CME
On the Frontlines of CLL/SLL: International Perspectives on First-line Treatments for CLL/SLL
Physicians: Maximum of 1.00 AMA PRA Category 1 Credit™
Released: February 10, 2026
Expiration: August 09, 2026
Activity
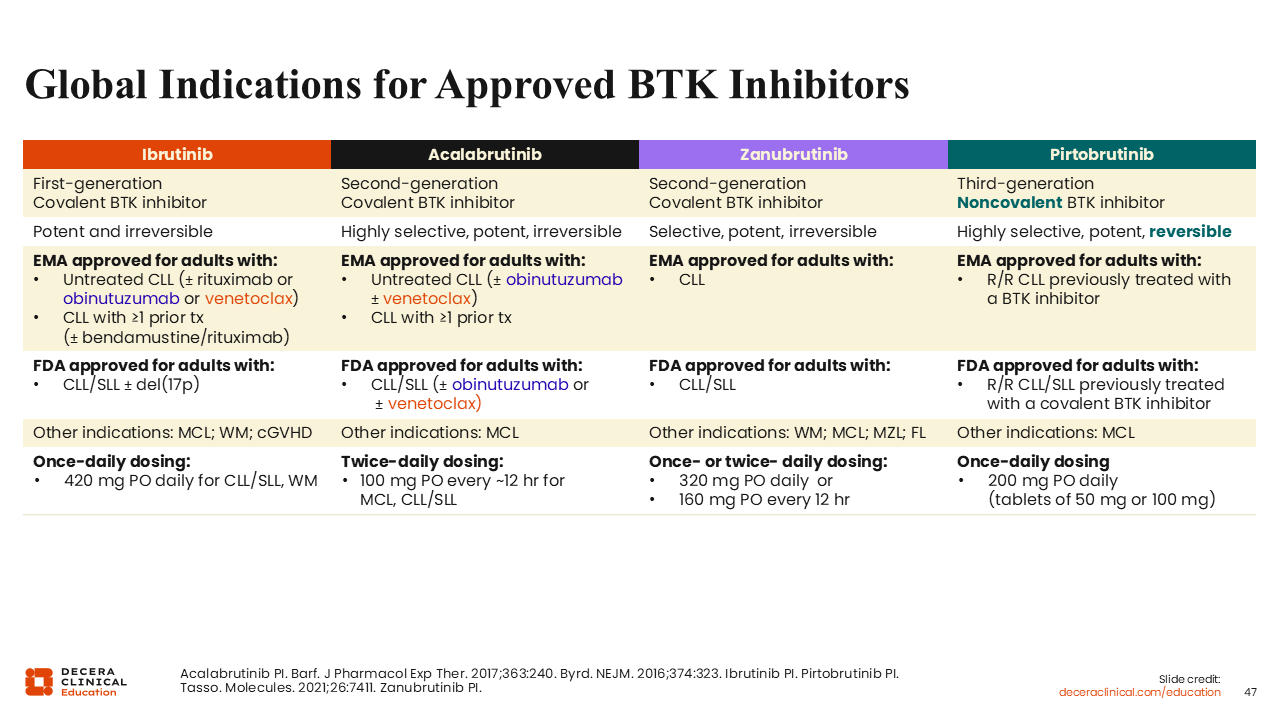
Global Indications for Approved BTK Inhibitors
As previously discussed, ibrutinib was the first-in-class BTK inhibitor available for the management of CLL and is currently approved as monotherapy in Europe and the United States for use in adult patients with CLL/SLL with or without del(17p), and in Europe in various combinations including with rituximab with or without bendamustine, or with obinutuzumab, or with venetoclax.37,47
Acalabrutinib is available in Europe and the United States as monotherapy for patients with previously untreated or relapsed CLL.44,48 In Europe, acalabrutinib is approved in combination with venetoclax and/or obinutuzumab in patients with previously untreated CLL.44 Acalabrutinib plus venetoclax is approved by the FDA,48 and the triplet combination is also listed in the NCCN guidelines.49
Zanubrutinib is approved in Europe and the United States as monotherapy for the treatment of patients with CLL.50,51
Pirtobrutinib is approved in Europe for the treatment of adults with R/R CLL who have been previously treated with a BTK inhibitor.3 In the United States, it is similarly approved after a covalent BTK inhibitor.4
It is worth mentioning that these agents are all approved for treating mantle cell lymphoma and some are approved for treating other B-cell malignancies. Zanubrutinib is the most broadly approved for these, including Waldenström’s macroglobulinemia, marginal zone lymphoma, and follicular lymphoma.
Which Is the Best Initial Therapy in CLL?
Cross-trial comparison of survival outcomes data is limited by many factors that contribute to variability in trial conduct. In clinical practice, however, we must do cross-trial comparisons because in the first-line setting, we have limited head-to-head clinical trial comparisons for the various treatment options. CLL17 is the only trial comparing fixed duration and continuous targeted treatment options (ibrutinib vs ibrutinib plus venetoclax vs venetoclax plus obinutuzumab) and BRUIN CLL-314 is the only other trial comparing targeted treatments (pirtobrutinib vs ibrutinib). Both studies currently have limited follow up. Looking at a chart of the approximate 4-year PFS rates of the different treatment regimens and trials to date in this disease setting, we see that there is no single best initial treatment in CLL.17,22,26,35,40,52-58
Available data also suggest that PFS varies mostly in relation to the age and fitness of the patient receiving treatment for CLL/SLL. For instance, 4-year PFS rates with percentages in the mid-70s are often seen in trials with a median patient age of approximately 70 years, whereas 4-year PFS rates with percentages in the mid-80s are associated with trials with a median patient age of 60 years (ie, in younger and more physically fit patients).
Ultimately, many of these first-line treatments achieve good outcomes, so multiple other factors influence our treatment choice including patient preference, comorbid conditions, toxicities, and efficacy data in patient subgroups.
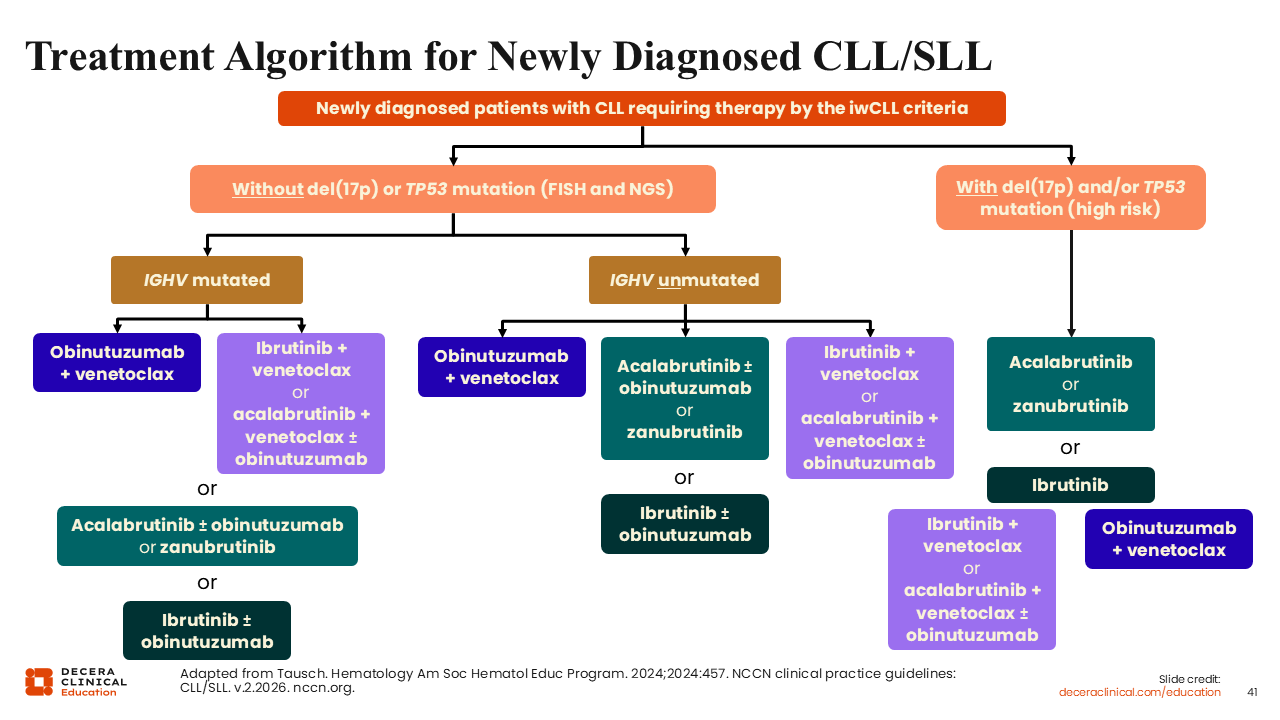
Treatment Algorithm for Newly Diagnosed CLL/SLL
The recommended treatment algorithm from the 2024 ASH annual meeting’s educational program shows treatment options based on priority.52 Patient genetic information (eg, del[17p], TP53, and IGHV mutation status) is the most important factor to consider when selecting therapy for a patient with newly diagnosed CLL.
For patients with mutated IGHV status, time-limited treatments are preferred. Time-limited therapy with venetoclax-based regimens is preferred because the efficacy in patients with mutated IGHV status has been shown to be superior to continuous BTK inhibitor therapy. That said, continuous therapy is also an option. For patients with unmutated IGHV, there is no preference between the 2 treatment types.
For now, for patients with a del(17p) and/or TP53 mutation, our recommendation is for continuous BTK inhibitor treatment rather than time-limited treatment. Generally, acalabrutinib and zanubrutinib are preferred over ibrutinib because of a better safety profile with similar efficacy (acalabrutinib) or improved efficacy (zanubrutinib) in the R/R setting.59,60 We can extrapolate that this efficacy extends to the first-line setting as well, although head-to-head comparison data in this setting are not available.
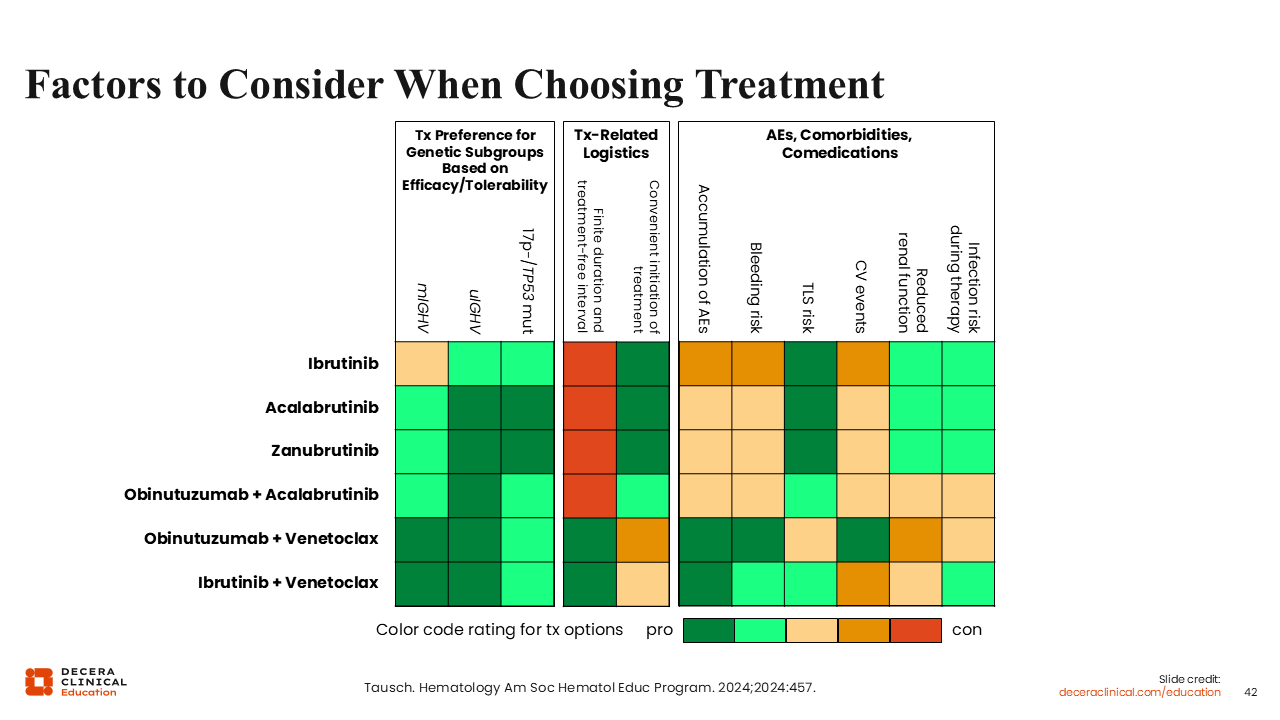
Factors to Consider When Choosing Treatment
However, the optimal choice of treatment for individual patients may further change based on comorbidities, treatment toxicities, treatment logistics, comedications, and patient preferences.
The chart shown here displays the weighted pros and cons for each treatment based on efficacy and tolerability for genetic subgroups of patients on the left (as discussed in the previous section), as well as treatment-related logistics in the middle, and AEs, comorbidities, and comedications on the right.52
For example, the recommended treatment for a patient with no del(17p) or TP53 mutation and mutated IGHV would be obinutuzumab plus venetoclax, ibrutinib plus venetoclax, or acalabrutinib plus venetoclax with or without obinutuzumab. However, if the patient also has a history of cardiovascular events, this chart suggests that obinutuzumab plus venetoclax may be the preferred option.
Of note, the addition of obinutuzumab to acalabrutinib plus venetoclax increases risk of infection and we are unsure of when to prefer the addition over acalabrutinib plus venetoclax without obinutuzumab.
In general, continuous BTK inhibitor therapy is more convenient to administer than venetoclax-based treatments because no intravenous infusions are involved and usually there is no need to monitor for TLS. By contrast, venetoclax regimens have a much lower risk for causing bleeding episodes and less accumulation of AEs because of the fixed-duration treatment.
Other factors to consider that are not shown in this chart are drug availability and cost. For instance, ibrutinib plus venetoclax is not approved by the FDA for use in the United States, but it can be used off-label.47,49 Other countries have approved only some of these BTK inhibitors for treating CLL. Also, the cost of continuous BTK inhibitor therapy is often much higher than the cost of fixed-duration venetoclax-based treatments.
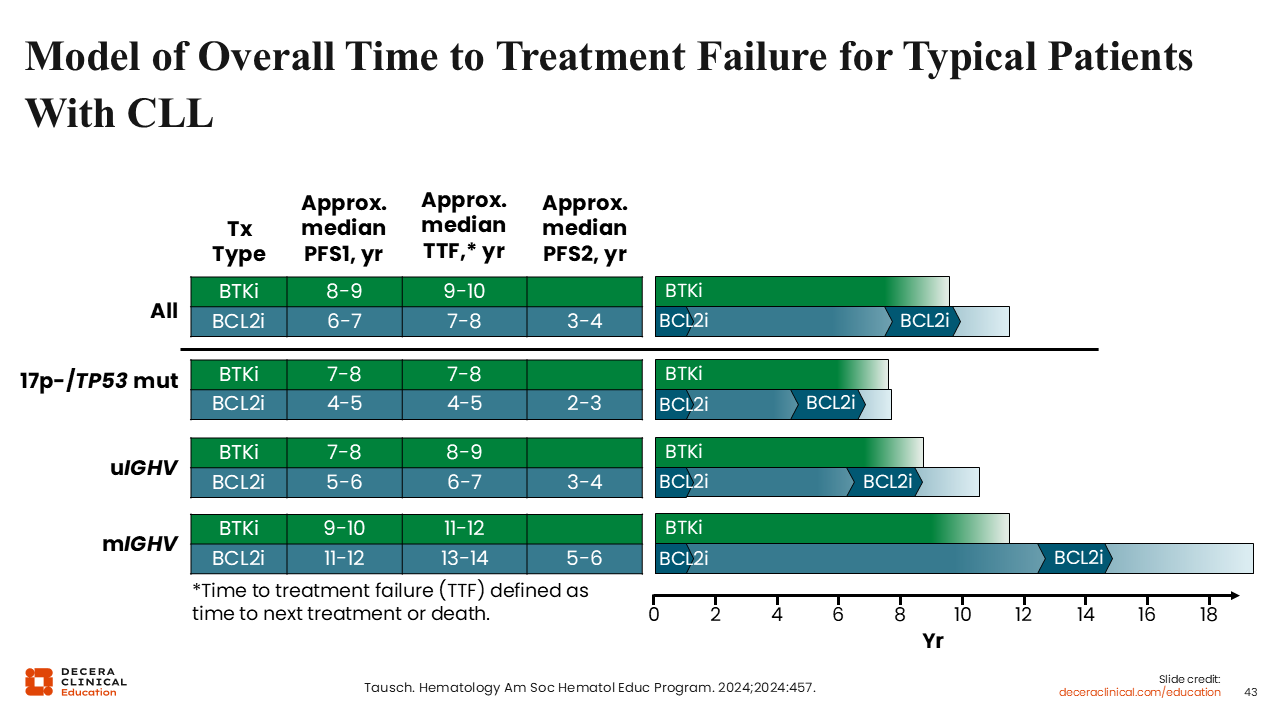
Model of Overall Time to Treatment Failure for Typical Patients with CLL
This figure illustrates estimated treatment outcomes over time for patients in 3 genetic subgroups.52 This is an estimation because we do not have clinical trial data for this. Continuous BTK inhibitor treatment, shown in green, often achieves good outcomes, with time to treatment failure of approximately 8-10 years when second-line treatment is required. Time-limited BCL-2 inhibitor treatment is shown in the dark blue color followed by light blue between treatments. For the general patient population, we observe that the first PFS event after first-line BCL-2 inhibitor therapy occurs sooner than BTK inhibitor treatment, at approximately 6-7 years. However, we have the option of retreatment with BCL-2 inhibitor therapy, which can extend the time to a second PFS event for an additional 3-4 years. Retreatment outcomes for patients initially treated with both a BTK inhibitor and BCL-2 inhibitor are also promising.62
As mentioned, treatment preference varies across the genetic subgroups, with a preference for BTK inhibitor for patients with a del(17p) and/or TP53 mutation. The 2 treatment options appear balanced regarding time to treatment failure for patients with unmutated IGHV. By contrast, patients with mutated IGHV tend to have longer times to progression with time-limited BCL-2 inhibitor-based treatment than with continuous BTK inhibitors.
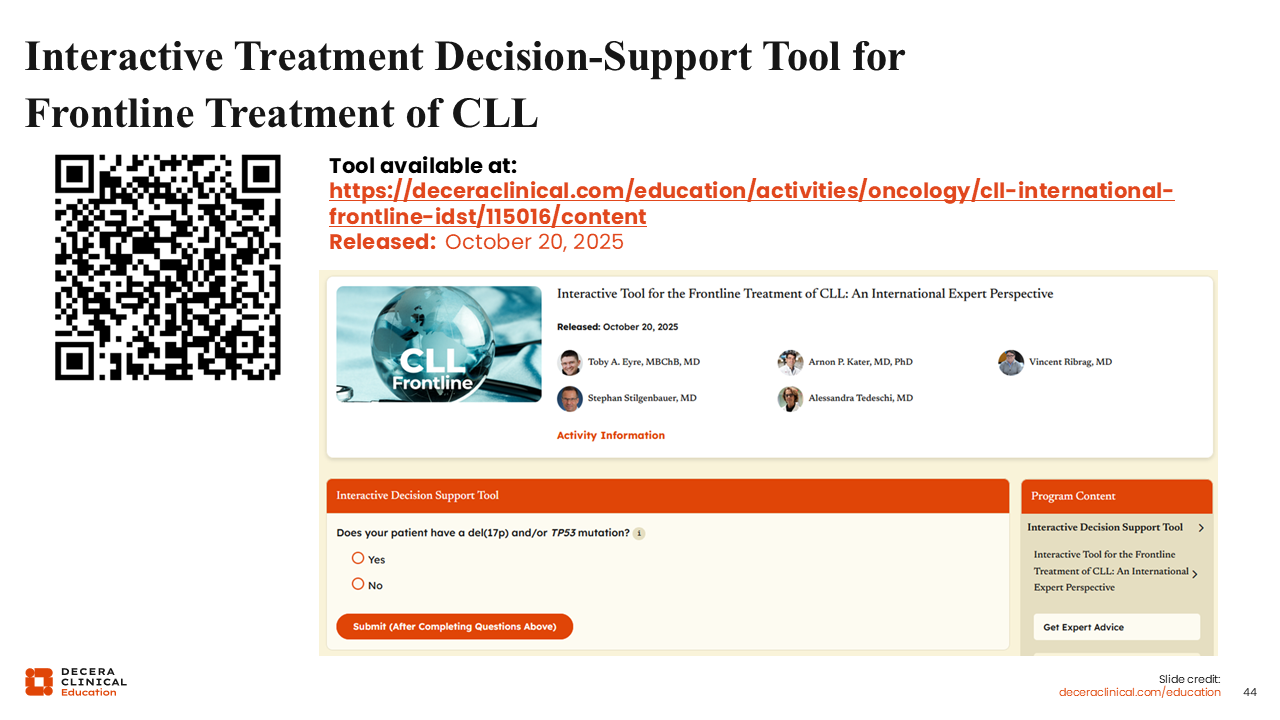
Interactive Treatment Decision Support Tool for Frontline Treatment of CLL
I along with 4 other EU-based experts have worked with Decera Clinical Education to develop an interactive treatment decision support tool to help healthcare professionals (HCPs) choose treatments for their patients with newly diagnosed CLL. The details of a patient case can be entered into the tool, including comorbidities, patient fitness, and risk factors, such as 17p deletion, TP53 mutation, and IGHV status. HCPs can then see the 5 expert recommendations based on those specific presenting characteristics. We will discuss some example patient cases and examine the expert recommendations we receive from the tool.
Patient Case 1: Del(17p) and Hypertension
Let’s consider treatment options for a 70-year-old male patient who presents with newly diagnosed CLL. Del(17p) is detected in his workup, and he has a history of arterial hypertension that is currently well-controlled with medication.
For this patient, I would begin treatment with acalabrutinib or zanubrutinib, because these options provide the best efficacy and good tolerability in this population.
If we enter the patient characteristics into the interactive treatment decision support tool, we receive a range of BTK inhibitor recommendations. Most experts recommend BTK inhibitor monotherapy, primarily acalabrutinib and zanubrutinib—some specify 1 over another—but some also would consider ibrutinib with venetoclax as an alternative based on the recent CLL17 study data.
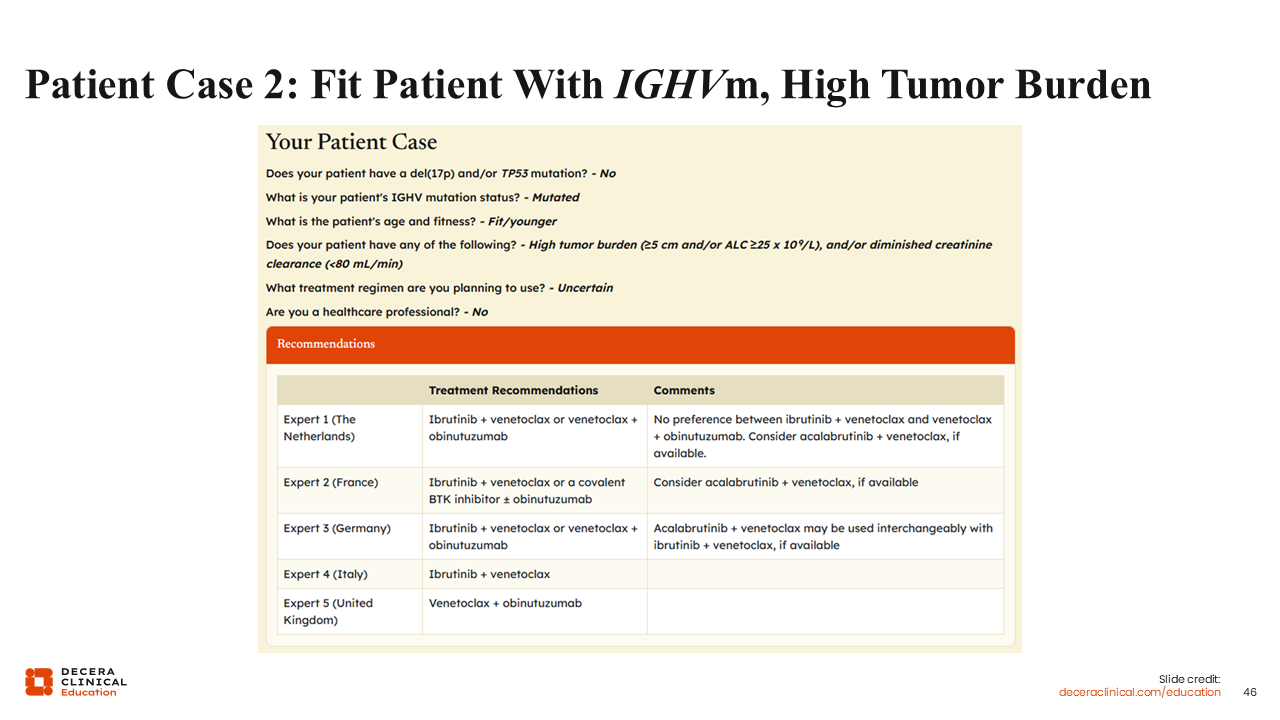
Patient Case 2: Fit Patient with IGHVm, High Tumor Burden
For a second example, let’s consider a 63-year-old female patient who presents with newly diagnosed CLL. No del(17p) or TP53 mutation is detected in her workup, but she does have mutated IGHV. Her tumor size is 5 cm, but she is otherwise generally fit.
I recommend ibrutinib or acalabrutinib combined with venetoclax, or venetoclax with obinutuzumab. We see somewhat of a consensus in expert recommendations in the tool with all recommending 1 or more of these regimens. One faculty also suggests a covalent BTK inhibitor with or without obinutuzumab.
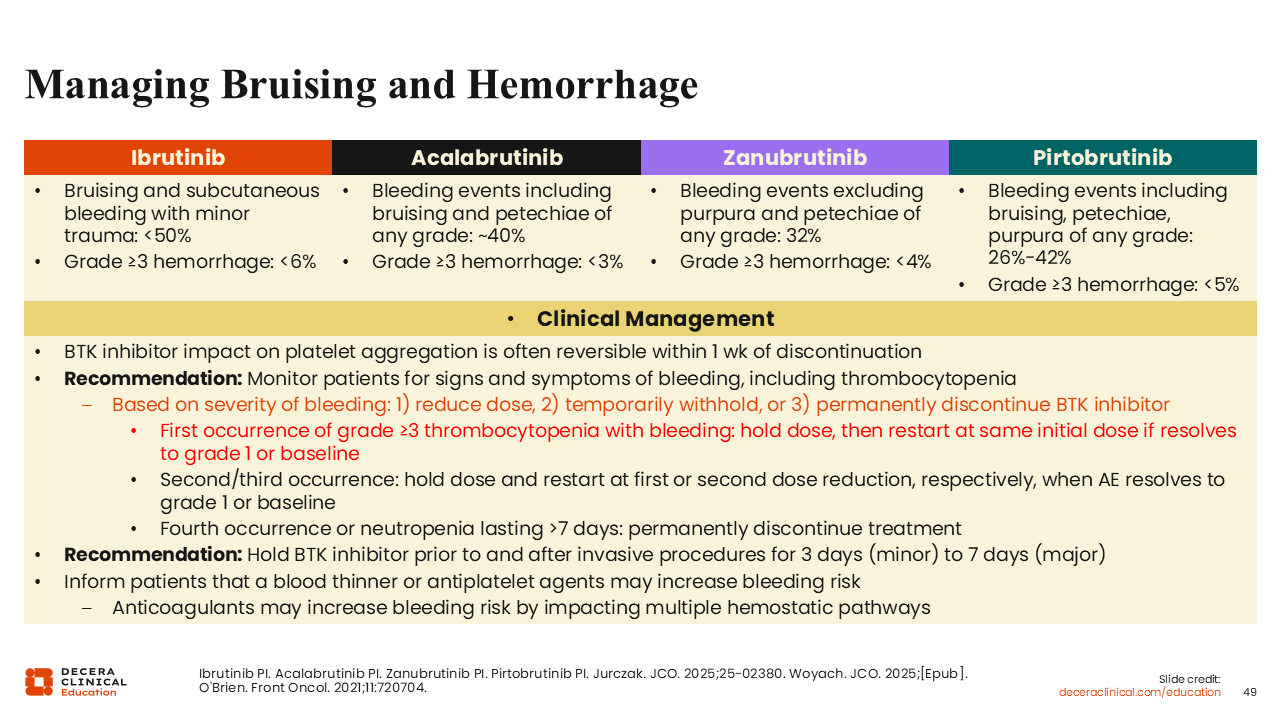
Managing Bruising and Hemorrhage
General recommendations for managing the hemorrhagic AEs associated with the BTK inhibitor drug class include monitoring patients for signs of bleeding and, at the first occurrence of grade ≥3 hemorrhage, to withhold the dose until the AE resolves to grade 1 or baseline.3,4,37,44,47,48,50,51 For subsequent occurrences of a hemorrhagic AE, it is recommended to hold treatment and reduce the dose or permanently discontinue treatment after 4 occurrences or a long-lasting episode of neutropenia.
Managing Atrial Fibrillation and Cardiac Arrhythmias
Atrial fibrillation is also a known class-related AE with BTK inhibitors, but it does not necessarily require complete discontinuation of the BTK inhibitor drug class.3,4,37,44,47,48,50,51 Cardiology consultation is recommended for patients experiencing atrial fibrillation. Low-grade occurrences can potentially be controlled with rate control–directed therapeutic interventions without halting BTK inhibitor therapy. However, this should be managed on a case-by-case basis.
Patient Case 3: Managing Atrial Fibrillation
Let’s consider a specific case of atrial fibrillation. A 68-year-old female patient is newly diagnosed with CLL and a del(17p) is detected on her workup. She has a history of arterial hypertension and has been experiencing fatigue and frequent infections. She is started on 100 mg twice daily oral acalabrutinib. Six months after beginning treatment, she develops grade 1 atrial fibrillation. What is the best approach to managing this patient going forward?
In most cases, patients with low-grade (grade ≤2) atrial fibrillation while receiving acalabrutinib can continue treatment. A cardiologist should evaluate whether rate control–directed therapeutic interventions should be initiated and whether anticoagulation treatment should be considered. Patients who develop atrial fibrillation while receiving acalabrutinib should be thoroughly assessed for the risk of thromboembolic disease.
If her atrial fibrillation were high grade, this would warrant dose interruption or reduction until resolution of the AE to grade 1 or baseline. Atrial fibrillation is not an indication to discontinue BTK inhibitors as long as it is well controlled.
Managing Other Covalent BTK Inhibitor–Related AEs
For the management of other BTK inhibitor–related AEs, my general recommendation is to monitor patients for known AEs of interest and interrupt or reduce dosing as necessary before discontinuing BTK inhibitors.63 In addition, prophylaxis to reduce the incidence of infections may also be considered.
Patient and Caregiver Discussions: What AEs to Expect/Monitor During BTK Inhibitor Treatment
It is important to inform patients and their caregivers about what AEs they should expect and how to monitor for their occurrence.
I usually start discussing the potential for diarrhea, because it is 1 of the first AEs that patients notice. I make it clear to them that diarrhea can be of low grade and is usually temporary.
I also discuss the potential for bleeding and bruising, and that this is usually not very harmful, but that they should watch for serious or prolonged bleeding incidents.
I tell my patients that AEs like headache, arthralgia, diarrhea, and bleeding episodes may be low grade and do not require intervention or can be managed without disrupting treatment. However, we also discuss options for screening patients for more serious AEs such as atrial fibrillation.
Understanding Challenges to Adherence and Persistence With Oral BTK Inhibitors
HCPs should also be aware of the challenges that patients face for adhering to BTK inhibitor treatment.
The cost of these agents can be a barrier for patients who have copayments for their medical care. We should discuss AE expectations with patients, ensuring they know that symptoms from their disease should improve soon after beginning treatment, but that AEs that make them feel worse can occur and need to be managed. They should feel confident in calling their HCP with any concerns without delay to avoid worsened AEs.
Patient education, shared decision-making regarding patient care, and involving an interdisciplinary team are critical to maximizing the benefit attained with BTK inhibitor treatment in patients with CLL.