CE / CME
Breaking Barriers in Obesity Care: Applying Emerging Evidence to Primary Care Practice
Physician Assistants/Physician Associates: 1.00 AAPA Category 1 CME credit
ABIM MOC: maximum of 1.00 Medical Knowledge MOC point
Physicians: maximum of 1.00 AMA PRA Category 1 Credit™
Nurse Practitioners/Nurses: 1.00 Nursing contact hour
Released: December 10, 2025
Expiration: December 09, 2026
Activity
OASIS 4: Oral Semaglutide 25 mg in Patients With Overweight or Obesity
Let us now shift to new data on emerging therapies for AOM.
Although most people are familiar with SC weekly semaglutide injections to treat obesity, as of December 2025 oral semaglutide for obesity is still under regulatory review with the FDA. To this end, 1 of the studies that will help inform the FDA’s review is the OASIS 4 study of oral semaglutide for treatment of obesity in adults. This study evaluated body weight changes and quality of life scores with 25 mg of oral semaglutide over 64 weeks (NCT05564117).19
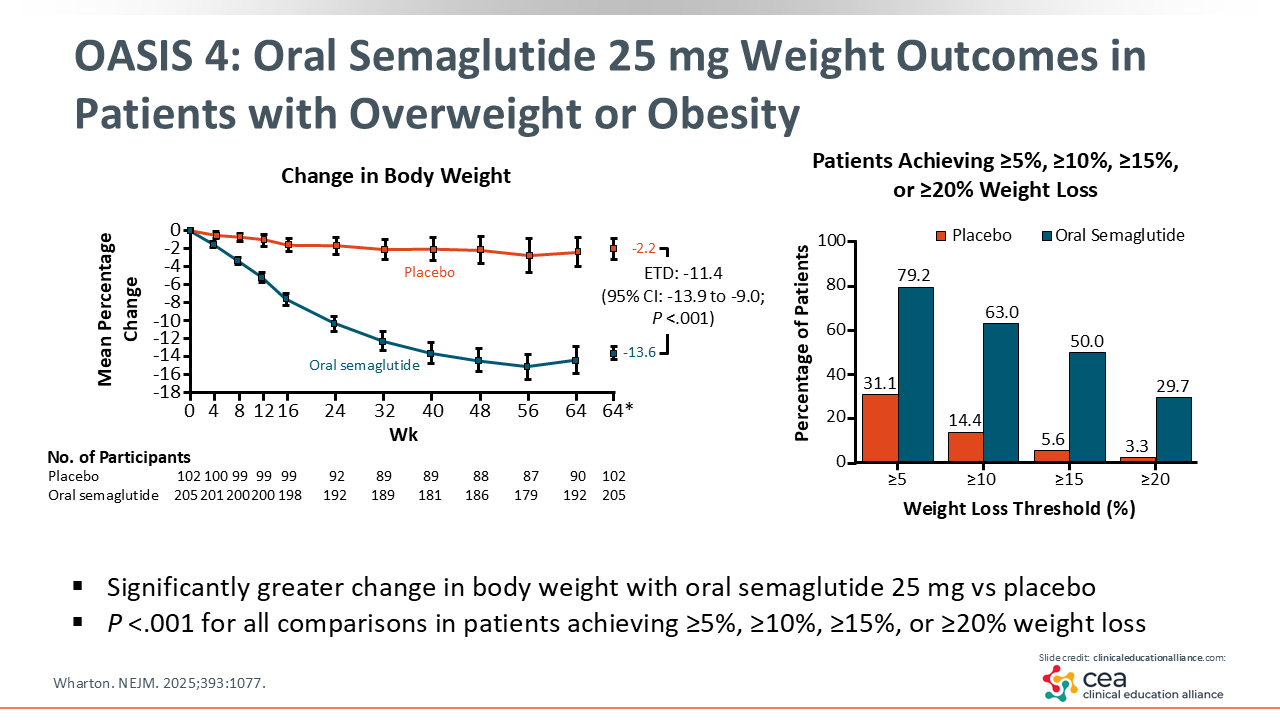
OASIS 4: Oral Semaglutide 25 mg Weight Outcomes in Patients With Overweight or Obesity
At the end of the 64-week follow-up, the intervention group had a mean -13.6% change in body weight compared to the placebo group, which only showed a mean 2.2% weight loss.19
Breaking down the data by weight percent targets showed that approximately 80% of people receiving oral semaglutide reached clinically significant weight loss of ≥5%, and approximately 30% lost ≥20% of their total body weight.19
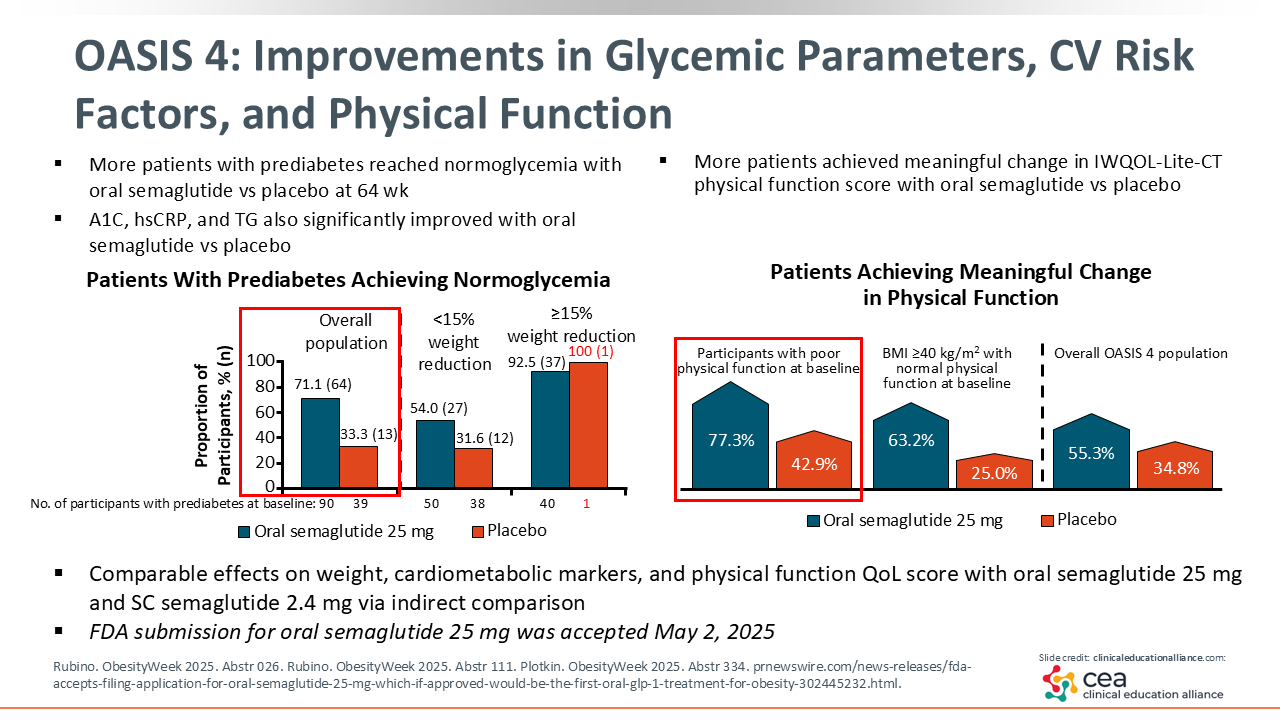
OASIS 4: Improvements in Glycemic Parameters, CV Risk Factors, and Physical Function
In addition to weight changes, oral semaglutide was also associated with greater improvements in quality-of-life scores, glycemic parameters, CV risk factors, and physical function. In particular, of participants receiving oral semaglutide, 71.1% achieved normoglycemia and 77.3% of those with poor physical function at baseline achieved meaningful improvement in physical function, compared to 33.3% and 42.9%, respectively, in the placebo group.20-23
ATTAIN-1: Oral Small-Molecule GLP-1 RA Orforglipron in Patients With Overweight or Obesity
Also under investigation for oral treatment of obesity is orforglipron, a novel small molecule, nonpeptide GLP-1 receptor agonist. This agent is currently being studied for weight loss in the ATTAIN-1 trial among participants with obesity and no diabetes. This trial compared 3 doses of orforglipron—6 mg, 12 mg, and 36 mg—to placebo (NCT05869903).24
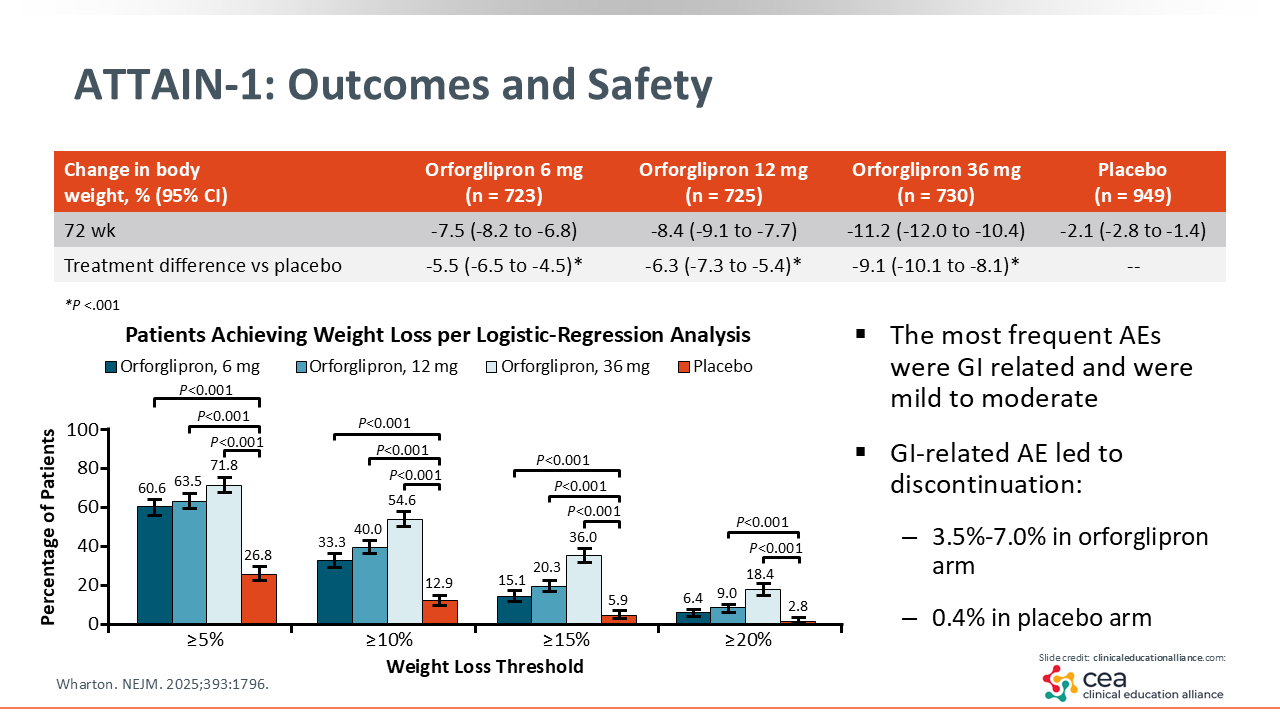
ATTAIN-1: Outcomes and Safety
Although all doses of orforglipron were effective for achieving weight loss, a key observation was that the proportion of weight lost scaled with the dose of orforglipron; more weight loss was observed with higher doses. With the 6-mg dose, the mean change in body weight was -7.5%, with the 12-mg dose it was -8.4%, and with the 36-mg dose, it was -11.2%.24
Orforglipron was also well tolerated. Like other GLP-1 receptor agonist-containing medications, gastrointestinal side effects or AEs were the most common.24
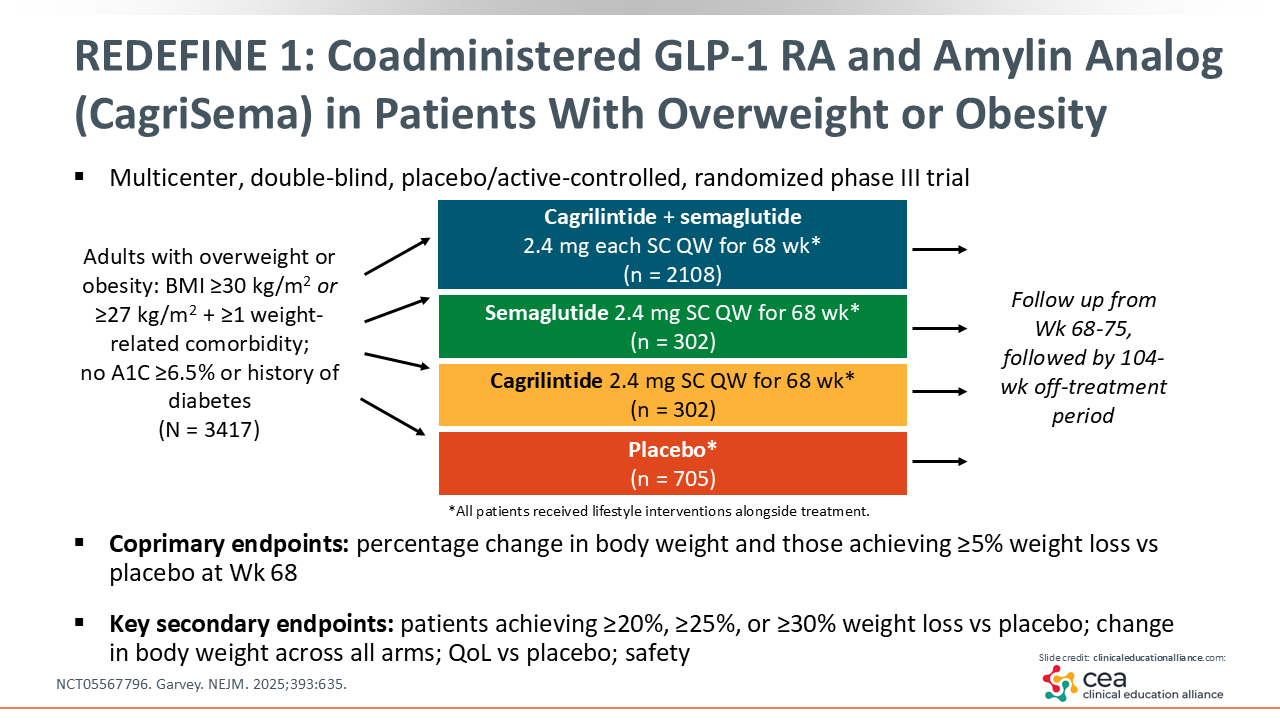
REDEFINE 1: Coadministered GLP-1 RA and Amylin Analog (CagriSema) in Patients With Overweight or Obesity
Next, the REDEFINE 1 trial examined cagrilintide 2.4 mg SC both coadministered with semaglutide 2.4 mg SC (CagriSema) and as a monotherapy for adults with obesity. Cagrilintide is a long-acting amylin analogue thought to act on amylin and calcitonin receptors in the brain to affect appetite regulation (NCT05567796).25
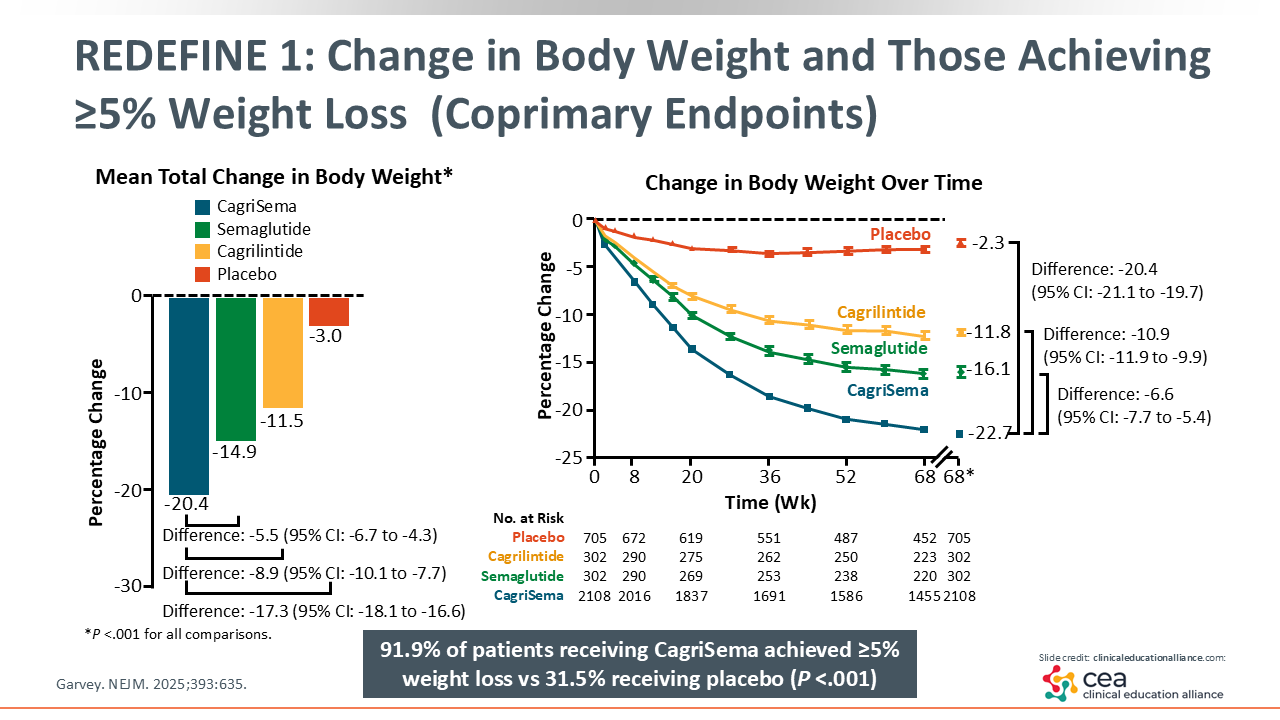
REDEFINE 1: Change in Body Weight and Those Achieving ≥5% Weight Loss (Coprimary Endpoints)
At the end of the 68-week follow-up, the mean total change in body weight with CagriSema was -20.4%. Of note, this is among the highest amounts of weight loss reported with AOMs so far.25
Another key aspect of this study is that 91.9% of participants reached the threshold of clinically significant weight loss (≥ 5%) with CagriSema, compared to 31.5% with placebo. That is an exceptionally high response rate. Furthermore, the CagriSema combination was associated with greater weight loss than semaglutide by itself, which in turn elicited greater weight loss than cagrilintide by itself.25
REDEFINE 1: Additional Outcomes
Aside from weight loss, additional outcomes of the REDEFINE trial included other metabolic health parameters, waist circumference, systolic blood pressure, lipid profile, and physical function scores, all of which were improved with CagriSema vs placebo.25
Overall, CagriSema was well tolerated, with AEs similar to what is seen with other GLP-1 medications. Gastrointestinal side effects were most common, and discontinuation due to AEs was low, just under 6%.25
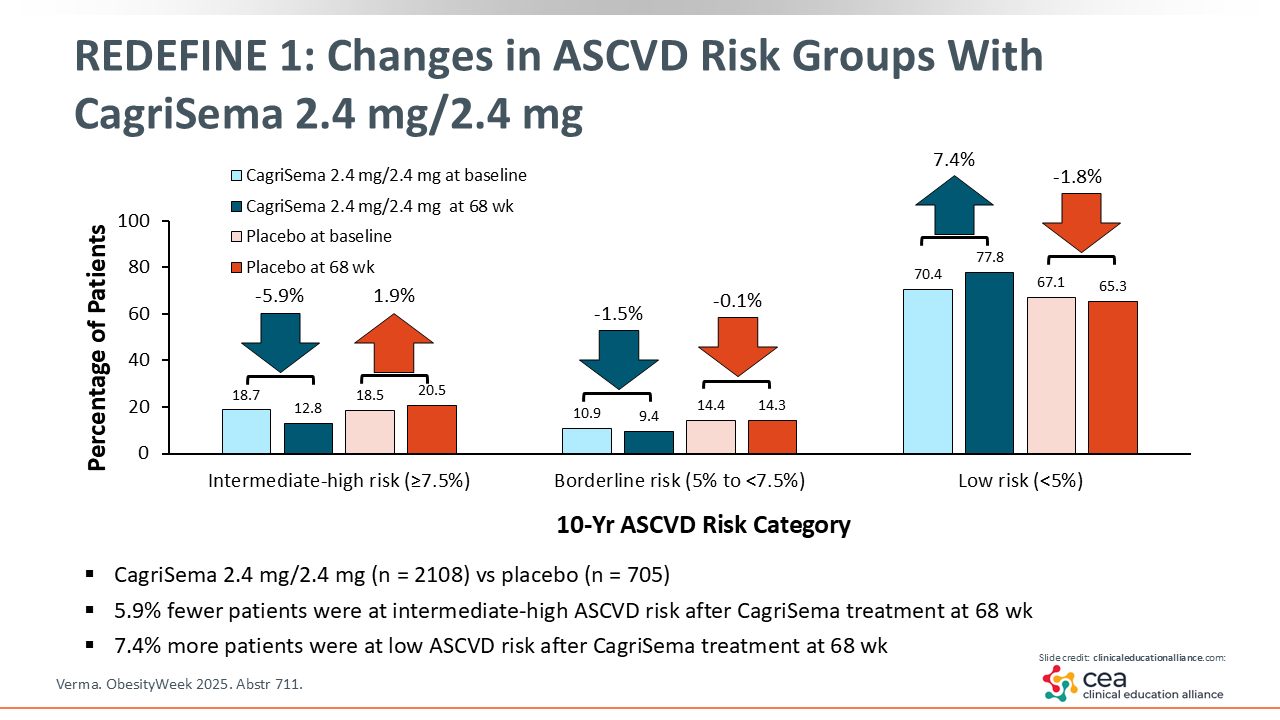
REDEFINE 1: Changes in ASCVD Risk Groups With CagriSema 2.4 mg/2.4 mg
An additional outcome of particular importance is ASCVD risk. Among patients receiving CagriSema, the proportion with intermediate or high risk for ASCVD decreased by 5.9%, and the proportion with low risk increased by 7.4%. That is, patients receiving CagriSema transitioned from higher ASCVD risk groups to lower risk groups. This effect was not observed with placebo and suggests that improvements in metabolic parameters and weight with CagriSema are accompanied by lower risk for CVD.26
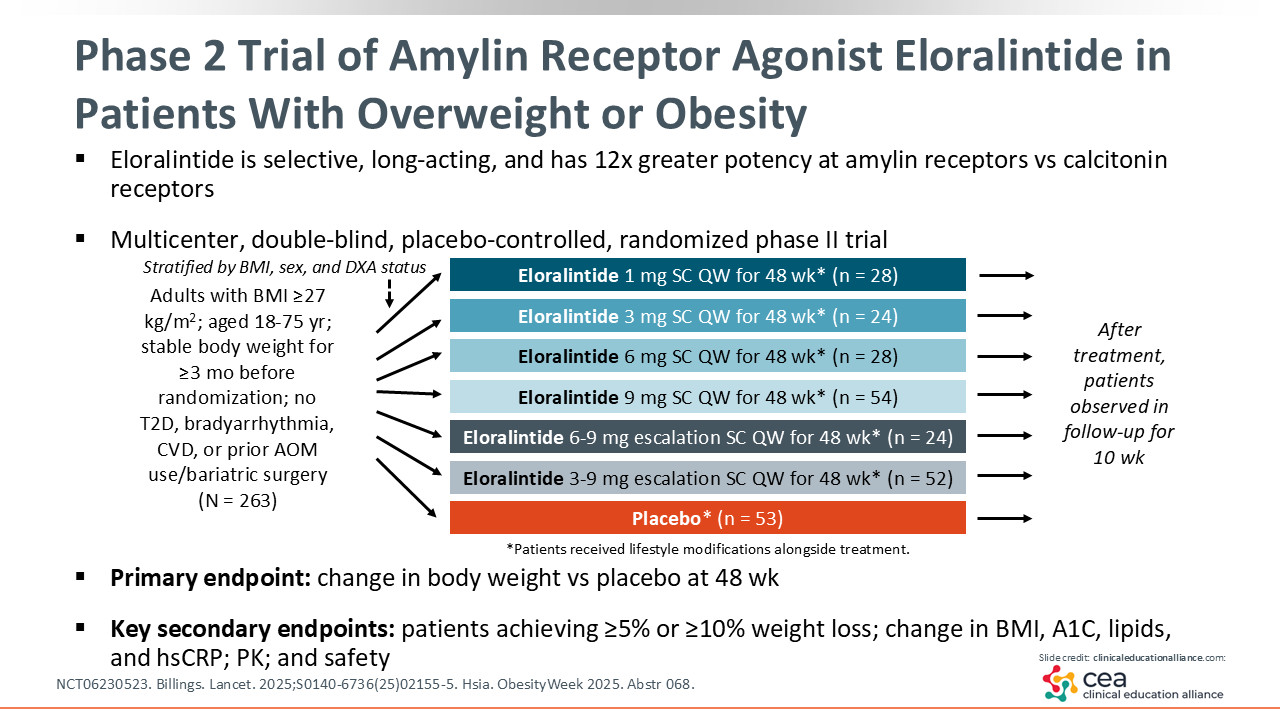
Phase II Trial of Amylin Receptor Agonist Eloralintide in Patients With Overweight or Obesity
Next, eloralintide is an exciting agent in earlier stages of development. This novel drug is a selective, long-acting amylin receptor agonist with much greater potency at the amylin receptors vs calcitonin receptors. A phase II trial of eloralintide evaluated the safety and efficacy of several different doses: 1 mg, 3 mg, 6 mg, and 9 mg, with dose escalation groups for 6 mg to 9 mg and 3 mg to 9 mg (NCT06230523).27,28
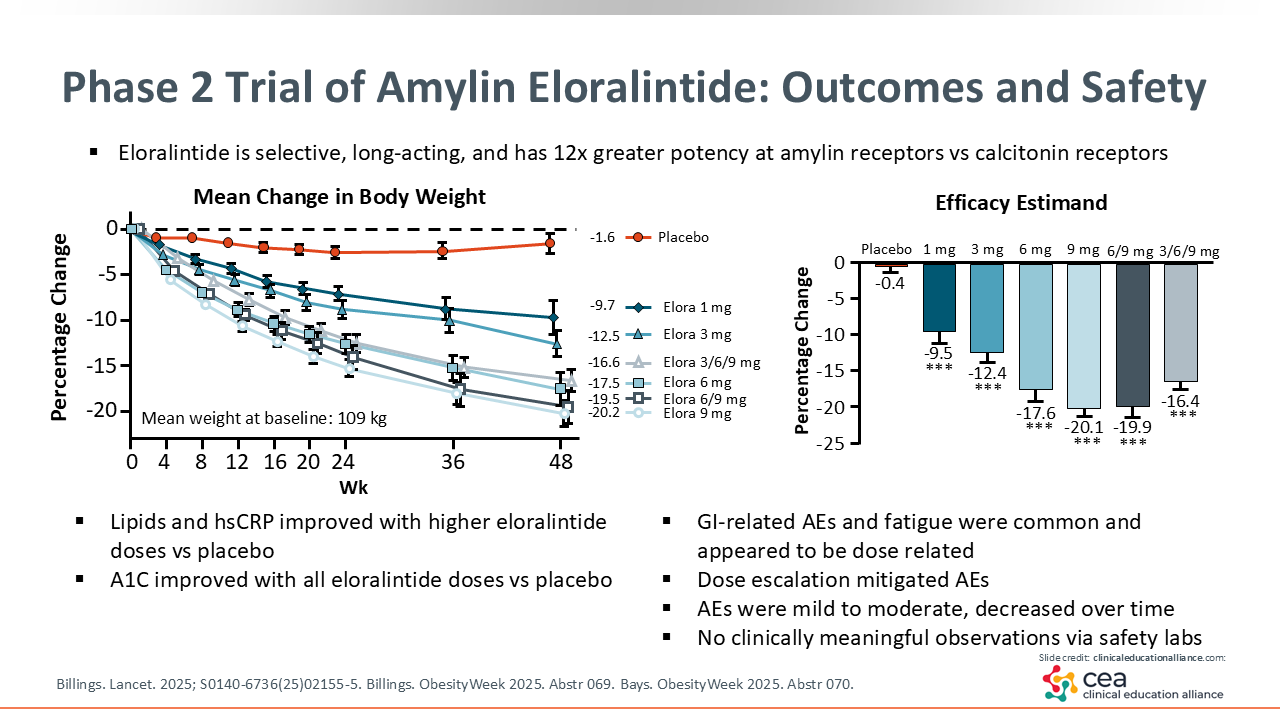
Phase II Trial of Amylin Eloralintide: Outcomes and Safety
Greater weight loss was seen with higher doses of eloralintide. The group that initiated treatment with the highest dose, 9 mg, exhibited the greatest mean weight loss: 20.2%. AEs were mild to moderate, and were mostly gastrointestinal, with the dose-escalation groups tending to have fewer AEs. Aside from weight loss, there were also improvements in lipid profile and high-sensitivity C-reactive protein levels at higher doses of eloralintide; A1C improved at all of the doses in the intervention groups.27,29,30
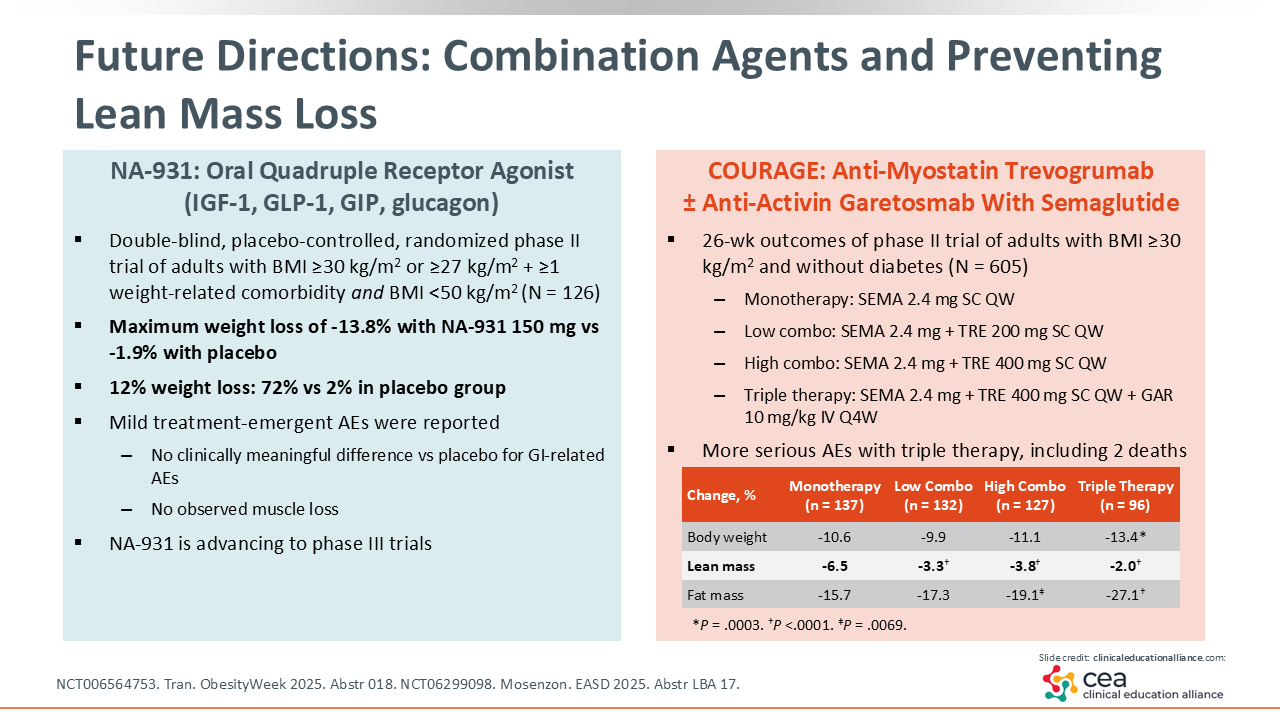
Future Directions: Combination Agents and Preventing Lean Mass Loss
Finally, I think that the future of obesity treatments will focus on combination agents and not just losing weight but losing excess adipose tissue while maintaining lean mass. To this end, there are 2 novel agents that appear to be especially promising.
First, NA-931 is a first-in-class, oral, small molecule receptor agonist of GLP-1, GIP, glucagon and IGF-1. In a 13-week phase II trial for patients with obesity, NA-931 elicited a maximum weight reduction of 13.8%, compared to 1.9% with placebo. AEs were mild and similar between the placebo and intervention groups. Of note, there was no muscle loss observed with NA-931. This is of particular interest, as a major concern with patients undergoing a significant amount of weight loss is how much of it is lean muscle mass vs adipose tissue. The fact that no muscle loss was observed with this agent indicates that the vast majority of weight lost was excess adipose tissue (NCT006564753).31
Trevogrumab is another novel agent, an antimyostatin, that was recently studied in a phase II trial with semaglutide and with or without garetosmab, an antiactivin,. Trevogrumab was evaluated with 2.4 mg semaglutide at 2 different doses: 200 mg or 400 mg SC each week. The triple-therapy combination consisted of 2.4 mg semaglutide and 400 mg trevogrumab weekly, and garetosmab 10 mg/kg IV monthly. More weight loss was observed with the higher dose of trevogrumab, and even higher weight loss was seen with the triple therapy. High proportions of fat mass loss were observed with minimal lean mass loss across all trevogrumab groups. However, there were some very significant AEs with the triple therapy, including 2 deaths. In all, these results are promising but definitely require more study before this combination is ready to be adopted in practice (NCT06299098).32
Key Points for Individualizing Obesity Care: Recent Evidence on Emerging Therapy
Ultimately, emerging therapies hold much promise for continued individualization of obesity care.
For one, oral therapies are advancing in development. Oral therapies for obesity have been available for decades but historically did not compare to the potency of the newer injectable medications. However, oral semaglutide, which is awaiting FDA approval for weight loss as of December 2025, may change that with potency similar to injectable semaglutide.
Orforglipron, a nonpeptide GLP-1 receptor agonist, is another promising oral agent.
Amylin analogs are also emerging as potentially viable and potent treatment options. Cagrisema— semaglutide combined with the amylin analog cagrilintide—was associated with significant weight loss and decreased risk for ASCVD. Eloralintide is a promising long-acting agent.
Finally, novel agents and combinations are in development to address the issue of preservation of lean muscle mass.