CE / CME
HER2-Directed ADCs in HER2-Mutant and HER2-Overexpressing NSCLC
Physician Assistants/Physician Associates: 1.00 AAPA Category 1 CME credit
Pharmacists: 1.00 contact hour (0.1 CEUs)
Physicians: maximum of 1.00 AMA PRA Category 1 Credit™
Nurse Practitioners/Nurses: 1.00 Nursing contact hour
Released: March 18, 2026
Expiration: September 17, 2026
Activity
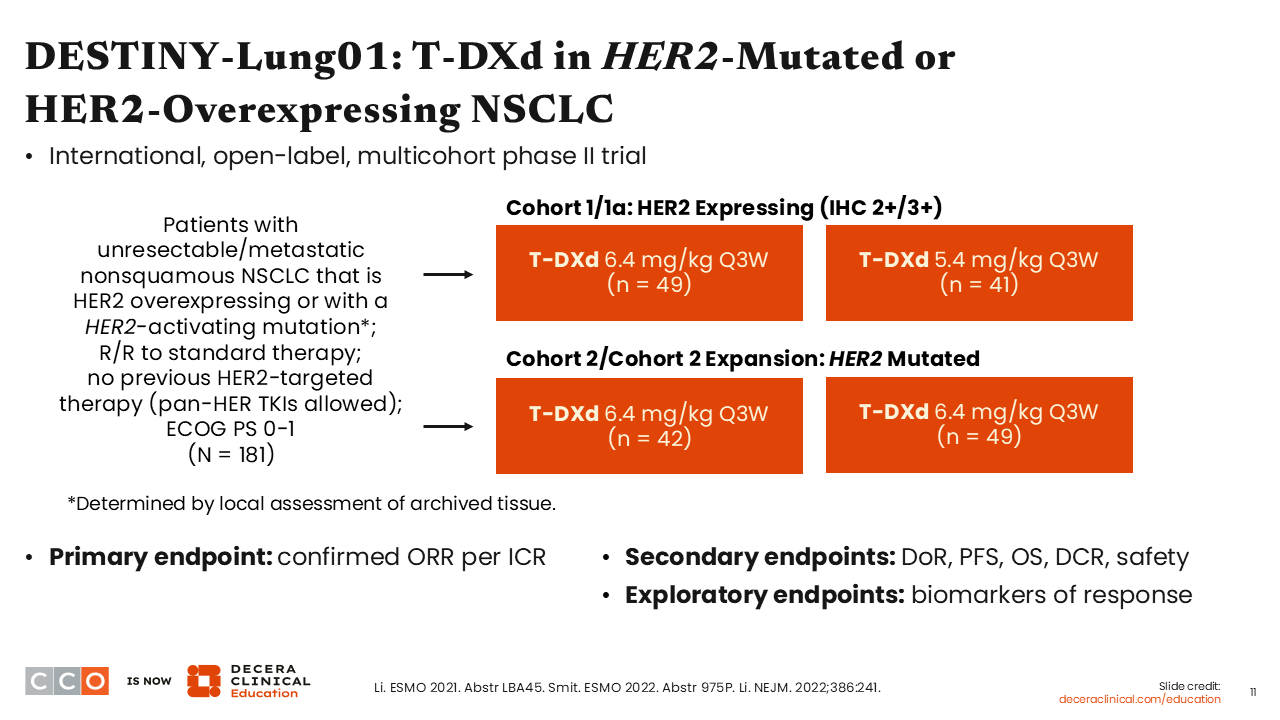
DESTINY-Lung01: T-DXd in HER2-Mutated or HER2-Overexpressing NSCLC
Estelamari Rodriguez, MD, MPH:
Now, I will delve into the available data on FDA-approved HER2-targeted ADCs in NSCLC. The DESTINY-Lung01 is a nonrandomized, multicohort phase II trial that investigated the efficacy and safety of 2 different doses of T-DXd (5.4 mg/kg and 6.4 mg/kg) for 181 patients with unresectable or metastatic nonsquamous NSCLC (NCT03505710). Patients in one of the cohorts had HER2-overexpressing disease and those in another cohort had HER2-activating mutations. All patients had progressed on a prior standard therapy but had not been previously exposed to a HER2-directed therapy. Previous exposure to pan-HER TKIs was allowed. The primary endpoint was confirmed objective response rate (ORR). The secondary endpoints were DoR, PFS, OS, disease control rate (DCR), and safety. The exploratory endpoint was the determination of biomarkers of response.
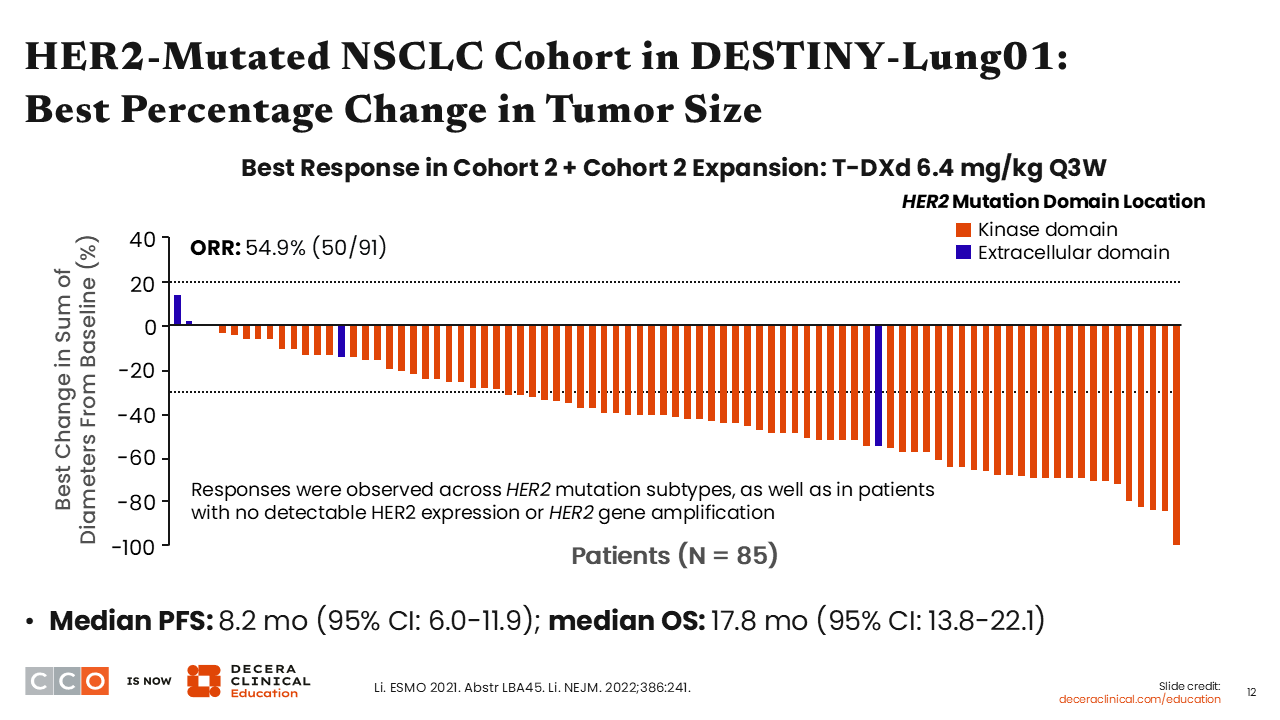
DESTINY-Lung01: Response
Estelamari Rodriguez, MD, MPH:
Among 91 patients with HER2-mutated NSCLC, the ORR was 54.9%.35 The median PFS was 8.2 months, and the median OS was 17.8 months. Remarkably, responses were observed across different HER2 mutation subtypes and also in patients with no detectable HER2 expression or HER2 gene amplification.
It is important to note that among 49 patients with HER2-overexpressing NSCLC, the confirmed ORR was 26.5%.36
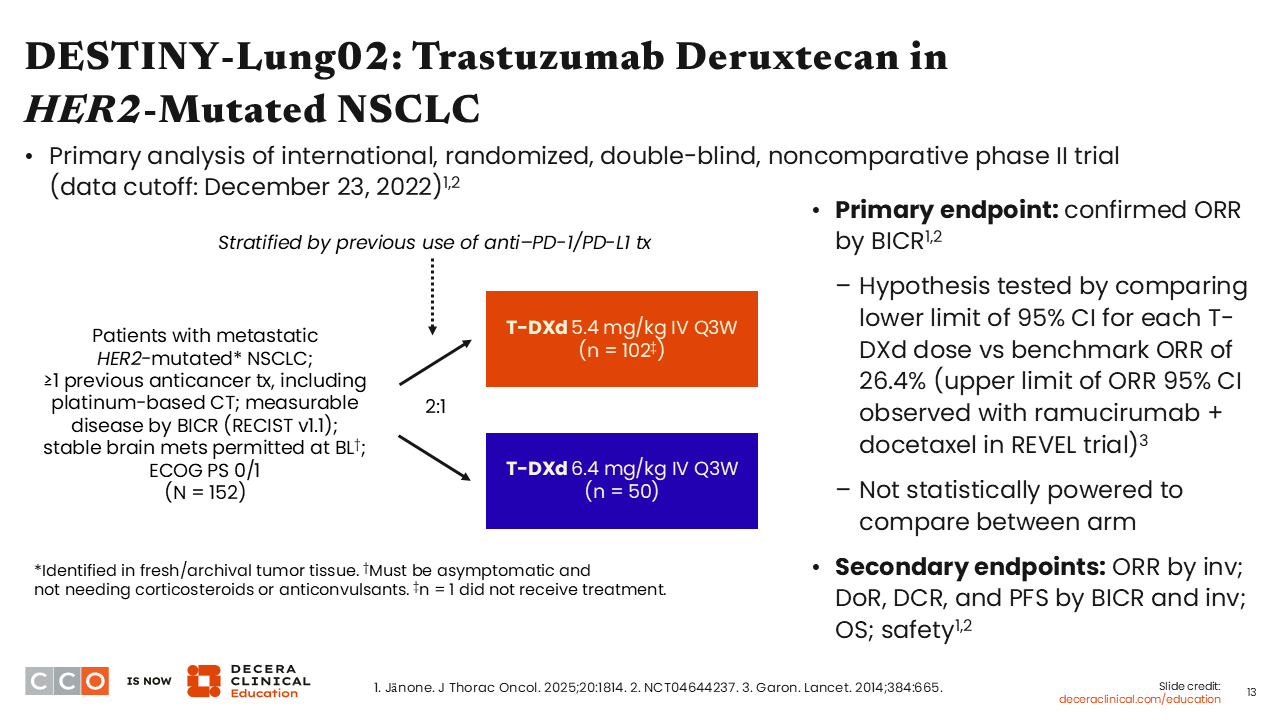
DESTINY-Lung02: Trastuzumab Deruxtecan in HER2-Mutated NSCLC
Estelamari Rodriguez, MD, MPH:
The randomized phase II DESTINY-Lung02 trial investigated 2 different doses of T-DXd (5.4 mg/kg vs 6.4 mg/kg) for 152 patients with metastatic HER2 mutation–positive NSCLC who have received ≥1 previous anticancer therapy including platinum-based chemotherapy (NCT04644237). Patients with stable brain metastases at baseline were allowed to participate. Patients were stratified by previous use of anti–PD-1/PD-L1 antibody treatment prior to randomization. In total, 102 patients were randomly assigned to receive 5.4 mg/kg of T-DXd vs 50 patients who were randomly assigned to receive the 6.4-mg/kg dose. The primary endpoint was confirmed ORR by blinded independent central review. The secondary endpoints included investigator-assessed ORR, DoR, PFS, OS, and safety.
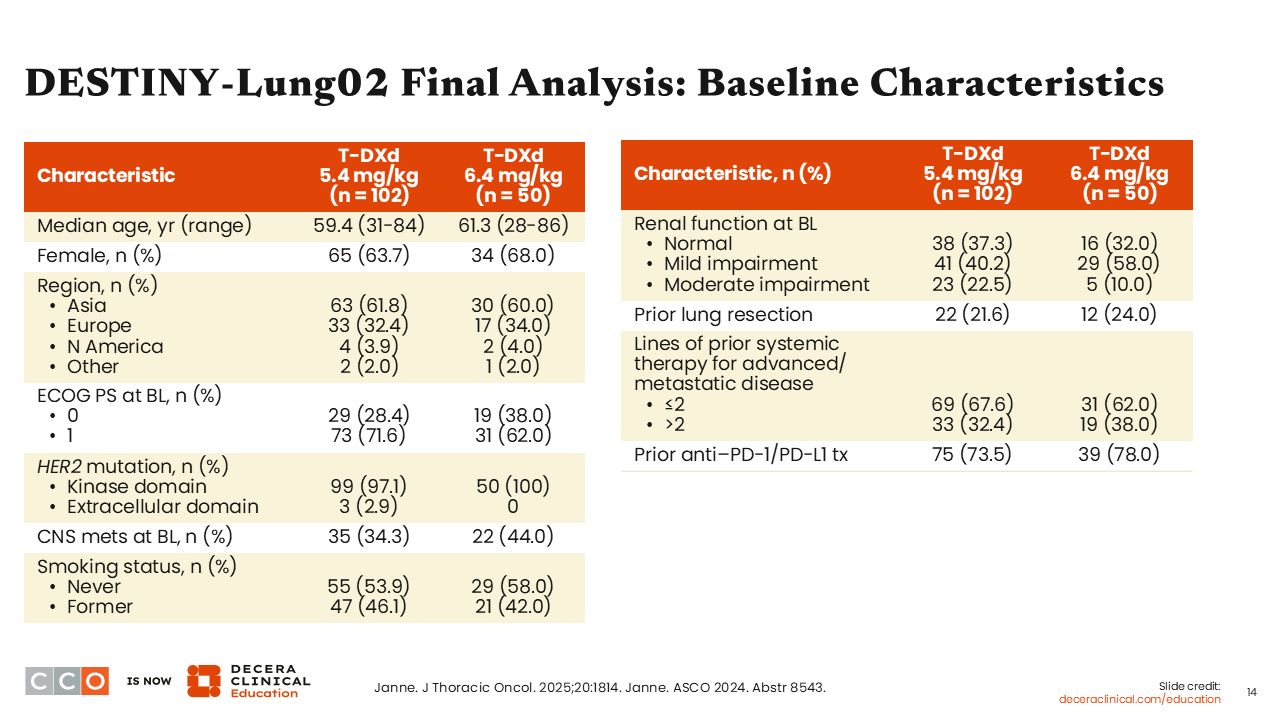
DESTINY-Lung02 Final Analysis: Baseline Characteristics
Estelamari Rodriguez, MD, MPH:
The baseline characteristics were fairly balanced between the 2 arms.37 The median age was approximately 60 years on both arms. All patients had an Eastern Cooperative Oncology Group performance status of either 0 or 1. The vast majority of the patients had disease harboring HER2 TKD mutations. Approximately 34% to 40% of the patients had brain metastasis at baseline, and approximately two thirds of the patients had received ≤2 prior lines of therapy. On either arm, 74% to 78% had previously received an anti–PD-1/PD-L1 antibody.
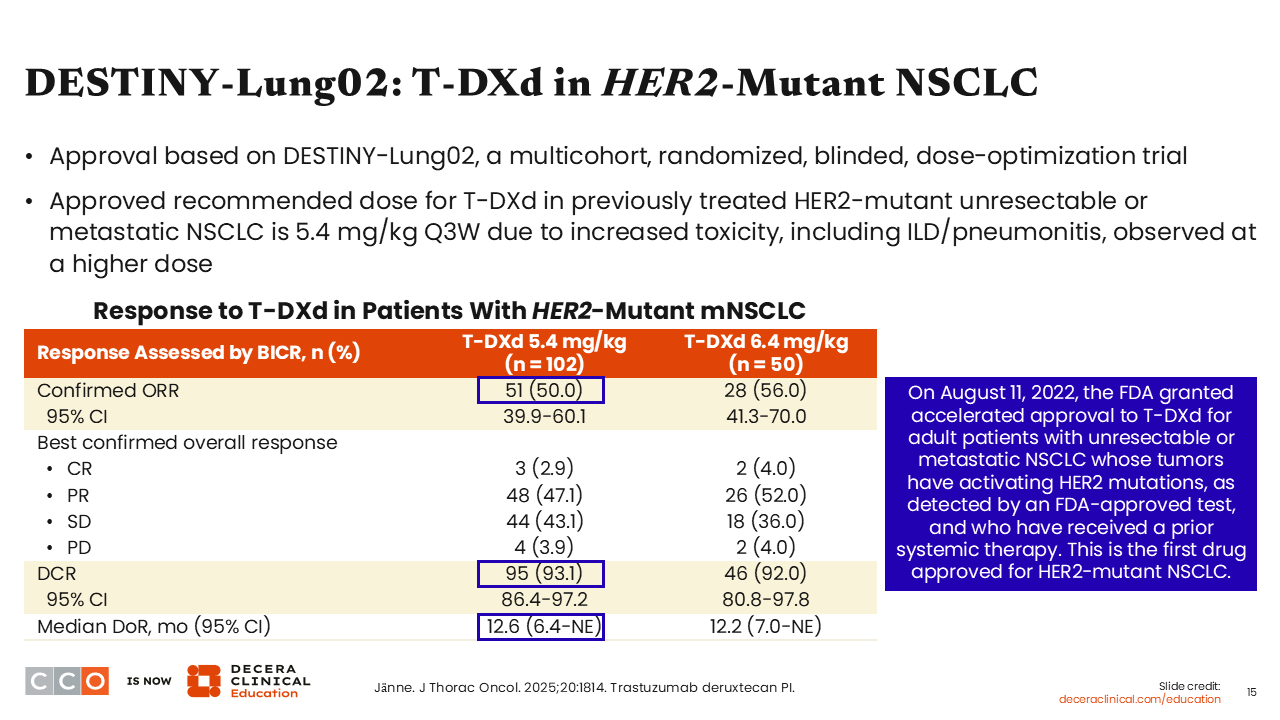
DESTINY-Lung02: T-DXd in HER2-Mutant NSCLC
Estelamari Rodriguez, MD, MPH:
The confirmed ORR was 50% in the 5.4-mg/kg arm vs 56% in the 6.4-mg/kg arm. The DCRs were 93.1% and 92.0%, and the median DoRs were 12.6 months and 12.2 months, respectively. However, toxicities, including ILD/pneumonitis, were higher in the 6.4-mg/kg T-DXd arm compared with the 5.4-mg/kg arm. These results demonstrated that at the lower dose of 5.4 mg/kg, T-DXd yields similar response rates to that achieved at the higher dose of 6.4 mg/kg, but the lower dose is associated with lower rates of adverse events (AEs). Based on these results, the FDA granted accelerated approval to T-DXd at the 5.4-mg/kg dose for adult patients with unresectable or metastatic NSCLC whose tumors have activating HER2 mutations, as detected by an FDA-approved test, and who have received a prior systemic therapy.22 Of importance, this is the first drug approved for HER2-mutant NSCLC.
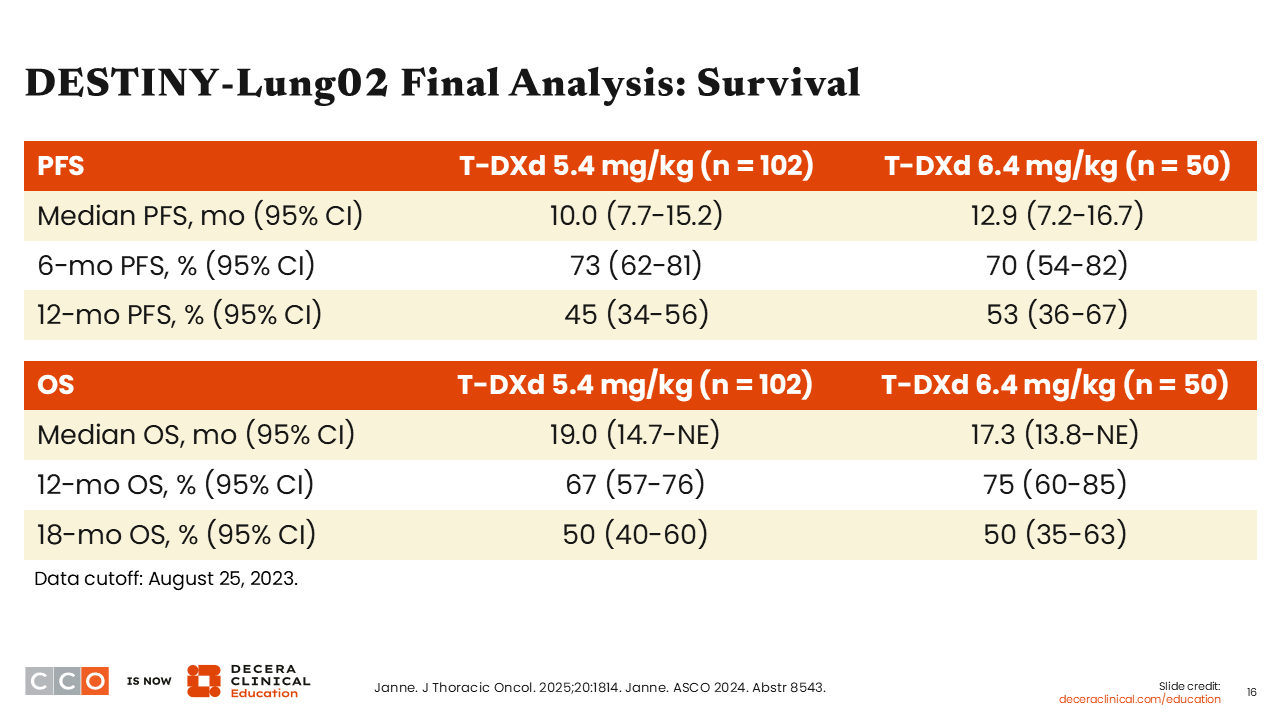
DESTINY-Lung02 Final Analysis: Survival
Estelamari Rodriguez, MD, MPH:
In the final analysis of the survival outcomes, the median PFS results were 10.0 months and 12.9 months with 5.4 mg/kg and 6.4 mg/kg of T-DXd, respectively.37 The 6-month and 12-month PFS rates with 5.4-mg/kg T-DXd were impressive at 73% and 45%, respectively. Similarly, the 6-month and 12-month PFS rates with 6.4-mg/kg T-DXd were 70% and 53%, respectively.
The median OS results were 19.0 months and 17.3 months with 5.4 mg/kg and 6.4 mg/kg of T-DXd, respectively. At the 5.4-mg/kg T-DXd dose, the 6-month and 12-month OS rates were 67% and 50%, respectively. At the 6.4-mg/kg dose, the 6-month and 12-month OS rates were 75% and 50%, respectively. Overall, these results are remarkable and clinically meaningful in a population of patients with previously treated advanced disease.
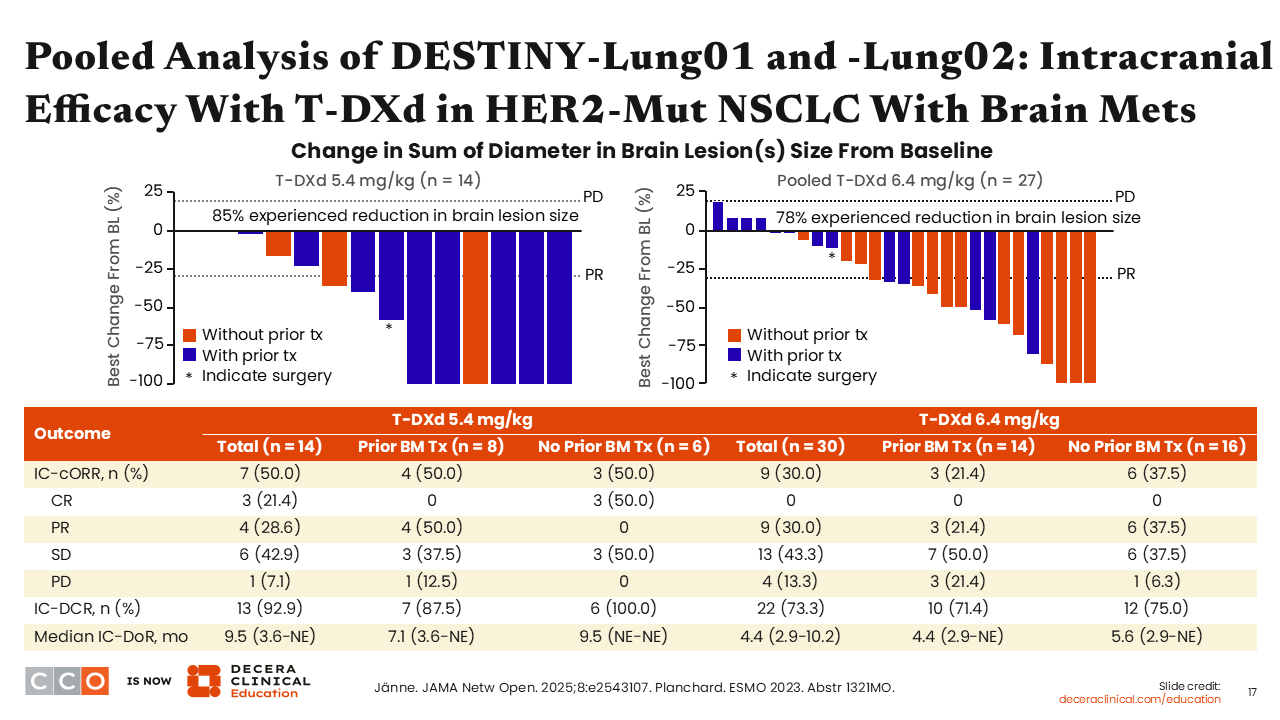
Pooled Analysis of DESTINY-Lung01 and DESTINY-Lung02: Intracranial Efficacy With T-DXd in HER2-Mut NSCLC With Brain Metastases
Estelamari Rodriguez, MD, MPH:
In the past, patients with NSCLC and brain metastasis were treated using whole brain radiation. More recently, these patients were managed with targeted radiation therapy using gamma knife to the sites of central nervous system (CNS) metastases and stereotactic body radiation therapy to other involved organs.38 Although the use of gamma knife prolongs survival, it is associated with a high toxicity rate. The development of less toxic targeted agents with the ability to cross the blood–brain barrier is important in that the approach will improve the quality of life for patients with CNS metastasis.
In a pooled analysis of data from the DESTINY-Lung01 and DESTINY-Lung02 trials, the intracranial activity of T-DXd in patients with HER2 mutation–positive NSCLC and brain metastasis at baseline was determined.39 At the 5.4-mg/kg T-DXd dose, the intracranial ORR was 50% among patients with brain metastases at baseline, and the median DoR of intracranial response was 7.1 months. At the 6.4-mg/kg T-DXd dose, the intracranial ORR was 21.4% among patients with brain metastases at baseline, and the median DoR of intracranial response was 4.4 months. Of important note, DESTINY-Lung02 allowed the participation of patients with stable brain metastases at baseline. Therefore, these results demonstrated that patients who may not have received treatment targeted to the brain because they had stable brain metastases are able to achieve intracranial response to T-DXd.
DESTINY-Lung02 Final Analysis: Safety Summary
Estelamari Rodriguez, MD, MPH:
It is important to keep in mind that the conjugation chemistry and mechanism of action of T-DXd involve the delivery of chemotherapy in a more targeted way to tumor cells. So, it is not surprising that T-DXd is commonly associated with nausea, neutropenia, and fatigue. Nausea was reported in approximately 65% of patients who received 5.4-mg/kg dose of T-DXd on DESTINY-Lung02.37 So, the use of prophylactic antiemetic medications per local institutional guidelines is recommended to prevent chemotherapy-induced nausea and vomiting.22
On the DESTINY-Lung02 trial, grade ≥3 hematologic AEs associated with T-DXd at the 5.4-mg/kg dose included neutropenia (18.8%), anemia (11.9%), and thrombocytopenia and leukopenia (5.9% each). Careful monitoring of patients receiving T-DXd for myelosuppression is important, and these AEs should be promptly managed following the prescribing instructions.
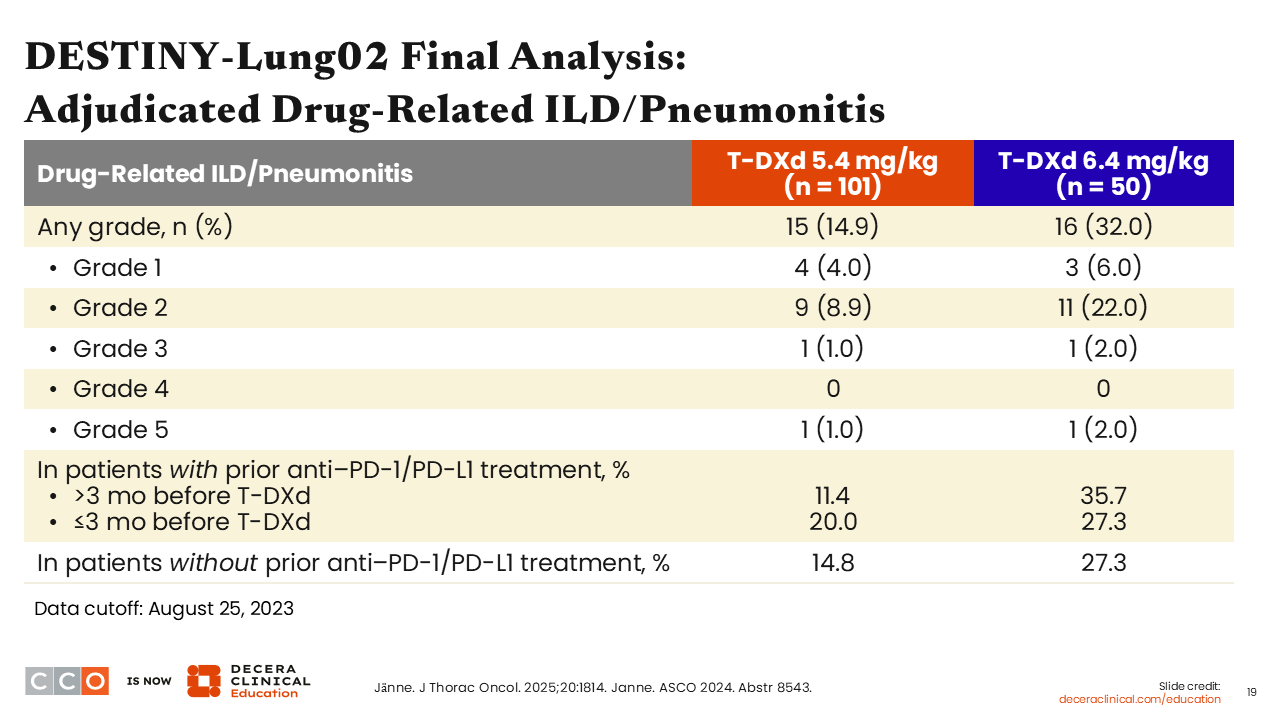
DESTINY-Lung02 Final Analysis: Adjudicated Drug-Related ILD/Pneumonitis
Estelamari Rodriguez, MD, MPH:
It is known that T-DXd is associated with the risk of developing ILD/pneumonitis. This AE is particularly important to monitor in patients with NSCLC because these patients are already experiencing shortness of breath because of their disease, and T-DXd is specifically targeting the cancer in the lungs. So, it is very important to be able to recognize early signs and symptoms of T-DXd–related ILD/pneumonitis, which can be fatal if not promptly treated. At the FDA-approved T-DXd dose of 5.4 mg/kg for patients with NSCLC, any-grade ILD/pneumonitis was reported in approximately 15% of patients on the DESTINY-Lung02 trial, and most were grade 1/2. Grade ≥3 ILD/pneumonitis was reported in only 2 of 101 patients (2%).
Among patients with prior exposure to an immune checkpoint inhibitor ≥3 months before receiving T-DXd, ILD/pneumonitis was reported in 20%. The incidence of ILD/pneumonitis in patients who had no prior exposure to an immune checkpoint inhibitor was approximately 15%.
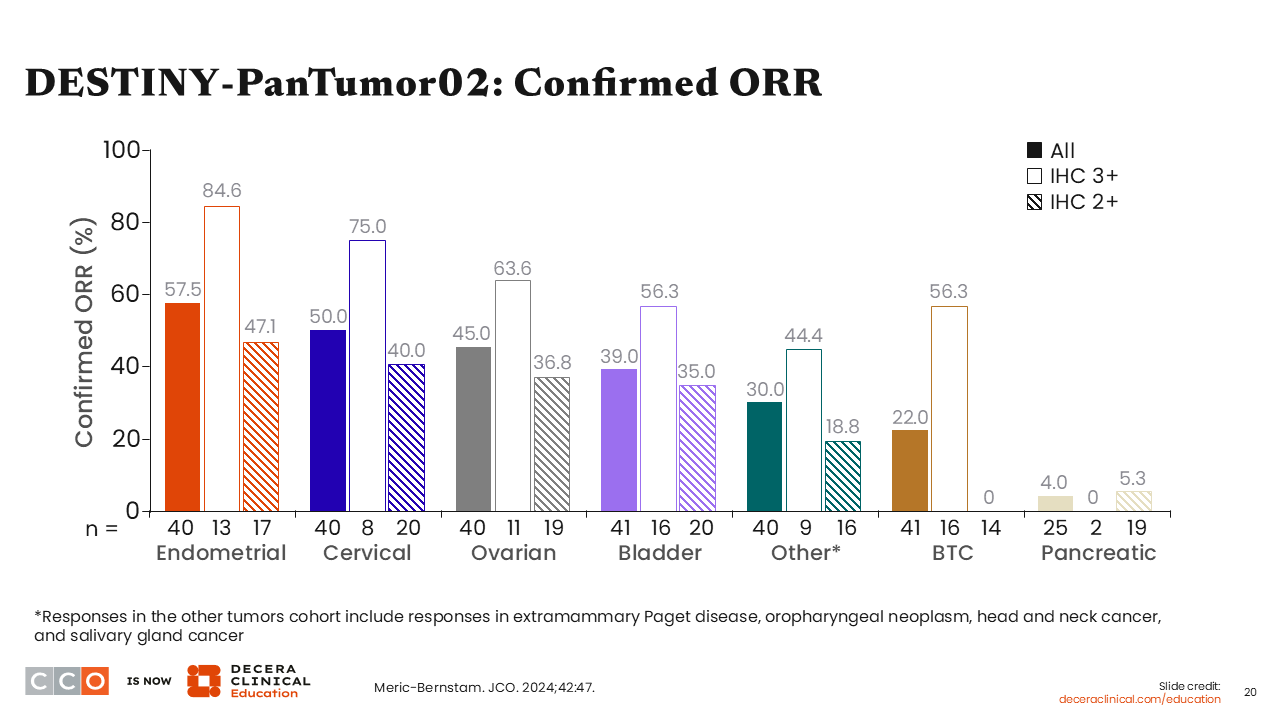
DESTINY-PanTumor02: Confirmed ORR
Estelamari Rodriguez, MD, MPH:
The multicohort phase II DESTINY-PanTumor02 trial investigated T-DXd for patients with HER2-expressing locally advanced, unresectable or metastatic solid tumors with disease progression following previous treatment and who have no satisfactory alternative treatment options (NCT04482309). Patients with somatic DNA mutations of HER2 without tumoral HER2 protein expression were excluded from participating in this trial. The ORR was significantly greater in patients with HER2 IHC 3+ compared with those with disease harboring HER2 IHC 2+ in patients with endometrial, cervical, ovarian, bladder, and biliary tract cancers and other solid tumor types such as head and neck cancer and salivary gland cancer.40 These results from the DESTINY-PanTumor02 trial contributed to the FDA’s approval of T-DXd for patients with unresectable or metastatic HER2-positive (IHC 3+) solid tumors who have received prior systemic treatment and have no satisfactory alternative treatment options.
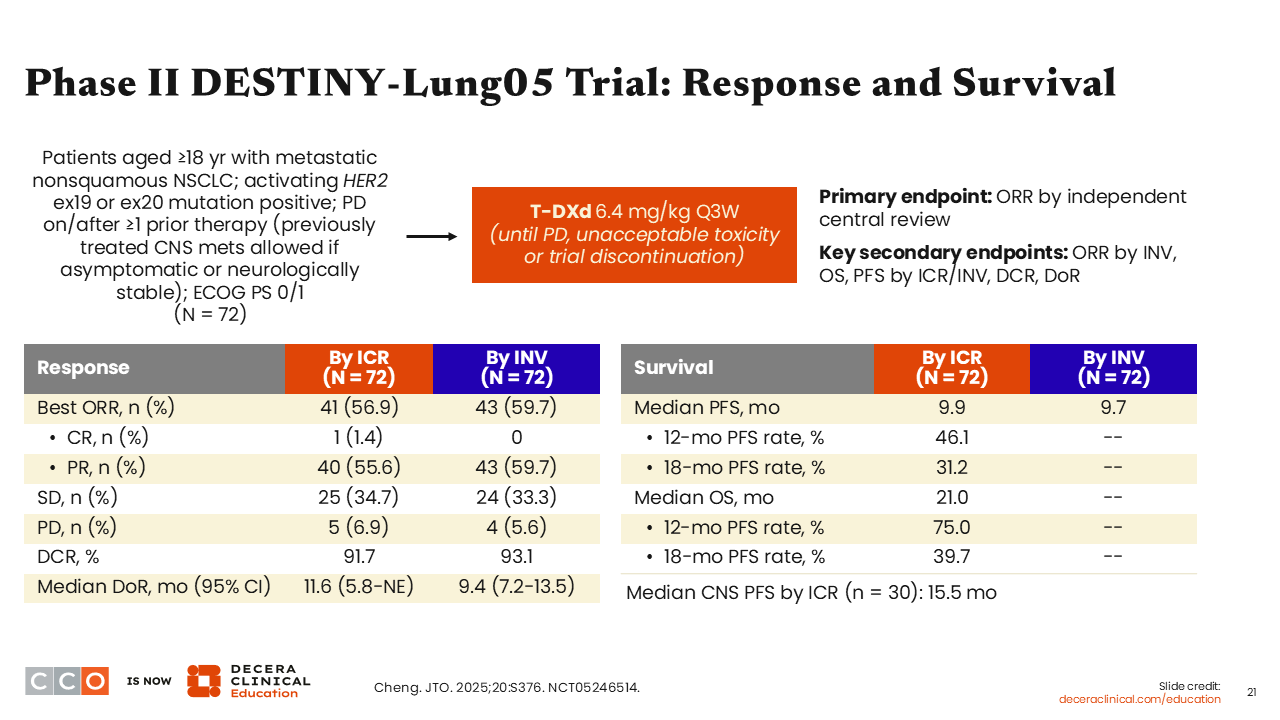
Phase II DESTINY-Lung05 Trial: Response and Survival
Estelamari Rodriguez, MD, MPH:
Another important trial is the single-arm phase II DESTINY-Lung05 trial that investigated T-DXd for patients with HER2 mutation–positive metastatic nonsquamous NSCLC with disease progression on or after ≥1 line of treatment (NCT05246514). Patients with previously treated brain metastases were allowed if asymptomatic or neurologically stable. In total, 72 patients received 6.4 mg/kg of T-DXd until disease progression, unacceptable toxicity, or trial discontinuation. The primary endpoint was ORR by independent central review (ICR), and the secondary endpoints included PFS and OS.
The ORR by ICR was 56.9%, including 1.4% who achieved a complete response and 55.6% who achieved a partial response.41 The DCR by ICR was impressive at 91.7% with a median DoR of 11.6 months. The median PFS by ICR was 9.9 months and the median OS was 21.0 months. Of note, the median CNS PFS among 30 patients with brain metastasis was 15.5 months. These impressive results underscore the efficacy of T-DXd in patients with HER2 mutation–positive NSCLC, including intracranial activity in those with stable CNS metastasis.
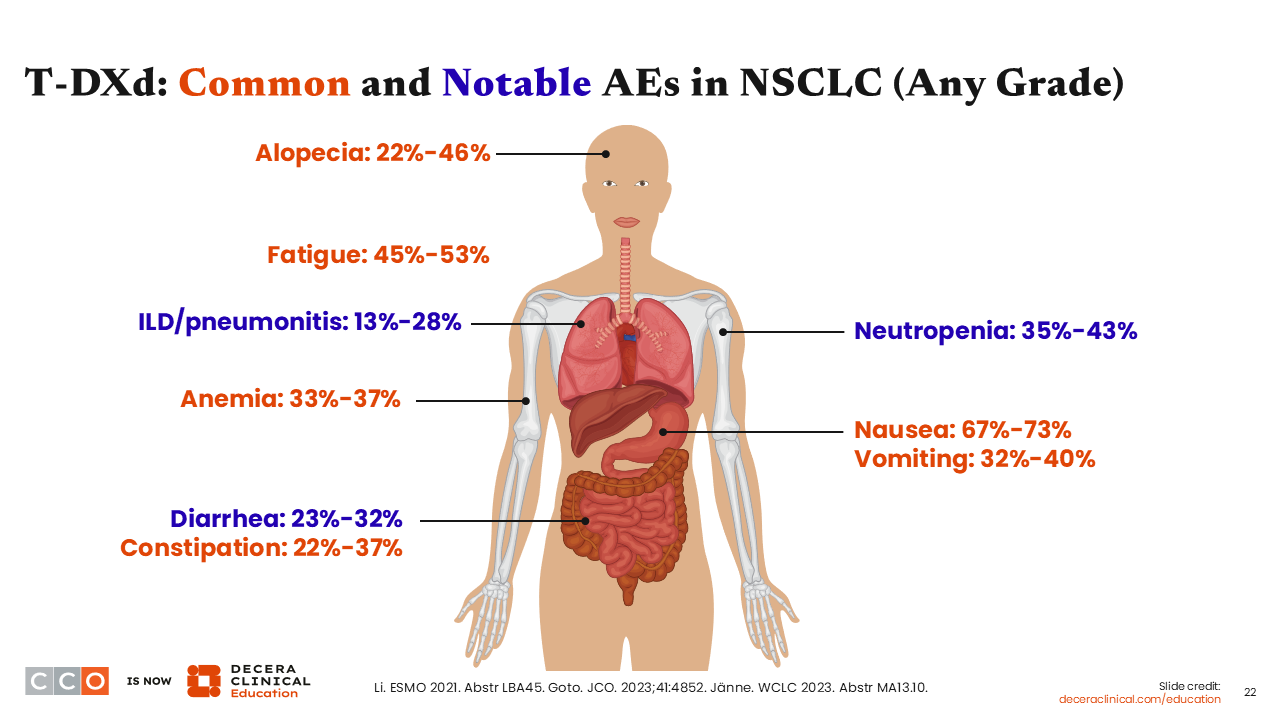
T-DXd: Common and Notable AEs in NSCLC (Any Grade)
Estelamari Rodriguez, MD, MPH:
Like any other agent used in cancer treatment, the use of T-DXd leads to some AEs. The common and notable AEs associated with T-DXd include alopecia, fatigue, nausea, vomiting, diarrhea, constipation, neutropenia, and anemia.22,42,43 Since these AEs are commonly reported, patients need to be educated on how to identify the initial signs and how to promptly manage them when they occur. Some patients may require dose interruption or the use of granulocyte colony-stimulating factor (G-CSF) when some T-DXd–associated toxicities occur.
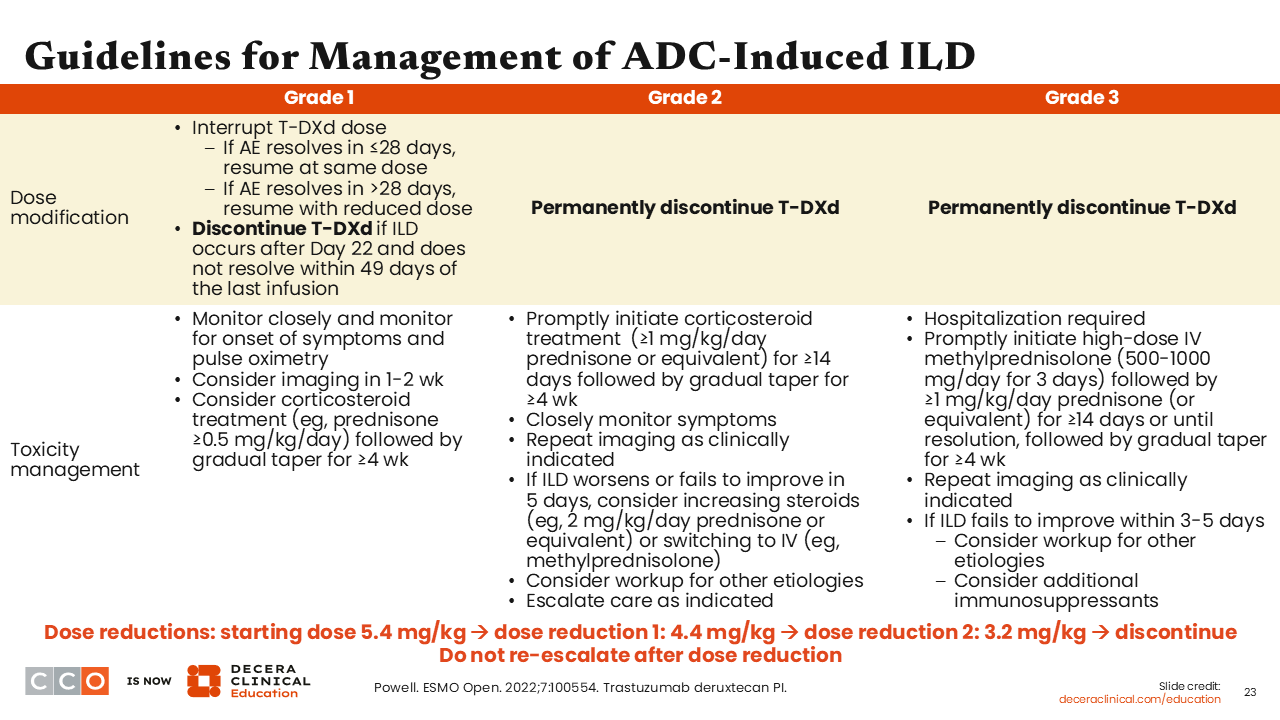
Guidelines for Management of ADC-Induced ILD
Estelamari Rodriguez, MD, MPH:
As I previously alluded to, ILD/pneumonitis is rare but this is an AE of special interest that is associated with T-DXd. So, as soon as ILD/pneumonitis is suspected, T-DXd should be held until diagnosis is confirmed. All other causes of ILD/pneumonitis, such as AEs associated with radiotherapy or use of other concomitant medications, and other pathologies, such as infection or pulmonary embolism, should be confidently ruled out using appropriate imaging techniques.22,44,45 Laboratory tests for complete blood counts (CBCs), tumor markers, and autoimmune antibodies should also be performed as indicated.
Screening for ILD/pneumonitis is important at baseline and regularly during treatment with T-DXd. It is crucial to ensure that the patient has no sign(s) or symptom(s) of ILD at baseline and ascertain that the patient has no known history of ILD/pneumonitis based on the most recent treatments received and on the patient’s most recent scans. Regularly monitoring for signs and symptoms of ILD/pneumonitis when receiving T-DXd is important.
For asymptomatic ILD/pneumonitis (grade 1), treatment with T-DXd should be interrupted until resolution to grade 0. If symptoms resolve ≤28 days from onset, T-DXd can be resumed at the same dose. However, if symptoms resolve >28 days from the time of onset, T-DXd dose should be reduced by 1 dose level as stated in the prescribing information. The use of corticosteroids should be considered as soon as ILD/pneumonitis is suspected. Of note, in NSCLC, the recommended starting dose of T-DXd is 5.4 mg/kg. The first dose reduction is to 4.4 mg/kg and the second dose reduction is to 3.2 mg/kg as indicated. For symptomatic ILD/pneumonitis (grade ≥2), T-DXd should be permanently discontinued with the prompt administration of corticosteroids as soon as ILD/pneumonitis is suspected.
In my practice, I typically administer corticosteroids as soon as I suspect ILD/pneumonitis, even when the patient is asymptomatic. By so doing, I am hoping that corticosteroid use will help with the rapid resolution of the ILD/pneumonitis, especially because my intention is to resume treatment with T-DXd in a timely fashion.
Monitoring Left Ventricular Dysfunction With T-DXd
Estelamari Rodriguez, MD, MPH:
Left ventricular dysfunction is another T-DXd–associated AE of special interest.22 Therefore, it is important to assess left ventricular ejection fraction (LVEF) before initiating T-DXd, and these assessments should also be performed at regular intervals when the patient is receiving T-DXd as clinically indicated. If the LVEF >45% and the absolute decrease from baseline is 10% to 20%, treatment with T-DXd should be continued. If the LVEF is 40% to 45% and the absolute decrease from baseline is <10%, treatment with T-DXd should be continued and LVEF assessments should be repeated within 3 weeks. If the LVEF is 40% to 45% and the absolute decrease from baseline is 10% to 20%, treatment with T-DXd should be interrupted and LVEF assessments should be repeated within 3 weeks. If the LVEF has not recovered to within 10% from baseline within 3 weeks, T-DXd should be permanently discontinued. However, if LVEF recovers to within 10% from baseline, treatment with T-DXd should be resumed at the same dose.
When the LVEF is <40% or absolute decrease from baseline is >20%, T-DXd should be interrupted with LVEF reassessment within 3 weeks. If LVEF is <40% or absolute decrease from baseline is >20%, T-DXd should be permanently discontinued. For patients with symptomatic congestive heart failure, T-DXd should be permanently discontinued.
Managing Neutropenia With T-DXd
Estelamari Rodriguez, MD, MPH:
Patients should be educated about the risk of developing neutropenia when receiving T-DXd, which is typically low grade. CBCs should be evaluated before T-DXd initiation, before each dose every 3 weeks, and as clinically indicated.22,46,47 For grade 3 neutropenia (absolute neutrophil count [ANC] 0.5 to <1.0 x 109/L), T-DXd should be interrupted until resolution to grade ≤2, and then restarted at the same dose. For grade 4 neutropenia (ANC <0.5 x 109/L), T-DXd should be interrupted until resolution to grade ≤2 and dose reduced by 1 level. For febrile neutropenia with ANC <1.0 x 109/L plus temperature >38.3ºC (>101°F) or a sustained temperature ≥38ºC (≥100.4°F) for more than 1 hour, T-DXd should be interrupted until symptom resolution and dose reduced by 1 level. Also, G-CSF prophylaxis should be considered for patients with a history of prior neutropenic complications.
It is important to educate patients about recognizing neutropenic fever. In my practice, we monitor patients’ CBCs regularly and inform patients of the median time to onset of 16-22 days from treatment initiation. For our patients with low baseline CBCs, we typically consider the use of G-CSF prophylaxis.