CE
APP Perspectives: Managing Toxicities With Bispecific Antibodies in Multiple Myeloma
Physician Assistants/Physician Associates: 0.50 AAPA Category 1 CME credit
Nurse Practitioners/Nurses: 0.50 Nursing contact hour
Released: November 07, 2025
Expiration: May 06, 2026
Activity
Using Bispecific Antibodies in MM
The treatment landscape for MM continues to evolve rapidly, with bispecific antibody–based strategies emerging as an important option for patients with R/R disease. This module will review current indications for bispecific therapies, highlight key safety considerations, and discuss strategies for managing AEs. This module will also include approaches to communication with patients and their caregivers, as well as ways to align treatment decisions with individual patient goals.
When to Treat R/R MM
When considering a change in therapy for a patient with MM, an important first step is to determine whether the patient is experiencing a clinical relapse or a biochemical relapse. This assessment is guided by the CRAB criteria, which focuses on symptoms such as hypercalcemia, renal dysfunction, and bone lesions.11,12
The focus of this discussion is patients who are more heavily pretreated and who may be candidates for bispecific antibodies. In practice, it is important to help patients and their care partners understand the difference between biochemical changes that signal disease progression and the point at which treatment changes are warranted. Preparing patients in advance allows for clearer expectations and smoother transitions when initiating a new therapy. A good model for these conversations follows an “if/then” format. For example, “If your lab results start to show signs that the disease is becoming more active and you develop symptoms, then we may need to talk about starting a new treatment.”
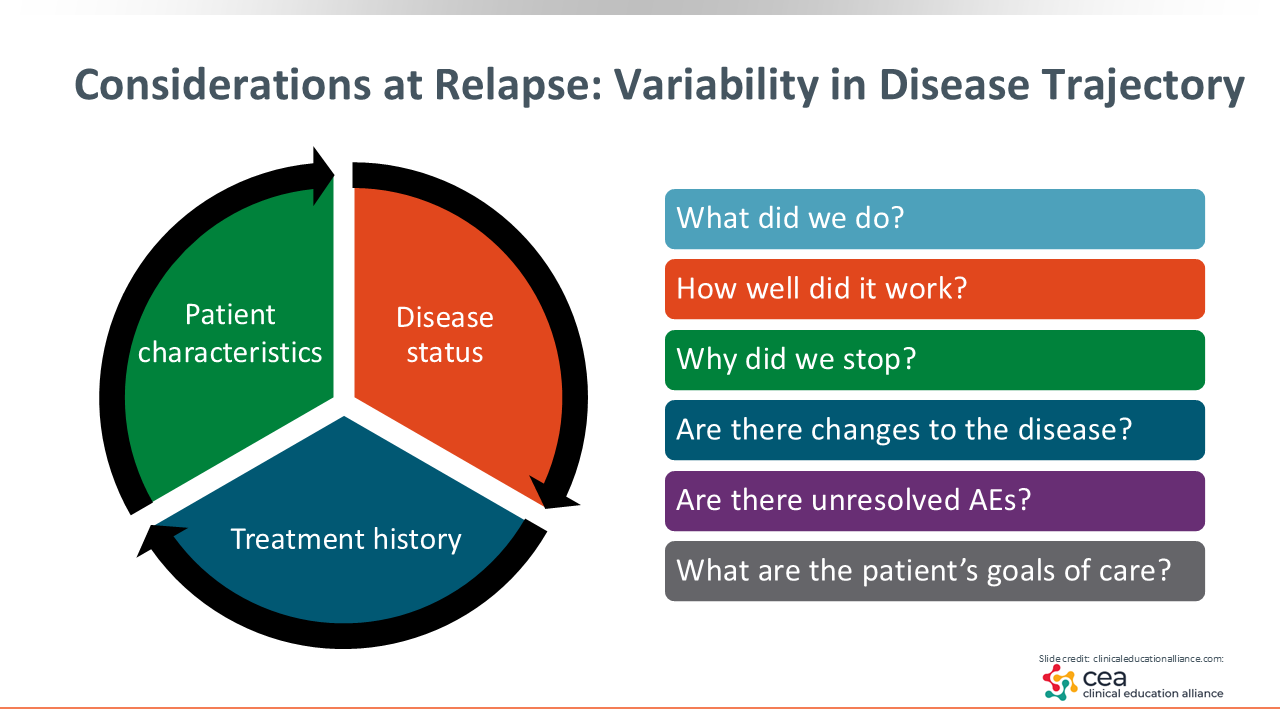
Considerations at Relapse: Variability in Disease Trajectory
When selecting a therapy for patients with MM, several key factors should be considered beyond the immediate disease status. We are looking at the patient in front of us. We are also looking at their disease burden and their prior treatment history. What did we give them previously? How did it work?
Evaluating these elements provides critical context for determining the most appropriate next line of therapy. This phase is also an opportunity to restage the disease and engage patients and their care partners in understanding the rationale for reassessment. Restaging helps identify new or evolving concerns, such as changes in disease biology, and can influence subsequent treatment choices.
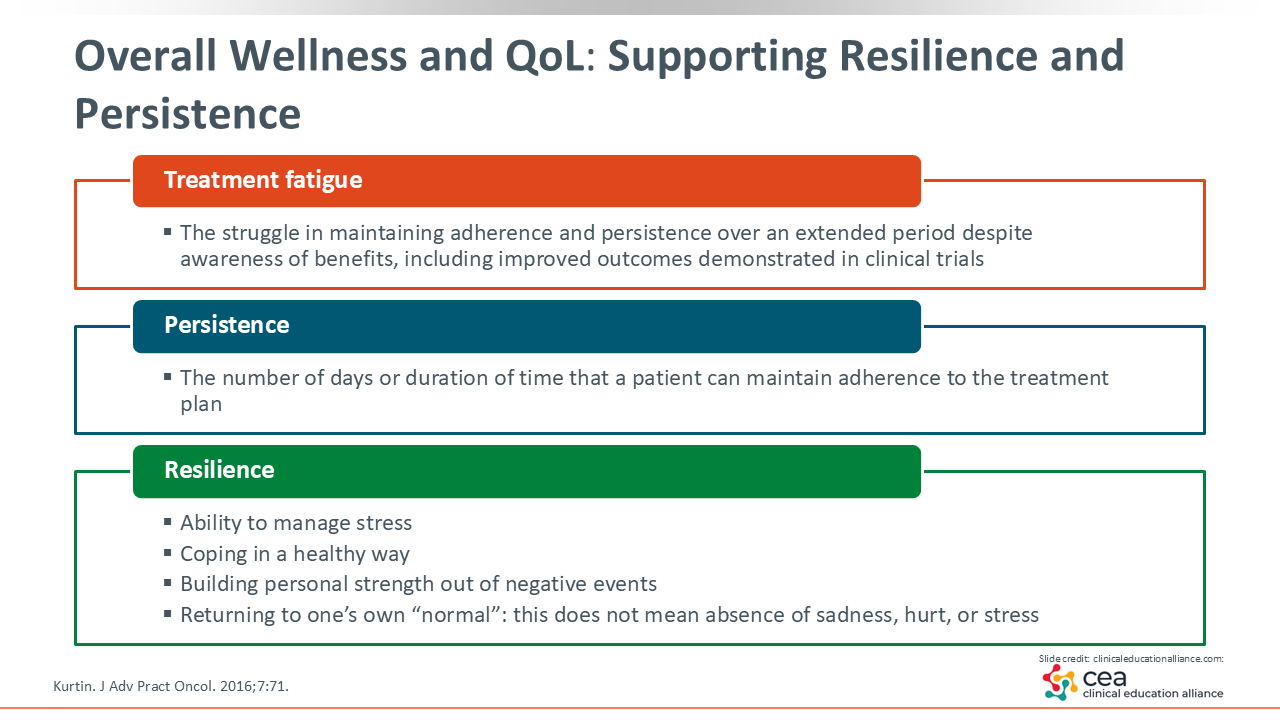
Overall Wellness and QoL: Supporting Resilience and Persistence
In addition to disease-related factors, we also want to evaluate each patient's overall wellness and quality of life (QoL), particularly for those who have received multiple prior lines of therapy. Healthcare professionals (HCPs) should assess how patients are coping with ongoing treatment, including signs of treatment fatigue, which is the struggle to maintain adherence and persistence over an extended period despite awareness of the benefits, as well as difficulties maintaining therapy schedules or the need for extended breaks.
Building on this assessment, it is also important to evaluate broader aspects of well-being. This includes assessing how patients are managing stress, whether they have adequate transportation and access to care, and identifying support needs. Engaging in these conversations at the time of therapy transition helps ensure comprehensive, patient-centered care and promotes sustained treatment success.13
These discussions help HCPs meet patients where they are and provide valuable context for framing conversations about potential next lines of therapy. For instance, if a patient is experiencing treatment fatigue or facing logistical challenges with their current regimen, introducing bispecific antibodies as a next option can be positioned around their potential convenience and QoL benefits. Bispecific antibodies are generally well tolerated once patients complete the initial step-up dosing phase, and treatment often transitions to biweekly or monthly maintenance dosing, which can further enhance convenience and overall QoL.14
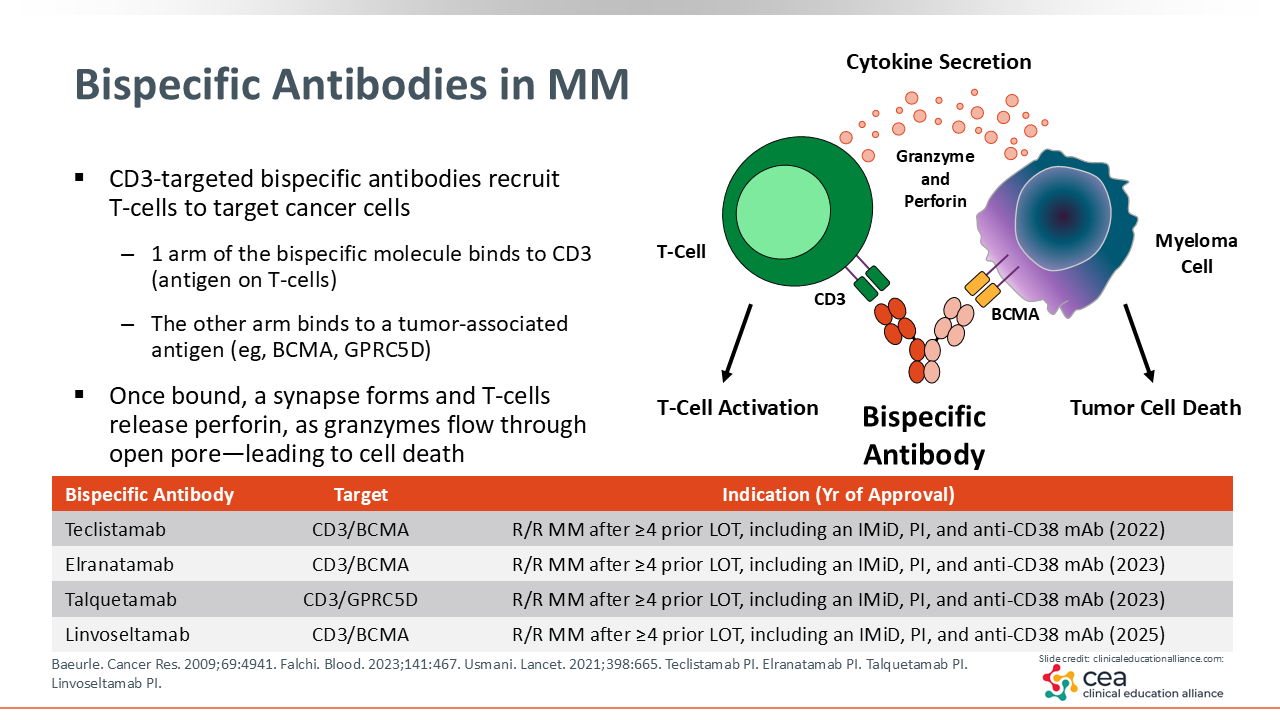
Bispecific Antibodies in MM
As of October 2025, there are 4 FDA-approved bispecific antibodies for the treatment of R/R MM. These agents are designed to bind distinct antigens, such as BCMA or GPRC5D, along with CD3 proteins on T-cells.15 Three FDA-approved bispecific antibodies target BCMA: teclistamab, elranatamab, and linvoseltamab. A GPRC5D-targeted bispecific antibody, talquetamab, is also FDA approved. All 4 agents are indicated for the treatment of adult patients with R/R MM who have received at least 4 prior lines of therapy, including a proteasome inhibitor, an immunomodulatory drug, and an anti-CD38 monoclonal antibody.10,16-18 Understanding the mechanisms and targets of bispecific antibodies is essential for managing AEs, as I will discuss later in this module. Although there are some common class effects in terms of CD3-binding bispecific antibody AEs, BCMA- andd GPRC5D-targeted agents differ in some anticipated AEs, and recognizing these differences can help guide monitoring and counseling strategies.
Bispecific Antibodies: Issues Affecting Access
When initiating bispecific antibody therapy, a key consideration is determining the appropriate timing and clinical context for treatment. In the current landscape, the positioning of CAR T-cell therapy relative to bispecific antibody use plays an important role. HCPs should assess whether a patient is relapsing after CAR T-cell therapy, as this can influence bispecific selection. Some patients may be CAR T-cell therapy naive but considered for future treatment or may receive a bispecific antibody as a bridging therapy approach to maintain disease control during the CAR T-cell manufacturing process. Others may not be suitable candidates for CAR T-cell therapy. Clarifying these scenarios is essential for guiding both agent selection and overall treatment planning.12,14
We also have to consider the logistics of administering bispecific antibodies while balancing safe monitoring practices with ease of access. Many large, academic institutions perform step-up dosing in the inpatient setting to monitor early toxicities, requiring brief hospitalization and caregiver presence. However, an increasing number of centers are successfully transitioning bispecific antibody initiation to the outpatient setting. Other potential barriers to access such as insurance coverage, travel costs, overnight stay, and long-term toxicity management should be considered proactively.19
Communication Between Academic and Community Centers
Many patients begin bispecific antibody therapy at academic centers and, when feasible, transition back to their referring community centers for ongoing care. This collaboration is essential to ensure continuity and patient convenience. A thorough handoff should include details about the step-up phase, observed side effects, and ongoing management needs, such as antiviral prophylaxis, IVIG replacement (either in-center or through home health), and use of growth factors when appropriate.
In cases where the community site cannot yet administer bispecific therapy, shared-care models can still work. Academic centers may give the bispecific antibody while the community team manages supportive measures like IVIG and local lab monitoring. Strengthening these partnerships helps patients receive care closer to home while maintaining oversight from experienced teams. Increasing familiarity with bispecific antibody management and navigating REMS requirements will continue to expand community access, and potential future changes to REMS programs may further streamline this process. Clear communication protocols, especially during the step-up phase, are critical so that patients know when and how to contact their care team.
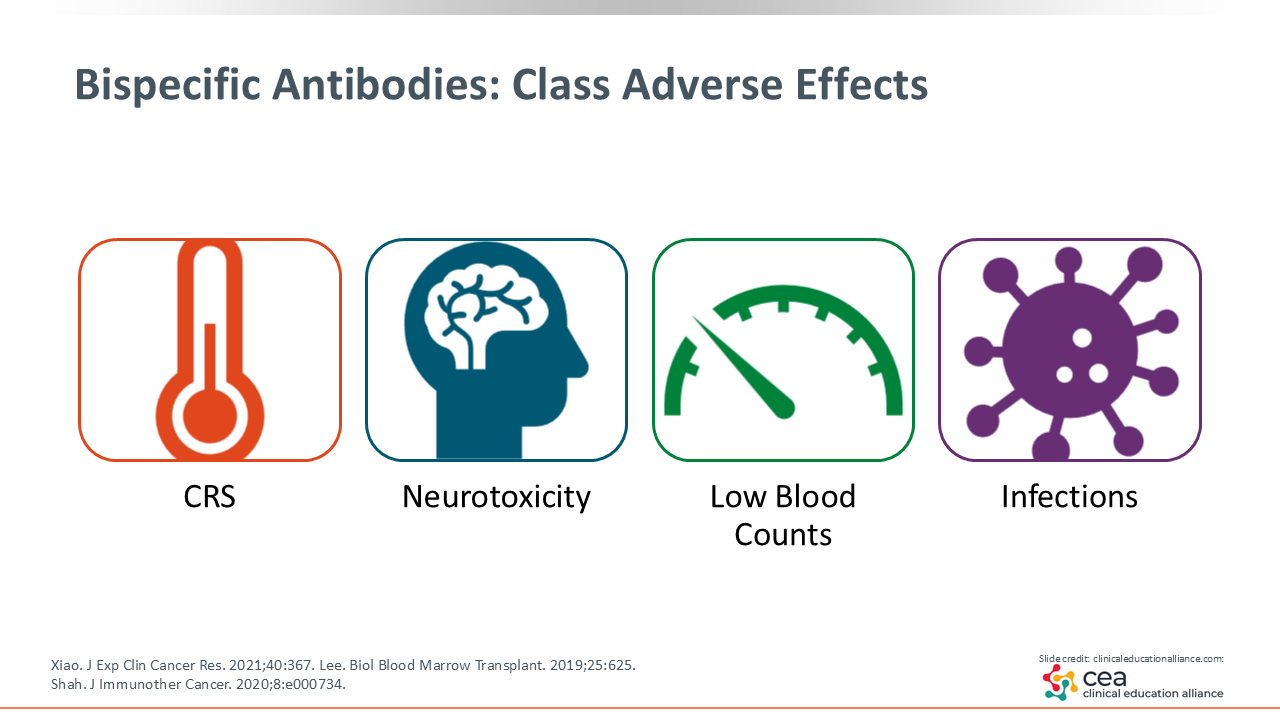
Bispecific Antibodies: Class Adverse Effects
One of the most important factors affecting the accessibility and effective use of bispecific antibodies is the proactive identification and management of AEs. These agents are generally well tolerated, and as HCPs gain more experience with them, AEs are typically manageable when appropriate monitoring and supportive care strategies are implemented.
Four class-wide AEs are particularly important when considering bispecific antibodies: neurotoxicity, CRS, cytopenias, and infections. Neurotoxicity is relatively uncommon, and cytopenias are generally manageable. CRS is also important, but with the right precautions and monitoring, it can be effectively controlled. Infections, however, are the most challenging to manage over the course of bispecific antibody therapy, especially when coordinating care between inpatient and outpatient settings, so I will spend more time reviewing key considerations for infection management.20-22
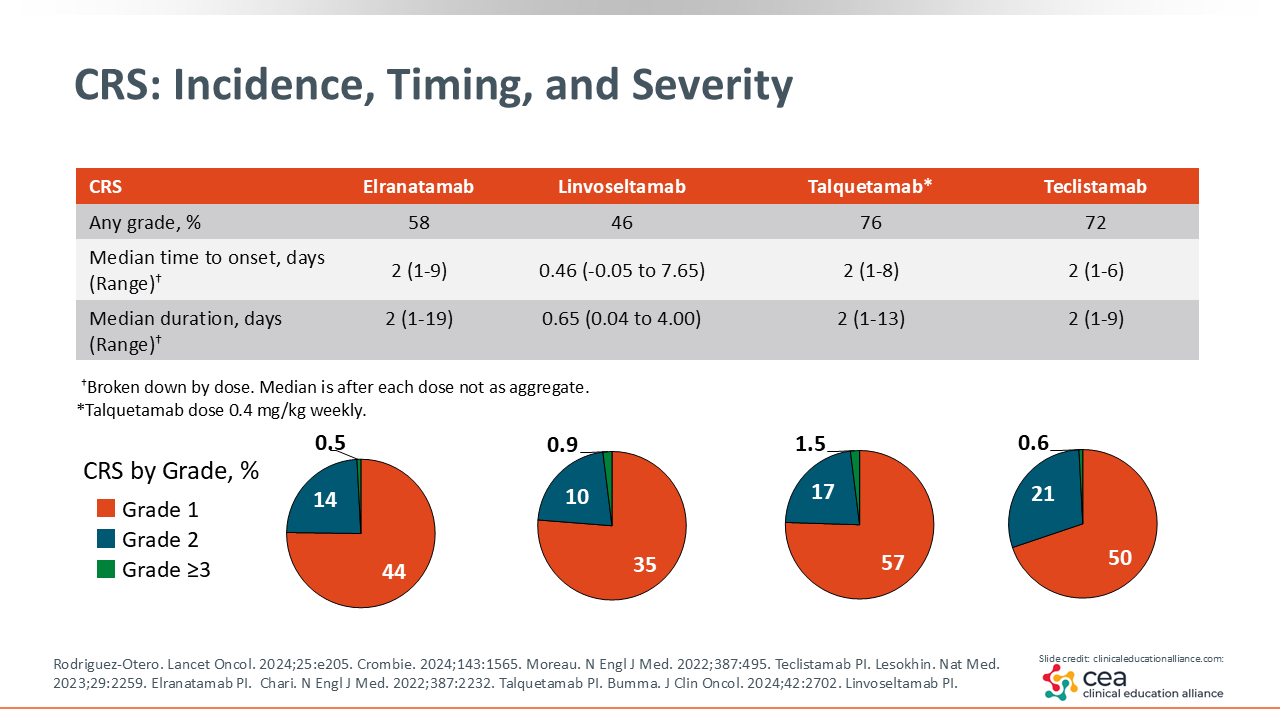
CRS: Incidence, Timing, and Severity
CRS associated with bispecific antibody therapy follows a more predictable onset pattern when compared with the onset and incidence of CRS seen in patients receiving CAR T-cell therapy.23,24 For agents such as elranatamab, talquetamab, and teclistamab, the median time to CRS onset is approximately 2 days. Linvoseltamab demonstrates even earlier onset, often within the first 12 hours of treatment. The majority of CRS events are grade 1 in severity and can be effectively managed with established supportive measures.2-6 At our center (Winship Cancer Institute), we administer prophylactic tocilizumab; some other centers have begun adopting this approach as well. By giving tocilizumab before the first dose, we rarely observe significant CRS.25
CRS: Grade-Based Management
There are several ways to manage CRS with several well-established strategies for risk mitigation and treatment. The hallmark symptoms are fever greater than 38°C without hypotension or hypoxia accompanied by other flu-like symptoms. Grade 1 CRS can be managed with dexamethasone and early use of tocilizumab alongside supportive care with hydration and antipyretics. You will also want to follow institutional guidelines for infection workup.
Grade 2 CRS is marked by fever and presence of hypotension, not requiring vasopressors or hypoxia requiring low-flow oxygen. Mainstays of treatment include supplemental oxygen, fluids, and tocilizumab. If the patient shows no improvement, consider adding dexamethasone.
Grades 3 and 4 CRS are characterized by an increase in severity, both warranting transfer to an intensive care unit.6,26
Grades 1 and 2 CRS are most commonly observed in patients undergoing treatment with bispecific antibodies. At this severity level, the reaction is typically predictable, resolving promptly with appropriate therapeutic intervention. Premedication is indicated during the step-up dosing phase of treatment and not typically given after a patient reaches full dose.19
We also use additional strategies to manage CRS that allow patients to remain outpatient and self-monitor. For example, patients are provided with dexamethasone to keep at home, so if they call with symptoms, we can instruct them to take the medication and come in for monitoring. This approach provides greater flexibility in treatment while maintaining safety.27
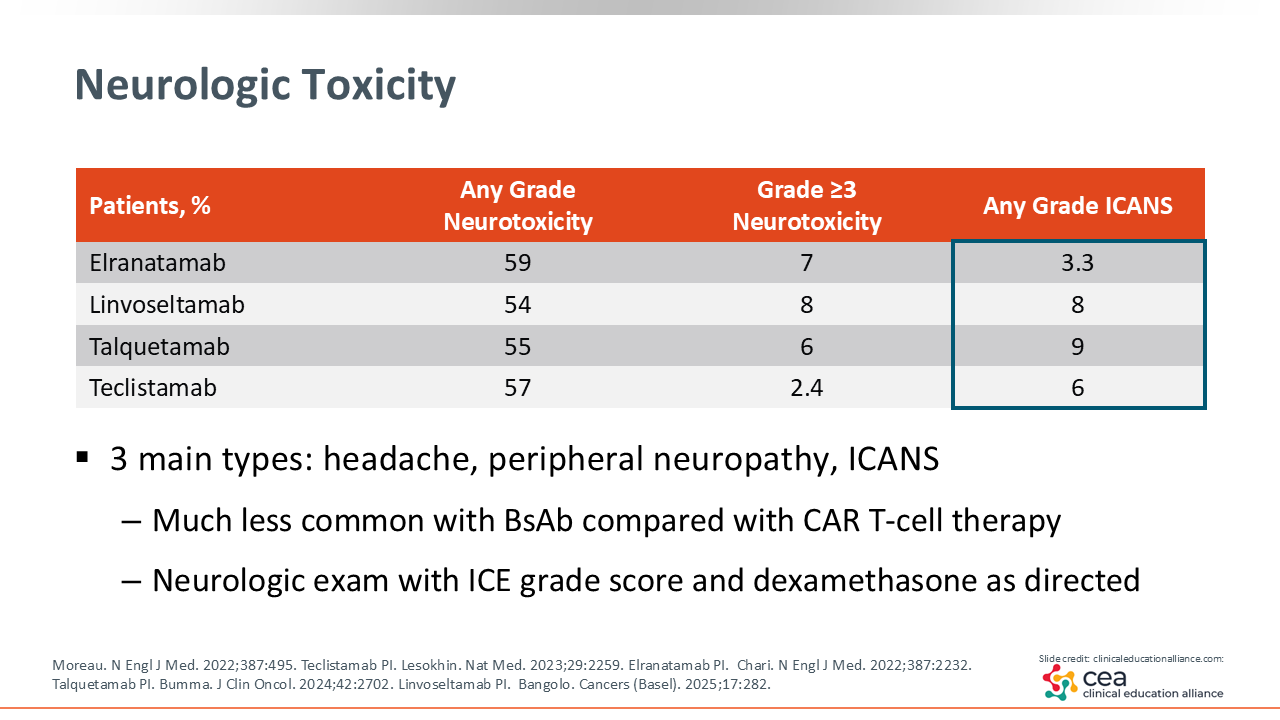
Neurologic Toxicity
Neurotoxicity is an AE more commonly associated with CAR T-cell therapy than with bispecific antibody treatment in patients with MM.28 The primary neurotoxic events include headache, peripheral neuropathy, and ICANS. ICANS may occur concurrently with or following CRS. However, it is observed more frequently in patients receiving CAR T therapy.29 Routine neurologic assessment using the ICE score is recommended for early detection, with dexamethasone administered as directed for management.
Class Effect: Incidence of Infection with Bispecific Antibodies in MM
A key consideration during bispecific antibody therapy is a patient’s susceptibility to infection throughout the course of treatment. Infections represent the most frequently observed AE and can significantly affect both treatment continuity and patient QoL. In clinical trials of BCMA-targeting bispecific antibodies (elranatamab, linvoseltamab, and teclistamab) any-grade infection rates have ranged from approximately 70% to 76%.2,3,5, The GPRC5D-targeting bispecific antibody talquetamab demonstrated a somewhat lower rate of infection: approximately 59% in clinical trials.4
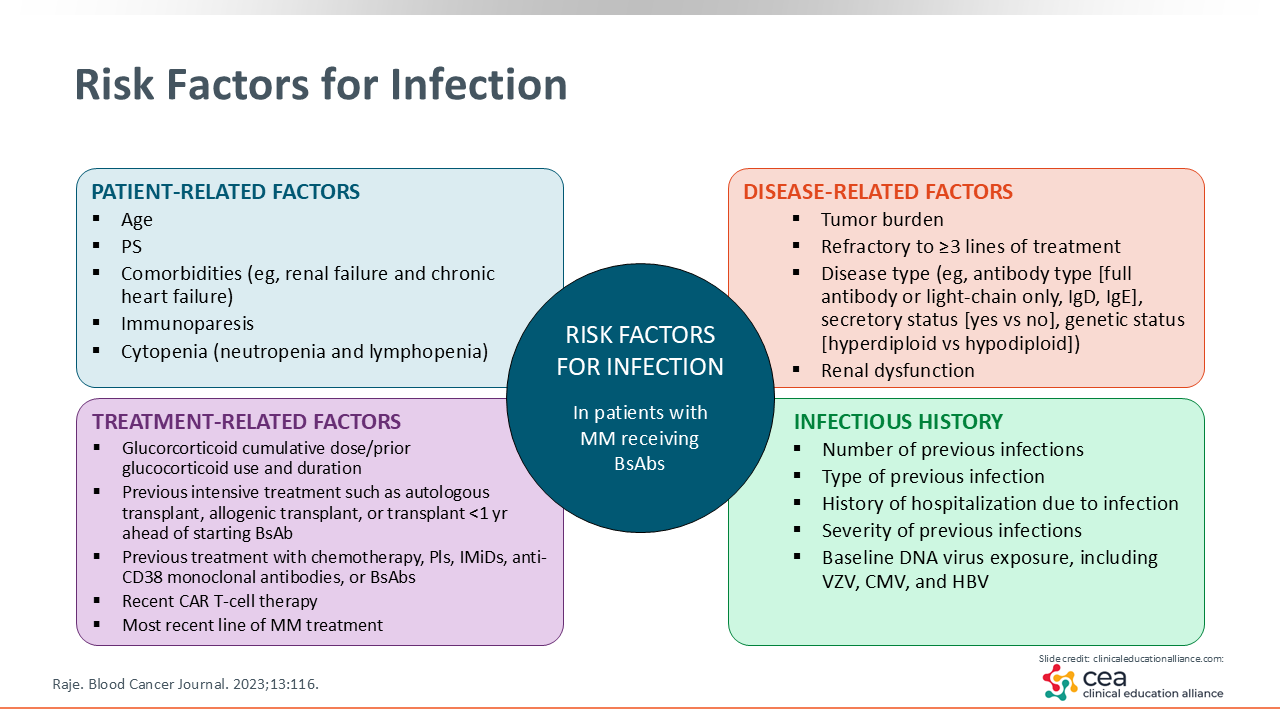
Risk Factors for Infection
Risk factors for infections can include patient-related factors such as age, comorbidities, immunosuppression status, and cytopenia, as well as disease-related factors including tumor burden. For early-stage treatment of myeloma, decreased immune cell counts may be more prevalent but usually improve with bispecific antibody treatment. Other factors to consider are a patient’s history, including previous treatments, past AEs, type and frequency of previous infections, prior hospital admissions, and risk of pneumonia. Along with this, patients that have recently undergone CAR T-cell therapy prior to the initiation of a bispecific antibody will be more immunosuppressed.1
Preventing Infection With Bispecific Antibody Therapy
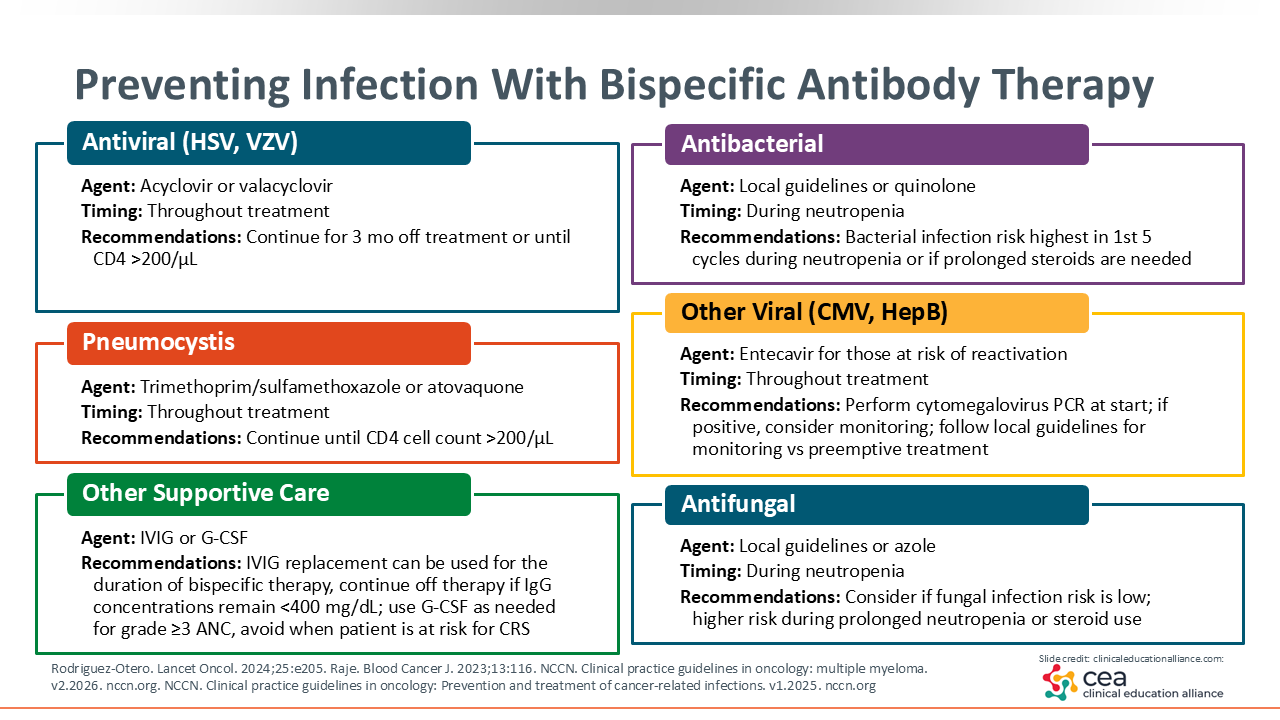
What are the recommendations for patients going on bispecific antibodies? Antiviral prophylaxis with acyclovir or valacyclovir is strongly recommended. As myeloma patients are typically on continuous therapy, antiviral prophylaxis is continued until 3 months post treatment or immune recovery (CD4 >200/μL).
Pneumocystis prophylaxis is another key consideration for patients with myeloma, with standard treatment including trimethoprim/sulfamethoxazole or atovaquone. Although not all institutions monitor CD4 count recovery, some HCPs recommend pneumocystis prophylaxis be continued until the patient’s CD4 count is above 200/μL. For agents like talquetamab, discontinuation can be considered following a patient’s sustained CD4 count recovery.
Supportive care includes monthly intravenous immunoglobulin (IVIG). Specific insurance coverage may require the use of subcutaneous self-administered IVIG. Although prior recommendations indicate IVIG therapy in patients with an IgG level of less than 400, recent clinical experience suggests that continued infection risk for patients with higher IgG levels warrants treatment. Because of this, some HCPs have begun initiating IVIG regardless of IgG level. Granulocyte colony-stimulating factor (G-CSF) can also be used for immune replacement therapy.
Cytomegalovirus (CMV) and hepatitis B (HBV) screenings and history impact aspects of bispecific therapy treatment. Although CMV-positive screenings may be observed, they do not always require treatment. Treatment protocols may vary by institution but usually include close monitoring of CMV viral load alongside temporary discontinuation of bispecific antibody therapy. If CMV levels persist following dose reduction or discontinuation, antiviral treatment can be initiated following protocol. Patients with a history of HBV are at risk for viral reactivation and should undergo baseline HBV serologic testing before starting therapy. Prophylactic treatment with entecavir should also be considered to reduce this risk.1,6,7,12
Management of Cytopenias
Management of cytopenia follows established protocol and clinical judgement. Neutropenia requires regular monitoring, with G-CSF administered as indicated to maintain adequate neutrophil counts. In the case of febrile neutropenia, bispecific antibody therapy should be held, with antibiotic therapy initiated as per institutional guidelines.8
Anemia in this setting tends to be resolved as disease control is established. However, red blood cell transfusion and erythropoietin-stimulating agents should be considered in symptomatic patients or those with hemoglobin less than 8 g/dL.
Finally, thrombocytopenia can be addressed with supportive transfusion when platelet counts fall below institutional thresholds. Current bispecific antibody product labeling indicates that therapy should be temporarily discontinued until platelet count is 25,000/mcL or higher and no evidence of bleeding.10,16-18
When performing an infection workup, HCPs should consider evaluating for additional viral etiologies such as adenovirus or Epstein-Barr virus. Comprehensive testing in patients with persistent, unexplained cytopenia or viral-like symptoms can help to identify co-occurring infections contributing to immune suppression.6,7,8,12
Adverse Events Associated With GPRC5D-Targeted Treatments
Talquetamab differs from other bispecific antibodies used in MM as it targets GPRC5D rather than BCMA. This unique mechanism of action is accompanied by target-related side effects as GPRC5D is expressed in hair follicles, oral mucosa, skin and nail beds.
As a result, patients may experience AEs such as taste changes or disturbances, weight loss, skin and nail changes, and rash during early phases of treatment. These events typically occur during the initial treatment cycles, often stabilizing with continued treatment or supportive care interventions.4,30
Pearls for Managing GPRC5D-Associated AEs
It is important to ensure that patients starting talquetamab are counseled on the unique GPRC5D-related AEs and that a clear management plan is in place to address these AEs should they occur.
Skin and nail care should emphasize oral and topical hydration. Sensitive areas such as the palms and feet may develop erythema and begin peeling, which can be managed with urea-based cream to reduce friction. Mild inflammation and pruritis can be treated with topical creams such as triamcinolone or ammonium lactate, and diffuse rash may require short-course oral steroids and antihistamines. Fungal infection must be routinely assessed and treated if identified. Presentation of nail changes may resemble those seen in patients with graft-vs-host disease, with symptoms such as jagged, cracked nails, or even nail loss. Advising patients to keep nails clean and avoid the use of nail hardeners or nail polish can lessen the risk and severity of nail dystrophy. Severe skin or nail changes may warrant temporary discontinuation or dose reduction in talquetamab therapy alongside dermatologic consultation, and side effects typically resolve following dose modification.
For mucosal irritation, emphasis should be placed on maintaining good oral hygiene and adequate hydration. Baking soda or sodium chloride rinses can also help soothe discomfort and promote healing. If mucosal irritation leads to difficulty eating or unintended weight loss, involving a dietitian as part of the care team can help support nutritional intake and overall well-being.
In my practice, I have found that patients often report higher sensitivity to sweet, sour, or bitter tastes, while salty and umami tastes are more palatable and increase saliva production. Sugar-free lozenges and artificial saliva spray can also assist in oral hydration.
Routine assessment for oral candidiasis and appropriate treatment is critical; budesonide oral slurry (“swish and swallow”) can be used for management. Candidiasis in this setting is graded based on symptom prevalence and is used to determine when to hold or decrease bispecific therapy.9,30
Transitioning patients to less frequent dosing schedules, such as monthly administration when feasible, has been associated with better tolerability and sustained adherence without compromising efficacy.6,24
New Achievements Reveal New Challenges
As the clinical landscape of oncology changes, breakthroughs and revolutionary treatments have helped to increase the lifespan of patients with myeloma. With this achievement, challenges and new considerations must be incorporated in treatment. As patients live longer, they often accumulate age-related comorbidities, and as treatment options expand, they are exposed to an increasing number of therapies over extended periods of time. Most MM treatments are continuous with rare treatment-free periods, with the exception of CAR T-cell therapy. Novel therapeutic mechanisms introduce new toxicity profiles, underscoring the importance of early AE recognition and management. Close collaboration with community partners is essential to ensure optimal supportive care and improve patient outcomes.32,33
Ultimately, one of the most critical components of successful therapy is comprehensive patient support, not only from the HCP, but the entire care team. Finally, it is important to remember that patient education, shared decision-making, and proactive counseling are key in maintaining QoL and positive outcomes.