CME
Risk Stratification in Polycythemia Vera and Essential Thrombocythemia: Applying Prognostic Scoring Systems in Practice
Physicians: Maximum of 0.25 AMA PRA Category 1 Credit™
Released: February 03, 2026
Expiration: August 02, 2026
Activity
Essential Thrombocythemia and Polycythemia Vera: Goals of Therapy
When we think about risk stratification in ET and PV, it is important to understand the outcomes for which we are risk stratifying, and what goals of therapy are reasonably achieved.1-3 In ET and PV, there are several complications that can develop, including thrombosis, bleeding, progression to myelofibrosis (MF) and transformation to acute myeloid leukemia (AML), and symptoms that impact quality of life, as well as death. These complications, however, are separate from one another; thrombotic risk and progression risk are different, and of most importance, therapies have differing effects on these outcomes. Ultimately, risk stratification is meaningful only if effective therapies can be tailored to each risk group. This is important to keep in mind as we review the different risk stratification tools at our disposal.
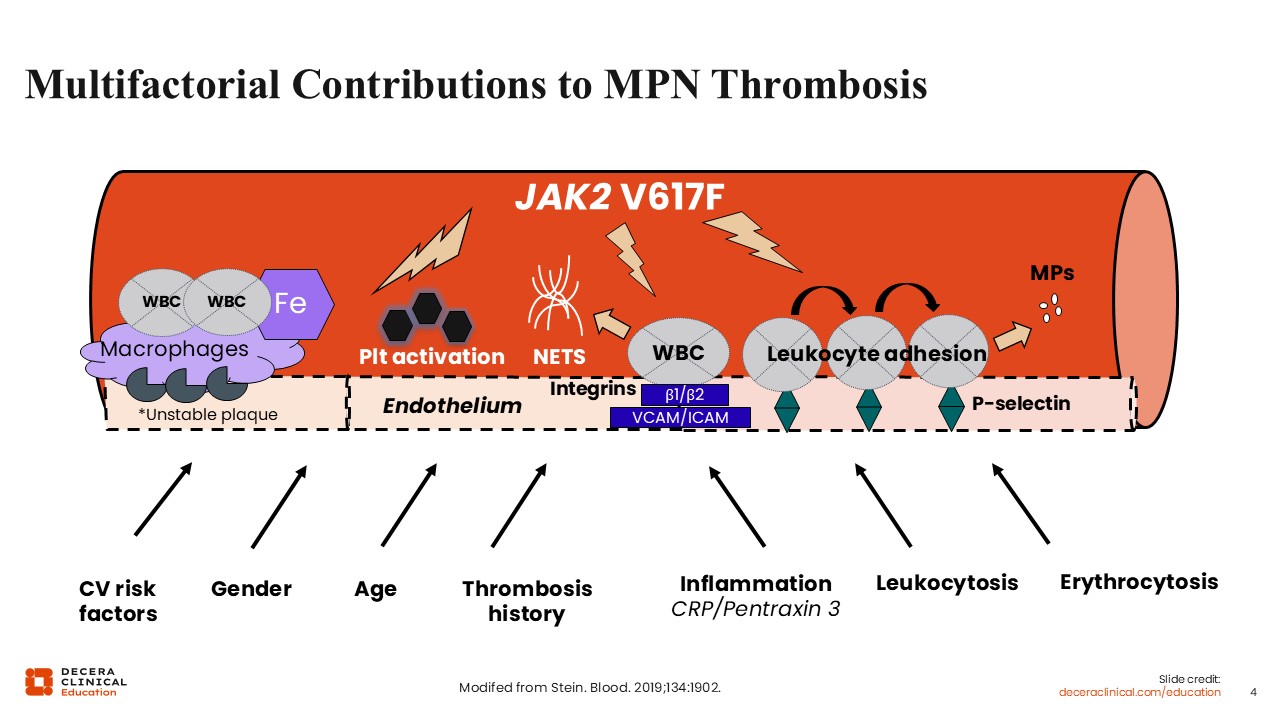
Multifactorial Contributions to MPN Thrombosis
Patients with myeloproliferative neoplasms (MPNs) have a 3- to 4-fold increased risk of arterial thrombotic events and a 10- to 12-fold increased risk of venous thrombotic events at diagnosis.4-6 Thrombosis is the most common cause of mortality in patients with PV and ET and, of most importance, something that can be treated, so most risk stratification tools with clinical utility center around thrombosis.
This slide summarizes the pathophysiology of thrombosis in MPNs.7 At the core is upregulated JAK-STAT signaling from the JAK2 V617F mutation, which drives a prothrombotic state through multiple mechanisms. JAK2 signaling promotes platelet activation, enhancing aggregation and thrombus formation, while also increasing neutrophil extracellular trap (NET) formation, which further supports thrombogenesis.7,8 Mutant leukocytes exhibit enhanced adhesion to the endothelium, mediated via integrins and adhesion molecules such as VCAM and ICAM, and elevated P-selectin expression, facilitating interactions between platelets, leukocytes, and endothelial cells. The vascular environment is influenced by systemic and local factors. Endothelial dysfunction is amplified by conventional cardiovascular risk factors (eg, hypertension, dyslipidemia) and sex-specific contributions. Inflammation, reflected by elevated C-reactive protein and pentraxin-3 levels, potentiates leukocyte activation and endothelial adhesion. Leukocytosis, a hallmark of MPNs, directly correlates with thrombosis risk, reinforcing the importance of myeloid cell burden. Erythrocytosis, particularly in PV, increases blood viscosity and shear stress, further promoting thrombotic events.
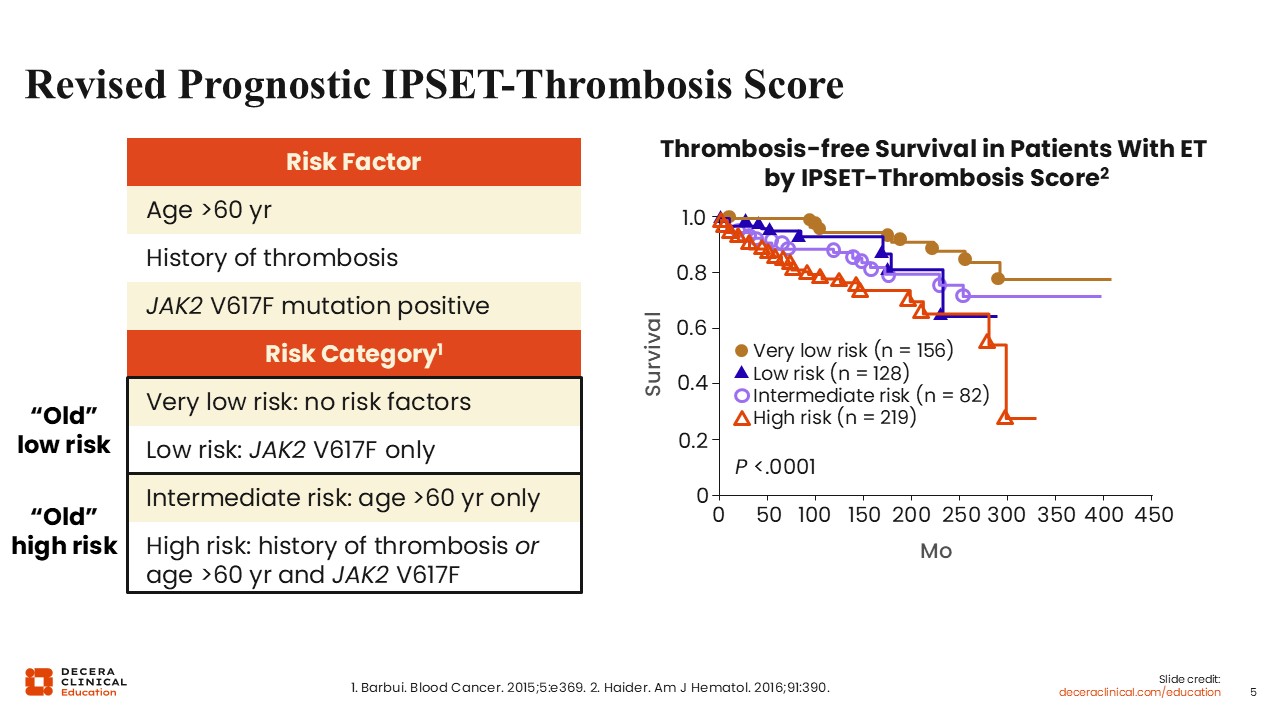
Revised Prognostic IPSET-Thrombosis Score
The revised IPSET is a standard risk stratification tool for thrombosis in patients with ET.9 The original version included older age, thrombosis history, JAK2 mutation, and cardiovascular risk factors.10 Re-analysis of the data used to establish the IPSET found that cardiovascular risk factors added no prognostic value.9 Thus, the revised version includes 4 risk categories based on 3 risk factors: age older than 60 years, history of thrombosis, and presence of a JAK2 mutation. Patients at very low risk are young without a thrombotic history or a JAK2 mutation; this includes, for instance, very young asymptomatic patients with a CALR mutation. Patients at low risk are younger with a JAK2 mutation and no thrombotic history. Patients at intermediate risk are older without a JAK2 mutation and no thrombotic history. Finally, patients at high risk are older with a JAK2 mutation or a thrombotic history regardless of age. The R-IPSET was validated, with thrombotic rates corresponding to approximately 1% per year in the very-low or low-risk group and up to approximately 4% per year in the high-risk group.11
Revised Prognostic IPSET-Thrombosis Score: Management
The R-IPSET helps determine how to treat ET to reduce the risk of thrombosis.2,9 All patients are generally treated with aspirin, although some data suggest that in patients at very low risk, aspirin does not significantly reduce thrombotic risk and may increase bleeding; therefore, observation is a reasonable option in this risk category. Cytoreduction is generally recommended in patients who are at high risk for thrombosis and can be omitted in patients who are at low risk. In patients who are at intermediate risk for thrombosis, cytoreduction may be more controversial and should depend on other risk factors present such as diabetes, hypertension, hyperlipemia, obesity, and smoking status.
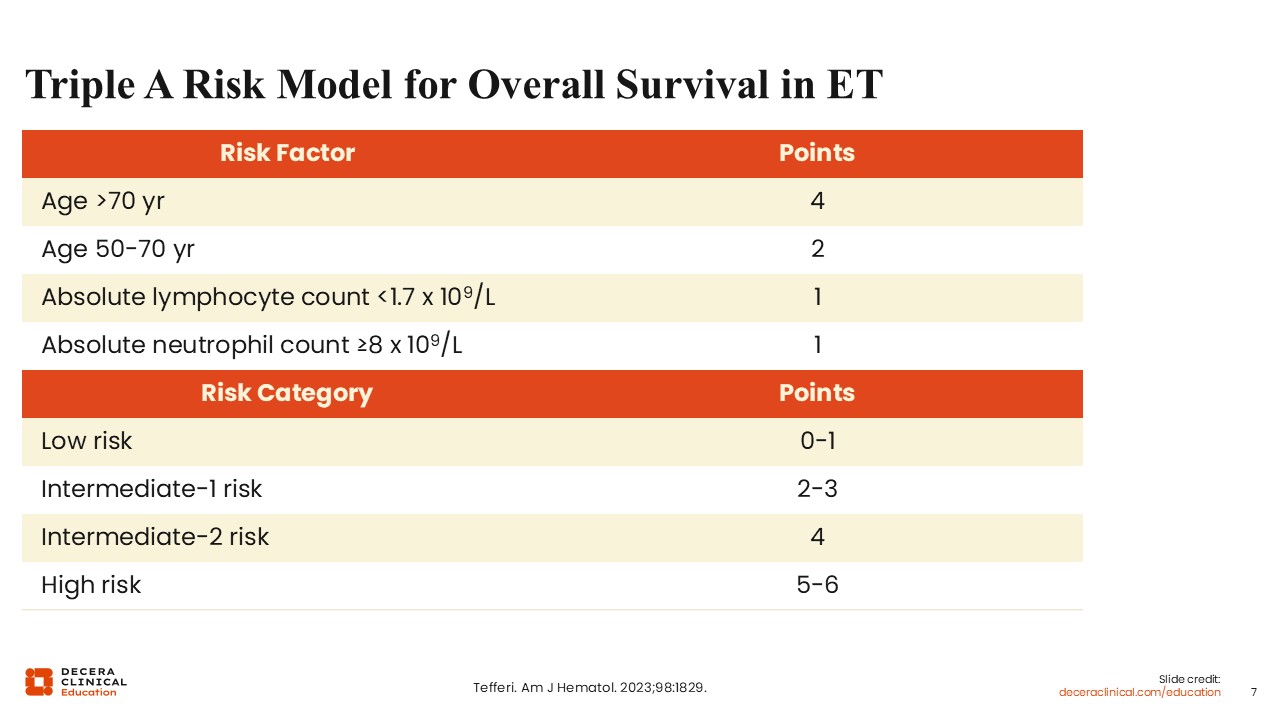
Triple A Risk Model for ET
Other risk stratification tools use overall survival (OS) as the primary outcome. The “triple A” (AgeAncAlc) risk model in ET is 1 such model.12 This model incorporates hematologic parameters that reflect immune and myeloid cell balance, such as absolute lymphocyte (ALC) and neutrophil counts (ANC), and are independently associated with survival in patients with ET.
The triple A model assigns points based on 3 variables: age, ALC, and ANC. Age contributes the most, with age older than 70 years assigned 4 points and age 50-70 years assigned 2 points. ALC <1.7 x 10⁹/L and an ANC ≥8 x 10⁹/L each contribute 1 point. Total points are used to stratify patients into 4 risk categories: low (0-1 points), intermediate-1 (2-3 points), intermediate-2 (4 points), and high (5-6 points).
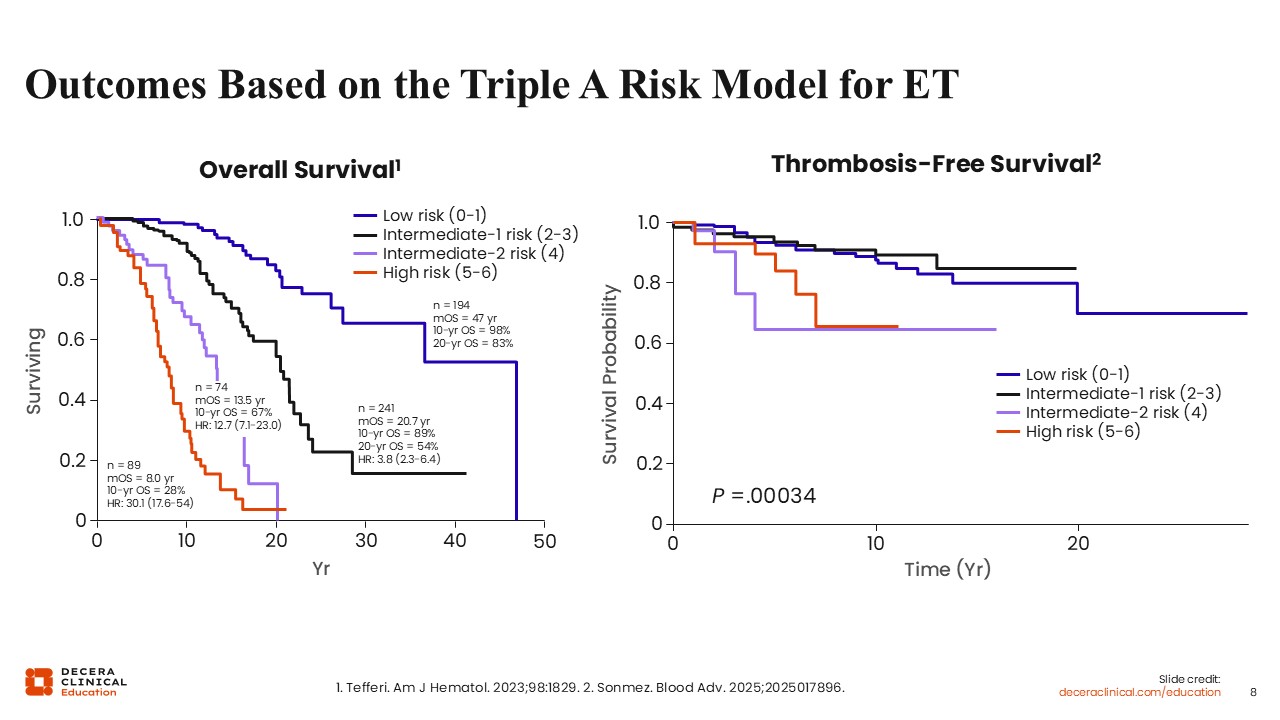
Outcomes Based on the Triple A Risk Model for ET
The triple A model results in median OS values of 47 years, 20.7 years, 13.5 years, and 8.0 years for low, intermediate-1, intermediate-2, and high risk groups, respectively among the discovery patient set.12 The model was validated in a second group of patients with similar results.
The triple A model was validated in an external cohort and also demonstrated prognostic significance for thrombosis-free survival.13 Patients at low risk experienced the highest probability of thrombosis-free survival over time, with minimal events. Intermediate-1 risk was associated with a modest reduction in survival probability. Intermediate-2 and high risk were associated with more rapid declines in thrombosis-free survival, with the high-risk group having the worst outcomes, particularly within the first 5-10 years. The triple A score was not found to discriminate bleeding or myelofibrosis progression risk, and monocytosis was identified as an adverse risk factor that could be incorporated into future iterations of this risk model.
Overall, the triple A model is a simple risk stratification tool that incorporates only age and complete blood count parameters. However, it is relatively new, and there is some uncertainty regarding how to structure therapeutic algorithms around its risk categories.
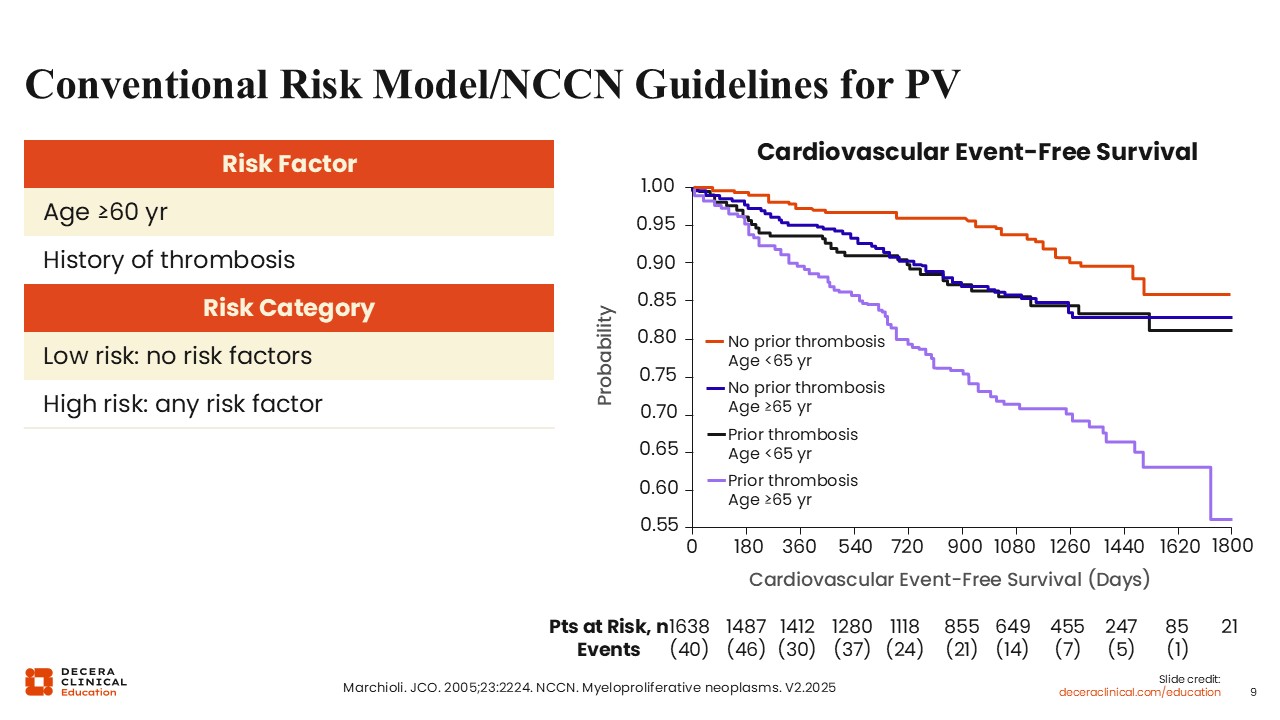
Conventional Risk Model/NCCN Guidelines for PV
The equivalent of the revised IPSET in PV is the conventional thrombotic risk stratification established by Marchioli and colleagues in 2005.14 This model is also utilized in expert consensus guidelines such as those established by the National Comprehensive Cancer Network (NCCN).15 Patients are classified into 2 principal risk groups based on 2 clinical factors: age 60 years or older and a history of thrombosis. Patients who are younger than 60 years of age and have no thrombosis history at diagnosis are considered low risk, and those who meet either or both criteria are considered high risk.
In Kaplan–Meier analyses comparing thrombotic outcomes between these 2 conventional risk groups, patients considered high risk demonstrated markedly lower thrombosis‑free survival over time (10.9 cardiovascular events per 100 persons per year in those with a history of thrombosis and age ≥65 years), whereas patients considered low risk had higher rates of cardiovascular event‑free survival (2.5 cardiovascular events per 100 persons per year).14
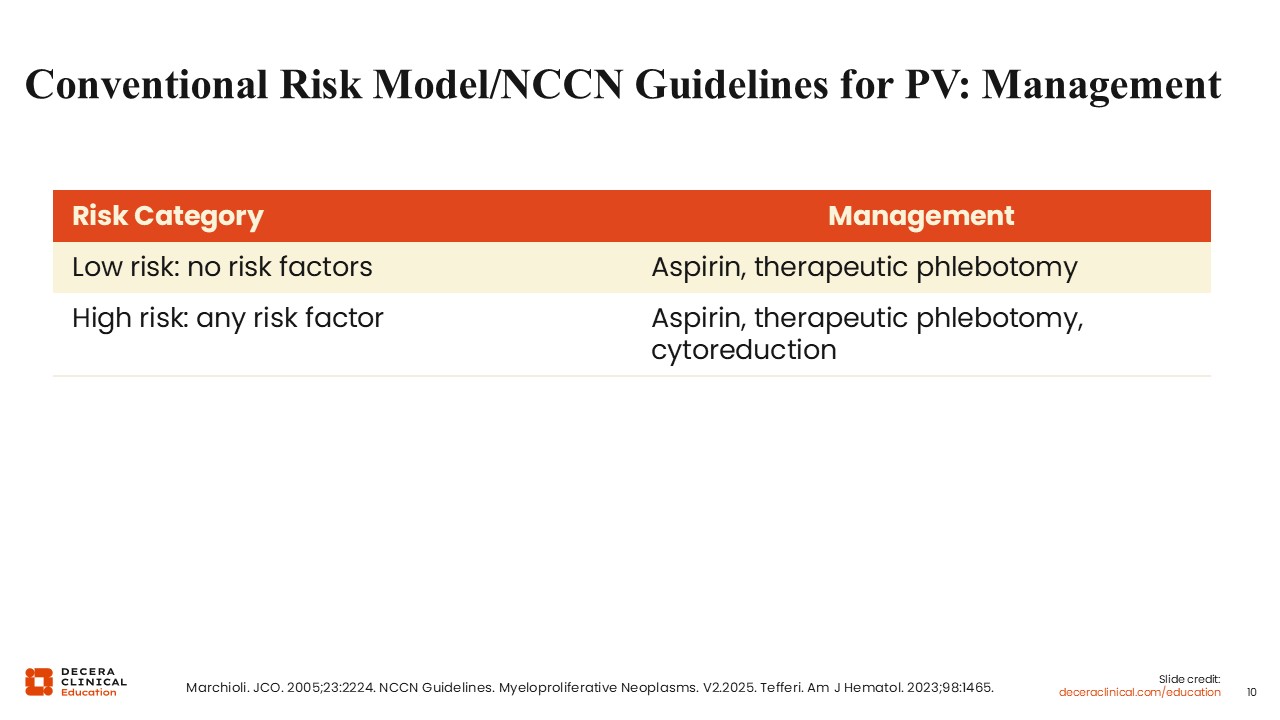
Conventional Risk Model/NCCN Guidelines for PV: Management
The conventional risk model for PV is used to guide therapy.1,15 Thrombosis remains the most common cause of morbidity and mortality, and patients with PV are at especially high risk for thrombosis. All patients with PV should be treated with aspirin and maintain a hematocrit <45%. The latter recommendation is based on clinical trial data demonstrating superior thrombotic-free survival in patients who maintained an aggressive (hematocrit <45%) goal compared to a liberal (hematocrit 45%-50%) goal.16 For those at low risk (ie, young patients without a history of thrombosis), this could be done through therapeutic phlebotomy. However, in those at high risk, cytoreduction is recommended to keep counts at goal. Cytoreduction provides even more control of hematocrit than periodic phlebotomy and can also reduce leukocytosis, which has been linked to poorer thrombosis-free survival and thrombocytosis.17,18
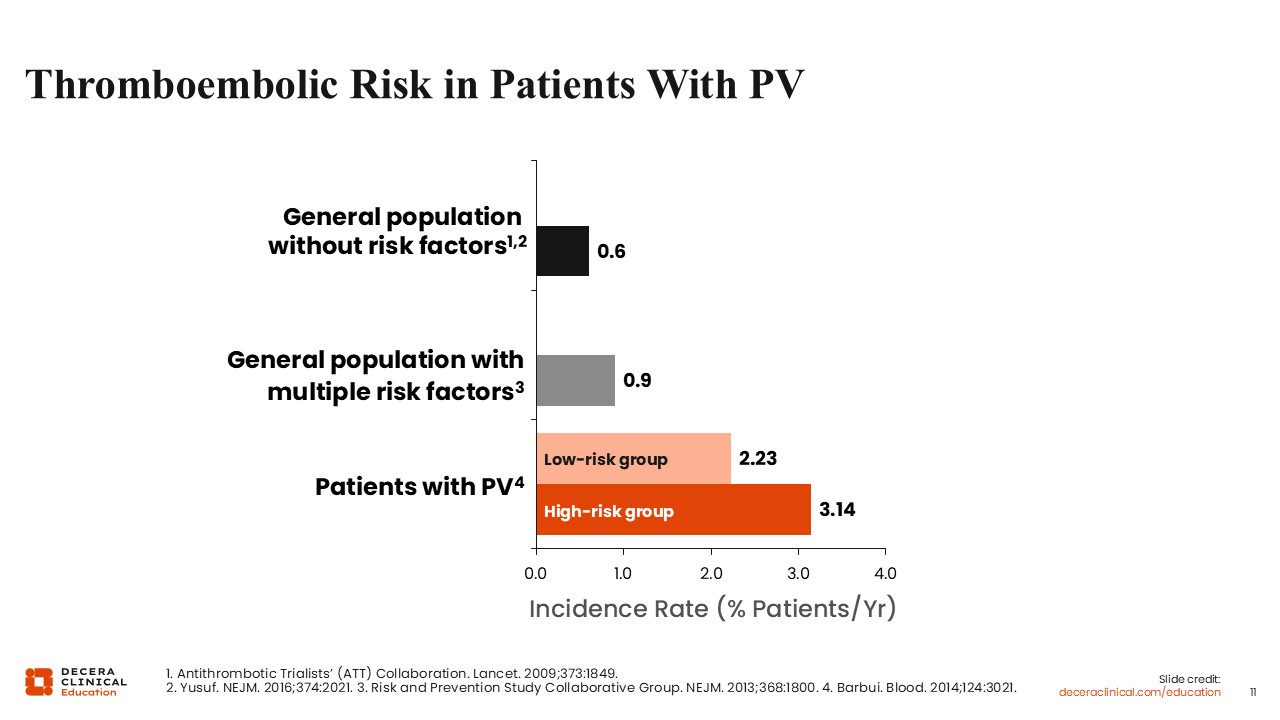
Thromboembolic Risk in Patients With PV
This slide highlights the elevated thromboembolic risk experienced by patients with PV. The incidence rate of thromboembolism in the general population is approximately 0.6% of patients per year19,20 and increases to 0.9% in the general population with multiple cardiovascular risk factors.21 In contrast, patients with PV are at significantly increased thrombotic risk: 2.23% per year among those considered low risk and 3.14% among those considered high risk (ie, those who are older or have a history of thrombosis).22 Again, treatments can reduce the risk of thrombosis in PV, so risk stratification has clinical utility in preventing thrombotic events in patients without overtreatment.
Additional Considerations for Thrombotic Risk
I consider these risk stratification models a starting point. Age is an arbitrary value and nothing magical happens when someone turns 61. For example, a 65-year-old without any other medical problems looks very different from a 55-year-old who smokes, has diabetes, high blood pressure, and is overweight. Although many risk stratification tools do not accurately capture the more complex medical comorbidities, they are still important to consider. Cardiovascular risk factors such as smoking, hypertension, hyperlipidemia, diabetes, and obesity do increase mortality risk substantially in patients with MPNs as well as thrombotic risk.23 The presence (or absence) of these risk factors may influence how aggressive I am with cytoreduction and can be a more powerful determinant than age.
Similarly, blood counts may influence decisions to start cytoreduction, as they can predict thrombotic risk. As discussed previously, there is good evidence for keeping the hematocrit <45% in patients with PV. Leukocytosis has repeatedly emerged as a significant risk factor for thrombosis; however, unlike hematocrit, there are no clear data specifying a target WBC count in these patients. Of interest, thrombocytosis has not emerged consistently as a risk factor for thrombosis, and I tend to be less aggressive with lowering platelet counts as a result. Extreme thrombocytosis, as defined as a platelet count >1.5 million, has been associated with an increased risk of bleeding due to acquired von Willebrand factor deficiency and may warrant cytoreduction to prevent bleeding more than thrombosis.1 Mutation profile also impacts thrombotic risk. A higher JAK2 variant allele frequency, which is more often seen in PV-like phenotypes, has been shown to increase thrombotic risk.24 Other pathogenic nondriver mutations may also increase thrombotic risk, particularly arterial thrombotic risk. Data suggest that patients with clonal hematopoiesis of indeterminate potential (CHIP), particularly with TET2 or DNMT3A mutations, have a 2- to 4-fold increased risk of coronary artery disease, likely due to mutated leukocytes producing proinflammatory cytokines and activating endothelial cells and platelets, thus promoting arterial plaque instability and endothelial dysfunction.25 As these mutations are also commonly found in patients with MPNs, they can also increase thrombotic risk.
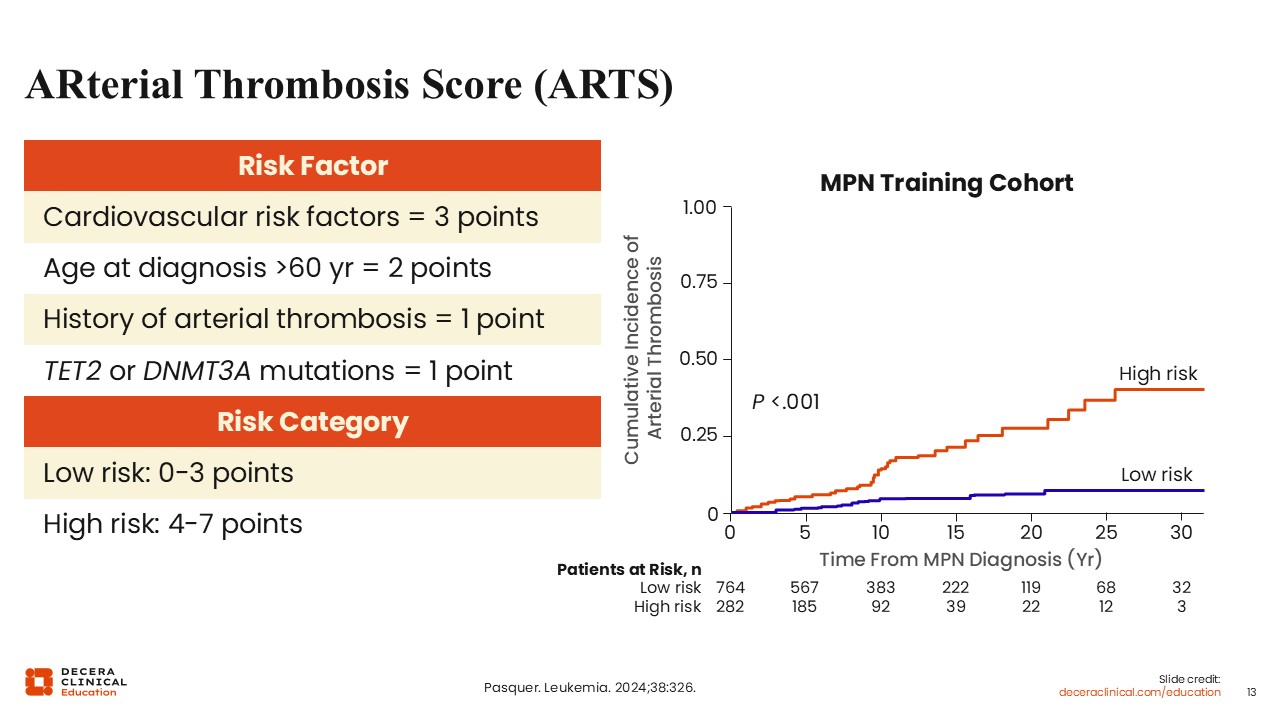
ARterial Thrombosis Score (ARTS)
The ARterial Risk Score (ARTS) tries to capture some of these clinical variables and was created specifically to estimate the risk of arterial vs venous thrombosis in patients with ET and PV.26 It was developed from a large, long‑term cohort, and multivariable analysis revealed 4 factors independently associated with arterial thrombosis: prior arterial thrombosis, age older than 60 years, traditional cardiovascular risk factors, and the presence of TET2 or DNMT3A mutations. In the ARTS, each factor contributes to a numeric score reflecting cumulative risk. Patients are stratified into low‑risk and high‑risk groups based on their total ARTS, with individuals considered low risk having a substantially lower annual incidence of arterial thrombosis (0.37% of patients per year) vs those considered high risk (1.19% of patients per year). The ARTS outperformed the traditional 2‑tier conventional thrombotic stratification (age and thrombosis history) across MPN subtypes and was validated in independent external cohorts, demonstrating improved discrimination for arterial events. However, this is a relatively new risk stratification system that requires further validation in other MPN cohorts and ideally more investigation regarding whether treatment strategies such as cytoreduction can ameliorate high-risk groups. The use of molecular testing in this model for nonpathogenic driver mutations may also limit its utility in more resource-scarce settings, particularly as comprehensive next-generation sequencing (NGS) beyond driver mutations is not universally recommended in ET or PV.
The authors also investigated risk factors for venous thrombosis and found only age and history of venous thrombosis as significant.
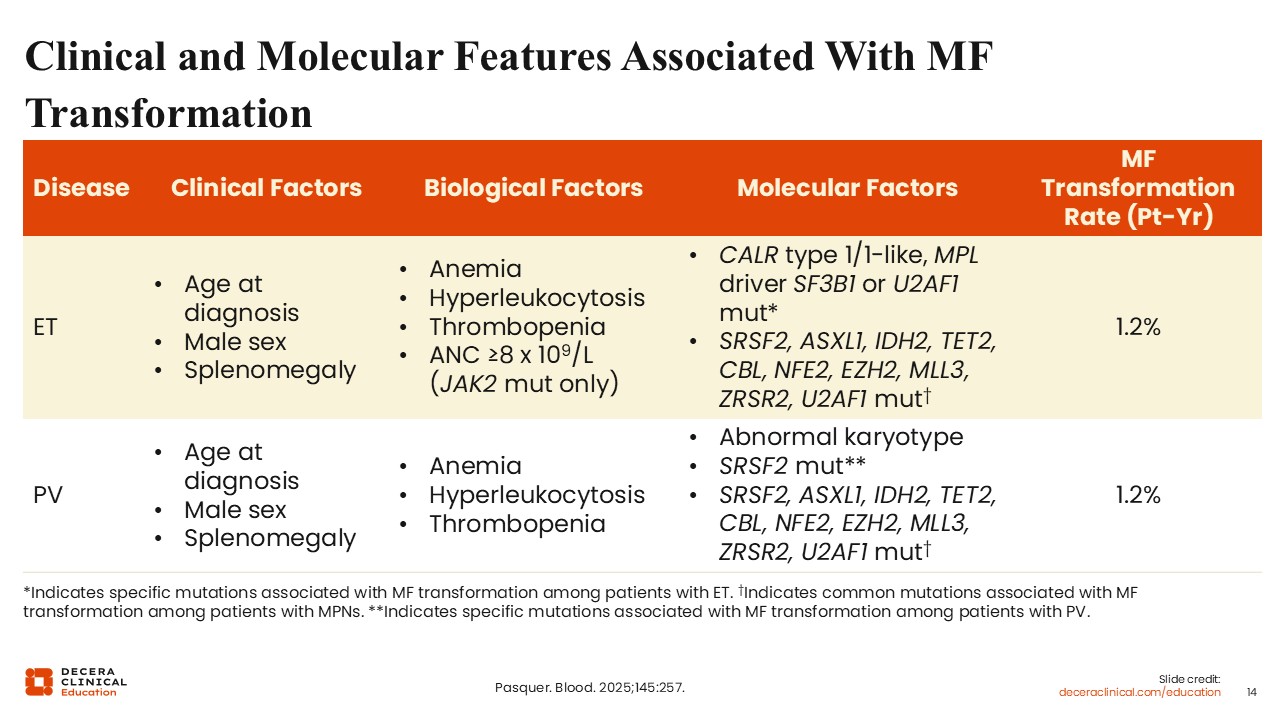
Clinical and Molecular Features Associated With MF Transformation
Although thrombosis is an important outcome that physicians often disproportionately value in treatment algorithms, primarily because it is a complication that can be mitigated, patients are often most concerned about progression. Multiple studies have looked at risk factors for MF transformation in ET and PV. These include clinical factors such as older age, male sex, and the presence of splenomegaly; biological risk factors such as cytopenias and leukocytosis; and, of most importance, molecular risk factors. CALR type 1 mutations may increase fibrotic progression although patients with MF who harbor CALR type 1 are considered lower risk; the presence of other pathogenic nondriver mutations listed in this table have also been variably associated with an increased rate of MF transformation.2
There are multiple problems with centering risk stratification tools around MF transformation, primarily because there are not yet definitive therapies that decrease the risk of progression. Patients can be categorized as higher risk, but the field has not yet reached the point where therapies can be tailored to effectively prevent progression. In addition, progression risk depends on molecular factors, and widespread NGS testing in ET and PV is not universally established.
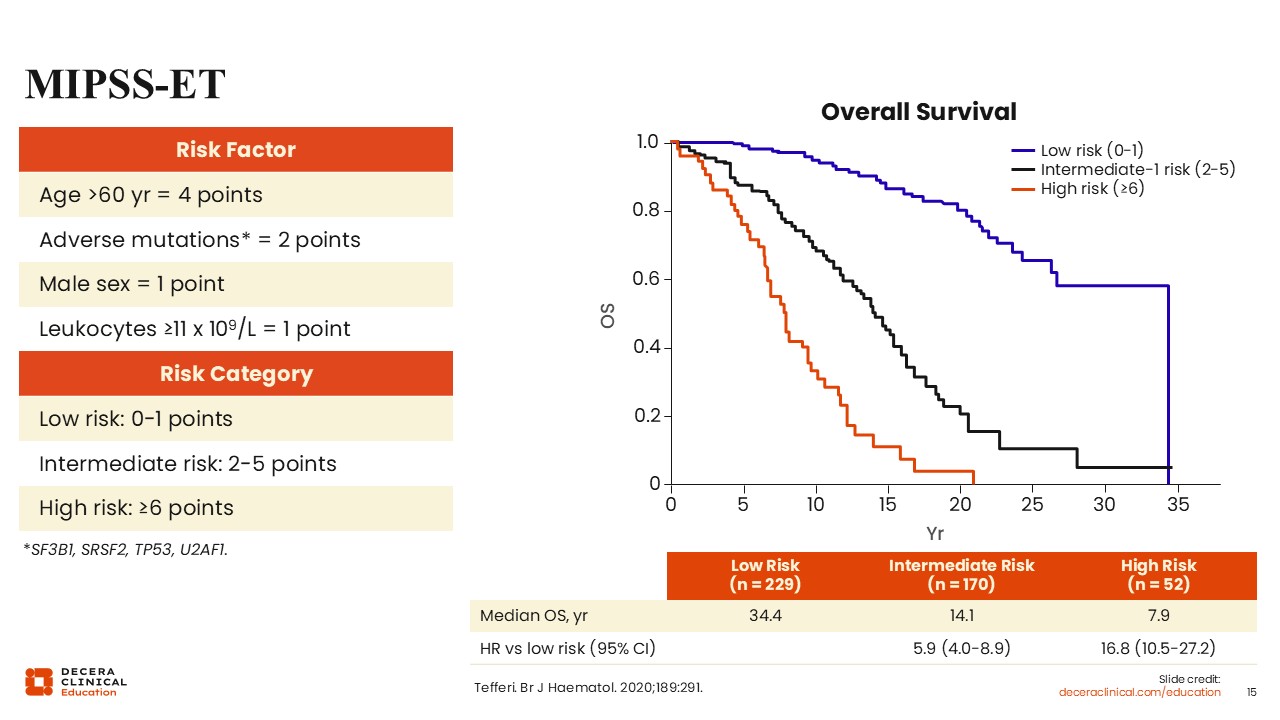
MIPSS-ET
It is possible to develop risk stratification tools around progression, which parallels some of the risk stratification tools used in MF. Chief among them are the mutation-enhanced international prognostic system for ET (MIPSS-ET) and MIPSS-PV, which integrate clinical and molecular risk factors to better predict OS and disease progression.27 Both models were derived from a cohort of over 900 molecularly annotated patients from the Mayo Clinic and University of Florence.
In the MIPSS‑ET, adverse prognostic factors and their point weights include age older than 60 years (4 points), mutations in SRSF2, SF3B1, U2AF1, or TP53 (2 points), male sex (1 point), and leukocytes ≥11×10⁹/L (1 point). Patients are stratified into low (0-1 points), intermediate (2-5 points), and high (≥6) risk, which reliably distinguishes groups with significantly different median survival durations (median OS: 34.4 years, 14.1 years, and 7.9 years, respectively).
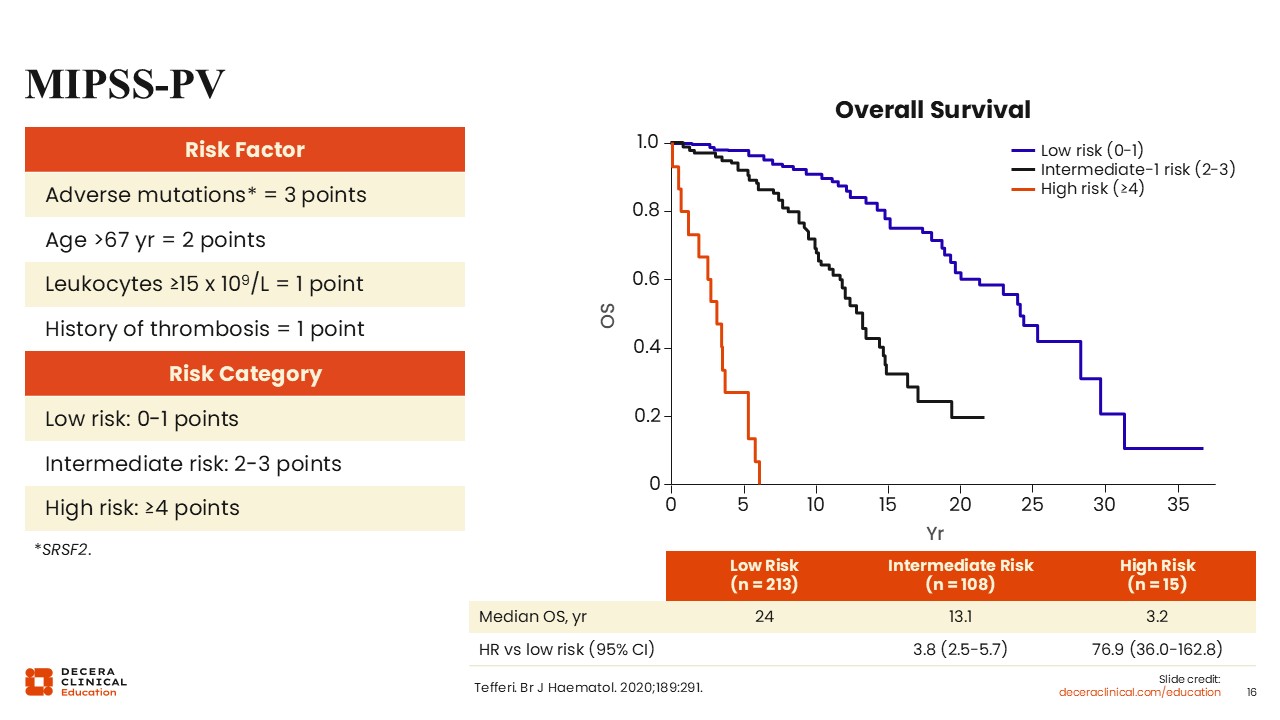
MIPSS-PV
In the MIPSS‑PV, 4 variables are independently associated with worse survival: age older than 67 years (2 points), leukocyte count ≥15 x 10⁹/L (1 point), history of thrombosis (1 point), and presence of an SRSF2 mutation (3 points).27 Patients are categorized as low‑risk (0-1 points), with prolonged survival, intermediate‑risk (2-3 points), with moderate OS, and high‑risk (≥4 points), with markedly reduced survival (median OS: 24 years, 13.1 years, and 3.2 years, respectively).
However, further studies are needed to validate the MIPSS, as there is uncertainty about how these results can be integrated into therapeutic algorithms.
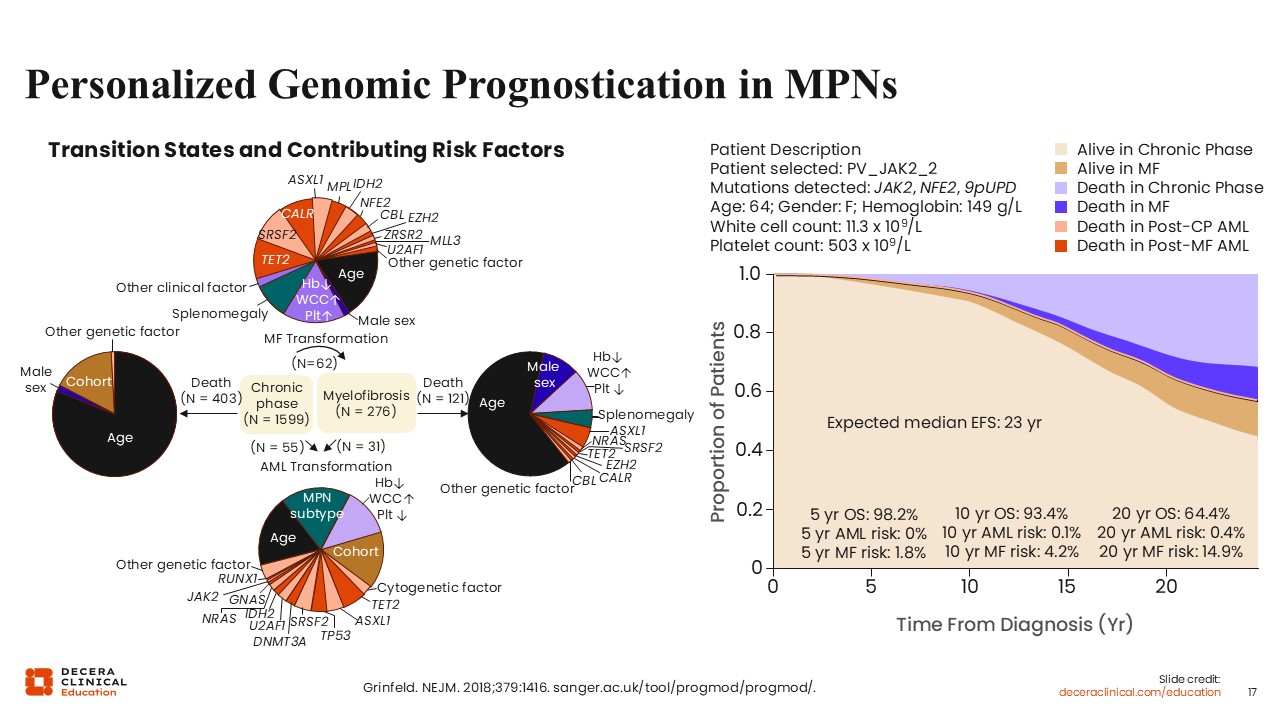
Personalized Genomic Prognostication
In 2018, Grinfeld and colleagues presented a novel framework for genetic prognostication in MPNs by integrating somatic mutations with clinical variables to model disease evolution, survival, and transformation risks at the individual patient level.28 Using large, well-annotated cohorts of patients with PV, ET, and MF, the authors showed that specific driver mutations (eg, those in JAK2, CALR, MPL), additional high-risk mutations (eg, ASXL1, SRSF2, EZH2, IDH1/2, TP53), and age/clinical features jointly shape prognosis, rather than diagnosis alone. Their models therefore use probabilistic, time-dependent predictions of outcomes such as MF or AML transformation and death. For instance, the figure on the left uses pie charts to depict transition states (chronic phase → MF → AML → death) and quantifies the relative contribution of age, sex, blood counts, splenomegaly, and individual mutations to each transition. Age dominates mortality risk, while specific mutations (eg, those in ASXL1, SRSF2, EZH2, IDH2) disproportionately influence progression to MF or AML.
The schematic on the right summarizes observed patient trajectories and competing risks across disease phases. Using an online tool, clinical and molecular variables are input for a given patient (for instance, a patient with PV and JAK2, NFE2, and 9pUPD; age 64 years; hemoglobin 14.9 g/L; white cell count 11.3 x109/L; platelets 503 x 109/L), and an individualized prognostic summary, including disease trajectory in chronic phase, then in MF phase, and risk of death or transformation to AML, is calculated based on the models of Grinfeld et al.28 Currently, these models are used for research purposes, not clinically, but they demonstrate the potential for individual risk stratification for outcomes outside of thrombosis.
Risk Stratification in ET and PV
In summary, many risk stratification tools have been developed and are still being developed.3 However, risk stratification is limited primarily by what current treatments can do. As most treatments have data to support preventing thrombosis, thrombotic risk stratification tools have the most practical implications. Established tools such as the R-IPSET, however, can still be improved upon as we learn how to integrate clinical and molecular data.9 Risk stratification for survival and progression will become more important as new treatments with disease-modifying potential emerge, leading to an exciting possibility where treatments can be tailored to patients at highest risk and possibly prevent MF progression. Genomic models may also be limited by the availability of NGS testing, but again, as the field and treatments evolve, there will also be emerging rationale for molecular testing in ET and PV, not just MF. Finally, the idea of individualized prognostication may ultimately be the best form of risk stratification and represents an area where further development is needed.