CME
HER2: Biology and Testing in Gastrointestinal, Genitourinary, and Gynecologic Malignancies
Physicians: Maximum of 0.25 AMA PRA Category 1 Credit™
Released: August 04, 2025
Expiration: February 03, 2026
Activity
Introduction
In this activity, Zev A. Wainberg, MD; Alexandra Leary, MD, PhD; and Catherine Fahey, MD, PhD, explore testing for HER2 alterations and the incidence of HER2-positive disease in the treatment of GU, GI, and GYN malignancies. This session aims to bridge foundational information on HER2 biology with practical clinical strategies across solid tumors as HER2-targeted antibody–drug conjugates (ADCs) emerge as potential treatment options in these settings. Experts will review biomarker interpretation, HER2 testing modalities, and how HER2 expression can inform treatment decisions in different cancer settings.
Please note that the slide thumbnails in this activity link to a PowerPoint slideset that also can be found here. The slideset also may be downloaded by clicking on any of the thumbnails within the activity.
Before continuing with this educational activity, please take a moment to answer the following question.
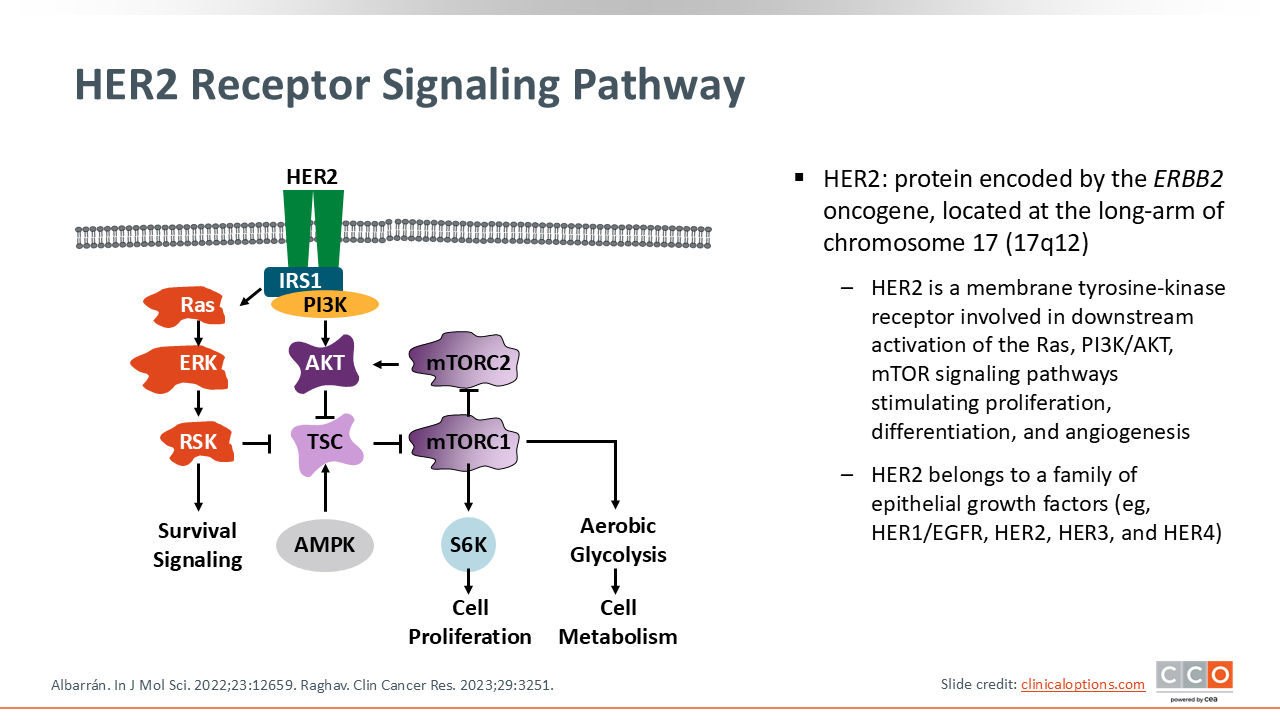
HER2 Receptor Signaling Pathway
HER2, or human epidermal growth factor receptor 2, is a transmembrane tyrosine kinase receptor involved in cell growth and survival.1,2 It is encoded by the ERBB2 gene and was initially characterized in breast cancer over 3 decades ago. HER2/ERBB2 gene amplification and HER2 overexpression was historically considered a poor prognostic biomarker.3 However, the development of HER2-targeted therapies, such as trastuzumab, dramatically changed the treatment landscape by demonstrating improved outcomes in patients with breast cancer and HER2 amplification.4 Similarly, HER2 mutations occur in approximately 1% to 3% of non-small-cell lung cancer cases, predominantly as exon 20 insertions, and were historically difficult to target until the FDA approval of the HER2-targeted ADC, trastuzumab deruxtecan (T-DXd), for patients with HER2 mutations after previous therapy.
HER2-targeted strategies have also expanded into gastric and gastroesophageal cancers, where HER2 overexpression is also clinically relevant.5 More recently, HER2 has been implicated in other tumor types such as colorectal, bladder, biliary tract, and endometrial cancers.2 HER2-targeting ADCs, which deliver cytotoxic agents directly to HER2-expressing cells, are driving renewed interest in this pathway, and T-DXd has been approved in a tumor-agnostic indication for patients with HER2-positive (IHC3+) solid tumors after previous systemic treatment. IN addition, emerging data have also suggested a role for HER2-targeted ADCs for patients with low HER2 expression.6
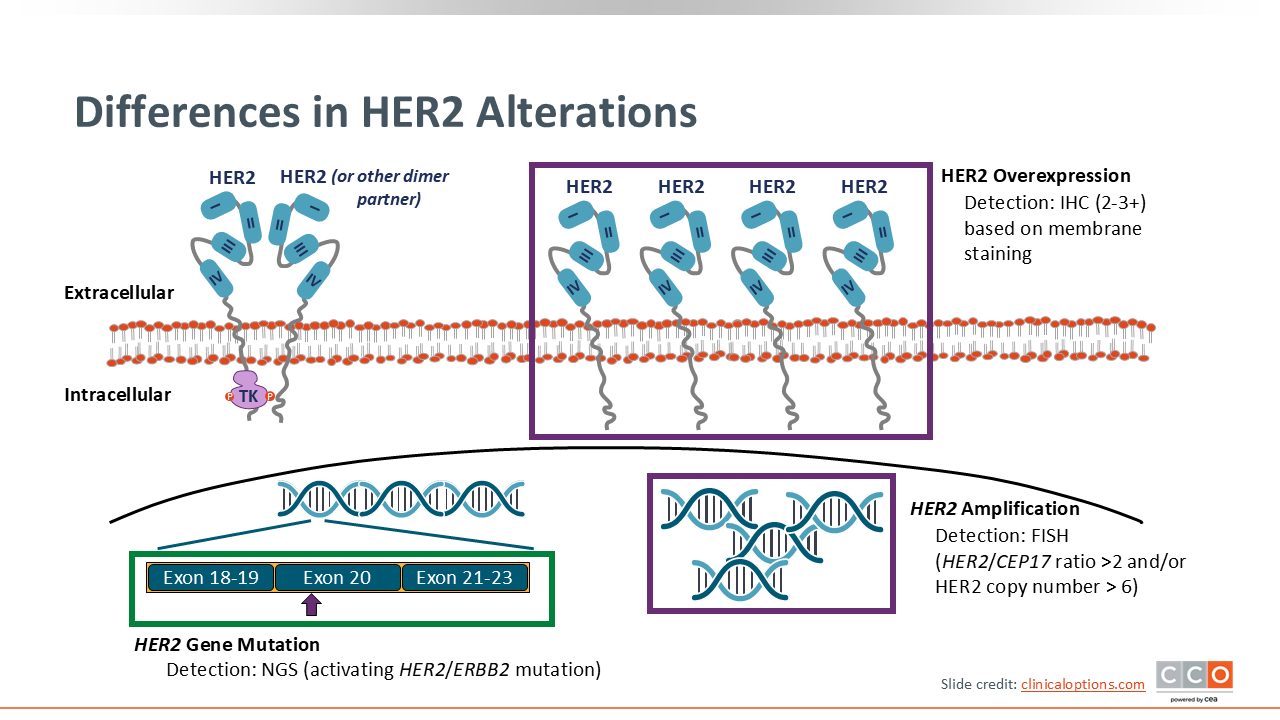
Differences in HER2 Alterations
Cancers have historically been identified as being “HER2 positive” by way of HER2 gene amplification and/or HER2 protein overexpression. However, activating HER2 gene mutations are also emerging as a targetable alterations in some tumor types.
HER2 alterations primarily include gene amplification, protein overexpression, and gene mutations. It is crucial to distinguish between these as they have different biological consequences and may require different testing strategies and treatments.
HER2 gene amplification refers to an increased number of copies of the ERBB2 gene within the cancer cell. This is typically detected using ISH, such as fluorescence in situ hybridization (FISH). Amplification is a common mechanism leading to HER2-positive disease in breast and gastric cancers.
HER2 protein overexpression is the direct result of gene amplification, where the increased number of gene copies leads to excessive production of the HER2 protein on the cell surface. Overexpression is assessed by IHC, which grades the intensity and completeness of membrane staining from 0 to 3+.
HER2 gene mutations are changes in the DNA sequence of the ERBB2 gene itself. Unlike amplification, mutations do not necessarily lead to HER2 overexpression but can lead to activating mutations that cause constitutive activation of HER2 signaling pathways. These mutations are typically identified through next-generation sequencing (NGS).7
Prevalence of HER2 Alterations in Gastric and Other GI Cancers
Gastric cancer emerged as 1 of the earlier GI malignancies where HER2 expression was identified as clinically actionable. Approximately 15%-20% of gastric cancers overexpress HER2,8,9 primarily in the intestinal subtype.10 However, HER2 expression in gastric tumors is highly heterogeneous.11
Beyond gastric cancer, HER2 alterations have also been observed in other GI cancers. In colorectal cancer, approximately 3%-5% of cases show HER2 amplification, particularly those that are left-sided and KRAS wild-type.1 These molecular characteristics led to the development of HER2-targeted regimens for colorectal cancer, especially in chemotherapy-refractory settings. In biliary tract cancers, HER2 overexpression is seen in up to 10% of patients.12
Prevalence of HER2 Alterations in Endometrial and Ovarian Cancers
HER2 also plays a significant role in certain subtypes of GYN malignancies, most notably endometrial cancer. Studies have shown that HER2 gene amplification is present in up to 30% of endometrial cancers,13,14 with enrichment seen in high-grade serous, p53-mutated, and high-grade endometrioid histologies. These subtypes tend to have more aggressive clinical behavior, making the identification of HER2 expression clinically relevant.
In contrast, HER2 overexpression in ovarian cancer is less common. It is rarely found in the more prevalent high-grade serous ovarian cancer but may be seen more frequently in uncommon histologies such as mucinous and clear-cell ovarian cancers. In mucinous subtypes specifically, HER2 overexpression may be present in up to 30%,15 prompting some centers to routinely test for HER2 in this context. HER2 overexpression by IHC is observed in a pooled 5.7% of invasive cervical cancers, and gene amplification in 1.2% based on ASCO/CAP-compliant analyses.16 Currently, the National Comprehensive Cancer Network guidelines recommend tumor molecular analysis, including HER2 status by IHC, to identify actionable biomarkers for targeted therapies in patients with persistent or recurrent disease.17
Prevalence of HER2 Alterations in Urothelial Carcinoma
Among GU malignancies, urothelial carcinoma has the most robust data supporting the role of HER2 as a therapeutic target. In urothelial carcinoma, HER2 overexpression is reported in 15%-20% of patients, although emerging evidence suggests that HER2-low expression—defined as IHC 1+—may be observed in up to one third of cases.18,19 This has important implications for expanding eligibility for HER2-targeted ADCs. Current trials are exploring the benefit of these agents even in patients with low levels of HER2 expression.20
ASCO/CAP Guidelines for HER2 Classification
The decision of when and how to test for HER2 in solid tumors depends on tumor type, disease stage, and therapeutic context.
Measurement of HER2 expression has traditionally been performed following ASCO/CAP guidelines for testing HER2 in breast cancer and in gastric cancer relying on IHC, scored as 0, 1+, 2+, or 3+.21 Tumors with IHC 3+ (strong, complete membrane staining in >10% of tumor cells) are classified as HER2-positive. IHC scores of 0 or 1+ remain HER2-negative. IHC 2+ (weak to moderate complete membrane staining in >10% of cells) remains equivocal and reflex ISH is required to determine HER2-positivity. Cases that are IHC 2+ with ISH-confirmed ERBB2 amplification are reported as HER2-positive; IHC 2+ that are ISH-negative were deemed HER2-negative.
Historically, HER2-targeted therapies were restricted to HER2-positive (IHC 3+ or IHC2+ and ISH-amplified) tumors. However, in January 2025, the FDA approved T-DXd for unresectable or metastatic HR-positive breast cancers with HER2-low or HER2-ultralow expression that have progressed on 1 or more endocrine therapy.22 Although definitions of HER2-low and HER2-ultralow have not yet been included in ASCO/CAP guidelines, HER2-low has been defined as IHC 1+ or 2+ with negative ISH and HER2-ultra-low as ≤10% of infiltrating cancer cells showing incomplete and faint/weak membrane staining.23[Shami 2025]
Although the determination of HER2 protein expression and HER2 gene mutation status is currently part of reflex testing for most patients with breast cancer, HER2 is a new biomarker in many other types of cancer. Therefore, it is important to understand what is being evaluated using the reflex testing system. Comprehensive molecular profiling via NGS can be used to identify activating HER2 mutations, typically occurring as exon 20 insertions in the HER2 gene or less commonly as single nucleotide variants involving exons 19-21 of the HER2 kinase domain. Beyond breast cancer, HER2 overexpression has emerged as an actionable biomarker in multiple additional solid tumors.24,25
Guideline and Expert Recommendations for Type of HER2 Testing Across Solid Tumors
When considering HER2 testing guidelines across these malignancies, it is important to remember that each tumor type has its own recommendations for testing. In gastric cancer, HER2 IHC testing is routinely performed in all patients with advanced or metastatic disease26 where HER2 positivity can guide first-line therapy. In colorectal27 and biliary tract28 cancers, HER2 alterations can also be detected through NGS, especially in left-sided, KRAS wild-type colorectal tumors.
In GYN cancers, HER2 testing is most commonly used in aggressive subtypes of endometrial cancer, particularly those with serous or p53-mutated histology. NCCN guidelines recommend HER2 testing, typically by IHC, for all patients with advanced or metastatic uterine serous carcinoma or other p53-aberrant subtypes. For recurrent ovarian cancer, tumor molecular analysis—which can include HER2 testing by IHC—is recommended to identify potential targets.29 In ovarian cancer, tumor molecular analysis is recommended at recurrence to identify potential benefit from targeted therapeutics that have tumor-specific or tumor-agnostic benefit including, but not limited to, HER2 status by IHC.17 However, HER2 overexpression is more common for rarer subtypes such as mucinous or clear-cell carcinoma.30
For metastatic bladder cancer, HER2 testing is increasingly conducted at diagnosis using both IHC and NGS, often alongside FGFR3 and PD-L1 testing,31 to determine clinical trial eligibility and to inform future therapy lines.
Challenges in HER2 Testing and Scoring
HER2 testing methodologies and interpretation criteria vary significantly across institutions and tumor types. In clinical trials such as DESTINY-PanTumor02, HER2 assessment was performed using gastric cancer scoring systems,32 while other institutions may apply breast cancer criteria regardless of tumor origin. These inconsistencies can lead to discordant HER2 status assignments, potentially affecting patient eligibility for trials and access to therapy.
Standardized scoring systems—such as those recommended by ASCO/CAP—exist for breast22 and gastric24 cancers, but harmonized guidance for other tumors remains lacking. Beyond scoring, variations in antibodies used, tissue processing, and reporting practices all contribute to variability in HER2 assessment. Healthcare professionals and pathologists must collaborate closely to ensure reliable interpretation of HER2 testing results.
Timing of HER2 Testing in Clinical Practice
Optimal timing of HER2 testing is also critical to ensuring patients receive appropriate therapy. In diseases like gastric cancer, HER2 testing is routine and conducted at diagnosis of advanced disease. For other tumors, such as bladder, endometrial, and colorectal cancers, HER2 testing may be initiated when NGS panels are ordered at metastatic presentation or progression, often as part of broader biomarker profiling.32
The rationale for early testing, even when HER2-targeted agents may be reserved for later lines, is that biomarker data inform not only current decisions but also future treatment options and clinical trial enrollment. As HER2-targeted ADCs become more widely available and approved across indications, earlier identification of HER2 expression—regardless of the level—will be essential to optimize care pathways.
HER2-Low: Expanding the Targetable Population
The emergence of HER2-targeted ADCs has led to a paradigm shift in how HER2 expression is defined and targeted. Historically, only tumors with HER2 3+ expression or HER2 gene amplification were considered suitable for HER2-directed therapy. However, recent clinical trial data have demonstrated that patients with HER2-low tumors (IHC 1+ or 2+ without amplification) may also respond to HER2-targeted ADCs.33-35
This has raised important questions for other solid tumors, such as gastric, colorectal, bladder, and endometrial cancers. Although HER2-low testing is not yet uniformly standardized across all tumor types, these findings suggest that expanding the definition of HER2 positivity could increase access to targeted therapies and broaden patient eligibility for clinical trials. Ongoing studies are evaluating the efficacy of ADCs in the HER2-low population.37,38
Closing Remarks
The understanding of HER2 as a biomarker and therapeutic target has rapidly evolved. With the introduction of ADCs capable of targeting both HER2-high and HER2-low tumors, there is a renewed need for precise and consistent HER2 testing across all relevant tumor types. Healthcare professionals should remain aware of tumor-specific testing practices, scoring guidelines, and emerging data supporting the role of HER2 in GI, GU, and GYN cancers.
Ongoing research continues to refine the definition of HER2 positivity, optimize testing methods, and evaluate ADC efficacy in less traditionally HER2-driven tumors. As new data emerge, multidisciplinary collaboration among oncologists, pathologists, and trial investigators will be essential to translating these insights into meaningful improvements in patient care.