CE / CME
MASH Matters: New Evidence and Its Application to Gastroenterology and Hepatology Practice
Physician Assistants/Physician Associates: 0.50 AAPA Category 1 CME credit
Nurse Practitioners/Nurses: 0.50 Nursing contact hour
Physicians: maximum of 0.50 AMA PRA Category 1 Credit™
Released: June 23, 2025
Expiration: June 22, 2026
Activity
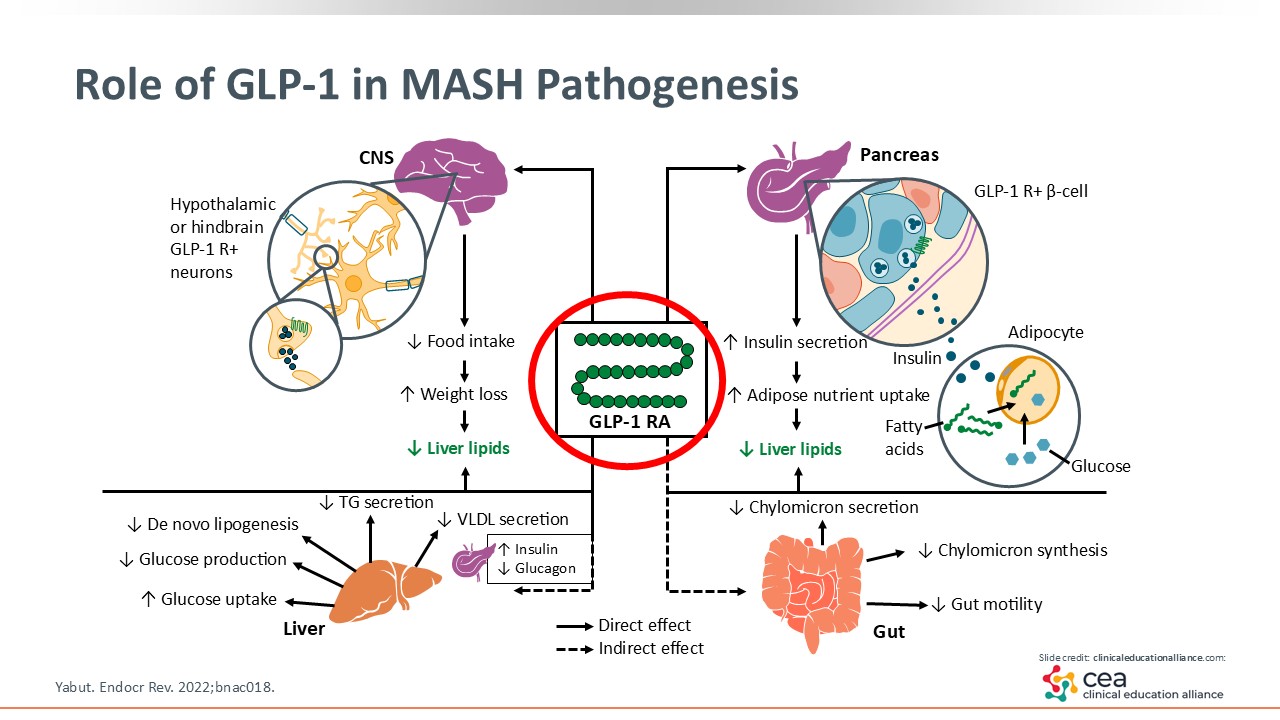
Role of GLP-1 in MASH Pathogenesis
Before getting into the FDA approval of resmetirom, let’s discuss GLP-1. This is now essential in managing T2D and obesity as shown in previous studies, and it is being studied in the treatment of MASLD/MASH.13 GLP-1 RAs lead to weight loss by decreasing food intake via multiple mechanisms, such as having an effect on the central nervous system, improving insulin resistance, and delaying gastric emptying. There are other effects that occur within the liver, including the decrease of de novo lipogenesis and glucose production as well as more efficiency in dealing with lipid overburden. GLP-1 RAs are essential in managing metabolic syndrome.14
ESSENCE: Semaglutide in Patients With MASH
Now let us look at an important study that was recently published. The phase III ESSENCE trial evaluated semaglutide 2.4 mg weekly vs placebo. The trial enrolled patients with MASH and fibrosis stage 2 or 3. They were randomized 2:1, with 533 patients in the semaglutide arm and 267 patients in the placebo arm. The interim analysis that was published evaluated the first in 800 patients at 72 weeks of treatment.15
Of note, MASH studies often look at histology as a surrogate endpoint to get accelerated approval (or Subpart H), which is a temporary approval granted by the FDA until the data related to the study’s outcomes are finalized and regular approval is granted or denied. That is how resmetirom was granted accelerated approval.12,16
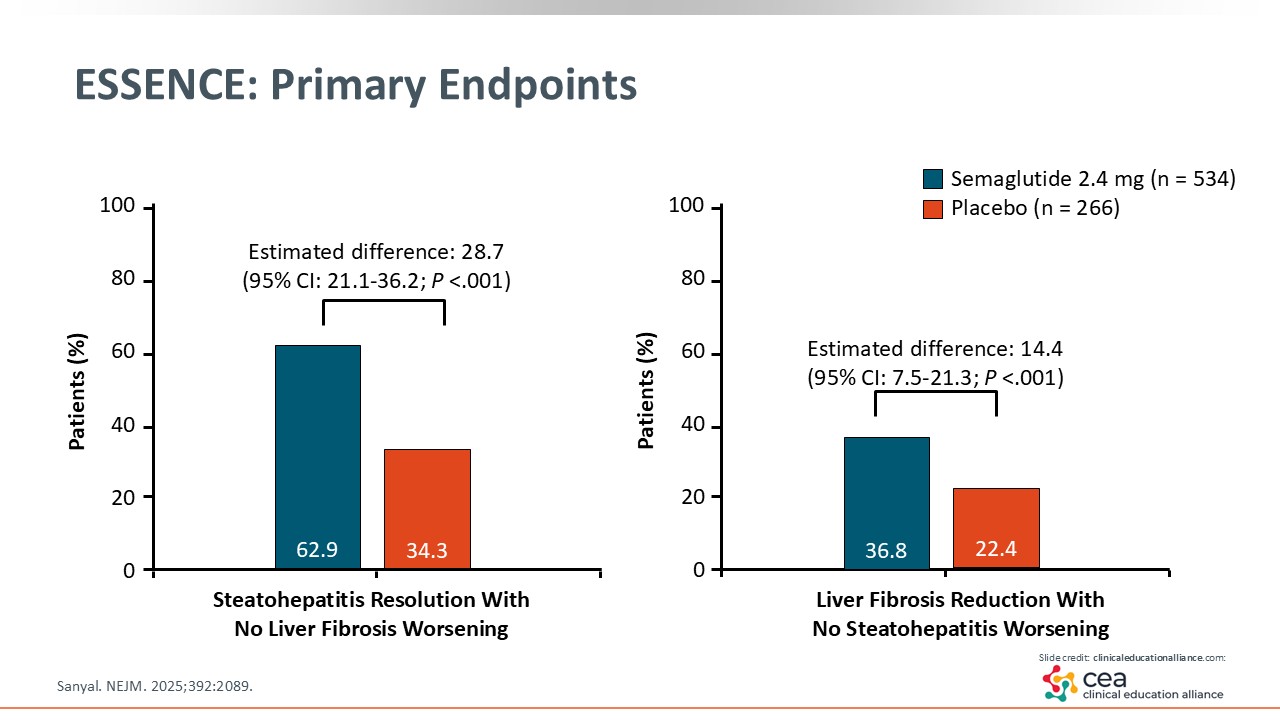
ESSENCE: Primary Endpoints
MASH resolution is defined as steatohepatitis resolution, which is the change in the score of inflammation, steatosis, and ballooning. The ESSENCE trial showed that a statistically significant number of patients achieved MASH resolution with semaglutide vs placebo. The estimated difference was almost 30%. This is an important endpoint that the FDA looks for and that is enough to submit for accelerated approval. The other endpoint illustrated on this slide is important to hepatology: liver fibrosis improvement by stage. That was met in almost 15% of patients in the semaglutide arm compared with those in the placebo arm, which was statistically significant. Because semaglutide achieved both primary endpoints in the ESSENCE trial, I am anticipating its approval soon.15
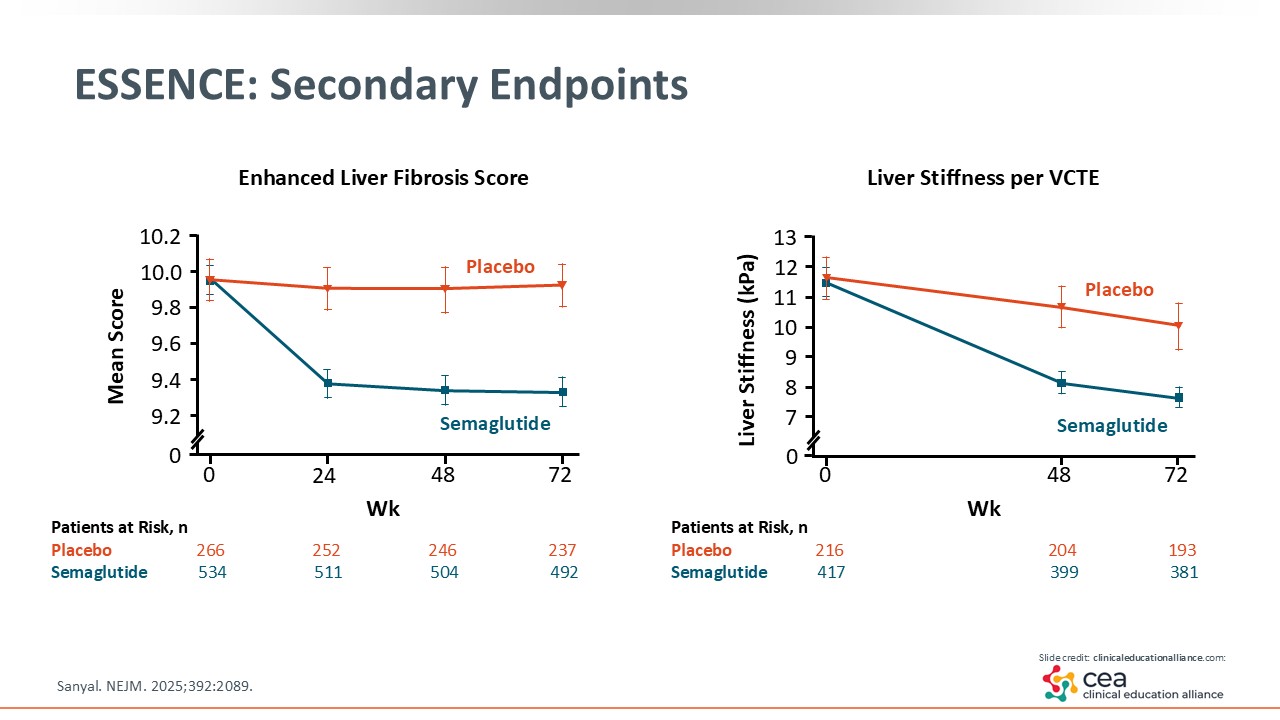
ESSENCE: Secondary Endpoints
Other noninvasive testing are key in evaluating MASH. The Enhanced Liver Fibrosis score shown on the left side of the slide is a blood test. As shown, patients treated with semaglutide saw an increased reduction in this fibrosis biomarker compared with placebo. On the right side is LSM measured by VCTE. Here, too, semaglutide had an increased reduction in liver stiffness vs placebo.15
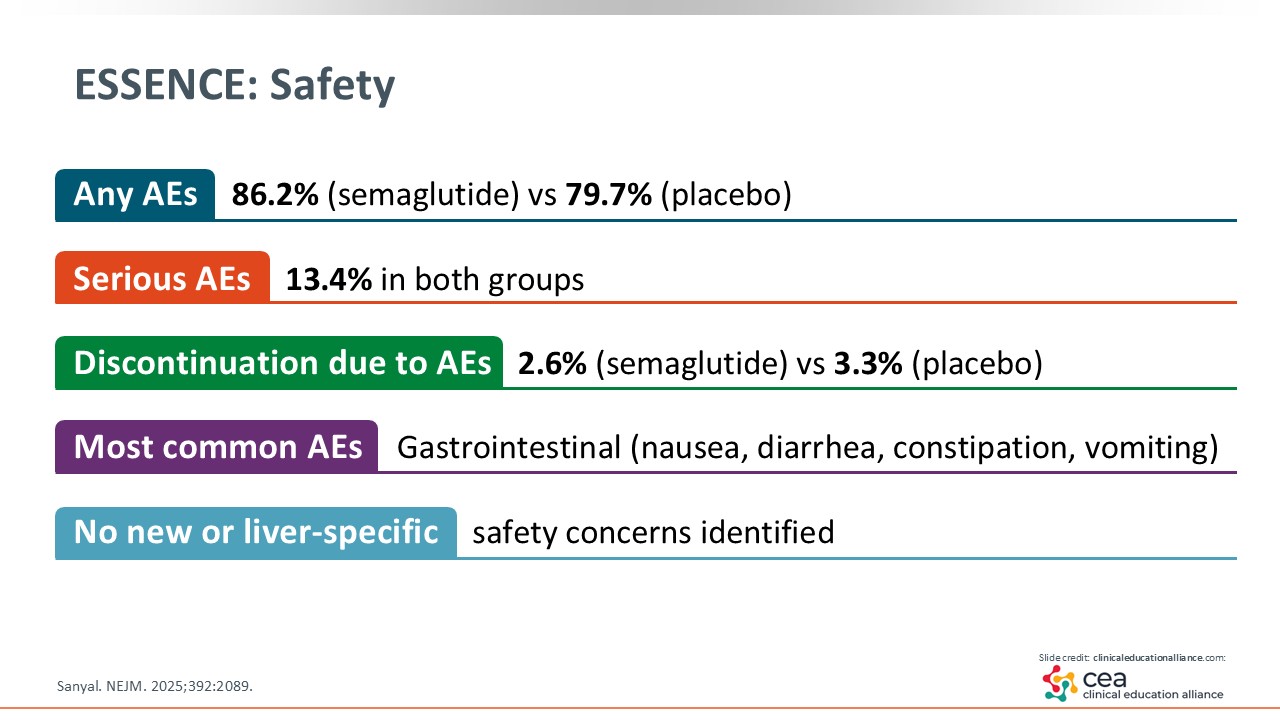
ESSENCE: Safety
In terms of safety, the ESSENCE trial reported adverse events (AEs) in 86% of patients in the semaglutide arm compared with 80% in the placebo arm. Serious AEs in both groups were reported in 13% of patients. Discontinuation rates because of AEs were low, and the most common AEs were gastrointestinal-related: nausea, diarrhea, and constipation. There were no new safety concerns identified.15
ESSENCE: Authors’ Conclusions
In conclusion, the ESSENCE trial found that semaglutide 2.4 mg weekly demonstrated significant histologic improvement in both MASH resolution and fibrosis improvement in patients with MASH and F2 and F3 fibrosis (also known as those with at-risk MASH). Semaglutide also improved cardiometabolic risk factors common in patients with MASH including obesity and T2D. The safety profile was acceptable and consistent with previous reports. These findings support semaglutide as a promising therapeutic option for patients with MASH and moderate to advanced fibrosis.15
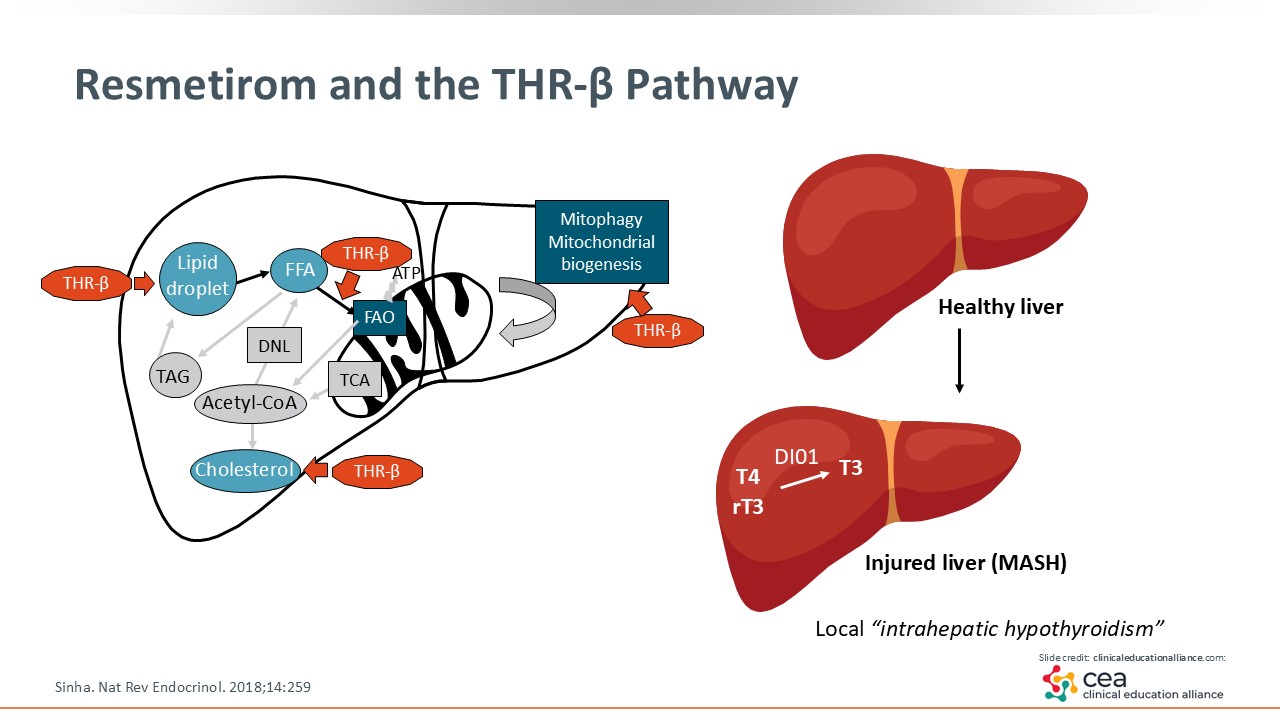
Resmetirom and the THR-β Pathway
As mentioned, resmetirom was approved in conjunction with diet and exercise to treat adults with noncirrhotic MASH with F2/F3 fibrosis. It is a THR-β agonist, and targets the thyroid receptors within the liver. It regulates lipid metabolism like de novo lipogenesis, synthesis of fat in the liver, and mitochondrial biogenesis, where it increases β-fatty oxidation.12,17
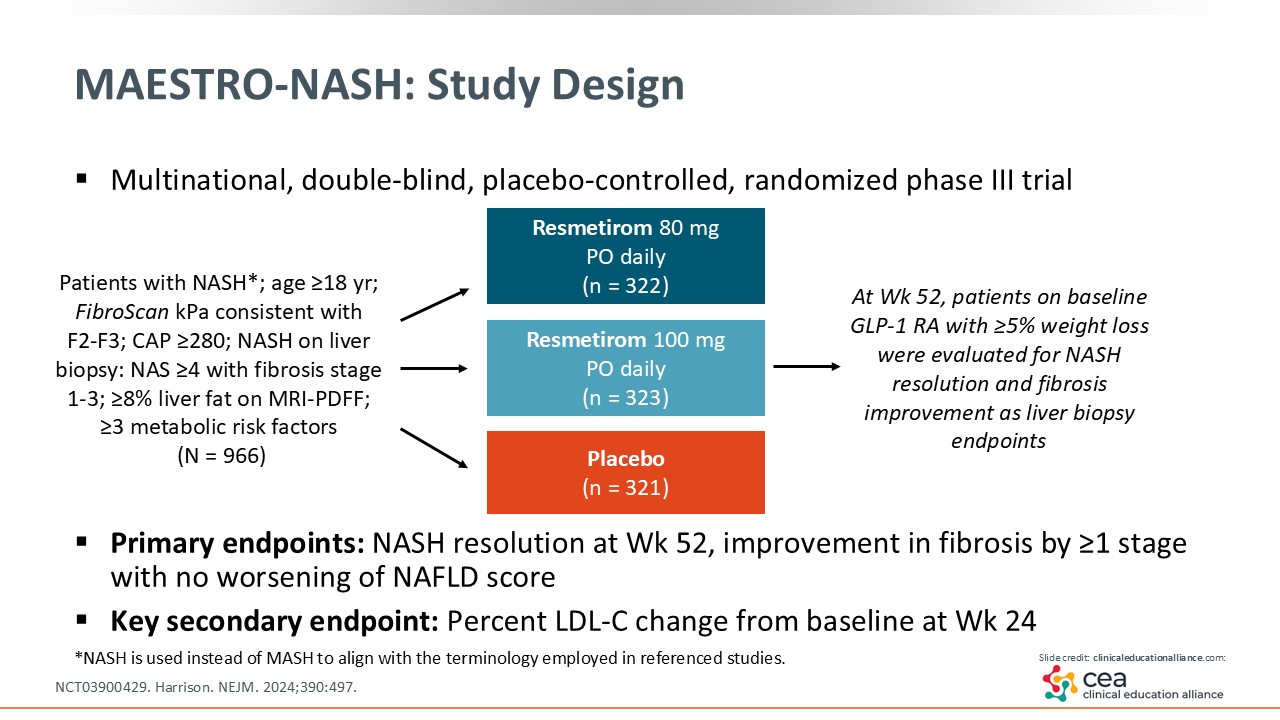
MAESTRO-NASH: Study Design
Resmetirom was studied in the phase III MAESTRO-NASH trial, where researchers compared resmetirom 80-100 mg with placebo treatment for 52 weeks. The trial enrolled patients with MASH (seen as “NASH” on this slide per previous nomenclature) and stage 1-3 fibrosis. The primary endpoints were MASH resolution and fibrosis improvement by one stage or more.18
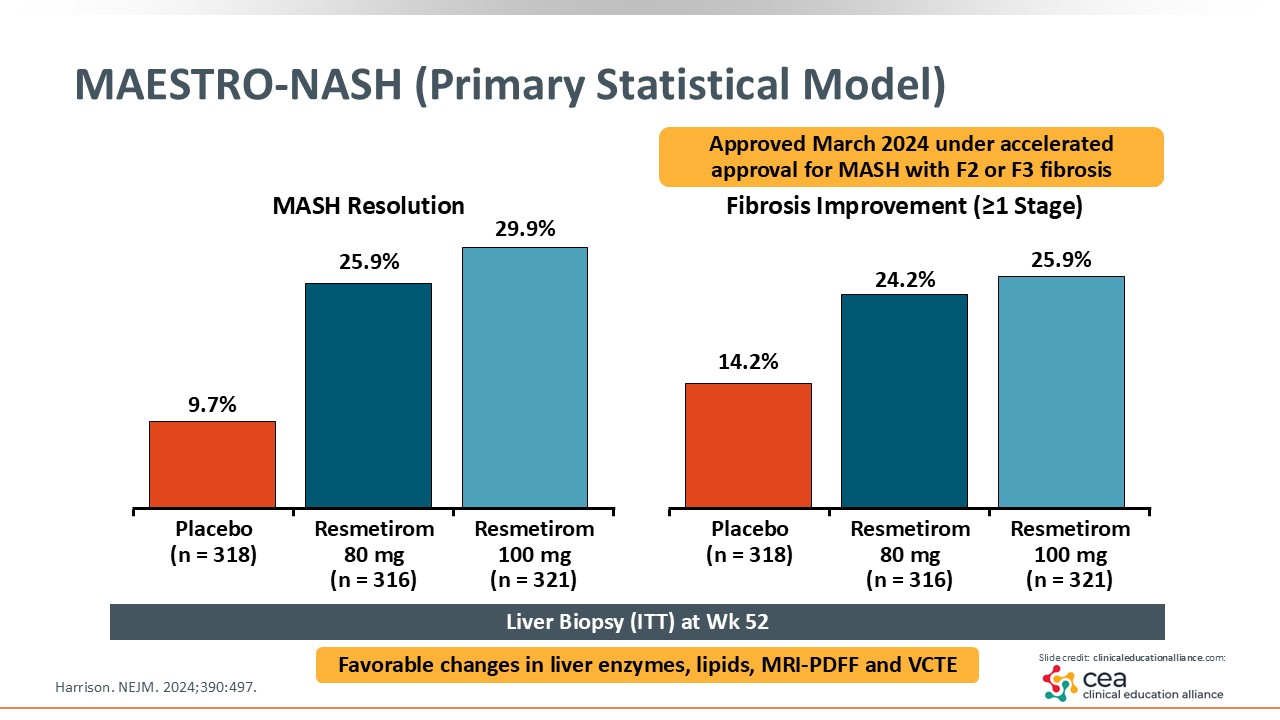
MAESTRO-NASH (Primary Statistical Model)
This slide shows the results. On the left, MASH resolution was achieved in approximately 30% of patients treated with resmetirom compared with 10% with placebo, which was a statistically significant finding. On the right, approximately 26% of patients treated with resmetirom had fibrosis improvement by one stage or more vs 14% with placebo.18 After this study was published, resmetirom was approved by the FDA under accelerated approval.
Phase III Trials for MASH F2-F3 Without Cirrhosis
Semaglutide and resmetirom trials are ongoing because researchers have to prove benefit to receive regular approval for MASH. In addition, there are other agents in the pipeline. The first one is lanifibranor, a pan-PPAR agonist being evaluated in the ongoing phase III NATiV3 trial (NCT04849728). Then there are 3 important injectables in phase III trials: pegozafermin and efruxifermin (FGF21 analogues) and survodutide (a dual GLP-1/glucagon RA) (NCT06318169, NCT06215716, NCT06632457).
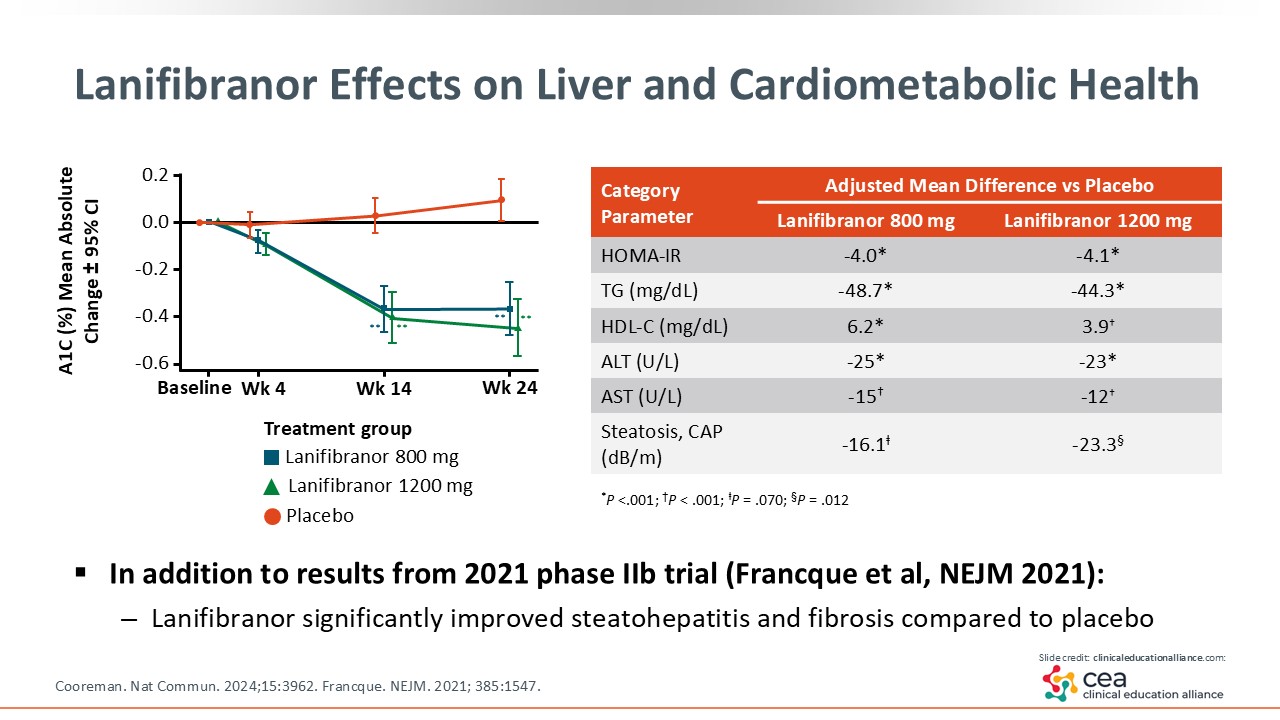
Lanifibranor Effects on Liver and Cardiometabolic Health
The phase IIb NATIVE trial compared lanifibranor with placebo, and it met the endpoints of MASH resolution and fibrosis improvement, so it is now in phase III study (NCT04849728). Analyses also showed significant improvement in metabolic parameters like homeostatic model assessment for insulin resistance, triglycerides, liver enzymes, and steatosis with lanifibranor.19, 20
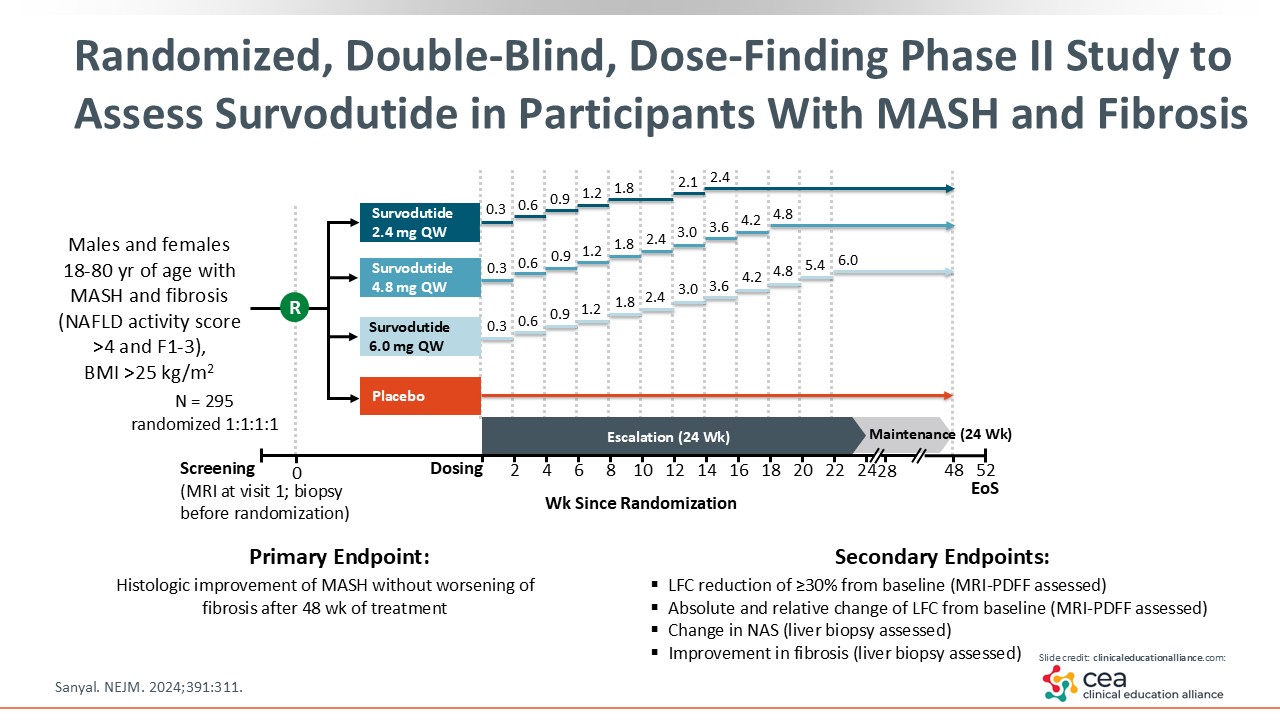
Randomized, Double-Blind, Dose-Finding Phase II Study to Assess Survodutide in Participants With MASH and Fibrosis
This slide shows the design of the dose-finding, double-blind, randomized phase II trial comparing survodutide 2.4, 4.8, and 6.0 mg with placebo. This was a 52-week trial, with up to 24 weeks of titration to overcome any dose-related AEs, that enrolled patients with MASH and fibrosis stage 1-3. In total, 295 patients were enrolled and randomized to each treatment in a 1:1:1:1 ratio.21
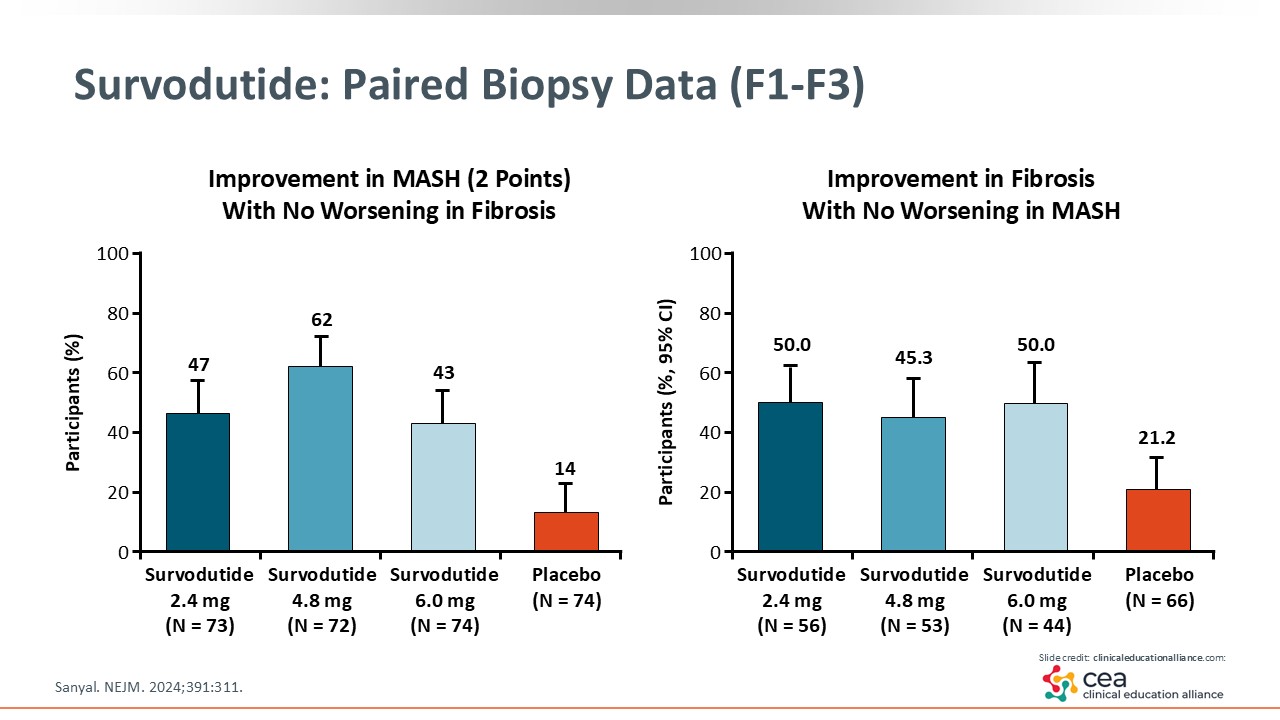
Survodutide: Paired Biopsy Data (F1-F3)
The first graph shows the MASH improvement by 2 points, which is a little bit lower of a bar. Yet all 3 survodutide doses significantly improved MASH by 2 points compared with placebo. The graph on the right illustrates the improvement in fibrosis seen with survodutide vs placebo. Survodutide also led to a greater percentage of patients achieving MASH resolution vs placebo, which is more difficult to achieve in terms of outcomes. The most common AEs were nausea, vomiting, and diarrhea.21 And now survodutide is in phase III development (NCT06632457).
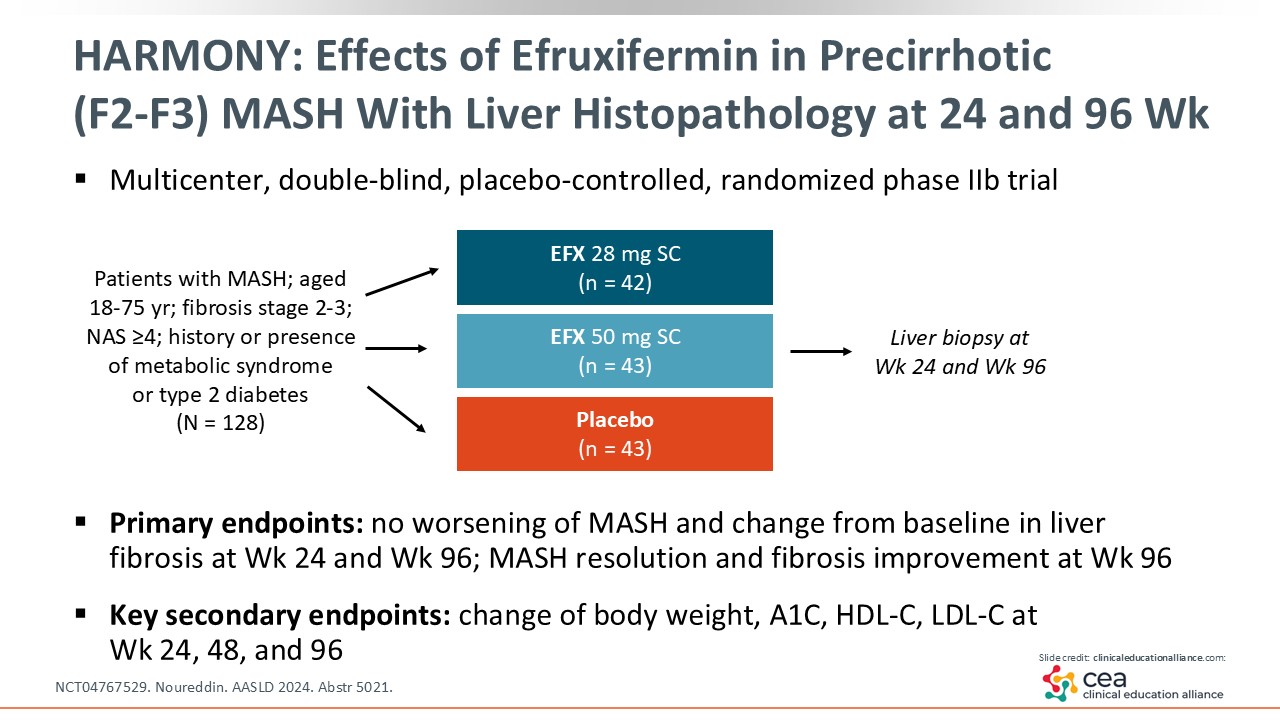
HARMONY: Effects of Efruxifermin in Precirrhotic (F2-F3) MASH With Liver Histopathology at 24 and 96 Weeks
Next I will discuss an FGF21 analogue. Of note, FGF21 plays a role in regulating glucose and lipid metabolism.22 A promising drug that replicates this hormone is called efruxifermin, which was studied in the phase IIb HARMONY trial. This study enrolled patients with MASH and stage 2-3 fibrosis to be treated with efruxifermin 28-50 mg vs placebo. At Weeks 24 and 96, liver biopsies were completed and compared with baseline.23 Let us take a look what happened between baseline and Week 24, and then what happened at Week 96.
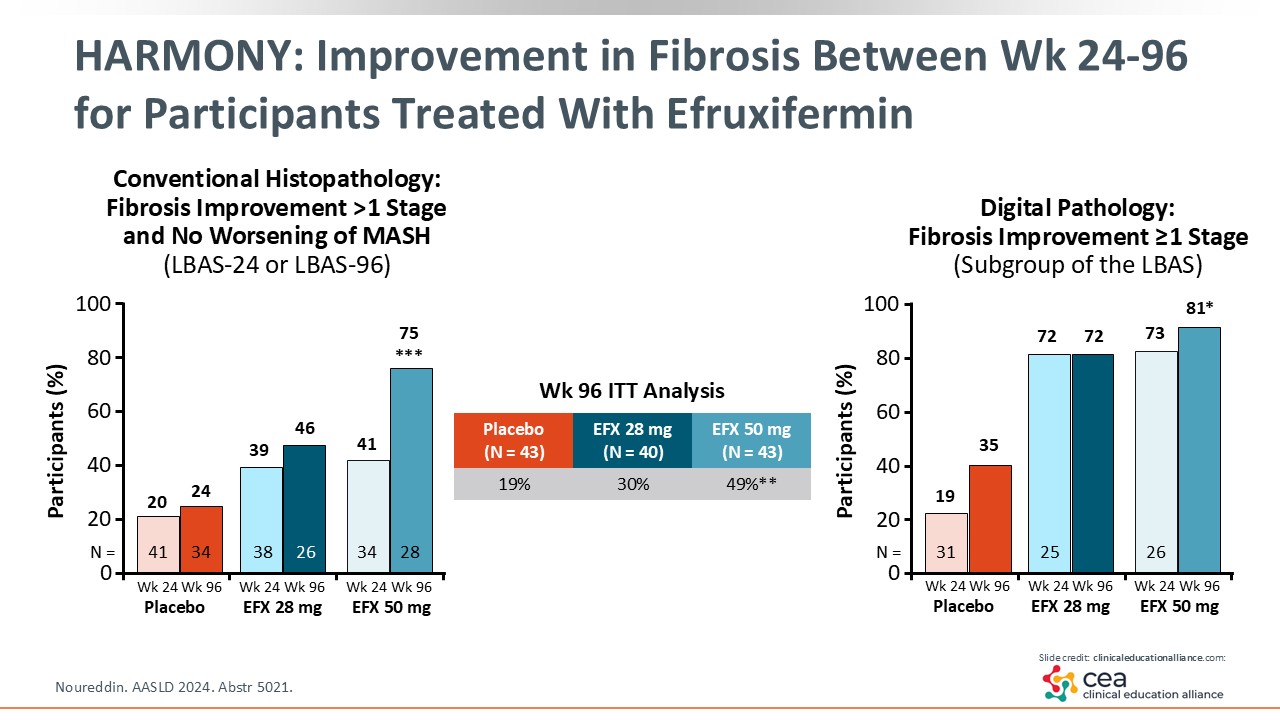
HARMONY: Improvement in Fibrosis Between Week 24 and 96 for Participants Treated With Efruxifermin
At Week 24, there was less of a response compared with Week 96, where almost 75% of patients had improvement in their fibrosis by one stage or more with efruxifermin 50 mg vs placebo. In the intention-to-treat population, almost 50% in the efruxifermin 50 mg arm achieved efficacy at Week 96 compared with 19% of those in the placebo arm. What does that tell us? There is a high bar for fibrosis improvement and the longer patients are treated, the more likely their fibrosis will improve. That is plausible because MASH is a chronic disease that takes a long time to progress; therefore, it needs time to regress, too.
Then on the right, the graph shows the digital pathology with similar results.23 This is a little bit more augmented because of the detailed assessment of fibrosis architecture.
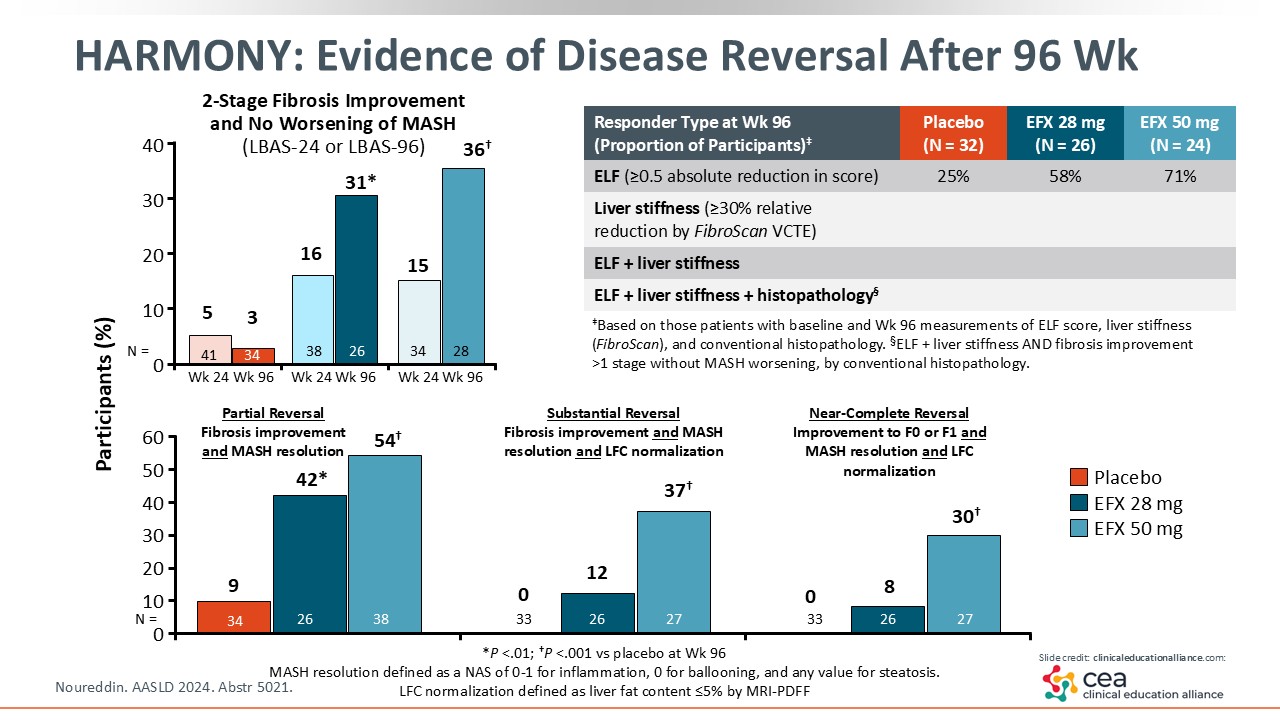
HARMONY: Evidence of Disease Reversal After 96 Weeks
There are other ways to look at this study. The top left graph now shows the 2-point improvement in fibrosis. There was quite a bit of improvement seen with 36% of patients in the efruxifermin arm achieving a 2-point improvement at Week 96. That is a very high bar to achieve. Other histology parameters were looked at and are included on this slide. The bottom graph shows normalization of the liver parenchyma in some patients. Furthermore, some fibrosis biomarkers that are noninvasive improved as well.23
In summary, the HARMONY study showed that treatment with the FGF21 analogue efruxifermin led to both MASH resolution and fibrosis improvement.
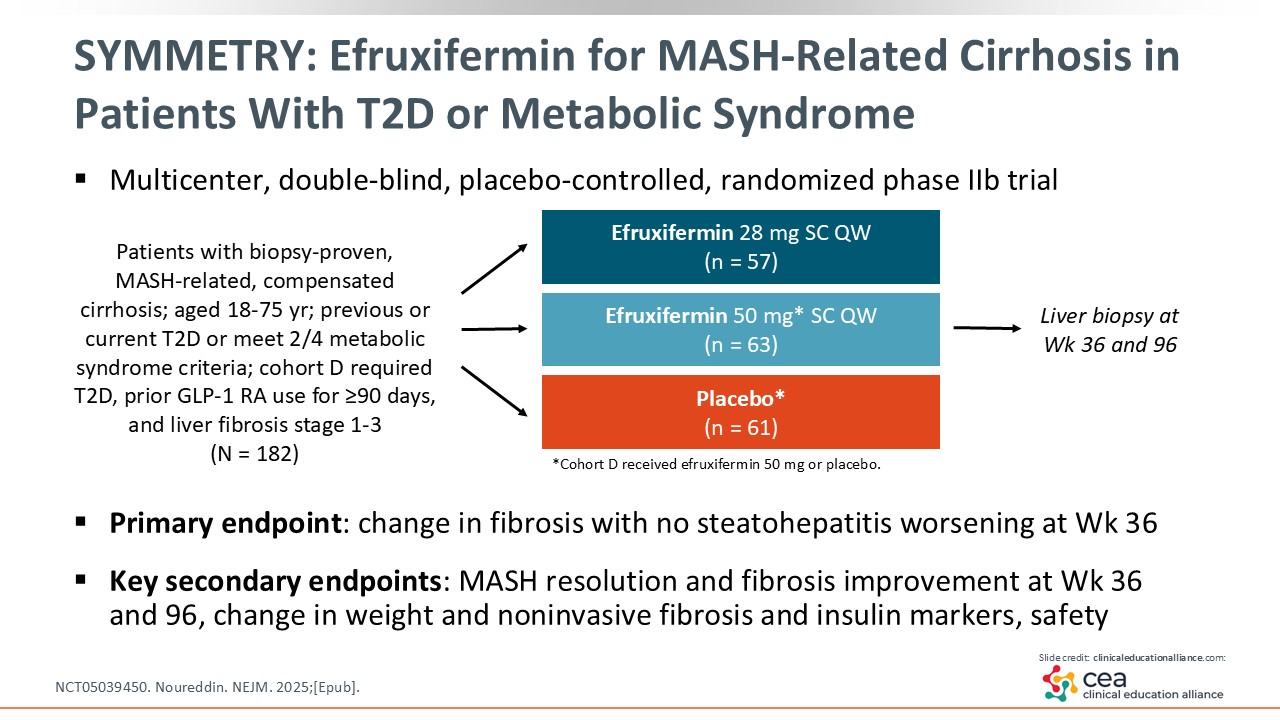
SYMMETRY: Efruxifermin for MASH-Related Cirrhosis in Patients With T2D or Metabolic Syndrome
Efruxifermin was also evaluated in a study called SYMMETRY, which enrolled patients with MASH-related, compensated cirrhosis and T2D or who met 2 of 4 metabolic syndrome criteria. This was a multicenter, double-blinded, placebo-controlled randomized phase IIb trial. Liver biopsy was performed at baseline, Week 36, and Week 96. Efruxifermin 28 and 50 mg were compared with placebo.24
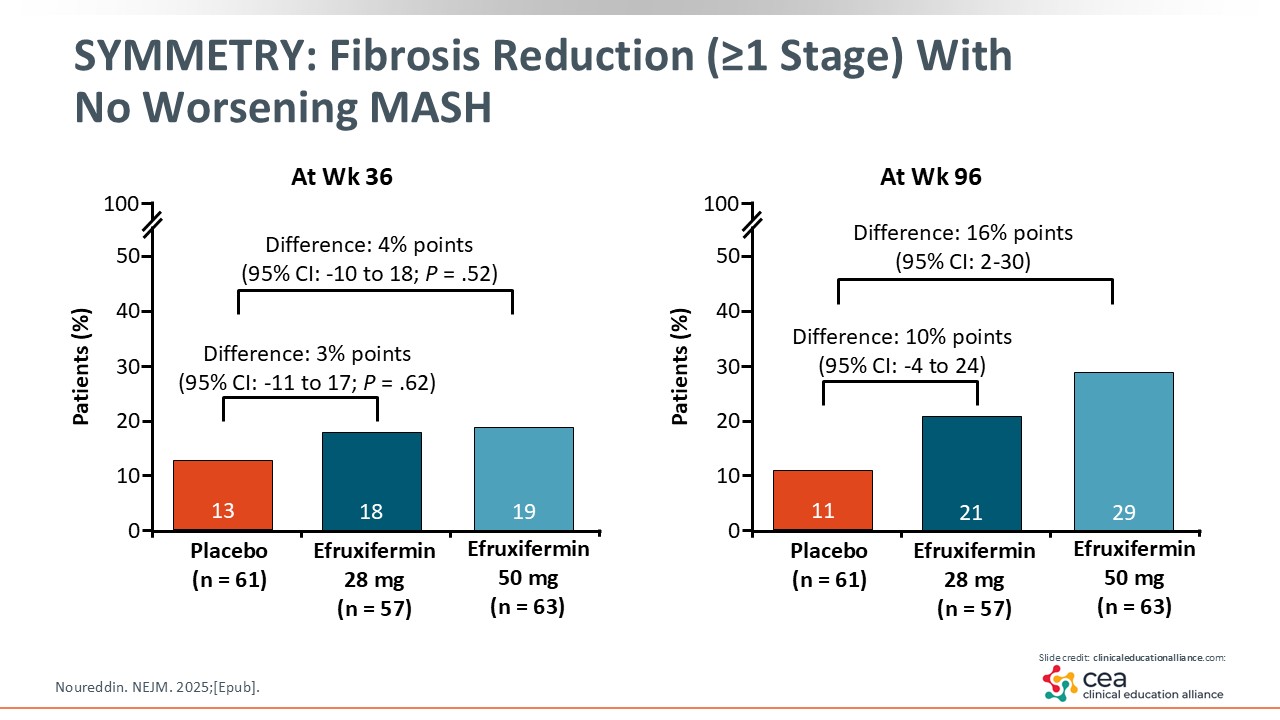
SYMMETRY: Fibrosis Reduction (≥1 Stage) With No Worsening MASH
In looking at the results, efruxifermin did not reach statistical significance in fibrosis improvement at Week 36 compared with placebo. However, at Week 96, efruxifermin 50 mg reached statistical significance for improving fibrosis by at least 1 stage vs placebo.24 This is the first time I have seen that benefit in patients with cirrhosis.
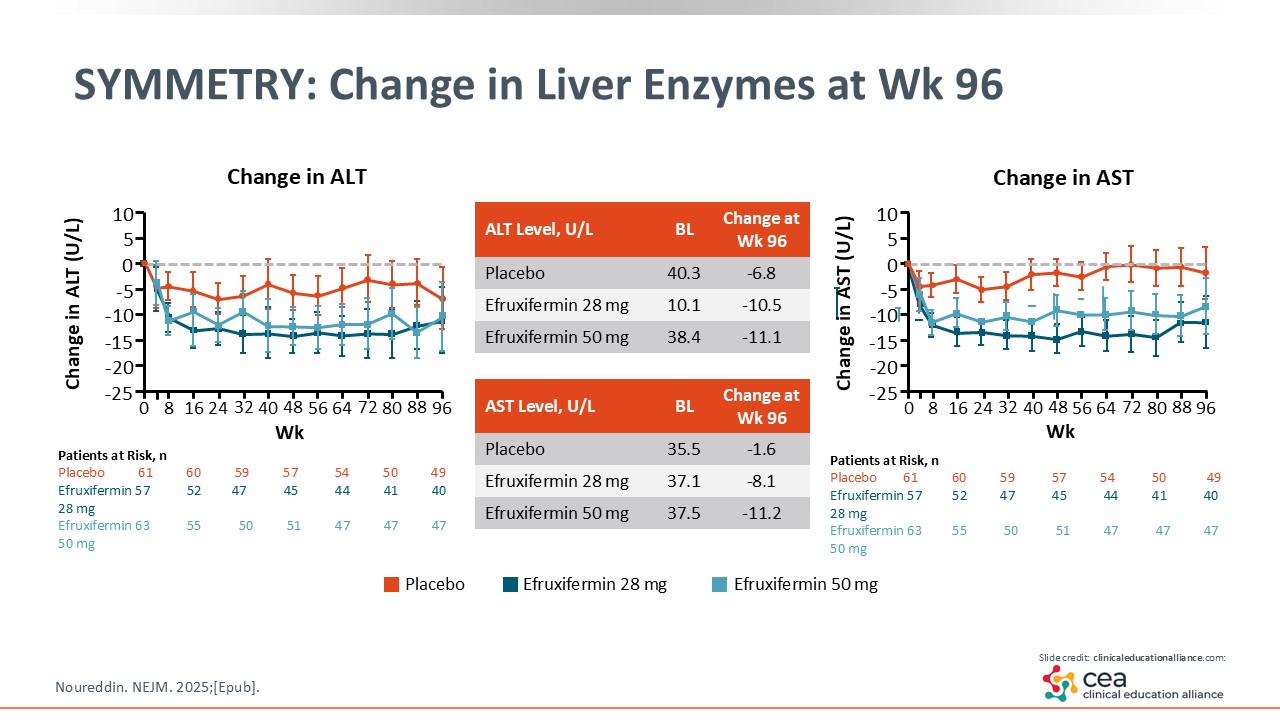
SYMMETRY: Change in Liver Enzymes at Week 96
To increase confidence in efruxifermin, SYMMETRY also looked at changes in biomarkers (ie, liver enzymes, fibrosis) with noninvasive testing. As shown on this slide, efruxifermin improved liver enzymes at Week 96.24
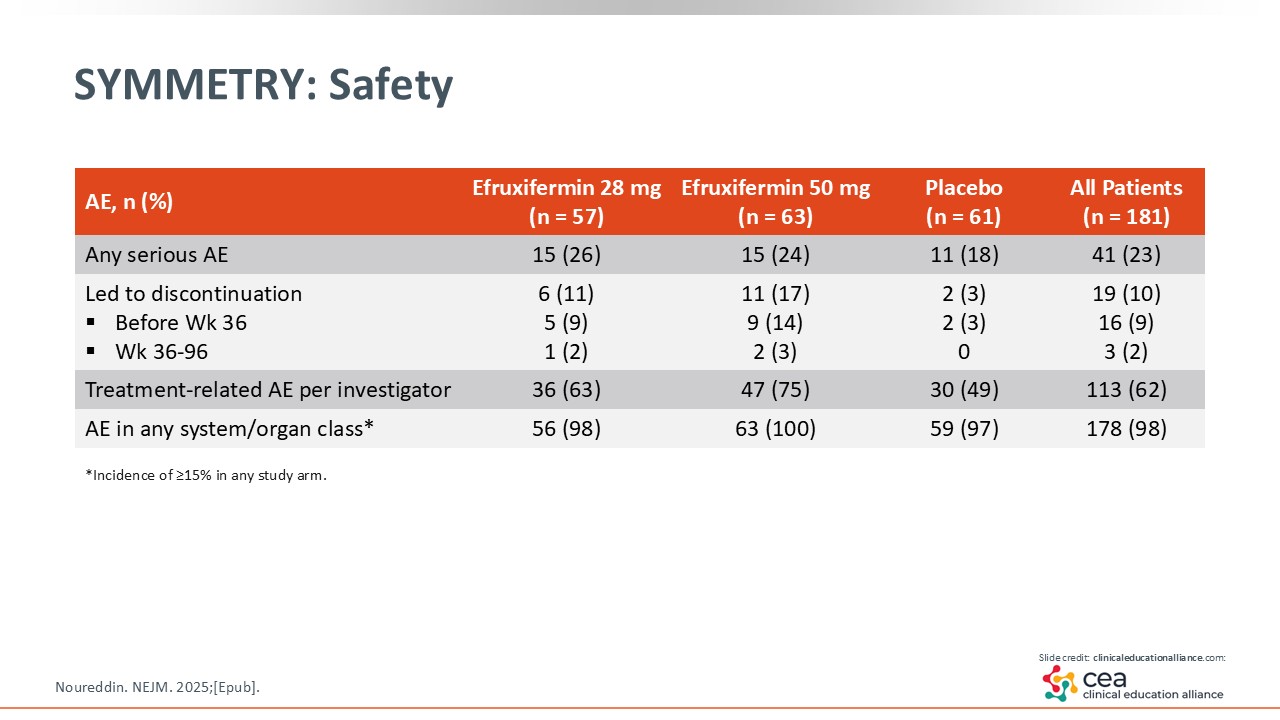
SYMMETRY: Safety
In terms of safety, the reported AEs in this trial were mostly gastrointestinal related. There was slight decrease in bone mineral density with efruxifermin, which is being evaluated in the phase III studies.24
SYMMETRY: Authors’ Conclusions
With the SYMMETRY trial, there was fibrosis improvement by at least one stage at Week 96 in the efruxifermin 50 mg arm vs placebo. Furthermore, the reported AEs were mostly gastrointestinal related and mild to moderate in severity.24
We will see in phase III trials if efruxifermin will improve clinical outcomes like ascites, hepatic encephalopathy, and variceal bleed.
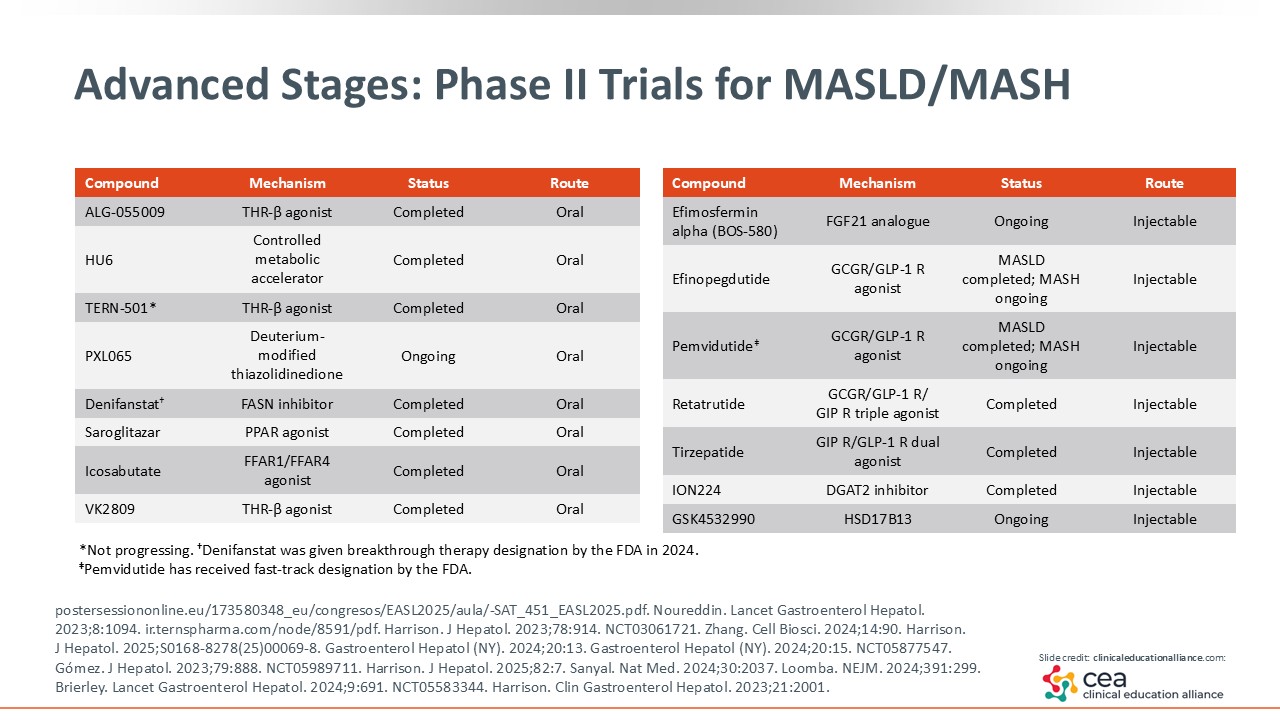
Advanced Stages: Phase II Trials for MASLD/MASH
There are many other studies in phase II. For example, efimosfermin is an FGF21 analogue that finished its phase II trial and is poised to begin phase III.25 Pemvidutide is dual GLP-1/glucagon RA that is undergoing phase II trials in MASH.26 If successful, it should move onto phase III. Finally, tirzepatide is a dual glucose-dependent insulinotropic polypeptide/GLP-1 RA. It had positive phase II data published and might move on to phase III.27
Phase III Trials for MASH Cirrhosis
This slide highlights the various phase III trials underway for patients with MASH-related cirrhosis. As shown with efruxifermin in phase II, we now have the evidence (or the proof of concept) that we can address cirrhosis in patients with MASH. Furthermore, there is a study called MAESTRO-NASH OUTCOMES that is very exciting. It is a phase III trial evaluating efruxifermin 80 mg in patients with MASH-related cirrhosis that is well compensated (NCT05500222). Pegozafermin is being studied in the phase III ENLIGHTEN-Fibrosis trial among patients with MASH and stage 2-3 fibrosis (NCT06318169). Finally, survodutide (the dual GLP-1/glucagon RA) is in 2 phase III studies: one dedicated to patients with MASH moderate/advanced fibrosis and the other to patients with MASH-related cirrhosis (NCT0663244, NCT06632457). It truly is an exciting time for our patients.
Faculty Discussion
When considering the effect these newer agents will likely have on the care of patients with MASH, there was a time when these patients had no options. Indeed, there were many failed trials. But now we have resmetirom as the first accelerated approval and many others in the pipeline that are being tested. The ESSENCE trial was positive, and I expect semaglutide will have an additional indication upon FDA approval. That will give patients even more options. These new therapies also will increase screening and recognition of MASLD/MASH in patients. Hopefully it will allow HCPs to diagnosis patients early enough to give them therapies so they do not progress to cirrhosis.
How can hepatology and gastroenterology HCPs engage their multidisciplinary colleagues to improve the care of patients with MASLD/MASH? I think it is a multidisciplinary approach. In general, patients at risk for MASH or with MASH and stage 2-3 fibrosis need treatment. I see a role for endocrinology to help treat these patients. In addition, it should be a collaboration among gastroenterology, hepatology, endocrinology, and primary care to improve the metabolic management of these patients, whether it be T2D, obesity, or cardiovascular risk. Everyone should be involved to help these patients.
MASH Therapies Conclusions
In conclusion, MASH therapies are really exciting. We finally have the first agent approved for use in MASH: resmetirom. Furthermore, there are multiple drugs in phase II/III trials. Semaglutide showed positive benefit in its phase III ESSENCE trial. There are others like survodutide and pemvidutide that hopefully will enter phase III soon. Efruxifermin also has promising data, and efimosfermin should go into phase III soon. Others include lanifibranor. But despite the limitation in options for patients with MASH-related cirrhosis, we are starting to see light at the end of the tunnel. There is some positivity and hopefully we will have agents to help these patients in the near future.