CE / CME
HER2-Directed ADCs in HER2-Mutant and HER2-Overexpressing NSCLC
Physician Assistants/Physician Associates: 1.00 AAPA Category 1 CME credit
Pharmacists: 1.00 contact hour (0.1 CEUs)
Physicians: maximum of 1.00 AMA PRA Category 1 Credit™
Nurse Practitioners/Nurses: 1.00 Nursing contact hour
Released: March 18, 2026
Expiration: September 17, 2026
Activity
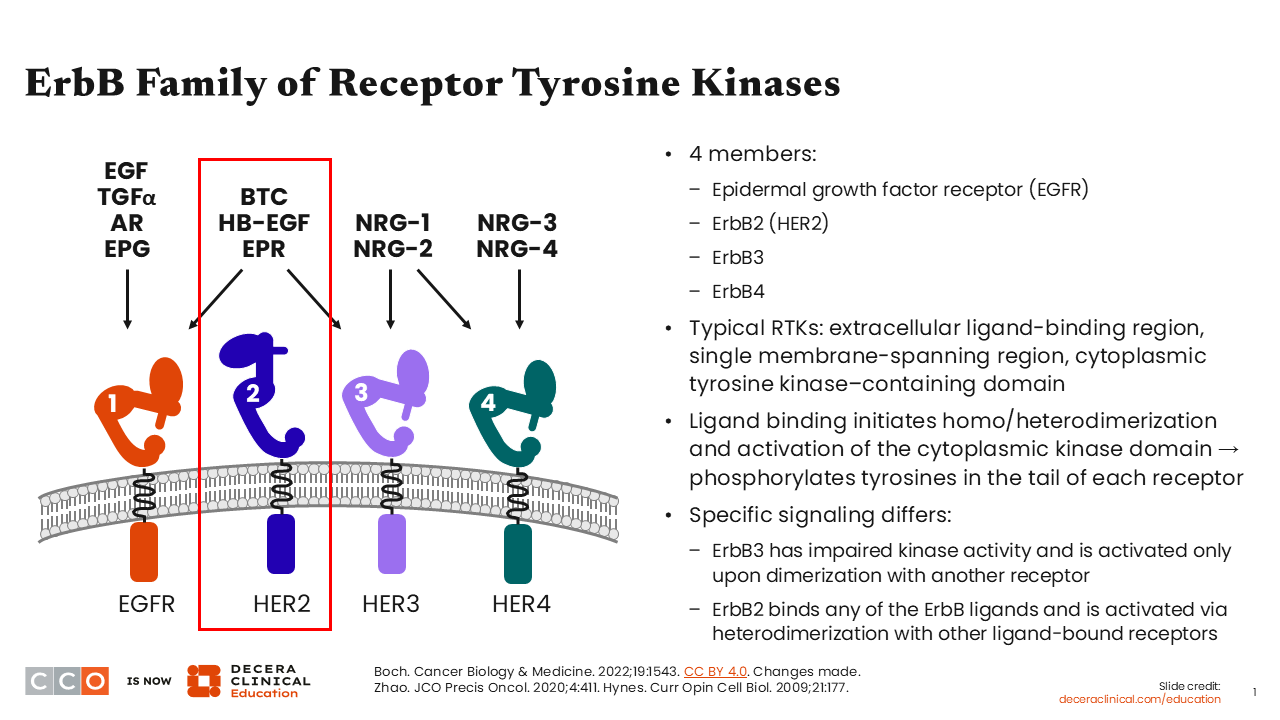
ErbB Family of Receptor Tyrosine Kinases
Matthew Gubens, MD, MS, FASCO:
Over the past 10 years, there has been a proliferation of opportunities to find targetable biomarkers in NSCLC, and this has led to the development of several targeted therapies with clinically meaningful activity in this heterogeneous disease. The incorporation of several targeted agents into the treatment armamentarium has significantly improved the overall treatment journey and experience of our patients with NSCLC.
Of note, the focus of this module is on the exciting changes in the setting of HER2-directed therapies for patients with HER2-positive (also referred to as HER2-altered) NSCLC.
The ErbB or HER family of receptor tyrosine kinases (RTKs) has 4 members: HER1 (also known as EGFR), HER2, HER3, and HER4.1-3 Typically, RTKs have an extracellular ligand-binding region, single membrane-spanning region, and a cytoplasmic tyrosine kinase–containing domain. Ligand binding initiates homodimerization or heterodimerization, which then leads to the activation of the cytoplasmic kinase domain, followed by the phosphorylation of tyrosine in the tail of each receptor.
Regarding HER2, it has the ability to bind to any of the HER ligands, and it is activated via heterodimerization with other ligand-bound receptors.
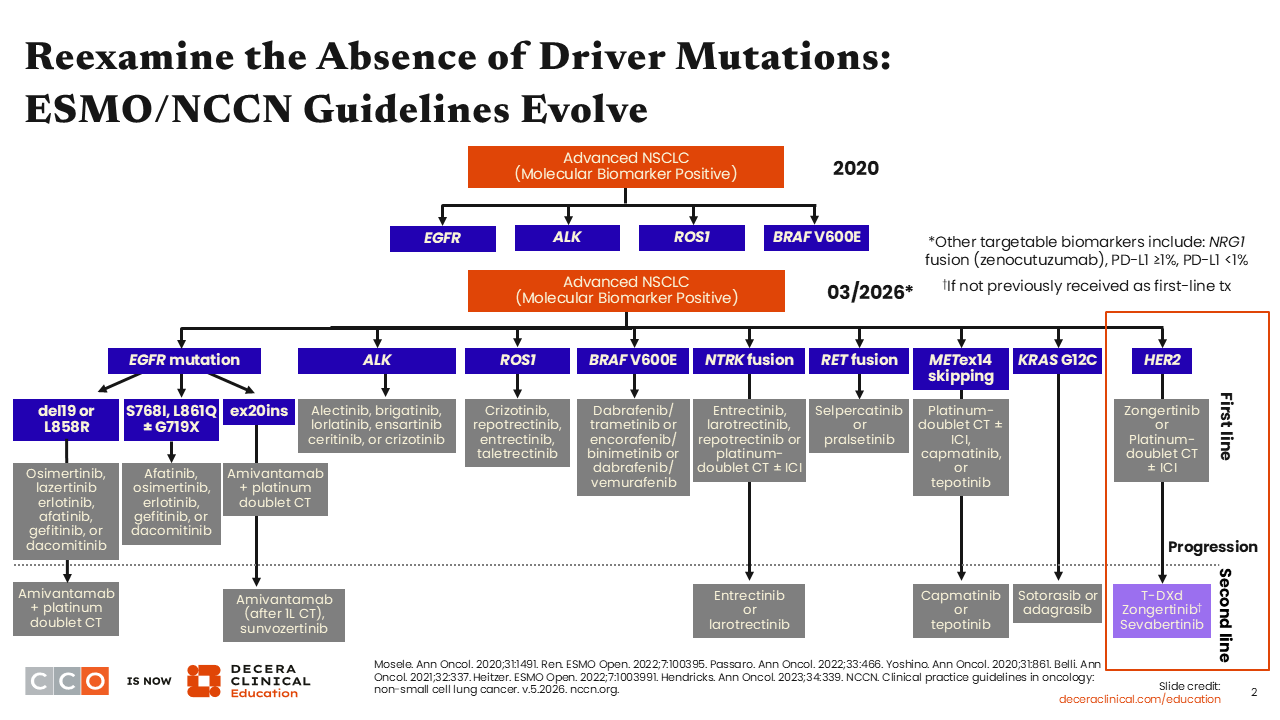
Reexamine the Absence of Driver Mutations: ESMO/NCCN Guidelines Evolve
Matthew Gubens, MD, MS, FASCO:
The identification and management of biomarker-positive NSCLC has evolved over the years. In 2020, there were only 4 prominent and targetable biomarkers, EGFR, ALK, ROS1, and BRAF V600E mutations.4,5 Today, there are more than 10 actionable biomarkers in NSCLC, namely, EGFR, ALK, ROS1, BRAF V600E, NTRK, RET, MET, KRAS, NRG1, PD-L1, and HER2.6 This is very exciting, and it is very important to determine if the patient’s disease harbors any of these driver alterations before making treatment decisions.
An important take-home message is that the treating oncologist needs to read the complete molecular report, especially with careful considerations for details on the sufficiency of the available sample tested and whether a rebiopsy or retesting is necessary to ensure that the report can be used to make the best treatment decision for the patient. In the days of minimally invasive biopsies through endobronchial ultrasound or fine needle aspirations, both of which result in smaller sample sizes, a deeper review and understanding of the molecular report is important. An interrogation of the report for insufficiencies in factors such as sample quantity and quality is necessary as opposed to a quick look for biomarker status. It is important that one is open to the idea of a rebiopsy or retesting, if necessary. Even if it takes more time than anticipated, a deeper review of the molecular report is critical and very important to maximize treatment outcomes. Liquid biopsy by circulating DNA can be an important tool to improve the diagnosis of actionable genomic alterations in the metastatic disease setting.
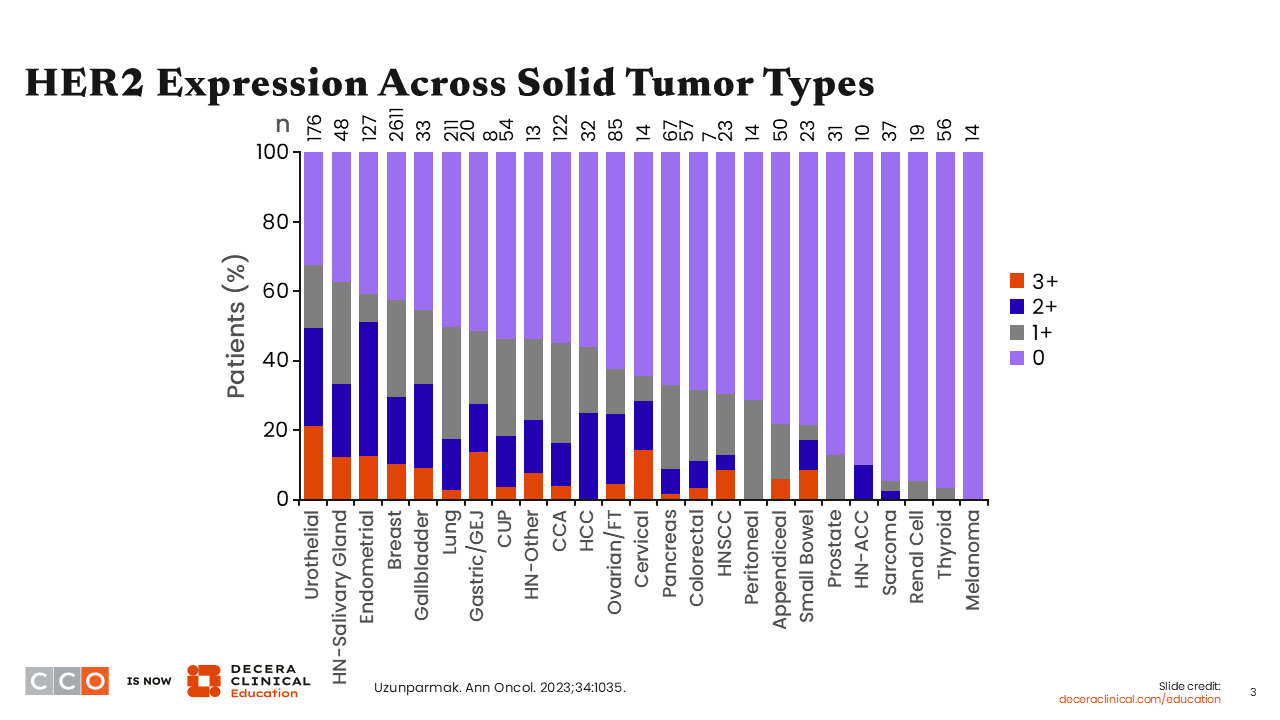
HER2 Expression Across Solid Tumor Types
Matthew Gubens, MD, MS, FASCO:
Regarding HER2 as a biomarker in NSCLC, a study that evaluated formalin-fixed paraffin-embedded (FFPE) tissue samples from 4701 patients with solid tumors showed that HER2 is differentially expressed across different solid tumor types.7 HER2 overexpression determined via IHC and scored as 3+ staining intensity, occurred more frequently in urothelial, head and neck/salivary gland, endometrial, breast, gallbladder, and cervical cancers compared with peritoneal, prostate, renal cell, thyroid, and melanoma. In this study, the incidence of HER2 overexpression (IHC 3+) among patients with lung cancer was approximately 3%.
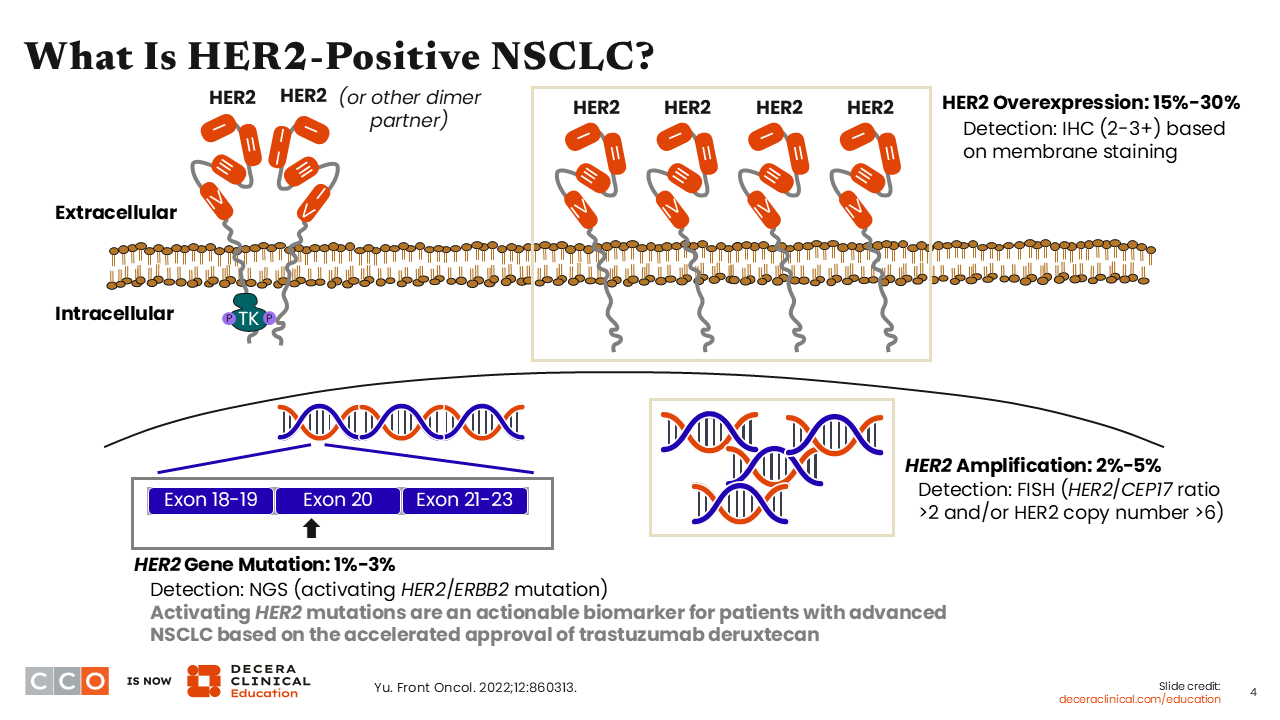
What Is HER2-Positive NSCLC?
Matthew Gubens, MD, MS, FASCO:
HER2-altered NSCLC is a heterogeneous group of diseases.8 In general, when considering the definition of HER2 positivity in NSCLC, there are 3 distinct types: HER2 gene mutations, HER2 protein overexpression, and HER2 gene amplification.
HER2 gene mutations in exons 18-19, exon 20, and exons 21-23 in the TKD of HER2 have been reported in 1% to 3% of patients with NSCLC. Activating HER2 mutations can be detected by NGS, and these mutations are actionable. In NSCLC, HER2 mutations are more commonly identified in never smokers or those with a mild history of tobacco use, in females, and among those with adenocarcinoma or adenosquamous histology. HER2 mutations in NSCLC are associated with higher rates of brain metastasis and worse overall survival (OS) outcomes. HER2 mutations are inherently distinct from HER2 overexpression and HER2 amplification.
In NSCLC, HER2 overexpression detected by IHC is also actionable when at a 3+ level. Finally, we have HER2 amplifications in NSCLC, and these can be detected by fluorescence in situ hybridization. HER2 amplifications have been reported in 2% to 5% of patients with NSCLC. De novo HER2 overexpression or HER2 amplification is more frequent in male smokers with NSCLC. It is not clear if the presence of HER2 overexpression and/or HER2 amplifications is associated with OS outcome in patients with NSCLC. As of March 2026, HER2 amplifications in NSCLC are not actionable.
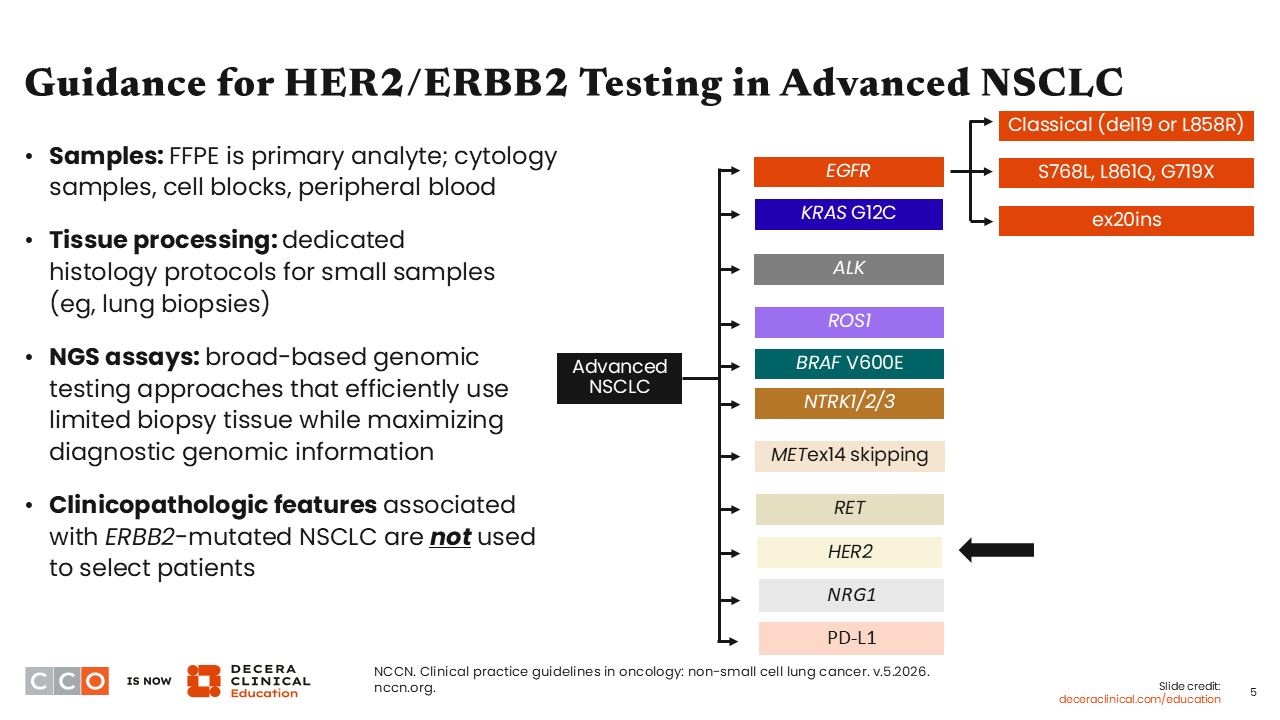
Guidance for HER2/ERBB2 Testing in Advanced NSCLC
Matthew Gubens, MD, MS, FASCO:
In lung cancer, there are primarily 2 different dimensions to HER2 testing. For all patients with metastatic nonsquamous NSCLC or NSCLC not otherwise specified, broad molecular testing by NGS is recommended by the National Comprehensive Cancer Network (NCCN) guidelines. Testing for HER2 mutations can also be considered for patients with metastatic squamous cell lung carcinoma. During initial evaluation, testing priority should be given to PD-L1 status determination and multigene panel testing. If sufficient tissue is available, then additional testing can be performed to include the use of IHC for HER2 overexpression status determination. This order of testing priority for PD-L1 status and multigene NGS is important because results will affect the choice of first-line therapy.
To determine the HER2 expression level in a malignant tumor, FFPE tissue is the primary sample. Cytology samples, cell blocks, and peripheral blood can also be assessed, as these sample types can be useful sources of data for NGS analysis. For tissue processing, it is important to have a standardized workflow with dedicated histology protocols, especially when only a small size of tumor tissue is available for testing. Broad panel NGS assays that efficiently use minimal tissue should be used for genomic testing. Of note, clinicopathologic features associated with HER2 mutation–positive NSCLC or factors that predispose patients to developing NSCLC, such as gender or smoking history, should not be used to select patients for treatment. The patient’s tumor-specific genomic information is needed to determine the optimal treatment choice.
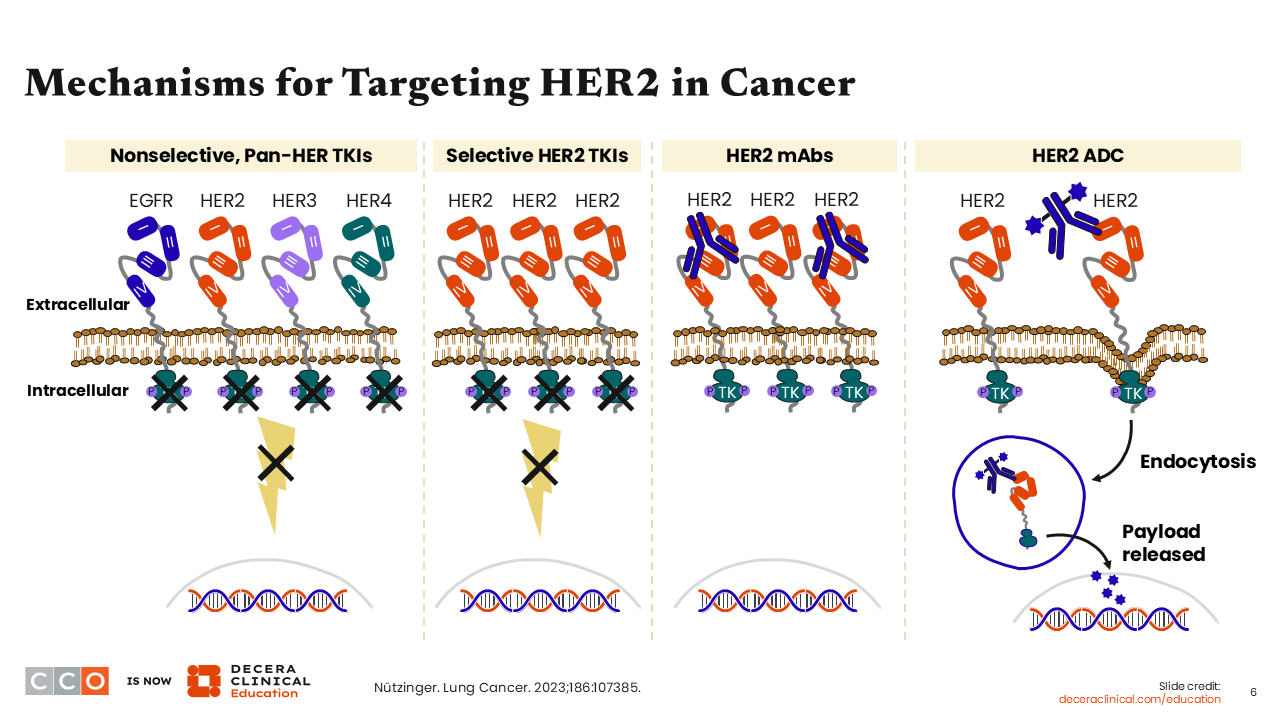
Mechanisms for Targeting HER2 in Cancer
Matthew Gubens, MD, MS, FASCO:
Different drug mechanisms have already been successfully used to target HER2 in the treatment of cancer.9 For instance, drugs such as afatinib, dacomitinib, and neratinib are nonselective pan-HER tyrosine kinase inhibitors (TKIs). Other drugs such as tucatinib, zongertinib, and sevabertinib are TKIs with potent selectivity for HER2. Also, the use of monoclonal antibodies such as trastuzumab and pertuzumab has been successfully used to target HER2 in cancer. More recently, a novel class of targeted agents, termed ADCs, capable of delivering cytotoxic chemotherapy to tumor cells was developed. HER2-directed ADCs such as T-DXd and ado-trastuzumab emtansine (T-DM1) were added to the treatment armamentarium for the treatment of different cancer types.
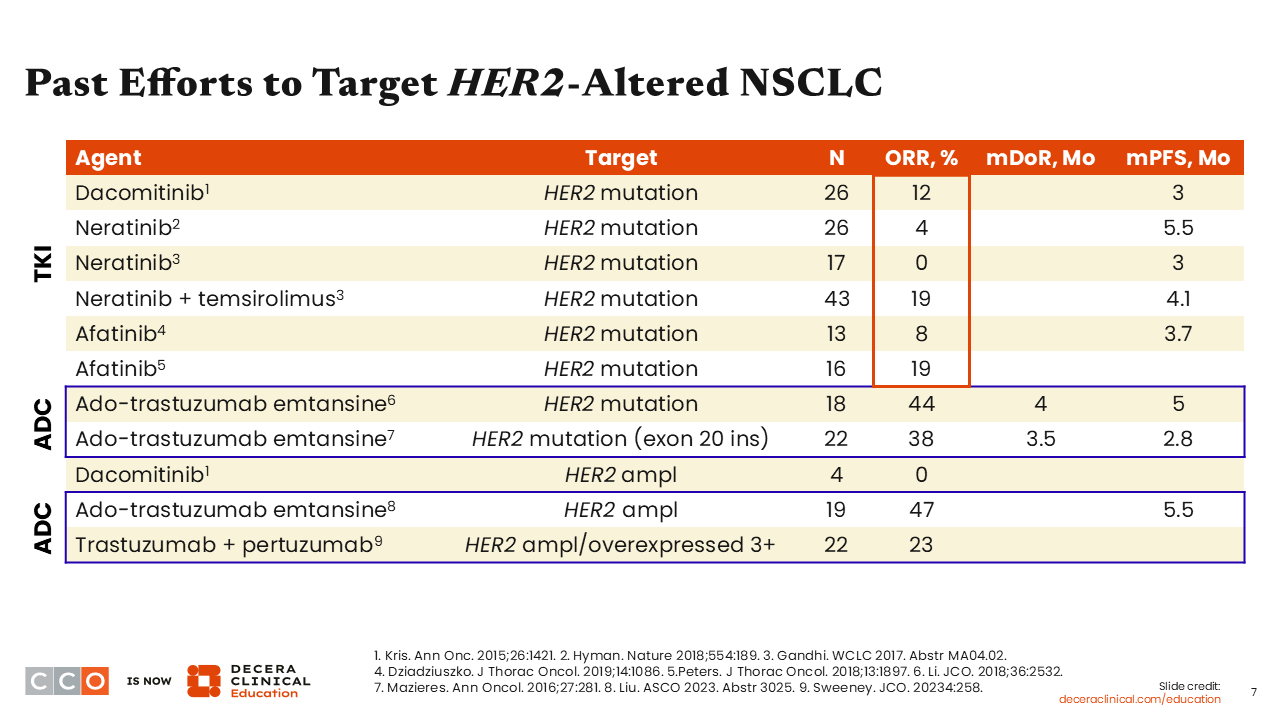
Past Efforts to Target HER2-Altered NSCLC
Matthew Gubens, MD, MS, FASCO:
Several past efforts to target HER2 in NSCLC have been somewhat disappointing.10-14 The use of the TKIs afatinib, dacomitinib, or neratinib to target HER2 mutations in NSCLC has resulted in quite modest progression-free survival (PFS) of approximately 3.0-5.5 months. These TKIs have elicited low response rates among patients with HER2 mutation–positive NSCLC, and they have also been limited by the toxicities associated with them. Of note, afatinib and dacomitinib are approved by the FDA for patients with EGFR mutation–positive NSCLC but not those with HER2-positive NSCLC.15,16
Previous studies of the ADC, T-DM1, in HER2 mutation–positive or HER2-amplified NSCLC resulted in a median PFS of approximately 3.0-5.5 months with a response rate of approximately 40% to 50%.17-19 However, T-DM1 is not approved by the FDA for patients with NSCLC. Also, investigations of a dual HER2-targeted approach with the combination of the monoclonal antibodies, trastuzumab and pertuzumab, resulted in disappointing response rates.20
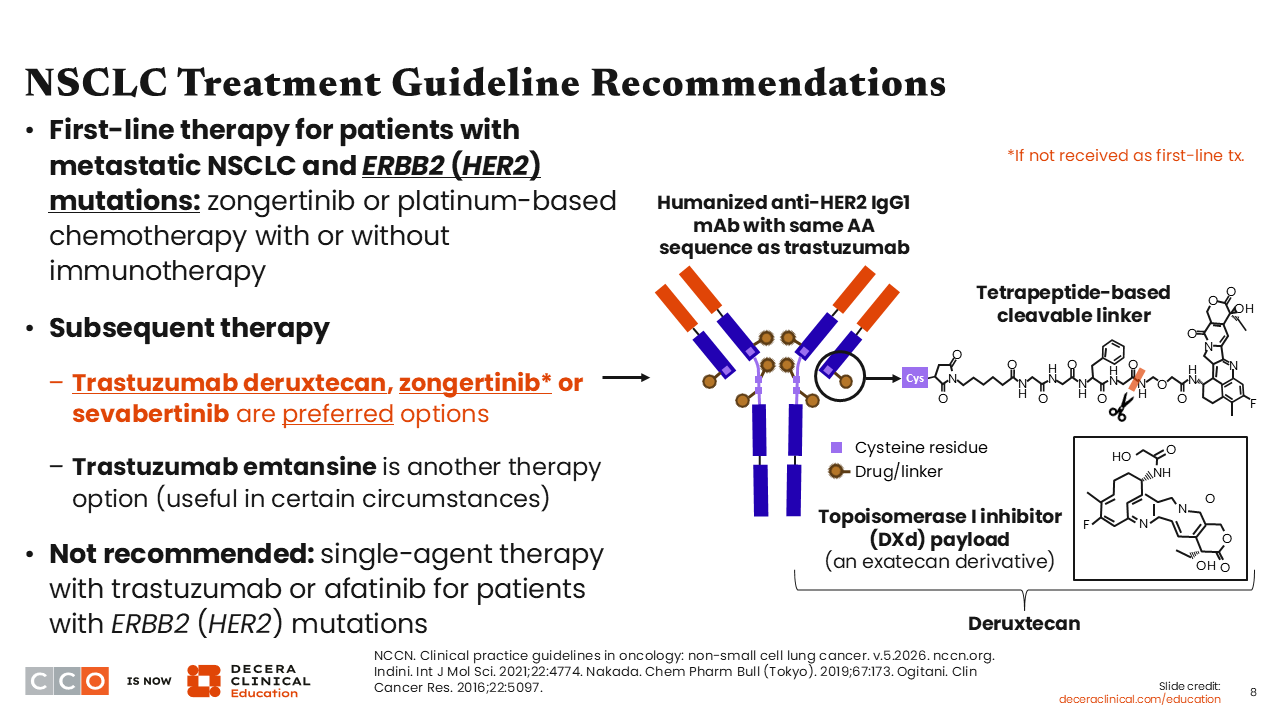
NSCLC Treatment Guideline Recommendations
Matthew Gubens, MD, MS, FASCO:
On February 26, 2026, the FDA granted accelerated approval to zongertinib for an expanded indication for adults with unresectable or metastatic nonsquamous NSCLC whose tumors have HER2 TKD–activating mutations, as detected by an FDA-authorized test.21 So, the recommended first-line therapy for patients with HER2 mutation–positive metastatic NSCLC is zongertinib. Upon disease progression, the use of T-DXd, T-DM1, or platinum-based chemotherapy with or without an immune checkpoint inhibitor is recommended.6
For patients with HER2 mutation–positive NSCLC who received platinum-based chemotherapy with or without an immune checkpoint inhibitor as first-line therapy, the NCCN-preferred second-line options are T-DXd, zongertinib, or sevabertinib. T-DM1 may be used under certain circumstances.
The recommended first-line therapy for patients with HER2-overexpressing (IHC 3+) metastatic NSCLC is platinum-based chemotherapy with or without an immune checkpoint inhibitor. Upon disease progression, T-DXd is recommended.
T-DXd is approved by the FDA for patients with unresectable or metastatic NSCLC whose tumors have activating HER2 mutations, as detected by an FDA-approved test, and who have received a prior systemic therapy.22 T-DXd is also approved for patients with unresectable or metastatic HER2-positive (IHC 3+) solid tumors who have received prior systemic treatment and have no satisfactory alternative treatment options. Zongertinib is approved by the FDA for patients with unresectable or metastatic nonsquamous NSCLC whose tumors have HER2 TKD–activating mutations, as detected by an FDA-authorized test.21 Also, sevabertinib is approved by the FDA for patients with locally advanced or metastatic nonsquamous NSCLC whose tumors have HER2 TKD–activating mutations, as detected by an FDA-approved test, and who have received a prior systemic therapy.23
These 3 HER2-directed agents are preferred for use as first-line or second-line therapy for patients with HER2 mutation–positive NSCLC, certainly to be used before the typical salvage treatment of docetaxel with or without ramucirumab.6 It is very important to note that the TKIs zongertinib and sevabertinib are approved only for HER2 mutation–positive advanced NSCLC.21,23 Among the 3 agents noted above, only T-DXd has FDA approval for use in patients with HER2-overexpressing (IHC 3+) advanced NSCLC after disease progression on systemic first-line therapy.22
T-DM1 is another option that may be considered for patients with HER2 mutation–positive NSCLC after progression on first-line therapy.6 Of importance, the single-agent use of trastuzumab or afatinib is not recommended for patients with HER2 mutation–positive advanced NSCLC.