CE / CME
Evolving Treatment Landscape for HER2-Positive/HER2-Low Early Breast Cancer
Physician Assistants/Physician Associates: 0.50 AAPA Category 1 CME credit
Pharmacists: 0.50 contact hour (0.05 CEUs)
ABIM MOC: maximum of 0.50 Medical Knowledge MOC point
Physicians: maximum of 0.50 AMA PRA Category 1 Credit™
Nurse Practitioners/Nurses: 0.50 Nursing contact hour
Released: May 01, 2026
Expiration: October 31, 2026
Activity
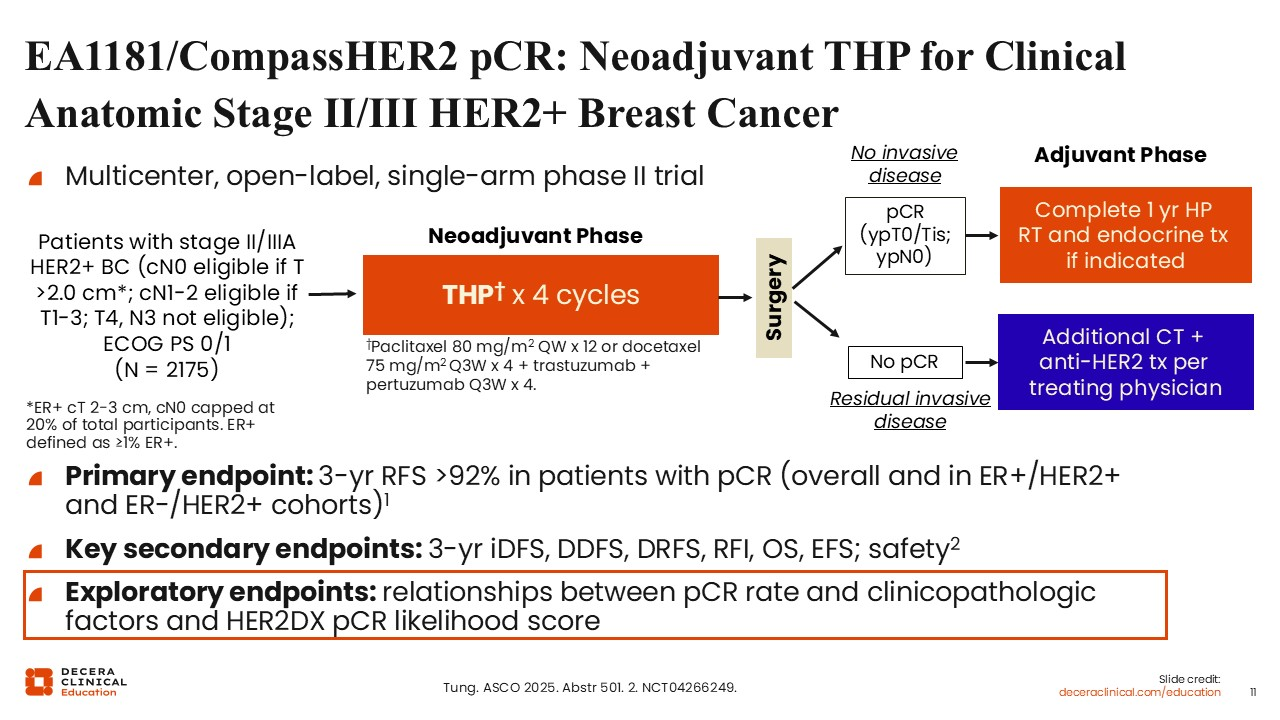
EA1181/CompassHER2 pCR: Neoadjuvant THP for Clinical Anatomic Stage II/III HER2+ Breast Cancer
Several key ongoing clinical trials are exploring contemporary treatment strategies for HER2‑positive EBC and continue to improve outcomes for neoadjuvant and adjuvant settings. The following trial summaries highlight representative approaches for multiple patient subgroups with HER2-positive EBC.
The first ongoing study I would like to discuss is the multicenter, open-label, phase II CompassHER2 pCR study, which is evaluating a response‑adapted neoadjuvant THP regimen for 4 cycles followed by surgery. Patients are then stratified into those with no invasive disease (pCR) vs those with residual invasive disease (no pCR) after THP.21 In the adjuvant phase, patients in the pCR group were given 1 year of HP with radiotherapy and endocrine therapy if indicated, and patients with no pCR received additional chemotherapy with anti-HER2 treatment based on physician’s decision. The primary endpoint of the study is 3-year relapse-free survival >92% in patients with pCR (overall and in estrogen receptor [ER]–positive/HER2-positive and ER-negative/HER2-positive cohorts). Key secondary endpoints for the study include 3-year iDFS, distant disease-free survival, distant recurrence-free survival, recurrence-free interval, OS, event-free survival (EFS), and safety. Of importance, exploratory endpoints looked at the relationships between pCR rate and clinicopathologic factors and HER2Dx pCR likelihood score.
CompassHER2 pCR Exploratory Endpoints
Investigators from CompassHER2 pCR reported that in patients with stage II or III HER2-positive EBC, THP was associated with a pCR rate of 44% in all participants, 64% in ER-negative, and 33% in ER-positive disease. Of note, a significantly higher pCR rate was associated with the following factors: ER-positive status of ≤70% or ER-negative; HER2 IHC 3+ (vs IHC 2+/ISH+); weekly paclitaxel (vs Q3W docetaxel); and higher HER2Dx pCR score. By contrast, pCR was not associated with baseline clinical T and N stage. Investigators also note that the HER2Dx pCR score provides a composite complementary estimate of pCR likelihood and that clinicopathologic factors and molecular tools may help select patients with HER2-positive EBC for less intensive chemotherapy.
DESTINY-Breast05: Adjuvant Trastuzumab Deruxtecan (T-DXd) in HER2-Positive High-Risk EBC
More recently, we have seen data emerge from the global multicenter DESTINY-Breast05 trial supporting its listing as a category 1 preferred option in guidelines in the adjuvant setting .7,20
The overall trial design for DESTINY-Breast05 was conceptually similar to that of the KATHERINE study19 but targeted a more stringently defined high‑risk population. To be eligible for DESTINY-Breast05, patients were required to have residual invasive disease in the breast and/or axillary lymph nodes after completion of neoadjuvant chemotherapy with HER2‑targeted therapy. High‑risk disease was defined based on clinical stage at presentation and response to neoadjuvant treatment, including patients with inoperable disease (such as clinical T4 tumors or extensive nodal involvement), as well as those with operable disease who had persistent nodal involvement following neoadjuvant therapy. Patients were randomized to receive either T-DXd or T‑DM1 for a total of 14 cycles in the adjuvant setting. The primary endpoint of the study was iDFS.
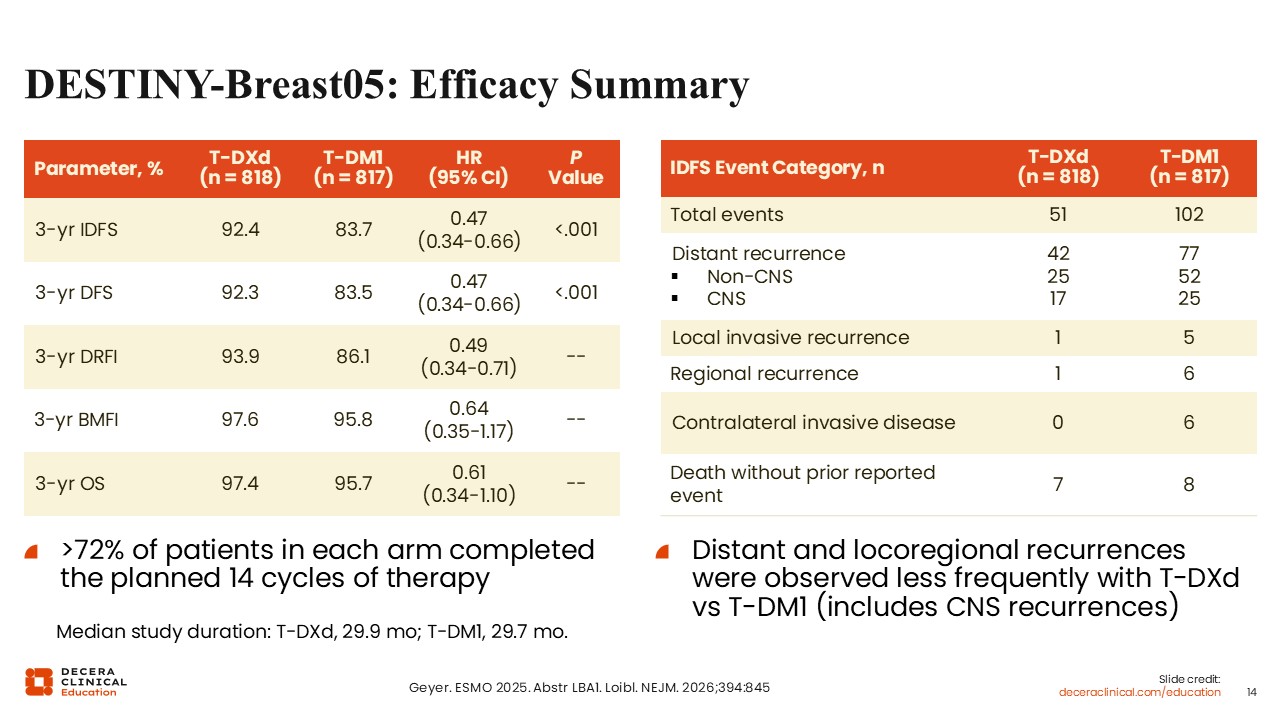
DESTINY-Breast05: Efficacy Summary
When compared to T-DM1, T-DXd significantly improved 3-year iDFS rates (92.4% vs 83.7%; hazard ratio: 0.47; 95% CI: 0.34-0.66; P <.001). Moreover, 3-year disease-free survival (92.3% vs 83.5%; hazard ratio: 0.47; 95% CI: 0.34-0.66; P <.001) and 3-year distant recurrence-free interval (93.9% vs 86.1%; hazard ratio: 0.49; 95% CI: 0.34-0.71) were also improved. OS data remain immature.
Treatment completion rates were favorable, with more than 72% of patients in each treatment arm completing all 14 planned cycles.
Analysis of recurrence patterns showed approximately twice as many recurrence events in the T‑DM1 arm compared with the T-DXd arm, with most events representing distant recurrence (77% vs 42%).
The number of central nervous system events in the T-DM1 arm was 25 compared with 17 in the T-DXd arm. There is ongoing interest in better defining the impact of T-DXd on central nervous system recurrence, although follow‑up to date remains limited and additional data are awaited.
These data led to an NCCN category 1 listing of adjuvant therapy with T-DXd in patients with high-risk HER2-positive EBC, defined as inoperable disease at presentation (cT4, N0-3, M0 or cT1-3, N2-3, M0) or operable disease with residual axillary node–positive disease (ypN1-3) following neoadjuvant therapy, reflecting its emerging role in response-adapted adjuvant treatment strategies.
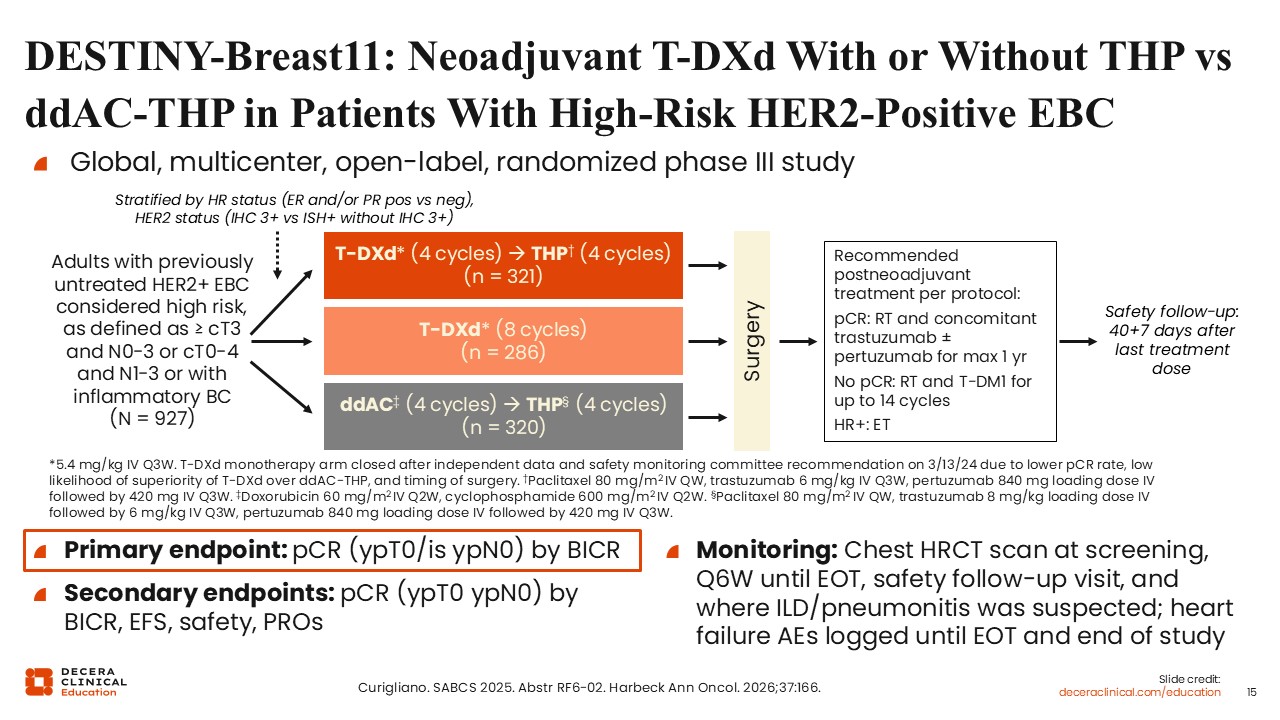
DESTINY-Breast11: Neoadjuvant T-DXd With or Without THP vs ddAC-THP in Patients With High-Risk HER2-Positive EBC
Building on the encouraging findings from the DESTINY-Breast05 trial in the adjuvant setting,22 we will now discuss the ongoing phase III DESTINY‑Breast11 trial. This study focused on a population with a substantial baseline risk of recurrence and therefore addressed an important area of therapeutic development within NCCN‑aligned neoadjuvant treatment paradigms.
DESTINY‑Breast11 is evaluating neoadjuvant T-DXd given for 4 cycles followed by 4 cycles of THP, compared with T-DXd alone or ddAC for 4 cycles followed by 4 cycles of THP in patients with previously untreated HER2-positive high-risk EBC (defined as ≥ cT3 and N0-3 or cT0-4 and N1-3 or with inflammatory phenotype).22 Patients with lower‑risk disease, including those with clinical T2N0 tumors, were intentionally excluded, underscoring that the findings of this trial are most applicable to patients with higher‑risk presentations. The primary endpoint of the study is pCR by blinded independent review committee (BICR). Secondary endpoints include pCR by BICR, EFS, safety, and patient-reported outcomes.
At an early interim analysis, the T-DXd monotherapy arm was discontinued, and only the T-DXd followed by THP and ddAC followed by THP arms are being compared.
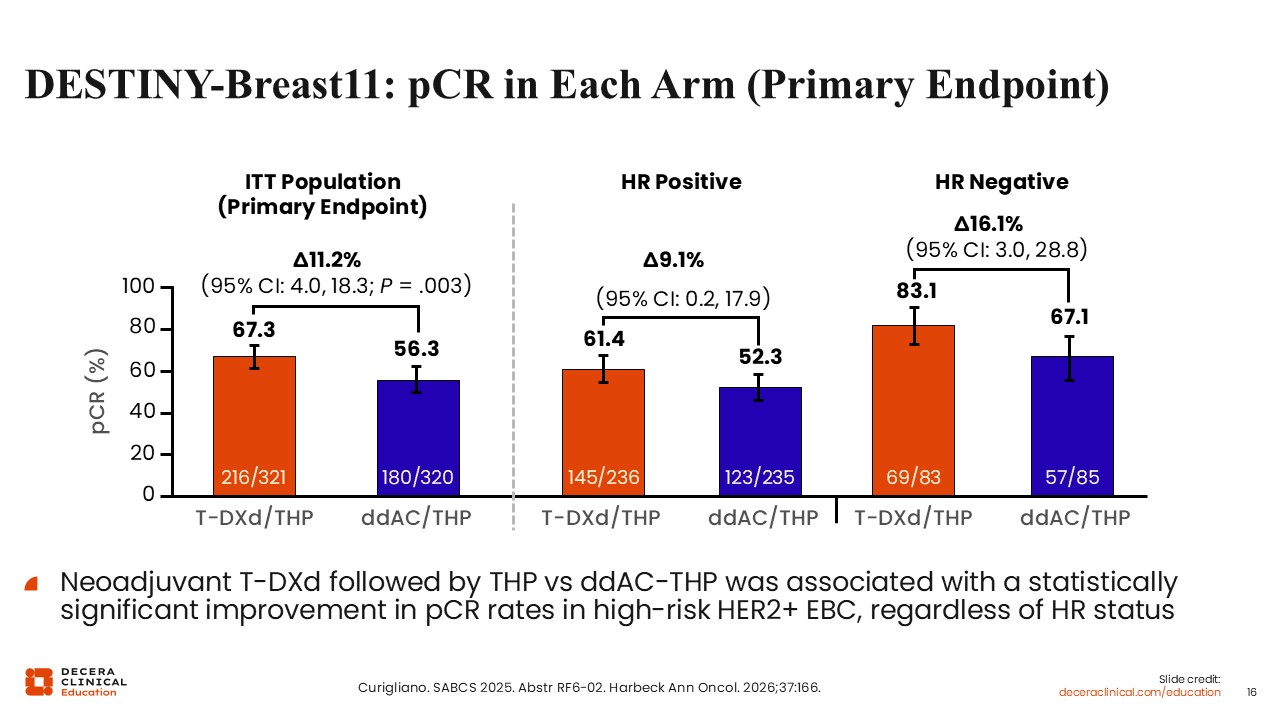
DESTINY-Breast11: pCR in Each Arm (Primary Endpoint)
With regard to the primary endpoint of pCR, the highest pCR rates were observed with T-DXd followed by THP compared with ddAC followed by THP (67.3% vs 56.3%; 95% CI: 4.0- 18.3; P = .003). The absolute magnitude of pCR improvement with T-DXd followed by THP compared with ddAC followed by THP was approximately 11%.22
Of note, improvements in pCR rates with T-DXd followed by THP were higher with ddAC followed by THP across prespecified subgroups (HR positive or negative), with higher pCR rates observed in HR-negative disease (83.1% vs 67.1%; 95% CI: 3.0-28.8) than in HR-positive disease (61.4% vs 52.3%; 95% CI: 0.2-17.9), consistent with prior observations in HER2‑positive breast cancer. Taken together, these findings support the antitumor activity of T-DXd in the neoadjuvant setting and highlight the importance of combination therapy in this setting. Moreover, careful patient selection is warranted when considering the application of these data in clinical practice, given the exclusion of lower‑risk populations and the evolving role of T-DXd across neoadjuvant and adjuvant treatment settings.
neoCARHP: De-escalated Neoadjuvant Taxane + Trastuzumab and Pertuzumab With or Without Carboplatin in HER2+ EBC
Another strategy involves identifying patients with HER2-positive EBC who might benefit from treatment de-escalation.
The multicenter, open-label, phase III neoCARHP study evaluated neoadjuvant treatment with THP for 6 cycles every 3 weeks vs THP with intravenous carboplatin AUC 6 mg/mL/min (TCHP) also for 6 cycles every 3 weeks in patients with previously untreated HER2-positive stage II-IIIC EBC and ECOG performance status of ≤1.23 The primary endpoint of the study was pCR among all patients receiving 1 dose of therapy. Secondary endpoints included EFS, DFS, OS, clinical response during neoadjuvant therapy, and safety.
This trial addressed an important clinical question regarding the incremental benefit of carboplatin when dual HER2 blockade is used in the neoadjuvant setting.
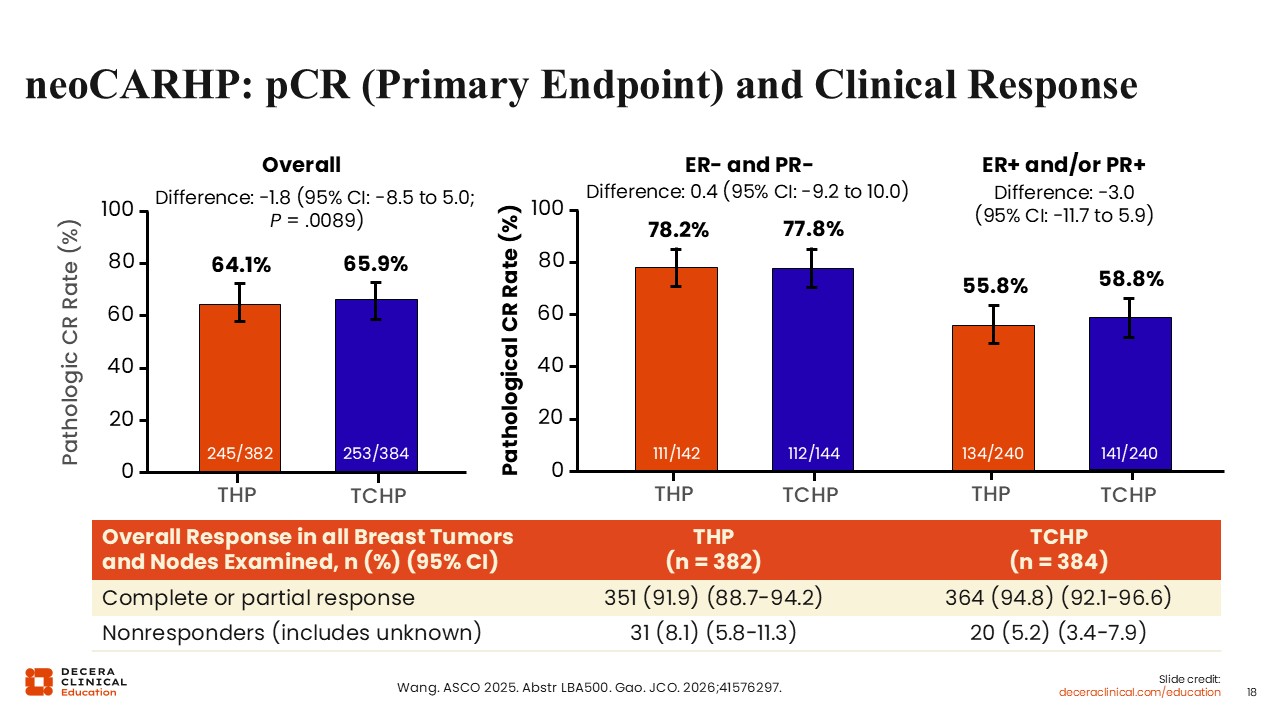
neoCARHP: pCR (Primary Endpoint) and Clinical Response
Results from neoCARHP showed the THP and TCHP treatment arms yielded similar pCR rates overall (64.1% vs 65.9%; 95% CI: -8.5 to 5.0). Moreover, similar pCR results were observed with THP compared with TCHP in patients with ER-negative disease (78.2% vs 77.8%) and ER-positive disease (55.8% vs 58.8%).
Overall response (complete or partial) in patients receiving THP vs TCHP were 91.9% vs 94.8%. The proportion of nonresponders in the THP vs the TCHP arm were 8.1% and 5.5%, respectively.
These findings suggest that the addition of carboplatin may not substantially improve short‑term pCR rates in this setting, and that THP can potentially achieve noninferior pCR rates when compared with TCHP across both ER-positive and ER-negative subgroups.
However, interpretation of these results requires caution, as long‑term outcomes, including EFS and OS, remain immature and the findings are not yet practice changing.
In addition, the study compared 6 cycles of THP with 6 cycles of TCHP, which differs from other neoadjuvant approaches commonly used in clinical practice, including regimens with shorter durations of THP or alternative sequencing strategies.
In real‑world practice, these findings may support consideration of THP in selected patients, particularly in situations where toxicity, comorbidities, or patient preferences influence treatment selection. The neoCARHP results contribute to broader efforts to evaluate chemotherapy de-escalation and balance treatment efficacy with tolerability in HER2‑positive EBC, while underscoring the need for longer follow‑up and additional data to guide decisions regarding chemotherapy de‑escalation.
Takeaways on Emerging Data for (Neo)adjuvant Trials in HER2-Positive EBC
ADC Neoadjuvant Options
Unanswered questions about where to integrate the most recent data for T-DXd include whether biomarkers may help identify patients who are more or less likely to benefit from T-DXd and THP and/or platinum-based chemotherapy. Additional follow-up will help better define the benefit of using T-DXd earlier within the treatment landscape, including the benefit of using 4 cycles of T-DXd in the neoadjuvant setting compared to 14 cycles in the adjuvant setting. Clinical trial data are immature at this time.
There are also outstanding questions regarding the optimal duration of neoadjuvant therapy (eg, 4 vs 6 cycles) and whether taxane selection influences outcomes. Smaller studies evaluating shorter duration regimens, including 4 cycles of THP, have demonstrated encouraging pCR rates and are being further explored in larger trials.24
Beyond carboplatin omission, other de-escalation strategies are being actively investigated in HER2-positive EBC. These include response-adapted approaches in which treatment intensity is tailored based on pCR, as well as regimens that reduce or omit components of cytotoxic chemotherapy in selected patients. Although these strategies aim to minimize toxicity without compromising efficacy, their application in routine clinical practice remains dependent on pending longer-term outcomes and careful patient selection.
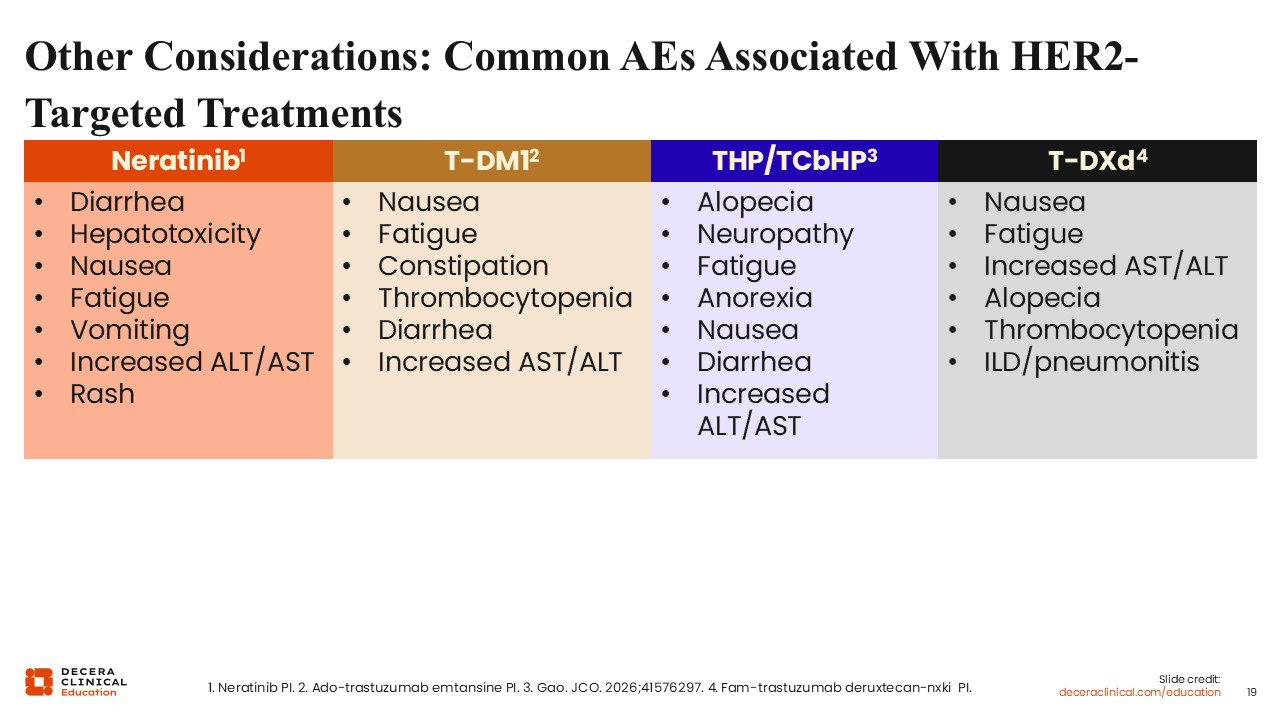
Other Considerations: Common AEs Associated With HER2-Targeted Treatments
HER2‑directed therapies are associated with distinct toxicity profiles, and patient comorbidities should be carefully considered when selecting and sequencing treatment. Individualizing therapy based on baseline health status and anticipated tolerability is essential to support adherence and completion of guideline‑concordant care.
Neratinib is most associated with gastrointestinal toxicity, particularly diarrhea, as well as rash and elevations in liver function tests.17,18 These AEs may impact treatment tolerability and adherence, especially in the extended adjuvant setting. Proactive management strategies, including dose escalation approaches and antidiarrheal prophylaxis (ie, loperamide), may help mitigate toxicity in selected patients.
T‑DM1 is associated with peripheral neuropathy, which may be concerning in patients with preexisting neuropathic symptoms.13,19 Close monitoring is recommended, and dose modifications may be required if neuropathy worsens during treatment.
Neoadjuvant combination regimens such as THP and TCHP differ in their overall toxicity profiles.21,23 Regimens that include carboplatin, such as TCHP, are generally associated with a higher toxicity burden, including fatigue, nausea, cytopenias, and gastrointestinal AEs. In patients with frailty or significant comorbidities, THP may represent a more tolerable alternative while still providing effective dual HER2 blockade.
T-DXd is associated with a clinically meaningful risk of ILD/pneumonitis, which is a key consideration when treating patients with preexisting pulmonary conditions or limited pulmonary reserve.20,22 Baseline pulmonary assessment and vigilance for new or worsening respiratory symptoms are essential when using this therapy.12
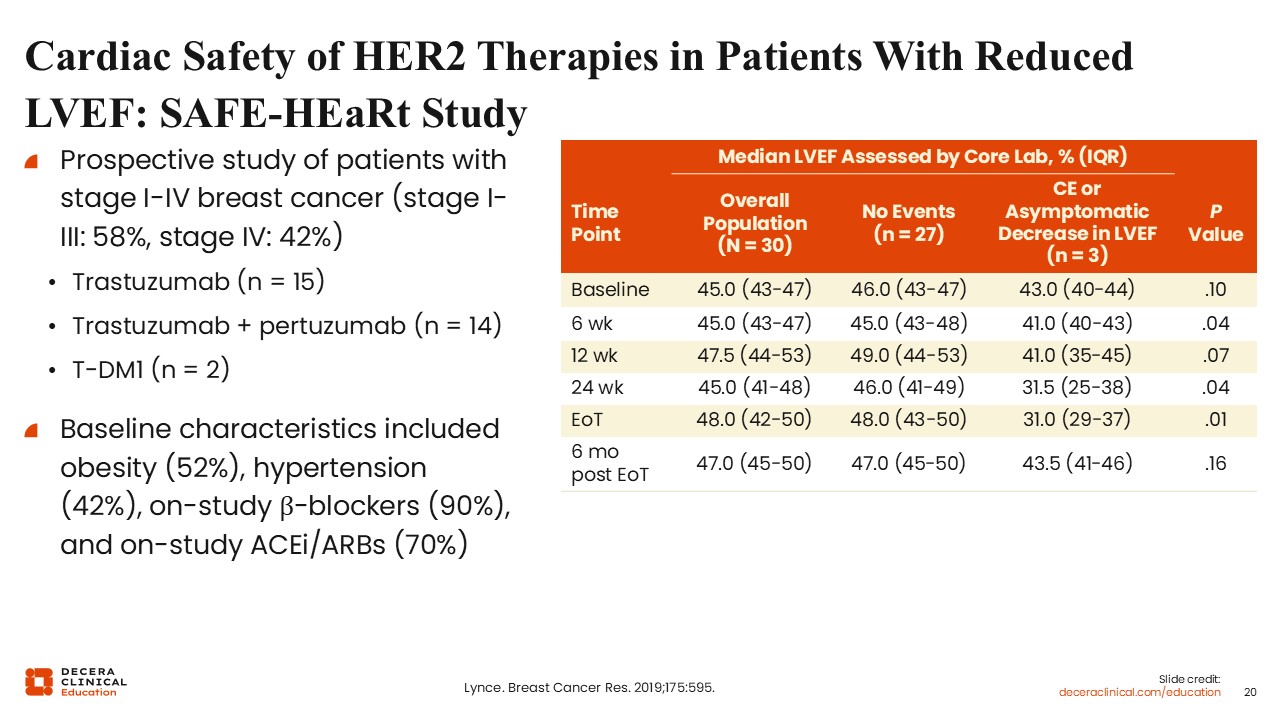
Cardiac Safety of HER2 Therapies in Patients With Reduced LVEF: SAFE-HEaRt Study
Cardiac function may also impact treatment selection and AE monitoring strategies. For instance, a patient with cardiac dysfunction requires close monitoring across all HER2‑directed therapies, including baseline and periodic assessment of LVEF. Collaboration with a cardiologist or cardio‑oncology specialists may help optimize cardiac function and support continuation of therapy when appropriate.25
In a prospective study of patients with breast cancer, 58% with stage I-III EBC receiving either trastuzumab (n = 15), trastuzumab with pertuzumab (n = 14), or T-DM1 (n = 2), an asymptomatic decrease in LVEF was observed between 24 weeks up to end of therapy, which appears to recover close to baseline at approximately 6 months from the end of therapy.26
Baseline characteristics associated with LVEF reduction risk may include high body mass index (obesity, 52%), high blood pressure/hypertension (42%), receiving β-blockers (90%), and on-study angiotensin-converting enzyme inhibitors/angiotensin receptor blockers (70%).
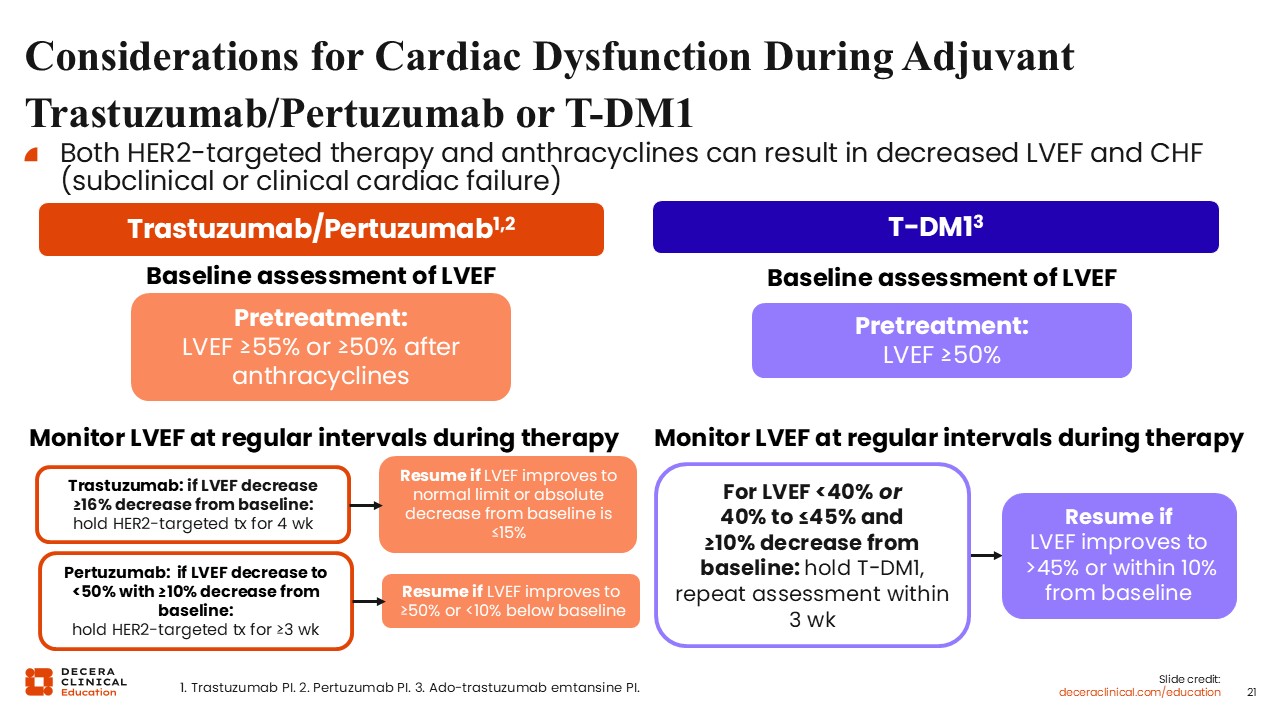
Considerations for Cardiac Dysfunction During Adjuvant Trastuzumab/Pertuzumab or T-DM1
Before beginning adjuvant therapy with trastuzumab and pertuzumab, a baseline cardiac function assessment is essential to ensure a pretreatment LVEF of ≥55%, or ≥50% for those who have previously received anthracyclines. Throughout the course of treatment, LVEF must be monitored at regular intervals to detect potential cardiac dysfunction.
For patients receiving trastuzumab, therapy should be held for 4 weeks if there is a ≥16% decrease in LVEF from the established baseline. Treatment can be resumed once the LVEF returns to normal limits or if the absolute decrease from baseline remains ≤15%.14 Similarly, for pertuzumab, therapy is held for at least 3 weeks if the LVEF drops below 50% alongside a ≥10% decrease from baseline. Resumption is permitted once the LVEF improves to ≥50% or reaches a level less than 10% below the baseline. These guidelines are supported by the respective prescribing information for each agent.
In the case of T-DM1, the pretreatment requirement is a baseline LVEF of ≥50%. Monitoring continues at regular intervals during therapy to ensure patient safety. If the LVEF falls below 40% or is measured between 40% and 45% with a ≥10% decrease from baseline, T-DM1 must be held and a repeat assessment performed within 3 weeks. Therapy may resume if the LVEF improves to >45% or within 10% of the baseline value. Clinical management should always be guided by the specific recommendations found in the prescribing information for T-DM1.
Other Relevant Comorbidities in Patients Receiving (Neo)adjuvant Therapy for HER2-Positive EBC
When thinking about giving patients all options available to them, we must first address how other relevant comorbidities like diabetes may influence chemotherapy selection and supportive care planning due to the use of corticosteroids, particularly with taxane‑based regimens. Cognitive impairment can also affect treatment adherence, especially with oral therapies such as neratinib.
Older adults with EBC may experience distinct patterns of treatment-related toxicity and often require additional consideration in treatment selection and management. Age-related changes in cardiac function, renal and hepatic reserve, and increased prevalence of comorbidities may increase susceptibility to AEs, including cardiotoxicity with HER2-targeted agents, cytopenias, and treatment-related fatigue.
Functional status, frailty, and polypharmacy are also important considerations, as these factors may influence both ADC tolerability and adherence. In clinical practice, treatment approaches for older patients are often individualized with consideration of less intensive regimens or lower/modified dosing strategies that remain efficacious, and when appropriate.
Comprehensive geriatric assessment and multidisciplinary care may help optimize treatment selection, anticipate toxicity, and support safe delivery of HER2-directed therapy in this population, such as with T-DXd.
Differences in AE risk and treatment tolerability have been reported across patient populations, which may further influence outcomes. However, based on limited data from a real-world, retrospective, registry study,27 T-DXd use in older patients (≥70 years) was not associated with new safety signals, with ILD/pneumonitis rates observed in line with previously reported data. Thus, age should not be a criterion to discourage treatment with T-DXd. That being said, a multidisciplinary approach, including oncology, nursing, pharmacy, and relevant subspecialists, is essential to support proactive AE management in all patients receiving HER2-targeted therapy, addressing comorbidities, and ensuring that patients can safely complete their HER2‑directed therapy.