CE / CME
From Evidence to Practice—Barriers to Access and Manufacturing Delays in CAR T-Cell Therapy
ABIM MOC: maximum of 0.50 Medical Knowledge MOC point
Physicians: Maximum of 0.50 AMA PRA Category 1 Credit™
Released: December 09, 2025
Expiration: June 08, 2026
Activity
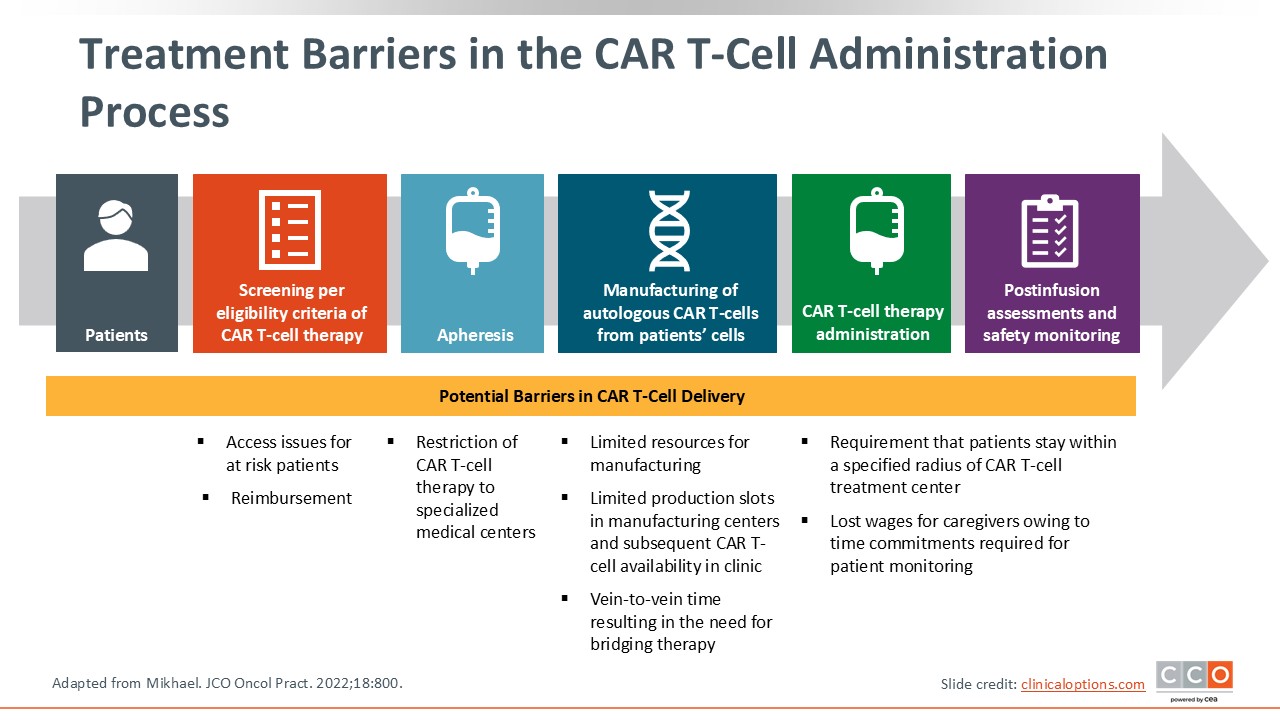
Treatment Barriers in the CAR T-Cell Administration Process
Barriers to access and manufacturing delays continue to pose major challenges in the delivery of CAR T-cell therapy. During the past several years, CAR T-cell therapies have significantly advanced the treatment landscape for MM, offering meaningful and durable responses for patients with R/R disease.2,3
Despite this progress, the path to receiving CAR T-cell therapy is not straightforward. With 2 products, idecabtagene vicleucel (ide-cel) and ciltacabtagene autoleucel (cilta-cel), now approved for MM, it is more important than ever to examine the obstacles that prevent eligible patients from receiving these treatments.4,5
Many factors can contribute to limited access, including manufacturing and logistical constraints to referral patterns, treatment center capacity, and broader systemic inequities within healthcare delivery.6
This discussion focuses on identifying and understanding those barriers. By addressing challenges in manufacturing and access, and by ensuring that patients are appropriately managed and monitored during the manufacturing period through the use of bridging therapy, the field can move closer to ensuring that every patient who may benefit from CAR T-cell therapy has a realistic opportunity to receive it.
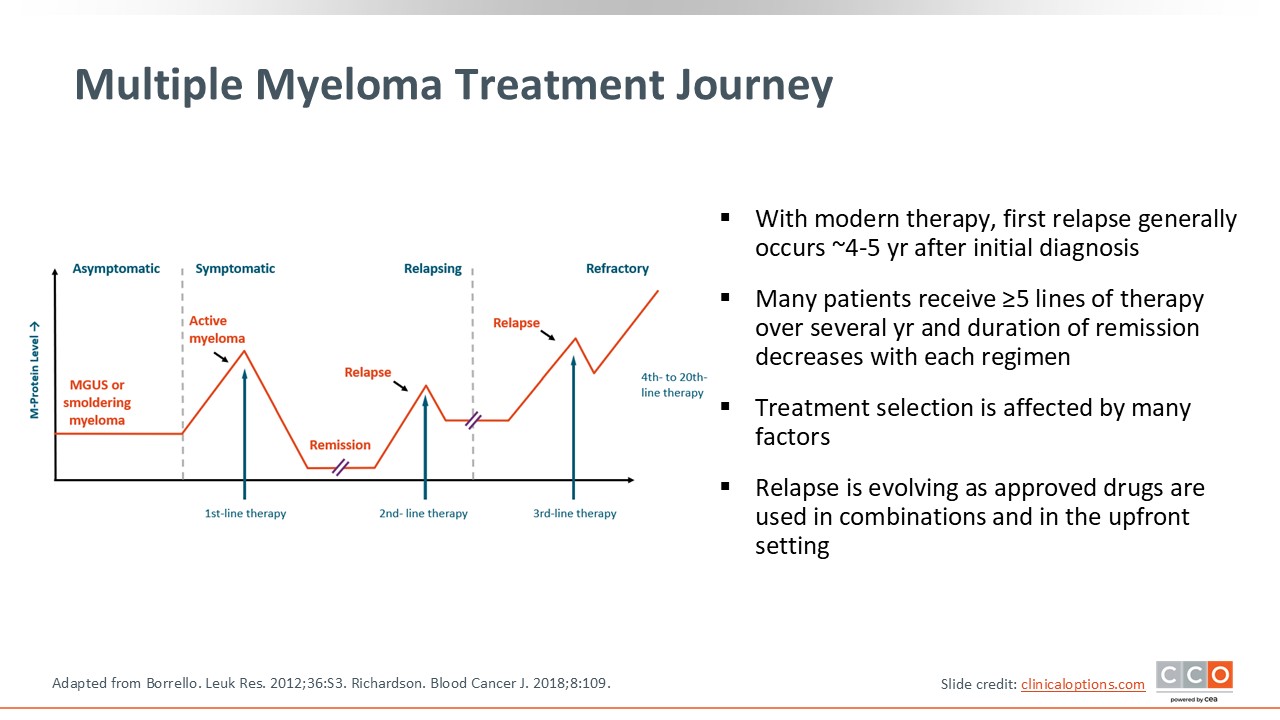
Multiple Myeloma Treatment Journey
In the current MM landscape, we often see the first relapse at a median of 4-5 years after initial diagnosis, particularly when patients are started on combination regimens early in the treatment cycle.7 Using a triplet therapy backbone, including a proteasome inhibitor (PI), an immunomodulatory agent (IMiD), and a corticosteroid, remains as a staple of initial treatment, with the use of anti-CD38–containing quadruplet regimens becoming increasingly common in newly diagnosed patients.8,9
Although these regimens have contributed to deeper and more durable responses, relapse remains inevitable for most patients, and the disease course involves exposure to multiple lines of therapy over time. With each successive relapse, treatment options become more limited, and the likelihood of achieving a meaningful response narrows.10,11
As treatment options diminish with each relapse, the challenge of ensuring access to the most effective and timely therapies becomes increasingly complex. Addressing these barriers to treatment, including logistical challenges, such as manufacturing delays or treatment center availability, as well as patient-level factors like fitness, treatment fatigue, and financial or geographic constraints, is critical to ensuring patients can receive advanced therapies.12
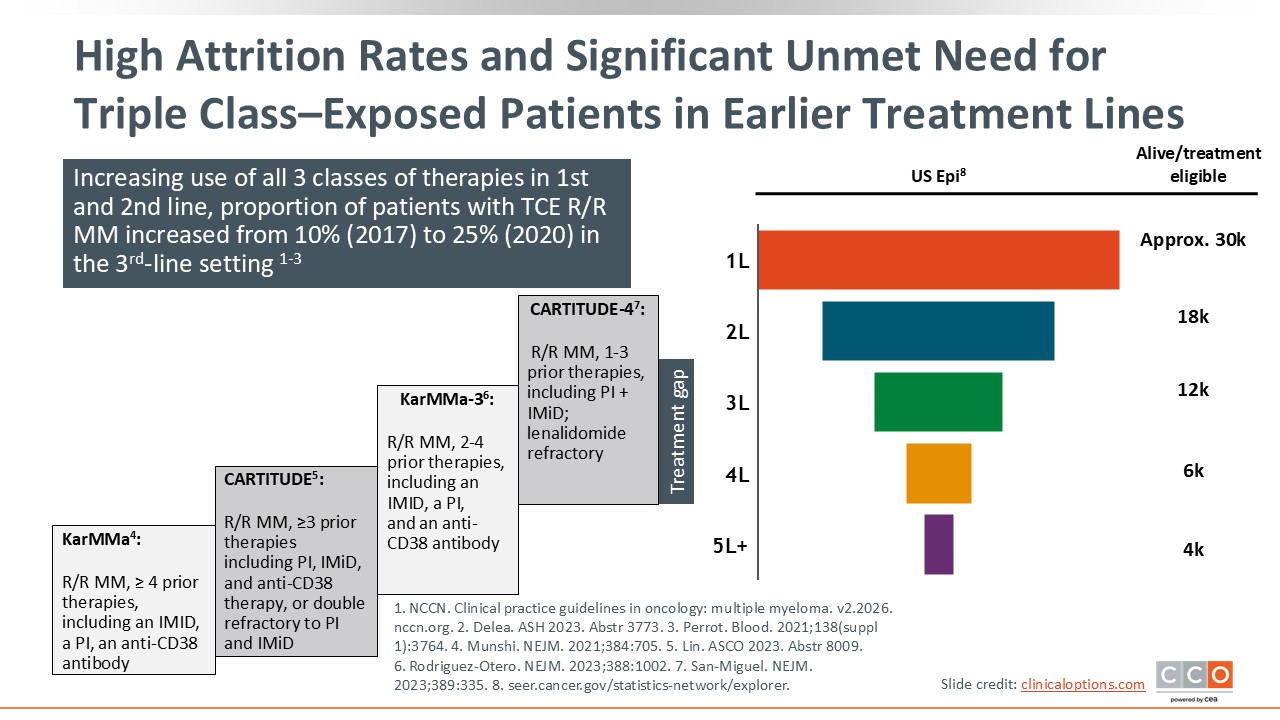
High Attrition Rates and Significant Unmet Need for Triple Class–Exposed Patients in Earlier Treatment Lines
The shift in practice to using combination approaches earlier in therapy leads to exhaustion of standard treatment options earlier in the treatment course. With each relapse, a substantial proportion of patients discontinue treatment because of disease progression, treatment-related toxicity, comorbidities, or declining performance status.13,14 This puts limits on how many patients are able to receive subsequent lines of therapy.
Evidence from clinical trials illustrates this attrition. For example, in the phase III KarMMa-3 trial, patients with R/R MM who had received 2-4 prior lines of therapy were randomized 2:1 to receive ide-cel or standard-of-care therapy, which included one of 5 investigator-selected regimens:
- Daratumumab/bortezomib/dexamethasone
- Daratumumab/pomalidomide/dexamethasone
- Elotuzumab/pomalidomide/dexamethasone
- Carfilzomib plus dexamethasone or
- Ixazomib/lenalidomide/dexamethasone
Per protocol, patients randomized to ide-cel could receive 1 cycle of standard-of-care therapy as bridging therapy on an as-needed basis, although the data on actual bridging approaches were not reported. Despite this, attrition remained substantial: Approximately one half of the patients did not proceed to a second line of treatment, and by the third line, more than two thirds discontinued treatment.2,15
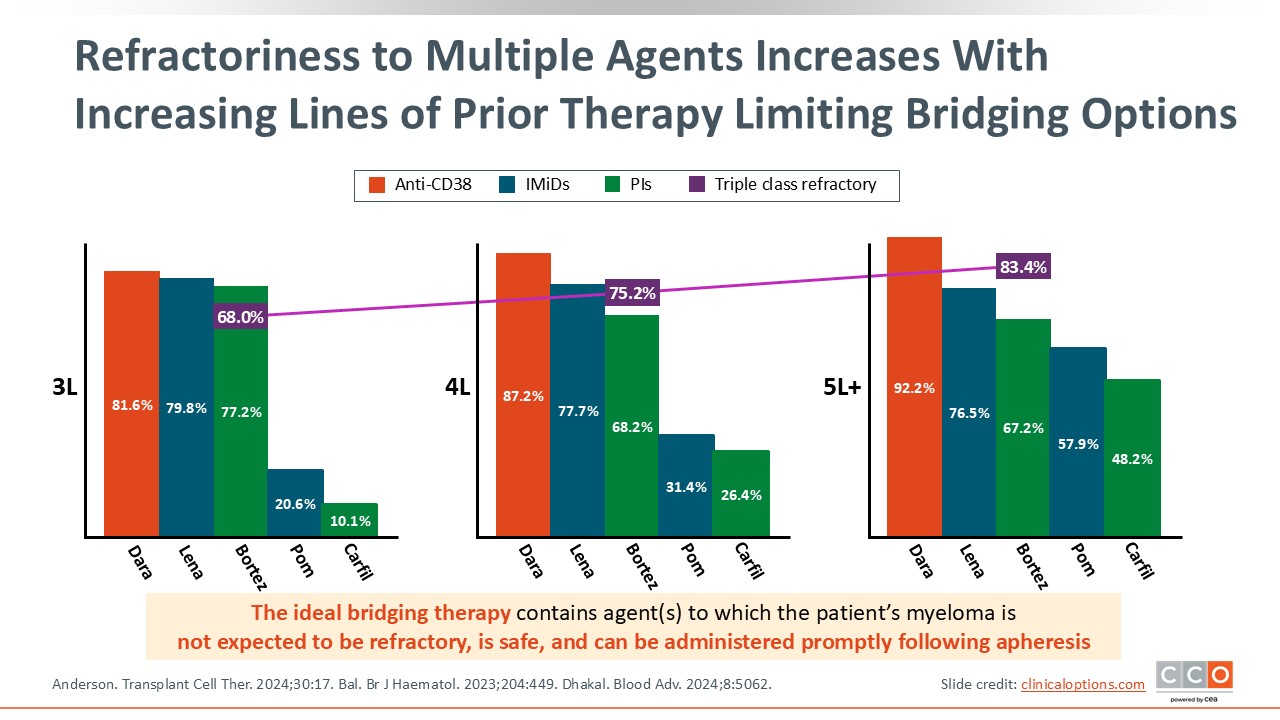
Refractoriness to Multiple Agents Increases With Increasing Lines of Prior Therapy Limiting Bridging Options
It is important to recognize that with each additional line of therapy, a patient’s refractoriness to treatment typically increases, limiting available options for effective bridging therapy.16 This challenge is particularly pronounced in patients who are triple-class refractory and have a high disease burden that must be controlled during the CAR T-cell manufacturing process. Ideally, bridging therapy should include agents to which the patient’s MM is likely still sensitive, should be safe, and should be started promptly after apheresis.17
CAR T-Cells in MM: Ide-cel and Cilta-cel
Both ide-cel and cilta-cel are highly effective products that have shown high response rates and durable responses in very heavily pretreated patients.2,3,18,19 Toxicity is predictable and manageable and includes cytopenias, cytokine-release syndrome, and immune effector cell–associated neurotoxicity syndrome.20,21 Real-world data also support the use of these CAR T-cell products, with no new safety signals,22 and an appreciation of this “one and done” approach by patients based on health-related quality-of-life data.23
FDA-Approved Autologous CAR T-Cell Therapy for R/R MM
Ide-cel was the first CAR T-cell therapy approved for R/R MM, initially receiving FDA approval in 2021 for patients who had received 4 or more prior lines of therapy, including an IMiD, a PI, and an anti-CD38 antibody, based on the KarMMa-1 trial. In 2024, this indication was expanded to patients with R/R disease after 2 or more prior lines of therapy, supported by results from KarMMa-3.2,4,24
Cilta-cel was first approved in 2022 based on the CARTITUDE-1 trial for adults with R/R MM after at least 4 prior lines of therapy, including an IMiD, a PI, and an anti-CD38. Its indication was expanded in 2024 following CARTITUDE-4 to include patients with R/R disease after 1 prior line of therapy, provided they previously received an IMiD and PI and are refractory to lenalidomide.3,5,24,25
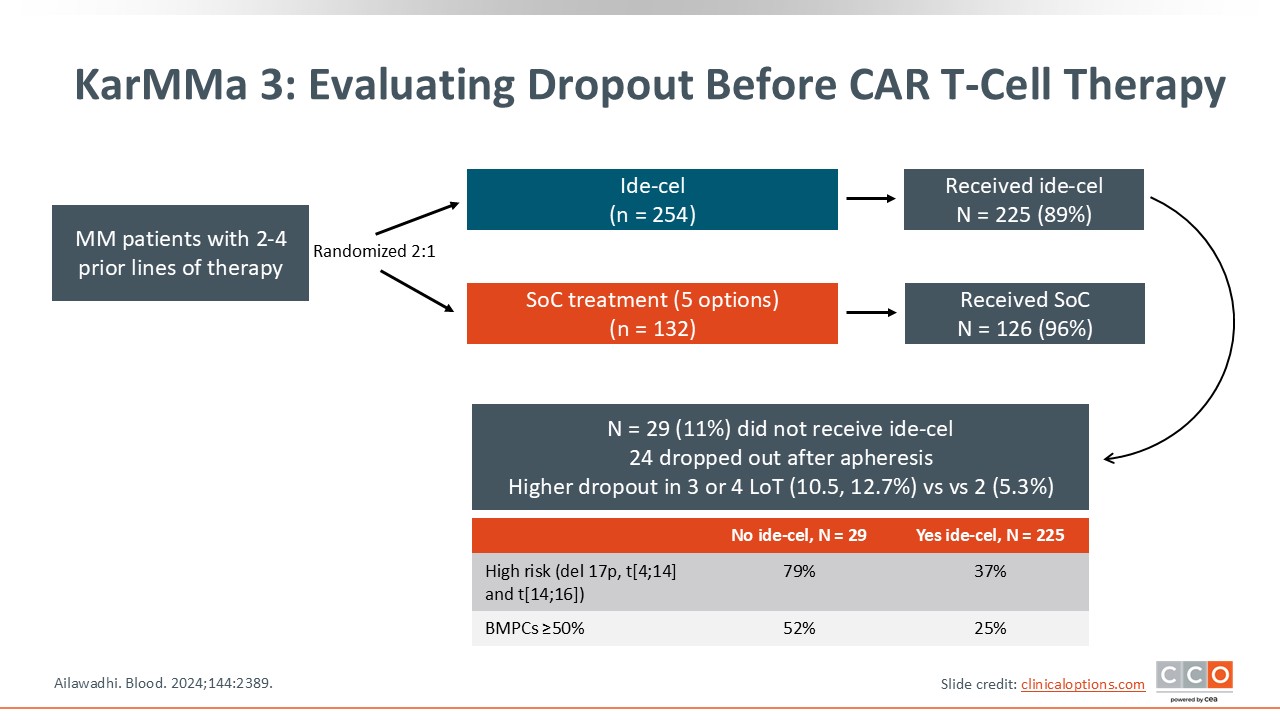
KarMMa-3: Evaluating Dropout Before CAR T-Cell Therapy
It is important to note that, in the intention-to-treat populations, there was an improvement in progression-free survival (PFS) associated with CAR T-cell therapy but not in overall survival (OS) early on in the treatment course, either in KarMMa-3 or CARTITUDE-4, largely because of the dropout rate. In KarMMA-3, the dropout rate was approximately 13% among patients who had received 4 prior lines of therapy vs approximately 5% among patients who had received 2 prior lines of therapy.26
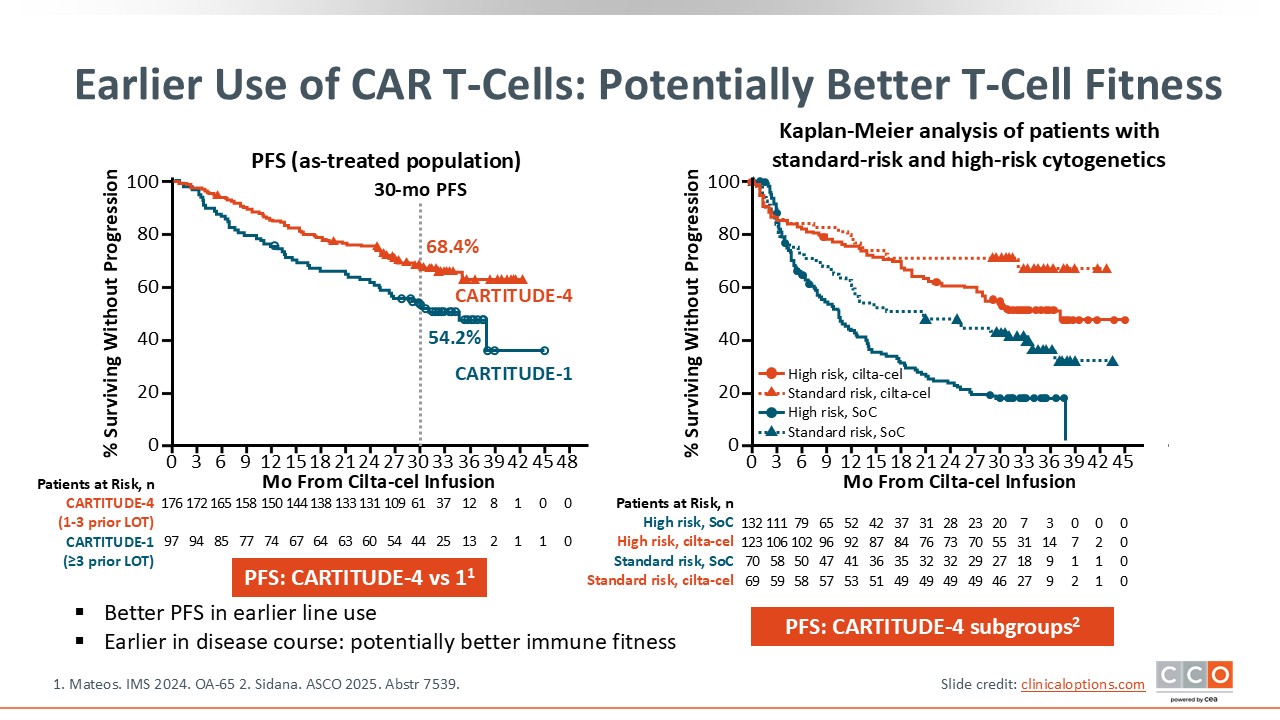
Earlier Use of CAR T-Cells: Potentially Better T-Cell Fitness
The data shown here further support the use of CAR T-cell therapy earlier in treatment. Among all patients enrolled in CARTITUDE-4, with an inclusion criterion of 1-3 prior lines of treatment, the 30-month PFS rate was 68.4% vs 54.2% among patients enrolled on the phase Ib/II CARTITUDE-1 trial, with an inclusion criterion of at least 3 prior lines of therapy.18 Thus, as CAR T-cell therapy was moved to earlier lines of treatment, PFS was improved, underscoring the importance of getting these effective agents into the treatment paradigm as early as possible to improve outcomes and prevent dropout.
Subgroup analyses of CARTITUDE-4 supported the positive benefits associated with cilta-cel in patients with lenalidomide-refractory MM as early as after first relapse, with PFS benefits across patients with standard-risk or high-risk cytogenetics.20
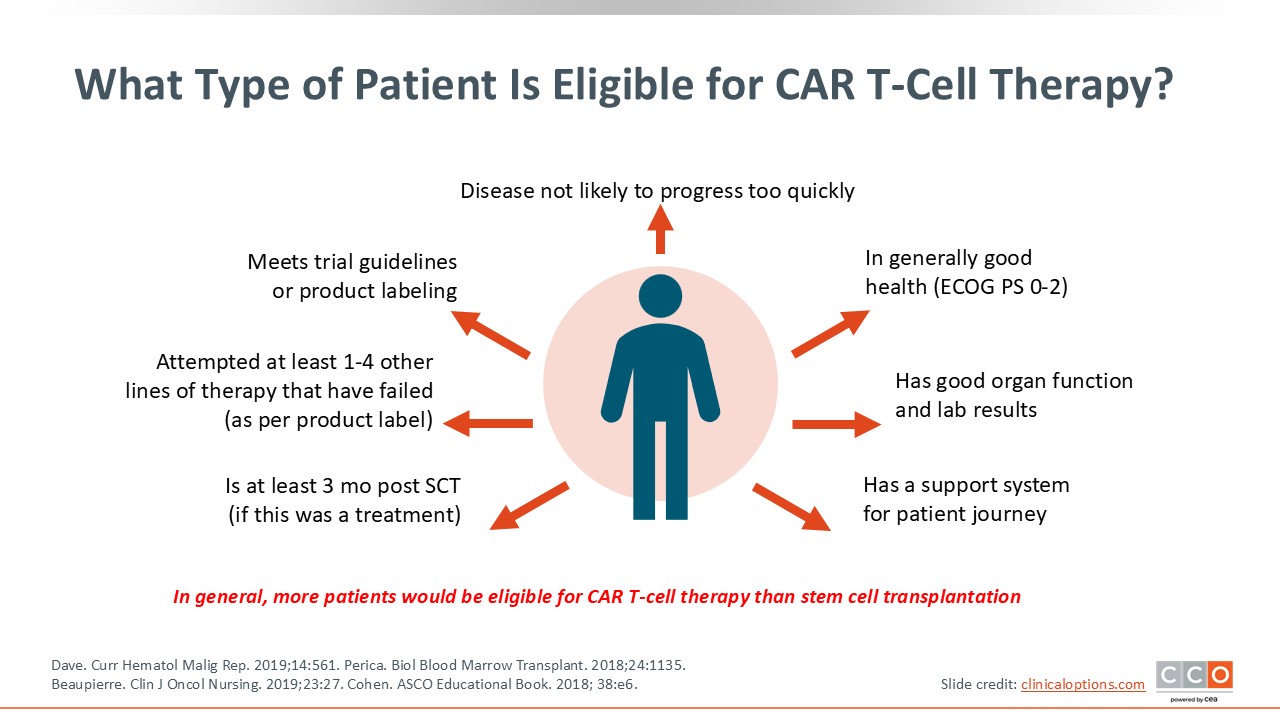
What Type of Patient Is Eligible for CAR T-Cell Therapy?
As long as patients are diagnosed with MM and their disease is not rapidly progressing, they are a candidate for CAR T-cell therapy. As mentioned earlier, with ide-cel, a patient must have received at least 2 prior lines of treatment, whereas for cilta-cel, a patient must have received at least 1 prior line of treatment and be refractory to lenalidomide. The patient should have a reasonable performance status and, in general, good organ function to be able to tolerate the lymphodepleting chemotherapy as well as the CAR T-cell product.
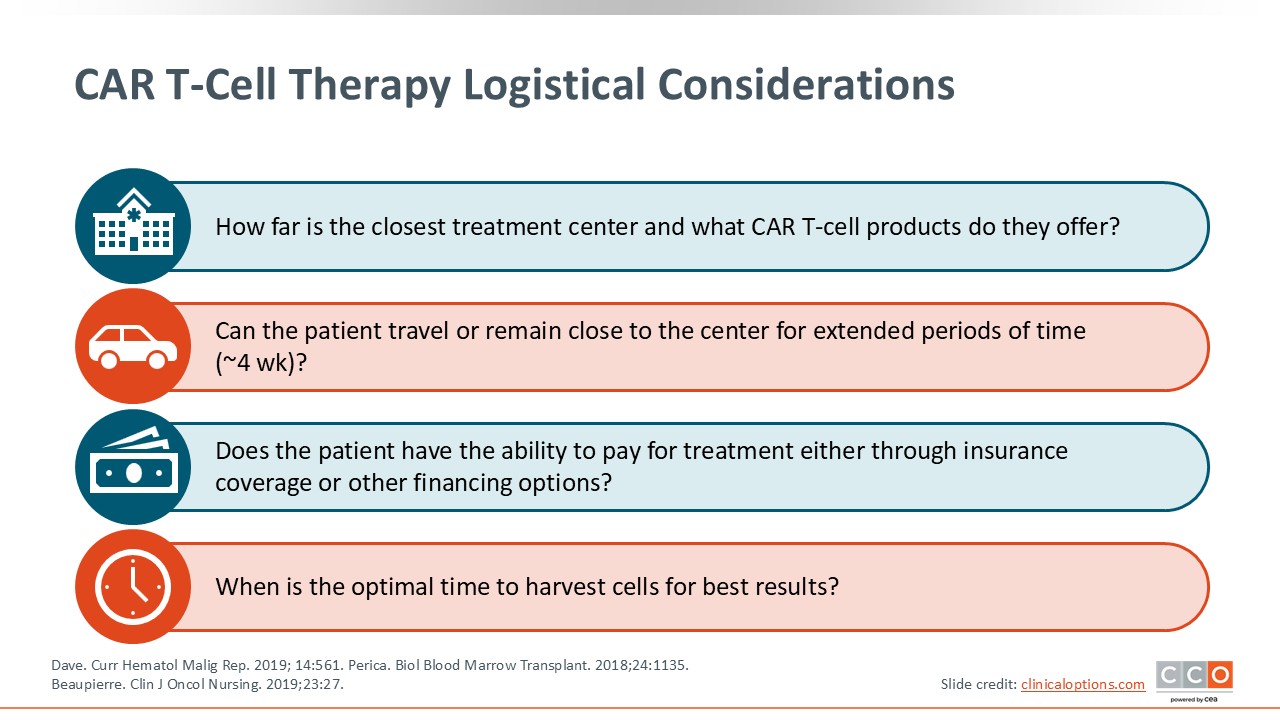
CAR T-Cell Therapy Logistical Considerations
There are many logistical considerations that should be taken into account when deciding whether to treat patients with CAR T-cell therapy. This is where shared decision-making with patients, their caregivers, and healthcare professionals comes into play. It is also important to consider the distance to the closest treatment center wherein the CAR T-cell product is offered and access to care. Can the patient travel to that center? Is the patient able to stay close to the center early on in the treatment course? These issues have become easier to address since the FDA removed the REMS requirement, but patients still need to be monitored, especially within the first month. Thus, it is critical for both the patients and their caregivers to be able to stay close to the center where the CAR T-cell product is offered.
Other important questions to consider include: Do the patients have the ability to pay for treatment? Are they able to take time away from work? Do they have a support system at home that allows them to be away for the initial 2-4 weeks often required for treatment? Many treatment centers have social workers, patient navigators, or financial counselors who can help assess these needs and support patients and their families throughout the process.
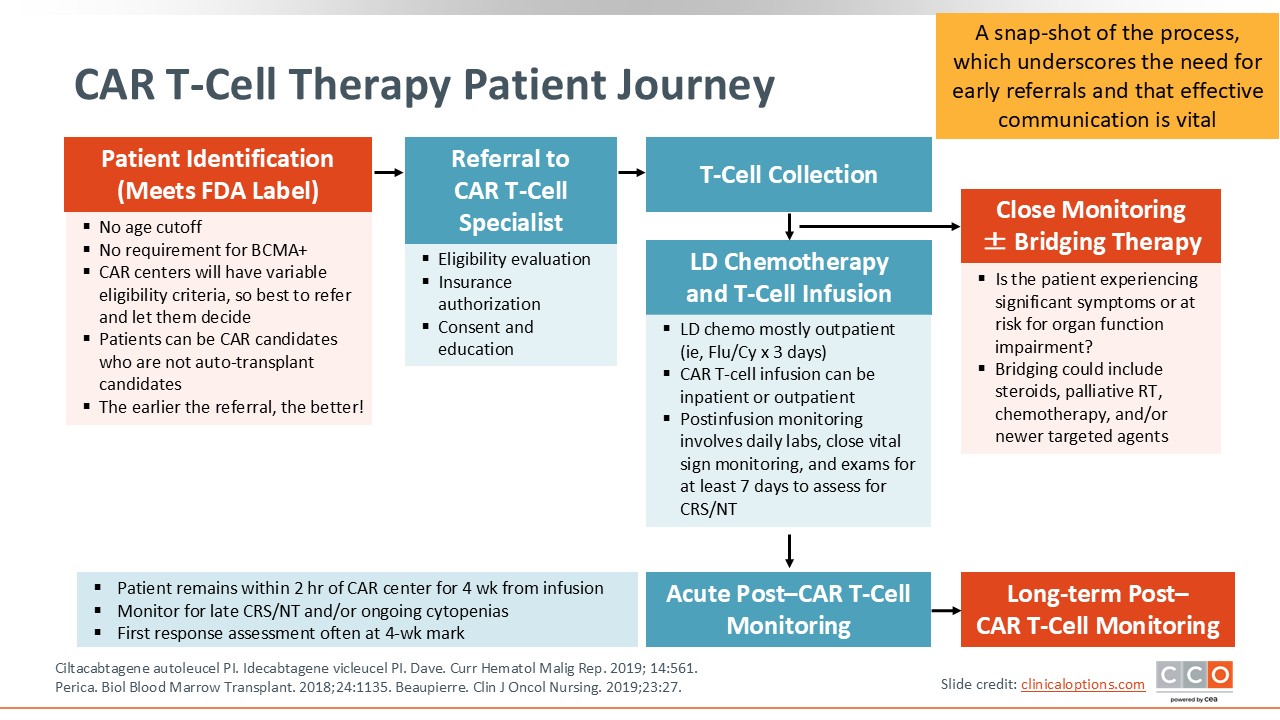
CAR T-Cell Therapy Patient Journey
This slide represents a snapshot of the CAR T-cell therapy treatment journey. Once a patient has been identified as a good candidate and meets all the logistical requirements noted earlier, the patient undergoes leukapheresis. Once complete, the patient’s T-cells go through a manufacturing process that typically takes 4-6 weeks. This period can be even longer because of manufacturing delays. During this time, the collection is enriched for white blood cells, and CAR T-cells are activated, engineered, grown, and expanded. These cells are then infused into the patient.
Prior to the infusion, patients are required to receive chemotherapy, which is typically lymphodepleting chemotherapy involving cyclophosphamide and fludarabine.26 After 3 days of this chemotherapy, patients are instructed to rest for 2 days, after which point CAR T-cells are infused.
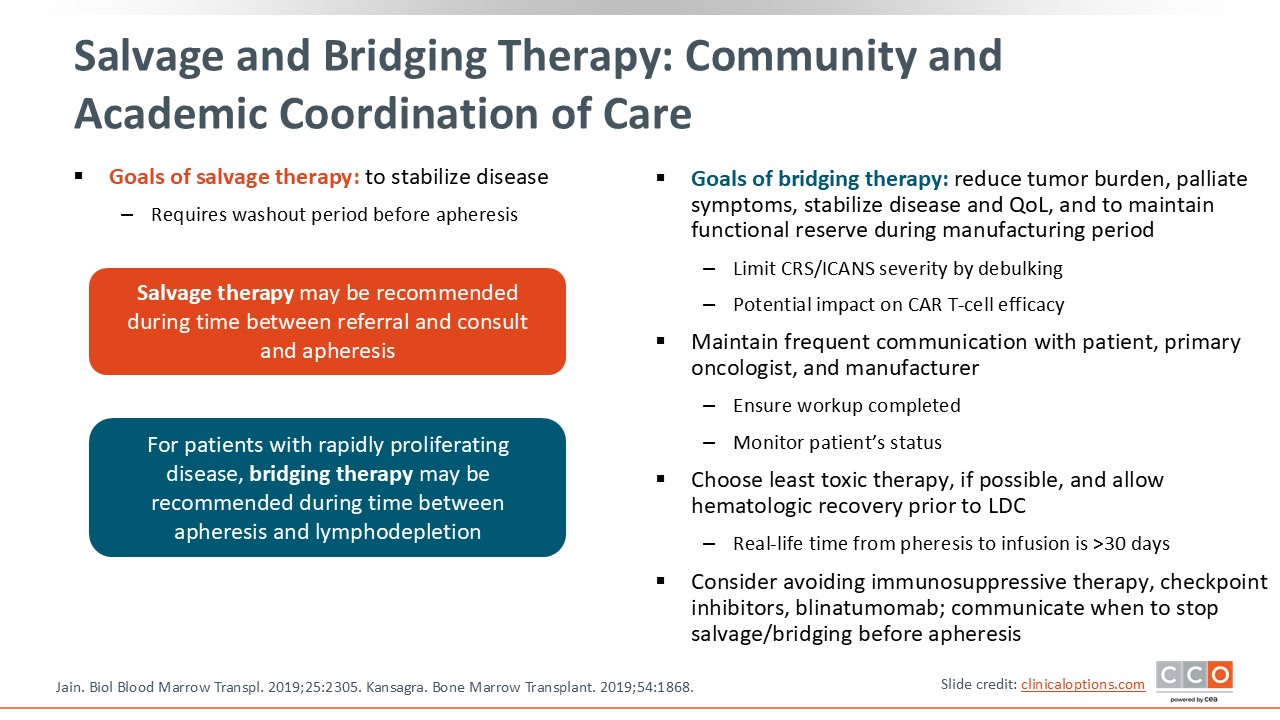
Salvage and Bridging Therapy: Community and Academic Coordination of Care
One gap in administering CAR T-cells successfully can be narrowed by establishing good relationships between the CAR T-cell treatment center and the community oncologists who refer patients for CAR T-cell therapy. The number of CAR T-cell centers is limited; therefore, it is important for healthcare professionals to work closely with their community colleagues, especially for bridging therapy, which would allow bridging therapy to be instituted by the referring physician in some cases. Close communication between the CAR T-cell center and the referring physician is critical. It is also important to develop a plan for both the best bridging therapy and optimal treatment duration. This requires effective communication between the community oncologist and CAR T-cell treatment center team as well as education, and this gap needs to be overcome by continued education around bridging therapy and its importance.
Questions and Challenges
There are several ongoing challenges associated with CAR T-cell therapy. Vein to vein time, which is the time from leukapheresis to infusion, can differ, and treatment slot access at a treatment center can be limited. As noted above, healthcare professionals should establish the optimal bridging treatment, especially in patients with R/R disease and those with a high disease burden. A plan for what should be done for patients who progress while on bridging therapy needs to be developed. Are certain washout periods needed in these patients before CAR T-cell infusion? These questions and challenges are commonly encountered in the clinic, so I want to review data that highlight the importance of bridging therapy in patients with a high disease burden to improve upon the outcomes of these highly effective treatment strategies.
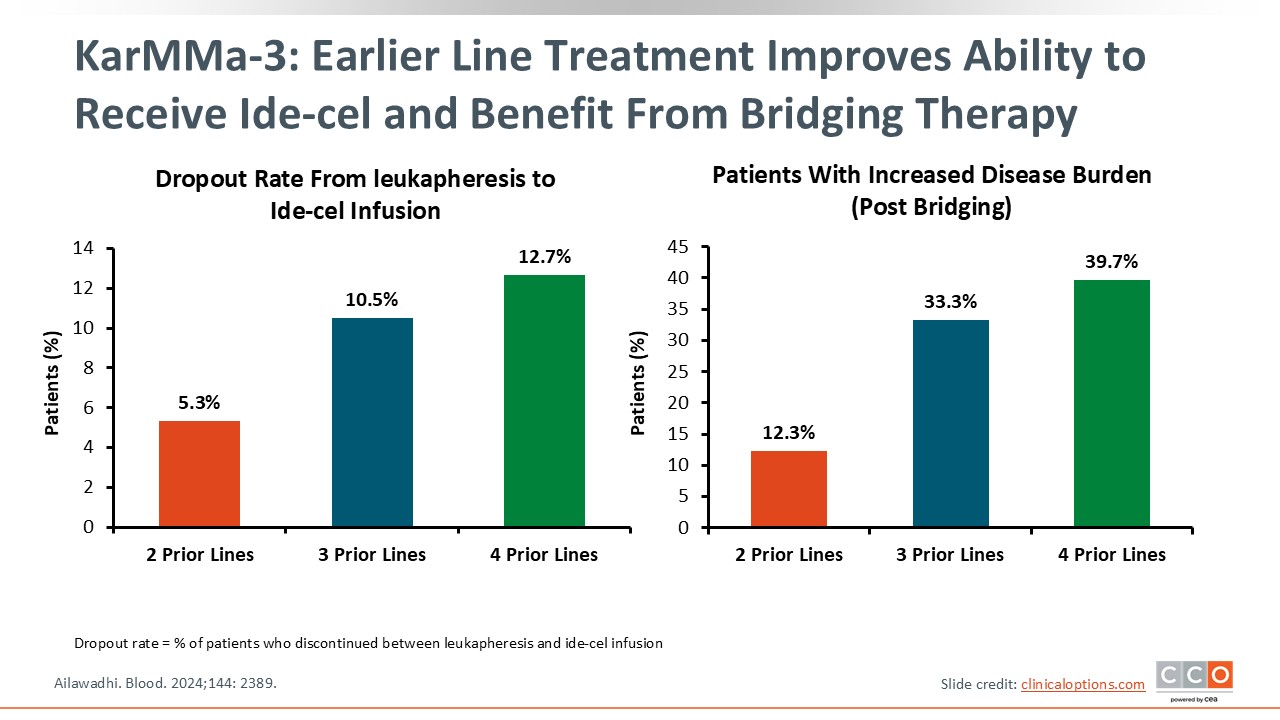
KarMMa-3: Earlier Line Treatment Improves Ability to Receive Ide-cel and Benefit From Bridging Therapy
As seen in KarMMa-3, with every new line of treatment comes an increased risk of dropout. Among patients who had received 2 prior lines of treatment, the dropout rate was low, approximately 5%, from leukapheresis to ide-cel infusion. Among those who had received 4 prior lines of treatment, the dropout rate was much higher—approximately 13%—because these patients are more refractory and do not respond to treatment. Rather, they progress rapidly. The same trend was observed in patients with an increased disease burden post bridging therapy.27 This is why it is important to identify candidates for CAR T-cells early on and to incorporate effective bridging treatment in this population.
Conclusions
In conclusion, it is very important to refer patients early for CAR T-cell treatment as the expanding indications allow for earlier incorporation. Earlier referral means more options for bridging therapy, expanding beyond talquetamab alone. The optimal bridging therapy may vary among patients, as discussed in additional text modules in this series. For example, if a patient is refractory to lenalidomide only, daratumumab with carfilzomib or daratumumab with pomalidomide could be an option for bridging therapy. Remember that these are patients who will get the best possible outcome from CAR T-cells. Therefore, education is critical in ensuring that more patients have access to these life-saving treatments in the context of R/R MM.