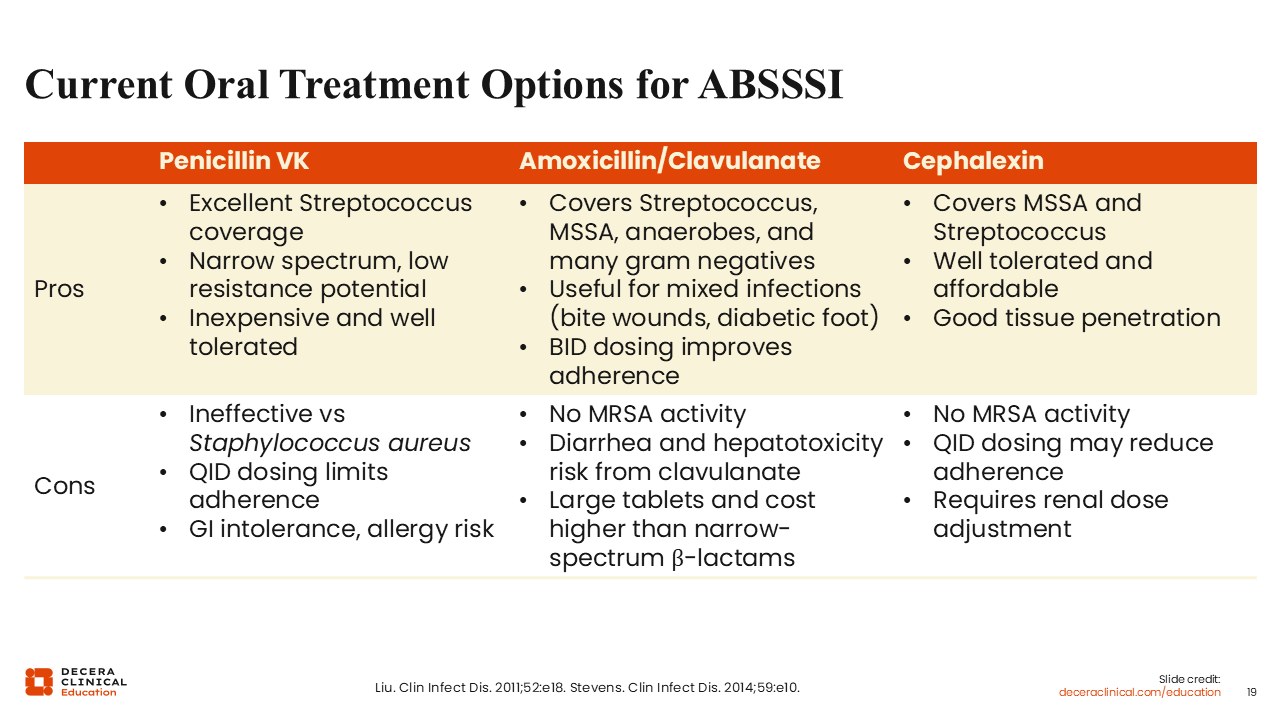
Current Oral Treatment Options for ABSSSI
Ultimately, I believe the treatment landscape is changing to favor oral options whenever possible.
In terms of oral treatment options for ABSSSI, there are 3 primary options. I only use penicillin for cases of classic Streptococcal infection: lymphedema, shaking chills, followed by skin redness, and only if I have the ability to monitor the patient during treatment. I would be hard-pressed to give oral penicillin to an outpatient with a soft tissue infection, but it remains in the guidelines as a recommended treatment.23,27
I would be more likely to reach for amoxicillin/clavulanate or a cephalexin to cover MSSA and Strep, particularly when treating ABSSSI empirically and if treating a nonpurulent infection. When treating a purulent infection, I would certainly want to augment these agents with an anti-MRSA agent, unless I had cultures identified.23,27
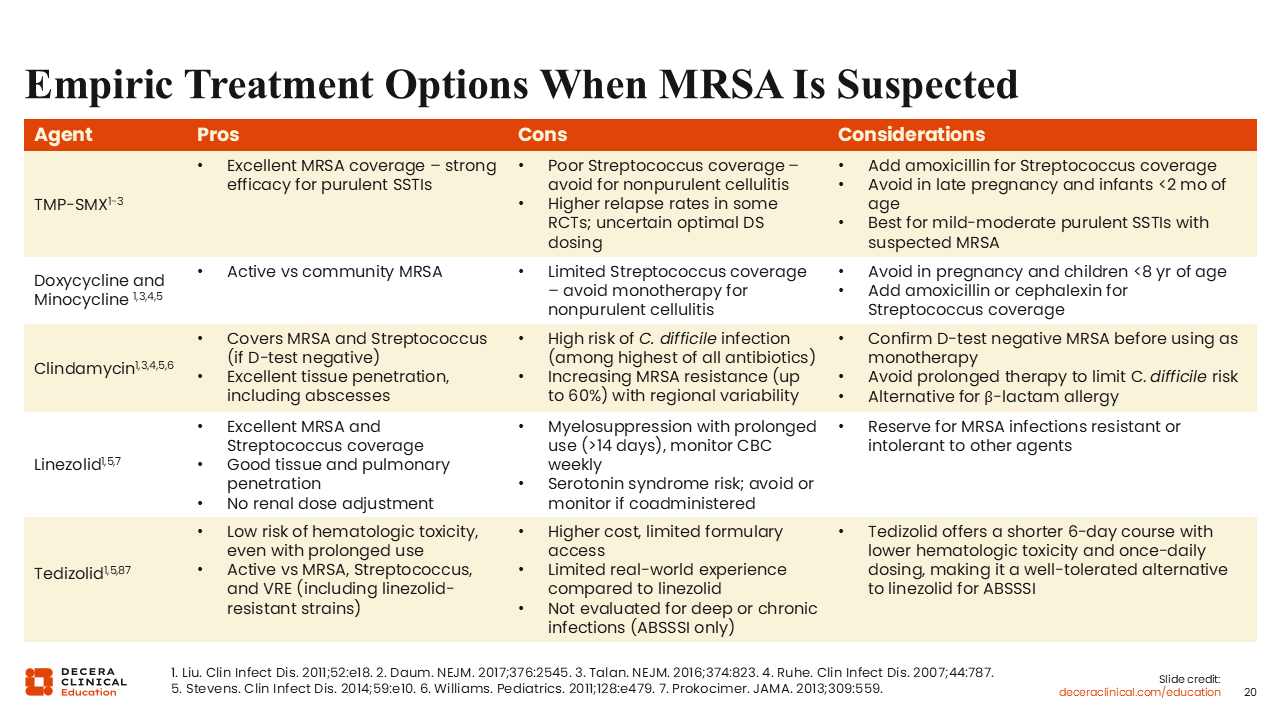
Empiric Treatment Options When MRSA Is Suspected
When MRSA is suspected, I would either add one of the following agents or perhaps use them first-line.
TMP/SMX is a good option because it has enhanced staphylococcal activity. However, even though the double-strength formulation is commonly prescribed, the question remains whether that dose is sufficient. In patients undergoing incision and drainage of an abscess, we should also ask whether adjunctive antibiotic therapy is necessary at all. Is source control alone enough? If a patient improves after debridement followed by TMP/SMX, I think it is fair to question how much of the clinical response is attributable to the antibiotic vs the drainage by itself.27,28,29
Beyond those considerations, I have additional reservations about TMP/SMX related to tolerability, particularly in older patients who are often receiving multiple concomitant medications. The potential for drug interactions and adverse effects in this population further tempers my enthusiasm for this agent in certain settings.
Doxycycline or minocycline are also good choices, although minocycline tends to be a little bit more active in vitro. The clinical relevance of this difference is not known. In a mixed infection with more resistant gram negatives, minocycline does offer broader coverage.23,27,29
There is also clindamycin, linezolid, and tedizolid. Linezolid can be associated with myelosuppression after prolonged use, but this is generally not a consideration when treating ABSSSI because patients are usually treated for 10 days or less. The greater concern, I think, is serotonin syndrome among people receiving selective serotonin reuptake inhibitors (SSRIs) as a result of monoamine oxidase inhibition. This is a significant concern because approximately 20% of the US population is receiving an SSRI. Personally, I do not use linezolid for patients receiving SSRIs, although these agents can be coadministered with close monitoring, especially in an inpatient setting.23,27,29-31
Why Clindamycin?
As alluded to earlier, some ribosomal agents, like clindamycin, possess toxin-inhibitory effects that are highly relevant for more severe forms of soft tissue infections, like necrotizing fasciitis. I typically would prescribe a β-lactam antibiotic, like penicillin, ceftriaxone, or another cephalosporin, along with clindamycin. The β-lactam provides reliable bactericidal activity, whereas clindamycin inhibits bacterial toxins.32,33
The addition of clindamycin has shown mortality benefit at the clinical trial level for invasive group A Strep. As a result, most HCPs today would will routinely use clindamycin to supplement the primary β-lactam therapy in group A Strep necrotizing infection.32
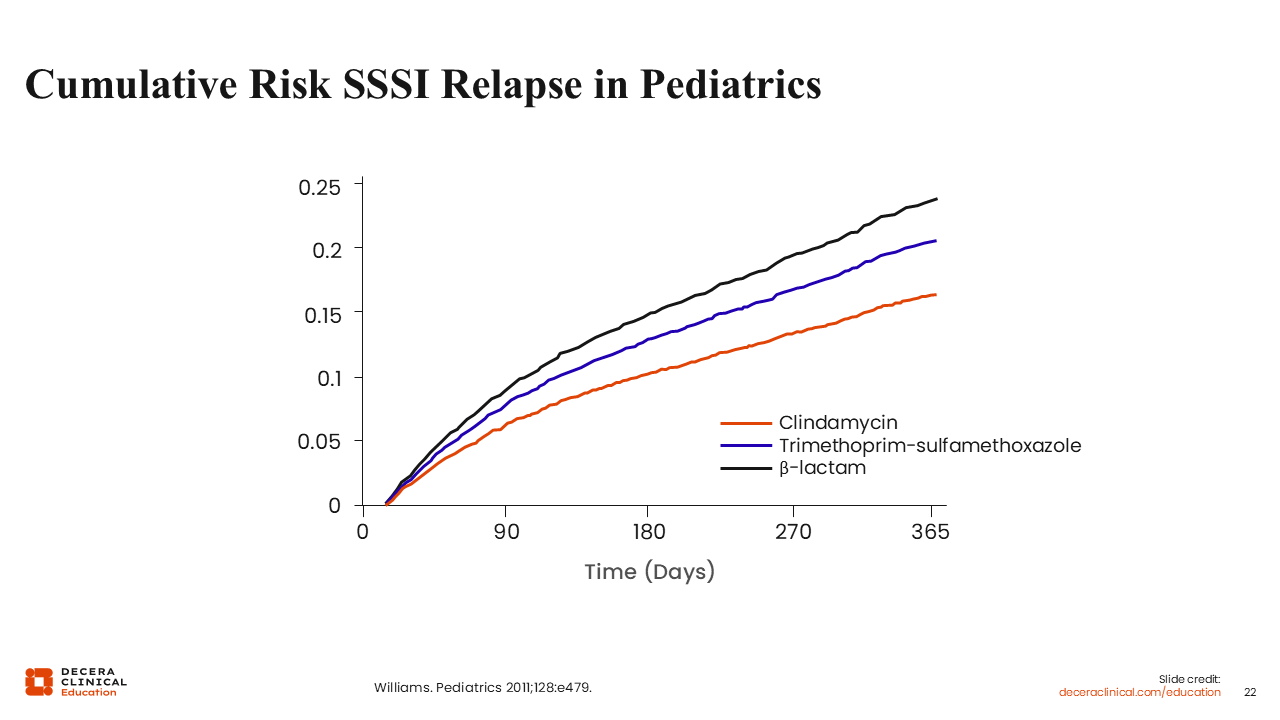
Cumulative Risk SSSI Relapse in Pediatrics
With all these options, there is the question of how clindamycin, TMP/SMX, and β-lactams compare with each other for the treatment of SSSI.
In this analysis of treatment for pediatric SSTIs, clindamycin was associated with the lowest relapse rates, whereas β-lactams had the highest relapse rates over time. This possibly speaks to clindamycin’s toxin inhibitory effect, which is not present in the β-lactam. These data highlight the fact that HCPs should consider taking advantage of specific antibiotic features when making treatment decisions.34
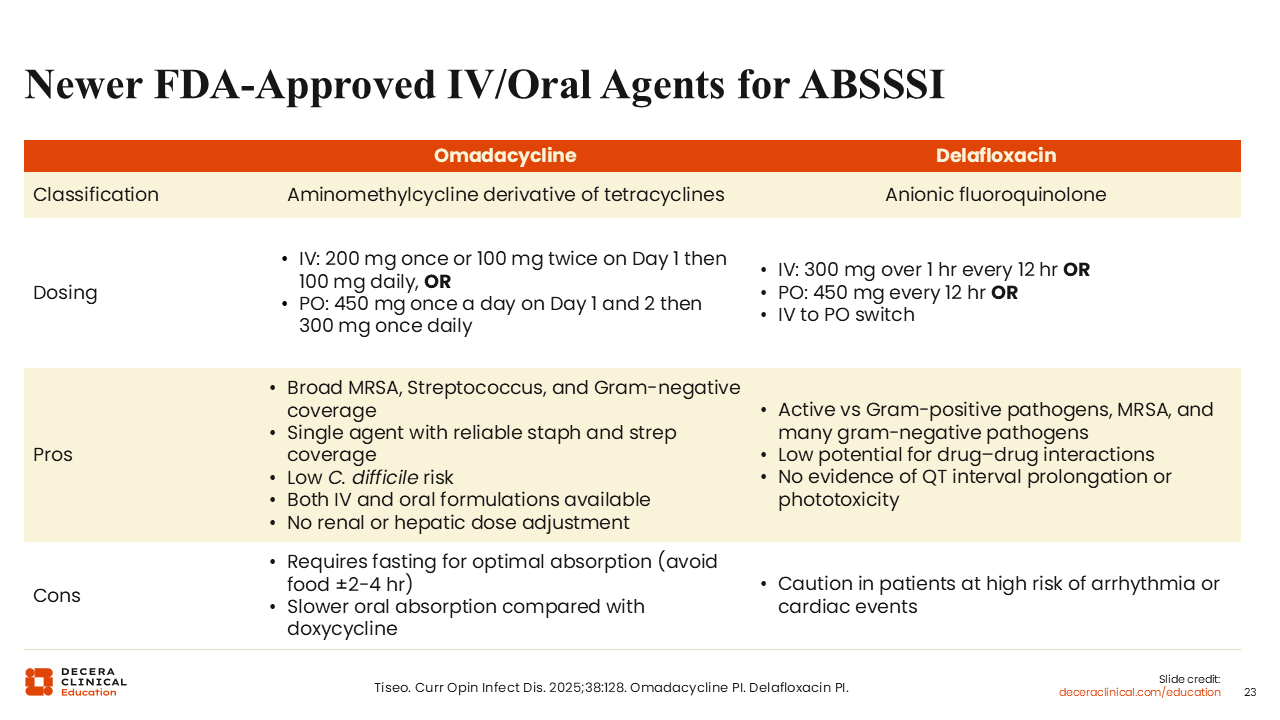
Newer FDA-Approved IV-Oral Agents for ABSSSI
Omadacycline and delafloxacin are newer FDA-approved oral agents for ABSSSI.35,36
I have not personally used delafloxacin for ABSSSI, but I would consider it for someone with a soft-tissue infection stemming from a water exposure where gram-negative pathogens, like Pseudomonas or Aeromonas, are a concern.35-37
In my experience, omadacycline is more broadly used, particularly when simultaneous coverage of S. aureusand Strep is desired. As a tetracycline derivative, omadacycline provides much more reliable coverage of Streptococci than older tetracyclines.35-37
A major difference between these 2 agents is the C. difficile risk. Omadacycline is associated with low C. difficile risk, whereas delafloxacin is associated with higher risk by virtue of being a quinolone. As a quinolone, delafloxacin also is associated with increased risk of cardiac events, and omadacycline is not. However, omadacycline is more slowly absorbed and requires fasting for optimal absorption.35-37
OASIS-1 and OASIS-2: Omadacycline Efficacy in ABSSSI
Omadacycline achieved FDA approval when it was found to be noninferior to linezolid in the OASIS-1 and OASIS-2 clinical trials. I found this to be a pretty robust comparison, as linezolid can be thought of as the gold standard and a very reliable treatment for community-acquired soft tissue infections.38
OASIS-1 and OASIS-2: Omadacycline Safety
In terms of safety and tolerability, omadacycline was also similar to linezolid. Oral linezolid was associated with nausea, vomiting, and gastrointestinal disturbance, but adverse effects were generally mild.38
Delafloxacin Efficacy vs Vancomycin Plus Aztreonam
Similarly, delafloxacin was given FDA approval when it demonstrated noninferiority to vancomycin plus aztreonam.39
Special Considerations for Omadacycline: Place in Therapy
For which patients would omadacycline be best? I tend to consider omadacycline more for higher-risk patients. This includes older patients and patients who are immunocompromised, with community-acquired infection and no prior antibiotic exposure, where S. aureus and Strep remain the primary concern.35,38,40-42]
Looking more closely at the omadacycline clinical trial data, particularly in subgroup analyses, the diabetic population is noteworthy. In that subgroup, outcomes in the omadacycline arm were superior to those in the linezolid arm, although the reasons for this are not entirely clear. There are theoretical explanations—possibly related to polymicrobial considerations or tissue penetration—but these remain speculative. Omadacycline may also be an attractive option in patients at high risk for CDI, or in those with a recent history of C. difficile.35,38,40-42
Finally, a simpler once-daily dosing schedule with oral omadacycline may allow for improved treatment adherence.35
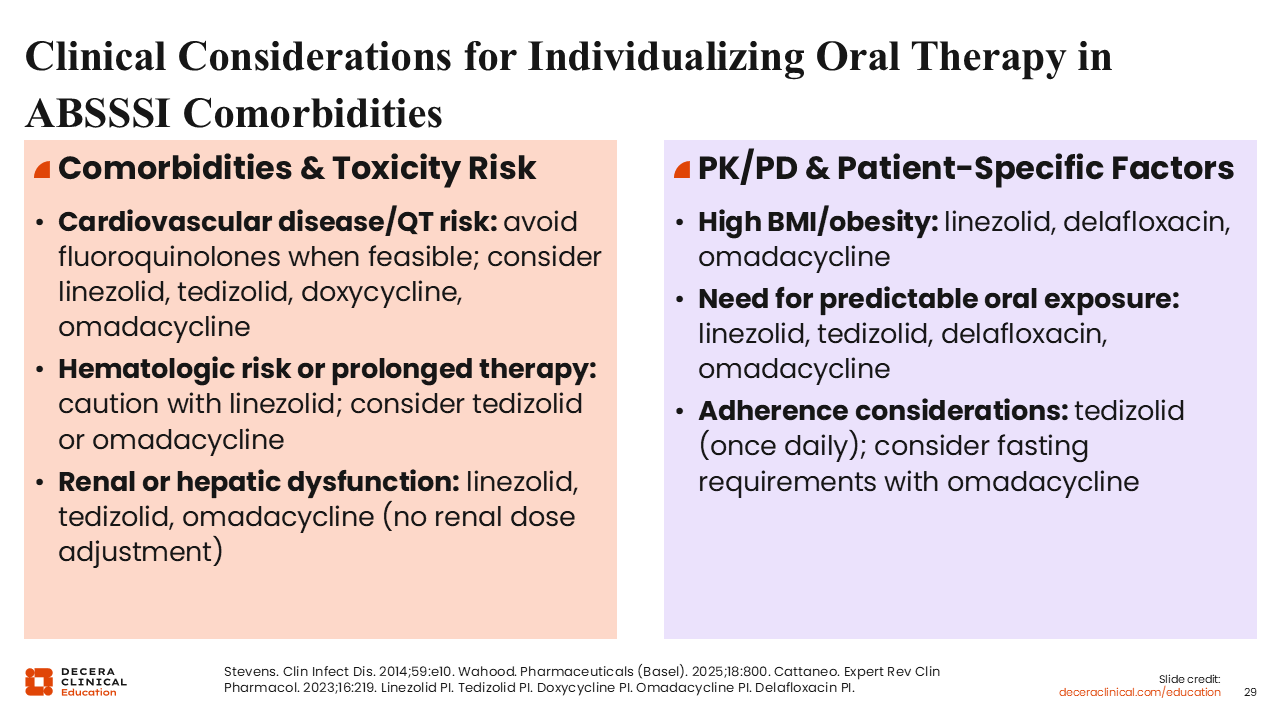
Clinical Considerations for Individualizing Oral Therapy in ABSSSI
In terms of individualizing oral therapy, the factors that come into play are patient age, previous infections and treatment history, and the risk of MRSA.23
Pathogen risk profile is also key. When there is a risk for MRSA, linezolid and tedizolid are options. Linezolid is dosed twice daily, whereas tedizolid offers the convenience of once-daily dosing, which may improve adherence in some patients.23
If there is concern for gram-negative mixed infections, that is, in patients who have received previous antibiotics, it is important to consider treatment for Pseudomonas and some of the other more resistant gram-negative pathogens. In these cases, antibiotic selection must reflect that expanded risk profile.23
For bite wounds, where anaerobic organisms are a concern, amoxicillin/clavulanate is typically the backbone of therapy, with or without an MRSA agent.23
Clinical Considerations for Individualizing Oral Therapy in ABSSSI Comorbidities
Among patient-specific factors, comorbidities are also a consideration. Drug–drug interactions are very important, and care must be taken to avoid exacerbating any preexisting conditions.23
Among patients with cardiovascular disease/QT risk, fluoroquinolones should be avoided. However, I have recently observed hospitalists order ECGs to measure QTcs in patients receiving quinolones. I found this quite profound, as quinolones have been in use since the 1990s, and this has never been standard practice. Clearly, the concern for QT prolongation with fluoroquinolones is heightened among prescribers, but there are ways to use these agents safely, even among patients with cardiovascular disease/QT risk.23,43
Hematologic toxicity and renal impairment may require dose adjustments in certain drugs, along with obesity and high BMI.
I think dosing in obesity can be complicated with some drugs, particularly cephalosporins. Sufficient oral exposure is another consideration.23,35,36,44-47
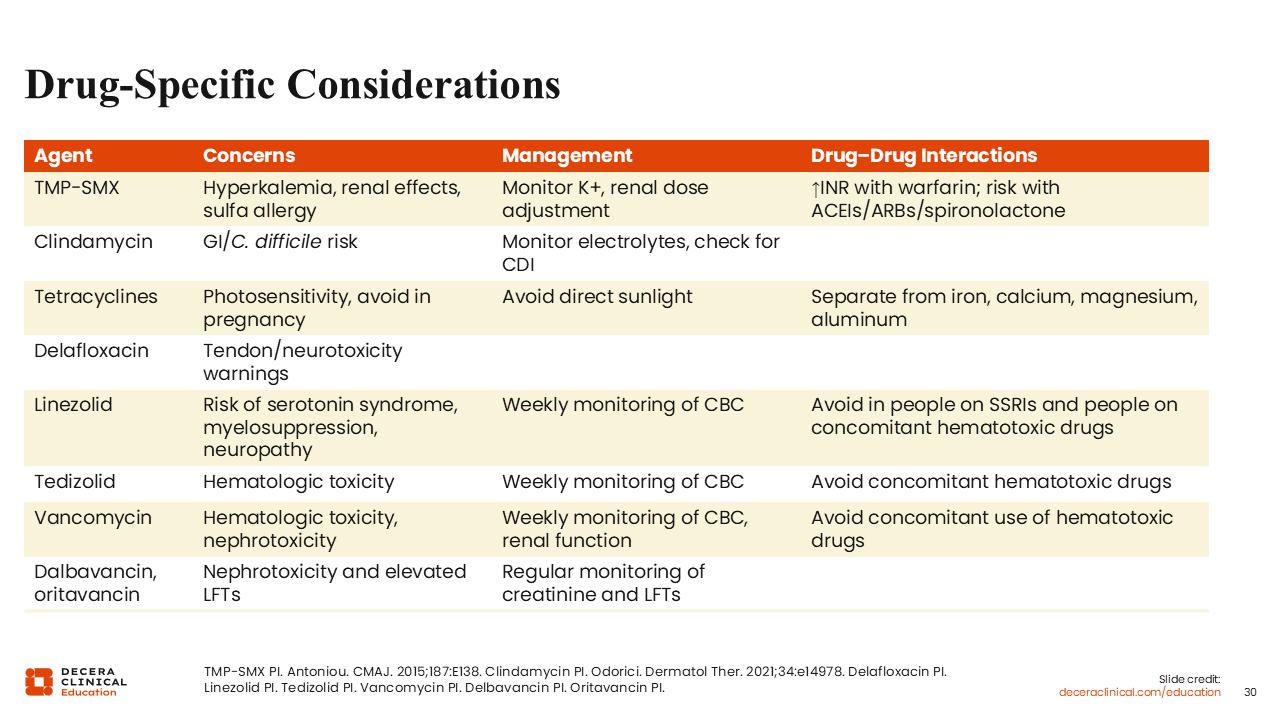
Drug-Specific Considerations
Regarding drug-specific considerations, I would not use TMP/SMX in a patient older than 60 years of age and concurrently receiving a coumarin or an angiotensin-converting enzyme inhibitor. The C. difficile risk should be considered with clindamycin. Photosensitivity with tetracyclines can be a real concern if the patient is in a sunny climate. Quinolones come with warnings about tendon rupture and neurotoxicity. Linezolid carries with it the risk of SSRI interaction.36,45,48-50
Vancomycin is known for its nephrotoxic potential, although I think that is less of a concern now with the shift from trough-based dosing—where higher trough levels were often targeted for many years—to area under the concentration curve–guided dosing strategies, which are associated with a lower risk of nephrotoxicity. However, I would still use caution when prescribing this to patients who are receiving other potentially nephrotoxic drugs.51
There is also a heightened awareness of hypoglycemia with linezolid, more so than with tedizolid. These drugs have monoamine oxidase–inhibitory activity, which can increase circulating catecholamines. They also increase sensitivity to insulin.45,46
Increasing reports of hypoglycemia with linezolid, particularly in patients with type 2 diabetes, are reflected now in the prescribing information. I think that is an additional concern, especially for treatment-naive patients.45
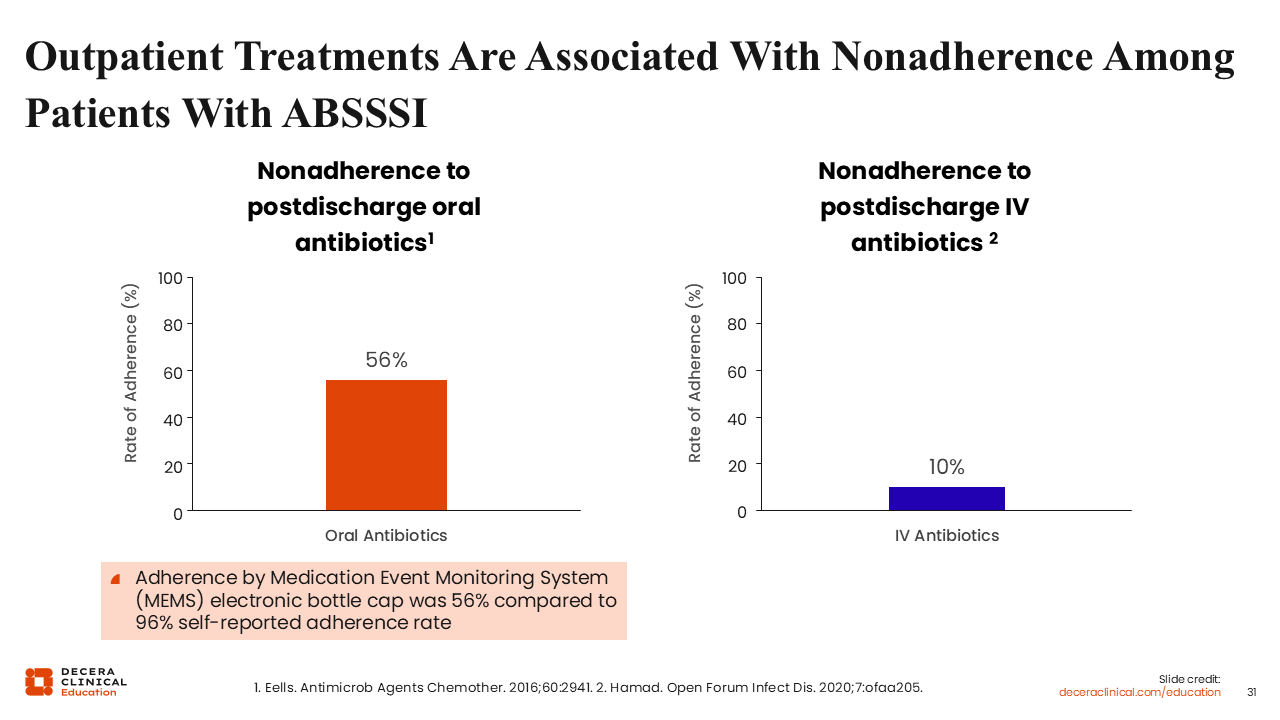
Outpatient Treatments Are Associated With Nonadherence Among ABSSSI Patients
Of note, although OPAT increases compliance, oral therapy is associated with nonadherence. Nonadherence to postdischarge oral antibiotics is more than 50% in many patients.52,53
This may be because of difficulty adhering to complicated dosing schedules, especially since some of these oral agents must be administered multiple times per day. For instance, combination therapy with cefalexin plus TMP/SMX entails 6 doses of antibiotics per day. Is it really practical to expect patients to take 6 doses per day for a week?23
In practice, some HCPs have adopted more practical dosing to encourage better adherence. For example, cefalexin is usually dosed at 500 mg 4 times daily. I generally prescribe my patients a 1-g dose of cefalexin every 8 hours, or 3 times daily, to eliminate that middle dose in the middle of the day.54
Simplified dose regimens also can be further justification for using newer agents with less frequent dosing.