CE / CME
Spot the Signal: Global Radiology Training for ARIA Detection in Alzheimer’s Care
Physician Assistants/Physician Associates: 1.00 AAPA Category 1 CME credit
Physicians: Maximum of 1.00 AMA PRA Category 1 Credit™
European Learners: 1.00 EBAC® CE Credit
Released: April 23, 2026
Expiration: April 22, 2027
Activity
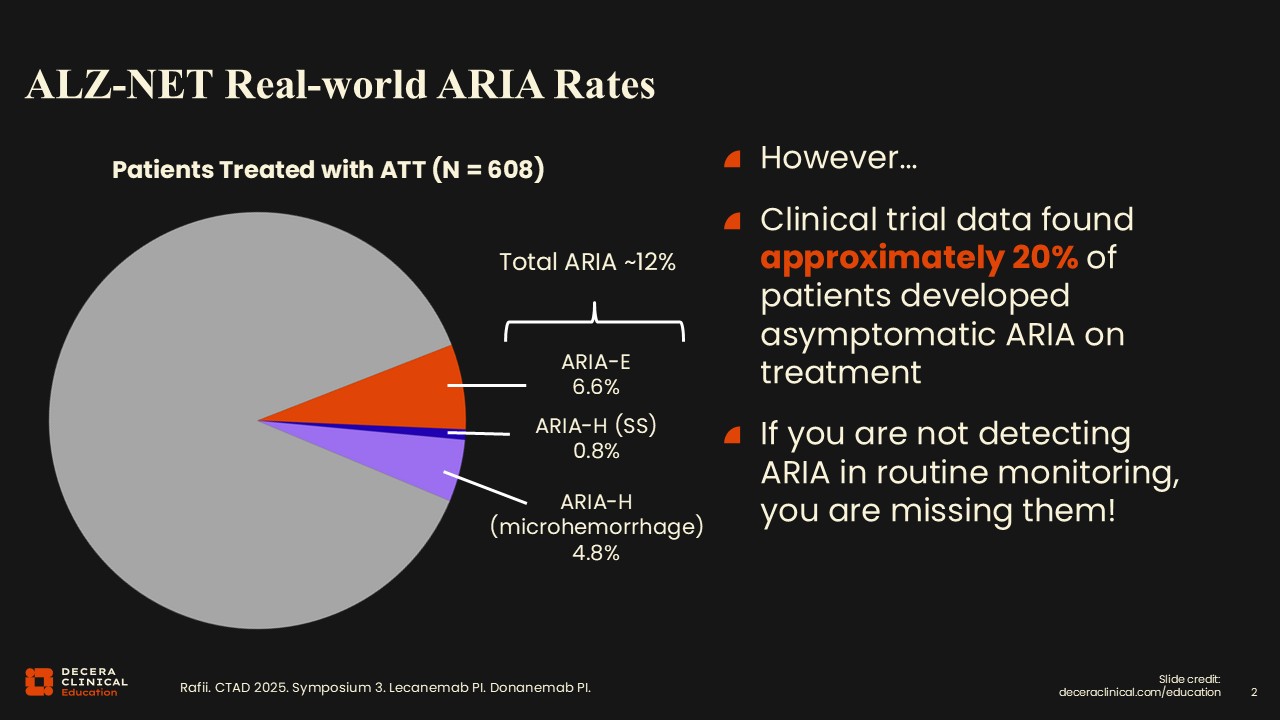
ALZ-NET Real-world ARIA Rates
Dr Benzinger:
Real-world data suggest that approximately 20% of ARIA cases will be asymptomatic, which is important to recognize.6 If you read 5 of these scans in a week, you should see 1 case of ARIA. It is not a rare occurrence but a rather common finding to see on a routine MRI for a patient treated with ATT. Radiologists who interpret MRIs for several patients treated with ATTs should expect to encounter ARIA relatively frequently, rather than viewing it as a rare complication.
Recognizing how common ARIA is in treated populations helps set expectations for radiologists and other healthcare professionals (HCPs) on the care team and reinforces the need for careful, systematic review of both eligibility and monitoring scans.
MRI Monitoring Schedule for ATTs
Dr Benzinger:
The 2 available ATTs, donanemab and lecanemab, have slightly different dosing regimens and also slightly different MRI schedules to monitor for ARIA.7,8 Typically, MRIs are scheduled at baseline (before the first infusion) and at multiple time points during treatment, which are often preplanned at the time of patient enrollment to start therapy. If we see a positive finding on any MRI, then we recommend additional scans for follow-up 1 or 2 months after that positive MRI until edema (for cases of ARIA-E) has completely resolved and/or hemorrhages (for cases of ARIA-H) have stabilized. Clearly, this is a substantial number of scans for the patients and additional workload for radiology practices.
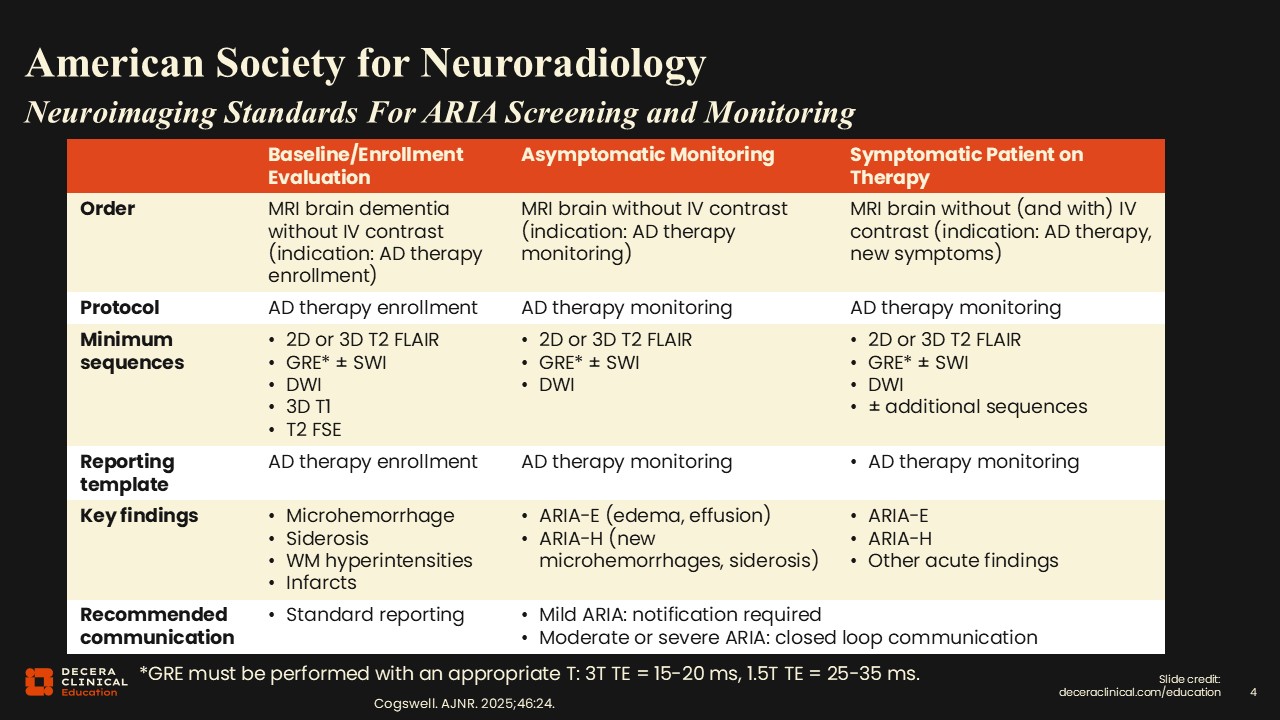
American Society for Neuroradiology (ASNR) Recommendations
Dr Benzinger:
There are some key reference guidelines radiologists can use to make sure that scans are being done correctly. The ASNR and related professional societies have developed detailed technical guidelines for ARIA monitoring.9,10 The key principle is consistency; scans should be performed using standardized protocols to allow reliable comparison over time.
If you go to the ASNR website, for example, you will find links to download vendor-specific protocols for Siemens, GE, and Philips scanners, so you don't have to build the protocols from scratch yourself.11 Adherence to these protocols supports reproducible detection of subtle ARIA findings and reduces inter-reader variability.
I want to add a little color to some details here, too. The question of SWI vs T2 comes up in nearly every conversation with radiologists. A related issue is field strength: 1.5 vs 3 Tesla (3T). For those outside neuroradiology, it may seem logical to think 3T SWI is always best, but in practice, it is not that straightforward. Because of advances in gradients, coils, software, and pulse sequences, higher field strength does not automatically mean better imaging. If I compare my oldest 3T scanner with my newest 1.5T scanner, I can often obtain better brain images from the newer 1.5T system.
In my experience, consistency is the most important factor. If a patient can remain on the same scanner over time, that provides the best longitudinal comparison. However, most of us do not practice in that kind of environment. In my health system alone, we have 60 MRI scanners serving patients across a 300-mile radius, so it is simply not feasible for every patient to return to the same scanner each time.
I do typically begin with 3T using both SWI and GRE. This gives a baseline sense of where hemorrhages are and what they look like as a grounding point for future comparisons. We are sometimes asked why we continue to acquire both sequences on follow-up. One key reason is motion. SWI is highly sensitive and excellent for detecting small findings, but only if the patient can remain still. It is important to remember that patients with dementia, particularly those experiencing ARIA symptoms, often have difficulty tolerating an MRI exam. SWI may fail entirely because of motion. In those situations, the faster GRE/T2* sequence can “save the day” and still allow us to identify critical findings. This is why, in our practice, we obtain both.
Using the ASNR protocols, we have streamlined our monitoring exams. We only acquire FLAIR, GRE, SWI, and DWI. We do not routinely obtain T1, T2, arterial spin labeling, or additional sequences. As a result, our monitoring exam takes about 10 minutes from start to finish. When I interpret the study, I focus only on those essential images. The findings can be subtle, so they require careful review, but they are not buried in thousands of unnecessary images. That is the philosophy guiding our approach.
Dr Lövblad:
This is exactly why we recommend limiting the exam to 4 or 5 sequences at most. Time matters, and patient tolerance matters. As noted, many of these patients cannot remain in the scanner for 30 minutes. The shorter we can make the exam, the better.
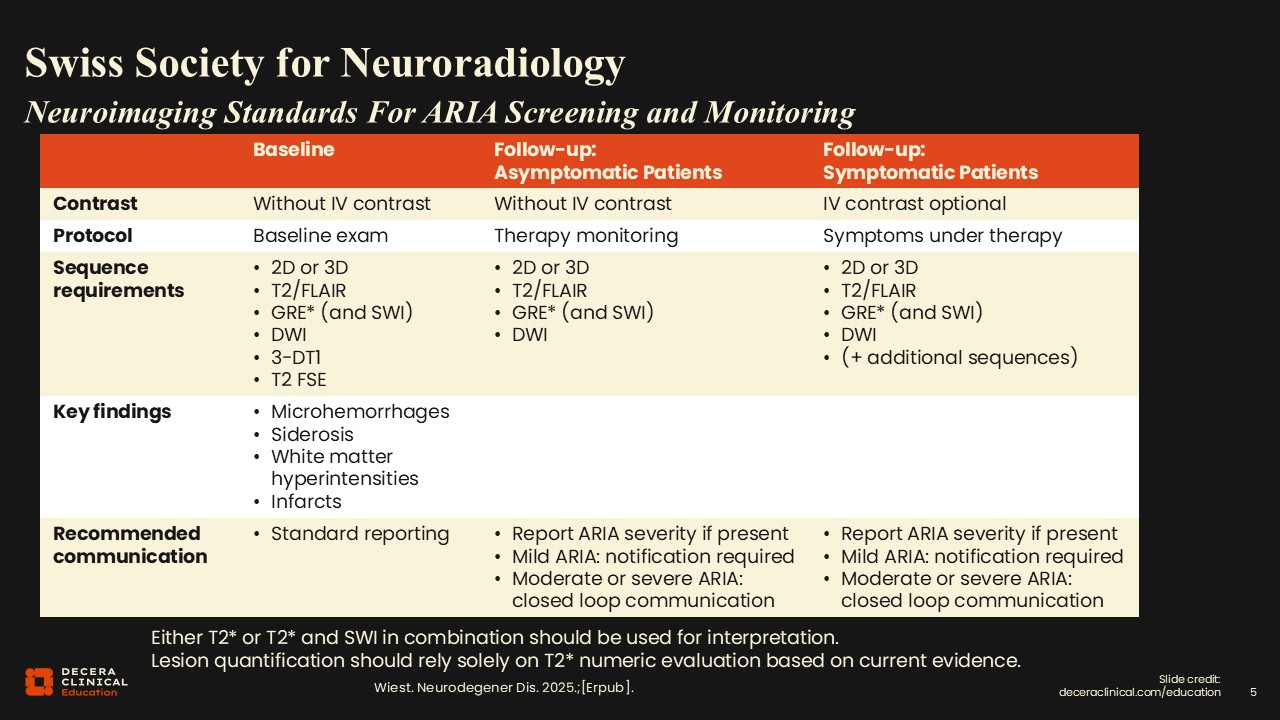
Swiss Society for Neuroradiology (SSNR) Recommendations
Dr Lövblad:
Recommendations from the SSNR are almost identical to those proposed by the ASNR, with gradient echo (GRE) and susceptibility-weighted (SWI) images to detect microhemorrhages.10,11 Using both sequences is particularly helpful. Diffusion-weighted imaging (DWI) is valuable for identifying alternative pathologies such as stroke, particularly when patients come in urgently. FLAIR imaging is used to look for the presence of hyperintensities that enter the white matter or infarcts that might be older and not necessarily detected by DWI. Baseline examinations follow standard reporting protocols, whereas follow-up reporting differs depending on whether patients are asymptomatic or symptomatic.
In asymptomatic patients, we will not use IV contrast, but we find it may be useful in symptomatic patients, and we perform the combination of the 4 basic sequences or 5, including DWI, GRE, SWI, T2, and 3D imaging. Everything is considered on a case-by-case basis, and depending on grading, additional sequences may be ordered. Communication of findings is essential so that treating HCPs can make clinical treatment decisions, which may be more cautious in some circumstances.
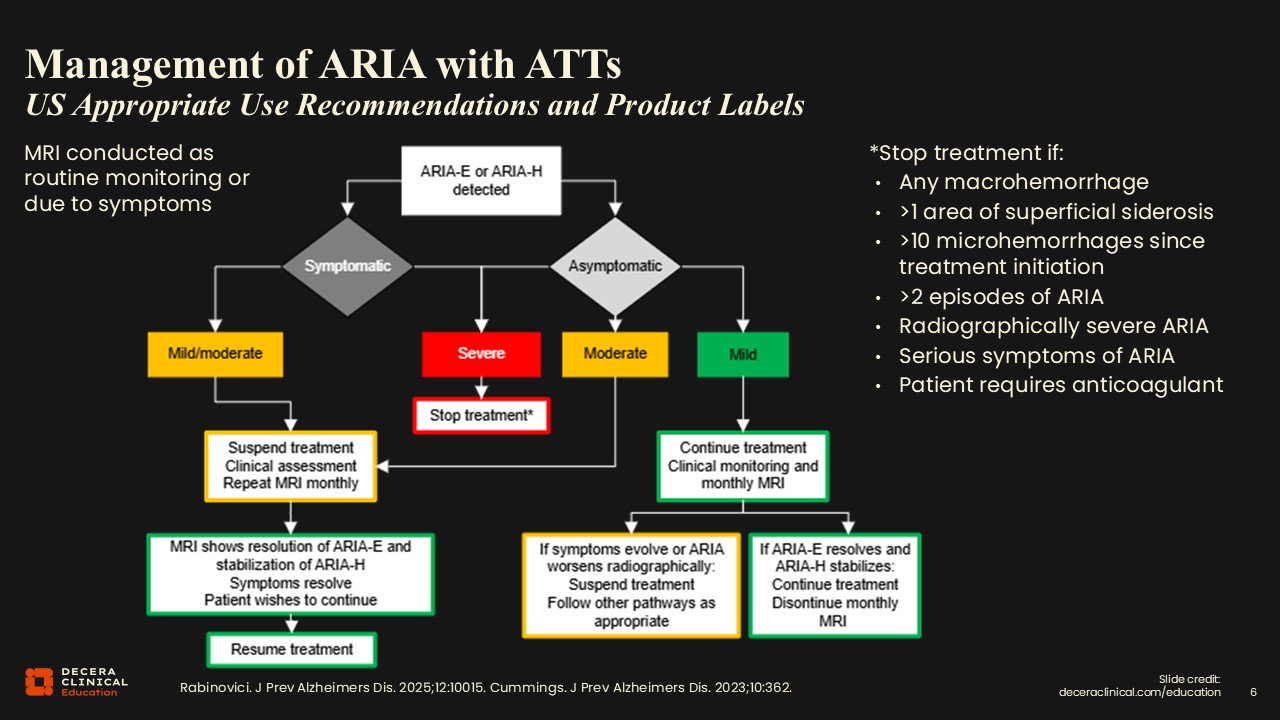
Management of ARIA With ATTs
Dr Benzinger:
When an MRI finding of ARIA-E or H is detected, that triggers the start of a treatment algorithm. The next step is correlation with symptoms. A positive or worsening case of ARIA necessitates a call to the referring physician's office so that they can get in touch with the patient and assess whether symptoms are present, if that is known.
Treatment is then guided by the ARIA severity as determined by standardized grading. For severe ARIA, treatment is stopped regardless of the presence or absence of symptoms. If the patient has mild or moderate ARIA with symptoms, they go down 1 side of this flowchart. If they are asymptomatic, the treatment differs for mild vs moderate ARIA. This demonstrates the importance of the radiologist applying the correct grading scale to the images. The terms mild, moderate, and severe here are not subjective measures (“oh, it looks pretty bad to me”). A standardized objective severity scale is used across all the ATTs to drive clinical management decisions.
Dr Lövblad:
As noted previously, ideally, scans should be done on the same scanner at the same field strength. That might not always be possible because sometimes patients go to clinics or hospitals other than the one where they were initially treated. Maximal standardization is recommended, as comparison with prior studies is frequently essential. If different scanners or protocols are used, ARIA detection and evaluation can be difficult even for radiologists specifically trained in this area. Appropriate scans include FLAIR for edema in ARIA-E and some type of SWI T2* images for hemorrhage in ARIA-H.
Dr Benzinger:
For ARIA-E, grading is based primarily on the number and size of involved regions on FLAIR. For ARIA-H, separate grading criteria exist for microhemorrhages and for superficial siderosis, largely based on lesion count. Management options range from continued treatment with routine monitoring to temporary suspension of therapy, enhanced MRI surveillance, dose modification, or permanent discontinuation, depending on ARIA severity and the presence or absence of symptoms.3,4,7,8
From the radiology perspective, it is essential not only to identify ARIA but also to apply the correct grading criteria in the report and explicitly recommend follow-up imaging when appropriate, as these elements directly influence clinical decision-making.