CE / CME
Enhancing Outcomes in Extensive-Stage Small-Cell Lung Cancer With Recent Therapeutic Advancements for First-line Therapy
Physician Assistants/Physician Associates: 0.25 AAPA Category 1 CME credit
ABIM MOC: maximum of 0.25 Medical Knowledge MOC point
Pharmacists: 0.25 contact hour (0.025 CEUs)
Nurse Practitioners/Nurses: 0.25 Nursing contact hour
Physicians: maximum of 0.25 AMA PRA Category 1 Credit™
Released: November 10, 2025
Expiration: May 09, 2026
Activity
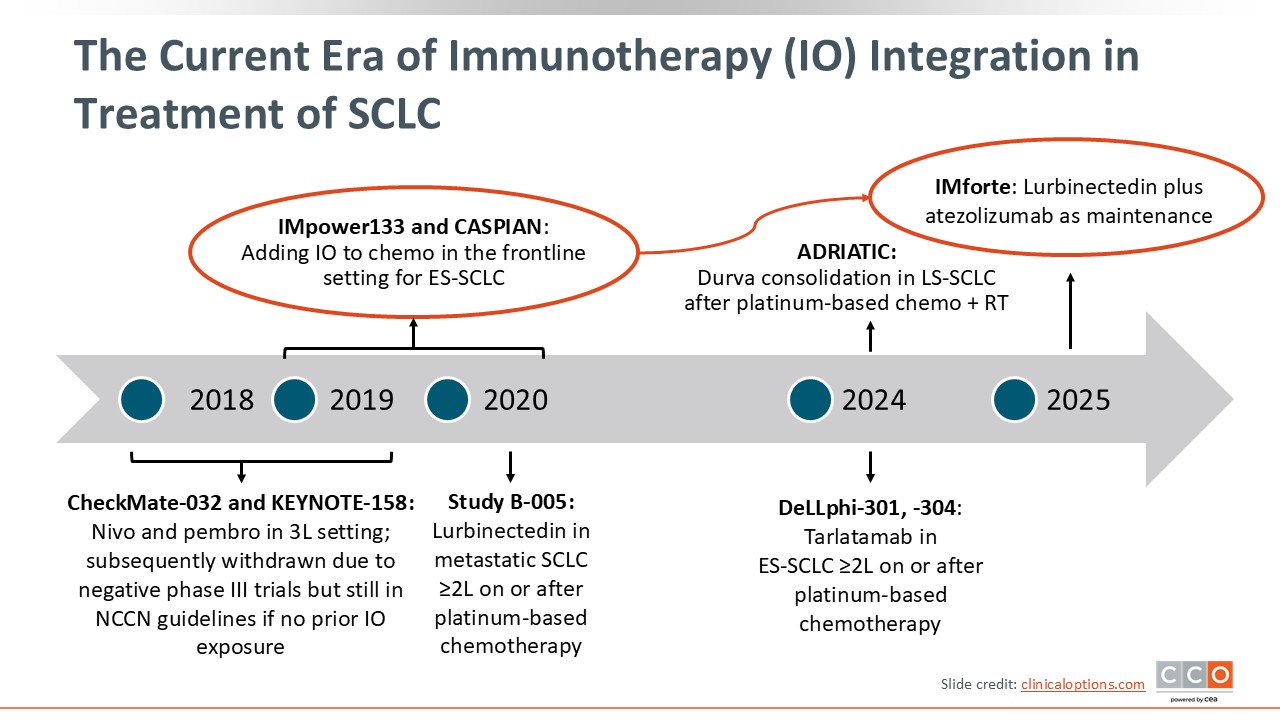
The Current Era of Immunotherapy (IO) Integration in Treatment of SCLC
Chemoimmunotherapy is the current standard of care in the frontline setting for patients with ES-SCLC. The phase III IMpower133 and CASPIAN trials, which showed clear survival benefits with the addition of atezolizumab or durvalumab, respectively, to chemotherapy (CT), led to FDA approval of these agents in combination with CT in this setting. Lurbinectedin was also recently FDA approved in combination with atezolizumab maintenance after first-line induction therapy with chemoimmunotherapy with atezolizumab, based on the IMforte trial.
In this text module, we will focus on the current data for first-line treatment approaches for patients with ES-SCLC. Additional information on limited-stage SCLC, treatment for ES-SCLC after progression, and how to manage adverse events (AEs) associated with newer treatment options can be found here.
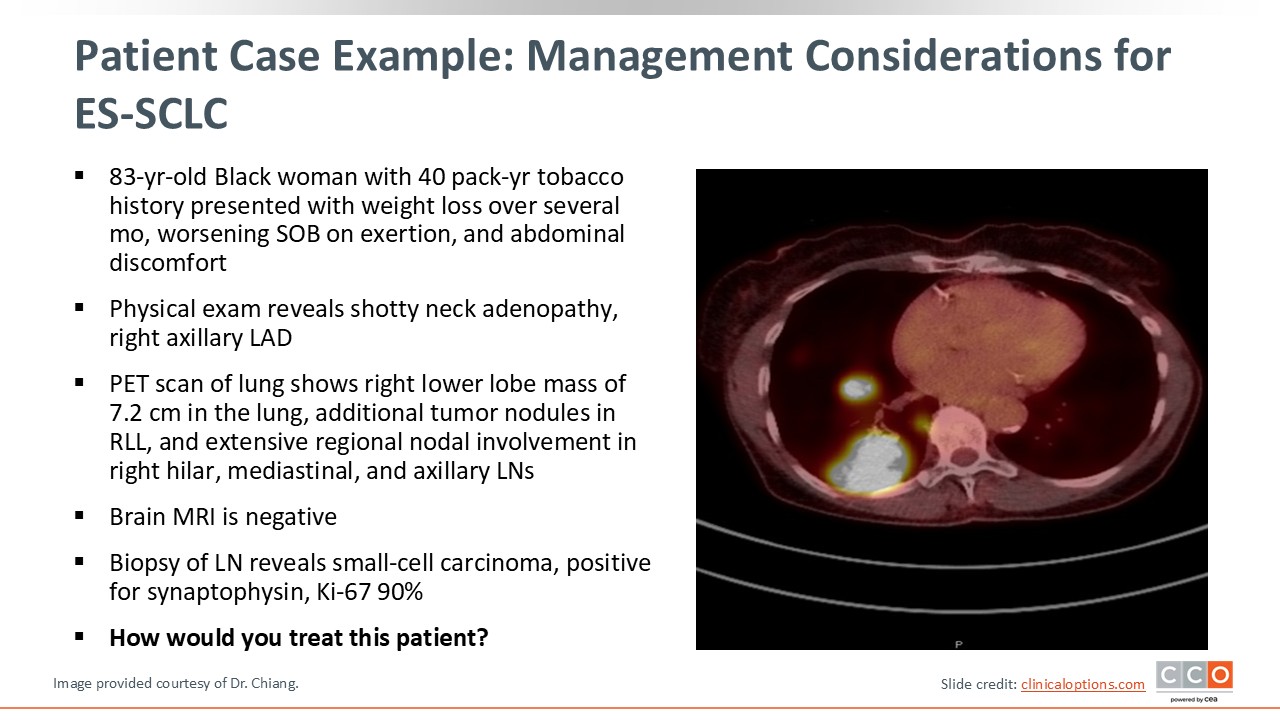
Patient Case Example: Management Considerations for ES-SCLC
To think through current management decisions for patients with ES-SCLC, let us consider a patient case example.
An 83-year-old Black woman with a 40 pack-year history of tobacco use presented with several concerning symptoms, including progressive weight loss over several months, worsening shortness of breath with exertion, and abdominal discomfort. Her physical exam showed shotty neck adenopathy and right axillary lymphadenopathy. A subsequent PET scan revealed a hypermetabolic 7.2 cm mass in the right lower lobe. Additional tumor nodules in the right lower lobe could also be seen. The patient also had extensive regional nodal involvement in the right hilar mediastinal and axillary lymph nodes. The brain MRI was negative for metastasis.
A biopsy of the axillary lymph node revealed small-cell carcinoma, positive for synaptophysin, with a Ki-67 proliferative index of 90%, indicating a highly proliferative ES-SCLC tumor.
Patient Case Example: Management Considerations for Newly Diagnosed ES-SCLC
Patients who are diagnosed with ES-SCLC often present with high disease burden and symptomatic decline requiring initial hospitalization for stabilization and initiation of systemic therapy. Standard first-line treatment typically consists of a platinum agent (carboplatin or cisplatin) and etoposide with an ICI targeting PD-L1 (atezolizumab or durvalumab), which can be introduced in the second cycle once the patient is clinically stable and discharged from the hospital.
Following induction therapy, patients who respond or achieve disease control should continue maintenance treatment until progression or unacceptable toxicity. As mentioned, lurbinectedin was recently FDA approved in combination with atezolizumab maintenance in this setting based on the IMforte trial and should be considered for eligible patients.
Regarding future options, data on CT plus anti–PD-1 antibodies from clinical trials primarily conducted in China look promising. These trials, such as ASTRUM-005 (serplulimab), CAPSTONE-1 (adebrelimab), and RATIONALE-312 (tislelizumab), have shown impressive survival benefits, although these agents are not yet approved in the United States. Other trials are exploring frontline combinations with novel agents such as bispecific antibodies and antiangiogenic inhibitors in various combinations with standard chemoimmunotherapy. Also, very exciting is the potential of replacing CT with antibody–drug conjugates (ADCs) in the frontline setting.
Now we will discuss some of the trials that led to the current management decisions mentioned in this case example.
Phase III Trials: Addition of Anti–PD-L1 Checkpoint Inhibitors to First-line Platinum + Etoposide in ES-SCLC
Two pivotal phase III trials that utilized anti–PD-L1 ICIs in combination with CT in the first-line setting led to the current standard of care.
The phase III IMpower133 trial included patients with measurable ES-SCLC, an Eastern Cooperative Oncology Group performance status (ECOG PS) of 0 or 1, and no prior systemic therapy. Patients with treated asymptomatic brain metastases were eligible to participate in this study. Participants were randomized to receive CT with carboplatin/etoposide plus IO with atezolizumab in the induction phase followed by maintenance with atezolizumab until disease progression or carboplatin/etoposide plus placebo followed by placebo maintenance. Coprimary endpoints were OS and progression-free survival.1,2
The phase III CASPIAN trial had a similar design. Eligible patients had treatment-naive ES-SCLC and an ECOG PS of 0 or 1. In this trial, patients could have asymptomatic or treated and stable brain metastases. Participants were randomized to 3 arms: CT with a platinum agent/etoposide plus durvalumab and tremelimumab followed by durvalumab and tremelimumab maintenance, CT plus durvalumab followed by durvalumab maintenance, or CT followed by observation. In this trial, either carboplatin or cisplatin could be used as the platinum agent, and the primary endpoint was OS.3
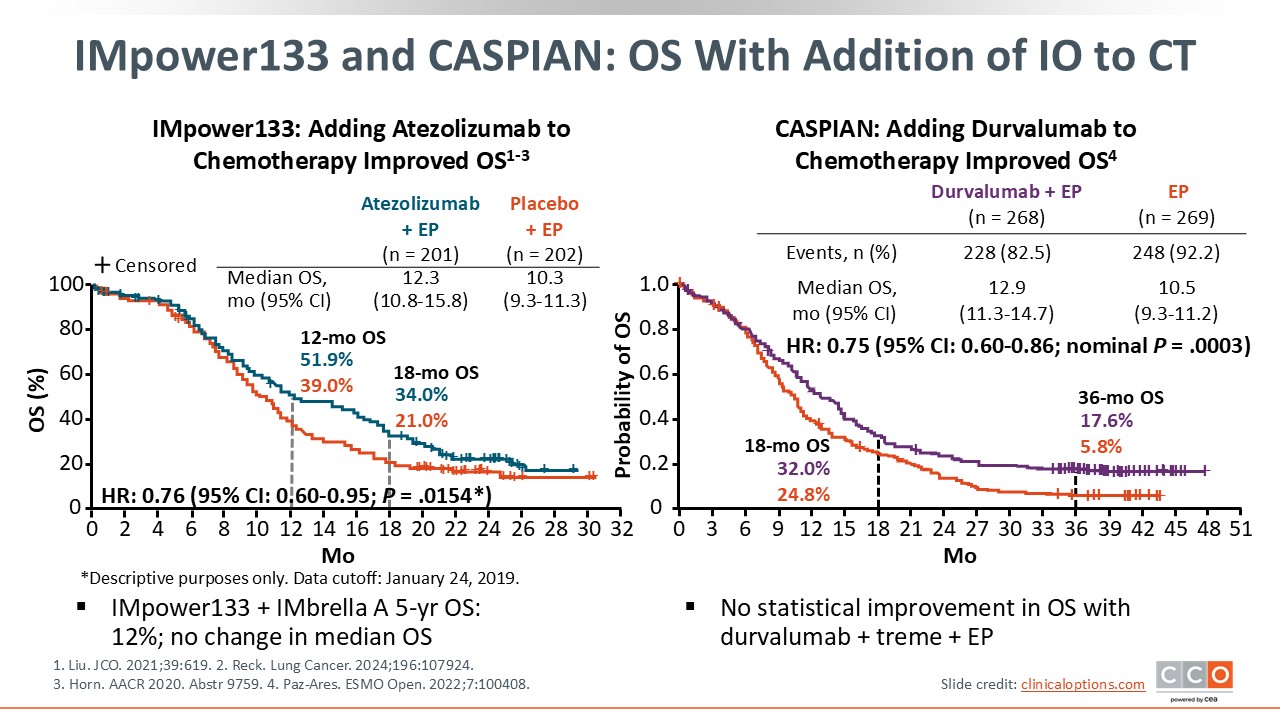
IMpower133 and CASPIAN: OS With Addition of IO to CT
In IMpower133, the hazard ratio for OS was 0.76 (95% CI: 0.60-0.95; P = .0154) with the addition of atezolizumab to CT. Median OS with CT plus atezolizumab was 12.3 months vs 10.3 months with CT plus placebo.1 The FDA approved atezolizumab in combination with carboplatin and etoposide for the first-line treatment of adult patients with ES-SCLC in 2019.4
After the protocol-defined follow-up period in Impower133 was completed, patients were offered enrollment in an extension trial called IMbrella A. Although the number of patients was small, the data from this trial report a 5-year OS rate of approximately 12%,5 confirming a durable long-term survival benefit for a subset of patients.
In CASPIAN, the hazard ratio was similar at 0.75 (95% CI: 0.60-0.86; nominal P = .0003). Median OS was 12.9 months with CT + durvalumab vs 10.5 months with CT alone.3 This trial extended to 3 years and reported a 3-year OS rate of 17.6% with the addition of durvalumab vs 5.8% for the platinum doublet only. Based on these results, the FDA approved durvalumab in combination with etoposide and either carboplatin or cisplatin for the first-line treatment of adult patients with ES-SCLC in 2020.6]
There were a few differences to emphasize between the Impower133 and CASPIAN trials: IMpower133 included patients with treated brain mets only, whereas CASPIAN also included patients with asymptomatic brain mets; and cisplatin or carboplatin was allowed in CASPIAN whereas only carboplatin was allowed in IMpower133.
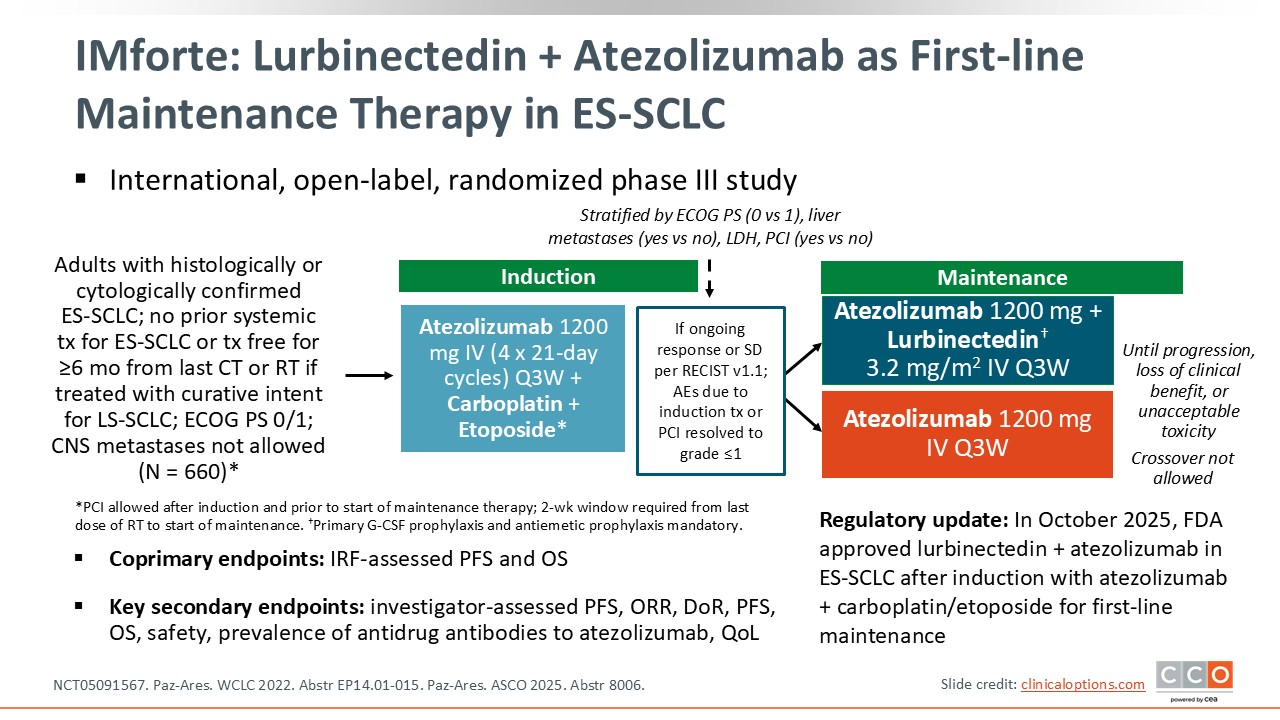
IMforte: Lurbinectedin + Atezolizumab as First-line Maintenance Therapy in ES-SCLC
The phase III IMforte trial explored the potential of using an active agent approved in the relapsed setting earlier in the disease course as a part of maintenance treatment. In this randomized study, patients with ES-SCLC without progression after 4 cycles of first-line carboplatin/etoposide plus atezolizumab were randomized to receive maintenance therapy with either lurbinectedin plus atezolizumab or atezolizumab alone. Patients in this trial had an ECOG PS of 0 or 1 and central nervous system metastases were not allowed. The primary endpoint was progression-free survival and OS assessed by an independent review facility.7
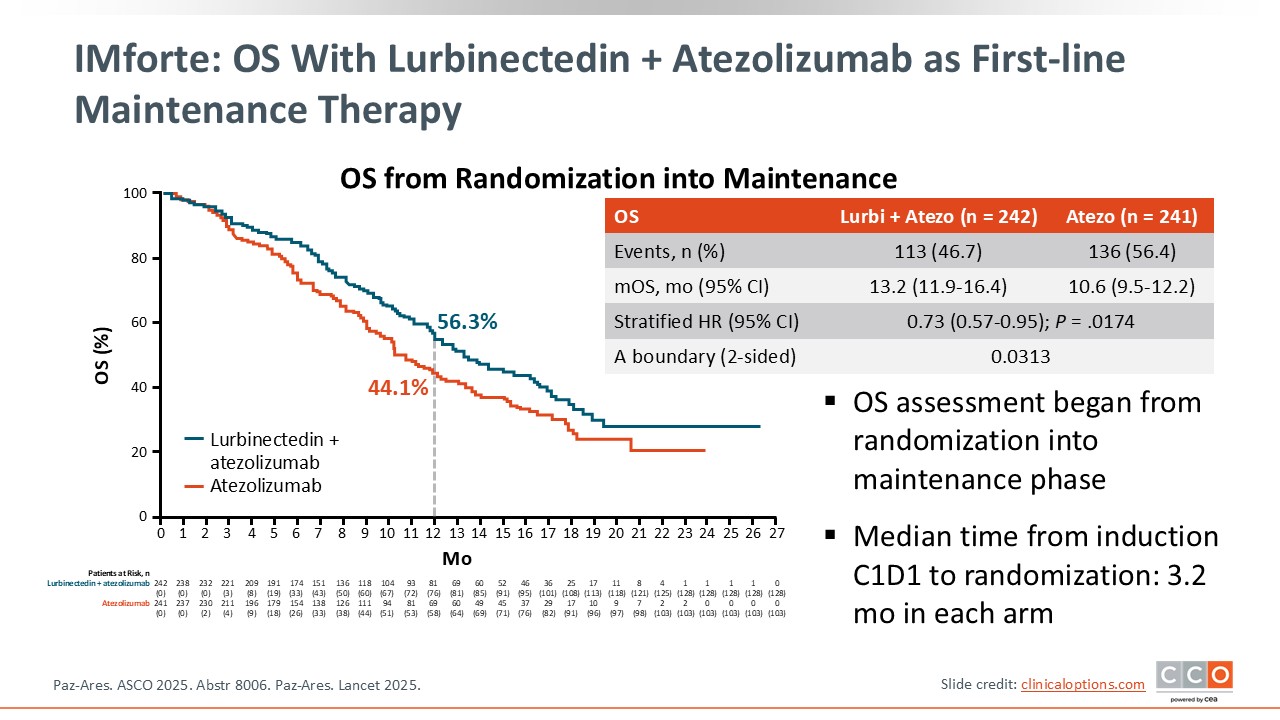
IMforte: OS with Lurbinectedin + Atezolizumab as First-line Maintenance Therapy
A significant OS benefit was observed with lurbinectedin plus atezolizumab vs atezolizumab alone, with a median OS from randomization of 13.2 months vs 10.6 months, respectively (HR: 0.73; 95% CI: 0.57-0.95; P = .0174).
At 12 months, the OS rate was 56.3% for the lurbinectedin + atezolizumab arm and 44.1% for the atezolizumab arm.7
In October 2025, the FDA approved lurbinectedin in combination with atezolizumab as a first-line maintenance therapy in ES-SCLC for patients whose disease has not progressed after induction treatment with atezolizumab plus carboplatin/etoposide.
Current NCCN Guidelines on Primary Treatment for ES-SCLC
In addition to considerations for systemic therapy, patients with ES-SCLC often require radiation therapy (RT) and/or steroids for symptomatic sites or brain metastases. According to current NCCN guidelines for the primary treatment of ES-SCLC, if a patient has brain metastases that are asymptomatic, systemic treatment should begin before any brain RT. Often, brain metastases will disappear in many of these patients and only monitoring is needed.
If a patient has symptomatic brain metastases, the NCCN guidelines recommend brain RT prior to systemic treatment unless immediate systemic treatment is required. Steroids can be used as needed.
If the disease is in other localized symptomatic sites and causes, for example, superior vena cava syndrome, lobar obstruction, or bone metastases, then the primary treatment is systemic therapy, and RT to symptomatic sites can be considered. If spinal cord compression is a factor, steroids should be initiated immediately, and systemic therapy plus RT (usually sequential) is indicated.
For patients without any localized symptomatic sites of brain metastases, a good PS, or a poor PS due to the disease, the NCCN guidelines recommend starting combination systemic therapy as discussed above, including supportive care. If the patient has a poor PS due to the disease, then the treatment plan is individualized, where the risks and benefits for each individual patient are weighed, and best supportive care is certainly an option.8
NCCN Guideline for Patients With SCLC Who Do Not Have a History of Smoking
There are several NCCN guidelines suggestions for patients with SCLC without a history of smoking. Comprehensive tumor profiling is not typically performed on patients with ES-SCLC. However, in patients who do not smoke tobacco, lightly smoke, or have a remote smoking history, genetic profiling can be considered because the results may change management.
If the patient has a targetable mutation, such as EGFR, osimertinib plus with CT can be considered.8
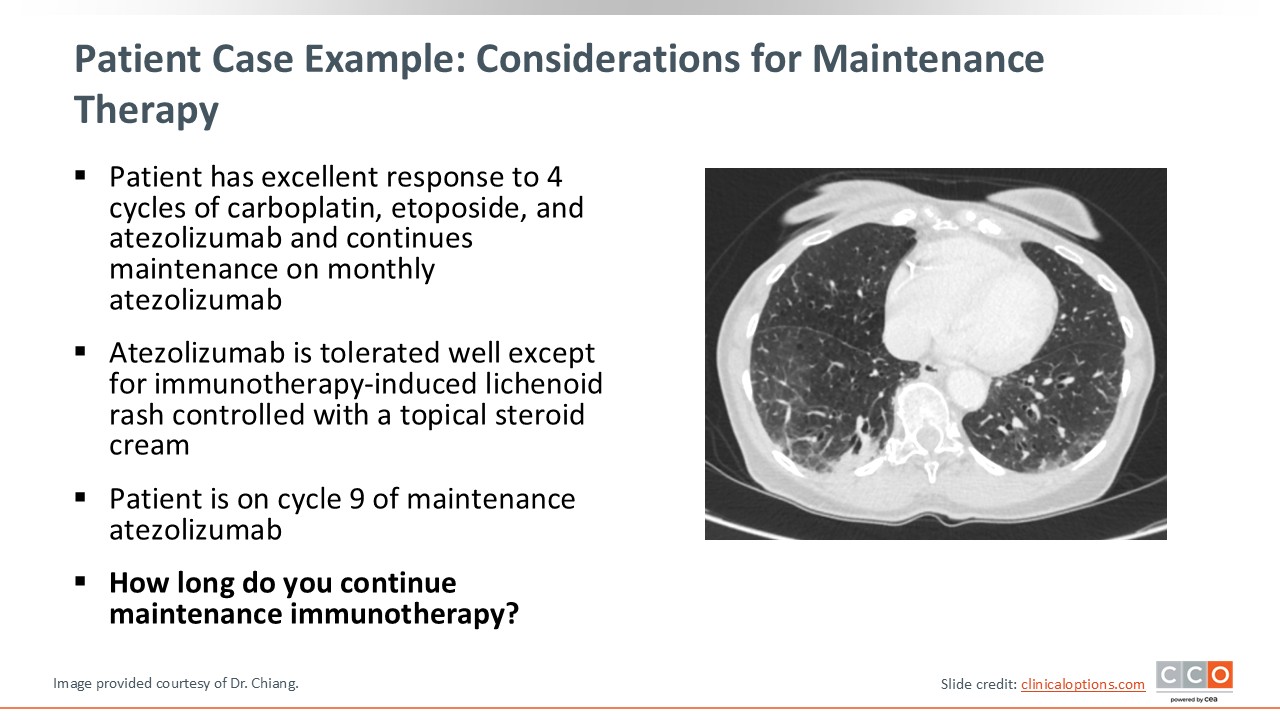
Patient Case Example: Considerations for Maintenance Therapy
Let us continue with our patient case. The patient had an excellent response to 4 cycles of carboplatin, etoposide, and atezolizumab using the IMpower133 regimen. This patient began atezolizumab maintenance before the new approval of lurbinectedin plus atezolizumab maintenance. At the time of this scan, she was continuing monthly atezolizumab and was on cycle 9 of maintenance.
The next question we must consider is how long to continue maintenance IO. The duration of maintenance IO is not fixed for patients with ES-SCLC, and treatment with IO therapy is generally continued until progression or unacceptable toxicity. However, practice patterns may vary and some consider holding or discontinuing IO therapy after 2 years. Now with the addition of lurbinectedin to atezolizumab maintenance until progression, the question of how long one should continue maintenance therapy remains.
In my clinical practice, we will often continue therapy depending on how well the patient tolerates the maintenance treatment. For patients with no side effects or minimal side effects that can be controlled with supportive care, we may consider continuing maintenance until progression. For patients that do experience side effects that may impact quality of life, having a conversation about discontinuing maintenance may also be a reasonable approach.
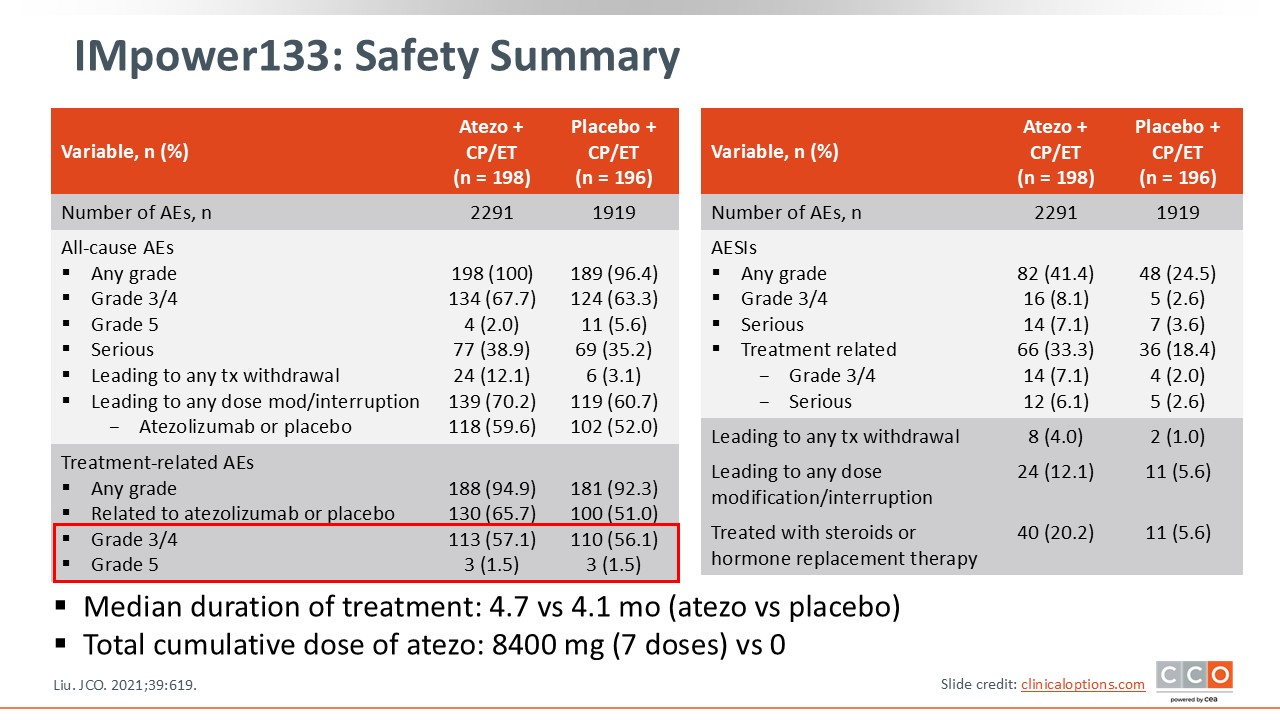
IMpower133: Safety Summary
When comparing the treatment-related adverse events (TRAEs) in the atezolizumab vs placebo arms of IMpower133, they appear to be very similar regardless of the addition of atezolizumab. The percentages of patients who experienced grade 3 or 4 TRAEs were 57% vs 56%. The median duration of treatment was 4.7 months for atezolizumab vs 4.1 months for placebo, and the total cumulative dose of atezolizumab was 8400 mg given in 7 doses. In this trial, 12.1% of patients receiving atezolizumab experienced AEs leading to dose modification/interruption vs 5.6% of patients receiving placebo. Similarly, 4.0% of patients receiving atezolizumab experienced AEs leading to discontinuation vs 1.0% of patients receiving placebo. Of note, our patient case example received 42 cycles of atezolizumab, stopped at that time due to COVID-19, and remains with no evidence of disease 2 years after stopping atezolizumab.1
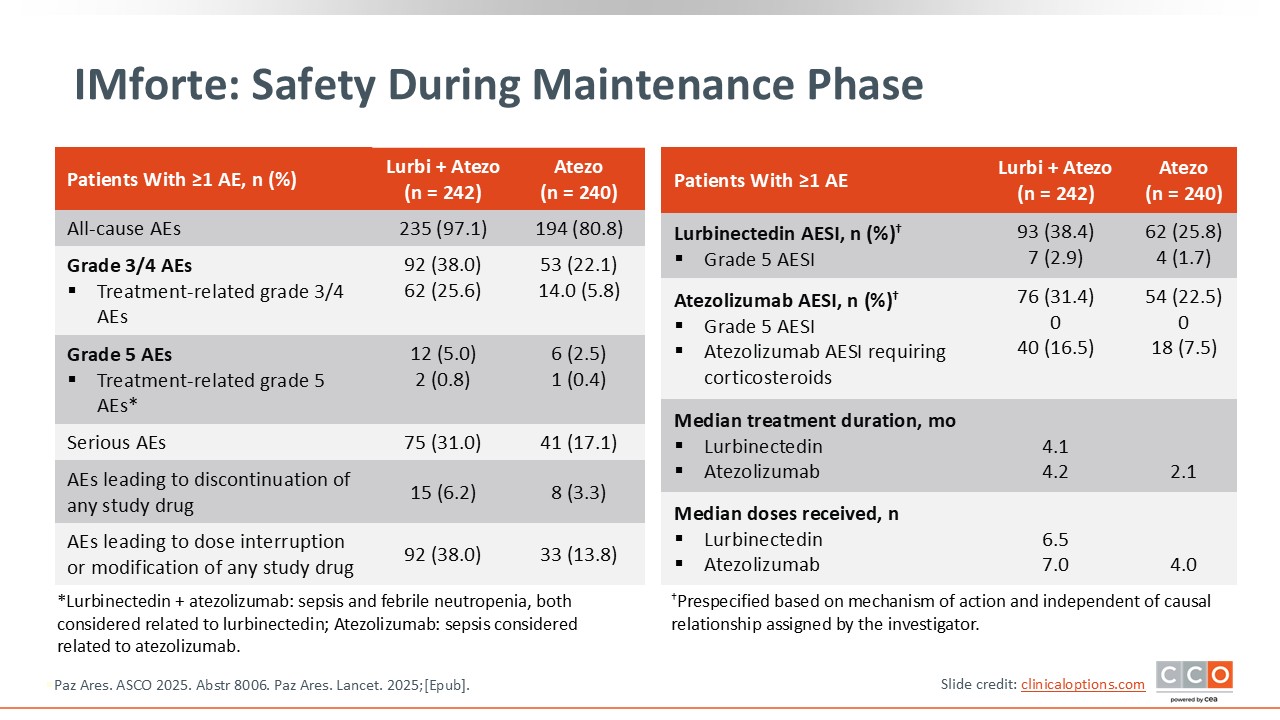
IMforte: Safety During Maintenance Phase
In the IMforte trial, the safety profile of lurbinectedin plus atezolizumab was manageable and consistent with the known profiles of each agent. Any grade AEs occurred in 97.1% vs 80.8%. Grade 3 or 4 TRAEs were 25.6% vs 5.8%, and grade 5 TRAEs were 0.8% vs 0.4%. Serious AEs occurred in 31.0% vs 17.1%. AEs leading to dose modification or interruption were 38.0% vs 13.8%, and discontinuations were 6.2% vs 3.3%. Median maintenance exposure was 4.1 months with lurbinectedin and 4.2 months with atezolizumab in the combination arm vs 2.1 months with atezolizumab monotherapy. Median doses received were 6.5 lurbinectedin and 7.0 atezolizumab in the combination arm vs 4.0 atezolizumab doses with monotherapy. Of importance, there were no new or unexpected safety signals.7
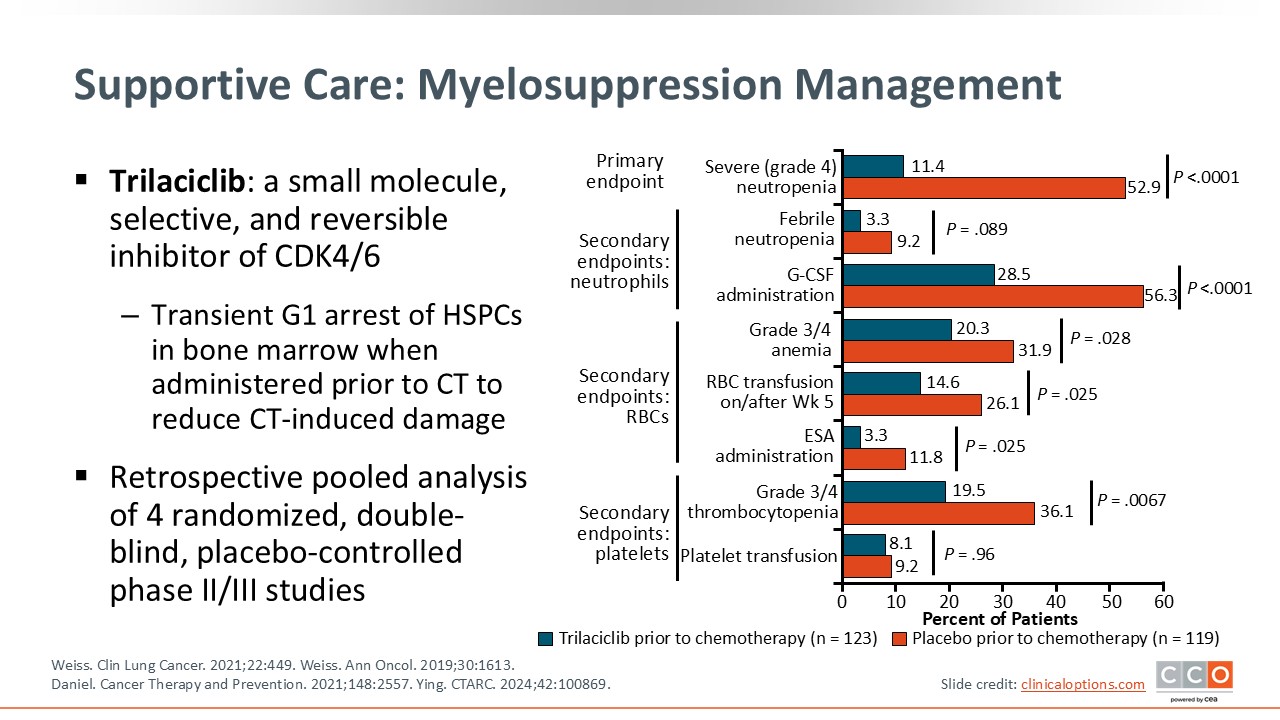
Supportive Care: Myelosuppression Management
As with any therapy regimen for patients with cancer, supportive care is an important consideration for helping patients maintain optimal quality of life and remain on therapy to optimize their survival outcomes.
For IO-related toxicities, management typically involves withholding the IO agent and administering corticosteroids, depending on the severity. Lurbinectedin toxicities are primarily hematologic and managed with dose modifications and growth factor support as needed.
For patients receiving CT, significant side effects often include cytotoxicity and myelosuppression. Subsequent cytopenias, including neutropenia, thrombocytopenia, and anemia, require supportive care such as G-CSF for neutropenia; platelet transfusions for thrombocytopenia; and erythropoiesis‐stimulating agents, red blood cell, and/or iron supplements for anemia. Trilaciclib can also be considered to protect from CT-induced damage. These agents are guideline recommended for reducing myelosuppression in patients during frontline therapy and other CT-containing regimens.8
However, GM-CSF or G-CSF are not recommended during concurrent systemic therapy with RT. Trilaciclib or G-CSF can be used as prophylaxis to decrease the incidence of CT-induced myelosuppression when platinum/etoposide with or without ICI-containing regimens or a topotecan-containing regimen is administered for ES-SCLC.
Trilaciclib is a CDK4/6 inhibitor that decreases myelosuppression by transiently arresting hematopoietic stem and progenitor cells in G1 phase of the cell cycle. Therefore, those cells are protected from CT-induced damage.
In a phase II trial exploring the use of trilaciclib vs placebo prior to carboplatin/etoposide and atezolizumab in patients with ES-SCLC, key endpoints of myelosuppression were significantly less frequent in patients treated with trilaciclib. Specifically, the primary endpoint, duration of severe neutropenia in cycle 1, was shorter (0 days vs 4 days; P <.0001), and the occurrence of grade ≥3 AEs like febrile neutropenia was lower in the trilaciclib arm.9 Real-world analyses have also shown less frequent cytopenias, reduced use of supportive care, fewer hospitalizations, and improved quality of life with trilaciclib.10
Approaches to Improve Long-term Outcomes
As we continue to explore new avenues for treatment for our patients with ES-SCLC, the question of how to continue to improve long-term outcomes for more patients remains.
One approach is to use active agents in earlier settings, such as in limited-stage disease as well as in frontline and/or maintenance therapy for ES-SCLC, either alone or in novel combinations. Another approach is to try to understand the heterogeneity and determinants of response and resistance so that a more targeted approach can be utilized to individualize treatment for our patients with SCLC.
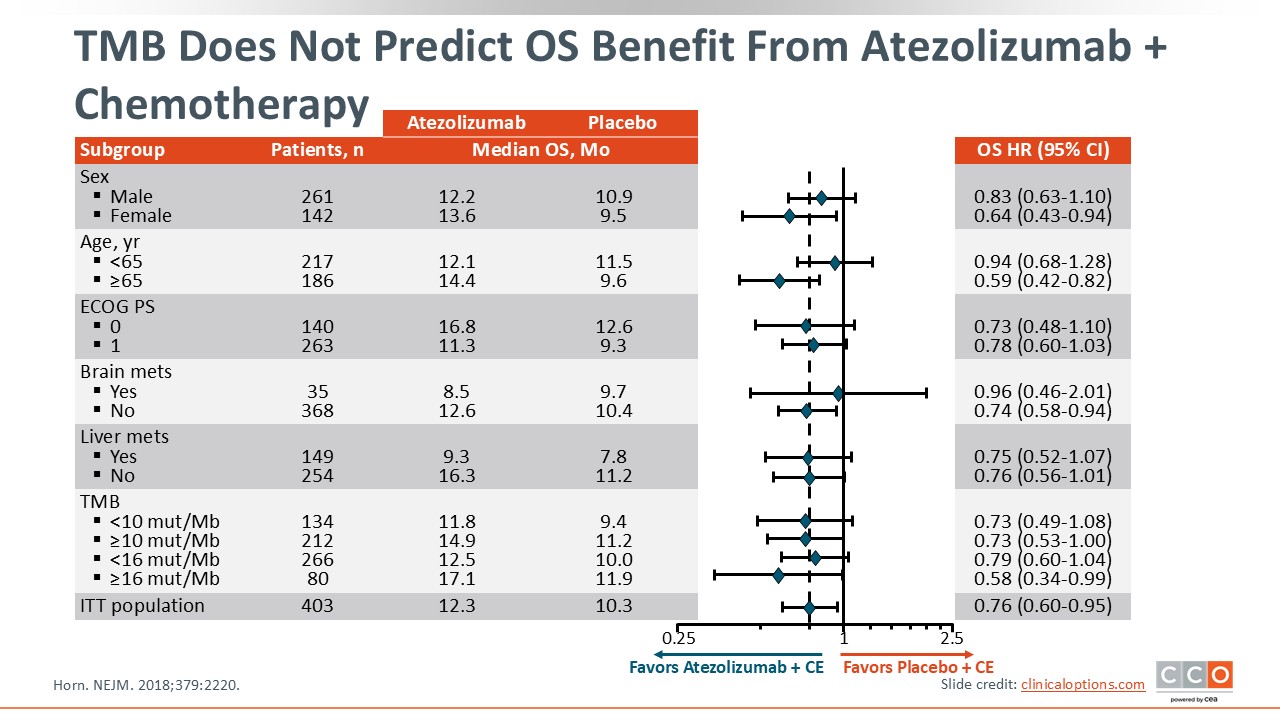
TMB Does Not Predict OS Benefit From Atezolizumab + Chemotherapy
In order to personalize therapy, research has attempted to understand potential biomarkers for response to ICIs. However, the search for an optimal biomarker continues.
Initially, CheckMate-032 suggested that a high tumor mutational burden (TMB) may be a good predictor of response because patients with a high TMB had a 46% objective response rate following treatment with nivolumab plus ipilimumab.11
However, in IMpower133, a randomized, retrospective trial of atezolizumab plus carboplatin and etoposide vs placebo plus carboplatin and etoposide for first-line ES-SCLC, an exploratory blood-based TMB analysis did not predict OS benefit. The survival advantage with atezolizumab plus CT was seen irrespective of TMB or PD-L1 status.2
In addition, PD-L1 status does not correlate with response in SCLC, regardless of whether measured by combined positive score or immune cell status, as evidenced by KEYNOTE-60412 and CASPIAN.3 Collectively, these findings suggest that neither TMB nor PD-L1 expression is a reliable biomarker of benefit in ES-SCLC.
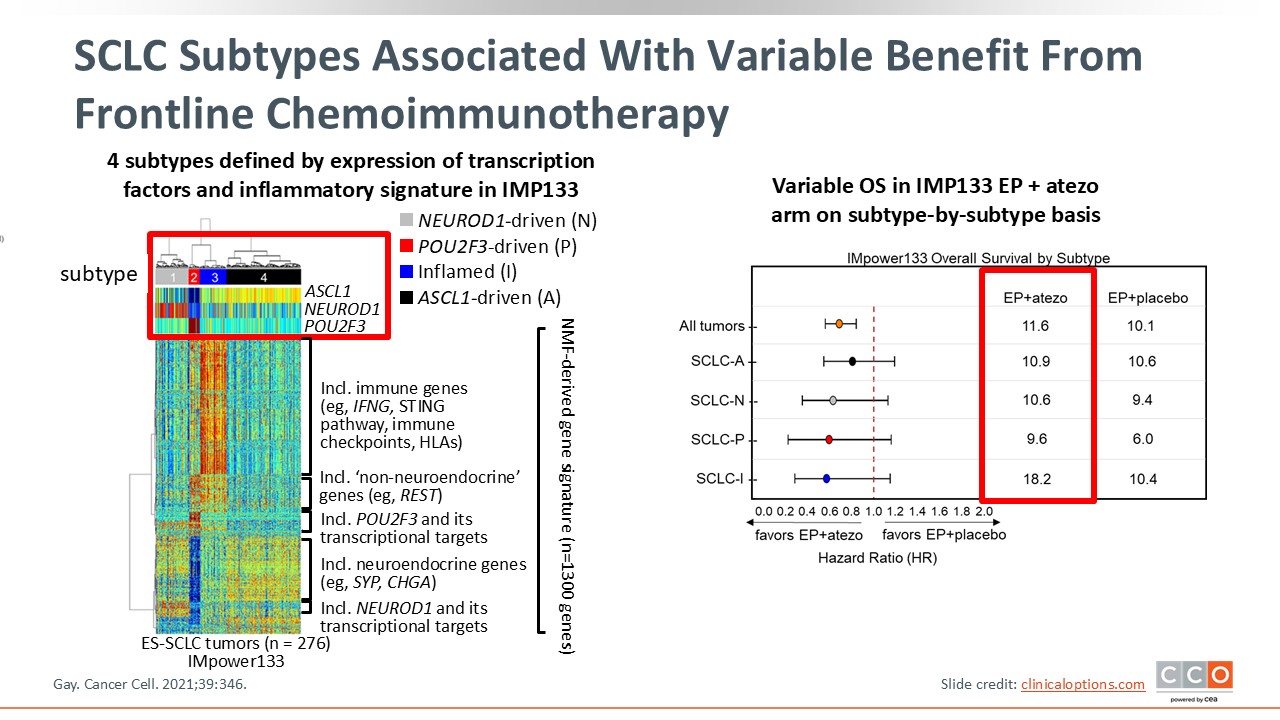
SCLC Subtypes Associated With Variable Benefit from Frontline Chemoimmunotherapy
We are beginning to better understand the transcriptomic profile of SCLC due, in large part, to a study conducted at MD Anderson Cancer Center. An unsupervised cluster analysis was performed on IMpower133 data in which 4 different subtypes were described, mostly defined by overexpression of transcription factors in ASCL1 (subtypes A), NEUROD1 (subtype N), and POU2F3 (subtype P), and an inflammatory signature in an inflammatory signature (subtype I).
Patients with subtype I in the etoposide plus atezolizumab arm experienced the longest OS (18.2 months), and this benefit was not observed in the placebo arm.13 These data suggest that subtype I may be a predictor of benefit of ICI therapy; however, more data are needed to confirm this.
SWOG S2409-PRISM: Durvalumab vs Biomarker-Directed Novel Agents + Durvalumab in ES-SCLC
Based on the results from cluster analysis in the IMpower133 trial data (subtype I), a cooperative group phase II trial, called S2409 (PRISM), was designed. In this trial, over 800 patients will receive frontline induction therapy with carboplatin, etoposide, and durvalumab, and their SCLC subtypes will be assessed. Based on those results and their Schlafen 11 (SLFN11) status, they will be randomized to a biomarker-directed targeted therapy plus durvalumab vs durvalumab alone. Patients with subtype I will receive durvalumab and monalizumab, a natural killer cell activator, or durvalumab alone. Patients with subtypes A/N and SLFN11 negative will receive the ATR inhibitor ceralasertib plus durvalumab vs durvalumab alone, and patients with subtypes A/N and SLFN11 positive or with subtype P will receive a PARP inhibitor plus durvalumab vs durvalumab alone (NCT06769126).
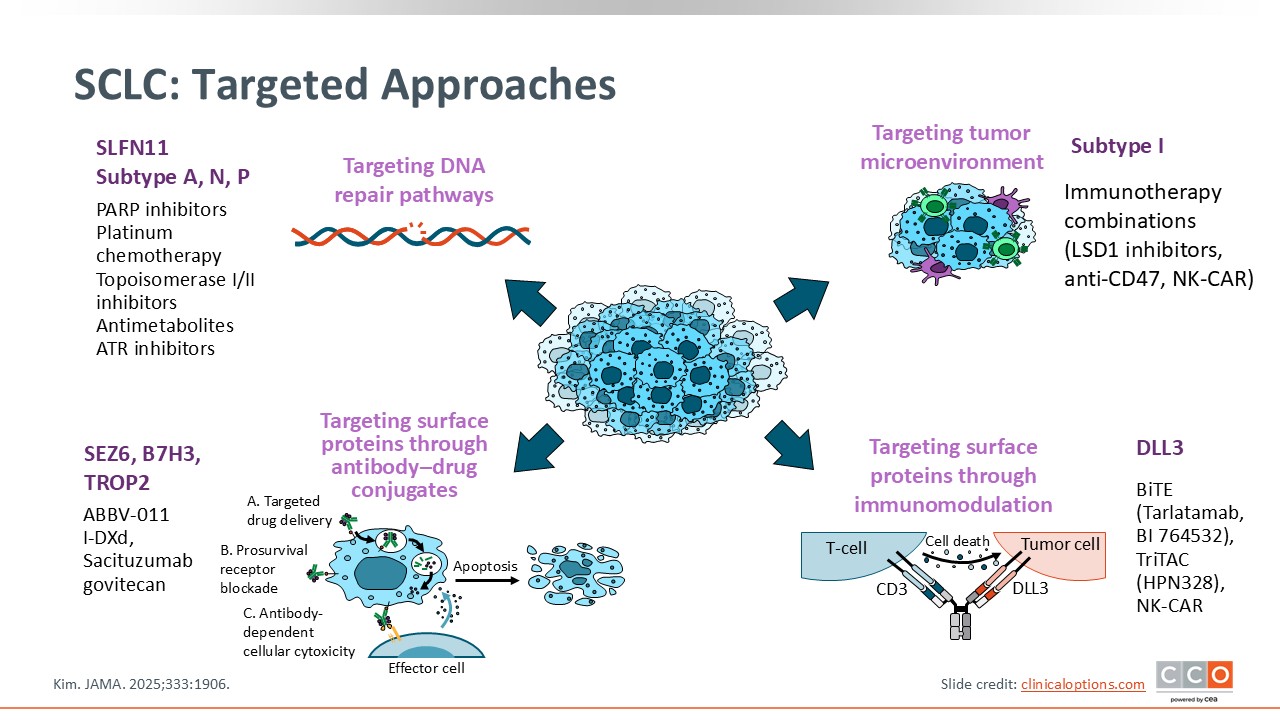
SCLC: Targeted Approaches
Although chemoimmunotherapy is the frontline standard, the treatment landscape for relapsed SCLC has been transformed by non-CT options like lurbinectedin and the DLL3-targeted BiTE tarlatamab. There are also ongoing trials of targeted approaches for SCLC, including in the frontline setting. Though it is not currently standard of care to perform profiling at diagnosis, emerging clinical evidence may help show that targeted frontline approaches can improve patient outcomes.14
Additional information on these targeted treatment options for relapsed ES-SCLC can be found here.
ES-SCLC: Key Points
The treatment landscape for ES-SCLC has evolved significantly with the integration of IO into frontline regimens. Chemoimmunotherapy combining platinum/etoposide with PD-L1 inhibitors such as atezolizumab (IMpower133) or durvalumab (CASPIAN) remains the foundation of care, providing consistent survival benefits and establishing durable responses in a subset of patients. Recently, the IMforte trial further advanced this paradigm by demonstrating a significant OS improvement with the addition of lurbinectedin to atezolizumab maintenance, leading to the newest FDA approval and NCCN recommendation for frontline use. Together, these studies define a new era in which IO-based approaches are central to initial management, with maintenance strategies now incorporating active agents previously reserved for relapsed disease.
Ongoing research continues to refine treatment personalization through transcriptomic subtyping, biomarker exploration, and targeted combinations. Although PD-L1 expression and TMB have not proven predictive of IO benefit, emerging molecular subtypes—particularly the inflammatory (I) subtype—may help identify patients most likely to respond. Novel strategies, including PD-1 inhibitors, bispecific antibodies, ADCs, and biomarker-directed therapies hold the promise of extending survival and improving long-term outcomes. Alongside these advances, supportive care measures such as myelosuppression management and proactive toxicity mitigation remain essential to maintaining treatment continuity and optimizing quality of life.