CME
On the Frontlines of CLL/SLL: International Perspectives on First-line Treatments for CLL/SLL
Physicians: Maximum of 1.00 AMA PRA Category 1 Credit™
Released: February 10, 2026
Expiration: August 09, 2026
Activity
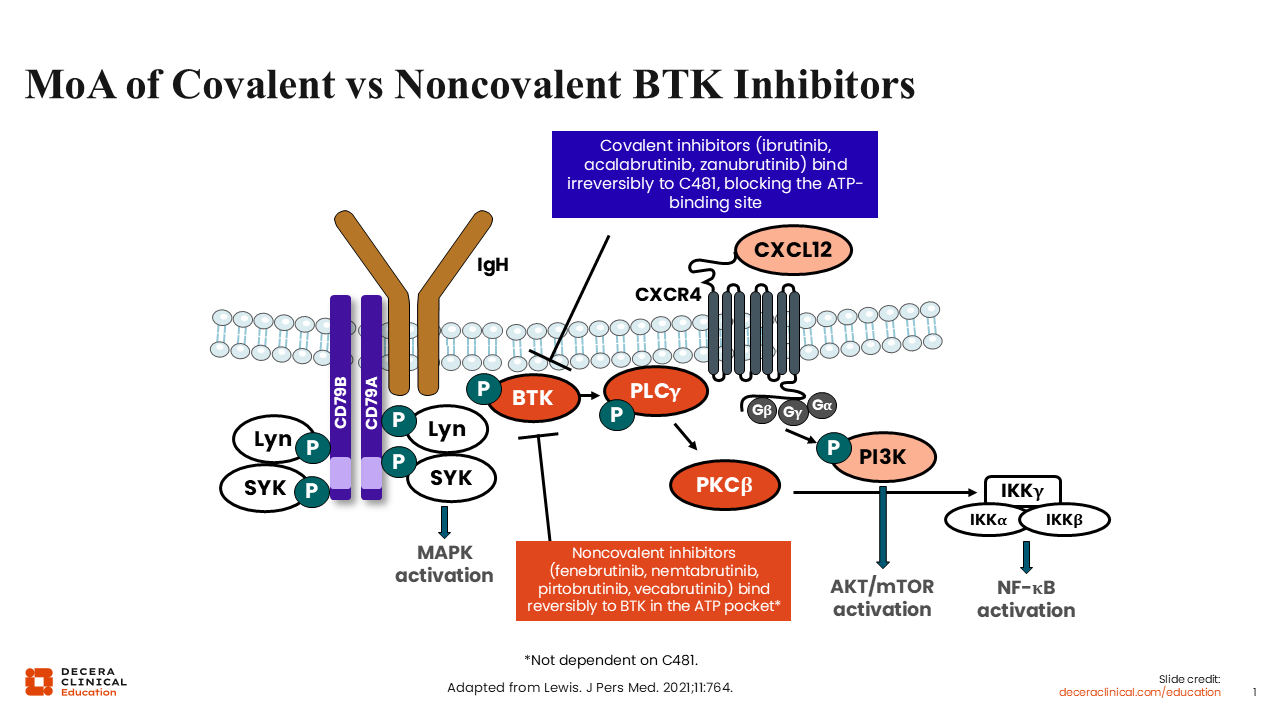
MoA of Covalent vs Noncovalent BTK Inhibitors
B-cell receptor signaling is important to the pathogenesis and progression of B-cell malignancies, with BTK playing a crucial role in the signaling pathway. Therefore, targeting BTK has proven to be a useful treatment strategy for managing patients with CLL/SLL.
The class of BTK inhibitors was pioneered with the covalent BTK inhibitors including ibrutinib, acalabrutinib, and zanubrutinib. Covalent BTK inhibitors bind irreversibly to the catalytic ATP-binding domain of the BTK protein on cysteine 481 (C481), thereby blocking autophosphorylation and downstream signaling through PLCγ2.1,2
The next development in this therapeutic drug class was the introduction of noncovalent BTK inhibitors, which reversibly bind to the ATP pocket regardless of C481 mutation status.1 To date, pirtobrutinib is the only noncovalent BTK inhibitor recommended in guidelines and approved by both EMA and FDA for CLL.3,4 Other noncovalent BTK inhibitors are in development, including fenebrutinib, nemtabrutinib, and vecabrutinib.2
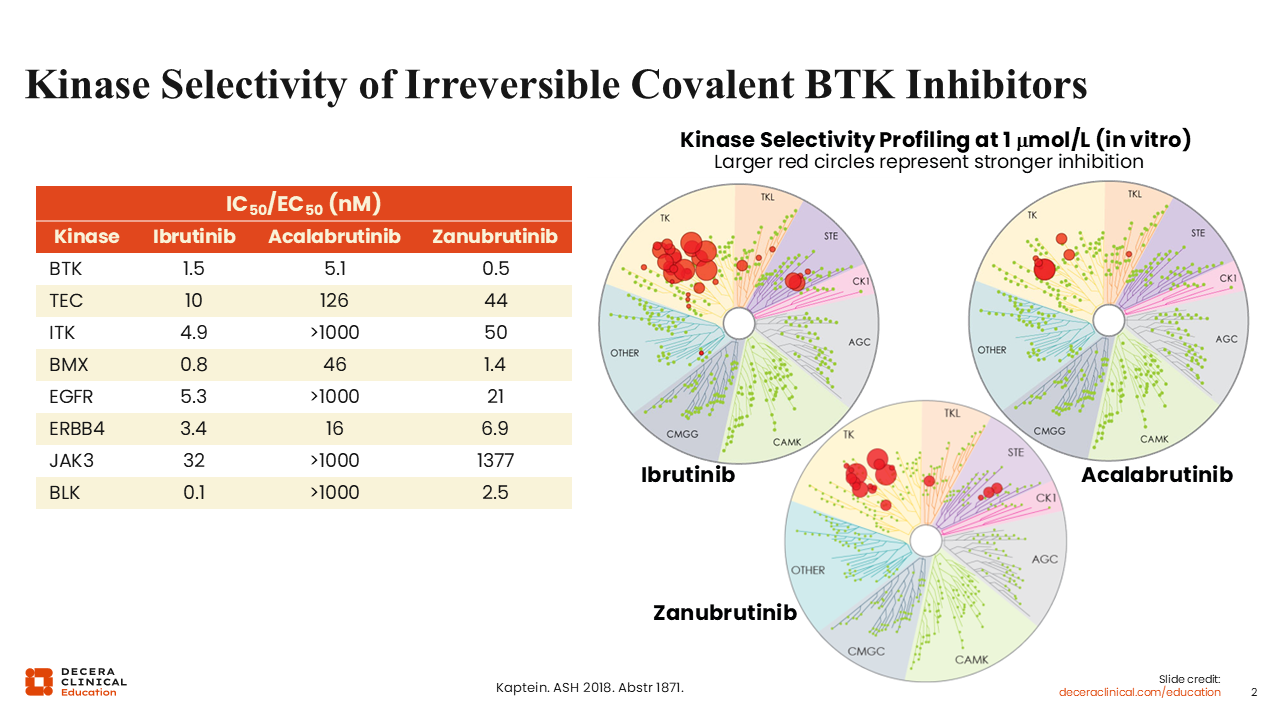
Kinase Selectivity of Irreversible Covalent BTK Inhibitors
The selectivity of the covalent inhibitors for BTK and other on-target and off-target kinases varies across the available BTK inhibitors (acalabrutinib, ibrutinib, and zanubrutinib). We see that acalabrutinib exhibits the highest selectivity for the BTK protein, followed by zanubrutinib, with ibrutinib being the least selective among the 3 approved covalent BTK inhibitors.5
A key objective in the development of more selective compounds targeting BTK has been to decrease off-target binding activity and to improve the AE profile of this drug class.
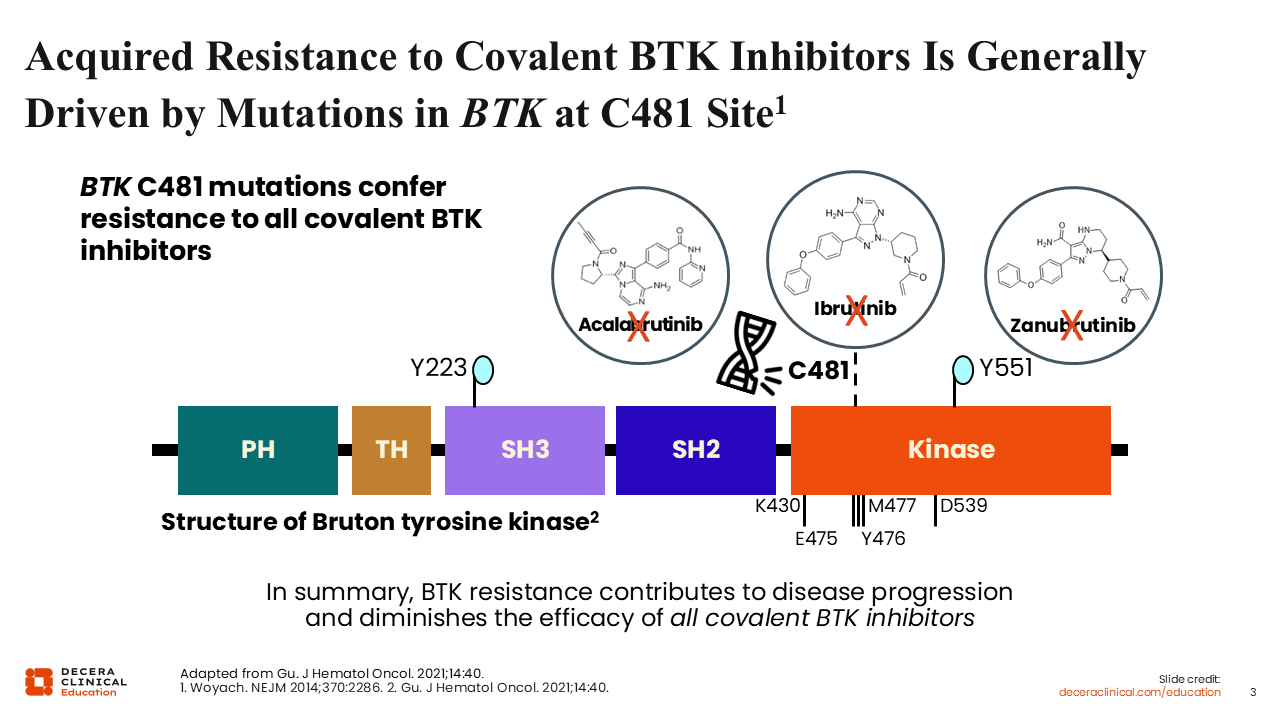
Acquired Resistance to Covalent BTK Inhibitors Is Generally Driven by Mutations in BTK at the C481 Site
Although the available covalent BTK inhibitors are very efficacious and some have been shown to provide prolonged overall survival (OS) in clinical trials, patients eventually develop resistance to covalent BTK inhibitors and experience disease progression.6-8
The most frequent cause of covalent BTK inhibitor resistance is gene mutations that affect the C481 site of the BTK protein, where the covalent BTK inhibitors bind.7,9 Mutations at C481 abrogate the covalent binding, conferring resistance to covalent BTK inhibitors and restoring the ATP-binding activity of BTK. Because of their short half-life in plasma, covalent BTK inhibitors can only achieve incomplete inhibition of the BTK pathway in patients with a confirmed C481 mutation.8,10 Of note, mutations in other locations on BTK occur much more rarely during treatment with these covalent BTK inhibitors.2
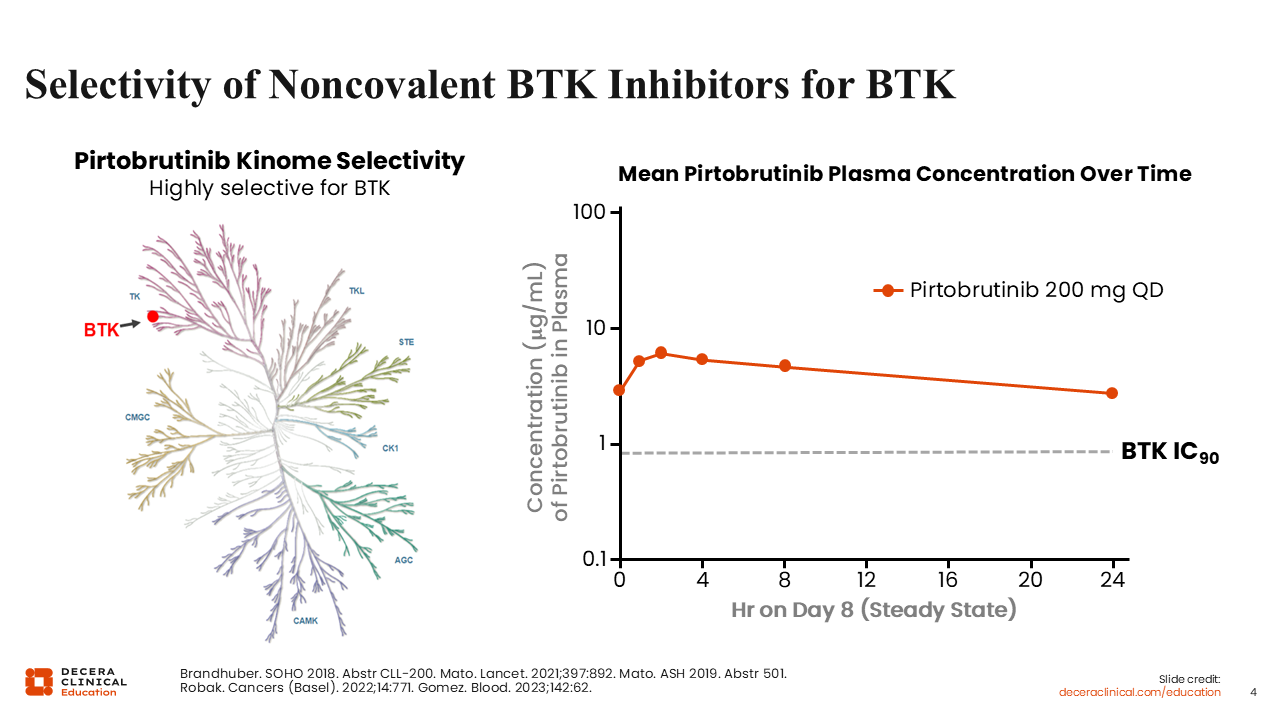
Selectivity of Noncovalent BTK Inhibitors for BTK
The mechanism of resistance to covalent BTK inhibitors via the emerging mutations at C481 led to the development of next-generation noncovalent BTK inhibitors that do not rely on C481, such as pirtobrutinib.11-13
Pirtobrutinib is a selective, noncovalent (reversible) BTK inhibitor currently approved by both the EMA and the FDA. The EMA approved pirtobrutinib for relapsed/refractory (R/R) CLL after previous treatment with a BTK inhibitor.3 The FDA approved pirtobrutinib for treating adults with CLL or SLL who have been previously treated with a covalent BTK inhibitor.4
If we look at the kinome selectivity map for pirtobrutinib, we can see high selectivity for BTK. Moreover, pirtobrutinib also has a long plasma half-life, allowing for complete BTK target occupancy with once-daily dosing.14,15
ALLIANCE A041202: First-line Ibrutinib ± Rituximab vs Bendamustine + Rituximab
I would like to briefly review the efficacy of these BTK inhibitors starting with the phase III ALLIANCE A041202 study evaluating ibrutinib alone or ibrutinib plus rituximab (IR) vs bendamustine plus rituximab (BR) in the first-line treatment of older or physically unfit patients with CLL (N = 547).16,17 BR was administered for 6 cycles whereas ibrutinib was administered as continuous therapy until disease progression occurred. The BR arm allowed crossover to the ibrutinib arm after disease progression. The primary endpoint of the study was PFS.
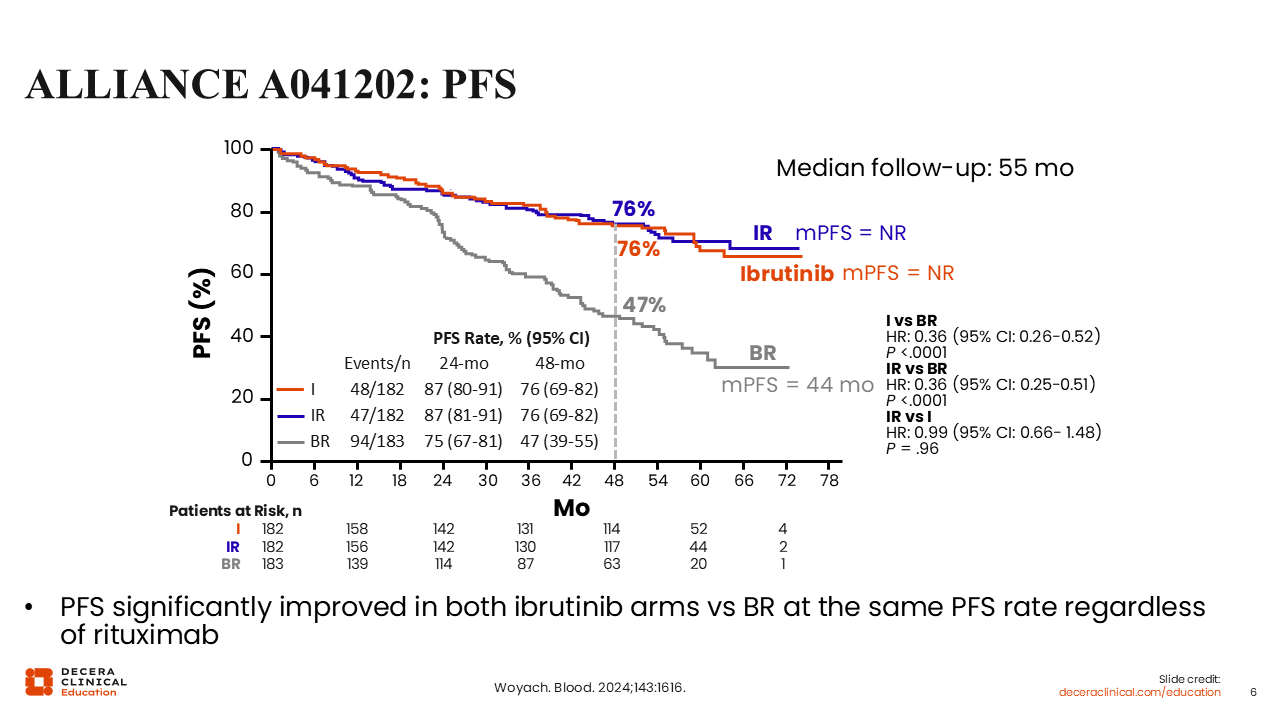
ALLIANCE A041202: PFS
After a median follow-up of 55 months, data for median PFS showed that ibrutinib alone and IR were superior to BR (HR for ibrutinib vs BR: 0.36; P <.0001; HR for IR vs BR: 0.36; P <.0001).17 Median PFS was NR for the arms containing ibrutinib and was 44 months for the BR arm. The 48-month PFS rates were 76% for both ibrutinib-containing arms vs 47% for the BR arm.
All patient subgroups also showed improved PFS with IR vs BR, except patients with mutated IGHV disease.
Taken together, these data showed that the addition of rituximab to ibrutinib was not superior to ibrutinib alone, so it is not a standard treatment addition to ibrutinib.
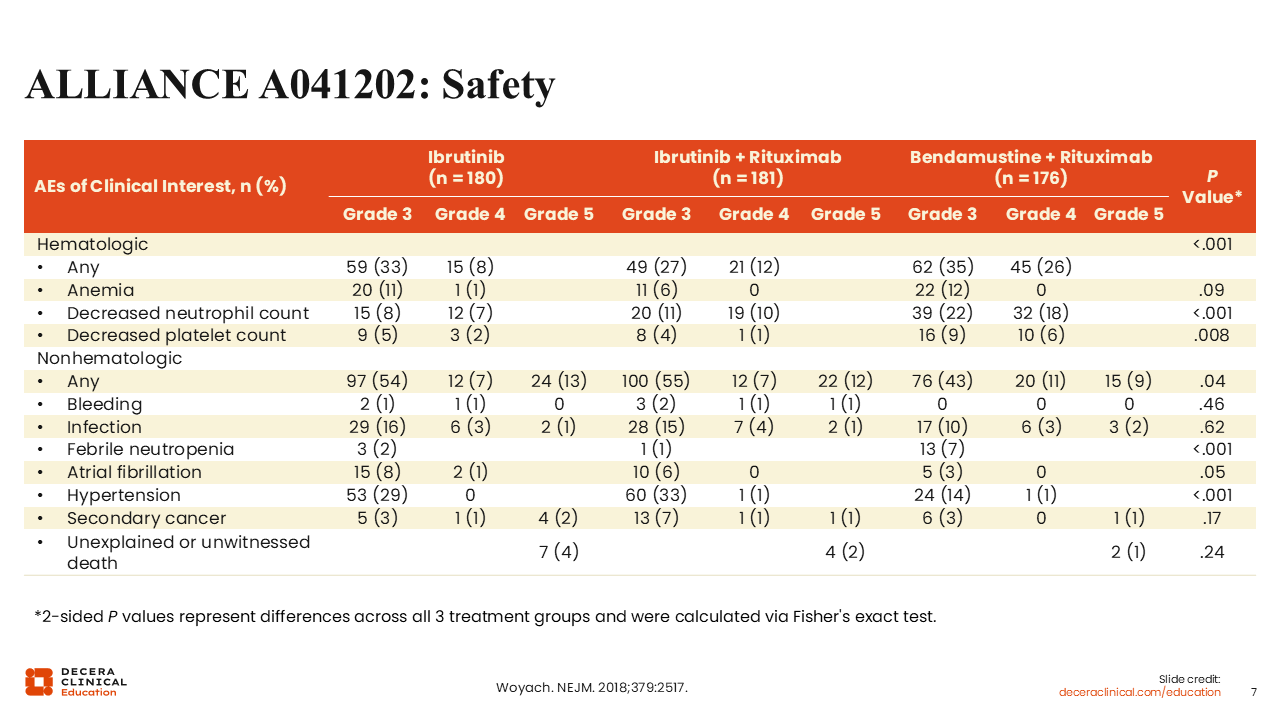
ALLIANCE A041202: Safety
BR treatment demonstrated the expected safety profile, consisting mostly of hematologic toxicity and infections.17 In the study arms containing ibrutinib, hematologic toxicity also occurred but at a lower frequency. With ibrutinib, we also see the class-associated AE of bleeding.18,19 However, higher-grade bleeding events with ibrutinib occurred infrequently (grade 3/4: 1%/1%).
Rates of grade 3 atrial fibrillation (ibrutinib vs IR vs BR: 8% vs 6% vs 3%, respectively) and hypertension (ibrutinib vs IR vs BR: 29% vs 33% vs 14%, respectively) were twice as high in the ibrutinib-containing arms compared with the BR arm. These AEs are also associated with the BTK inhibitor class, so we need to closely monitor for them when treating patients with BTK inhibitors.
When discussing the safety profiles of the ALLIANCE A041202 trial, I must note that the capture period of AEs here is different for the BR and ibrutinib arms because BR is a time-limited treatment and corresponding AEs were captured during the 6 cycles of treatment.20 Only unsolicited AEs were recorded after the 6 cycles. By contrast, for the 2 ibrutinib arms, AEs were solicited and captured for the entire duration of treatment. These facts should be considered when looking at these AE data.
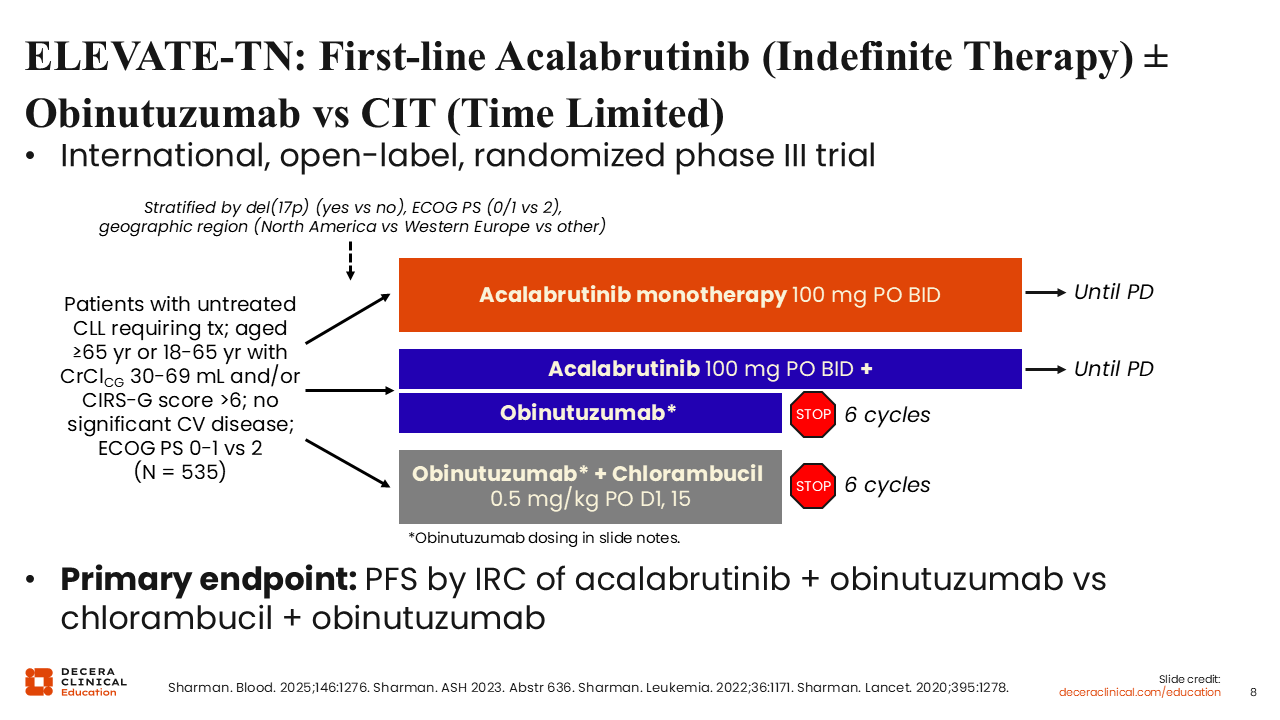
ELEVATE-TN: First-line Acalabrutinib (Indefinite Therapy) ± Obinutuzumab vs Chemoimmunotherapy (Time Limited)
The next development in the BTK inhibitor drug class was the next-generation agent acalabrutinib, a more selective covalent BTK inhibitor that became available based on the results of the randomized phase III ELEVATE-TN trial. This trial compared continuous treatment with acalabrutinib alone or continuous acalabrutinib plus obinutuzumab (AO) for 6 cycles vs OC for 6 cycles in older patients with CLL or physically unfit patients with CLL and decreased renal function or other comorbidities (N = 535).21-24 The primary endpoint was PFS by independent review committee (IRC) assessment.
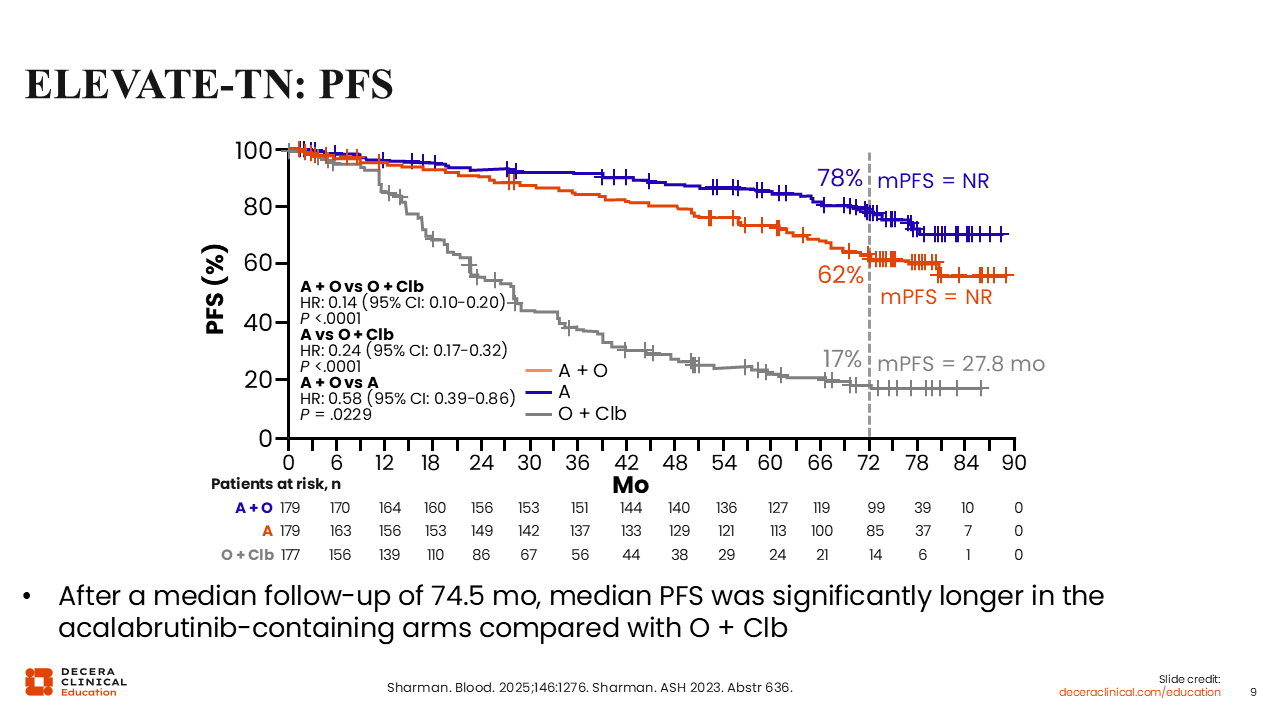
ELEVATE-TN: PFS
The primary endpoint was met, with acalabrutinib and AO demonstrating clinically meaningful and statistically significant improvement in median PFS vs OC (HR for acalabrutinib vs OC: 0.24; P <.0001; HR for AO vs OC: 0.14; P <.0001).24 In addition, median PFS was higher with acalabrutinib-containing regimens vs OC in all patient subgroups, including those with or without del(17p), TP53, or IGHV mutations.
Of note, there was an improvement in PFS with the addition of obinutuzumab to acalabrutinib vs acalabrutinib monotherapy (HR: 0.58; P = .0229). The median PFS was NR in both arms containing acalabrutinib and was 27.8 months in the OC arm.
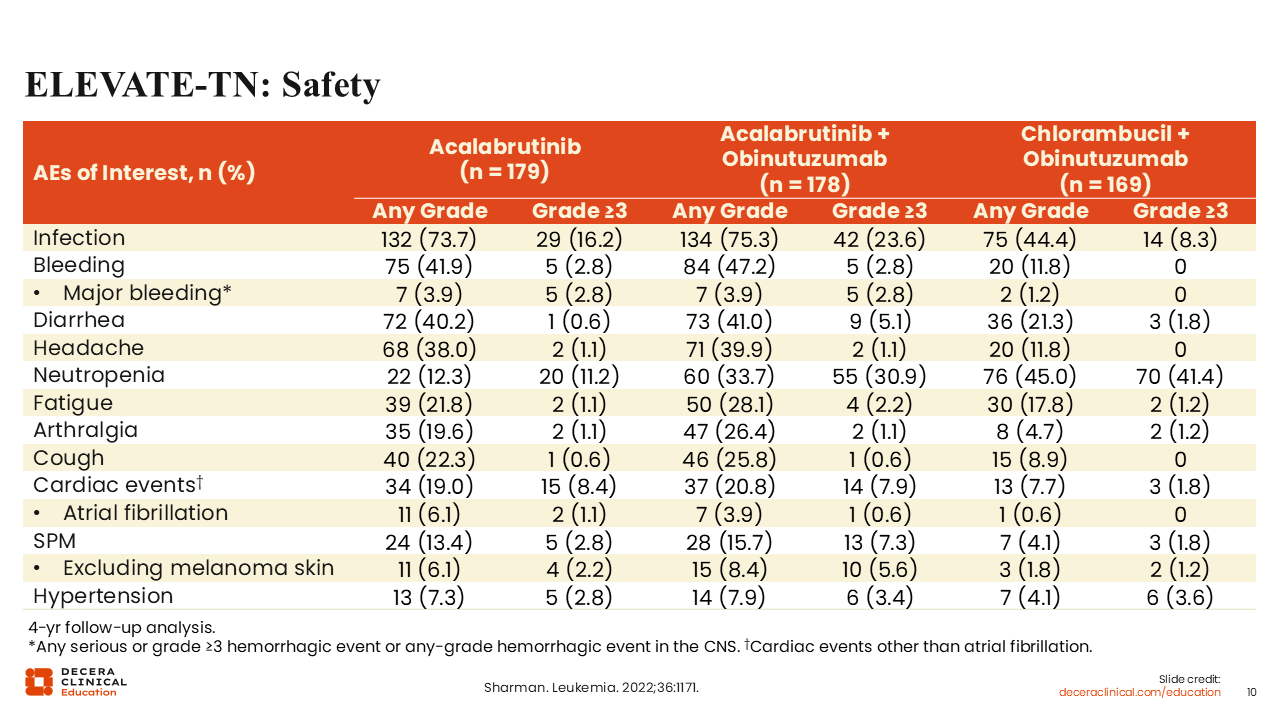
ELEVATE-TN: Safety Analysis
When we look at the safety analysis, we should again consider the impact of different treatment durations on the capture of AE data. In ELEVATE-TN we still see the AEs associated with BTK inhibitors in the acalabrutinib-containing arms, such as cardiac events, bleeding, and hypertension, but at a lower frequency and severity (fewer grade ≥3).22 Of note, the addition of obinutuzumab to acalabrutinib increased the incidence of grade 3 neutropenia (30.9% with obinutuzumab vs 11.2% without obinutuzumab) and infection (23.6% vs 16.2%, respectively).
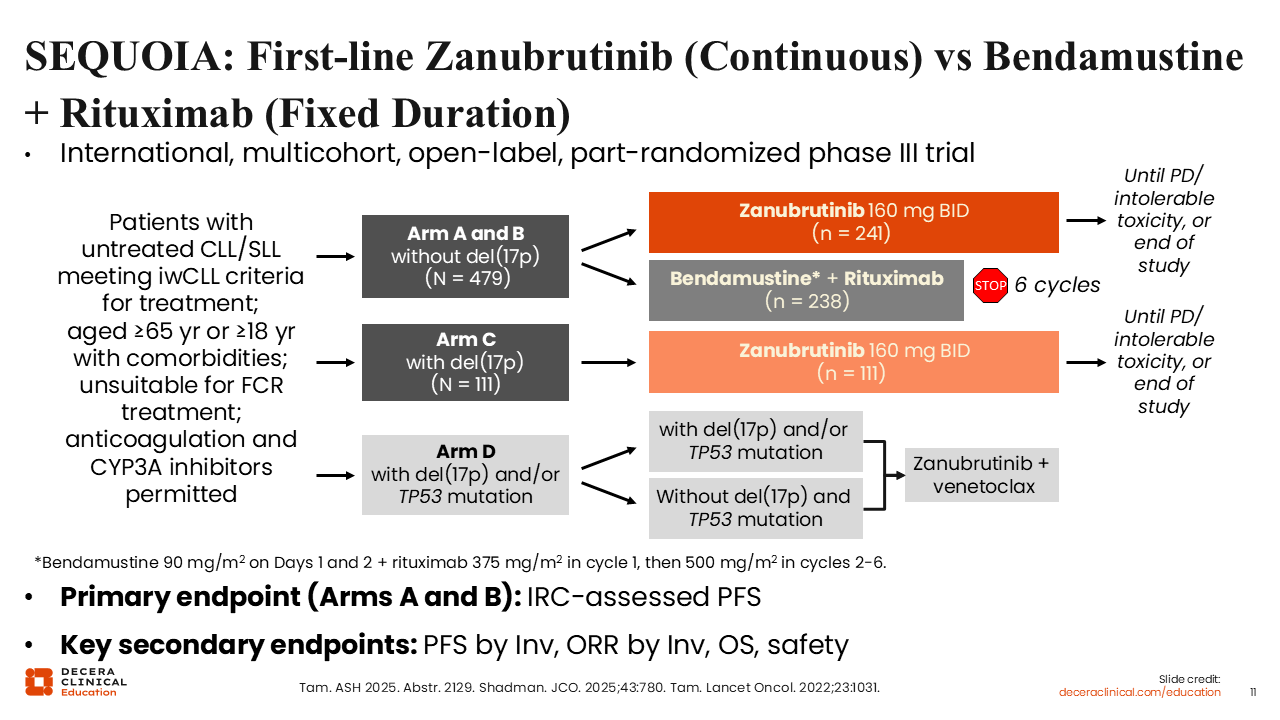
SEQUOIA: First-line Zanubrutinib (Continuous) vs Bendamustine + Rituximab (Fixed Duration)
Next, I would like to move on to discuss data from the multicohort phase III SEQUOIA study in patients with previously untreated CLL/SLL who meet the International Workshop on Chronic Lymphocytic Leukemia criteria for treatment (N = 590).25,26 Updated results were recently presented for cohort 1 and 2 which enrolled patients without del(17p) and with del(17p), respectively. Patients in cohort 1 were randomized to receive continuous zanubrutinib (arm A; n = 241) or BR for 6 cycles (arm B; n = 238) whereas all patients in cohort 2 received zanubrutinib (arm C; n = 111). All cohorts accepted patients with CLL/SLL who were older, had comorbidities, or were unsuitable for fludarabine/cyclophosphamide/rituximab (FCR) treatment. The primary endpoint was PFS by IRC assessment.
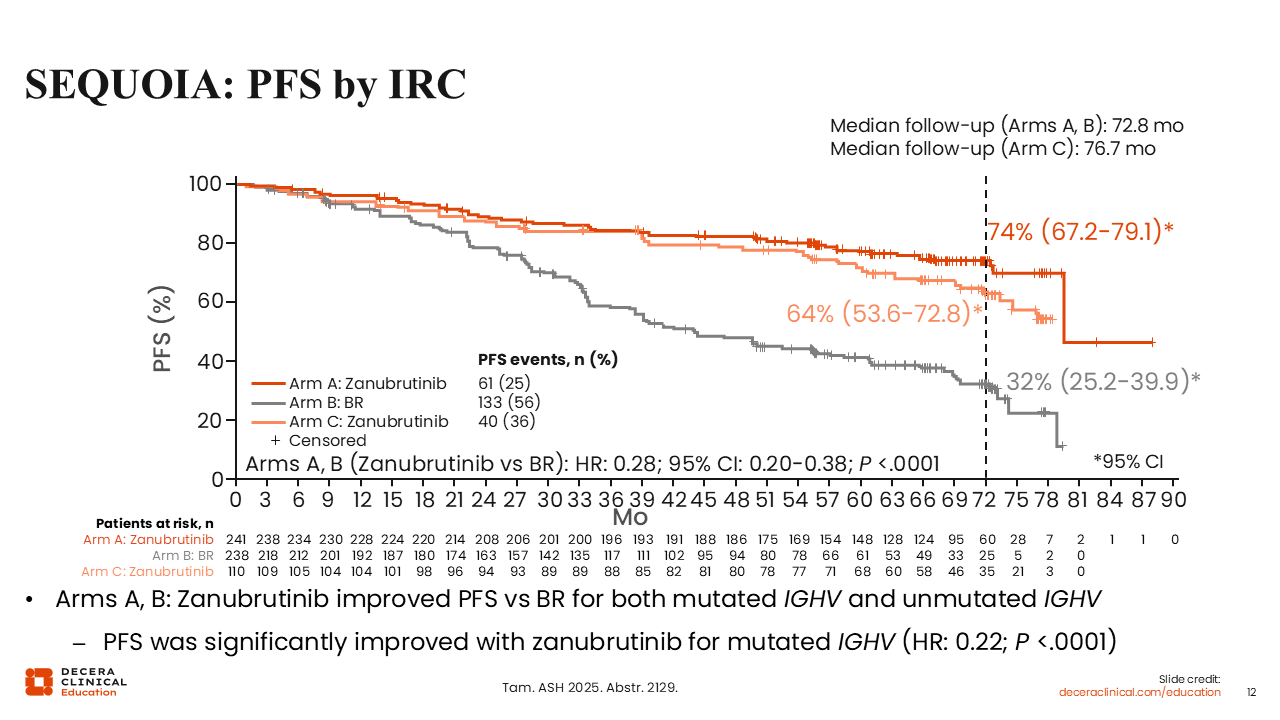
SEQUOIA: PFS by IRC
At the 6-year update, patients without del(17p) receiving continuous zanubrutinib therapy had significantly better PFS by IRC assessment (the primary endpoint) compared with patients receiving BR (HR: 0.28; P <.0001), with an estimated 72-month PFS rate of 74% vs 32%, respectively.25 In patients with del(17p) receiving zanubrutinib, the 72-month PFS rate was 64%.
In addition, PFS was improved with zanubrutinib over BR regardless of patient IGHV mutation status, and was significantly improved in IGHV unmutated disease, (HR: 0.22; P <.0001). In arm C, IGHV-mutated and IGHV-unmutated patients treated with zanubrutinib had similar PFS.
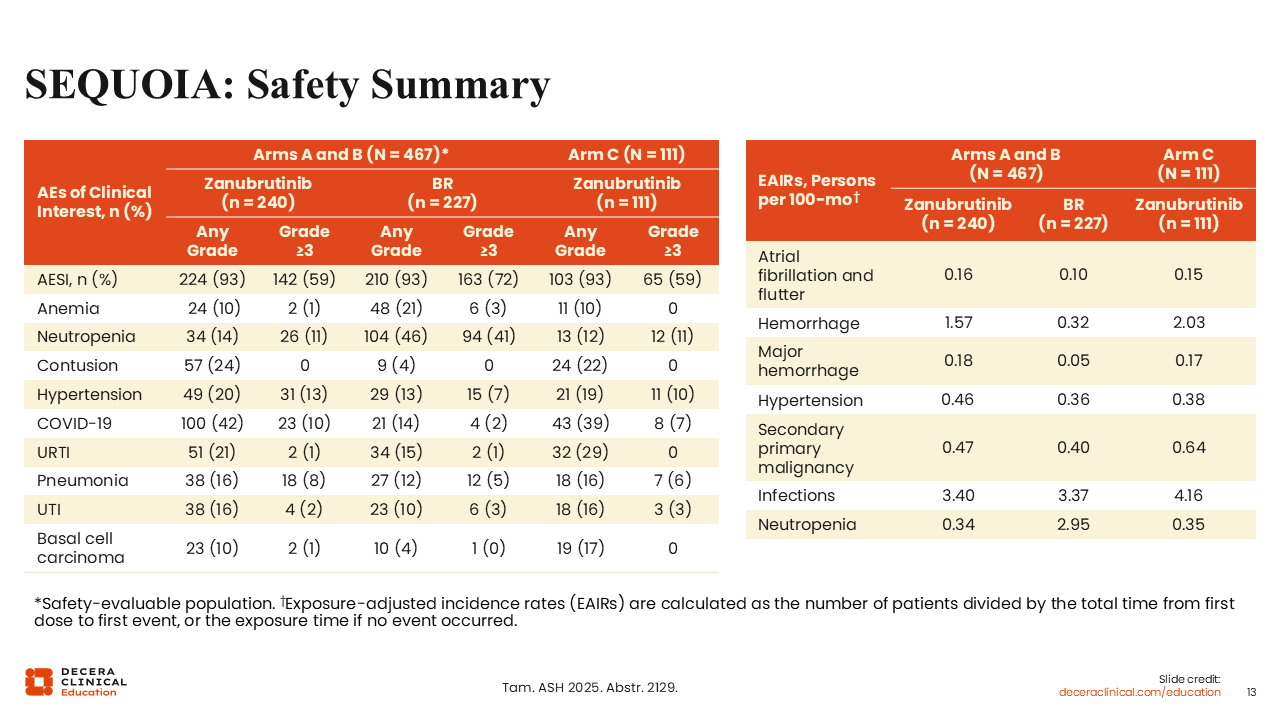
SEQUOIA: Safety Analysis Summary
The safety profile of zanubrutinib was encouraging. The rate of grade ≥3 treatment-emergent AEs was similar across all arms (72% in arm A; 74% in arm B; and 74% in arm C).25 Although any grade anemia (10% vs 21%) and neutropenia (14% vs 46%) occurred at lower rates in arm A compared to arm B, contusion (24% vs 4%), hypertension (20% vs 13%), and COVID-19 (42% vs 14%) occurred at higher rates. Atrial fibrillation also occurred at a slightly higher rate in arm A with zanubrutinib vs arm B with BR (exposure-adjusted incidence rates of 0.16 vs 0.10).
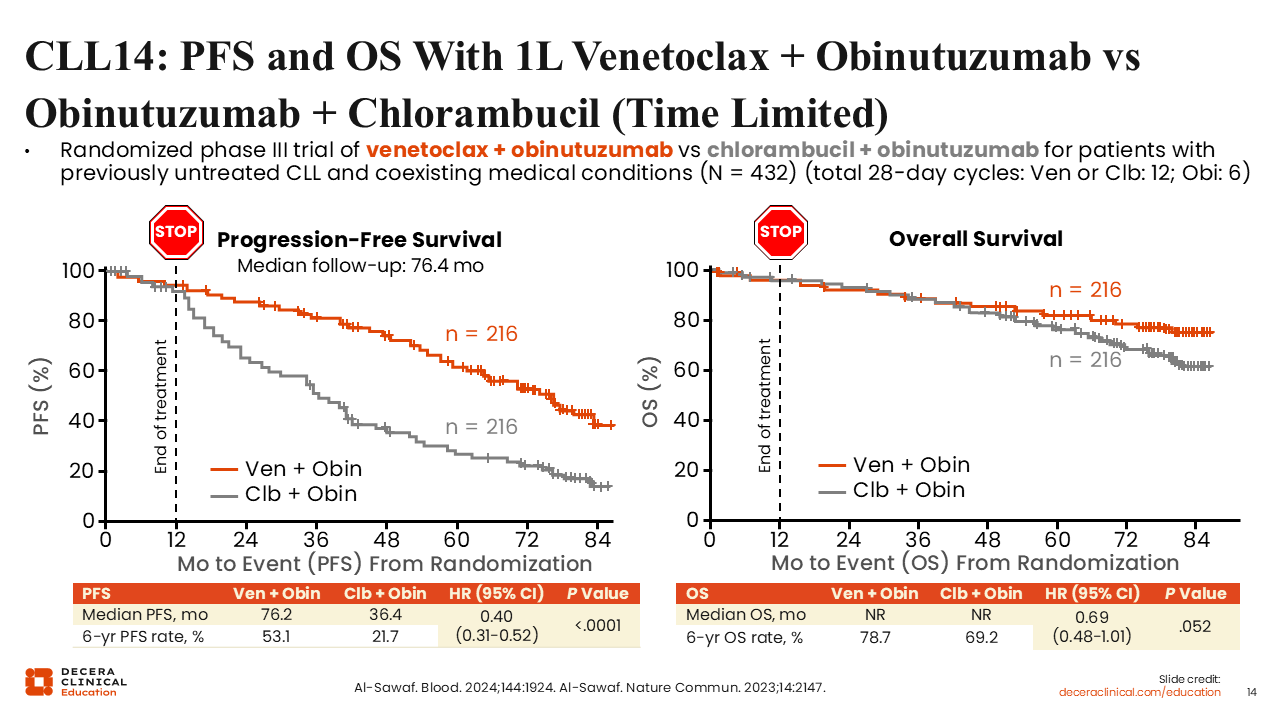
CLL14: PFS and OS With First-line Venetoclax + Obinutuzumab or Obinutuzumab + Chlorambucil (Time Limited)
To provide some context, I would also like to highlight data for other standard-of-care options in CLL/SLL, including data from the randomized phase III CLL14 trial evaluating time-limited therapy with the BCL-2 inhibitor venetoclax plus obinutuzumab vs OC in patients with previously untreated CLL and coexisting medical conditions (N = 432).27,28 Obinutuzumab was administered for six 28-day cycles, chlorambucil for 12 cycles, and venetoclax was administered with step-up dosing during the first 2 of 12 total cycles. The primary endpoint of the study was PFS, and a key secondary endpoint was OS.
After a median follow-up of 76.4 months, we see that the venetoclax plus obinutuzumab arm had a statistically significant prolonged median PFS vs the OC arm (PFS: 76.2 vs 36.4 months; HR: 0.4; P <.0001). By contrast, and although median OS was NR in both arms, no statistically significant difference in median OS was reported (HR: 0.69; P = .052).
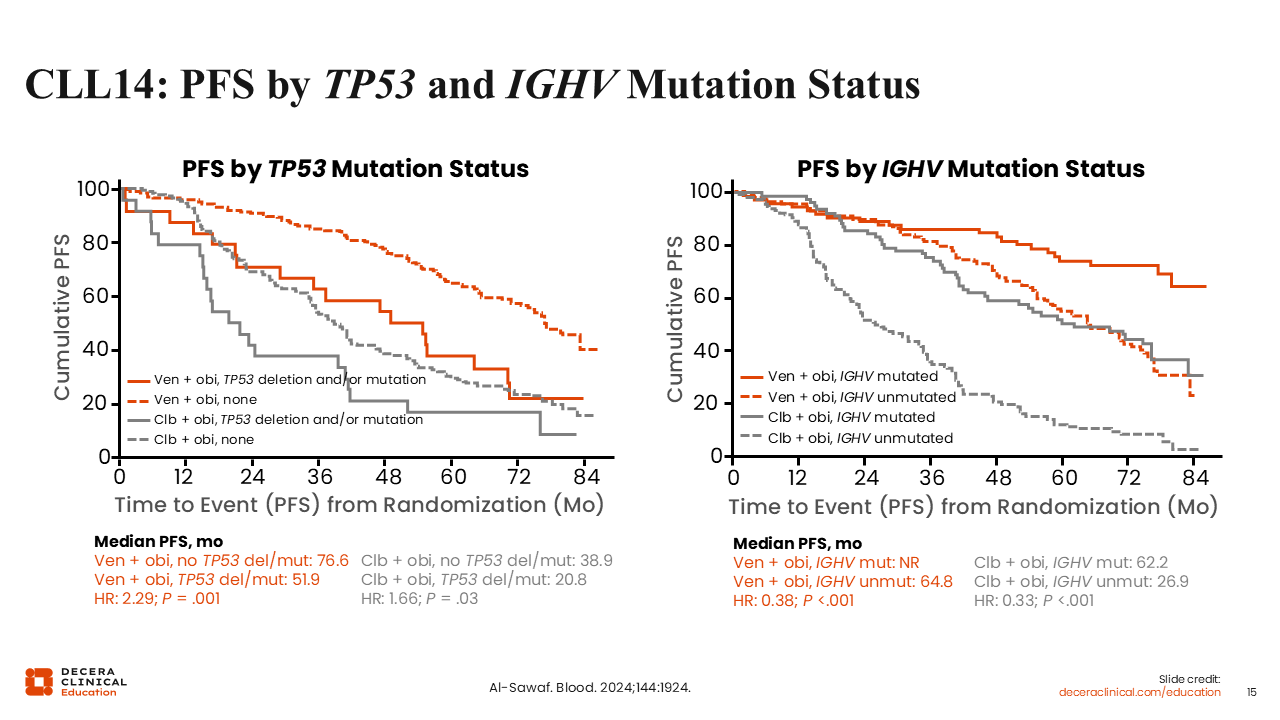
CLL14: PFS by TP53 and IGHV Mutation Status
Looking at PFS results for subgroups defined by TP53 and IGHV status, here we also see that limited duration treatment with venetoclax plus obinutuzumab vs OC resulted in a median PFS benefit across subgroups, particularly for the higher-risk subgroups of TP53 mutation status (51.9 vs 20.8 months, respectively; HR: 0.56) and IGHV unmutated status (64.8 vs 26.9 months, respectively; HR: 0.30).28
These data may also be useful to inform the prognostic impact of del(17p) and/or TP53 mutation on shorter PFS compared with the PFS in patients without del(17p) and/or TP53 mutation and in those with unmutated IGHV vs mutated IGHV for either of the treatment arms evaluated.
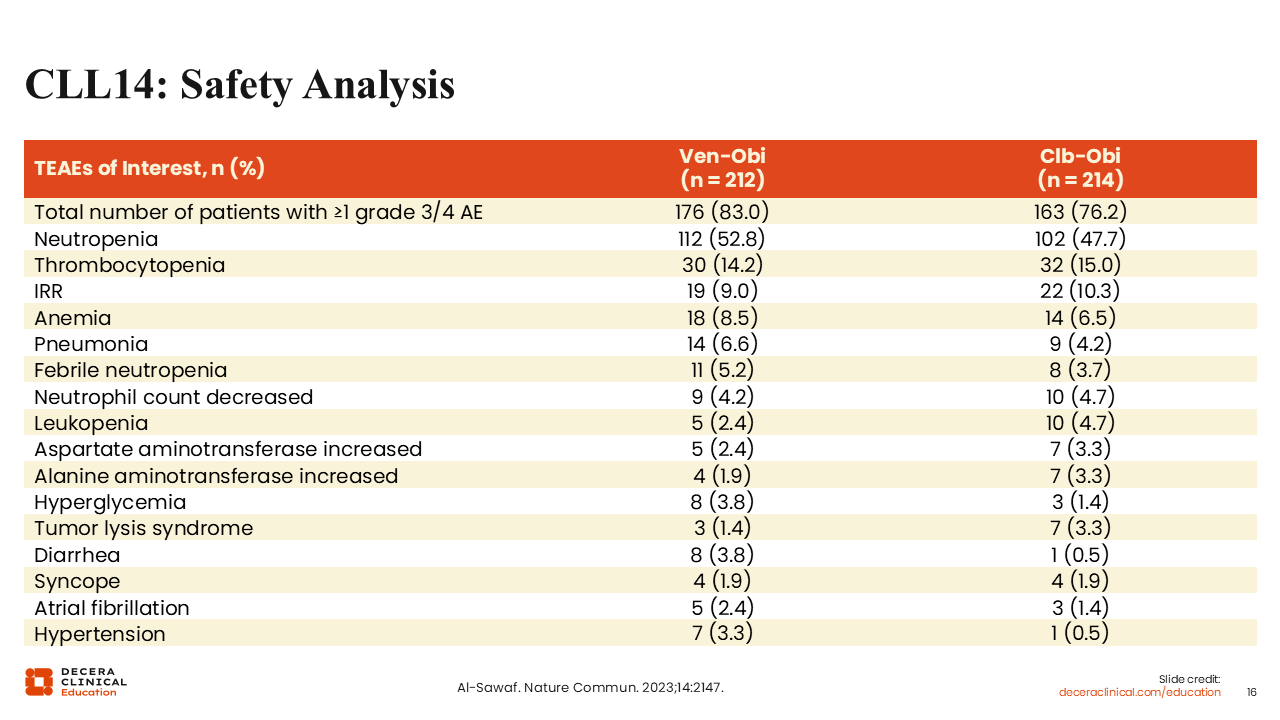
CLL14: Safety Analysis
The most common AEs in both treatment arms were hematologic and included neutropenia, thrombocytopenia, and anemia.27
I would also note here that tumor lysis syndrome (TLS) is a serious potential AE with venetoclax. Prophylaxis and step-up dosing of venetoclax when initiating therapy is required to reduce the risk of patients developing TLS as much as possible.
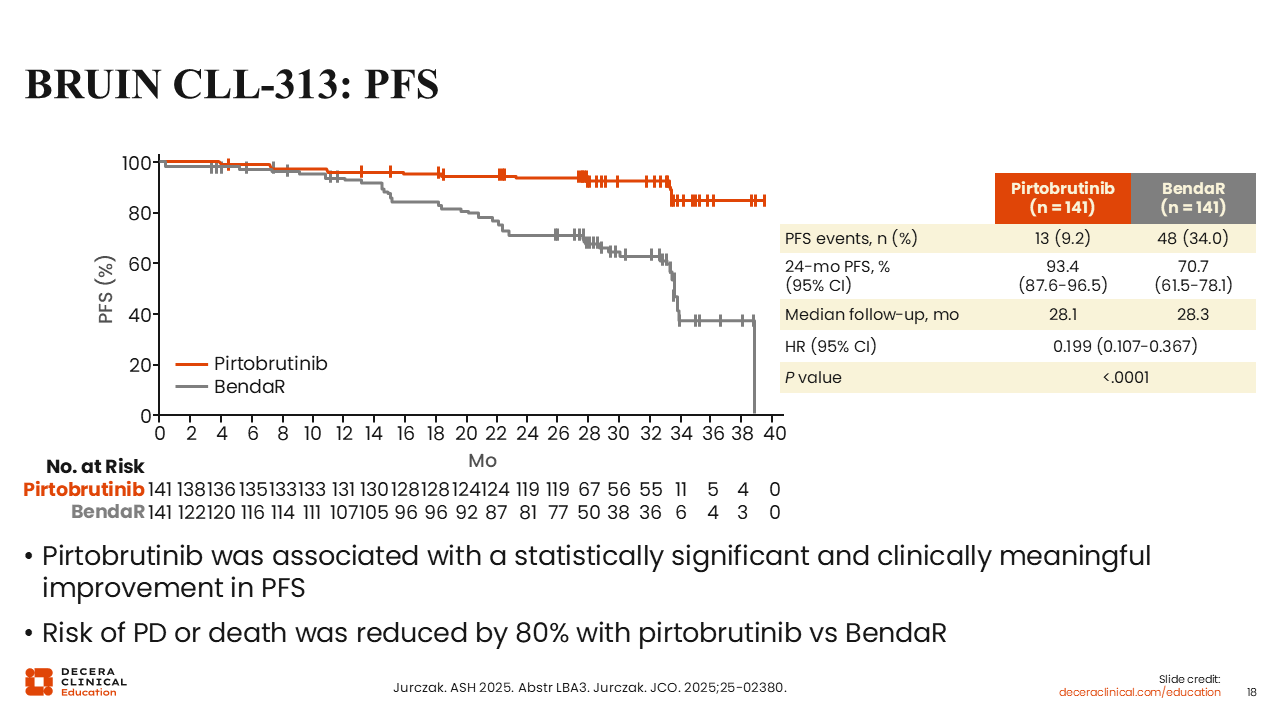
BRUIN CLL-313: First-line Pirtobrutinib (Continuous) vs Bendamustine/Rituximab (Fixed Duration)
At the 2025 American Society of Hematology (ASH) annual meeting, randomized data for the noncovalent BTK inhibitor pirtobrutinib as first-line treatment for CLL were reported.29,30 The phase III BRUIN CLL-313 study randomized 282 patients with previously untreated CLL to continuous pirtobrutinib treatment (n = 141) or fixed duration bendamustine plus rituximab (n = 141). Crossover was allowed after progressive disease in the control arm, and the primary endpoint was PFS.
BRUIN CLL-313: PFS
With a median follow-up of approximately 28 months, pirtobrutinib significantly improved PFS vs bendamustine plus rituximab (HR: 0.199; P <.0001).29,30 24-month PFS rate was 93.4% vs 70.7%. Improved PFS was observed across subgroups.
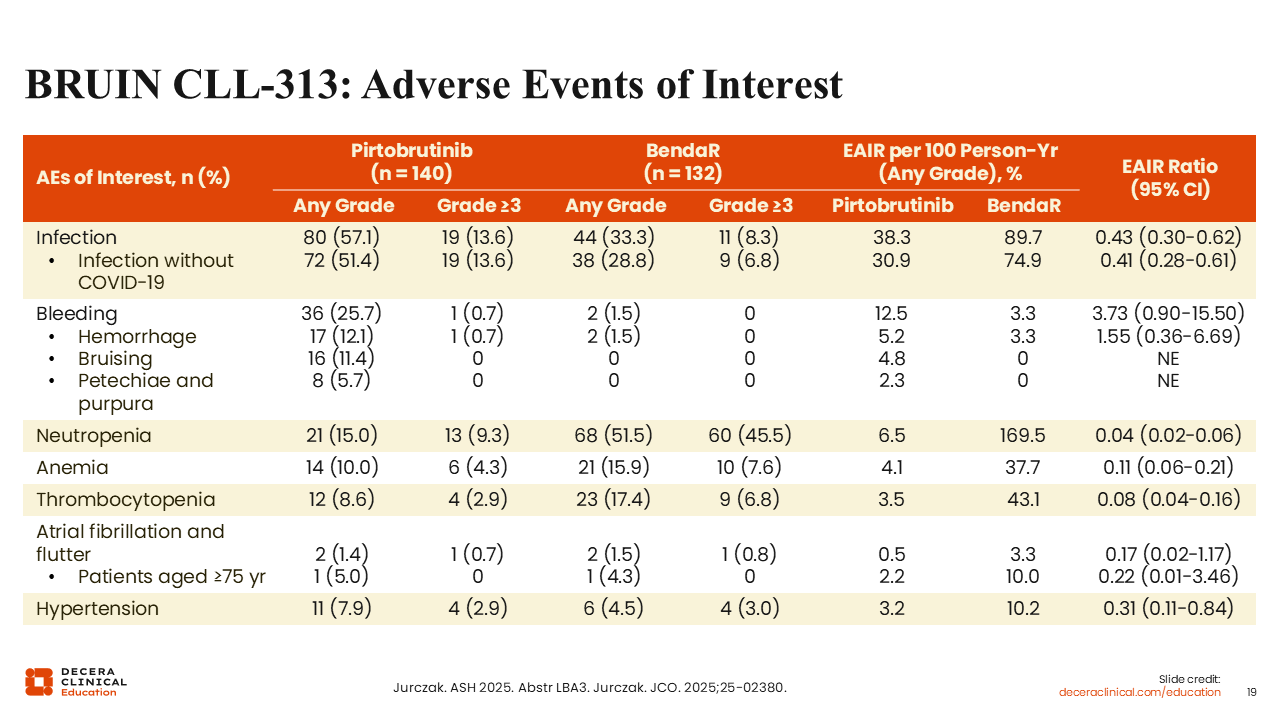
BRUIN CLL-313: Adverse Events of Interest
Pirtobrutinib was associated with higher incidence of infection (57% vs 33%) and bleeding (26% vs 1.5%) compared with bendamustine plus rituximab, but had lower incidence of cytopenias (eg, neutropenia: 15% vs 52%).29,30 Of note, rates of atrial fibrillation were similarly low in both arms (1.4% vs 1.5%).
BRUIN CLL-314: Pirtobrutinib vs Ibrutinib for Treatment-Naive and R/R CLL
Results for the phase III BRUIN CLL-314 trial of pirtobrutinib vs ibrutinib for both untreated and R/R CLL were also reported at the 2025 ASH annual meeting.31,32 This is the first randomized trial to directly compare covalent and noncovalent BTK inhibitors. Primary endpoints were the noninferiority of overall response rate (ORR) in the overall intent-to-treat population and the R/R population. Here we will focus on the outcomes in the treatment-naive population which were exploratory endpoints.
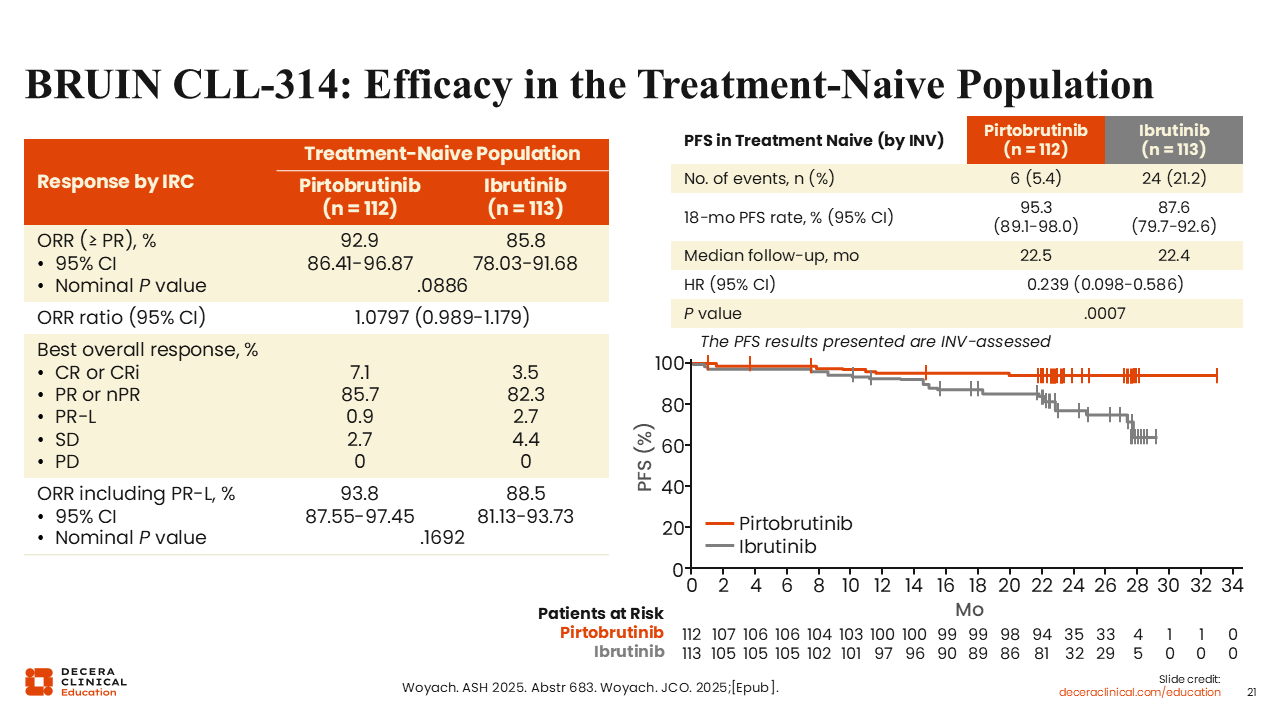
BRUIN CLL-314: ORR in the Treatment-Naive Population
There were 112 and 113 patients with treatment-naive disease randomized to the pirtobrutinib and ibrutinib arms, respectively.31,32 ORR assessed by IRC was improved with pirtobrutinib (93% vs 86%; P = .0886). At a median follow-up of approximately 22.5 months, investigator-assessed PFS was significantly improved (HR: 0.239; P = .0007); 18-month PFS rate was 95% vs 88%. Of note, PFS was most significantly improved in the treatment-naive population as compared to the intent-to-treat and R/R populations.
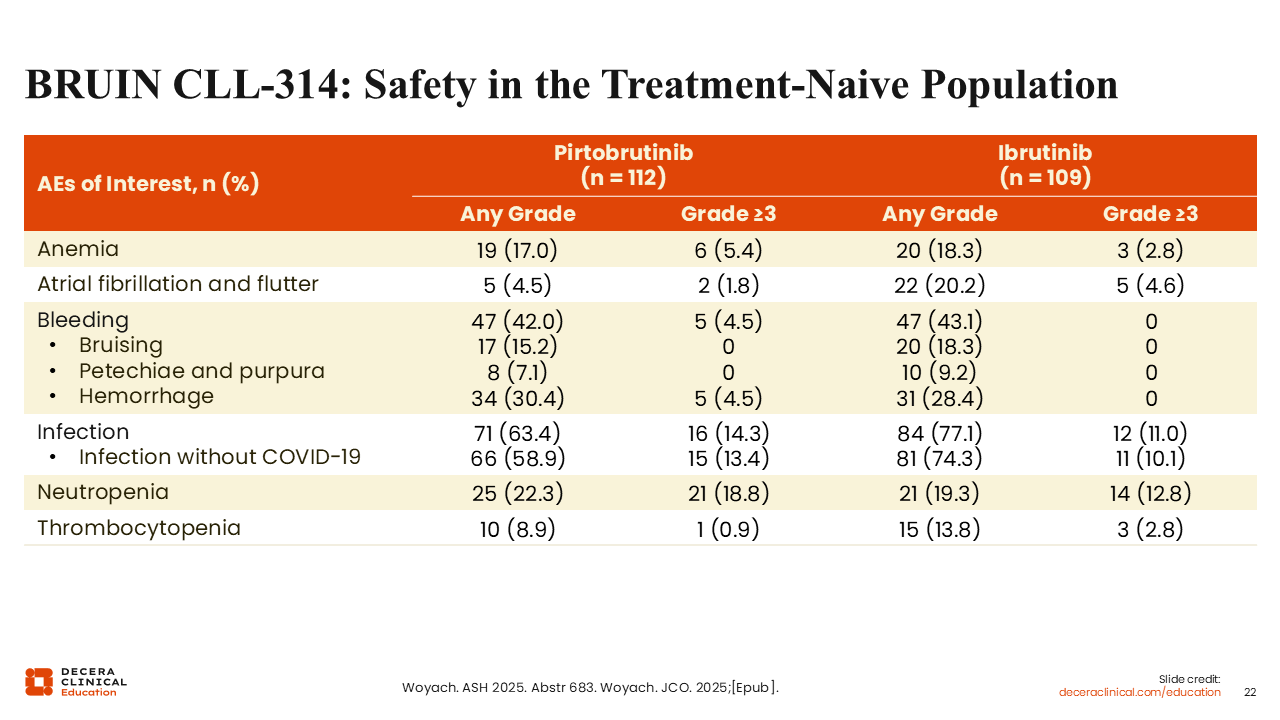
BRUIN CLL-314: Safety in the Treatment-Naive Population
Rates of cytopenias (eg, anemia: 17% vs 18%) and bleeding (42% vs 43%), in the treatment-naive population were similar between the pirtobrutinib and ibrutinib arms.31 However, pirtobrutinib was associated with lower rates of atrial fibrillation and flutter (5% vs 20%). Safety results reported in the treatment-naive population were similar to the overall population.