CE / CME
Chronic Obesity Management: Expert Perspectives on Long-term Weight Loss Support and Monitoring
Physician Assistants/Physician Associates: 0.25 AAPA Category 1 CME credit
Nurse Practitioners/Nurses: 0.25 Nursing contact hour
Physicians: maximum of 0.25 AMA PRA Category 1 Credit™
Released: September 25, 2025
Expiration: September 24, 2026
Activity
Obesity as a Chronic Disease
Obesity is a chronic disease, much like diabetes and hypertension, and therefore, it should be treated like a chronic disease. Patients with obesity need continued, ongoing, and enduring treatment. This is because there are biological mechanisms with compensatory response, such as metabolic adaptation, at play. What happens with this? Unfortunately, as increased weight loss is achieved, the greater impact this has on metabolism—hence the metabolic adaptation.
There also are hormonal and neural adaptations. These occur when weight loss ensues. There is clinical evidence of the chronicity of obesity in that it is hard to lose weight and keep it off without adjunctive obesity treatment like antiobesity medications (AOMs) and/or bariatric surgery. These forces usually occur within 1 year.1
In addition, evidence shows that more than 80% of one’s weight is regained within 5 years after stopping treatment.2 Biology persists; compensatory changes persist for years. That is why continued ongoing and enduring treatment for obesity is required.
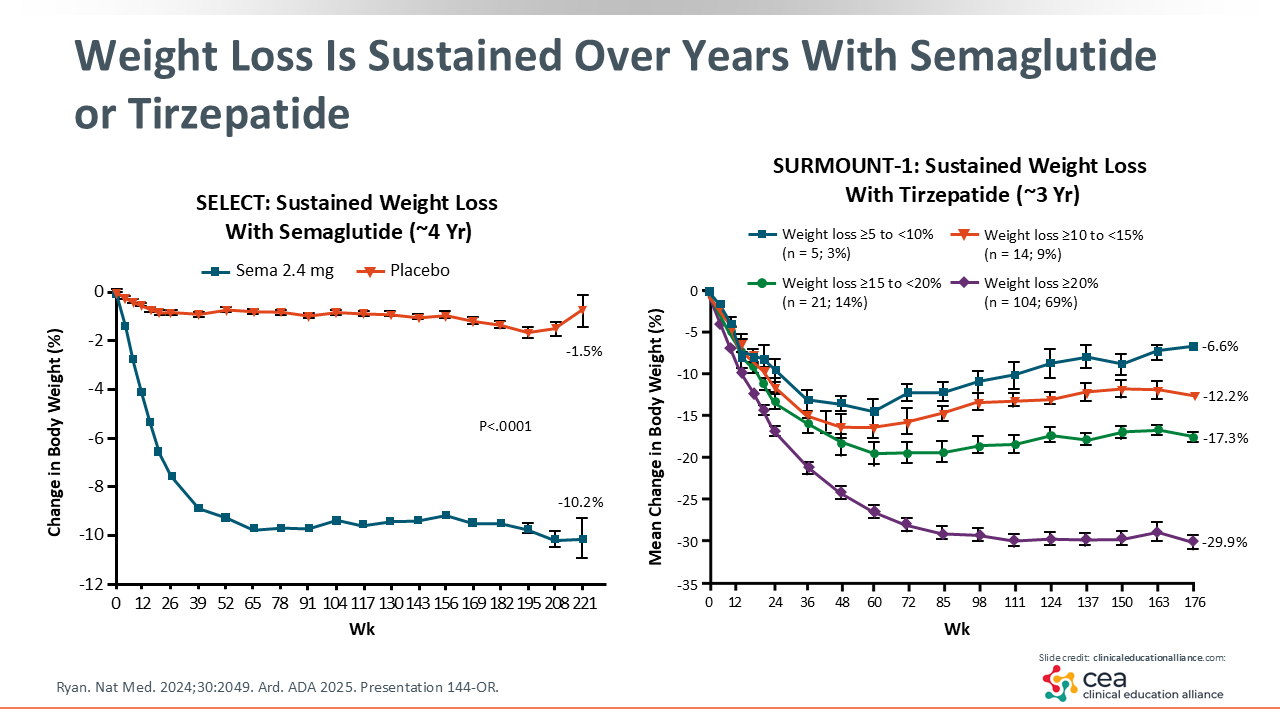
Weight Loss Is Sustained Over Years With Semaglutide or Tirzepatide
Looking at 2 pivotal studies, the SELECT trial evaluated the long-term weight loss induced by semaglutide vs placebo and was a cardiovascular outcomes trial. The figure on left side of the slide shows that there was a significant improvement in weight loss (-10.2%) at 221 weeks. This showed that semaglutide offers continued weight loss to patients over approximately 4 years of treatment.3
The right figure illustrates the sustained weight loss seen with tirzepatide in the SURMOUNT-1 trial. The figures shows that with 3 varying doses of tirzepatide (5.0, 10.0, and 15.0 mg), patients also achieved significant weight loss, ranging from -6.6% to -29.9%.4
Long-term Outcomes With AOMs
Long-term outcomes with AOMs are impressive, but use of AOMs must be sustained. For instance, studies on AOMs have reported an average weight loss of 12% to 15% with semaglutide, 8% with liraglutide, and approximately 20% with tirzepatide over 1-2 years of treatment. In addition to this weight loss, AOMs have positive impacts on cardiometabolic markers and/or anthropometric measures, as seen with all 6 major AOMs.5-7 As alluded to before, however, discontinuing an incretin-based therapy, in particular, leads to significant weight gain starting at approximately 8 weeks and reaching an average of 2.5 kg regained by 20 weeks.5-8
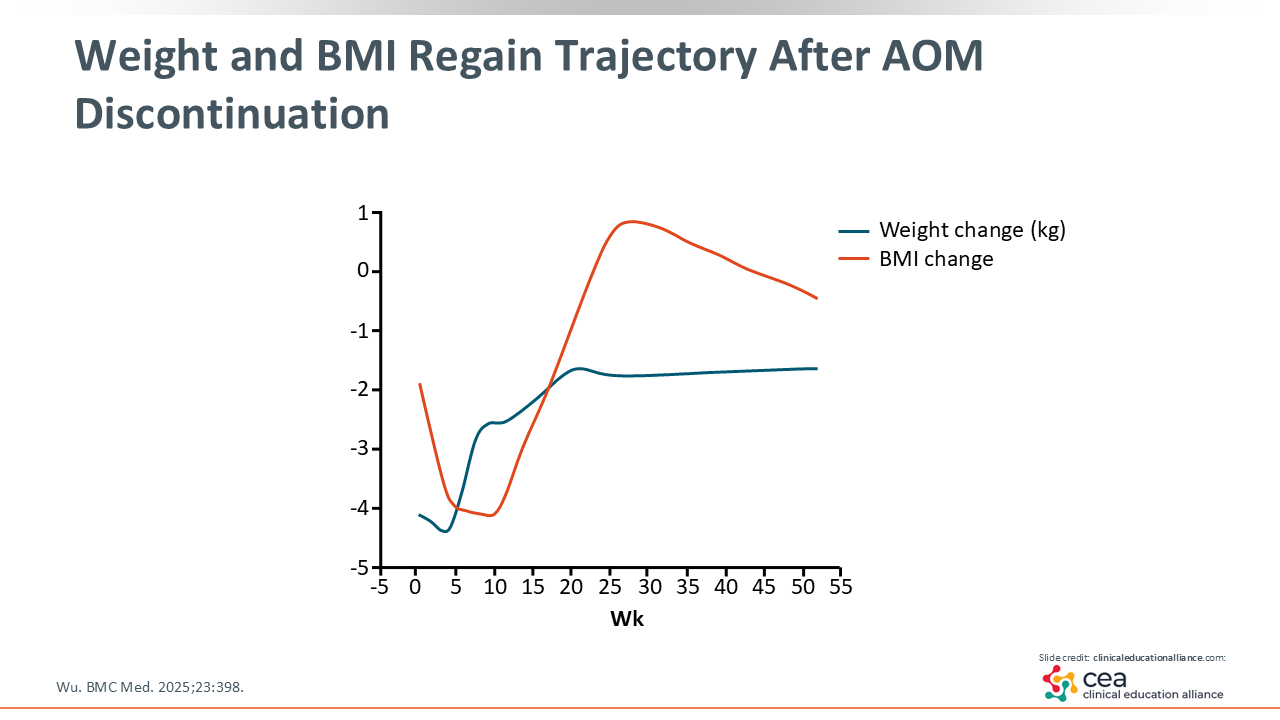
Weight and BMI Regain Trajectory After AOM Discontinuation
The weight and BMI regain traction after AOM discontinuation are depicted on the slide. After stopping incretin-based therapy, there was a significant amount of weight regained and increases in BMI reported.8
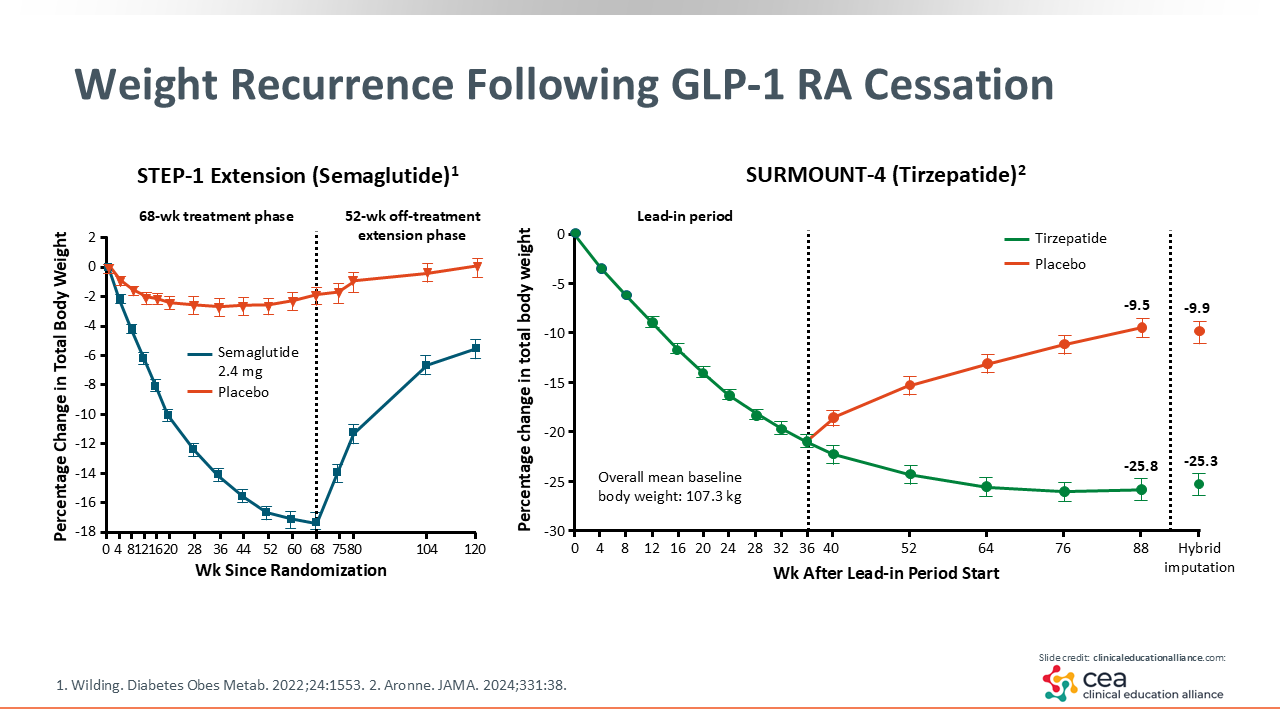
Weight Recurrence Following GLP-1 RA Cessation
This slide specifically highlights weight recurrence following semaglutide or tirzepatide cessation. The STEP-1 study found that after patients stopped semaglutide, there was significant weight regained within 1 year. This is important because incretin-based therapies are incredibly effective at reducing appetite. When taking that physiologic appetite suppression away, that initial weight loss is negatively affected.9
The SURMOUNT-4 study looking at tirzepatide was slightly different. These patients were treated with tirzepatide 10 or 15 mg for 36 weeks. Then they were randomized either to continue tirzepatide or switch to placebo for 52 weeks. The mean weight change for the patients who continued tirzepatide was -5.5% vs 14.0% for those who switched to placebo. The average weight loss for those patients treated with tirzepatide for the full study period was approximately -26.0%.10
Again, this supports the theory that obesity is a chronic disease that needs to be treated as such, especially with GLP-1 receptor agonists. Abrupt cessation of these agents leads to significant weight regain.
Additional Indications for Semaglutide and Tirzepatide Obesity Management Formulations
Regarding some GLP-1 receptor agonists, there are additional indications and health improvements reported for both semaglutide and tirzepatide in obesity management.
In addition to reducing body weight, semaglutide is also indicated to reduce risk of major adverse cardiovascular events (MACE) in adults with established cardiovascular disease and overweight or obesity. Most recently, semaglutide was approved to treat metabolic dysfunction–associated steatohepatitis (MASH) with moderate to advanced fibrosis in adults. Therefore, it is apparent through these indications that semaglutide significantly improves patients’ cardiometabolic health, too.11
In addition to reducing body weight, tirzepatide was recently approved to treat of moderate to severe obstructive sleep apnea (OSA) in adults with obesity.12 Like semaglutide, tirzepatide shows significant cardiometabolic improvement in patients and is not just used for weight loss.
Clinical Benefits of AOMs Beyond Weight Loss
This slide highlights the studies that led to the additional indications for semaglutide and tirzepatide discussed previously. The SELECT study enrolled adults with obesity and cardiovascular disease, without type 2 diabetes. In this population, semaglutide reduced risk of MACE (ie, composite cardiovascular death, nonfatal myocardial infarction, and nonfatal stroke) by 20% vs placebo.13 An ongoing study called the ESSENCE then evaluated liver outcomes with semaglutide in adults with MASH and moderate to severe fibrosis. This study found that semaglutide significantly improved liver histology (steatohepatitis resolution and fibrosis improvement) compared with placebo in these patients.14
Finally, the SURMOUNT-OSA trial reported improved sleep outcomes with tirzepatide. It showed that tirzepatide significantly reduced apnea–hypopnea index scores and bettered sleep-related patient-reported outcomes vs placebo in adults with obesity and moderate to severe OSA.15
Strategies for Long-term Weight Maintenance
Continuing AOMs in the long term is crucial because stopping these agents leads to weight regain. This was well proven in the STEP-1 extension and SURMOUNT-4 trials.9,10 Behavioral reinforcement also is critical and includes nutrition counseling, physical activity, sleep optimization, and structured follow-up visits. This support is imperative for ongoing success.
I think nutrition counseling is particularly important because healthcare professionals (HCPs) must ensure patients receiving long-term AOMs have a good diet quality. Since these agents are effective in reducing calorie and food intake, HCPs must make sure that what food patients are eating is of high quality. For example, patients still need to intake enough protein, fruits, and vegetables while receiving AOMs.
Other strategies for long-term weight maintenance include multidisciplinary support with collaboration among behavioral health professionals, dietitians, and group programs to address any emotional and environmental triggers. Obesity management is a group effort; therefore, everybody who can be involved will help for the betterment and the success of patients going forward.
Monitoring follow-up is important. HCPs should at the least measure patients’ weight frequently. I recommend regularly measuring their waist circumferences, labs, and comorbidity improvements as well. Finally, relapse management is necessary since there are—and will be—fluctuations in weight loss and gain. Continued accountability is imperative here.16,17
Communicating Obesity as a Chronic Disease
It also is critical to communicate with patients that obesity is a chronic disease. We need to set early and realistic expectations that obesity is a chronic, relapsing condition like hypertension and diabetes. Therefore, long-term management is expected. This should be treated as a marathon, not a sprint. HCPs should set realistic expectations with patients of 5% to 10% weight loss goals. Once these goals are met, then we can move on to the next 5% to 10% weight loss.
HCPs must normalize treatment duration, meaning AOMs often need to be continued lifelong like antihypertensive and cholesterol therapy. Stopping AOMs can lead to rapid weight gain and might undo the cardiometabolic improvements that were seen while on the treatment.9,10 Also of importance is ensuring patients understand that a lack of willpower does not cause this to happen; rather, this is a result of biology and physiology. If patients stop taking their medications, it is not as if they can just say, “Well, I am not going to eat.” AOMs are incredibly effective at decreasing appetite. So when patients stop taking them, their appetite returns and weight gain ensues.
Finally, HCPs must emphasize health, not just weight loss. As I discussed, some AOMs are effective at reducing risk of MACE, diabetes, liver disease, and OSA. They also improve patients’ quality of life and energy. This is why HCPs should not only focus on the improvements seen on a scale, but highlight the improvements seen in their quality of life.17
Communicating Obesity as a Chronic Disease Cont.
Of note, communicating obesity as a chronic disease also addresses issues related to stigma and language. HCPs should use patient-first language, meaning we do not say “obese patients” but “patients with obesity.” We should avoid using the word “obese” altogether. In addition, HCPs should eliminate words like “fat,” “morbidly obese,” and the like from their vocabulary. This is because we should be reinforcing the idea to patients that obesity is not simply about willpower, but about biology and the environment.
Those triggers of hormonal and metabolic adaptations described earlier come into play lifelong. Therefore, HCPs should empower with partnership. As I said before, the more we can be accountable with patients, the better they are going to do in the long term. We want to encourage patient engagement and shared decision-making on all fronts—on what meal plan is best for them, on what exercise schedule they should follow, and on what AOM meets their individual needs.17,18
Framing Chronic Treatment to Patients
For how to frame chronic treatment to patients, your communication strategy can follow the chronic care model.19 That is, we do not stop patients’ antihypertensive agents when their blood pressure normalizes, nor do we stop blood sugar therapy when their blood sugar normalizes. Therefore, we should not stop AOMs when patients have “finished losing weight."
Again, we also should focus on health outcomes—improvements in all the comorbid medical issues, such as hypertension, OSA, degenerative joint disease, that patients deal with daily. HCPs should then address common concerns among patients, especially regarding AOMs. These agents come with some toxicities, so we need to look at safety and costs.
Furthermore, HCPs should ensure that patients understand that these are long-term treatments. I often am asked, “Do I need to take this forever?” I respond by relating obesity to those other conditions I have discussed. Once they stop their antihypertensive agents, their blood pressure will increase, for example. There also are many patient education tools available, so HCPs should ensure that patients are tracking weight, waist circumference, and serologies or blood work that need to be done on a regular basis.
Strategies for Maintenance Treatment
Here are some take-home strategies for long-term weight maintenance treatment. Pharmacotherapy should be continued long-term and optimized to the maximal therapeutic dose needed; HCPs can consider combination therapy if needed. Furthermore, lifestyle modifications, behavioral support, self-monitoring, and relapse prevention are critical to comprehensive obesity management. There are many apps out there that patients can use. We also can use tracking from our phones. Anything that is used for self-monitoring will help keep us accountable.
Lifestyle modifications include sustainable dietary patterns with adequate protein, fruits, vegetables, and fluids intake as well as regular physical activity to maintain weight loss. It is currently recommended that patients engage in 200-300 minutes per week of physical activity, including resistance training.
Finally, regarding regular follow-up and monitoring, it is critical to do this on a regular basis. It is recommended that follow-up visits should occur every 1-3 months initially, then at least every 3-6 months thereafter. I think it is important patients are seen regularly to titrate their AOM as needed and ensure that they are getting adequate protein and diet quality intake, as discussed. In these regular visits, HCPs should track weight, body composition, cardiometabolic markers, quality of life, and treatment adherence. Doing that will help with maintenance treatment or allow weight loss maintenance to continue long term.17,20