CE / CME
Identifying Patients Likely to Benefit From Bridging Therapy Before CAR T-Cell Infusion in R/R MM
ABIM MOC: maximum of 0.50 Medical Knowledge MOC point
Physicians: Maximum of 0.50 AMA PRA Category 1 Credit™
Released: December 29, 2025
Expiration: June 28, 2026
Activity
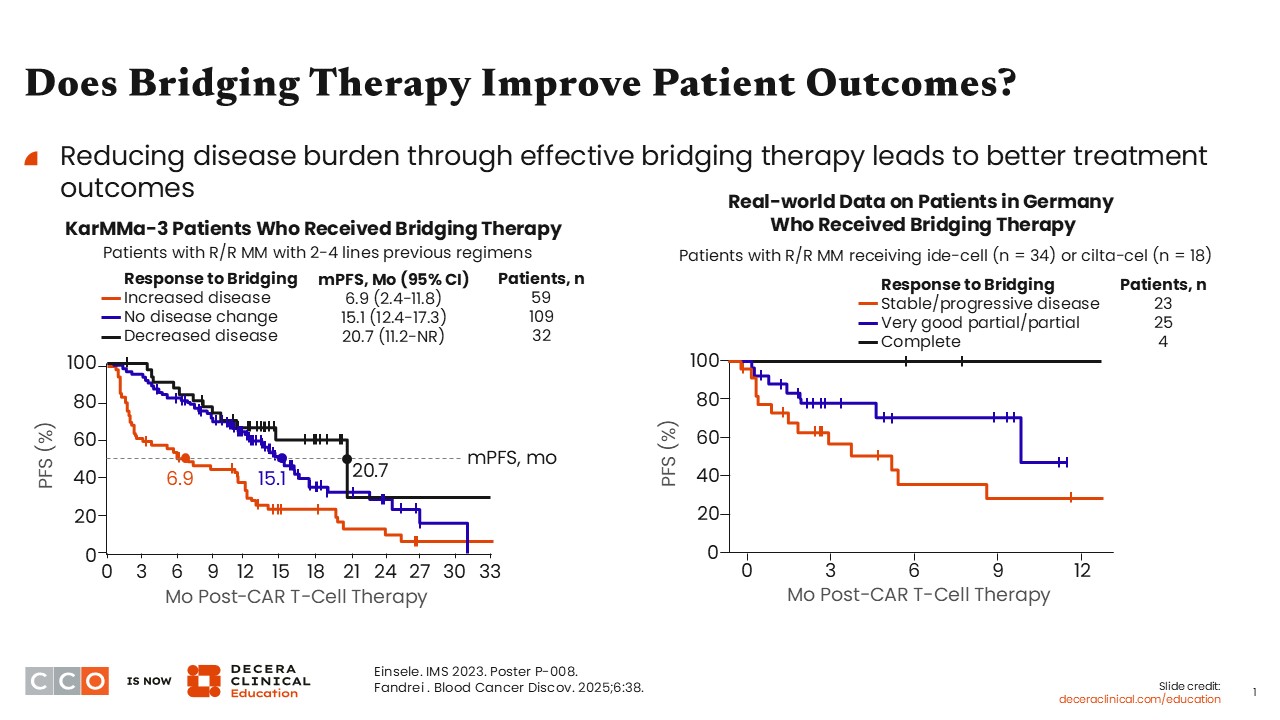
Does Bridging Therapy Improve Patient Outcomes?
Since the approvals of CAR T-cell therapy in R/R MM, first with ide-cel in 2021 and then cilta-cel in 2022, we have learned so much about how we can improve outcomes, both efficacy and safety, for patients undergoing CAR T-cell therapy.7,8
One of the modifiable aspects of CAR T-cell therapy is the bridging therapy. Most of us used to not think that bridging therapy had any impact on the outcomes of patients undergoing CAR T-cell treatment, but it does. It actually improves efficacy and reduces toxicity.9
When asking whether bridging therapy actually will help my patients, I think it is important to look at some data from patients receiving bridging therapy in KarMMa-3, an open-label, randomized phase III trial evaluating ide-cel vs standard-of-care regimens in triple class–exposed R/R MM. In this study, patients who received bridging therapy and showed response to the bridging therapy, and improvement in disease during the period of bridging therapy actually ended up with the longer median progression-free survival (PFS).4,5
This was true for patients with even just a minimal response to bridging therapy. As long as the disease burden decreased with the use of bridging therapy, the median PFS was 20.7 months vs 6.9 months among those with increasing disease burden during bridging therapy (ie, bridging therapy was not working).
When looking at real-world data, for both ide-cel and cilta-cel, those patients who had a complete response prior to going to CAR T-cell therapy had the longest PFS so far vs those with stable or progressive disease, who tended to have a much lower PFS.6
This is our first inkling that, yes, in randomized and retrospective studies, we really do see improvement in PFS if patients go in with decreasing disease.
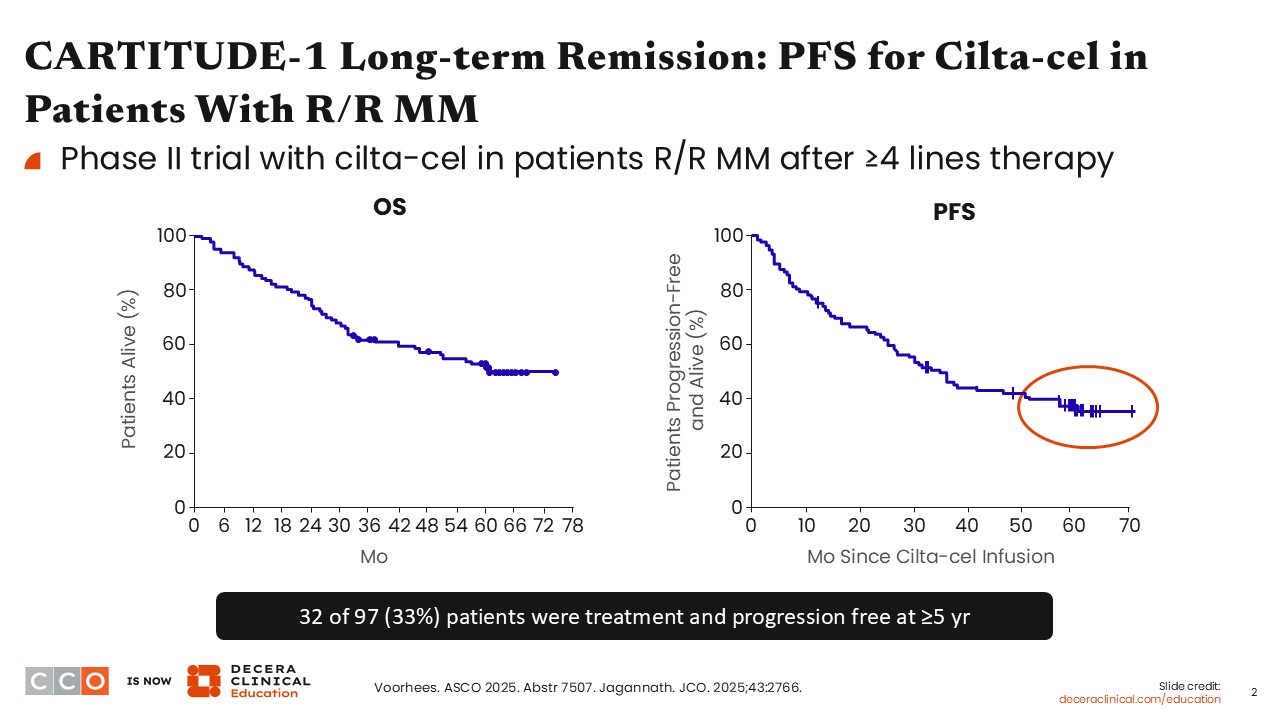
CARTITUDE-1 Long-term Remission: PFS for Cilta-cel in Patients With R/R MM
Long-term follow-up from the phase I/II CARTITUDE-1 trial of cilta-cel in heavily pretreated patients with R/R MM demonstrated a median PFS of 34.9 months among the 97 treated patients. Of note, approximately one third of patients remained progression free for at least 5 years without requiring additional MM therapy, reflected by a clear plateau in the PFS curve. Subsequent analyses examined clinical and disease-related characteristics associated with these durable responses, revealing meaningful differences between patients who achieved long-term remission and those who did not.1
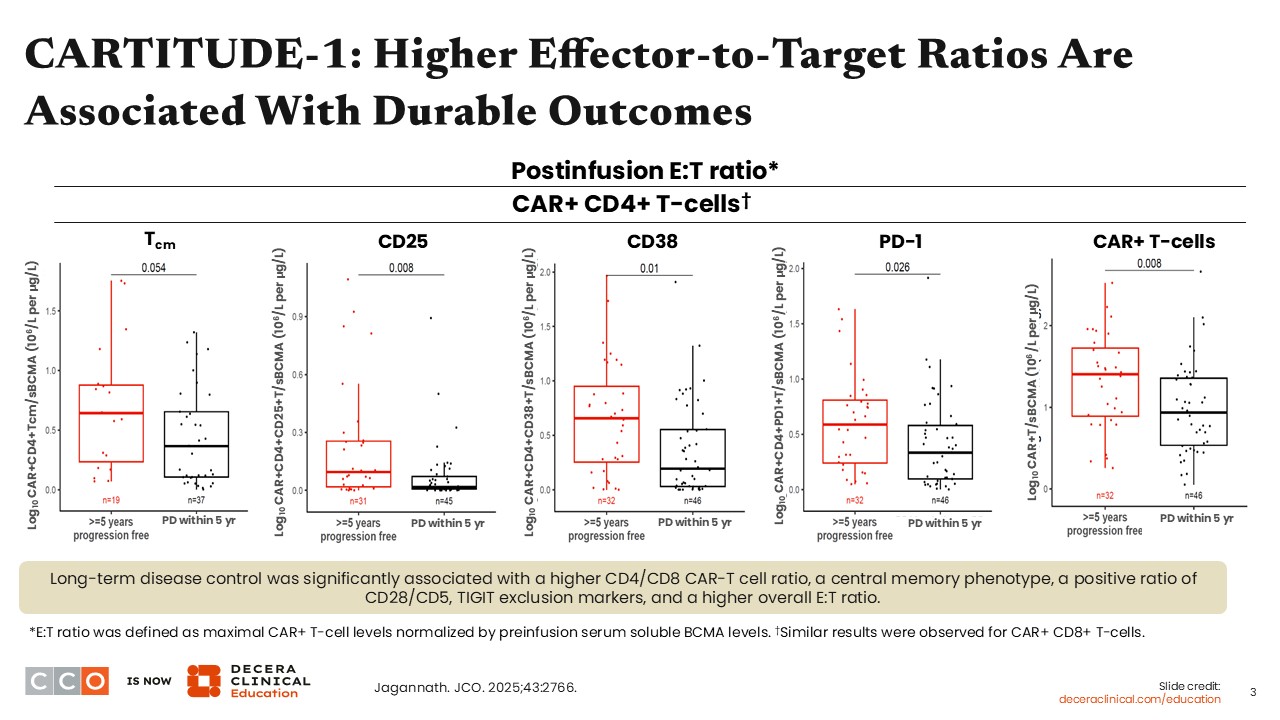
CARTITUDE-1: Higher Effector-to-Target Ratios Are Associated With Durable Outcomes
Most people assume that factors such as high-risk cytogenetics or the presence of extramedullary disease drive these long-term outcomes seen with cilta-cel in CARTITUDE-1. In reality, those features did not appear to matter. There were similar proportions of patients with high-risk cytogenetics and extramedullary disease among those who remained progression free at 5 years and those who did not.
What emerged as more important came from the translational analyses, specifically the effector-to-target ratio. After CAR T-cell infusion, patients who had a higher proportion of early, fitter T-cell phenotypes tended to have better outcomes. In addition, patients with a higher number of T-cells relative to MM tumor cells also did better overall. Put simply, the lower the MM burden and the greater the CAR T-cell presence, the better the response.1,2
This highlights a key modifiable factor in clinical practice. The main opportunity to influence this balance is during bridging therapy, where reducing MM burden before CAR T-cell infusion may improve outcomes once patients ultimately receive CAR T-cell therapy.
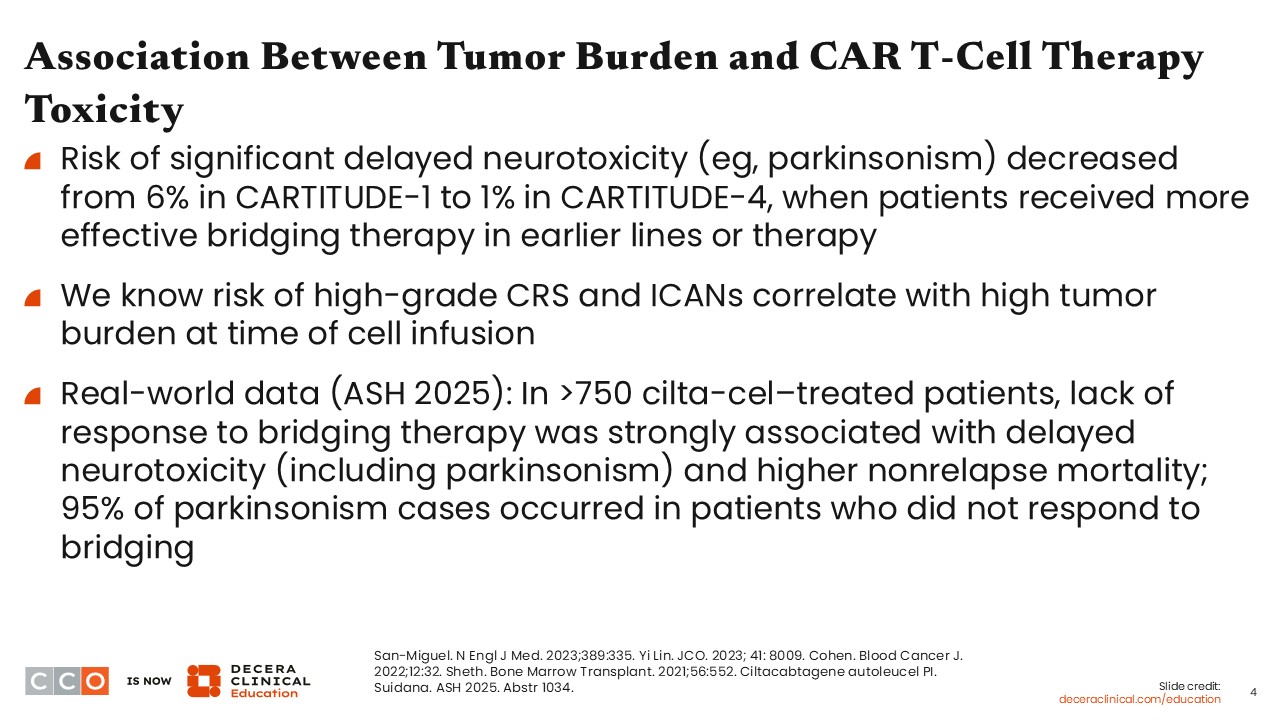
Association Between Tumor Burden and CAR T-Cell Therapy Toxicity
We have focused heavily on efficacy to this point, but toxicity also appears to correlate with tumor burden. Data show that the risk of significant delayed neurotoxicity, such as parkinsonism, is lower when disease burden is reduced. In CARTITUDE-4, the phase III trial evaluating cilta-cel in an earlier-line setting after 1-3 prior lines of therapy, the incidence of this delayed neurotoxicity was approximately 1% of patients. By contrast, in CARTITUDE-1, which enrolled a heavily pretreated population with ≥4 previous lines of therapy, the incidence was closer to 5%.3,8,10
One proposed explanation is that patients treated earlier in the disease course can receive more effective bridging therapy compared with those treated later. Supporting this, a higher proportion of patients in the CARTITUDE-4 trial responded to bridging therapy before receiving their CAR T-cells compared with patients in the CARTITUDE-1 trial.11,12
In addition, higher tumor burden at the time of cell infusion is associated with an increased risk of severe acute toxicities, including grade 3/4 cytokine-release syndrome (CRS), grade 3/4 immune effector cell–associated neurotoxicity syndrome, and immune effector cell–associated hemophagocytic lymphohistiocytosis-like syndrome. If we can reduce tumor burden prior to infusion, we may be able to lower the risk of these toxicities.12,13
This concept is supported by emerging real-world data. In large multicenter retrospective analysis from the US Multiple Myeloma Immunotherapy Consortium recently presented at ASH 2025, response to bridging therapy emerged as an important factor linked to toxicity risk among more than 750 patients treated with cilta-cel. Although most patients received some form of bridging therapy, only approximately one third achieved a response, and those who did not respond were significantly more likely to develop delayed neurotoxicity, including parkinsonism, as well as experience higher nonrelapse mortality. Of note, nearly all (21/22; 95%) patients who developed parkinsonism had disease that failed to respond to bridging therapy, even though many responded well to CAR T-cell treatment.14 Together, these findings suggest that how well disease is controlled before infusion, rather than CAR T-cell activity alone, plays a meaningful role in toxicity risk and highlight the importance of effective bridging strategies to reduce tumor burden prior to CAR-T infusion.
Who Needs Bridging Therapy Before CAR-T Therapy?
Who needs bridging therapy prior to CAR T-cell therapy in MM? In some cases, the answer is obvious, but in others it is less clear. Over time, my approach has shifted such that most patients receive some form of bridging therapy. There are very few situations in which I would simply observe, particularly because CAR T-cell manufacturing currently takes 4-6 weeks.
During that time, a great deal can change for patients, especially those with refractory disease in later lines of therapy or with a high disease burden. To put this into perspective, let’s walk through a few illustrative cases.
Case Study: Bridging Therapy in a Patient With High-Disease Burden
Mr S is a 64-year-old man with R/R MM who was originally diagnosed with IgG kappa MM in 2017. He initially received 8 cycles of bortezomib/lenalidomide/dexamethasone and deferred transplant, then transitioned to lenalidomide maintenance for 2 years before stopping because of toxicity. In 2023, he experienced biochemical progression and was started on daratumumab/pomalidomide/dexamethasone, achieving a partial response but again discontinuing therapy within 6 months because of toxicity. He presented for a second opinion after being off all therapy for more than a year because of concerns about adverse effects. At this presentation:
- M protein was elevated at 8.5 g/dL
- Light chain ratio remained relatively low at 10
- Hemoglobin 7 g/dL
- Creatinine 1.2 mg/dL
- PET/CT revealed diffuse lytic lesions, but no risk of fracture or extramedullary disease
Given his history of treatment-related toxicity, he expressed a strong preference for a one-and-done approach and elected to proceed with cilta-cel. He underwent apheresis, but this was an obvious case where bridging therapy was necessary, given his high disease burden and prolonged time off treatment. The challenge was selecting a regimen that he would tolerate, as he was extremely concerned about toxicity and there was even concern for tumor lysis. We chose a carfilzomib-based approach that he had not received previously, combined with daratumumab, but administered at low doses because of his concerns. Although his disease had been relatively indolent over the prior year without major organ damage, we anticipated that response would be gradual with this approach.
He ultimately received bridging therapy for 6 months, recognizing that collected CAR T-cells remain viable for up to 9 months and that a longer bridging period would allow for maximal disease reduction. He had an excellent response to bridging therapy, proceeded to CAR T-cell infusion, and experienced only grade 1 CRS. He recovered well, returned home on time, and is now doing very well clinically, approaching a complete response and currently in a very good partial response. This case highlights that some patients require extended or additional bridging therapy, particularly when aggressive regimens are not feasible and rapid disease control within 4-6 weeks is not realistic.
Case Study: When Minimal Disease Still Requires Bridging
The decision to use bridging therapy is not always straightforward. For example, consider Mr P, a 47-year-old Black man with IgA kappa MM diagnosed in 2024 with bone disease and renal insufficiency related to elevated kappa levels. His bone marrow demonstrated 70% aberrant plasma cells, with fluorescence in situ hybridization revealing t(14;16) and 1q amplification.
He started on a quadruplet therapy (daratumumab/bortezomib/lenalidomide/dexamethasone) for 4 cycles and achieved a very good partial response. That was followed by an autologous stem cell transplant and then bortezomib/lenalidomide maintenance because of his high-risk cytogenetics. He initially had a complete response after transplant and maintenance, but he was still measurable residual disease positive. Unfortunately, he had a biochemical progression with M-protein 0.5 g/dL (from 0 g/dL at nadir) 8 months after autologous stem cell transplant.
At that point, some might argue that this represented minimal disease, with stable hemoglobin and no active disease on PET imaging, and question whether bridging therapy was truly necessary if the CAR T-cell infusion was planned within 4-6 weeks. However, these are precisely the patients in whom I believe bridging therapy is critical. High-risk disease can progress rapidly, and what appears to be minimal disease can evolve into widespread bone or extramedullary involvement in a short period of time.
For newly diagnosed patients with high-risk features, I do not feel comfortable withholding all therapy during the CAR T-cell manufacturing window. In this case, we were able to initiate daratumumab/pomalidomide/dexamethasone quickly as bridging therapy. Once his CAR T-cells were ready, treatment was stopped, he had a 1 week break without toxicity, and he proceeded directly to CAR T-cell infusion. He responded well to bridging therapy, with a modest reduction in M-protein prior to infusion, and is now in a complete response with autologous stem cell transplant measurable residual disease undetectable for the first time. He continues to be monitored closely and is doing very well clinically.
Taken together, these 2 cases reinforce my general approach that most patients require some form of bridging therapy, with only a small number of exceptions. Patients with standard-risk disease, very indolent biochemical progression, and a long prior remission may be reasonable candidates for observation, although I still consider whether low-dose steroids are needed for disease control during the manufacturing period. Any patient with clinical relapse should receive bridging therapy to prevent worsening bone disease, renal dysfunction, or anemia before CAR T-cell therapy infusion.
Clinical Pearls for Choosing Bridging Therapy
The following considerations form the framework I use when deciding whether bridging is needed and, if so, how to select the most appropriate bridging regimen.
Disease biology and disease dynamics: Both overall disease burden and the anticipated rate of progression are critical factors. One patient may be standard risk but have a very high disease burden, whereas another may have lower disease volume but high-risk cytogenetics with the potential for rapid M-protein increase. Clinical risks and symptom burden also matter. Patients with bone pain or declining performance status because of progressive MM often require treatment to improve functional status before CAR T-cell therapy, particularly to ensure that they can tolerate any treatment-related complications. Reversing MM-related end-organ damage, including hypercalcemia or renal dysfunction, is an important goal before proceeding with CAR T-cell therapy.
Patient-specific and functional factors: Performance status is closely tied to disease burden, and comorbidities such as cardiac or renal disease may be exacerbated by active MM. Poorly controlled comorbidities can worsen tolerance of CAR T-cell therapy, especially in the setting of inflammatory toxicities that may require interventions such as fluid resuscitation, such as with CRS. Baseline cytopenias and marrow reserve are also critical considerations, as aggressive bridging therapy can worsen cytopenias and increase toxicity both from the bridging regimen itself and from subsequent CAR T-cell therapy, which is known to cause prolonged cytopenias. Prior therapies and patterns of resistance must be considered, since agents to which a patient is already refractory are unlikely to be effective for disease control during bridging therapy.
Immune and hematologic considerations: After cell collection, I am less concerned about transient effects on lymphocytes or neutrophils, although cytopenias still increase infection risk and should be minimized when possible. It is also important to avoid approaches that could impair CAR T-cell expansion or persistence. Although concerns about bispecific antibodies during bridging have lessened, intensive chemotherapy or excessive lymphodepletion can interfere with immune recovery and may promote excessive CAR T-cell expansion. Finally, it is important to distinguish between holding therapy and bridging therapy. Holding therapy refers to MM treatment given between CAR T-cell indication and apheresis and is primarily used to maintain disease control when delays occur because of insurance authorization or scheduling. During this period, therapies that may impair T-cell fitness (such as high-dose alkylator-based regimens or high-dose cyclophosphamide) should be avoided whenever possible. By contrast, bridging therapy refers to treatment administered after apheresis but before CAR T-cell infusion. Because T-cells have already been collected, bridging therapy can be selected with fewer constraints related to T-cell preservation and should focus on achieving disease control prior to infusion.9,11
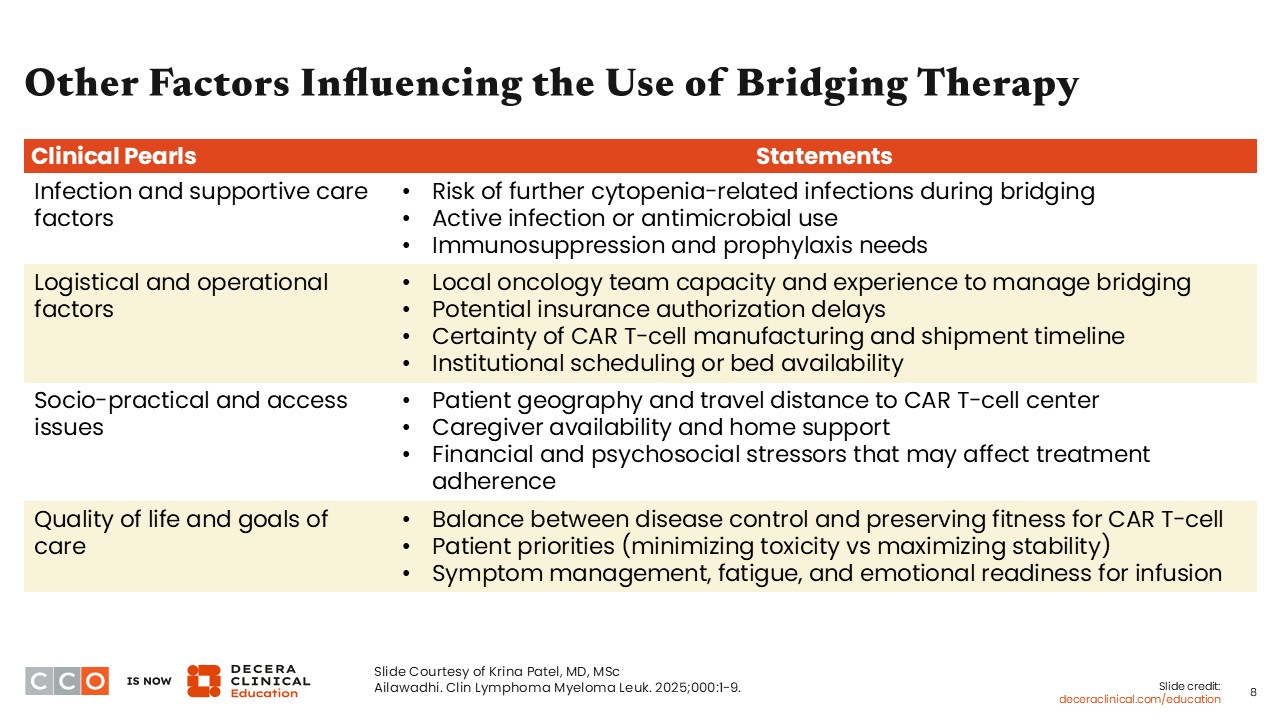
Other Factors Influencing the Use of Bridging Therapy
Additional factors center on infection risk, supportive care, and practical considerations. We do not want patients to have active infections during bridging therapy or at the time of CAR T-cell infusion. Overly aggressive bridging therapy that worsens cytopenias can increase the risk of infection and contribute to greater toxicity once CAR T-cell therapy is administered.
Logistical and operational issues are also important. Some patients live far from the treatment center and may not want or be able to receive 1-2 cycles of bridging therapy on site. In these situations, it is important to consider what treatments are available locally and whether the local team has the capacity to deliver them safely and efficiently.
Access to specific therapies can be a limitation, particularly for late-line patients. Many referring treatment centers do not yet have the ability to administer bispecific antibodies, which may require patients to return to the CAR-T treatment center for bridging therapy. This can add substantial burden in terms of travel, time away from work, and demands on caregivers. Broader socioeconomic and access issues also come into play, including travel costs, caregiver availability, and whether local teams can initiate therapy promptly.15
Finally, quality of life and patient goals of care are central to these decisions. Many patients choose CAR T-cell therapy because of the possibility of a one-and-done approach and a desire to minimize time spent receiving treatment. Effective bridging therapy supports this goal by helping patients arrive at their CAR T-cell therapy administration in better condition and return home on time with fewer complications. Achieving this often requires close coordination with local care teams to ensure that the chosen bridging regimen can be delivered as intended.9,11
Current Challenges With Bridging Therapy Before CAR T-Cell Administration
One of the major challenges in late-line disease is that most patients are already penta exposed to carfilzomib, bortezomib, pomalidomide, lenalidomide, and anti-CD38 antibodies, and many are refractory to some or all of these agents. Novel targeted bispecific therapies, such as talquetamab, represent an important option, but not all referring community centers currently have the infrastructure to administer them, although access is gradually improving.6,16 When these options are not available locally, patients may need to travel to specialized centers or rely on alternative regimens that their local teams can deliver.
In those situations, options may include hyperfractionated cyclophosphamide–based therapy, dexamethasone/cyclophosphamide/etoposide/cisplatin, or bortezomib/dexamethasone/thalidomide plus cisplatin/doxorubicin/cyclophosphamide/etoposide, all of which typically require hospitalization and can significantly affect quality of life. These regimens often lead to severe cytopenias requiring transfusions and granulocyte colony-stimulating factor support, which can delay CAR T-cell infusion because of treatment-related toxicities or infections. This creates challenges with washout timing, particularly in high-risk patients with a large disease burden who require aggressive disease control. Although the goal is to avoid prolonged periods off therapy, cytopenias can paradoxically extend the washout period and undermine the original intent of bridging therapy.9,11
Ultimately, reducing tumor burden remains critical, but this must be balanced against the risks of increased toxicity, longer washout times, and potential negative effects on CAR T-cell therapy outcomes. Another challenge arises when patients do not respond to bridging therapy. When alternative options are available, switching to a different approach is appropriate, particularly if it may offer better disease control with less toxicity. However, in patients who have exhausted available therapies, shared decision-making becomes essential. In these cases, it is important to have an open discussion about the higher risks associated with proceeding to CAR T-cell therapy and to decide together whether moving forward remains the best option.
The Need for Better Biomarkers to Assess Disease Burden
These challenges highlight how central disease burden is to bridging therapy decisions, but they also expose a critical limitation in our current approach. We frequently talk about reducing tumor burden to improve safety and outcomes, but in many patients receiving late-line therapy, we lack reliable tools to accurately measure how much disease is truly present or how it is changing over time.
In patients receiving late-line therapy, disease is often oligosecretory or nonsecretory, making M-protein, light chains, and Bence-Jones protein unreliable measures of tumor volume, and although markers such as baseline ferritin and bone marrow plasma cell percentage have been explored, ferritin is a nonspecific acute phase reactant, and marrow involvement alone does not capture disease outside the marrow.17 Although PET/CT imaging can identify bony and extramedullary disease, we currently lack standardized, quantitative assessments of total disease burden, and radiology reports rarely provide this level of detail, highlighting a significant unmet need in biomarkers to guide CAR T-cell therapy decision making and toxicity risk assessment.18
Final Thoughts: The Evolving Role of Bridging Therapy in the CAR T-Cell Process
We have made substantial progress since the first CAR T-cell therapy approvals in 2021. With the move toward earlier-line use, patients are achieving better outcomes, in part because bridging strategies are more effective and disease responses to bridging therapy are improving. However, there remain specific patient populations in whom we need a better understanding of how to optimize bridging therapy to reduce disease burden and improve outcomes before CAR T-cell infusion, as well as ongoing limitations in the ability of referring community treatment centers to administer the full range of available bridging therapies.
Looking ahead, advances such as faster manufacturing or off-the-shelf CAR T-cell products may shift some of these challenges. For now, however, bridging therapy remains a central component of CAR T-cell therapy and requires deliberate, thoughtful planning, including coordination with community partners, rather than being viewed as a secondary step in the CAR T-cell therapy process.