CE / CME
Evolving Treatment Landscape for HER2-Positive/HER2-Low Early Breast Cancer
Physician Assistants/Physician Associates: 0.50 AAPA Category 1 CME credit
Pharmacists: 0.50 contact hour (0.05 CEUs)
ABIM MOC: maximum of 0.50 Medical Knowledge MOC point
Physicians: maximum of 0.50 AMA PRA Category 1 Credit™
Nurse Practitioners/Nurses: 0.50 Nursing contact hour
Released: May 01, 2026
Expiration: October 31, 2026
Activity
Healthcare Disparities Among Patients With Breast Cancer
Despite substantial advances in systemic therapy and the availability of guideline‑directed treatment, disparities in outcomes remain evident among patients with HER2‑positive EBC.1 Across multiple studies comparing outcomes for Black vs White patients with EBC, differences have been observed with regard to lower rates of mammogram examination, a 65% higher rate of incidence of any hormone receptor (HR)–negative breast cancer subtype, a higher likelihood of not having adequate healthcare coverage, and a 40% higher risk of mortality from breast cancer.2,3
Disparities in Prognosis/Outcomes for Patients With HER2-Positive EBC
Data across various studies suggest that Black patients were less likely to receive neoadjuvant chemotherapy or achieve a pathologic complete response (pCR).4 Of note, Black women also experience significantly higher rates of cardiotoxicity and poorer cardiac recovery during HER2-targeted therapy.5
A comparison of key side effects in Black vs White patients shows more6:
- Chemotherapy-related complications (20.9% vs 12.2%)
- Cardiac toxicity, including acute myocardial infarction (3.1% vs 0.2%)
- Pneumonitis (7.8% vs 2.0%)
- Impacted by cognitive decline or dementia (13.6% vs 6.7%)
Current clinical practice guidelines7 emphasize completion of HER2‑directed therapy when clinically feasible. However, the aforementioned safety considerations may have downstream implications for long‑term outcomes.
Together, these data highlight the need for continued efforts to ensure equitable access to National Comprehensive Cancer Network (NCCN) guideline‑concordant care across the entire breast cancer continuum, as well as improved representation of diverse populations in clinical trials to better inform evidence‑based management strategies.
Unmet Needs in HER2-Positive and HER2-Low EBC
Response to neoadjuvant therapy is a key determinant of recurrence risk, and patients with residual invasive disease following neoadjuvant systemic treatment represent a particularly high‑risk group.8 Despite receipt of HER2‑directed therapy, patients with residual disease face a substantial risk of recurrence, underscoring the importance of optimizing adjuvant treatment strategies, consistent with guideline recommendations.7 On the other hand, patients with HER2-positive status who achieve a pCR after neoadjuvant treatment generally experience more favorable outcomes and are typically managed with continuation of trastuzumab with or without pertuzumab to complete 1 year of HER2‑directed therapy.7
Management of HER2‑low EBC remains less well defined. At present, treatment approaches often align with those used for HER2‑negative breast cancer. Increasing recognition of biologic heterogeneity within the HER2‑low population has led to ongoing investigation of more individualized and tumor‑informed strategies.9
Toxicity and treatment adherence are critical considerations across HER2‑directed therapies. Cardiac dysfunction, including decreases in left ventricular ejection fraction (LVEF), may occur with multiple agents and requires routine monitoring in accordance with guideline recommendations.7,10,11
ADCs are associated with a clinically meaningful risk of interstitial lung disease (ILD)/pneumonitis, which appears to occur at higher rates than with other HER2‑targeted therapies and warrants early detection and prompt management.12,13 Gastrointestinal toxicity and peripheral neuropathy are additional concerns, particularly with ado‑trastuzumab emtansine (T‑DM1).13 Thus, proactive monitoring and supportive care are essential, as these toxicities may negatively impact treatment adherence, persistence, and completion of cancer therapy.
Evidence gaps remain regarding limited representation of older patients, patients with significant comorbidities, and underrepresented patients of colors (Black/Latina) in clinical research for whom limited data exist.
Emerging approaches, including ADCs and circulating tumor DNA–guided strategies, may help overcome some of the aforementioned unmet needs by enabling improved detection of residual disease and risk-adapted escalation or de-escalation of therapy.
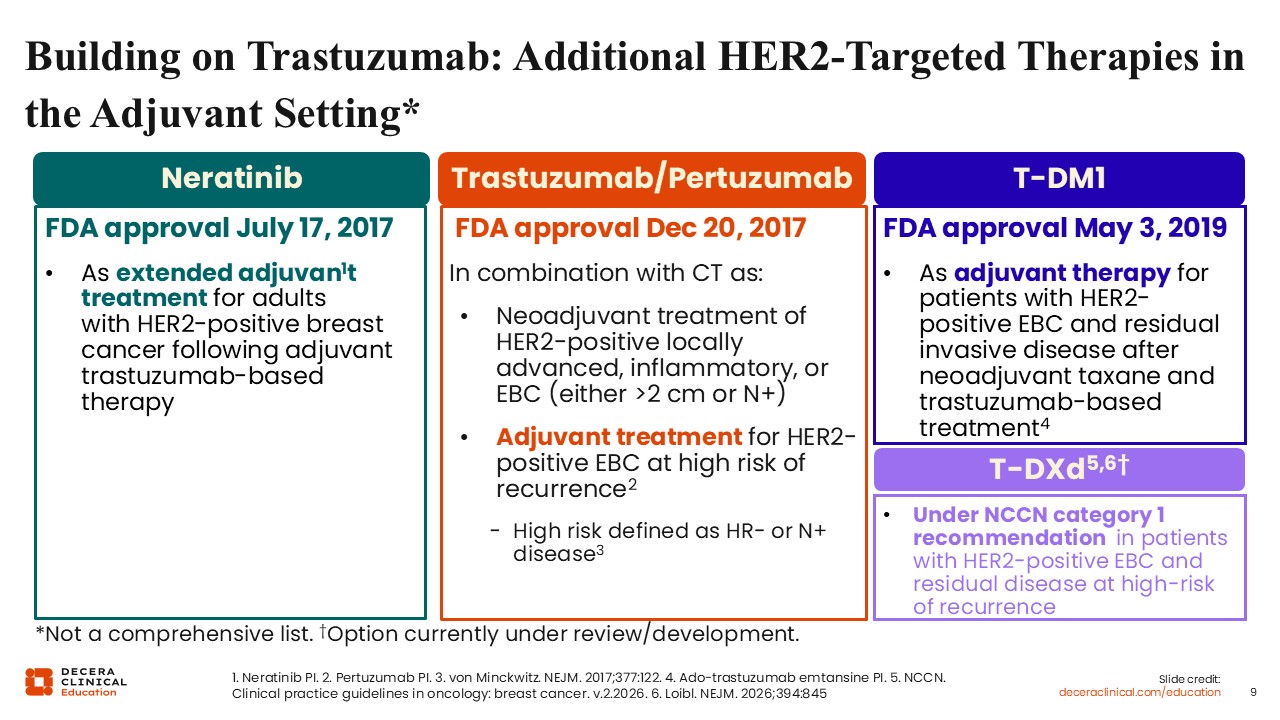
Building on Trastuzumab: Additional HER2-Targeted Therapies in the Adjuvant Setting
The standard of care for HER2‑positive EBC has evolved substantially in the last decade, resulting in marked improvements in disease‑free and overall survival (OS) outcomes.
These advances have occurred through the sequential development of HER2‑targeted therapies, each building on the foundation established by trastuzumab. Initially approved in the metastatic setting, trastuzumab was subsequently incorporated into the management of early‑stage disease, where it became the cornerstone of HER2‑directed therapy and remains central to current guideline‑recommended treatment strategies.7,14
As experience with trastuzumab expanded, additional agents were introduced to improve outcomes in higher‑risk populations. In 2017, pertuzumab received FDA approval15 in combination with chemotherapy for the neoadjuvant treatment of patients with HER2‑positive breast cancer whose tumors were ≥2 cm or who had node‑positive disease. This approval reinforced the role of neoadjuvant systemic therapy as a standard approach for patients with higher‑risk HER2‑positive disease, consistent with current NCCN recommendations.7
Pertuzumab may also be used in the adjuvant setting for patients at increased risk of recurrence, typically continued in combination with trastuzumab following neoadjuvant therapy when tolerated.14,15
Neratinib is an irreversible pan-ErbB tyrosine kinase inhibitor that binds and blocks the signaling of HER2, EGFR, and HER4 receptors.16 Neratinib received FDA approval in 2017 as extended adjuvant therapy following completion of trastuzumab‑based treatment.17 Commonly referred to as “Year 2” therapy, neratinib is administered after completion of the initial year of HER2‑directed therapy and has demonstrated benefit in select patient populations, particularly those with HR-positive disease.
The randomized phase III ExteNET trial evaluated extended adjuvant therapy with neratinib compared to placebo following completion of trastuzumab‑based treatment in patients with HER2-positive EBC who had completed neoadjuvant and adjuvant chemotherapy plus trastuzumab. At the 5.2 year median follow-up, the study demonstrated improvement in invasive disease‑free survival (iDFS) favoring use of neratinib (hazard ratio: 0.73; P = .0083), but it did not show improvement in OS rates after 8 years of follow-up (90.1% vs 90.2%; hazard ratio: 0.86; 95% CI: 0.75-1.24; P = .6914).18
The use of neratinib in this setting reflects a strategy of prolonged HER2 pathway inhibition to further reduce the risk of late recurrence.
T‑DM1 represented a significant shift toward response‑adapted therapy in EBC. In patients with HER2-positive status and residual disease following neoadjuvant treatment, T-DM1 is an adjuvant treatment option to consider based on the results from the phase III KATHERINE study.19 The KATHERINE study was conducted in 1486 patients with HER2-positive EBC and residual invasive disease following trastuzumab with chemotherapy (with or without anthracycline). Patients were randomized to receive either adjuvant T-DM1 or trastuzumab for 14 cycles. The primary study endpoint of iDFS was shown to significantly improve with T-DM1 compared with trastuzumab (hazard ratio: 0.50; 95% CI: 0.39-0.64; P <.001) whereas distant recurrence occurred in fewer patients (10.5% vs 15.9%, respectively).19 T‑DM1 became the standard adjuvant therapy for patients with residual invasive disease following neoadjuvant taxane‑ and trastuzumab‑based treatment.
Emerging data are evaluating whether further treatment intensification may improve outcomes for selected patients, particularly those at high risk of disease recurrence. The phase III DESTINY‑Breast05 trial evaluating adjuvant trastuzumab deruxtecan (T-DXd) compared with T‑DM1 demonstrated improved 3‑year iDFS (92.4% vs 83.7%), suggesting a potential role for this agent pending longer follow‑up and careful safety assessment.20 Although not yet FDA approved, T-DXd is recommended by clinical guidelines in this setting.7