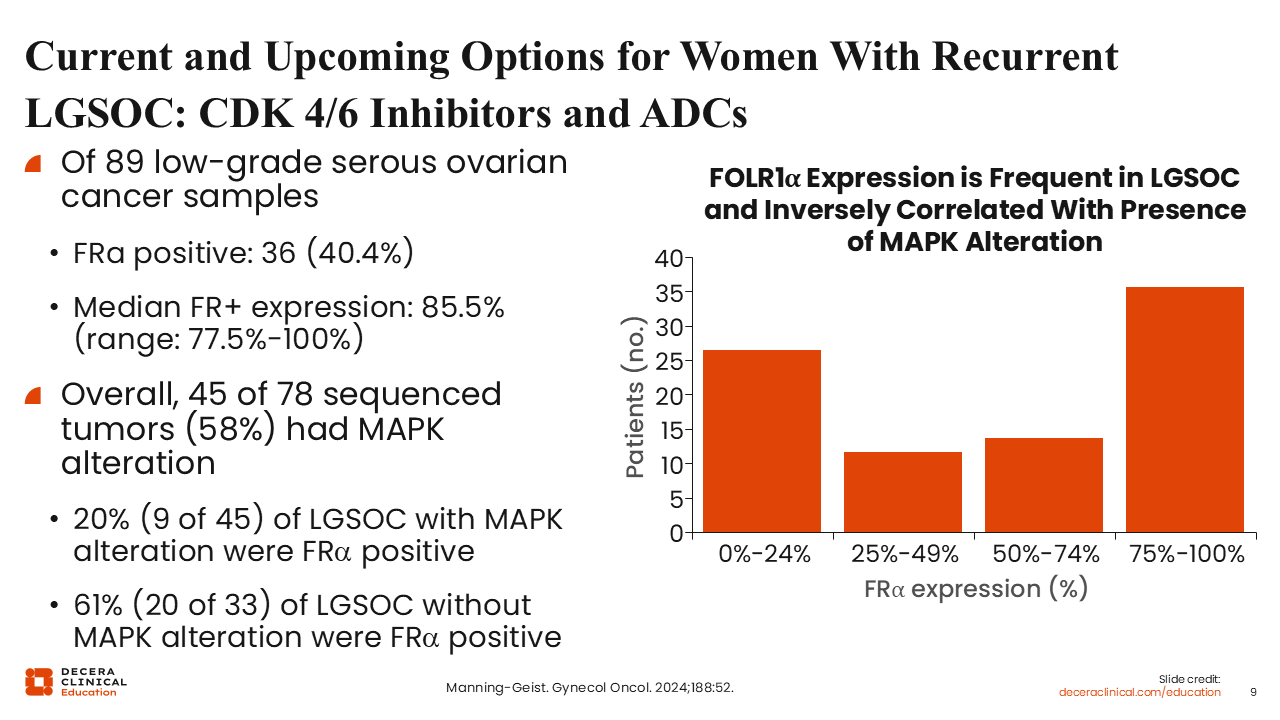
CE / CME
Clinical Focus in KRAS-Mutated Low-Grade Serous Ovarian Cancer: Integrating a Novel Dual MEK/FAK Inhibitor Into Practice
Physician Assistants/Physician Associates: 0.75 AAPA Category 1 CME credit
Pharmacists: 0.75 contact hour (0.075 CEUs)
ABIM MOC: maximum of 0.75 Medical Knowledge MOC point
Physicians: maximum of 0.75 AMA PRA Category 1 Credit™
Nurse Practitioners/Nurses: 0.75 Nursing contact hour
Released: February 17, 2026
Expiration: August 16, 2026
Activity
Molecular and Clinical Features of LGSOC and HGSOC
Kathleen N. Moore, MD, MS, FASCO:
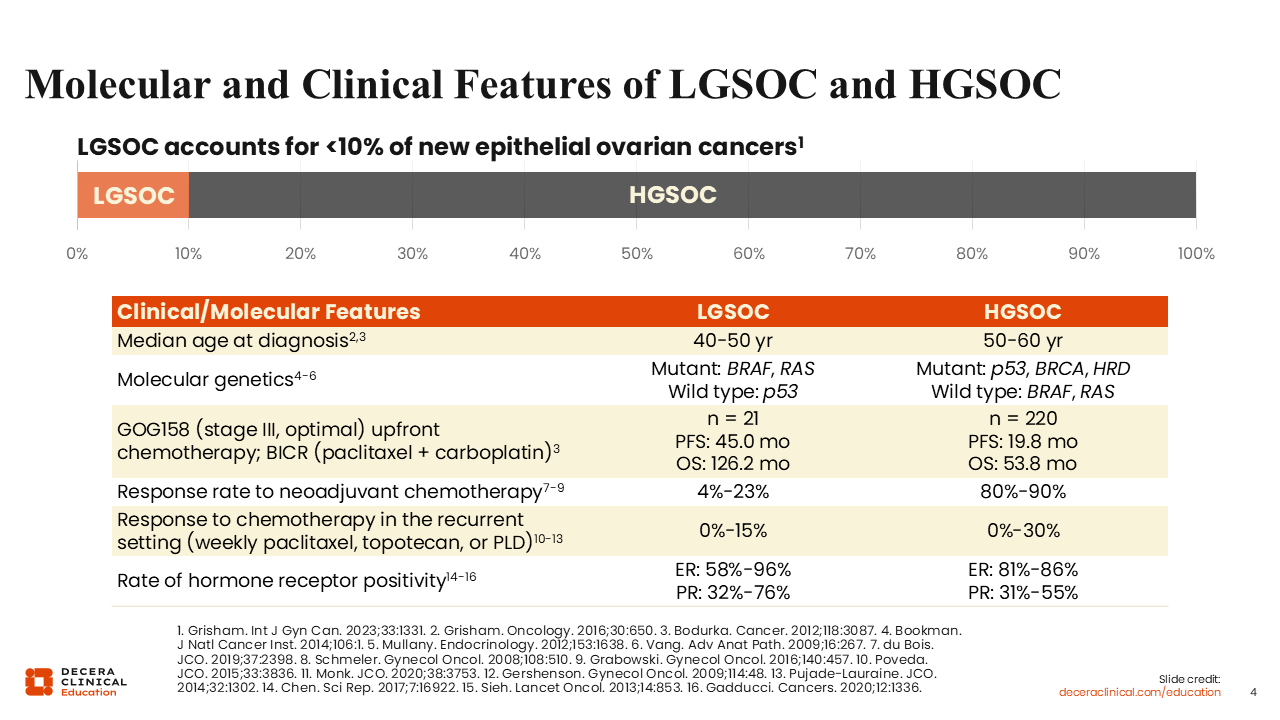
It is critical to understand how LGSOC differs from HGSOC, as they are different diseases with distinguishing features. Patients with LGSOC tend to be about a decade younger than those with HGSOC. These tumors also differ in terms of their natural history and site of origin. LGSOC is referred to as a borderline tumor or a tumor with low malignant potential. Most do not undergo malignant transformation. These tumors do not lack the tumor suppressor gene TP53. Other alterations commonly seen in LGSOC include MAP kinase (MAPK) pathway mutations, most often KRAS, and BRAF mutations.1 In contrast, HGSOC starts predominantly in the fallopian tube and is most often characterized by loss of TP53. Homologous recombination deficiency, which suggests inherent vulnerability within the tumor to how the DNA is fixed, is also noted, with such changes including loss of the BRCA1 gene.2
These tumors also differ in their response to standard therapies. Patients with HGSOC tend to respond well to neoadjuvant chemotherapy, with rates of up to 80% to 90%. However, in patients with LGSOC, responses to neoadjuvant chemotherapy are very uncommon.3 These tumors are not entirely chemoresistant, but they are far more chemoresistant than HGSOCs. The same is true when assessing response to chemotherapy in the recurrent setting, which involves treatment with weekly paclitaxel, with a response rate of up to 30% in patients with HGSOC4 compared with up to 15% in patients with LGSOC.5 This means that LGSOC tumors are more chemoresistant,6 although they do maintain some sensitivity to endocrine therapies.
Estrogen receptor positivity is very common across both LGSOC and HGSOC. However, LGSOCs are hormonally driven tumors, so endocrine therapy is sometimes more effective than chemotherapy.
Why Is Somatic Tumor Testing Important in LGSOC?
Kathleen N. Moore, MD, MS, FASCO:
As with any solid tumor, it is important to perform a careful histologic evaluation when diagnosing ovarian cancer. Sufficient tumor tissue is incredibly important for making a definitive diagnosis. Differentiating LGSOC from HGSOC often requires central review because diagnosis can be challenging. Diagnosis from either a surgically resected specimen or core biopsy is
the gold standard. It is recommended to test for somatic mutations at a minimum, as identification of actionable molecular mutations will help guide personalized care and optimize outcomes.
MAPK Pathway Alterations in LGSOC
Kathleen N. Moore, MD, MS, FASCO:
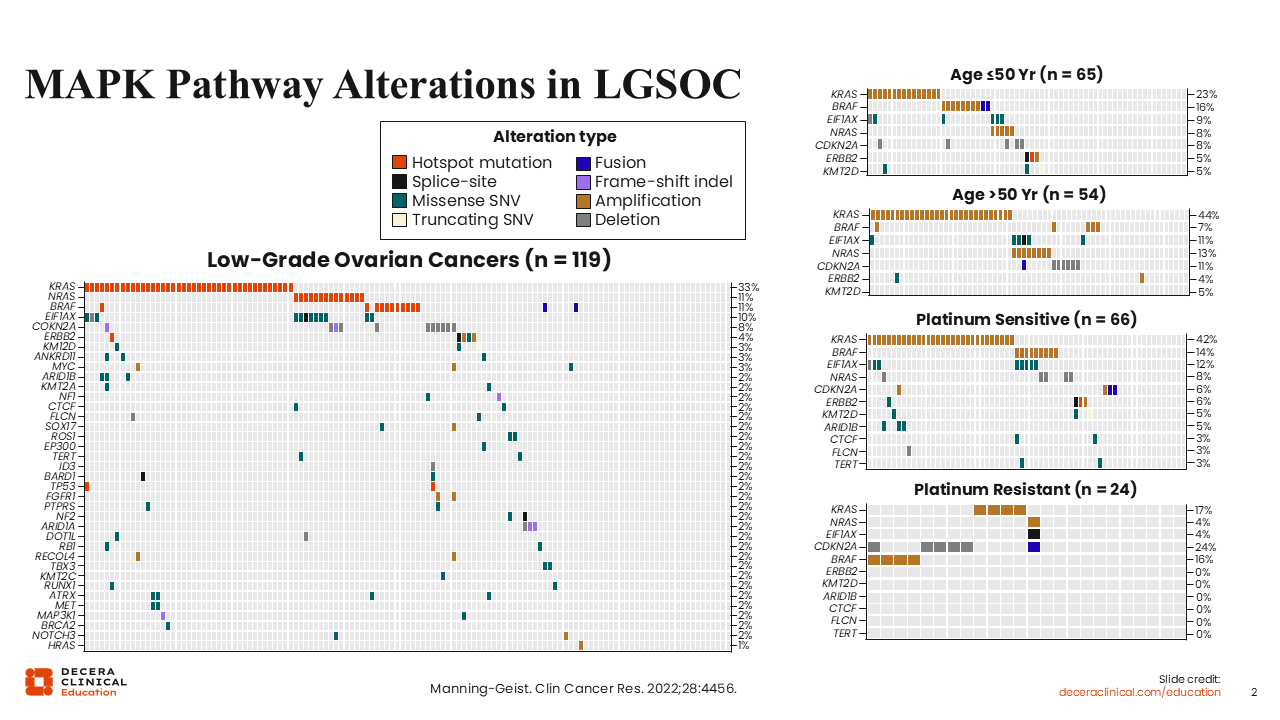
Although potentially predictive biomarkers for novel therapeutics in the LGSOC setting are still being evaluated, the vast amount of evidence focuses on alterations in the MAPK pathway. Shown here are heatmaps from nearly 120 patients with LGSOC. KRAS is the most commonly altered gene, accounting for 33% of alterations, followed by NRAS (11%), and BRAF (11%).7
Biomarkers Used to Guide Targeted Therapy in LGSOC
Kathleen N. Moore, MD, MS, FASCO:
There are a few biomarkers currently used to guide targeted therapy in the setting of LGSOC. Again, BRAF alterations are found in approximately 11% of these tumors. Such tumors can be treated with dabrafenib and trametinib. The FDA approved the use of dabrafenib and trametinib the treatment of adult and pediatric patients 1 year of age or older with unresectable or metastatic solid tumors with BRAF V600E mutation who have progressed following prior treatment and have no satisfactory alternative treatment options.8 For patients with confirmed LGSOC who also harbor a BRCA mutation, though rare, a PARP inhibitor can be considered. As previously mentioned, the presence of estrogen and progesterone receptors drives the selection of endocrine-based therapies for patients with LGSOC. However, one must be careful referring to this as a predictive biomarker. RAS or KRAS mutations are predictive biomarkers for response to MEK inhibitors. Patients with KRAS-altered recurrent LGSOC can be treated with avutometinib and defactinib, a combination that gained FDA approval based on results from the phase II RAMP-201 trial.9
How Do We Sequence Therapy in Patients With Recurrent Low-Grade Serous Ovarian Cancer?
Kathleen N. Moore, MD, MS, FASCO:
Currently, biomarkers do not inform frontline therapies for patients diagnosed with LGSOC; biomarkers come into play at recurrence. When deciding on sequencing, the first consideration is biomarker status. Does this patient have a KRAS mutation? Do they have a BRAF mutation? Protein expression of other biomarkers should also be considered. For this reason, immunohistochemistry testing is being increasingly incorporated into treatment decisions. We also want to identify clinical features of recurrence. Is the patient experiencing recurrence that is asymptomatic years after initial diagnosis? Does the patient have bulky disease that is growing quickly? Therapy sequence depends on the patient, how the disease is behaving, biomarkers, and the patient’s ability to tolerate oral medications, along with other factors. Treatment sequencing is the art of medicine, and additional information on biomarkers will help us better understand the clinical behavior of the tumor to optimize therapeutic outcomes.
Can Liquid Biopsies Be Used to Assess Somatic Mutations?
Kathleen N. Moore, MD, MS, FASCO:
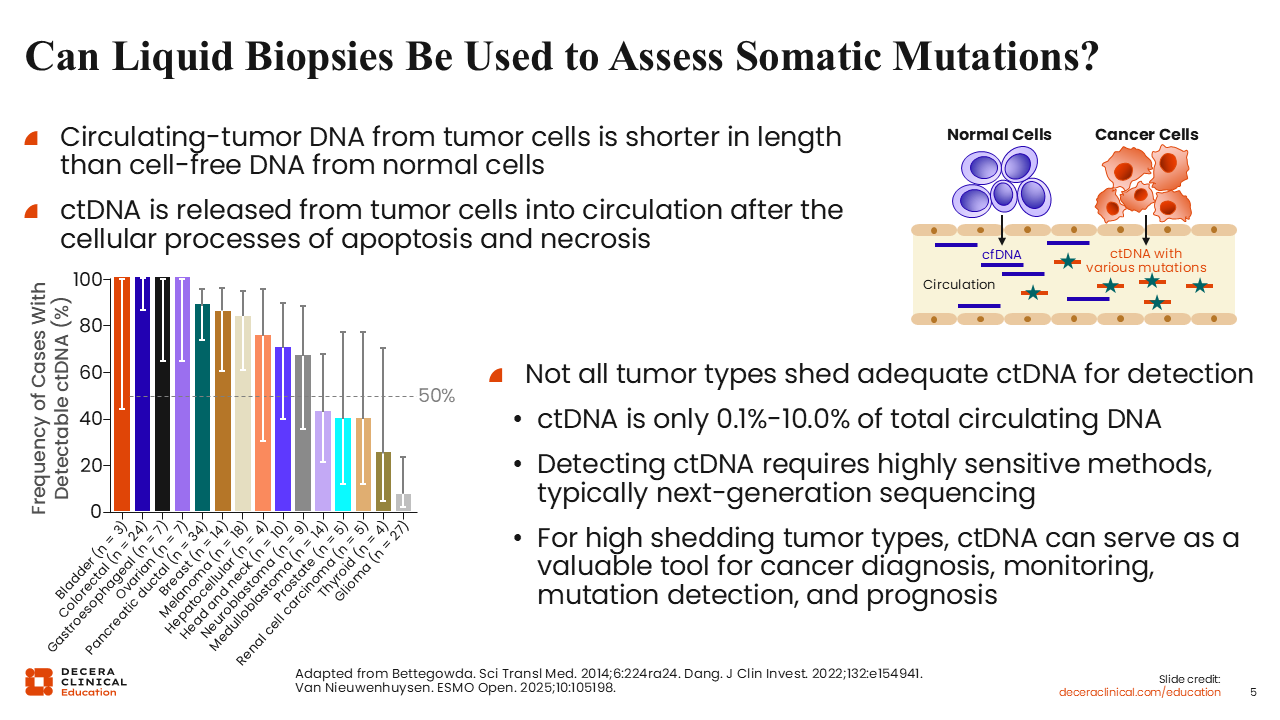
Response to therapy can be assessed using liquid biopsies, which can help identify resistance mechanisms. Healthcare professionals (HCPs) in the field of ovarian cancer are learning a lot from our colleagues across the solid tumor landscape. However, at present, there is not enough evidence to suggest that ctDNA can be used to assess somatic mutations in LGSOC.10
Liquid Biopsies Are NOT a Substitute for Detecting Somatic mutations in LGSOC
Kathleen N. Moore, MD, MS, FASCO:
The concordance of identifying KRAS mutations between liquid biopsy and blood samples is only 44%,11 so over half will be missed. More than half of patients with LGSOC harbor a KRAS, NRAS, HRAS or BRAF mutation. We want to offer effective therapy, but if we rely on liquid biopsies, many mutations will be missed. Thus, tumor tissue is required, as it allows opportunities to access MEK inhibitors, which may be highly effective in this population.10
GOG 281 and MILO/ENGOT-ov11: Single Agent MEK Inhibition for Recurrent LGSOC
Rachel Grisham, MD:
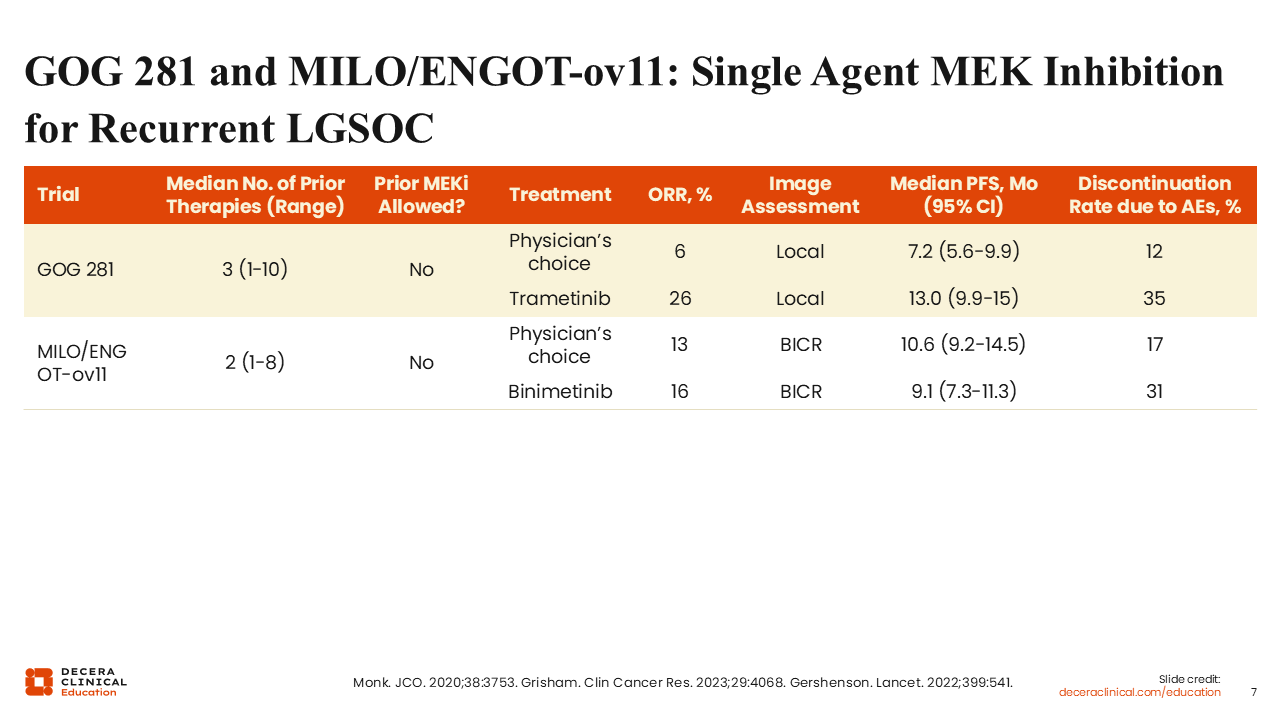
Two phase III studies showed the activity of single-agent MEK inhibitors in patients with recurrent LGSOC. In GOG 281, patients with recurrent LGSOC were randomized to either the single-agent MEK inhibitor trametinib or physician's choice standard of care (SoC). The overall response rate (ORR) to trametinib was 26%.12 In MILO/ENGOT-ov11, patients with recurrent LGSOC were randomized to either physician's choice of chemotherapy or the single-agent MEK inhibitor binimetinib. This study closed prematurely, but at the time of the interim analysis, the ORR to binimetinib was 16%.13
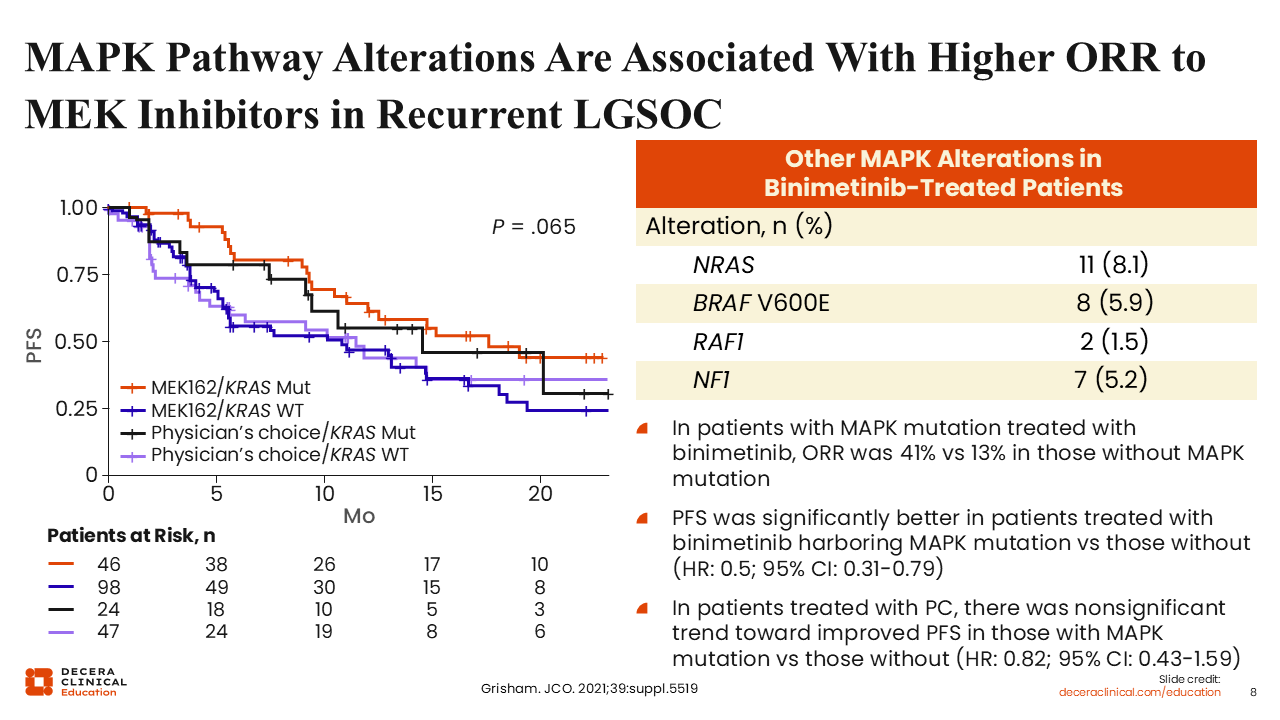
MAPK Pathway Alterations Are Associated With Higher ORR to MEK Inhibitors in Recurrent LGSOC
Rachel Grisham, MD:
An analysis of the MILO study showed that patients with a MAPK pathway alteration had higher response rates, with an ORR of 41% to binimetinib, vs those without a MAPK pathway alteration (13%). There was a similar trend when comparing responses between patients with a KRAS mutation and those without. A post hoc analysis of patients treated with trametinib enrolled in the GOG 281 study also showed a similar trend (ORR: 50% vs 8.3% among patients with a MAPK pathway mutation vs those without).13
Current and Upcoming Options for Women With Recurrent LGSOC: CDK 4/6 Inhibitors and ADCs
Rachel Grisham, MD:
Beyond MEK inhibitors, there are several new types of drugs in development for LGSOC, including novel hormonal therapy combinations and agents that target proteins on cancer cells like antibody–drug conjugates (ADCs). Approximately 40% of patients with LGSOC have high levels of folate receptor alpha (FRα) expression,14 making available ADCs such as mirvetuximab soravtansine and new ADCs targeting FRα exciting options for these patients.
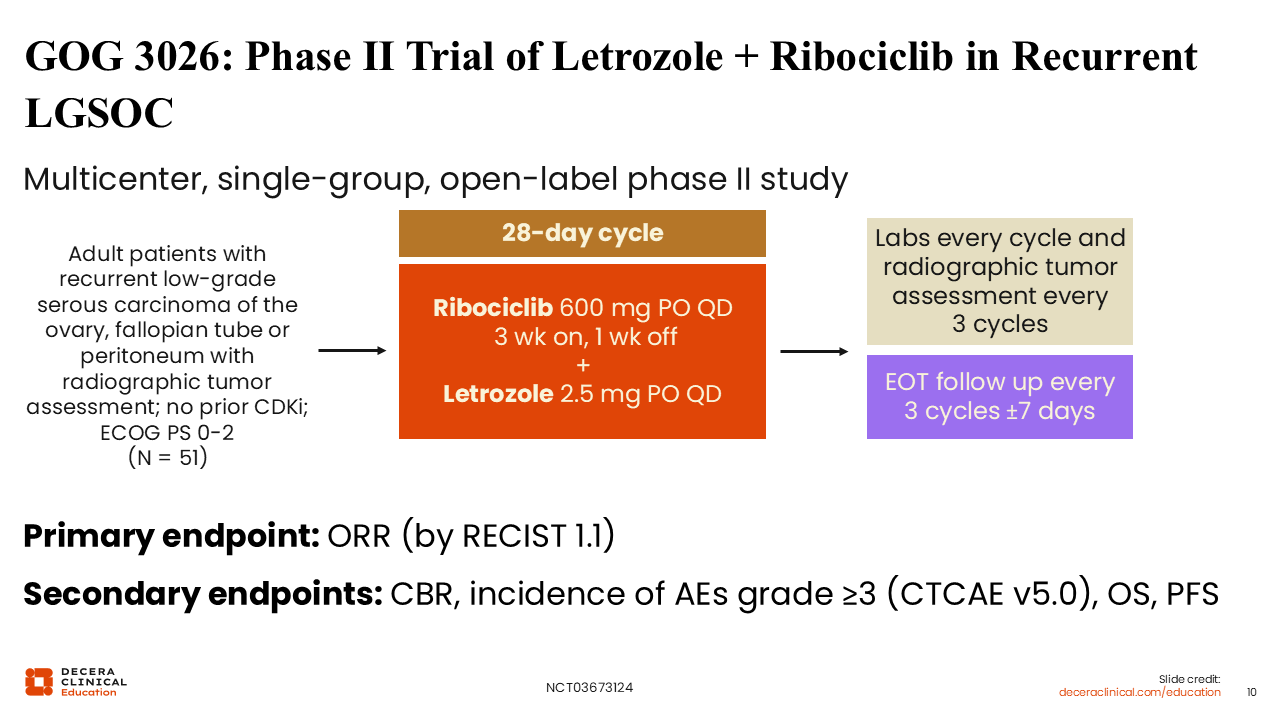
GOG 3026: Phase II Trial of Letrozole + Ribociclib in Recurrent LGSOC
Rachel Grisham, MD:
GOG 3026 is a phase II study evaluating letrozole combined with the CDK4/6 inhibitor ribociclib in patients with recurrent LGSOC who have not previously progressed on an aromatase inhibitor and not previously received a CDK4/6 inhibitor. Patients were treated with ribociclib 600 mg daily every 21 days followed by 7 days off in combination with letrozole 2.5 mg daily, and remained on treatment until disease progression or unacceptable toxicity. The primary endpoint was investigator-assessed ORR by RECIST v1.1 (NCT03673124).
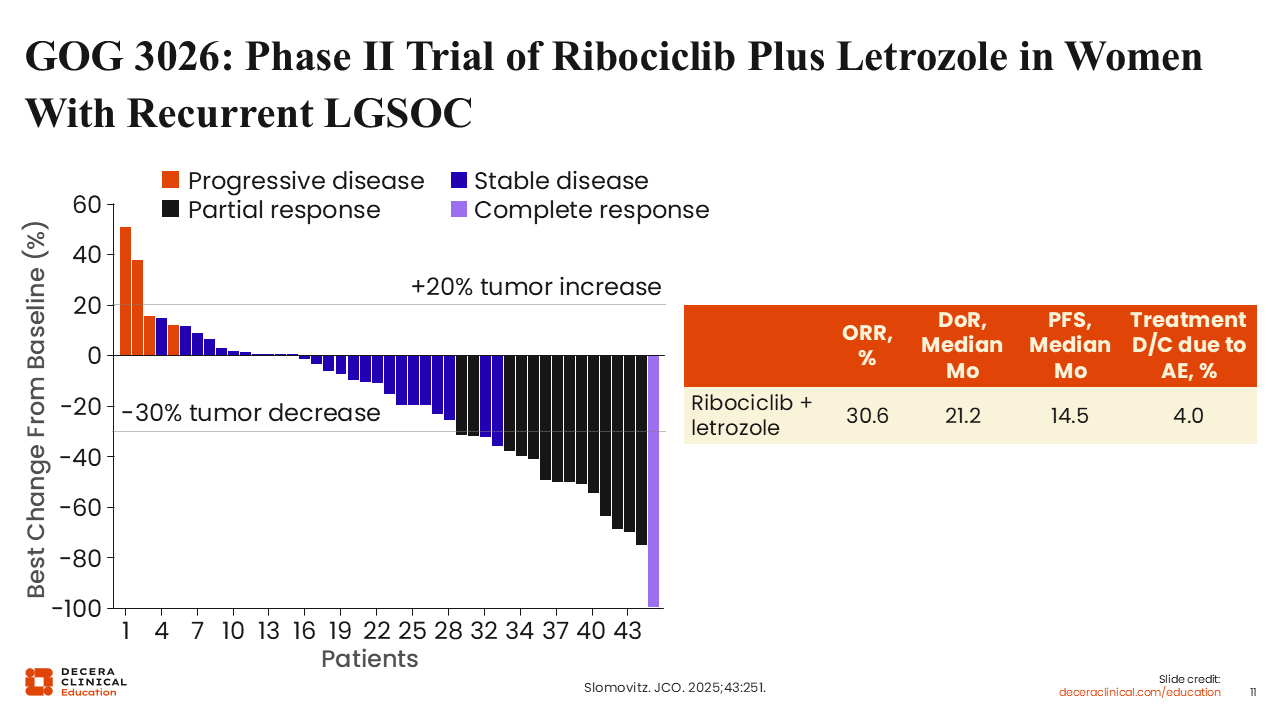
GOG 3026: Phase II Trial of Ribociclib Plus Letrozole in Women With Recurrent LGSOC
Rachel Grisham, MD:
This study showed a promising response rate of 30.6% and a median duration of response (DoR) of 21.2 months in patients treated with ribociclib and letrozole. Of note, the rate of treatment discontinuation due to adverse events (AEs) was 4%—much lower than that observed in single-agent MEK inhibitor studies, where approximately one third (34%) of patients treated with trametinib discontinued treatment due to AEs.15
BOUQUET: Phase II Biomarker-Directed Cobimetinib or Atezolizumab + Bevacizumab in Persistent/Recurrent Rare EOC
Rachel Grisham, MD:
Another study looking at targeted therapy in patients with recurrent LGSOC is BOUQUET, a platform study that is assessing multiple biomarker-selected treatments, including cobimetinib 60 mg/day 3 weeks on and 1 week off, in patients with a BRAF-, KRAS-, or NRAS-activating mutation and/or NF1 loss-of-function alteration. If there was no available biomarker, patients could be treated with atezolizumab in combination with bevacizumab (nonmatched arm). The primary endpoint was investigator-assessed confirmed ORR per RECIST v1.1.16
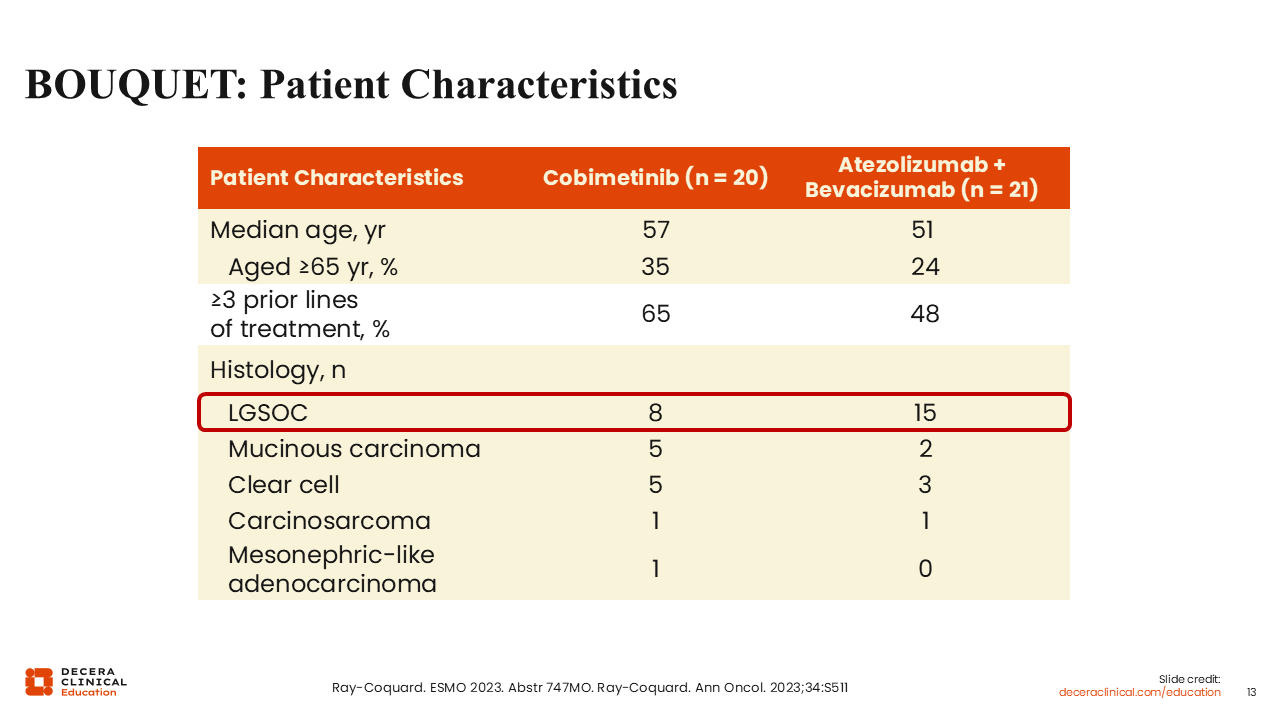
BOUQUET: Patient Characteristics
Rachel Grisham, MD:
Eight patients with LGSOC were treated with cobimetinib, and 15 were treated with atezolizumab and bevacizumab.16
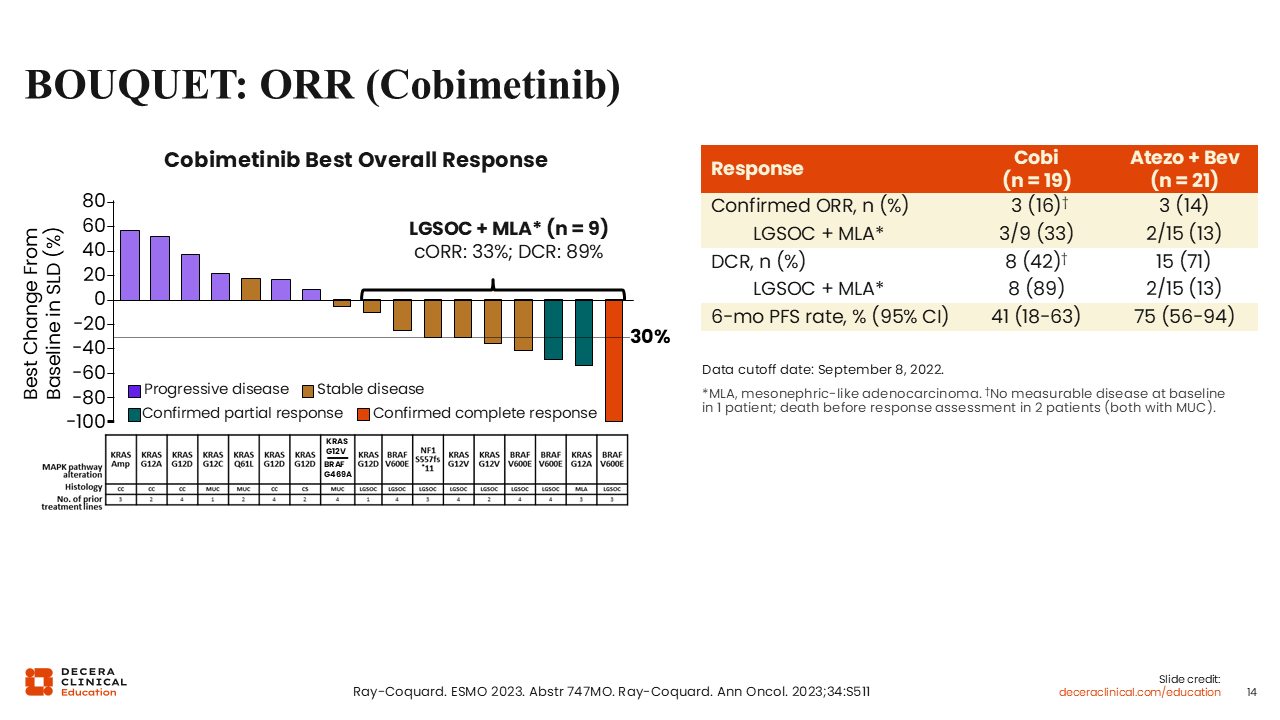
BOUQUET: ORR (Cobimetinib)
Rachel Grisham, MD:
Interim results showed a promising response rate in patients with LGSOC or mesonephric-like adenocarcinoma treated with cobimetinib (33%). It is important to note that this was a biomarker-selected population, so all patients harbored a MAPK alteration.16
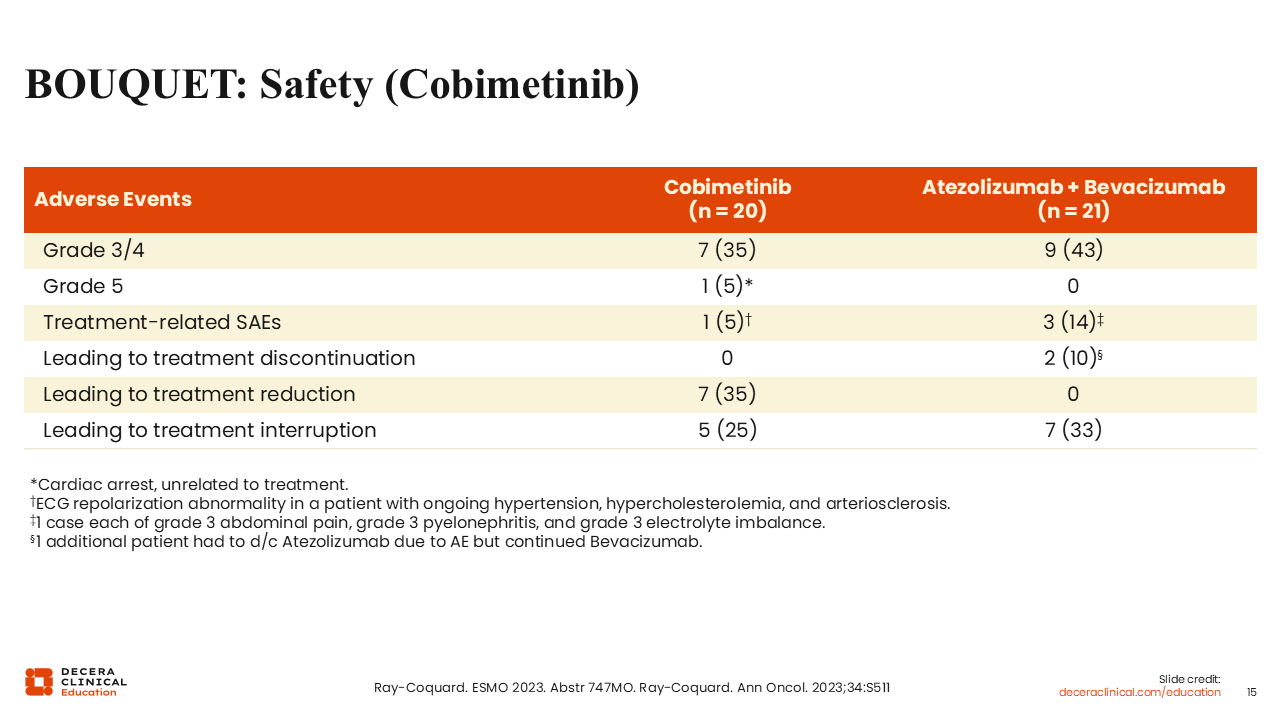
BOUQUET: Safety (Cobimetinib)
Rachel Grisham, MD:
Shown here are the AEs reported in patients on the study. Overall, cobimetinib was well tolerated. In total, 25% of patients had treatment interruption due to AEs, and 35% had treatment reduction due to AEs.16
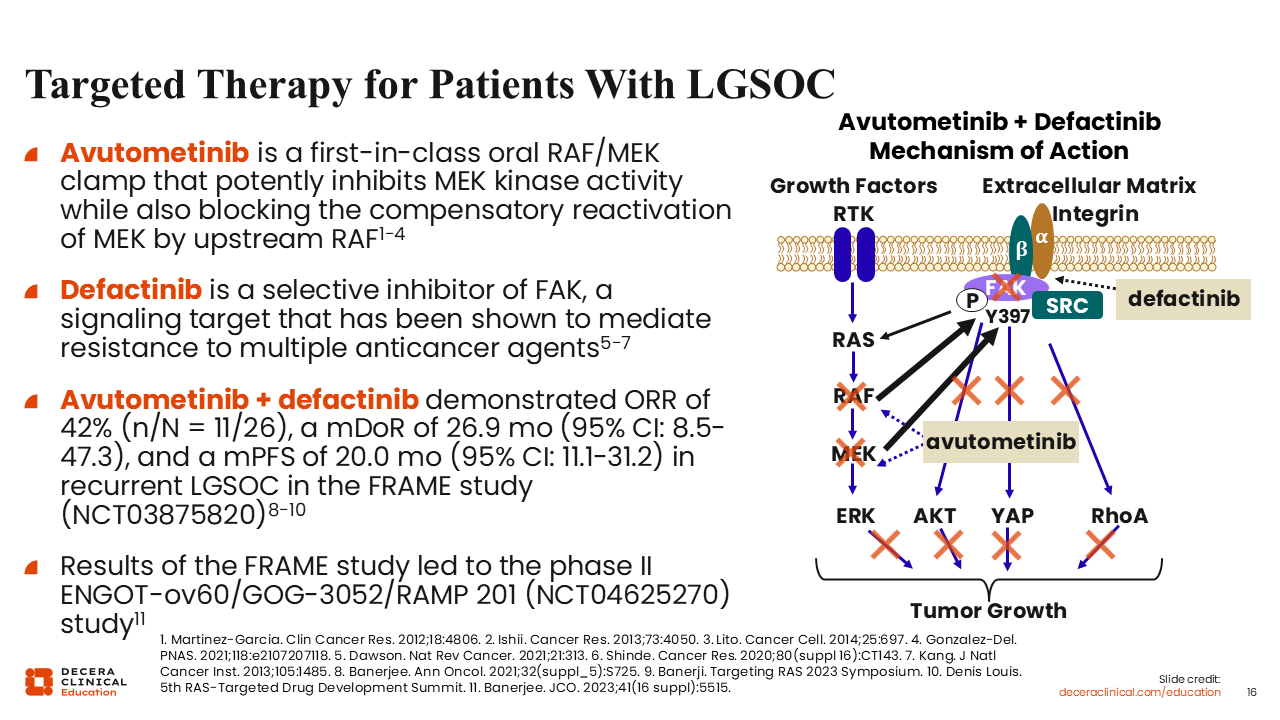
Targeted Therapy for Patients With LGSOC
Rachel Grisham, MD:
As previously mentioned, MAPK alterations are the most common drivers of LGSOC. Currently available therapies target this pathway with single-agent MEK inhibitors, but a recently approved combination therapy targets the MAPK pathway through multiple mechanisms.
Avutometinib is a first-in-class oral RAF/MEK clamp that potently inhibits MEK kinase activity while blocking the compensatory reactivation of MEK by upstream RAF. Defactinib, also an oral therapy, is a selective inhibitor of FAK, a signaling target that mediates resistance to multiple anticancer agents. The phase I FRAME study showed preliminary activity of avutometinib plus defactinib in patients with LGSOC,17 which led to the phase II ENGOT-ov60/GOG-3052/RAMP 201 trial.
ENGOT-ov60/GOG-3052/RAMP 201: Phase II Registration-Directed Trial of Avutometinib ± Defactinib in Patients With Recurrent LGSOC
Rachel Grisham, MD:
This study enrolled women with recurrent LGSOC who had received 1 or more prior line of platinum chemotherapy. Initially, patients were randomized to avutometinib 3.2 mg twice weekly in combination with defactinib 200 mg BID or avutometinib alone. Initial analysis showed a higher response rate and similar toxicity profile for patients treated with the combination (ORR: 28%) vs single-agent avutometinib (ORR: 7%), leading to the decision to move forward with combination therapy for subsequent study cohorts.
An expansion phase enrolled additional patients who were treated with avutometinib plus defactinib. Finally, a dose optimization phase looked at a lower dose of avutometinib (1.6 mg twice weekly) in combination with defactinib to determine its effectiveness.18
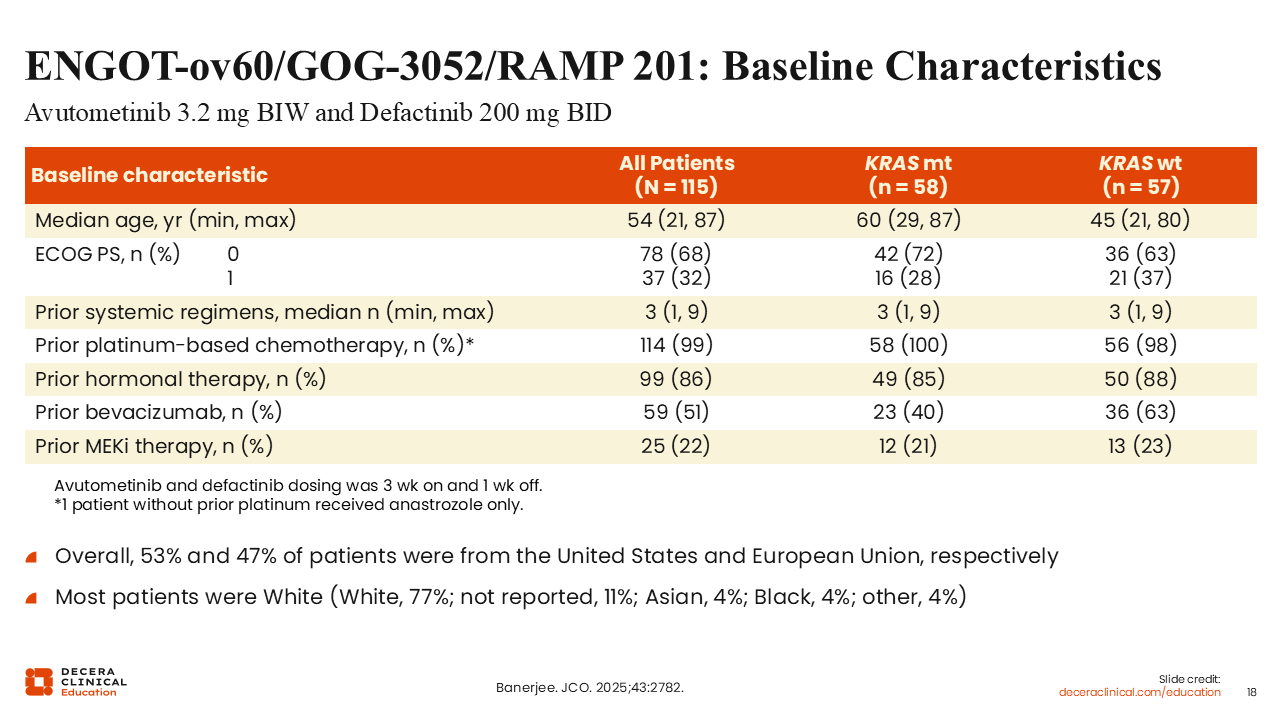
ENGOT-ov60/GOG-3052/RAMP 201: Baseline Characteristics
Rachel Grisham, MD:
The patients enrolled in this study had received a median of 3 prior lines of therapy, and 22% had received a single-agent MEK inhibitor before study entry. Just over half (51%) of the patients had previously been treated with bevacizumab, and most patients (86%) had received prior hormonal therapy.18
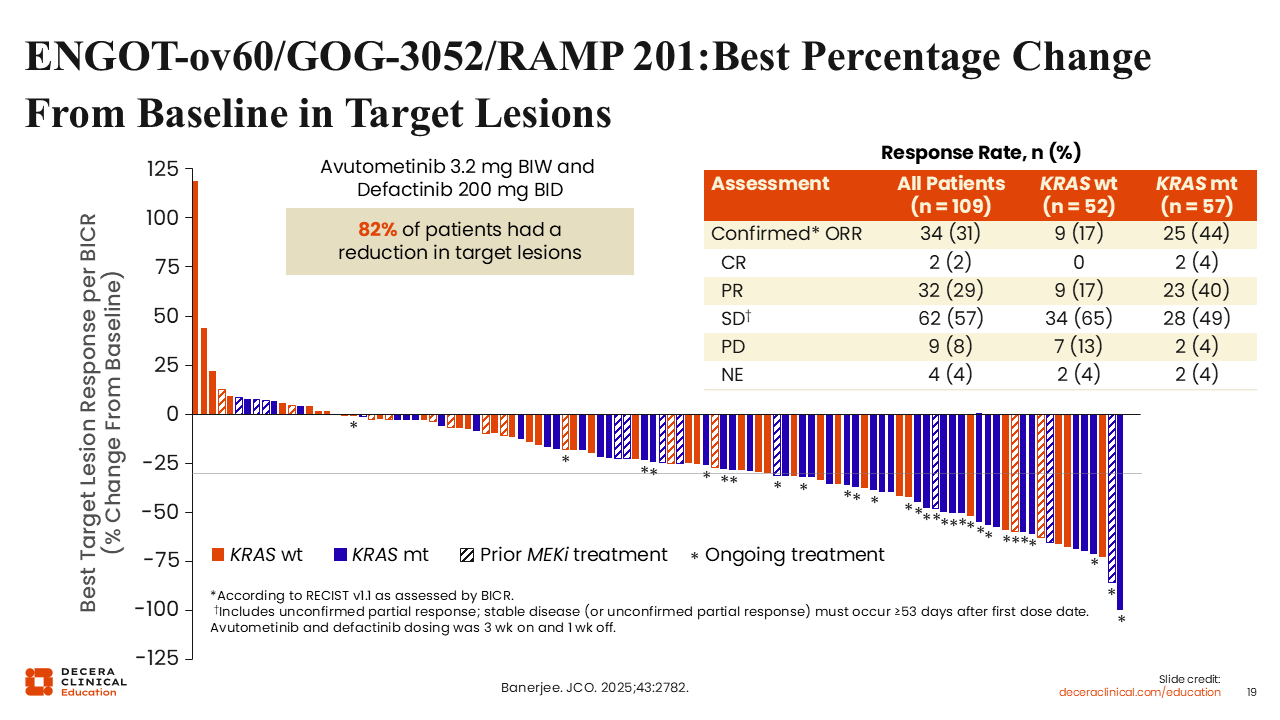
ENGOT-ov60/GOG-3052/RAMP 201: Best Percentage Change From Baseline in Target Lesions
Rachel Grisham, MD:
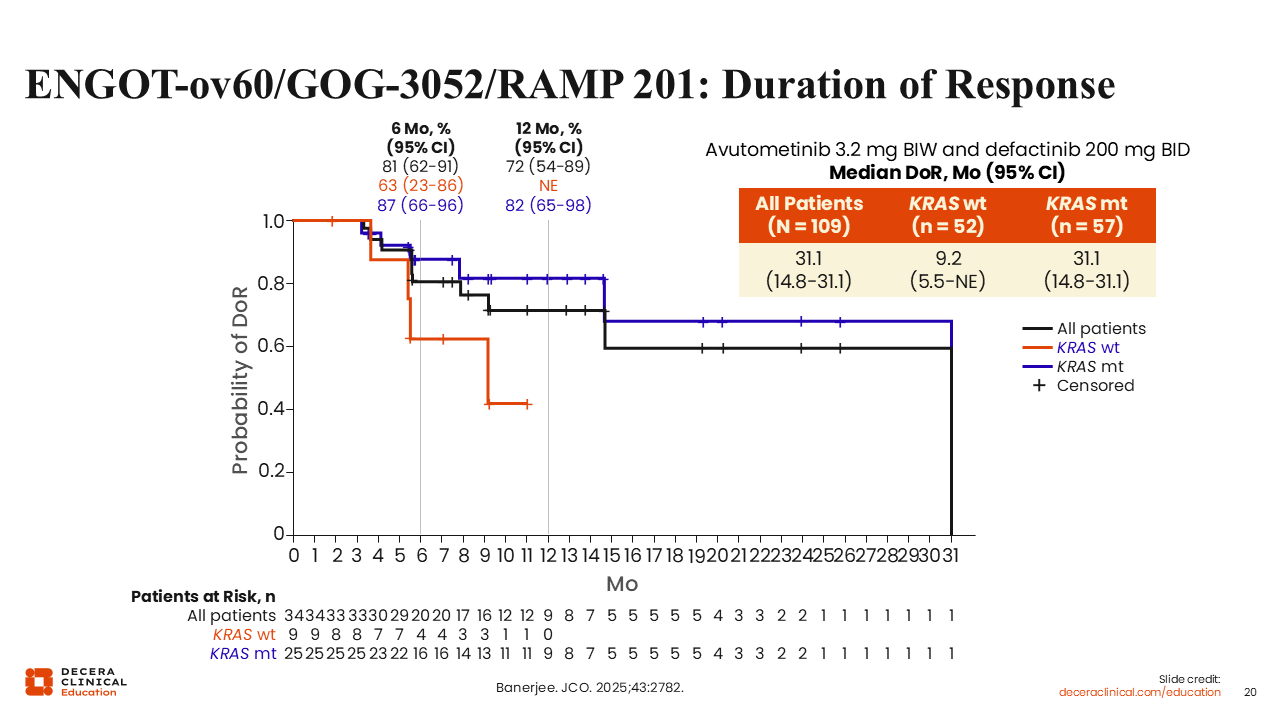
This study showed a confirmed ORR of 31% in the overall population, which is very exciting, with a response rate of 44% in patients who harbored a KRAS mutation. As expected and consistent with prior studies showing that patients with LGSOC without a KRAS mutation have lower response rates and a poor prognosis, the response rate was lower in the KRAS wild-type population (17%). However, despite this finding, 82% of patients experienced some reduction in target lesions regardless of KRAS status.18
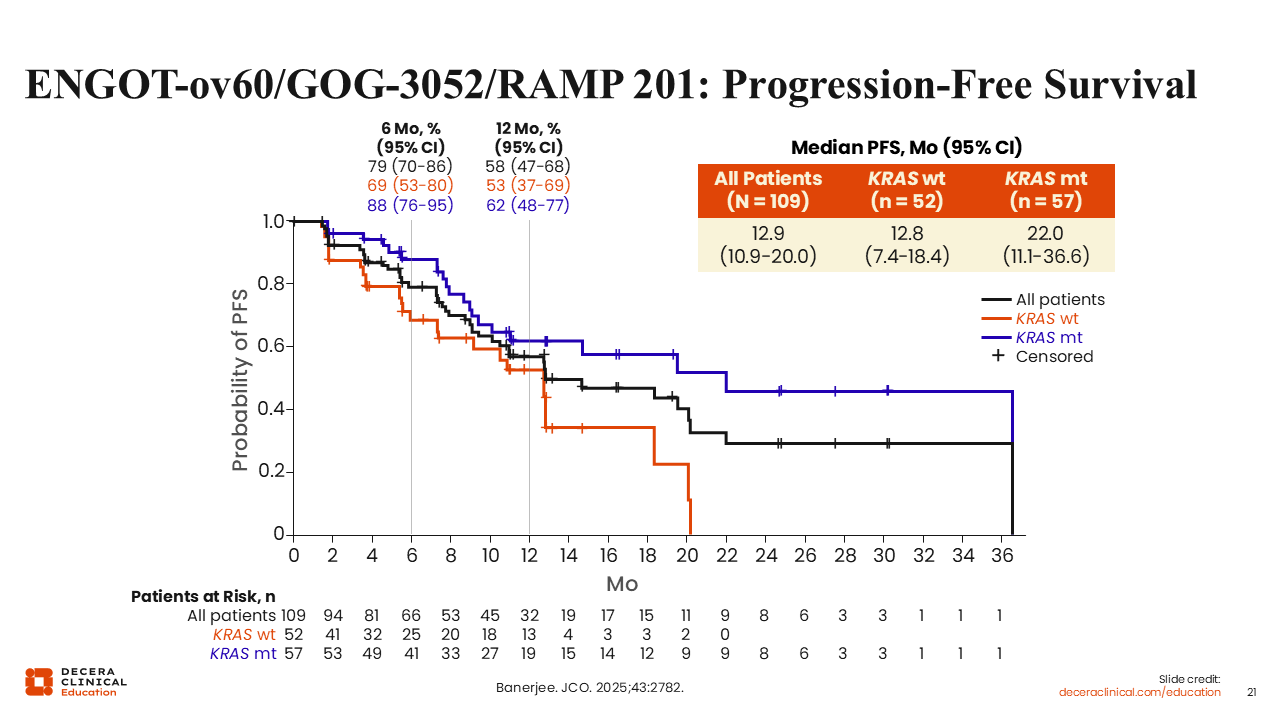
ENGOT-ov60/GOG-3052/RAMP 201: Progression-Free Survival
Rachel Grisham, MD:
The median progression-free survival was 12.9 months for all patients, 12.8 months for patients with wild-type KRAS, and 22.0 months for patients with mutant KRAS.18
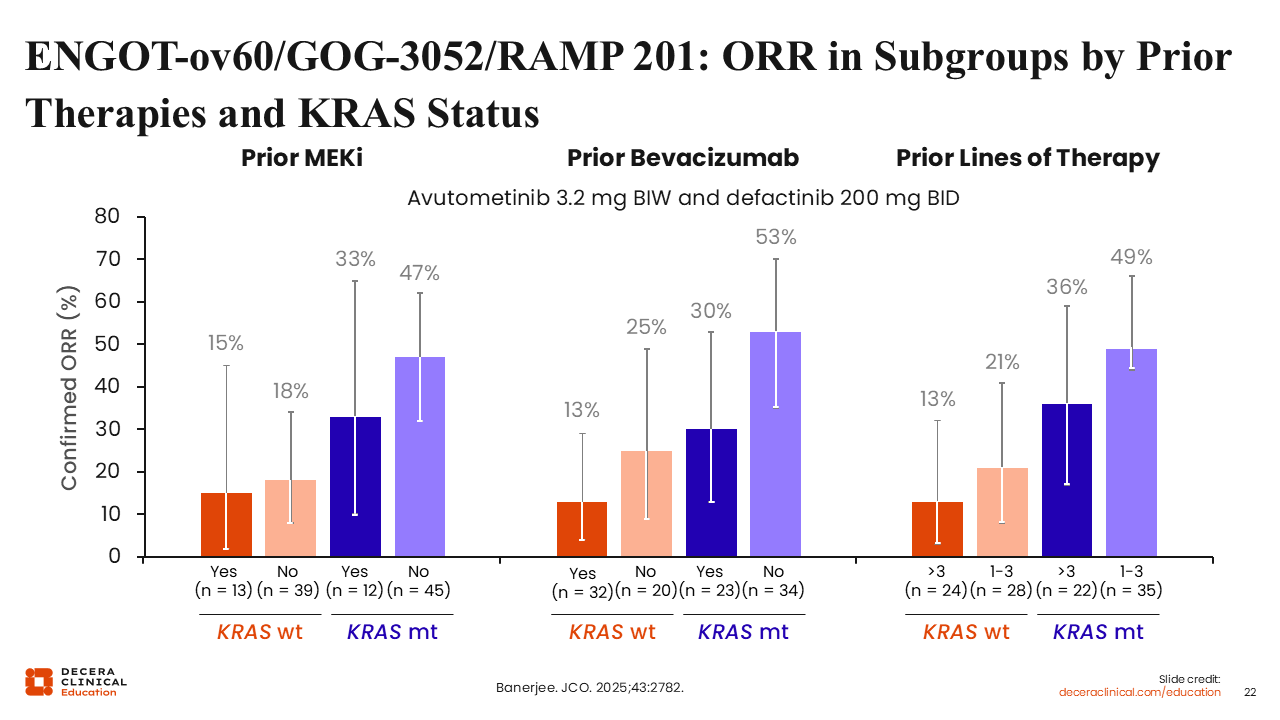
ENGOT-ov60/GOG-3052/RAMP 201: ORR in Subgroups by Prior Therapies and KRAS Status
Rachel Grisham, MD:
As one may expect, patients who were less heavily pretreated had higher response rates. For example, patients who had not received prior treatment with bevacizumab had higher response rates than those who had regardless of KRAS mutation status. It is important to note that even among patients treated with a single-agent MEK inhibitor before study entry, responses were still seen with the combination of avutometinib and defactinib. The response rate was 33% in patients with a KRAS mutation who had received a prior MEK inhibitor.18
ENGOT-ov60/GOG-3052/RAMP 201: AEs
Rachel Grisham, MD:
AEs were manageable with dose holds or reductions.
Overall, 10% of patients treated with avutometinib and defactinib had a dose reduction due to an AE. However, only 10% of patients ultimately had to discontinue combination treatment due to AEs, primarily because of increased creatine phosphokinase (CPK), a muscle enzyme that is commonly elevated when patients are treated with a MEK inhibitor or MEK inhibitor combinations. This elevation is generally asymptomatic but should be monitored in all patients on treatment to ensure there are no signs of rhabdomyolysis. Some of the most common treatment-related nonlaboratory AEs related to the combination treatment included nausea, diarrhea, peripheral edema, and rash.18
ENGOT-ov60/GOG-3052/RAMP 201: Adverse Events of Interest
Rachel Grisham, MD:
AEs of interest included blurred vision and rash. Blurred vision occurred most during the first week of treatment. Fortunately, this AE often resolved without any need for dose interruption and did not lead to treatment discontinuation. However, it is important to educate patients and their caregivers about the possibility of blurred vision and for patients to be followed appropriately by an eye care professional. Rash is also a common side effect of MEK inhibitors, including avutometinib and defactinib. This is primarily an acneiform rash occurring on the face and upper chest area. Rash occurred at a median onset of 15 days following treatment, and 6% of patients had a treatment-related skin reaction that resulted in a dose interruption or reduction.18
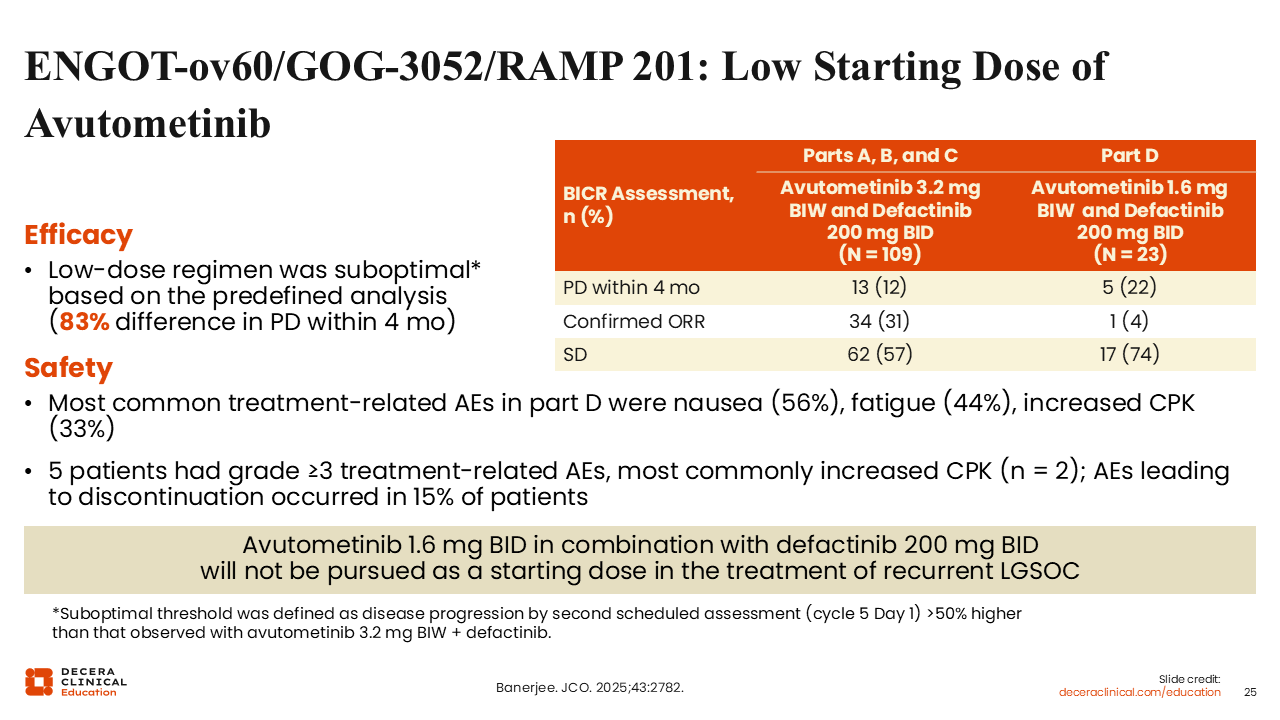
ENGOT-ov60/GOG-3052/RAMP 201: Low Starting Dose of Avutometinib
Rachel Grisham, MD:
As previously mentioned, the low-dose cohort was given avutometinib at half dose (1.6 mg) vs the standard dose (3.2 mg). Given in combination with defactinib, this dose optimization phase demonstrated that low-dose avutometinib plus defactinib was not as effective as the standard dose, with a confirmed ORR of 31% with the standard dose vs 4% with the low dose, so the decision was made to move forward with standard dosing of avutometinib and defactinib.18
Avutometinib + Defactinib AE Management Guidelines
Rachel Grisham, MD:
Based on the results of RAMP 201, a combination of avutometinib and defactinib, each kinase inhibitors, is indicated for the treatment of adult patients with KRAS-mutated recurrent LGSOC who have received prior systemic therapy. It was approved under accelerated approval based on tumor response rate and DoR. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial.
Now that this combination is commercially available, it is important that HCPs know how to best counsel patients about potential toxicities and provide them with mitigation strategies. In terms of ocular toxicity, it is advised that all patients see an eye care professional prior to starting treatment, after their first 28-day/4-week cycle, and then every 3 cycles thereafter or as clinically indicated. In clinical studies, some patients who experienced ocular adverse reactions had dose interruptions and 1 patient had a reaction that resulted in dose reduction.
To help prevent skin toxicity, it is advised that patients consider using prophylactic antibiotics. Minocycline or doxycycline is often used to help to prevent skin rash from MEK inhibitors during at least the first 2 cycles of treatment. Topical application of corticosteroids on the face, neck, and upper body can also be initiated at the start of treatment through at least the first 2 cycles. Patients should also be advised to limit unnecessary sun exposure, as UV can worsen skin rash.
Avutometinib and defactinib can also cause elevations in liver function tests (LFTs) or bilirubin. Therefore, LFTs should be monitored during treatment with this combination. For example, a 52-year-old woman with recurrent LGSOC receiving combination therapy with avutometinib and defactinib achieved a partial response. Although she reported feeling well at her appointment and denied abdominal pain, nausea, pruritus, jaundice, or fatigue, her laboratory tests revealed: ALT at 7 × upper limit of normal (ULN); AST at 5 × ULN; normal total bilirubin; and normal alkaline phosphatase. She was experiencing grade 3 transaminitis (ALT >5–20 × ULN). For this patient, we responded by holding both agents and closely monitoring liver tests. Once the patient’s transaminases improved to ≤ grade 1, then the treatment resumed at a reduced dose.
As previously mentioned, it is common to observe CPK elevations during treatment with a MEK inhibitor or avutometinib in combination with defactinib. During clinical studies, 75% of patients experienced elevated CPK and 18% had grade 3-4 elevations. Therefore, CPK should be monitored throughout treatment.19
Avutometinib + Defactinib Dosing and Modification Guidelines
Rachel Grisham, MD:
As avutometinib and defactinib are oral therapies given intermittently, it is important that patients understand the dosing schedule and have appropriate tools like a pill diary, an app, or a refrigerated pill box in place to allow them to maintain compliance. Avutometinib is taken orally twice weekly for the first 3 weeks of each 4-week cycle, whereas defactinib is taken orally twice daily for the first 3 weeks of each 4-week cycle.19
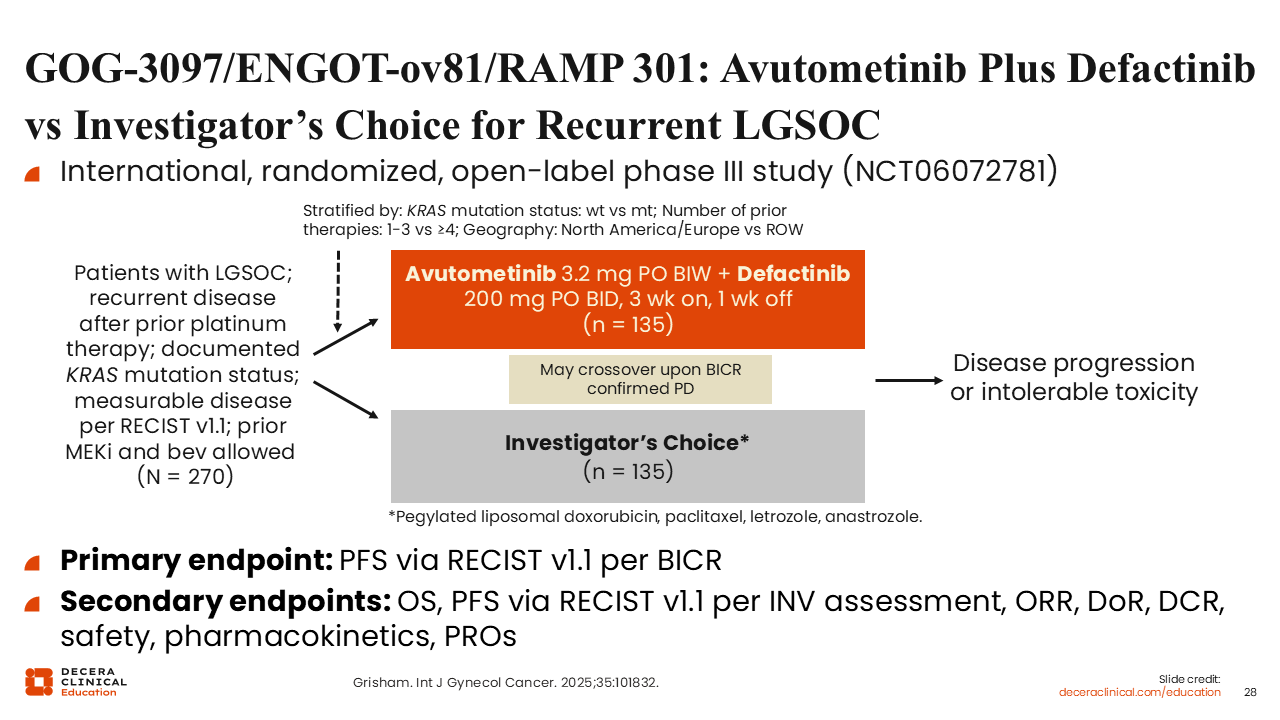
GOG-3097/ENGOT-ov81/RAMP 301: Avutometinib Plus Defactinib vs Investigator’s Choice for Recurrent LGSOC
Rachel Grisham, MD:
The ongoing phase III GOG-3097/ENGOT-ov81/RAMP 301 study is a confirmatory study wherein patients with recurrent LGSOC were randomized to either avutometinib and defactinib or SoC. We await the results of this pivotal study and are interested to see the results for patients with KRAS wild-type and KRAS-mutant LGSOC treated with avutometinib and defactinib or SoC.20
Multidisciplinary Approach to Managing TRAEs in LGSOC
Rachel Grisham, MD:
Whether we are treating patients with recurrent LGSOC with single-agent MEK inhibitors, endocrine therapy, chemotherapy, avutometinib in combination with defactinib, or an ADC, it is important that HCPs work together with the goal of providing the best quality of life and outcomes for our patients. Therefore, it is important to empower our nurses to understand the management schedule and potential toxicities of new and emerging treatments. It is also vital to engage with our consults if an ocular or dermatologic assessment is needed. Finally, a thorough review of current medications also helps allow the best outcomes possible.