CE / CME
Translating Evidence to Action in Relapsed ES-SCLC: Integrating New Targeted Agents Into Clinical Practice
Physician Assistants/Physician Associates: 0.50 AAPA Category 1 CME credit
Pharmacists: 0.50 contact hour (0.05 CEUs)
ABIM MOC: maximum of 0.50 Medical Knowledge MOC point
Physicians: maximum of 0.50 AMA PRA Category 1 Credit™
Nurse Practitioners/Nurses: 0.50 Nursing contact hour
Released: November 25, 2025
Expiration: May 24, 2026
Activity
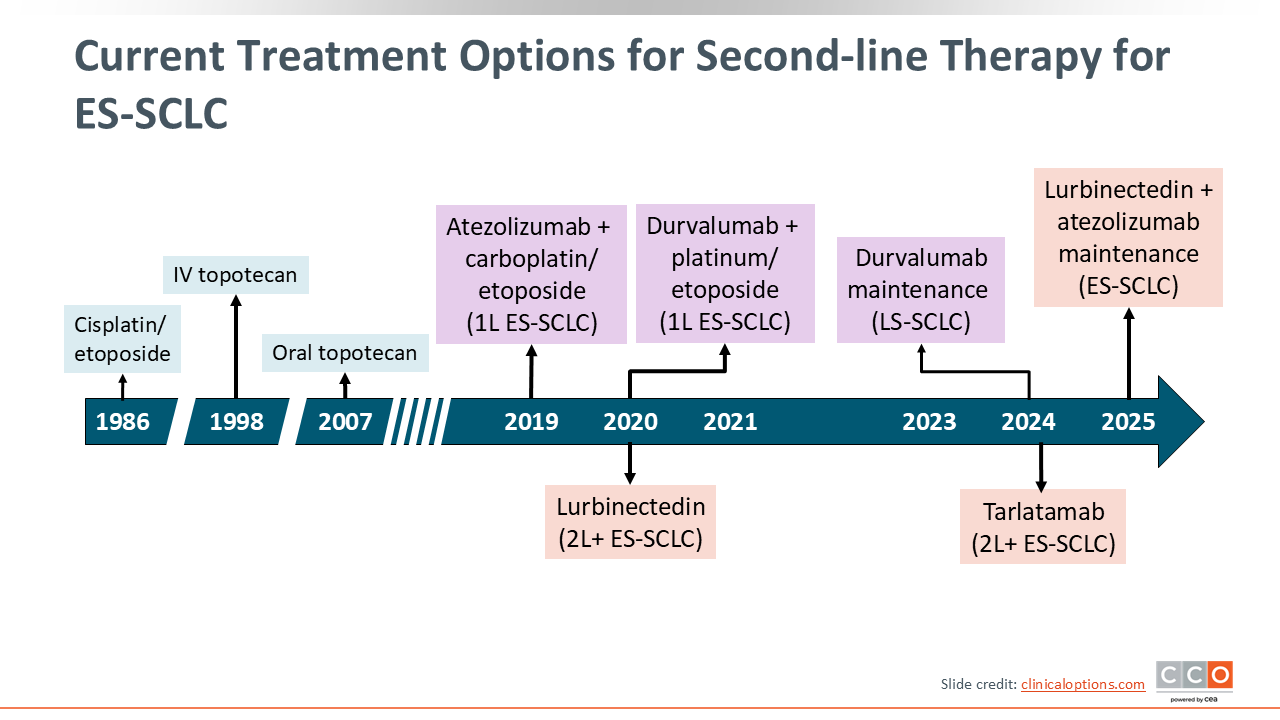
Current Treatment Options for Second-line Therapy for ES-SCLC
Despite uptake of first-line chemoimmunotherapy and new maintenance strategies, most patients with ES-SCLC relapse and require second-line therapy. In the United States, historical activity with topotecan has been modest (ORR: ~18%-22%; median overall survival [OS]: ~7.6-8.0 months) for patients with relapsed ES-SCLC.1 However, recent therapeutic advances have significantly reshaped the management of SCLC, including in the relapsed setting. Most notably, lurbinectedin (an alkylating transcription inhibitor), tarlatamab (a DLL3-targeting BiTE), and novel immunotherapy combinations or new agents on the horizon have demonstrated significant efficacy for patients with SCLC.
In the relapsed ES-SCLC setting, lurbinectedin monotherapy improved on these benchmarks (ORR: ~35%; median progression-free survival [PFS]: ~3.5 months; median OS: ~9.3 months).2 The phase II DeLLphi-301 showed similar efficacy results (ORR: 40%; median duration of disease control: 6.9 months) with 10 mg dosing and, more recently, the phase III DeLLphi-304 showed tarlatamab significantly improved OS (13.6 vs 8.3 months) and PFS (4.2 vs 3.7 months) vs investigator’s choice of chemotherapy, establishing a new evidence-based second-line option for eligible patients.3
Clinical trial participation remains important as ongoing studies continue to evaluate strategies to further improve second-line outcomes.
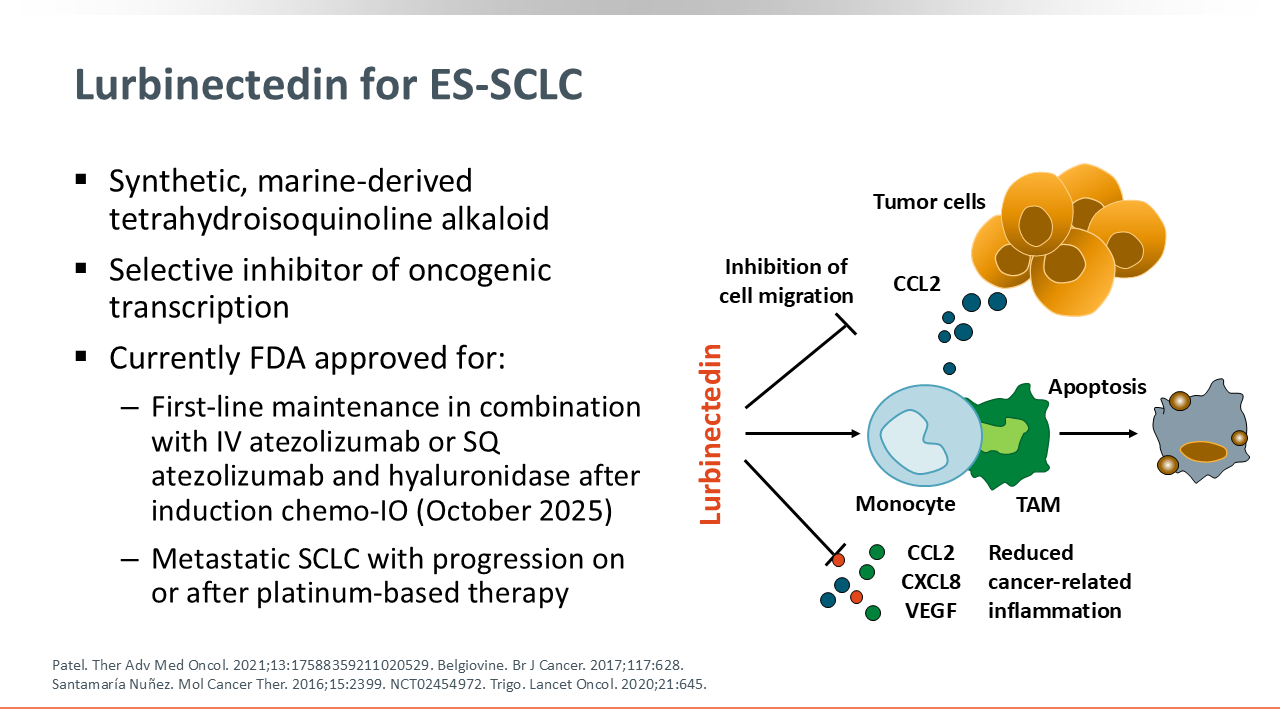
Lurbinectedin for ES-SCLC
Lurbinectedin is a synthetic, marine-derived tetrahydroisoquinoline alkaloid and selective inhibitor of oncogenic transcription. Based on current clinical trials, lurbinectedin is approved by the FDA as first-line maintenance in combination with IV atezolizumab or SQ atezolizumab and hyaluronidase after induction chemoimmunotherapy for patients with ES-SCLC whose disease responded to initial therapy with chemoimmunotherapy and as a single-agent treatment for patients with metastatic SCLC and disease progression on or after platinum-based therapy.4
Additional information on treatment options for limited-stage SCLC and first-line treatment for ES-SCLC can be found here.
The approval for lurbinectedin for relapsed SCLC was based on the phase II basket study Study B-005.
Phase II Basket Study (Study B-005): Lurbinectedin in ES-SCLC Cohort
The basket study Study B-005 was a single-arm, open-label phase II trial evaluating lurbinectedin in patients with ES-SCLC after 1 prior platinum-based chemotherapy regimen. Enrolled patients had an Eastern Cooperative Oncology Group performance status of ≤2 and no brain metastases. In total, 105 patients were treated with lurbinectedin at a dose of 3.2 mg/m² administered intravenously every 3 weeks. Treatment continued until disease progression (as defined by Response Evaluation Criteria in Solid Tumors) or the occurrence of unacceptable toxicity, per investigator discretion.
The study’s primary endpoint was the investigator-assessed ORR. Secondary endpoints included duration of response (DoR), DCR, PFS, OS, and safety outcomes.
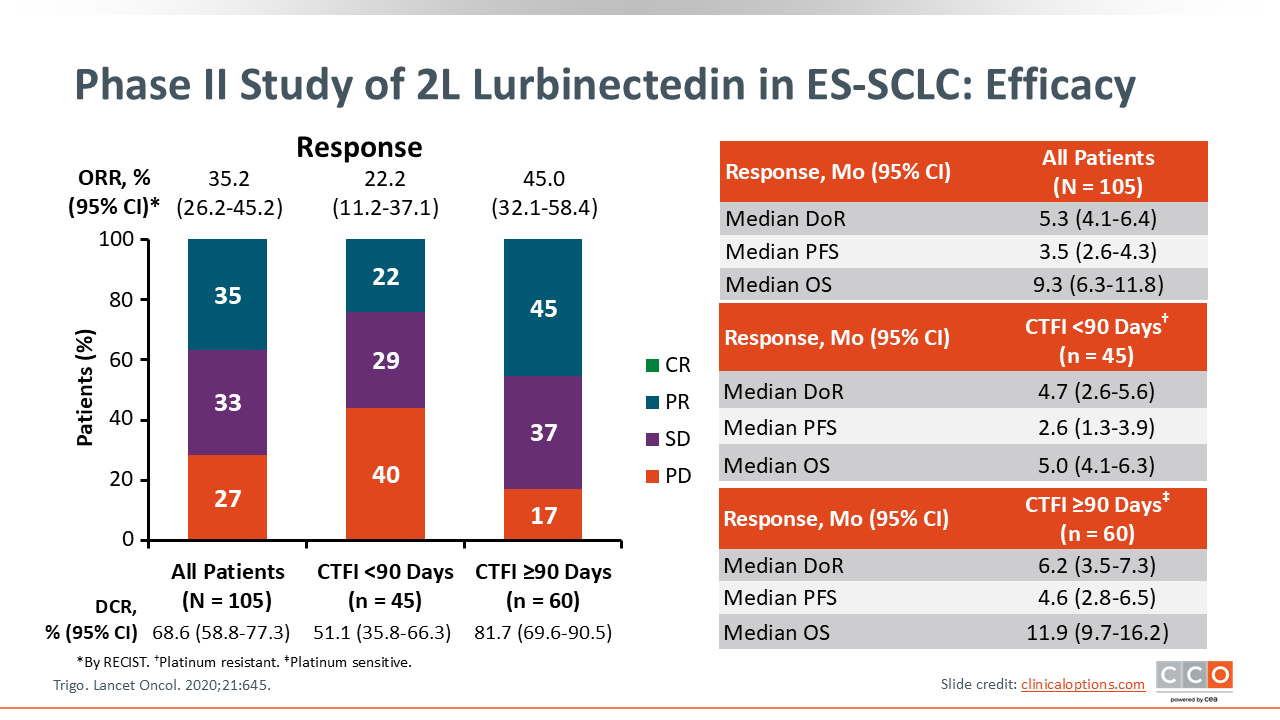
Phase II Study of 2L Lurbinectedin in ES-SCLC: Efficacy
The ORR with lurbinectedin was 35.2% (95% CI: 26.2%-45.2%), with a DCR of 68.6%. When stratified by chemotherapy-free interval (CTFI)—a measure of sensitivity to prior platinum therapy—patients with CTFI <90 days (platinum resistant) had a lower ORR of 22.2%, whereas those with CTFI ≥90 days (platinum sensitive) achieved a higher ORR of 45.0%. The distribution of responses shows a larger percentage of PD among the resistant group (40%) compared with the sensitive group (17%).
Among all patients, the median DoR was 5.3 months, median PFS was 3.5 months, and median OS was 9.3 months. In the platinum-resistant population, outcomes were shorter, with a median DoR of 4.7 months, median PFS of 2.6 months, and median OS of 5.0 months. Conversely, patients with platinum-sensitive disease experienced improved outcomes, with median DoR of 6.2 months, median PFS of 4.6 months, and median OS of 11.9 months. Lurbinectedin showed meaningful activity in relapsed SCLC, with particularly favorable efficacy in patients with platinum-sensitive relapse.
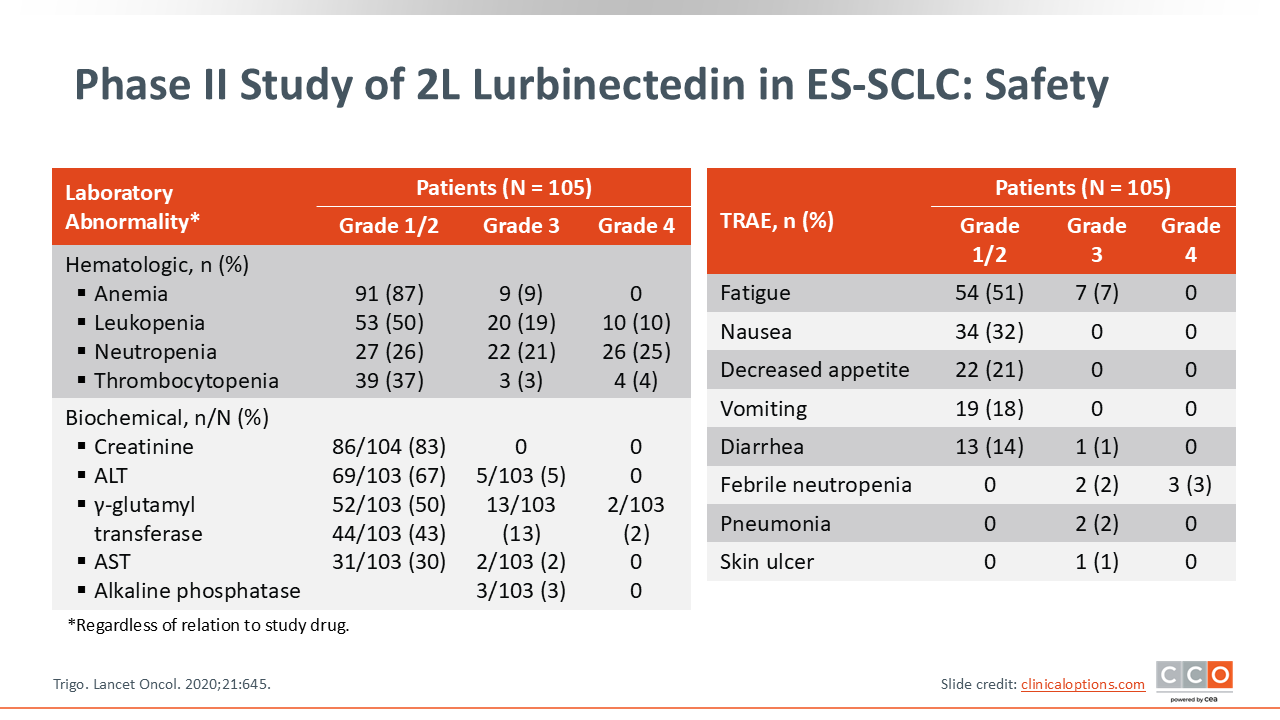
Phase II Study of 2L Lurbinectedin in ES-SCLC: Safety
Overall, lurbinectedin treatment was associated with manageable hematologic toxicities and generally mild nonhematologic adverse events (AEs).
Among hematologic laboratory abnormalities, the most common were anemia (87%), leukopenia (50%), neutropenia (26%), and thrombocytopenia (37%). Of note, grade 3/4 neutropenia occurred in nearly one half of patients (21% grade 3; 25% grade 4), and grade 3/4 leukopenia occurred in 29% (19% grade 3; 10% grade 4). Severe anemia and thrombocytopenia were less frequent.
Biochemical abnormalities were also observed but were mostly low grade. These included elevations in creatinine (83%), alanine aminotransferase (67%), γ-glutamyl transferase (50%), and aspartate aminotransferase (30%), with few grade ≥3 AEs.
The most common treatment-related AEs were fatigue (51%), nausea (32%), decreased appetite (21%), and vomiting (18%), all primarily grade 1/2. Serious AEs such as febrile neutropenia (5%), pneumonia (2%), and skin ulcer (1%) were infrequent.
Overall, these findings suggested that the toxicity profile of lurbinectedin is largely driven by myelosuppression, consistent with its mechanism of action, but other AEs are typically mild and manageable with supportive care and dose adjustments. Additional information on managing AEs associated with lurbinectedin and other treatment options for ES-SCLC can be found here.
Lurbinectedin Combinations: Lurbinectedin Plus Irinotecan in Relapsed SCLC
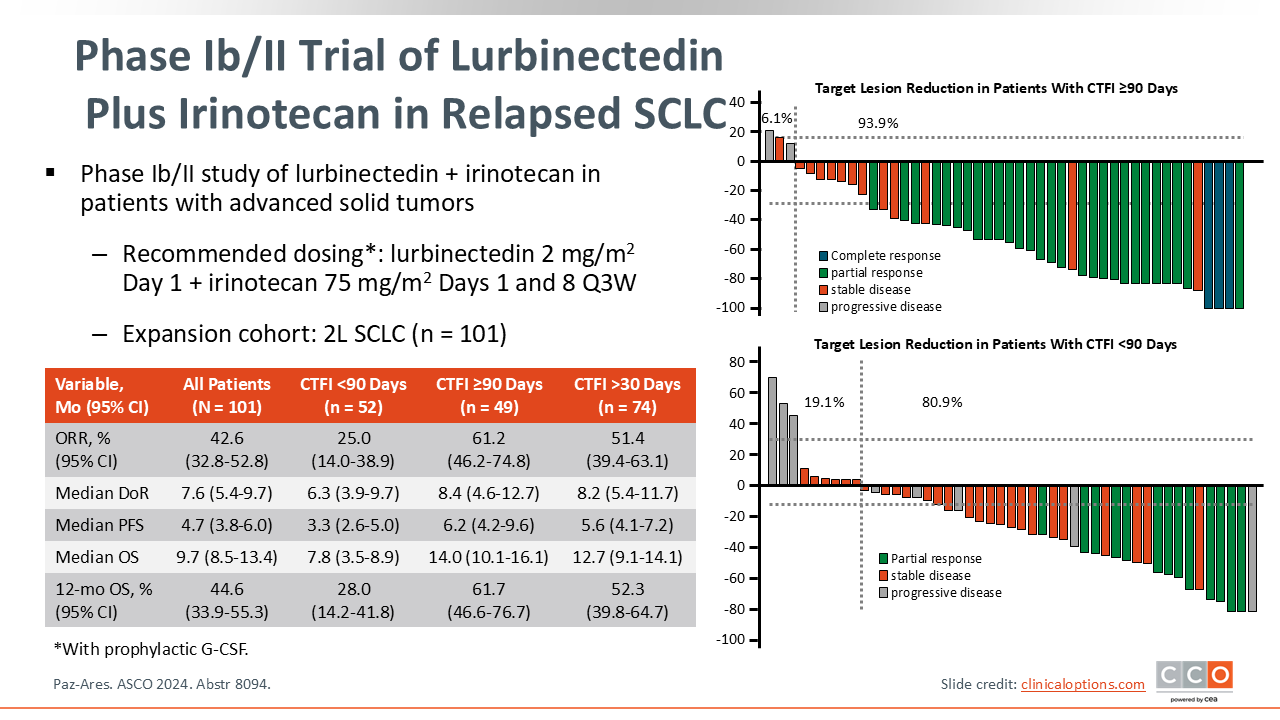
Novel approaches are also exploring lurbinectedin in various new combinations. A phase I/II study evaluated lurbinectedin plus irinotecan in patients with solid tumors, including an expansion cohort of 101 patients with relapsed SCLC after 1 prior line of therapy with a platinum-based chemotherapy with or without immunotherapy.
The combination showed promising results in the SCLC cohort, with an overall ORR of 42.6% (95% CI: 32.8-52.8). The benefit was most pronounced in patients with a CTFI of ≥90 days, who achieved an ORR of 61.2% (95% CI: 46.2-74.8) compared with those with a CTFI <90 days who achieved an ORR of 25.0% (95% CI: 14.0-38.9). The median OS for the overall cohort was 9.7 months (95% CI: 8.5-13.4), and the median OS in the platinum-sensitive group was 14.0 months (95% CI: 10.1-16.1). This trial also assessed efficacy in patients with CTFI >30 days and showed promising efficacy, with an ORR of 51.4 (95% CI: 39.4-63.1) and median OS of 12.7 months (95% CI: 9.1-14.1).5
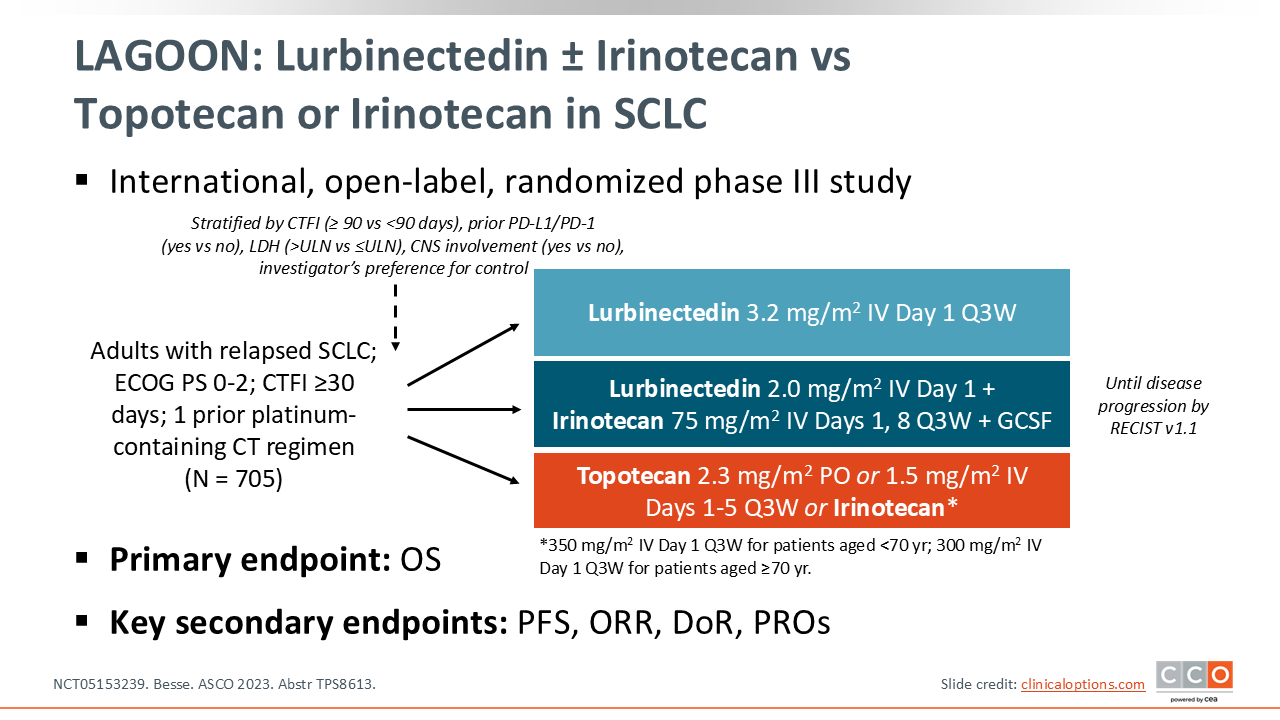
LAGOON: Lurbinectedin ± Irinotecan vs Topotecan or Irinotecan in SCLC
Based on the promising phase I/II data, the randomized phase III LAGOON trial is currently underway. This international, open-label, randomized study is enrolling patients with relapsed SCLC who have received 1 prior platinum-containing and have a CTFI of ≥30 days.
The 705 patients who enroll in this trial will be randomized to 1 of 3 arms: lurbinectedin monotherapy 3.2 mg/m2, lurbinectedin 2.0 mg/m² plus irinotecan 75 mg/m2, or investigator's choice of standard of care (SoC) with topotecan or irinotecan. The primary endpoint is OS and secondary endpoints include PFS, ORR, DoR, and patient-reported outcomes (NCT05153239).6
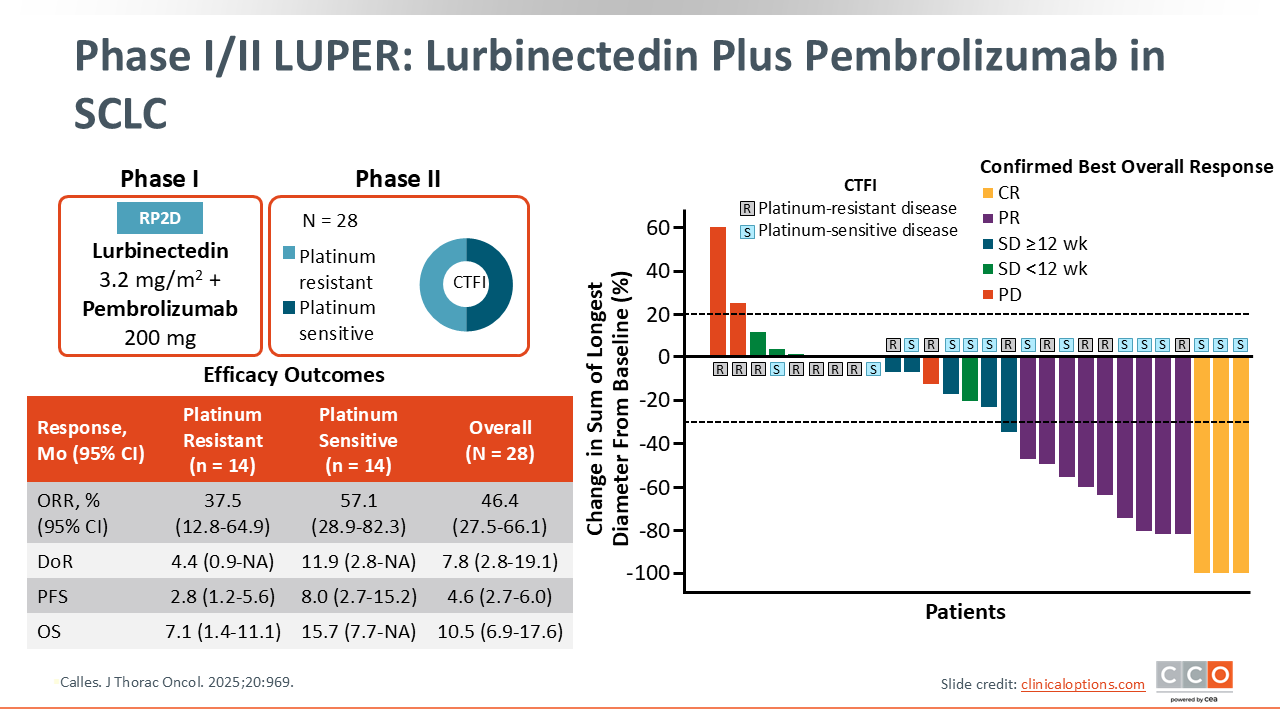
Phase I/II LUPER: Lurbinectedin + Pembrolizumab in SCLC
Lurbinectedin is also being evaluated in combination with checkpoint inhibitors. The phase I/II LUPER study evaluated lurbinectedin plus pembrolizumab in patients with relapsed SCLC after prior chemotherapy who had not received prior immunotherapy.
Overall, the combination was associated with an ORR of 46.4% (95% CI: 27.5-66.1), a median PFS of 4.6 months (95% CI: 2.7-6.0), and a median OS of 10.5 months (95% CI: 6.9-17.6).7
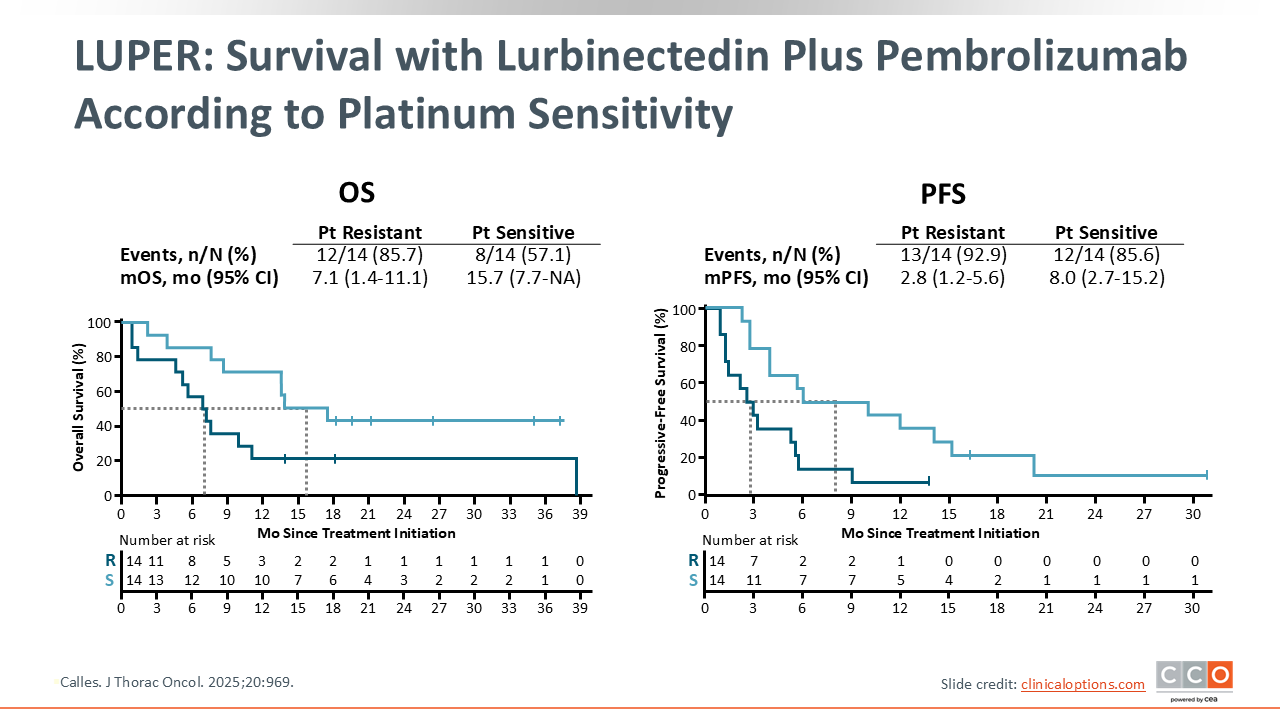
LUPER: Survival with Lurbinectedin Plus Pembrolizumab According to Platinum Sensitivity
A clear separation in outcomes was seen based on platinum sensitivity. Patients with platinum-sensitive disease (CTFI ≥90 days) had a median OS of 15.7 months (95% CI: 7.7-not estimable [NE]) and a median PFS of 8.0 months (95% CI: 2.7-15.2). This contrasts with the platinum-resistant cohort, who had a median OS of 7.1 months (95% CI: 1.4-11.1) and median PFS of 2.8 months (95% CI: 1.2-5.6).7
2SMALL: Lurbinectedin Plus Atezolizumab as 2L Treatment for Advanced SCLC
The phase I/II 2SMALL study evaluated lurbinectedin plus atezolizumab as a second-line treatment for patients who progressed after first-line platinum-based chemotherapy with or without immunotherapy who achieved CTFI ≥30 days. The trial stratified patients into 2 cohorts: immunotherapy naive (cohort 1) and immunotherapy exposed (cohort 2) (NCT04253145).8
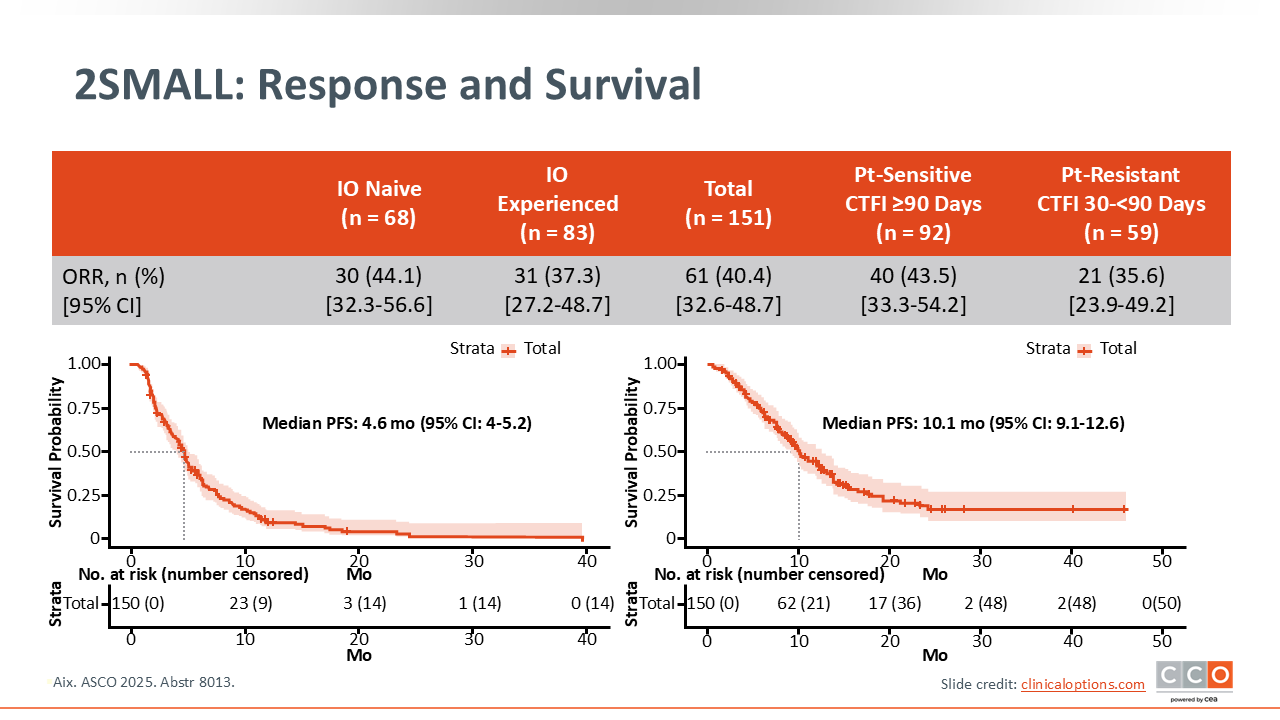
2SMALL: Response and Survival
The combination of lurbinectedin and atezolizumab showed consistent activity in both cohorts of patients. The ORR was 44.1% (95% CI: 32.3-56.6) in the immunotherapy-naive cohort and 37.3% (95% CI: 27.2-48.7) in the immunotherapy-exposed cohort. Across all 151 patients, the median PFS was 4.6 months (95% CI: 4.0-5.2), and the median OS was 10.1 months (95% CI: 9.1-12.6) (NCT04253145).8
Taken together, these data suggest a potential role for combination with lurbinectedin and various chemotherapy or immunotherapy agents for patients with relapsed ES-SCLC.
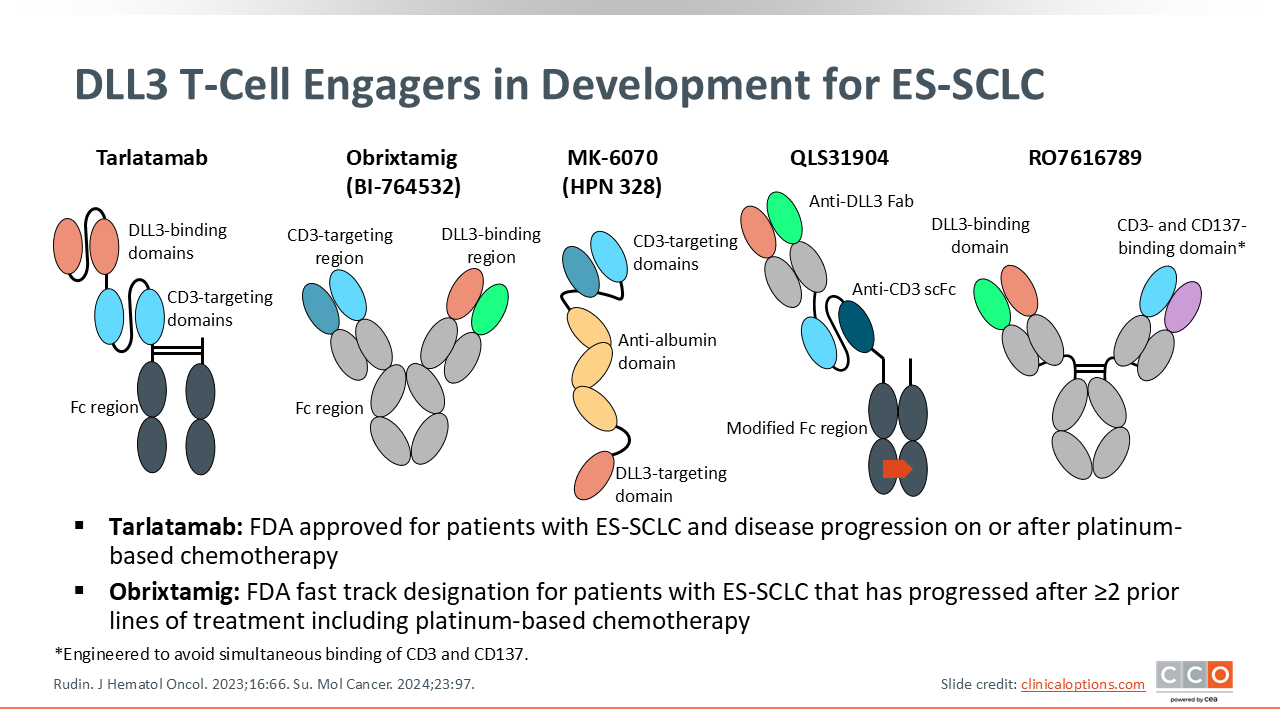
DLL3 T-Cell Engagers in Development for ES-SCLC
T-cell engagers (TCEs) targeting DLL3 represent a major advance in SCLC. DLL3 is a protein aberrantly overexpressed on the surface of most SCLC cells but is normally restricted to the Golgi apparatus in healthy cells.
Tarlatamab, a BiTE, links endogenous T-cells to these tumor cells. It binds to CD3 on T-cells and DLL3 on tumor cells, forcing an immunologic response. This leads to T-cell activation, proliferation, and the release of perforin and granzymes, resulting in serial, T-cell–mediated killing of the SCLC cells. Tarlatamab is currently approved by the FDA for the treatment of adult patients with ES-SCLC and disease progression on or after platinum-based chemotherapy, under accelerated approval.
Obrixtamig is a DLL3 x CD3 TCE with an IgG-like architecture, which is distinct from tarlatamab, and has received FDA Fast Track designation for patients with ES-SCLC that has progressed after ≥2 prior lines of treatment including platinum-based chemotherapy, for those with advanced or metastatic extrapulmonary neuroendocrine carcinoma (NEC) with disease progression after at least 1 prior line of treatment including platinum-based chemotherapy, and for those with advanced or metastatic DLL3-expressing large-cell NEC of the lung who experienced disease progression after ≥1 line of treatment including platinum-based chemotherapy.9,10
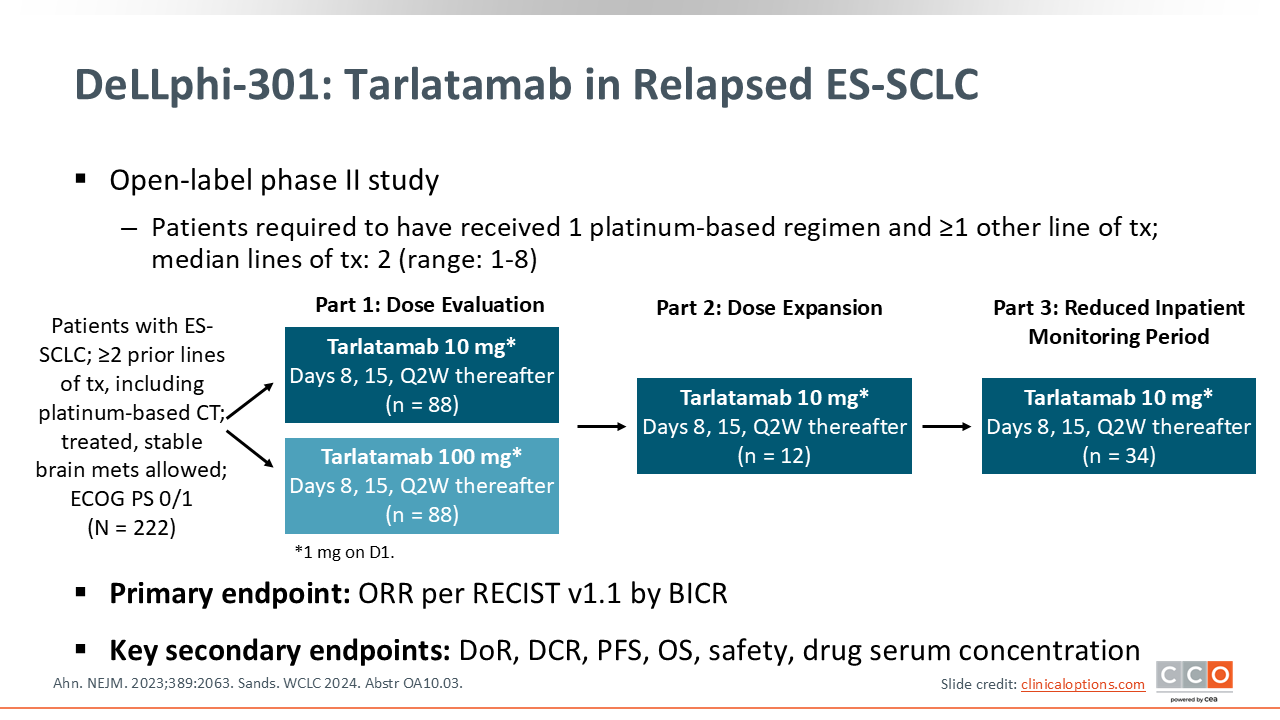
DeLLphi-301: Tarlatamab in Relapsed ES-SCLC
First, let’s talk about the data that led to the approval of tarlatamab. The phase II DeLLphi-301 study evaluated tarlatamab in patients with relapsed SCLC who had received at least 2 prior lines of therapy. The trial compared 2 doses—10 mg and 100 mg—administered every 2 weeks after a step-up dose. The primary endpoint was ORR and secondary endpoints included DoR, DCR, PFS, OS, and safety.11,12
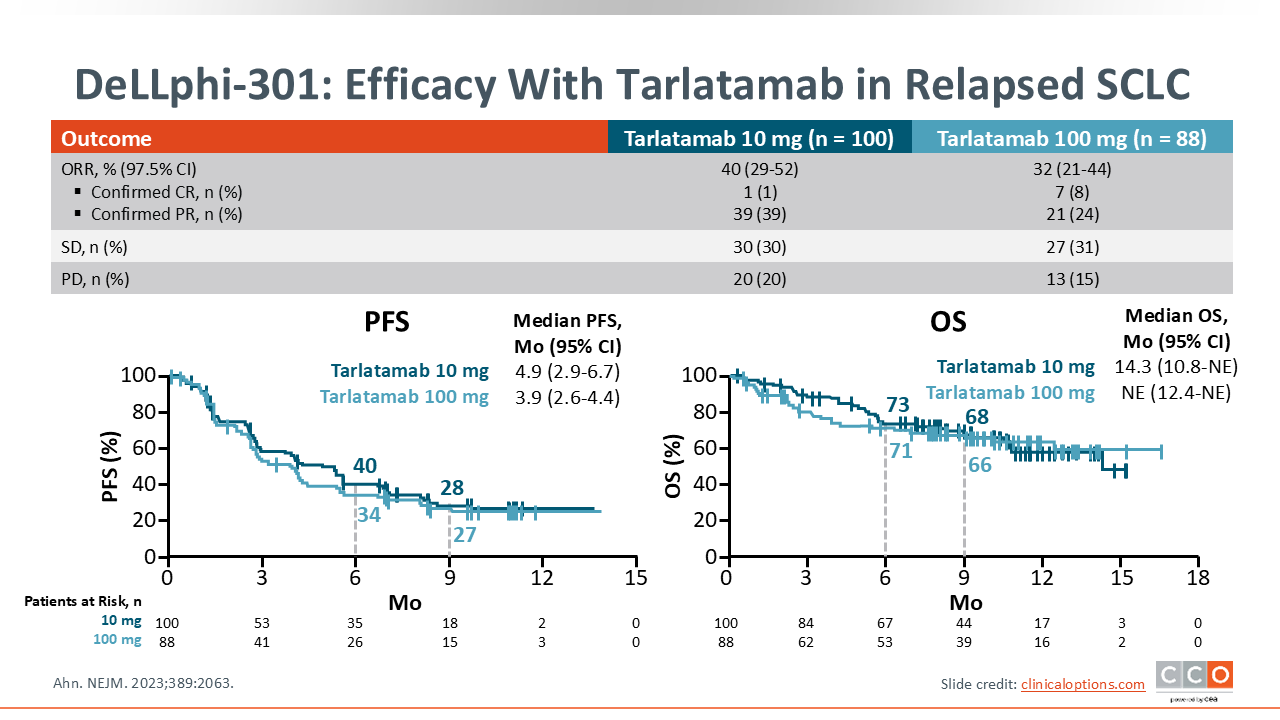
DeLLphi-301: Efficacy With Tarlatamab in Relapsed SCLC
In this trial, tarlatamab showed antitumor activity with a confirmed ORR of 40% (97.5% CI: 29-52) with 10-mg dosing and 32% (97.5% CI: 21-44) with 100-mg dosing. Median PFS was 4.9 months (95% CI: 2.9-6.7) with the 10-mg dose and 3.9 months (2.6-4.4) with the 100-mg dose, and median OS was 14.3 months (95% CI: 10.8-NE) 10-mg dose and not estimable with the 100-mg dose.
Based on this superior efficacy and a better safety profile, 10 mg was chosen as the recommended dose, leading to an FDA accelerated approval in 2024.11,12
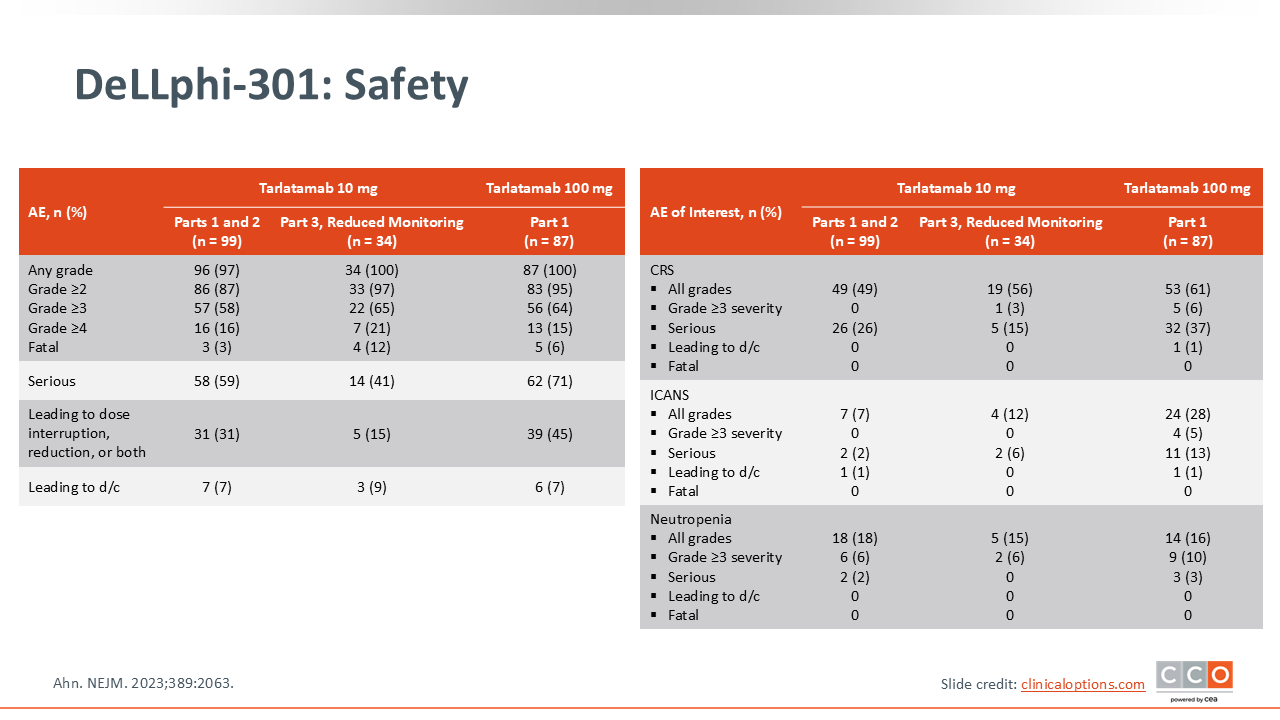
DeLLphi-301: Safety
The DeLLphi-301 trial established the manageable safety profile of the 10-mg tarlatamab dose. A key AE of interest is CRS, which occurred in 49% of patients (any grade) but was predominantly low-grade (no grade ≥3 AEs). Most CRS events were grade ½, occurred with the first or second dose, and were manageable with standard mitigation strategies.
Immune effector cell–associated neurotoxicity syndrome (ICANS) was much less frequent, occurring in 7% of patients (any grade), with no grade ≥3 AEs. Neutropenia occurred in 18% of patients (6% grade ≥3).11
Additional information on managing AEs associated with tarlatamab and other treatment options for ES-SCLC can be found here.
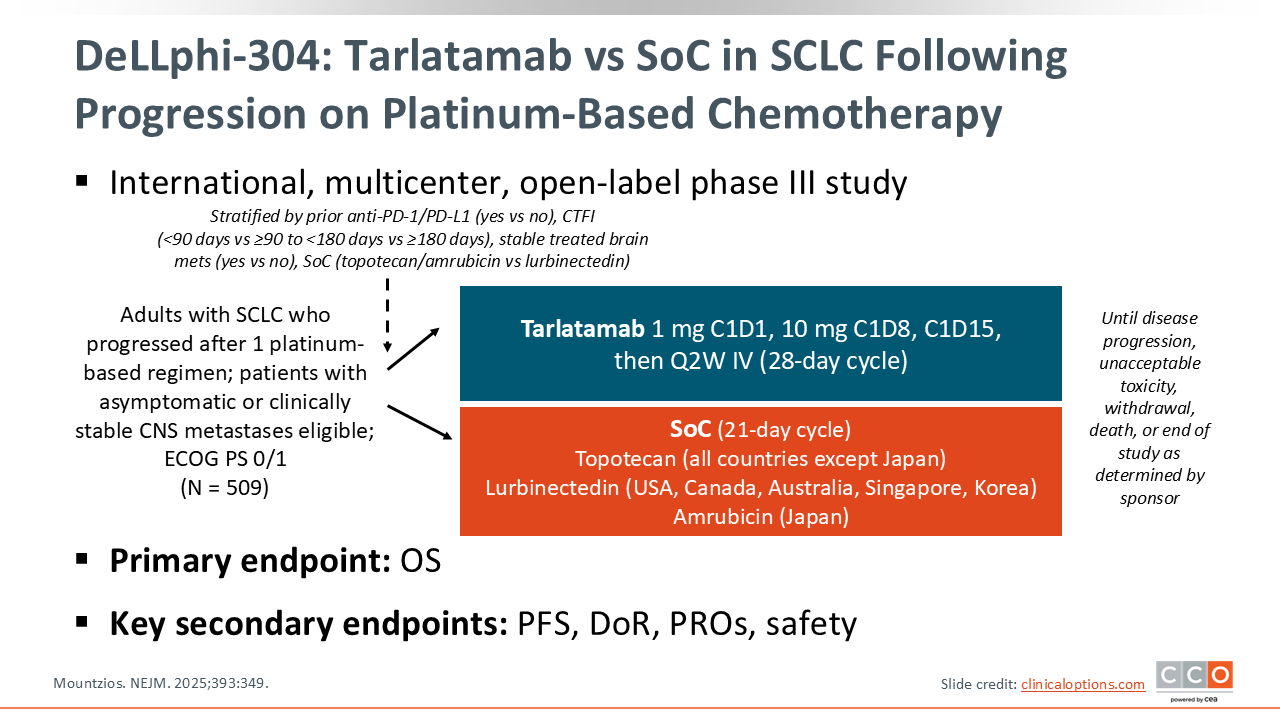
DeLLphi-304: Tarlatamab vs SoC in SCLC Following Progression on Platinum-Based Chemotherapy
The efficacy of tarlatamab has been confirmed in the phase III DeLLphi-304 trial. This international, multicenter, open-label study randomized 509 patients with ES-SCLC who progressed after 1 prior platinum-based regimen to receive either tarlatamab 10 mg every 2 weeks or SoC chemotherapy (topotecan, lurbinectedin, or amrubicin [Japan only]). The primary endpoint of this study was OS and key secondary endpoints included PFS, DoR, patient-reported outcomes, and safety.3
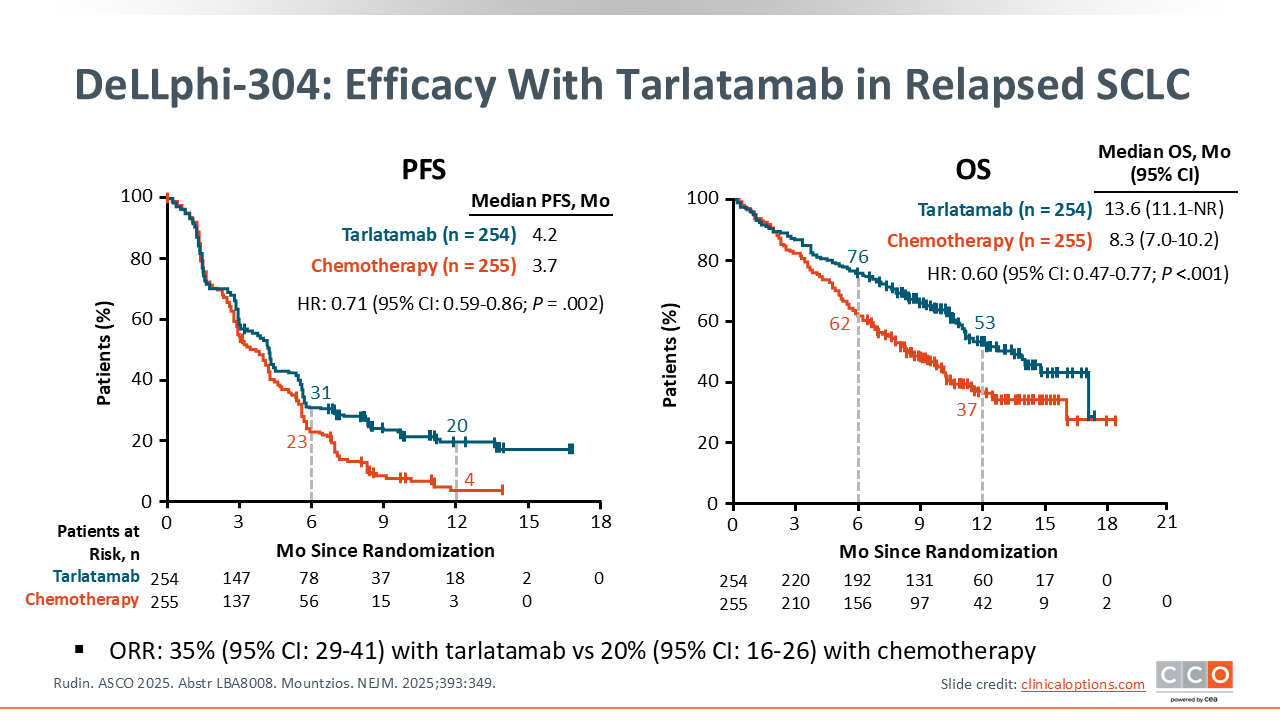
DeLLphi-304: Efficacy With Tarlatamab in Relapsed SCLC
The DeLLphi-304 study met its primary endpoint, demonstrating a statistically significant and clinically meaningful improvement in OS with tarlatamab compared with SoC: Median OS was 13.6 months (95% CI: 11.1-NE) with tarlatamab vs 8.3 months (95% CI: 7.0-10.2) with SoC (HR: 0.60; 95% CI: 0.47-0.77; P <.001).
Tarlatamab also showed significantly improved median PFS at 4.2 months vs 3.2 months with SoC (HR: 0.71; 95% CI: 0.59-0.86; P = .002) and ORR 35% (95% CI: 29-41) vs 20% (95% CI: 16-26).3
DeLLphi-303: Tarlatamab + PD-L1 Inhibitor as First-line Maintenance in ES-SCLC
Given its success in the relapsed setting, tarlatamab is being explored in earlier lines of therapy. The phase Ib DeLLphi-303 study evaluated tarlatamab 10 mg every 2 weeks plus a PD-L1 inhibitor (atezolizumab or durvalumab) as first-line maintenance therapy for patients who did not progress after 4-6 cycles of chemoimmunotherapy. The coprimary endpoints were rate of dose-limiting toxicities, treatment-emergent AEs, and treatment-related AEs, and key secondary endpoints included DCR, PFS (per local assessment by Response Evaluation Criteria in Solid Tumors v1.1), and OS.13
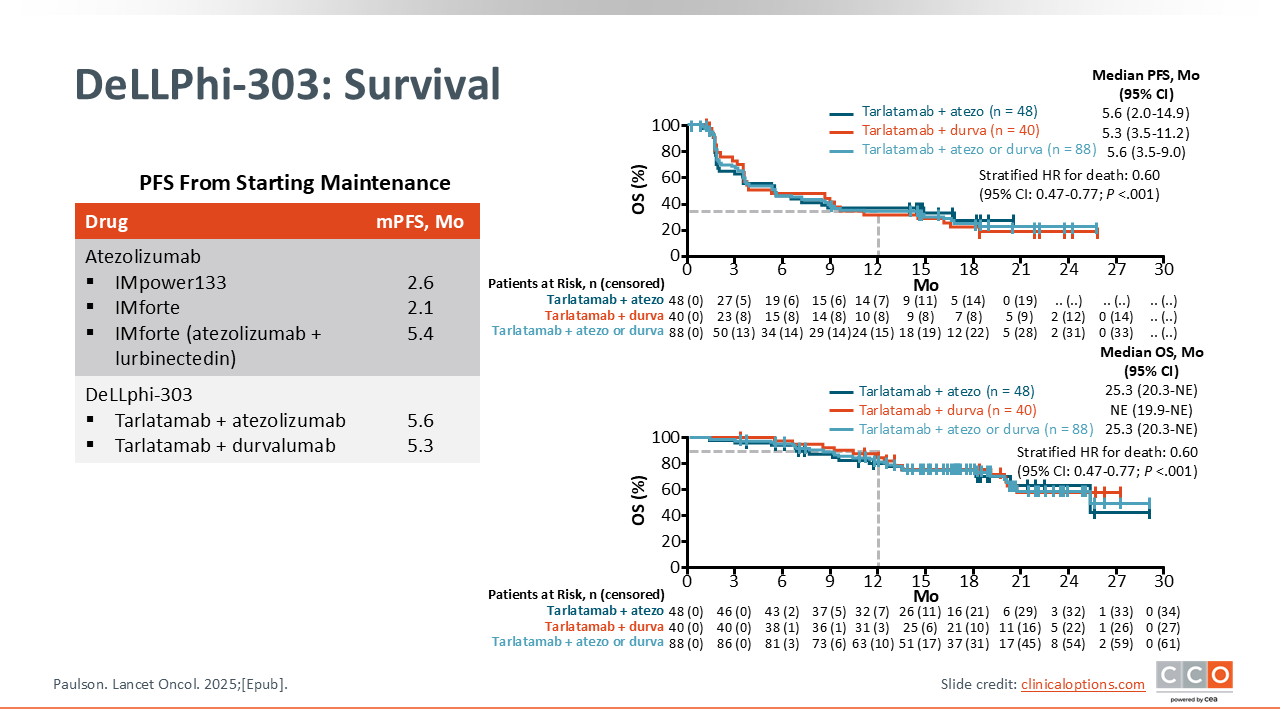
DeLLPhi-303: Survival
The combination of tarlatamab and a PD-L1 inhibitor was feasible and showed promising efficacy. The median PFS from the start of maintenance was 5.6 months (95% CI: 3.5-9.0) with tarlatamab plus atezolizumab or tarlatamab plus durvalumab. This compares favorably with the median PFS of 2.1-2.6 months seen with immunotherapy monotherapy maintenance in IMforte and IMpower133 and is similar to the 5.4 months seen with lurbinectedin plus atezolizumab in IMforte. The median OS with tarlatamab plus atezolizumab or tarlatamab plus durvalumab was 25.3 months (95% CI: 20.3-NE), supporting further investigation of this strategy.13
The phase III DeLLphi-305 trial is ongoing to evaluate tarlatamab as a first-line maintenance therapy in combination with durvalumab vs durvalumab alone for patients with ES-SCLC following initial treatment with platinum agent/etoposide and durvalumab (NCT06211036).
Obrixtamig in Patients With DLL3+ SCLC or Neuroendocrine Carcinomas
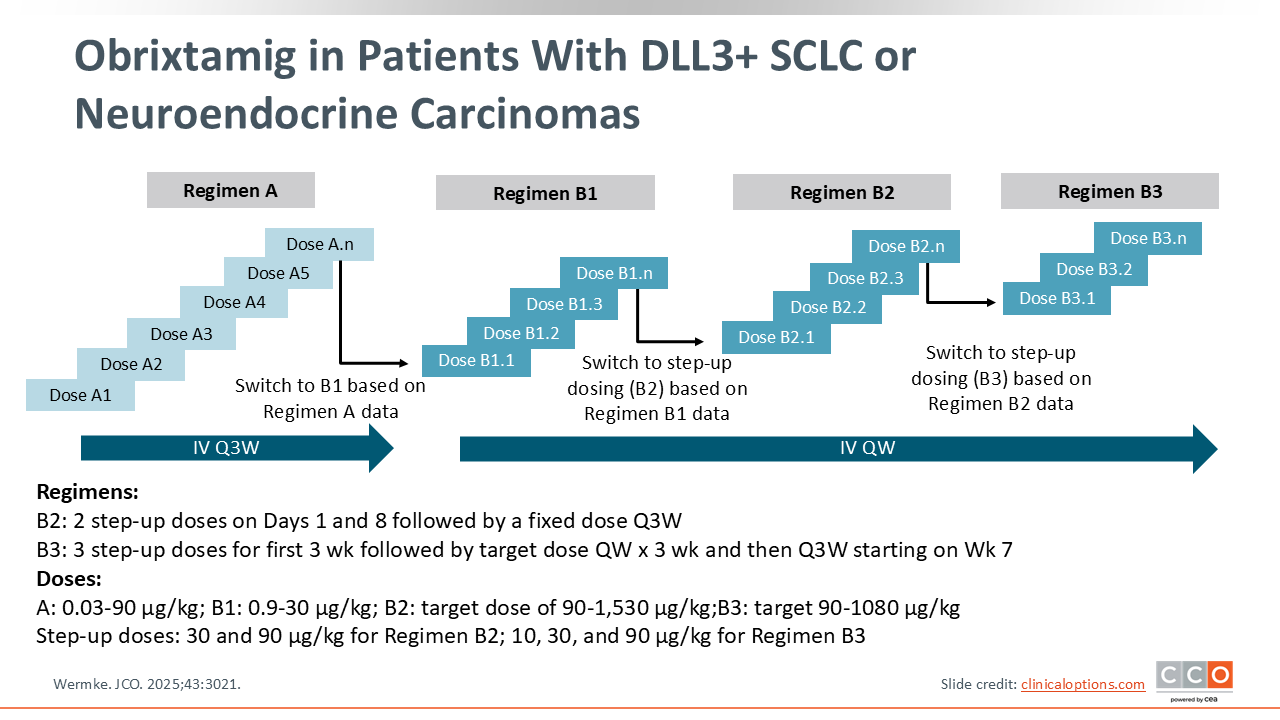
Obrixtamig is another DLL3-targeted TCE but with an IgG-like structure, distinct from the BiTE format of tarlatamab. A phase I dose-escalation and expansion study evaluated obrixtamig in patients with DLL3+ SCLC, extrapulmonary NECs, or large cell neuroendocrine carcinoma of the lung. Patients received escalating doses of obrixtamig IV in one of 4 regimens, including a fixed dose once every 3 weeks (regimen A); a fixed dose once weekly (regimen B1); a step-up dose once weekly for 2 weeks and then the target dose once weekly (regimen B2); and a step-up dose once weekly for 3 weeks, the target dose once weekly for 3 weeks, and then once every 3 weeks (regimen B3). The primary endpoint was finding the maximum tolerated dose (MTD) and secondary endpoints included safety, pharmacokinetics, and tumor response.14
Obrixtamig: Efficacy in Patients With Relapsed SCLC
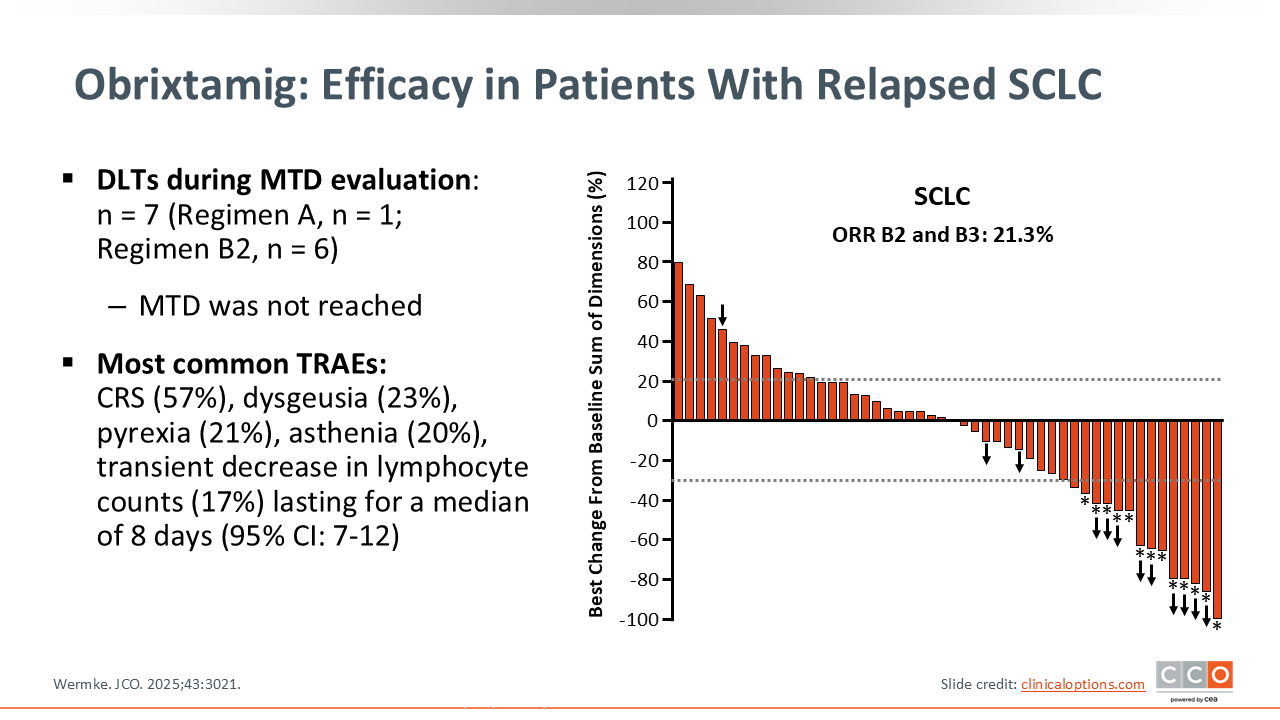
The study explored multiple regimens and doses, including 2-step (regimen B2) and 3-step (regimen B3) step-up dosing to mitigate CRS. During the MTD evaluation, 7 dose-limiting AEs were observed but the MTD was not reached. The most common treatment-related AEs included CRS (57%), dysgeusia (23%), pyrexia (21%), asthenia (20%), and transient lymphocyte decreases (17%), with lymphocyte recovery occurring after a median of 8 days.
Among patients with SCLC treated with the optimized regimens (regimen B2/regimen B3, doses ≥90 µg/kg), the confirmed ORR was 21.3%. The data show that approximately one half of the patients experienced some degree of tumor reduction.14
Contextually, this ORR for monotherapy is modest. It appears to be lower than the activity reported with tarlatamab, which had an ORR of 35% in phase III DeLLphi-304 study, which also demonstrated a significant OS benefit vs chemotherapy. However, cross-trial comparison must be taken with caution and no head-to-head study is available.15 Obrixtamig monotherapy activity is comparable to historical US second-line benchmarks (topotecan 10%-25% and lurbinectedin 35% in platinum-sensitive SCLC).2,16 These findings indicate credible early activity and support the continued investigation of obrixtamig, including its promising activity in combination setting.
DAREON-9: Phase Ib Study of Obrixtamig + Topotecan in Relapsed SCLC
In this open-label phase Ib dose-escalation/confirmation study of obrixtamig plus topotecan in adults with relapsed SCLC after ≥1 prior platinum-based line, the MTD was not reached across 3 dose levels. Dose-limiting toxicities were infrequent and manageable (grade 3 tumor pain during step-up dosing; grade 3 fatigue at target dose; grade 2 creatinine increase). As expected with a topotecan backbone, grade ≥3 hematologic AEs were common (neutropenia: 60%; thrombocytopenia: 52%; anemia: 24%) whereas no grade ≥3 immune-mediated AEs were observed (CRS: 0%; obrixtamig-related neurologic toxicity/ICANS: 0%). These data indicate a tolerable, component-consistent safety profile and support ongoing dose optimization and efficacy evaluation of the combination.14
DAREON-9: Efficacy with Obrixtamig + Topotecan in Relapsed SCLC
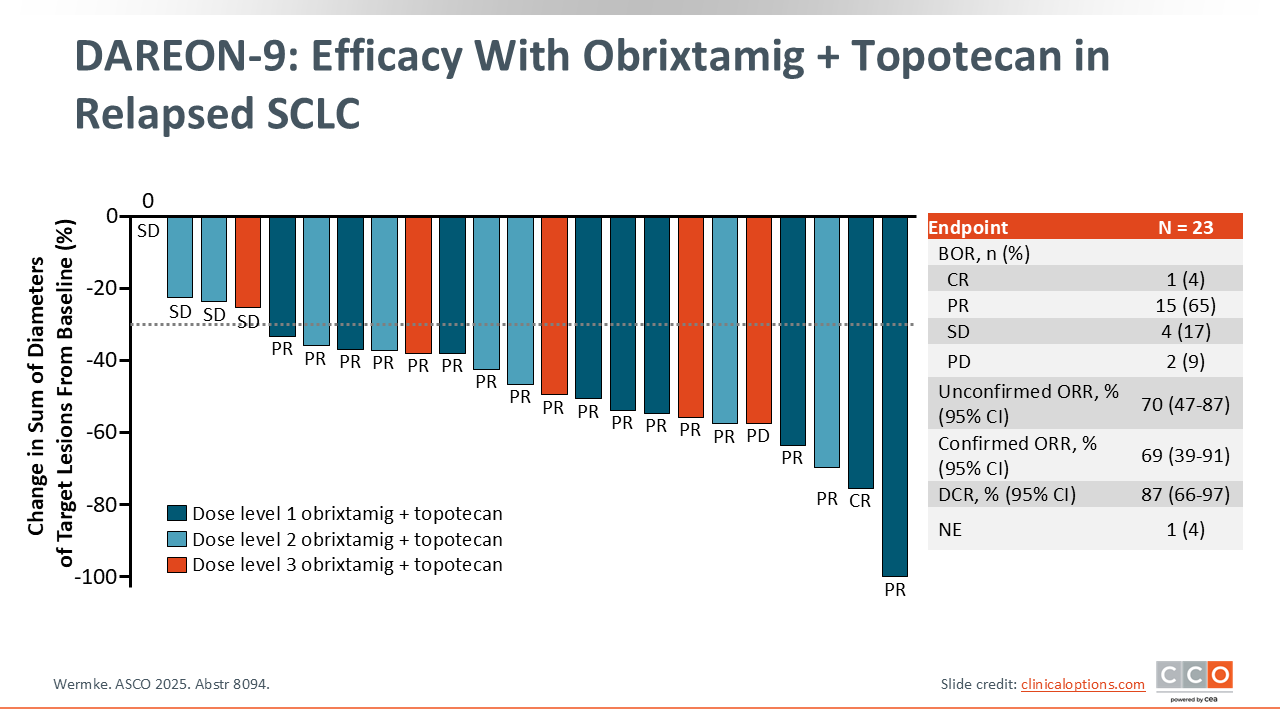
In this open-label phase Ib study of obrixtamig plus topotecan for relapsed SCLC, the efficacy-evaluable population was 23 patients. Confirmed ORR was 69% (95% CI: 39%-91%), including complete response of 4% and partial response of 65%, with stable disease of 17%, PD of 9%, and NE of 4%. Unconfirmed ORR was 70% (95% CI: 47%-87%), and the DCR was 87% (95% CI: 66%-97%). The data show tumor shrinkage in most patients across all 3 dose levels, including several deep responses.14
ADC Mechanism of Action
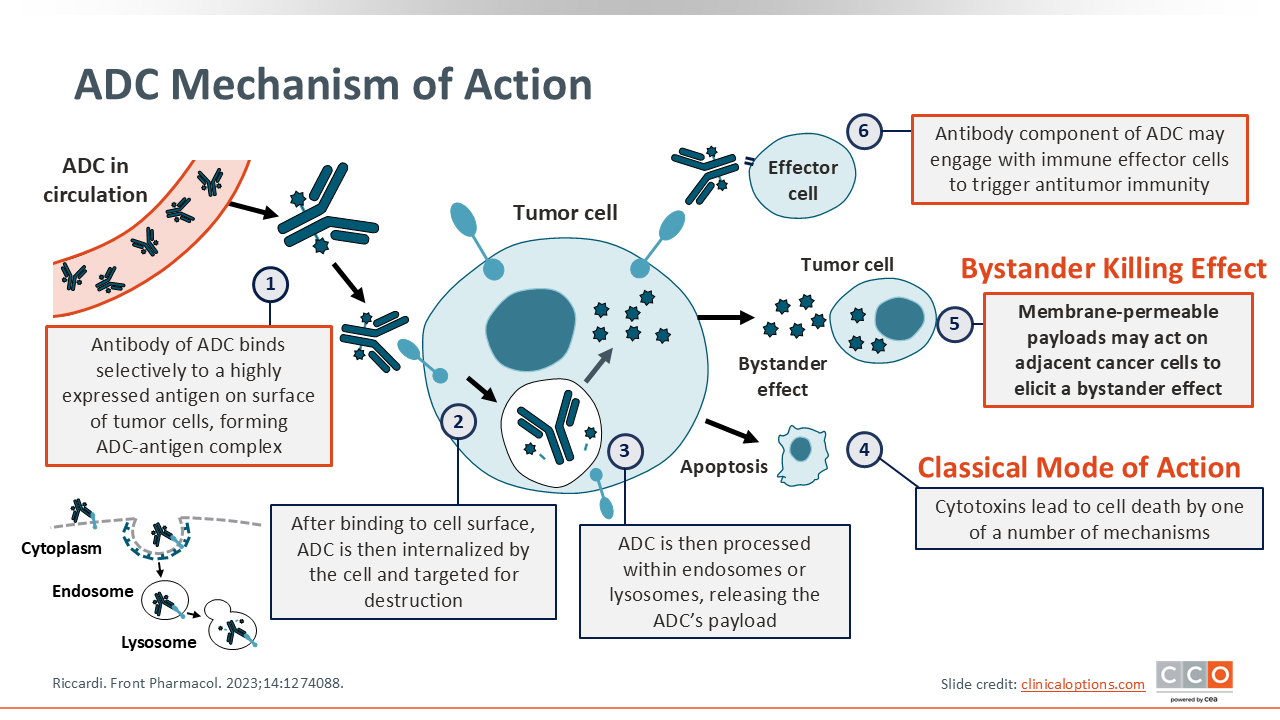
Another promising treatment strategy for SCLC is the use of antibody–drug conjugates (ADCs). ADCs comprise 3 elements: the antibody, linker, and payload. First, the ADC binds to the antigen on the cell surface. Next, the ADC–antigen complex is internalized. Then, the antibody portion undergoes lysosomal degradation. The payload is then released into the cytoplasm, and finally, the ADC interacts with the target (ie, DNA, microtubules, Topo-I). Certain payloads create a “bystander effect,” enabling the destruction of adjacent low-antigen cells.17
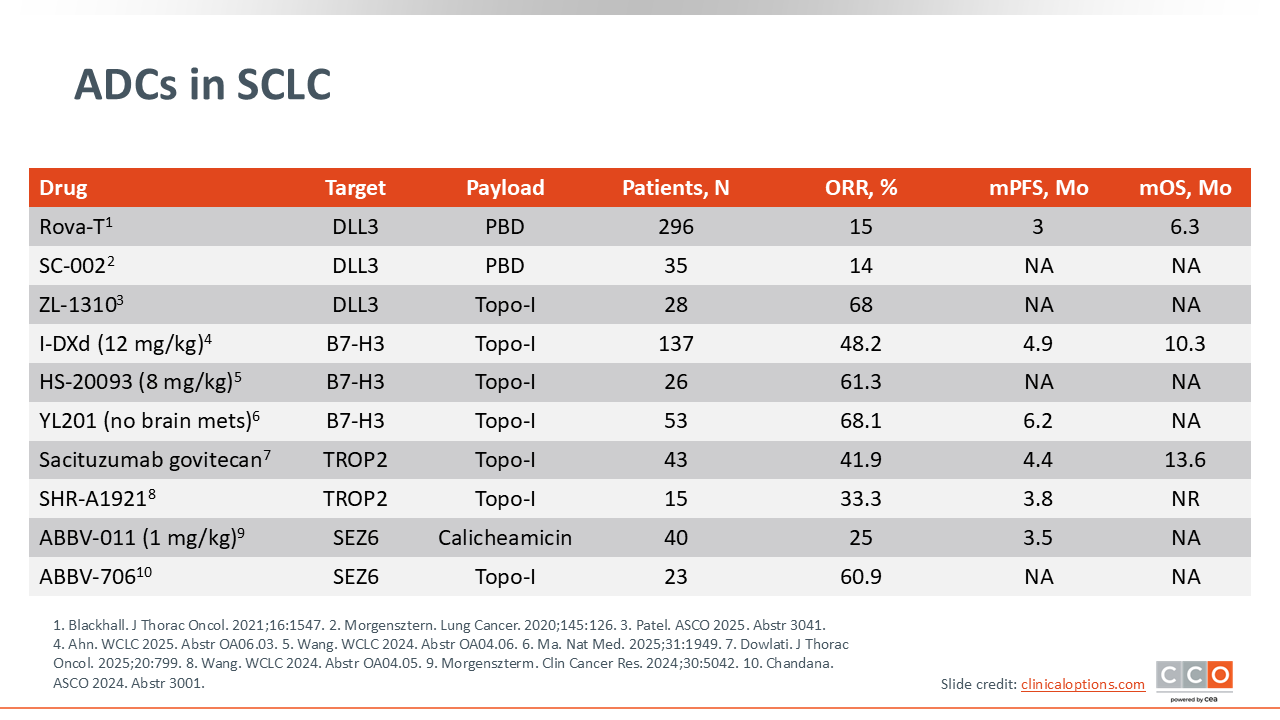
ADCs in SCLC
Several ADCs are also being tested against various targets in SCLC. New ADCs targeting B7-H3, TROP2, and SEZ6 with Topo-I payloads have shown unprecedented response rates for patients with relapsed SCLC. Thus far, the best results with ADCs in SCLC have been reported in agents with a Topo-I payload.18-20
Two prior DLL3-targeting ADCs with a Pyrrolobenzodiazepine payload (Rova-T and SC-002) were not very effective for patients with relapsed SCLC,20 but zocilurtatug pelitecan (ZL-1310), which is an ADC targeting DLL3 with a Topo-I payload, has shown promising results.21 Similarly, results from the first SEZ6-targeting ADC with a calicheamicin payload (ABBV-011) were modest in patients with SCLC, whereas the ADC ABBV-706, which uses a Topo-I payload, showed a markedly higher response rate (60.9% vs 25.0%) in this patient population.19
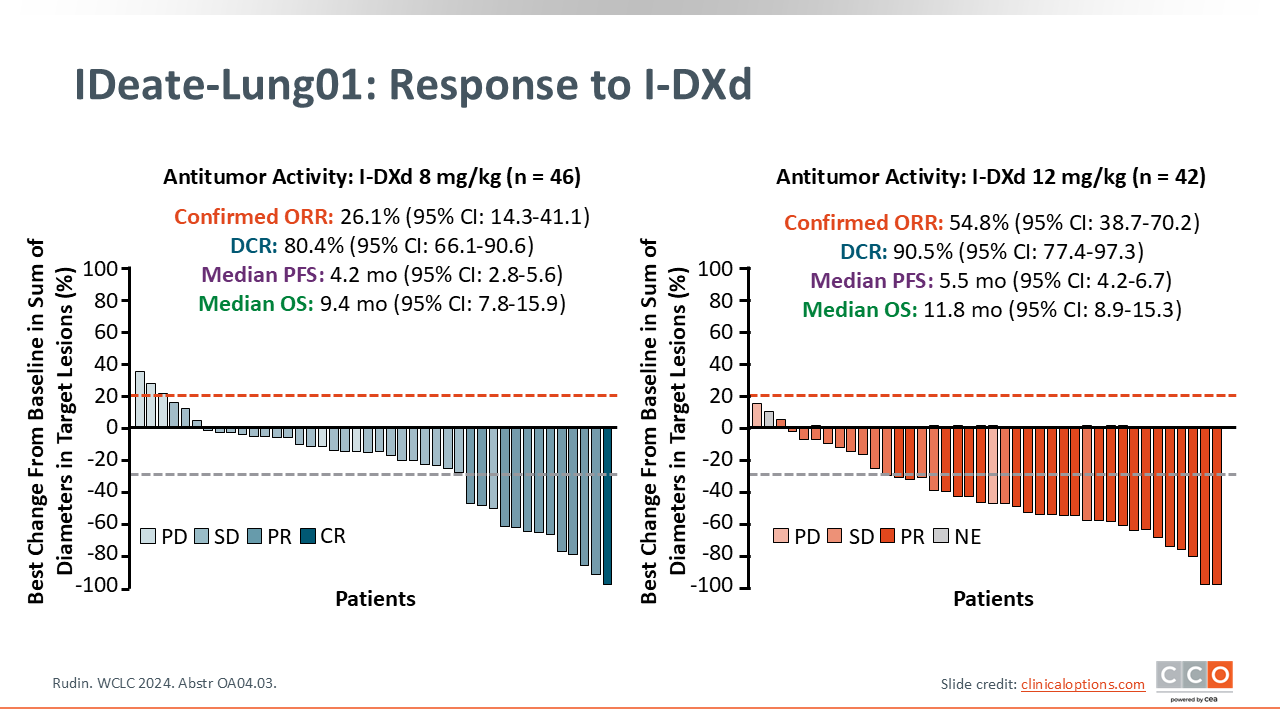
IDeate-Lung01: I-DXd in Patients With Pretreated ES-SCLC
Ifinatamab deruxtecan (I-DXd) is a humanized anti–B7-H3 IgG1 monoclonal antibody with a cleavable linker and a Topo-I payload.22 The randomized phase II IDeate-Lung01 trial evaluated I-DXd in 88 patients with ES-SCLC who had progressed after at least 1 prior platinum-based therapy. Patients were randomized 1:1 to I-DXd 8 mg/kg or 12 mg/kg, both administered every 3 weeks. The primary endpoint was ORR.21,22
IDeate-Lung01: Response to I-DXd
The 12-mg/kg dose of I-DXd resulted in a confirmed ORR of 54.8% (95% CI: 38.7%-70.2%) and the 8-mg/kg dose resulted in an ORR of 26.1% (95% CI: 14.3%-41.1%). The DCR was 90.5% (95% CI: 77.4%-97.3%) at the 12-mg/kg dose and 80.4% (95% CI: 66.1%-90.6%) at the 8-mg/kg dose.22
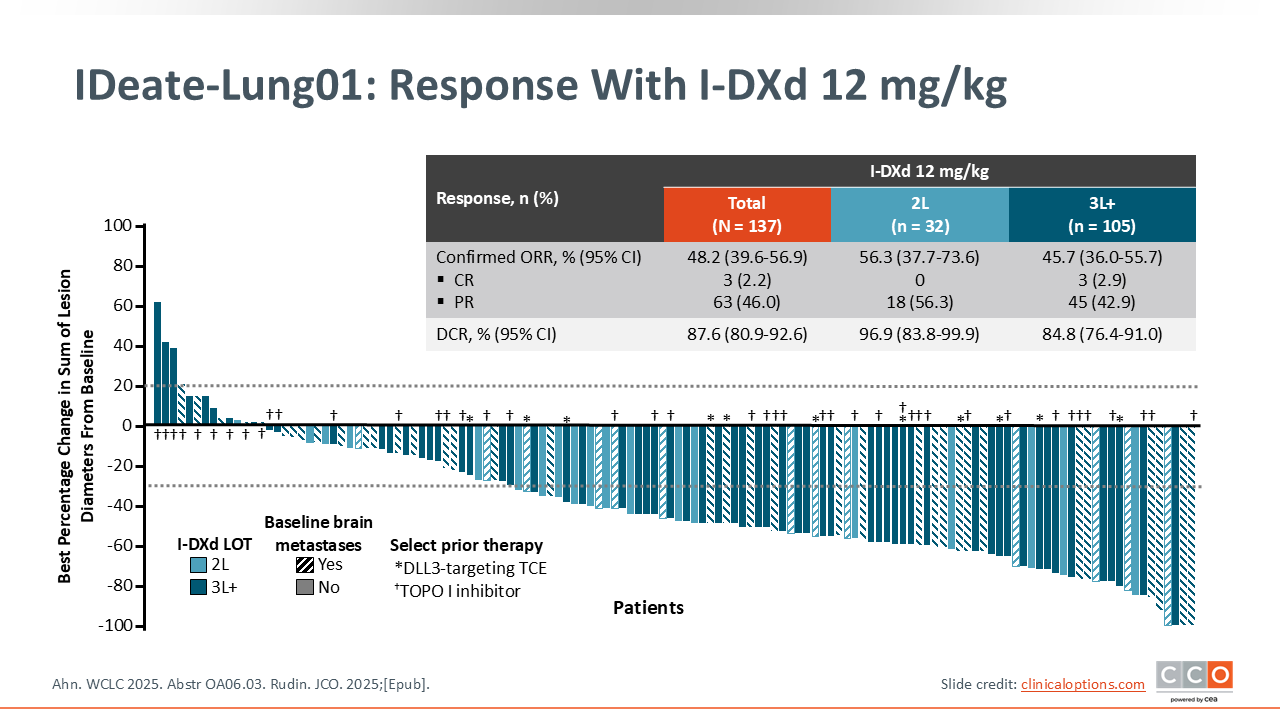
IDeate-Lung01: Response With I-DXd 12 mg/kg
In the dose-extension phase with I-DXd 12 mg/kg, 95 additional patients were enrolled. The confirmed ORR was 48.2% (95% CI: 39.6%-56.9%) in the overall cohort, with a confirmed ORR of 56.3% in the second-line setting, and 45.7% in the third-line and beyond settings.21
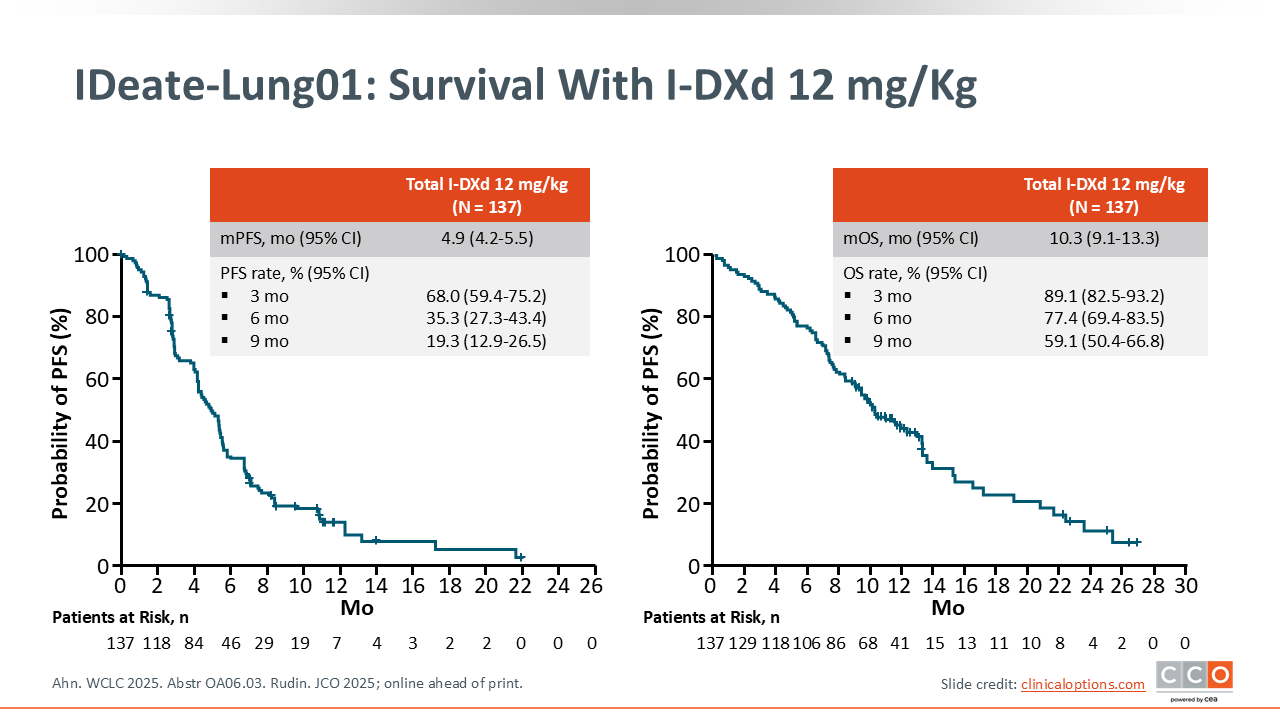
IDeate-Lung01: Survival With I-DXd 12 mg/kg
The median PFS for all patients who received I-DXd 12 mg/kg was 4.9 months (95% CI: 4.2-5.5), and the median OS was 10.3 months (95% CI: 9.1-13.3).21 Based on these data, I-DXd 12 mg/kg every 3 weeks shows promising efficacy in patients with previously treated ES-SCLC.
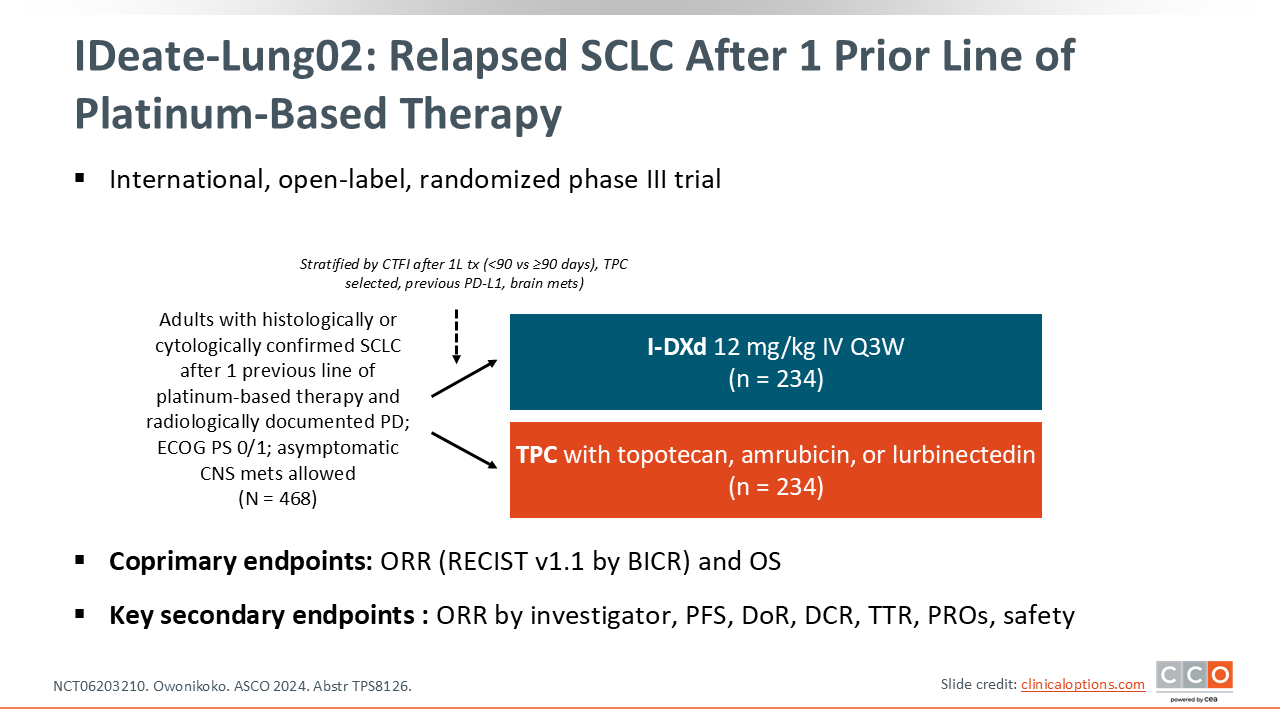
IDeate-Lung02: Relapsed SCLC After 1 Prior Line of Platinum-Based Therapy
The ongoing, confirmatory phase III IDeate-Lung02 study is evaluating I-DXd in patients with relapsed SCLC after 1 prior line of platinum-based therapy.
Patients are being randomized 1:1 to receive either I-DXd 12 mg/kg every 3 weeks or the treating physician’s choice of SoC (topotecan, amrubicin [Japan only], or lurbinectedin). The coprimary endpoints are ORR and OS (NCT06203210).23
ABBV-706 in Patients With R/R SCLC
ABBV-706 is another promising ADC that targets SEZ6 and has a Topo-I payload. SEZ6 is a protein that is highly expressed in SCLC and other neuroendocrine tumors, making it a target for various cancer therapies. A dose-escalation and dose-expansion phase I trial evaluated ABBV-706 in patients with relapsed/refractory SCLC after at least 1 prior platinum-based regimen.
The dose optimization part of the study randomized 80 patients to 2 doses: 1.8 mg/kg or 2.5 mg/kg, both administered every 3 weeks. The primary endpoints were safety and tolerability and to determine the recommended phase II dose (NCT05599984).24
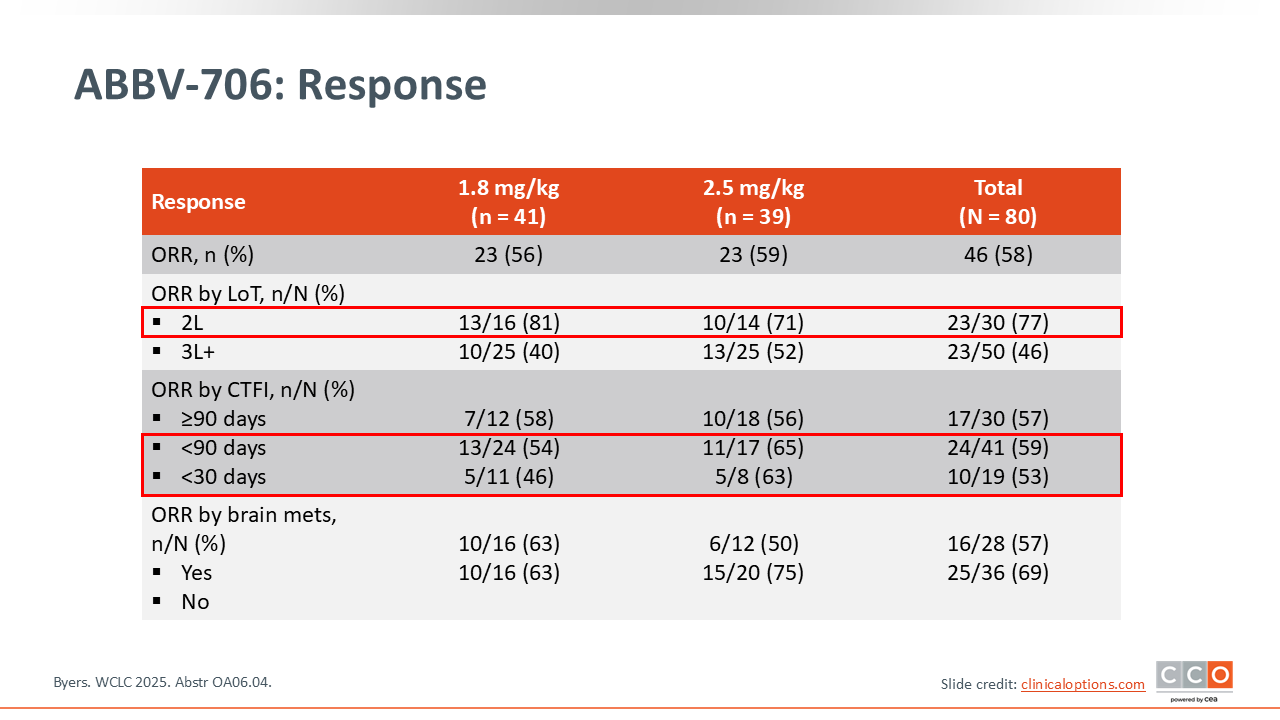
ABBV-706: Response
Among the patients participating in the dose-optimization phase, ABBV-706 demonstrated exceptionally high response rates. The confirmed ORR was 58% (46/80) across both dose cohorts.
Efficacy was particularly high in the second-line setting, with an ORR of 77% (23/30). In the 1.8-mg/kg cohort, the second-line ORR was 81% (13/16) and the ORR was 71% (10/14) with 2.5-mg/kg dose. This activity was seen regardless of platinum sensitivity, with an ORR of 57% (17/30) in platinum-sensitive (CTFI ≥90 days) patients and 59% (24/41) in platinum-resistant (CTFI <90 days) patients. Almost all patients at both dose levels achieved some degree of tumor reduction.24
Select Ongoing Studies With TCEs and ADCs in SCLC
Several ongoing studies are evaluating TCEs and ADCs in SCLC. In the first-line maintenance setting, DeLLphi-305 is evaluating durvalumab with or without tarlatamab. In the first-line induction setting, DeLLphi-312 is adding tarlatamab to standard chemoimmunotherapy. Combination studies in the second-line and beyond settings include DeLLphi-310, which is testing tarlatamab plus the B7-H3 ADC YL201, and DAREON-9, which is testing obrixtamig plus chemotherapy (NCT05990738).14
Conclusions
After decades with few advances, the management of ES-SCLC is rapidly evolving. The first-line setting has been improved with the first successful maintenance strategy, lurbinectedin plus atezolizumab, which has shown a significant OS benefit. Despite these advances, most patients with ES-SCLC relapse and require second-line therapy.
Lurbinectedin remains an important option in the relapsed setting, demonstrating clinically meaningful activity in both platinum-sensitive and platinum-resistant disease, and ongoing investigations—including the LAGOON phase III trial—are assessing its integration into combination strategies to further improve survival. Parallel development of checkpoint inhibitor combinations, such as lurbinectedin with atezolizumab or pembrolizumab, has shown encouraging response rates and survival signals, reinforcing the value of immunotherapy-based combinations in select patients.
DLL3-targeted therapies have emerged as one of the most significant breakthroughs in SCLC. Tarlatamab, the first DLL3-directed TCE to achieve regulatory approval, has demonstrated robust and durable responses in heavily pretreated populations, culminating in an OS benefit over standard chemotherapy in the phase III DeLLphi-304 study. Building on these results, ongoing trials are pushing tarlatamab into earlier lines of therapy and exploring rational combinations with PD-L1 inhibitors and ADCs. Obrixtamig, a next-generation DLL3 x CD3 TCE, has also shown early signs of clinical activity, supporting its continued clinical development both as monotherapy and in combination regimens such as the DAREON-9 topotecan combination study.
ADCs represent another promising pillar of innovation, with several agents delivering promising efficacy in relapsed SCLC. B7-H3–targeted I-DXd and the SEZ6-targeted ADC ABBV-706 have produced high ORRs across both platinum-sensitive and platinum-resistant disease, with sustained benefit and manageable toxicity profiles. With multiple confirmatory phase III trials underway, these agents are poised to change the SoC. The next steps will be to combine these novel TCEs and ADCs and test them in the first-line setting, either during induction or maintenance, to further improve outcomes.