CME
On the Frontlines of CLL/SLL: International Perspectives on First-line Treatments for CLL/SLL
Physicians: Maximum of 1.00 AMA PRA Category 1 Credit™
Released: February 10, 2026
Expiration: August 09, 2026
Activity
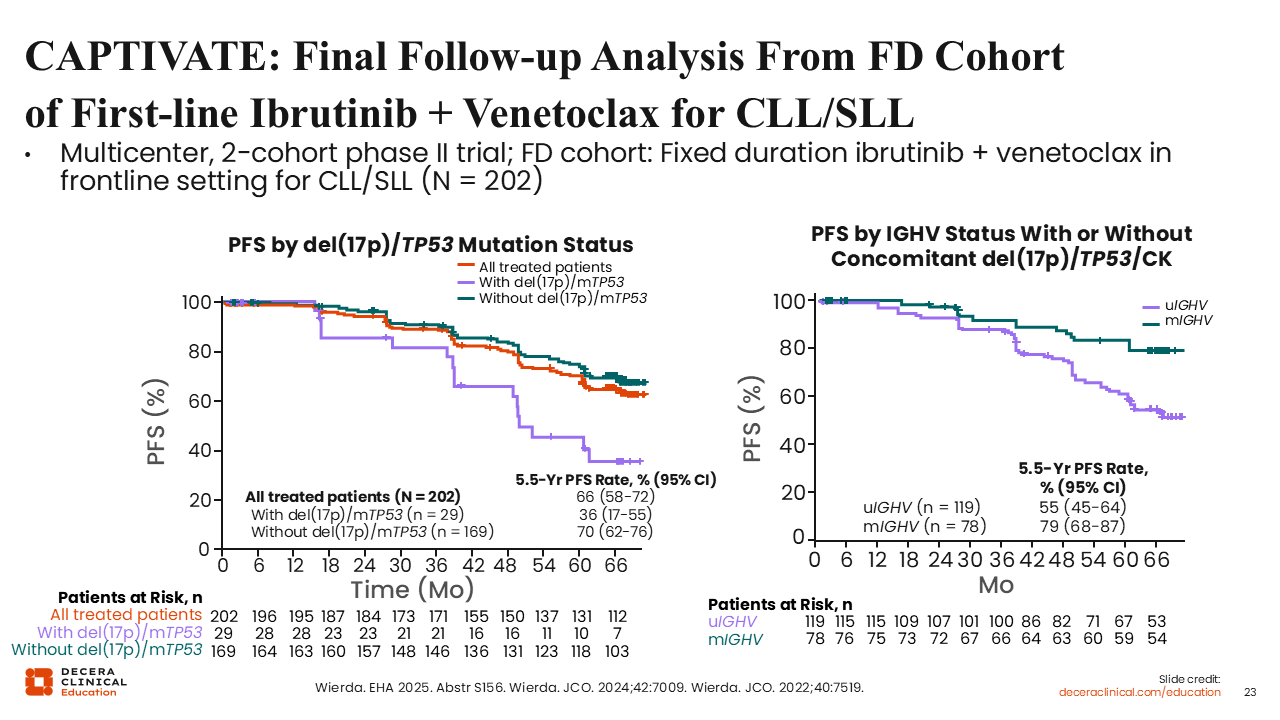
CAPTIVATE: Final Follow-up Analysis From Fixed-Duration Cohort of First-line Ibrutinib + Venetoclax for CLL/SLL
Now we will focus on the available data for combinations of the 2 treatment modalities that revolutionized CLL/SLL treatment, namely BTK inhibitors and BCL-2 inhibitors.33,34
The phase II CAPTIVATE trial evaluated ibrutinib plus venetoclax as first-line therapy in young and physically fit patients with CLL/SLL (N = 202). The study showed good efficacy for the drug combination, with a 5.5-year PFS rate of 66% (95% CI: 58-72) and OS of 97% (95% CI: 93-99) for all treated patients.34 There also appears to be some prognostic impact of del(17p) status, TP53 status, complex karyotype, and IGHV status regarding PFS. In patients with unmutated IGHV, the 5.5-year PFS rate was 55% compared to 79% in patients with mutated IGHV.
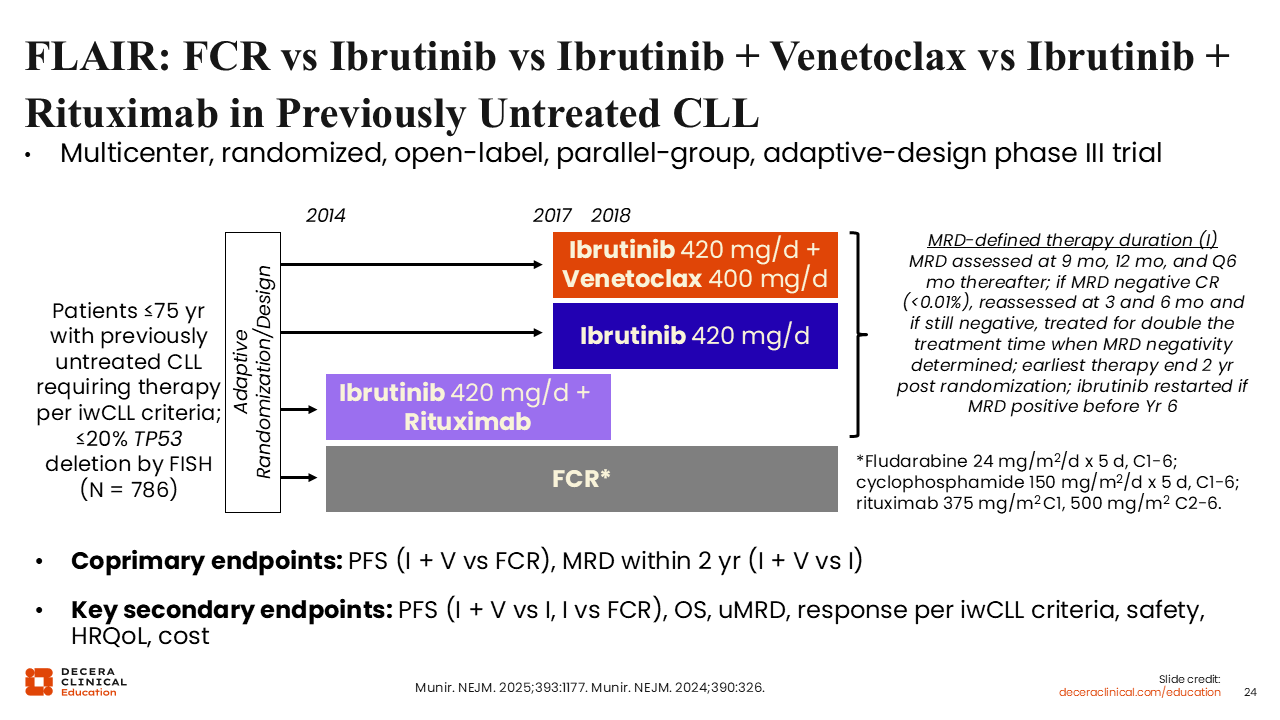
FLAIR: FCR vs Ibrutinib vs Ibrutinib + Venetoclax in Previously Untreated CLL
The randomized phase III FLAIR study evaluated the ibrutinib plus venetoclax combination vs ibrutinib monotherapy; IR; or FCR as first-line therapy in young and physically fit patients with CLL.35,36 The coprimary endpoint of the study was PFS and measurable residual disease (MRD). Here, I focus on the comparison of ibrutinib plus venetoclax (n = 260) vs FCR (n = 263).
Of importance, ibrutinib plus venetoclax was not given with a fixed treatment duration but was instead guided by MRD status. MRD in peripheral blood (PB) and bone marrow (BM) was assessed with multiparameter flow cytometry 9 months after randomization, then PB MRD was measured at 12 months and every subsequent 6 months. If a patient was found to have a complete response and be PB MRD negative (MRD <0.01%), the patient was reassessed for PB MRD 3 months later and was reassessed for PB MRD and BM MRD after an additional 3 months. At that point, if both PB and BM MRD were negative, the treatment time was doubled, up to the first negative PB MRD assessment in the sequence. The earliest therapy completion time was 2 years following randomization. Ibrutinib treatment could be reinitiated if the patient became MRD positive before Year 6. Note that this treatment approach is only experimental. Ibrutinib plus venetoclax is only approved by the EMA with a fixed duration of 12 months of use as combined therapy (three 28-day cycles of ibrutinib followed by 12 cycles of combination therapy).37
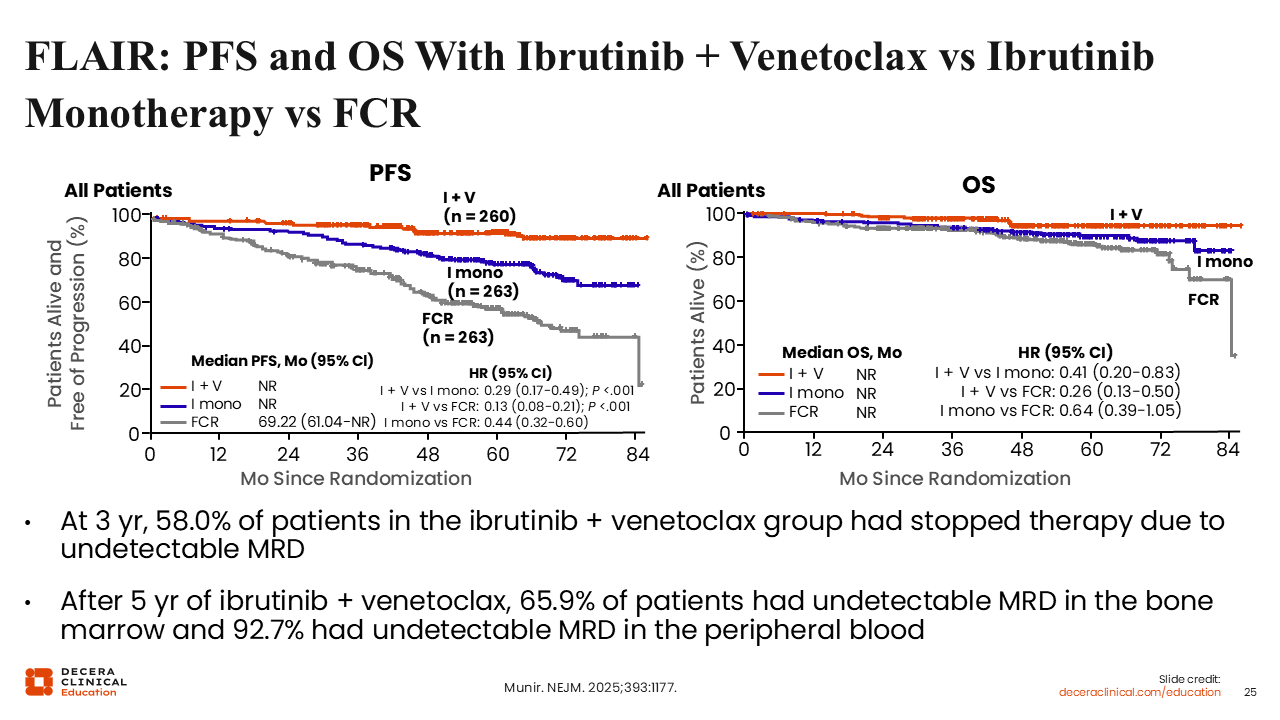
FLAIR: PFS and OS With Ibrutinib + Venetoclax vs FCR
MRD-guided treatment in the FLAIR trial resulted in significantly longer PFS (HR: 0.13; 95% CI: 0.08-0.21; P <.001) and OS (HR: 0.26; 95% CI: 0.13-0.50) in patients treated with ibrutinib plus venetoclax vs FCR.36 However, this was at the cost of prolonged treatment with ibrutinib plus venetoclax, because at 3 years only 56% of patients had stopped their treatment because of undetectable MRD. The duration of the combined treatment was nearly triple the EMA-approved duration for the majority of patients, leading to good outcomes, but also prolonged toxicity from treatment. Of note, MRD-guided reinstitution of treatment in the ibrutinib plus venetoclax arm was not considered a PFS event, so the PFS data reflect both continuous treatment and two lines of treatment in this arm. Additionally, MRD-guided ibrutinib plus venetoclax is not currently approved by the EMA or FDA and has not been directly compared to the approved fixed duration regimen.
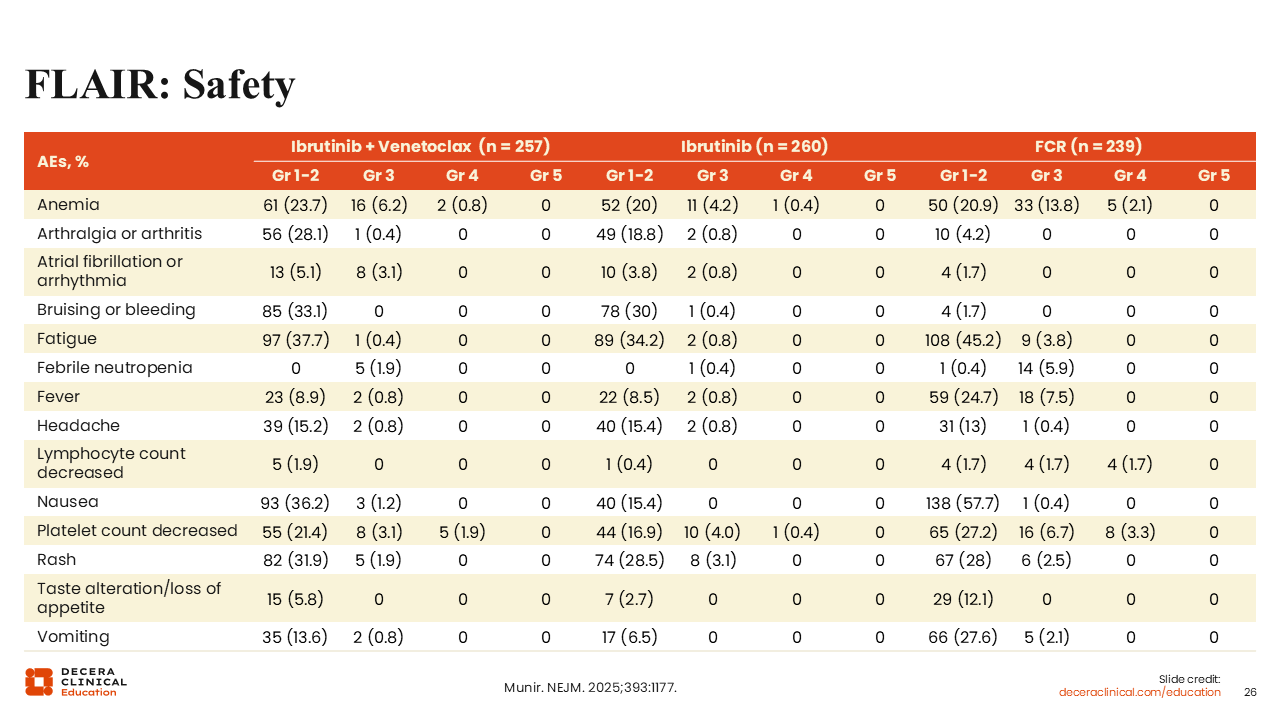
FLAIR: Safety
The safety data show that ibrutinib plus venetoclax was better tolerated than FCR and high-grade AEs were infrequent.36 In both the CAPTIVATE and FLAIR trials and across all grades of AEs, we see that ibrutinib plus venetoclax is generally well tolerated. The class-associated atrial fibrillation/arrhythmia of grade 1/2 was observed in 5.1% of patients and grade ≥3 in 3.1% of patients treated with ibrutinib plus venetoclax.
GLOW: Ibrutinib + Venetoclax vs OC in Previously Untreated CLL
The randomized phase III GLOW study evaluating ibrutinib plus venetoclax vs OC in patients with previously untreated CLL led to marketing authorization of this combination in Europe.37-39 Patients in the experimental arm received 3 lead-in cycles of ibrutinib monotherapy followed by 12 cycles of ibrutinib plus venetoclax combination therapy. Patients in the standard-of-care arm received OC for six 28-day cycles. The trial included patients who were older and/or physically unfit with previously untreated CLL and no del(17p) alteration. The primary endpoint was PFS by IRC assessment.
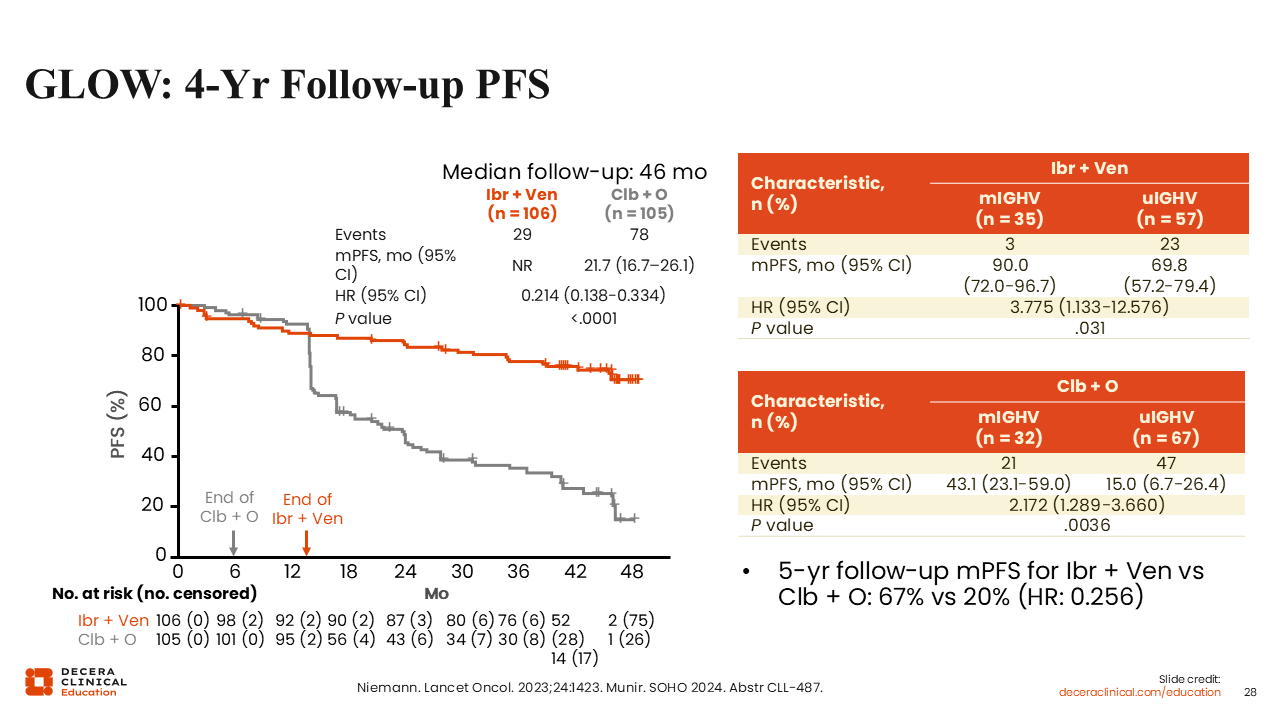
GLOW: 4-Year Follow-up PFS
The GLOW trial demonstrated statistically significantly longer median PFS by IRC assessment at the 4-year follow-up in favor of ibrutinib plus venetoclax vs OC (NR vs 21.7 months, respectively; HR: 0.214; P <.0001) with similar improvement at the 5-year PFS update (67% vs 20%, respectively; HR: 0.256).40,41 A PFS benefit was also seen in patients with mutated and unmutated IGHV, and although unmutated IGHV carried a worse prognosis overall, those patients still did substantially better with ibrutinib plus venetoclax treatment vs OC treatment regarding median PFS (69.8 vs 15.0 months, respectively).40
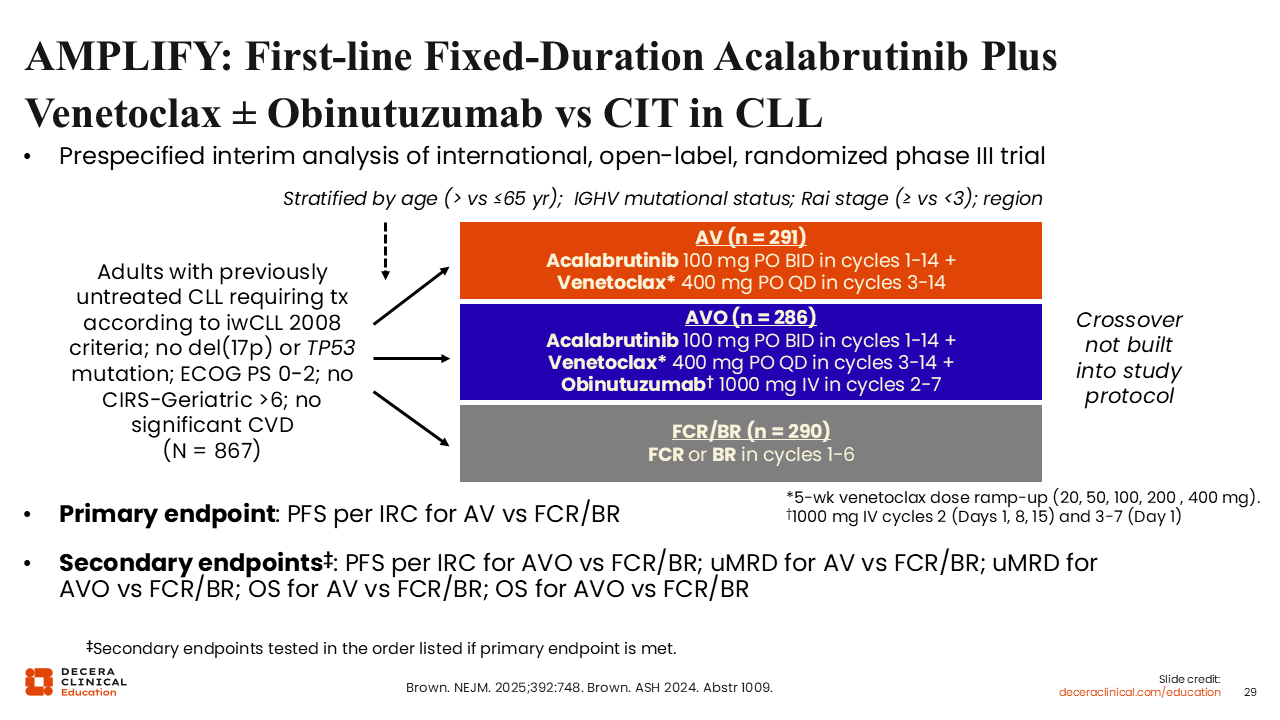
AMPLIFY: First-line Fixed-Duration Acalabrutinib + Venetoclax ± Obinutuzumab vs Chemoimmunotherapy in CLL
The interim results from the randomized phase III AMPLIFY trial evaluating acalabrutinib plus venetoclax with or without obinutuzumab vs FCR or BR in patients with previously untreated CLL and no del(17p) or TP53 mutation, but who required treatment according to 2008 International Workshop on Chronic Lymphocytic Leukemia criteria, were presented at the ASH 2024 annual meeting.42,43 The primary endpoint of the study was PFS by IRC assessment for acalabrutinib plus venetoclax vs FCR or BR.
Unlike the GLOW trial, patients in AMPLIFY could have some comorbidities, but they were fit enough to receive either FCR or BR in the standard-of-care arm. The median patient age in AMPLIFY was 61 years, which is 10 years younger than it was for patients enrolled in the GLOW trial, where the median patient age was 71 years.39,42
The age of patients included in these 2 trials is of consequence because it may affect treatment outcomes.
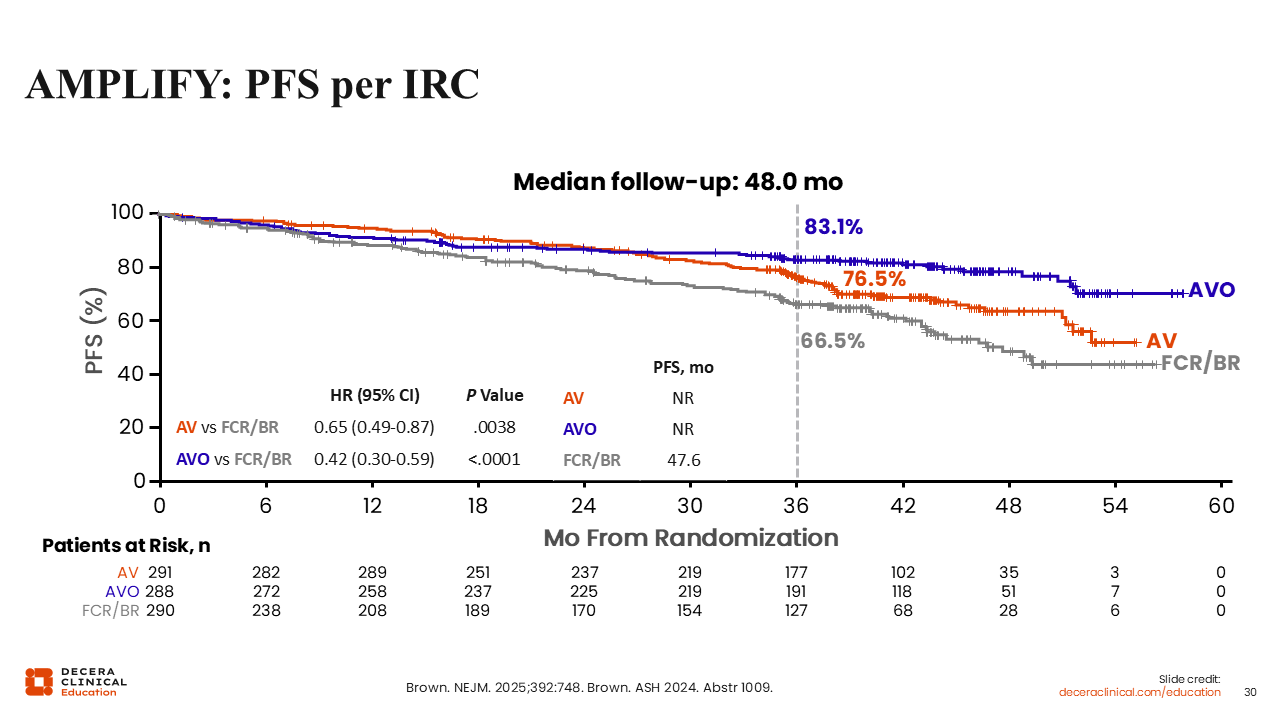
AMPLIFY: PFS per IRC
Treatment with acalabrutinib plus venetoclax resulted in superior median PFS vs FCR or BR (NR vs 47.6 months, respectively; HR: 0.65; P = .0038). Moreover, the benefit in median PFS was further enhanced when obinutuzumab was added to the acalabrutinib plus venetoclax arm (NR vs 47.6 months with FCR or BR; HR: 0.42; P <.0001).42
AMPLIFY: PFS Subgroup Analyses
Subgroup analyses in the AMPLIFY trial also showed that the combination of acalabrutinib plus venetoclax was beneficial in patients with unmutated IGHV, improving the median PFS to 51.5 months vs 43.3 months with FCR or BR (HR: 0.69). Here again, adding obinutuzumab further extended the benefit in median PFS vs FCR or BR (NR vs 43.3 months; HR: 0.35).42
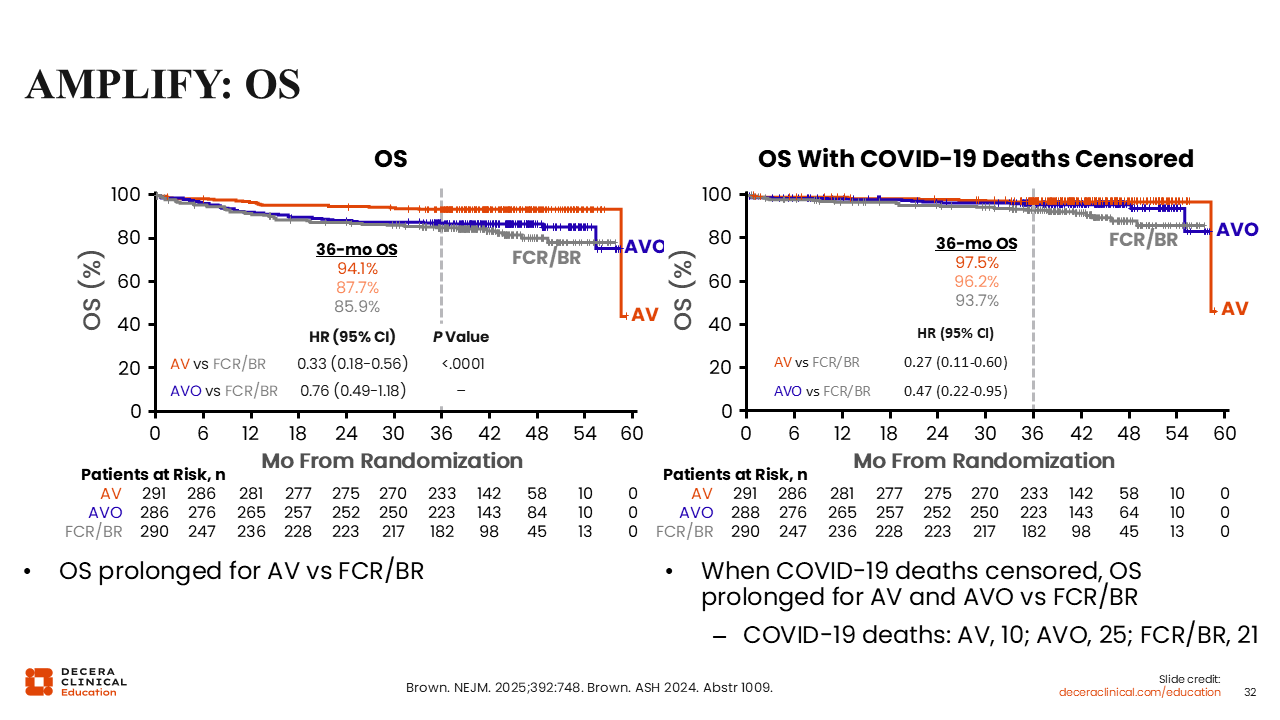
AMPLIFY: OS
Regarding the OS results, it is important to note that the trial was conducted during the COVID-19 pandemic, so the acalabrutinib, venetoclax, and obinutuzumab and FCR/BR arms were particularly affected by an increase of COVID-19–related deaths.
When deaths related to COVID-19 are censored in the OS analysis, the outcome of the acalabrutinib, venetoclax, and obinutuzumab and FCR/BR arms is improved, whereas the outcome of the acalabrutinib/venetoclax arm is not affected because of a lower rate of COVID-19–related deaths in that arm.42
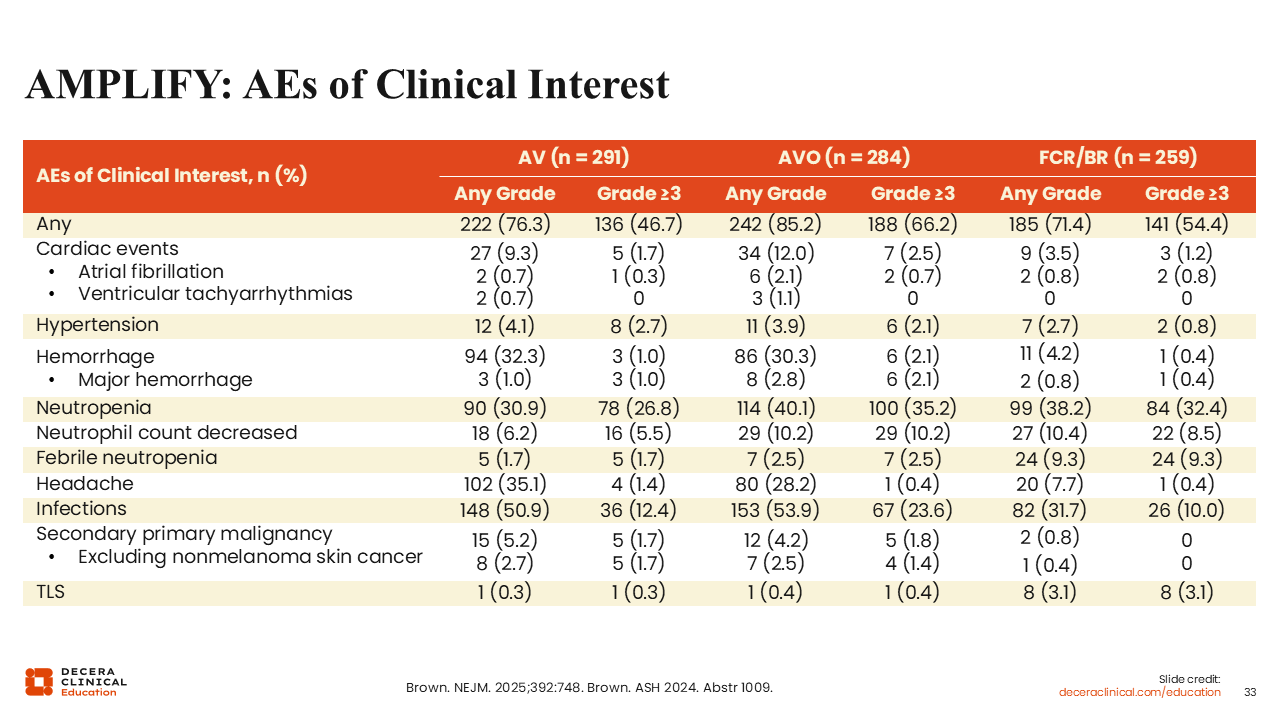
AMPLIFY: AEs of Clinical Interest
The AEs of interest in the acalabrutinib-containing arms included cardiac events (eg, atrial fibrillation/ ventricular tachyarrhythmias), hemorrhage, neutropenia, and infections. With the exception of neutropenia/neutrophil count decrease and infections, these AEs of interest were primarily grade 1/2.42 Of note, the frequency of neutropenia and infections was greater with the addition of obinutuzumab. By contrast, the FCR or BR arm showed higher rates of hematologic toxicity, with decreased neutrophil counts, thrombocytopenia, and febrile neutropenia overall.
AMPLIFY: Summary
The recently EMA approved combination of acalabrutinib, venetoclax, with or without obinutuzumab provides another valuable treatment option of finite duration for our patients.44 Acalabrutinib with venetoclax was also recently approved by the FDA for adults with CLL/SLL.48 The combination of acalabrutinib, venetoclax, and obinutuzumab leads to improved efficacy but also increased toxicity and may be an option for fit patients. The place of acalabrutinib/venetoclax and acalabrutinib/venetoclax/obinutuzumab as compared to ibrutinib/venetoclax, obinutuzumab/venetoclax, and continuous BTK inhibitor therapy may be settled with longer follow up of AMPLIFY.
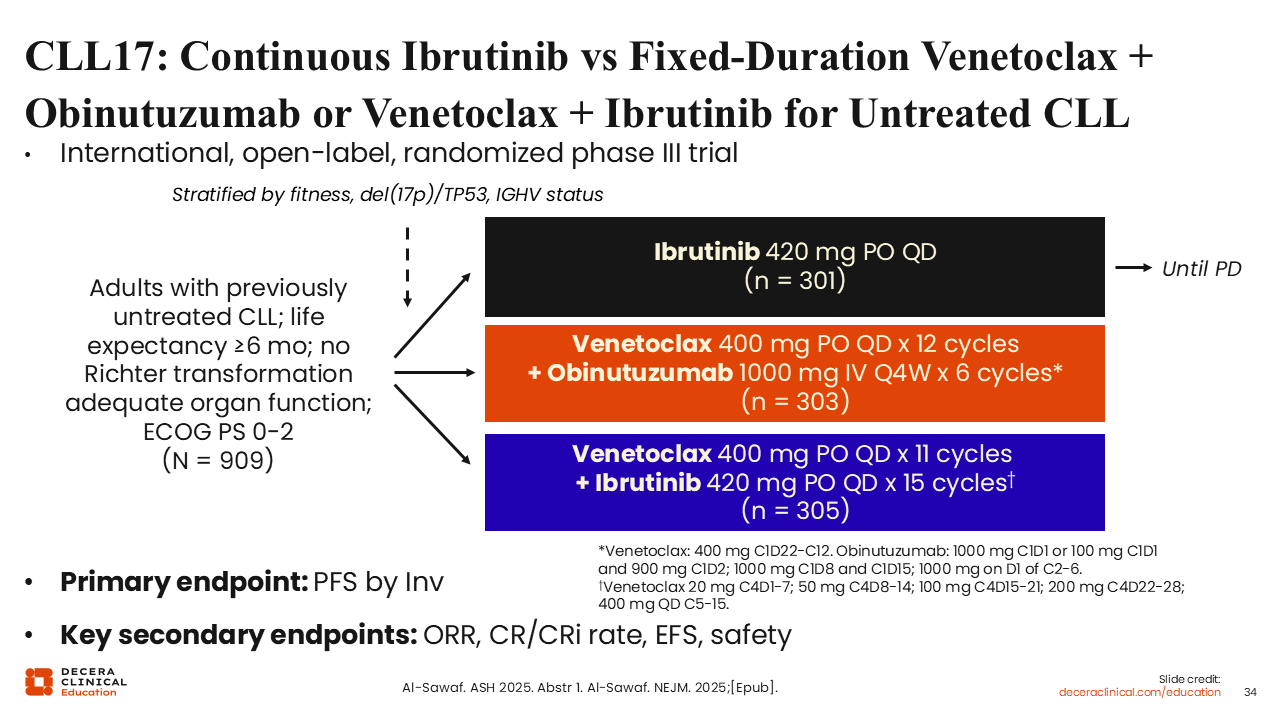
CLL17: Continuous Ibrutinib vs Fixed-Duration Venetoclax + Obinutuzumab or Venetoclax + Ibrutinib for Untreated CLL
Results from the open-label, randomized phase III CLL17 trial evaluating continuous ibrutinib vs fixed-duration venetoclax plus obinutuzumab or venetoclax plus ibrutinib for untreated CLL were presented at the 2025 ASH annual meeting.45,46 There has been no randomized study directly comparing fixed-duration vs continuous treatment methods. The primary endpoint of the study was noninferior investigator-assessed PFS and the key secondary endpoints included ORR, event-free survival and safety.
A total of 909 patients were randomized to venetoclax plus obinutuzumab (N = 303), venetoclax plus ibrutinib (N = 305), and continuous ibrutinib (N = 301). Median follow-up at the first analysis was 34.2 months.
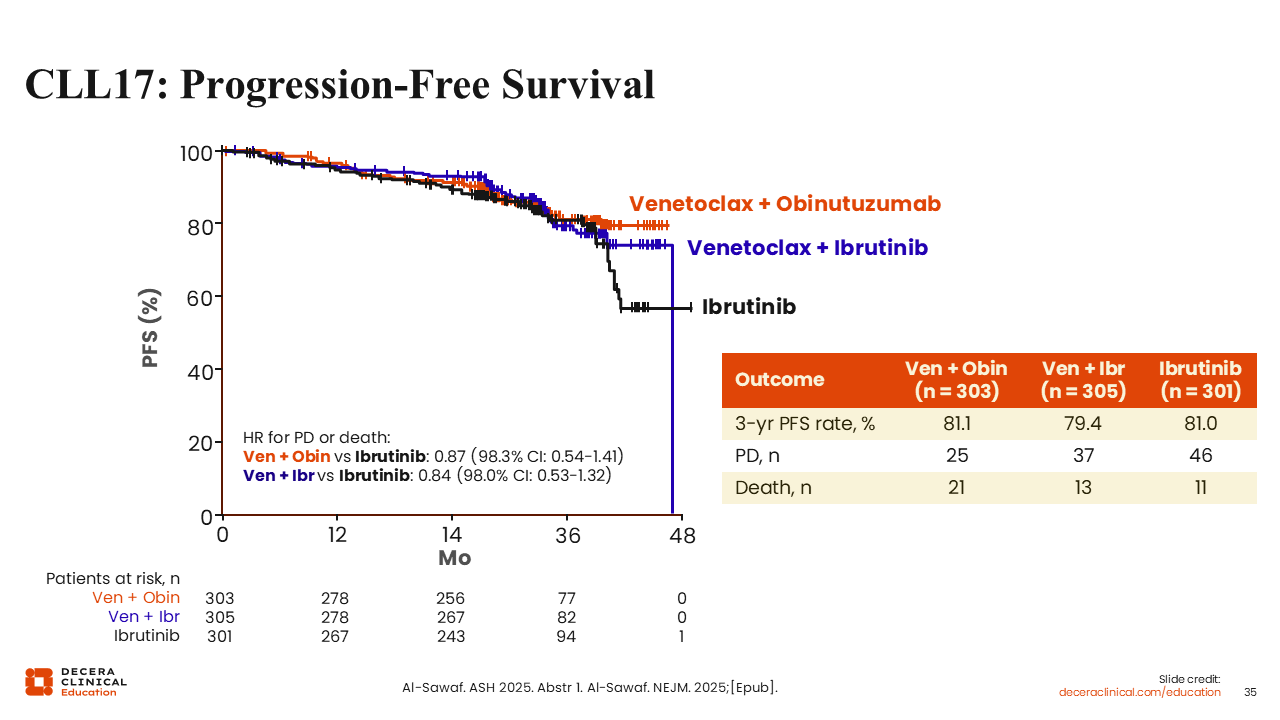
CLL17: Progression-Free Survival
The results from CLL17 indicate that fixed-duration treatment with venetoclax plus obinutuzumab or venetoclax plus ibrutinib are noninferior to continuous treatment with ibrutinib. The 3-year PFS rate was 81.1% in the venetoclax plus obinutuzumab arm compared to 81.0% in the ibrutinib arm (HR: 0.87) and 79.4% in the venetoclax plus ibrutinib arm (HR: 0.84).45,46
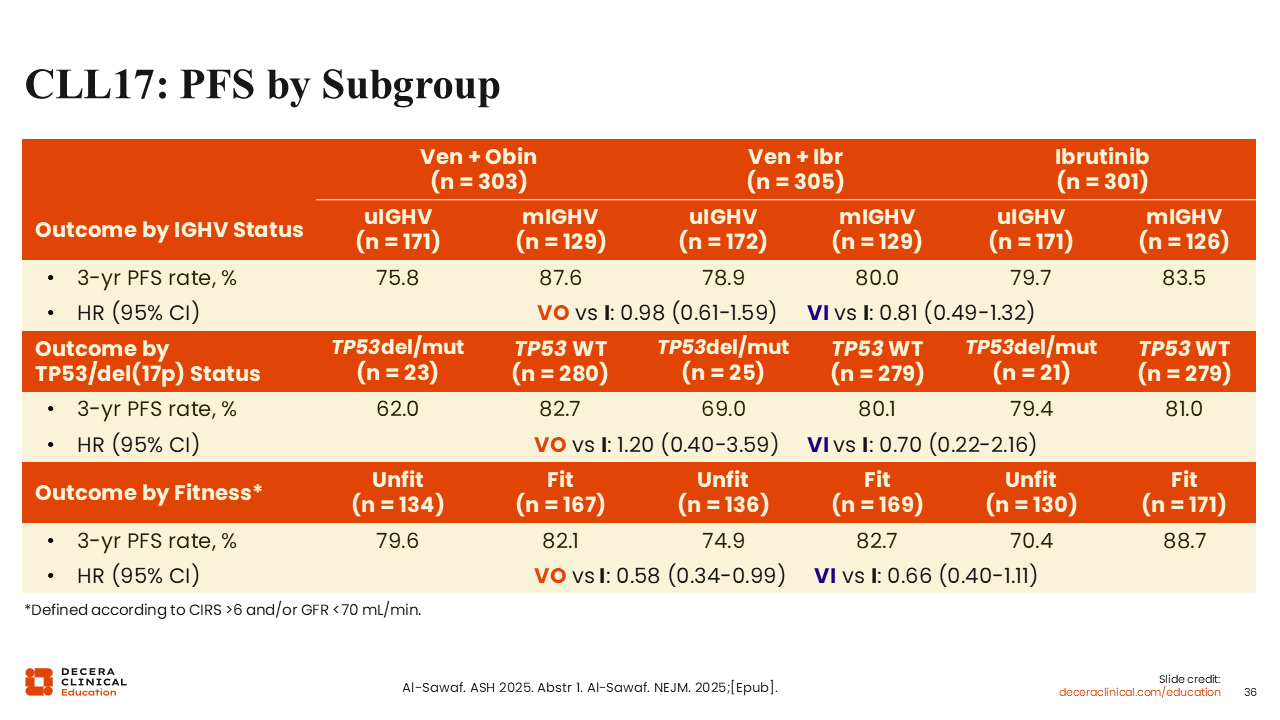
CLL17: PFS by Subgroup
The 3-year PFS rate was generally similar for patients with mutated IGHV compared to those with unmutated IGHV and between arms (approximately 76%-88% in all arms).45,46 The 3-year PFS rate of patients with del(17p)/TP53 mutations was slightly improved for patients treated with ibrutinib-based regimens (venetoclax plus obinutuzumab vs ibrutinib HR: 1.20; venetoclax plus ibrutinib vs ibrutinib HR: 0.70).
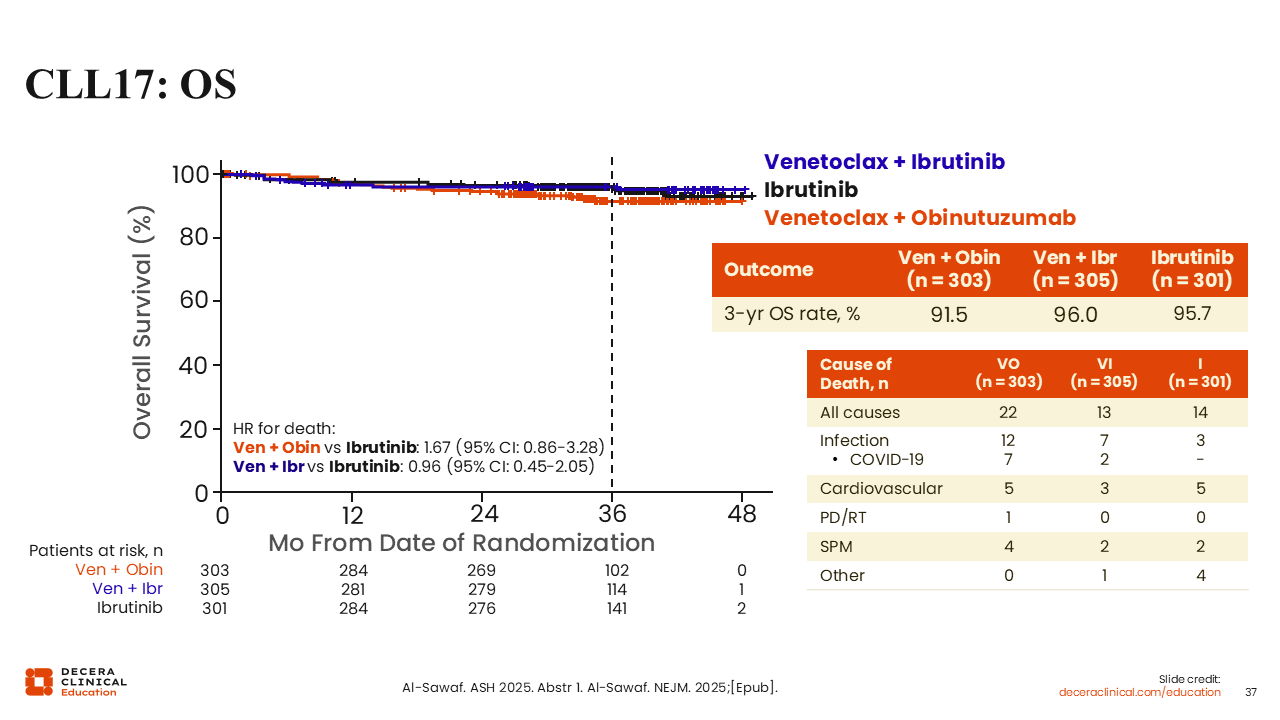
CLL17: Overall Survival and Causes of Death
The OS rate at 3 years was 91.5% in the venetoclax plus obinutuzumab arm compared to 95.7% in the ibrutinib arm (HR: 1.67; 95% CI: 0.86-3.28) and 96.0% in the venetoclax plus ibrutinib arm (compared to ibrutinib: HR: 0.96; 95%CI: 0.45-2.05), respectively.45,46 The most common cause of death in the combination arms was infection and the most common in the ibrutinib monotherapy arm was cardiovascular causes.
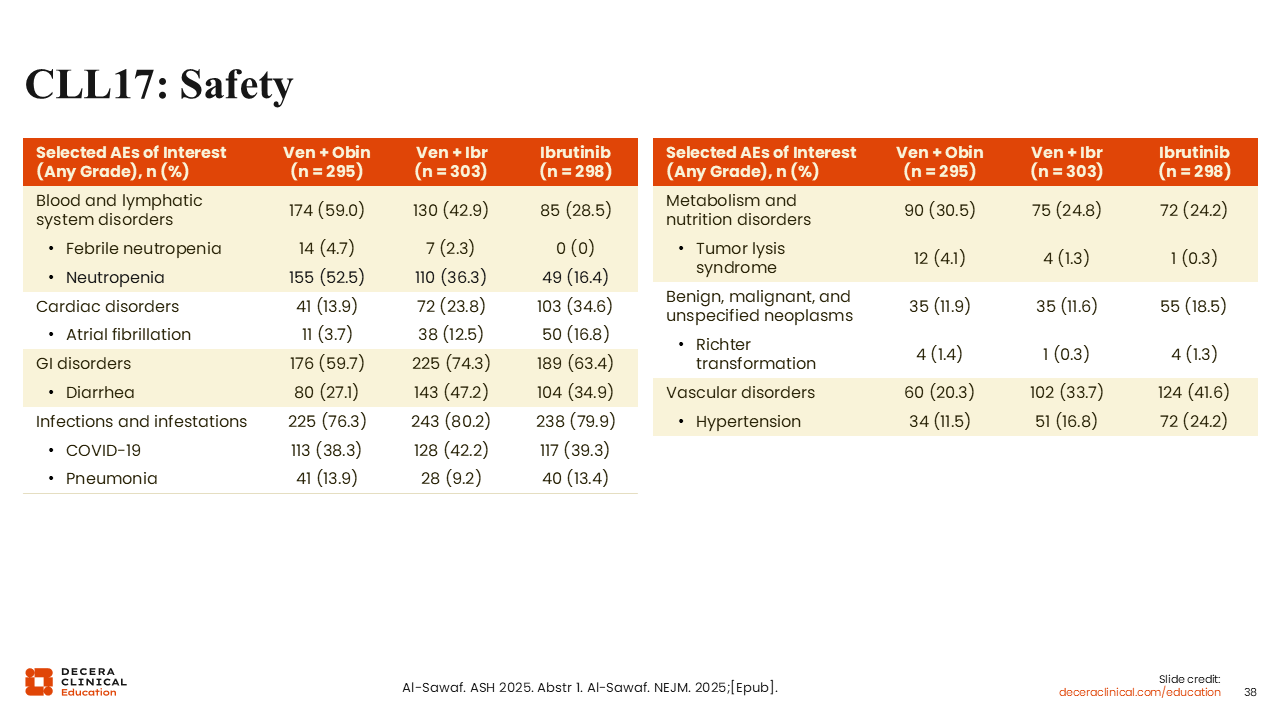
CLL17: Safety
The most frequent AEs in all treatment arms were infections and infestations which included COVID-19 and pneumonia.45,46 COVID-19 infection was observed in 38.3%, 42.2%, and 39.3% of patients with venetoclax plus obinutuzumab, venetoclax plus ibrutinib, and ibrutinib, respectively. As expected, atrial fibrillation was more common in the ibrutinib-containing arms (venetoclax plus obinutuzumab: 3.7%; venetoclax plus ibrutinib: 12.5%; ibrutinib: 16.8%).