CE / CME
You’re in the Know: Laying the Foundation for Optimal Care of Complicated UTIs
Pharmacists: 1.00 contact hour (0.1 CEUs)
ABIM MOC: maximum of 1.00 Medical Knowledge MOC point
Physicians: maximum of 1.00 AMA PRA Category 1 Credit™
Nurse Practitioners/Nurses: 1.00 Nursing contact hour
Released: December 31, 2025
Expiration: December 30, 2026
Activity
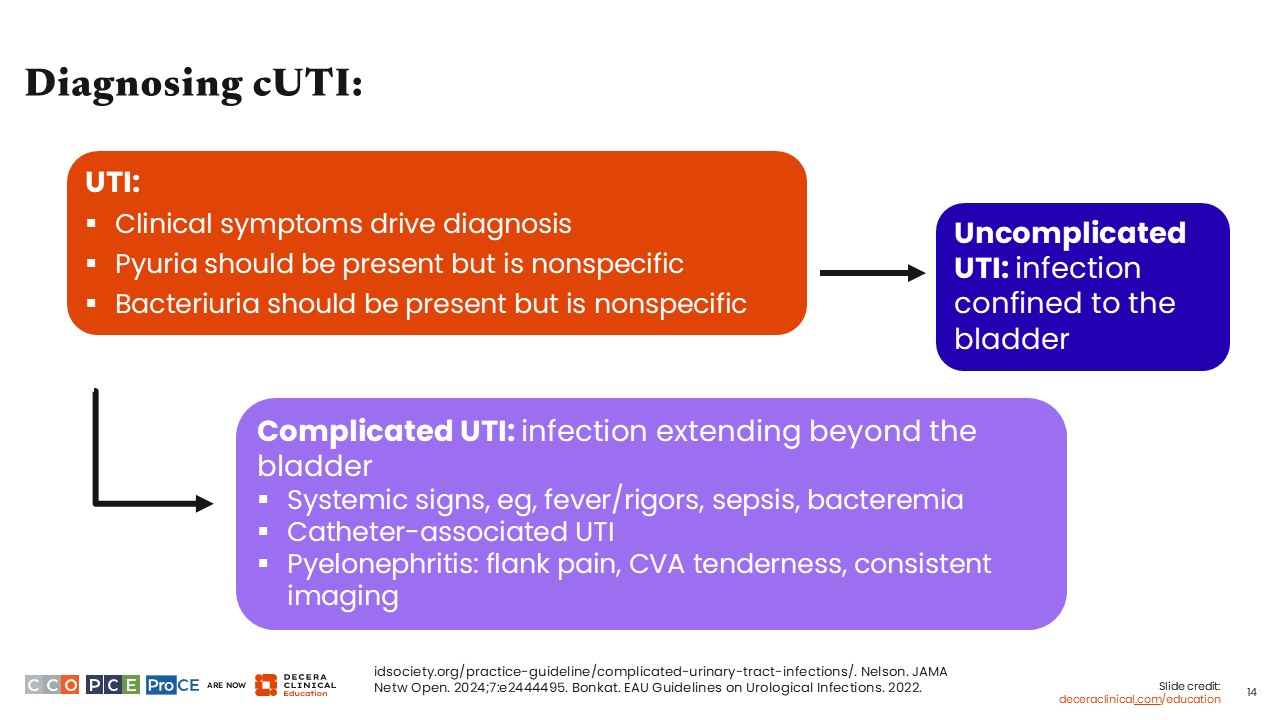
Diagnosing cUTI:
Next, we will explore strategies for the management of cUTI.
First, and perhaps of most importance, UTI is not a diagnosis that is made by looking at lab results. It is made by talking to and examining your patient. So far, no laboratory test can perfectly discriminate between infection of the urinary tract and asymptomatic bacteria. That is something that HCPs have to determine by assessing for clinical symptoms. The presence of clinical symptoms should drive the diagnosis of UTI.1,13,14
In addition to clinical symptoms, pyuria and bacteriuria should be present, but nonspecific. This is frequently seen in patients with urinary catheters and other core reasons to have urinary retention and asymptomatic bacteria.1,13,14
Once the criteria for UTI have been met, whether an individual has cUTI depends on whether there is evidence that the infection has extended beyond the bladder. That would include systemic signs of illness, such as fever, sepsis, and bacteremia. Catheter-associated UTIs are also considered complicated.1,13,14
If there is no evidence of infection beyond the bladder and no evidence of systemic infection, whether or not the patient truly has cUTI should be reconsidered.1,13,14
Finally, pyelonephritis characterized by flank pain, costovertebral angle tenderness, tenderness, or imaging concerning for renal infection would also be considered cUTI.1,13,14
Patient Case
A case study is depicted here to illustrate these concepts. Based on the details presented here, we know right away that this individual has a cUTI. He has fever and flank pain, which are likely indicative of infection extending outside the bladder.
Another factor to consider for empiric therapy is the recent exposure to a bacteria that was susceptible only to carbapenems. Carbapenems are a preferred agent for cUTI, particularly for a patient with sepsis and in whom the margin of error is lower. So, what do the guidelines tell us to do?
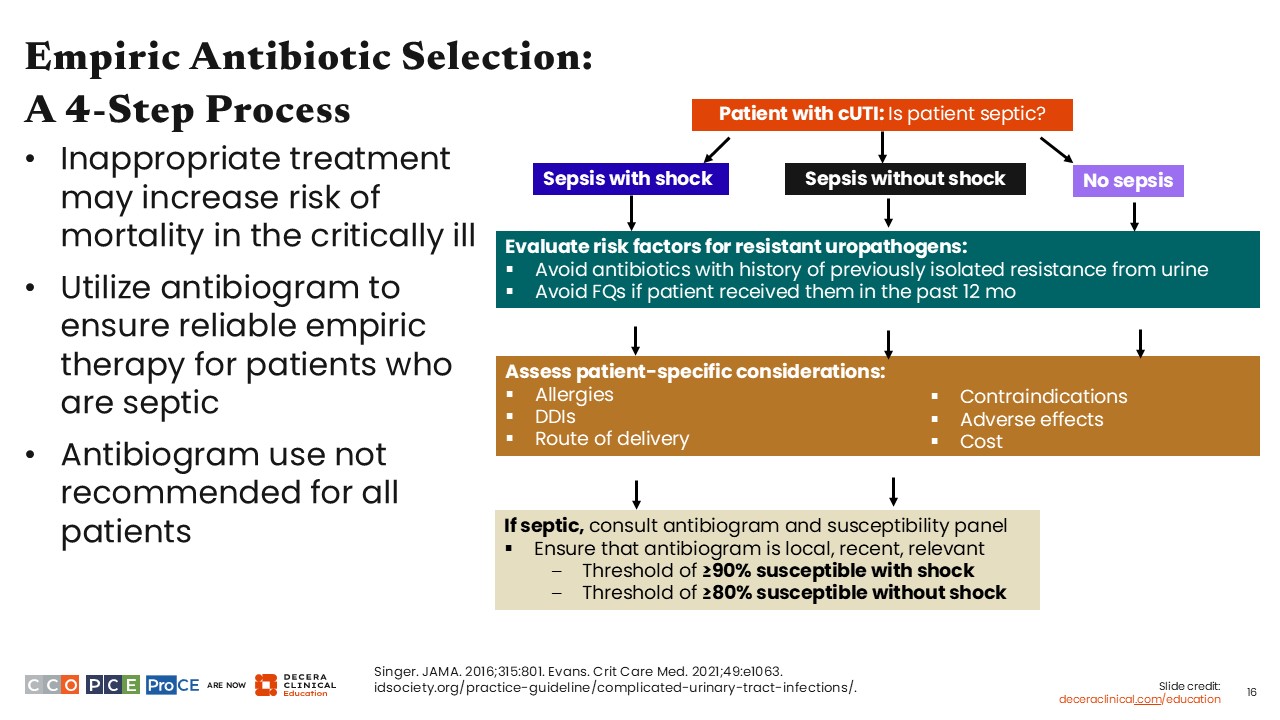
Empiric Antibiotic Selection: A 4-Step Process
The IDSA cUTI guidelines provide HCPs with a framework for how to think about choosing antibiotics for empiric therapy in cUTI.
This was done for 2 reasons. First, the randomized controlled trials used to inform these guidelines were generally noninferiority trials. That is, these recommendations were written based on studies showing that drug A is just as good as drug B. Second, we knew from experience with the prior IDSA UTI guidelines that specific drug recommendations can quickly become obsolete because of the increasing prevalence of antimicrobial resistance. Presenting recommendations as a decision workflow will allow them to remain relevant as antimicrobial resistance evolves.1,15,16
The decision tree is a 4-step process, consisting of a couple of simple questions.1
First, do my patients have sepsis? If they do have sepsis, do they have septic shock?
Second, what risk factors do my patients have for resistance to specific agents? Here, HCPs should consider prior urine cultures that have identified resistant uropathogens. In addition, the guidelines advise against fluoroquinolones if patients have received a fluoroquinolone in the past 12 months. This is based on strong evidence from multiple studies that prior exposure to fluoroquinolones predicts future resistance.
The third question is which antibiotic option would be best for my patients’ specific needs? This encompasses considerations such as drug–drug interactions, allergies, contraindications, adverse effects, route of delivery, and cost. This acknowledges that there is no one-size-fits-all treatment. HCPs must consider many other individual factors aside from simply what antibiotics are effective to identify what is the most appropriate treatment for each unique patient.
Finally, the guidelines advise consulting an antibiogram for patients with sepsis, assuming that the HCP’s facility has a local, up-to-date antibiogram that includes relevant uropathogens.1
The guidelines recommend only consulting the antibiogram for patients with sepsis, with or without shock, because the data that antibiograms actually lead to improved choice of empiric therapy are limited. In fact, there is increasing concern in the antimicrobial stewardship and infectious disease communities that overreliance on antibiograms may actually be driving overprescription and, paradoxically, promoting more antimicrobial resistance.1
However, for patients who have sepsis, the margin for error in treatment is lower. Accordingly, based on mortality modeling conducted during guideline development, we recommend that empiric therapy, guided by a local antibiogram, cover at least 80% of isolates for patients with sepsis and at least 90% of isolates for patients with septic shock.1
Approach to Empiric Antibiotic Selection: Initial Selection of Antibiotics
The guidelines provide 2 pools of antibiotics, preferred agents and alternative agents, for each of the following settings: sepsis (with or without shock), IV therapy for individuals without sepsis, and oral therapy for individuals without sepsis.
The preferred and alternative IV therapies differ for patients with and without sepsis. For patients with sepsis, carbapenems are included in the preferred category. However, carbapenems are an alternative antibiotic for patients without sepsis.1,17-20
This is because cUTI without sepsis has greater room for error in choice of empiric therapy. There is no definitive evidence that a 24-hour delay in appropriate antibiotic treatment makes a significant difference in clinical outcomes for patients without sepsis. So, in the interest of antimicrobial stewardship, we recommend using narrower-spectrum agents in these patients, like the third-generation or fourth-generation cephalosporins, piperacillin/tazobactam, or fluoroquinolones.1,17-20
For patients without sepsis, in whom oral therapy is preferred, such as patients with cUTI who do not otherwise need hospital admission, the guidelines recommend fluoroquinolones or trimethoprim/sulfamethoxazole as preferred treatment. If neither of those is an option, alternative options consist of amoxicillin/clavulanate or oral cephalosporins. The guidelines acknowledge that there are a few highly reliable options for empiric oral therapy: Fluoroquinolones and trimethoprim/sulfamethoxazole are listed as preferred in part because of the much more robust body of literature for these agents in cUTI.1,17-20
Finally, I want to highlight a caveat regarding use of fluoroquinolones as a preferred option. This recommendation is supported by clinical trial, but I want to emphasize that there are many factors that must be considered to determine whether fluoroquinolones are appropriate for one’s specific patient in one’s specific facility. Ultimately, it depends on individual risk factors and the local prevalence of fluoroquinolone resistance. There are now many areas of the world where fluoroquinolone resistance is highly prevalent, and I would certainly not advise empirical treatment with a fluoroquinolone in one of those settings.1,17-20
Approach to Selecting Definitive Therapy
The next thing to think about is moving from empiric to definitive therapy, meaning treatment selection based on culture data and antimicrobial susceptibilities. First, the guidelines recommend targeting antibiotic therapy as soon as urine and blood culture results are available, with antibiotic stewardship at the forefront of this decision.1
In this setting, I define antibiotic stewardship as the best balance between a narrow-spectrum drug, one with lower risk of promoting antimicrobial resistance, lower risk of toxicity and drug–drug interactions, lower cost, and ideally an oral route of administration, to avoid the potential complications and cost of prolonged IV antibiotics.
With a multidrug-resistant infection, it is always reasonable to consult an infectious disease specialist, particularly if the HCP is not experienced or comfortable with antibiotic selection for definitive therapy. A multidrug-resistant infection does not necessarily affect duration of therapy or the route of administration, but it will certainly affect what the optimal choice of therapy.1,20
In terms of when to transition to oral therapy, the criteria set by the guidelines are simple: Patients should be able to take oral therapy and should be clinically improving. They need to be either afebrile or headed toward afebrile, hemodynamically stable, with any urgent procedures needed to address the source of infection already performed.1,20-22
Having bacteremia is not itself a contraindication for switching into oral antibiotics, as long as there is clinical improvement. However, one specific situation in which I would hesitate to transition to oral antibiotics is in patients who have had extensive small bowel resections. There are limited data, even for highly bioavailable oral antibiotics, as to what drug level is actually being achieved in these patients.1,20-22
Finally, if patients are not clinically improving after several days of antibiotics, the answer is not simply extending the duration of therapy. Rather, the answer should be further workup to determine the root cause of the lack of improvement.1,20-22
Duration of Therapy
Essentially, for most patients with cUTI, with or without bacteremia, the guidelines recommend 7 days of therapy, as long as they are clinically improving. The data support as little as 5 days of therapy, specifically when using a fluoroquinolone.1,2
However, some populations may need an individualized duration of therapy, such as men with febrile UTI or other symptoms that suggest acute bacterial prostatitis. In this specific subpopulation, longer course of therapy, between 10 and 14 days, may be more effective.1,2
What If the Patient Is Not Clinically Improving?
What about patients who are not improving after several days of antibiotics? What evaluation should be done to determine the cause, and what needs to be done to address it?
There are 3 things HCPs should consider. First is whether or not the patients are truly receiving optimal antimicrobial therapy. Is the antibiotic active against their uropathogen? Is the dose appropriate to reach therapeutic levels in urine and tissue and blood?1
Second is reexamining the source control. As an infectious diseases HCP consulted on patients with cUTI who are not improving on therapy, what I probably encounter most often is an issue with source control. A key question I would ask is whether this patient could have a urinary obstruction that needs to be relieved. An obstruction could be why antibiotics alone are not controlling the infection. HCPs should also determine if there are perinephric or renal abscesses that need to be drained, and they should ensure that imaging has been done to identify any other source of infection in that area.1
Third, if a patient is not clinically improving with treatment, HCPs should reassess the cUTI diagnosis entirely—perhaps something else is going on. This is particularly something I consider when a patient has systemic symptoms without urinary symptoms, with pyuria or bacteriuria. In those cases, it is worth asking whether the source of infection could be outside of the urinary tract.1
Similarly, if patients’ urinary symptoms improve, but they still have a fever, there may be a noninfectious cause, such as a drug fever. For example, β-lactams are some of the most common drugs used to treat cUTI and are also some of the drugs most likely to cause drug fever.1
Patient Case: Discussion
Let’s continue our case. The patient described earlier was admitted to the hospital. We previously categorized this patient as having a cUTI. The infectious disease team was consulted, and IV meropenem was initiated because of his previous history of carbapenem-resistant uropathogens.
On Day 3 of therapy, his symptoms improved and he was afebrile. His urine culture grew an isolate of Pseudomonas aeruginosa that was resistant to fluoroquinolones but susceptible to cefepime and meropenem.
Overall, this patient has demonstrated an early response to therapy. He is now afebrile, with clinical symptom improvement. Ideally, the next step would be to switch him to an oral antibiotic. Unfortunately, his isolate is resistant to fluoroquinolones, which are essentially the only effective oral option for Pseudomonas aeruginosa is the only kind of active oral option. As a result, he must be discharged on IV therapy.
At this point, there are 2 options: cefepime or meropenem. In this case, I would de-escalate him from meropenem to cefepime, as cefepime is a little more narrow in spectrum, a little more targeted, and of importance, a bit more robust against development of antimicrobial resistance in Pseudomonas compared with meropenem.
Many see meropenem as the stronger drug or the “bigger gun” because it has a broader spectrum of activity. However, Pseudomonas can very rapidly develop resistance to meropenem via efflux pumps and porin mutations. As such, in this situation, cefepime is the more reliable agent.
Given the patient’s early clinical improvement, there is no concern for uncontrolled infection, making a 7-day duration of therapy more appropriate than a longer duration.
Newer Agents for cUTI
Of course, it would be ideal to have other oral options for Pseudomonas and other multidrug-resistant organisms. Fortunately, these drugs are certainly in the pipeline. During the past 10-20 years, there has been something of a renaissance in β-lactam–β-lactamase inhibitor development. There are numerous new antibiotics with activity against ESBL-producing organisms, AmpC-β-lactamase–producing organisms, and even carbapenemase-producing organisms.
In fact, even more β-lactam–β-lactamase inhibitor combinations, additional carbapenems, and other novel agents are currently in the pipeline, and we hope to have many of these available for clinical use in the near future.