CE / CME
Emerging Data and Clinical Implications in Nonmalignant Hematologic Disorders: ASH 2025 Highlights
Physician Assistants/Physician Associates: 1.00 AAPA Category 1 CME credit
Pharmacists: 1.00 contact hour (0.1 CEUs)
Physicians: maximum of 1.00 AMA PRA Category 1 Credit™
Nurse Practitioners/Nurses: 1.00 Nursing contact hour
Released: February 20, 2026
Expiration: August 19, 2026
Activity
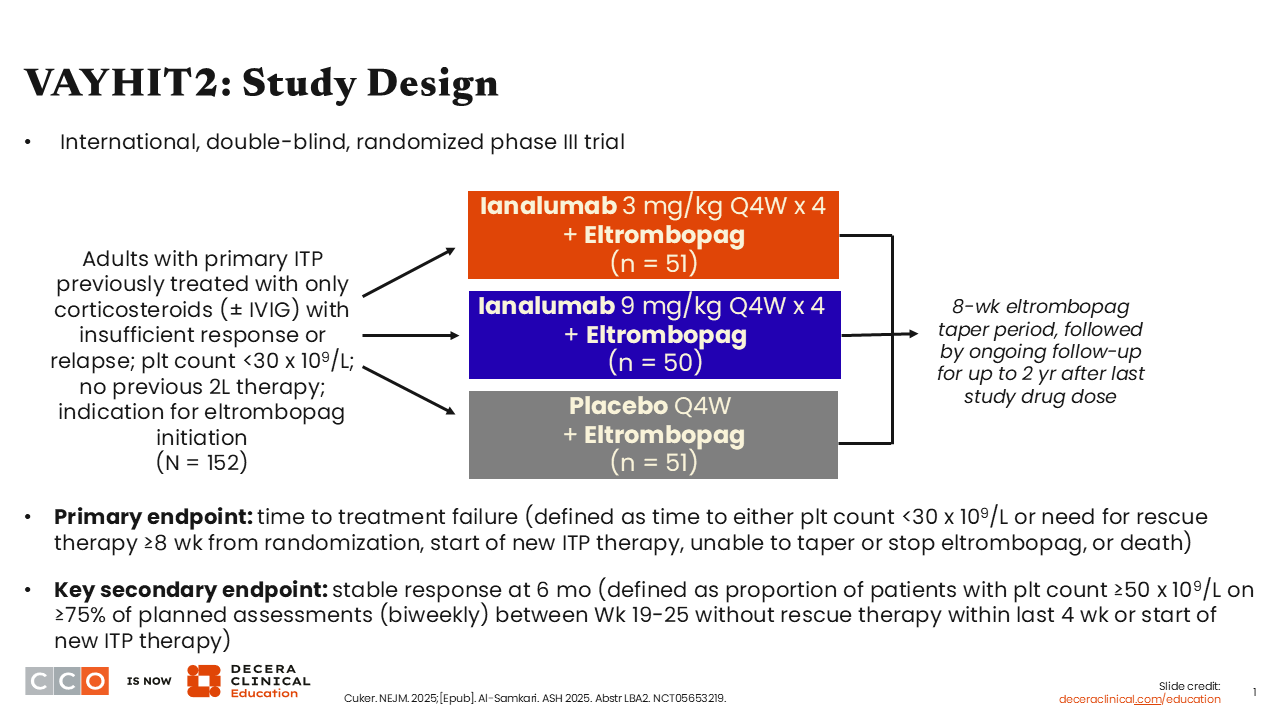
VAYHIT2: Study Design
Hanny Al-Samkari, MD:
VAYHIT2 is a pivotal randomized controlled phase III trial of ianalumab, a novel antibody that binds to the BAFF receptor, plus eltrombopag vs placebo plus eltrombopag in patients with previously treated ITP who received only 1 previous line of therapy consisting of corticosteroids with or without intravenous immunoglobulin (IVIG). Ianalumab works via a dual mechanism, acting as both a very potent depletor of B-cells and preventing signaling through the BAFF pathway, which is important in preventing the maturation and development of B-cells and ultimately their differentiation into long-lived plasma cells that likely drive chronic, refractory ITP.
Different than most ITP studies, VAYHIT2 was designed to intervene early in the disease course with the goal of giving patients a long period off therapy and possibly modifying the disease course. There were 3 study arms: high-dose ianalumab (9 mg/kg) plus eltrombopag, low-dose ianalumab (3 mg/kg) plus eltrombopag, and placebo plus eltrombopag. The primary endpoint was time to treatment failure.1,2
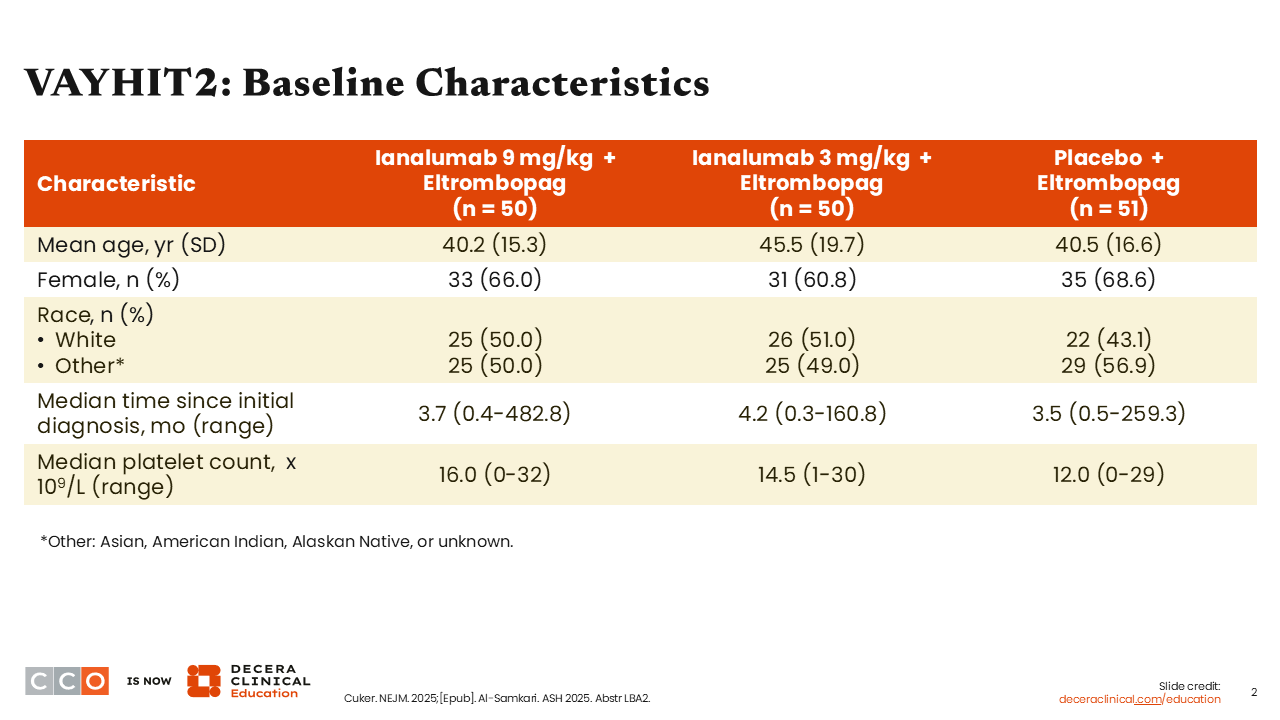
VAYHIT2: Baseline Characteristics
Hanny Al-Samkari, MD:
Because of the study design, the majority of patients were younger than those in most ITP studies. The average time since diagnosis across all arms was approximately 3-4 months.1,2
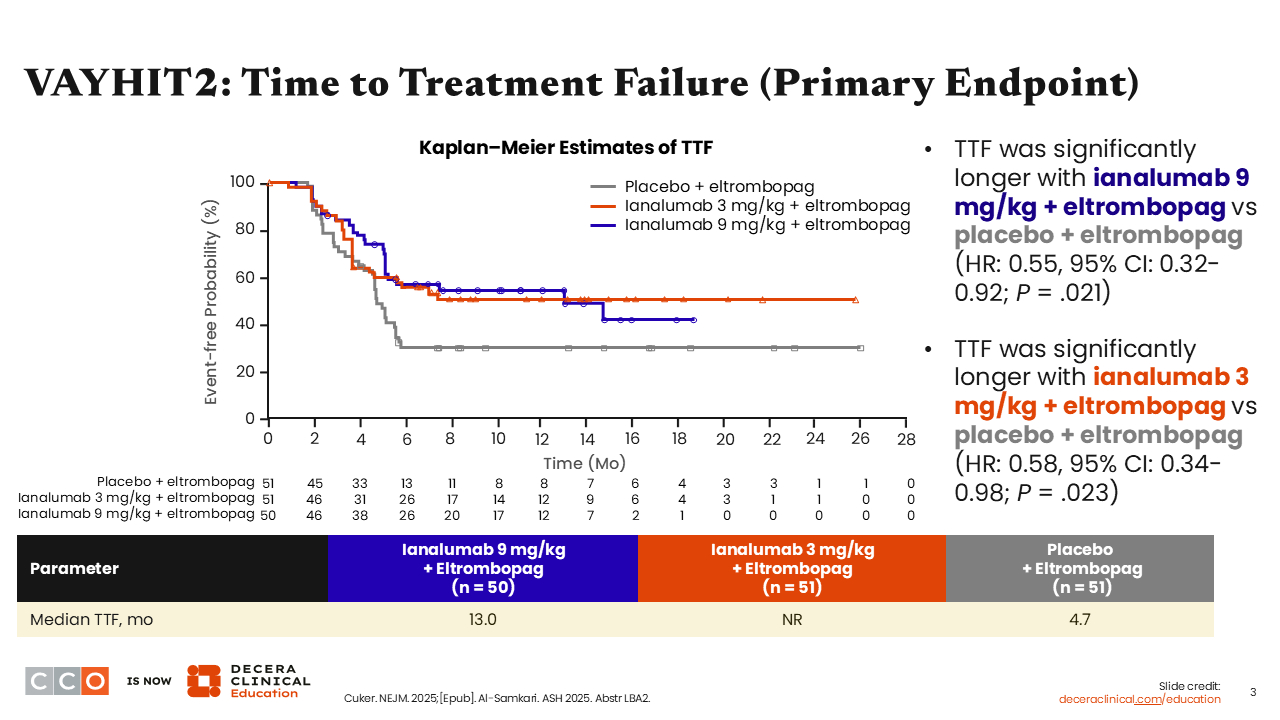
VAYHIT2: Time to Treatment Failure (Primary Endpoint)
Hanny Al-Samkari, MD:
Median time to treatment failure—the primary endpoint—was significantly longer with both low-dose (not reached [NR]) and high-dose (13.0 months) ianalumab vs placebo (4.7 months). It is important to keep in mind that every patient received eltrombopag and was required to taper off eltrombopag by Month 6. Patients receiving ianalumab had both clinically significant and statistically significant longer time to treatment failure than those receiving placebo (ianalumab 3 mg/kg vs placebo: P = .023; ianalumab 9 mg/kg vs placebo: P = .021). Here, treatment failure was defined as a platelet count <30,000, need for rescue therapy at least 8 weeks after randomization, inability to taper or discontinue eltrombopag, start of an alternative ITP therapy at any time, or death.1,2
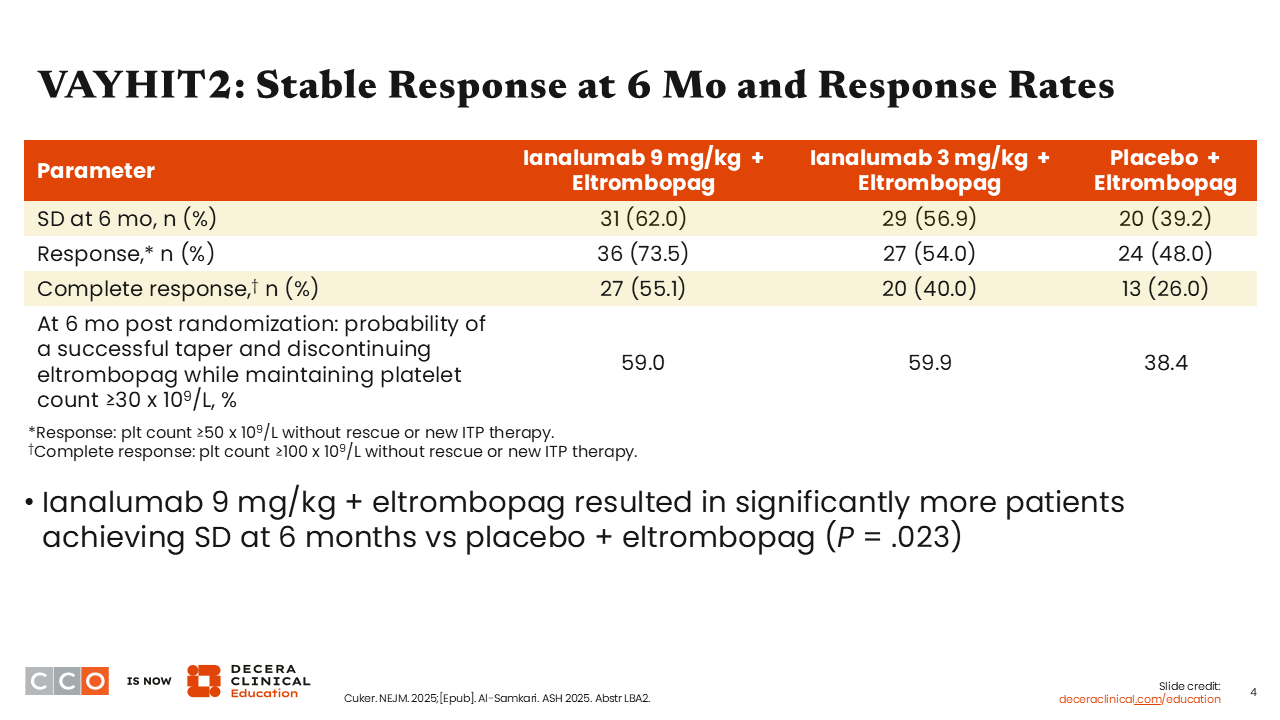
VAYHIT2: Stable Response at 6 Mo and Response Rates
Hanny Al-Samkari, MD:
More patients in the ianalumab arms (approximately 59% to 60%) maintained platelet counts ≥30,000 than those in the placebo arm (38.4%). The ianalumab 9 mg/kg arm had the highest rate of stable response at 6 months (62.0%), the highest rate of response (73.5%), and the highest rate of complete response (55.1%).1,2
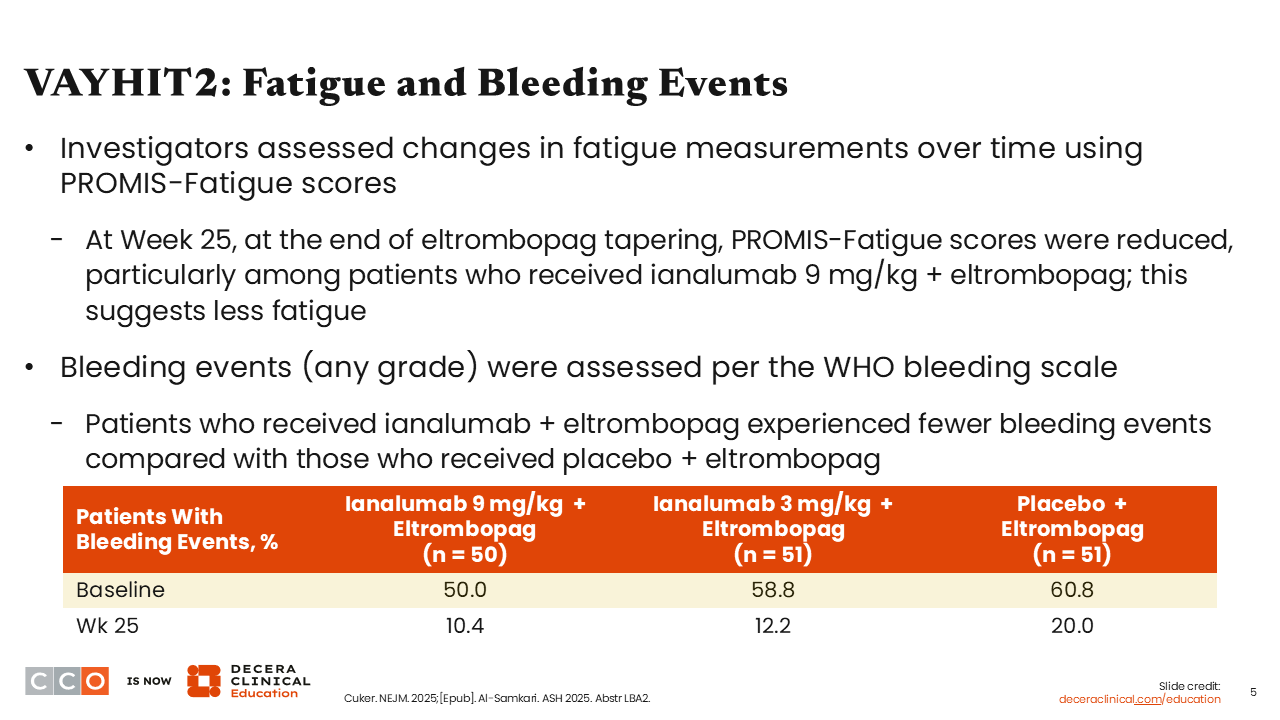
VAYHIT2: Fatigue and Bleeding Events
Hanny Al-Samkari, MD:
Treatment with ianalumab improved fatigue and reduced bleeding events. It is important to recognize that constitutional symptoms of ITP, like fatigue, are very problematic for patients. Patients in the ianalumab 9 mg/kg arm experienced the greatest improvements in fatigue and lowest rates of bleeding events at Week 25 (10.4%).1,2
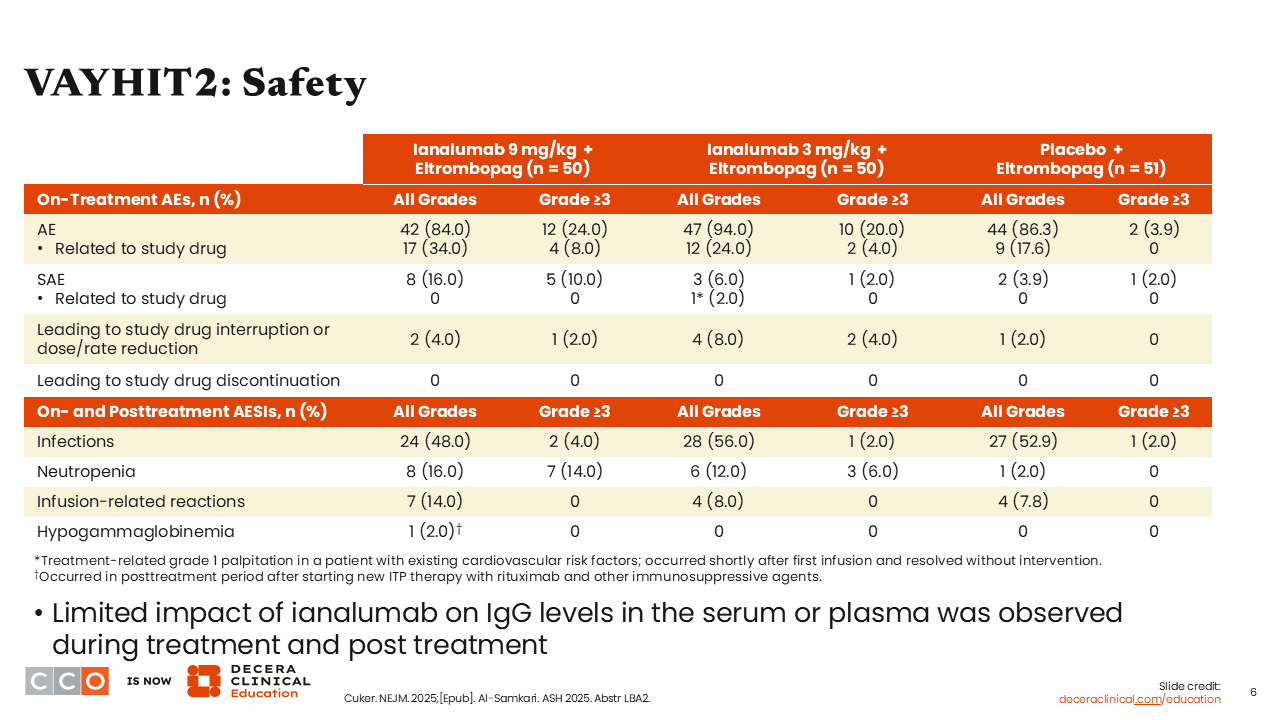
VAYHIT2: Safety
Hanny Al-Samkari, MD:
Ianalumab was administered as 4 once-monthly infusions. The biggest concern with use of a drug with this mechanism of action is infection. Of importance, there was no increase in either the frequency or severity of infections in patients receiving ianalumab vs placebo. There was a higher incidence of neutropenia, but it was transient, lasting a few days to 1 or 2 weeks. The difference was not clinically relevant or clinically significant and did not result in a higher incidence of infections.1,2
VAYHIT2: Summary
Hanny Al-Samkari, MD:
The investigators concluded that combination therapy with ianalumab plus standard-of-care eltrombopag early in the course of ITP, particularly in the second-line setting, was effective at giving patients a prolonged duration of time off therapy without any significant increase in adverse events (AEs). The main question remaining to be answered is whether this addition early in the disease course is disease modifying. VAYHIT2 is well designed to answer this question.
Joseph J. Shatzel, MD, MCR:
I agree. The study design is innovative because each arm is receiving treatment, and it’s nice to see a doublet being investigated in ITP. I was also pleased to see the improvement in fatigue because patients with this autoimmune disease often experience fatigue and related symptoms.
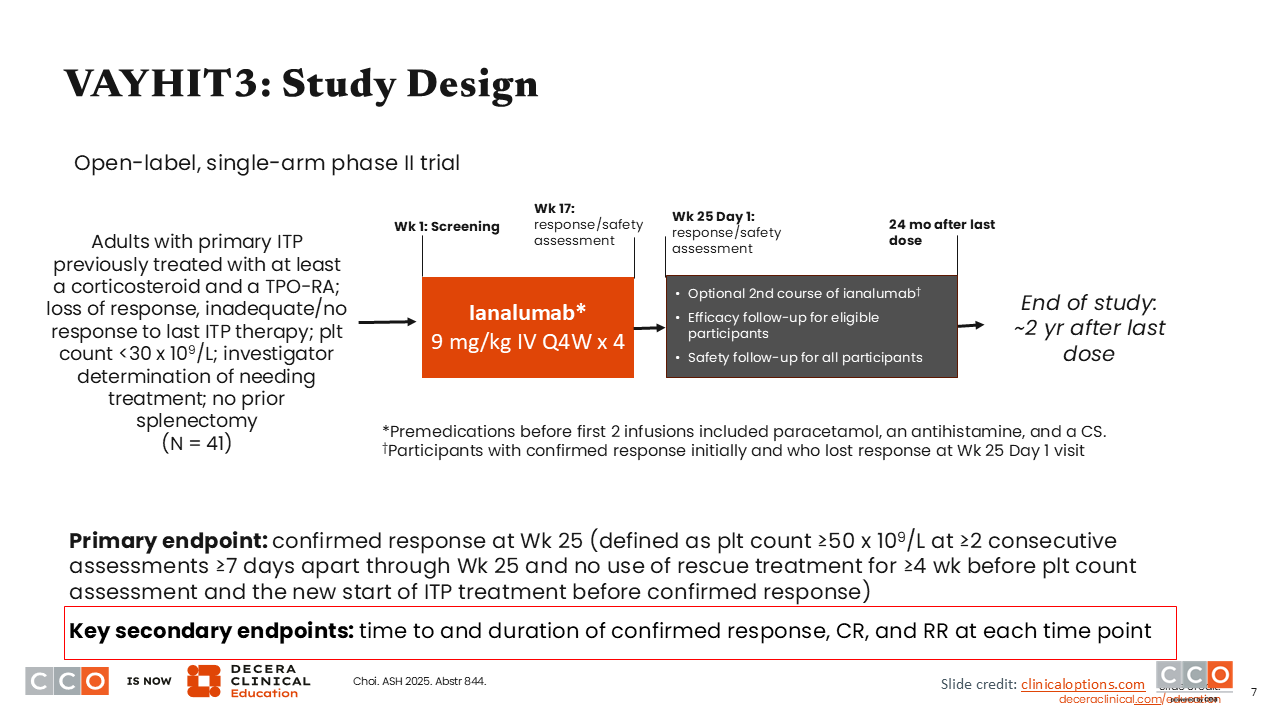
VAYHIT3: Study Design
Hanny Al-Samkari, MD:
The phase II VAYHIT3 trial is looking at ianalumab as a single agent in patients with more heavily pretreated primary ITP. This study enrolled 41 patients who had previously been treated with at least corticosteroid and a thrombopoietin receptor agonist (TPO-RA), which is typical of patients with ITP enrolled in clinical trials. Participants received 4 doses of ianalumab 9 mg/kg intravenously every 4 weeks. The primary endpoint was confirmed response at Week 25.3
VAYHIT3: Baseline Characteristics
Hanny Al-Samkari, MD:
The patients enrolled in VAYHIT3 were older than those enrolled in VAYHIT2 because they had relapsed or been refractory to prior treatment, with the median number of prior lines of therapy being 6.3
VAYHIT3: Efficacy Summary (Primary Analysis)
Hanny Al-Samkari, MD:
The results were positive, with 44% of patients having a confirmed response at Week 25. The median time to confirmed response in this population was approximately 1 month. In total, 90% of patients achieved complete response.3
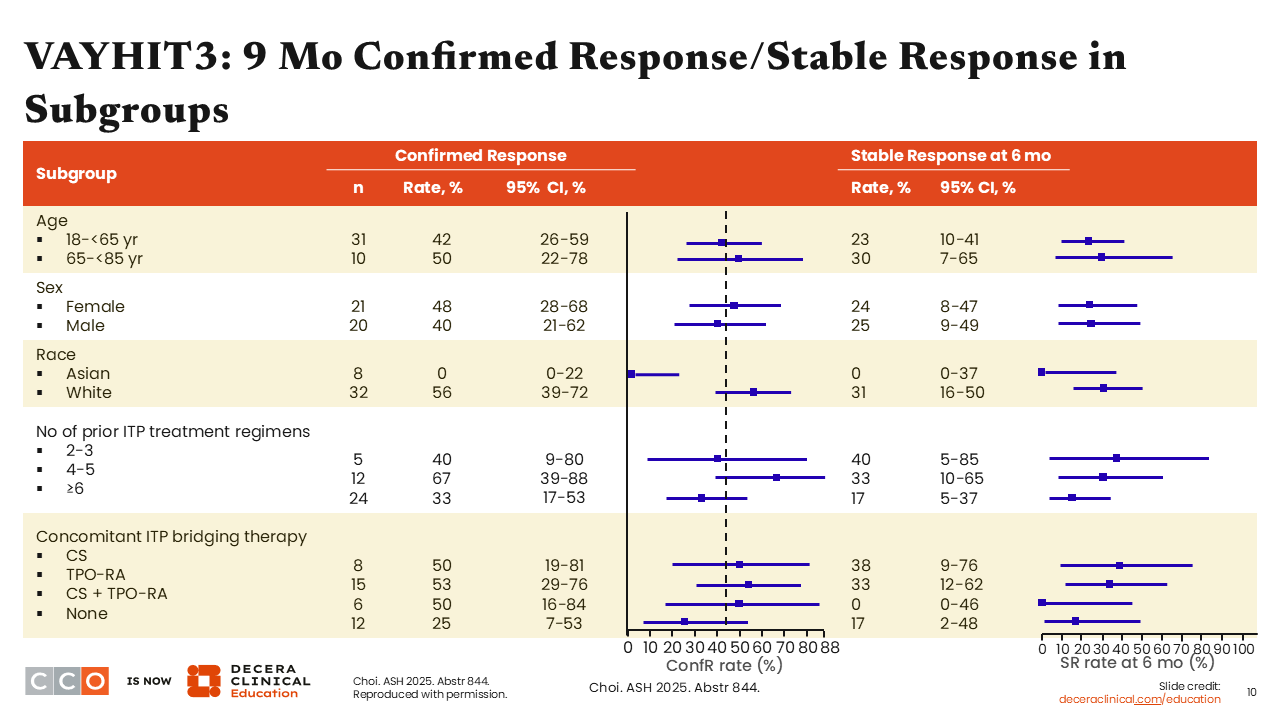
VAYHIT3: 9 Mo Confirmed Response/Stable Response in Subgroups
Hanny Al-Samkari, MD:
The investigators wanted to determine whether there were any predictors of response. As shown on this slide, the stable response rate in Asian patients was low but was an artifact of the data, and very specific circumstances led to this result. We do not expect this agent to be ineffective in Asian patients, because in VAYHIT2, nearly half of the patients on study were Asian, and they responded well to ianalumab. There were no obvious differences in subgroups regarding response.3
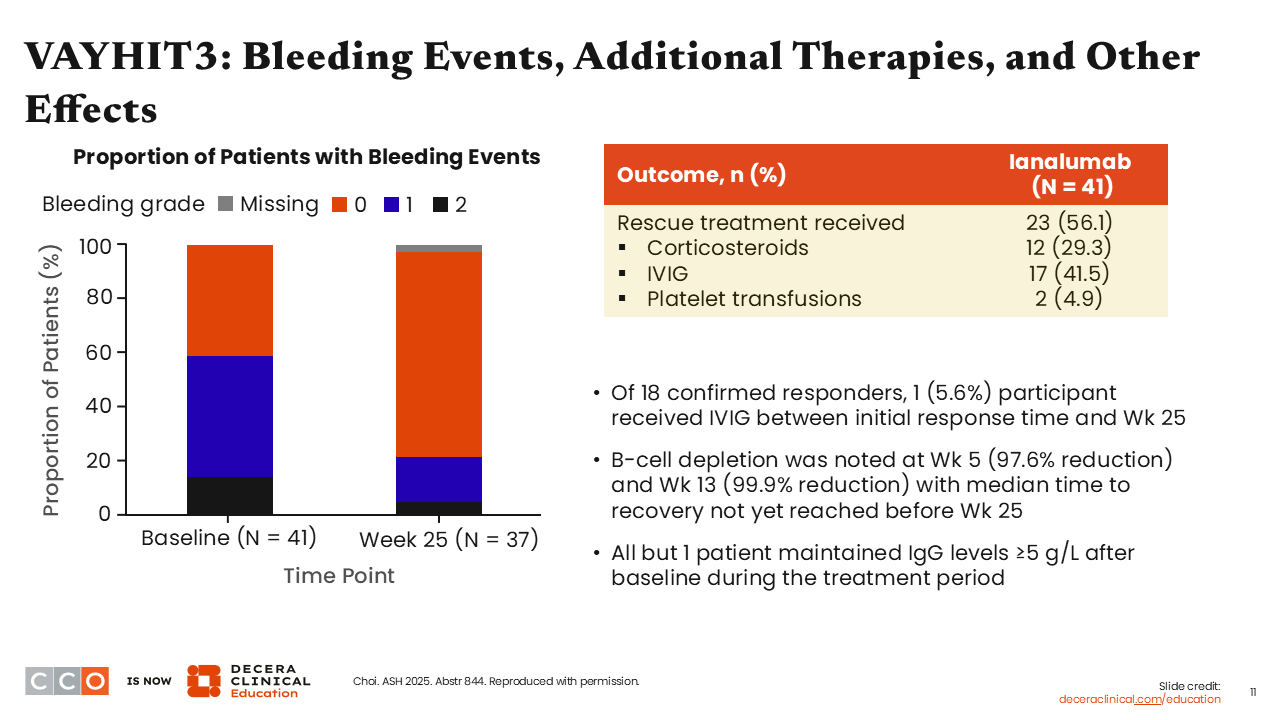
VAYHIT3: Bleeding Events, Additional Therapies, and Other Effects
Hanny Al-Samkari, MD:
The proportion of patients with bleeding events was lower at Week 25 after receipt of ianalumab than at baseline. The overall concomitant therapy burden was also lower at Week 25. The rate of B-cell depletion was profound: 99.9% by Week 13, keeping with the expectation for this kind of drug. Despite the fact that there was significant B-cell depletion, as was also shown in VAYHIT2, there was not a drastic drop in immunoglobulin levels. IgG concentrations remained ≥5 g/L in nearly all patients.3
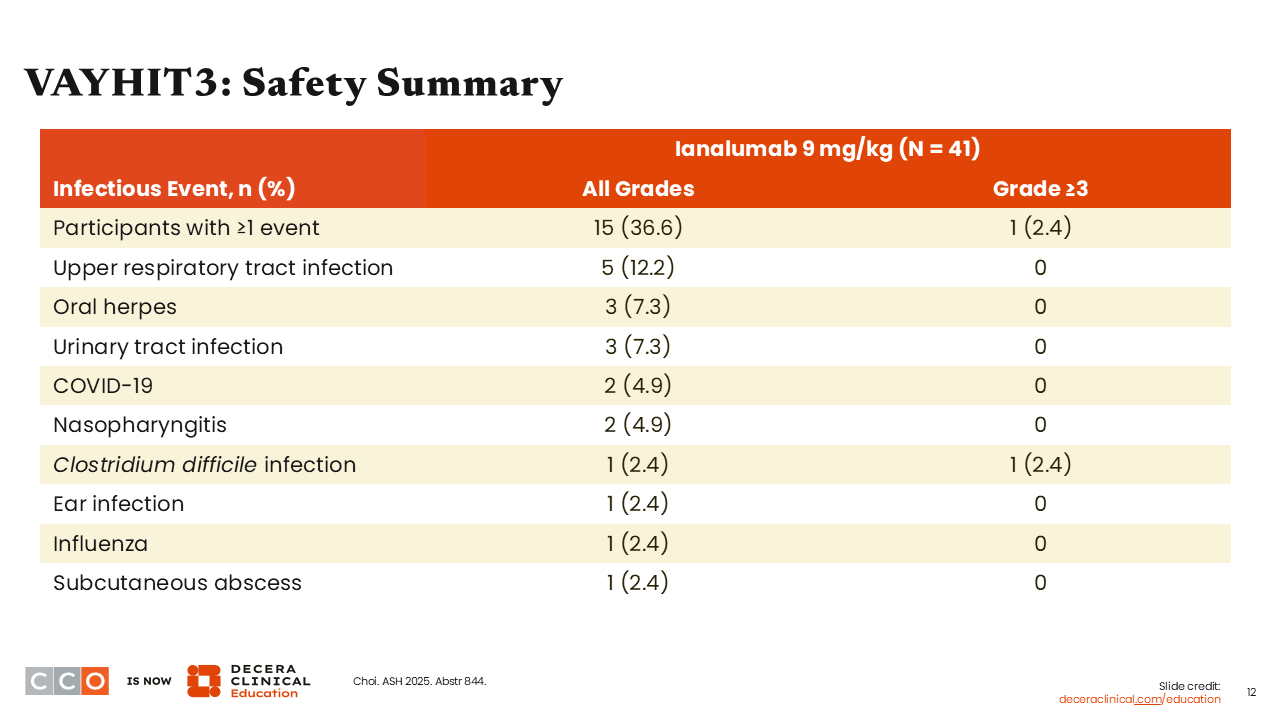
VAYHIT3: Safety Summary
Hanny Al-Samkari, MD:
Overall, the safety profile of ianalumab in VAYHIT3 was relatively similar to that in VAYHIT2, showing that ianalumab was well tolerated.3
VAYHIT3: Summary
Hanny Al-Samkari, MD:
Ultimately, this study, which is a counterpart to VAYHIT2, showed that ianalumab is also effective in more heavily pretreated patients, reduced bleeding, resulted in profound, deep, and rapid B-cell depletion, but did not dramatically impact IgG levels. From a clinical standpoint, these results suggest that prior responses to vaccinations will be preserved. One outstanding question however is how patients will respond to vaccination after having previously received ianalumab.
Joseph J. Shatzel, MD, MCR:
I was very impressed with these data because it is not uncommon to see patients with refractory, heavily pretreated ITP in the clinic. The current treatment landscape includes fostamatinib, which has a very low response rate, so patients need a different thrombopoietin mimetic for a response but have often already been treated with one. Patients can be offered surgery to remove their spleen, but this approach is not ideal.
The key result here was the high response rate in a heavily pretreated population. If these results are confirmed and ianalumab is approved for use, I would be excited to use this agent, particularly because it seems safe, but I would want to know exactly what treatment(s) my patient had already received.
Hanny Al-Samkari, MD:
I agree completely. In thinking about how these data are going to change clinical practice, I believe they will allow more options in the later-line setting and possibly change the way we treat ITP early on to minimize the number of patients who require long-term chronic therapy. In the later-line setting, shared decision-making will be very important, because there is not and will likely never be a study comparing rilzabrutinib to ianalumab. Some patients will value treatment-free disease control above all else, and ianalumab would be ideal for them. Others will value platelet count stability while taking a medication, and rilzabrutinib may be the better choice for them. Both of these agents appear to improve fatigue, which is an improvement over older therapies.