CE
APP Perspectives: Safe and Effective Use of Bispecific Antibodies in Multiple Myeloma
Physician Assistants/Physician Associates: 0.50 AAPA Category 1 CME credit
Nurse Practitioners/Nurses: 0.50 Nursing contact hour
Released: November 17, 2025
Expiration: May 16, 2026
Activity
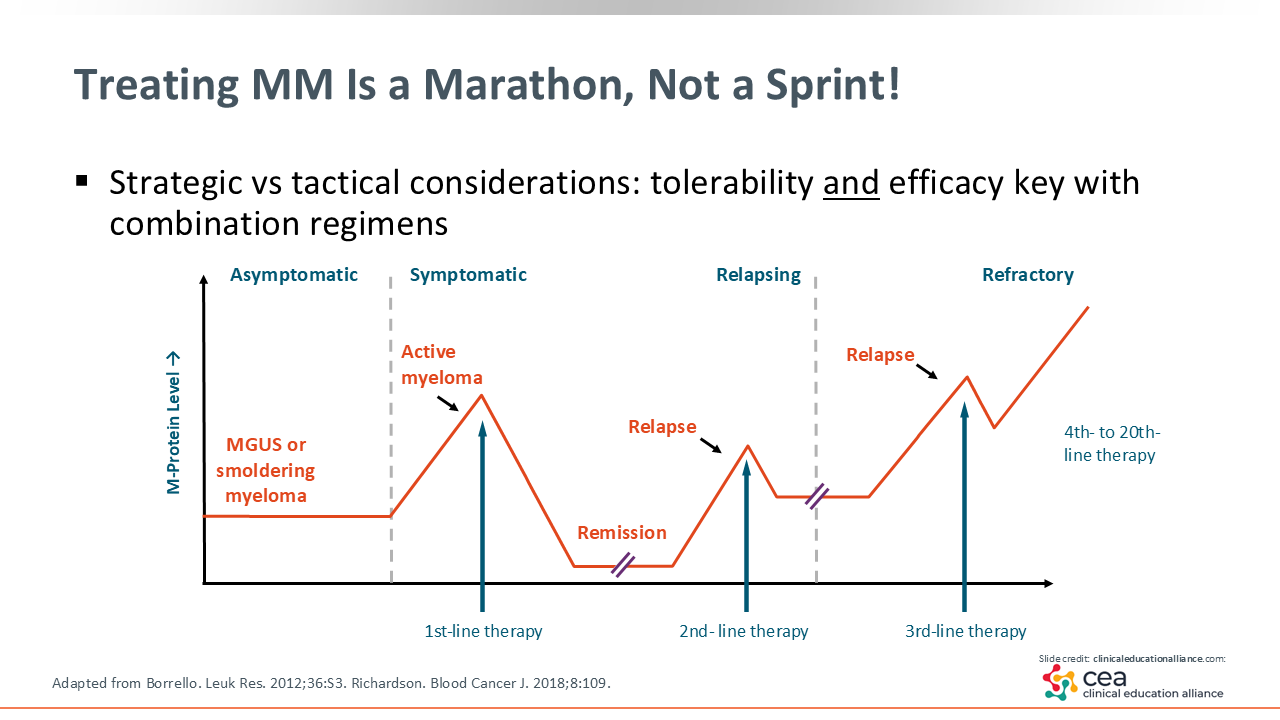
Treating MM Is a Marathon, Not a Sprint
With new treatment options extending survival for patients with MM, advanced practice providers are often at the center of helping patients navigate what has become a long and often complex treatment journey: Treating MM is a marathon, not a sprint. As the disease remains incurable, management often resembles that of a chronic condition, with treatment decisions evolving over many years.8
Before diving into newer therapies like bispecific antibodies, it is helpful to take a step back and review how MM treatment traditionally has been approached, beginning with first-line therapy for symptomatic disease and progressing through subsequent lines as patients relapse or become refractory to prior treatments.
Treatment of MM typically begins with first-line therapy for symptomatic patients. Prior to symptom onset, almost all cases of MM begin as either monoclonal gammopathy of undetermined significance (MGUS) or smoldering MM. Patients may stay at MGUS or smoldering MM for years or even through their lifetimes without developing symptomatic MM.
For those who do transform to active, symptomatic MM, first-line therapy typically involves induction therapy followed by autologous stem cell transplant in eligible patients. Although this method can produce a durable response for years, most patients eventually experience relapse events, requiring second-line therapy. Those who relapse again will move on to third-line therapy and subsequently progress to treatment with multiple lines of therapy as their disease becomes refractory.9,10
Definition of R/R Myeloma
What does it mean to have R/R MM? The International Myeloma Working Group defines this as disease that is nonresponsive to therapy or progress within 60 days of the last line of treatment. Primary refractory MM refers to cases where disease is unresponsive to initial therapy and patients never achieve a minimal or better response. Relapsed MM describes cases where disease becomes nonresponsive to a chosen line of therapy after a patient experiences minimal or better response.9,11 When we think about treatment goals for our patients in the relapsed setting, the first goal is always to reestablish disease control. With that, we are hoping to prolong overall survival and maintain and improve quality of life.
What does this mean for us in practice? It means we always need to think a few steps ahead, knowing what options are available next, preparing patients for potential transitions in therapy, and reinforcing that even if one treatment stops working, there is always a next step in their journey.
When to Initiate Therapy at Relapse
Each case of relapse is a little different, and not every patient will need to start treatment right away. Some patients experience what we call biochemical relapse, which means there is evidence of disease progression based on an increase in M-protein levels but no new symptoms or MM-related organ dysfunction.
In these cases, if patients are standard risk and asymptomatic with a low disease burden, then it is often reasonable to continue their current maintenance regimen or keep them on observation with close monitoring. The key is to watch closely for changes that might signal disease acceleration.
On the other hand, if patients are high risk, develop new or worsening symptoms, or show signs of rapid progression, then we need to start thinking about adjusting their treatment plan or initiating a new line of therapy.12,13 The goal is to stay one step ahead by recognizing when observation is safe and when it is time to act.
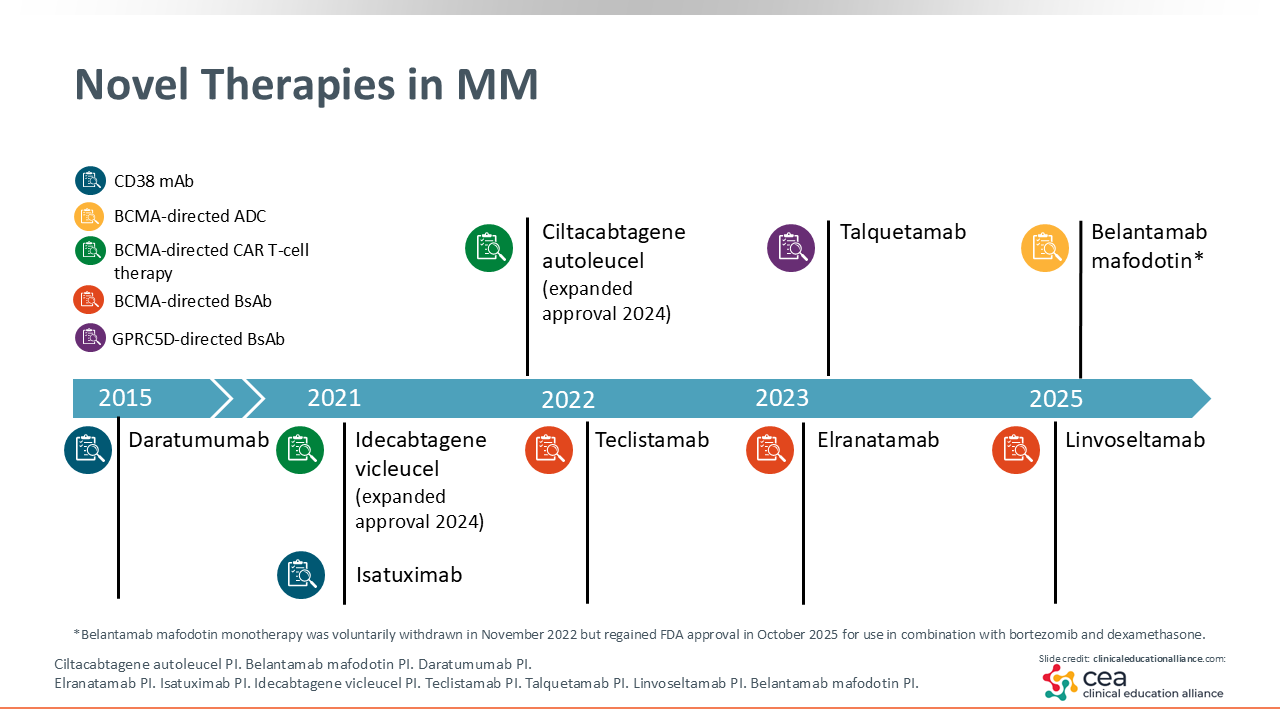
Novel Therapies in MM
We are fortunate in the MM space to have seen an incredible amount of progress in a short period of time. Over the past 3 years, more new drugs have been approved for MM than for any other tumor type.
In that time, 4 bispecific antibodies have received approval for use in the relapsed or refractory setting, starting with teclistamab in 2022, followed by elranatamab, talquetamab, and linvoseltamab1-4 We have also seen the approval of 2 CAR T-cell therapies, idecabtagene vicleucel and ciltacabtagene autoleucel, which were initially approved in later-line settings but are now becoming clinically relevant earlier in the disease course, even as early as second-line therapy.14,15
That is a remarkable amount of change in just a few years, and it has completely reshaped how we think about sequencing and selecting therapy. As healthcare professionals, it is important to pause and take stock of where we are in the treatment landscape and to understand how these advances are influencing day-to-day decisions.
In this module, I focus primarily on bispecific antibodies, their place in current practice, and how their introduction into the R/R MM space has transformed our approach to treatment decisions.
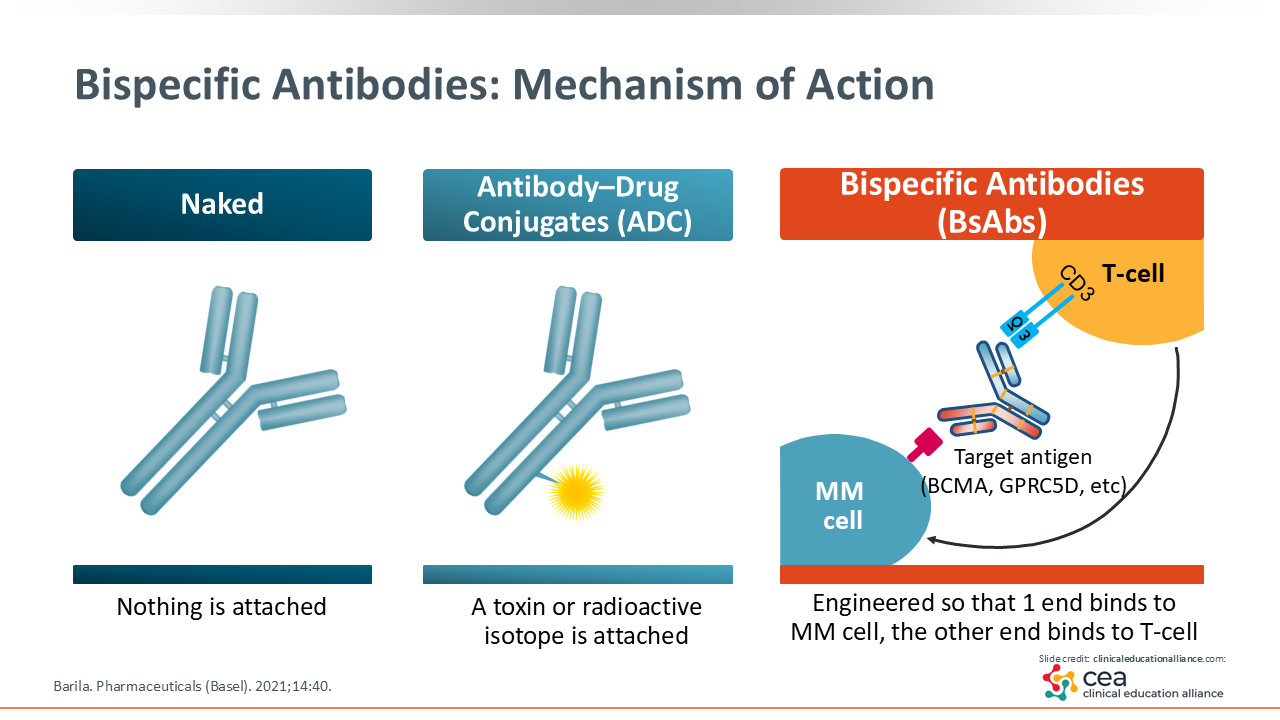
Bispecific Antibodies: Mechanism of Action
Bispecific antibodies are designed to activate the immune system’s T-cells and direct them to bind to a specific target on MM cells. Following this, breakdown of the cancer cells occurs through redirected lysis and cytologic synapse.16
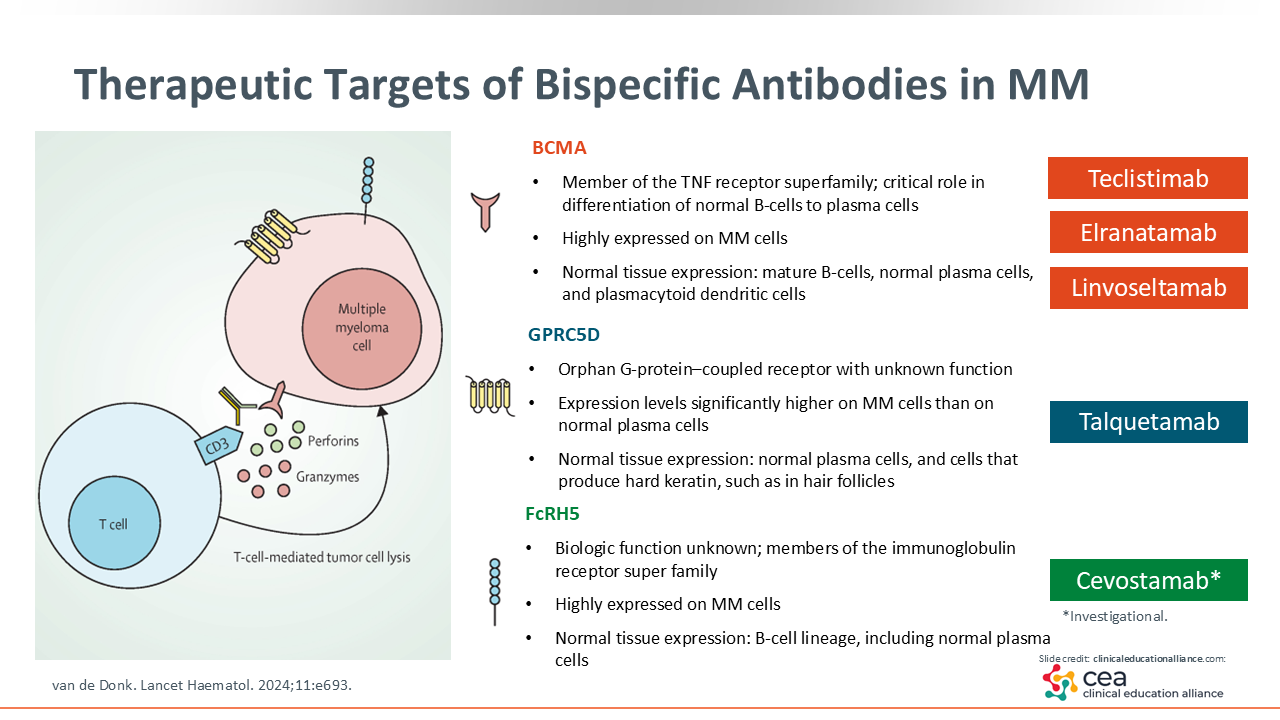
Therapeutic Targets of Bispecific Antibodies in MM
Currently, there are 4 bispecific antibody products that are approved by the FDA for MM. Three of these (teclistamab, elranatamab, and linvoseltamab) target B-cell maturation antigen (BCMA), which is a member of the TNF super receptor family. BCMA plays a critical role in the differentiation of normal B-cells into plasma cells and is highly expressed in MM cells. Adverse effects, such as infection, occur because BCMA is also expressed in normal tissues.
The target of the fourth FDA-approved therapy, talquetamab, is GPRC5D, which has a higher expression on MM cells compared with normal plasma cells. GPRC5D is also expressed in hard keratin cells and hair follicles, which relates to the unique adverse effect profile of talquetamab.17
Teclistamab, elranatamab, linvoseltamab, and talquetamab are all approved by the FDA for R/R MM after ≥4 prior lines of therapy, including an immunomodulatory drug, proteasome inhibitor, and anti-CD38 monoclonal antibody.1-4
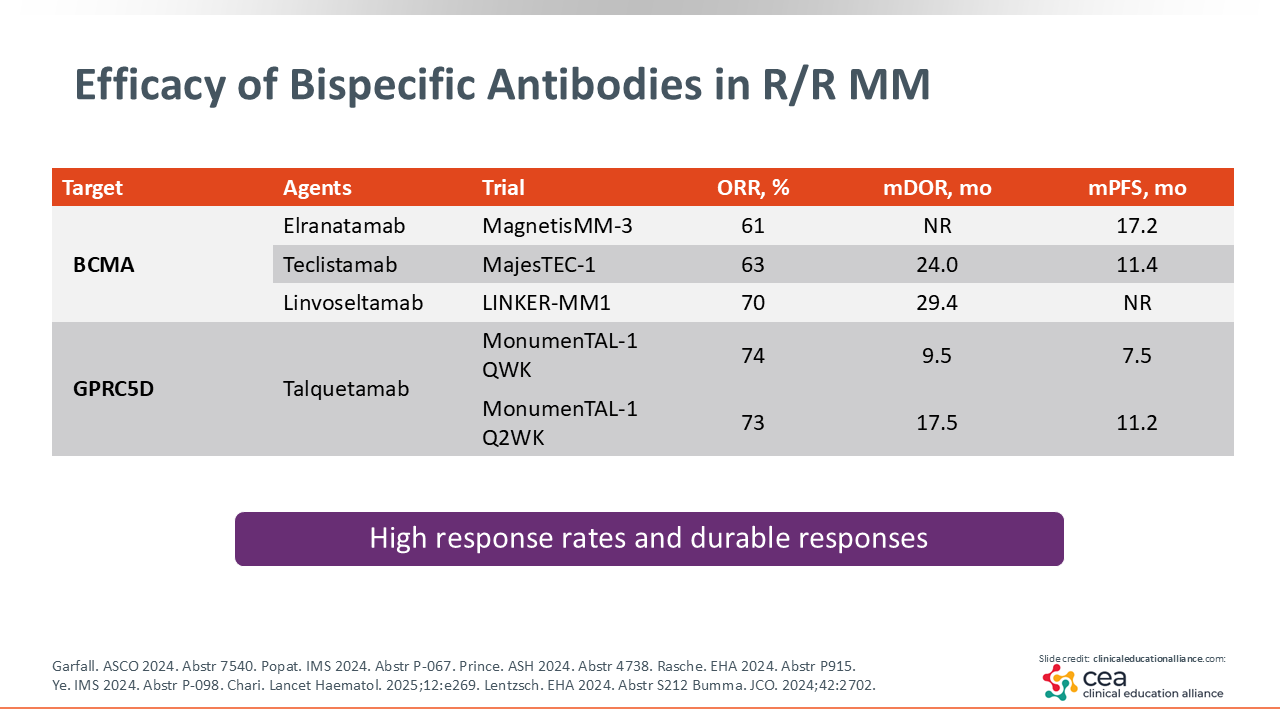
Efficacy of Bispecific Antibodies in R/R MM
When treating patients who have already received multiple lines of therapy, one of our main goals is to select options that offer the best chance for a longer and more durable response. The 4 bispecific antibodies currently approved by the FDA for R/R MM have all shown impressive efficacy in their phase I/II trials: MajesTEC-1 (teclistamab), MagnetisMM-3 (elranatamab), MonumenTAL-1 (talquetamab), and LINKER-MM1 (linvoseltamab).
Across these studies, overall response rates (ORRs) range from approximately 61% to 74%, with median progression-free survival between 11.0 and 17.2 months.18-20 Because linvoseltamab was approved most recently, survival data are still maturing.21 Clinically, we are seeing that many patients achieve durable responses with manageable adverse effects and good overall tolerability with these agents.
However, it is important to consider the adverse effect profile of each medication, and this is discussed in a separate module.
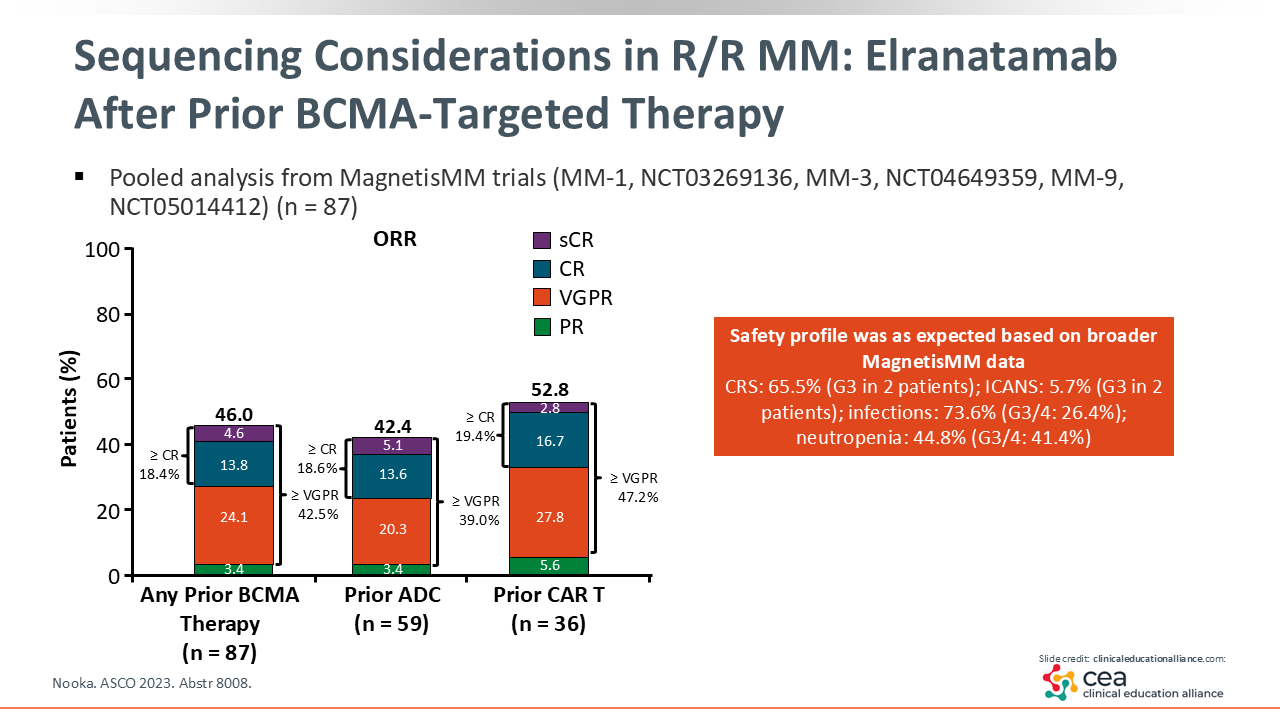
Sequencing Considerations in R/R MM: Elranatamab After Prior BCMA-Targeted Therapy
When thinking about how to use bispecific antibody therapies in practice, it is important to choose an approach that considers what to use first, what alternatives to consider if a patient cannot tolerate a particular agent, and how to move forward after a relapse. Sequencing in this area is not one-size-fits-all. It varies based on patient-specific factors, disease characteristics, and provider experience.
We also have emerging data on how well BCMA-targeting bispecific therapies work after prior exposure to other BCMA-directed treatments.22 For example, a pooled analysis of patients treated with elranatamab across the MagnetisMM trial program evaluated a subgroup that had previously received BCMA-directed therapy (n = 87). In this group, elranatamab still produced responses, with an ORR of 46%.23 Although this is lower than the response rate observed in BCMA-naive patients in the phase I/II MagnetisMM-3 trial, who had an ORR of 61%, it still represents meaningful clinical activity.19
These findings highlight that prior exposure to BCMA-directed therapy does not eliminate the possibility of benefit from a BCMA-targeting bispecific, although the depth of response may be affected. It also raises an important question about whether switching to a different target can offer an advantage for certain patients.
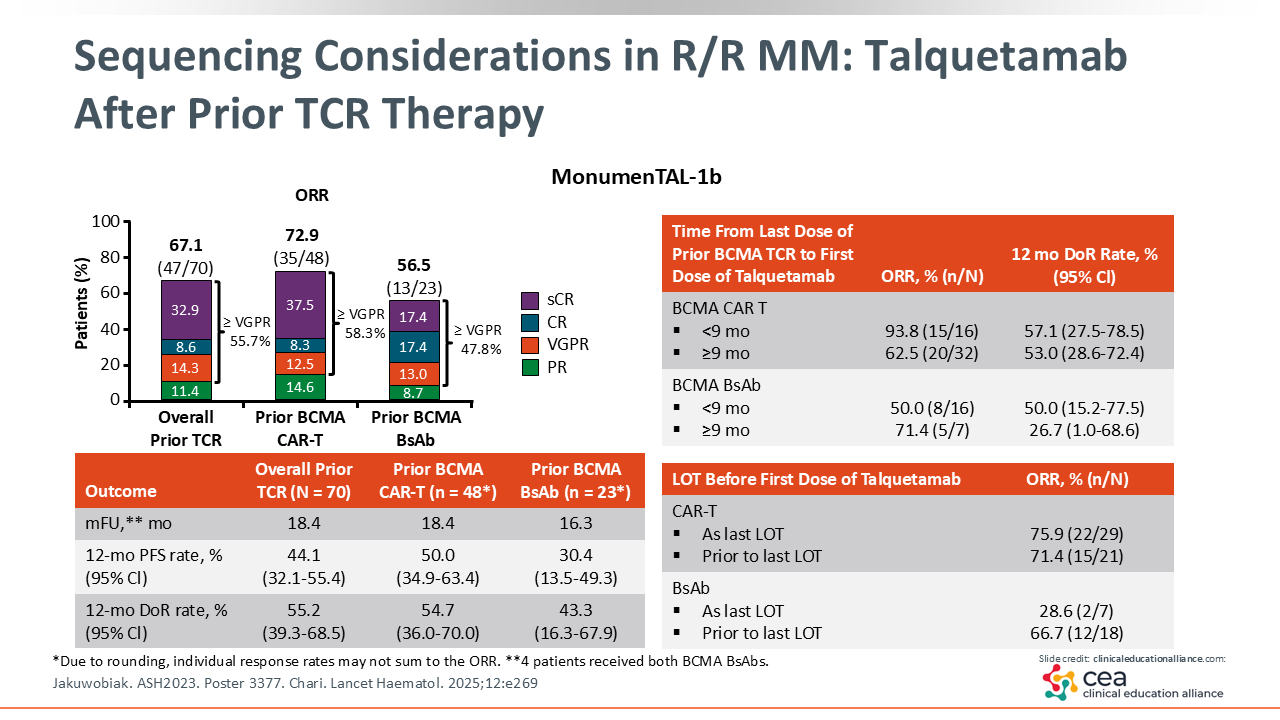
Sequencing Considerations in R/R MM: Talquetamab After Prior TCR Therapy
Another area of interest is the potential for T-cell exhaustion after bispecific antibody use. This has led to ongoing discussion about whether patients might benefit from switching to a non–T-cell engaging therapy before starting another bispecific antibody to allow time for T-cell recovery. This question also comes up when considering talquetamab as an option between BCMA-directed bispecific antibodies, with the idea that using a GPRC5D-targeting agent may help preserve BCMA receptor availability for future therapy.24
Data from the phase I/II MonumenTAL-1 study provide helpful insight here. This trial evaluated talquetamab, a GPRC5D-targeting bispecific antibody, in patients with R/R MM. Among patients who had previously received T-cell redirecting therapies (n = 70), most of which were anti-BCMA, the ORR was 67% compared with 74% in T-cell redirecting–naive patients.
In the post–CAR T-cell setting specifically, talquetamab demonstrated an ORR of 73% and a median duration of response of more than 1 year.20,25 These results support its potential role as a sequencing option after prior BCMA-directed therapy; however, these concepts are still evolving and will likely become clearer as more real-world experience and clinical trial data emerge.
Finally, another consideration in sequencing is the use of bispecific antibodies as a bridge to future CAR T-cell therapy. Some institutions may use talquetamab as bridging therapy, since its GPRC5D target allows BCMA to remain available for subsequent CAR T-cell treatment, which may help maintain the effectiveness of future BCMA-directed approaches.26-28
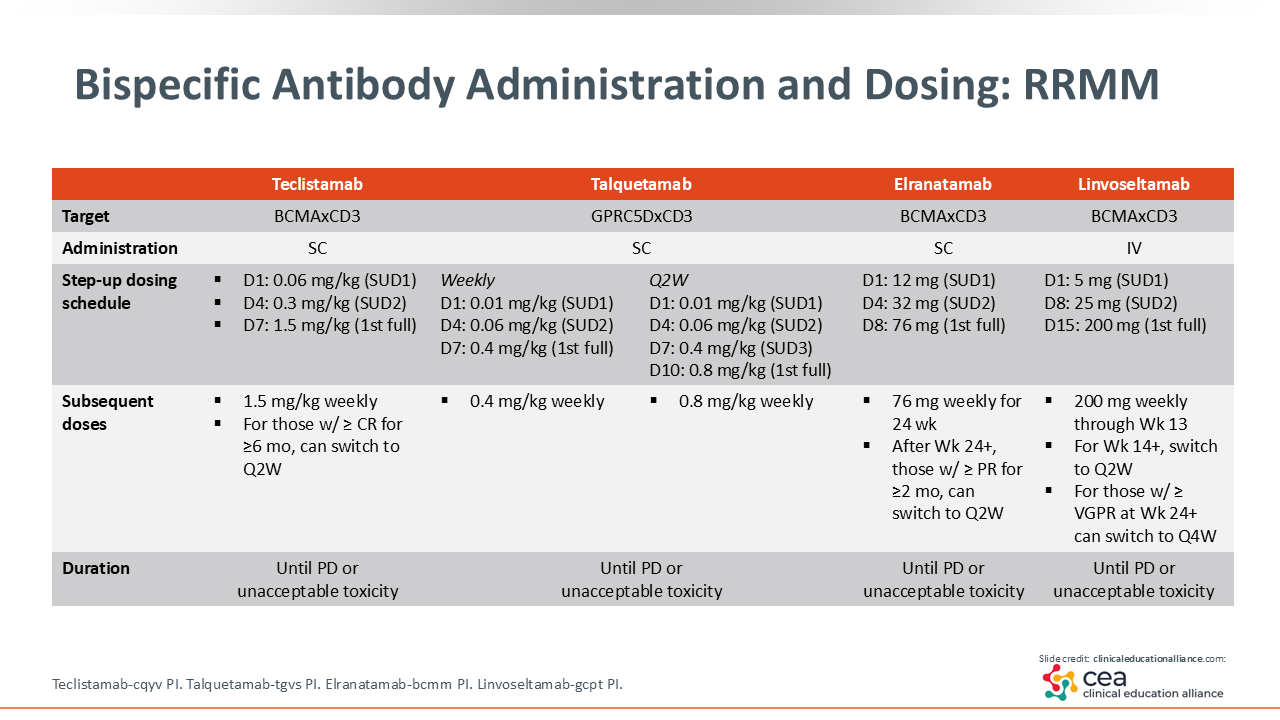
Bispecific Antibody Administration and Dosing: R/R MM
Once we decide that a bispecific antibody is the right next step for a patient, the next questions are usually very practical ones: How do we give these drugs? What does the dosing schedule look like? What should patients expect during those first few doses?
When we look at the 4 FDA-approved bispecific antibodies, teclistamab, talquetamab, and elranatamab are all administered subcutaneously, and linvoseltamab is the only one given intravenously.
All 4 agents use a small starting dose and a ramp-up schedule. For teclistamab, talquetamab, and elranatamab, patients typically receive a very small “baby” dose on Day 1. This is followed by a slightly larger “toddler” dose, and then the full treatment dose, usually all within the first week.
Linvoseltamab follows a similar concept but on a longer schedule. The “baby” dose is given on Day 1, the “toddler” dose on Day 8, and the first full dose on Day 15.
After step-up dosing, each agent differs slightly in its prescribing information regarding when a patient can transition from weekly dosing to every other week or even monthly. These transitions depend on the specific product and the patient’s response. In general, patients remain on weekly dosing for at least the first few months before spacing out intervals.
It is also important to note that talquetamab can be initiated as either a weekly or a biweekly regimen. The step-up schedule is slightly different for the biweekly option, with one additional step-up dose, so it is helpful to discuss the most appropriate regimen with the patient from the beginning.1-4
Some institutions will transition patients to monthly dosing once a good response is achieved to help reduce the adverse effects associated with ongoing bispecific therapy or lessen the burden of traveling to and from treatment.
Delivery Models
When we think about delivery models for bispecific antibody therapy, we need to consider how we are actually going to treat our patients. Are we giving these drugs entirely in the inpatient setting? Are we planning for full outpatient administration? Or are we using a hybrid model that combines both approaches?
If you look at the prescribing information for these medications, they state that the first doses should be administered in a hospital setting with observation. The keyword here is “should.” It does not say “must,” but it does emphasize that inpatient monitoring is recommended so that any early toxicity, such as CRS or immune effector cell–associated neurotoxicity syndrome (ICANS), can be managed quickly.1-4
In real-world practice, we have learned that there is more flexibility. Data from centers like Emory University and the Mayo Clinic have shown that outpatient administration is feasible when the right infrastructure and safety protocols are in place. In these outpatient models, patients receive their step-up doses in the clinic and are only admitted to the hospital if toxicity develops. These programs rely on clear after-hours pathways, weekend coverage plans, and tools such as wearable temperature monitors to detect fevers early.5,6
Hybrid models are another option. In this approach, patients receive the bispecific antibody doses in the outpatient clinic but have a planned hospital admission for observation on specific days. This setup requires close coordination with local hospitals and clear communication among the care team so that monitoring and management are seamless.7
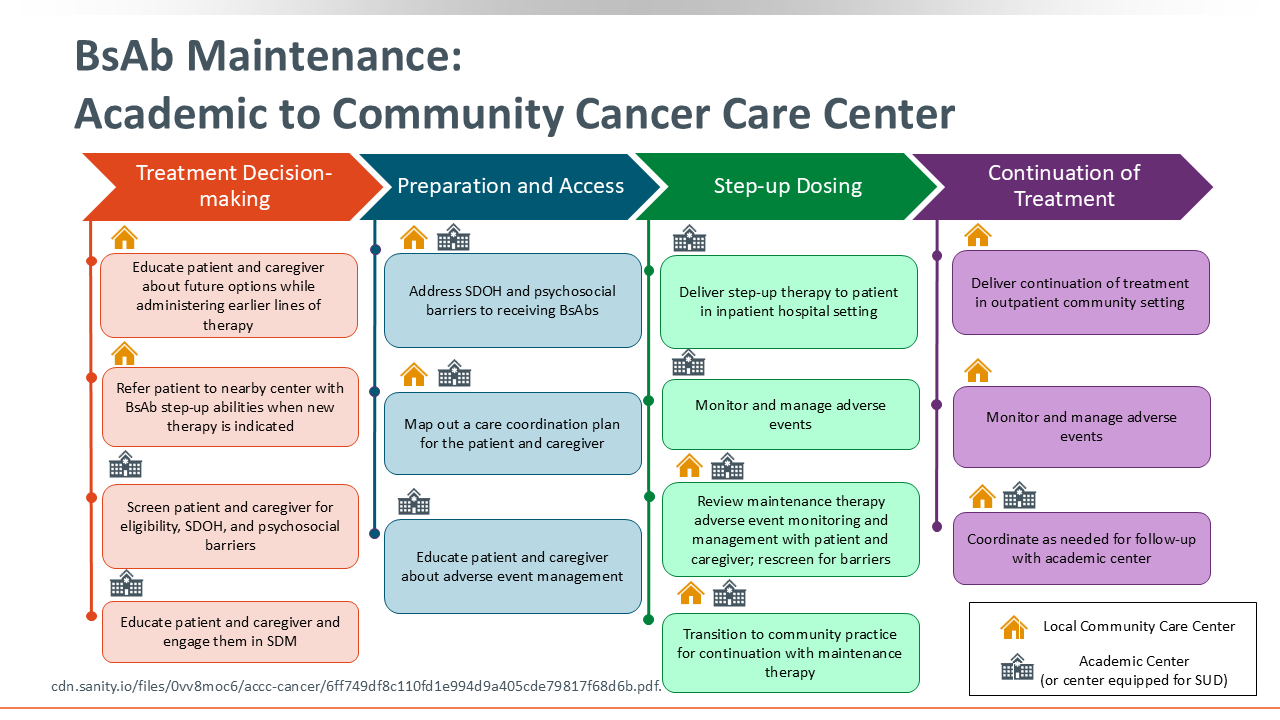
BsAb Maintenance: Academic to Community Cancer Care Center
When we think about how bispecific antibodies are used in the real world, the approach can look very different in an academic center than in a community cancer center. I work at an academic, university-based tertiary center, and many of our patients come from community clinics across the Southeast. Because of this, we spend a lot of time thinking about how to start therapy safely and then transition care back to local teams.
Here are some of the key considerations we think through:
- Availability of Products. Not every community center has all bispecific antibodies on formulary. At our center, we intentionally keep all 4 approved bispecific antibodies available so that patients can start therapy with us and then move back to their local teams without needing to switch products.
- Coordinating Step-up Dosing. Many community centers prefer that the first doses be administered at an academic center with experience managing CRS and ICANS. After the completion of step-up, we work closely with community practices to plan a smooth transfer of care. If a community center is new to bispecific therapy, medical science liaisons can help support training and site onboarding.
- Assessing Psychosocial and Logistical Needs. Before starting treatment, we screen patients and caregivers for barriers such as travel, housing, transportation, caregiver availability, and financial considerations. These conversations help us plan whether step-up dosing should occur at our center or at a site closer to home.
- Transitioning Back to Community Care. Once patients are established on therapy and tolerating treatment, many continue their maintenance dosing in the community setting. I still see these patients every 3 months to assess response, review adverse effects, and coordinate any needed therapy adjustments.29
Outpatient Model for Administration of Bispecific Antibodies: Patient Selection and Step-up Dosing
Every institution is going to approach bispecific antibody administration a little differently, and there is no single model that fits everyone. What matters most is having a clear plan, communicating that plan across the entire care team, and creating enough standardization so that everyone knows the process and patients receive consistent, safe care.29 With that in mind, I can share what this looks like where I practice.
At Emory, bispecific antibody therapy is routinely initiated in the outpatient setting for carefully selected patients. Eligibility for outpatient administration depends on both disease characteristics and social factors. Patients with high disease burden, poor performance status, or a prior history of severe CRS or ICANS are typically managed as inpatients.
For outpatient administration, patients must live within 30 minutes of the treatment center and have a dedicated caregiver available 24 hours per day. They are also restricted from driving for 48 hours after receiving the first full dose, in accordance with Risk Evaluation and Mitigation Strategy program requirements.
Treatment is delivered in our ambulatory infusion center. On Day 1, all patients receive prophylactic tocilizumab along with a small step-up dose of the bispecific antibody. A slightly larger dose is given on Day 4, followed by the first full dose on Day 8. Patients are discharged with “pocket medications,” including dexamethasone, diphenhydramine, and acetaminophen for both premedication and management of early CRS or ICANS symptoms at home.6
Outpatient Model for Administration of Bispecific Antibodies: AE Management
This outpatient model allows many of our patients to transition back to their community centers, but we still have to monitor closely for new or worsening adverse events, especially ICANS and CRS early in the treatment course. After patients receive their doses and go home, we ask them to check their temperature every 8 hours while awake or anytime new symptoms develop.
We review the common early warning signs with both the patient and their caregiver. These include fever, headache, shortness of breath, body aches, new fatigue, dizziness, or any change in mental status. They also monitor for neurologic changes such as confusion, difficulty speaking, or delayed responses. If any of these symptoms occur, the patient takes the 3 prescribed “pocket medications” at home: dexamethasone, acetaminophen, and diphenhydramine.
From there, they call our immediate care center and have their caregiver drive them directly to the immediate care center. Our center functions like an oncology urgent care and is staffed 24 hours per day by advanced practice providers, which helps us avoid sending patients to the emergency department whenever possible.
Once the patient arrives, we formally assess and grade CRS with vital signs and an Immune Effector Cell Encephalopathy (ICE) score to evaluate neurologic function. If fever is the only symptom and the presentation is consistent with grade 1 CRS, we administer tocilizumab and monitor for 8 hours to ensure symptoms resolve. Often the fever has already improved because of the home medications, but a patient-reported fever still warrants tocilizumab to help prevent recurrence.
If patients present with grade ≥2 CRS or neurologic toxicity, or if grade 1 CRS persists despite tocilizumab, they are admitted to the hospital for further management.6
Outpatient Model for Administration of Bispecific Antibodies: Prophylactic Tocilizumab
As we think about all the moving parts involved in coordinating bispecific antibody therapy, another key component is how we approach the use of tocilizumab. This is an area where practice patterns vary widely from one institution to another, so I can share what we are doing at Emory and why.
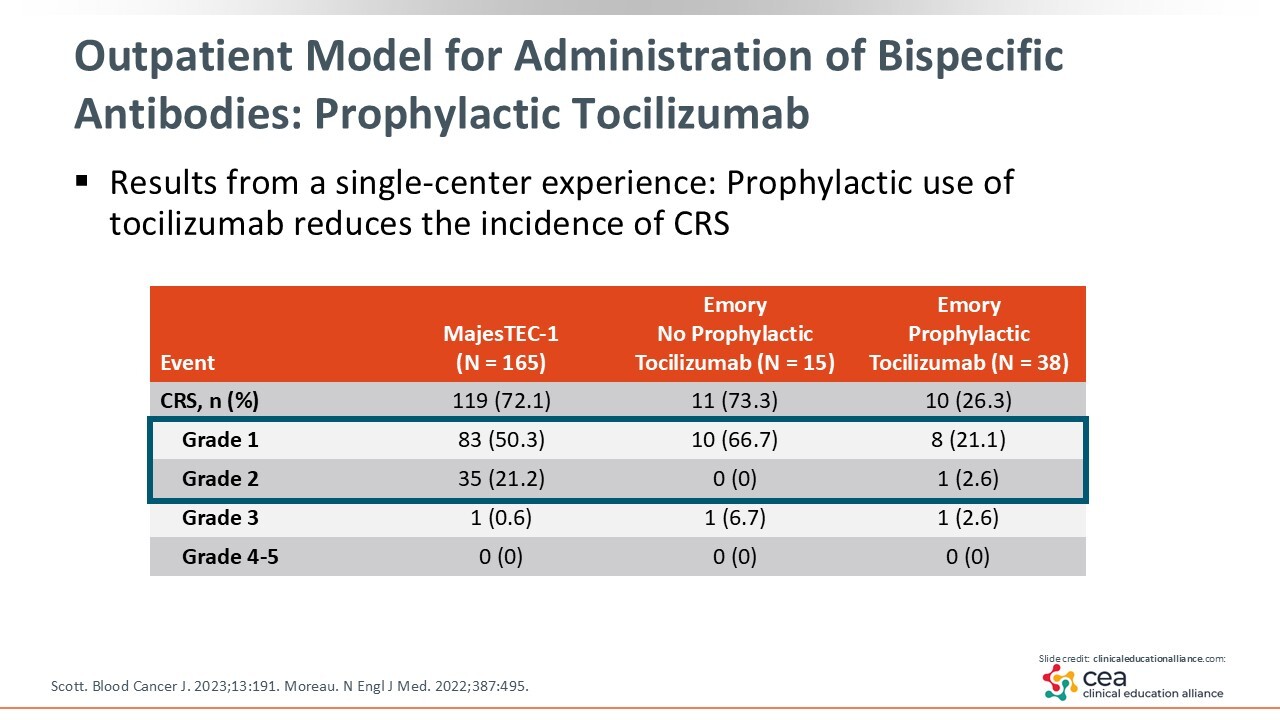
Many healthcare professionals are understandably hesitant about prophylactic tocilizumab because of concerns that it might blunt treatment efficacy or increase infection risk. However, the data we have so far are reassuring. Results from our single-center experience have shown prophylactic tocilizumab to reduce the overall rate of CRS to approximately 26.3% without affecting response rates or increasing notable toxicities such as neutropenia or infection during the early months of therapy.
Taking into consideration that CRS and ICANS rates may be overestimated in settings with continuous inpatient monitoring, this becomes a meaningful data set to guide outpatient decision-making.30 These findings were a major factor in our decision to move select patients to outpatient bispecific antibody administration, and we now use tocilizumab prophylaxis routinely across all bispecific antibody therapy we administer.
We initially adopted this approach using teclistamab, since it was the first FDA-approved bispecific antibody. Over time, we have broadened it to all bispecific antibody agents in our formulary. Although tocilizumab is an expensive medication, we think about it similarly to using primary granulocyte colony-stimulating factor prophylaxis for chemotherapy regimens that carry a 25% risk of febrile neutropenia.31 The goal is to prevent serious complications up front, especially in an older patient population where reducing CRS-related events like tachycardia, hypotension, or shortness of breath can make a big difference.
We have also refined the timing of administration. Giving tocilizumab before the first step-up dose has reduced our CRS incidence even further. When we initially administered it before the second step-up dose, CRS rates were higher. Since making this adjustment, most CRS events we see are grade 1/2. We have had no grade 4/5 CRS, and only 1 patient has experienced grade 3 CRS.6,32
Conclusions
These are the approaches and lessons we have learned at our center as we have integrated bispecific antibodies into the care of patients with R/R MM. For advanced practice providers who are considering incorporating bispecific antibodies into your own practice, I would strongly encourage you to explore these therapies and begin conversations within your team about creating a realistic, standardized workflow that fits the needs of your institution. Clear processes and communication are key to making these treatments both safe and accessible.
There are excellent resources available to help guide implementation, including the Successful Integration of Bispecific Antibodies into Community Oncology Practice materials offered by the Association of Cancer Care Centers. These tools can support both academic and community practices as they build or refine their bispecific programs.29
Overall, we have found bispecific antibody therapy to be well tolerated, even among older and more frail patients. When used thoughtfully and with the right support systems in place, they can make a significant impact on patient outcomes.