CE / CME
The Latest in Obesity Management: Current Evidence for Primary Care
Physician Assistants/Physician Associates: 1.00 AAPA Category 1 CME credit
ABIM MOC: maximum of 1.00 Medical Knowledge MOC point
Nurse Practitioners/Nurses: 1.00 Nursing contact hour
Physicians: maximum of 1.00 AMA PRA Category 1 Credit™
Released: September 25, 2025
Expiration: September 24, 2026
Activity
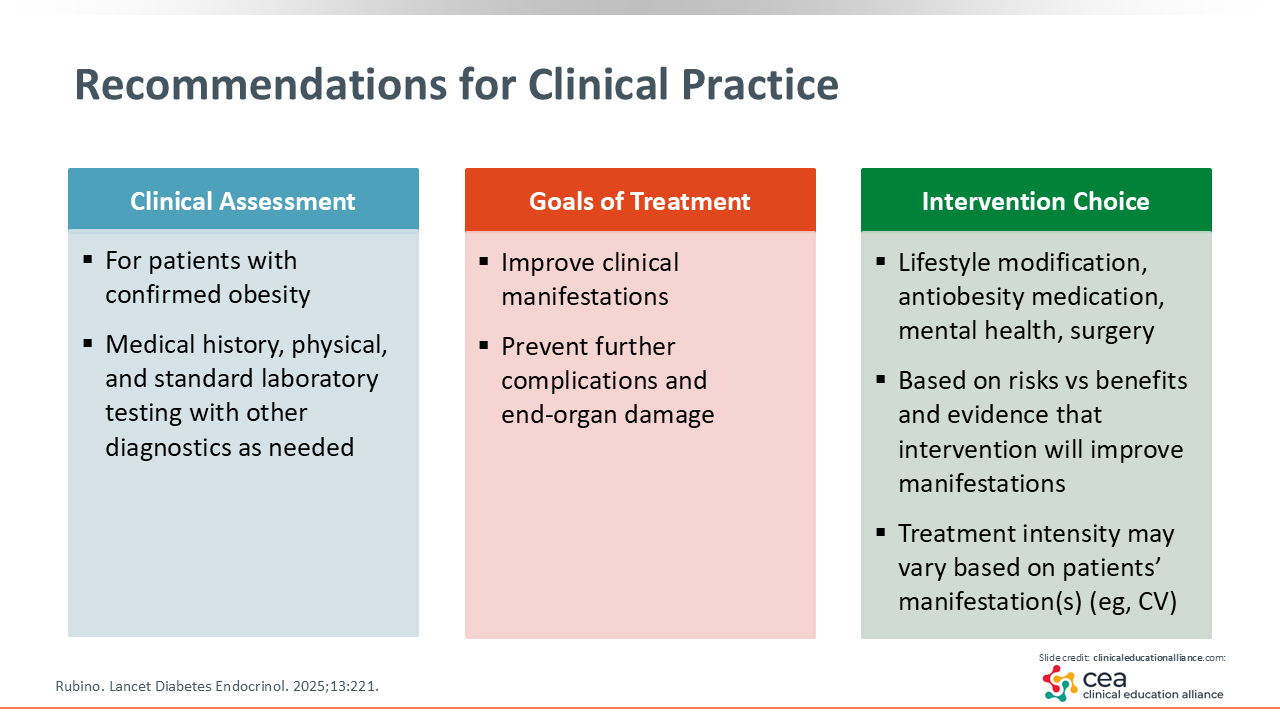
Recommendations for Clinical Practice
For all patients with preclinical or clinical obesity, HCPs should do structured and thorough therapeutic lifestyle recommendation counseling. For HCPs who have the skill set to do this, they should ensure that they set aside sufficient time and space to work with their patients to develop an actionable plan that is realistic. For HCPs who do not have this skill set, they should use a team-based approach so they can maximize their patients’ ability to be successful in making lifestyle changes. Too often, patients receive an empty prescription of “eat less and do more.” That direction is neither actionable nor reproducible. Know where patients are and what they are capable of when developing a treatment plan. This will give them a chance to have therapeutic lifestyle modification as a major part of their obesity-related treatment program.
It also is important to recognize where patients are on the continuum of obesity. There are lifestyle, pharmacologic, psychologic, and surgical approaches to obesity that are all effective.3 Hopefully, every patient will receive an intervention combination that is right for that individual. HCPs should ensure that patients are receiving the tools and treatment options that they feel good about and can implement.
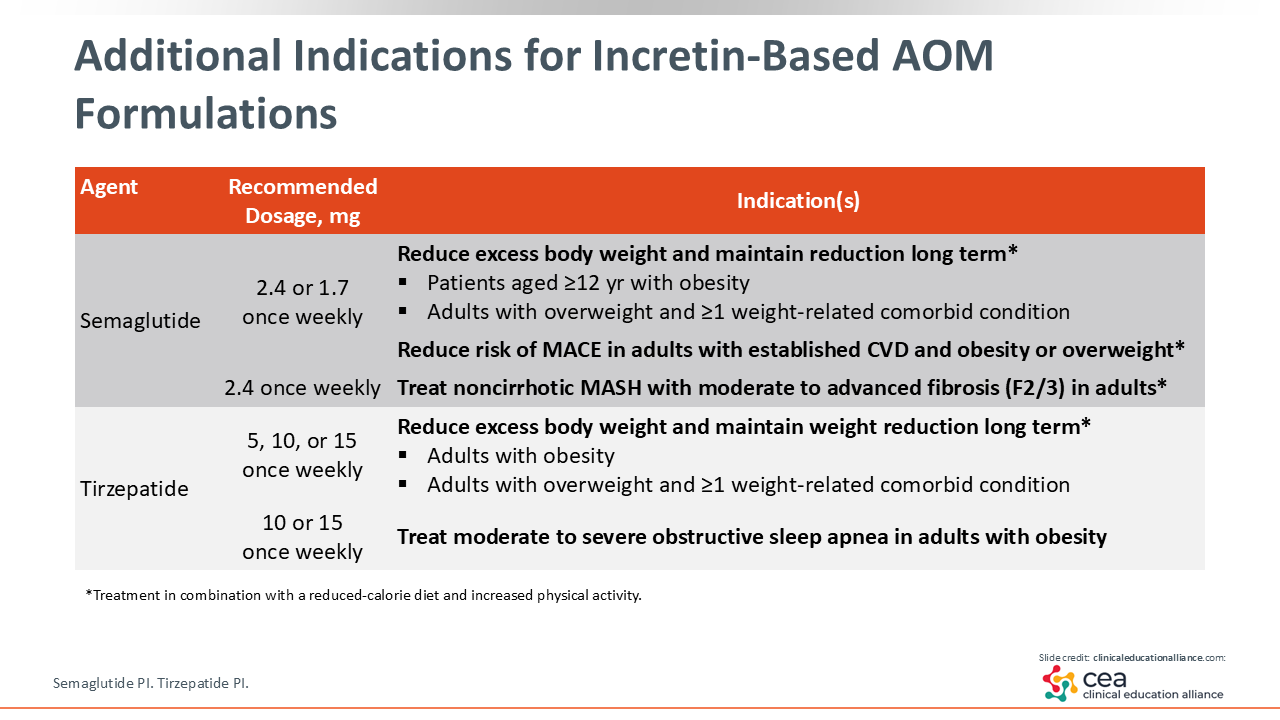
Additional Indications for Incretin-Based AOM Formulations
In the past, multiple weight loss medications were available, but they did not necessarily improve obesity-related complications and/or conditions and did not have indications to do so. However, the emergence of incretin-based AOMs with demonstrated benefits for additional obesity-associated conditions provides the opportunity to improve excess adiposity and other obesity-related health outcomes. For example, formulations of both semaglutide and tirzepatide that are approved to reduce excess body weight are also approved for additional indications.
Semaglutide is a GLP-1 receptor agonist. The formulation approved for obesity management is dosed at either 1.7 or 2.4 mg once weekly and is specifically indicated to reduce excess body weight and maintain long-term weight loss in patients aged 12 years or older with obesity or adults with overweight and ≥1 weight-related condition. The same formulation, at either 1.7 or 2.4 mg once weekly, is also indicated to reduce the risk of MACE in adults with overweight or obesity who have established CVD, and it is approved at a recommended dose of 2.4 mg once weekly to treat MASH in adults with moderate to advanced liver fibrosis. Therefore, the formulation of semaglutide approved for weight reduction is not only an AOM but also a CV risk reduction medication and a MASH treatment for certain patients.11
Tirzepatide is a dual GIP/GLP-1 receptor agonist. The recommended dosage of the formulation approved for obesity management 5, 10, or 15 mg once weekly, and the specific indication is for reduction of excess body weight and long-term maintenance of weight loss in adults with obesity or overweight and ≥1 weight-related comorbidity. It also is indicated at recommended doses of 10 mg or 15 mg once weekly to treat moderate to severe OSA.12 Again, there is a benefit with tirzepatide that goes beyond weight loss.
Both semaglutide and tirzepatide are approved in combination with reduced calorie diet and increased physical activity for the indications described in the previous paragraphs.11,12 Of note, different formulations of both semaglutide and tirzepatide are approved to improve glycemic control, in combination with diet and exercise, in people with T2D. For a full list of approved indications for each formulation, please refer to the respective prescribing information.13,14
Children and adolescents with obesity may also benefit from AOMs. Evidence has demonstrated that those with obesity as a child, even those 6 years of age, are much more likely to have obesity as an adult.15 Furthermore, most adolescents become physiologically more insulin resistant during their growth period, potentially contributing to metabolic health problems; obesity can exacerbate this situation, but weight loss may help.16
Therefore, the availability of AOMs, including semaglutide, that are approved for use for patients as young as12 years of age has the potential to improve obesity management and health outcomes for many children.17
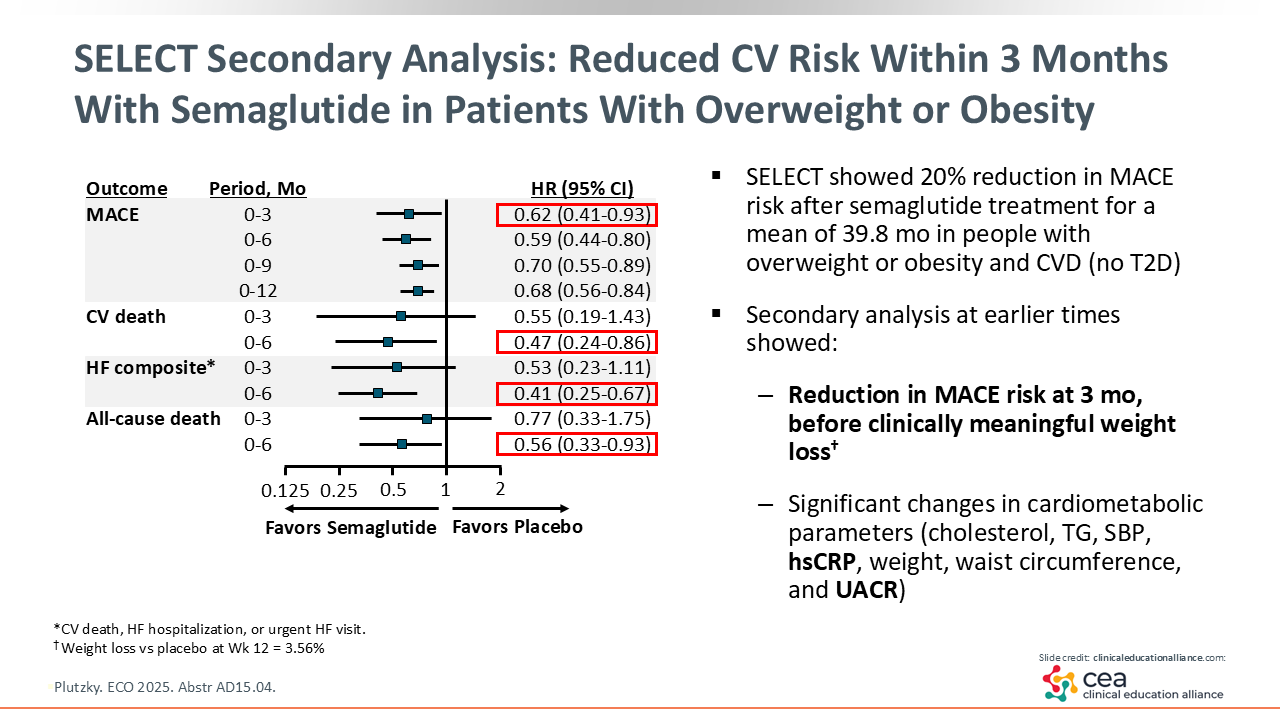
SELECT Secondary Analysis: Reduced CV Risk Within 3 Months With Semaglutide in Patients With Overweight or Obesity
As obesity management moves beyond just weight-centric care, it has been exciting to see the effects of AOMs on CV outcomes in patients with overweight or obesity. The SELECT trial showed a 20% reduction in MACE after a mean 39.8 months of treatment with semaglutide vs placebo in patients with overweight or obesity and established CVD and without T2D. Results of the SELECT trial led to a semaglutide indication for reducing the risk of MACE in this population.
More recently, a secondary analysis of the SELECT trial data revealed very early CV outcome benefit in patients who received semaglutide. In fact, there was a significant reduction in MACE risk as early as 3 months, before mean weight loss reached a clinically significant level of 5%. By 6 months, reductions were observed in risk of heart failure composite outcome (CV death, hospitalization for heart failure, or urgent heart failure visit), all-cause death, and CV death. In addition, early improvements were observed in biomarkers, such as cholesterol, systolic blood pressure, and inflammatory markers like high-sensitivity C-reactive protein and urine albumin–to-creatinine ratio (UACR). These observations indicate that mechanisms other than weight loss contribute to semaglutide benefits on CV outcomes.18
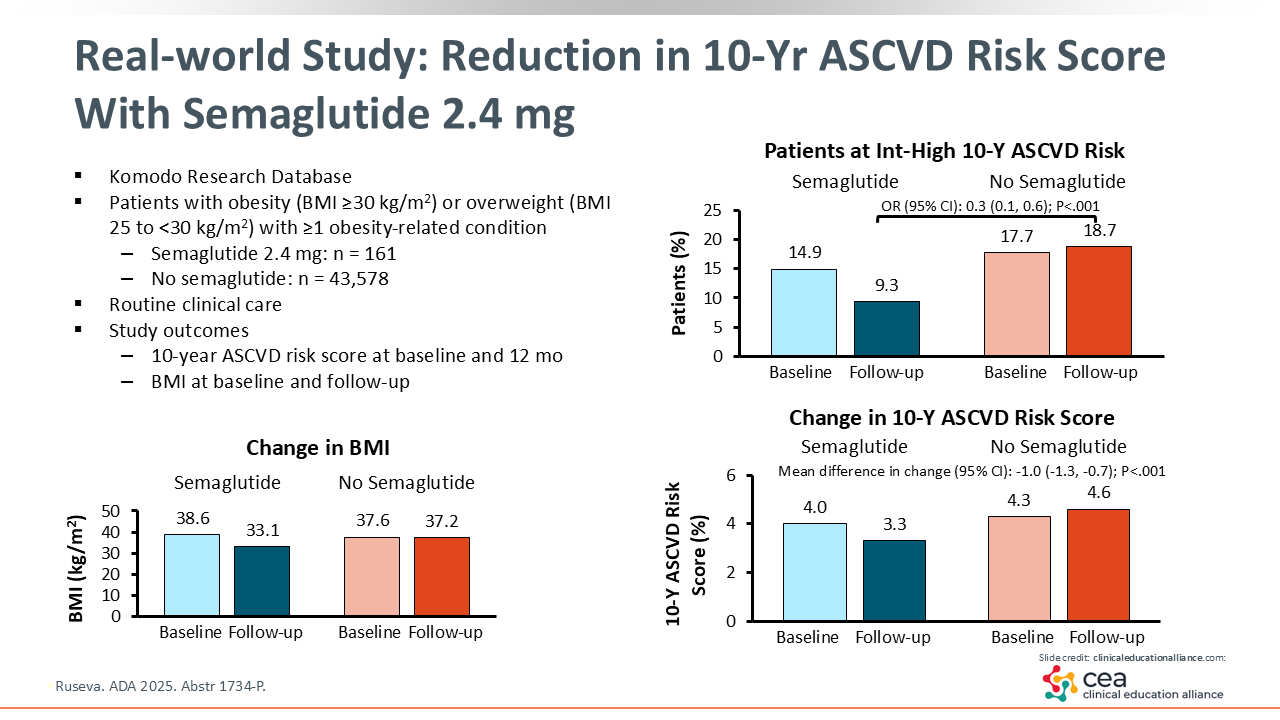
Real-world Study: Reduction in 10-Year ASCVD Risk Score With Semaglutide 2.4 mg
Clinical trials provide critical information for guiding medication use, but it is also valuable to confirm the results in real-world practice, where the patient population and treatment approaches are more diverse. Retrospective database reviews are one source of real-world practice data.
The effect of 1 year of semaglutide 2.4 mg on CV risk was investigated in a real-world database study of people with BMI ≥25 kg/m2. Average BMI decreased in patients receiving semaglutide and remained essentially unchanged in patients who did not receive semaglutide. In addition, the mean 10-year ASCVD risk score and proportion of people at intermediate to high 10-year ASCVD risk were significantly reduced in people receiving semaglutide vs those not receiving semaglutide.19 Results like these can help HCPs as they develop treatment plans that not only help with weight loss but can also reduce morbidity and mortality risk and help people live longer.
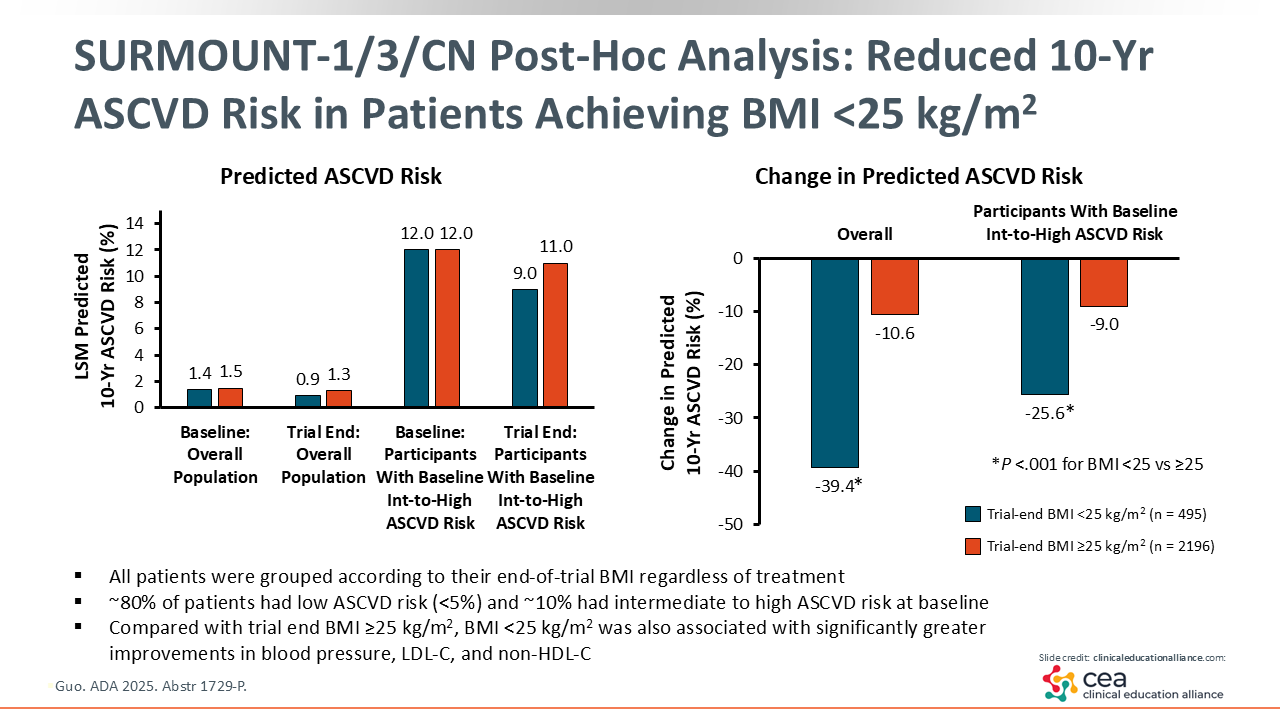
SURMOUNT-1/3/CN Post Hoc Analysis: Reduced 10-Year ASCVD Risk in Patients Achieving BMI <25 kg/m2
A CV outcomes trial of tirzepatide in patients with obesity or overweight and without T2D has not been completed, and tirzepatide does not yet have an indication for reduction of CV risk at this time. However, a post hoc analysis of phase III SURMOUNT trials investigated the relationship between achieved BMI, regardless of treatment, and ASCVD risk in patients with elevated BMI and no T2D at baseline. In particular, the analysis examined the benefit of achieving BMI <25 kg/m2. The analysis showed greater improvement in 10-year ASCVD risk score in patients who achieved BMI <25 kg/m2 vs those who achieved BMI ≥25 kg/m2.20
Of note, evidence suggests that achieving weight loss to reach a BMI in the normal range (<25 kg/m2) may not be unusual, as many patients with obesity and without T2D achieved ≥20% weight loss in tirzepatide clinical trials.21
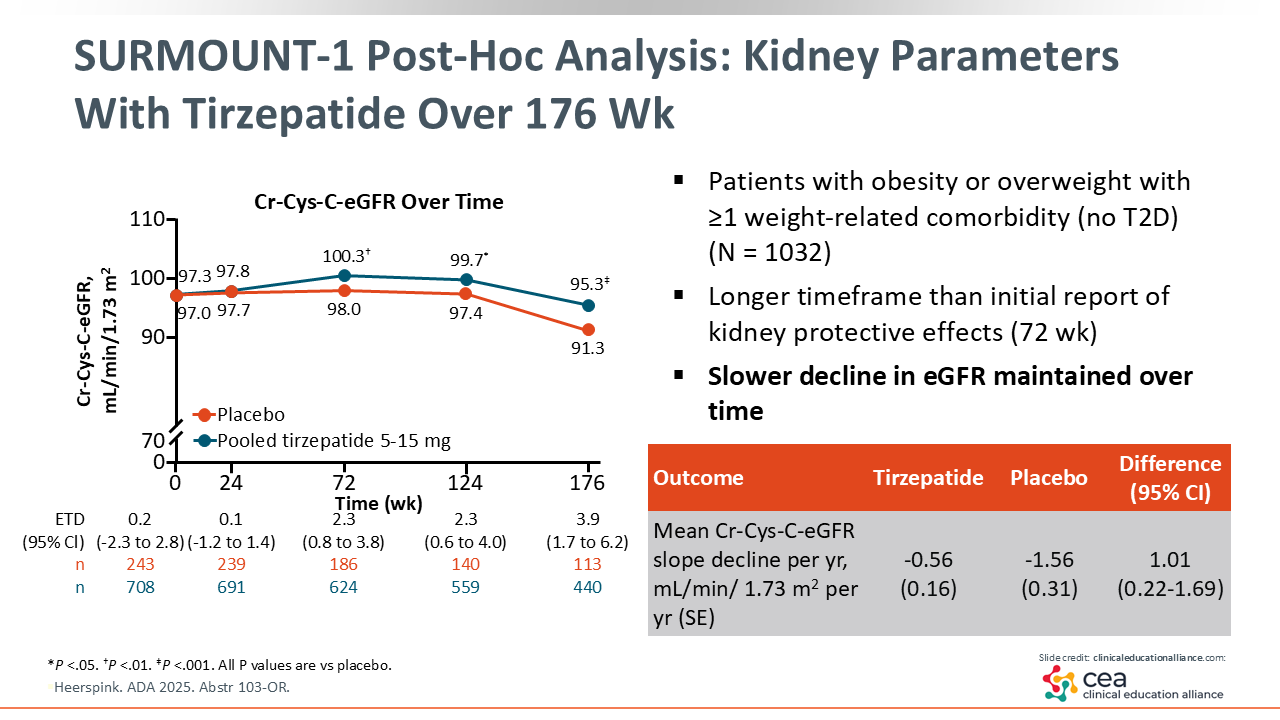
SURMOUNT-1 Post Hoc Analysis: Kidney Parameters With Tirzepatide vs Placebo Over 176 Weeks
There are additional data from the SURMOUNT-1 trial, which evaluated tirzepatide vs placebo in patients with obesity or overweight and ≥1 weight-related comorbidity but without T2D. In a post hoc analysis, renal benefits were reported with tirzepatide treatment. Over 176 weeks, there was a slower decline in estimated glomerular filtration rate (eGFR) with tirzepatide vs placebo. Again, these are results showing benefit for multiple systems, including the kidneys.22
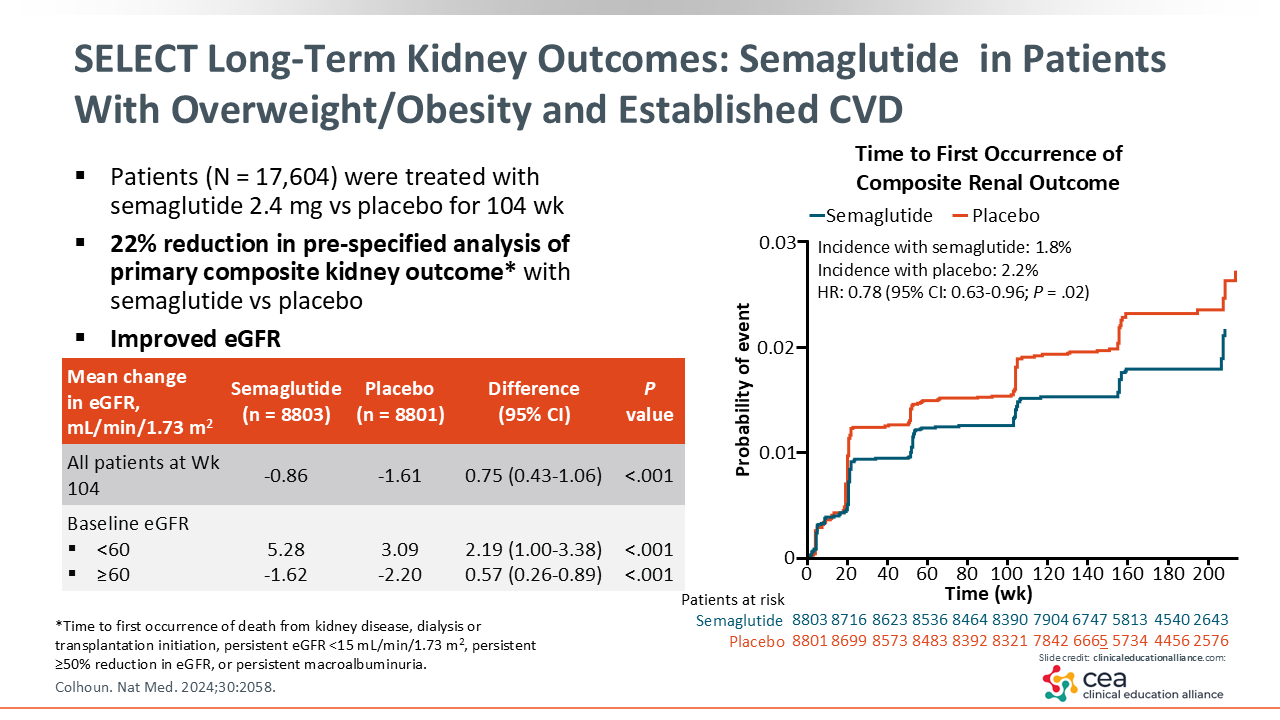
SELECT Long-term Kidney Outcomes: Semaglutide in Patients With Overweight/Obesity and Established CVD
Kidney benefits also have been reported for the SELECT trial of semaglutide 2.4 mg. Compared with placebo, in patients with obesity or overweight, established CVD, and no T2D, semaglutide was associated with a 22% reduction in risk for the prespecified composite kidney outcome (time to first occurrence of death from kidney disease, dialysis or transplantation initiation, persistent eGFR <15 mL/min/1.73 m2, persistent ≥50% reduction in eGFR, or persistent macroalbuminuria).23
These data indicate potential renal benefits with these agents. The observation of improved outcomes in both CV and renal systems, which are functionally tied together, suggests that these treatments may be addressing root causes of obesity-associated diseases in these patients.
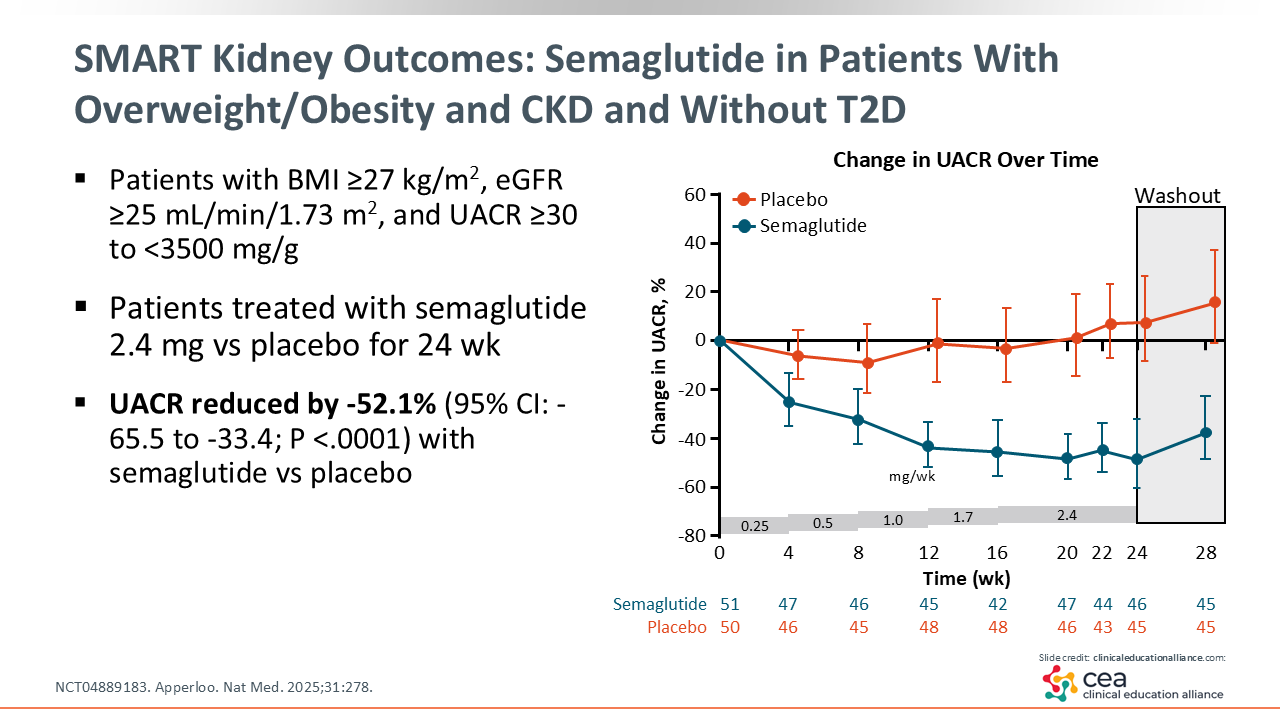
SMART Kidney Outcomes: Semaglutide in Patients With Overweight/Obesity and CKD
The SMART trial investigated kidney outcomes with semaglutide in patients with overweight or obesity, CKD, and without T2D. Investigators reported a 52% reduction in UACR with semaglutide compared with placebo.24 Of note, UACR is not only a risk identifier for T2D or hypertension; it is a marker of systemic disease.25 HCPs are doing the right thing for their patients if they can provide them with a treatment that reduces systemic disease.
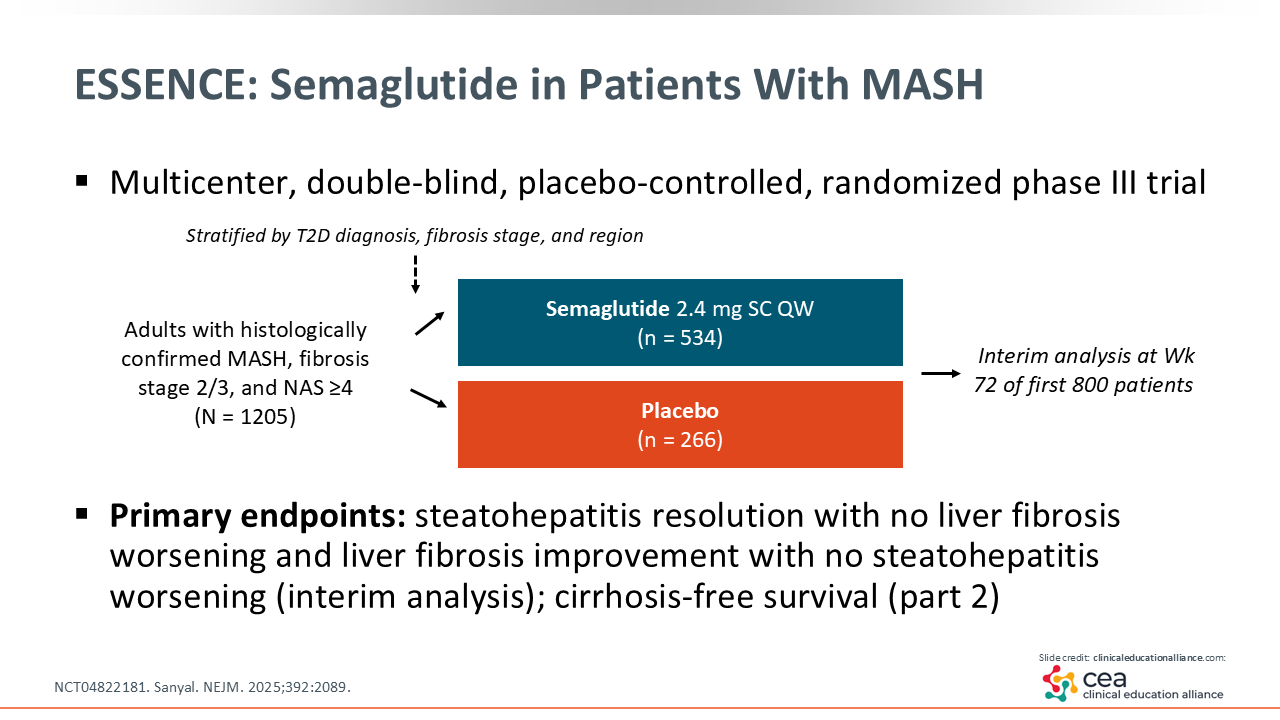
ESSENCE: Semaglutide in Patients With MASH
Moving on to liver disease, as previously mentioned, approximately one third of the global population and approximately 70% of patients in the United States with T2D have MASLD.9,10 This used to be termed nonalcoholic fatty liver disease, and MASH was previously known as nonalcoholic steatohepatitis (or NASH). It is important to recognize that MASLD/MASH can not only lead to liver complications, such as cirrhosis or hepatocellular cancer, but also increases the risk of all other nonhepatic cancers and CVD.26
Furthermore, MASH is a condition that will progress if left untreated. This progression may be faster among those with obesity, so positive results provide an opportunity to do something about that. Treatments for MASLD/MASH continue to be an unmet need, but there is growing evidence that semaglutide and tirzepatide provide benefit.
The multicenter, double-blind, placebo-controlled randomized phase III ESSENCE trial was designed to evaluate semaglutide vs placebo. It enrolled adults with histologically confirmed MASH, fibrosis stage 2 or 3, and a NASH activity score of ≥4.27
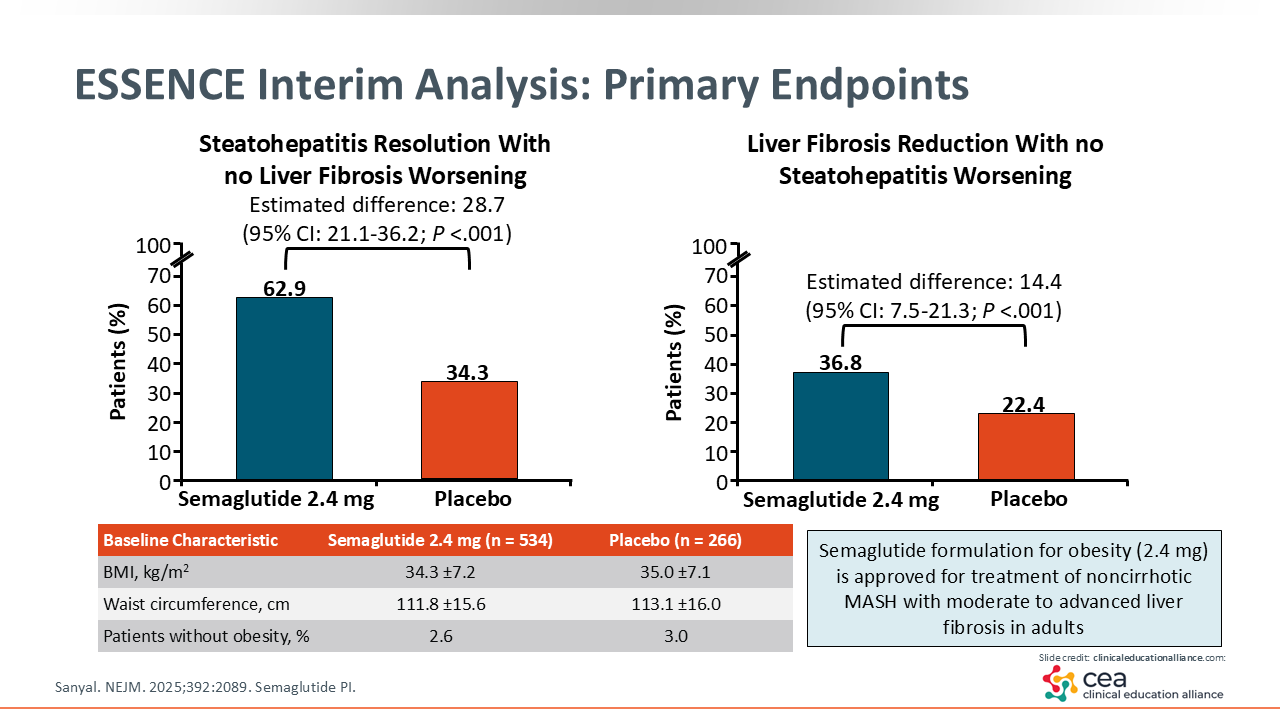
ESSENCE Interim Analysis: Primary Endpoints
At a prespecified interim analysis of ESSENCE results, investigators reported significant benefits with semaglutide vs placebo. Considering the primary endpoints, resolution of steatohepatitis with no liver fibrosis worsening occurred in 63% of patients in the semaglutide arm compared with 34% in the placebo arm (P <.001). Reduction in liver fibrosis with no steatohepatitis worsening, which is an important outcome, occurred in 37% of patients in the semaglutide arm vs 22% in the placebo arm (P <.001).27 Results of the ESSENCE trial supported approval of semaglutide, in combination with reduced calorie diet and increased physical activity, for treatment of noncirrhotic MASH with moderate to advanced liver fibrosis.11
ESSENCE: Additional Findings
In a post hoc analysis of ESSENCE, other MASLD outcomes were improved with semaglutide, and both weight loss–dependent and weight loss–independent mechanisms contributed to these improvements. So benefits may be mediated by effects in addition to weight loss, such as reduction in inflammation, although additional research is needed to characterize contributing weight loss–independent mechanisms.28
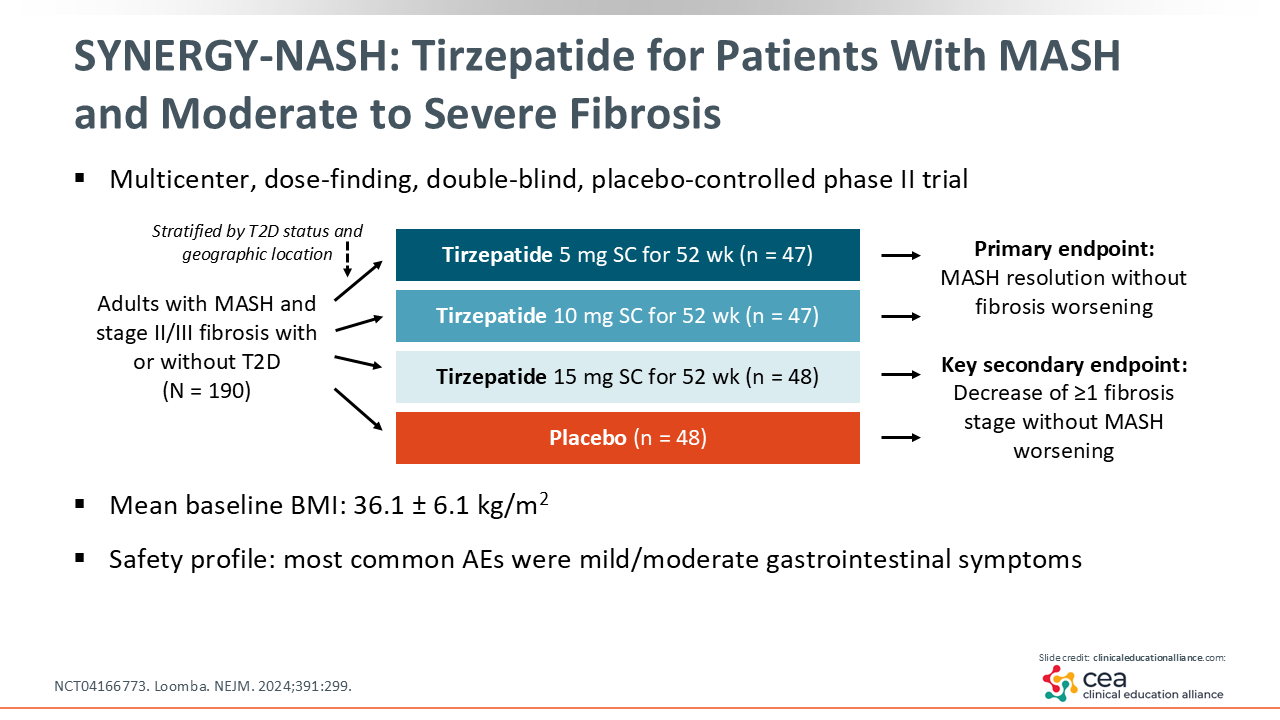
SYNERGY-NASH: Tirzepatide for Patients With MASH and Moderate to Severe Fibrosis
The SYNERGY-NASH trial enrolled adults with MASH and stage 2/3 fibrosis, with or without T2D. Patients were randomized to 3 doses of tirzepatide (5, 10, or 15 mg) or placebo. The trial’s primary endpoint was MASH resolution without fibrosis worsening.
In terms of safety, and like other tirzepatide studies, gastrointestinal-related adverse effects (AEs) were the most commonly reported AEs, but they were relatively self-limited and time dependent.29
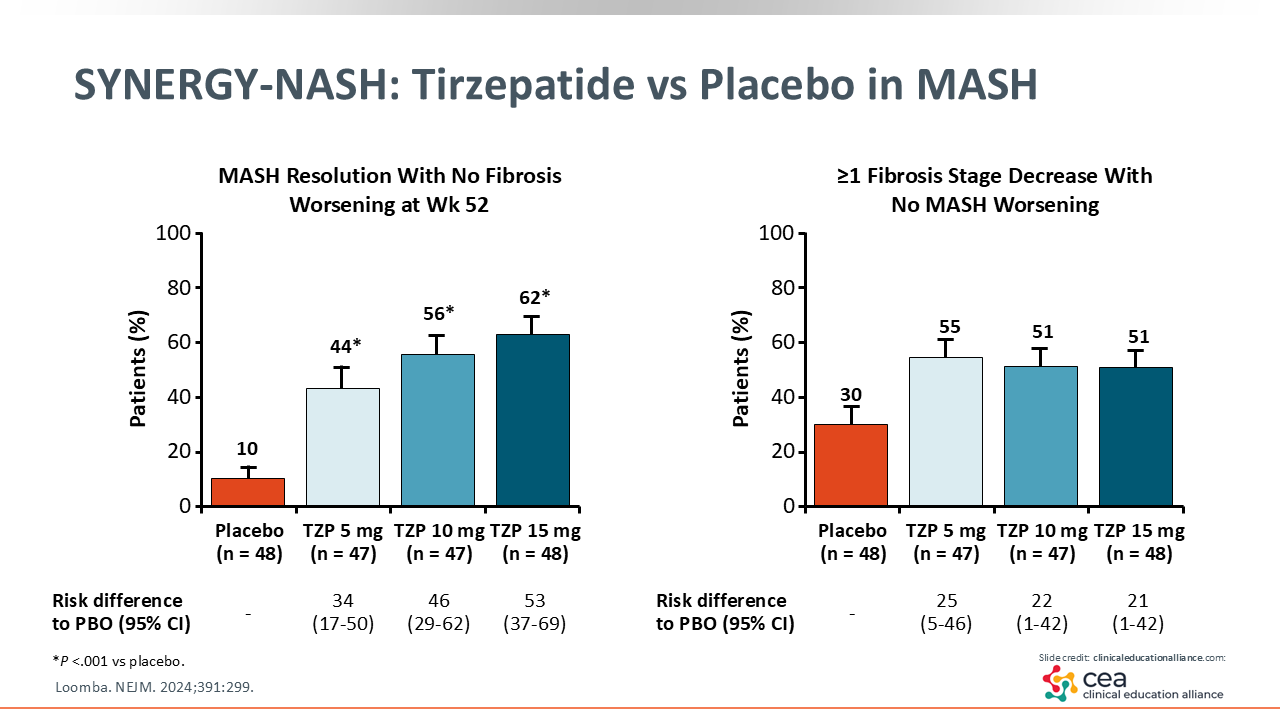
SYNERGY-NASH: Tirzepatide vs Placebo in MASH
In the tirzepatide arms of the SYNERGY-NASH trial, 44% to 62% of patients achieved the primary endpoint compared with 10% of patients in the placebo arm.29
Furthermore, in looking at the figure for the primary endpoint, there is a dose-dependent response up to 62% for MASH resolution with no fibrosis worsening for the 15-mg dose of tirzepatide.29
The key secondary endpoint reflects researchers’ focus on achieving improvement in fibrosis score by at least one stage, regardless of where patients are on the liver disease spectrum. In this trial, up to 55% of patients in the tirzepatide arms saw an improvement in fibrosis stage by at least one stage vs 30% of patients in the placebo arm.
Therefore, in addition to promoting significant weight loss, semaglutide and tirzepatide also provide benefits in MASH, with semaglutide having approval in adults, in combination with reduced calorie diet and increased physical activity, for the treatment of noncirrhotic MASH with moderate to advanced fibrosis.11
Case 2: Key Points on Individualized Care—MASH
Thinking about this patient case, it is important to recognize that weight loss is the main treatment for MASLD/MASH.30 Weight loss of 3% to 5% can improve hepatic steatosis and transaminase levels, weight reduction of 7% to 10% can stop disease progression, and weight loss >10% can lead to fibrosis regression.31-33
For patients with MASLD/MASH who do not have cirrhosis, consider starting treatment right away, ideally with an agent that supports weight loss and provides liver benefit, such as a GLP-1–based therapy. It is also worth noting that patients with MASLD or MASH have a higher CV risk, and semaglutide is an AOM that can reduce this risk in people with overweight/obesity and established CVD. Then work closely with hepatology. Whereas a primary care professional manages incretin-based therapy, the hepatologist can decide if patients should be offered liver-directed therapy.