CE / CME
Clinical Focus: Translating Discoveries Into Clinical Practice to Advance the Care of Patients With Cutaneous Squamous Cell Carcinoma
Physician Assistants/Physician Associates: 0.50 AAPA Category 1 CME credit
Pharmacists: 0.50 contact hour (0.05 CEUs)
Physicians: maximum of 0.50 AMA PRA Category 1 Credit™
Nurse Practitioners/Nurses: 0.50 Nursing contact hour
Released: January 26, 2026
Expiration: July 25, 2026
Activity
Overview of cSCC
cSCC is the second most common skin cancer and a leading cause of mortality among keratinocyte carcinomas.1 Although the majority of tumors are small, well-differentiated, and effectively managed with standard surgical excision or Mohs micrographic surgery, a clinically significant subset of patients presents with high-risk or advanced disease.1,2
High-risk features including large tumor size, deep invasion, perineural or lymphovascular involvement, and regional nodal disease are associated with substantially higher rates of locoregional recurrence, distant metastasis, and disease-specific mortality.2,3 Historically, management of these tumors relied on extensive surgery followed by adjuvant RT, with cytotoxic chemotherapy used selectively for unresectable cases. However, these traditional approaches often increased treatment-related morbidity without consistently improving long-term oncologic outcomes, leaving a significant unmet need for durable systemic options.3,4
The introduction of ICIs, specifically those targeting the PD-1 pathway, has fundamentally transformed the care of advanced cSCC. These therapies leverage the high tumor mutational burden characteristic of cSCC to generate meaningful and durable responses in both locally advanced and metastatic settings.4,5
The current gold standard of care is now expanding beyond the metastatic setting to include multidisciplinary-led neoadjuvant and adjuvant strategies. In particular, the phase III C-POST trial established adjuvant cemiplimab as a new standard of care for resected high-risk disease, demonstrating a 68% reduction in recurrence risk following definitive surgery and radiation.2,6 This module reviews the integration of these emerging evidence-based strategies into clinical practice to optimize outcomes for patients across the advanced cSCC spectrum.
Patient With Newly Diagnosed cSCC
Gordie is an 80-year-old man who presents with a large scalp lesion that has progressively enlarged. Biopsy confirms cSCC. Cross-sectional imaging reveals a deeply invasive primary lesion with suspected involvement of the calvarium and enlarged cervical lymph nodes, features typical of very high–risk head and neck cSCC. His past medical history is notable for hypertension and mild chronic kidney disease, and he lives independently with limited family support.
This case illustrates the common scenario of an older patient with advanced cSCC, complex anatomy, and competing risks, raising questions about operability, perioperative systemic therapy, and the balance between aggressive treatment and preservation of quality of life (QoL).
Multidisciplinary Care in Nonmelanoma Skin Cancers
Patients like Gordie benefit from early referral to a multidisciplinary tumor board. Optimal management of advanced cSCC often involves dermatologists and Mohs surgeons, head and neck or surgical oncologists, radiation oncologists, and medical oncologists, supported by radiologists, pathologists, and advanced practice providers.7 Complex cases may require combined resections with reconstruction, adjuvant radiation therapy (RT), and systemic immunotherapy. Multidisciplinary care facilitates coordination of sequencing and timing, helps anticipate functional and cosmetic outcomes, and ensures that treatment decisions align with the patient’s values and goals.2,3 Involving palliative care, social work, and rehabilitation services early can further support older or frail patients through treatment.8
Rationale for PD-1 Inhibition in cSCC
cSCC is characterized by very high tumor mutational burden due to chronic ultraviolet damage, creating abundant neoantigens that can be recognized by T-cells.1 Many cSCCs express PD-L1, and higher PD-L1 expression has been associated with more aggressive tumor behavior.3 Immunosuppression, such as that seen in solid organ transplant recipients, dramatically increases the incidence and aggressiveness of cSCC, highlighting the importance of intact immune surveillance.1 These features collectively provide biologic rationale for targeting the PD-1/PD-L1 pathway in cSCC and help explain the robust and durable responses observed with PD-1 inhibitors in advanced disease.4,5
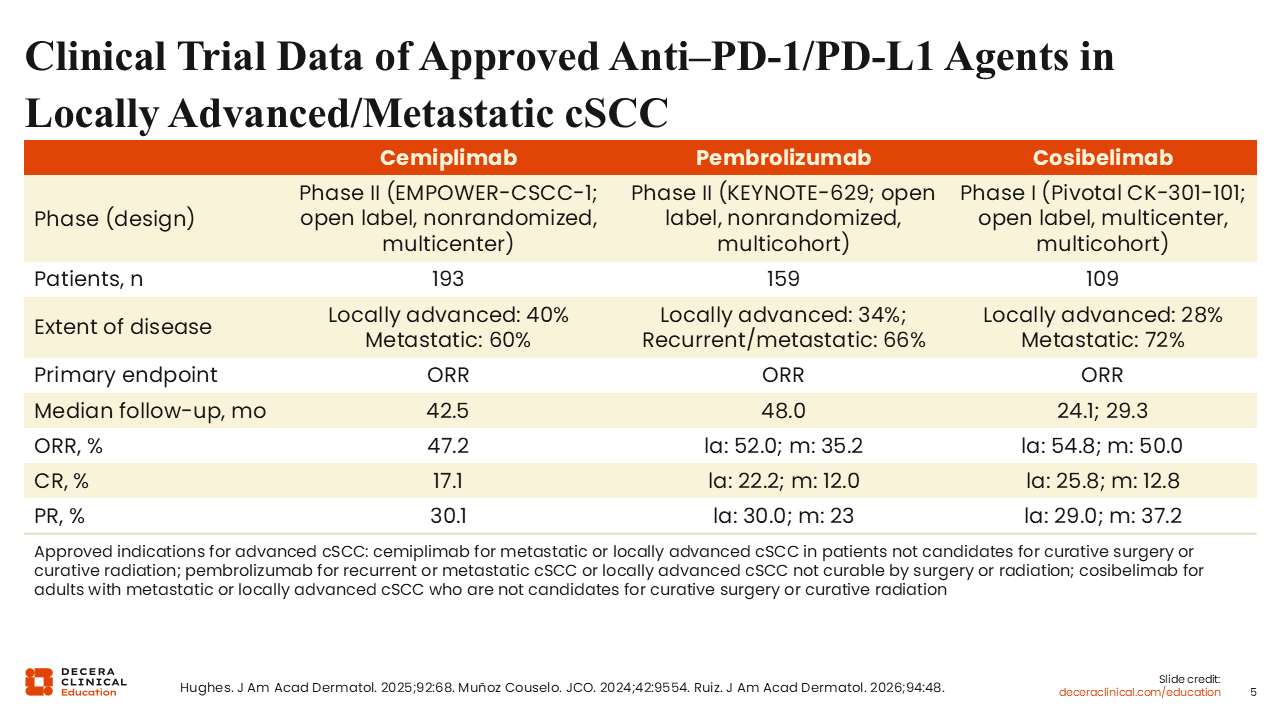
Clinical Trial Data of Approved Anti–PD-1/PD-L1 Agents in Locally Advanced/Metastatic cSCC
PD-1 blockade is the standard systemic therapy for unresectable or metastatic cSCC, supported by multiple pivotal clinical trials. In EMPOWER-CSCC-1, cemiplimab produced a 47.2% objective response rate (ORR) in 193 patients, with durable responses at a median follow-up of 42.5 months.5,9 KEYNOTE-629 showed similarly meaningful activity for pembrolizumab, with ORRs ranging from approximately 35% to 52% across locally advanced and recurrent/metastatic cohorts and follow-up extending beyond 50 months.5,10 Cosibelimab demonstrated ORRs of 54.8% in locally advanced disease and 50.0% in metastatic disease, with sustained responses at 24-29 months of follow-up in a pivotal phase I trial.11 Real-world data further support the effectiveness and tolerability of PD-1 inhibitors in older patients with advanced cSCC.12,13 These successes in advanced disease laid the groundwork for exploring PD-1 blockade in the perioperative setting for high-risk resectable cSCC.
Transition to the Perioperative Setting
With PD-1 inhibitors now established in unresectable/metastatic cSCC, the next logical question became whether earlier use of immunotherapy—either before or after surgery and RT—could improve long-term outcomes for patients with high-risk but resectable disease.2 Two complementary strategies emerged.
- Neoadjuvant PD-1 therapy involves administering a short course of PD-1 blockade before planned surgery. Potential advantages include:
- Tumor shrinkage and improved resectability, leading to a higher likelihood of negative margins
- The ability to evaluate pathologic response as an early surrogate for long-term outcomes
- Treating the intact tumor to provide a rich antigen source for immune priming
- Adjuvant PD-1 therapy administered after definitive surgery and RT is a strategy that aims to:
- Eradicate micrometastatic disease and reduce recurrence risk6,14
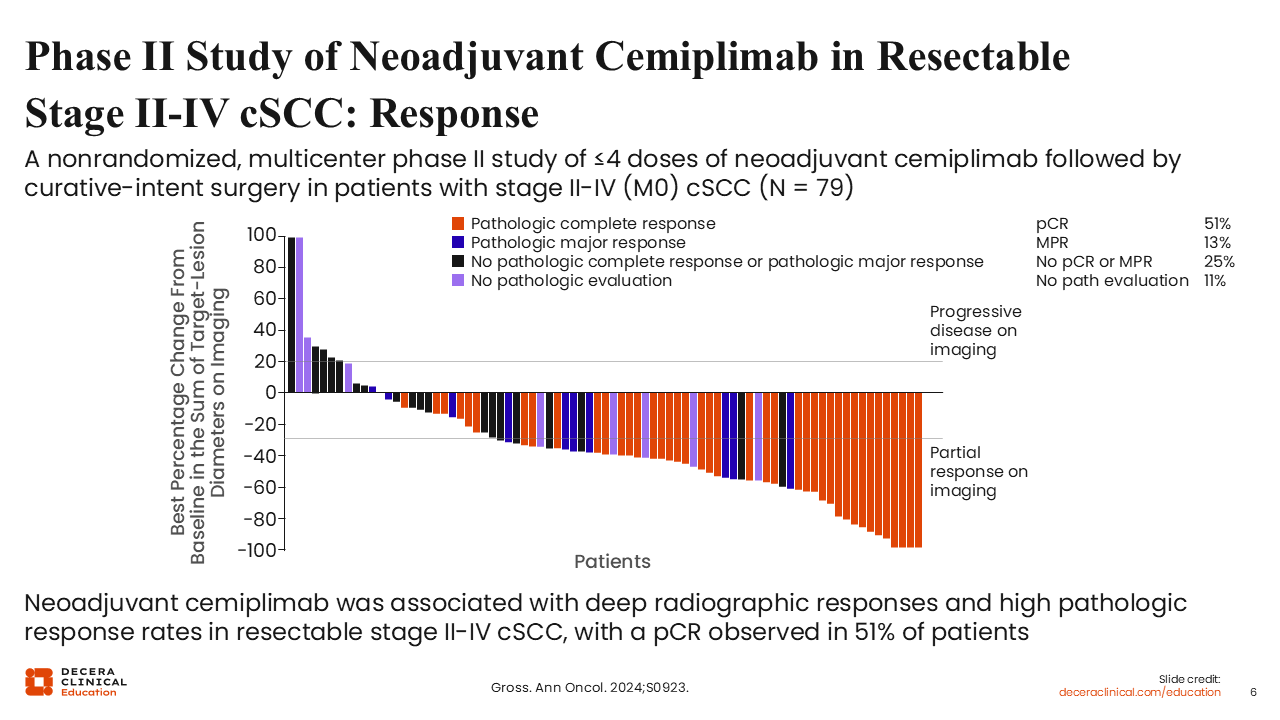
Phase II Study of Neoadjuvant Cemiplimab in Resectable Stage II-IV cSCC: Response
A multicenter phase II trial evaluated cemiplimab in resectable stage II-IV (M0) cSCC. Eligible patients had tumors considered resectable with curative intent but associated with substantial expected morbidity. Participants received cemiplimab 350 mg IV every 3 weeks for up to 4 preoperative doses followed by surgery at a predefined time point. Postoperative RT and further systemic therapy were allowed at investigators’ discretion, mimicking real-world practice. The primary endpoint was pathologic complete response (pCR) in the resection specimen, with major pathologic response (MPR) and radiographic response as key secondary endpoints, along with event-free survival (EFS) and overall survival (OS).14
In the phase II multicenter study, neoadjuvant cemiplimab demonstrated substantial activity in patients with resectable stage II-IV cSCC. The primary analysis showed that 50.6% of patients achieved a pCR, defined as no viable tumor cells in the surgical specimen, and an additional 12.7% achieved an MPR, defined as ≤10% viable tumor. Collectively, nearly two thirds (63.3%) of patients experienced 1 of these deep pathologic responses.14
Follow-up analyses confirmed that these responses correlate with excellent early disease control. At a median follow-up of 18.7 months, no patients who achieved a pCR experienced a recurrence, and only a single patient with an MPR relapsed. In contrast, disease recurrences were significantly more frequent among nonresponders.14 These results strongly support the immunogenicity of cSCC and the potential for neoadjuvant PD-1 therapy to change the surgical landscape in selected cases.
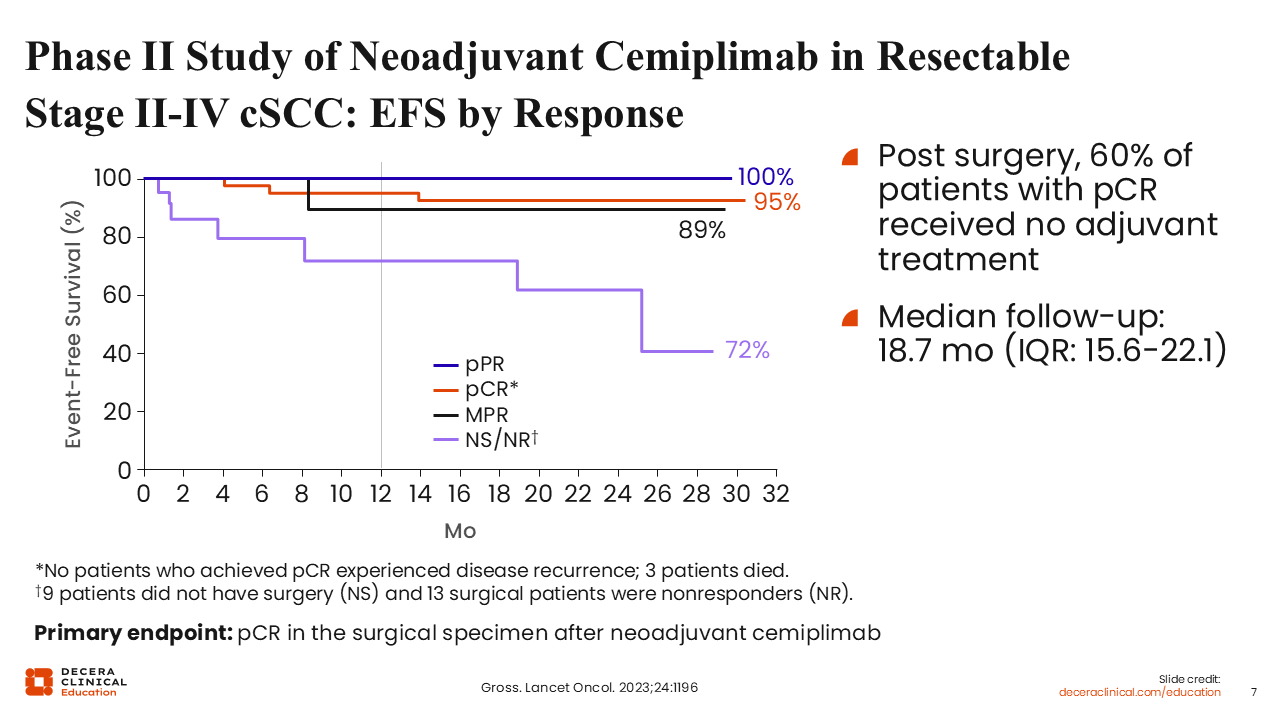
Phase II Study of Neoadjuvant Cemiplimab in Resectable Stage II-IV cSCC: EFS by Response
Long-term follow-up from the phase II study of neoadjuvant cemiplimab in resectable stage II-IV cSCC has demonstrated favorable and durable survival outcomes. At a median follow-up of 18.7 months, the estimated 12-month EFS was 89% and OS was 92%.15
These outcomes were highly correlated with pathologic response: no recurrences were observed among patients who achieved a pCR, and high EFS rates were maintained in those with MPR or partial pathologic response (pPR). Of note, the depth of response allowed for significant treatment de-escalation; 60% of patients with a pCR received no adjuvant treatment post-surgery yet remained disease free. In contrast, EFS was substantially lower for nonresponders and those who did not proceed to surgery. Most patients underwent surgery as scheduled, confirming that neoadjuvant cemiplimab is both feasible and highly effective in changing the management of high-risk cSCC.14,15
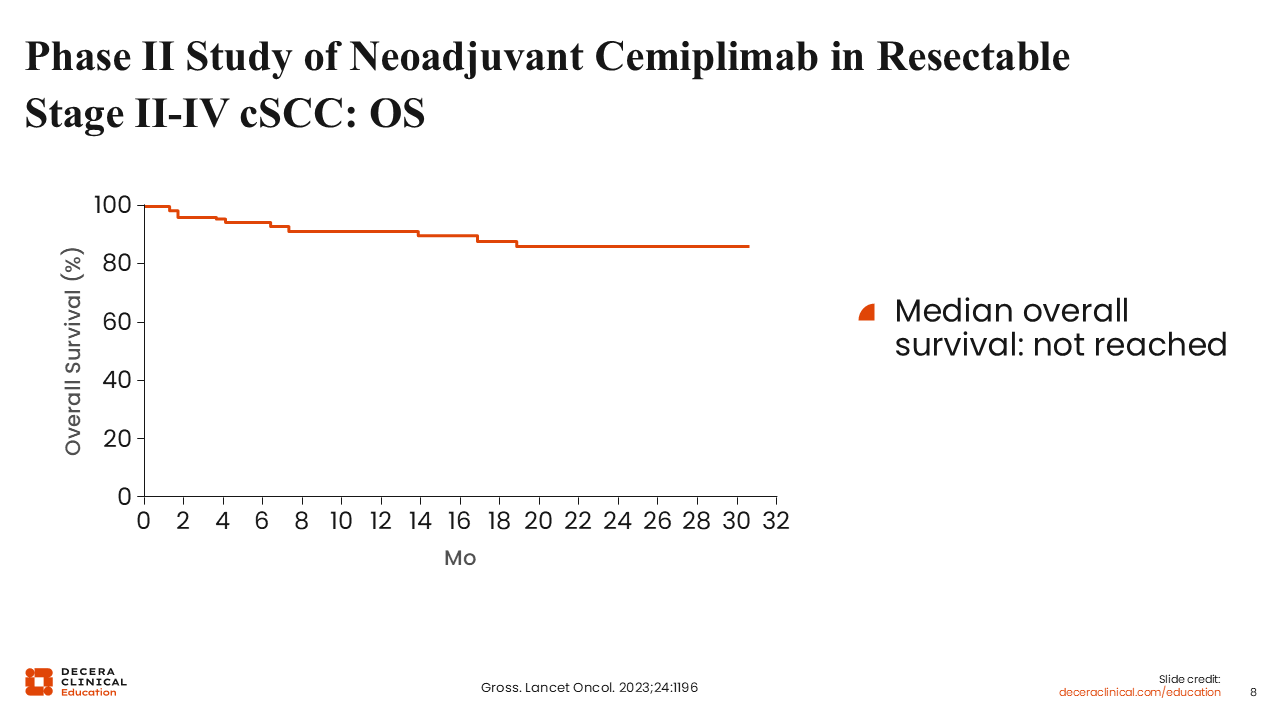
Phase II Study of Neoadjuvant Cemiplimab in Resectable Stage II-IV cSCC: OS
The phase II study of neoadjuvant cemiplimab followed by surgery in patients with resectable stage II-IV (M0) cSCC continues to show high OS. Among the 79 treated patients, the estimated 12-month OS rate was 92% (95% CI: 83%-96%), and the Kaplan–Meier curve remains high and flat over time, with median OS not reached at the time of analysis.15
Most deaths observed in the study were not attributed to cSCC, consistent with the curative-intent treatment setting and the older population with substantial comorbidities. Patients who achieved pCR or MPR showed particularly favorable survival outcomes, reflecting the association between deep pathologic response and improved prognosis in this cohort.15
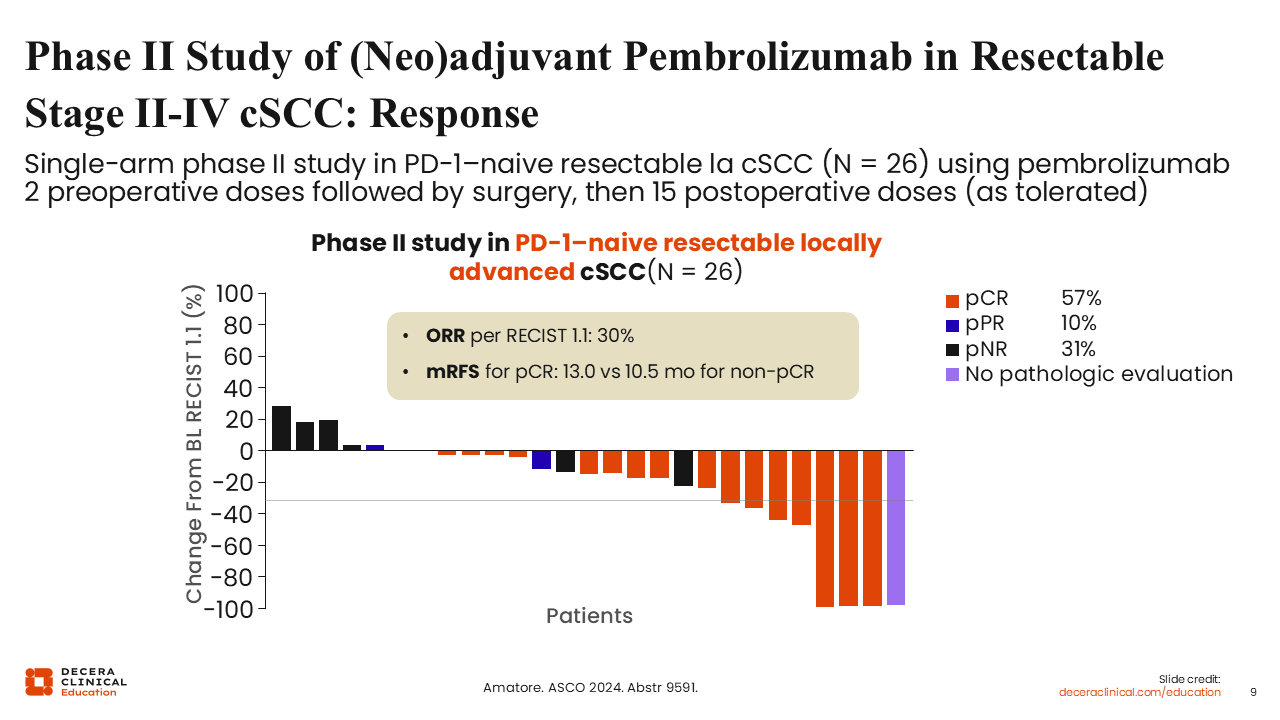
Phase II Study of (Neo)adjuvant Pembrolizumab in Resectable Stage II-IV cSCC: Response
A phase II single-arm trial conducted by Amatore and colleagues in 2024 evaluated a perioperative regimen of pembrolizumab in patients with resectable, locally advanced cSCC. The study enriched for a very high–risk population, including those with T3 disease or node-positive status. Among 26 evaluable patients, the ORR was 30% by imaging, but pathologic responses were far more pronounced: 57% achieved a pCR, and 8% and 31% showed pPR and pathologic non-response (pNR), respectively.16
Median RFS was 13 months for patients with a pCR and 10.5 months for those without pCR, based on the available follow-up.16 Although the study is single-arm with a small cohort and limited follow-up, the high pCR rate and early RFS differences support further evaluation of perioperative PD-1 blockade in resectable, locally advanced cSCC.
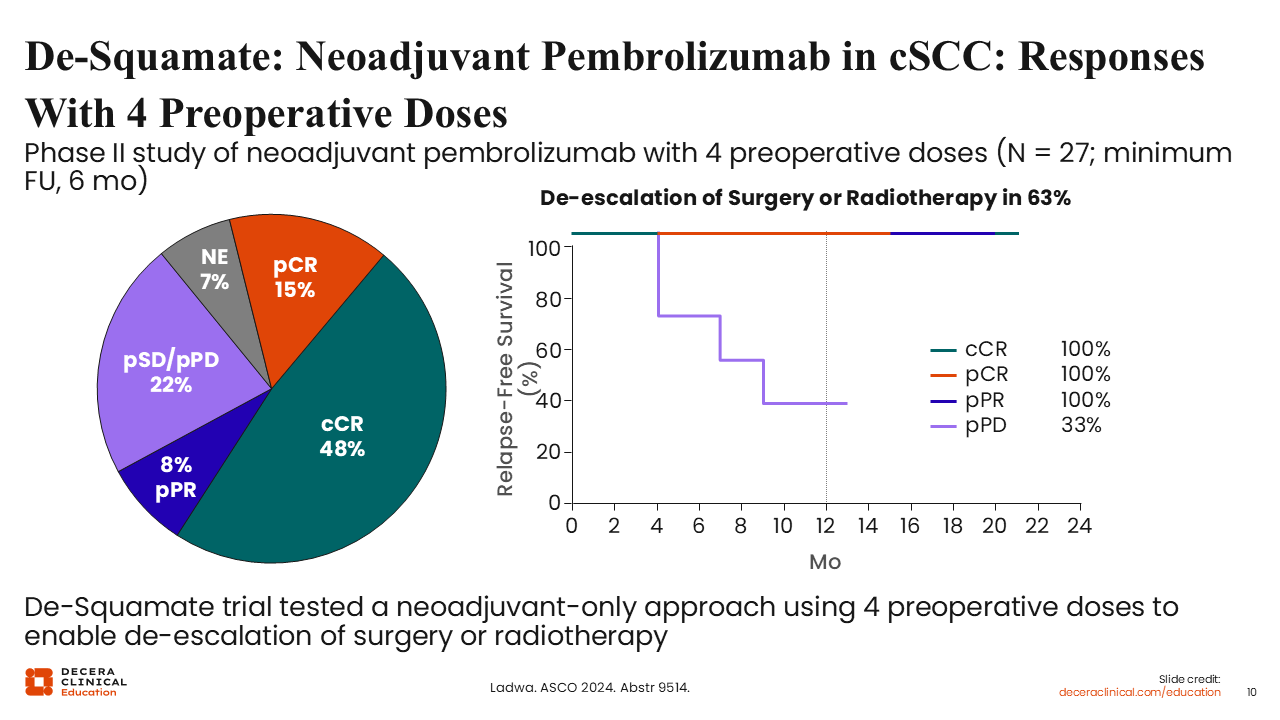
De-Squamate: Neoadjuvant Pembrolizumab in cSCC: Responses With 4 Preoperative Doses
The phase II trial evaluated a risk-adapted de-escalation strategy using neoadjuvant pembrolizumab in patients with resectable, locally advanced stage II-IV cSCC who were otherwise facing highly morbid surgery and/or adjuvant RT. Patients received 4 doses of pembrolizumab 200 mg every 3 weeks, followed by restaging with CT/MRI and PET imaging. Patients who achieved a clinical complete response (cCR) and had negative mapping biopsies were eligible to omit surgery and RT, while those with residual disease proceeded to surgery with the option for de-escalated postoperative RT if a pCR or MPR was achieved.17
Neoadjuvant pembrolizumab produced cCR in approximately half of patients, with additional pCR and pPR observed. Only approximately one fifth of patients had pathologic stable or progressive disease. Early RFS patterns favored deep responders: patients with cCR or pCR maintained 100% RFS at 6-12 months, whereas outcomes were less favorable among patients with only partial or no pathologic response. Overall, 63% of patients were able to de-escalate surgery and/or RT, directly linking the depth of neoadjuvant response to clinically meaningful reductions in treatment intensity.17
As a small, single-arm study with early follow-up, these findings remain exploratory, and additional prospective data will be required to confirm the safety and durability of this de-escalation approach.
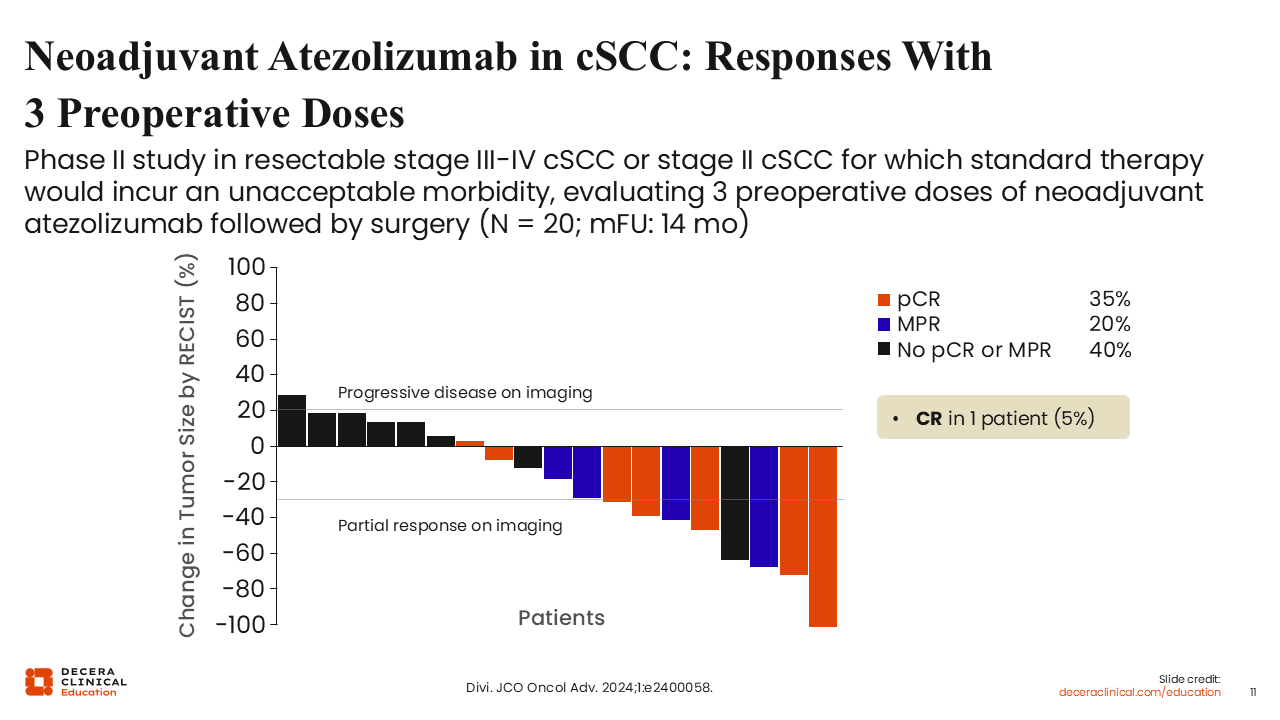
Neoadjuvant Atezolizumab in cSCC: Responses With 3 Preoperative Doses
This single-arm phase II study evaluated 3 doses of neoadjuvant atezolizumab in patients with resectable stage III-IV cSCC or stage II disease in which standard surgery and/or RT would result in unacceptable morbidity (N = 20; median follow-up, 14 months).18 Patients received atezolizumab 1200 mg every 21 days prior to planned curative-intent surgery.
Most patients experienced some degree of tumor shrinkage. RECIST responses occurred in 40%, and only 1 patient (5%) demonstrated radiographic progression before surgery. Thirty five percent of patients achieved a pCR and 20% achieved an MPR; 40% had neither pCR nor MPR despite radiographic improvements, and 1 patient (5%) achieved a confirmed radiographic complete response.18
Collectively, these results indicate that neoadjuvant atezolizumab is feasible and biologically active in advanced, surgically resectable cSCC, producing meaningful pathologic responses with a low rate of preoperative progression. However, as a small, single-arm feasibility study, these findings are preliminary and not sufficient to change clinical practice.18
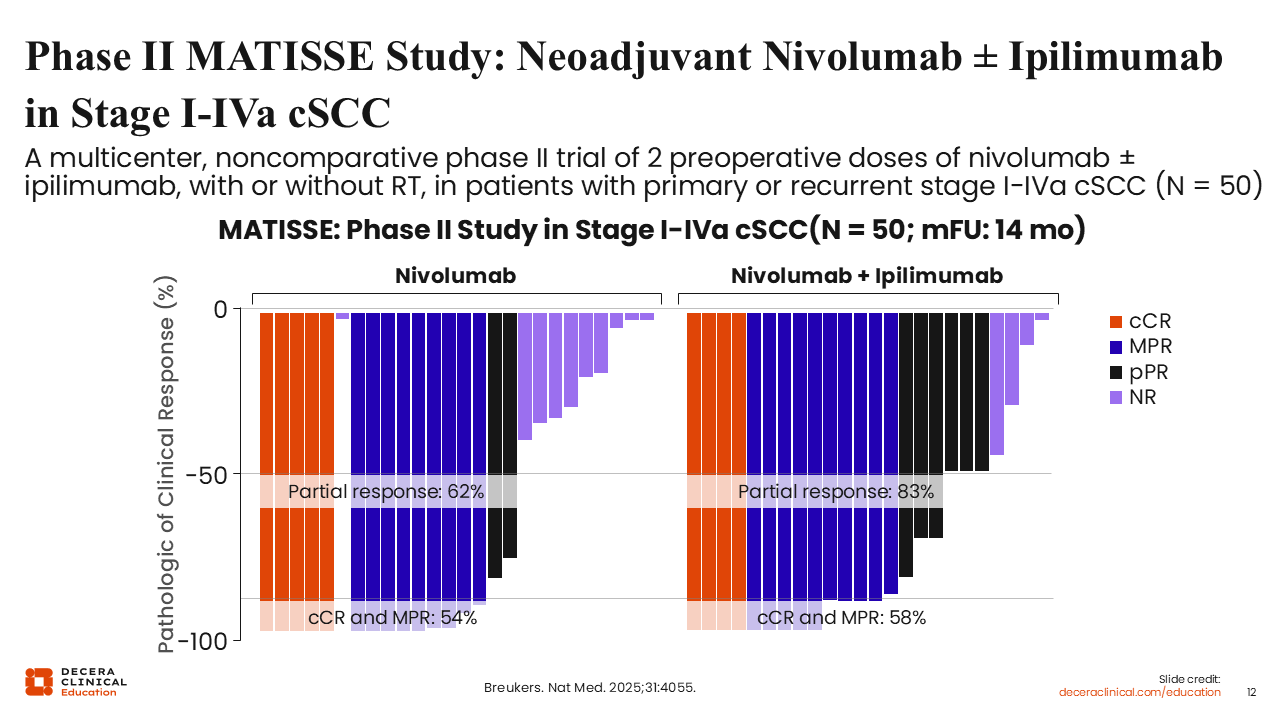
Phase II MATISSE Study: Neoadjuvant Nivolumab ± Ipilimumab in Stage I-IVa cSCC
The randomized, multicenter phase II MATISSE trial investigated an "ultra-short" neoadjuvant regimen in patients with resectable stage I-IVa cSCC facing extensive surgery. Patients received either 2 doses of nivolumab or a combination of 2 doses of nivolumab plus 1 low dose of ipilimumab over just 4 weeks.19
The data demonstrate substantial reductions in viable tumor across both arms. In the nivolumab monotherapy arm, 62% of patients achieved at least a partial response, and 54% had either a cCR or an MPR. In the nivolumab plus ipilimumab arm, responses were deeper: 83% of patients achieved at least a partial response, and 58% reached cCR or MPR. Among patients who proceeded to surgery, total pathologic response (defined as MPR plus pPR) was 55% with nivolumab alone and 80% with nivolumab plus ipilimumab. Early follow-up shows excellent disease-specific survival among patients who achieved a meaningful clinical or pathologic response.19
Taken together, MATISSE provides evidence that an ultra-short, 2-dose neoadjuvant regimen of nivolumab with or without ipilimumab can induce high rates of deep response in patients otherwise facing surgery. The findings raise important questions for future study regarding the necessary extent of surgery and RT in patients who achieve robust neoadjuvant responses, though such de-escalation strategies remain investigational.
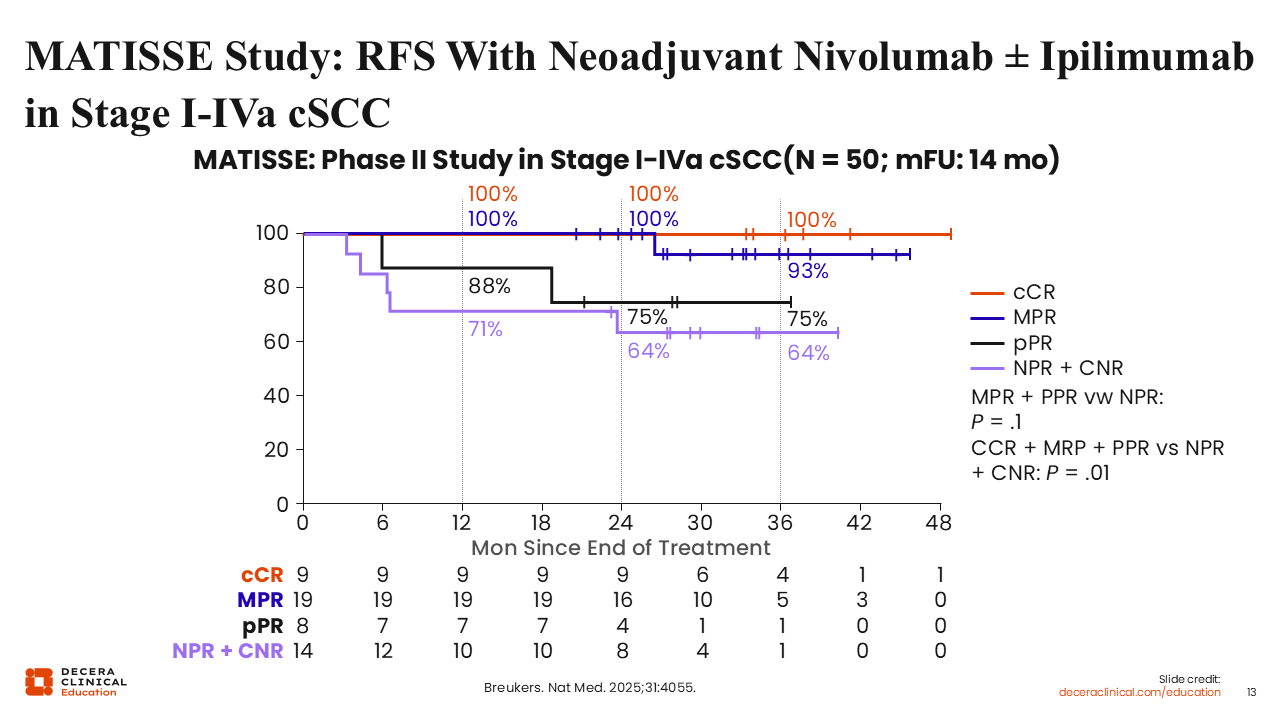
MATISSE Study: RFS With Neoadjuvant Nivolumab ± Ipilimumab in Stage I-IVa cSCC
In the MATISSE trial, RFS strongly correlated with the depth of response following just 2 doses of neoadjuvant nivolumab with or without ipilimumab. Outcomes were analyzed across 4 response categories: clinical complete response (cCR), major pathologic response (MPR), pathologic partial response (pPR), and no response (no pathologic response [NPR] plus clinical non-response [CNR]).
Patients who achieved cCR or MPR had outstanding outcomes, with 2-year RFS of 100%. Those with pPR had intermediate outcomes (2-year RFS approximately 75%-88%), whereas patients with NPR plus CNR had the poorest RFS of roughly 64% to 71% at 2 years. When cCR, MPR, and pPR were grouped together and compared with NPR plus CNR, the difference in RFS was statistically significant (P = .01).19 These findings underscore that any meaningful response to an ultra-short neoadjuvant regimen serves as a powerful surrogate for long-term disease control, while nonresponders may require more intensive or alternative interventions to avoid relapse.
Very High–Risk CSCC: When to Consider Neoadjuvant Cemiplimab
The NCCN guidelines now state that neoadjuvant cemiplimab may be considered for select patients with very high–risk cSCC following a multidisciplinary review.2 This recommendation is specifically tailored for patients with rapidly growing tumors, in-transit metastases, borderline resectable disease, or cases where surgical resection alone is unlikely to be curative or would result in substantial functional impairment.
The NCCN emphasizes that neoadjuvant cemiplimab therapy remains an optional strategy, and treatment decisions should be individualized based on institutional expertise and tumor-specific factors.2 Although neoadjuvant PD-1 blockade offers a new path for organ preservation and improved local control, surgical resection—often followed by adjuvant RT—remains the primary curative-intent cornerstone for the majority of high-risk patients.
Patient Case (cont’d)
Gordie undergoes neoadjuvant treatment with cemiplimab. After 2 doses, he demonstrated a clinical response with no visible or palpable tumor on physical examination and no residual disease identified on CT imaging. Following multidisciplinary evaluation, Gordie elected to discontinue systemic therapy and defer surgical intervention. He continued to show no evidence of disease 1 year after his final immunotherapy dose.
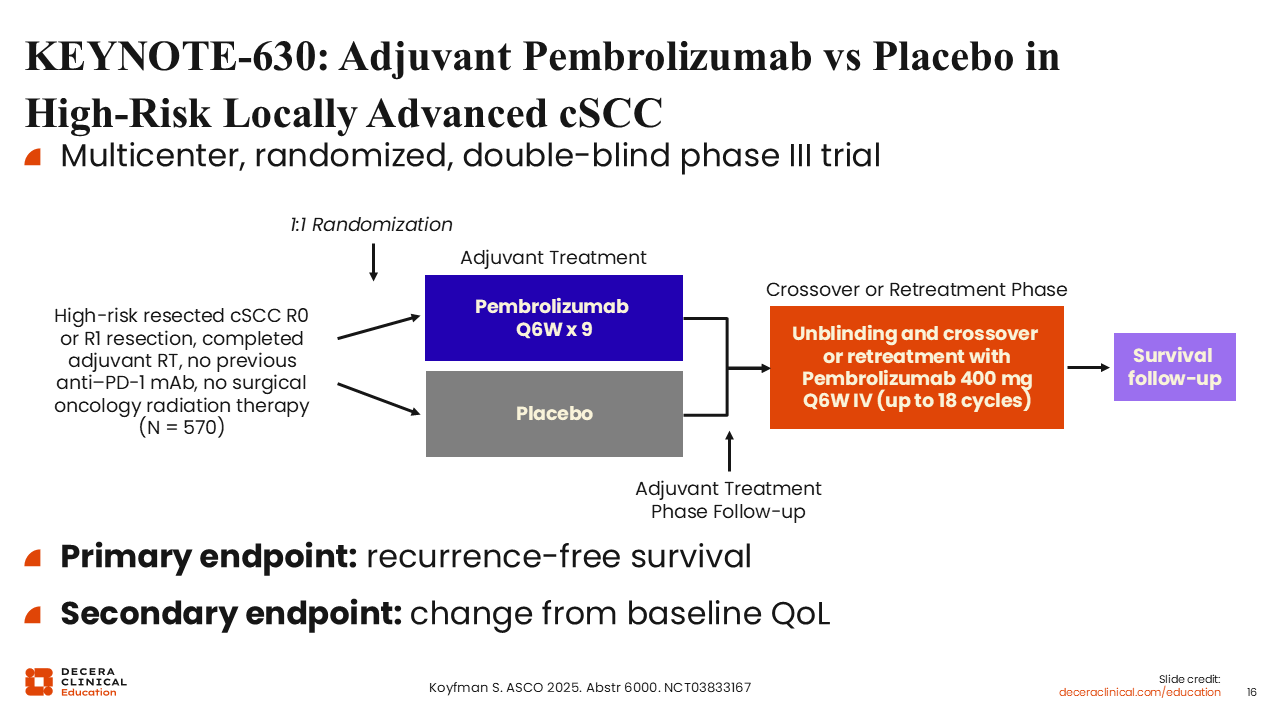
KEYNOTE-630: Adjuvant Pembrolizumab vs Placebo in High-Risk Locally Advanced cSCC
KEYNOTE-630 was a randomized, double-blind, placebo-controlled phase III trial evaluating adjuvant pembrolizumab in patients with resected, high-risk locally advanced cSCC who had completed adjuvant RT. The study enrolled patients with high-risk pathologic features, such as large or multiple positive lymph nodes and extranodal extension (ENE). The primary endpoint was RFS, with OS as a key secondary endpoint.20
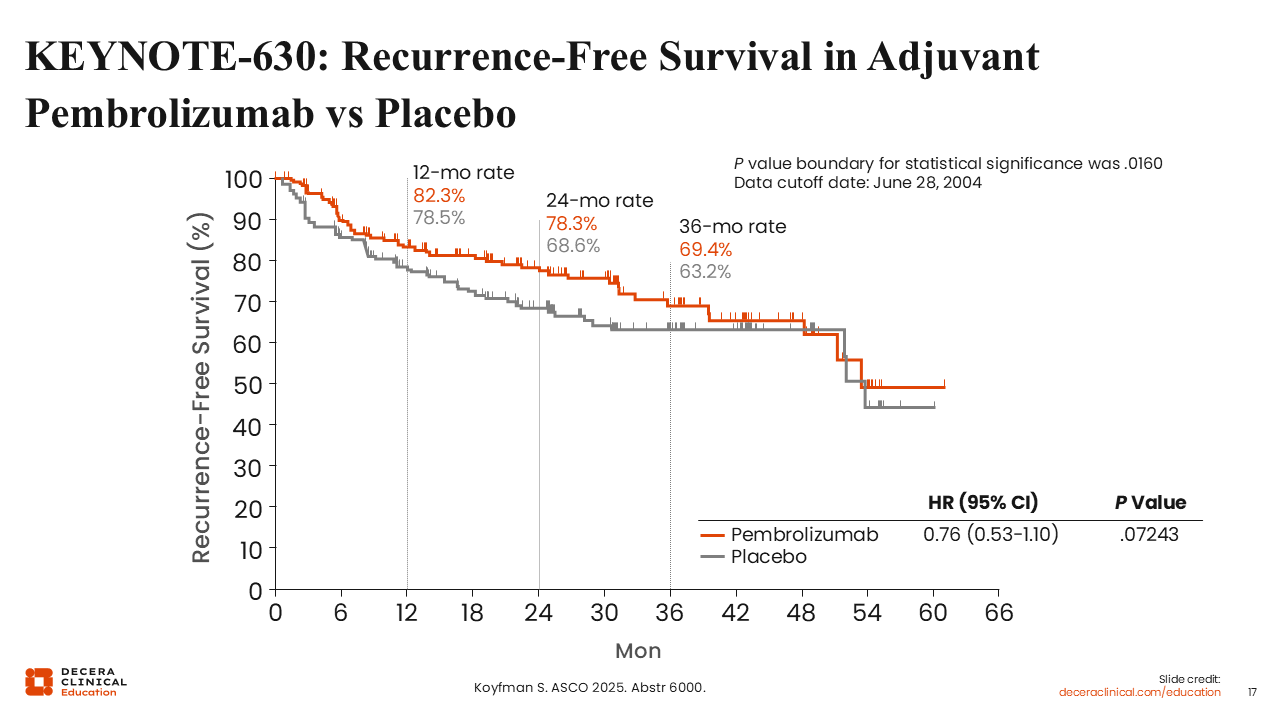
KEYNOTE-630: Recurrence-Free Survival in Adjuvant Pembrolizumab vs Placebo
An independent data monitoring committee recommended stopping the study for futility at a preplanned interim analysis. Although pembrolizumab showed a 24% reduction in the risk of recurrence with a hazard ratio of 0.76 (95% CI: 0.53-1.10), it failed to reach the prespecified boundary for statistical significance (P = .072). Of note, the 24-month OS rate was numerically lower in the pembrolizumab arm (87.3%) compared to placebo (90.7%), partly due to an unexplained higher incidence of non–treatment-related deaths in the immunotherapy group. These findings, which contrast with the positive results of the C-POST trial (adjuvant cemiplimab), reinforce that a PD-1 “class effect” cannot be assumed and that adjuvant benefit requires agent-specific, phase III evidence before adoption into routine practice. The overarching conclusion is that adjuvant pembrolizumab should not be considered standard of care following full-course RT in this specific setting.21,22
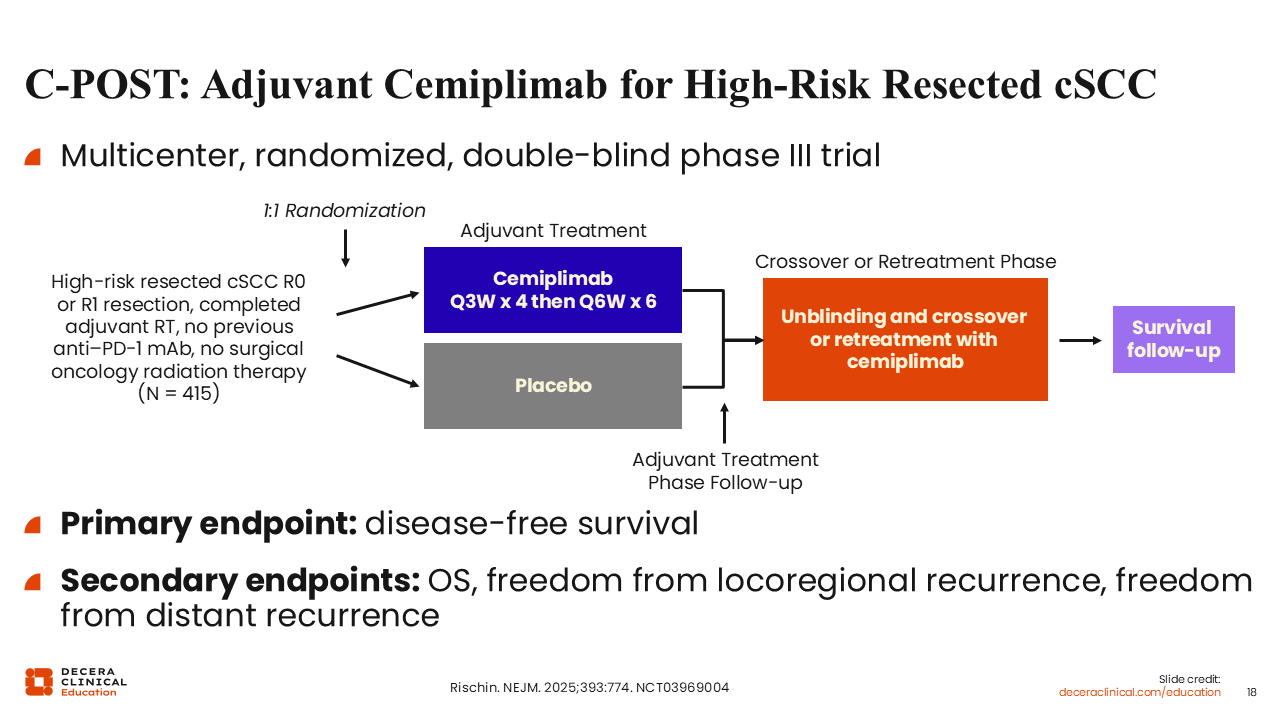
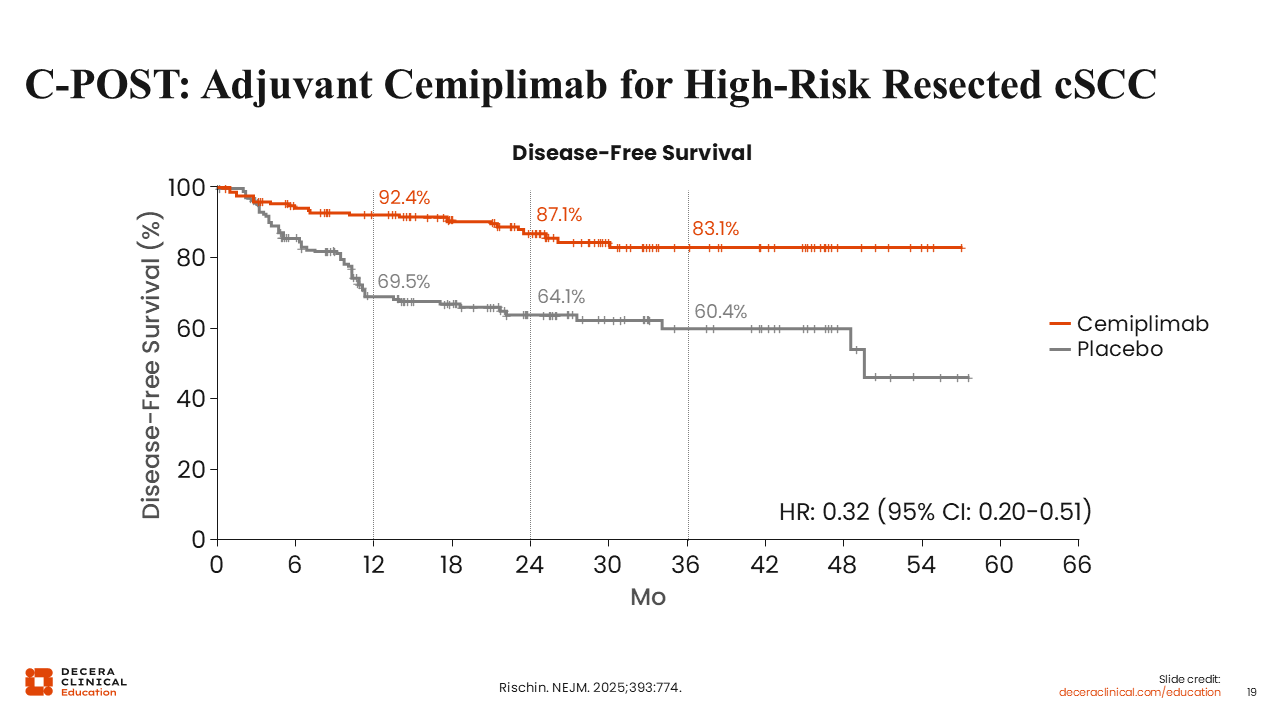
C-POST: Adjuvant Cemiplimab for High-Risk Resected cSCC
The phase III C-POST trial evaluated adjuvant cemiplimab vs placebo in patients with resected high-risk cSCC who had completed postoperative RT. Eligible patients had either high-risk nodal disease, including multiple positive lymph nodes or ENE or high-risk non-nodal features, such as deeply invasive primary tumors, perineural invasion, or high-risk histologic features. Treatment consisted of cemiplimab administered every 3 weeks during the induction phase followed by every 6 weeks during maintenance for a total planned duration of approximately 1 year, compared with matched placebo. The primary endpoint was disease-free survival (DFS), and secondary endpoints included OS and patterns of locoregional and distant recurrence.6
C-POST: Adjuvant Cemiplimab for High-Risk Resected cSCC
C-POST met its primary endpoint, demonstrating a substantial improvement in DFS with adjuvant cemiplimab in patients with high-risk cSCC. At a median follow-up of 24 months, cemiplimab-treated patients exhibited a markedly lower risk of recurrence or death compared to those receiving placebo, with a hazard ratio of 0.32 (95% CI: 0.20-0.51), corresponding to an approximately 68% risk reduction.6 The estimated 24-month DFS rate was 87.1% for the cemiplimab group vs 64.1% for the placebo group, representing a clinically significant difference of 23 percentage points. Furthermore, the therapeutic benefit remained consistent across all prespecified high-risk subgroups—including both nodal and non-nodal features—and did not appear to depend on PD-L1 expression. These data support the use of adjuvant cemiplimab as a potential new standard of care in appropriately selected patients regardless of PD-L1 status.6,23,24
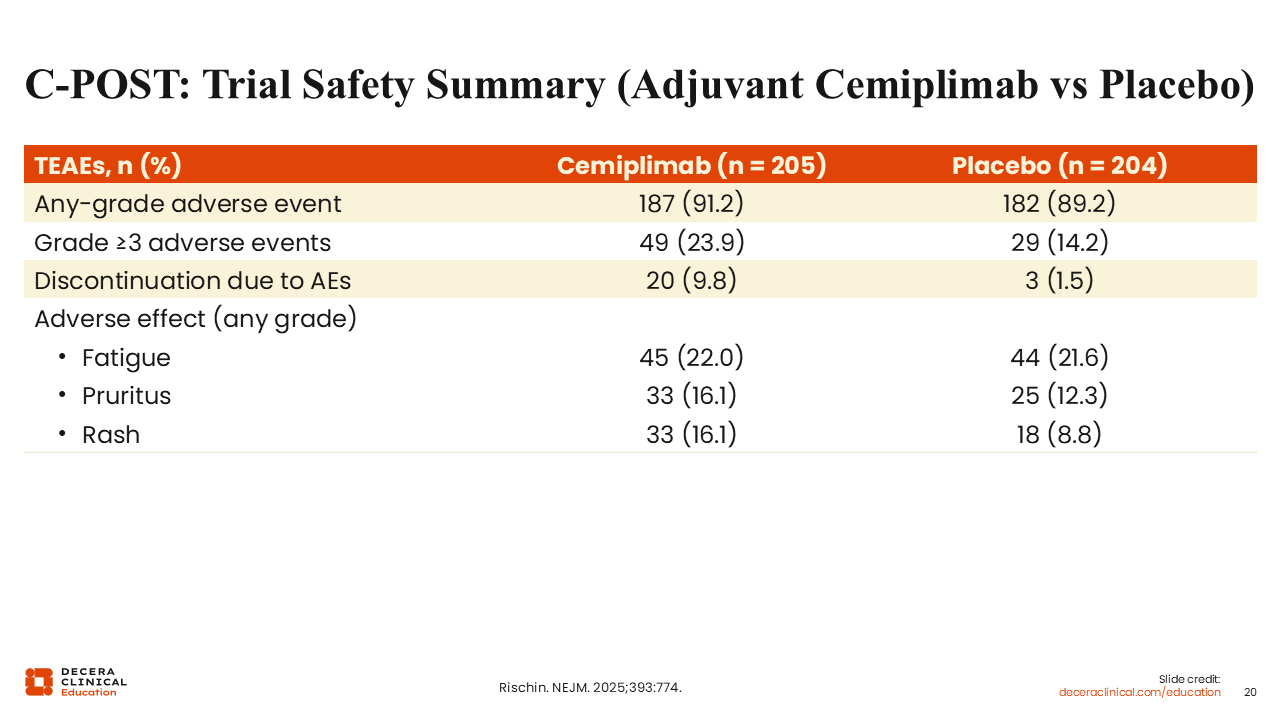
C-POST: Trial Safety Summary (Adjuvant Cemiplimab vs Placebo)
The overall treatment-emergent adverse event (TEAE) burden in C-POST was similar between arms: 91.2% with cemiplimab vs 89.2% with placebo, consistent with an older, comorbidity-heavy population. However, higher-grade toxicity was more frequent with cemiplimab, with grade 3-5 TEAEs in 23.9% vs 14.2%. Treatment discontinuation due to TEAEs occurred more often with cemiplimab (9.8%) vs placebo (1.5%), aligning with the expected tolerability tradeoff of adjuvant PD-1 blockade.6
The pattern of differences was driven largely by immune-mediated/class-consistent events, particularly rash (16.1% vs 8.8%). Fatigue was the most frequently reported event in both arms, occurring in 22.0% of patients receiving cemiplimab and 21.6% of patients receiving placebo. Low-grade pruritus was also prevalent in both groups, occurring in 16.1% of patients receiving cemiplimab vs 12.3% receiving placebo, and rash occurred in 16.2% and 8.8%, respectively.6
Eligibility Criteria in C-POST vs KEYNOTE-630
The safety profile of adjuvant cemiplimab in C-POST was consistent with prior clinical experience in patients with advanced cSCC. Although the majority of patients experienced at least 1 TEAE, grade ≥3 events occurred in approximately one quarter of patients and were generally manageable with standard supportive measures.6 irAEs such as thyroid dysfunction, hepatitis, colitis, and pneumonitis occurred at rates typical for PD-1 blockade and were managed using established toxicity algorithms.6,25 Approximately 10% of patients discontinued cemiplimab because of adverse events (AEs), and treatment-related deaths were rare.6 These findings support the feasibility of delivering approximately 1 year of adjuvant cemiplimab after major surgery and RT in appropriately selected patients, provided that proactive monitoring and guideline-based management of irAEs are in place.25,26
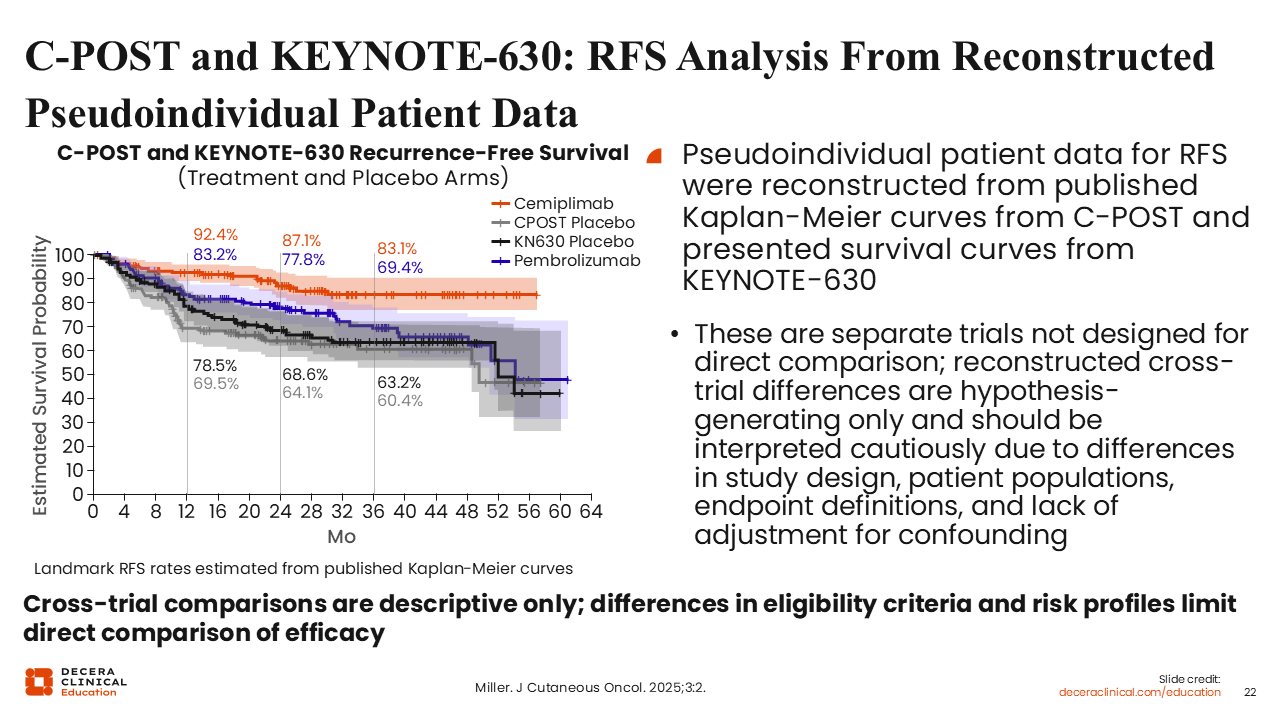
C-POST and KEYNOTE-630: RFS Analysis From Reconstructed Pseudoindividual Patient Data
The KEYNOTE-630 and C-POST trials exemplify that a PD-1 “class effect” cannot be assumed in the adjuvant setting for cSCC. Although both phase III trials evaluated adjuvant PD-1 blockade following surgery and RT in high-risk patients, they differed in eligibility criteria, risk enrichment, and statistical design, which likely contributed to their divergent outcomes.24,27 As noted on the previous eligibility slide, differences in nodal involvement and ENE requirements remain key variables.
The C-POST trial demonstrated a robust, statistically significant DFS benefit with cemiplimab, yielding a hazard ratio of 0.32 (approximately 68% risk reduction), whereas KEYNOTE-630 failed to meet its primary endpoint, as pembrolizumab did not significantly improve RFS compared to placebo.6,22 Comparative analyses using reconstructed Kaplan–Meier data showed that although the placebo arms behaved similarly across both trials, the cemiplimab curve separated distinctly while the pembrolizumab curve tracked closely to placebo.27 This suggests that the C-POST results reflect a true treatment effect rather than a weak control arm. Based on these data and current NCCN guidelines, adjuvant cemiplimab is the evidence-based standard for resected high-risk cSCC, whereas pembrolizumab is not recommended for adjuvant use outside of a clinical trial.2,6
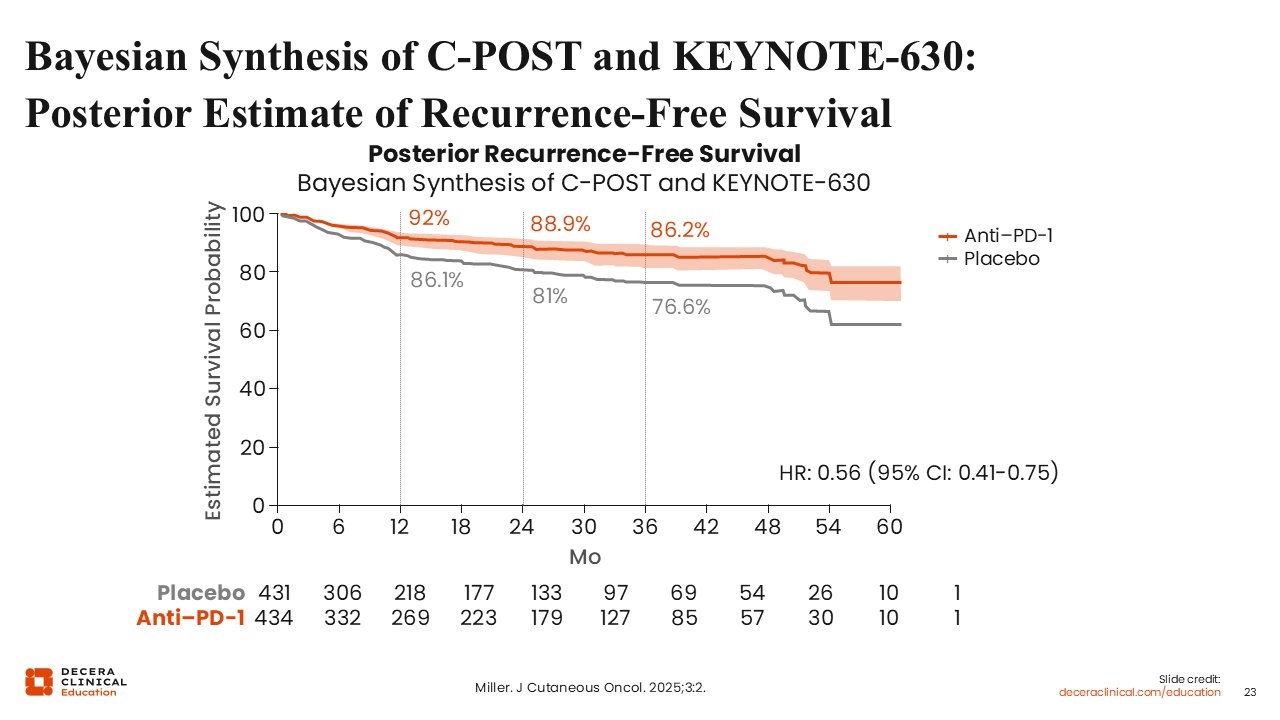
Bayesian Synthesis of C-POST and KEYNOTE-630: Posterior Estimate of Recurrence-Free Survival
From a Bayesian perspective, the totality of data suggests that adjuvant PD-1 blockade improves outcomes in appropriately selected patients with high-risk cSCC, though the strength of evidence is not uniform across agents. The C-POST trial demonstrated a robust 68% relative reduction in recurrence or death with cemiplimab (HR: 0.32; 95% CI: 0.20-0.51), whereas KEYNOTE-630 was stopped early for futility, as pembrolizumab failed to show a significant DFS benefit.6,24 A Bayesian synthesis incorporating prior evidence from metastatic cSCC supports a high posterior probability that the cemiplimab results reflect a true treatment effect, rather than a class-wide certainty for all PD-1 inhibitors.27
This raises the critical question of therapeutic sequencing: preoperative (neoadjuvant) vs postoperative (adjuvant) immunotherapy. Although adjuvant cemiplimab is now the evidence-based standard of care following surgery and RT for very high–risk resected patients, the neoadjuvant approach is gaining momentum.6 Drawing from successes in melanoma trials like SWOG S1801 and NADINA, starting PD-1 therapy in the neoadjuvant setting can enhance EFS and allow for response-adapted therapy.28,29 In cSCC, neoadjuvant cemiplimab has yielded high pCR rates in phase II studies, though this remains investigational.14 Currently, adjuvant cemiplimab remains the standard of care, and neoadjuvant strategies should be prioritized within multidisciplinary centers or clinical trials for locally advanced disease.2
Adjuvant Cemiplimab in NCCN Guidelines for Very High–Risk cSCC
Reflecting the positive findings from the C-POST trial, the FDA approved cemiplimab in October 2025 for the adjuvant treatment of adult patients with cSCC at high risk of recurrence after surgery and RT. In alignment with this landmark approval, the NCCN guidelines now list adjuvant cemiplimab as a preferred option for patients with extremely high–risk nodal or non-nodal features after surgery and RT.2 For these patients, healthcare professionals (HCPs) should discuss the potential benefits of adjuvant cemiplimab in reducing recurrence risk, while also addressing the commitment required for regular clinic visits, laboratory monitoring, and the possibility of irAEs that may necessitate immunosuppression or treatment interruption.23 Integration of adjuvant cemiplimab should be highly individualized, with careful attention paid to the patient's performance status, comorbidities, personal preferences, and prior treatment tolerance.2
Preoperative vs Postoperative Immunotherapy
A natural question emerging from the evolving data is whether preoperative (neoadjuvant) or postoperative (adjuvant) immunotherapy is the superior strategy. For now, there is no single correct answer, but the roles for each are becoming clearly defined. The C-POST trial demonstrated that adjuvant cemiplimab significantly improves DFS in high-risk resected cSCC following surgery and RT.6
In contrast, landmark melanoma trials such as SWOG S1801 and NADINA illustrate that initiating immunotherapy in the neoadjuvant setting can enhance EFS and allow for response-adapted adjuvant therapy.28,29 Although neoadjuvant cemiplimab has produced high pCR rates and encouraging EFS in single-arm cSCC studies, these approaches remain investigational.14 Ultimately, both perioperative strategies have a role: adjuvant cemiplimab is the established standard for selected very high–risk resected patients, whereas neoadjuvant PD-1 therapy is best reserved for multidisciplinary centers or clinical trials involving locally advanced, borderline-resectable disease.2,6
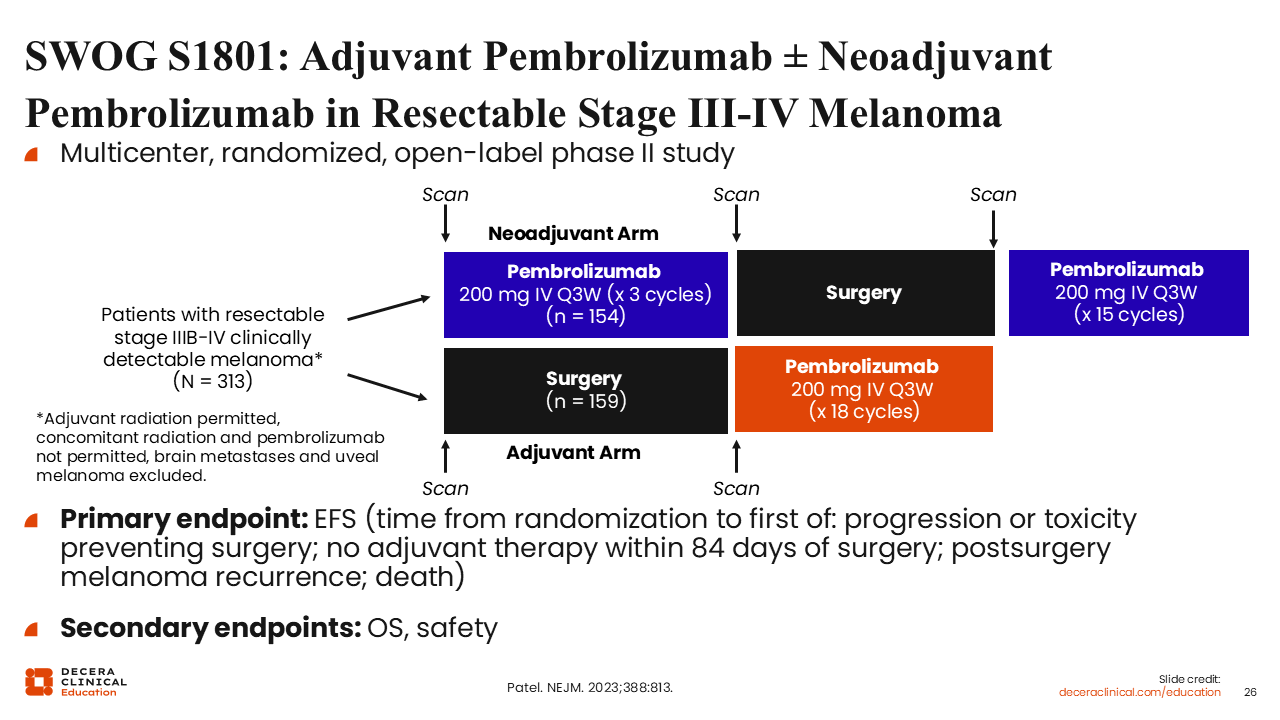
SWOG S1801: Adjuvant Pembrolizumab ± Neoadjuvant Pembrolizumab in Resectable Stage III-IV Melanoma
Experience in melanoma helps frame why neoadjuvant immunotherapy is attractive for high-risk cSCC, even though the diseases are biologically distinct. In the SWOG S1801 trial, patients with resectable stage IIIB-IV melanoma were randomized to either 3 cycles of neoadjuvant pembrolizumab followed by surgery and 15 additional adjuvant cycles, or upfront surgery followed by adjuvant pembrolizumab alone. Of importance, both arms were planned to receive the same total number of doses of pembrolizumab; the only variable was the timing of treatment initiation.28
This design isolates the effect of starting PD-1 blockade while the bulk tumor is still present and antigen rich. This allows for the in-situ priming of tumor-reactive T-cells before the primary source of neoantigens is surgically removed.28,30 The S1801 schema underpins the rationale now motivating neoadjuvant cemiplimab trials in resectable cSCC: engaging the immune system early to potentially improve cure rates through a coordinated perioperative strategy rather than relying on adjuvant therapy alone.6,14
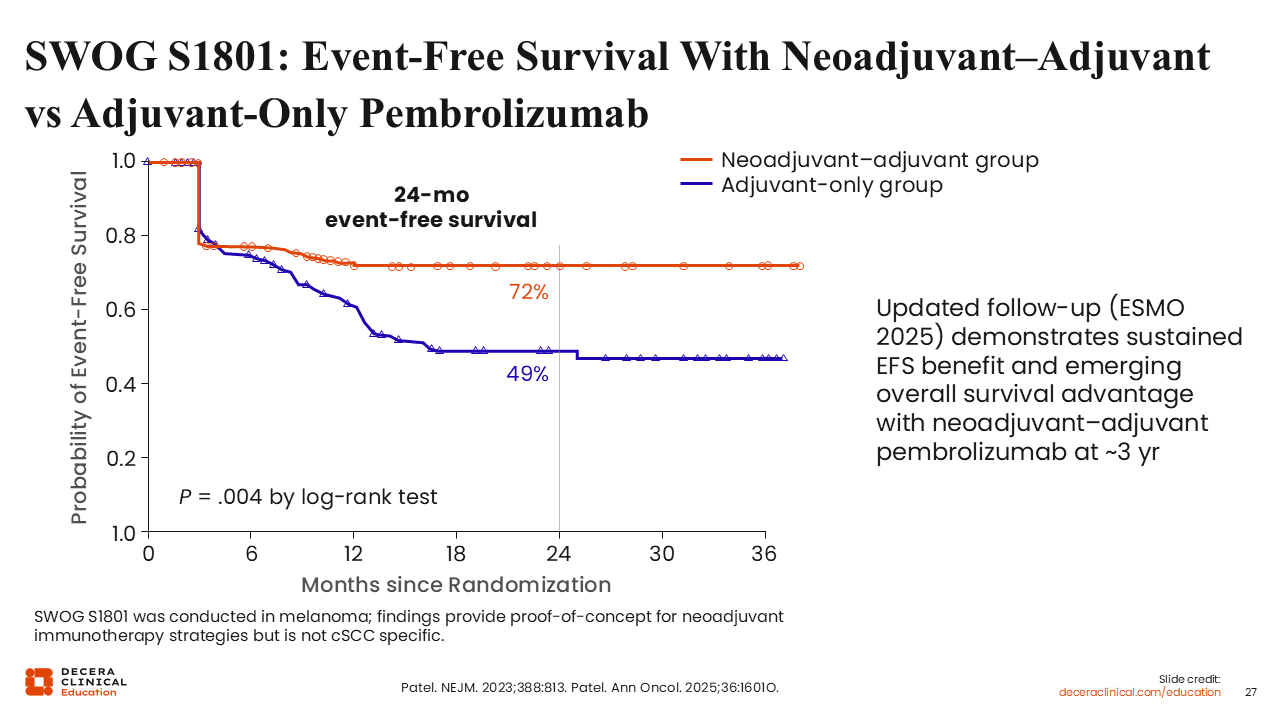
SWOG S1801: Event-Free Survival With Neoadjuvant–Adjuvant vs Adjuvant-Only Pembrolizumab
The updated data from SWOG S1801 presented at ESMO 2025 demonstrate that the clinical advantage of the neoadjuvant–adjuvant pembrolizumab sequence is durable over the long term. With a median follow-up of 3 years, the neoadjuvant–adjuvant arm continued to show a statistically significant improvement in EFS compared to the adjuvant-only group.31
The 3-year EFS rate was 68% in the neoadjuvant–adjuvant group vs 43% in the adjuvant-only group, maintaining a robust hazard ratio of 0.59 (95% CI: 0.47-0.94; P = .0003). These results further validate the "timing matters" hypothesis, as both arms received the same total number of doses. Of importance, no new safety signals emerged in the long-term follow-up, and grade ≥3 treatment-related AEs remained consistent with earlier readouts at approximately 12%.31 This 25 percentage point difference at 3 years reinforces the rationale for evaluating similar neoadjuvant strategies in cSCC, suggesting that early immune priming before surgery may be superior to the current adjuvant-only standard.6,30
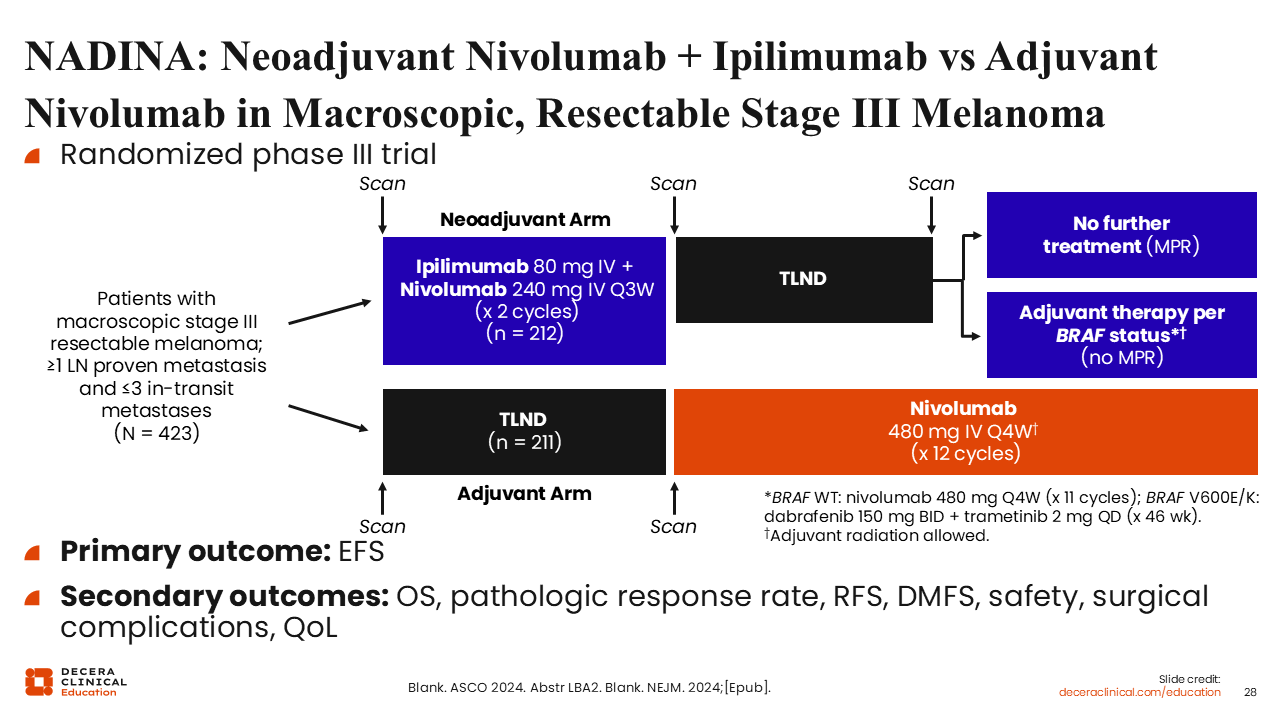
NADINA: Neoadjuvant Nivolumab + Ipilimumab vs Adjuvant Nivolumab in Macroscopic, Resectable Stage III Melanoma
The phase III NADINA trial in macroscopic stage III melanoma represents a significant advancement in the neoadjuvant paradigm through a response-adapted treatment strategy. Patients were randomized to receive either 2 cycles of neoadjuvant ipilimumab plus nivolumab followed by surgery and selective adjuvant therapy only when pathologic response was suboptimal or upfront surgery followed by 12 cycles of adjuvant nivolumab.29
In the neoadjuvant arm, patients who achieved a pCR or MPR frequently required no further systemic therapy after surgery. Patients with residual tumor, however, received adjuvant nivolumab or, when a BRAF V600 mutation was present, adjuvant BRAF/MEK inhibitor therapy—a targeted combination approach that suppresses MAPK pathway signaling by pairing a BRAF inhibitor (such as dabrafenib, vemurafenib, or encorafenib) with a MEK inhibitor (such as trametinib, cobimetinib, or binimetinib).29
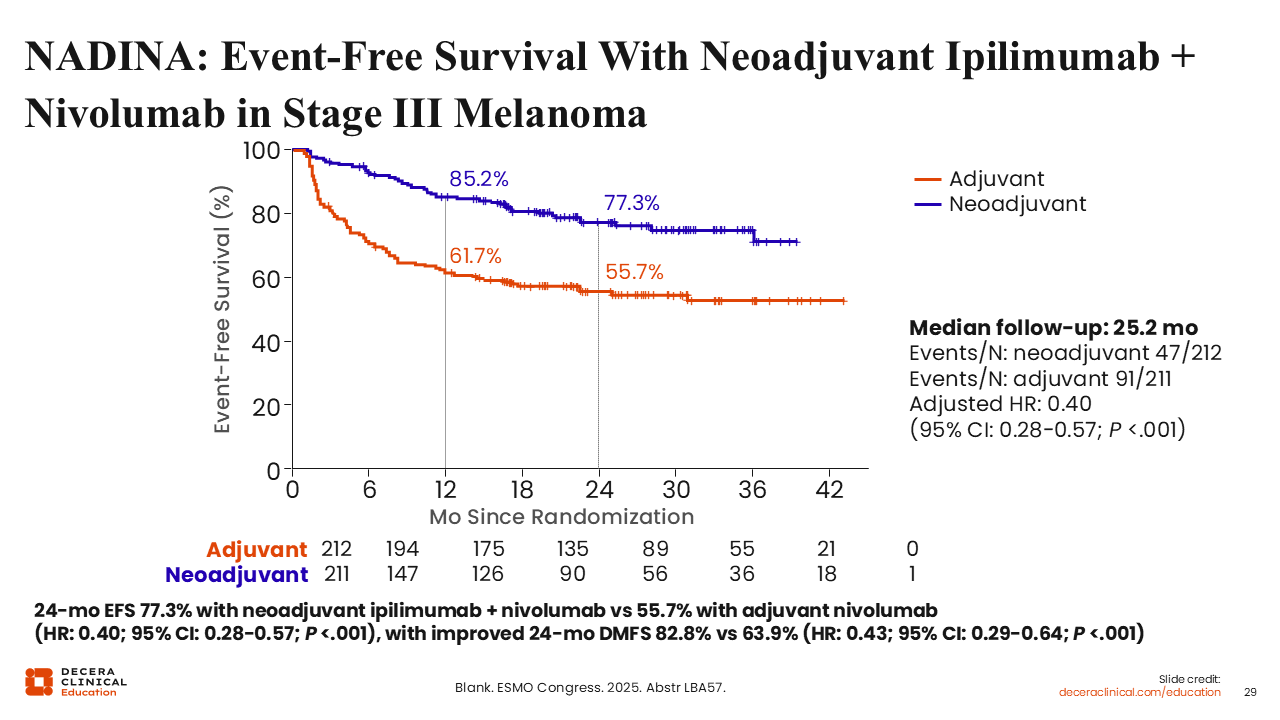
NADINA: Event-Free Survival with Neoadjuvant Ipilimumab + Nivolumab in Stage III Melanoma
The phase III NADINA trial results demonstrate a striking and durable clinical benefit for neoadjuvant ipilimumab plus nivolumab. At a median follow-up, the estimated 24-month EFS rate was 77.3% in the neoadjuvant group vs 55.7% in the adjuvant nivolumab group, representing a 22-percentage-point advantage. This corresponds to a hazard ratio of 0.40 (95% CI: 0.28-0.57; P <.001), reflecting a 60% reduction in the risk of progression, recurrence, or death.29,32
For cSCC, the key transferable insight is that short-course, high-intensity neoadjuvant immunotherapy—in this case, ipilimumab plus nivolumab—can meaningfully improve EFS in node-positive skin cancer when followed by response-adapted adjuvant therapy. These results support ongoing studies aiming to evaluate whether similar neoadjuvant paradigms can benefit patients with resectable, high-risk cSCC.32
Biomarkers in Immune Checkpoint Inhibitor Decision-making
In many solid tumors, PD-L1 expression is used as a predictive biomarker for PD-1/PD-L1 blockade. In cSCC, however, the story is more nuanced. Although PD-L1 expression is common and may correlate with aggressive features, analyses from advanced-disease trials and from C-POST show that patients with both PD-L1–high and PD-L1–low or –negative tumors can derive significant benefit from PD-1 blockade.4-6 No validated PD-L1 cutoff exists to guide adjuvant cemiplimab selection in cSCC. As a result, PD-L1 status should not be used to deny patients adjuvant cemiplimab or advanced-stage PD-1 therapy when otherwise clinically appropriate.2 In current practice, clinical risk features—including tumor diameter, depth of invasion, large-nerve perineural invasion, and nodal involvement—remain the primary determinants of decision-making for adjuvant intervention.2,6
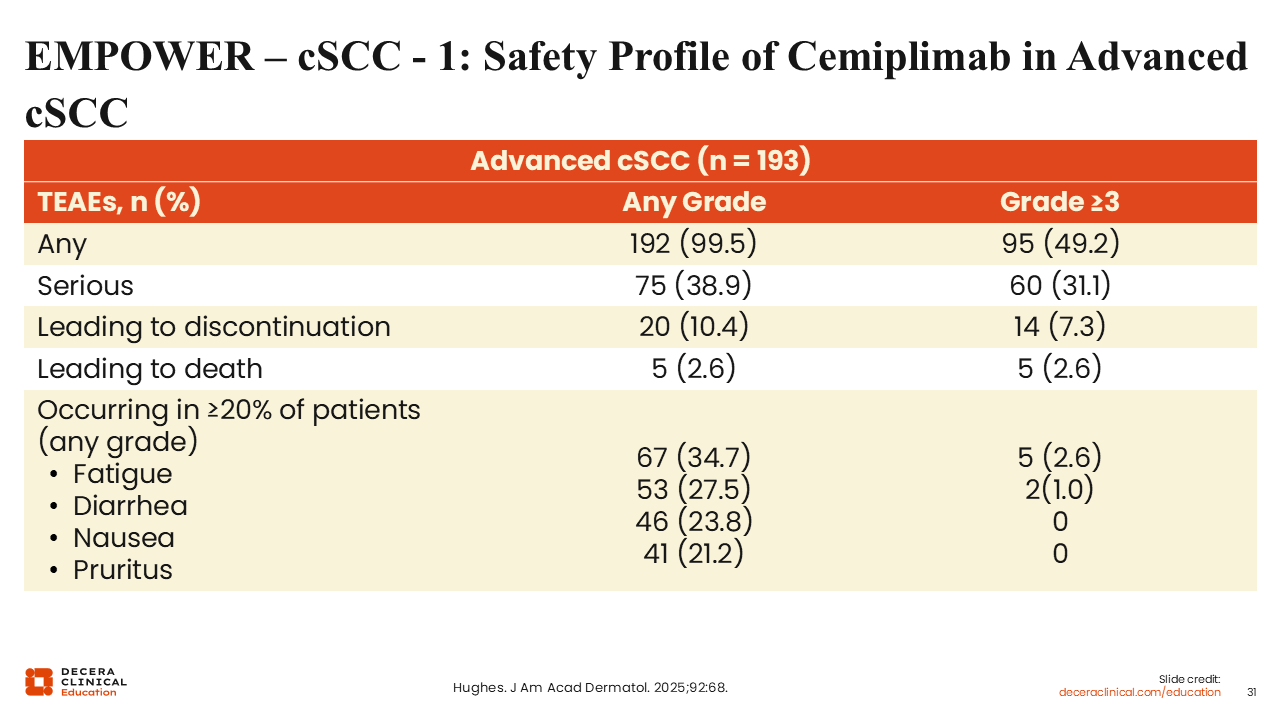
EMPOWER – cSCC - 1: Safety Profile of Cemiplimab in Advanced cSCC
Although neoadjuvant and adjuvant strategies represent the new frontier in resectable disease, our understanding of the long-term safety and durability of PD-1 inhibition is rooted in the pivotal trials for advanced cSCC. Shifting our focus to the final analysis of the EMPOWER-cSCC-1 study, we see that cemiplimab maintains a well-characterized safety profile over multi-year follow-up in patients with metastatic or locally advanced disease. In this foundational setting, cemiplimab continued to demonstrate durable antitumor responses in roughly half of the patient population, with toxicities that remained consistent with the established PD-1 inhibitor class. Most TEAEs were generally low-grade—including fatigue, diarrhea, nausea, and rash—with only a small minority of patients necessitating treatment discontinuation.33
Serious TEAEs occurred in a subset of patients, though relatively few were deemed treatment related. Immune-mediated toxicities such as pneumonitis, thyroid dysfunction, and hepatitis fell within the expected range for PD-1 blockade. Long-term follow-up and pooled safety analyses reinforce that grade ≥3 irAEs with cemiplimab remain in the low- to mid-teens percentage range with no new safety signals emerging over multi-year monitoring.33 These findings indicate that the counseling, monitoring, and management strategies standard for other PD-1 antibodies can be applied directly to cemiplimab in the advanced and adjuvant cSCC settings.
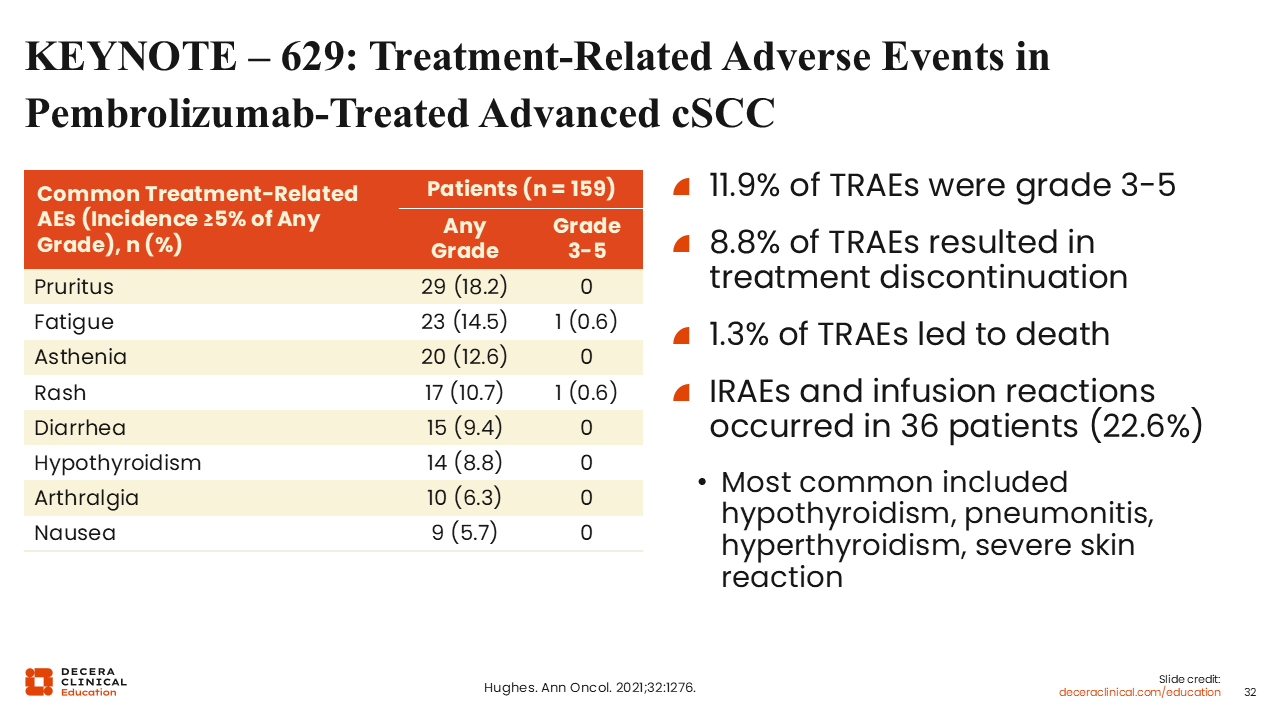
KEYNOTE – 629: Treatment-Related Adverse Events in Pembrolizumab-Treated Advanced cSCC
Safety findings from the KEYNOTE-629 trial support the use of PD-1 blockade in advanced cSCC when accompanied by careful monitoring. Across the locally advanced and recurrent/metastatic cohorts, 69% of patients experienced a treatment-related AE with pembrolizumab, the majority of which were low grade and manageable.5,10 Grade 3-5 treatment-related AEs occurred in approximately 12% of patients, with a toxicity spectrum characteristic of PD-1 inhibition—most notably fatigue, pruritus, diarrhea, and endocrinopathies.5
Health-related QoL assessments demonstrated that overall functioning and global health scores generally remained stable or improved during treatment.34 This suggests that pembrolizumab does not substantially impair daily functioning for appropriately selected patients, even in an older, comorbid population. Within cSCC treatment pathways, KEYNOTE-629 reinforces that anti–PD-1 therapy is feasible when care teams proactively educate patients, employ standardized toxicity checklists, and intervene early for emerging immune-mediated events.5,10
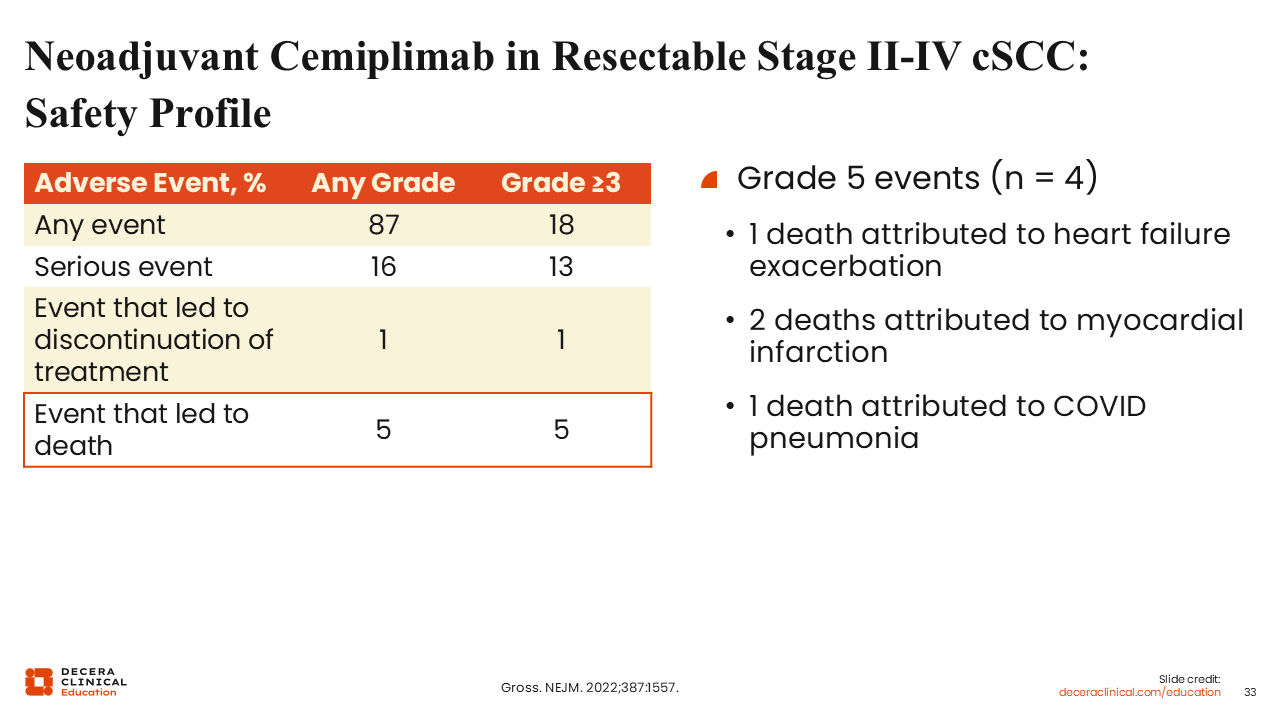
Neoadjuvant Cemiplimab in Resectable Stage II-IV cSCC: Safety Profile
In the phase II neoadjuvant cemiplimab trial for resectable stage II-IV (M0) cSCC, AEs were frequent but largely manageable and did not generally preclude surgery. Any-grade AEs occurred in 87% of patients, and grade ≥3 AEs in 18%.14 Immune-mediated toxicities, such as hypothyroidism, hepatitis, pneumonitis, and colitis, were consistent with the broader PD-1 class and were typically reversible with standard immunosuppression.14,27 Of importance, there were no treatment-related deaths and no grade 4 treatment-related AEs reported in this study.14,35 Only a small minority of patients experienced delays to surgery due to toxicity, underscoring that a short neoadjuvant course of cemiplimab can be delivered safely in experienced multidisciplinary centers with appropriate pretreatment education and rapid access pathways for AE evaluation.
Mechanistic Link Between Immune Checkpoint Inhibition and Atherosclerotic Cardiovascular Risk
ICIs have clear antitumor benefits in cSCC, but they also remove critical “brakes” on vascular inflammation. A growing cardio-oncology literature suggests that PD-1 and CTLA-4 signaling normally restrain atherosclerotic plaque formation and destabilization. Blocking these checkpoints can accelerate plaque progression and increase the risk of myocardial infarction and other atherosclerotic events compared with non–ICI-treated populations. In practice, this means that older patients with preexisting cardiovascular risk factors, hypertension, diabetes, or prior coronary disease may be particularly vulnerable. Before initiating adjuvant or neoadjuvant PD-1 therapy, HCPs should optimize traditional cardiovascular risk factors, review baseline symptoms, and consider involving cardio-oncology colleagues for patients with substantial vascular disease. During treatment, a low threshold for evaluating new chest pain, dyspnea, or exercise intolerance is essential. These steps do not negate the benefits of PD-1 therapy in high-risk cSCC, but they help us deliver immunotherapy in a way that is safer and more sustainable for patients who are already at elevated cardiovascular risk.36
Real-world Toxicity of Immunotherapy: Beyond Grades 3/4― Hidden Burden of “Moderate” AEs
ICIs have transformed the management of advanced cSCC, yet they are not without meaningful toxicity.5,37 Real-world data of cemiplimab and pembrolizumab consistently show that most patients experience treatment-related AEs, with the majority being grade 1-2 but still clinically significant.37,38 These moderate toxicities including grade 2 diarrhea, dermatitis, thyroid dysfunction, and arthralgias are frequently underrecognized in routine practice yet often necessitate corticosteroids, temporary treatment interruptions, and additional laboratory monitoring.11,38 This pattern contributes to substantial “time toxicity,” characterized by repeated appointments and care coordination layered on top of complex local therapies.6 Even when technically low-grade, persistent symptoms can diminish QoL and functional independence, particularly in the elderly population where cSCC most commonly arises.11,38
From the patient’s perspective, chronic low- to moderate-grade irAEs can be as impactful as occasional high-grade toxicities. In the KEYNOTE-629 trial, approximately 70% of patients receiving pembrolizumab experienced treatment-related AEs, with 11% to 12% developing grade 3-5 events. However, even persistent grade 1-2 symptoms such as fatigue, pruritus, or diarrhea were sufficient to disrupt daily functioning in older or frail individuals.38 Real-world studies of cemiplimab similarly report that a notable proportion of patients discontinue or interrupt therapy due to cumulative toxicity, despite comparatively low rates of severe (grade ≥3) events.15,37
In the C-POST trial, adjuvant cemiplimab significantly improved DFS but also introduced a full year of systemic therapy for patients who were otherwise clinically disease free following surgery and radiation.6 Observational analyses confirm that most high-risk cSCC recurrences occur within the first several years, a period that now overlaps significantly with the burden of ongoing immunotherapy.11 When counseling patients about adjuvant or neoadjuvant PD-1 therapy, it is therefore essential to balance the quantified efficacy benefit against the real-world time and symptom burden, using these cSCC-specific data to guide a truly shared decision-making process.6
Radiation as an Immune Primer
RT functions not only as a local cytotoxic modality but also as a potential immune sensitizer that may augment the activity of PD-1 blockade. Preclinical and translational studies suggest that moderately hypofractionated regimens, such as 8 Gy × 3 fractions, can induce immunogenic cell death, activate type I interferon signaling, and enhance tumor antigen presentation, thereby increasing tumor susceptibility to immune-mediated clearance.39 In several experimental models, combining hypofractionated RT with PD-1 inhibition has been associated with increased CD8⁺ T-cell infiltration and, in some cases, abscopal responses, where nonirradiated lesions also regress.39
For cSCC, these observations offer a biologic rationale for investigating RT–immunotherapy combinations across adjuvant, definitive, and potentially neoadjuvant settings. However, data specific to cSCC remain limited, and much of the mechanistic understanding is extrapolated from broader solid-tumor research. Prospective studies are underway to define the optimal dose, fractionation, and sequencing of RT relative to PD-1 blockade.39 Until such evidence is available, deliberate attempts to use RT for immune “priming” should be approached within the context of a clinical trial or with multidisciplinary evaluation to balance potential synergy with the risks of added toxicity and increased treatment complexity.
CLEAR-CSCC: Intralesional Cemiplimab vs Primary Surgery in Early-Stage cSCC
Intralesional approaches provide a potential means of leveraging immunotherapy for cSCC while limiting systemic exposure. Early phase studies of intralesional cemiplimab in nonmelanoma skin cancers, including cSCC, have demonstrated encouraging local activity with high pCR rates and no unexpected safety findings beyond what is typically observed with systemic PD-1 inhibition.33 Because the drug is administered directly into the tumor, systemic drug levels may be lower, a feature that may be particularly relevant for frail patients, those with autoimmune conditions, or individuals who have difficulties tolerating additional systemic therapy.
Building on these preliminary results, the phase III CLEAR-CSCC trial is evaluating low-dose intralesional cemiplimab compared with Mohs micrographic surgery for early-stage cSCC, including scenarios where surgery may be technically complex or poorly tolerated (for example, lower extremity lesions in very elderly patients).33 Should CLEAR-CSCC confirm high local control with an acceptable safety profile, intralesional PD-1 therapy could represent a valuable option for individualizing treatment intensity and potentially reducing the need for disfiguring or function-limiting procedures in carefully selected patients.
Tailoring Therapy for Older and Frail Patients With cSCC
Most individuals with advanced cSCC are older adults, including a substantial proportion 80 years of age or older with multiple comorbidities. Clinical trial and real-world data with cemiplimab and pembrolizumab indicate that treatment efficacy is generally preserved in older populations, with response rates and durability comparable to outcomes observed in younger patients.5,37,38 However, even moderate irAEs—such as fatigue, thyroid dysfunction, dermatitis, or diarrhea—may disproportionately affect independence, mobility, and overall QoL in this age group.
Health-related QoL analyses from KEYNOTE-629 demonstrate that many patients maintain or improve global functioning during PD-1 therapy, although a subset experienced declines that mirror treatment-emergent toxicity.38 For patients who are very frail or highly comorbid, alternative strategies such as shorter neoadjuvant PD-1 courses, intralesional or localized treatments, or a focus on best supportive care may provide a more appropriate balance between tumor control and the risks of prolonged low-grade toxicity, increased time in care, and potential functional deterioration.15,37
Future Directions and Outstanding Questions
We have made significant advances in the management of cSCC. However, several key questions remain. In the neoadjuvant setting, randomized trials are needed to determine whether neoadjuvant PD-1 blockade offers advantages over adjuvant therapy alone and to define the optimal treatment duration, number of doses, and pathologic response thresholds that could guide postoperative decision-making.14,16 In the adjuvant setting, extended follow-up from the C-POST trial will be important to clarify any OS benefit and to identify patient subgroups who derive the greatest reduction in recurrence risk.6
Further research is also necessary to refine the integration of RT and ICIs, to validate predictive biomarkers beyond PD-L1 expression, and to clarify the role of intralesional therapies and emerging systemic combinations within the evolving treatment landscape.24
Key Takeaways for Clinical Practice
The treatment landscape for cSCC has evolved substantially with the introduction of PD-1 blockade. For patients with unresectable or metastatic cSCC, PD-1 inhibitors represent the current standard systemic therapy based on the pivotal EMPOWER-CSCC-1 trial for cemiplimab as well as confirmatory evidence from the KEYNOTE-629 trial for pembrolizumab.4,5 In the postoperative setting, the treatment paradigm has recently expanded. Adjuvant cemiplimab is now an FDA-approved and evidence-based option to reduce the risk of recurrence following definitive surgery and RT in patients with resected high-risk disease. This approval is supported by the phase III C-POST trial, which demonstrated a significant improvement in DFS, and has been formally incorporated into the NCCN guidelines 2,6
Interest in neoadjuvant PD-1 therapy continues to grow for patients with locally advanced, resectable cSCC, with phase II data demonstrating substantial pathologic responses. However, this approach remains investigational pending further validation.14 Across all stages of disease, optimal care depends on multidisciplinary coordination, vigilant immune-related toxicity monitoring, and the integration of geriatric assessment principles to support treatment decision-making in an older population.8,25 Through thoughtful application of these strategies, HCPs can incorporate emerging evidence into practice and improve outcomes for patients with high-risk and advanced cSCC.