CE / CME
Advancing Inflammatory Bowel Disease Management With Precision Medicine and Emerging Data
Physician Assistants/Physician Associates: 0.75 AAPA Category 1 CME credit
Nurse Practitioners/Nurses: 0.75 Nursing contact hour
Physicians: maximum of 0.75 AMA PRA Category 1 Credit™
Released: May 08, 2025
Expiration: May 07, 2026
Activity
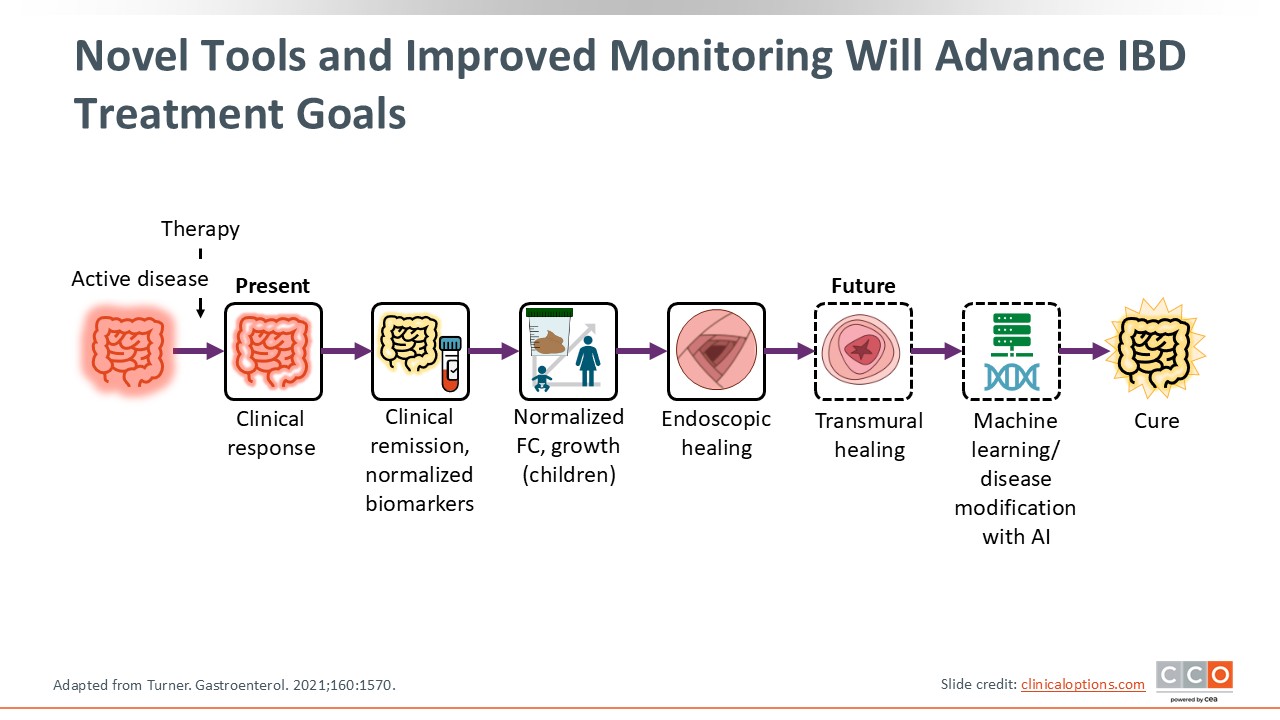
Novel Tools and Improved Monitoring Will Advance IBD Treatment Goals
We have several biomarkers at our disposal right now, including serum and stool biomarkers like CRP and FCP. However, there are several other surrogates of disease activity, such as endoscopy, that can help along the way. Then there are some aspirational targets like transmural healing and perhaps molecular healing that will further be associated with improvements in disease activity and reduction in future complications. This slide highlights the current and aspirational tools for monitoring IBD activity beyond endoscopic assessment. This is important to consider as we move forward and our thinking about treat to target evolves.10
Noninvasive Imaging Modalities
Let us review the imaging modalities that are available and can be integrated into treat to target. Again, here, we are not just talking about symptoms, biomarkers, or endoscopic improvement; rather, we are talking about a noninvasive approach to monitoring that will inform treatment adjustments to achieve transmural or mucosal healing.
First is intestinal ultrasound, which is finally increasing in utilization among US HCPs. This is a noninvasive, bedside, and real-time imaging technique that can accurately assess bowel wall thickness and vascularization, which are surrogates for active inflammation. Data show these findings closely correlate with endoscopic severity in UC and are effective in monitoring disease complications. Furthermore, intestinal ultrasound is an effective monitoring strategy for assessing treatment response. Before initiating treatment, if patients have a thickened bowel wall in UC or thickened ileum in CD, HCPs can assess them at 3-month to 6-month intervals to evaluate improvement. If patients do not have improvement at these time points, HCPs need to make treatment adjustments.
MRE is an extremely sensitive and detailed image of the gastrointestinal tract. It is the preferred method for evaluating complications of CD in particular. Of note, MRE is expensive, takes time, and requires intravenous and oral contrast. However, no radiation exposure is required with this technology. It is much more complicated, cumbersome, and expensive vs bedside intestinal ultrasound.
Like MRE, CT enterography is highly sensitive for detecting transmural complications. Because it requires radiation exposure, HCPs should limit its use, so patients do not have repeated exposures. Whereas intestinal ultrasound operators need training and MRE has limited ability, CT is widely available in the United States and provides a lot of valuable information.14-18
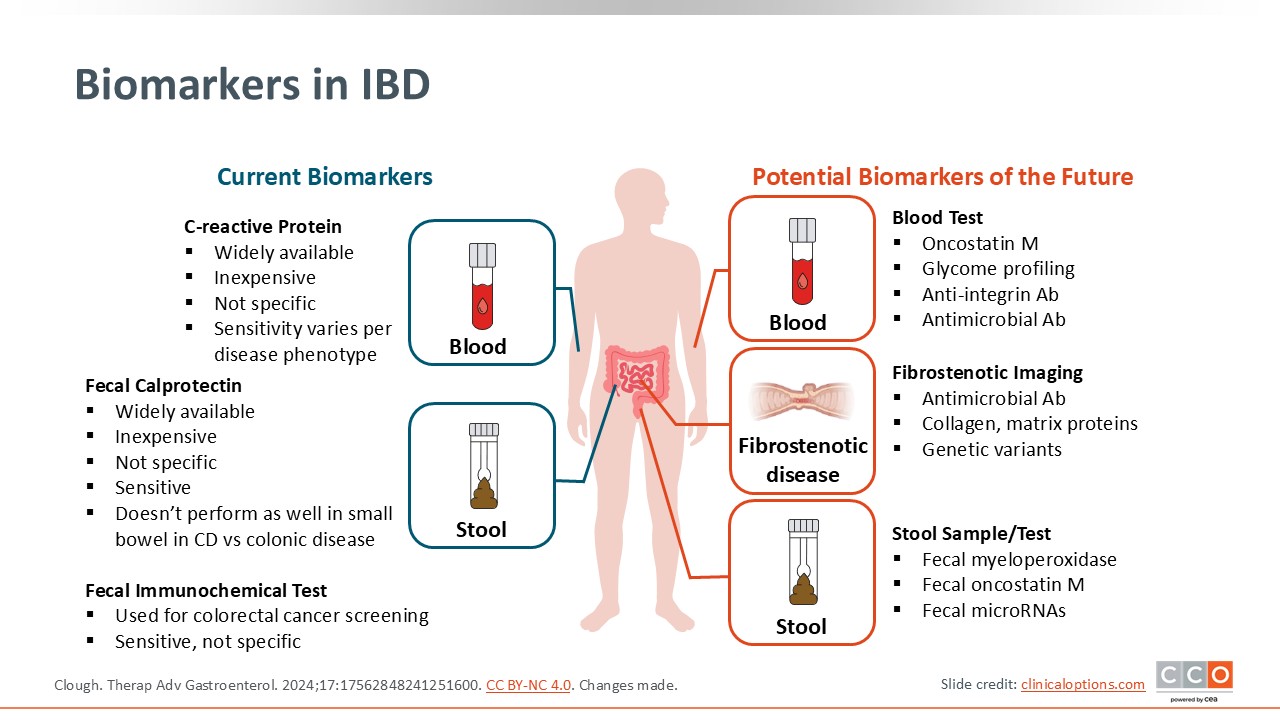
Biomarkers in IBD
Currently, HCPs use serum CRP, which can be checked anywhere and everywhere. In addition, FCP is also broadly available but can be cumbersome because patients need to either produce a stool sample in the office or lab or they need use a stool kit at home and bring that back to the office or lab. However, FCP is more sensitive and specific for detecting intestinal inflammation. Whereas patients with an infection or another inflammatory condition can have an elevated CRP, FCP is specific to the intestinal tract.
In the future, there may be many other stool and serum biomarkers that can be measured, as these are being explored now. Research is underway to understand how they correlate with disease activity and complications like fibrosis. We are likely to get much more sophisticated serum and stool biomarkers in the future that will help us understand more in depth how patients are doing and whether treatment adjustments will improve their outcomes.19
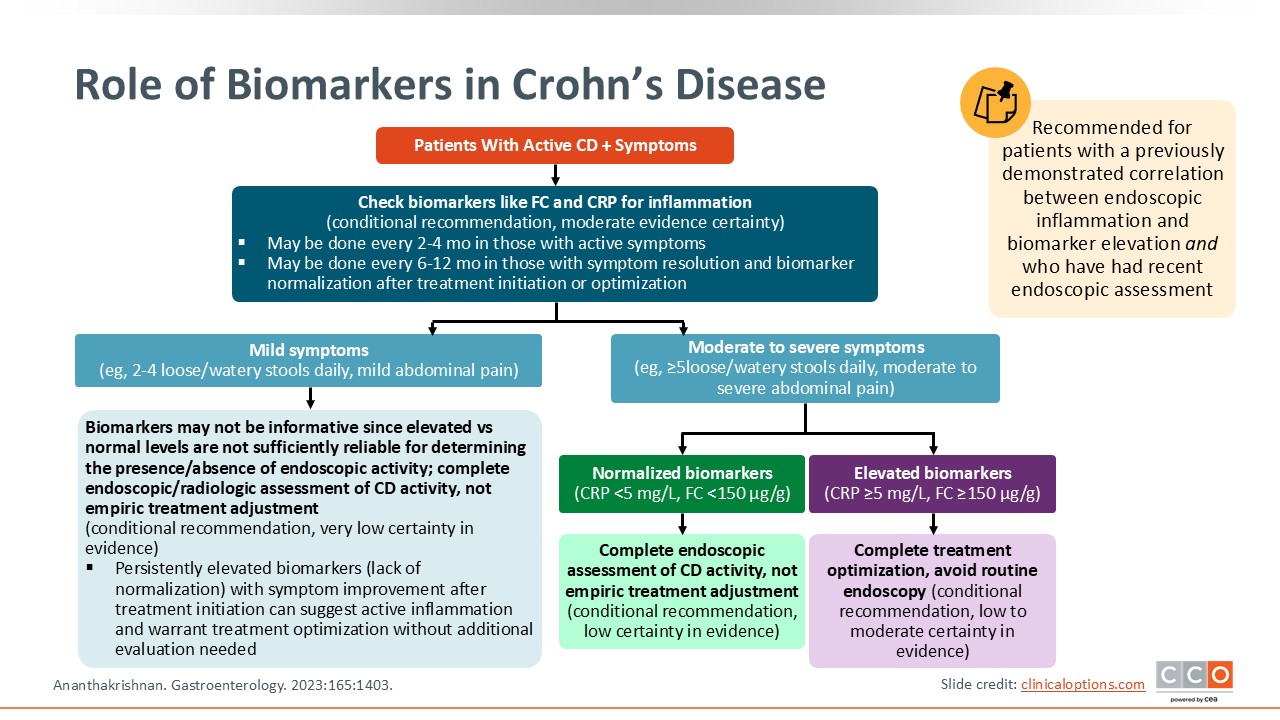
Role of Biomarkers in Crohn’s Disease
Biomarkers are critical in managing CD, and patients should be assessed both when they are not doing well (ie, there is symptom burden) as well as when they are doing well to prevent or mitigate relapse. When patients are doing well, HCPs typically monitor biomarkers every 6-12 months. Early in the treatment paradigm or when patients are symptomatic, HCPs should check biomarkers more frequently to inform treatment adjustments and better achieve treatment targets. If patients’ biomarkers remain elevated despite treat-to-target approaches, HCPs can pursue other techniques, such as cross-sectional imaging, to determine how the biomarkers are correlating with disease activity and to provide more detailed information for informing further treatment adjustments.20
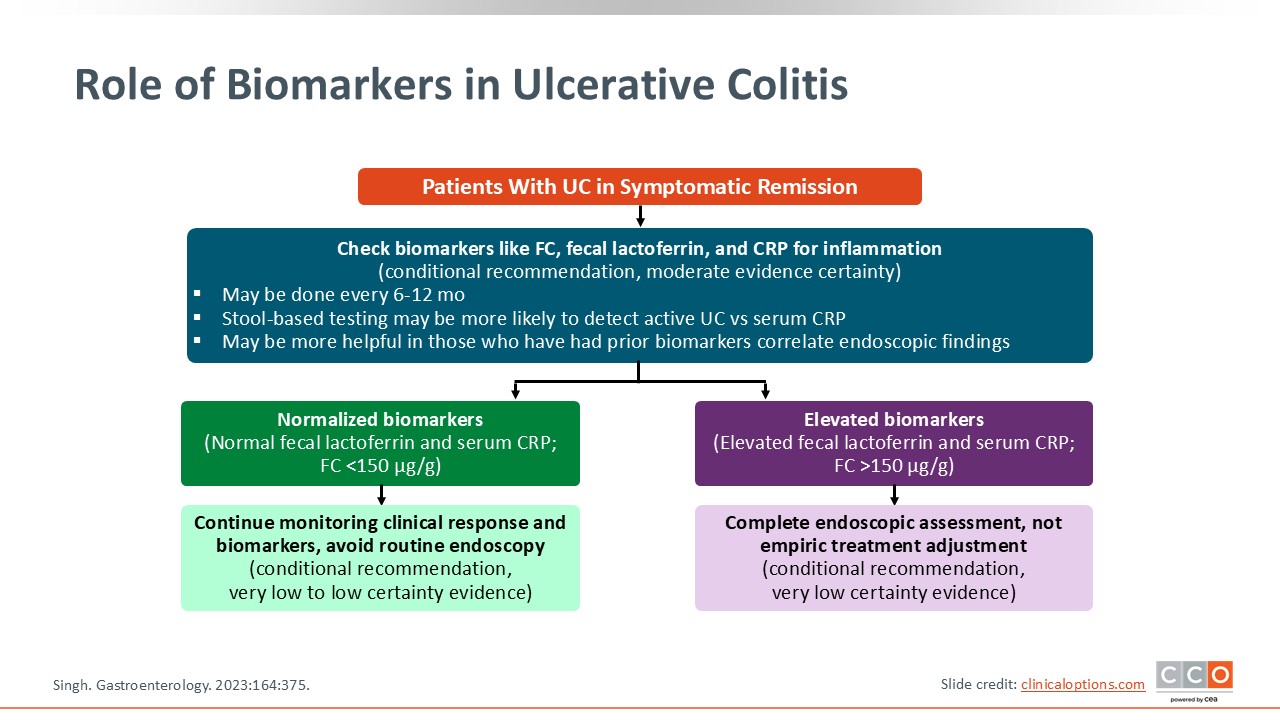
Role of Biomarkers in Ulcerative Colitis
In UC, imaging with axial CT or MRE is not part of the general monitoring strategy; however, biomarkers are, and endoscopy, colonoscopy, or sigmoidoscopy are important assessments. Of course, HCPs should gather baseline data by understanding what the patient’s colon looks like and what the biomarkers are at that point. Then, they can follow biomarkers over time via a noninvasive strategy. For example, if patients have a Mayo 3 pan-colitis, FCP of 200 μg/g, CRP of 20 μg/g at baseline, instead of repeating colonoscopy or sigmoidoscopy every couple of months, HCPs can follow those biomarkers because their baselines measured objective disease activity via endoscopy. This approach sets the stage for how HCPs can follow patients and what improvement should look like over time.21
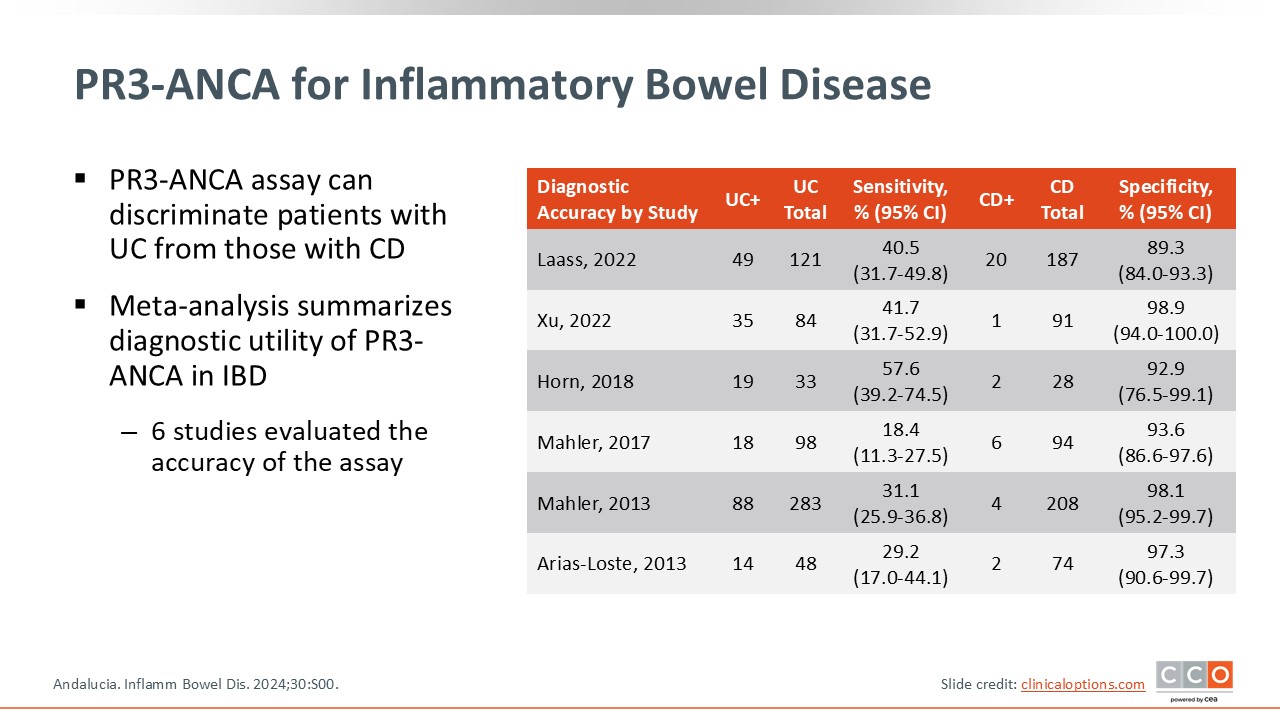
PR3-ANCA for Inflammatory Bowel Disease
There are other biomarkers that could be considered in IBD. For example, antineutrophil cytoplasmic antibodies (ANCA) in patients with IBD is a serum antibody measure. This has been associated with UC and less so with CD. Of importance is that the PR3-ANCA assay can differentiate patients with UC vs those with CD. This is helpful in clinical practice because some of these serum antibody biomarkers can differentiate patients who present with overlapping UC and CD symptoms, especially when surgical interventions are considered. Although antibody testing is not required as part of patient assessment and monitoring, these could be helpful if there are diagnostic and other clinical questions.22
PROFILE: Infliximab vs Standard of Care for Patients With Newly Diagnosed and Active CD
Next I review data on the emerging early and aggressive treatments for CD, including how these may be beneficial compared with a standard approach to care.
This is the multicenter, randomized PROFILE trial.23 Patients with newly diagnosed and active CD were randomized to an aggressive treatment combination of infliximab, a tumor necrosis factor (TNF) blocker,24 plus an immunomodulator or standard of care, where treatment escalation was determined by flares. The primary endpoint for this trial was surgery-free and steroid-free remission. Key secondary endpoints were quality of life, endoscopic remission, and flare frequency.25
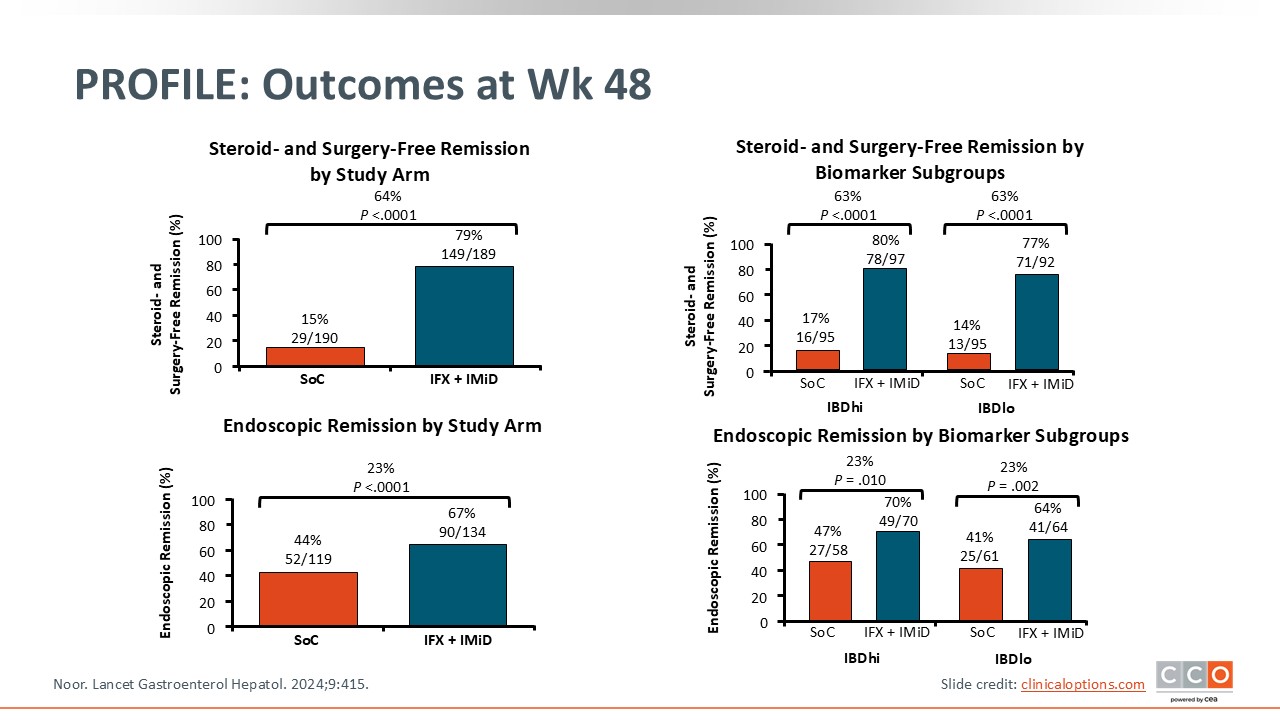
PROFILE: Outcomes at Week 48
The results of this study show that early and aggressive combination therapy was significantly better than standard of care. Across all outcomes (steroid-free, surgery-free, and endoscopic remission) whether patients were treated with aggressive therapy upfront or they were at high or low risk for disease severity over time, combination treatment with infliximab plus an immunomodulator was associated with significant improvements vs a step-up/watch-and-wait approach. This study provides good evidence, as I previously talked about that window of opportunity, that early and aggressive therapy can really help patients and improve outcomes.25
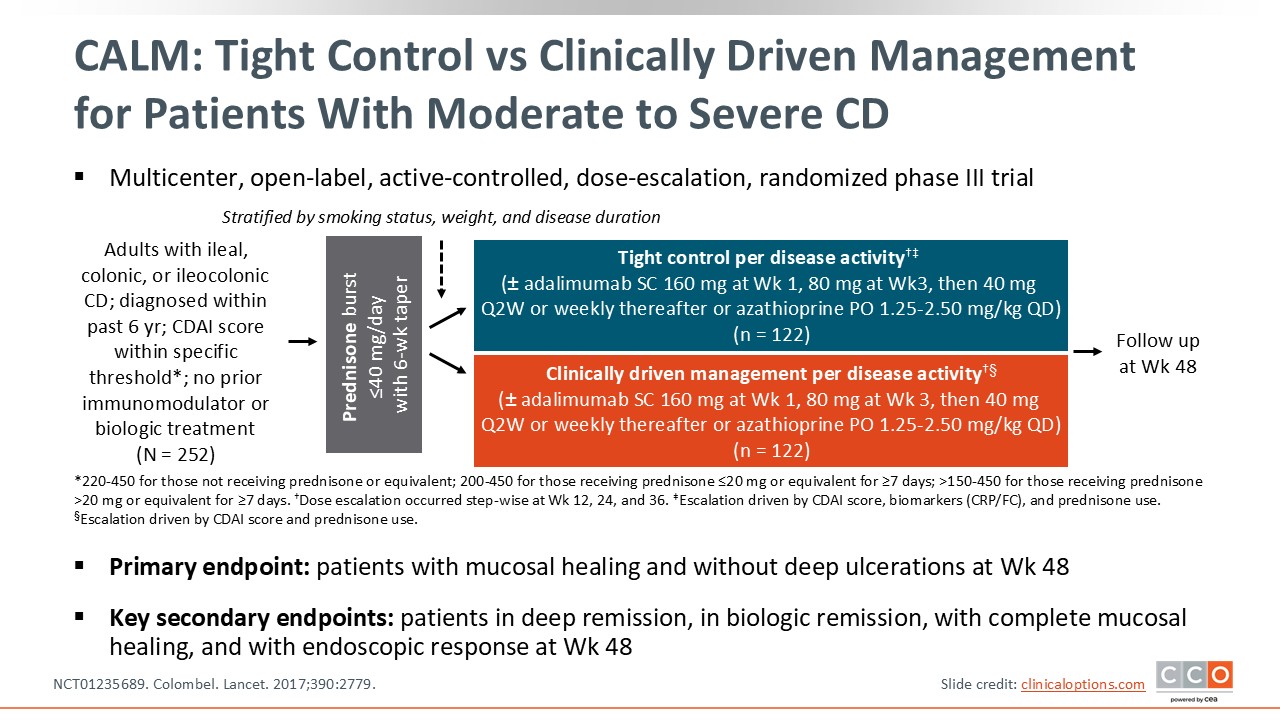
Tight Control vs Clinically Driven Management for Patients With Moderate to Severe CD
CALM was a similar trial that looked at tight control vs clinically driven management per disease activity for patients with ileal, colonic, or ileocolonic CD who were diagnosed within prior 6 years (NCT01235689). This multicenter, dose-escalation phase III trial randomized patients to tight control based on biomarker assessment vs monitoring disease over time based on symptoms. For both treatment arms, patients could receive adalimumab or azathioprine.26
CALM: Study Outcomes
Similar outcomes to PROFILE, the CALM study looked at mucosal healing and endoscopic response. As expected, tight control with treatment adjustments based on biomarkers resulted in better outcomes vs clinically driven management. This study underscores that biomarker driven adjustments to a treatment strategy is directly associated with improved long-term disease management.26
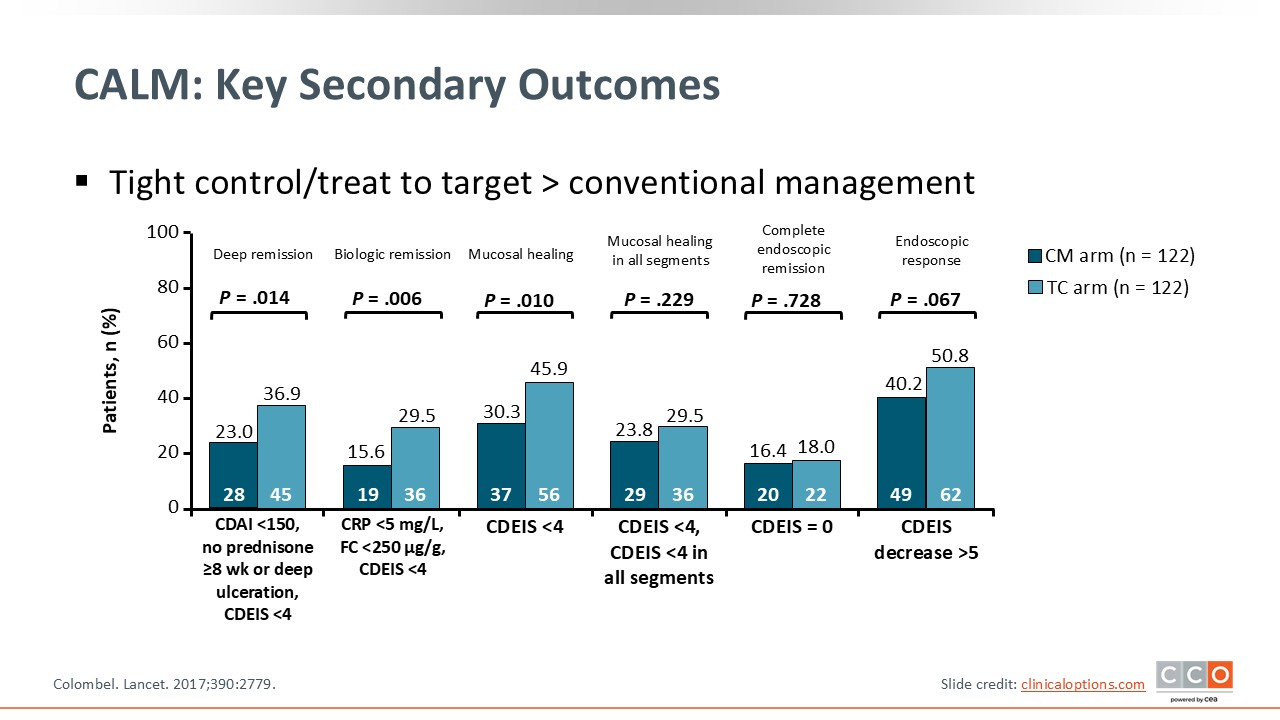
CALM: Key Secondary Outcomes
This slide provides a closer look at the CALM study’s outcomes, including deep remission, biologic remission, and mucosal healing, among others. One can see that tight control led to improvement vs clinical management for most outcomes.26
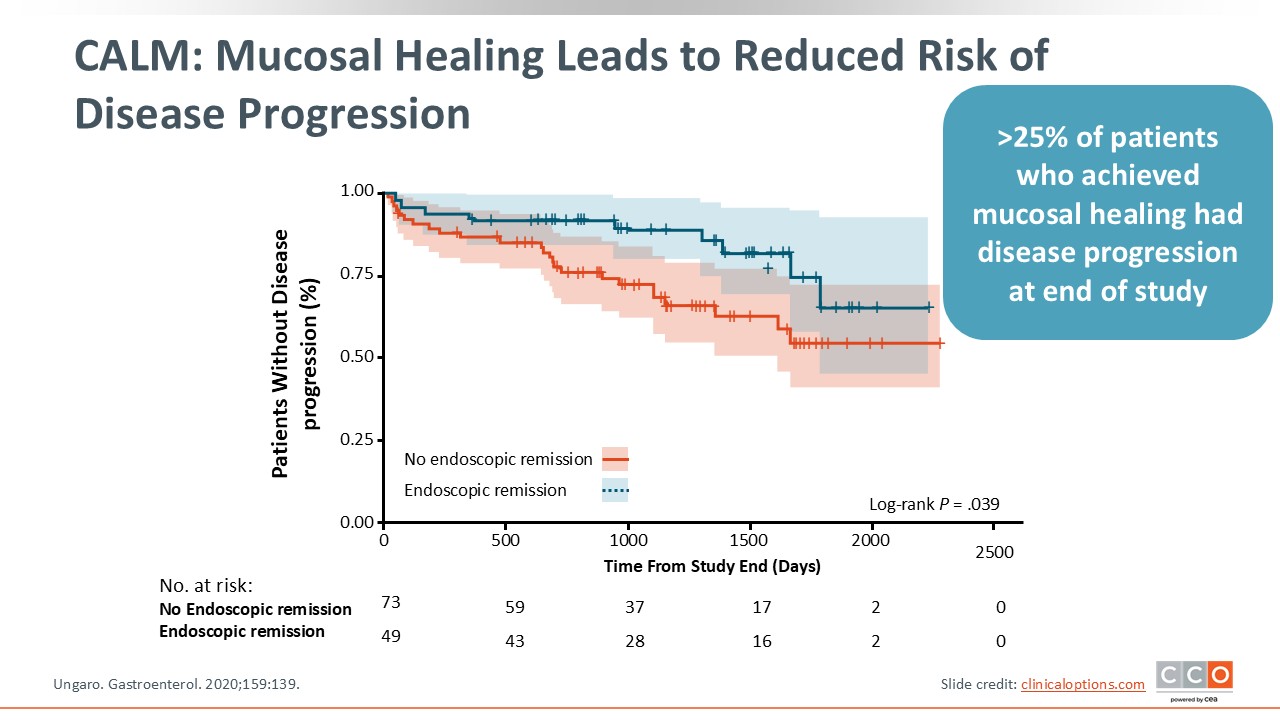
CALM: Mucosal Healing Leads to Reduced Risk of Disease Progression
In a follow-up analysis of the CALM study, it was found that patients were far less likely to develop disease progression after achieving mucosal healing, underscoring again how early aggressive treatment led to more improved outcomes and decreased disease progression over time vs clinical management.27
Looking Beyond STRIDE-II: Future Considerations
In looking beyond STRIDE-II, what does the future hold? As I have mentioned, transmural and histologic healing are aspirational treatment targets. This is something that is being considered in how we may further improve outcomes and reduce complications in IBD. Furthermore, integrating patient-reported quality-of-life outcomes is also being considered. Beyond getting patients to feel better, normalize biomarkers, and improve endoscopy, HCPs should measure quality-of-life outcomes and ensure patients are feeling better beyond just symptom improvement.
Then, as new therapies emerge, treatment strategies may change based on how these agents are administered or how quickly they work. We want patients to be treated effectively, early, and with tight control, which is associated with reduced complications and disease progression.28,29
Click here to listen to expert viewpoints on early evidence for companion diagnostics assessing genetic markers.