CE / CME
Mitigating and Managing AEs Associated With Newer Agents for Relapsed ES-SCLC
Physician Assistants/Physician Associates: 0.25 AAPA Category 1 CME credit
Pharmacists: 0.25 contact hour (0.025 CEUs)
ABIM MOC: maximum of 0.25 Medical Knowledge MOC point
Physicians: maximum of 0.25 AMA PRA Category 1 Credit™
Nurse Practitioners/Nurses: 0.25 Nursing contact hour
Released: December 02, 2025
Expiration: June 01, 2026
Activity
Considerations for Managing AEs Associated With Newer Agents Used to Treat SCLC
Recent therapeutic advances have significantly reshaped the management of SCLC, including in the challenging extensive-stage setting. Most notably, tarlatamab (a DLL3-targeting bispecific T‑cell engager), lurbinectedin (an alkylating transcription inhibitor), and novel immunotherapy combinations have demonstrated significant improvements in overall survival (OS) for patients with SCLC. However, as with all cancer treatments, these novel therapies are associated with distinct and potentially serious AEs. Understanding and proactively managing these AEs is essential to optimizing patient outcomes and ensuring safe, effective use of these innovative agents.
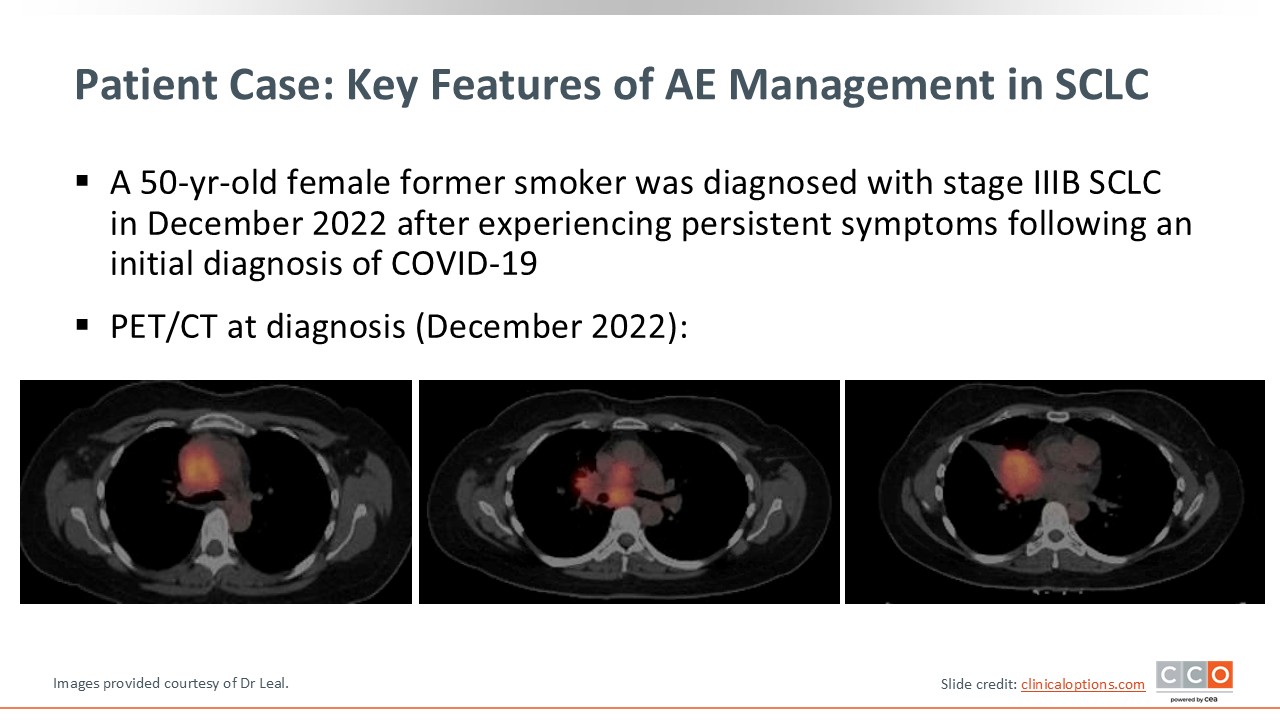
Patient Case: Key Features of AE Management in SCLC
Let’s start by reviewing a patient case example that details some of the key features of AE management associated with a recently approved targeted therapy. A 50-year-old female former smoker was diagnosed with stage IIIB SCLC in December 2022 after experiencing persistent respiratory symptoms following an initial diagnosis of COVID-19. The PET/CT scan at diagnosis demonstrated bulky mediastinal and hilar adenopathy, as well as a right middle lobe mass leading to lung collapse.
Patient Case: Clinical Course
Because the patient’s disease was stage IIIB, she underwent chemoradiation with cisplatin/etoposide but experienced side effects from the cisplatin, which included worsening fatigue, neutropenia, nausea, and vomiting. She switched to carboplatin and completed the chemotherapy course in February 2023.
A posttreatment PET scan was notable for a partial response, but there was decreased residual activity in all treated disease sites in June 2023. Four months later in October 2023, she demonstrated evidence of progression on an updated PET scan, with new sites of disease with right supraclavicular lymph node involvement, and a right flank soft tissue mass. She had stable right paratracheal, subcarinal, and right perihilar masses, although increasing PET avidity was noted.
This patient was treated in early 2023, prior to the widespread adoption of maintenance immunotherapy or availability of combination strategies with lurbinectedin. Her disease course may have differed had these options been available, as trial data now support improved progression-free survival (PFS) and OS with these newer approaches, which might have offered more durable disease control following her initial response.
Additional information on treatment options for limited-stage SCLC, first-line treatment for ES-SCLC, and treatment selection for ES-SCLC after progression can be found here.
Patient Case: Second-line Therapy Selection
At progression after first-line therapy, the patient’s main symptoms included decreased appetite, weight loss, weakness, shortness of breath, and cough. After a discussion about her disease progression and symptoms, the decision was made to proceed with tarlatamab.
Given her high burden of symptoms, high burden of disease, and decline in ECOG PS to 2, inpatient tarlatamab administration with 24-hour monitoring was recommended for the initial step-up dosing.
Patient Case: Inpatient Monitoring of CRS and ICANS With Tarlatamab
The patient was admitted for cycle 1 Day 1 dosing of tarlatamab at 1 mg IV. Overnight, she developed grade 1 CRS, which consisted of fever with sinus tachycardia (heart rate in the 130s). CRS was managed with acetaminophen and a 500 cc bolus of normal saline, with clinical improvement.
She also developed grade 2 immune effector cell–associated neurotoxicity syndrome (ICANS), with an Immune Effector Cell Encephalopathy (ICE) score of 4 out of 10, after having been 10 out of 10 at baseline. She received dexamethasone 10 mg IV every 6 hours per institutional protocol.
By morning, her ICE score had improved to 9/10, but she experienced an additional episode of fever and tachycardia, that was managed with acetaminophen, dexamethasone, and IV fluids. She was subsequently discharged within 48 hours with her caregiver, feeling improved, with fever resolution.
Patient Case: Step-Up Dosing With Tarlatamab
She returned for cycle1 Day 8 administration of tarlatamab at 10 mg IV, using the approved step-up dosing strategy to minimize AEs. Following cycle 1 Day 8 treatment, overnight, she developed grade 1 CRS with fever that was treated with acetaminophen and dexamethasone, resulting in improvement. She also had grade 1 ICANS at midnight with an ICE score of 9 out of 10, that was back to normal (10 out of 10) the following morning. She was discharged home with her caregiver within 24 hours, feeling improved and without fever.
Patient Case: Outpatient Monitoring of CRS and ICANS
Given that she had done well with grade 1 CRS and ICANS after Day 8 administration, for cycle 1 Day 15, she received tarlatamab 10 mg and was monitored for 6 hours in an outpatient urgent care setting, per institutional protocol. Of note, she reported a decrease in size of visible masses in the subcutaneous areas of her right leg, left neck, and right lower ribs. She also noted clinical improvement, with reduction in cough and shortness of breath upon exertion since starting tarlatamab.
She had no significant complications with the cycle 1 Day 15 dose of tarlatamab and was discharged home after 6 hours of monitoring with her caregiver.
After 2 cycles of tarlatamab, she demonstrated significant clinical benefit, and CT scans showed significant improvement in her disease. Of importance, neither CRS nor ICANS recurred after cycle 2 Day 1.
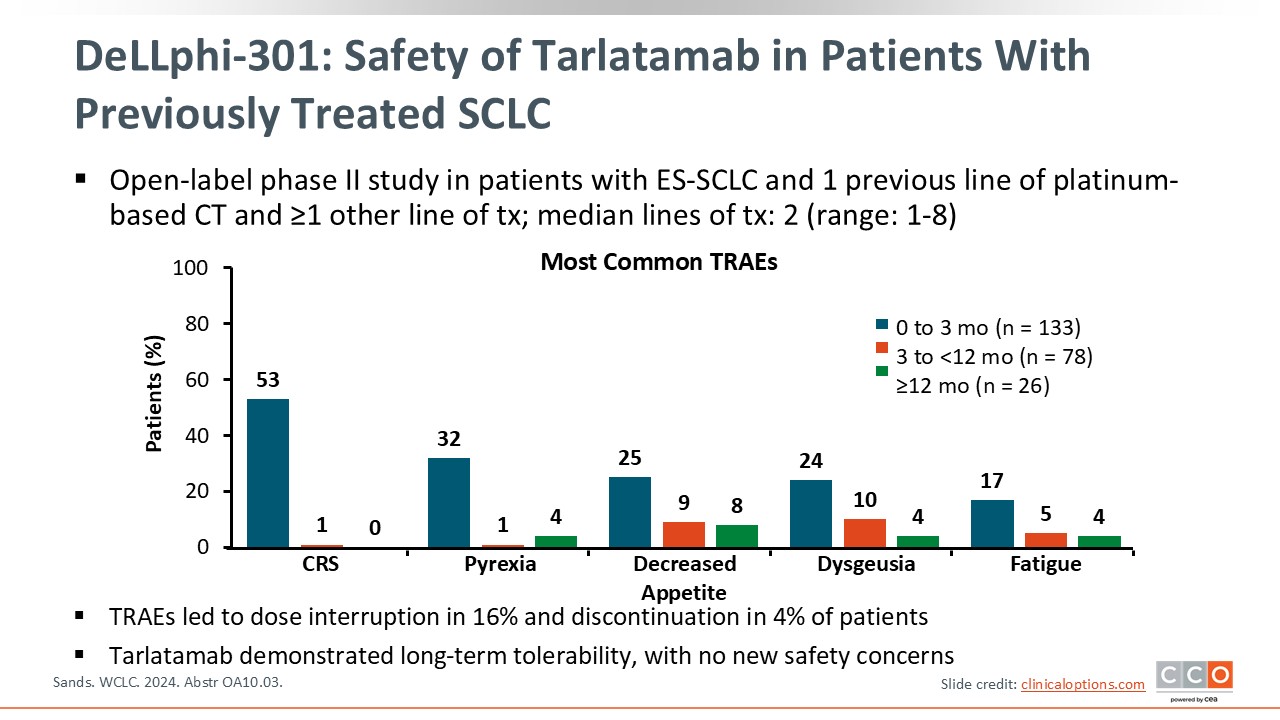
DeLLphi-301: Safety of Tarlatamab in Patients With Previously Treated SCLC
Tarlatamab is a DLL3-targeting T-cell engager approved for ES-SCLC with disease progression on or after platinum-based chemotherapy.1 The phase II DeLLphi-301 study of tarlatamab in patients with previously treated ES-SCLC showed strong clinical benefit with regards to the overall response rate (ORR) as well as durable responses.2 In DeLLphi-301, the most common treatment-related adverse events (TRAEs) included CRS, pyrexia, decreased appetite, dysgeusia, and fatigue.
When assessing the rate of TRAEs that occurred within 3 months, between 3 and 12 months, and after at least 12 months, the majority of AEs occurred early in the treatment course, especially CRS, which occurred mostly within the first 2 doses.3
TRAEs led to dose interruption in 16% of patients and discontinuation in only 4% of patients, demonstrating the long-term tolerability of tarlatamab in patients with ES-SCLC.
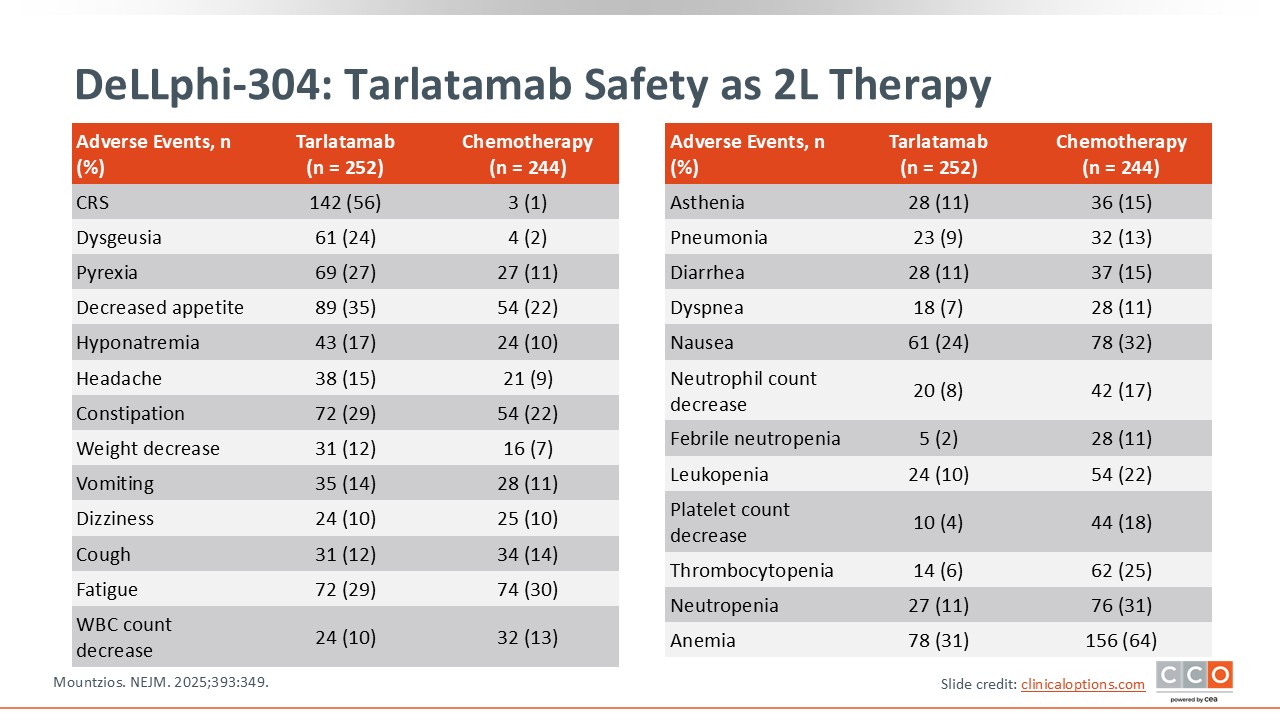
DeLLphi-304: Tarlatamab Safety as 2L Therapy
The confirmatory phase III DeLLphi-304 trial compared tarlatamab vs standard of care therapy with topotecan, lurbinectedin, or amrubicin in patients with ES-SCLC who had disease progression after 1 platinum-based regimen. This randomized trial found that tarlatamab improved OS as second-line therapy vs chemotherapy.4
The safety profile was similar to that observed in DeLLphi-301. The most common grade 3 or greater TRAEs were cytopenias, hyponatremia, and fatigue. CRS was observed in 56% of patients and was primarily grade 1 or 2. ICANs was seen in 6% of patients. The rate of discontinuation due to AEs was 5% in the in the tarlatamab arm.
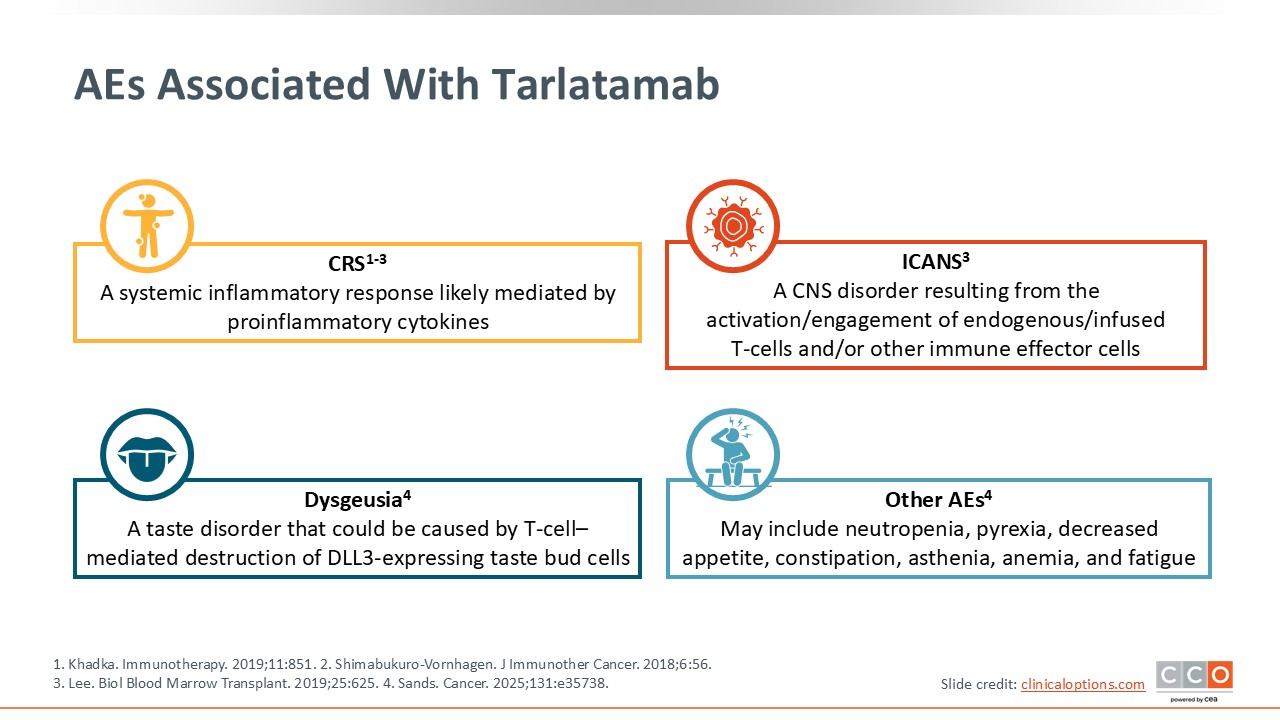
AEs Associated With Tarlatamab
Shown here are some of the AEs associated with tarlatamab. CRS is a systemic inflammatory response mediated by proinflammatory cytokines.5,6 T-cell engagers such as tarlatamab trigger the release of proinflammatory cytokines, which then leads to immune-mediated AEs, including CRS. ICANS is a disorder of the CNS that results from the activation or engagement of endogenous T-cells and other immune effector cells.7 Both CRS and ICANS are potentially serious complications and appropriate monitoring and management are needed.
Dysgeusia is a taste disorder that may be caused by T-cell–mediated destruction of DLL3-expressing taste bud cells. Other important AEs that are observed with tarlatamab include pyrexia, neutropenia, decreased appetite, anemia, constipation, asthenia, and fatigue.2,8
Clinical Presentation of CRS
Of importance, the presenting and often defining symptom of CRS is fever. Fever related to CRS can be high grade and last for multiple days. Other associated symptoms include hypoxia and hypotension.7,9
Tachycardia, tachypnea, and headache may also be noted, and these symptoms can escalate with potentially life-threatening complications such as cardiac dysfunction, acute respiratory distress syndrome, and renal or hepatic dysfunction.6,7 However, rapid identification and management of CRS can mitigate its life-threatening complications. As previously stated, CRS is largely confined to the first administration of tarlatamab during step-up dosing, and the median time to CRS occurrence is typically 4-16 hours after infusion.8
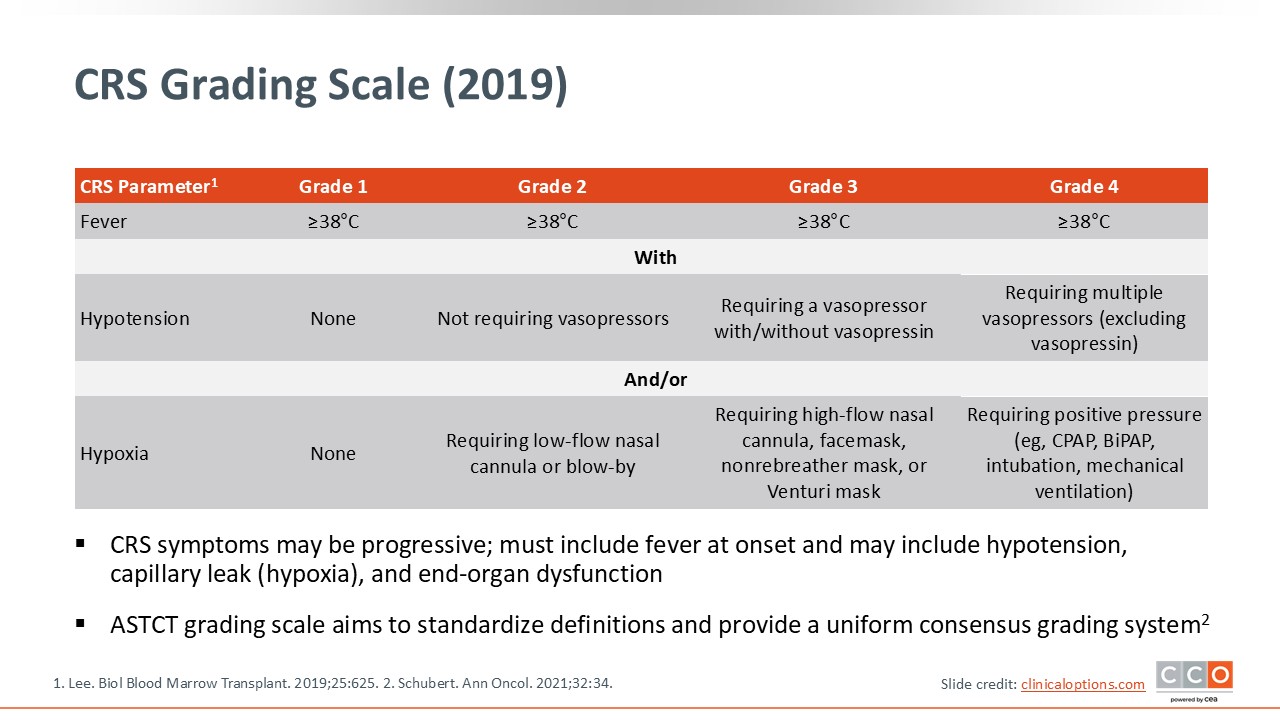
CRS Grading Scale (2019)
The CRS grading scale is important for standardizing definitions and providing a uniform consensus grading system to better manage CRS.10 With grade 1 CRS, patients present with fever and no other symptoms.7 Patients with grade 2 CRS present with fever and hypotension not requiring vasopressors and/or requirements of oxygen for hypoxia. Grade 3 CRS includes fever, hypotension requiring vasopressors with or without vasopressin, and hypoxia requiring more oxygen, such as a high-flow nasal cannula, face mask, or nonrebreather mask. Grade 4 CRS includes fever, hypotension requiring multiple vasopressors, and hypoxia requiring more invasive support, including positive pressure (ie, intubation and mechanical ventilation).
It is important to recognize, grade, and treat appropriately because these symptoms may be progressive and could lead to end-organ dysfunction.
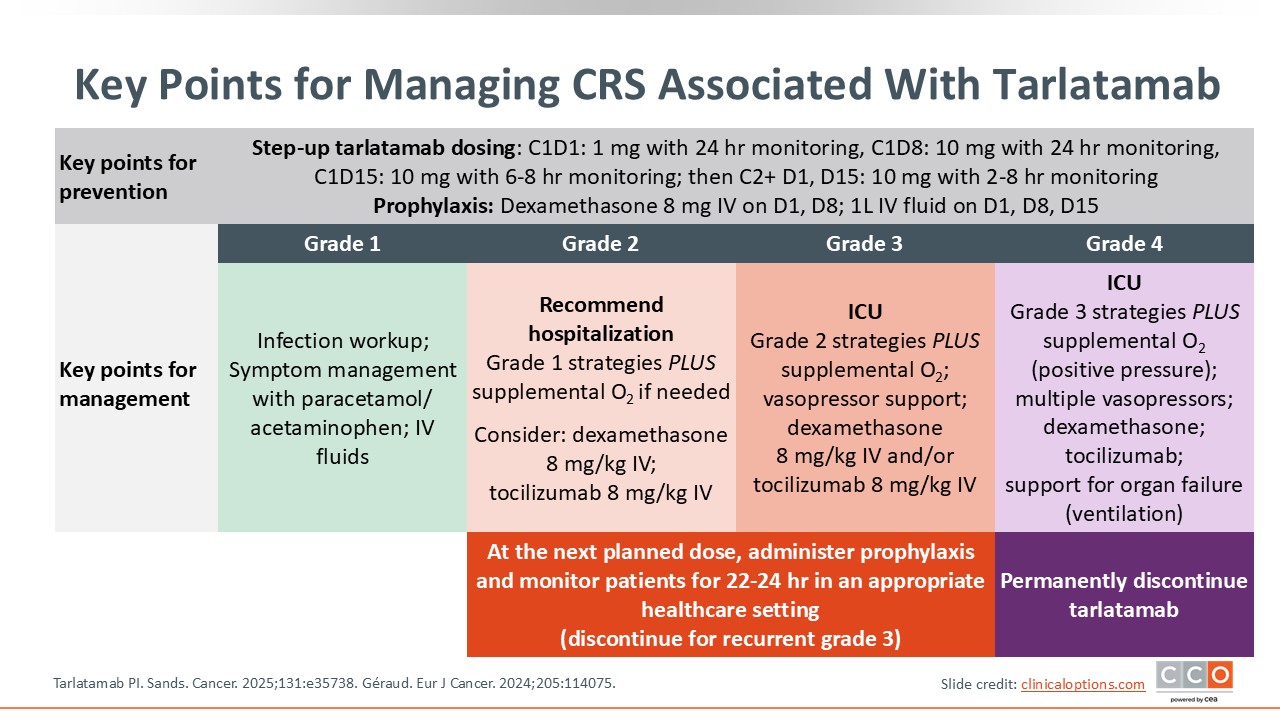
Key Points for Managing CRS Associated With Tarlatamab
The key point for managing CRS associated with T-cell engagers is prevention. Step-up dosing, as mentioned in our patient case example, along with the use of prophylactic dexamethasone and IV fluids, are important components of this strategy.1,3,9 Step-up dosing with tarlatamab occurs within cycle 1. On Day 1, tarlatamab 1 mg is administered and the patient should be monitored for 22-24 hours, with appropriate medical support to manage any CRS or neurotoxicity that could occur. The patient should remain within 1 hour of the healthcare facility for 48 hours. On Day 8, the tarlatamab dose is increased to 10 mg, with similar monitoring as on Day 1. On Day 15, tarlatamab 10 mg is administered and then the patient should be monitored for 6-8 hours. In cycle 1 on Days 1 and 8, concomitant dexamethasone 8 mg IV should be administered within 1 hour or tarlatamab dosing and on Days 1, 8, and 15, 1 liter of normal saline should be administered IV over 4-5 hours after tarlatamab infusion. After step-up dosing, tarlatamab is administered every 2 weeks (Day 1 and Day 15 of each cycle), with observation tapering down from 6-8 hours in cycle 2 to 3-4 hours in cycle 3 and 4 and then 2 hours in cycle 5 and beyond.
If CRS does occur, it is managed according to grade with each higher grade adding additional components to management. It is always important to rule out infection or other causes of fever. For grade 1 CRS (fever not due to infection or other causes), patients can be managed with supportive care, including acetaminophen and IV fluids.
For grade 2 CRS, escalate care to include supplemental oxygen and, if needed, dexamethasone 8 mg/kg IV (steroid to limit inflammation) or tocilizumab 8 mg/kg IV (a monoclonal antibody that blocks the proinflammatory cytokine IL-6 receptor).
For grade 3 CRS, patients require escalation of care to the ICU setting and vasopressor support in addition to high-flow oxygen. For grade 2 or grade 3 CRS, prophylaxis should be administered at the next planned dose of tarlatamab and patients should be monitored for 22-24 hours in an appropriate healthcare setting. For recurrent grade 3 CRS, tarlatamab should be discontinued.
Patients with grade 4 CRS are critically ill. Management includes ICU, IV fluids, vasopressor support, dexamethasone, tocilizumab, and support for organ failure, which includes ventilation. For patients who experience grade 4 CRS, tarlatamab should be permanently discontinued.
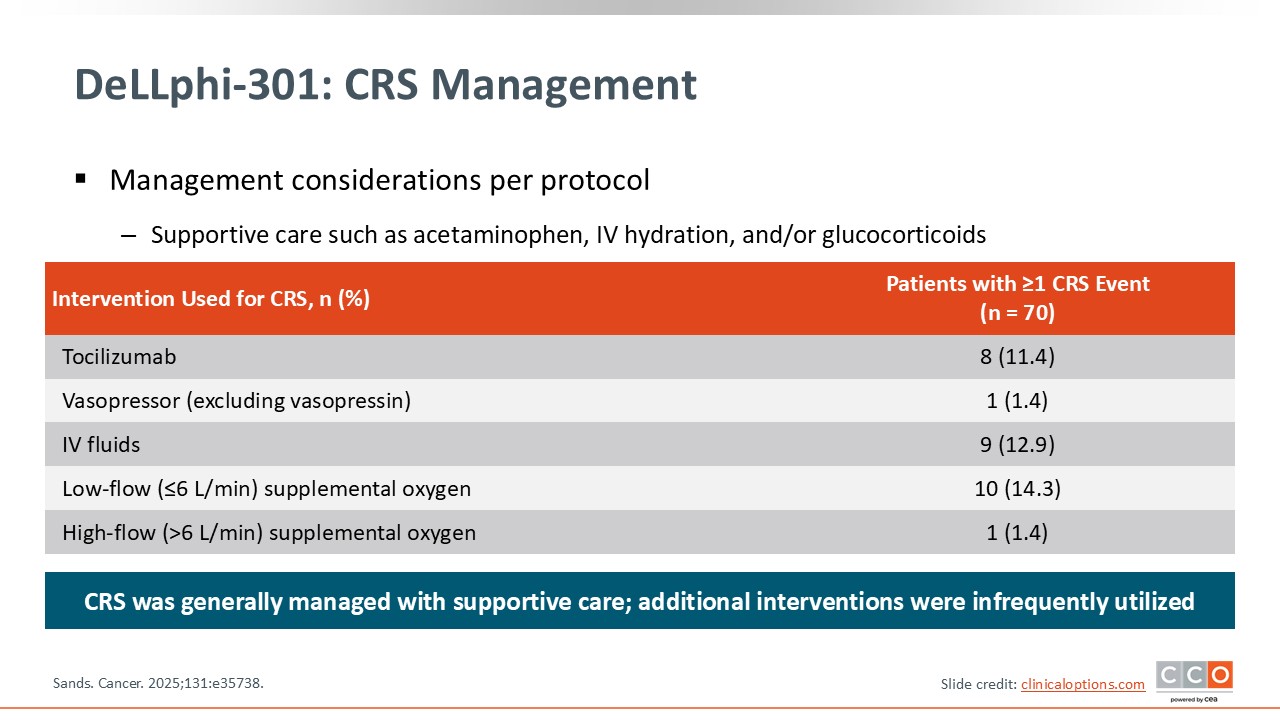
DeLLphi-301: CRS Management
In DeLLphi-301, the process for CRS management was well outlined, and the majority of patients were managed with supportive care. Additional interventions with escalating care were infrequently utilized. For example, tocilizumab was only used in approximately 11% of patients. Vasopressors were used in 1.4% of patients, IV fluids in 12.9%, low-flow supplemental oxygen in 14.3%, and high-flow supplemental oxygen in 1.4%.8
ICANS: Symptoms and Timing
ICANS is a neurologic complication of T-cell engagers that may occur more commonly after CRS but can also overlap with CRS, and the symptoms are highly variable.7,11
Symptoms of ICANS include confusion, impaired attention, tremor, and motor findings including trouble writing and weakness. These can progress to altered consciousness, seizures, and cerebral edema. Symptoms develop a median of 5 days after the final dose of tarlatamab, with a median time to resolution of 6.5 days.2
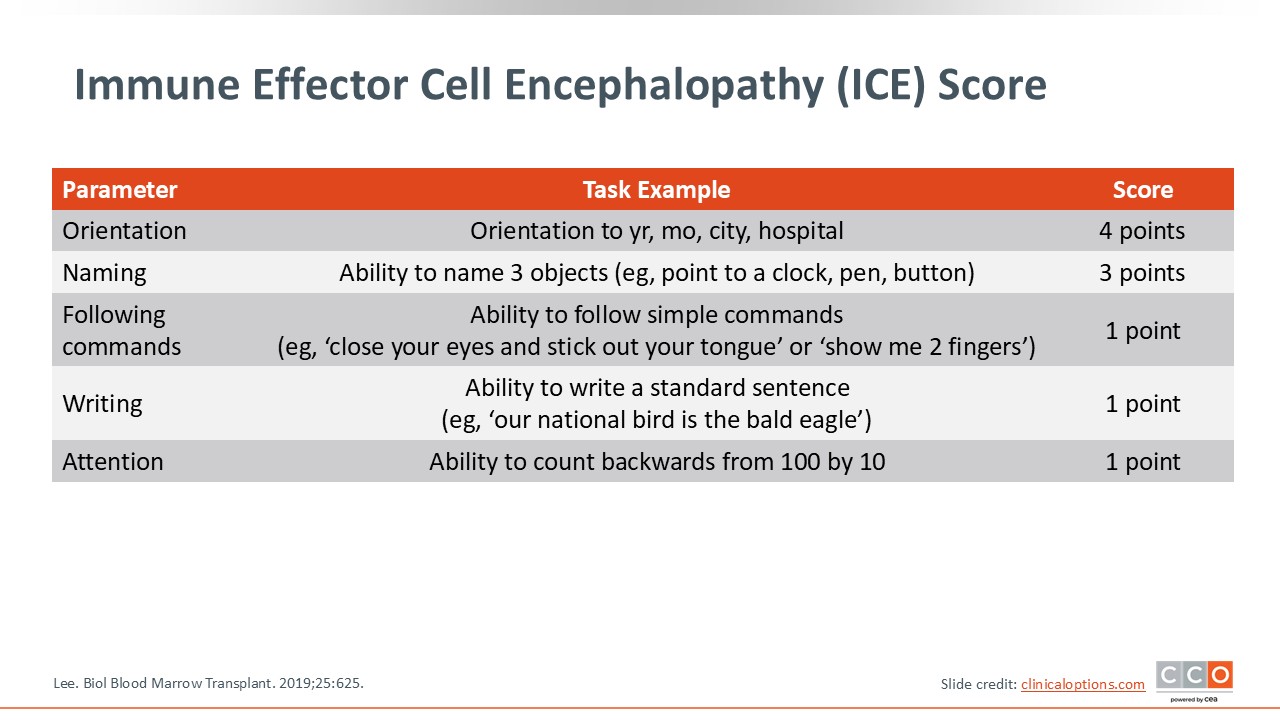
Immune Effector Cell Encephalopathy (ICE) Score
The ICE score is an important tool to standardize the approach to diagnosing ICANS. The ICE score includes 5 parameters: orientation, naming, following commands, writing, and attention. Patients are provided with tasks and then given a score, and the total ICE score is then calculated.7
ICANS Grading Scale (2019)
The ICANS grading scale incorporates the ICE score along with other features.7 If a patient has grade 1 ICANS, they have an ICE score of 7-9. Such patients have a depressed level of consciousness, but they awaken spontaneously. For grade 2 ICANS, patients have an ICE score of 3-6. Such patients have a depressed level of consciousness and awaken to a voice. For grade 3 ICANS, the ICE score is 0-2. Patients awaken only with tactile stimuli, and they may have other symptoms such as clinical seizures. These patients may also have nonconvulsive seizures on EEG that resolve with intervention. Patients with grade 4 ICANS are critically ill in the ICU, unarousable, and unable to perform the tests used to calculate the ICE score. These patients have life-threatening or prolonged seizures and may have focal motor weakness such as hemiparesis and diffuse cerebral edema on neuroimaging.
The key point is to determine the ICE score as well as screen for alternative causes of confusion or seizures, including performing brain MRI early and consulting with neurology as needed. It is important to implement protocols using corticosteroids, nonsedative anticonvulsants, and certainly escalating care appropriately and promptly with ICU management for grade 3 or 4 ICANS. Getting multidisciplinary input from critical care and neurology can also be helpful.
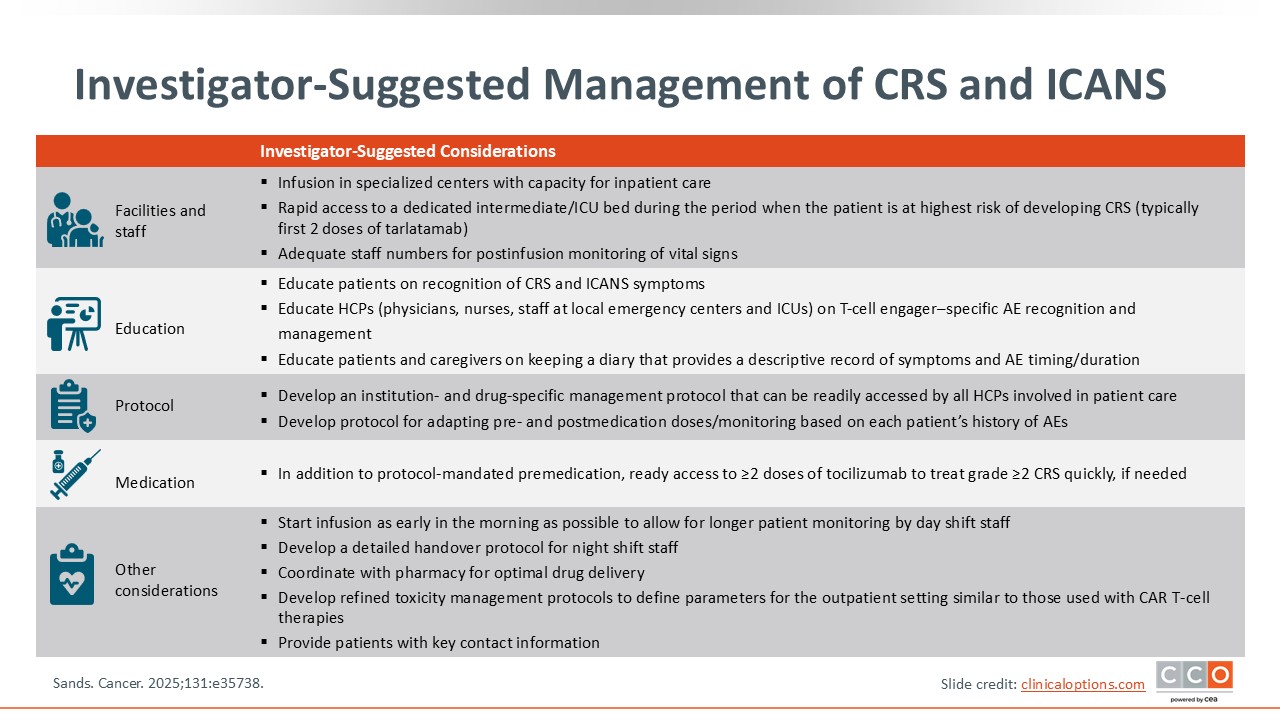
Investigator-Suggested Management of CRS and ICANS
This slide details a practical approach to CRS and ICANS management suggested by the investigators of DeLLphi-301.8 From a facility and staff standpoint, it is important to have infusion and specialized centers that have capacity for inpatient care and rapid access to dedicated intermediate or ICU beds during the period when the patient is at greatest risk for developing CRS (ie, typically within the first 2 doses of tarlatamab), ensuring adequate staffing for postinfusion monitoring of vital signs and to assess the ICE score.
From an education standpoint, it is critical to educate patients and their caregivers on symptom recognition for CRS and ICANS, especially when they return home. It is also important to educate the appropriate healthcare professionals (HCPs), including physicians, nurses and staff, not only in the inpatient setting but also at the emergency centers and ICU.
From a protocol standpoint, institution-specific and drug-specific management protocols that are applicable to the appropriate setting are crucial, and premedication and postmedication protocols based on the patient's history of AEs should be developed. In addition to protocol-mandated premedications, ready access to at least 2 doses of tocilizumab in case of grade 2 or higher CRS is essential.
Other practical considerations include starting the infusion early in the morning to allow longer patient monitoring, a detailed handover when appropriate, and coordinating with pharmacy for optimal drug delivery. Additional considerations are the development of refined toxicity management protocols to define parameters for the outpatient setting (similar to those used with CAR T-cell therapies) and providing patients with key contact information and wallet cards so they can present them to other HCPs if necessary.
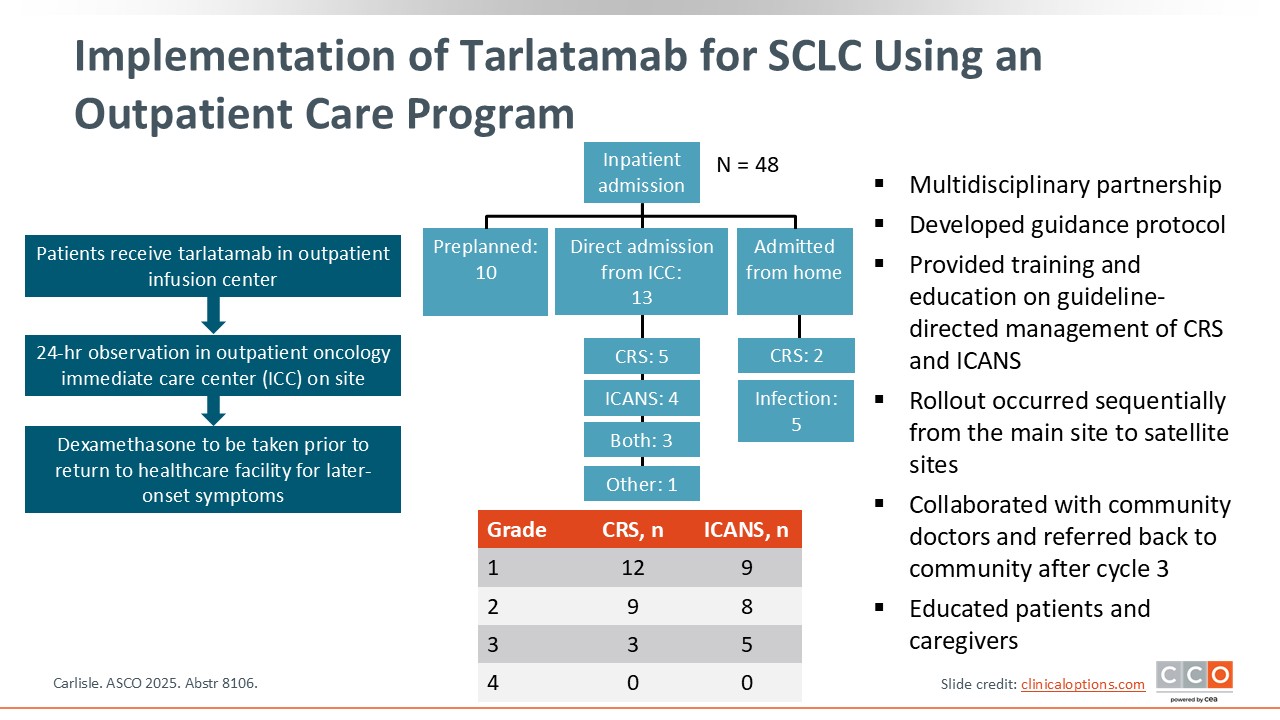
Implementation of Tarlatamab for SCLC Using an Outpatient Care Program
Outpatient administration of tarlatamab and monitoring of patients can reduce healthcare resource utilization and treatment costs associated with inpatient care, as well as enhance patient convenience and quality of life. A recent study assessed tarlatamab administration via an outpatient care program to treat SCLC.12
Forty-eight patients received tarlatamab in an outpatient infusion center and then 24-hour observation in the on-site outpatient oncology intermediate care center. When ready for discharge, patients received a prescription for dexamethasone to be taken prior to their return to a healthcare facility for later-onset symptoms.
Overall, the outpatient care program was feasible, even for patients with an ECOG performance status of 2. Approximately 25% of patients required admission from the outpatient setting. These findings suggest that multidisciplinary partnership is key. Guidance protocols for staff and HCPs were developed, and training and education on guideline-directed management of CRS and ICANS were provided.
The study was rolled out sequentially from the main site to satellite sites, and collaborations with community doctors ensured that cycles 1 and 2 were given at the main site while cycle 3 was given at community sites. Patient and caregiver education and partnership were also critical, especially information regarding when to call and when to return, either to the outpatient urgent care or the emergency room.
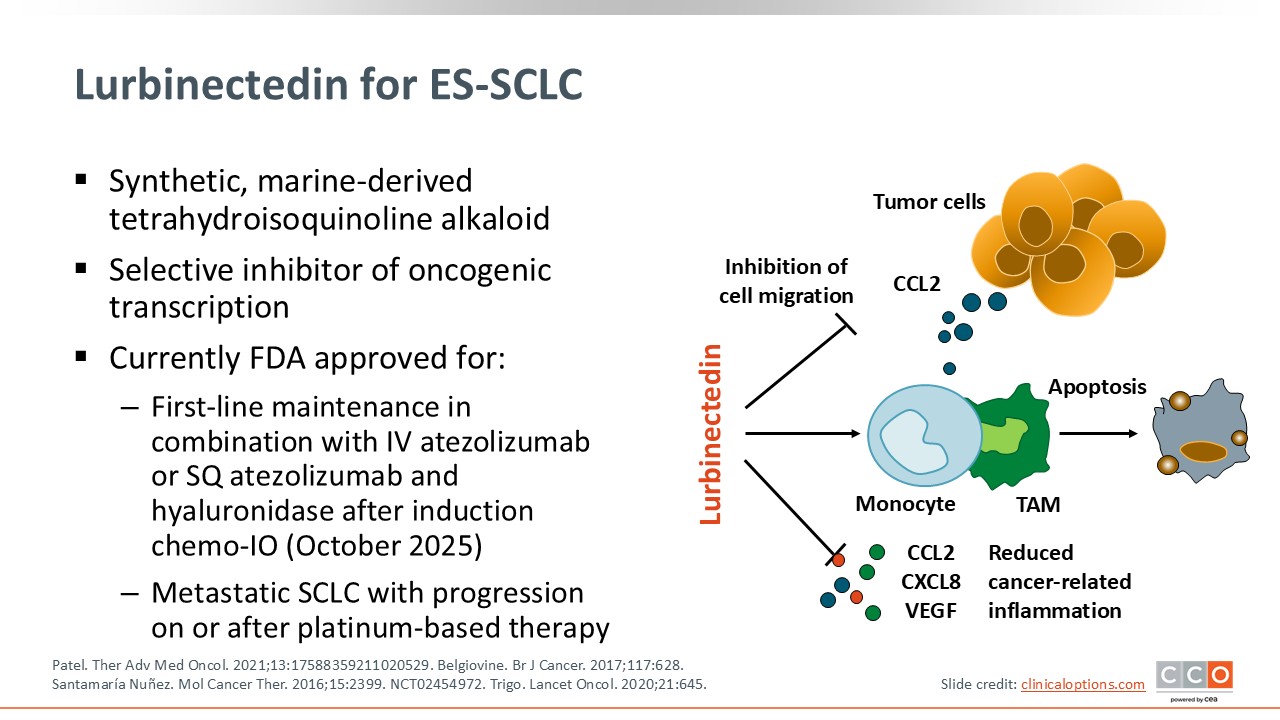
Lurbinectedin for ES-SCLC
Lurbinectedin is a synthetic marine-derived alkaloid with a multimodal mechanism of action. It is a selective inhibitor of oncogenic transcription and may also have effects on the tumor immune microenvironment.13
This agent was approved in the second-line and beyond setting for adult patients with metastatic SCLC who had disease progression after platinum-based chemotherapy, based on the phase II B-005 basket trial.14,15
More recently, based on the phase III IMforte trial, lurbinectedin was approved in combination with atezolizumab or atezolizumab and hyaluronidase-etqjs for the maintenance treatment of adult patients with ES-SCLC whose disease has not progressed after first-line induction therapy with atezolizumab or atezolizumab and hyaluronidase-etqjs, carboplatin, and etoposide.14,16
Current guidelines include single-agent lurbinectedin for second- or later-line therapy and lurbinectedin in combination with atezolizumab for maintenance.17
Phase II Study of 2L Lurbinectedin in ES-SCLC: Safety
Lurbinectedin has been well established as a safe treatment option for patients with ES-SCLC, either as monotherapy or in combination with immunotherapy. Shown here are safety data from the phase II study of second-line lurbinectedin that established the recommended dose of 3.2 mg/m2.15 The primary AEs seen with lurbinectedin are myelosuppression including anemia, leukopenia, neutropenia, and thrombocytopenia, with the majority of events being grade 1 and 2. The rate of grade 3 neutropenia was 21%, but the rate of febrile neutropenia was low. It is important to monitor for myelosuppression and ensure that secondary prophylaxis is given for patients who develop neutropenia.
Other common AEs include fatigue and nausea. Fatigue was observed in 51% of patients but the rate of grade 3 fatigue was low, at 7%. Grade 1/2 nausea was observed in 32% of patients. Important AEs to be monitored are elevations in creatinine, AST, and ALT, with most presenting as grade 1 or 2.
IMforte: Safety During Maintenance Phase
The phase III IMforte study found improved OS with the addition of lurbinectedin to atezolizumab as maintenance after first-line atezolizumab plus chemotherapy.16 The most common grade 3/4 AEs with lurbinectedin plus atezolizumab were similar to those observed in the phase II trial, with hematologic AEs being the most common. The discontinuation rate due to AEs was 6% in the lurbinectedin arm.
Practical Considerations on Dosing Lurbinectedin
To prevent nausea and vomiting, premedication with corticosteroids and serotonin antagonists should be considered when beginning therapy with lurbinectedin.14 Prophylactic G-CSF is recommended during treatment with lurbinectedin plus atezolizumab to reduce the risk of febrile neutropenia, or as secondary prophylaxis after an initial decrease in white blood cell count with single agent lurbinectedin. Also ensure that patients contact their HCPs for any new comedications.
Of importance, lurbinectedin AEs can be managed with dose modifications, dose interruptions, or dose reductions to manage the myelosuppression, as well as other notable AEs, including fatigue and laboratory abnormalities such as elevated creatinine and liver function tests.
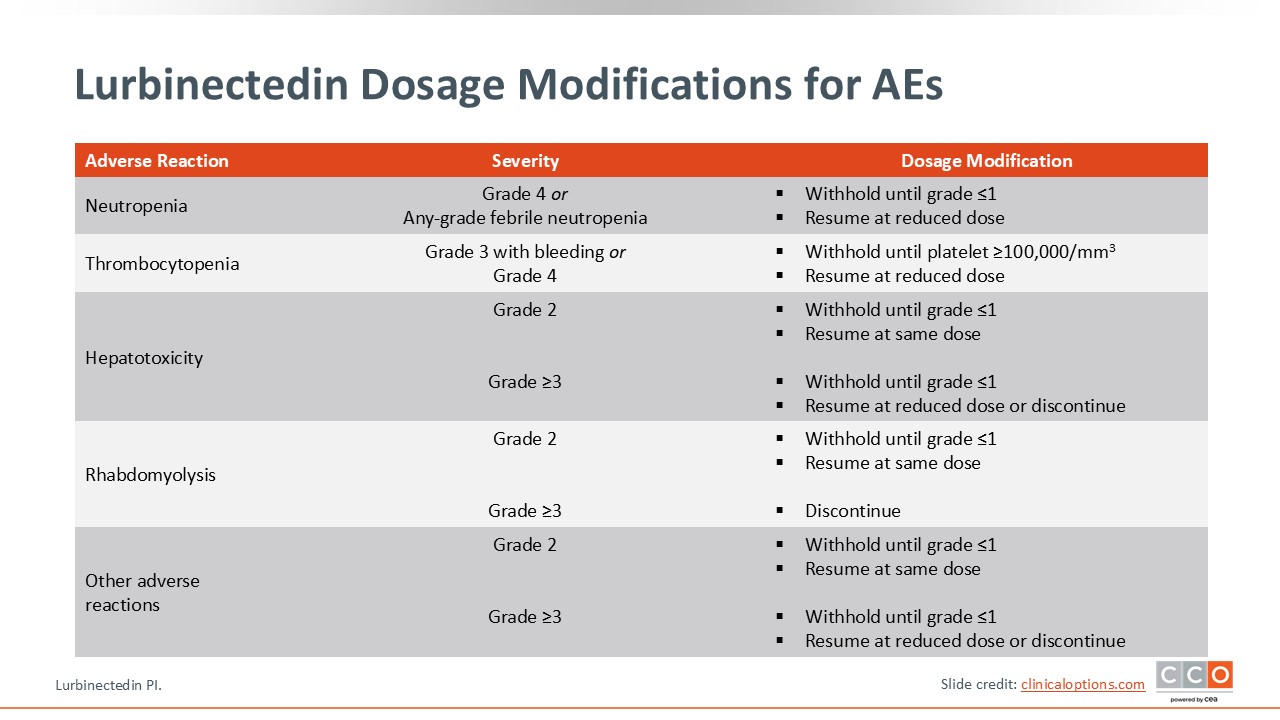
Lurbinectedin Dosage Modifications for AEs
Standard dose modifications for lurbinectedin due to AEs can help mitigate AEs and keep patients on therapy long-term.14
If patients develop grade 4 neutropenia or any-grade febrile neutropenia, hold lurbinectedin until resolution to grade 1 or less and then resume at a reduced dose. For grade 3 thrombocytopenia with bleeding or grade 4 thrombocytopenia, hold lurbinectedin until recovery of platelets to 100,000/mm3 or greater, and resume lurbinectedin at a reduced dose.
For hepatotoxicity, rhabdomyolysis, or other AEs, if grade 2, hold lurbinectedin until grade 1 or less and then resume at the same dose. For grade ≥3 hepatotoxicity or other AEs, hold lurbinectedin until grade 1 or less but resume at a reduced dose or permanently discontinue. For grade ≥3 rhabdomyolysis, permanently discontinue lurbinectedin.
Practical Considerations on Dosing Atezolizumab
Two PD-L1 inhibitors, atezolizumab and durvalumab, are indicated in the frontline setting in combination with chemotherapy for ES-SCLC.18,19 Both agents are also approved in the maintenance setting—atezolizumab in combination with lurbinectedin as previously described and durvalumab as monotherapy. Current guidelines also list single-agent atezolizumab for maintenance.17
Atezolizumab can be used at 3 different dosing schedules: 840 mg every 2 weeks, 1200 mg every 3 weeks, or 1680 mg every 4 weeks. The IMpower133 trial found improved OS with the addition of atezolizumab to carboplatin etoposide vs placebo plus carboplatin etoposide.20 In this study the 1200 mg every 3 weeks schedule was used, but the other strategies can be useful in clinical practice. In the IMforte trial, atezolizumab 1200 mg every 3 weeks was used in combination with lurbinectedin 3.2 mg/m2 IV every 3 weeks.21
In terms of dosing considerations, when atezolizumab is used in combination with chemotherapy, atezolizumab is given first, followed by carboplatin-etoposide, and antiemetics are administered for the chemotherapy portion per local guidelines.17,18 Premedication with antihistamines can be helpful if a patient experiences an infusion-related reaction during any prior infusion with atezolizumab.
Practical Considerations on Dosing Durvalumab
There are 2 approved dosing schedules for durvalumab based on weight.19 For patients weighing at least 30 kg, 1500 mg every 3 weeks plus chemotherapy, followed by 1500 mg every 4 weeks as a single agent is approved. For patients weighing less than 30 kg, the dosing is adjusted to 20 mg/kg every 3 weeks during the chemotherapy portion for 4 cycles, followed by 10 mg/kg every 2 weeks as a single agent. When used in combination with chemoimmunotherapy, durvalumab should be administered before chemotherapy. In case of infusion-related reactions, which are uncommon, premedication with corticosteroids can be helpful.21
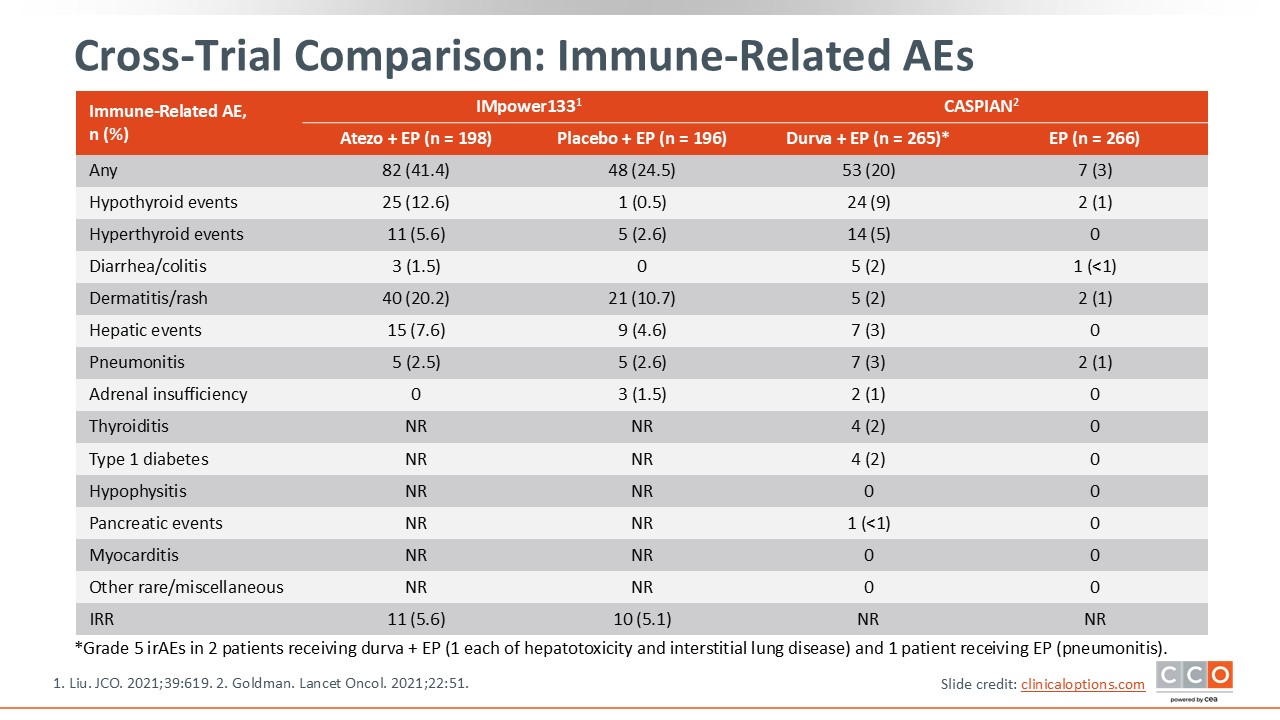
Cross-Trial Comparison: Immune-Related AEs
The majority of AEs noted in both IMpower133 and CASPIAN were related to the chemotherapy component.20,22 In terms of immune-related adverse events (irAEs), the frequency was generally low. The most common grade 3 or higher irAEs were related to hypothyroid or hyperthyroid events, dermatitis or rash, diarrhea and colitis, hepatic events, and pneumonitis. In my opinion, the overall safety profile of these agents is very well established.
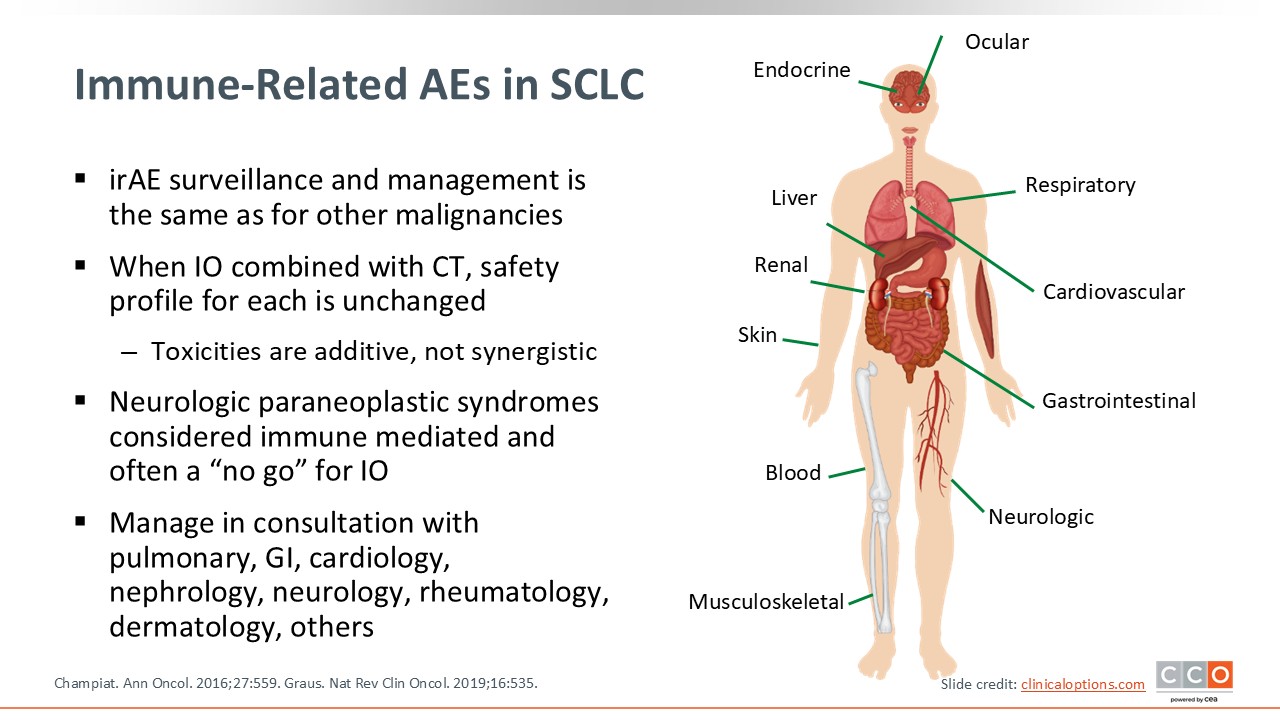
Immune-Related AEs in SCLC
The safety profiles of these agents are well established, but it is important to maintain vigilance with regards to irAEs.23,24 Although uncommon and typically grade 1 and 2 in nature, some can be significant and life-threatening, especially if unrecognized. When chemotherapy is combined with immunotherapy, it is important to note that the toxicities are additive, not synergistic. It is also important to differentiate between neurologic paraneoplastic syndromes and irAEs. If irAEs are escalating, consider consultation with different subspecialists depending on the organs affected.
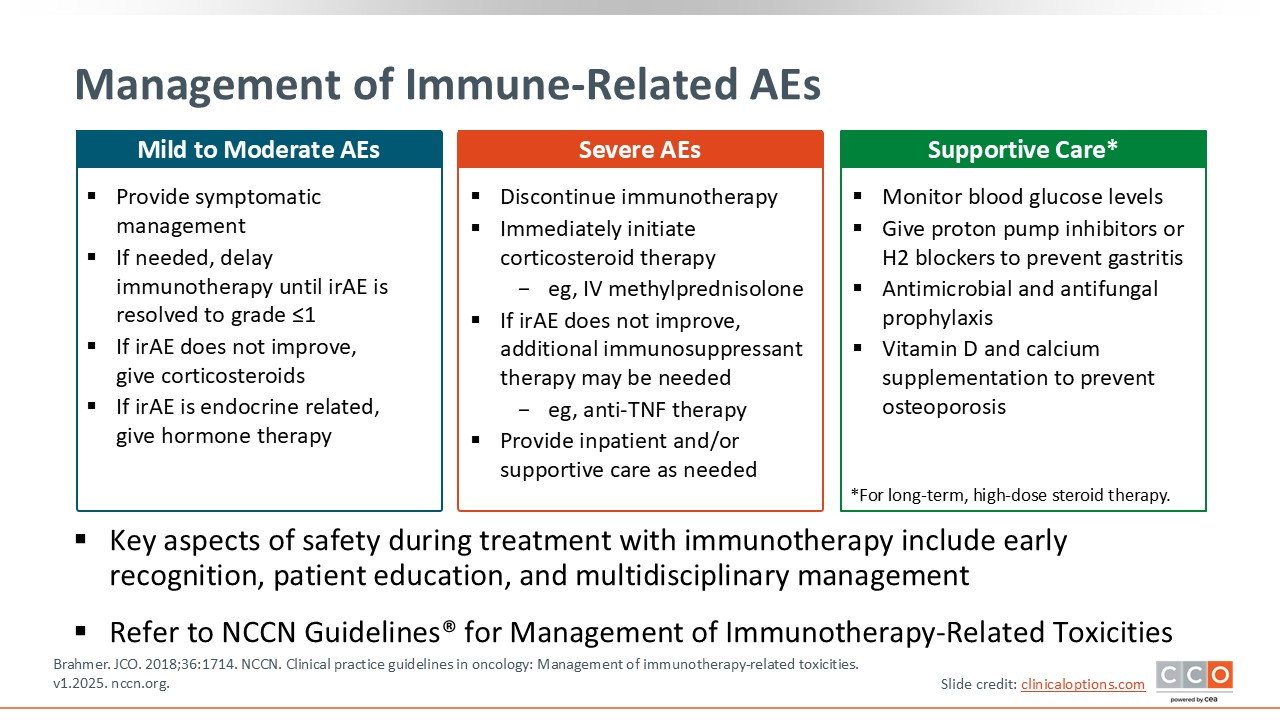
Management of Immune-Related AEs
For mild to moderate AEs, depending on the organs affected, provide symptomatic management and delay immunotherapy until resolution to grade 1 or less.25,26 In the absence of improvement and if escalating to grade 2, corticosteroids are key. For an endocrine-related AE such as hypothyroidism, thyroid hormone replacement can be helpful, and for adrenal insufficiency, replace with hydrocortisone.
For severe AEs, depending on severity, consider discontinuing immunotherapy, especially with grade 3 or higher pneumonitis. Immediately initiate corticosteroid therapy and consider an inpatient setting for management with IV steroids. If the irAE does not improve, additional immunosuppressant therapy may be needed. Over time, for patients who will be receiving at least a prolonged course of corticosteroids, it is important to monitor blood glucose levels. Give proton pump inhibitors or H2 blockers to prevent gastritis, use antimicrobial and antifungal prophylaxis, and use vitamin D and calcium supplementation to prevent osteoporosis. Please refer to the NCCN guidelines for further details for the management of immunotherapy-related toxicities.26
It is also critical to educate patients and their caregivers on when to call and when to come back to the clinic. Patients need to be well informed and encouraged to participate in treatment decisions to best cope with AEs, including irAEs as some can lead to a chronic need for steroids or hormone replacement therapy.
Take-home Messages
In conclusion, new agents have shown improved outcomes for patients with SCLC. Managing AEs is critical not only to allow patients to remain on therapy if beneficial but also to minimize the impact on quality of life.
Tarlatamab has a manageable safety profile with a low discontinuation rate due to TRAEs (4%-5%, depending on the study).3,4 CRS usually occurs after the first or second dose and is managed primarily with supportive care. ICANS are primarily grade 1 and 2 in nature and occur mostly in the first or second cycle of tarlatamab. Management strategies include supportive care for grade 1 and steroids for grade 2.3
Lurbinectedin has an established favorable safety profile, but it does require monitoring and dose adjustment, mainly for myelosuppression.15
Key aspects of safety during treatment with immunotherapy include early recognition, patient education, and multidisciplinary management, depending on the organ(s) affected and grade of the AE.