CME
Research Focus in Advanced NSCLC: Emerging Insights on Next-Generation KRAS G12C Inhibitors
Physicians: Maximum of 0.75 AMA PRA Category 1 Credit™
Released: April 30, 2026
Expiration: October 29, 2026
Activity
Introduction
In this research-focused text module, Luis Paz-Ares, MD, PhD, and Christine Bestvina, MD, review the landscape of KRAS G12C–targeted therapies in non-small-cell lung cancer (NSCLC). They highlight current limitations in clinical care and discuss next-generation KRAS G12C–targeted therapies designed to address the shortcomings of existing treatment standards for KRAS-mutated NSCLC.
The key points discussed in this module are illustrated with thumbnails from the accompanying downloadable PowerPoint slideset, which can be found here or downloaded by clicking on any of the slide thumbnails in the module alongside the expert commentary.
Before continuing with this educational activity, please take a moment to answer the following questions.
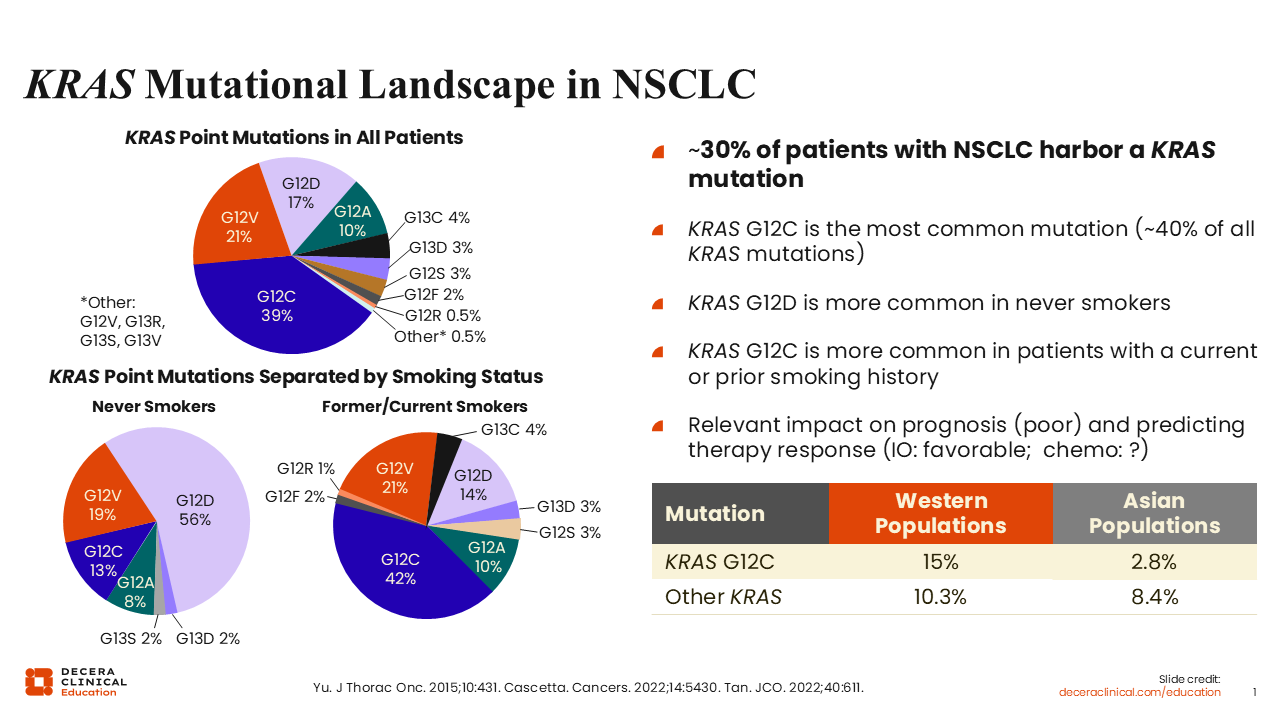
KRAS Mutational Landscape in NSCLC
Luis Paz-Ares, MD, PhD:
KRAS is one of the most frequently mutated oncogenes in solid tumors. It is present in >90% of pancreatic cancers and in approximately 30% of non-small-cell lung cancer (NSCLC).4 These mutations involve several allelic variants, including G12C, G12D, and others. KRAS G12C accounts for approximately 40% to 45% of KRAS mutations in NSCLC and is most commonly observed in patients with a history of smoking.
By contrast, among never-smokers, KRAS G12D is the most frequent mutation.2 These distinctions are clinically relevant because KRAS mutations activate multiple signaling pathways involved in key cellular processes. These include proliferation pathways such as BRAF, MEK, and ERK, as well as survival pathways mediated by PI3K. KRAS signaling also influences cytoskeletal organization, invasion, and other tumor-promoting cellular functions.5
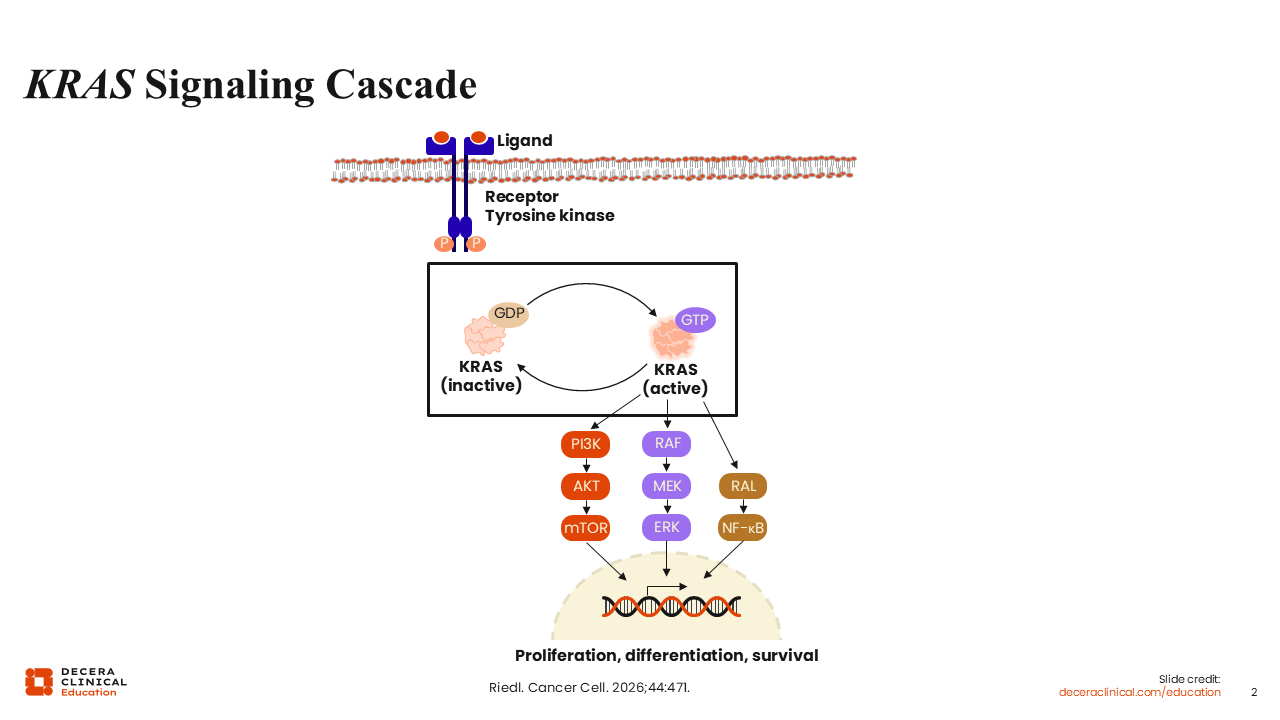
KRAS Signaling Cascade
Luis Paz-Ares, MD, PhD:
The development of KRAS-targeted therapies was enabled by structural insights obtained more than a decade ago. Resolution of the crystallographic structure of mutant KRAS led to the identification of the switch II pocket, which contains the cysteine residue characteristic of the G12C mutation. This structural feature allows for covalent binding of specific small-molecule inhibitors.5
KRAS G12C (OFF) inhibitors were designed to bind selectively to the inactive, GDP-bound form of KRAS. Agents such as sotorasib and adagrasib stabilize KRAS in this inactive conformation, thereby preventing signaling through downstream pathways. When KRAS remains in the GTP-bound active state, it continues to drive oncogenic signaling; therefore, trapping KRAS in the GDP-bound state is critical for therapeutic activity.2,3
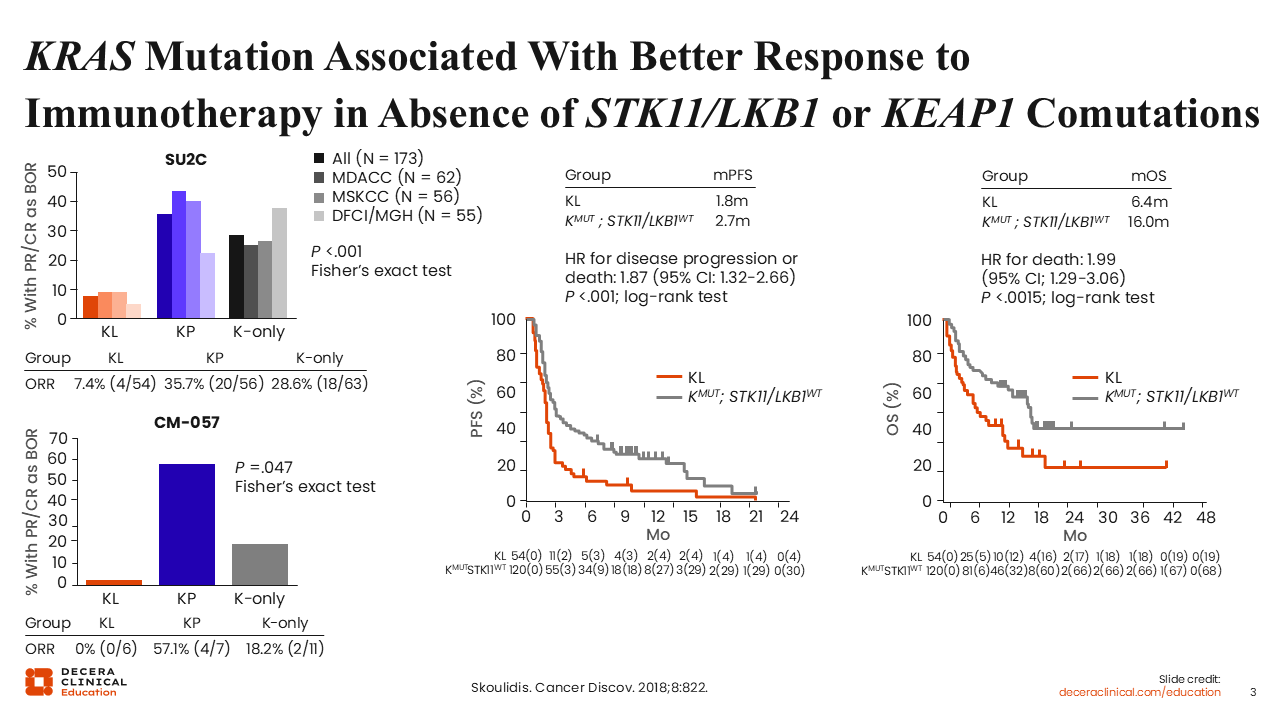
KRAS Mutation Associated With Better Response to Immunotherapy in Absence of STK11/LKB1 or KEAP1 Comutations
Luis Paz-Ares, MD, PhD:
KRAS G12C mutations are associated with poorer prognosis compared with KRAS wild-type disease, both in early-stage and advanced NSCLC. These tumors also tend to have suboptimal outcomes with chemotherapy.4
The benefit of immunotherapy varies across patient subsets. Improved responses have been observed in patients with co-occurring KRAS and TP53 mutations. By contrast, patients with concurrent KRAS or STK11/LKB1 or KEAP1 mutations tend to have inferior outcomes with immunotherapy.6,7
An analysis of multiple independent retrospective cohorts demonstrated that STK11/LKB1 inactivation is a major driver of immune escape and primary resistance to PD-1 blockade in KRAS-mutant lung adenocarcinoma.8
Similarly, KRAS and KEAP1 comutations are associated with inferior outcomes, including progression-free survival (PFS) (HR: 2.05; P <.0001) and overall survival (OS) (HR: 2.24; P <.0001).7 Of note, the adverse prognostic impact of STK11/LKB1 and KEAP1 alterations appears to be specific to tumors harboring KRAS mutations and is not observed in KRAS wild-type disease.
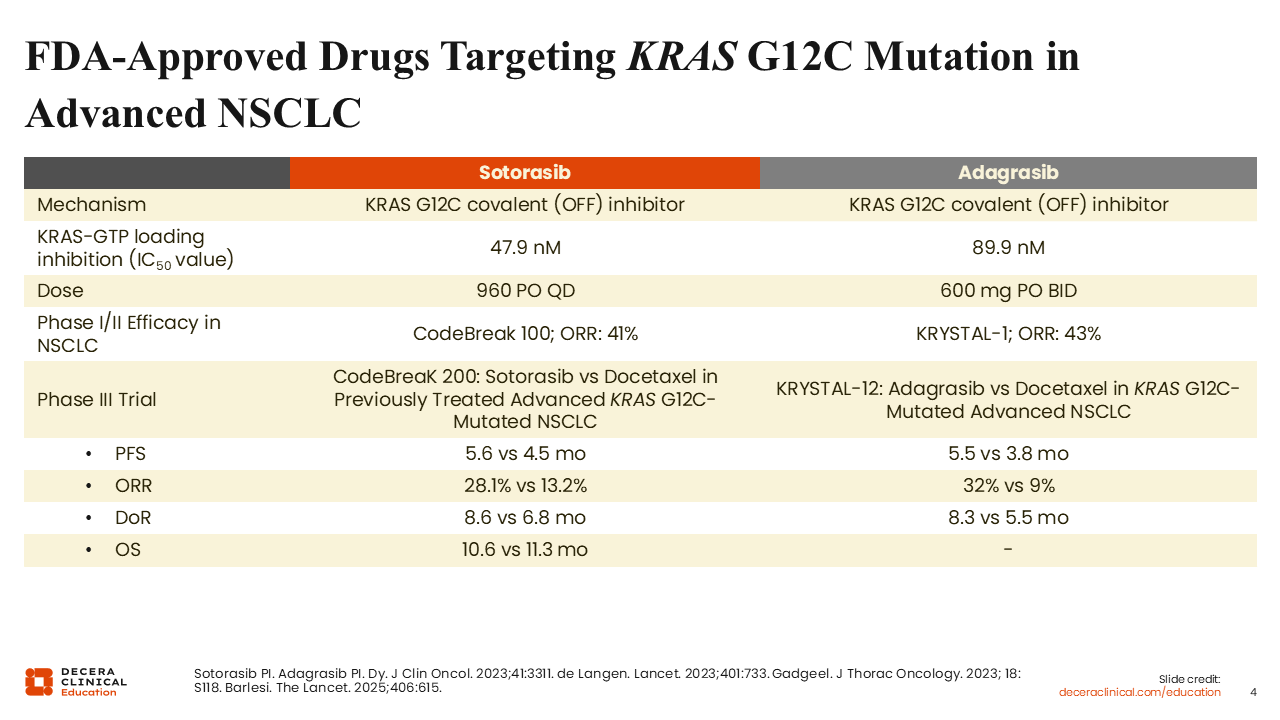
FDA-Approved Drugs Targeting KRAS G12C Mutation in Advanced NSCLC
Luis Paz-Ares, MD, PhD:
The identification of the switch II pocket in KRAS G12C enabled the development of targeted therapies after decades of unsuccessful attempts to directly inhibit KRAS. This structural insight revealed a cysteine residue amenable to covalent binding, forming the basis for KRAS G12C (OFF) inhibitors.2,3
Christine Bestvina, MD:
Currently, 2 KRAS G12C–targeted therapies, sotorasib and adagrasib, are approved by the FDA as single agents for the treatment of locally advanced or metastatic KRAS G12C–mutated NSCLC in patients who have received at least 1 prior systemic therapy.9,10
Early-phase clinical trials demonstrated meaningful activity for both agents. In the phase I CodeBreaK-100 trial of sotorasib and the phase I KRYSTAL-1 trial of adagrasib, objective response rates (ORRs) were 41% and 43%, respectively.11,12
Both agents were subsequently evaluated in randomized phase III trials comparing them with docetaxel (CodeBreaK-200 and KRYSTAL-12). In these studies, most patients had previously received both chemotherapy and immunotherapy. Both sotorasib and adagrasib improved median PFS compared with docetaxel, with median PFS values of 5.6 months and 5.5 months, respectively. However, no improvement in OS was observed with sotorasib, and OS data for adagrasib remain immature.13,14
Safety and Tolerability of Sotorasib and Adagrasib in Advanced NSCLC
Christine Bestvina, MD:
Both sotorasib and adagrasib have demonstrated manageable safety profiles in clinical trials. In phase III studies, grade 3/4 treatment-related adverse events (AEs) occurred in 33% of patients receiving sotorasib and 47% of those receiving adagrasib.
Of importance, relatively few high-grade AEs led to treatment discontinuation, occurring in approximately 10% of patients treated with sotorasib and 8% of those treated with adagrasib. These findings indicate that most patients are able to remain on therapy for a clinically meaningful duration.13,14
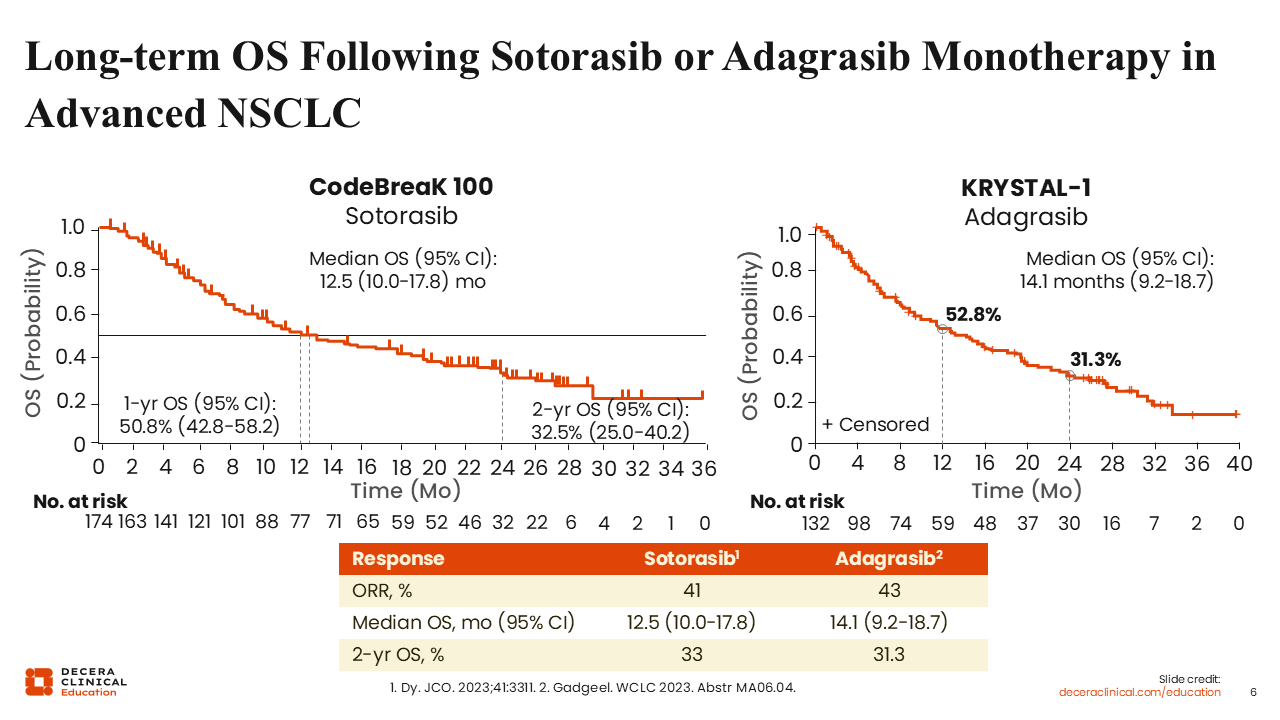
Long-term OS Following Sotorasib or Adagrasib Monotherapy in Advanced NSCLC
Christine Bestvina, MD:
Although these therapies represent an important advance, the magnitude and durability of benefit remain limited. Although improvements in PFS have been demonstrated, these have not yet translated into significant OS benefits.
Long-term follow-up from the phase I CodeBreaK-100 and KRYSTAL-1 trials showed 2-year OS rates of 33% for sotorasib and 31% for adagrasib.13,14 These findings highlight the need for improved therapeutic strategies to enhance the durability of response and long-term outcomes in this patient population.
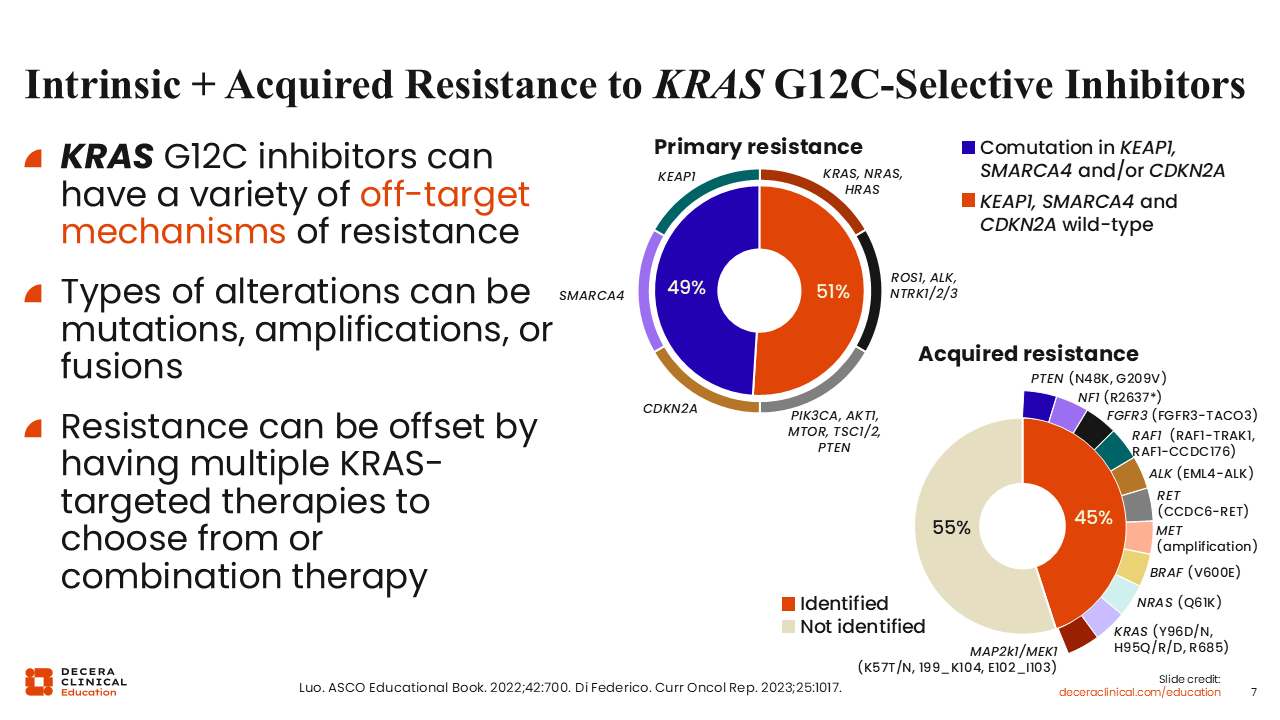
Intrinsic Plus Acquired Resistance to KRAS G12C–Selective Inhibitors
Christine Bestvina, MD:
As KRAS G12C inhibitors have been incorporated into clinical practice, there have been significant efforts to better understand mechanisms of resistance.
Broadly, primary (intrinsic) resistance and acquired resistance describe 2 distinct failure patterns with different biological underpinnings. Primary resistance is defined as a lack of meaningful clinical response to a targeted therapy from the outset, despite the presence of the presumed targetable alteration.
Acquired resistance is defined as disease progression after an initial period of clinical benefit (response or stable disease) on targeted therapy.
Understanding these mechanisms is critical as combination strategies are developed, particularly to determine whether resistance patterns differ between monotherapy and combination approaches.
Mechanisms of Resistance to KRAS G12C Inhibitors
Christine Bestvina, MD:
These resistance mechanisms can be broadly categorized into 3 groups.
First, acquired alterations within the KRAS gene itself may emerge, leading to reduced drug binding or reactivation of signaling. Second, alterations in RTK-RAS/MAPK or PI3K pathways can serve as bypass mechanisms, restoring downstream signaling despite KRAS inhibition. Third, acquired gene fusions may also contribute to resistance.15,16
Luis Paz-Ares, MD, PhD:
The relatively short duration of response observed with first-generation KRAS G12C inhibitors has prompted the development of strategies to overcome resistance. These strategies can be broadly divided into horizontal and vertical approaches.
Horizontal strategies target signaling pathways that operate in parallel with KRAS signaling. These include upstream regulators such as SHP2, SOS1, and EGFR, as well as downstream effectors such as MEK. Among these, combinations with EGFR inhibitors and SHP2 inhibitors have shown particular promise in early studies, especially in certain tumor types.17
Vertical strategies target parallel or downstream signaling pathways that may compensate for KRAS inhibition. These include inhibition of mTOR, CDK4/6, and WEE1. To date, these approaches have shown limited efficacy.18
Combinations with chemotherapy have demonstrated potential synergy in preclinical models, particularly with taxane-based regimens. In addition, combinations with immunotherapy are of interest, as KRAS inhibition has been shown to modulate the tumor microenvironment. Specifically, KRAS inhibition may reduce immunosuppressive myeloid cells, increase infiltration of activated T-cells, and enhance the production of proinflammatory cytokines, thereby increasing tumor immunogenicity.16
Combination Strategies With First-Generation KRAS G12C–Selective Inhibitors
Luis Paz-Ares, MD, PhD:
These observations provide a strong rationale for combination strategies involving KRAS G12C inhibitors. The goal is not only to improve response rates but also to enhance the durability of response.
Several approaches are under investigation, including combinations with chemotherapy and immunotherapy.
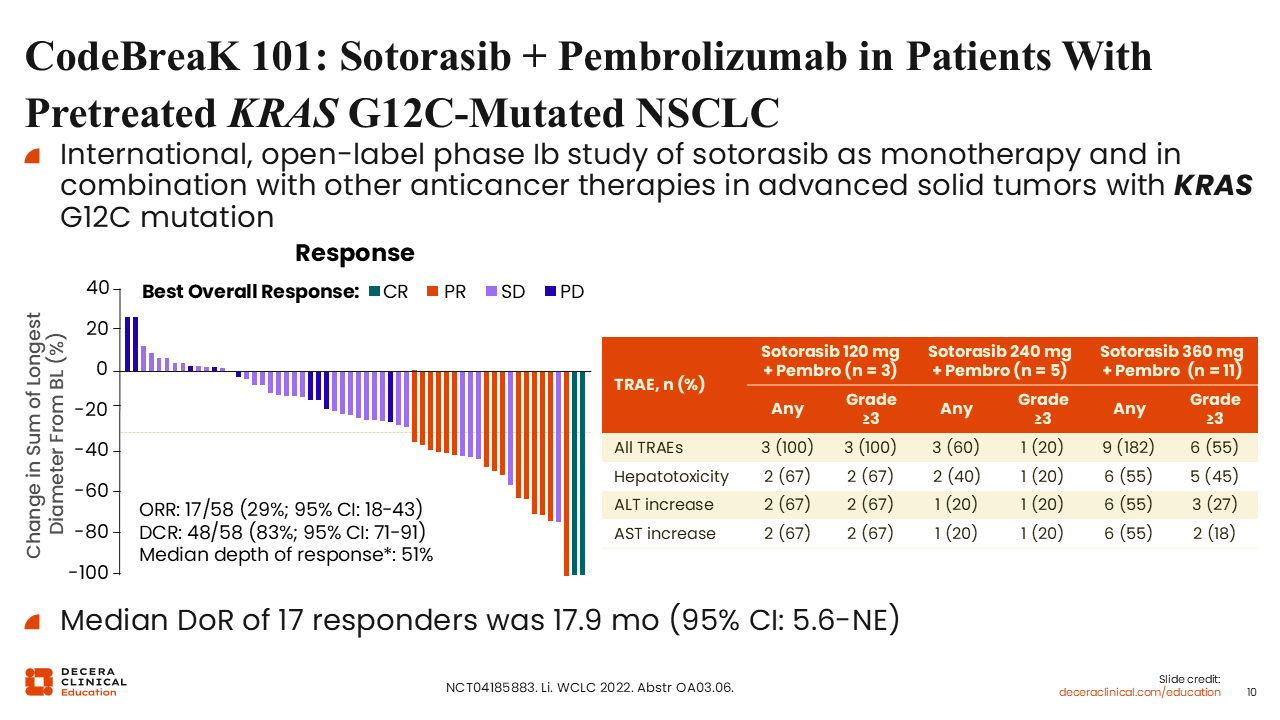
CodeBreaK 101: Sotorasib Plus Pembrolizumab in Patients With Pretreated KRAS G12C–Mutated NSCLC
Luis Paz-Ares, MD, PhD:
CodeBreaK 101 is an international, open-label phase Ib study evaluating sotorasib as monotherapy and in combination with other anticancer therapies in patients with advanced solid tumors harboring KRAS G12C mutations.
In the cohort evaluating sotorasib in combination with pembrolizumab, patients received a lead-in dose of sotorasib followed by the addition of pembrolizumab. This combination demonstrated an ORR of 29% and a median duration of response of 17.9 months.
However, the incidence of grade 3/4 hepatotoxicity was significant and considered unacceptable, leading to discontinuation of further development of this combination. These findings highlight the balance between potential improvements in efficacy with combination therapy and the increased risk of treatment-related AEs.19
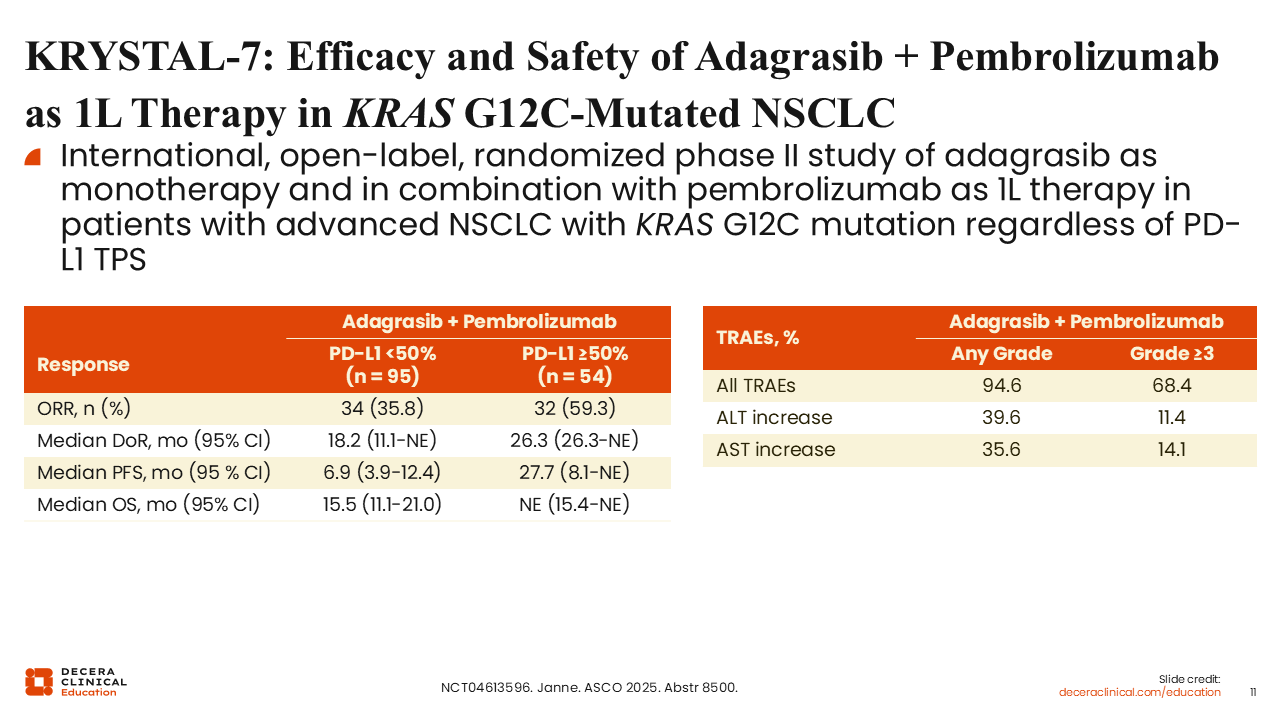
KRYSTAL-7: Efficacy and Safety of Adagrasib Plus Pembrolizumab as First-line Therapy in KRAS G12C–Mutated NSCLC
Luis Paz-Ares, MD, PhD:
KRYSTAL-7 is an international, open-label, randomized phase II study evaluating adagrasib as monotherapy and in combination with pembrolizumab as first-line therapy in patients with advanced NSCLC harboring KRAS G12C mutations, regardless of PD-L1 tumor proportion score (TPS).
In early results from this study, the combination of adagrasib plus pembrolizumab demonstrated promising activity. In untreated patients, the ORR was approximately 44%, with a median duration of response of 26.3 months. Among patients with PD-L1 TPS <50%, the ORR was 35.8%, with a median duration of response of 18.2 months. In those with PD-L1 TPS ≥50%, the ORR was 59.3%, with a median duration of response of 26.3 months.
The safety profile was manageable, with fewer than 20% of patients requiring treatment discontinuation because of immune-related AEs, including hepatotoxicity.20
Ongoing phase III trials are further evaluating this approach. The phase III portion of KRYSTAL-7 is comparing adagrasib plus pembrolizumab with pembrolizumab monotherapy in patients with unresectable, locally advanced or metastatic NSCLC with KRAS G12C mutations and PD-L1 TPS ≥50%. In addition, the KRYSTAL-4 trial is evaluating adagrasib plus pembrolizumab plus chemotherapy compared with pembrolizumab plus chemotherapy in previously untreated nonsquamous NSCLC, regardless of PD-L1 status.21
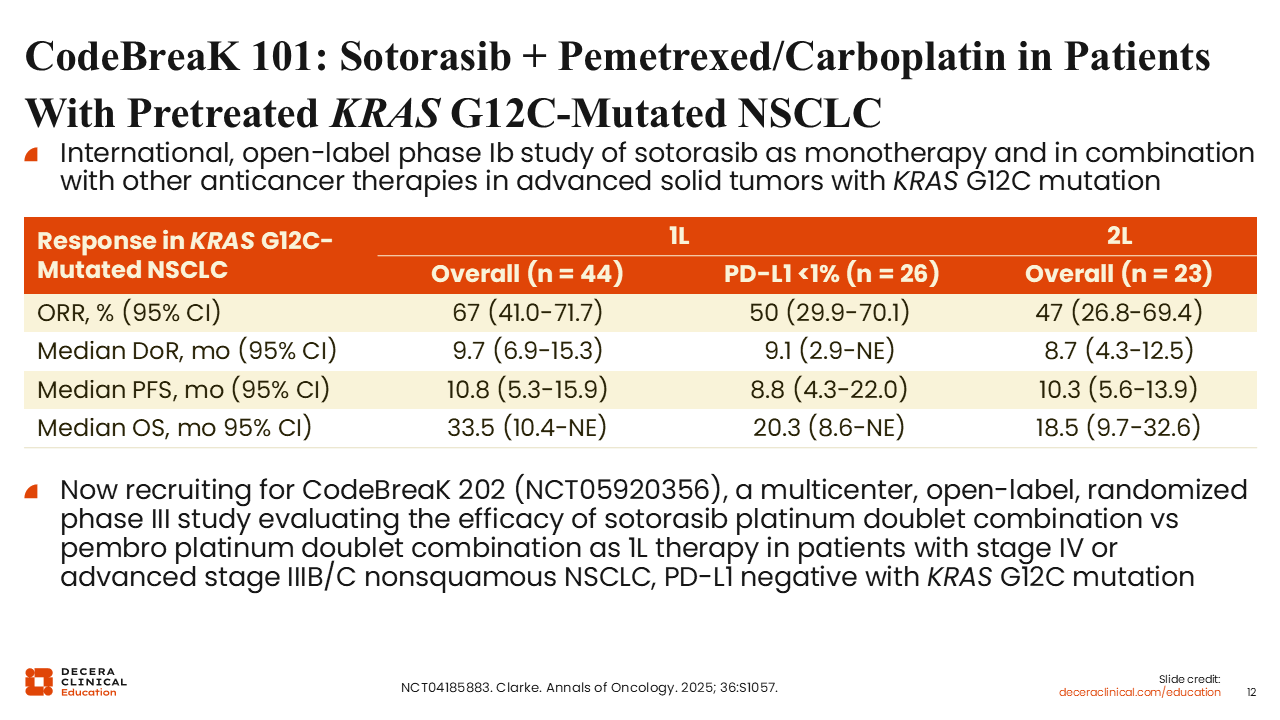
CodeBreaK 101: Sotorasib Plus Pemetrexed/Carboplatin in Patients with Pretreated KRAS G12C–Mutated NSCLC
Luis Paz-Ares, MD, PhD:
Sotorasib has also been evaluated in combination with chemotherapy. In the phase II CodeBreaK 101 study, the combination of sotorasib with carboplatin and pemetrexed demonstrated ORRs of approximately 67% in untreated patients and 47% in previously treated patients. Median PFS was approximately 10 months in both groups.22
However, these results were not consistently reproduced in the phase II SCARLET trial, in which the same combination demonstrated a high ORR of 89% but a shorter median PFS of 6.6 months.23
The phase III CodeBreaK 202 trial is ongoing and is evaluating sotorasib plus platinum-based chemotherapy compared with pembrolizumab plus platinum-based chemotherapy as first-line treatment for patients with metastatic or locally advanced, PD-L1–negative KRAS G12C–mutated NSCLC.24
Select Next-Generation KRAS G12C–Selective Inhibitors Currently in Development
Christine Bestvina, MD:
Given the limitations observed with first-generation KRAS G12C (OFF) inhibitors, there has been a focused effort to develop next-generation agents with improved potency and durability. Several next-generation KRAS G12C inhibitors are currently in development, including divarasib, olomorasib, calderasib, and opnurasib.
These agents are designed to retain the mechanism of targeting the inactive, GDP-bound state of KRAS G12C while achieving greater target inhibition. The goal is to improve response rates, prolong PFS, and reduce the incidence of primary resistance.25,26
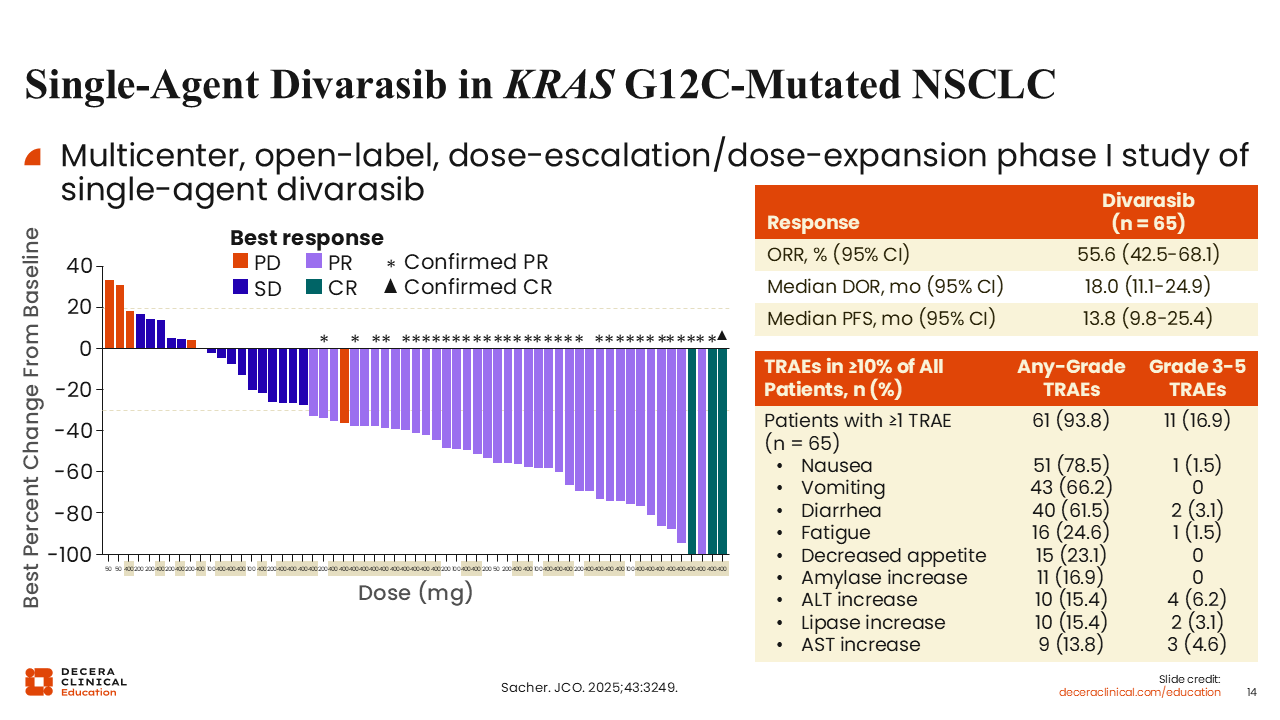
Single-Agent Divarasib in KRAS G12C–Mutated NSCLC
Christine Bestvina, MD:
Divarasib is a next-generation KRAS G12C (OFF) inhibitor designed to achieve high potency and sustained target engagement. In a phase I study, divarasib demonstrated an ORR of 56% and a median PFS of 14 months in patients with KRAS G12C–mutated NSCLC. The majority of patients enrolled (98%) had received at least 1 prior line of systemic therapy.
The safety profile was favorable, with a low incidence of grade ≥3 treatment-related AEs. Hepatic toxicity, including elevations in alanine aminotransferase (ALT) and aspartate aminotransferase (AST), occurred in approximately 5% of patients, which appears lower than rates reported with earlier-generation agents.27
KRAScendo 1: Divarasib vs Sotorasib or Adagrasib in KRAS G12C–Mutated Advanced or Metastatic NSCLC
Christine Bestvina, MD:
The phase III KRAScendo 1 trial is evaluating divarasib compared with sotorasib or adagrasib in patients with advanced or metastatic KRAS G12C–mutated NSCLC. Eligible patients must have received prior platinum-based chemotherapy and a PD-1 or PD-L1 inhibitor.
This randomized study is designed to directly compare divarasib and first-generation KRAS G12C inhibitors, with the goal of determining whether improved potency translates into superior clinical outcomes (NCT06497556).
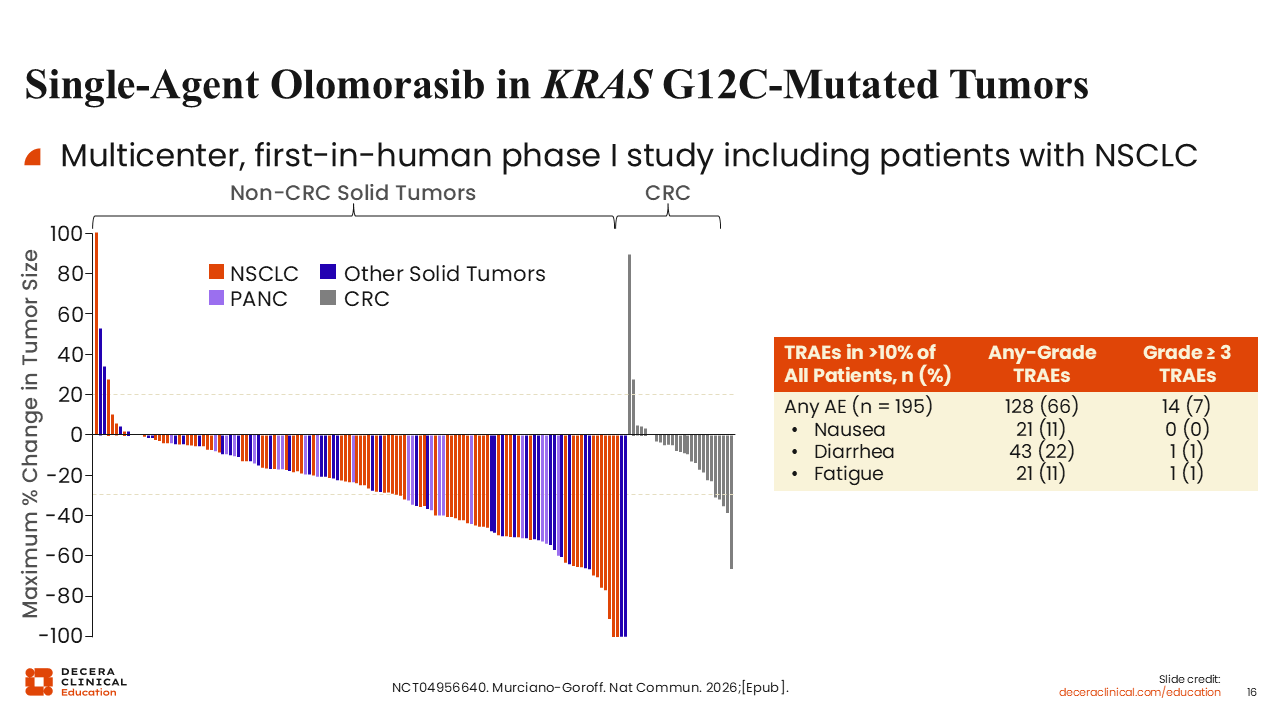
Single-Agent Olomorasib in KRAS G12C–Mutated Tumors
Christine Bestvina, MD:
Olomorasib is another next-generation KRAS G12C inhibitor designed to enhance target occupancy at lower drug exposures. In a first-in-human phase I study, olomorasib demonstrated an ORR of approximately 33% among patients with NSCLC. Among the 94 patients treated with olomorasib, 23% had previously received a KRAS G12C inhibitor.
The safety profile was favorable, with commonly reported AEs including diarrhea, nausea, fatigue, and constipation. Rates of hepatic toxicity, including ALT and AST elevations, were low.28
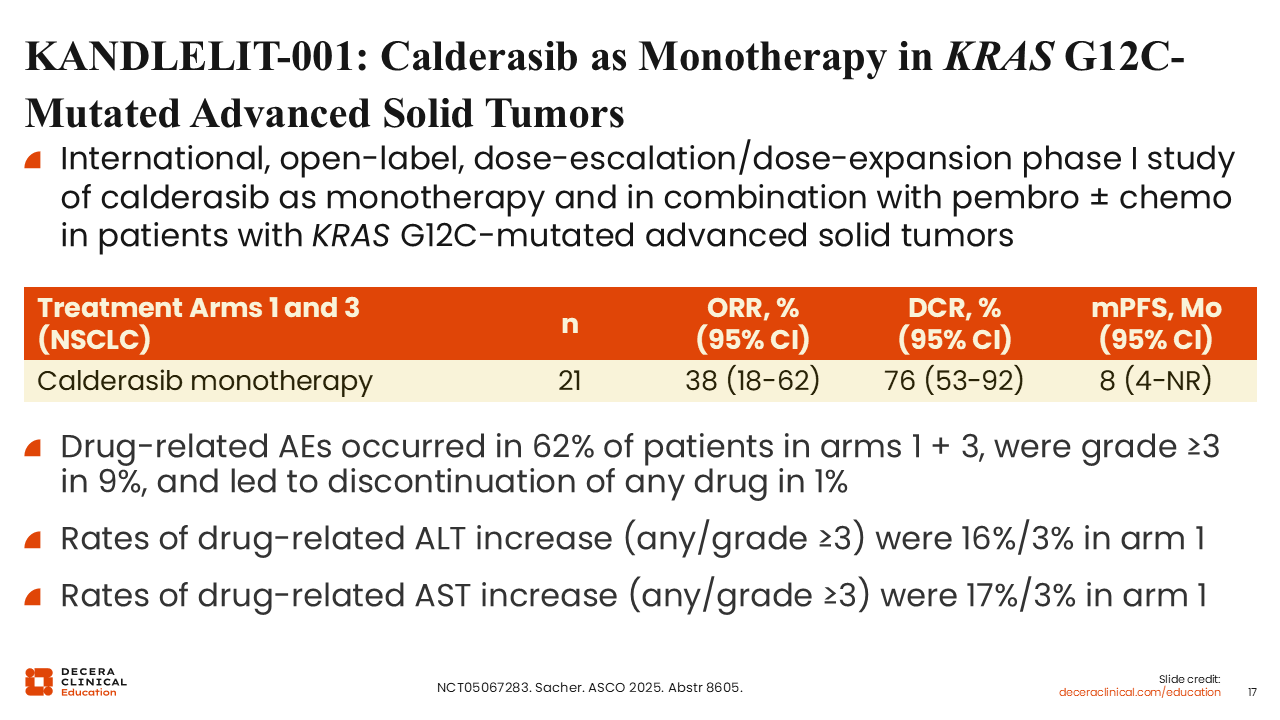
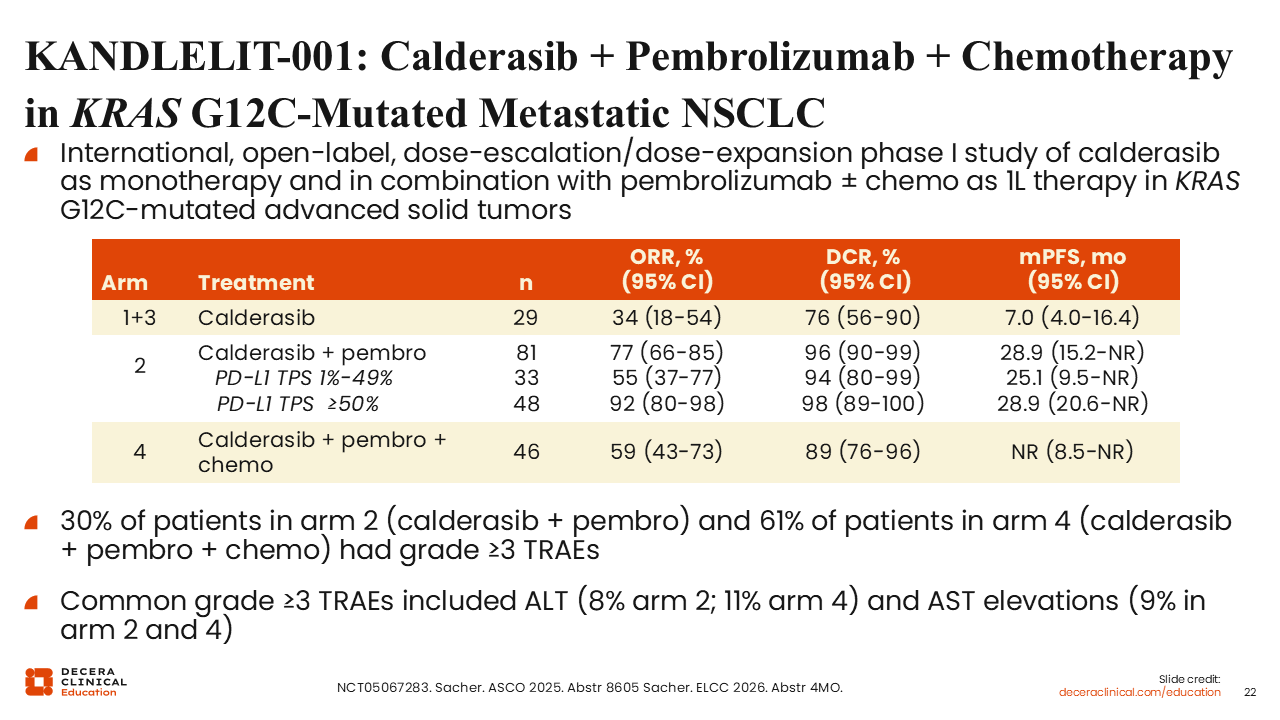
KANDLELIT-001: Calderasib as Monotherapy in KRAS G12C–Mutated Advanced Solid Tumors
Christine Bestvina, MD:
Calderasib has also demonstrated activity in early-phase clinical trials. In the phase I KANDLELIT-001 study, calderasib monotherapy produced an ORR of 38% in patients with KRAS G12C–mutated advanced solid tumors.
Among patients with NSCLC, the median duration of response was approximately 22 months, suggesting improved durability compared with earlier-generation agents.29
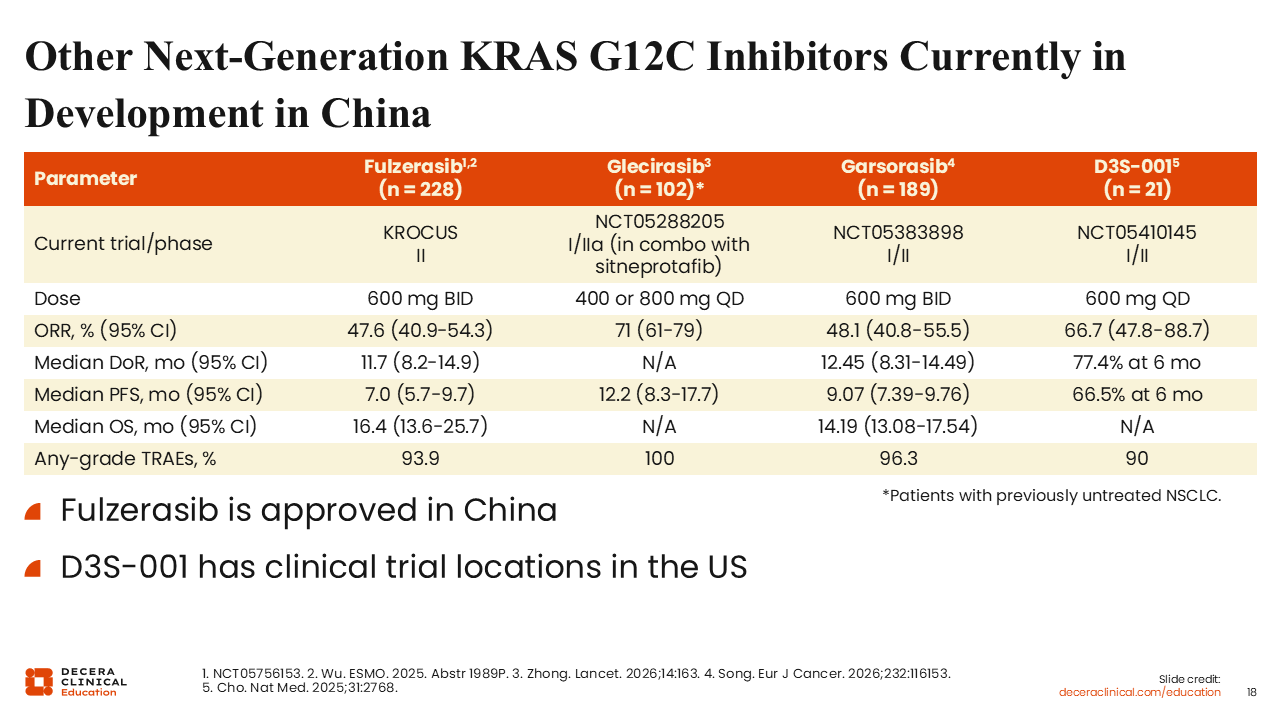
Other Next-Generation KRAS G12C Inhibitors Currently in Development in China
Christine Bestvina, MD:
Several other KRAS G12C inhibitors are being developed that should be briefly mentioned. Fulzerasib has demonstrated an ORR of 48% and a median PFS of 7 months in early studies.30 It is currently approved in China for KRAS G12C–mutant NSCLC. Other agents under investigation include glecirasib and garsorasib, with ongoing evaluation in clinical trials.31,32
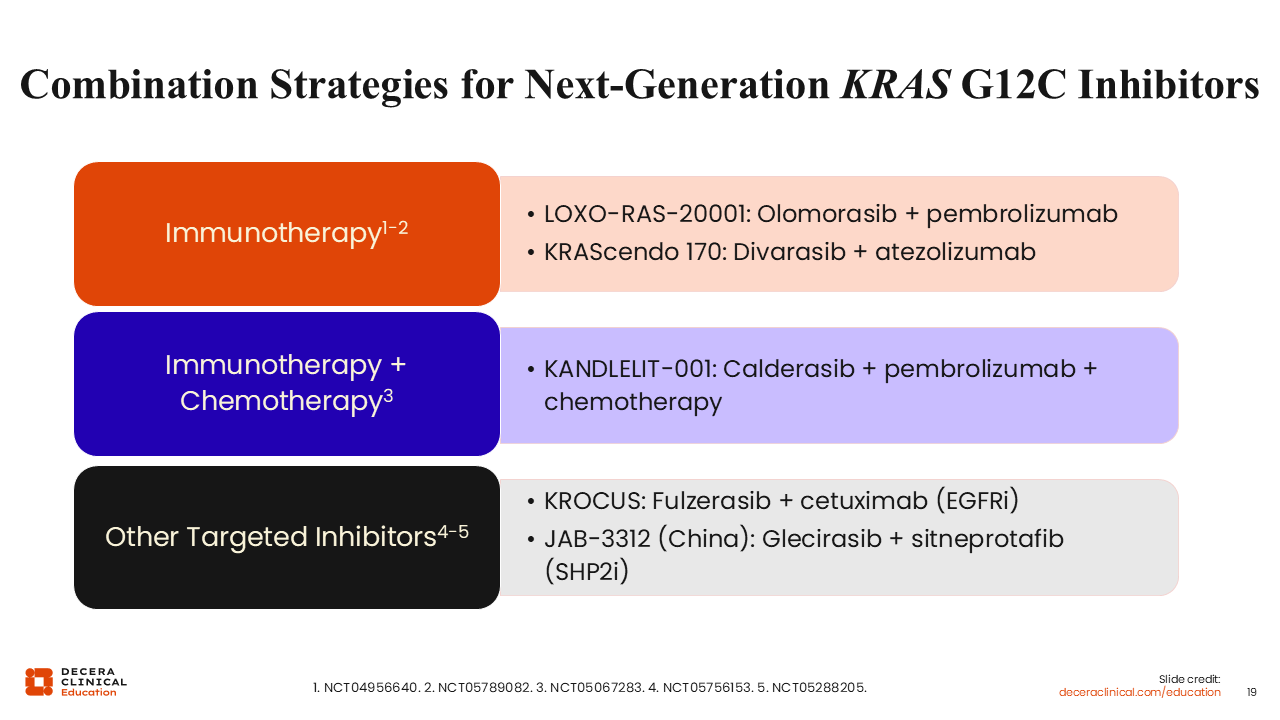
Combination Strategies for Next-Generation KRAS G12C Inhibitors
Christine Bestvina, MD:
Similar to first-generation agents, next-generation KRAS G12C inhibitors are being evaluated in combination with other therapies, particularly immunotherapy. These newer agents appear to have improved tolerability profiles, which may enhance their ability to be combined with immune checkpoint inhibitors.
Early observations suggest lower rates of hepatic toxicity, including ALT and AST elevations, compared with first-generation inhibitors, supporting further investigation of these combinations.
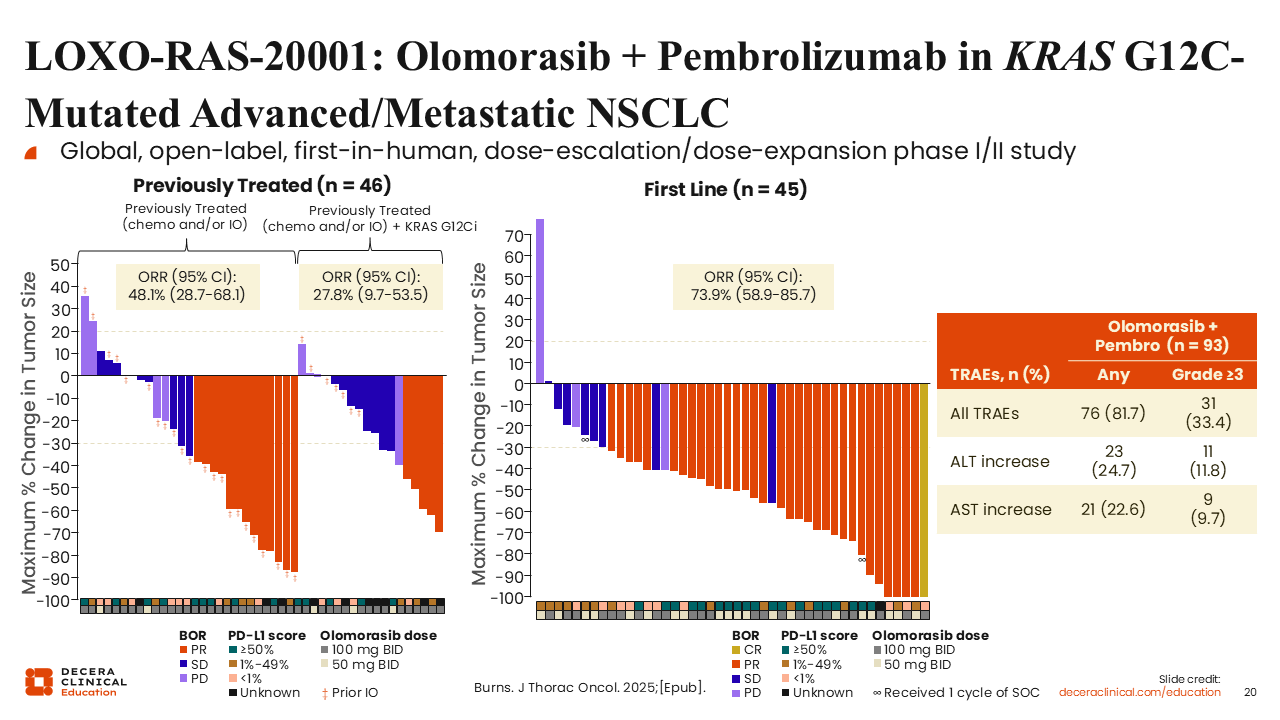
LOXO-RAS-20001: Olomorasib Plus Pembrolizumab in KRAS G12C–Mutated Advanced/Metastatic NSCLC
Christine Bestvina, MD:
LOXO-RAS-20001 is a global, open-label phase I/II study evaluating olomorasib in combination with pembrolizumab in patients with advanced or metastatic KRAS G12C–mutated NSCLC across multiple lines of therapy, including the first-line setting.
Eligible patients included those with any PD-L1 expression level, and patients could have received prior KRAS G12C inhibitors and/or immunotherapy. Among 93 patients treated at selected dose levels, grade ≥3 treatment-related AEs were observed in 33.3%. The most common AEs included diarrhea and elevations in ALT and AST.
Among 91 efficacy-evaluable patients in any line, the ORR was 57.1% across all PD-L1 expression levels. In the first-line setting, the ORR was 73.9%, and among patients with PD-L1 expression ≥50%, the ORR reached 90%. These findings suggest promising antitumor activity with manageable safety in this combination.33
KRAScendo 170: Safety of Divarasib Plus Atezolizumab in KRAS G12C–Mutated NSCLC
Christine Bestvina, MD:
In the KRAScendo 170 study, the combination of divarasib and atezolizumab demonstrated encouraging early activity. Reported outcomes included an ORR of approximately 61% and a median PFS of 15 months.
Among patients with measurable disease who had not received prior KRAS G12C inhibitors, the confirmed ORR was 55.6%. Grade 3 treatment-related AEs occurred in 25.6% of patients, with no grade 4/5 events reported. Hepatic AEs, including ALT and AST elevations, were infrequent and manageable.34
KANDLELIT-001: Calderasib Plus Pembrolizumab and Chemotherapy in KRAS G12C–Mutated Metastatic NSCLC
Christine Bestvina, MD:
Combination strategies incorporating calderasib with pembrolizumab, with or without chemotherapy, are under investigation in the KANDLELIT-001 study.
In this trial, response rates varied across treatment arms. The combination of calderasib plus pembrolizumab demonstrated ORRs ranging from 33% to 48%, with disease control rates up to 98% depending on PD-L1 expression. By contrast, the addition of chemotherapy resulted in an ORR of 59%.
Grade ≥3 treatment-related AEs were observed in approximately 30% of patients receiving calderasib plus pembrolizumab and 61% of those receiving the quadruplet combination. Common high-grade AEs included ALT and AST elevations. These findings reflect the combined toxicity of a KRAS inhibitor, immunotherapy and chemotherapy.35
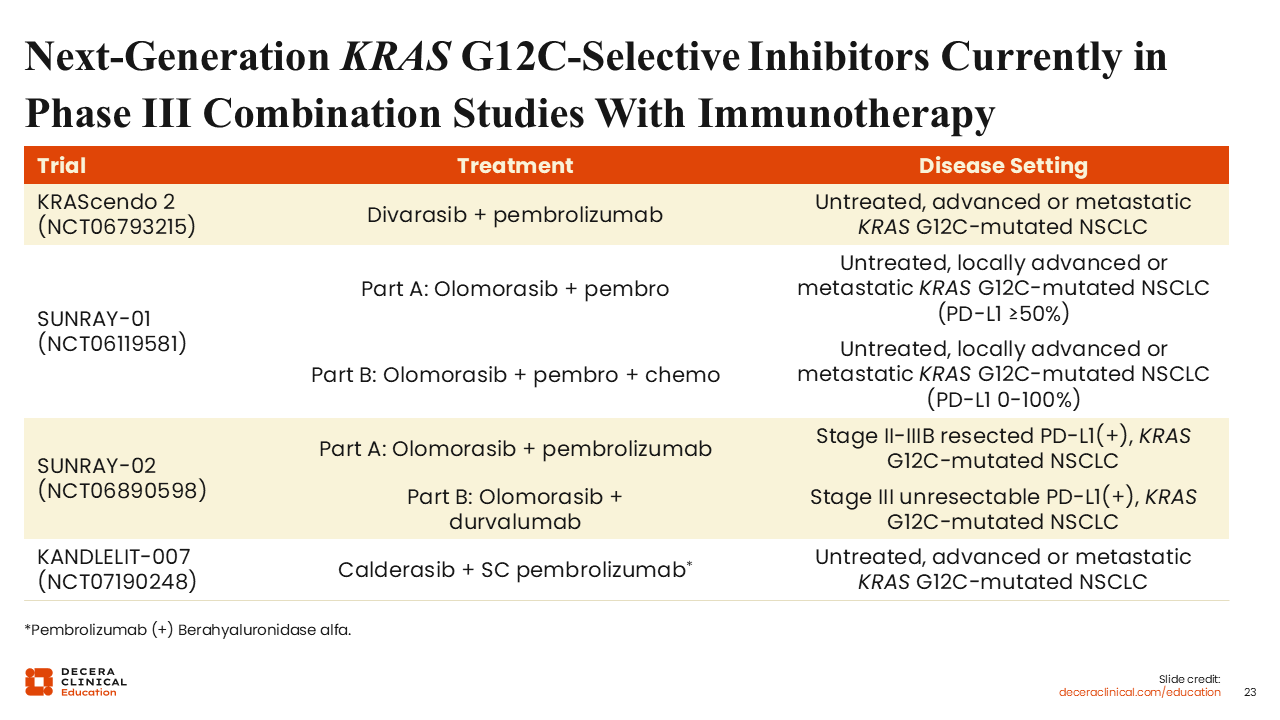
Next-Generation KRAS G12C–Selective Inhibitors Currently in Phase III Combination Studies With Immunotherapy
Christine Bestvina, MD:
Several phase III trials are ongoing to evaluate next-generation KRAS G12C inhibitors in combination with immunotherapy, with or without chemotherapy.
The KRAScendo 2 trial is comparing pembrolizumab plus chemotherapy with divarasib plus pembrolizumab. This study is designed to assess whether chemotherapy can be omitted in the frontline setting by incorporating a next-generation KRAS G12C inhibitor.36
Olomorasib combinations are also being evaluated across multiple phase III trials in KRAS G12C–mutated NSCLC. SUNRAY-01 is testing olomorasib with standard first-line therapy: Patients with PD-L1 ≥50% receive pembrolizumab with olomorasib or placebo (part A), while a broader population receives chemoimmunotherapy with or without olomorasib regardless of PD-L1 status (part B).37
Building on this, SUNRAY-02 extends evaluation into earlier-stage disease following definitive treatment.
This trial assesses olomorasib plus immunotherapy in stage II/III NSCLC, with pembrolizumab used in resected PD-L1–positive disease (part A) and durvalumab in unresectable stage III PD-L1–positive disease (part B).1 These studies aim to define optimal treatment strategies for patients with KRAS G12C–mutated NSCLC.
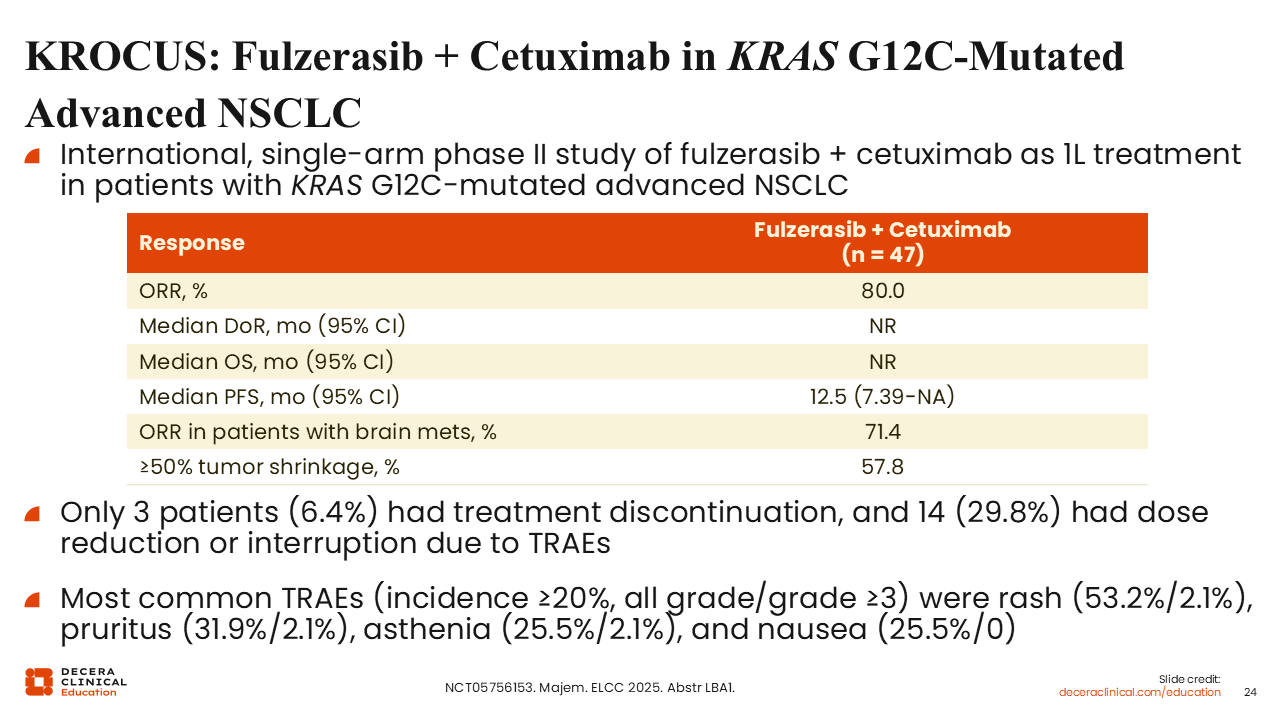
KROCUS: Fulzerasib Plus Cetuximab in KRAS G12C–Mutated Advanced NSCLC
Christine Bestvina, MD:
The KROCUS trial is an international, single-arm phase II study evaluating fulzerasib in combination with cetuximab as first-line treatment in patients with KRAS G12C–mutated advanced NSCLC.
In this study of 47 patients, the combination demonstrated an ORR of 80% and a median PFS of 12.5 months. Cetuximab, a monoclonal antibody targeting the EGFR, provides complementary pathway inhibition when combined with KRAS G12C inhibition, which may contribute to the high response rates observed.38
Phase I/IIa Study of Glecirasib (JAB-21288) Plus Sitneprotafib (JAB-3312), a SHP2 Inhibitor, in KRAS G12C–Mutated NSCLC
Christine Bestvina, MD:
A phase I/IIa study is evaluating the combination of glecirasib, a KRAS G12C inhibitor, with sitneprotafib, a SHP2 inhibitor, in patients with KRAS G12C–mutated advanced solid tumors, including NSCLC.
This combination targets both KRAS signaling and upstream pathway activation. Early data have demonstrated response rates of up to 73%, with more than one half of patients achieving deep responses, defined as >50% tumor reduction.39 These findings support the potential role of SHP2 inhibition in overcoming both primary and acquired resistance mechanisms.
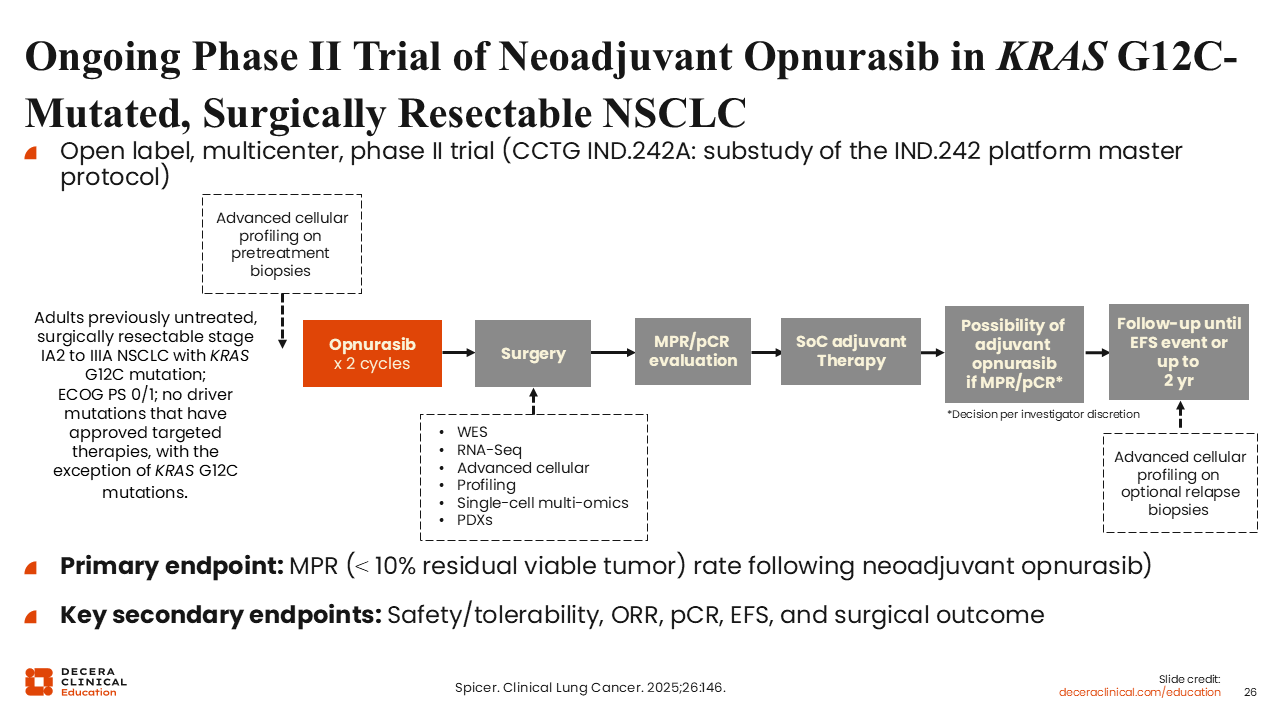
Ongoing Phase II Trial of Neoadjuvant Opnurasib in KRAS G12C–Mutated, Surgically Resectable NSCLC
Christine Bestvina, MD:
Efforts are underway to evaluate KRAS G12C inhibitors in earlier stages of disease. A phase II trial is investigating neoadjuvant opnurasib in patients with surgically resectable, KRAS G12C–mutated NSCLC.
In this study design, patients receive opnurasib prior to surgery, followed by standard-of-care adjuvant chemotherapy, with the option to continue opnurasib in the adjuvant setting. The primary endpoint includes pathologic response at the time of surgery, along with safety and feasibility assessments.40
This approach may provide important insights into tumor biology and response to KRAS inhibition in the early-stage setting.
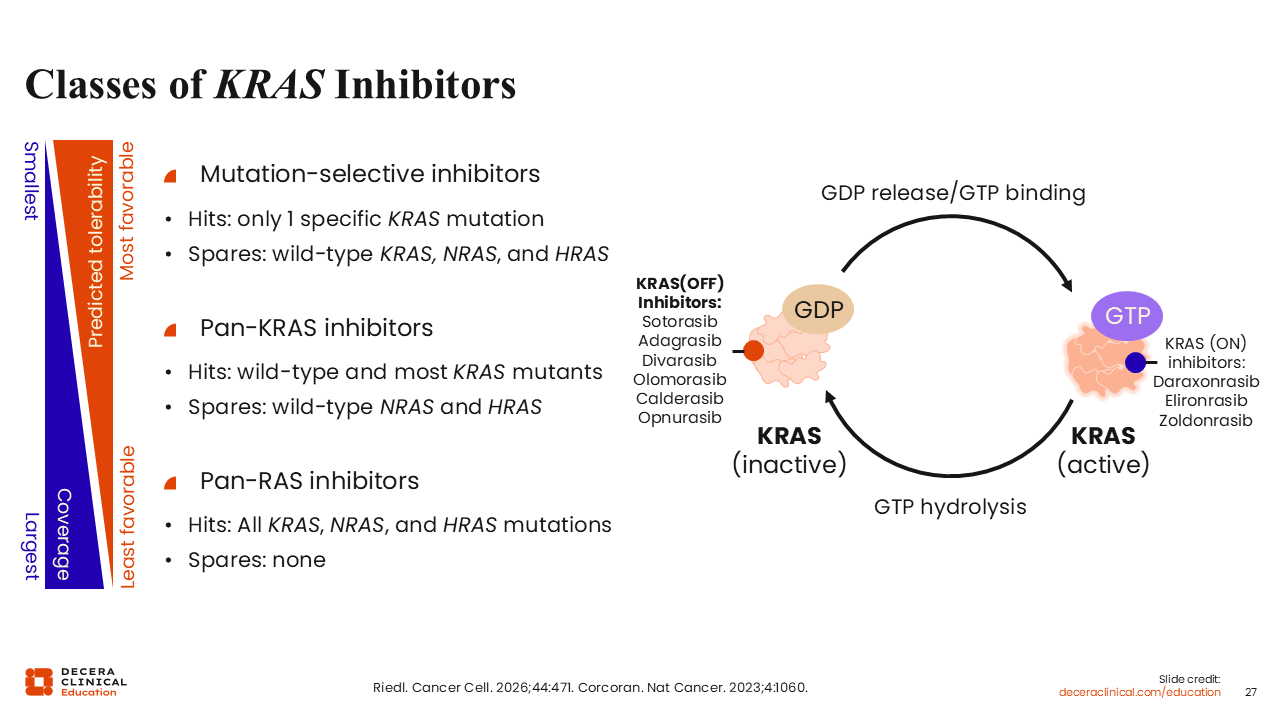
Classes of KRAS Inhibitors
Luis Paz-Ares, MD, PhD:
To date, most clinical development has focused on mutation-selective KRAS G12C (OFF) inhibitors. However, additional classes of KRAS and RAS inhibitors are under investigation.
These include KRAS G12C (ON) inhibitors, pan-KRAS inhibitors, and pan-RAS inhibitors. The degree of target coverage varies across these classes, ranging from selective inhibition of specific KRAS mutations to broader inhibition of KRAS, NRAS, and HRAS proteins.
Broader inhibition may be associated with increased toxicity because of effects on normal RAS signaling. Many KRAS (ON) inhibitors function by forming a complex with KRAS in its active state and a chaperone protein, cyclophilin A, thereby inhibiting downstream signaling.3
RAS(ON) Inhibitors in Clinical Trials: Elironrasib, Zoldonrasib, and Daraxonrasib
Luis Paz-Ares, MD, PhD:
Among RAS(ON) inhibitors, elironrasib is a mutant-selective, covalent inhibitor that targets the active KRAS G12C(ON) state. It has shown clinical activity in KRAS G12C–mutated NSCLC, with response rates of approximately 42% in pretreated patients (median PFS: 6.2 months) and >56% in untreated patients (median PFS: 9.9 months).41.
Additional RAS(ON) inhibitors include daraxonrasib, a multiselective noncovalent RAS(ON) inhibitor with broader mutant coverage, and zoldonrasib, a tricomplex inhibitor that selectively targets KRAS G12D(ON).
These agents are being evaluated in combinations (eg, with pembrolizumab or other RAS inhibitors) to enhance pathway suppression, overcome resistance, and address tumor heterogeneity (NCT06162221).
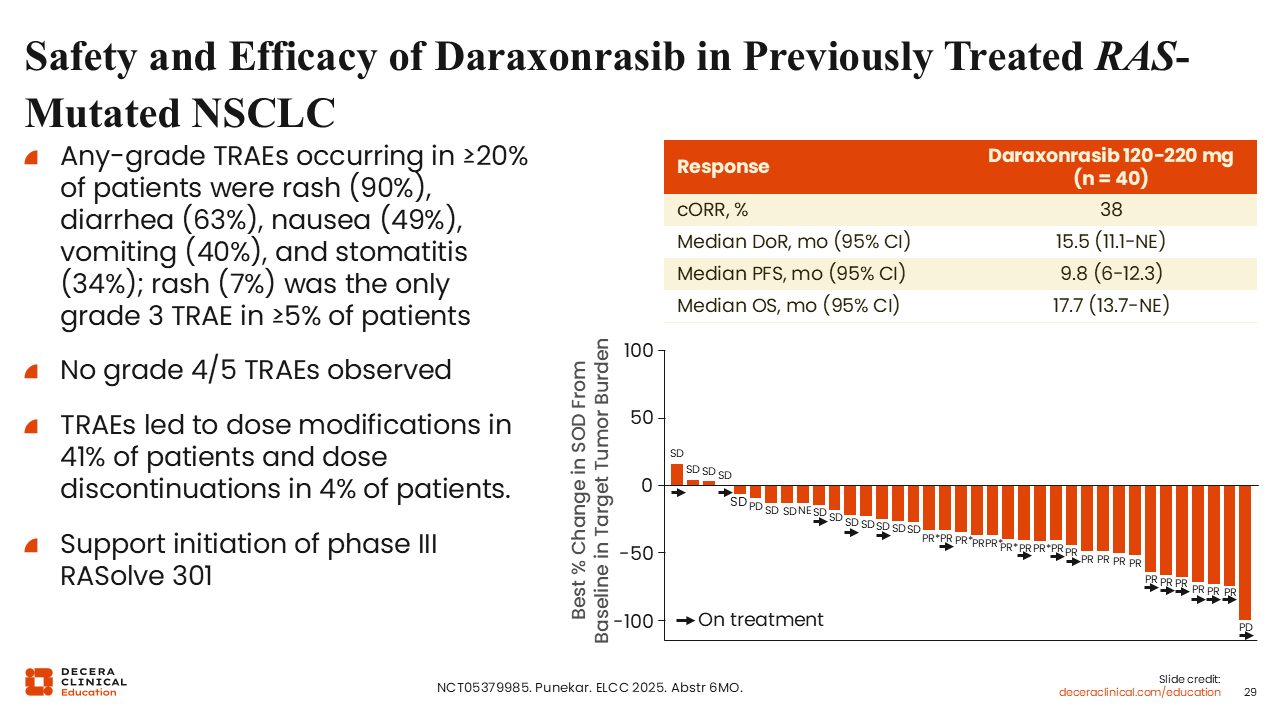
Safety and Efficacy of Daraxonrasib in Previously Treated RAS-Mutated NSCLC
Luis Paz-Ares, MD, PhD:
Daraxonrasib is a pan-RAS (ON) inhibitor that has demonstrated activity in previously treated patients with RAS-mutated NSCLC. In early studies, the ORR was approximately 38%, with a median duration of response of 15.1 months. Median PFS was 9.8 months, and median OS was 17.7 months.42
A phase III trial (RASolve 301) is ongoing to further evaluate daraxonrasib compared with docetaxel in previously treated patients.43
Take-home Points
Luis Paz-Ares, MD, PhD:
Targeting KRAS signaling in NSCLC has historically been challenging, but recent advances have led to the development of effective targeted therapies. Selective KRAS G12C inhibitors are now available for clinical use, although their benefit is often limited by the development of resistance.
Next-generation KRAS inhibitors and combination strategies, particularly those incorporating immunotherapy with or without chemotherapy, have demonstrated promising activity in early studies. Ongoing phase III trials will help define the role of these approaches in clinical practice.
Additional therapeutic strategies, including KRAS (ON) inhibitors and pan-RAS inhibitors, are also in development. A key unmet need is the identification of biomarkers to guide treatment selection and sequencing to optimize outcomes for individual patients.