CE / CME
Current, Novel and Emerging Therapies on the Horizon for Cancer Cachexia
Physician Assistants/Physician Associates: 0.25 AAPA Category 1 CME credit
Pharmacists: 0.25 contact hour (0.025 CEUs)
Physicians: maximum of 0.25 AMA PRA Category 1 Credit™
Nurse Practitioners/Nurses: 0.25 Nursing contact hour
European Learners: 0.25 EBAC® CE Credit
Released: March 24, 2026
Expiration: September 23, 2026
Activity
Cancer Cachexia: Brief Overview
Cancer cachexia is increasingly recognized as a complex, multiorgan syndrome involving interactions among tumor-derived signals, host inflammatory responses, and multiple metabolic pathways.1 This syndrome remains a major unmet medical need in the management of patients with cancer.
The release of inflammatory cytokines in tumor cells plays a central role in the development and progression of cancer cachexia. Tumor-derived factors and inflammatory mediators influence appetite regulation in the brain, promote skeletal muscle proteolysis, and drive alterations in adipose tissue metabolism. These processes contribute to cachexia-related symptoms such as anorexia, adipose tissue wasting, muscle atrophy, and multiple organ dysfunction.
NCCN Guidelines: Palliative Care for Patients With Anorexia/Cachexia
The National Comprehensive Cancer Network (NCCN) in the United States has guidelines for palliative care. These guidelines are patient specific and include recommendations for the assessment and interventions for patients with anorexia/cachexia.2
Based on the estimated life expectancy for the individual patient, the NCCN guidelines for palliative care recommend evaluations for weight loss, screening for conditions that interfere with food intake, and a review/modification of medications that may interfere with food consumption. Patient, family member, and caregiver education on the patient’s condition and needs, particularly related to the risk vs benefit of treatment options, is also emphasized in the NCCN guidelines.
These guidelines also highlight the importance of addressing the psychiatric, emotional, and nutritional needs of each individual patient. Of note, enrollment on a clinical trial investigating novel approaches should be considered if the patient meets the eligibility criteria and stands to benefit from trial participation. It is important to note that the patient needs to be regularly reassessed to ensure that continuous positive outcomes are achieved.
In general, the NCCN guidelines for palliative care do not primarily focus on the need to evaluate weight loss. Instead, it has a focus on screening for conditions and symptoms that may interfere with food intake, such as shortness of breath, fatigue, pain, early satiety, gastrointestinal conditions, mouth-related issues such as mucositis, and psychiatric conditions such as anxiety, depression, and eating disorders.
If the individual patient continues to require treatment, or if the condition progresses, reevaluation of the interventions being used is important.
ASCO Guidelines Management of Cancer Cachexia
The American Society of Clinical Oncology (ASCO) also has guidelines for cancer cachexia, even though these guidelines are quite restrictive. ASCO recommends that dietary counseling be offered with the goal of providing patients and their caregivers with advice on cachexia management.3,4
The ASCO guidelines note that enteral feeding tubes and parenteral nutrition should not be routinely used in the management of patients with cancer cachexia. Per the ASCO guidelines, no specific pharmacologic intervention can be recommended as the standard of care in the absence of more robust evidence.
Therefore, healthcare professionals (HCPs) may choose not to prescribe medications specifically for the treatment of cancer cachexia. When the HCP decides to recommend a drug to improve appetite and/or improve weight gain, currently available pharmacologic interventions that may be used include progesterone analogs and short-term (weeks) use of corticosteroids.
In a recent update to the ASCO guidelines, oncologists may now offer low-dose daily olanzapine, a second-generation antipsychotic agent.4,5
ASCO and ESMO Recommendations: Treatment Considerations for Patients With Cancer and Cachexia
Besides ASCO, the European Society for Medical Oncology (ESMO) is another professional body with guidelines for cancer cachexia. Both ESMO and ASCO have considered megestrol acetate (a synthetic progestin), olanzapine, and corticosteroids for use as appetite stimulants.3,4,6 However, it is very important to note that none of these agents is directly indicated for the treatment of cancer cachexia. Although in some cases these agents may exert limited but positive effects on weight gain and improvements in appetite, they are also associated with safety concerns that HCPs need to be aware of. In essence, none of these 3 agents really address the underlying biology of cachexia in patients with advanced cancer. So, there is substantial room for improvement in finding agents that directly treat cachexia.
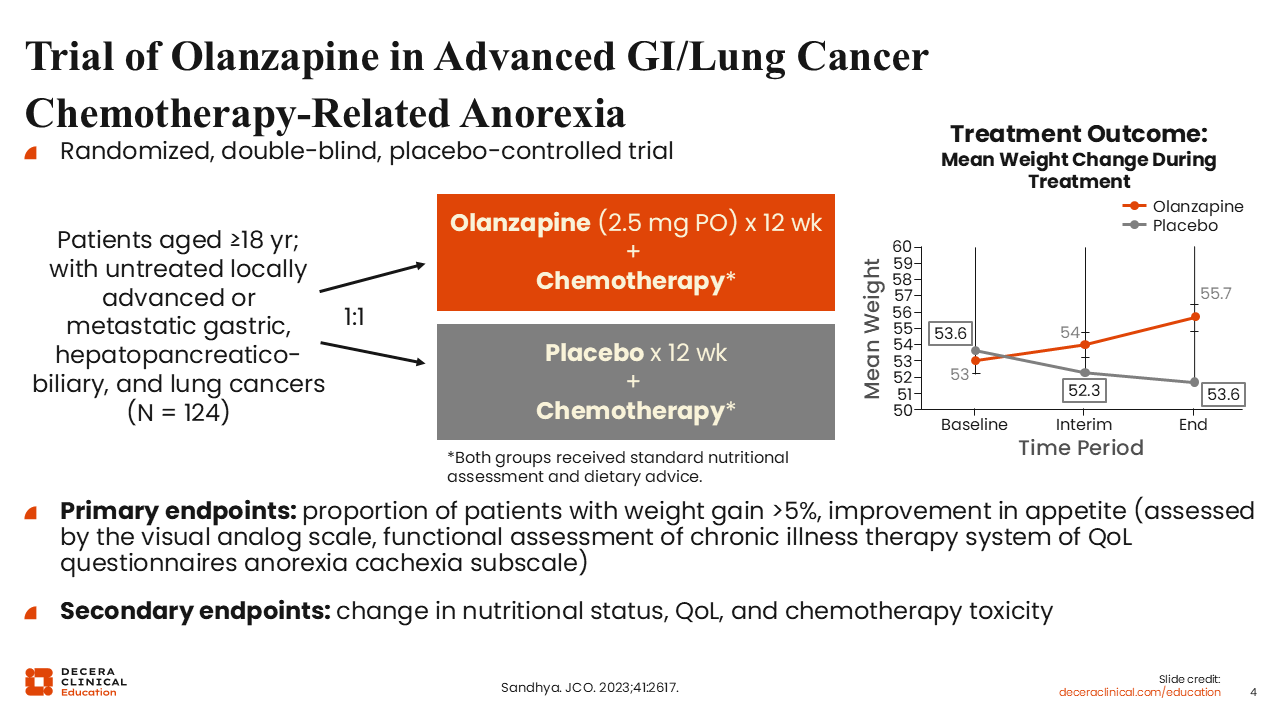
Trial of Olanzapine in Advanced GI/Lung Cancer Chemotherapy-Related Anorexia
A randomized, double-blind, placebo-controlled trial conducted in India investigated chemotherapy with or without low-dose olanzapine (2.5 mg PO for 12 weeks) for 124 adult patients with untreated locally advanced or metastatic gastric, hepatopancreaticobiliary, and lung cancers.7 The primary endpoints were the proportion of patients with weight gain >5% and improvement in appetite.
This trial demonstrated a significant improvement in weight gain over time, and the results led to the recommendation of olanzapine as a potential option for weight gain by many organizations.
ALLIANCE A222004 Trial of Olanzapine vs Megestrol Acetate for Cancer-Associated Anorexia
Of interest is the ongoing multicenter, randomized phase III ALLIANCE A222004 trial that is investigating olanzapine (5 mg PO for ≤4 weeks) vs megestrol acetate (600 mg PO for ≤4 weeks) for adult patients with a diagnosis of advanced cancer and anorexia (NCT04939090). It is anticipated that 360 patients will be enrolled on this trial. The primary endpoint of this head-to-head comparison between olanzapine and megestrol acetate is change in appetite. The secondary endpoints include the proportion of patients with a ≥5% weight gain, and a change in well-being status and cachexia/anorexia symptoms.
This is an interesting study that will shed more light on the impact of olanzapine vs megestrol acetate on cancer-related symptoms.
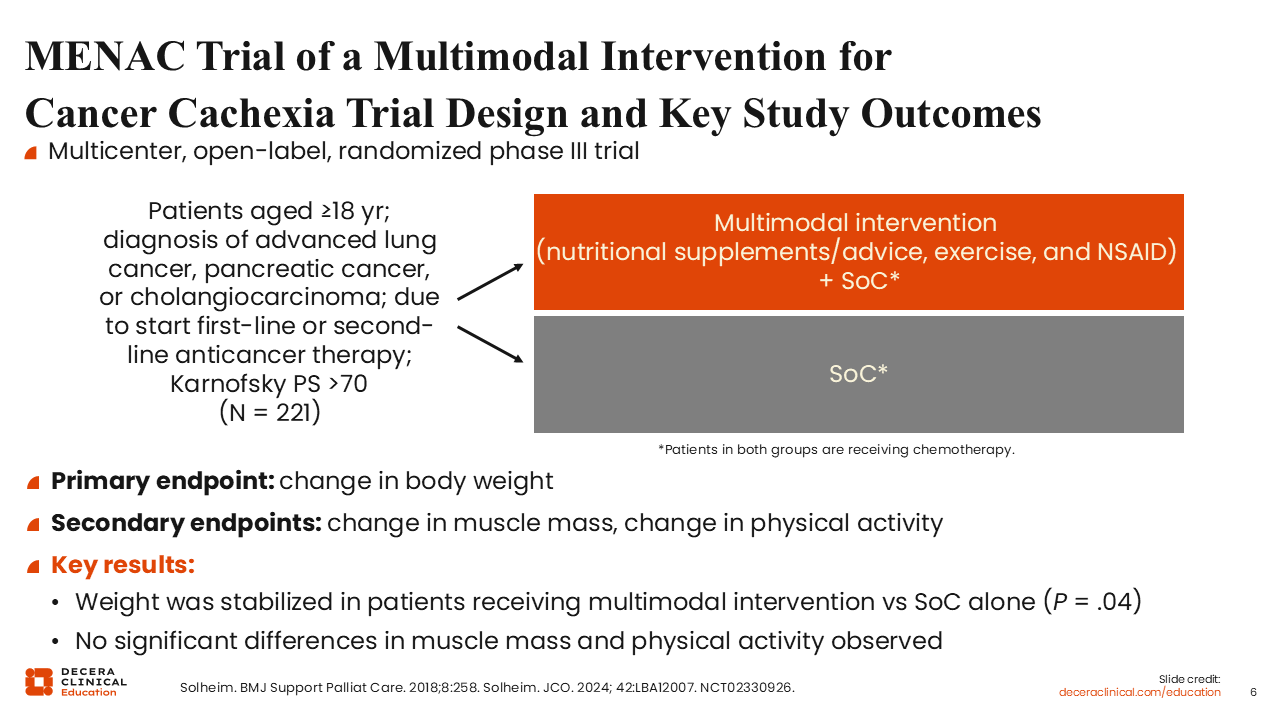
MENAC Trial of a Multimodal Intervention for Cancer Cachexia Trial Design and Key Study Outcomes
It is now widely recognized and recommended that a multimodal treatment approach is important to yield optimal outcomes in the management of patients with cancer cachexia. The MENAC trial uniquely evaluates a multimodal treatment of patients with cancer and cachexia (NCT02330926).
The multicenter, randomized, phase III MENAC trial investigated multimodal intervention using standard palliative care with or without nutritional supplements, exercise, and anti-inflammatory medication for patients with advanced lung cancer, pancreatic cancer, or cholangiocarcinoma who are due to start first-line or second-line anticancer therapy. The primary endpoint was change in body weight. The secondary endpoints were change in muscle mass and change in physical activity.
Even though weight gain was not achieved, weight was stabilized in patients receiving multimodal intervention vs standard palliative care alone (P = .04).8,9 This is a positive outcome for patients with cancer and cachexia. Unfortunately, there was no significant difference in muscle mass or physical activity.
These results demonstrated that better and improved clinical outcomes are still needed for our patients with cancer and cachexia.
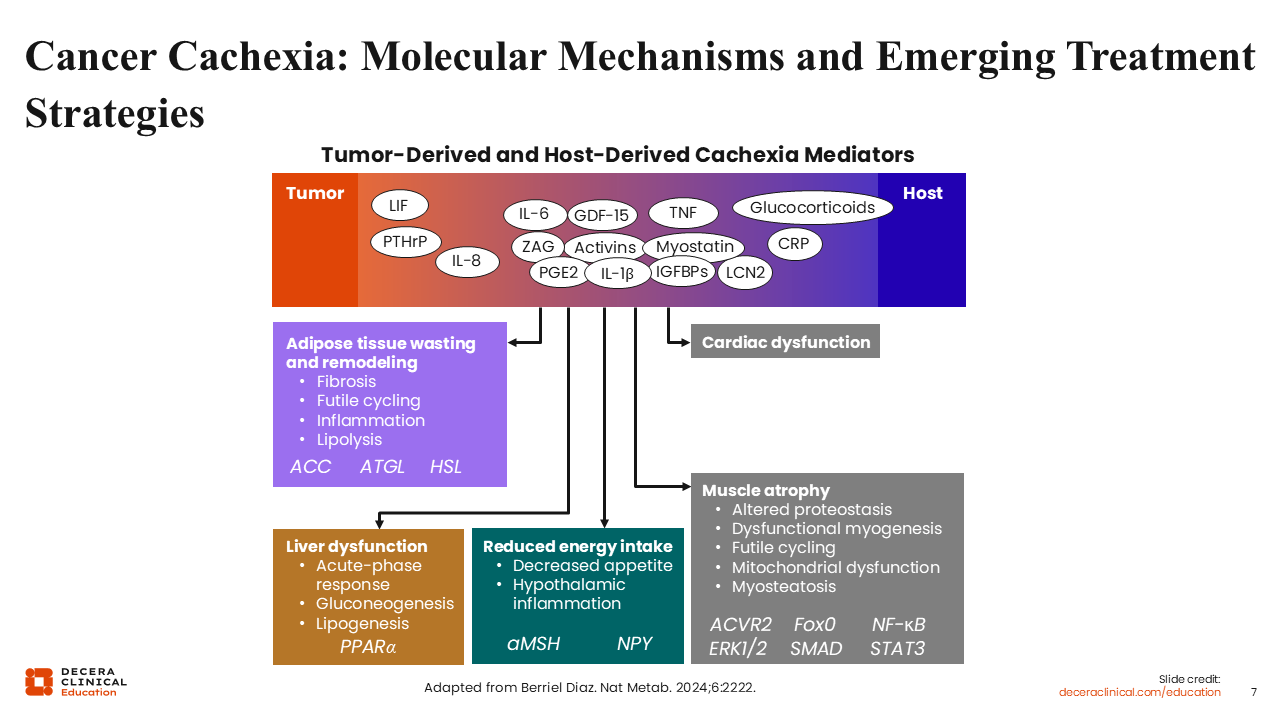
Cancer Cachexia: Molecular Mechanisms and Emerging Treatment Strategies
Although complex, the biology and pathobiology of cancer cachexia are increasingly becoming better understood.10 The molecular mechanisms contributing to the development of cancer cachexia include a variety of tumor- and host-derived mediators that disrupt normal metabolic regulation and promote adipose tissue wasting and remodeling.
Tumor- and host-associated factors released into the system can cause cardiac and liver dysfunction, reduce food intake, and promote muscle atrophy. The increasing recognition and understanding of the complexities and interactions among these biological pathways underlying cancer cachexia has the potential to lead to the development of new and novel therapeutic approaches for the syndrome itself, rather than symptom management.
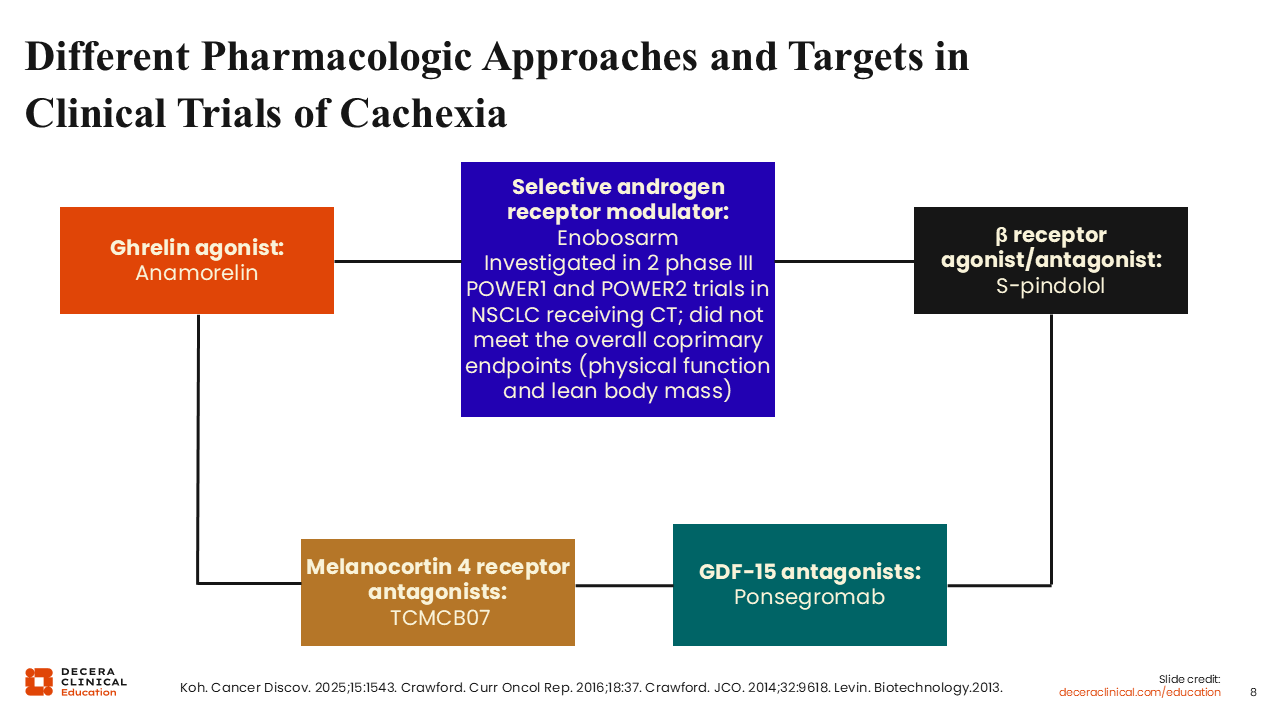
Different Pharmacologic Approaches and Targets in Clinical Trials of Cachexia
Due to its complexity and the myriad of signaling pathways influencing its development and progression, different pharmacologic approaches have been and are currently being tested clinically in the treatment of cancer cachexia.11 For example, anamorelin is an oral selective agonist of the ghrelin receptor that is approved in Japan for patients with cancer cachexia.12 In other parts of the world, anamorelin is still under investigation for cancer cachexia.
Enobosarm is a selective androgen
receptor modulator that was investigated in 2 identical randomized phase III trials (POWER1 and POWER2) for patients with advanced non-small-cell lung cancer (NSCLC) initiating first-line chemotherapy (NCT01355484, NCT01355497). Both trials assessed the efficacy of enobosarm for the prevention and treatment of muscle wasting. Unfortunately, both POWER1 and POWER2 failed to meet the prespecified criteria for the primary endpoints of physical function and lean body mass.13-16
Even though decreases in body weight compared with weight at baseline were reported in both trials with enobosarm, increases in lean body mass were observed with enobosarm vs placebo. Therefore, it appears that enobosarm is a muscle-specific agent that has no effect on appetite stimulation or weight gain.
Other investigational agents in the treatment of patients with cancer cachexia include S-pindolol, a nonselective β-adrenoceptor antagonist that possesses intrinsic sympathomimetic activity.17 TCMCB07, a melanocortin 4 receptor antagonist, is also under investigation for patients with cancer cachexia. Another exciting agent currently in late-phase clinical trials is ponsegromab, a humanized monoclonal antibody that targets GDF-15, a circulating cytokine.18,19
Cancer cachexia remains an unmet medical need. As the pharmacologic landscape continues to evolve, I am hopeful that these and other approaches being investigated will continue to lead to improvements in appetite, lean body mass, and physical function without significant adverse events.
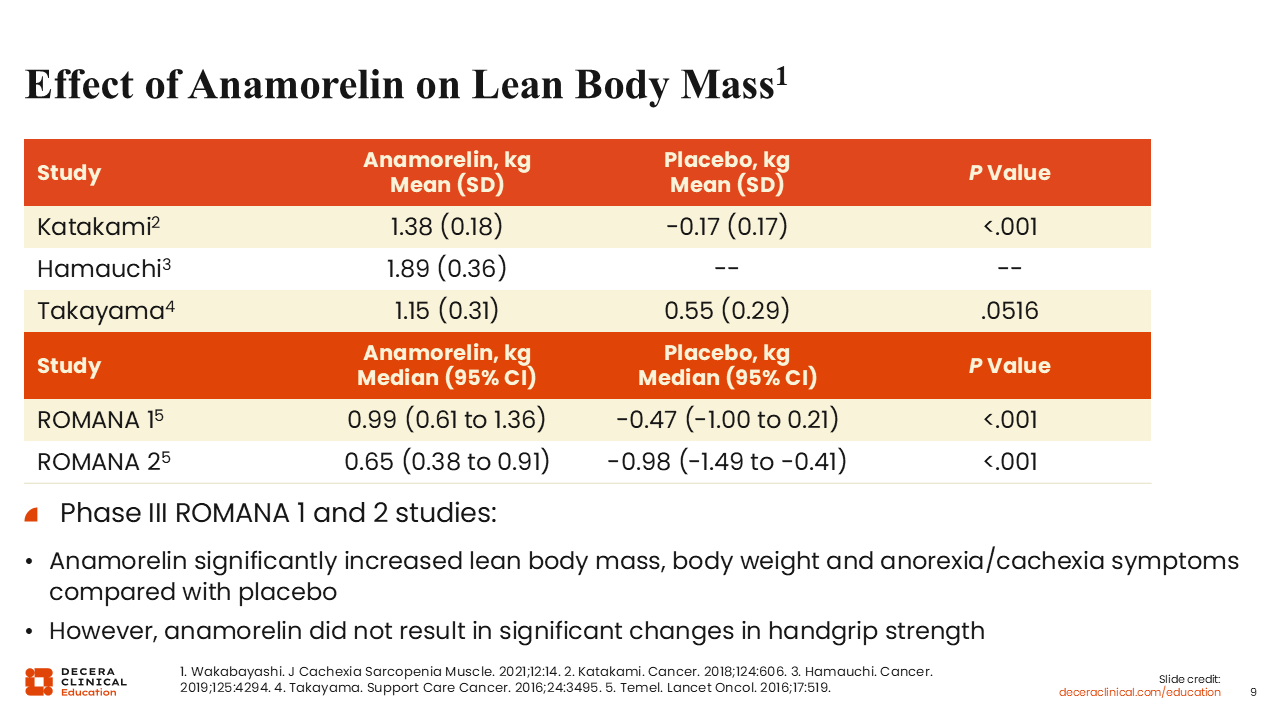
Effect of Anamorelin on Lean Body Mass
Several trials have investigated the effect of anamorelin on lean body mass in patients with cancer and cachexia.20-24 Of particular interest are the randomized phase III ROMANA 1 and ROMANA 2 trials, which investigated anamorelin vs placebo for patients with advanced NSCLC and cachexia.23 The coprimary efficacy endpoints were the median change in lean body mass and handgrip strength over 12 weeks measured in all study participants (intention-to-treat population).
In both trials, anamorelin significantly increased lean body mass (P <.0001), body weight (P <.0001), and anorexia/cachexia symptoms (P <.005) compared with placebo. However, anamorelin did not result in any significant changes in handgrip strength compared with placebo. In other words, even though appetite was improved with anamorelin, there was no functional improvement as determined by handgrip strength.
Therefore, in the United States and in Europe, anamorelin failed to receive approval for use in the treatment of cachexia in patients with cancer. In contrast, however, anamorelin received approval in Japan, where the focus is placed more on weight gain and lean body mass, symptoms for which improvements were seen. To date, Japan is the only country with regulatory approval for the use of anamorelin for cancer cachexia.12,24
ACT-ONE: Phase II Trial Espindolol (S-pindolol) in CRC or NSCLC
ACT-ONE is a randomized phase II trial that compared the effects of low-dose or high-dose S-pindolol vs placebo for 87 patients with cachexia and advanced-stage colorectal cancer (CRC) or NSCLC who have a life expectancy >3 months.25 All patients were receiving or had received chemotherapy-based therapy for advanced cancer. The primary endpoint was weight change rate over 16 weeks.
Compared with patients who received placebo, treatment with high-dose S-pindolol resulted in a significant reversal of weight loss (P <.0001). High-dose S-pindolol also led to lean body mass improvement (P <.02) and increased hand-grip strength (P = .0134) with a numerical improvement observed in fat mass compared with placebo. The study also showed trends toward improvements in functional tests for stair climbing power, ability to walk for 6 minutes, and a trend towards improvement in performing the short physical performance battery test.
Based on these findings, a global phase IIb/III IMPACT trial of S-pindolol for patients with CRC and cachexia is planned.
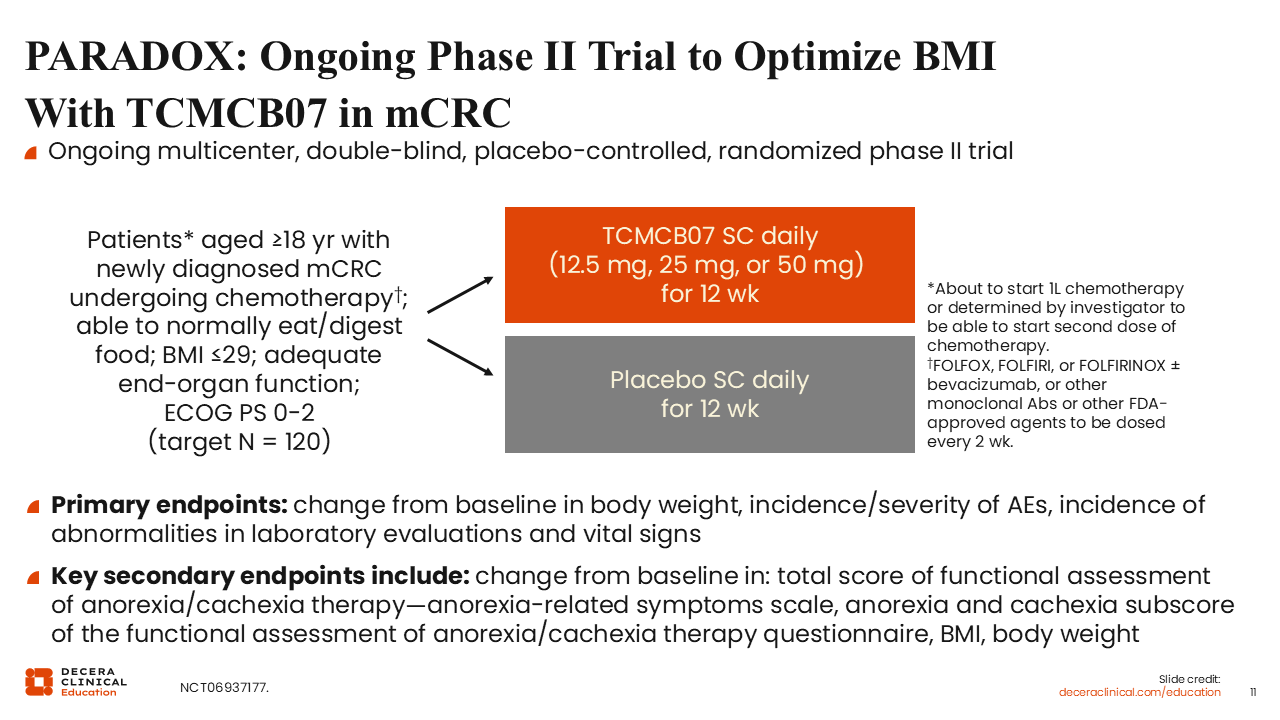
PARADOX: Ongoing Phase II Trial to Optimize BMI With TCMCB07 in mCRC
Melanocortins in the hypothalamus are potent appetite suppressors.26 So, pathophysiologic processes that increase melanocortin signaling cause anorexia and subsequently cachexia. In a preclinical study, treatment with TCMCB07, a melanocortin-4 receptor antagonist, led to increased food intake, increased body weight, and preserved fat/lean mass in rat models of anorexia and cachexia.
Therefore, PARADOX is an ongoing randomized phase II trial that is investigating TCMCB07 vs placebo for patients with newly diagnosed metastatic CRC receiving chemotherapy (NCT06937177). This proof-of-concept trial will investigate 3 different daily doses of TCMCB07 (12.5 mg, 25 mg, or 50 mg) vs placebo for 12 weeks. The primary endpoints are change from baseline in body weight, incidence/severity of adverse events, and incidence of abnormalities in laboratory evaluations and vital signs. The secondary endpoints include functional assessments of anorexia/cachexia and body mass index.
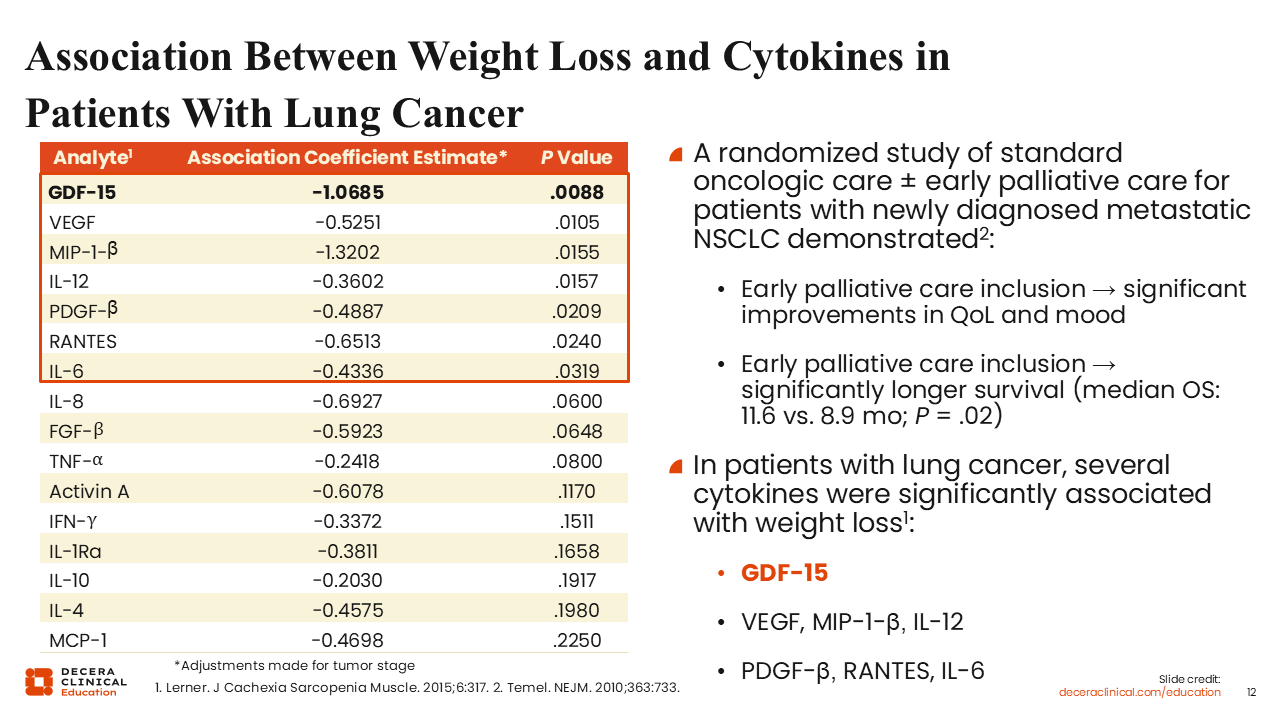
Association Between Weight Loss and Cytokines in Patients With Lung Cancer
As previously alluded to, multiple inflammatory cytokines play a central role in the development and progression of cancer cachexia.1,10 Of note, weight loss is commonly observed in patients with advanced cancer receiving treatment, and this is associated with increased inflammation and reduced survival.27
Along with other inflammatory markers, GDF-15 has been linked to weight loss and reduced appendicular lean body mass and handgrip strength.28,29 Also, the activation of the GDF-15 signaling axis has been identified as a potent inducer of cancer cachexia.
In a study of patients with lung cancer, multiple cytokines were found to correlate with weight loss, including GDF-15, vascular endothelial growth factor, and several interleukins. Of these inflammatory cytokines, GDF-15 was identified as the only predictor of weight loss.28
In another study, patients with newly diagnosed metastatic NSCLC were randomly assigned to receive either early palliative care plus standard anticancer therapy or standard anticancer therapy alone (NCT01038271).30 The addition of early palliative care to standard anticancer treatment demonstrated significant improvements in quality of life and mood and longer survival vs standard anticancer care alone.
Together, these observations highlight the importance of addressing symptom burden and providing anticancer therapy in the management of patients with cancer, particularly those experiencing cancer-related cachexia symptoms such as weight loss, muscle atrophy, and reduced physical function. By so doing, the patient’s overall well-being is improved with a significant improvement in quality of life and the prolongation of survival.
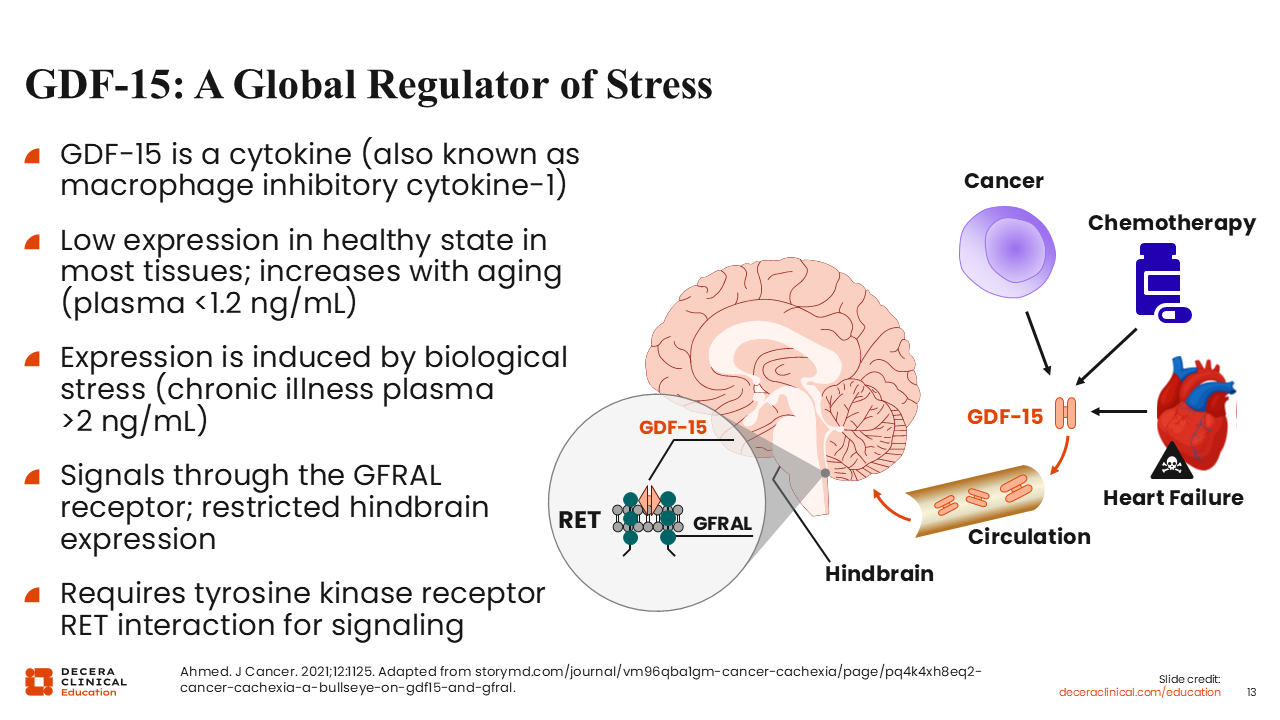
GDF-15: A Global Regulator of Stress
To reiterate, among several inflammatory cytokines associated with cancer cachexia, GDF-15 has emerged as an important regulator of stress response. It is a novel inflammatory cytokine that is detected at significantly higher concentrations in the plasma of patients with cancer compared with those without a cancer diagnosis.28,31 Its expression is triggered by biologic stress.
The biologic effects of GDF-15 are mediated through binding to the GFRAL receptor, which is expressed in the hindbrain. This signaling pathway requires interaction with the RET tyrosine kinase receptor, a coreceptor, to activate downstream signaling pathways.
The activation of the GDF-15/GFRAL signaling pathway influences eating behavior and triggers metabolic changes that impact food intake and weight changes. Reduction in food intake and weight loss caused by appetite suppression play an important role in the development of anorexia and cachexia in patients with cancer.
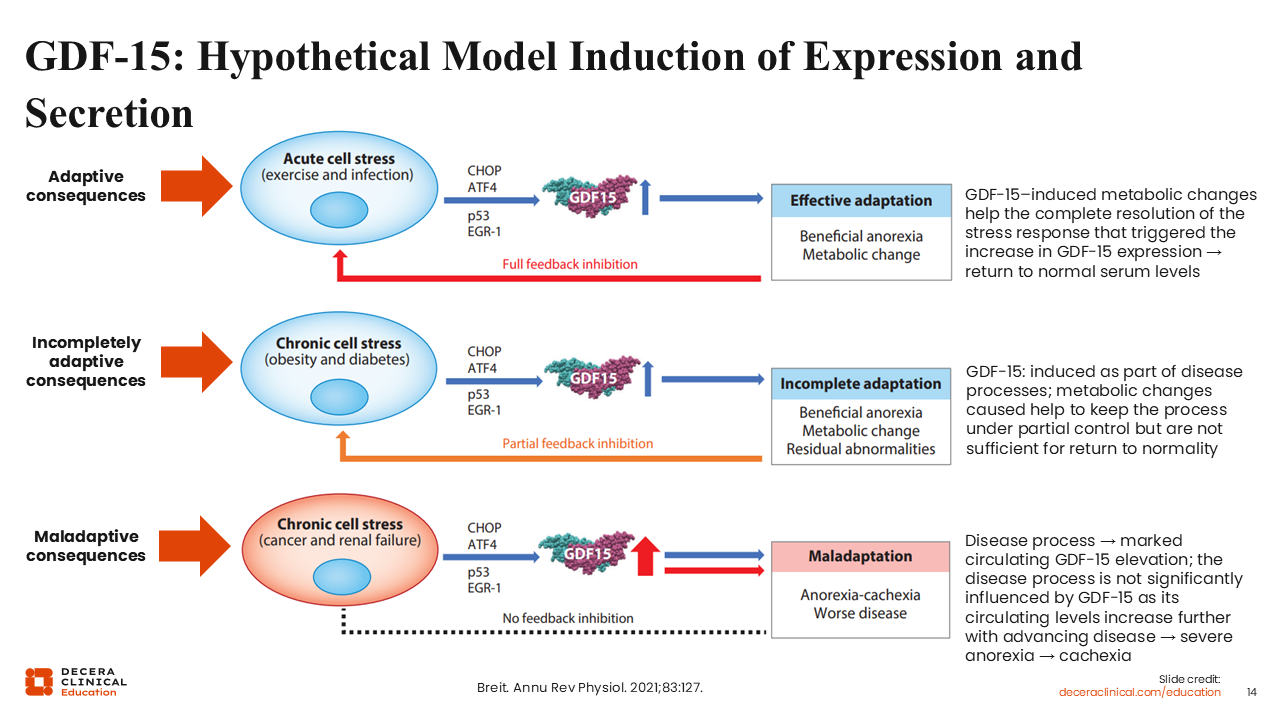
GDF-15: Hypothetical Model Induction of Expression and Secretion
It is important to note that the biologic effects of GDF-15 vary depending on the context and duration of the stress response.32 In acute cell stress conditions such as infection or exercise, transient increases in GDF-15 levels may produce adaptive metabolic responses. These responses can include temporary appetite suppression and metabolic adjustments that help the body to respond to short-term physiologic stress. Once the stress is eliminated, circulating GDF-15 levels typically return to normal serum levels.
In chronic cell stress conditions such as obesity and diabetes, sustained elevation of GDF-15 levels results in partial metabolic adaptation. Under metabolic conditions such as obesity and diabetes, elevated GDF-15 concentrations work to keep the process(es) under partial control. Here, circulating GDF-15 levels often remain elevated in the incomplete adaptation phase.
In the presence of debilitating disease conditions such as cancer or renal failure, GDF-15 levels may continue to rise substantially as the disease progresses. In this setting, the continuous increase in the levels of circulating GDF-15 triggers maladaptive consequences such as severe anorexia and subsequently cachexia.
Altogether, these observations have led to increasing interest in targeting the GDF-15 signaling pathway as a potential therapeutic strategy for patients with cancer-related cachexia.
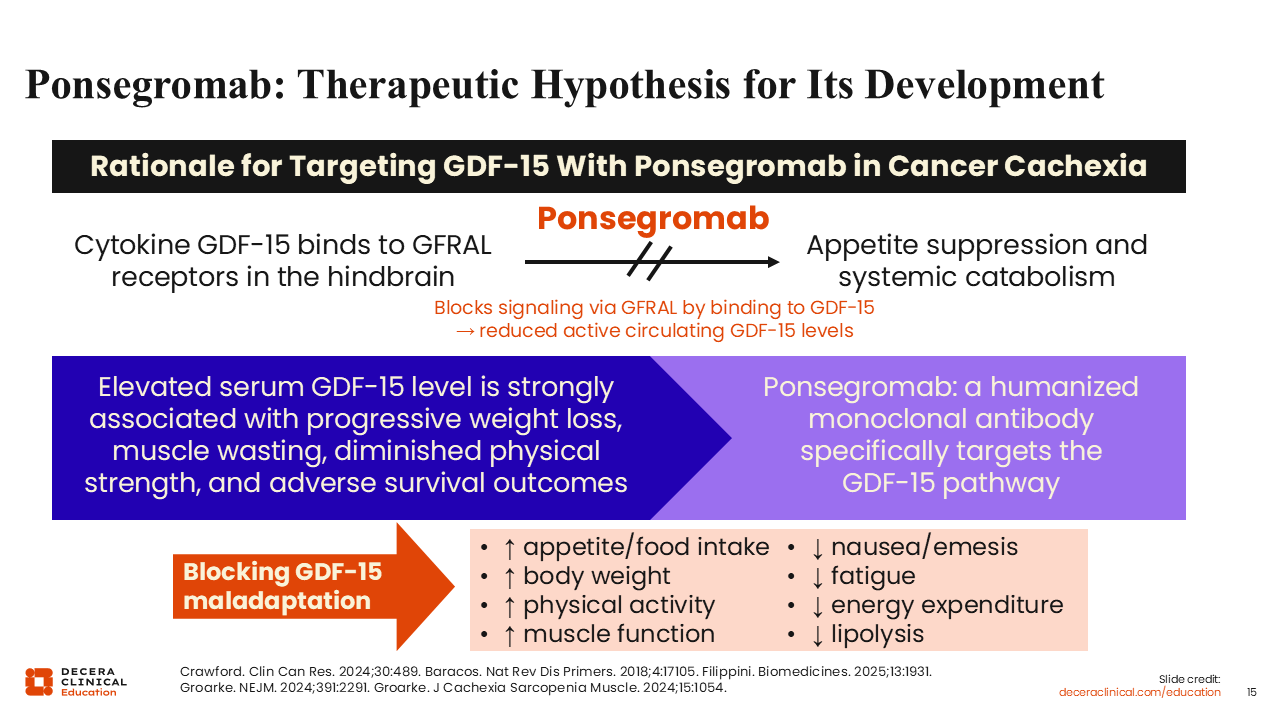
Ponsegromab: Therapeutic Hypothesis for Its Development
Ponsegromab is a humanized monoclonal antibody that targets GDF-15.33 Ponsegromab blocks the binding of GDF-15 to GFRAL receptors in the hindbrain.
Of special interest, GDF-15 blockade led to improved appetite, increased food intake, weight gain, and improvement in muscle and physical function in preclinical models mimicking GDF-15-mediated cachexia.29,34 Additional effects observed with GDF-15 blockade include reduction in nausea, fatigue, and energy expenditure, along with decreases in lipolysis.
These findings provide further support for clinical investigations of ponsegromab in patients with cancer cachexia.
Phase Ib Study of Ponsegromab in Patients With Cancer, Cachexia, and Elevated GDF-15 Levels: Summary of Findings
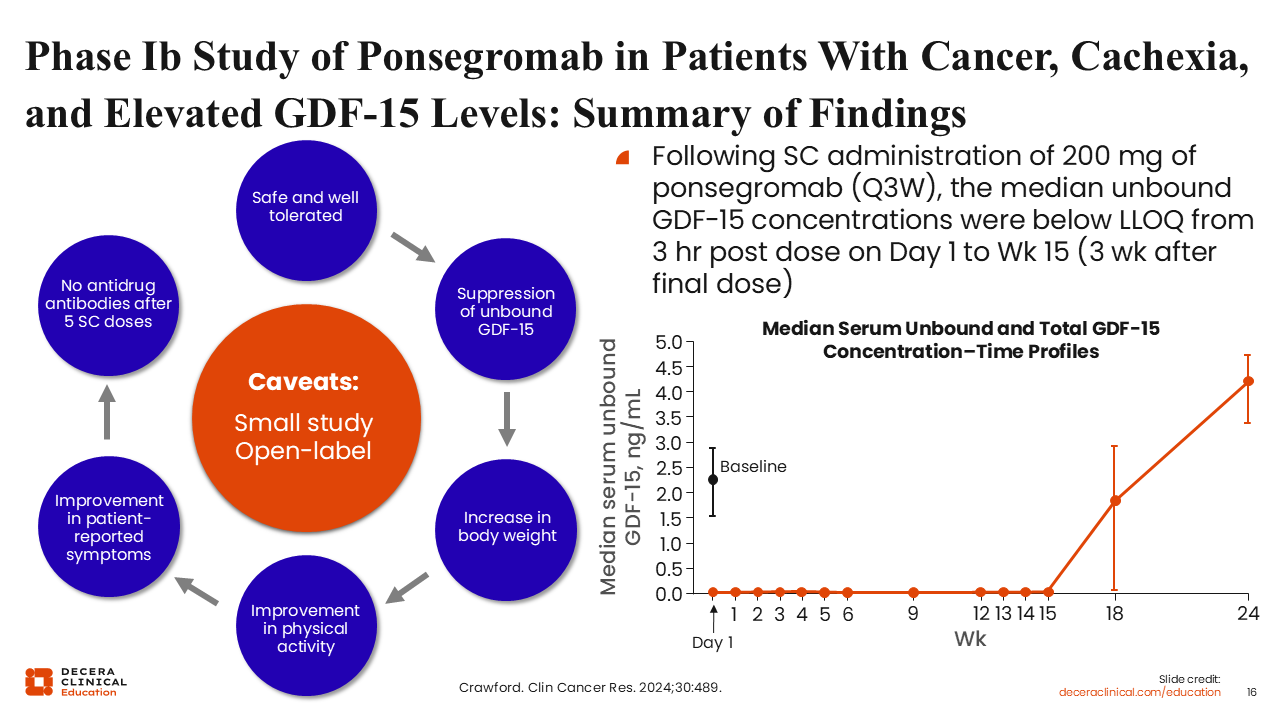
The first-in-human phase Ib trial evaluated the safety, tolerability, and pharmacokinetics/pharmacodynamics of ponsegromab in patients with cancer and cachexia (NCT04299048). In this trial, a total of 10 patients received 200 mg of ponsegromab every 3 weeks for 12 weeks in addition to standard anticancer therapy.33 In this small study, ponsegromab was well tolerated and suppressed circulating levels of GDF-15 to undetectable concentrations during the course of treatment. This effect was accompanied by increased body weight and improved physical activity, recapitulating observations noted in preclinical models of cancer cachexia.
It is important to note, however, that this is a small study without a placebo as control.
Phase II Trial of Ponsegromab in Patients With Cancer Cachexia
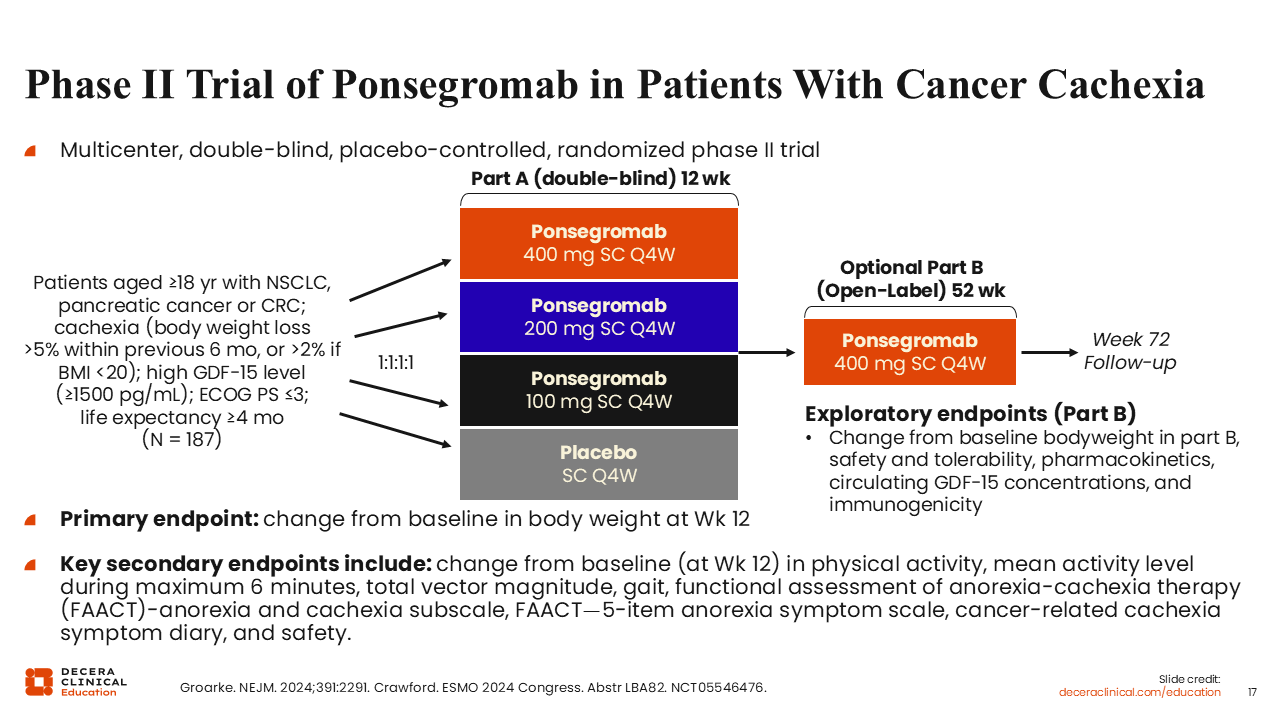
The encouraging results from the small first-in-human phase Ib trial led to the conduct of a multicenter, multipart, double-blind, placebo-controlled, randomized phase II trial (NCT05546476). Patients 18 years of age or older with NSCLC, pancreatic cancer, or CRC and cachexia were eligible to participate if they had body weight loss >5% within the previous 6 months, or body weight loss >2% and a body mass index <20 kg/m2. All patients had a high GDF-15 level (≥1500 pg/mL) and a life expectancy ≥4 months.
In total, 187 patients were randomly assigned in a 1:1:1:1 ratio to receive 100 mg, 200 mg, or 400 mg of ponsegromab subcutaneously every 4 weeks for 12 weeks vs placebo (Part A). The study included an optional phase (Part B) to continue to receive 400 mg of ponsegromab every 4 weeks for up to 1 year.18
The primary endpoint was change from baseline in body weight at Week 12. The key secondary endpoints included changes in physical activity, appetite-related symptom assessment, and safety.
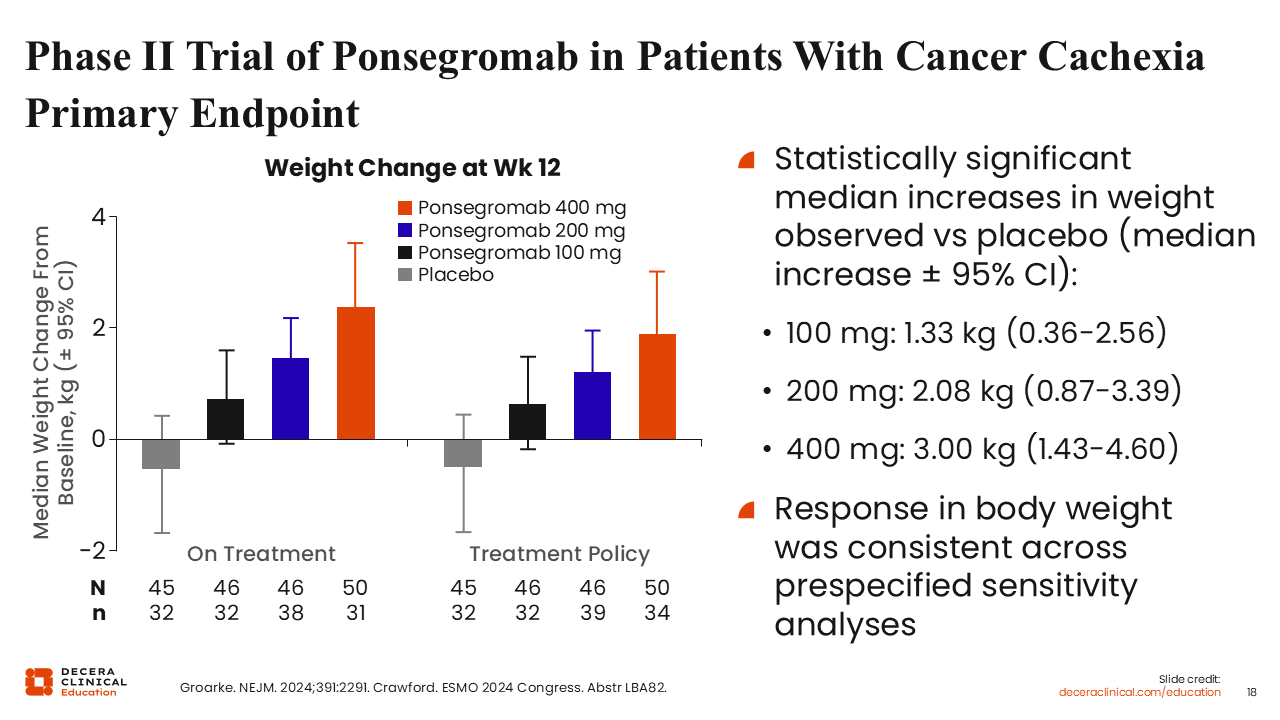
Phase II Trial of Ponsegromab in Patients With Cancer Cachexia Primary Endpoint
As expected, treatment with ponsegromab led to an increase in weight gain, and overall increase in functional activity with a reduction in symptoms of cachexia.18 Body weight was consistent across ponsegromab dose levels, with the largest increase in weight gain observed at the 400-mg dose level.
The observed improvements were consistent across prespecified sensitivity analyses, further supporting the robustness of the treatment effect.
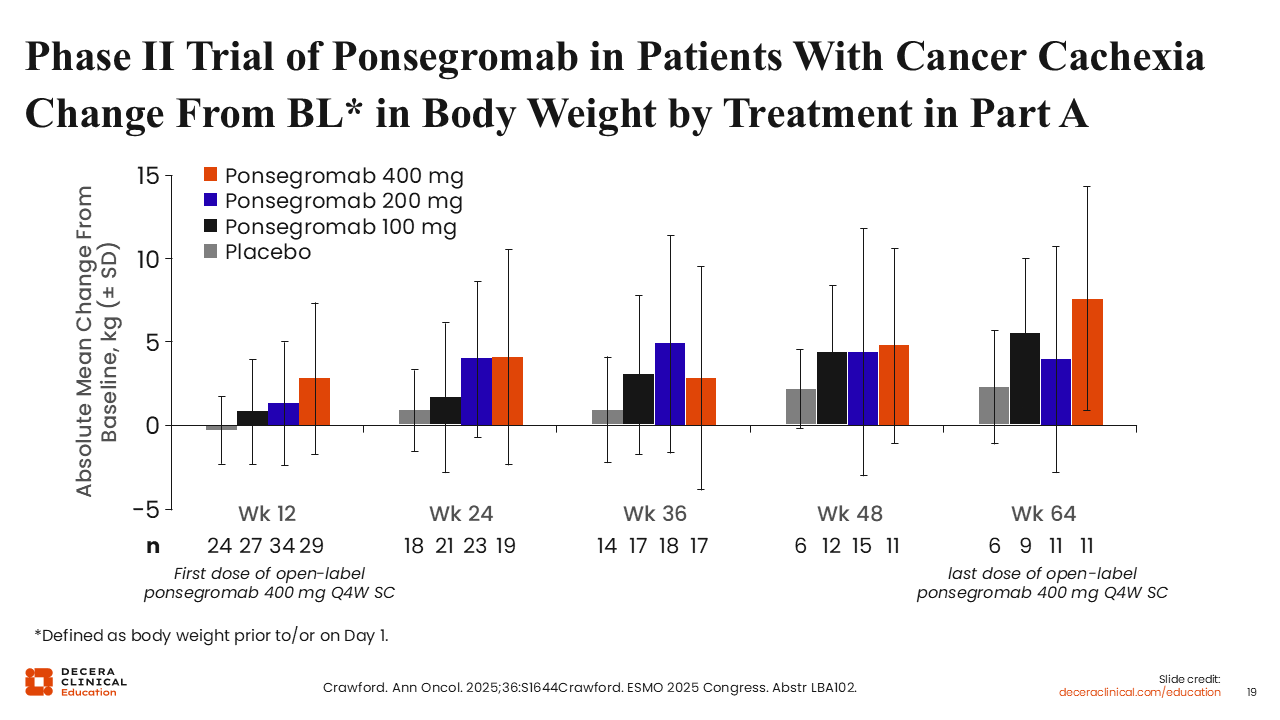
Phase II Trial of Ponsegromab in Patients With Cancer Cachexia – Change From Baseline in Body Weight by Treatment in Part A
Longer-term outcomes were evaluated in the extension phase (Part B) of the phase II trial.35 As previously stated, patients who completed Part A were eligible for treatment in Part B, where they continued to receive 400 mg of ponsegromab every 4 weeks for up to 52 weeks for a maximum of 64 weeks in Part A plus Part B.
Regardless of the initial ponsegromab dose received in part A, improvement in weight gain continued to Week 64. Although patients in the placebo group also experienced some weight gain, the gain was significantly lower compared with those who received ponsegromab, further demonstrating the durability of response with continued ponsegromab therapy.
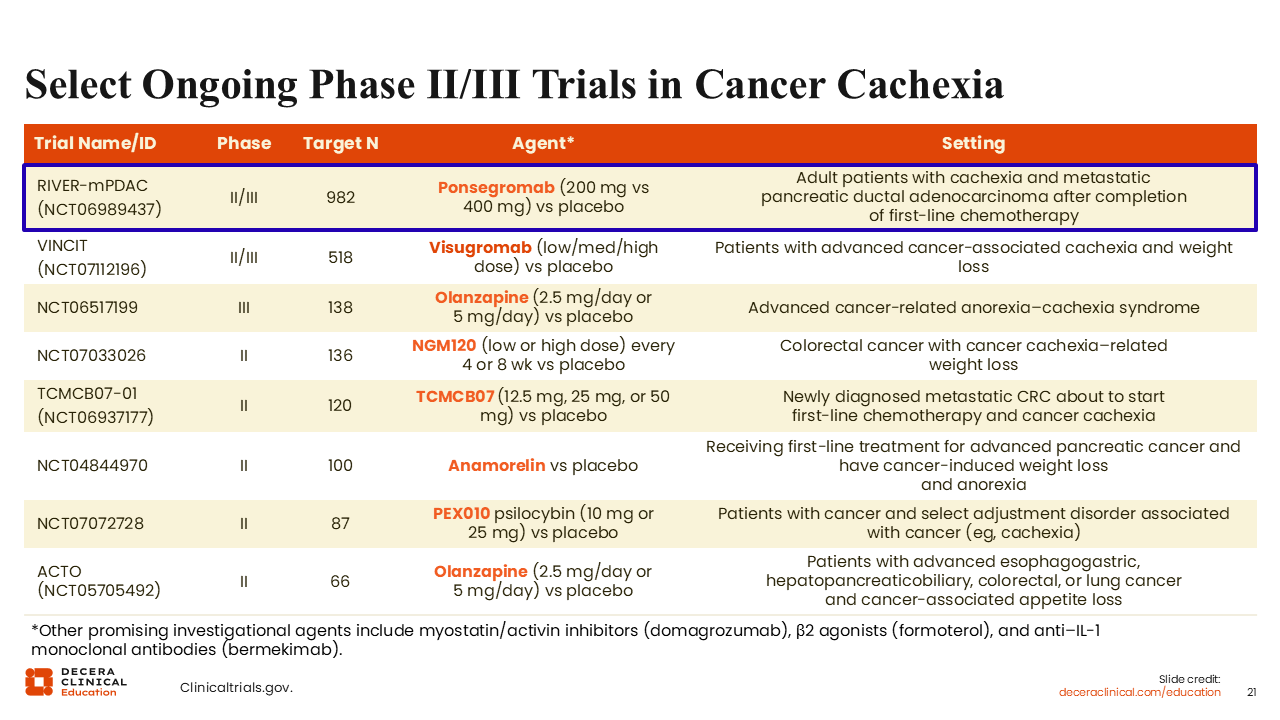
Based on these promising results, the randomized, double-blind, multicenter phase II/III RIVER-mPDAC trial is investigating the efficacy, safety, and tolerability of standard chemotherapy plus ponsegromab vs standard chemotherapy alone for adult patients with newly diagnosed metastatic pancreatic ductal adenocarcinoma and cachexia (NCT06989437).
The primary endpoints are change from baseline in body weight for ponsegromab compared to placebo and change from baseline in functional assessment of anorexia/cachexia. The key secondary endpoints include overall survival, progression-free survival, and objective response rate. I think the readouts from the RIVER-mPDAC trial will be interesting and will certainly extend our knowledge in the field.
Other Anti–GDF-15/GFRAL Therapies in Development for Cancer Cachexia
In addition to ponsegromab, several other agents targeting GDF-15/GFRAL are in the clinical pipeline. For example, the anti–GDF-15 humanized monoclonal antibodies, rilogrotug and visugromab, are being investigated in clinical trials for patients with cancer and cachexia (NCT05865535, NCT07112196). Of interest, visugromab is also being investigated in a randomized phase II trial in combination with an immune checkpoint inhibitor (nivolumab) with or without docetaxel as second-line therapy for patients with advanced NSCLC (NCT07246863). Although the primary endpoint of this trial is objective response rate, an important secondary endpoint of the study is the assessment of the participants' well-being using an NSCLC symptom assessment questionnaire.
Other therapeutic strategies under investigation include the study of the anti-GFRAL humanized monoclonal antibodies, NGM-120 and JMT-203, both of which are currently in early-phase clinical development (NCT07033026, NCT06868849).
Select Ongoing Phase II/III Trials in Cancer Cachexia
The phase II/III VINCIT trial is investigating visugromab (low/med/high dose) vs placebo for patients with advanced cancer-associated cachexia and weight loss (NCT07112196).
Finally, it is important to re-emphasize that there are other approaches under investigation for patients with cancer cachexia besides those targeting GDF-15 or GFRAL. For example, there is an ongoing phase III trial investigating olanzapine (2.5 mg/day or 5 mg/day) vs placebo for patients with advanced cancer-related anorexia/cachexia syndrome (NCT06517199).
Other investigational agents in early-phase clinical trials for patients with cancer and cachexia include TCMCB07, anamorelin, and PEX010, a natural botanical psilocybin agent.
Overall Conclusions
With an increasing number of investigational agents in cancer cachexia, more clinical trials will become available for patient participation. These studies have the potential to improve our understanding of optimal approaches for the management of our patients and ultimately, increase the number of available treatment options for cancer cachexia in the future.
Cancer cachexia is a complex, multifactorial condition and remains a major unmet medical need. Monitoring weight loss in patients can help with early detection and prompt intervention. Being a multifactorial syndrome, management requires a multimodal approach including nutritional counseling and support, as well as exercise and pharmacologic interventions tailored to each patient’s symptoms and needs.
With improved understanding of the biology and multifaceted nature of cancer cachexia, we will become better able to promptly recognize and appropriately mitigate the clinical consequences of cancer-associated cachexia. The multimodal treatment approach stands to improve quality of life, prolong survival, and as improve the overall well-being of our patients with cancer and cachexia.
I am hopeful that continued research will lead to the development of novel pharmacologic agents targeting the biologic mechanisms driving this syndrome, ultimately leading to significantly improved treatment outcomes for our patients with cancer and cachexia.