CE / CME
HBV in Key Communities: Strategies to Overcome Barriers and Elevate Care
Pharmacists: 0.75 contact hour (0.075 CEUs)
Physicians: maximum of 0.75 AMA PRA Category 1 Credit™
Nurse Practitioners/Nurses: 0.75 Nursing contact hour
Released: March 12, 2026
Expiration: March 11, 2027
Activity
US Data
Now, let’s move into the hepatitis B treatment recommendations, current approaches, and how the treatment landscape is evolving.
In the past, many people who met the criteria for treatment per the American Association for the Study of Liver Disease (AASLD) guidelines did not actually get treated.10
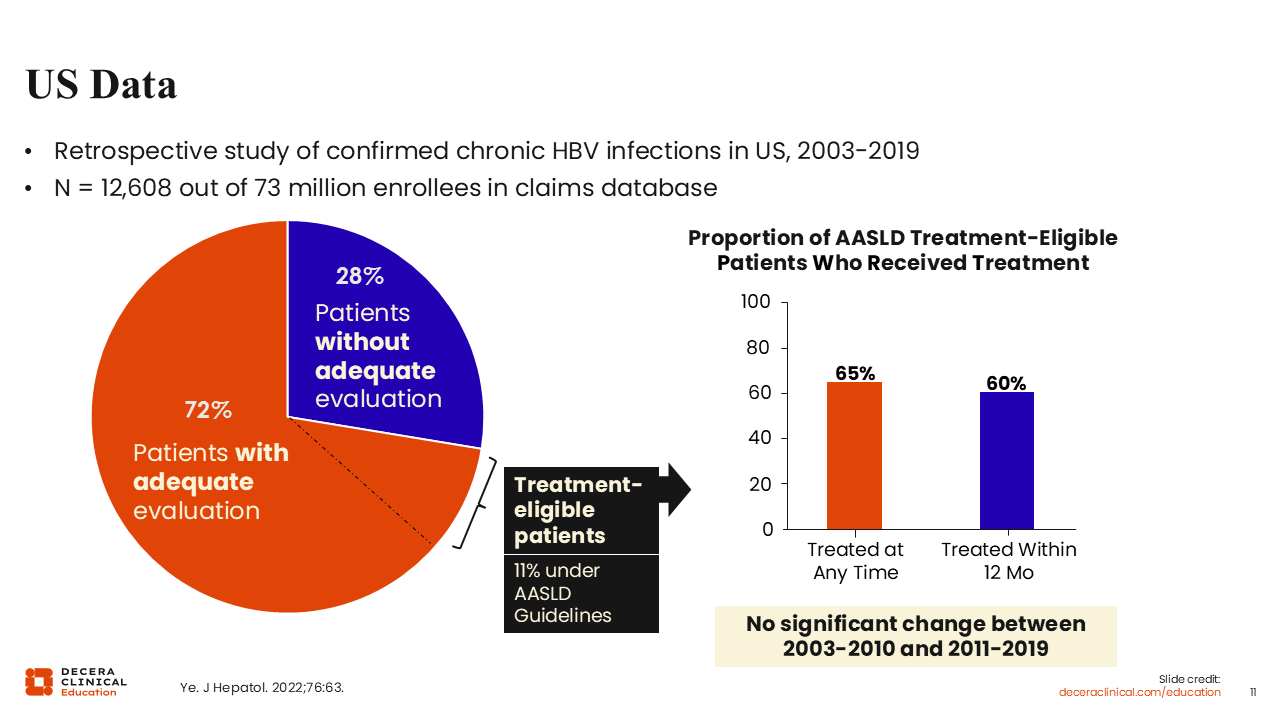
In this study using insurance claim data from 2003-2019, of approximately 12,000 people living with hepatitis B, only 72% had received adequate evaluation. Of those who did receive evaluation, only 11% were deemed treatment-eligible according to the AASLD guidelines. Then, of this 11% who were eligible for treatment, only 65% were treated at any time. An even lower percentage, 60%, received treatment within 12 months of evaluation.
These data demonstrate significant gaps between qualifying for treatment and actually receiving it. Furthermore, investigators did not see a significant change over time, indicating that there was no improvement in linkage to care.10
Who Should Be Considered for Treatment: The Traditional Approach
To determine how we can close the treatment gap, we must first consider the traditional approach to HBV treatment.
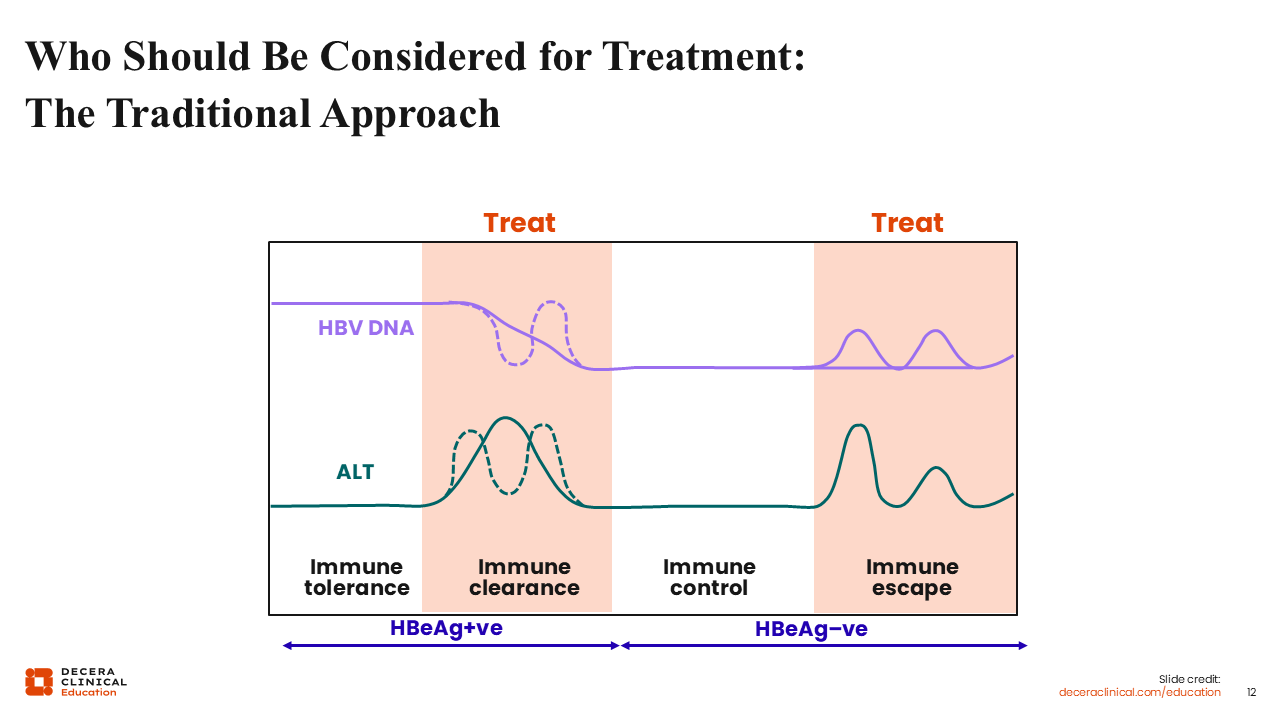
This diagram depicts the dynamic phases of hepatitis B. The point of this graph is to show that the traditional approach to HBV treatment is to treat only when we see signs of abnormal alanine transaminase (ALT) or inflammation.35
The phases of infection can also be divided into hepatitis B e-antigen (HBeAg)–positive or HBeAg-negative phases, which can be used to determine treatment eligibility. However, this can be a real barrier for HCPs, as all the serology tests start to resemble alphabet soup. Confusion on what test to order or how to interpret the results can mean that a lot of times, even if patients do get the tests, they don't actually get treated.35
Fortunately, as we’ll see later, the new WHO guidelines have removed this confusing e-antigen criteria as part of the workup and evaluation for treatment.36 This is 1 of the many ways that new, simpler guidelines may help with HBV management.
Faster HBV DNA Suppression Improves Clinical Outcomes
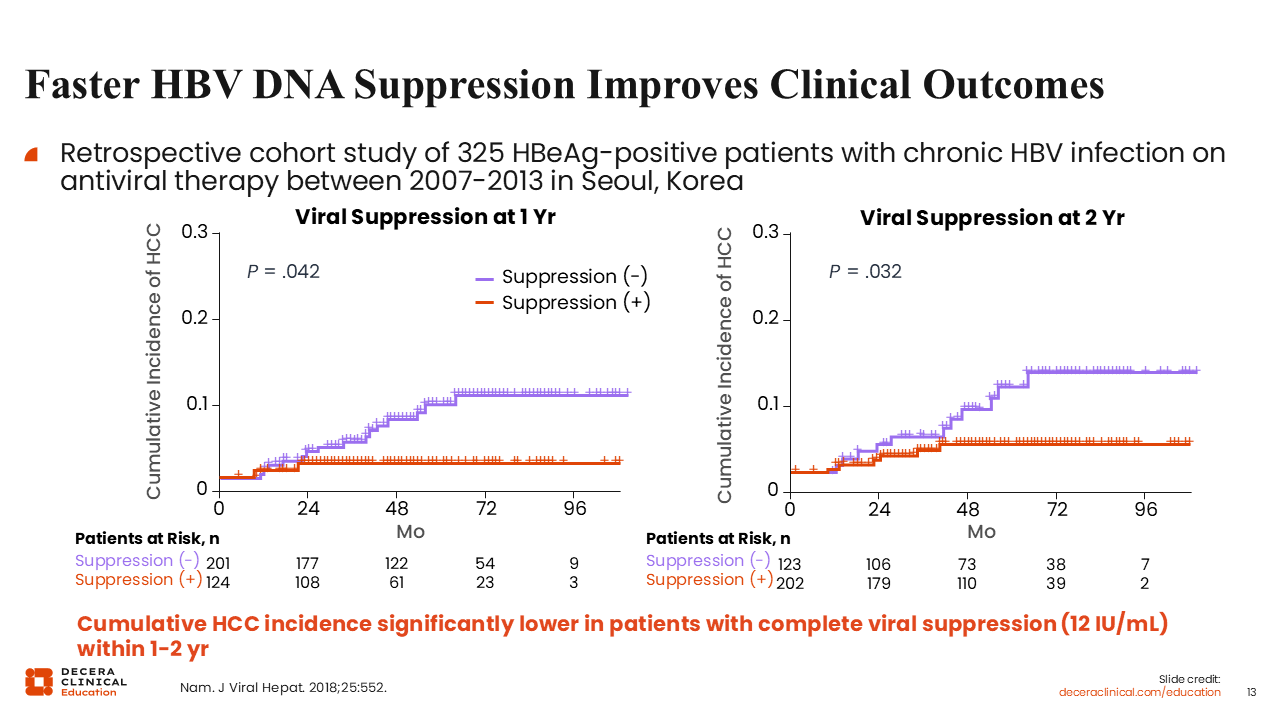
The key point is that the faster we can get somebody to DNA suppression, the more it improves outcomes.37
So in this study, a lower cumulative incidence of HCC was associated with quicker suppression of HBV DNA. I think that's a good takeaway for prioritizing timely treatment.37
New WHO Guidelines: HBV Treatment
It is exciting that over the past couple of years, we've seen a real shift in our approach to HBV treatment and updates to guidelines around the world.
The WHO hepatitis B guidelines were updated in 2024 to include expanded treatment recommendations for everyone 12 years of age or older with advanced fibrosis or cirrhosis, elevated HBV DNA or ALT, and certain risk factors listed in green on this slide. For example, immunosuppression from medication is also an indication to treat regardless of HBV DNA levels.36
The evidence for comorbidities as indications for treatment is not as strong, but the guidelines still recommend treatment in the presence of certain comorbidities. Diabetes and metabolic dysfunction–associated steatotic liver disease (MASLD), or what used to be called fatty liver, could be reasons to start treatment. Extrahepatic manifestations such as glomerulonephritis and vasculitis have always been indications for treatment.36
Previously, treatment eligibility was also determined by whether a patient was e-antigen positive or e-antigen negative. The new WHO guidelines have removed the e-antigen criteria as part of the workup and evaluation for treatment.36
Finally, I think it’s important that the WHO recommendations make special provisions for areas that have limited resources and limited access to molecular testing for hepatitis B DNA. The new guidelines indicate that if there is no access to an HBV DNA assay, patients can be treated simply based on persistently abnormal ALT levels. That is, HCPs don't need DNA test results in order to treat somebody. I think this will go a long way towards closing the gap between people who get tested and people who get treated.36
AASLD Guideline Changes: HBV Treatment Indications
The updated AASLD guidelines, which just came out in November of 2025, are important because they include expanded treatment criteria, with the addition of more groups who would be eligible for treatment with shared clinical decision-making.38
The goal is to target those at risk for disease progression. On this slide, the box that says "Treat Now," is focused on identifying patients who clearly need to be treated right away. These are people with active chronic hepatitis B who meet at least 1 of the following criteria: ALT 2 times the upper limit of normal (regardless of e-antigen status), e-antigen positive with HBV DNA >20,000, or e-antigen negative with HBV DNA >2000.35,38
Other indications to treat include evidence of advanced fibrosis or cirrhosis or coinfection with HIV.
The guidelines also specify special populations who should be treated immediately, such as those receiving immunosuppressive or immunomodulatory therapy and people who are pregnant.35,38
In addition to these groups, we now also have an expanded group of people who can be considered for treatment, with shared decision-making.
2025 AASLD Guidelines: Expanded Treatment Criteria, With Use of Shared Clinical Decision-Making
Let’s take a closer look at some of these special populations, and populations who could be treated with shared clinical decision-making.
For pregnant people, treatment with TDF or TAF should be initiated at gestational Week 28 if HBV DNA exceeds 200,000 copies/mL. The recommendation for TAF is a new addition, based on a recent meta-analysis.38
For people in the immune-tolerant phase who are 40 years of age or older, or who have grade 2 liver inflammation, or fibrosis greater than stage F2, the recommendation is to treat.
If they are younger than 40 years of age, HCPs should use shared clinical decision-making regarding treatment, taking into consideration patient preference, if they want to be treated, if they are at risk of transmission, and the benefits and risks of treatment.38
I don’t think the age distinction is clear-cut. Even in their 30s, the risk of negative outcomes starts to go up for people in the immune-tolerant phase. So, for example, if a patient is 39 and their fibrosis or liver inflammation is on the borderline of treatment eligibility, you may actually decide to treat them, even if they're not exactly 40.38
I think what's interesting about the immune-tolerant phase is that it is now more strictly defined as HBV DNA ≥ log10 7 IU/mL or 10 million IU/mL with a normal ALT. I think this is because we discovered that many people had lower HBV DNA and were not truly immune tolerant, and therefore still had poor outcomes when they were not treated.38
Shared clinical decision-making should also be used for people in the indeterminate phase, which includes up to 50% of people with hepatitis B. According to the AASLD guidelines, the indeterminate phase comprises people who are HBsAg positive and HBeAg negative, without cirrhosis. They don't yet fall into the treatment indication threshold in terms of ALT and HBV DNA levels. So, the recommendation now is that we use shared decision-making, taking into account other risk factors and patient preferences to determine if they should be treated or monitored for the time being.38
The AASLD also recommends using shared clinical decision-making for people who have a high risk for transmission of HBV to others. This is a novel recommendation that has not been implemented by the European Association for the Study of the Liver (EASL) or the WHO: Treatment as a way to prevent transmission of hepatitis B.38-40
According to the AASLD guidelines, this category includes both people who are clinically more likely to transmit the virus (such as those with high levels of HBV DNA) and those who could have high risk exposures (such as people who are having sex with multiple partners, sharing injection needles, or living with somebody who is immunocompromised, undergoing chemotherapy, or has had a transplant and cannot mount a response to hepatitis B even if they've been vaccinated).38
Finally, if patients decide to defer treatment via shared decision-making, the guideline recommendation is to make sure this decision is reassessed at the next visit.38
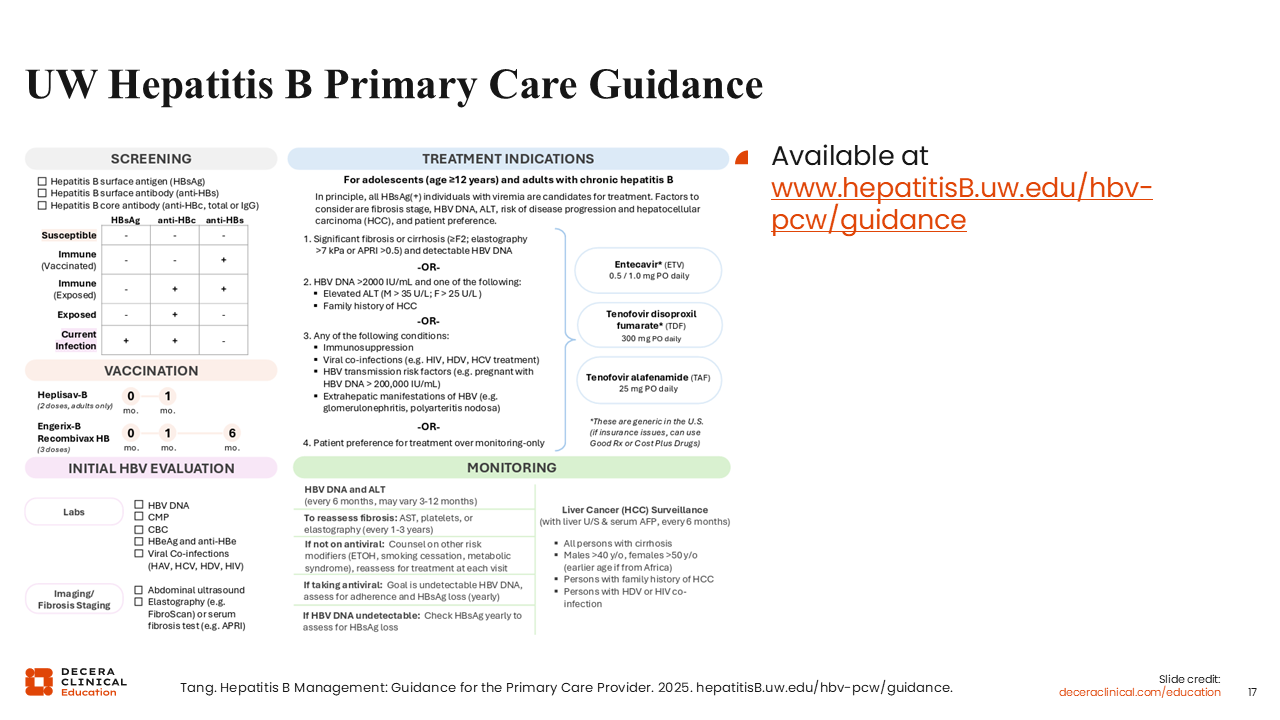
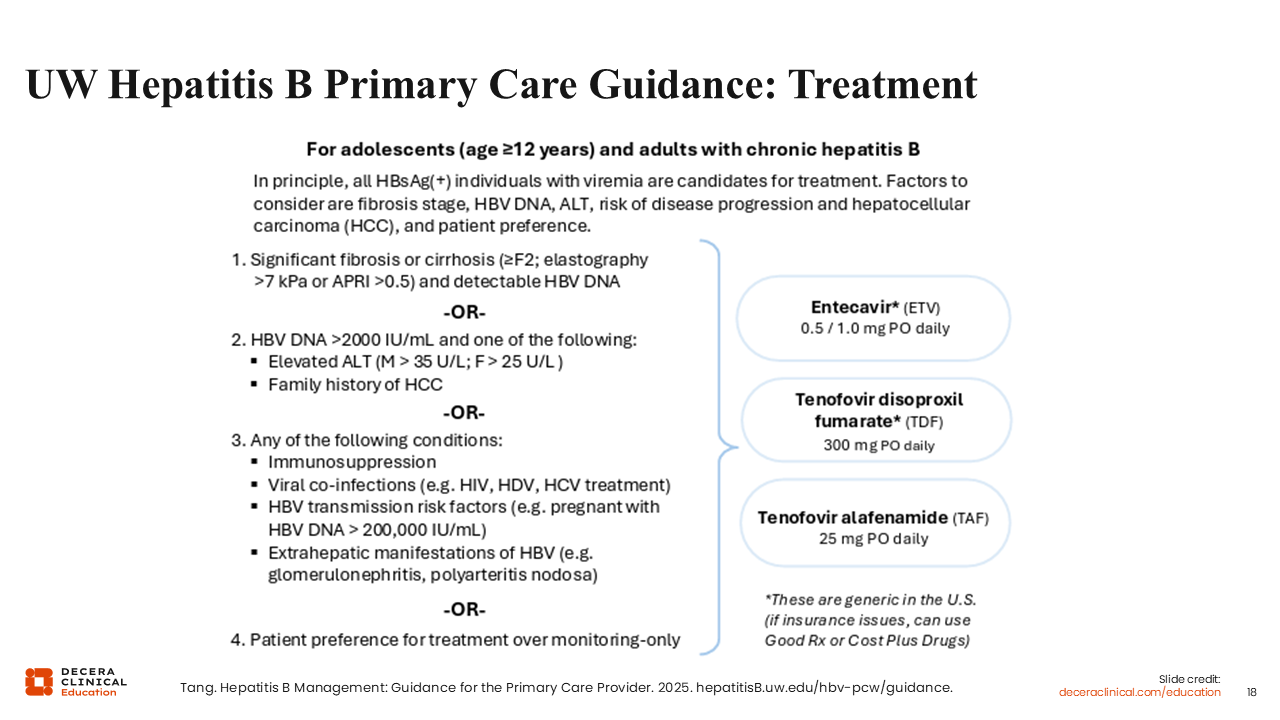
UW Hepatitis B Primary Care Guidance
I’m excited to also share updated guidance, built by a multidisciplinary workgroup that I am part of, in collaboration with the University of Washington, HBV. This is an excellent resource specifically for primary care providers and nonspecialists, including a simple 1-page summary of HBV care and management.
UW Hepatitis B Primary Care Guidance: Treatment
This guidance simplifies treatment eligibility into 4 categories, as shown on this slide.