CME
Novel Agents, New Combinations: An Expert Overview of Unmet Needs and Emerging Therapies for DLBCL
Physicians: Maximum of 1.50 AMA PRA Category 1 Credits™
Released: January 30, 2026
Expiration: July 29, 2026
Activity
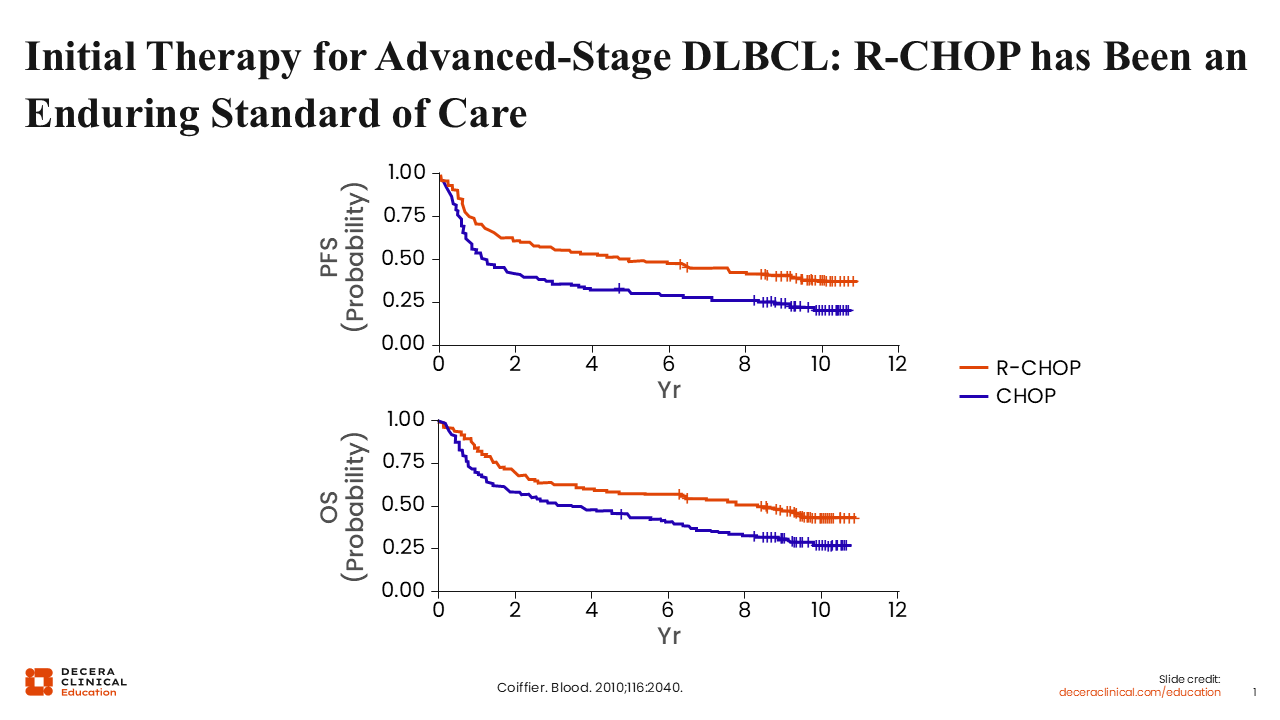
Initial Therapy for Advanced-Stage DLBCL: R-CHOP Has Been an Enduring Standard of Care
Let’s begin by looking at initial management of DLBCL. R-CHOP was the standard frontline regimen for many patients with advanced-stage DLBCL. The last major transformation in initial therapy was the introduction of rituximab to the CHOP regimen, which significantly improved both progression-free survival (PFS) and overall survival (OS).1 This paradigm remained a standard of care for approximately 20 years. However, recently, a new drug entered the treatment paradigm for patients with newly diagnosed DLBCL.
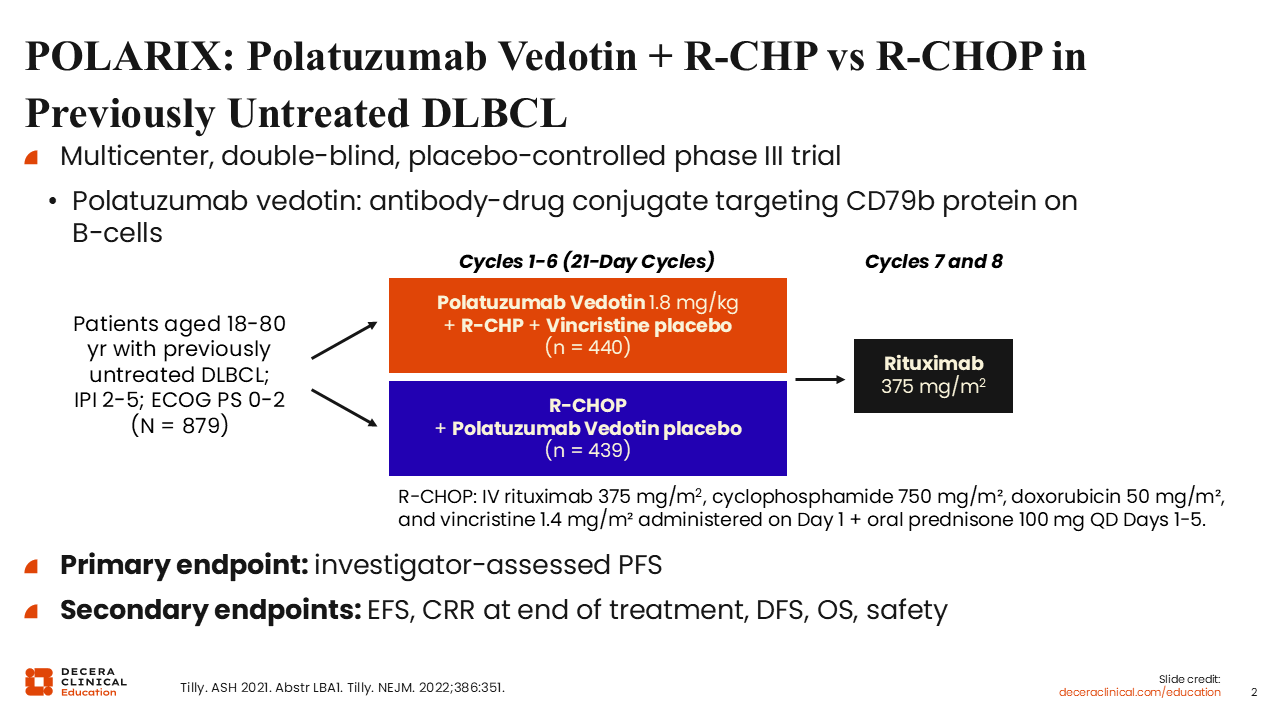
POLARIX: Polatuzumab Vedotin + R-CHP vs R-CHOP in Previously Untreated DLBCL
In the phase III POLARIX trial, patients with previously untreated DLBCL were randomized to receive polatuzumab vedotin plus R‑CHP or R-CHOP.2 Polatuzumab vedotin is an antibody–drug conjugate consisting of an anti‑CD79b antibody linked to the microtubule inhibitor monomethyl auristatin E. Eligible adult patients were up to 80 years of age and had an IPI score of 2-5.
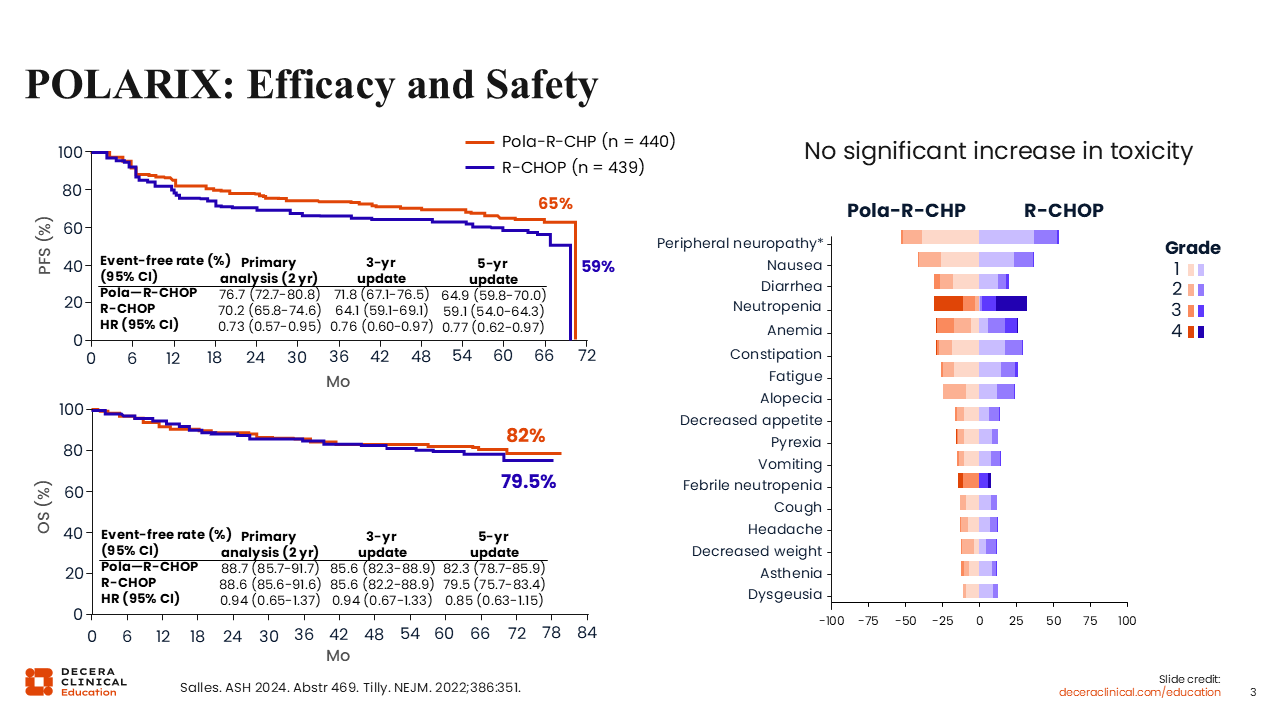
POLARIX: Efficacy and Safety
Updated results from the POLARIX trial demonstrate a PFS benefit with pola-R-CHP vs R-CHOP, showing an absolute improvement of approximately 7% at 5 years.3 This advantage was not associated with increased toxicity, as safety profiles were comparable between the 2 arms. No difference in OS has been observed. These findings have supported the adoption of the pola-R-CHP regimen for patients with previously untreated DLBCL and an IPI score of ≥2.
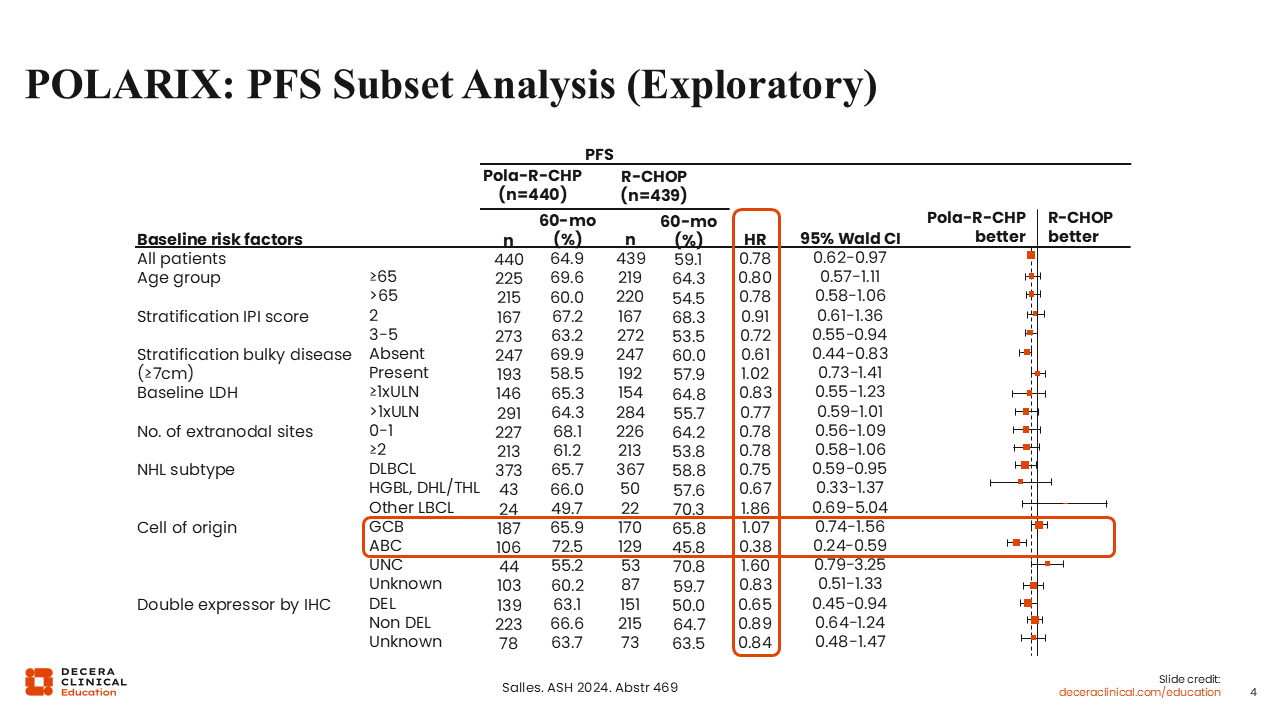
POLARIX: PFS Subset Analysis (Exploratory)
A subset analysis from the POLARIX trial, although exploratory and underpowered, provides insight into potential biological differences between DLBCL subtypes. Although the trial demonstrated a 7% improvement in PFS, this exploratory analysis suggested that the benefit was primarily observed in the ABC subtype, rather than in the germinal center B-cell (GCB) subtype.3 Some healthcare professionals have proposed restricting pola-R-CHP use to patients with the ABC or non-GCB subtype; however, such interpretations should be made with caution.
More recently, evidence with comprehensive molecular clustering suggested benefit is observed predominantly within higher-risk ABC tumors, which may constitute up to a quarter of DLBCLs.4 As we develop additional newer therapies for the upfront management of DLBCL, we will have to look more closely within molecular substructure to determine which novel treatment approach might be best for individual patients.
Settings Where Regimens Other Than R-CHOP or Pola-R-CHP May Be Preferable in LBCLs
Certain subgroups of patients with LBCLs require other treatment regimens. Very old or frail individuals may receive less intensive options such as R-mini-CHOP or nonanthracycline-containing regimens.5 Specific DLBCL subtypes, including primary mediastinal B-cell lymphoma and double-hit lymphomas, often benefit from DA-EPOCH-R. Primary central nervous system (CNS) DLBCL is distinct, requiring CNS-penetrating regimens. Primary testicular DLBCL, carries a higher risk of CNS and contralateral testicular relapse, warranting CNS-directed therapy and prophylactic scrotal radiation. Therefore, treatment selection should be individualized based on patient characteristics and disease subtype.
Overall, in the frontline setting, pola-R-CHP is selected for most patients with an IPI score of ≥2, particularly IPI ≥3. DA-EPOCH-R is used for specific LBCL subtypes such as double-hit lymphoma and primary mediastinal lymphoma, whereas R-CHOP remains an option for low-risk patients.
Before continuing, let’s revisit a question from earlier in the activity.
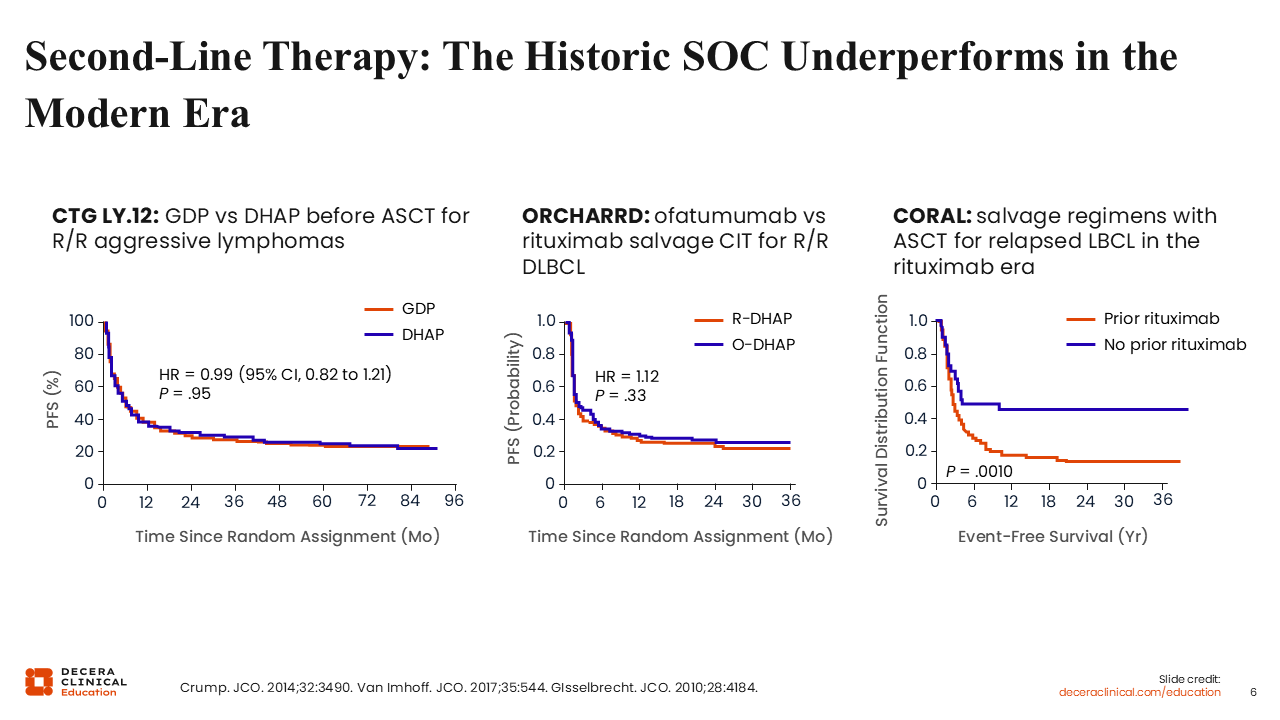
Second-line Therapy: The Historic SoC Underperforms in the Modern Era
Where I think we have seen the most advances recently is in the R/R setting, particularly in second-line therapy for DLBCL. In this setting, there was a long-standing standard of care involving platinum-based chemotherapy followed by high-dose chemotherapy and autologous stem cell transplantation (ASCT) in eligible patients. In the pre-rituximab era, potential cure rates were 50% to 60% with this approach. However, in the rituximab era, when more patients were potentially cured with upfront therapy, long-term remission after ASCT decreased to <25%, particularly among those with early relapsed or primary refractory disease. The CORAL trial demonstrated potential cure rates of ≤15% in patients relapsing within 1 year, emphasizing the need for improved therapeutic strategies.6-8
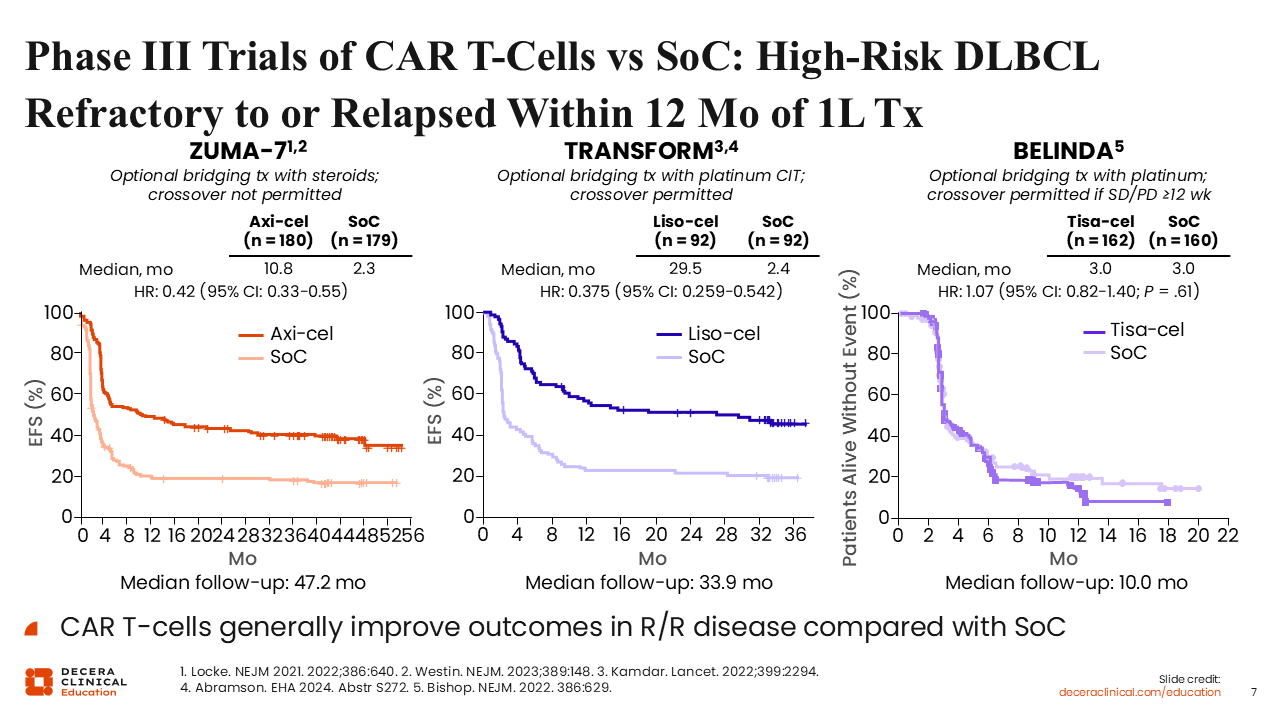
Phase III Trials of CAR T-Cells vs SoC: High-Risk DLBCL Refractory to or Relapsed Within 12 Months of First-line Tx
Three randomized trials evaluated CAR T-cell therapy as a potential replacement for second-line chemotherapy and ASCT in transplant-eligible patients with primary refractory or early relapsed DLBCL. The 3 CAR T-cell therapies investigated were axi-cel, liso-cel, and tisagenlecleucel (tisa-cel). Both the ZUMA-7 (axi-cel) and TRANSFORM (liso-cel) met their primary endpoints and led to regulatory approval, whereas BELINDA (tisa-cel) did not meet its primary endpoint.9-11
ZUMA-7 and TRANSFORM completely changed our standard of care, making either liso-cel or axi-cel now the preferred second-line treatment for patients relapsing after frontline chemoimmunotherapy, particularly if they had primary refractory disease or relapsed disease within 1 year. These 2 studies showed substantial improvement in the primary endpoint of event-free survival and PFS, with both leading to a >50% reduction in risk of progression or death, making these substantially better than a traditional chemotherapy and transplant-based approach.
With CAR T-cell therapies, specific toxicities related to the T-cell activation process are observed, and there were differences between liso-cel, which is a 4-1BB costimulated CAR T-cell, and axi-cel, which is a CD28 costimulated CAR T-cell. These different costimulation domains lead to different kinetics of CAR T-cell expansion, leading to lower incidence and severity of cytokine-release syndrome (CRS) and neurologic toxicity with liso-cel compared with axi-cel, although these therapies have not been directly compared in a prospective clinical trial.
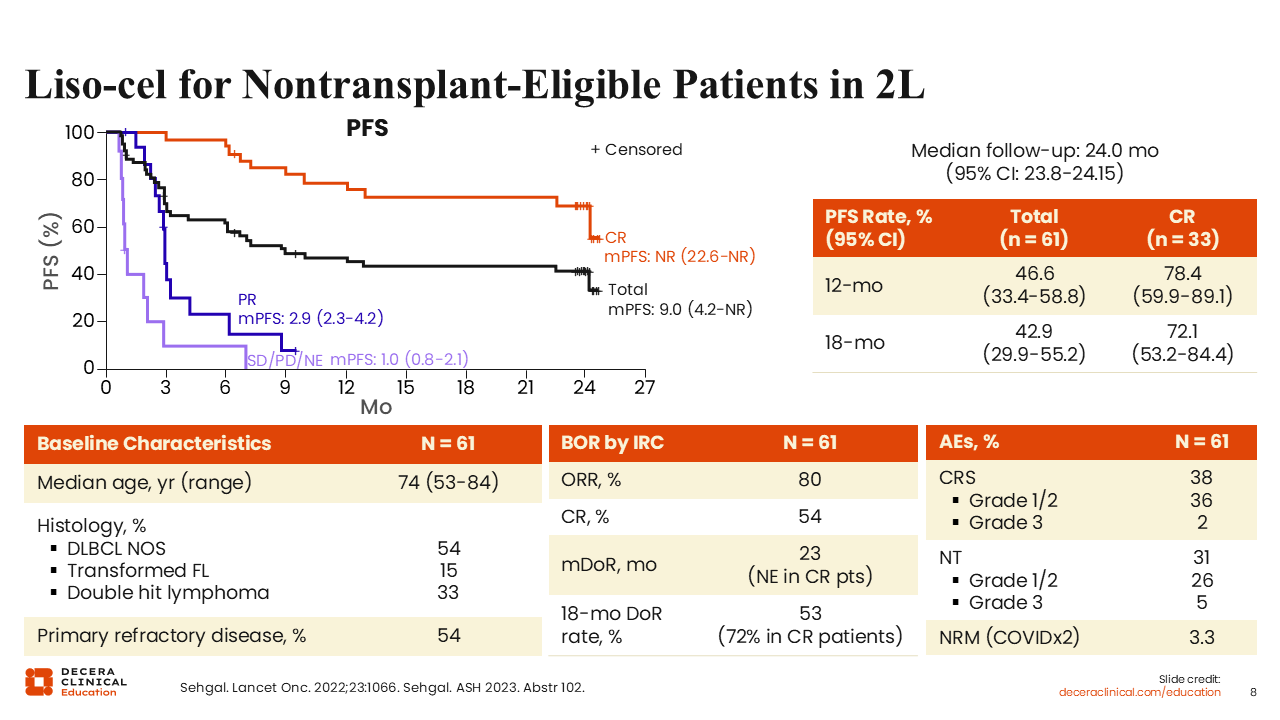
Liso-cel for Nontransplant-Eligible Patients in the Second Line
Historically, patients with DLBCL who are ineligible for ASCT have lacked potentially curative treatment options. Given that most of these patients remain eligible for CAR T-cell therapy, this approach was evaluated in the pivotal PILOT trial, a phase II study that led to approval of liso-cel as a second-line treatment for patients with refractory disease to first-line chemoimmunotherapy or relapse after first-line chemoimmunotherapy and who are not eligible for transplantation. The ORR was 80% with a CR of 54%. Responses were durable, with a median PFS of 9 months and a plateau in the PFS curve indicating that >40% of patients remained progression free at 2 years, suggesting potential cure in this population previously lacking curative intent therapies. Treatment was well tolerated, even among older and frailer patients. High-grade CRS and neurotoxicity were uncommon (2% and 5%, respectively), and no grade 4/5 events were observed.12
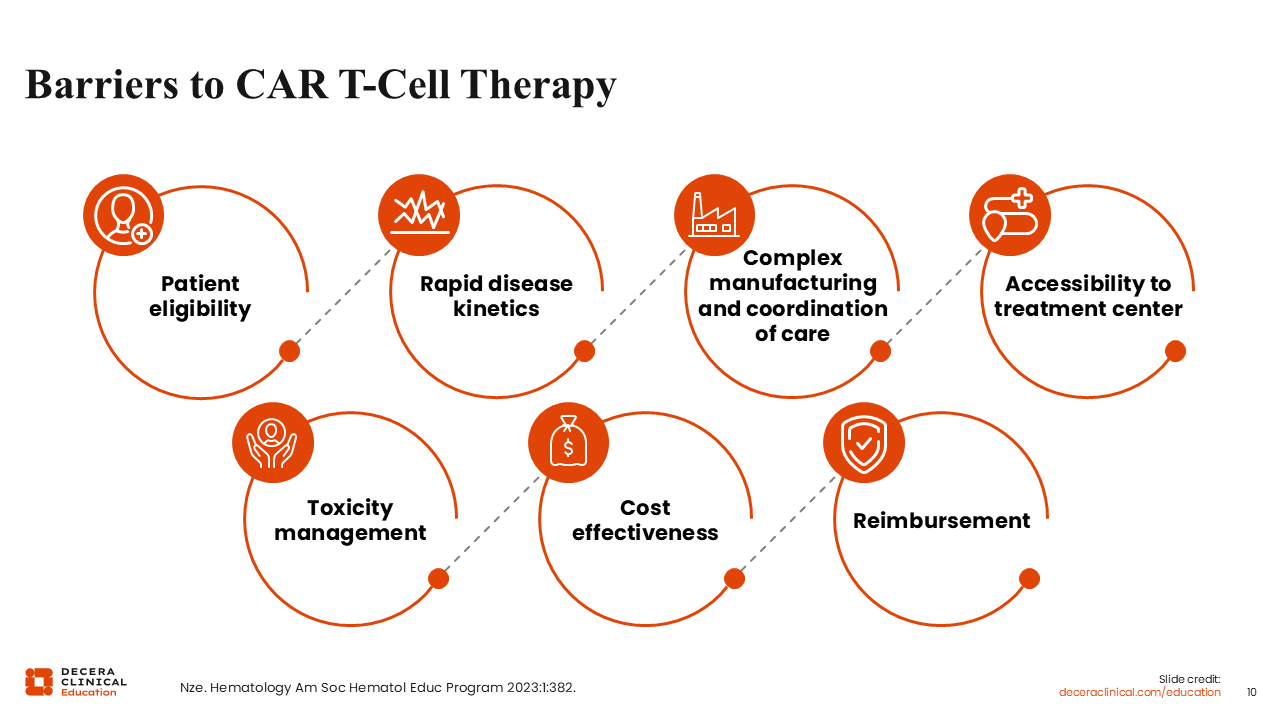
Barriers in CAR T-Cell Therapy
Despite demonstrating efficacy, <25% of eligible patients in the United States currently access CAR T-cell therapy. Barriers include limited geographic availability (with approximately 200 centers nationwide), the need for prolonged relocation, socioeconomic constraints, and the aggressive nature of DLBCL that may not allow the time required for the CAR T-cell process in a patient with rapidly progressive, bulky, or symptomatic disease. Cost and logistical challenges further limit accessibility, underscoring the need for alternative, readily available therapeutic options to treat patients.13
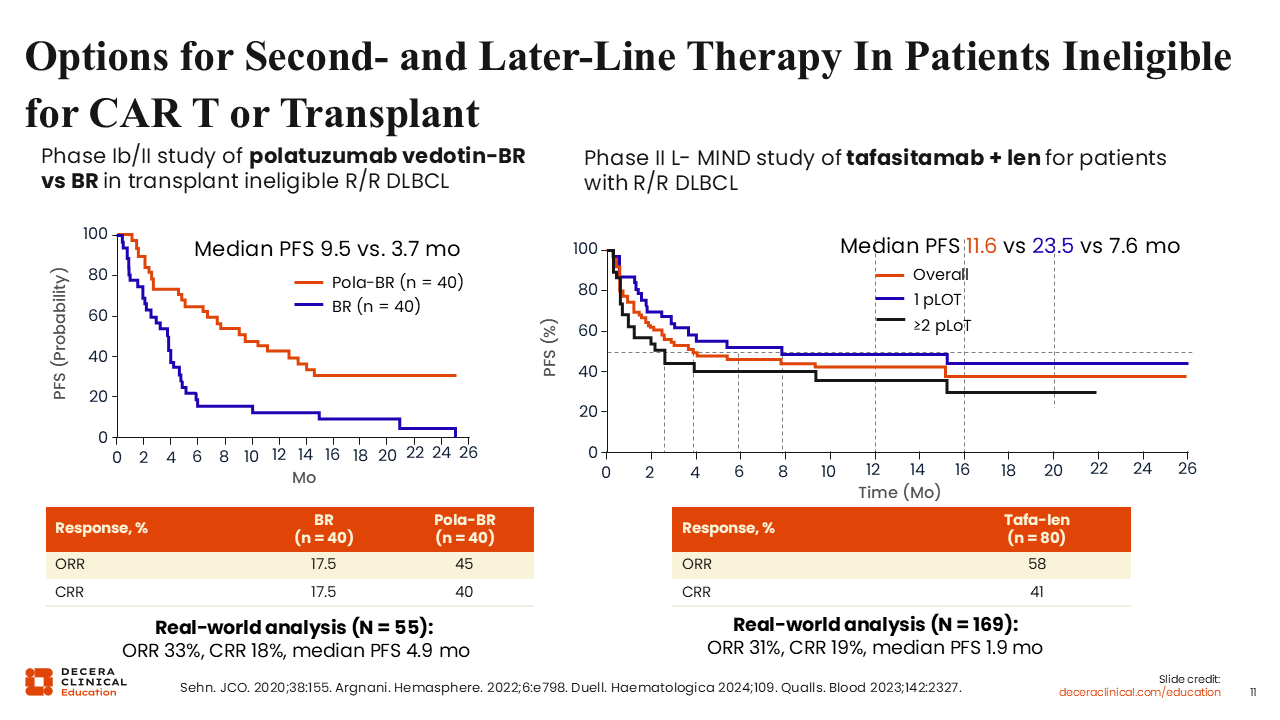
Options for Second-line and Later-Line Therapy in Patients Ineligible for CAR T-Cell Therapy or Transplant
In this space, novel alternative regimens such as polatuzumab vedotin plus bendamustine/rituximab and tafasitamab/lenalidomide achieve CR rates of approximately 40% in clinical trials, with considerably lower CR rates in real-world analyses.14-17 This suggests that these more conventional options are not serving the majority of our patients who so not have access to CAR T-cell therapy. This raises the question of wouldn't it be great, then, if we could bring a highly active next-generation immunotherapy into second-line treatment setting for these patients?
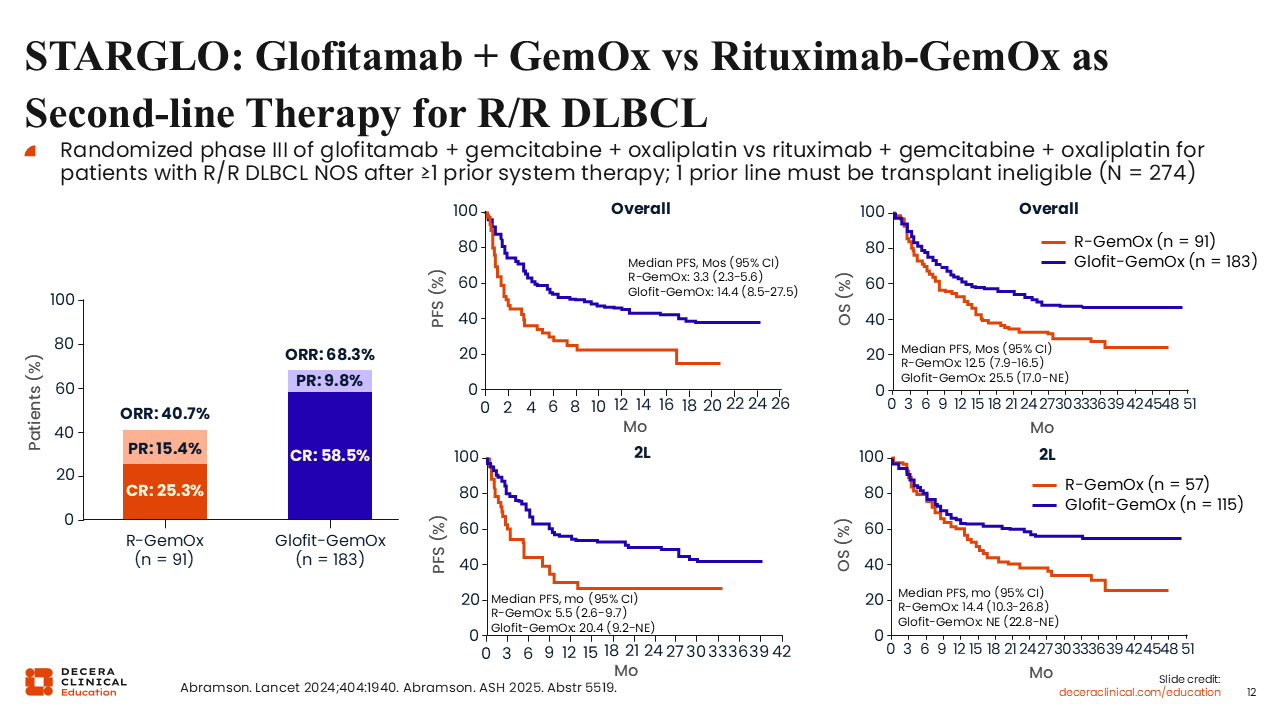
STARGLO: Glofitamab + GemOx vs Rituximab-GemOx as Second-line Therapy for R/R DLBCL
Bispecific antibodies, particularly glofitamab and epcoritamab, were initially approved by the FDA in the third-line and later setting for DLBCL. Recent trials sought to determine if these agents might provide an off-the-shelf option to induce deep and durable remissions as second-line therapy, particularly for patients who are not eligible for transplant and may not have access to CAR T-cell therapy.
The STARGLO trial randomized patients with DLBCL who were considered transplant-ineligible to glofitamab-GemOx or rituximab-GemOx (R-GemOx).18,19 Most patients—63%—were treated in the second-line setting.
Updated data show substantial improvement in ORR, CR rate, PFS, and the primary endpoint of OS favoring glofitamab-GemOx over R-GemOx in second-line or later-line treatment for DLBCL. The CR rate for glofitamab-GemOx was nearly 60% in this study, compared with only 25% with R-GemOx. The median PFS was 14.4 months with glofitamab-GemOx vs 3.3 months with R-GemOx, amounting to a 60% reduction in the risk of progression or death; similarly, the median OS was 25.5 months with glofitamab-GemOx vs 12.5 months with R-GemOx.
Among patients treated in the second-line setting in STARGLO, the median PFS was 20.4 months with glofitamab-GemOx vs 5.5 months with R-GemOx. Similarly, the median OS was not reached with glofitamab-GemOx vs 14.4 months with R-GemOx.
This demonstrates the potential for long-term remission and possible cures in a large patient population, without CAR T-cell therapy.
There is a risk of CRS with the bispecific antibody. That risk was modest at 44% and almost entirely low-grade, occurring predictably during the very first cycle of step-up dosing used to minimize the risk of CRS.
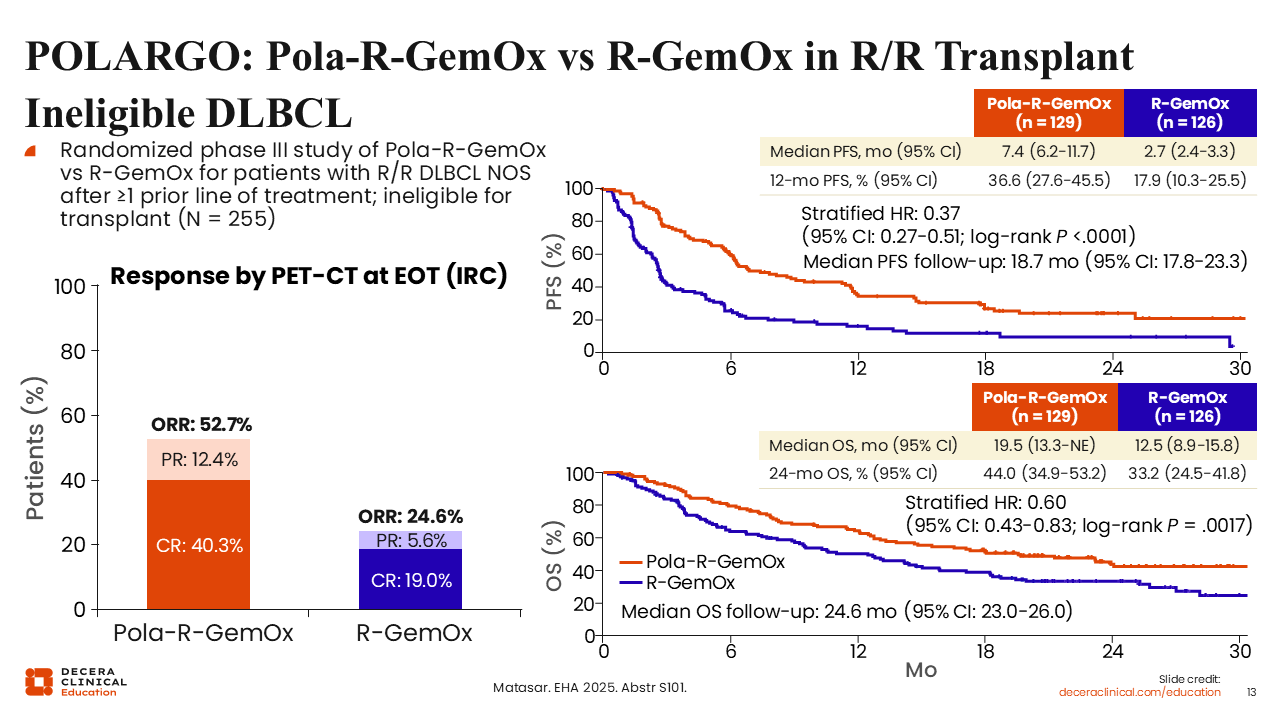
POLARGO: Pola-R-GemOx vs R-GemOx in R/R Transplant-Ineligible DLBCL
There have been advances also for those who do not have access to bispecific antibodies or CAR T-cell therapy. There are data from the POLARGO trial that compared polatuzumab vedotin with R-GemOx vs R-GemOx in patients with R/R DLBCL after ≥1 prior line of treatment who were considered not transplant eligible.20
Overall, there was an improvement in response rate, PFS, and OS. The CR rate for pola-R-GemOx was 40%, which was perhaps not quite as good as what was seen with either CAR T-cell therapy or with glofitamab-GemOx but still better than R-GemOx, where it was 20%. The median PFS was 7 months compared with 2 months, and the OS endpoint showed a median of approximately 20.0 months with pola-R-GemOx compared with 12.5 months for R-GemOx.
This regimen becomes an option, particularly as second-line therapy for patients without access to either bispecific antibodies or CAR T-cell therapy. From a clinical perspective, if one has access to bispecific antibodies and/or CAR T-cell therapy, then I would favor those approaches over a polatuzumab-based regimen. In addition, as polatuzumab vedotin continues to be used as initial therapy with pola-R-CHP, the less appealing a polatuzumab-based regimen will become in the R/R setting.
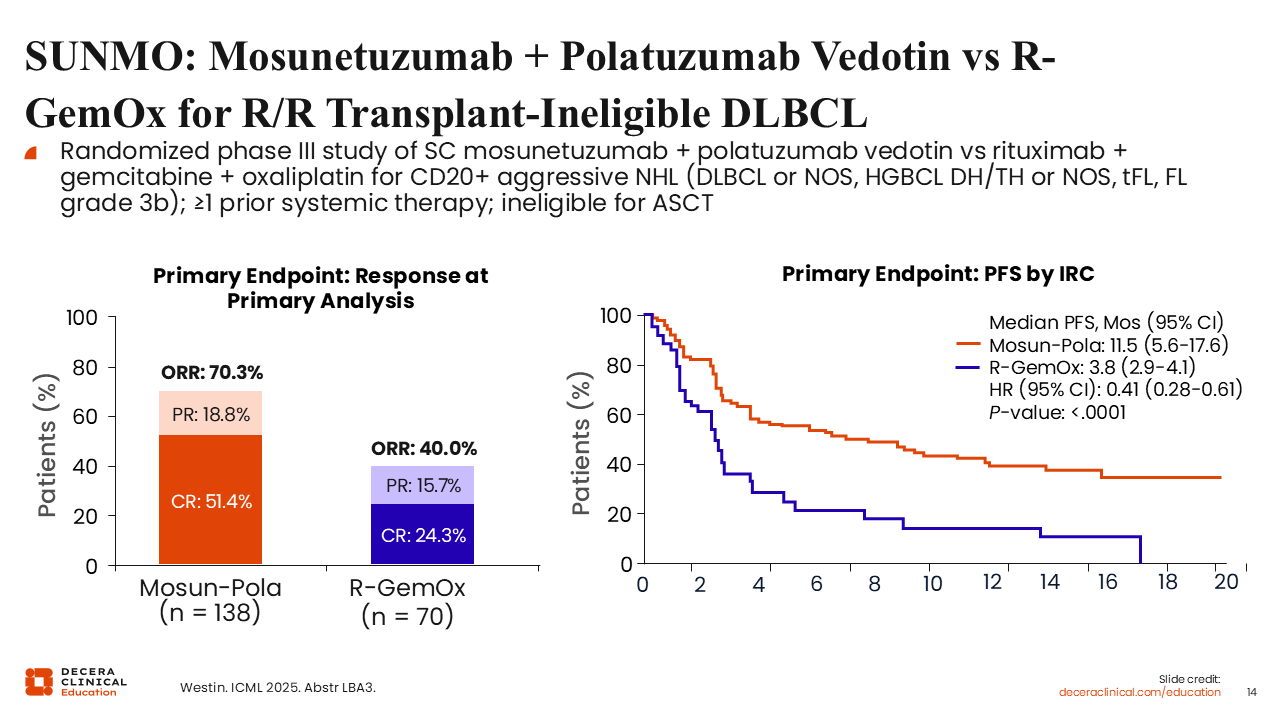
SUNMO: Mosunetuzumab + Polatuzumab Vedotin vs R-GemOx for R/R Transplant-Ineligible DLBCL
Another treatment option includes subcutaneous mosunetuzumab plus polatuzumab, a novel antibody–drug conjugate/bispecific antibody combination without traditional chemotherapy. This regimen was compared with R-GemOx in transplant ineligible R/R DLBCL in the SUNMO trial.21
Most patients were treated in the third-line or later-line setting, given that the median prior line of therapy was 2, but most patients had primary refractory disease and refractory disease to their last prior treatment.
The coprimary endpoints of improved response and PFS were both met. A CR rate for mosunetuzumab plus polatuzumab vedotin of 51% compared with 24% with R-GemOx was observed, and the median PFS was 11.5 months vs 3.8 months, which was a 60% reduction in the risk of progression or death with mosunetuzumab plus polatuzumab vedotin. Mosunetuzumab plus polatuzumab vedotin was well tolerated, with a fairly low incidence of CRS at 26%, that were almost entirely low grade.
From a clinical perspective, mosunetuzumab plus polatuzumab vedotin becomes an option, particularly for patients who are not good candidates for any GemOx or chemotherapy backbone and are polatuzumab vedotin naive. The glofitamab-GemOx or CAR T-cell–based approaches may be a bit more potent than this combination, but the combination of mosunetuzumab plus polatuzumab vedotin becomes an appealing option, particularly for older, frailer patients who are not particularly good candidates for a chemotherapy-containing approach.
TRANSCEND and ZUMA-7: Curative Potential of CAR T-Cell Therapy in the Third-line or Later-Line Settings
For patients with DLBCL that has progressed past 2 previous lines of therapy, knowledge of third-line and later-line options is crucial.
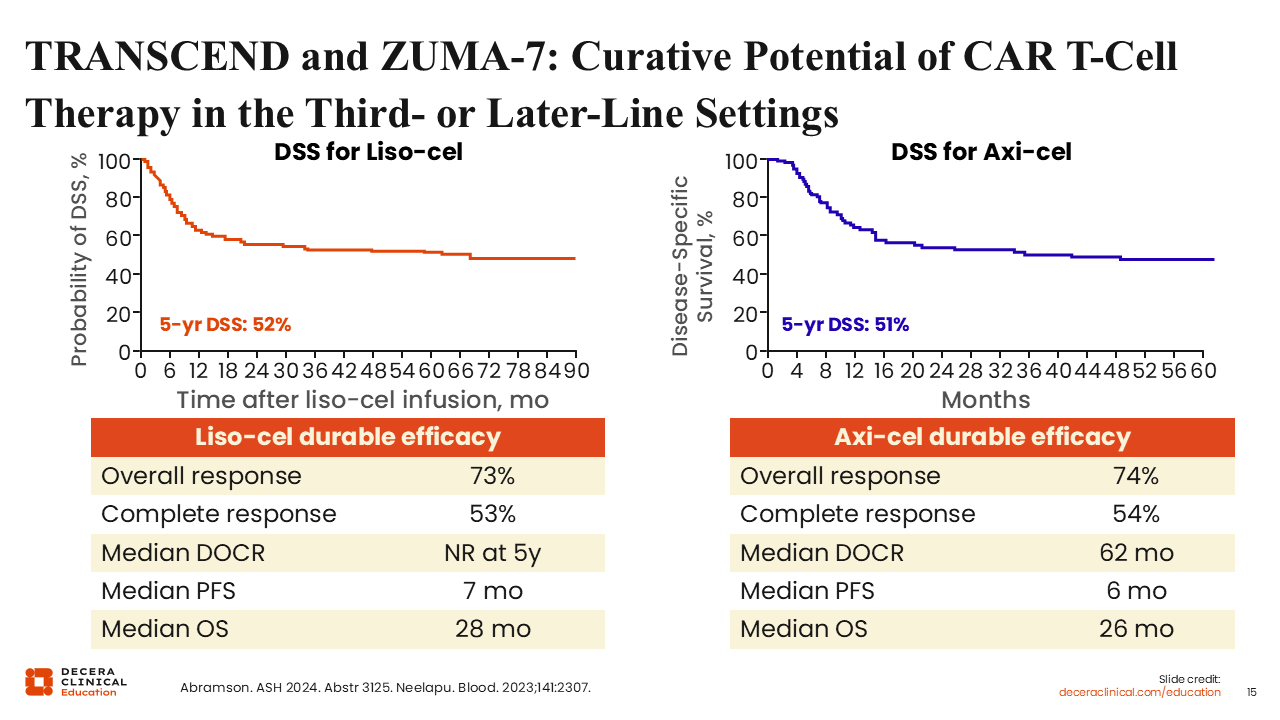
Long-term follow-up data from pivotal trials evaluating liso-cel and axi-cel in patients with a median of 3 prior lines of therapy have provided valuable data on 5-year outcomes in this population. Both CAR T-cell therapies show sustained plateaus in disease-specific survival curves, with a survival rate of approximately 50% at 5 years.18,22
During the pre–CAR T-cell era, median OS for this patient population was approximately 6 months.23 The long-term data from these 5-year follow-ups show the dramatic improvement in the natural history of the disease and underscore that CAR T-cell therapy should be strongly considered in the third-line setting for patients who have not previously received it.
However, as CAR T-cell therapy is increasingly incorporated into second-line treatment, there is a need for effective non–CAR T-cell therapy options for the third-line and later-line settings.
Anti-CD20/CD3 Bispecific Antibodies in the Third-line or Later-Line Settings
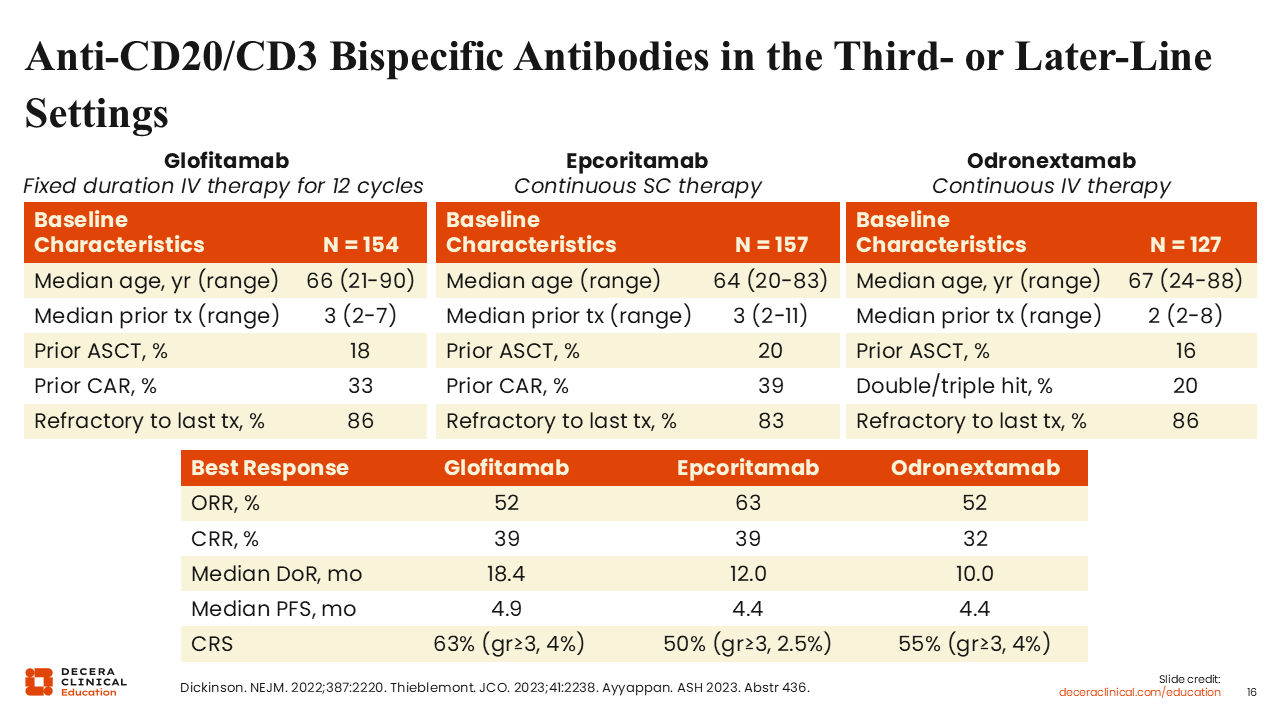
The most common non–CAR T-cell therapy options for the third-line and later-line settings are the bispecific antibodies. Currently, glofitamab and epcoritamab have been approved by the FDA in the United States, each based on pivotal phase II trial data.
- Glofitamab is approved for adult patients with R/R DLBCL NOS) or LBCL arising from follicular lymphoma, after ≥2 lines of systemic therapy.
- Epcoritamab is approved for adult patients with R/R DLBCL NOS, including DLBCL arising from indolent lymphoma, and high-grade B-cell lymphoma, after ≥2 lines of systemic therapy.
- Odronextamab is not currently approved by the FDA but is approved in the European Union for adult patients with R/R DLBCL after ≥2 lines of systemic therapy.
Glofitamab and epcoritamab were evaluated in patients with a median of 3 prior lines of therapy and a median age in the mid-to-late 60s.24,25 Approximately 1 in 5 participants had received an ASCT, and more than one third had previously been treated with CAR T-cell therapy. Most patients were refractory to their most recent line of therapy.
Across both studies, glofitamab and epcoritamab achieved a CR rate of approximately 40%. The median duration of response was approximately 18 months for glofitamab and 12 months for epcoritamab, with a median PFS of between 4 and 5 months. Durable remissions were observed with both products, particularly among complete responders.
Glofitamab is given intravenously as a fixed-duration therapy for 12 cycles over the span of 8.5 months. Epcoritamab is given subcutaneously as continuous therapy, with a frequent and long-term dosing schedule until disease progression or intolerance. Healthcare professionals may prefer a time-limited treatment strategy, such as glofitamab, to allow B-cell recovery and to reduce the risk of long-term infections.
Bispecific antibodies have become the standard go-to option for patients relapsing after CAR T-cell therapy.
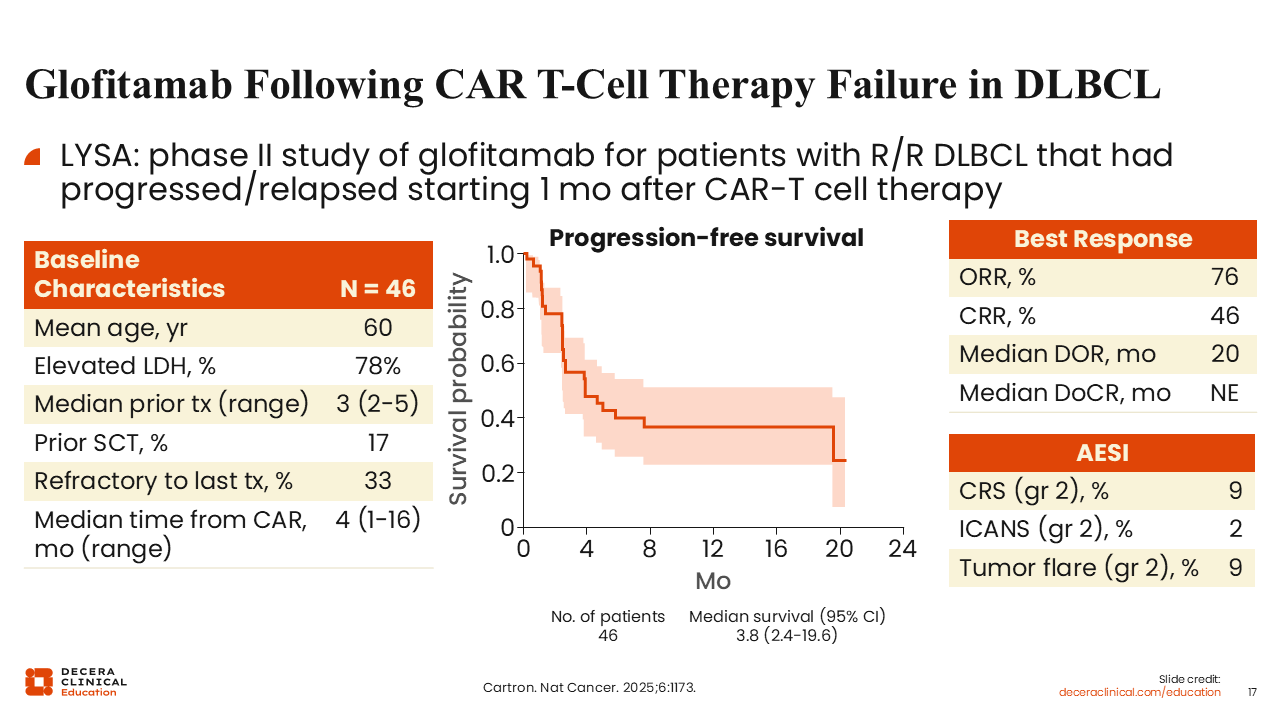
Glofitamab Following CAR T-Cell Therapy Failure in DLBCL
Glofitamab was evaluated in patients who had relapsed after second-line or third-line CAR T-cell therapy. Patients had received a median of 3 prior lines of therapy, with approximately one third being refractory, and 100% having had a prior CAR T-cell therapy with a median interval of 4 months between infusion and enrollment.26
The ORR was 76%, with 46% of patients achieving CR. The median duration of response approached 2 years, and the median duration of CR had not yet been reached at the time of reporting. PFS data suggested a plateau in the survival curve, and early estimates indicate that nearly 40% of patients may experience durable PFS, although follow-up is limited and longer-term data are needed to confirm these outcomes.
The incidence of CRS and immune effector cell–associated neurotoxicity (ICANS) was low. Tumor flare may also be observed with bispecific antibody monotherapy, although this is typically manageable with corticosteroid treatment over time.
Additional Third-Line and Beyond Treatment Options for R/R DLBCL
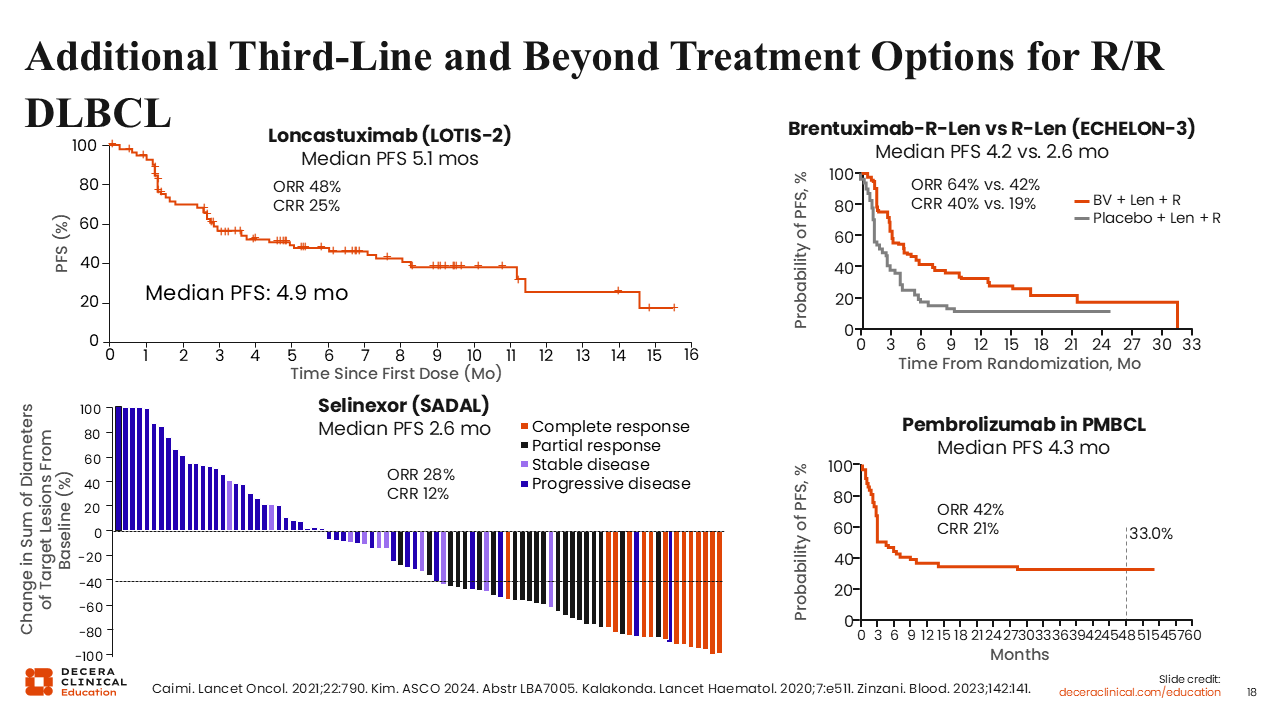
For patients without access to bispecific antibodies, several alternative therapies are available. Loncastuximab tesirine is an antibody–drug conjugate with a pyrrolobenzodiazepine dimer payload, different from that used with polatuzumab or brentuximab vedotin. The LOTIS-2 trial showed that this agent achieves a CR in approximately one fourth of patients in the third-line and later settings.27
More recently, the ECHELON-3 trial evaluated brentuximab vedotin plus lenalidomide/rituximab independent of CD30 expression. Results showed an encouraging 40% CR rate, outperforming lenalidomide/rituximab alone and improving both PFS and OS. However, the median PFS was disappointing at 4.2 months.28
Selinexor is also approved in this setting, although it is not a particularly active or well-tolerated drug. The SADAL pivotal trial reported a modest CR rate of 12% and a median PFS of <3 months.29
For patients with primary mediastinal B-cell lymphoma, 9p24 amplifications drive PD-L1/PD-L2 upregulation and thus may be uniquely sensitive to PD-1 inhibitors. In a phase II trial, pembrolizumab showed an overall and CR rate of 42% and 21%, respectively, in this population. The survival curve also showed a durable plateau, with one third of these patients remaining progression free at 4 years.30
Collectively, these data highlight the progress made in the setting of R/R DLBCL but also underscore a significant unmet need, with most patients unable to achieve durable, sustained outcomes despite current advances.
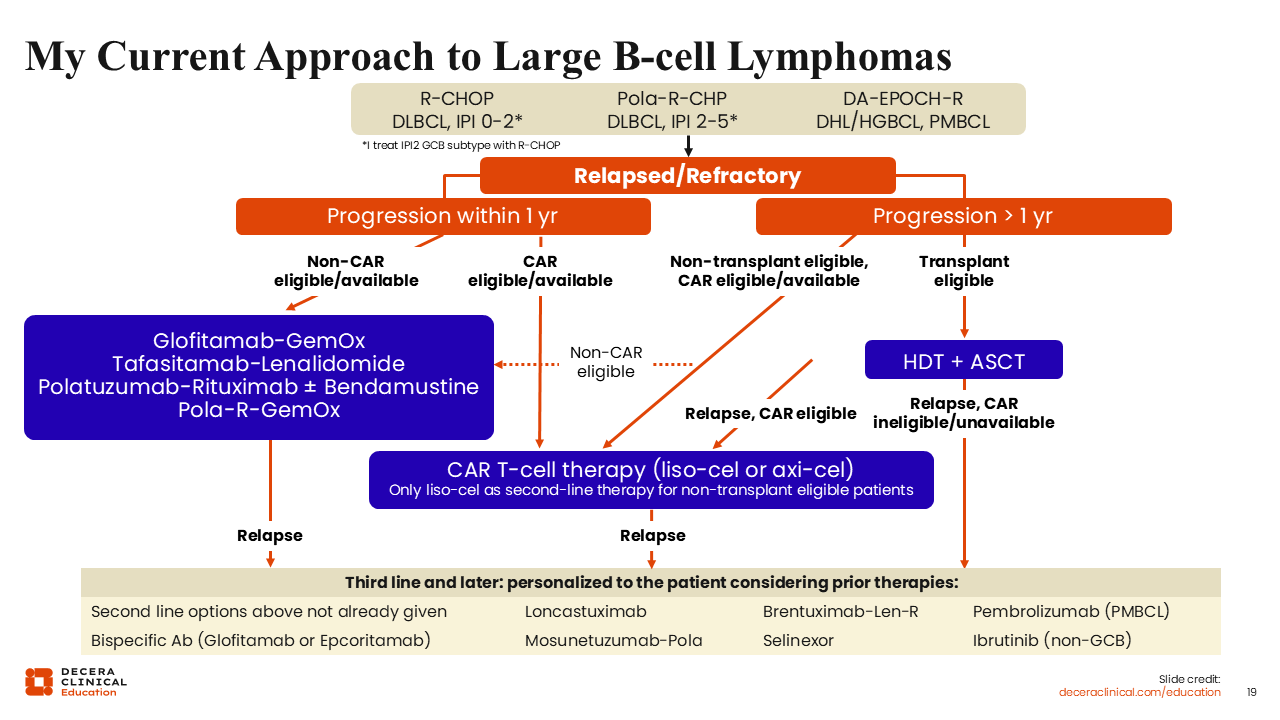
My Current Approach to Large B-Cell Lymphomas
Overall, in the frontline setting, pola-R-CHP is selected for most patients with an IPI score of ≥2, particularly IPI ≥3. DA-EPOCH-R is used for specific LBCL subtypes such as double-hit lymphoma and primary mediastinal lymphoma, whereas R-CHOP remains an option for low-risk patients. For patients with R/R disease that is primary refractory or an early relapse within 1 year, liso-cel or axi-cel CAR T-cell therapy is recommended, but if that therapy is not accessible, glofitamab-GemOx is an alternative. For patients with disease relapse beyond 1 year, transplant eligibility should be assessed. Late relapsing young, fit patients should proceed with high-dose chemotherapy and ASCT if they have chemotherapy-sensitive disease. Those who relapse late but who are not transplant eligible should be considered for CAR T-cell therapy, ideally with liso-cel or axi-cel, if they have access.
Advances on the Horizon
As we move forward, there are multiple opportunities to improve outcomes for patients with DLBCL. One strategy may be to introduce CAR T-cell therapy and bispecific antibodies earlier in the treatment paradigm. These approaches have already been explored in the second-line setting but may have utility in the frontline setting as well.
Another approach involves combining bispecific antibodies with other drugs. Antibody–drug conjugates or costimulatory molecules may help enhance cell-mediated cytotoxicity in combination with bispecific antibodies.
Development of novel bispecific antibodies and new drug classes such as CELMoDs or immunomodulatory drugs may help to improve efficacy in the future. Ongoing innovation in CAR T-cell therapies include rapid manufacturing, dual-targeting therapies, and even allogeneic off-the-shelf CAR T-cells that may help expand access and improve outcomes.
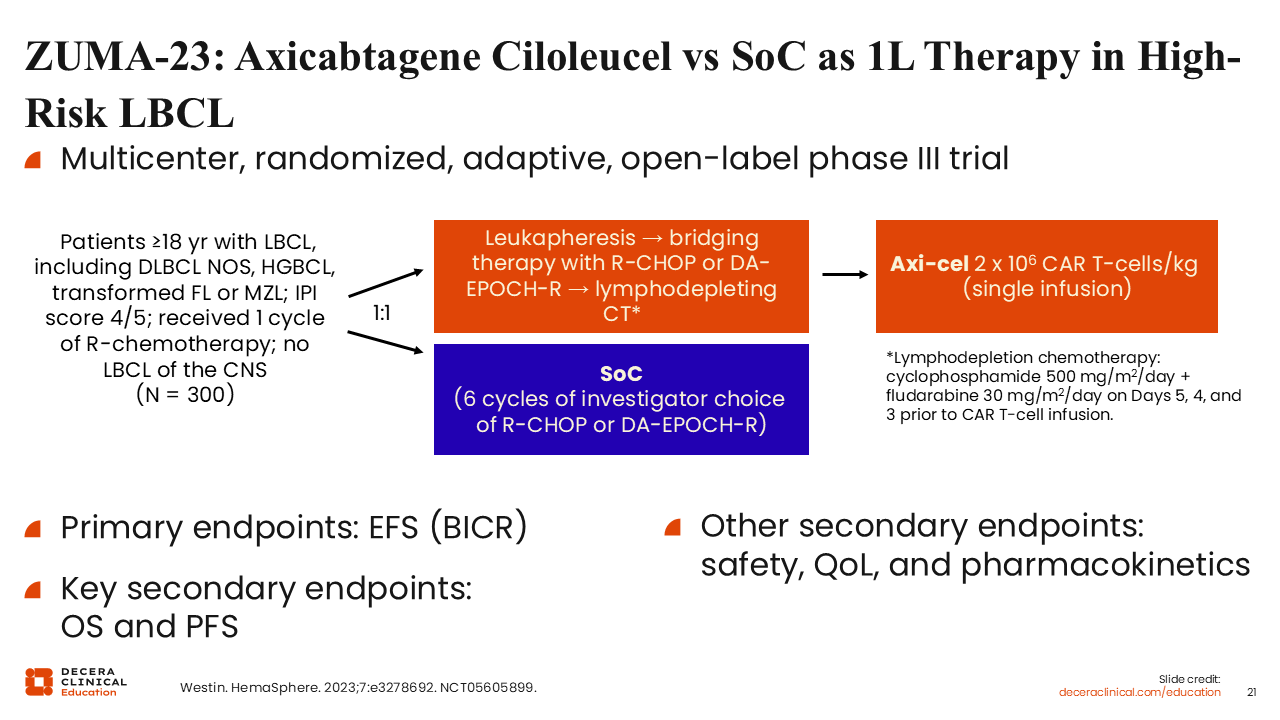
ZUMA-23: Axicabtagene Ciloleucel vs SoC as First-line Therapy in High-Risk LBCL
Moving CAR T-cell therapy earlier in patients with very high–risk disease may optimize the likelihood of a long-term remission or possibly even cure. This strategy is currently being evaluated in the ZUMA-23 trial, which is enrolling patients with an IPI score of 4-5, representing a very high–risk population. These patients are randomized to receive either standard-of-care therapy (R-CHOP or DA-EPOCH-R) or axi-cel CAR T-cell therapy. In the CAR T-cell therapy arm, patients undergo leukapheresis and then a bridging cycle of either R-CHOP or DA-EPOCH-R, followed by axi-cel infusion. Through this, the study aims to determine whether an upfront CAR T-cell–containing approach offers better outcomes compared with the traditional chemoimmunotherapy outcomes in the highest IPI–risk patients.31
Let’s return to a question from earlier in the activity.
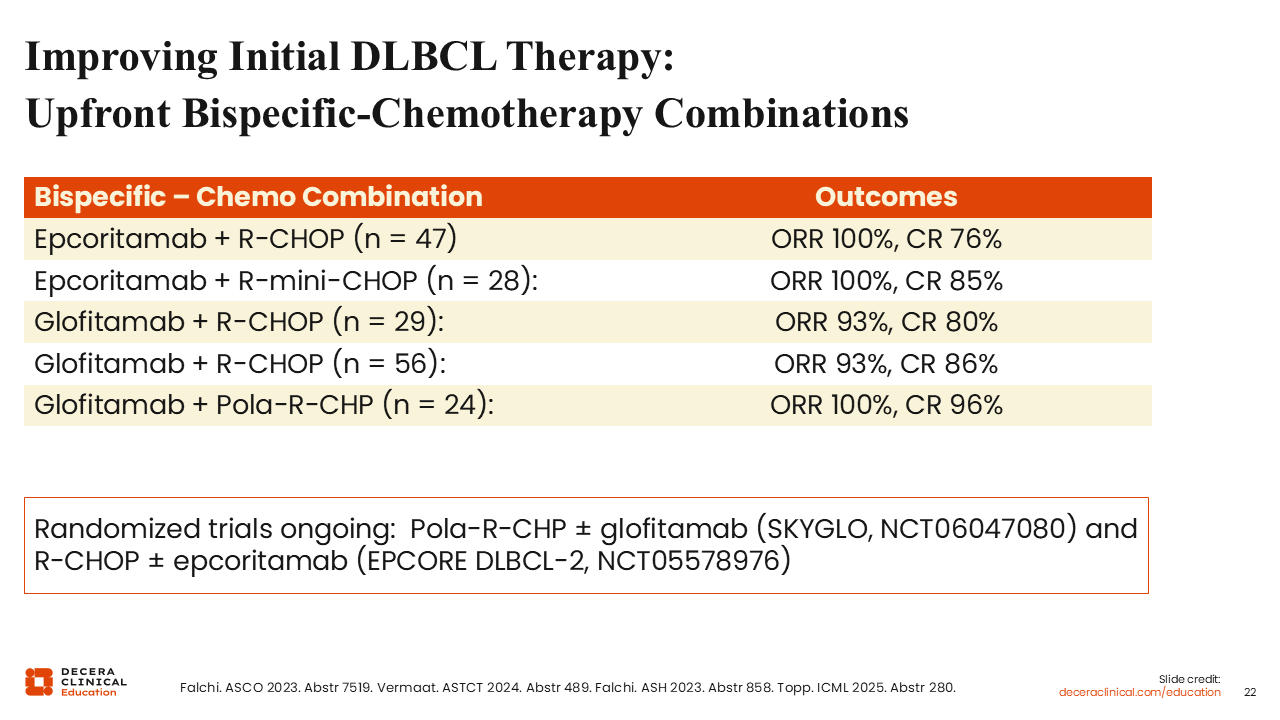
Improving Initial DLBCL Therapy: Upfront Bispecific-Chemotherapy Combinations
In addition to CAR-T cell therapies, bispecific antibodies are also being assessed as first-line therapy. Multiple smaller combination studies have shown that combining either epcoritamab or glofitamab with standard frontline chemoimmunotherapy produces exceptionally high overall and CR rates. This has led to 2 randomized controlled trials evaluating these bispecific antibodies for initial therapy in DLBCL: the ongoing SKYGLO trial, which is evaluating pola-R-CHP with and without glofitamab, and the EPCORE DLBCL study assessing R-CHOP with and without epcoritamab.32-35
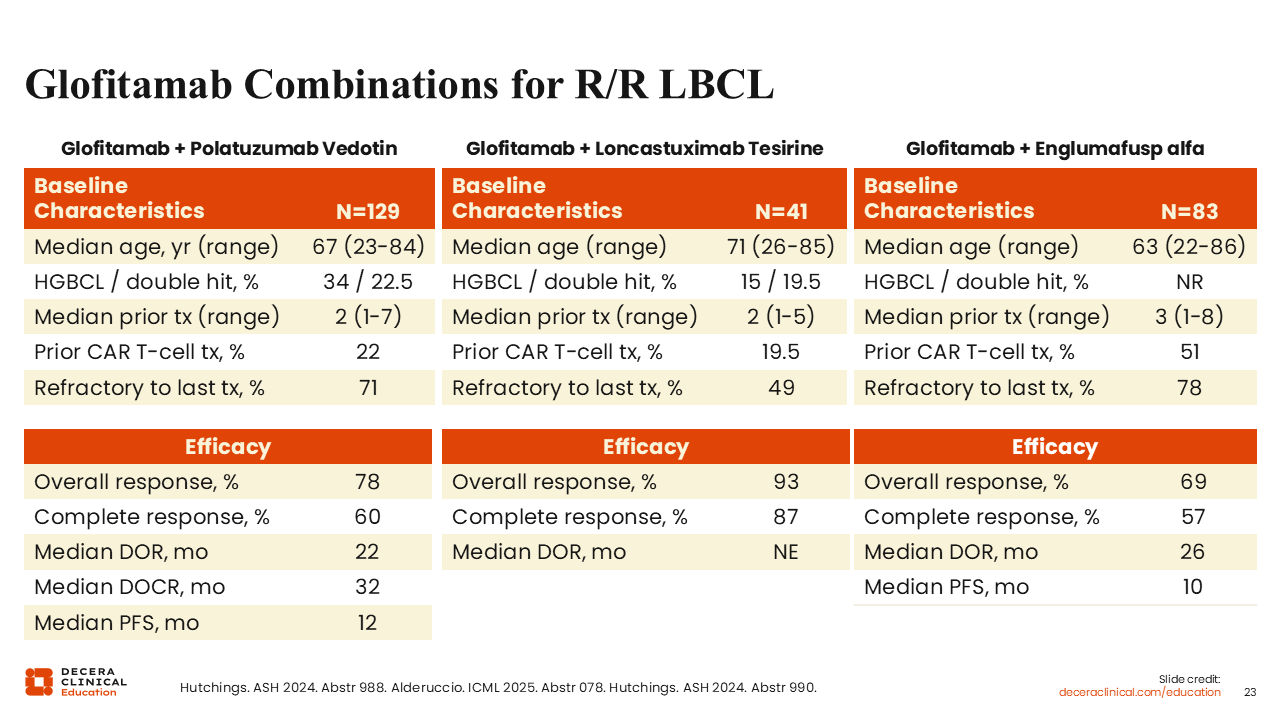
Glofitamab Combinations for R/R LBCL
Emerging data have shown that combinations of glofitamab with other novel agents can lead to improvements in CR and PFS in patients with R/R DLBCL.
Glofitamab plus polatuzumab vedotin was associated with a CR rate of 60%, an improvement >40% rate seen with glofitamab monotherapy.36
A smaller study evaluated glofitamab in combination with the anti-CD19–targeted antibody–drug conjugate loncastuximab tesirine and reported a 87% CR rate.37 In addition, the combination of glofitamab and the costimulatory bispecific antibody englumafusp alfa showed a CR rate approaching 60% in heavily pretreated patients with a median of 3 prior lines of therapy.38
These findings underscore the potential for bispecific antibody combinations to become a next novel approach in patients with R/R DLBCL.
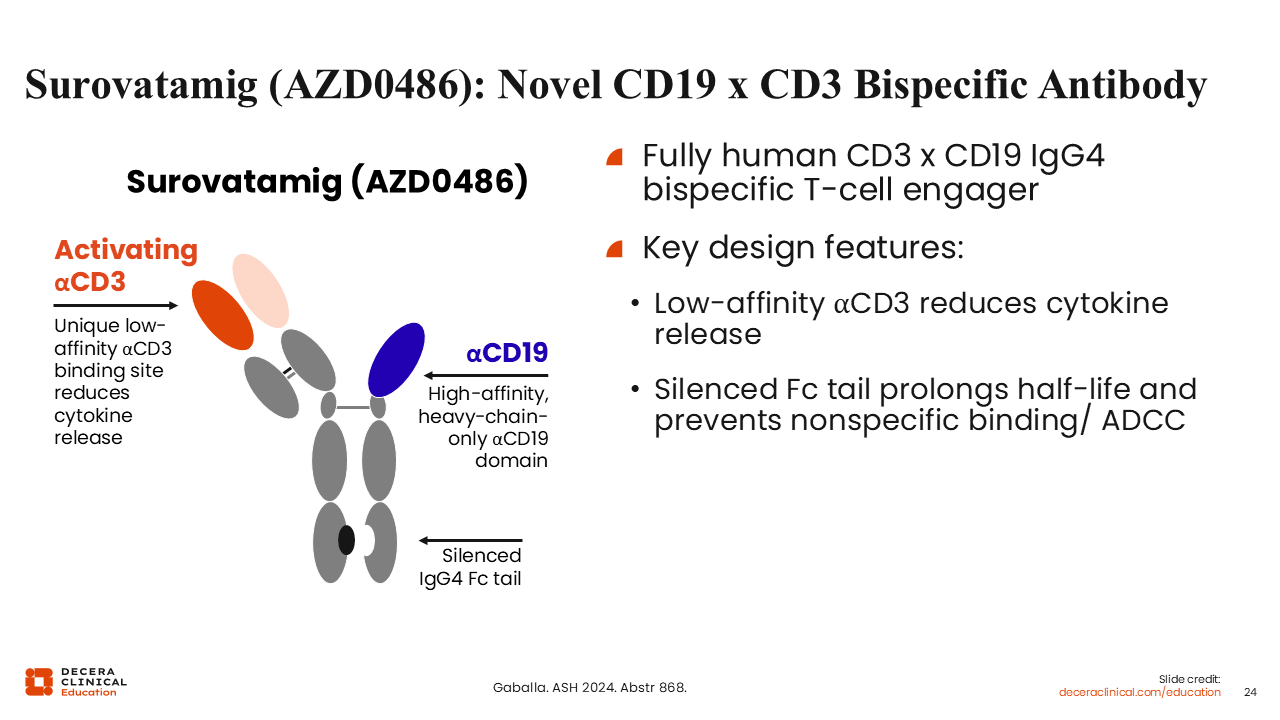
Surovatamig (AZD0486): Novel CD19 x CD3 Bispecific Antibody
Novel bispecific antibodies are also in clinical development. All of the approved bispecific antibodies discussed above target CD20. Surovatamig (AZD0486) is a novel bispecific antibody targeting CD19 along with CD3.39
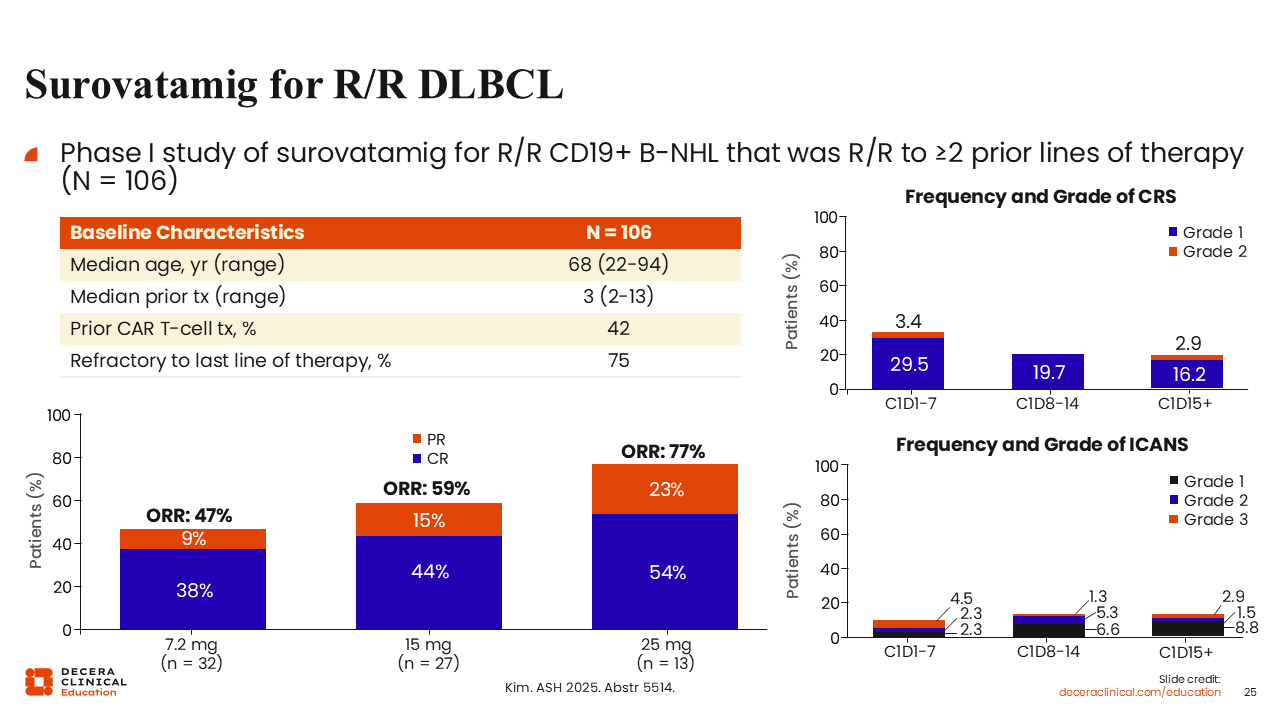
Surovatamig for R/R DLBCL
This CD19/CD3 bispecific has been tested in 106 patients with R/R DLBCL with a median of 3 prior lines of therapy. Approximately 42% of these patients had prior CAR T-cell therapy, and 75% were refractory to their last line of treatment.40
ORR reached 77% with a CR rate of 54% at the highest dose. The frequency of CRS and ICANS was low, and these were generally low-grade events.
For patients heavily pretreated with CD20-directed therapies, the data for surovatamig are very promising. Of importance, this would be our only CD19 bispecific antibody, which could be very appealing for patients who lose CD20 expression after multiple CD20-directed therapies. This agent is now being assessed in the phase III SOUNDTRACK-D2 study, in which older or unfit patients with newly diagnosed LBCL will receive either R-mini-CHOP for 2 cycles followed by surovatamig or R-mini-CHOP for 6 cycles (NCT07215585).
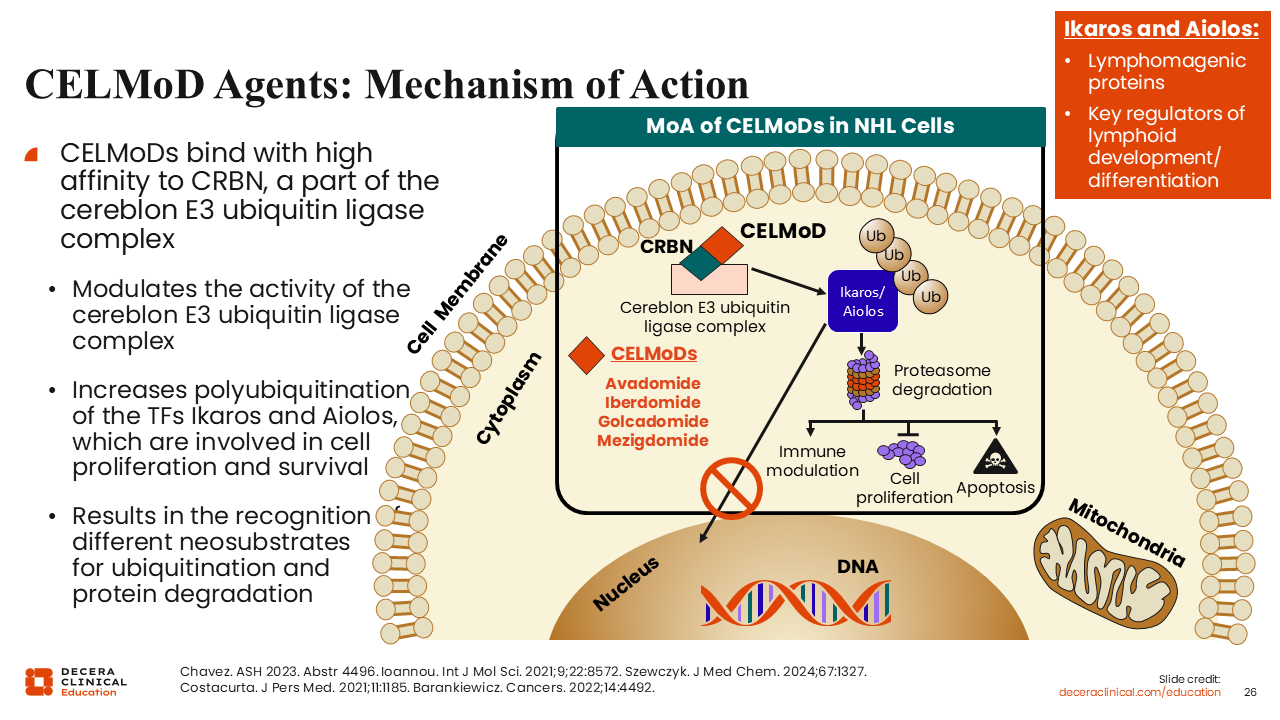
CELMoD Agents: Mechanism of Action
CELMoDs represent an exciting innovation in immunomodulatory therapy. CELMoDs are cereblon-dependent agents that promote degradation of the lymphogenic regulators Ikaros and Aiolos. Along with this, these agents have a predominate oral administration method, which adds to their clinical appeal.41
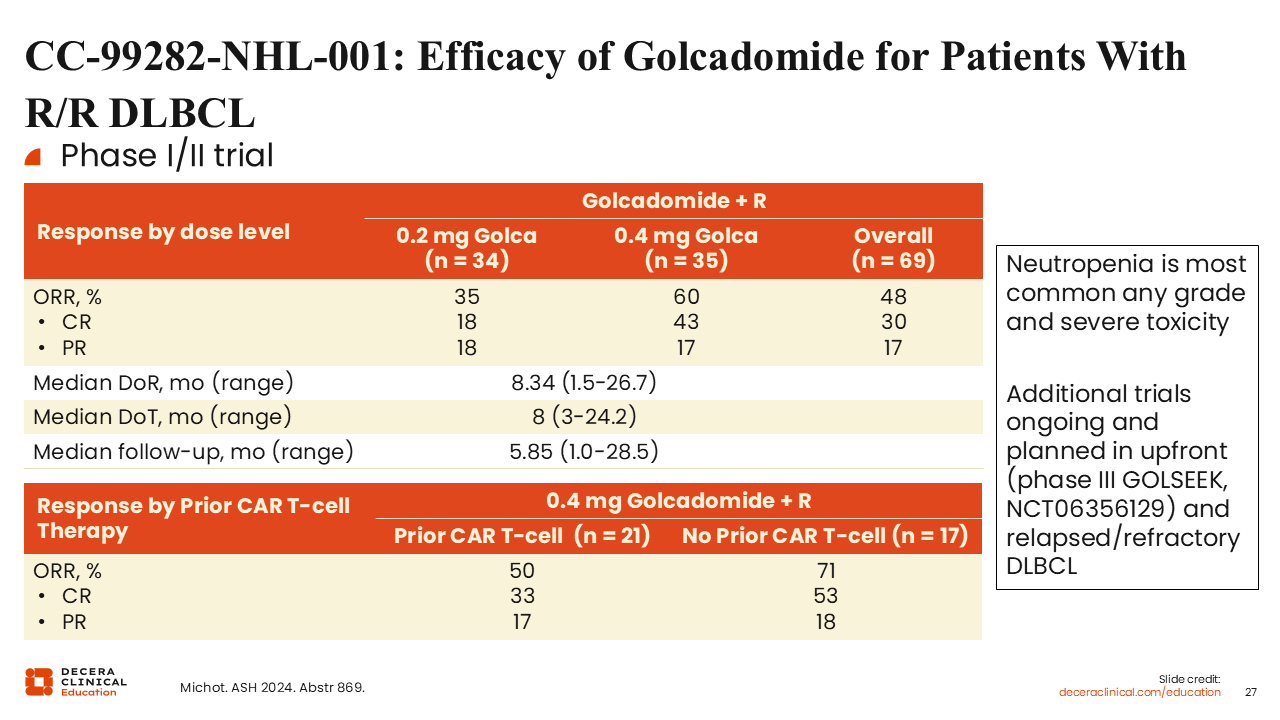
CC-99282-NHL-001: Efficacy of Golcadomide for Patients With R/R DLBCL
Golcadomide is a CELMoD that has been studied in combination with rituximab for the treatment of both aggressive and indolent B-cell tumors. Phase I/II data in patients with R/R DLBCL have shown an ORR of 48% with a CR rate of 30% and median duration of response of 8 months across all dose levels.42
At the recommended phase II dose (RP2D) of 0.4-mg golcadomide plus rituximab, patients without prior CAR T-cell therapy demonstrated an ORR of 71% with a CR rate of 53%. Patients with prior CAR T-cell treatment had slightly lower response rates with an ORR of 50% and a CR rate of 35%. Currently, golcadomide is being studied with other combination strategies, including in the phase III GOLSEEK trial of R-CHOP with or without golcadomide for previously untreated DLBCL.
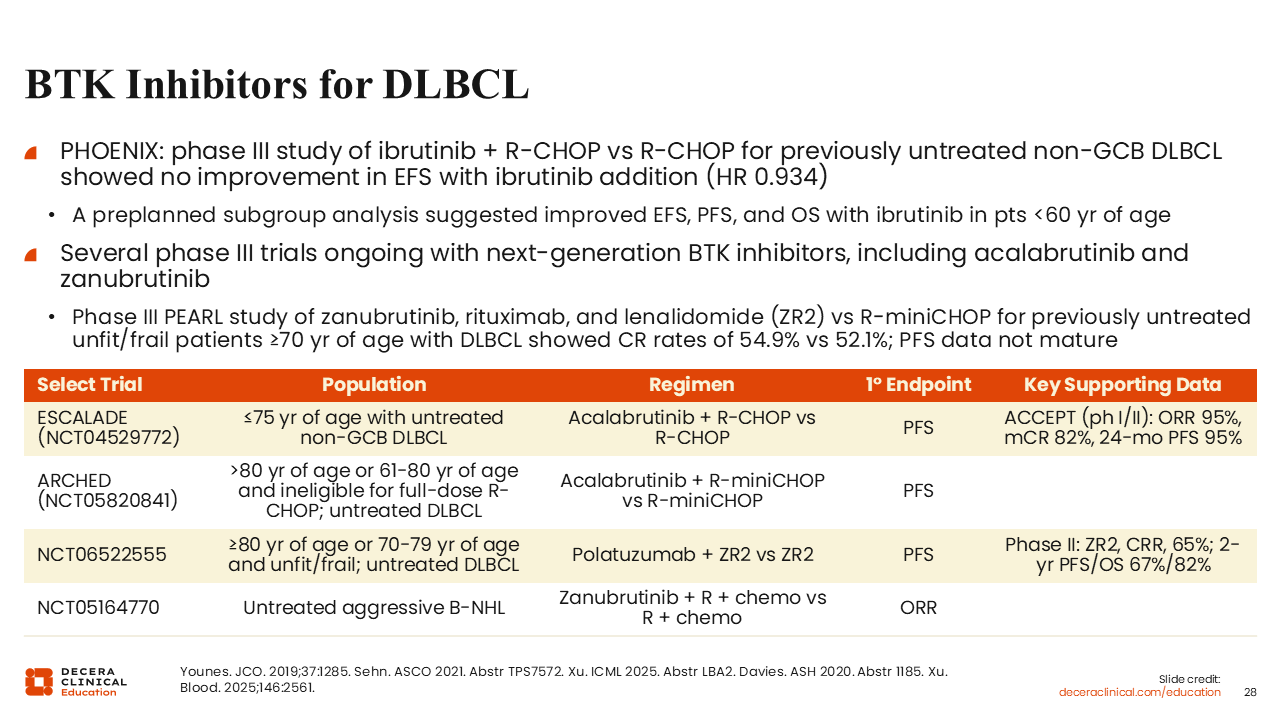
BTK Inhibitors for DLBCL
BTK inhibitors are also being studied for patients with DLBCL. Of note, in a 2019 report from the phase III PHOENIX trial, the addition of ibrutinib to R-CHOP did not improve event-free survival compared with R-CHOP alone (HR: 0.934) for patients with previously untreated non-GCB DLBCL.43 However, a preplanned subgroup analysis suggested improved event-free survival, PFS, and OS among patients younger than 60 years of age, suggesting a population that might benefit from BTK inhibitor therapy. Clinical development has shifted toward next-generation BTK inhibitors with improved selectivity and tolerability, including acalabrutinib and zanubrutinib. Multiple phase III studies are ongoing in the frontline DLBCL setting, including older or less fit patients.
Acalabrutinib demonstrated activity with R-CHOP in a phase I/II trial, showing high response rates and durable disease control.44 The ongoing phase III ESCALADE trial is evaluating frontline acalabrutinib plus R-CHOP vs R-CHOP in patients younger than 75 years of age with non-GCB DLBCL, and the ARCHED study is assessing acalabrutinib plus R-mini-CHOP vs R-mini-CHOP in patients aged 80 years or older, or younger patients ineligible for full-dose R-CHOP.
Zanubrutinib is also being evaluated in DLBCL. Recently, the phase III PEARL study, which enrolled previously untreated unfit or frail patients aged 70 years or older, showed that zanubrutinib plus rituximab/lenalidomide was associated with CR rates comparable to R-mini-CHOP (54.9% vs 52.1%), although PFS data remain immature.45 Ongoing phase III trials are assessing frontline polatuzumab vedotin plus zanubrutinib plus rituximab/lenalidomide for older/unfit patients with DLBCL and frontline zanubrutinib plus R-CHOP or other chemoimmunotherapy for aggressive B-cell non-Hodgkin lymphoma.
Let’s return to a question from earlier in the activity.
Optimizing CAR T-Cell Therapy
Optimizing CAR T-cell therapy can involve several strategies. Enhancement of the stem memory phenotype allows for those cells to be less prone to exhaustion and could improve efficacy. Acceleration of the manufacturing process would also be important as many patients have rapidly progressive disease in need of timely treatment. Prevention of antigen escape, such as the loss of CD19 expression from repeat exposure to CD19-directed therapy, could lower the risk of relapse. Finally, strategies to reduce the incidence of toxicities, particularly CRS and neurologic adverse effects, would greatly improve a patient's quality of life.
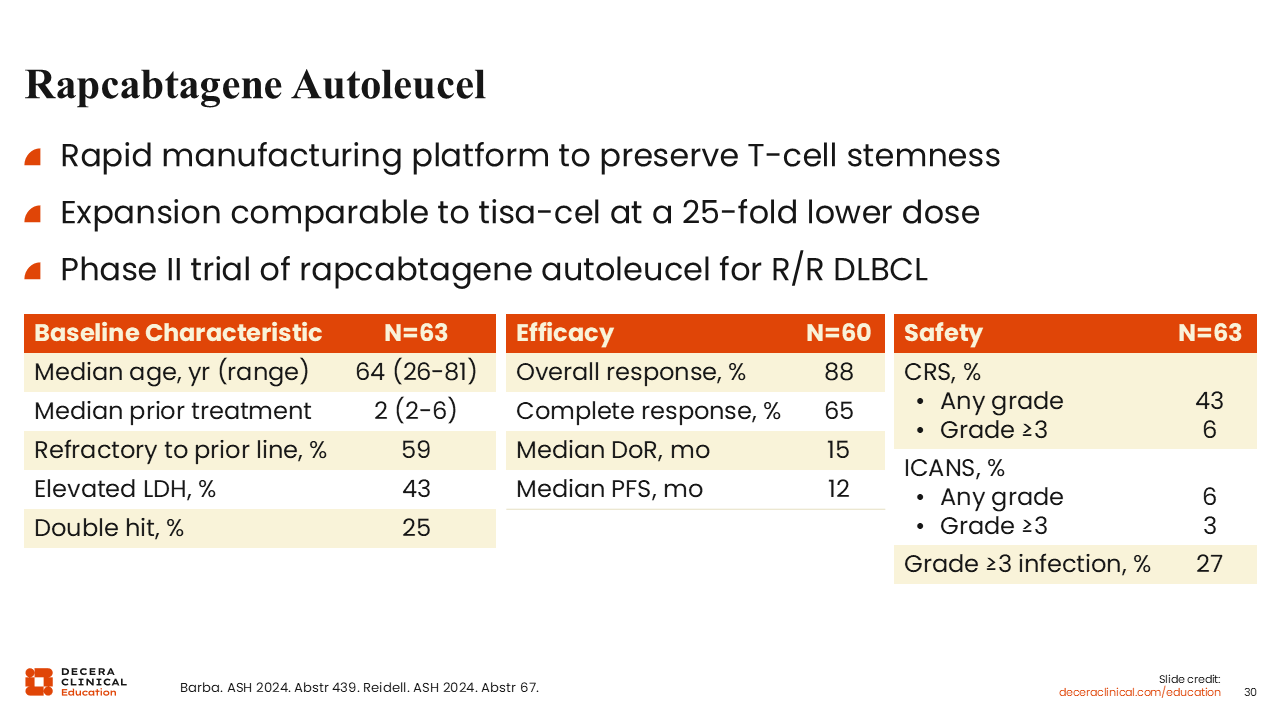
Rapcabtagene Autoleucel
Rapcabtagene autoleucel is a CD19- directed autologous CAR T-cell product and is the more potent, next-generation form of tisa-cell. It is manufactured using a platform that allows rapid manufacturing of the CAR T-cell product in fewer than 10 days. In an initial study of 63 patients with DLBCL with a median of 2 prior lines of therapy, rapcabtagene autoleucel produced a CR rate of 65% and a median PFS of 1 year. The study reported low rates of any-grade CRS at 43%, with relatively low rates of ICANS at 6%.46
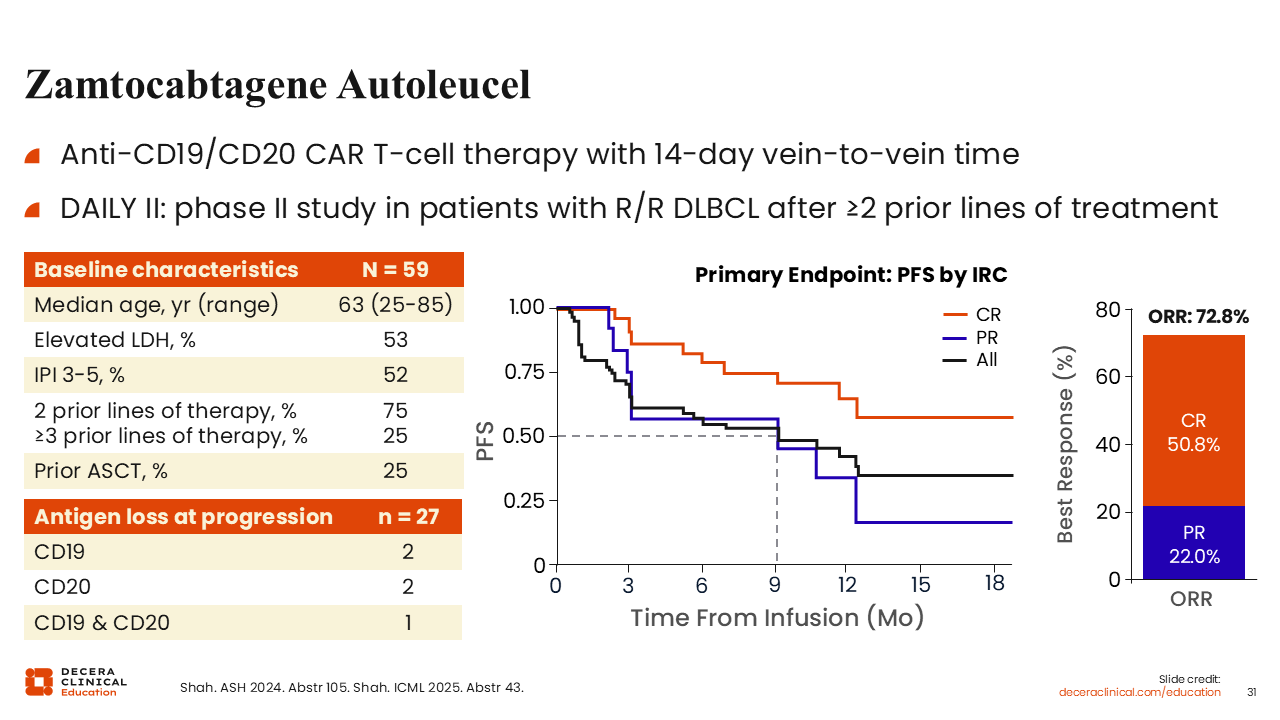
Zamtocabtagene Autoleucel
Another rapidly manufactured CD19/CD20-directed CAR T-cell agent is zamtocabtagene autoleucel. This therapy targets 2 antigens and is manufactured with a 14-day vein-to-vein time.
In the phase II DAILY II study, 59 patients were enrolled with a median age of 63 years, with many of these patients having received 2 prior lines of therapy. The study reported a CR of 51% and a median duration of response of 11 months. Relapse biopsy data were fairly limited but indicated that only a single biopsied patient lost both CD19 and CD20 expression, suggesting that this agent may be a way to overcome antigen loss as a mechanism of CAR T-cell resistance. Although follow-up data are also limited, the median PFS of 9 months is encouraging. Low-grade CRS occurred in 46% of patients and ICANS in 71%, with virtually no severe CRS or ICANS reported.47
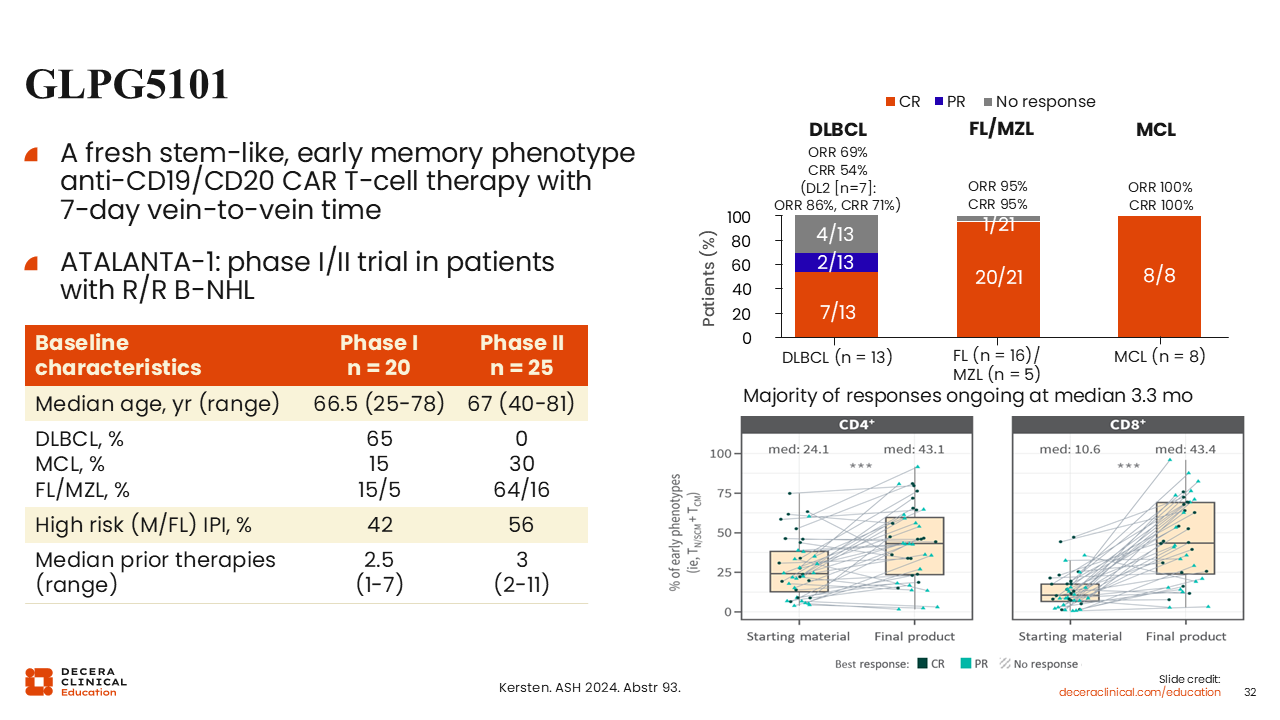
GLPG5101
The CD19-targeting agent, GLPG5101, is an even more rapidly manufactured CAR T-cell product, with a vein-to-vein time of only 7 days. Preliminary data from a phase I/II study evaluated its use in a mostly high-risk population of 45 patients with R/R B-cell non-Hodgkin lymphoma with a median age of 67 years and a median of 3 prior lines of therapy.
Focusing on patients with DLBCL, the study reported a CR rate of 54%, an ORR of 60%, and modest CRS rate of 40%. CRS rates were entirely low-grade reactions, with no grade ≥3 or higher events, and ICANS occurred in 16% of patients with only 4% high-grade ICANS. These results demonstrate that rapid manufacturing can lead to encouraging efficacy outcomes with manageable toxicity. Along with this, the study showed that this manufacturing process enhances the T-cell stem memory phenotype, producing more active, less exhaustible, and an overall healthier T-cell than what was extracted from the patient.48
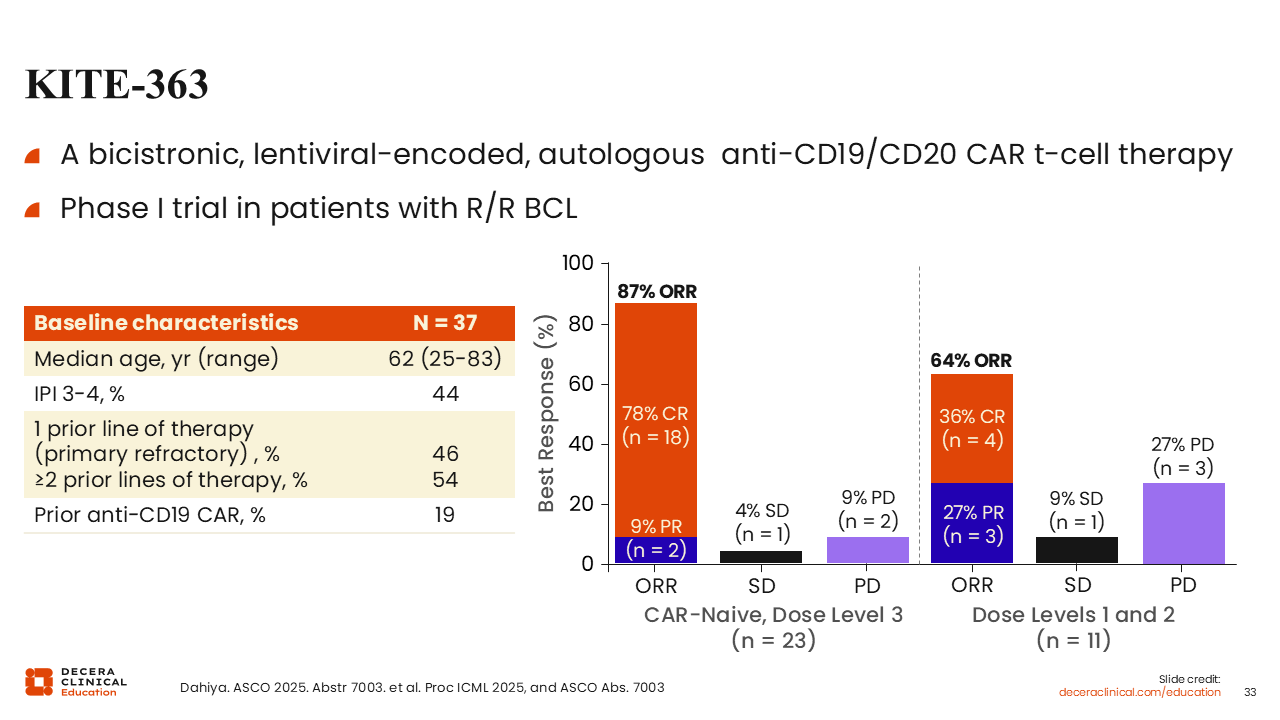
KITE-363
Another innovation in the CAR T-cell landscape is the dual-targeted, bicistronic CD19/CD20 CAR T-cell, KITE-363. In a recent small study of 37 patients with R/R BCL, KITE-363 achieved a CR rate of 78% in CAR T-cell therapy–naive patients at a higher dose level, with lower doses reporting a rate of 36%. Compared with agents in the previous studies that I have discussed, higher toxicities were reported with this agent, with rates of 92% and 46% CRS and ICANS, respectively, at dose level 3.49 KITE-363 is under active development and exploration relative to the single-target CAR T-cell therapies such as axi-cel.
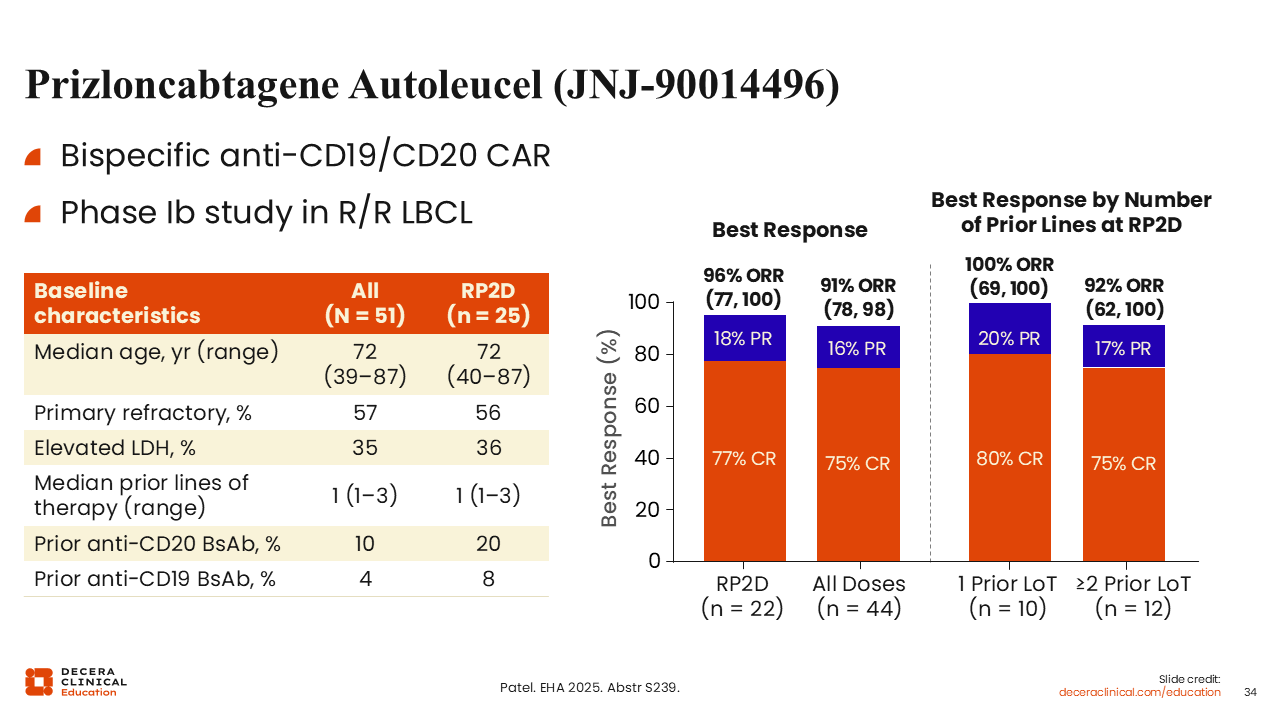
Prizloncabtagene Autoleucel (JNJ-90014496)
Prizloncabtagene autoleucel (JNJ-90014496) is a bispecific anti-CD19/CD20 CAR T-cell therapy that was evaluated in a phase Ib study in R/R LBCL. Among 51 treated patients (25 at the RP2D), the median age was 72 years, with a median number of prior lines of therapy of 1. At the RP2D, the ORR was 96%, including a 77% CR rate, with similarly high activity across all dose levels. Responses were consistent regardless of treatment history, including for patients with multiple prior lines of therapy. CRS was observed in most patients but was predominantly low grade; at RP2D, CRS was mainly grade 1 (68%) or grade 2 (20%), with no grade ≥3 events. ICANS was infrequent, occurring in 8% of patients overall and 4% at RP2D.50
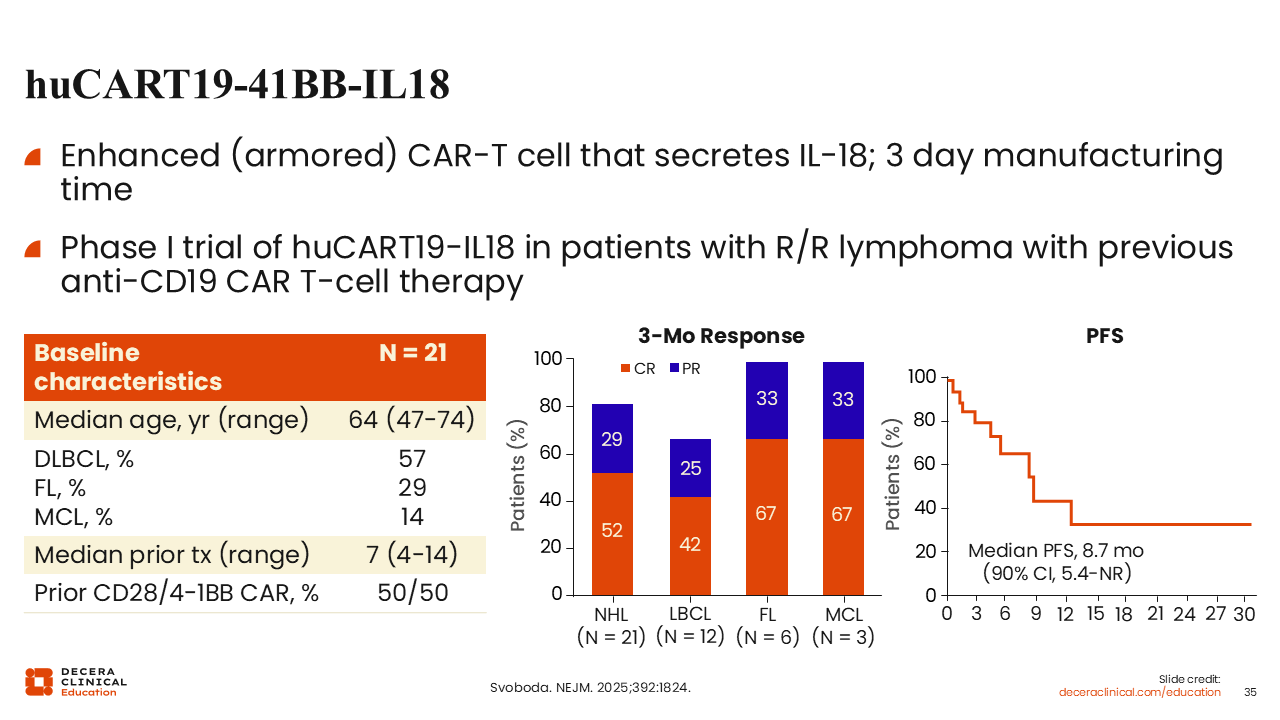
huCART19-41BB-IL18
Another strategy to enhance CAR T-cell outcomes is through the use of cytokine-secreting CARs that can overcome the immune-suppressive tumor microenvironment. The CD19-directed CAR T-cell therapy, huCART19-41BB-IL18, secretes IL-18, which decreases immune suppression and enhanced CAR T-cell activation through autocrine signaling.
In a small study, huCART19-41BB-IL18 was evaluated in a population exclusively consisting of patients that had previously received CD19-directed CAR T-cell therapy, with a median of 7 prior lines of therapy and including more than one half of patients diagnosed with DLBCL. In this heavily pretreated population, this therapy achieved an ORR of 81%, with a CR rate of 52%. Among all 21 patients, the CR rate was 42%.
The median PFS reported in this study was 9 months, which was an encouraging outcome considering the extensive past treatment history of this patient population. These findings underscore the potential of “armored” CARs as a way of overcoming the hostile immunosuppressive microenvironment that limits traditional CAR T-cell therapy.51
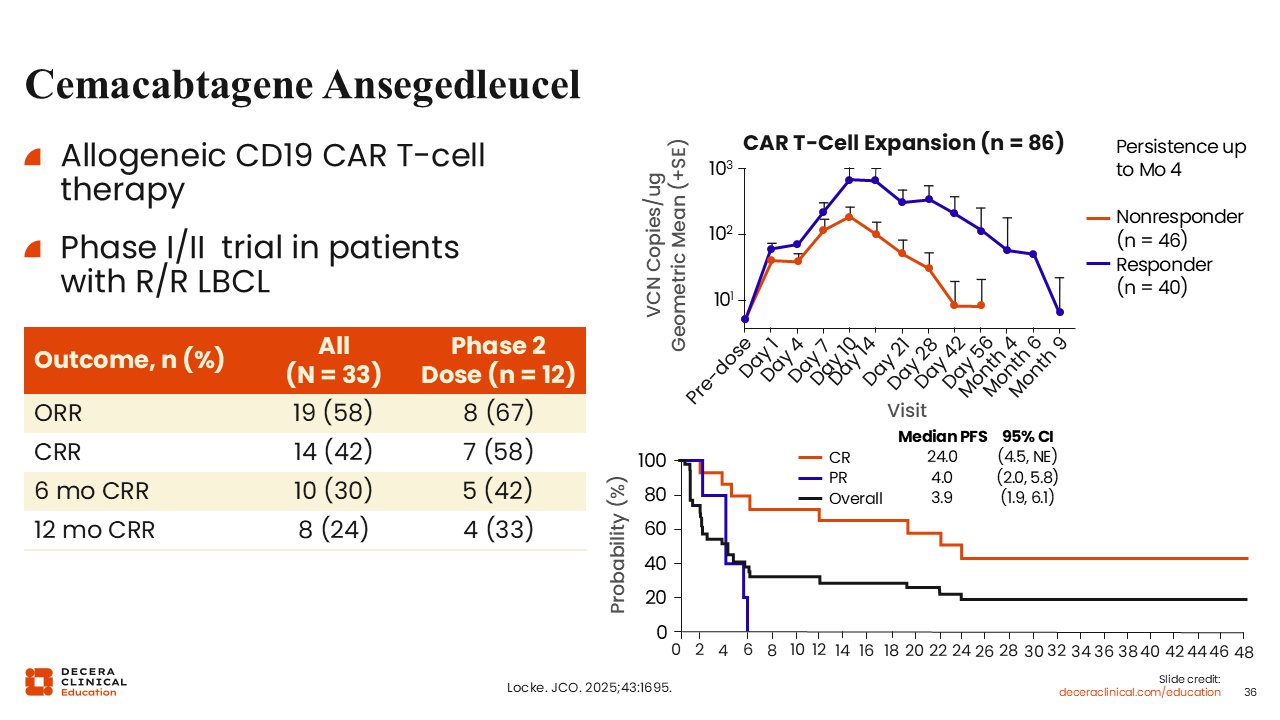
Cemacabtagene Ansegedleucel
Allogeneic CAR T-cells, or off-the-shelf CAR T-cells, are derived from healthy donors and may provide the benefit of availability, eliminating the need to manufacture of CAR T-cells from chemotherapy-exposed patients with lymphoma.
Data from the ALPHA2 study evaluated cema-cel, an allogeneic CD19-directed CAR T-cell therapy with a T-cell receptor knockout to reduce immunogenicity and a CD52 knockout to allow the use of an alemtuzumab-containing lymphodepleting agent.
Among all treated patients, the ORR was 58% with a CR rate of 42%, which rose to 58% at the RP2D. Compared with autologous products, the duration of response with cema-cel was shorter, which may be because of higher immunogenicity in allogeneic products leading to faster clearance. These findings suggest that although allogeneic products may not be as effective in patients with high disease burden, they may be effective options for eradication in those with measurable residual disease (MRD).52
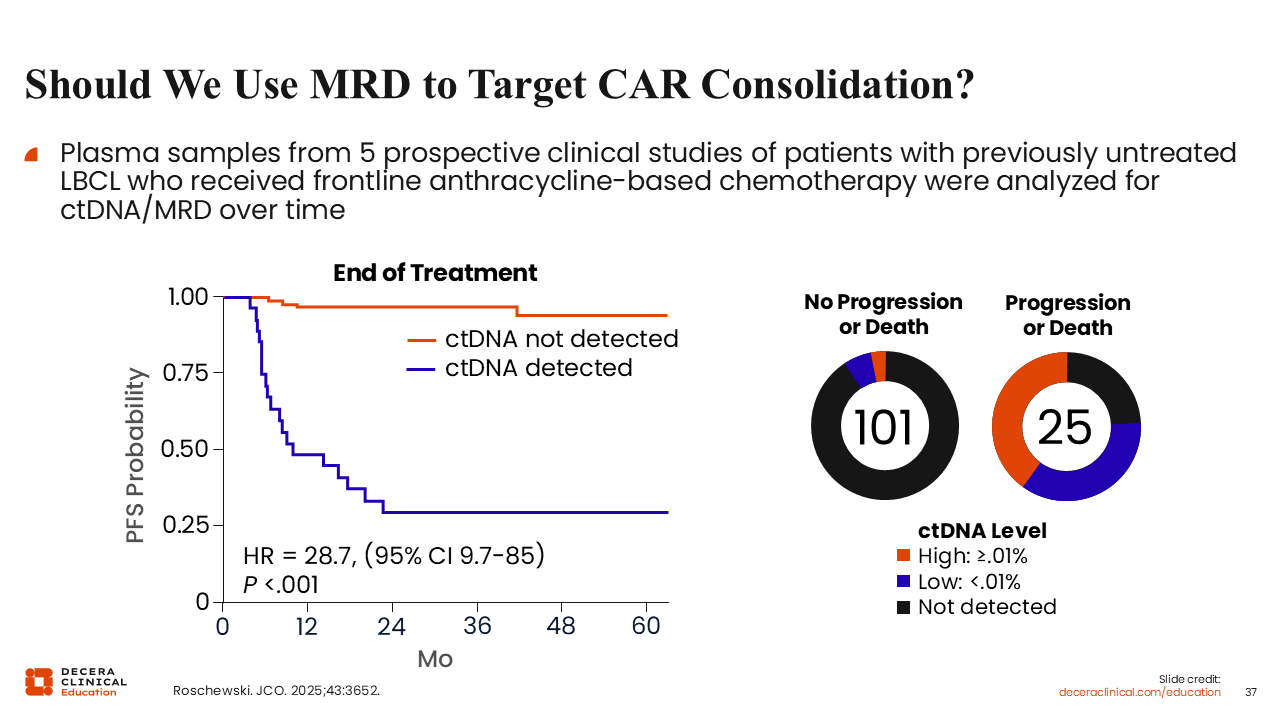
Should We Use MRD to Target CAR Consolidation?
Recent data have highlighted the role of MRD in DLBCL. In an assessment of 5 prospective studies of first-line chemoimmunotherapy for LBCL, circulating tumor DNA/MRD was examined in 137 patients with 409 plasma specimens gathered during the course of treatment. At the end of therapy, patients with undetectable MRD had an almost 100% PFS rate. By contrast, PFS was observed in only 25% of patients with detectable MRD. These findings underscore that detectable circulating tumor cells at the end of treatment can be a strong predictor of future relapse or treatment failure.53
ALPHA-3: Cema-cel as an “MRD Eraser”
New treatment options are needed for the patients who do not achieve undetectable MRD at the end of frontline therapy. The ALPHA-3 trial is exploring a new strategy by randomizing these patients to CAR T-cell consolidation therapy using the off-the-shelf CAR T-cell therapy cema-cel. As these patients have low disease burden, cema-cel can be especially suitable for this patient population. In this study, all patients will undergo MRD testing with the PhasED-Seq assay after induction therapy, and those who were MRD undetectable will be randomized 1:1 to either observation or cema-cell treatment. Results from this trial are anticipated in the next few years and could provide valuable data for preventing relapse in patients with DLBCL.54
Conclusions
Novel therapies have transformed the treatment landscape for LBCL. For second-line and later treatments, CAR T-cell therapies and bispecific antibodies have been observed to induce deep and durable remissions for patients independent of whether they were previously considered eligible for transplant. The emergence of new treatments such as novel bispecific antibodies, targeted therapies, CELMoDs, and novel CAR T-cell and bispecific technologies are promising. Collectively, these novel therapies hold the potential to optimize outcomes and improve durable remission and potentially cure rates in both the upfront and relapsed settings in the years to come.