CE / CME
PROTAC ER Degraders in ER-Positive/HER2-Negative MBC Progressing After Endocrine Therapy: Current Treatment Landscape and Addressing Unmet Needs
Physician Assistants/Physician Associates: 1.00 AAPA Category 1 CME credit
Pharmacists: 1.00 contact hour (0.1 CEUs)
Physicians: maximum of 1.00 AMA PRA Category 1 Credit™
Nurse Practitioners/Nurses: 1.00 Nursing contact hour
Released: March 12, 2026
Expiration: September 11, 2026
Activity
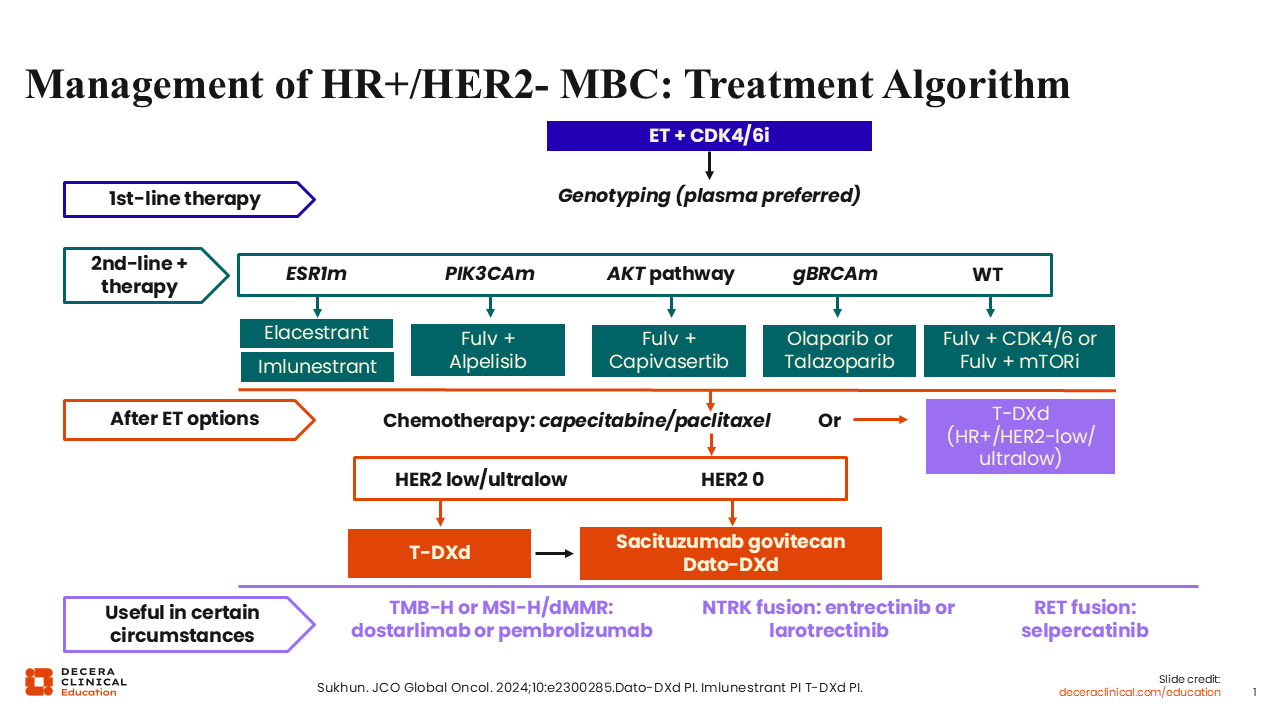
Management of HR+/HER2- MBC: Treatment Algorithm
Adrienne G. Waks, MD:
The current SoC first-line treatment for ER-positive/HER2-negative advanced or metastatic breast cancer remains ET with a CDK4/6 inhibitor.2 However, development of ET resistance mechanisms, such as mutation in the estrogen receptor 1 gene (ESR1), or alterations in PIK3CA, AKT1, and PTEN genes, remain critical challenges. Of note, ESR1 mutations are the more prominent mechanism of resistance that develops during or following treatment with ET.1 These mutations can lead to constitutive activation of ERs despite estrogen depletion, which enables or facilitates tumor progression. ESR1 mutations are detected in approximately 4% of patients receiving first-line treatment for ER-positive/HER2-negative MBC and in approximately 40% of such patients after progression on AIs.
ESR1-Altered MBC
At the time of this publication, there are 2 FDA approved single-agent oral SERDs for patients with ESR1 mutations: elacestrant and imlunestrant. Elacestrant is indicated for the treatment of postmenopausal women or adult men with ER-positive/HER2-negative, ESR1-mutated advanced or metastatic breast cancer with disease progression following at least 1 line of ET.3 Imlunestrant is indicated for treatment of adults with ER-positive/HER2-negative, ESR1-mutated advanced or metastatic breast cancer with disease progression following at least 1 line of ET.4
PIK3CA-Altered MBC
Alpelisib plus fulvestrant is indicated for adults with locally advanced or metastatic hormone receptor (HR)–positive/HER2-negative PIK3CA-mutated breast cancer following progression on or after an endocrine-based regimen.5
Inavolisib is currently indicated in combination with palbociclib and fulvestrant for the treatment of adults with ET-resistant, PIK3CA-mutated, HR-positive/HER2-negative, locally advanced or metastatic breast cancer, as detected by an FDA-approved test, following recurrence on or after completing adjuvant ET.6
PIK3CA/AKT1/PTEN-Altered MBC
Capivasertib in combination with fulvestrant is indicated for the treatment of adult patients with HR-positive/HER2-negative, locally advanced or metastatic breast cancer with 1 or more PIK3CA/AKT1/PTEN alterations as detected by an FDA-approved test following progression on 1 or more endocrine-based regimen in the metastatic setting or recurrence on or within 12 months of completing adjuvant therapy.7
BRCA-Altered MBC
PARP inhibitors are an option in patients with germline BRCA mutations following treatment with ET. Olaparib is indicated for the treatment of adult patients with deleterious or suspected deleterious germline BRCA mutation, based on an FDA-approved companion diagnostic test, and HER2-negative MBC who have been treated with chemotherapy in the neoadjuvant, adjuvant, or metastatic setting; patients with HR-positive breast cancer should have been treated with a prior ET or be considered inappropriate for ET.8
Talazoparib is indicated for the treatment of adult patients with deleterious or suspected deleterious germline BRCA mutation, HER2-negative locally advanced or metastatic breast cancer based on FDA-approved companion diagnostic testing.9
Wild-Type MBC
In patients with progression on an AI and no actionable mutations, SoC options may include fulvestrant with a different CDK4/6 inhibitor not previously received or with the mTOR inhibitor everolimus. Trastuzumab deruxtecan (if HER2-low/ultralow) or sacituzumab govitecan (if HER2 zero), or chemotherapy may also be considered.2,10
Summary Data for First-line Combinations of CDK4/6 Inhibitors and ET in HR+/HER2- Breast Cancer: PFS and OS
Adrienne G. Waks, MD:
For patients with ET-sensitive disease, we have available clinical data for 3 approved treatment options consisting of a CDK4/6 inhibitor (ie, abemaciclib, palbociclib, or ribociclib) in combination with either an AI or fulvestrant. mPFS for most of these combinations is approximately 24 months.11-15 With regard to median overall survival, only the combinations using the CDK4/6 inhibitors abemaciclib or ribociclib have demonstrated a survival benefit and thus are generally preferred by experts for patients who are thought to be able to tolerate these agents.
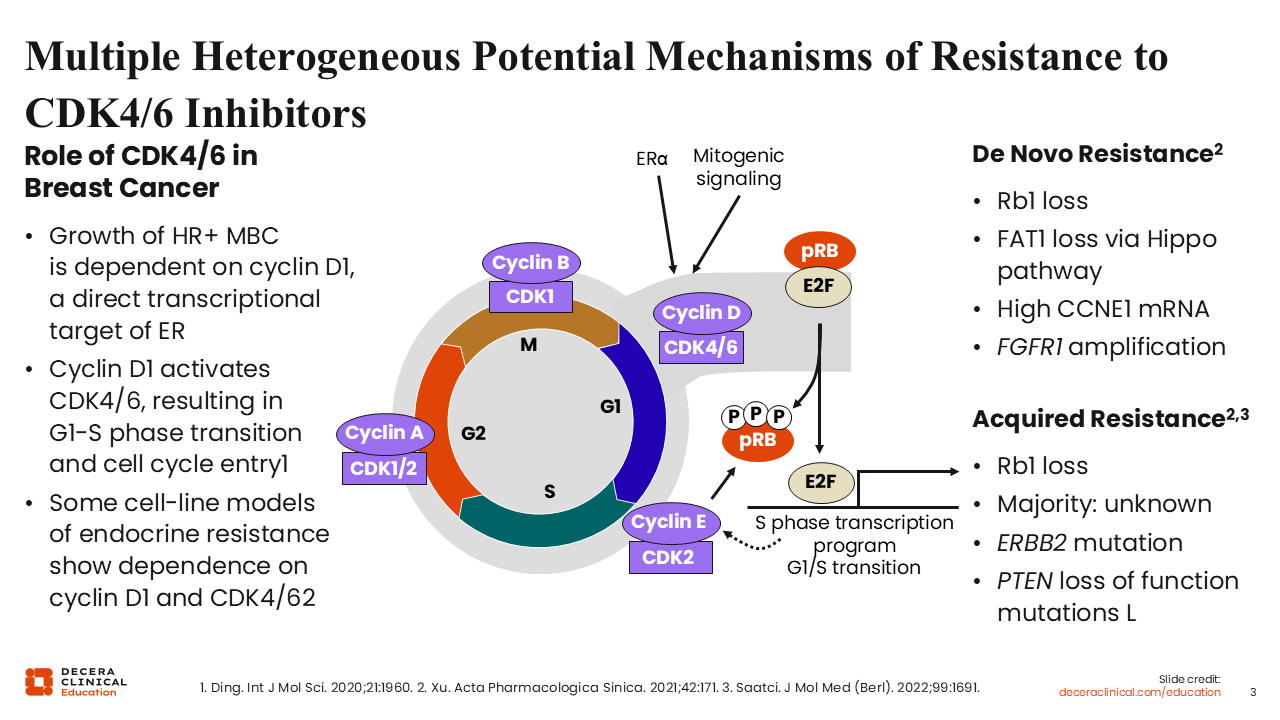
Multiple Heterogeneous Potential Mechanisms of Resistance to CDK4/6 Inhibitors
Adrienne G. Waks, MD:
In general, CDK4/6 inhibitor–based regimens are used in the first-line setting. An important unmet need in this population is identifying treatment options that are effective after the first-line CDK4/6 inhibitor no longer achieves disease control. There have been efforts to understand resistance mechanisms to CDK4/6 inhibitors and how we can translate that shift in tumor biology into novel targeted pathways in the second-line setting. Although there is still much to learn, we do understand a number of de novo (ie, Rb1 and/or FAT1 loss, upregulation of CCNE1 mRNA, and FGR1 amplification) and acquired resistance mechanisms (Rb1 loss, ERBB2/HER2 gene mutation, and PTEN loss-of-function mutations) to first-line CDK4/6 inhibitors and are currently working to develop targeted approaches.
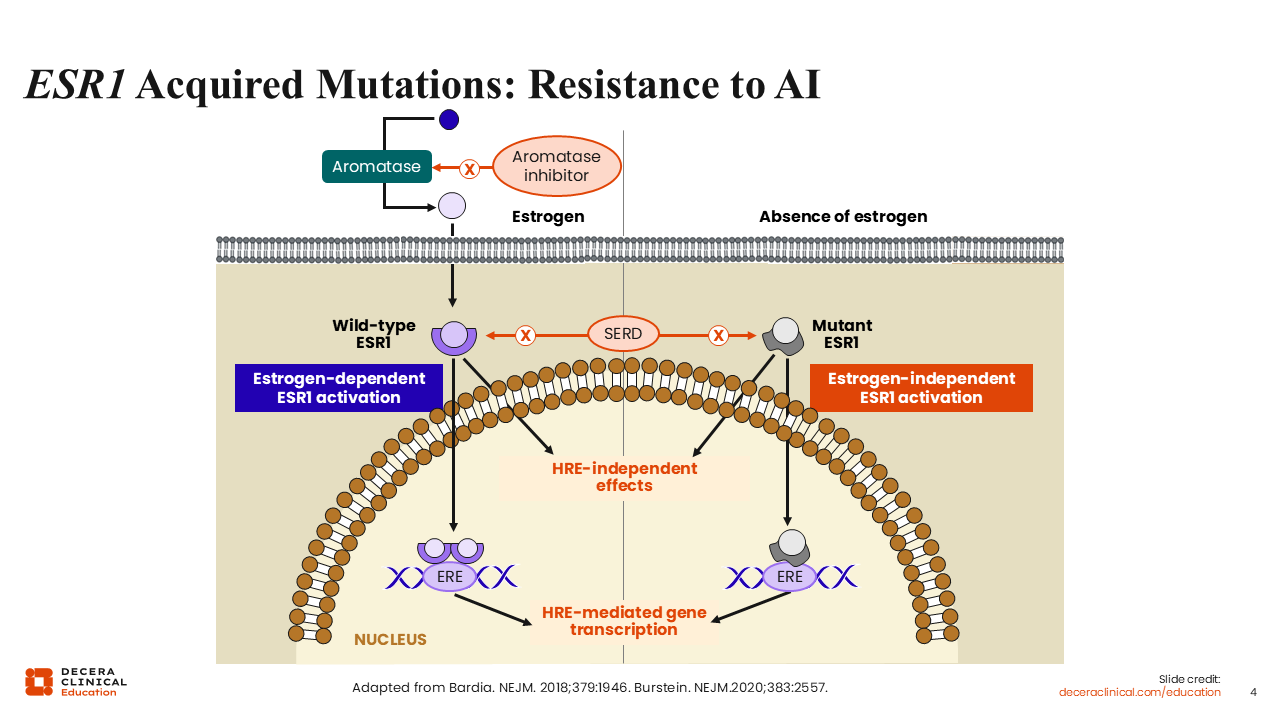
ESR1 Acquired Mutations: Resistance to AI
Adrienne G. Waks, MD:
Despite improved outcomes with oral SERDs, many patients continue to experience disease progression within 12 months of starting an oral SERD, including those with an ESR1 mutation.16,17
To gain a better understanding of ER-related therapy resistance mechanisms, I would like to dive deeper into the various ways the ER-receptor pathway escapes inhibition, including from aromatase inhibition and estrogen-independent ER activation.
Under normal breast cell function, the enzyme aromatase converts androgen precursors into estrogen, which in turn can enter the cell and dock into the ESR1 protein—helping drive its relocation into the cell nucleus to engage transcriptional programs via estrogen-responsive elements.18 Activation of the ESR1 pathway leads to cell proliferation, which we know can lead to cancer cell progression. This estrogen-dependent ESR1 activation can be blocked with AIs (anastrozole, letrozole, exemestane). AIs work by inhibiting the conversion of an androgen precursor into estrogen.19 Obviously, if estrogen is not present by virtue of aromatase inhibition, this cascade cannot happen. However, breast cancer cells find other ways to continue to thrive in the absence of estrogen. One such way is by acquired somatic mutations in the ESR1 gene that render the receptor active even in the absence of estrogen.
Global guidelines recommend ESR1 mutation testing for patients with early-stage breast cancer that has progressed to advanced breast cancer while receiving ET using liquid biopsy and ctDNA testing, as well as for PIK3CA, AKT, and PTEN pathway alterations.20,21
EMERALD: Elacestrant vs Investigator’s Choice SoC ET in ER+/HER2- MBC
Adrienne G. Waks, MD:
Now I want to focus on the data for patients with ESR1 mutations as a common mechanism of resistance to adjuvant or first-line ET. These are the patients who are potential candidates for the approved oral SERDs.
Patients with disease progression after AI or ET with a CDK4/6 inhibitor and confirmed ESR1 mutation may receive elacestrant based on the results from the phase III EMERALD trial.3 The study compared the oral SERD elacestrant vs investigator’s choice of single-agent ET in men and postmenopausal women with ER-positive/HER2-negative MBC with disease progression after 1-2 lines of ET (1 line with CDK4/6 inhibitor) and ≤1 line of chemotherapy for advanced disease (N = 478). The coprimary endpoints were progression-free survival (PFS) in all patients and PFS in patients with ESR1 mutation, both by blinded independent central review (BICR).16
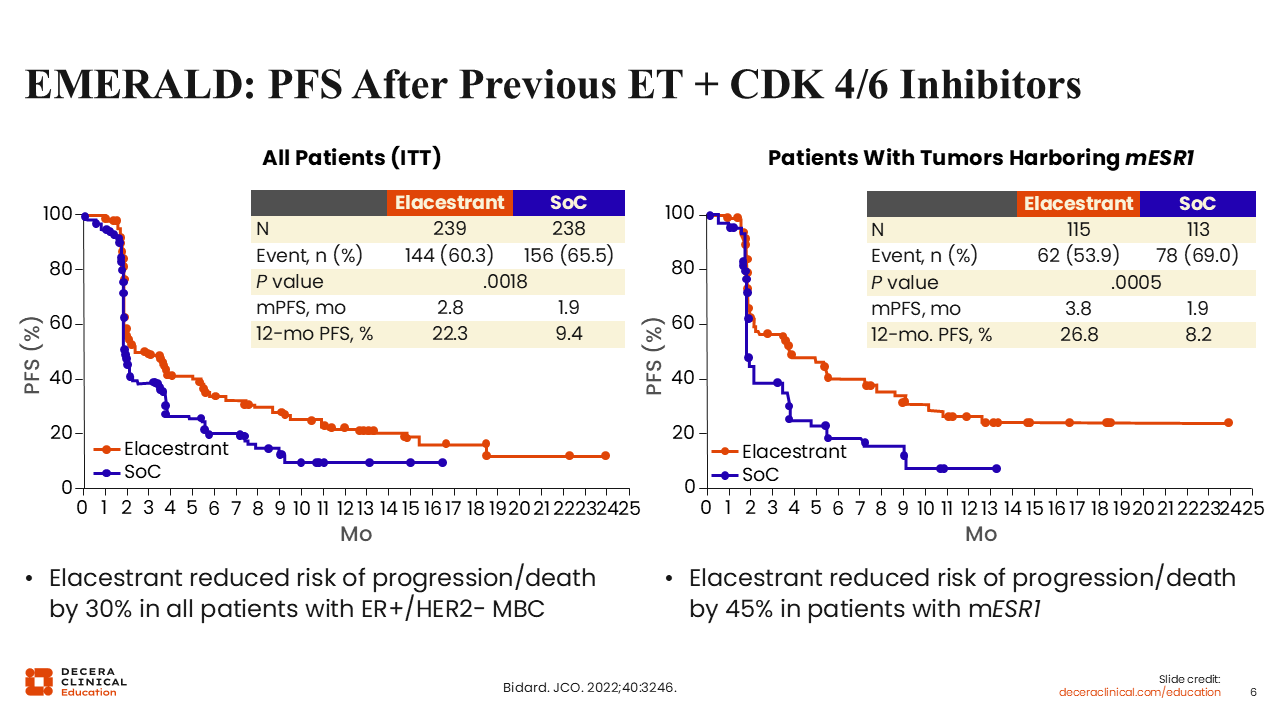
EMERALD: PFS After Previous ET + CDK 4/6 Inhibitors
Adrienne G. Waks, MD:
In the EMERALD trial, investigators showed that elacestrant statistically improved mPFS in all patients (2.8 vs 1.9 months; P = .0018), as well as in patients with ESR1 mutation status (3.8 vs 1.9 months; P = .0005), albeit the magnitude of benefit was greater in those with ESR1-mutated tumors.
Based on these data, elacestrant received FDA approval as previously discussed.3 However, we also know from the EMERALD trial that approximately 50% of patients experienced disease progression at first restaging.16
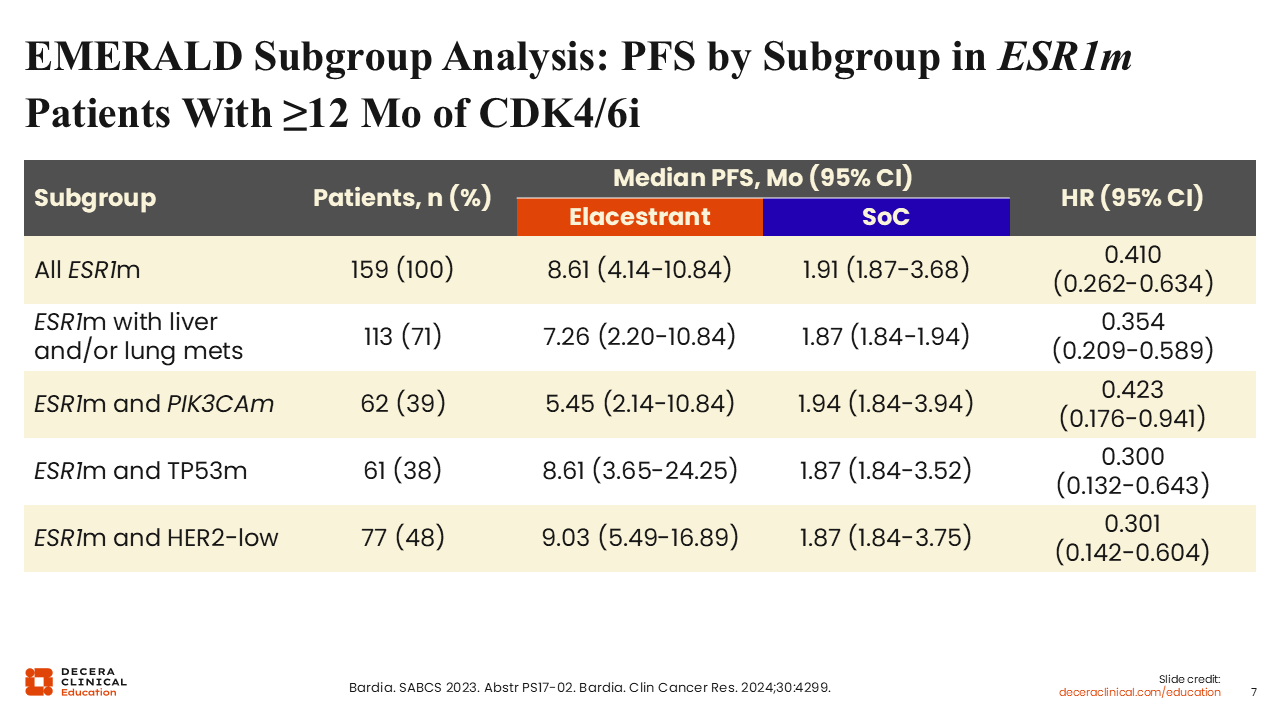
EMERALD Subgroup Analysis: PFS by Subgroup in ESR1m Patients With ≥12 Mo of CDK4/6i
Adrienne G. Waks, MD:
Subsequent subgroup analyses from the EMERALD trial aimed to identify the patients who would achieve more durable benefit with elacestrant monotherapy. Investigators showed that in patients with ESR1 mutation who received CDK4/6 inhibitor for ≥12 months, subsequent treatment with elacestrant yielded more durable and clinically meaningful benefit vs SoC (exemestane or injectable fulvestrant) (mPFS: 8.61 vs 1.91 months; hazard ratio: 0.410; 95% CI: 0.262-0.634). This trend was observed in patients with liver and/or lung metastases (mPFS: 7.26 vs 1.87 months; hazard ratio: 0.354; 95% CI: 0.209-0.589), in patients with PIK3CA mutations (mPFS: 5.45 vs 1.94 months; hazard ratio: 0.423; 95% CI: 0.176-0.941), those harboring a TP53 mutation (mPFS: 8.61 vs 1.87 months; hazard ratio: 0.300; 95% CI: 0.132-0.643), and in those with HER2-low disease (mPFS: 9.03 vs 1.87 months; hazard ratio: 0.301; 95% CI: 0.142-0.604).
EMBER-3 Update: Imlunestrant ± Abemaciclib vs SoC ET in ER+/HER2- Advanced Breast Cancer
Adrienne G. Waks, MD:
In September 2025, we saw the approval of a second oral SERD, imlunestrant.4 Imlunestrant’ s approval is supported by data from the phase III EMBER-3 trial.17 The study evaluated treatment with imlunestrant with or without abemaciclib compared with SoC (investigator’s choice of exemestane or intramuscular fulvestrant) in adult men and women with ER-positive/HER2-negative advanced or metastatic breast cancer with disease recurrence within 12 months of completing adjuvant AI with or without CDK4/6 inhibitor or who had disease progression while on AI with or without CDK4/6 inhibitor as first-line treatment for MBC. The coprimary endpoints were PFS in the intention-to-treat (ITT) population and PFS among patients with ESR1 mutations.
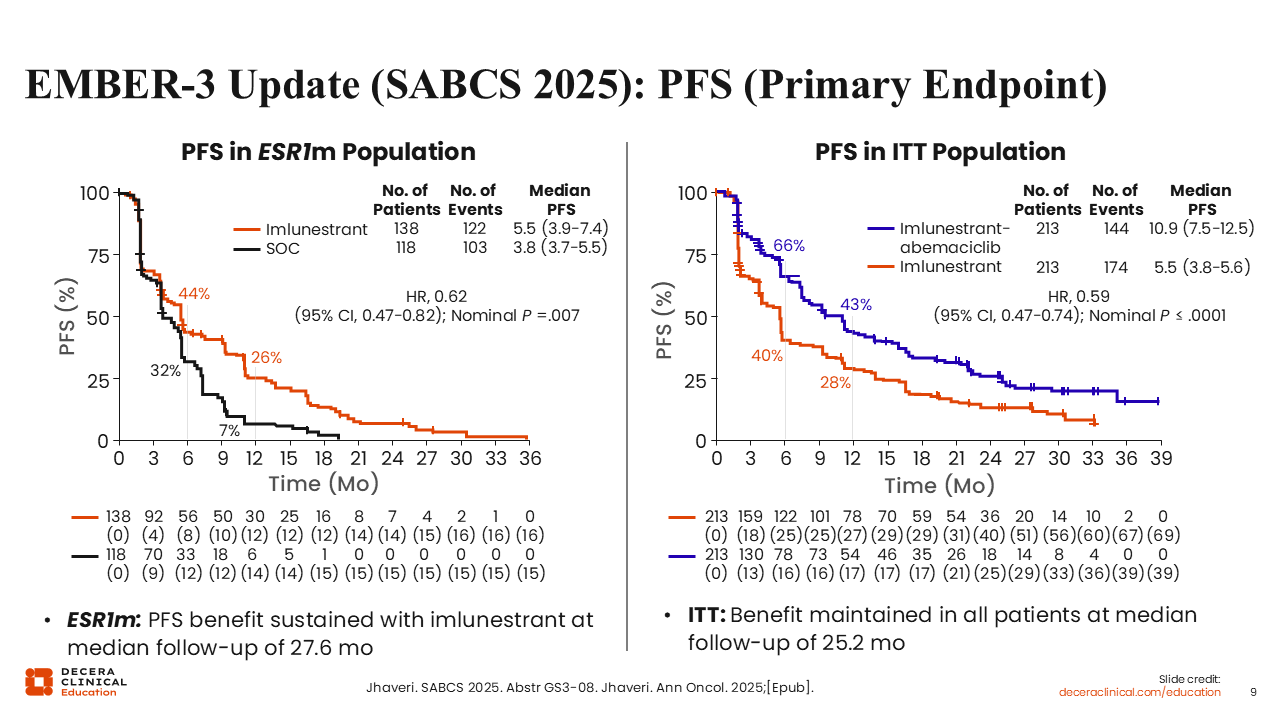
EMBER-3 Update (SABCS 2025): PFS (Primary Endpoint)
Adrienne G. Waks, MD:
Updated results for EMBER-3 were presented at the San Antonio Breast Cancer Symposium (SABCS) 2025. These updated results reassert the benefit of imlunestrant vs SoC in the ESR1-mutated population, demonstrating that patients receiving imlunestrant experienced better mPFS (5.5 vs 3.8 months; hazard ratio: 0.62; 95% CI: 0.47-0.82; nominal P =.007). In addition, in the arm evaluating the combination of imlunestrant with abemaciclib vs SoC in the ITT population, we also observe an mPFS benefit (10.9 vs 5.5 months; hazard ratio: 0.59; 95% CI: 0.47-0.74; nominal P ≤.0001).
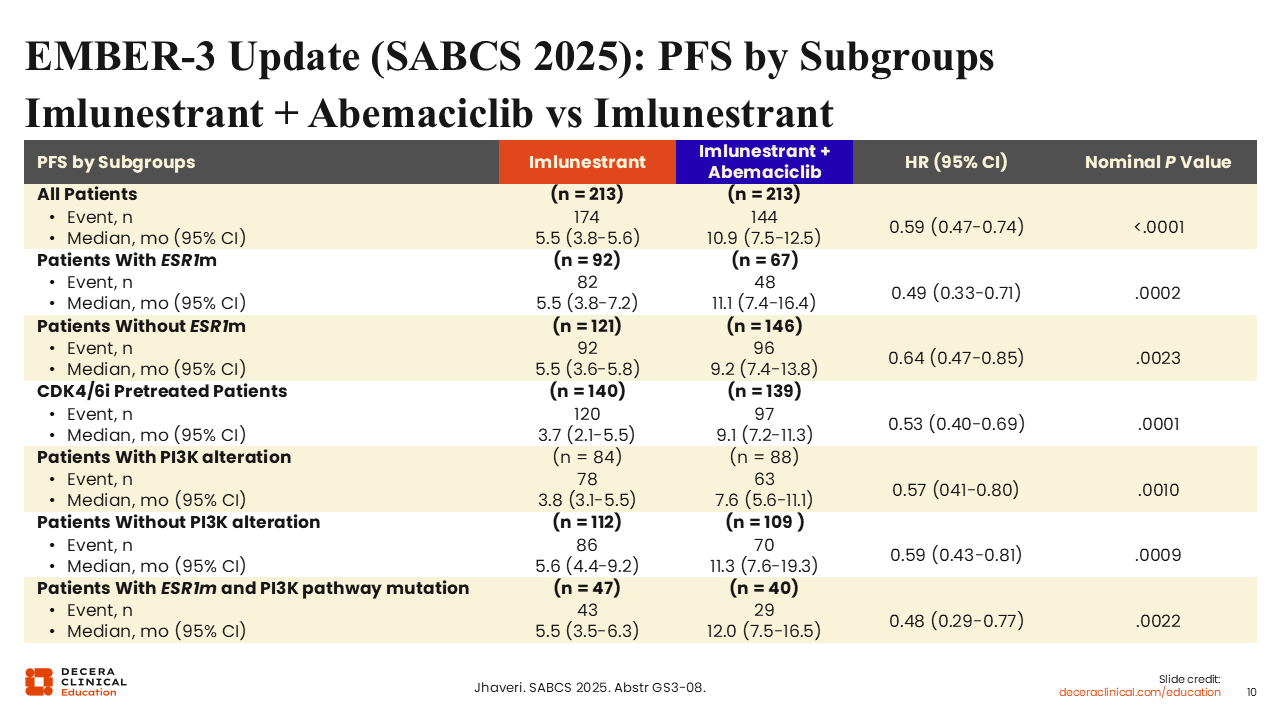
EMBER-3 Update (SABCS 2025): PFS by Subgroups Imlunestrant + Abemaciclib vs Imlunestrant
Adrienne G. Waks, MD:
If we look at subgroup analyses from the EMBER-3 trial presented at SABCS 2025 for both the imlunestrant with abemaciclib arm and the imlunestrant monotherapy arm, the mPFS for those with ESR1 mutations was 11.1 months with imlunestrant plus abemaciclib vs 5.5 months with imlunestrant monotherapy (hazard ratio: 0.49; 95% CI: 0.33-0.71; nominal P = .0002) and for those without an ESR1 mutation the mPFS was 9.2 months with imlunestrant plus abemaciclib vs 5.5 months with imlunestrant (hazard ratio: 0.64; 95% CI: 0.47-0.85); nominal P = .0023).
Other important subgroups include patients pretreated with a CDK4/6 inhibitor, where the PFS benefit was preserved. There is also a benefit seen regardless of patients’ PIK3CA mutation status, particularly in patients with comutated ESR1 and PI3K pathway. Of note, that is a small patient population, making up approximately 20% of all patients in these cohorts. This is important because healthcare professionals can encounter a situation where a patient’s tumor has both of these mutations, and a choice of which mutation to target must be made. Here, we see a preservation of benefit with an mPFS of 12.0 months with imlunestrant plus abemaciclib and of 5.5 months with imlunestrant monotherapy (hazard ratio: 0.48; 95% CI: 0.29-0.77; nominal P = .0022).22 This might also suggest that it is reasonable and appropriate to target the ESR1 mutation with an oral SERD in patients with mutations in both ESR1 and PIK3CA.
evERA Breast Cancer: Giredestrant + Everolimus vs Exemestane + Everolimus in ER+/HER2- ABC
Adrienne G. Waks, MD:
Another trial in which an oral SERD was combined with a targeted agent was presented at the European Society for Medical Oncology (ESMO) conference in October 2025. This was the randomized phase III evERA trial evaluating the oral SERD giredestrant plus everolimus vs SoC ET plus everolimus in patients with ER-positive, HER2-negative advanced breast cancer previously treated with a CDK4/6 inhibitor and no prior treatment with chemotherapy (n = 373). The primary endpoint from evERA showed giredestrant plus everolimus improved PFS by investigator in patients with ESR1 mutation (mPFS: 9.99 vs 5.45 months; hazard ratio: 0.38; 95% CI: 0.27-0.54; P <.0001) and in ITT (mPFS: 8.77 vs 5.49 months; hazard ratio: 0.56; 95% CI: 0.44-0.71; P <.0001).23
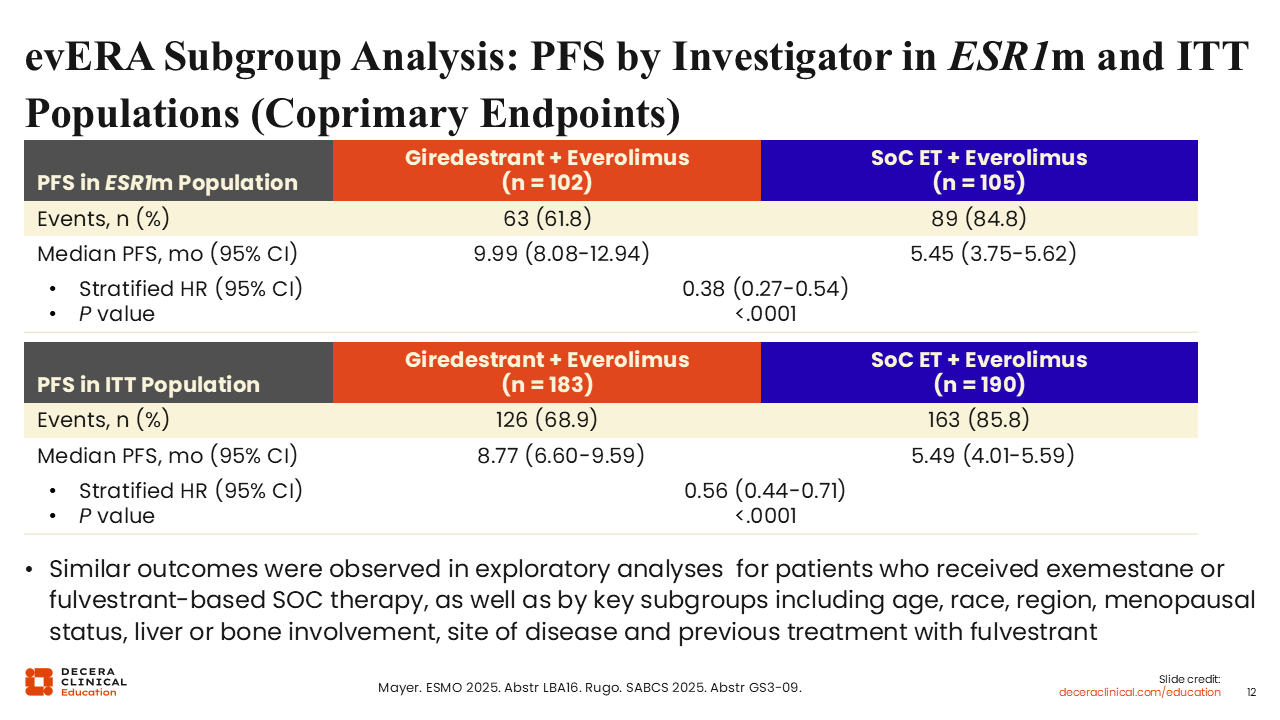
evERA Subgroup Analysis: PFS by Investigator in ESR1m and ITT Populations (Coprimary Endpoints)
Adrienne G. Waks, MD:
At SABCS 2025, we saw updated results from the evERA trial.24 In the ESR1-mutated population, there was a significant improvement in mPFS with giredestrant and everolimus vs SoC ET and everolimus (9.99 vs 5.45 months; stratified hazard ratio: 0.38; 95% CI: 0.27-0.54; P <.0001). Similarly, in the ITT population, which included some participants with ESR1 mutations, there was also a significant improvement in mPFS for the giredestrant-containing arm vs SoC ET (8.77 vs 5.49 months; stratified hazard ratio: 0.56; 95% CI: 0.44-0.71; P <.0001). Again, it appears that single-agent oral SERDs work best in a patient population with an ESR1 mutation and that if combined with a targeted agent, the presence of an ESR1 mutation may be less impactful.
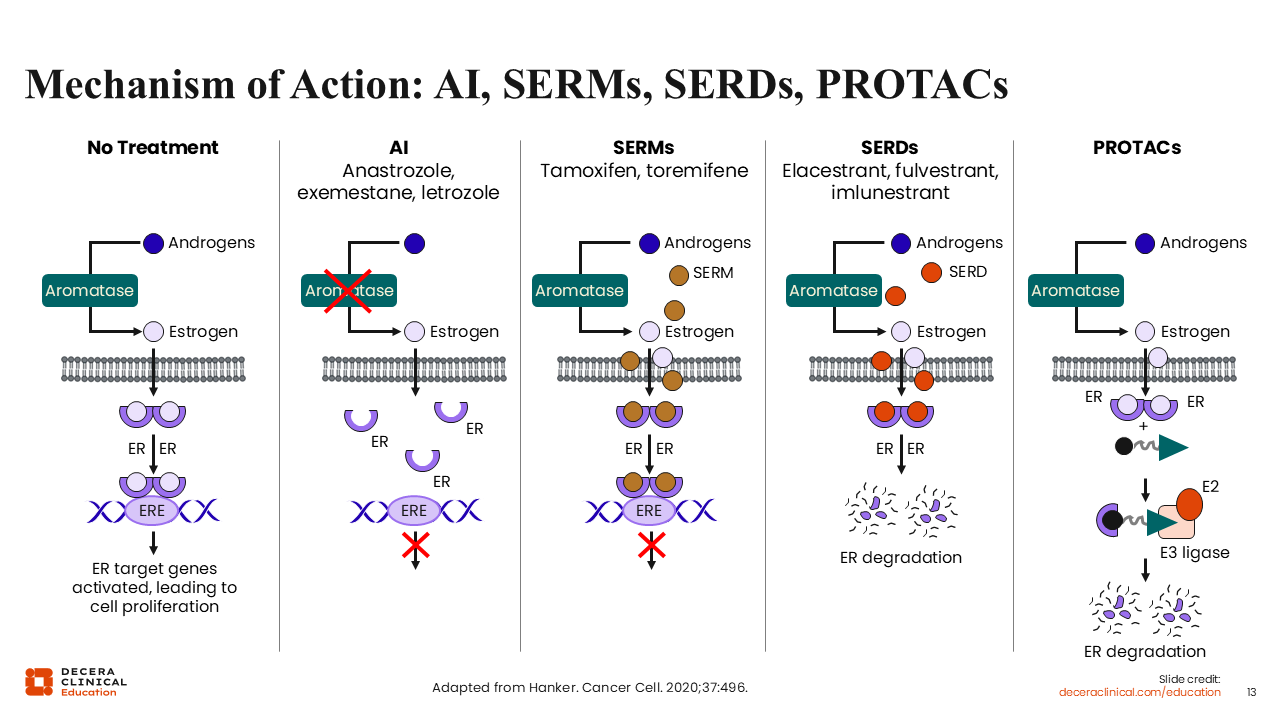
Mechanism of Action: AI, SERMs, SERDs, PROTACs
Adrienne G. Waks, MD:
Another way to inhibit the ER while circumventing aromatase and estrogen itself is with the use of selective estrogen receptor modulators (SERMS: tamoxifen or toremifene). SERMS are a class of competitive ER-binding molecules that can bind to the ER in the place of estrogen and prevent its activation of estrogen-dependent transcription elements in the cell nucleus.25,26
Like elacestrant, fulvestrant, and imlunestrant, SERDs work by binding to the ER and bringing about a conformational change in the ER that ultimately causes it to be recognized by the cell as an abnormal protein and thus targeted for proteasomal degradation.
An emerging new class of ER-targeted agents known as proteolysis targeting chimeras (PROTACs) is engineered so that 1 end of the molecule acts like an estrogen molecule that can dock into the ER receptor, while at the other end of the molecule is a docking site for ubiquitin E3 ligase. This system differs from SERDs in that it does not depend on the conformational change of the ESR1 protein and for the cellular machinery to identify and subsequently target it for degradation. In other words, PROTACs directly bind to ER receptors and facilitate recruitment of protein degradation machinery. Emerging data using preclinical models also suggest that PROTACs generally offer higher degradation efficiency and more potent, sustained, and complete degradation of the ER compared with fulvestrant and oral SERDs.27,28
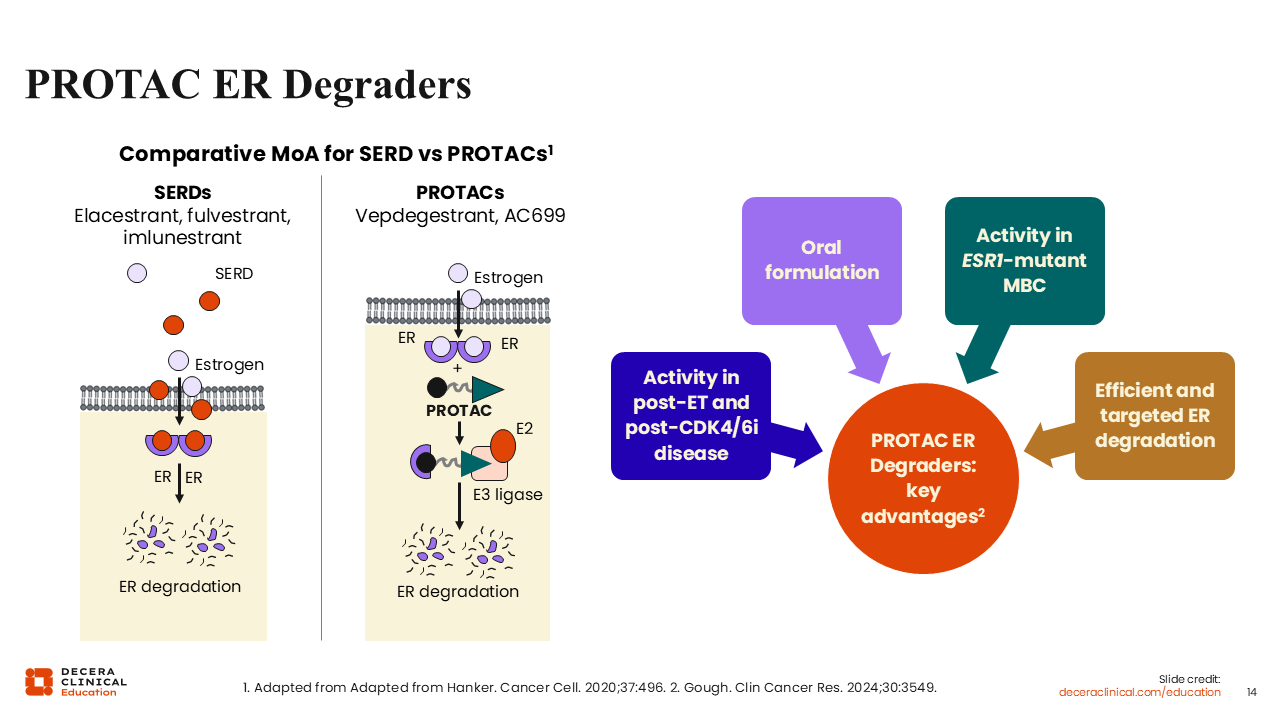
PROTAC ER Degraders
Adrienne G. Waks, MD:
PROTAC ER degraders aim to address an unmet need related to how we can better target the ER. They are theorized to have a number of advantages over traditional SERDs, including oral formulation, activity in the post-ET and CDK4/6 inhibitor disease setting, activity in ESR1-mutated MBC, as well as a more efficient targeted ER degradation.26,28,29
Because of their modular design, PROTAC degraders can be engineered to target multiple different proteins of interest. This opens the door to targeting other pathways and previously nondruggable targets, representing an exciting drug class under investigation.
Vepdegestrant (ARV-471) Background
Rita Nanda, MD:
Vepdegestrant is a selective oral PROTAC ER degrader that targets both wild-type and mutated ER. As a reminder, PROTACS like vepdegestrant bind to the ER and an E3 ubiquitin ligase to induce ubiquitination, which allows proteasomal degradation of the ER. SERDs, on the other hand, induce ER immobilization and/or conformational changes, indirectly recruiting the ubiquitin-proteasome degradation machinery inside cells.
We know that the SERD fulvestrant has some limitations with regard to its intramuscular administration, as well as having limited ER degradation. Even at the optimal dose of fulvestrant, approximately 40% to 50% of the ER is degraded.
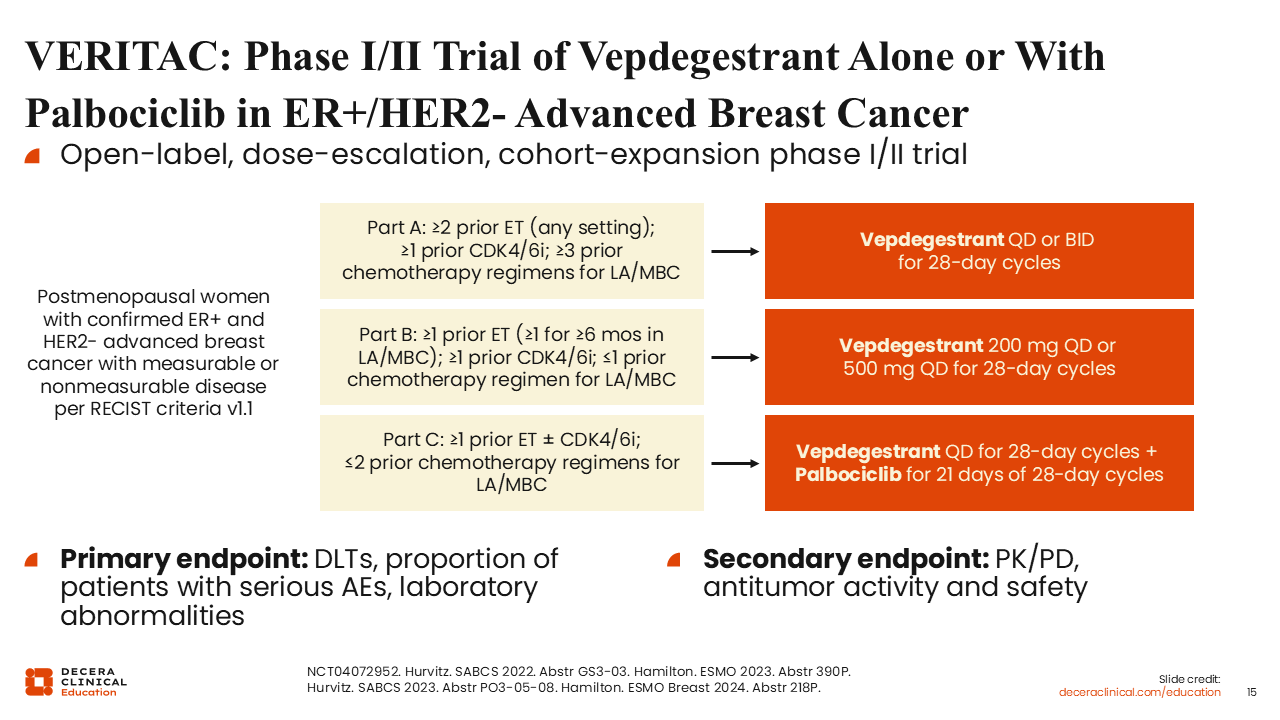
VERITAC: Phase I/II Trial of Vepdegestrant Alone or With Palbociclib in ER+/HER2- Advanced Breast Cancer
Rita Nanda, MD:
I would like to go over some of the data available for vepdegestrant, including for its combination with a CDK4/6 inhibitor.
The ongoing phase I/II VERITAC trial is evaluating vepdegestrant with or without palbociclib in patients with ER-positive/HER2-negative advanced breast cancer.
The study was conducted in 3 parts. Part A included patients with ≥2 prior ET (any setting), ≥1 prior CDK4/6 inhibitor, and ≥3 prior chemotherapy regimens for locally advanced/MBC. In part B, patients had received ≥1 prior ET (≥1 for ≥6 months in the locally advanced/MBC setting), ≥1 prior CDK4/6 inhibitor, and ≤1 prior chemotherapy regimen in the locally advanced/MBC setting. In part C, patients had received ≥1 prior ET with or without a CDK4/6 inhibitor and ≤2 prior chemotherapy regimens for locally advanced or metastatic breast cancer.
The goal of part A was to determine whether once-daily or twice-daily dosing was more efficacious. The goal of part B was to determine the recommended phase II dose, looking at different once-daily schedules of 200 mg or 500 mg. Ultimately, 200 mg was selected. The goal of part C was to evaluate the combination of vepdegestrant plus palbociclib. The primary focus of the cohorts in parts B and C was to look at dose-limiting toxicities to determine the recommended phase II dose and to perform pharmacokinetic and pharmacodynamic analyses.
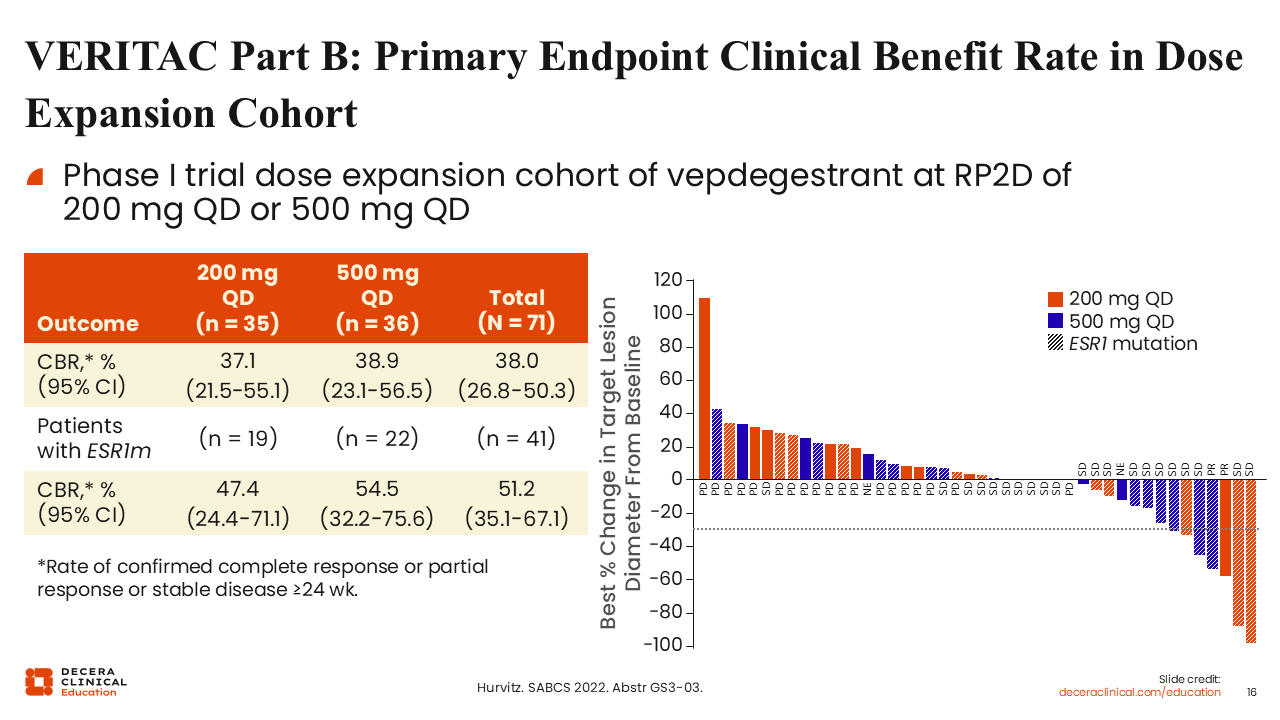
VERITAC Part B: Primary Endpoint Clinical Benefit Rate in Dose Expansion Cohort
Rita Nanda, MD:
The clinical benefit rate (CBR) was defined as the rate of confirmed complete response, partial response, or stable disease ≥24 weeks. The CBR for both of the 2 different doses selected as the potential recommended phase II dose (200 mg QD and 500 mg QD) for patients who had received prior ET, some even chemotherapy, was approximately 38%. The CBR did not vary substantially between the 2 doses, but in patients with ESR1 mutations, it was 47.4% (95% CI: 24.4%-71.1%) for the 200 mg QD group and 54.5% (95% CI: 32.2%-75.6%) for the 500 mg QD group.30
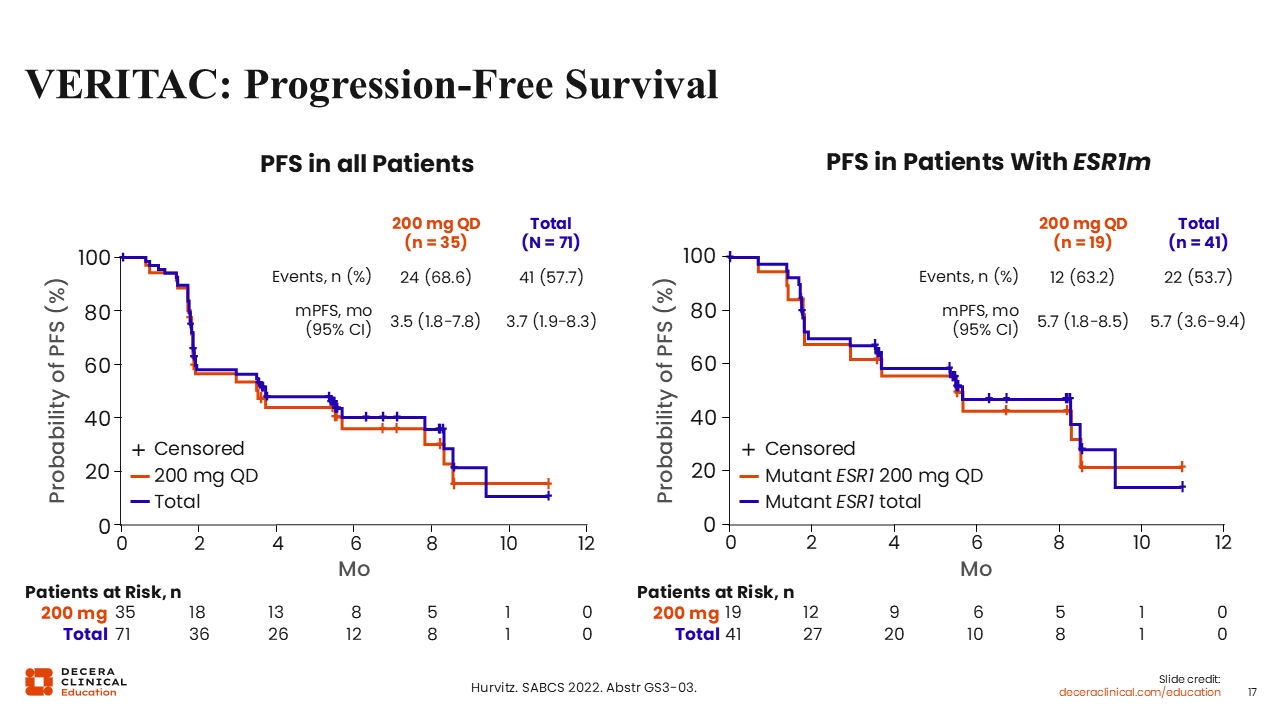
VERITAC: Progression-Free Survival
Rita Nanda, MD:
This was not a randomized study in terms of being able to compare efficacy outcomes between all patients dosed (n = 71) vs those who received the 200 mg QD dose (n = 35). However, looking at the mPFS for all patients vs that of patients receiving the 200 mg QD dose, they were not much different from each other (mPFS: 3.7 vs 3.5 months). Moreover, the mPFS was consistently similar in patients who had ESR1 mutated disease when comparing all patients dosed (n = 41) vs those who received the 200 mg QD dose (mPFS: 5.7 vs 5.7 months).
Taken together, these results demonstrate that PROTAC ER degraders are effective and yield remarkable disease control, including in patients with heavily pretreated disease, albeit in small numbers of patients in this early study.
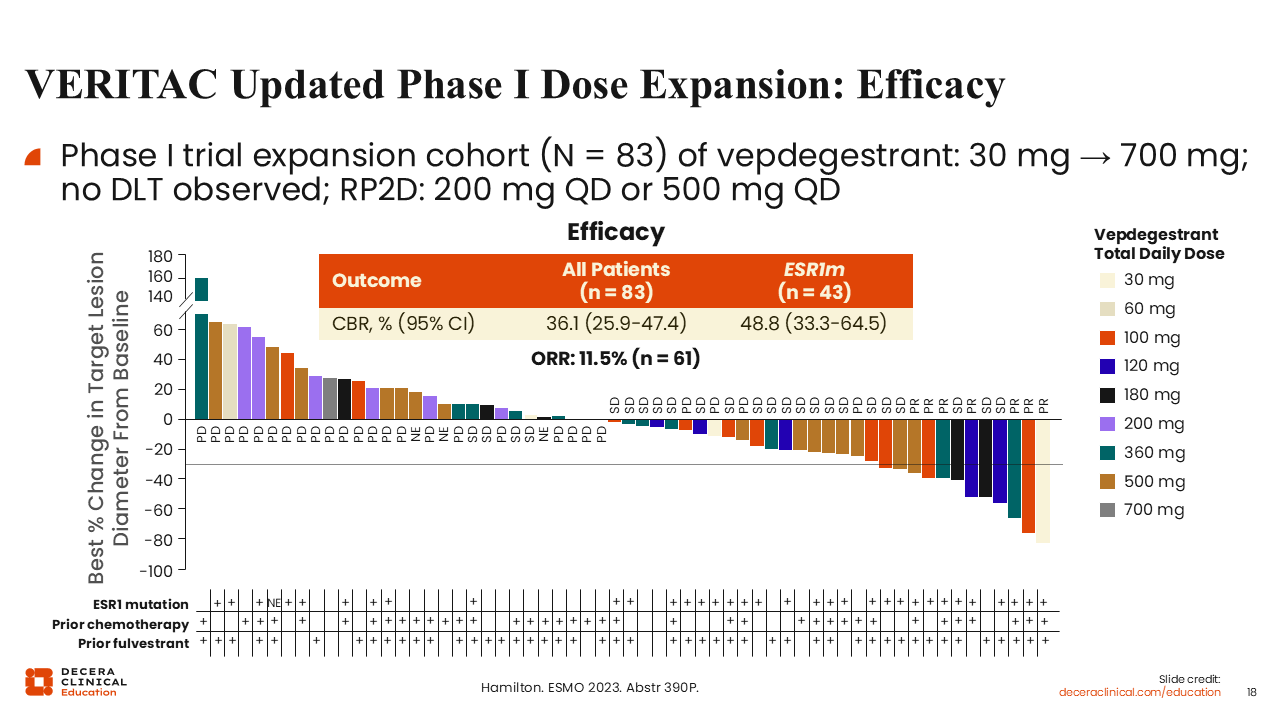
VERITAC Updated Phase I Dose Expansion: Efficacy
Rita Nanda, MD:
If we look at the waterfall plot of the 83 patients who participated in the dose expansion phase—these were patients who had received anywhere from 30 mg QD to 700 mg QD of vepdegestrant—there was no dose-limiting toxicity reported at the highest dose.
Responses were observed regardless of the dose. These results are encouraging, particularly because these patients' tumors were heavily pretreated.
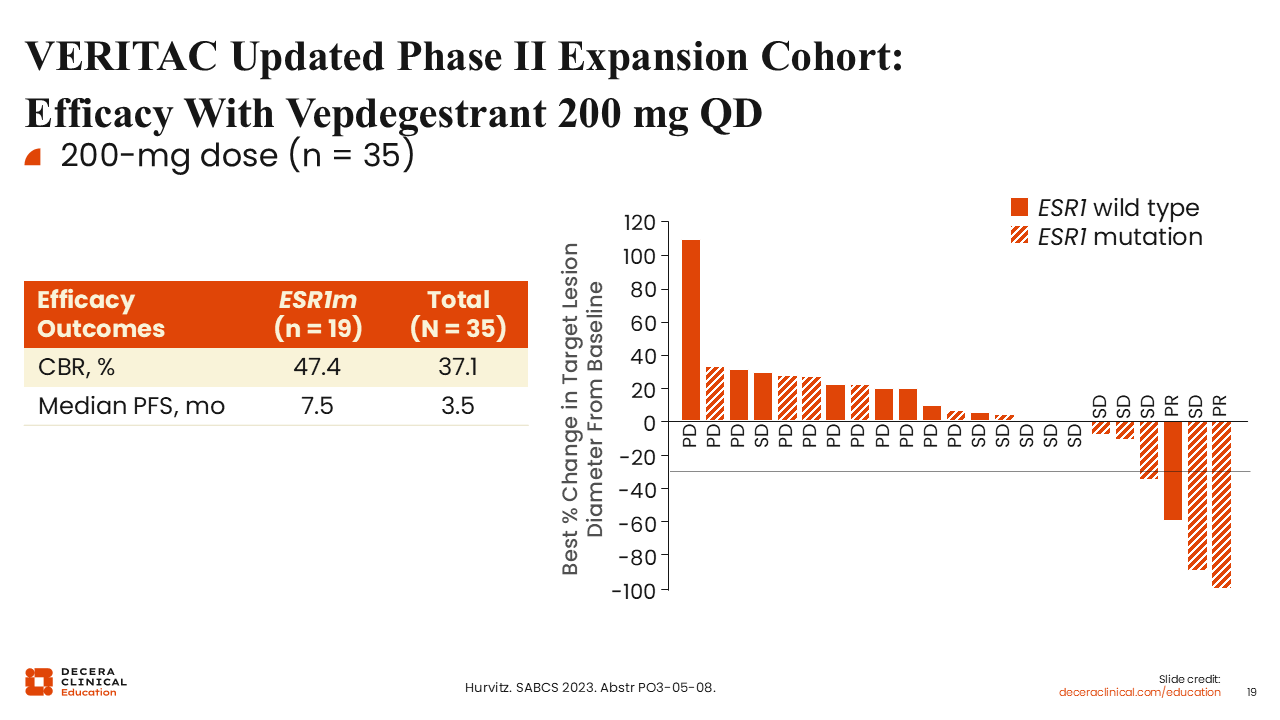
VERITAC Updated Phase II Expansion Cohort: Efficacy With Vepdegestrant 200 mg QD
Rita Nanda, MD:
Shifting our attention to the phase II dose expansion cohort evaluating vepdegestrant 200 mg QD (n = 35), including in patients with ESR1 mutations (n = 19), the CBR was 47.4% for those with an ESR1 mutation and 37.1% overall. mPFS was reported to be 7.5 months in the ESR1-mutated group and 3.5 months overall. These data suggest that similar to oral SERDs, the bulk of the benefit will likely be seen in patients with ESR1 mutations.
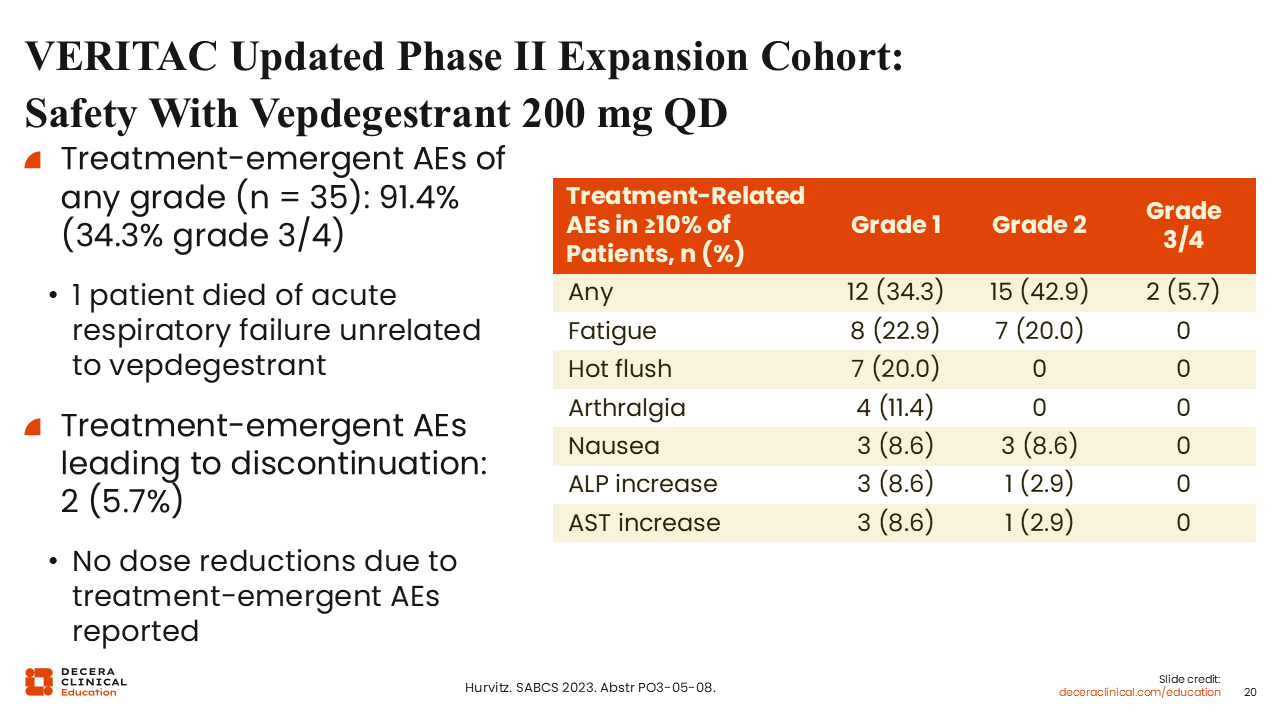
VERITAC Updated Phase II Expansion Cohort: Safety With Vepdegestrant 200 mg QD
Rita Nanda, MD:
Looking at treatment-emergent adverse events (AEs), we see that vepdegestrant is very well tolerated overall. Treatment-related AEs that occurred in at least 10% of patients were predominantly grade 1 and included fatigue (22.9%), hot flush (20.0%), and arthralgia (11.4%), which are often quite manageable. There were no dose reductions reported due to treatment-emergent AEs. There were very few discontinuations, with only 2 of 35 patients (5.7%) discontinuing treatment because of a treatment-emergent AE.
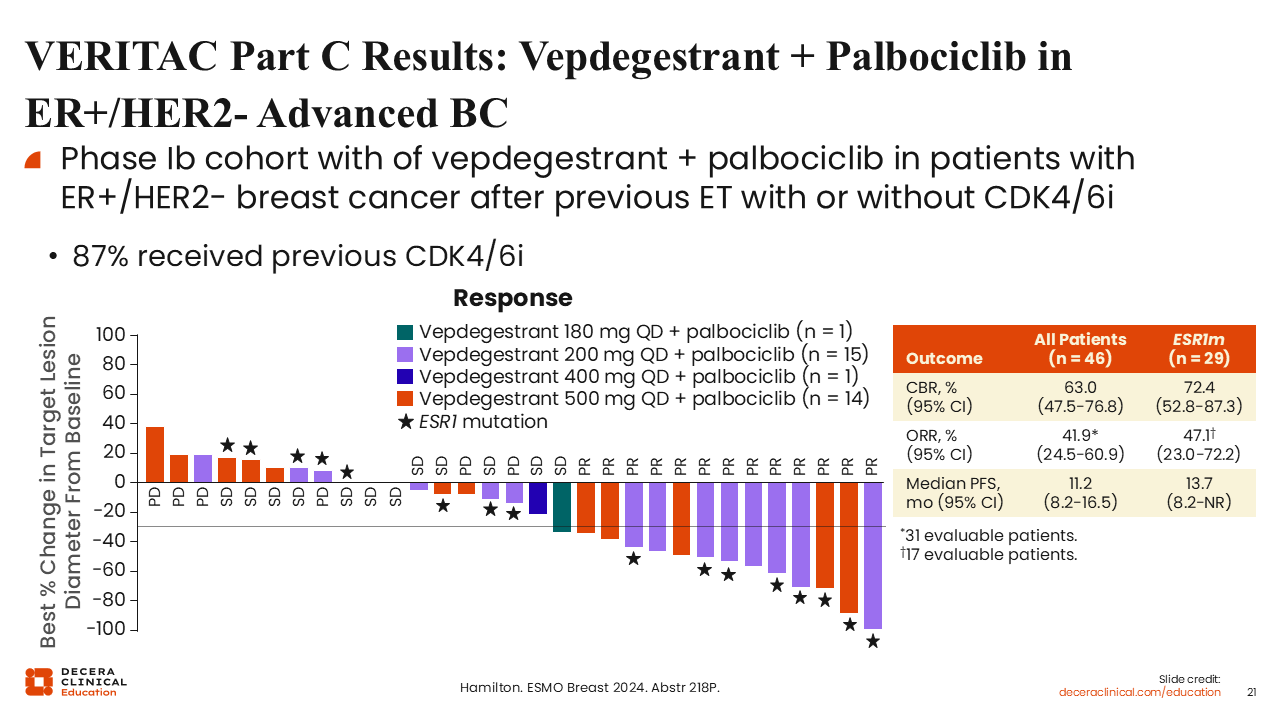
VERITAC Part C Results: Vepdegestrant + Palbociclib in ER+/HER2- Advanced BC
Rita Nanda, MD:
Part C of the study examined vepdegestrant in combination with palbociclib in ER-positive/HER2-negative breast cancer after previous ET with or without a CDK4/6 inhibitor.31
It is important to note that 87% of these patients had received a prior CDK4/6 inhibitor. In this group of patients, the combination of vepdegestrant with palbociclib yielded a CBR of 63% in all patients (n = 46) and approximately 73% in patients with ESR1-mutated tumors (n = 29). The mPFS was 11.2 months in all treated patients and 13.7 months for those with ESR1-mutated tumors. These early efficacy outcomes are very promising.
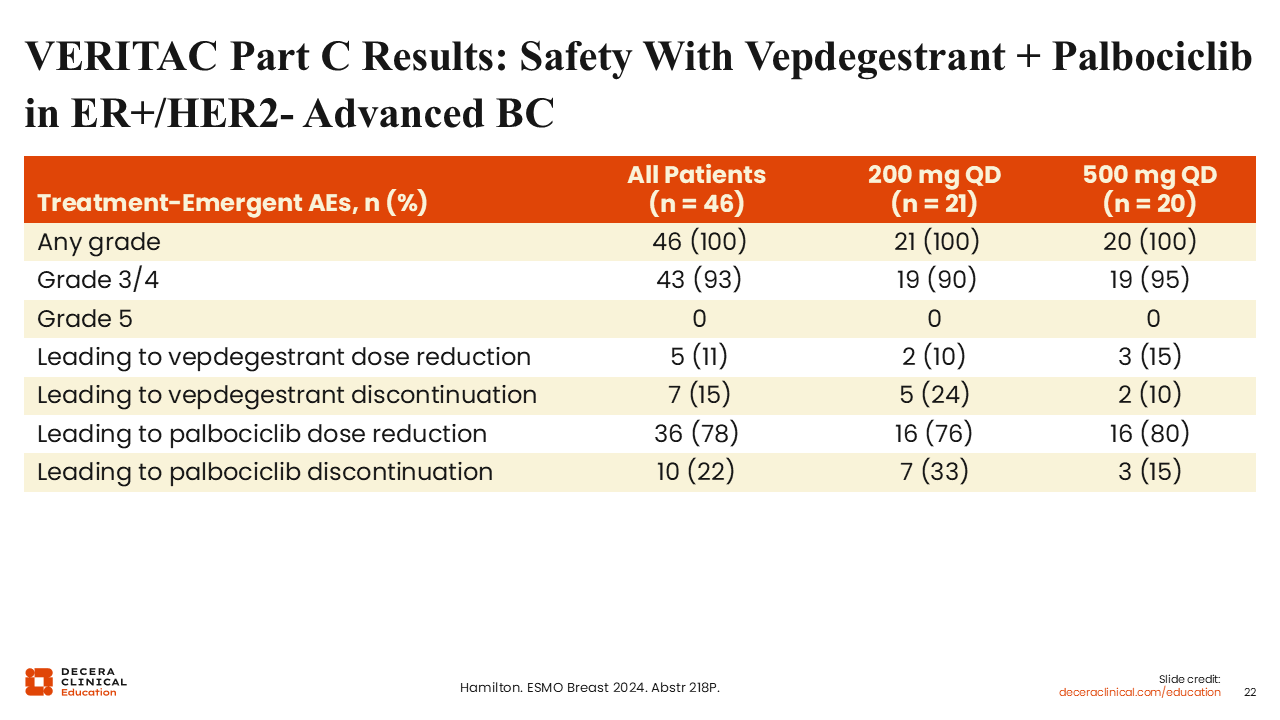
VERITAC Part C Results: Safety With Vepdegestrant + Palbociclib in ER+/HER2 Advanced BC
Rita Nanda, MD:
Regarding safety for the combination of vepdegestrant and palbociclib, the toxicities observed were largely related to palbociclib. All patients experienced AEs. Of 46 patients treated, 43 (93%) experienced grade 3/4 AEs. Overall, 11% of patients required vepdegestrant dose reduction because of AEs, and 15% had vepdegestrant discontinued. Palbociclib dose reductions occurred in 78% of patients and discontinuation was reported in 22%.
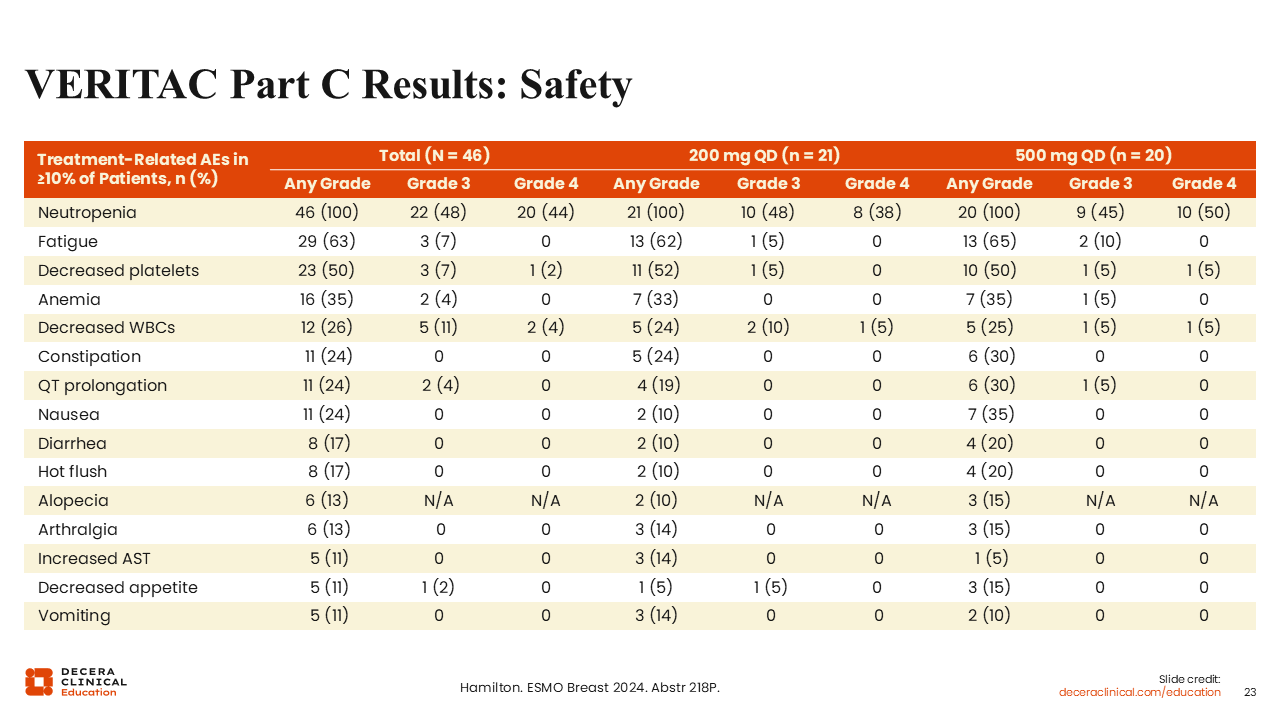
VERITAC Part C Results: Safety
Rita Nanda, MD:
Among the 46 patients treated with the vepdegestrant and palbociclib combination, the rates of any grade, grade 3, or grade 4 AEs did not appear to differ substantially for those receiving vepdegestrant at the 200 mg dose compared with the 500 mg dose.
When we look at the overall safety summary for TRAEs, we see the most common were cytopenias: neutropenia (100%), decreased platelets (50%), anemia (35%), and decreased white blood cell count (26%), all largely related to palbociclib.
Fatigue, which can be caused by palbociclib and certainly by vepdegestrant, was also noted in 63% of patients. For the most part, this combination was well tolerated.
VERITAC-2 Update: Phase III Study of Vepdegestrant vs Fulvestrant in ER+/HER2- Advanced BC
Rita Nanda, MD:
The phase III VERITAC-2 trial compared the activity of vepdegestrant vs fulvestrant in patients with ER-positive/HER2-negative MBC (n = 624), including patients with ESR1 mutations (40%). The primary endpoint was PFS by BICR in all patients and then separately in the ESR1-mutated population. PFS by subgroup was presented at the American Society of Clinical Oncology (ASCO) 2025.
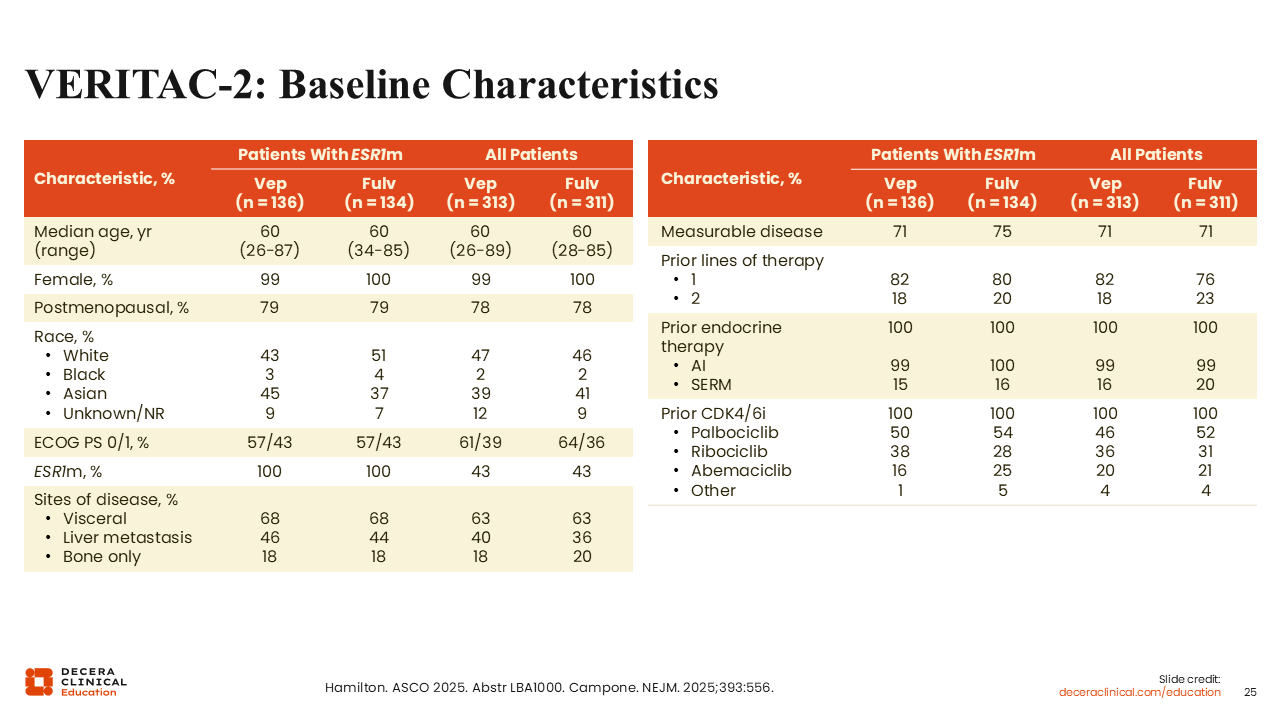
VERITAC-2: Baseline Characteristics
Rita Nanda, MD:
The baseline characteristics were well balanced.32 The median patient age was 60 years in all subgroups, which is typical for this patient population. As previously mentioned, approximately 40% of patients in the study had ESR1-mutated tumors, approximately 70% of patients had visceral disease, 18% had bone-only disease, and approximately 80% had received 1 prior line of systemic therapy (including all patients previously receiving a CDK4/6 inhibitor). Across all patients, in the arm treated with vepdegestrant vs fulvestrant, approximately 46% vs 52% had previously been treated with palbociclib, 36% vs 31% had received ribociclib, and 20% vs 21% had previously received abemaciclib.
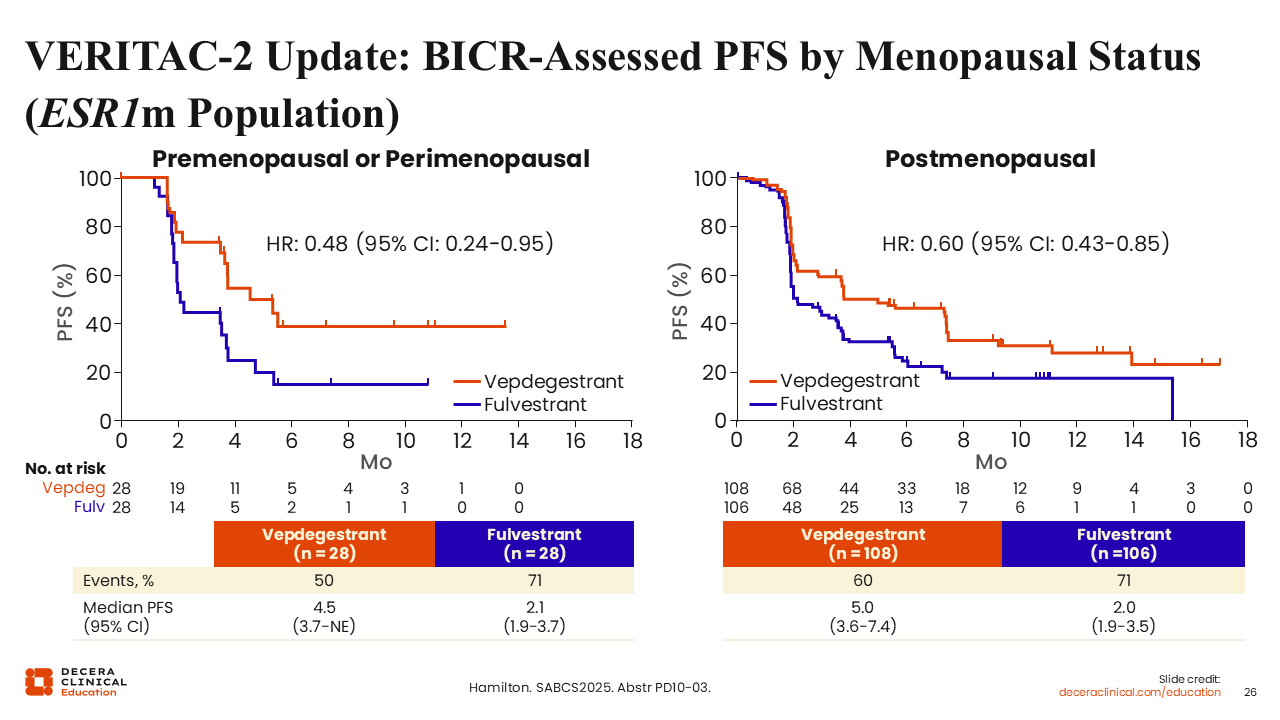
VERITAC-2 Update: BICR-Assessed PFS by Menopausal Status (ESR1m Population)
Rita Nanda, MD:
At the 2025 ASCO annual meeting, investigators showed PFS benefit with vepdegestrant compared with fulvestrant in patients whose tumors harbored an ESR1 mutation (mPFS: 5.0 vs 2.1 months; hazard ratio: 0.57; 95% CI: 0.42-0.77; 2-sided P <.001).33
An update from VERITAC-2 presented at SABCS 2025 reported the efficacy of vepdegestrant compared with fulvestrant by menopausal status in the ESR1-mutated population specifically. The mPFS in patients with premenopausal or perimenopausal status with vepdegestrant vs fulvestrant was 4.5 vs 2.1 months (hazard ratio: 0.48; 95% CI: 0.24-0.95), and in patients with postmenopausal status it was 5.0 vs 2.0 months (hazard ratio: 0.60; 95% CI: 0.43-0.85).
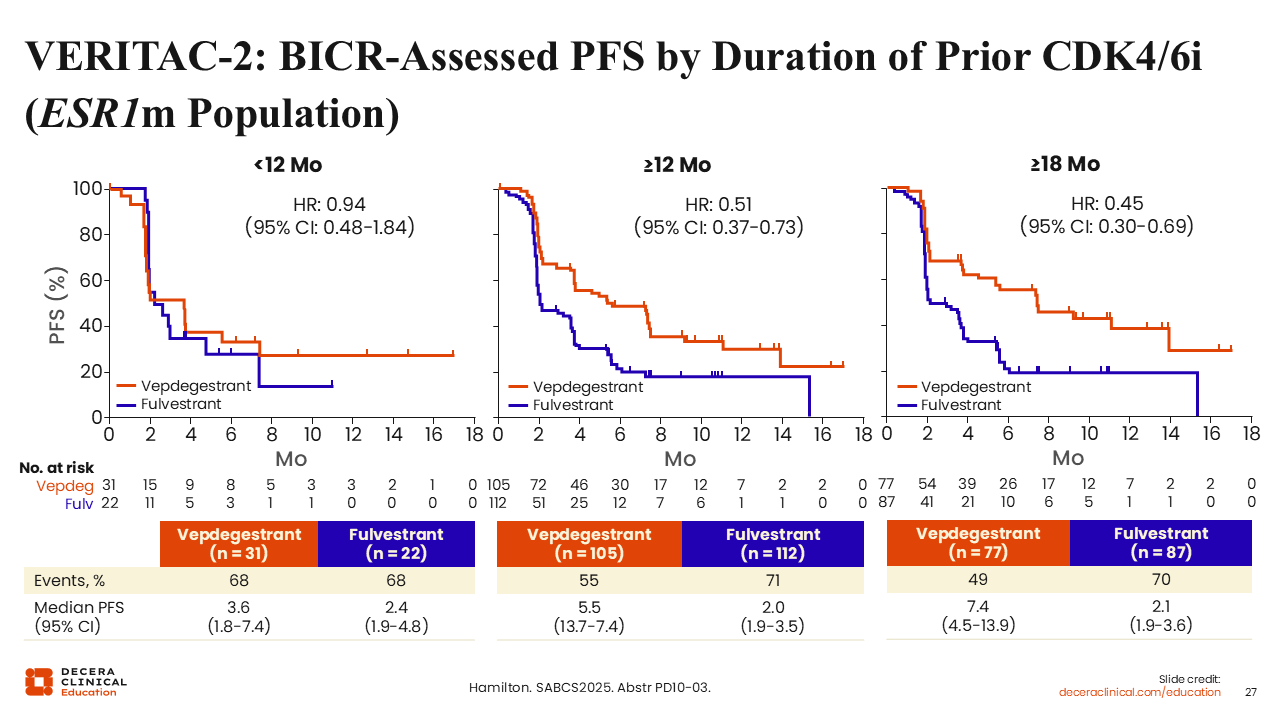
VERITAC-2: BICR-Assessed PFS by Duration of Prior CDK4/6i (ESR1m Population)
Rita Nanda, MD:
Investigators also looked at the mPFS for vepdegestrant compared with fulvestrant by duration of prior CDK4/6 inhibitor (<12 months, ≥12 months, and ≥18 months). In the <12 months group, mPFS for vepdegestrant vs fulvestrant was 3.6 vs 2.4 months (hazard ratio: 0.94; 95% CI: 0.48-1.84); in the ≥12 months group it was 5.5 vs 2.0 months (hazard ratio: 0.51; 95% CI: 0.37-0.73); and in the ≥18 months group it was 7.4 vs 2.1 months (hazard ratio: 0.45; 95% CI: 0.30-0.69).
In other words, the longer the patient was on endocrine-based therapy, the better they responded to vepdegestrant compared with fulvestrant in the ESR1-mutated population.
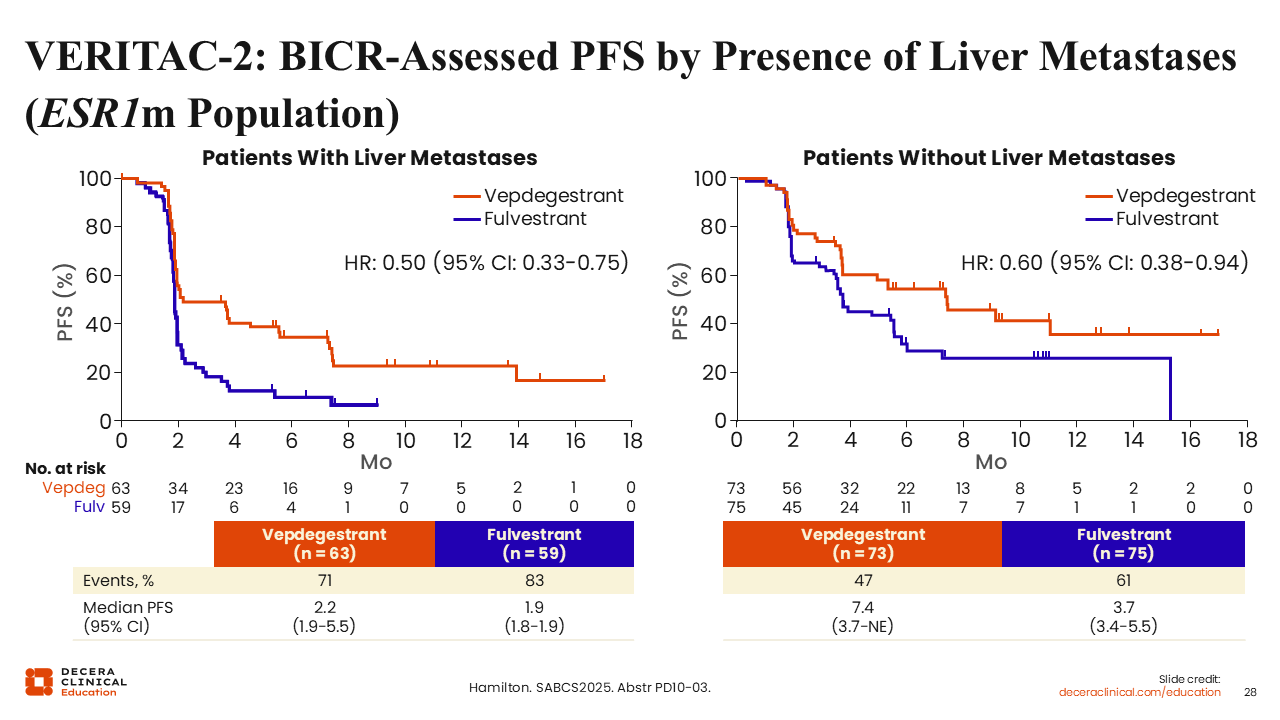
VERITAC-2: BICR-Assessed PFS by Presence of Liver Metastases (ESR1m Population)
Rita Nanda, MD:
Looking at the ESR1-mutated population with liver metastases, patients also did better with vepdegestrant. In the patient subgroup with liver metastases, mPFS for vepdegestrant vs fulvestrant was 2.2 vs 1.9 months (hazard ratio: 0.50; 95% CI: 0.33-0.75).
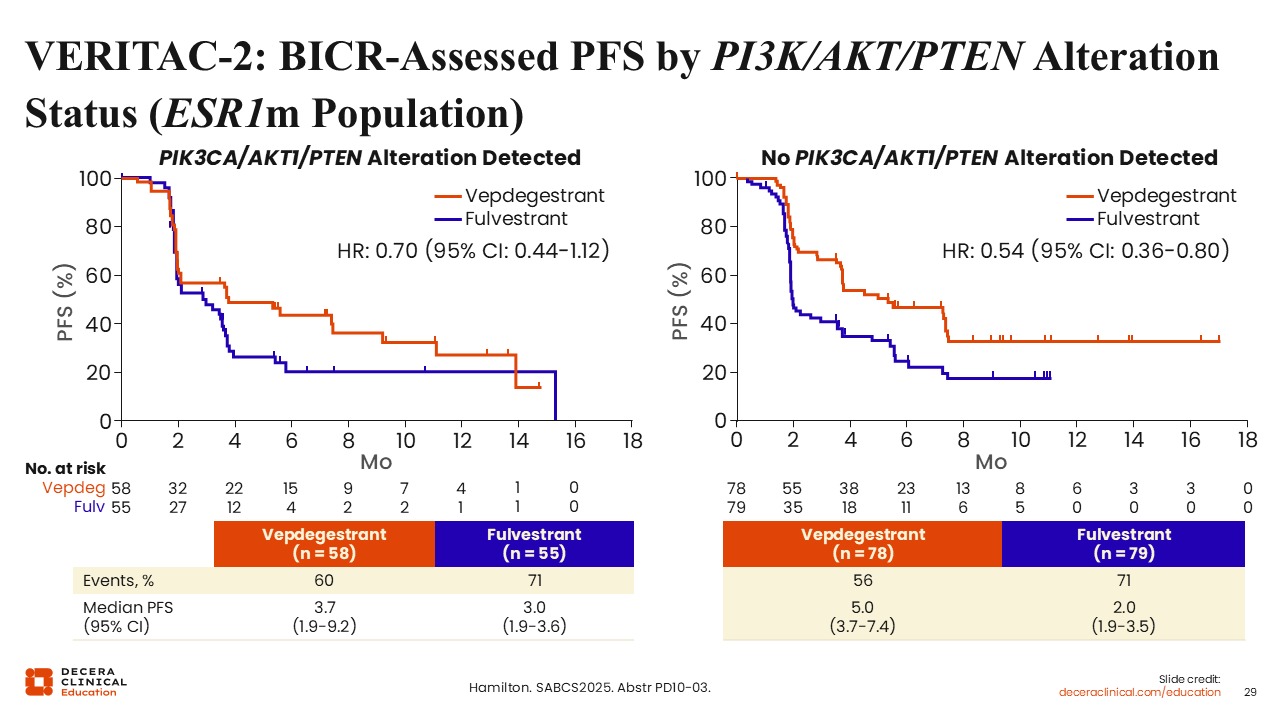
VERITAC-2: BICR-Assessed PFS by PI3K/AKT/PTEN Alteration Status (ESR1m Population)
Rita Nanda, MD:
Investigators also reported outcomes by presence or absence of a PIK3CA/AKT/PTEN alteration within the ESR1-mutated population. The mPFS was 3.7 vs 3.0 months (hazard ratio: 0.70; 95% CI: 0.44-1.12) for the subgroup with a PIK3CA/AKT/PTEN alteration detected, and mPFS was 5.0 vs 2.0 months (hazard ratio: 0.54; 95% CI: 0.36-0.80) in those where PIK3CA/AKT/PTEN alterations were not detected.
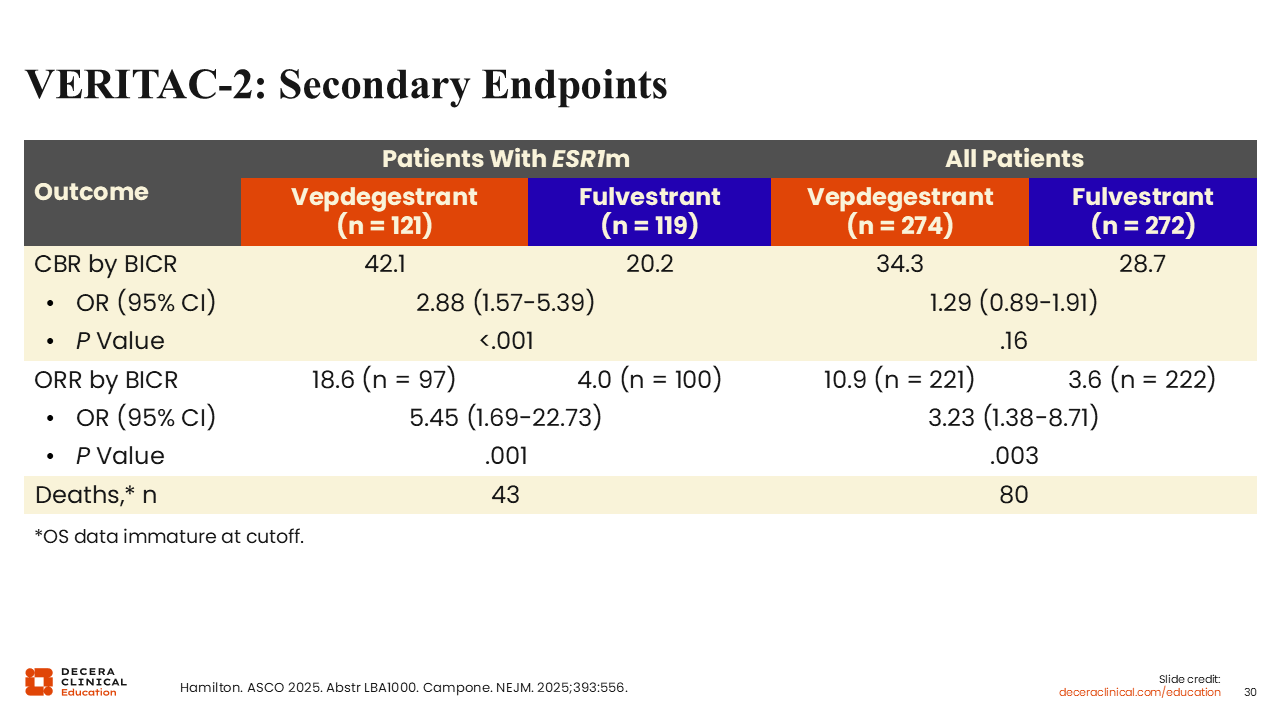
VERITAC-2: Secondary Endpoints
Rita Nanda, MD:
In the ESR1-mutated population, the CBR with vepdegestrant was 42.1% vs 20.2% with fulvestrant, and the overall response rate (ORR) was18.6% vs 4.0%, respectively.
In the overall patient population, however, the CBR with vepdegestrant was 34.3% vs 28.7% with fulvestrant, and the ORR was 10.9% vs 3.6%, respectively. Again, more modest differences in the overall population.32,33
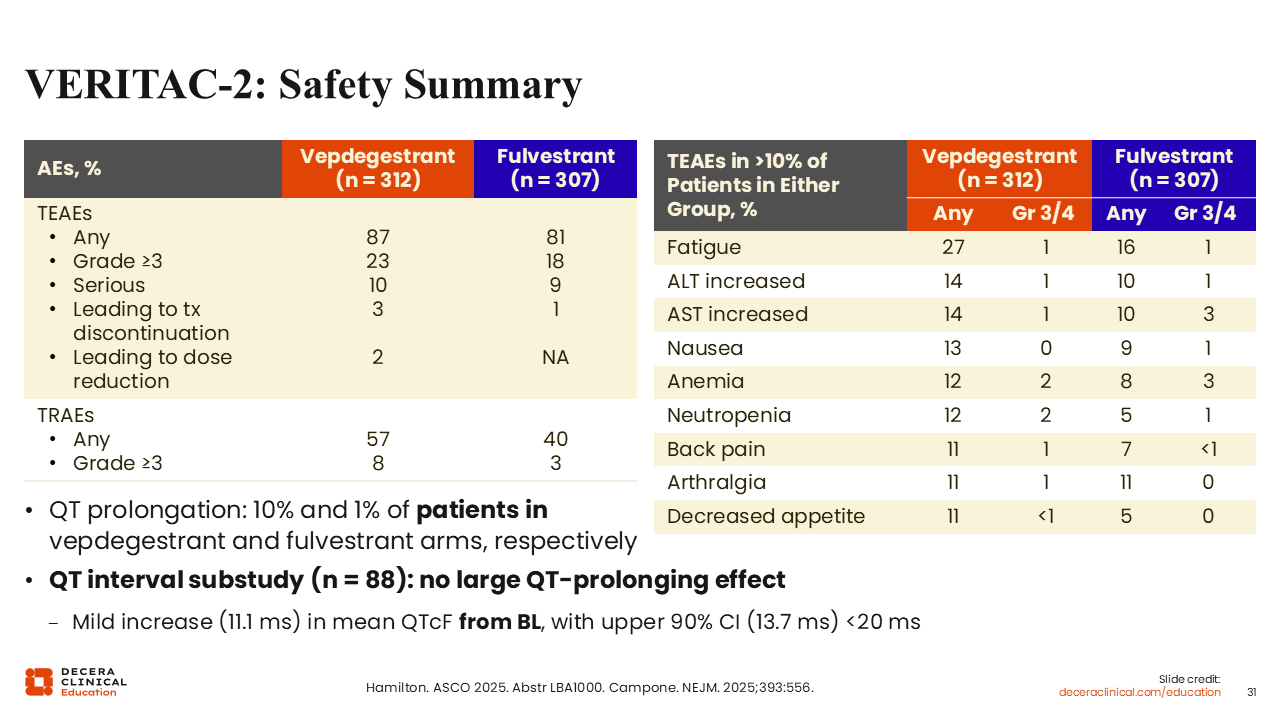
VERITAC-2: Safety Summary
Rita Nanda, MD:
Looking at safety, toxicities were mostly grade 1/2 and required discontinuation in 3% and 1%, respectively, in the vepdegestrant arm and fulvestrant arm, with dose reductions in 2% of patients treated with vepdegestrant. Fatigue was the most common TEAE seen with both vepdegestrant (27%) and fulvestrant (16%), followed by ALT (14% vs 10%) and AST (14% vs 10%) elevation, nausea (13% vs 9%), anemia (12% vs 8%), and neutropenia (12% vs 5%). Most cases were grade 1 or 2 and manageable. The proportion of patients in the vepdegestrant vs fulvestrant arm with grade ≥3 AEs was 23% and 18%, respectively.
Of note, QT prolongation was reported in 10% and 1% of patients receiving vepdegestrant and fulvestrant, respectively.
VERITAC-3: Vepdegestrant + Palbociclib vs Letrozole +Palbociclib in ER+/HER2- Advanced BC
Rita Nanda, MD:
The global, multicenter, open-label, phase III VERITAC-3 trial is evaluating vepdegestrant with palbociclib vs letrozole with palbociclib in patients with
ER-positive/HER2-negative locally recurrent or metastatic breast cancer with no prior treatment in the advanced disease setting, and no previous treatment in any setting with a CDK4/6 inhibitor, vepdegestrant, fulvestrant, elacestrant, or other investigational ER degraders or antagonists, and no disease recurrence ≤12 mo after completing adjuvant ET (N = 1130). The primary endpoint is PFS, and secondary endpoints include quality of life, ORR, duration of response (DoR), CBR, and overall survival. The rationale for the addition of palbociclib is related to concerns about QT prolongation and challenges associated with combining vepdegestrant with other CDK4/6 inhibitors.
Other related trials include the ongoing phase I/II trial, which is evaluating vepdegestrant combined with a KAT6 inhibitor (NCT04606446). This trial is currently enrolling. Participants must have experienced disease progression following at least 1 prior line of CDK4/6 inhibitor therapy and at least 1 prior line of ET; participants also must have not received more than 3 lines of systemic therapies, including up to 1 line of cytotoxic chemotherapy for visceral disease in the advanced or metastatic setting, but previous use of fulvestrant is allowed.
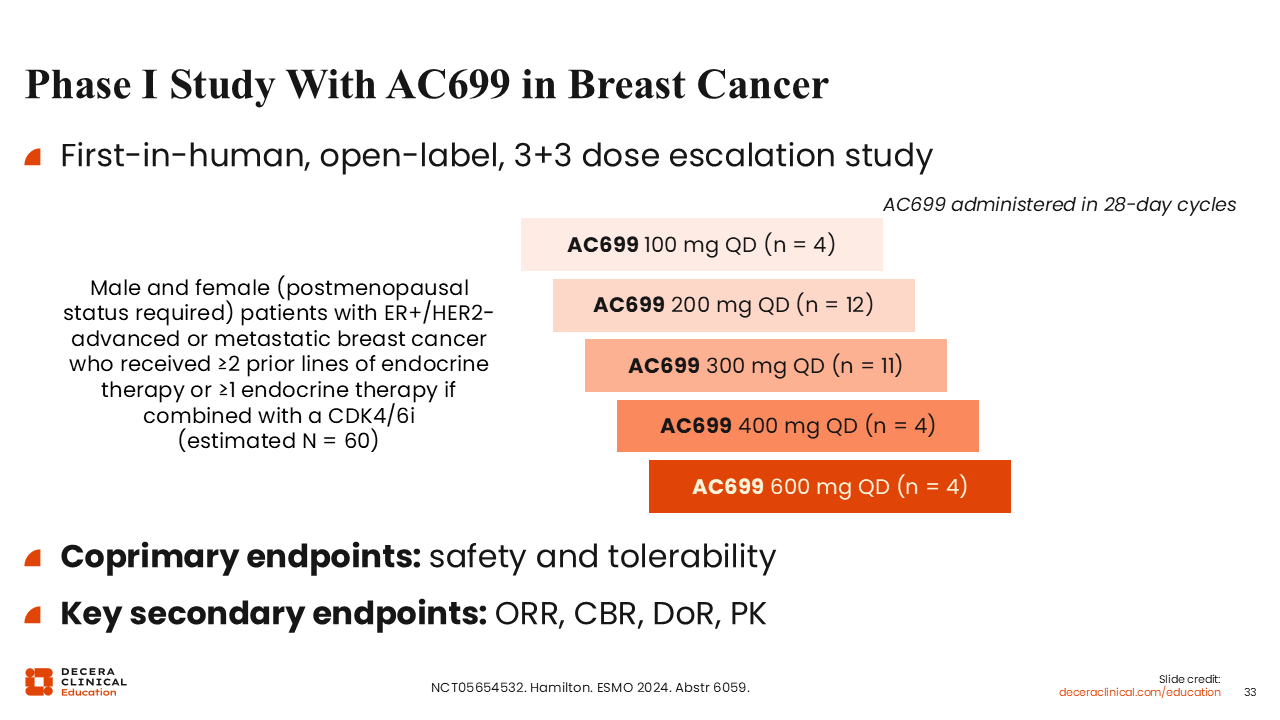
Phase I Study With AC699 in Breast Cancer
Rita Nanda, MD:
Another PROTAC ER degrader called AC699 is also under development (NCT05654532). Although the development strategy for this PROTAC agent moving forward remains unclear, we have access to early data from a first-in-human phase I study presented at ESMO 2024 including safety and tolerability analyses, as well as secondary efficacy endpoints.34
The study explored AC699 doses ranging from 100-600 mg QD in male and female patients (postmenopausal status required) with ER-positive/HER2-negative advanced or metastatic breast cancer who received ≥2 prior lines of ET or ≥1 line of ET if combined with a CDK4/6 inhibitor (Planned N = 60). The coprimary endpoints were safety and tolerability.
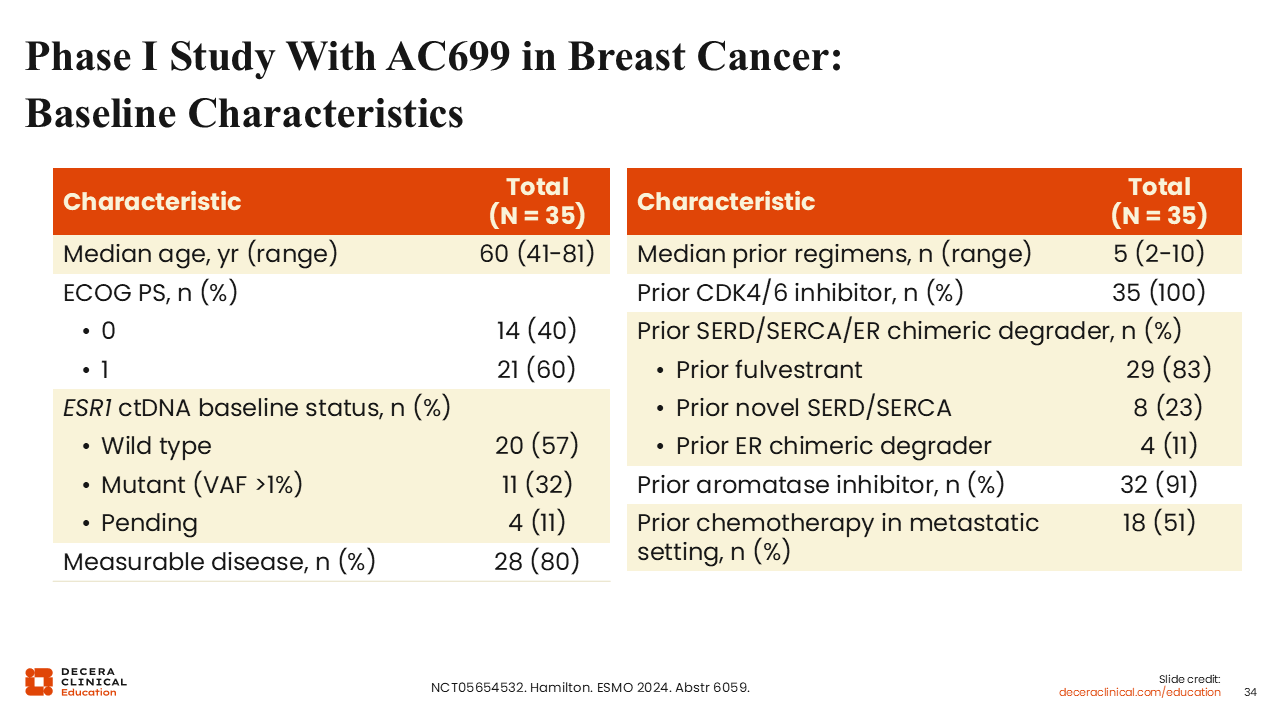
Phase I Study With AC699 in Breast Cancer: Baseline Characteristics
Rita Nanda, MD:
A total of 35 patients were enrolled, with a median age of 60 years (range: 41-81 years). The median number of previous lines of therapy was 5 (range: 2-10). Patients had received previous treatment with fulvestrant (83%), novel SERD/selective estrogen receptor covalent antagonist (23%), and previous ER chimeric degrader (11%). All patients had previously received a CDK4/6 inhibitor, 91% had received a prior AI, and 51% had received chemotherapy in the metastatic setting.
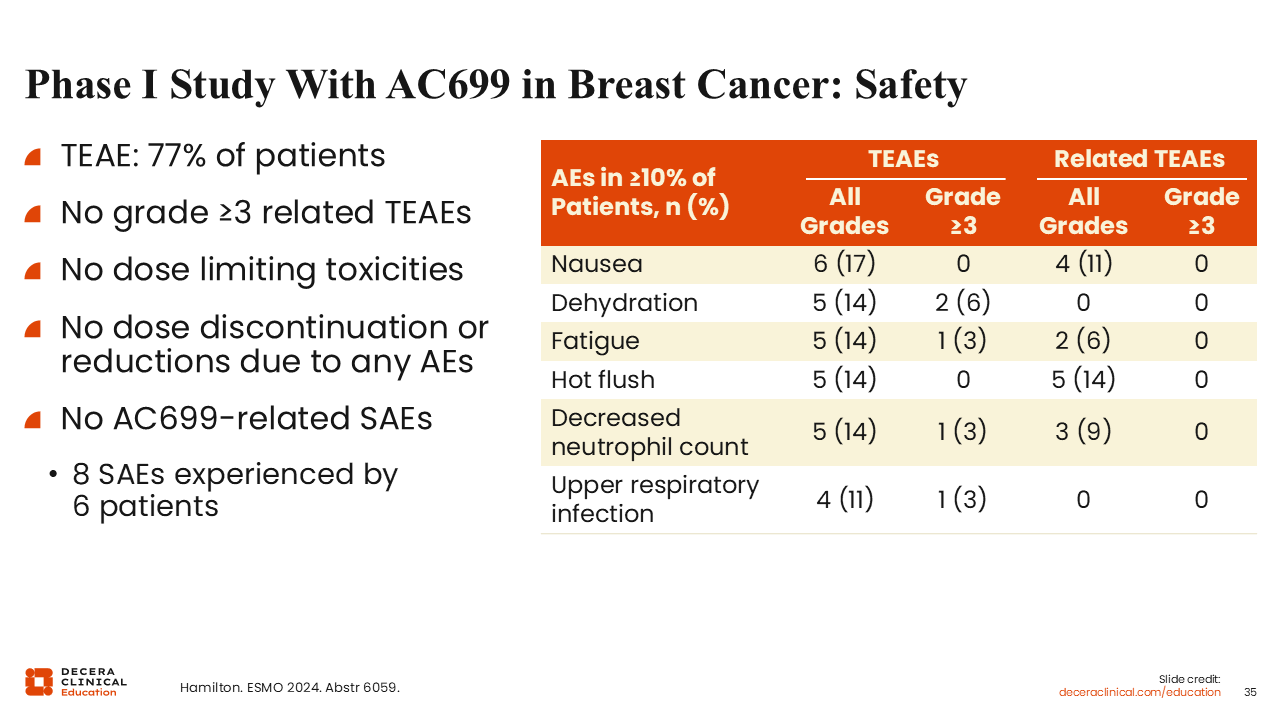
Phase I Study With AC699 in Breast Cancer: Safety
Rita Nanda, MD:
The overall rates of AEs observed with AC699 were consistent with those reported with vepdegestrant, with most being grade 1-2. The most common AEs reported in ≥10% of patients included nausea (17%), fatigue (14%), dehydration (14%),
and hot flush (14%). Grade ≥3 AEs reported included dehydration (6%), fatigue (3%), decreased neutrophil count (3%), and upper respiratory infection (3%).
Investigators also reported there were no dose-limiting toxicities, discontinuations, or reductions due to AEs.
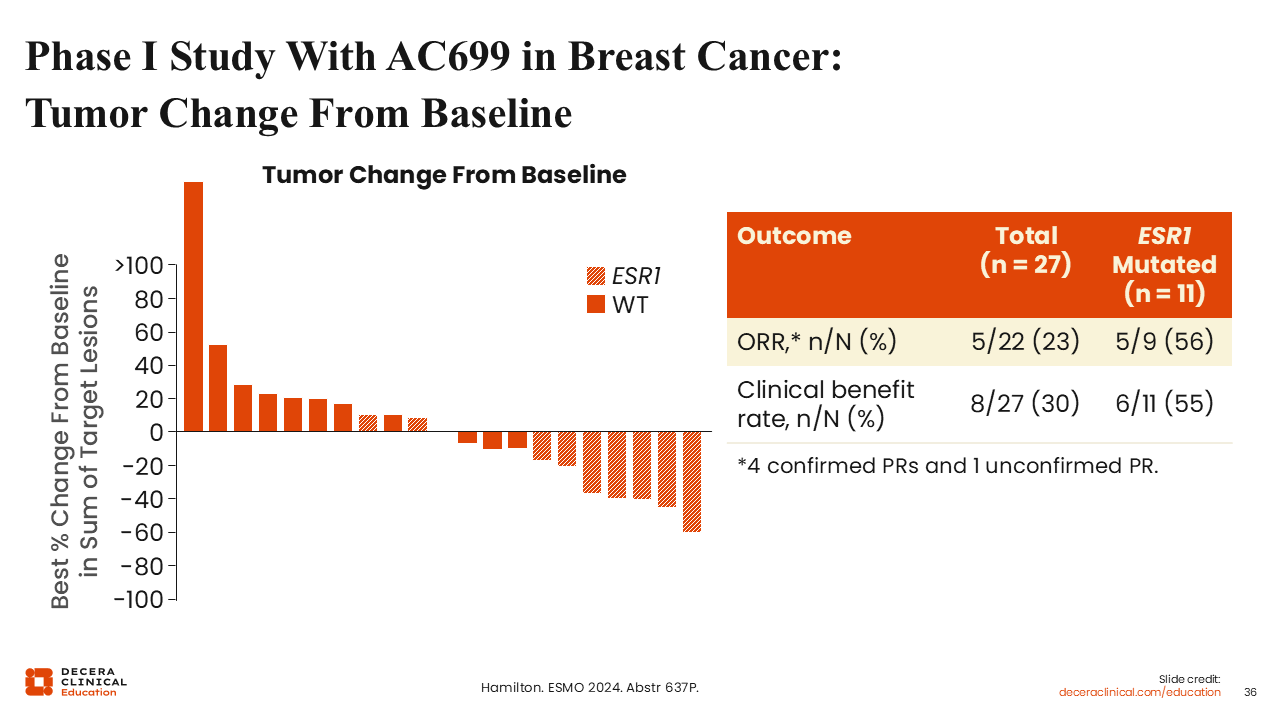
Phase I Study With AC699 in Breast Cancer: Tumor Change From Baseline
Rita Nanda, MD:
Regarding responses with AC699 in this previously treated population, 11 of 27 patients who were evaluable for response had ESR1 mutations, and 5 of 9 evaluable for response had an ORR of 56% and a CBR of 55%. In the overall population, 22 patients were evaluable for analysis, and these patients achieved an ORR of 22% and a CBR of 30%. These data are from very small numbers, but overall, we can discern a common theme for these novel PROTAC ER degraders, appearing to have higher activity in patients with ESR1-mutated disease.
What Is Next?
Rita Nanda, MD:
Predictive biomarkers are crucial for improving patient selection, not just for PROTAC ER degraders but for a variety of other therapies used to treat patients with ER-positive/HER2-negative disease.
We need to better understand which patients with ESR1 mutations do well following progression on or after ET with or without CDK4/6 inhibitors. As we are shifting more to combination strategies, we should remember that some patients do very well on ET alone. At this time, we identify these patients by asking how long they were on their initial ET, and based on that, whether they would benefit more from a combination approach.
At this time, long-term efficacy data for PROTAC ER degraders are immature and/or yet to be reported. It will be very important to see these results. PROTAC clinical trials may be designed in ways that improve upon the approaches used in oral SERD trials.
Questions on PROTAC Therapies From the Audience
If approved, where might PROTAC ER degraders be implemented in the treatment landscape?
Rita Nanda, MD:
The answer is certainly anywhere we have considered oral SERDs and where other endocrine-based therapies currently play a role—in the adjuvant setting or, I would argue, even in the neoadjuvant setting. The VERITAC-3 trial will begin to help address this question.
Is there a role for PROTACs in the frontline setting with a CDK4/6 inhibitor or in the second line and beyond?
Rita Nanda, MD:
Most earlier studies I reviewed here were performed in previously treated patients, and we definitely saw efficacy in patients with ESR1 mutations. Combination strategies do appear to work better. Thus, some of the ongoing trials will be looking at combination strategies moving forward.
Conclusion and Key Takeaways
Rita Nanda, MD:
Despite progression on standard ETs including CDK4/6 inhibitors, a number of patients remain endocrine sensitive. Data from the EMERALD trial and vepdegestrant studies indicate that a 1-year threshold may help predict which patients will benefit from additional endocrine-based therapy. Patients who have a longer DoR to first-line ET treatment appear more likely to benefit from another endocrine-based strategy. Novel estrogen-targeting drugs are needed to improve outcomes for patients with progression on or after previous ET. The mechanism of action of PROTAC agents that degrade the ER is very distinct from SERDs. Moving forward, we will be investigating combination strategies with other targeted therapies.
Vepdegestrant has shown activity both alone and in combination with palbociclib. The combination resulted in an mPFS of more than 11 months in patients regardless of ESR1 mutation status. Of note, 87% of these patients had previously received a CDK4/6 inhibitor. Combination with CDK4/6 inhibitors appears safe and could represent a new therapeutic approach in the frontline setting if the results of the VERITAC-3 trial are positive. As the treatment landscape continues to evolve, it is important that we watch for updates on ongoing trials and evolving treatment strategies involving PROTAC ER degraders for patients with advanced HR-positive disease.