CE / CME
Novel RAS-Targeted Therapies for Pancreatic Ductal Adenocarcinoma
ABIM MOC: maximum of 0.75 Medical Knowledge MOC point
Physicians: Maximum of 0.75 AMA PRA Category 1 Credit™
Released: April 15, 2026
Expiration: October 14, 2026
Activity
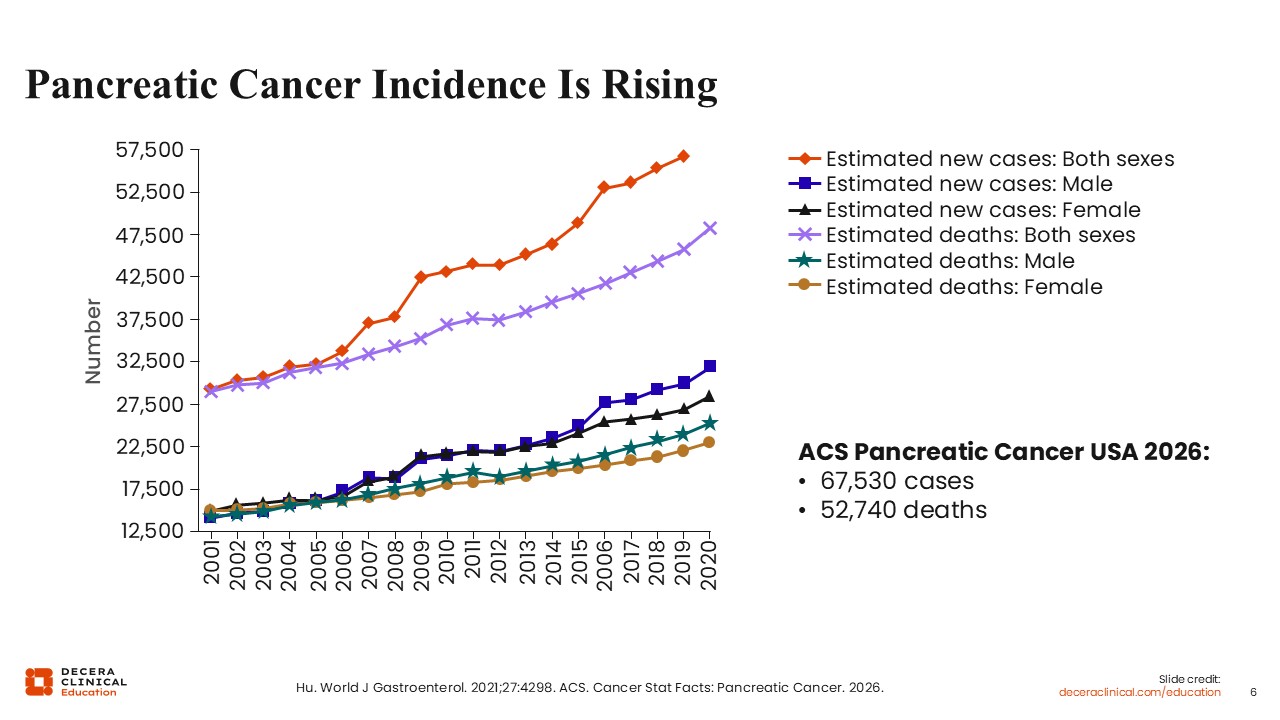
Pancreatic Cancer Incidence Is Rising
Christopher Lieu, MD, FASCO: In the United States, both the incidence and mortality of pancreatic cancer have increased over the past 2 decades, affecting both males and females. In 2026, an estimated 67,000 new cases of pancreatic cancer will be diagnosed, with approximately 52,000 deaths attributed to the disease.1,2
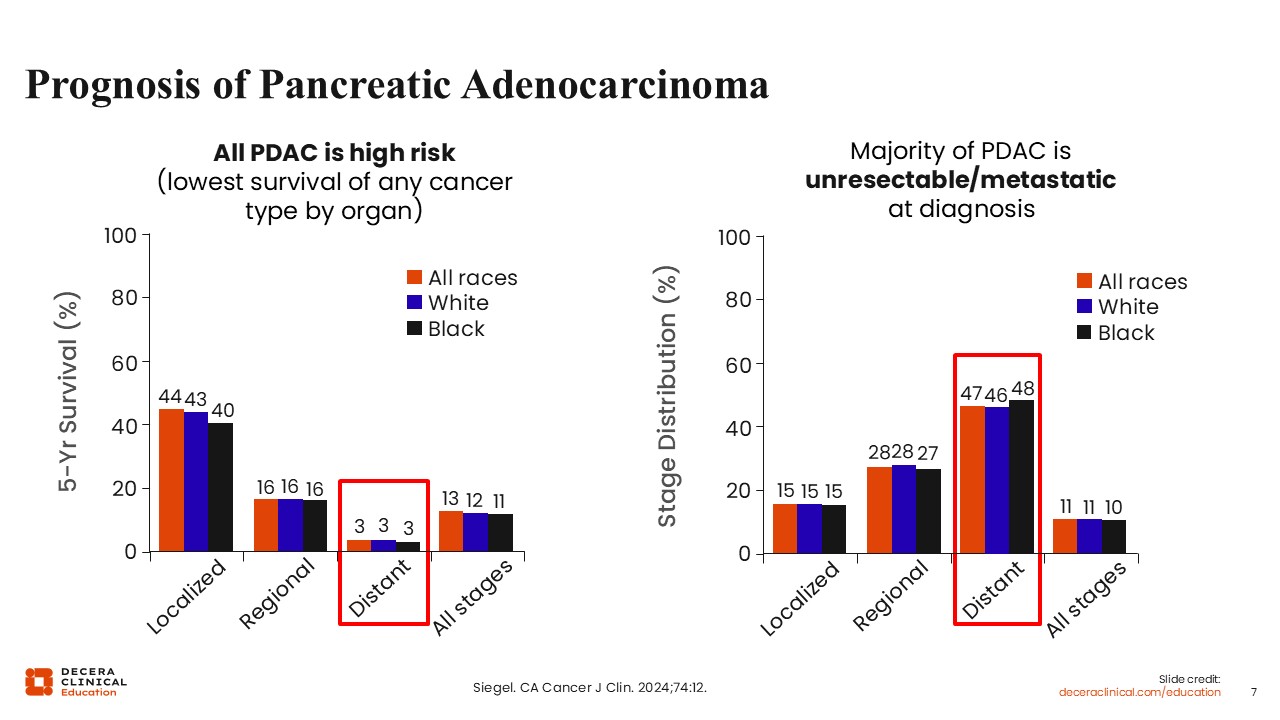
Prognosis of Pancreatic Adenocarcinoma
Christopher Lieu, MD, FASCO: Pancreatic adenocarcinoma remains associated with a poor prognosis, with limited screening tests causing most patients to be diagnosed with the disease at an advanced stage. This contributes to one of the lowest overall survival (OS) rates among solid tumors.3
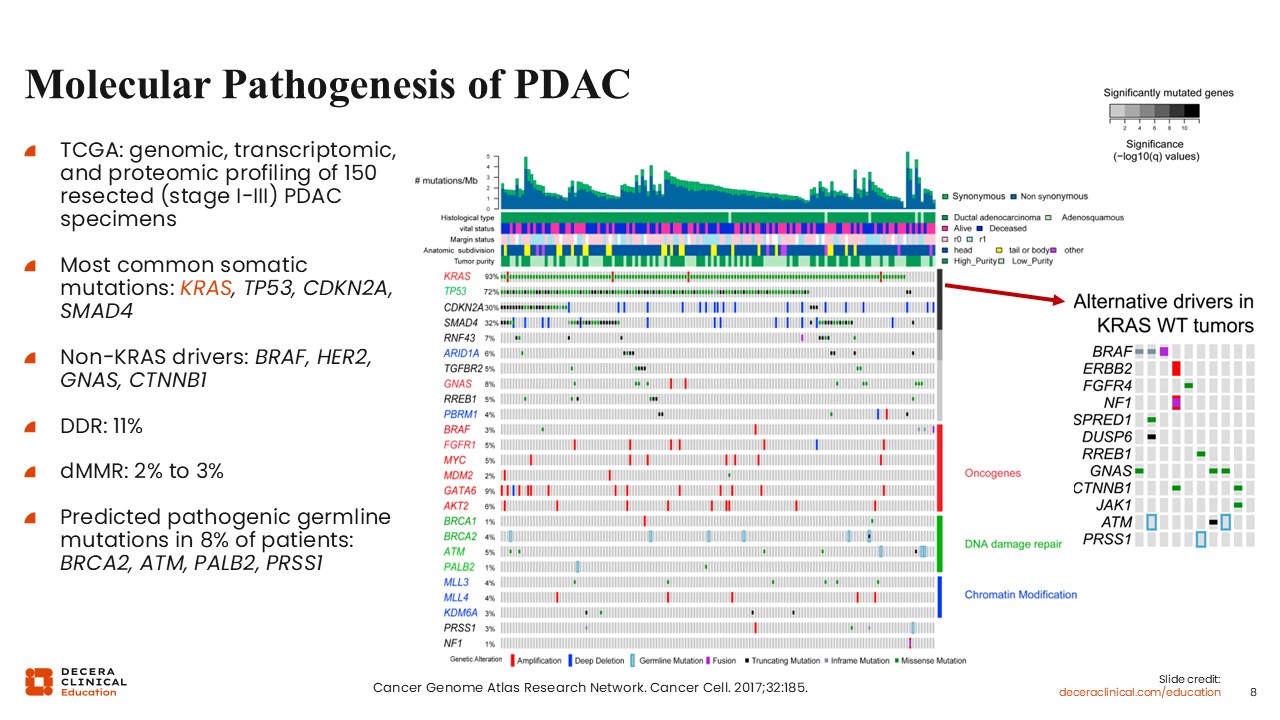
Molecular Pathogenesis of PDAC
Christopher Lieu, MD, FASCO: The Cancer Genome Atlas (TCGA) provided important insights into the molecular characteristics of pancreatic adenocarcinoma through a genomic, transcriptomic, and proteomic profiling study of 150 resected tumors, primarily of stage I through III disease.4
KRAS mutations were the most common somatic alterations in PDAC, occurring in approximately 93% of tumors. TP53 mutations are also highly prevalent, and CDKN2A and SMAD4 mutations are observed in a substantial proportion of cases.
Less frequent non-KRAS drivers include BRAF and HER2 alterations. DNA damage repair deficiencies are present in approximately 10% of tumors. Deficient mismatch repair is rare, occurring in approximately 2% of cases.
Of importance, pathogenic germline mutations are identified in approximately 7% to 8% of patients, supporting the role of genetic evaluation in this population.
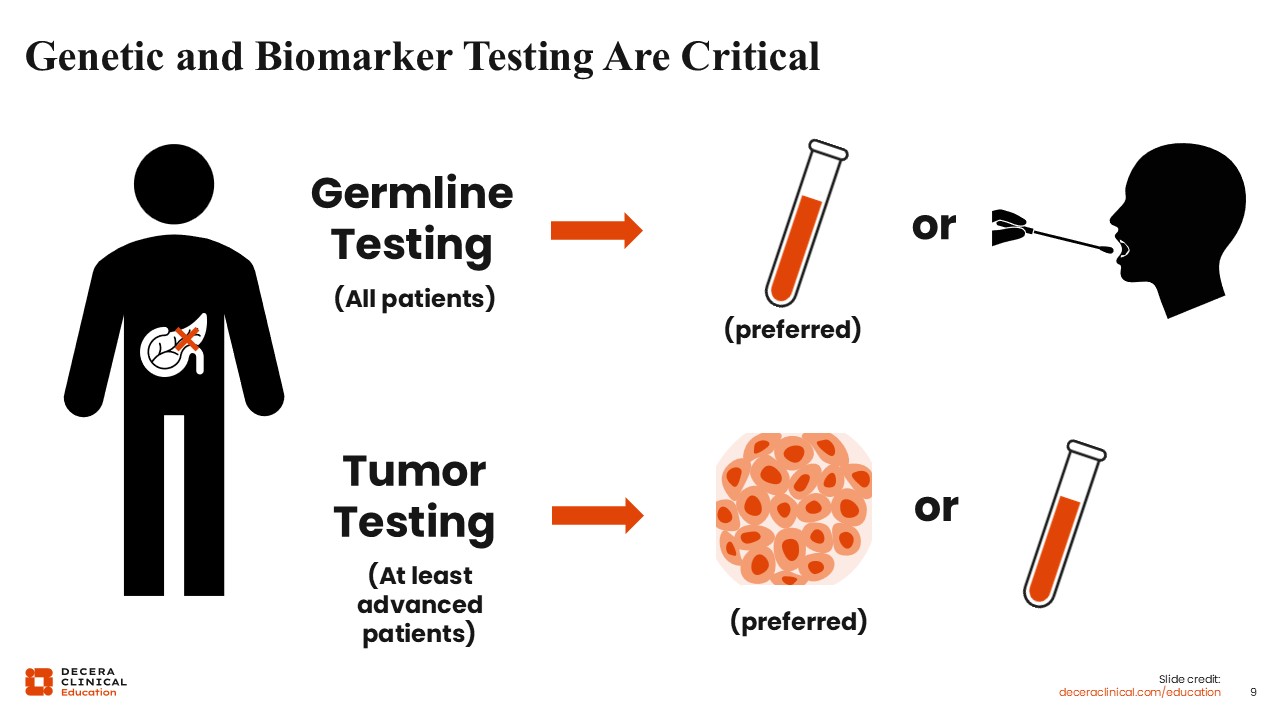
Genetic and Biomarker Testing Are Critical
Christopher Lieu, MD, FASCO: Genetic and biomarker testing are critical in the management of PDAC. Germline testing is recommended by the National Comprehensive Cancer Network (NCCN) for all patients diagnosed with PDAC. This has implications not only for familial risk but also for treatment selection.5
Tumor-based biomarker testing is also essential, particularly in patients with advanced or metastatic disease. Although actionable alterations are relatively uncommon, the identification of molecular targets is increasingly important with the development of RAS-directed therapies.
Wungki Park, MD, MS: This remains a very challenging disease. However, there is increasing momentum in research that offers hope for patients. In clinical practice, I am seeing more patients diagnosed at younger ages, including individuals in their 40s and 50s, rather than the traditional median age in the 70s. This trend is concerning but underscores the importance of continued research and collaboration.
Christopher Lieu, MD, FASCO: I agree. We are also seeing younger patients in the clinic, which further emphasizes the importance of germline testing in this population. Although treatment progress has historically been limited, the outlook is evolving, and we will discuss some emerging developments shortly.
KRAS Biology
Christopher Lieu, MD, FASCO: I will now briefly discuss KRAS mutations in pancreatic cancer.
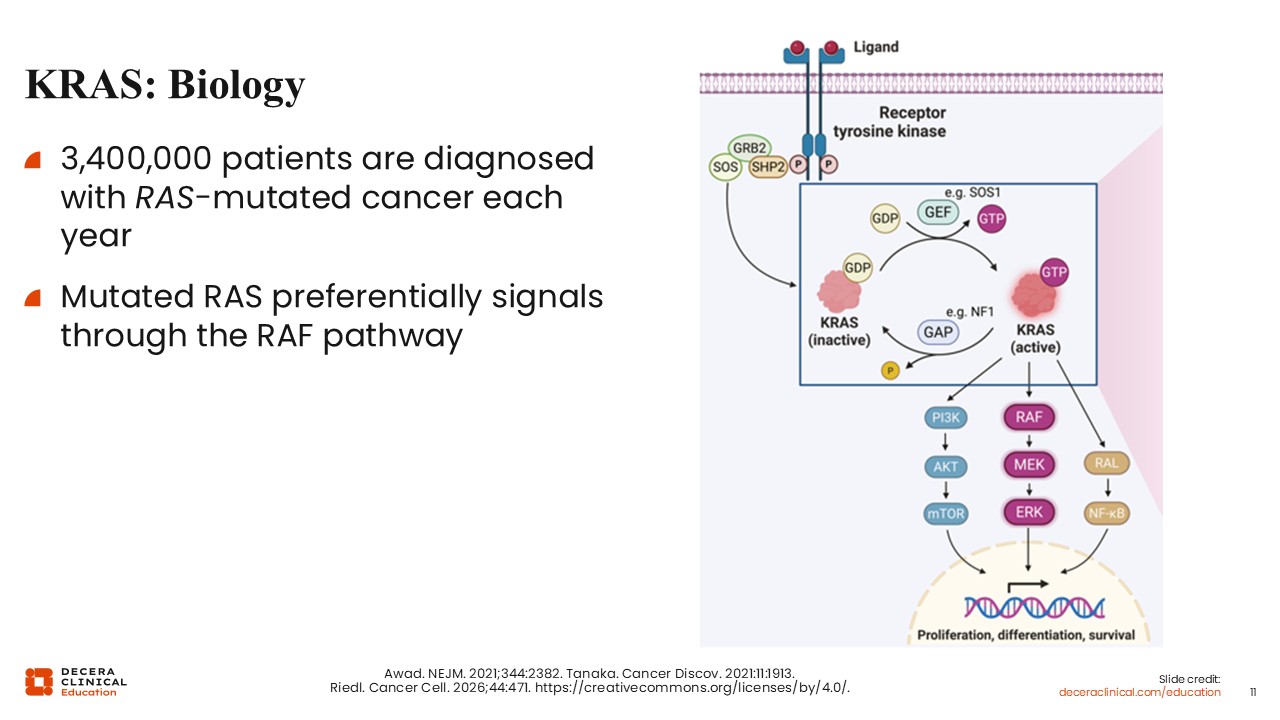
KRAS is a highly prevalent oncogenic driver across multiple cancer types. It is estimated that more than 3.4 million patients are diagnosed with RAS-mutated cancers each year.
Mutated RAS proteins preferentially signal through the RAF pathway, as illustrated in the accompanying diagram, ultimately driving downstream signaling that promotes tumor growth and survival. In pancreatic cancer, KRAS mutations are present in the vast majority of cases.4
KRAS: Distribution and Prognosis
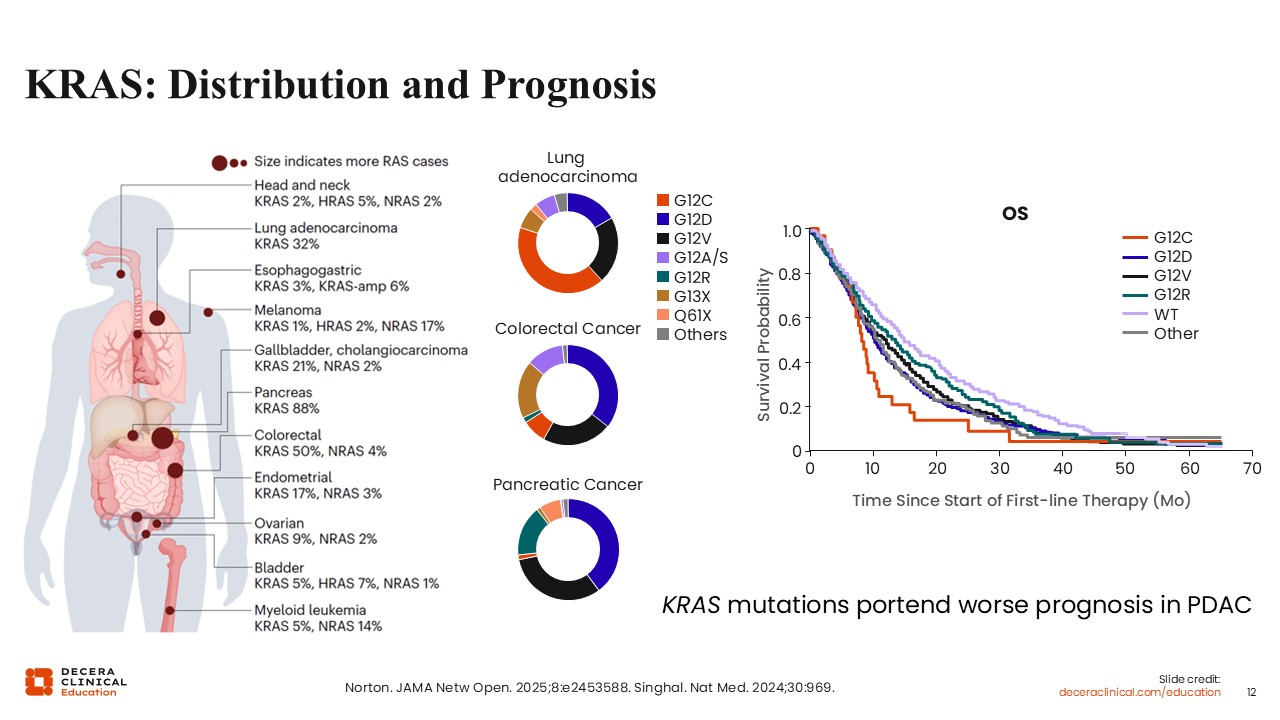
Christopher Lieu, MD, FASCO: The distribution of KRAS mutations varies across tumor types, but pancreatic cancer has the highest prevalence among solid tumors.6,7 Within pancreatic adenocarcinoma, the most common KRAS mutations are G12D and G12V, which together are present in the majority of cases.
From a prognostic standpoint, KRAS mutations are associated with worse outcomes. Although pancreatic cancer overall carries a poor prognosis, certain KRAS mutations, including G12D and G12C, are associated with particularly unfavorable survival.
The RAS-GTPase Effector Cycle
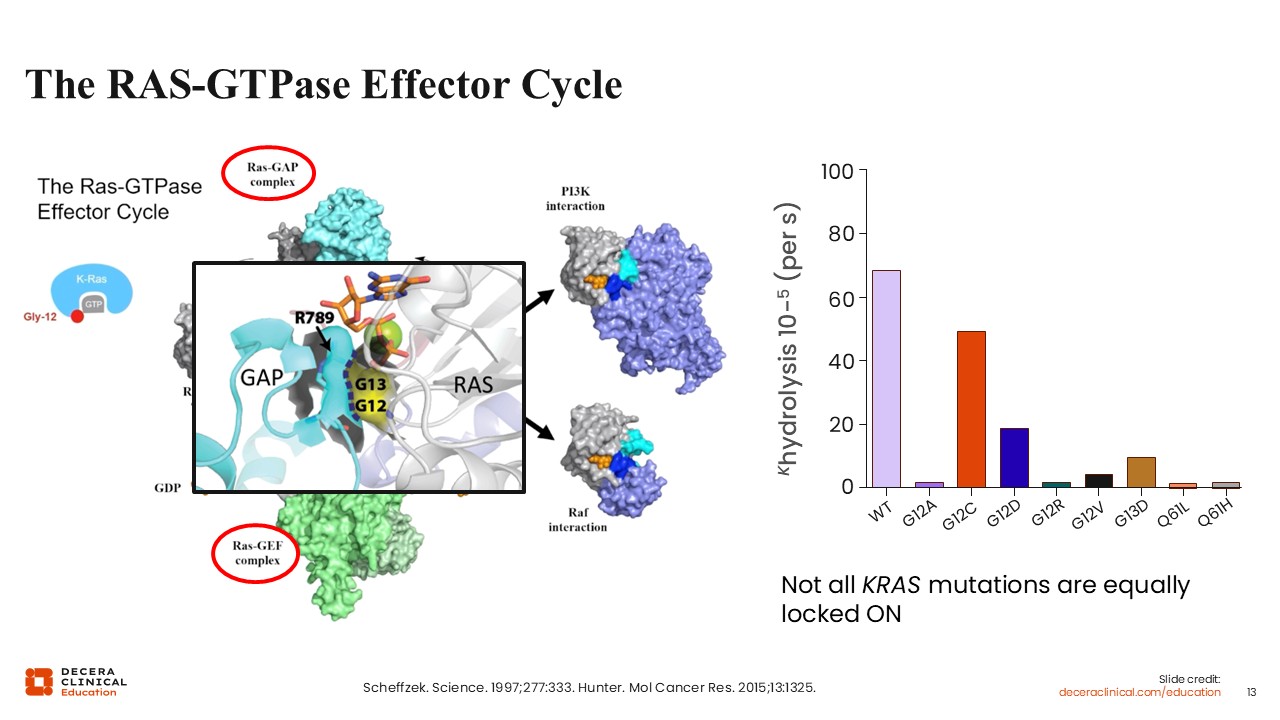
Christopher Lieu, MD, FASCO: One of the major challenges in targeting KRAS has been the complexity of RAS-GTPase signaling. Historically, this pathway has been considered difficult to inhibit, and drug development efforts have progressed slowly.
This longstanding challenge is a key reason for the current level of excitement in the field. We are now seeing the emergence of therapies that can effectively target KRAS, representing a significant shift in the treatment landscape for pancreatic cancer.8,9
Wungki Park, MD, MS: I agree. KRAS has long been considered one of the most important yet historically “undruggable” targets in pancreatic cancer. Now, that paradigm is changing. We are beginning to see meaningful progress, with multiple agents demonstrating activity. The field is rapidly evolving, and we are gaining a better understanding of allele-specific biology and signaling dynamics, including the importance of different nucleotide-bound states. Although we do not yet fully understand how these biological differences translate into clinical outcomes, this is clearly an exciting time in drug development.
PRODIGE 4-ACCORD 11: 1L FOLFIRINOX vs Gemcitabine for mPDAC
Christopher Lieu, MD, FASCO: Before we examine targeted therapies for PDAC, let’s briefly review the historical and current chemotherapy options for advanced pancreatic cancer.
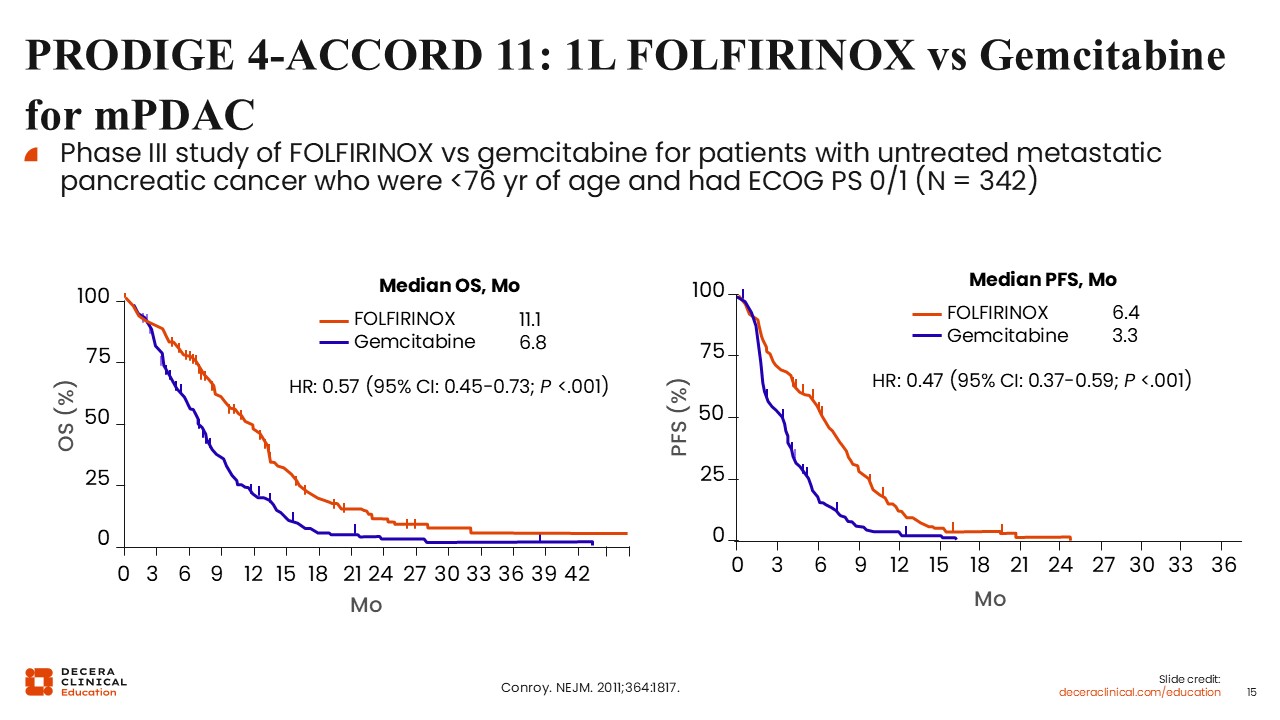
The PRODIGE 4/ACCORD 11 trial, published approximately 15 years ago, remains a foundational study in this setting. This phase III trial compared FOLFIRINOX with single-agent gemcitabine for patients with previously untreated metastatic pancreatic cancer.10
FOLFIRINOX demonstrated a significant improvement in both progression-free survival (PFS) and OS. Median OS was 11.1 months with FOLFIRINOX compared with approximately 7 months with gemcitabine. PFS was nearly doubled.
Although FOLFIRINOX is associated with increased toxicity due to its triplet chemotherapy backbone, it established a standard-of-care option in the first-line setting.
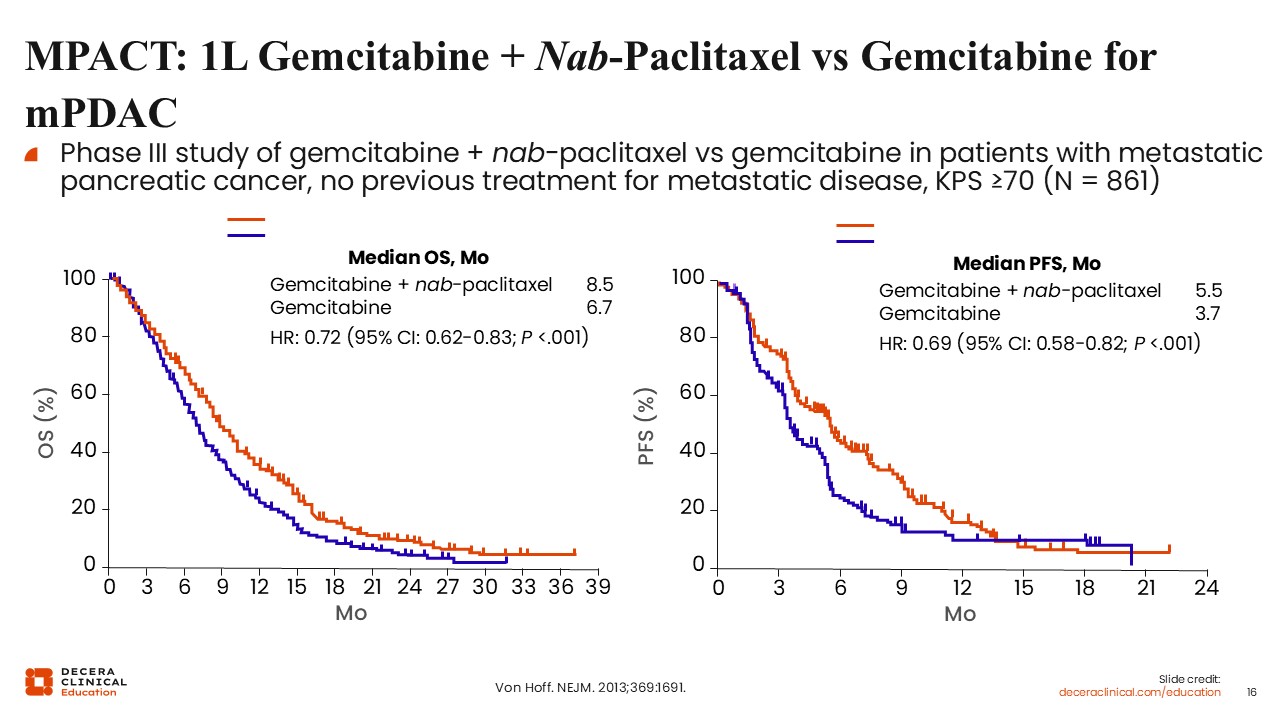
MPACT: 1L Gemcitabine + Nab-Paclitaxel vs Gemcitabine for mPDAC
Christopher Lieu, MD, FASCO: The MPACT trial evaluated gemcitabine in combination with nab-paclitaxel compared with gemcitabine alone in patients with previously untreated metastatic pancreatic adenocarcinoma.11
This phase III study demonstrated an improvement in median OS from 6.7 months with gemcitabine alone to 8.5 months with the combination. PFS improved from 3.7 months to 5.5 months. This regimen represents another standard first-line treatment option for mPDAC.
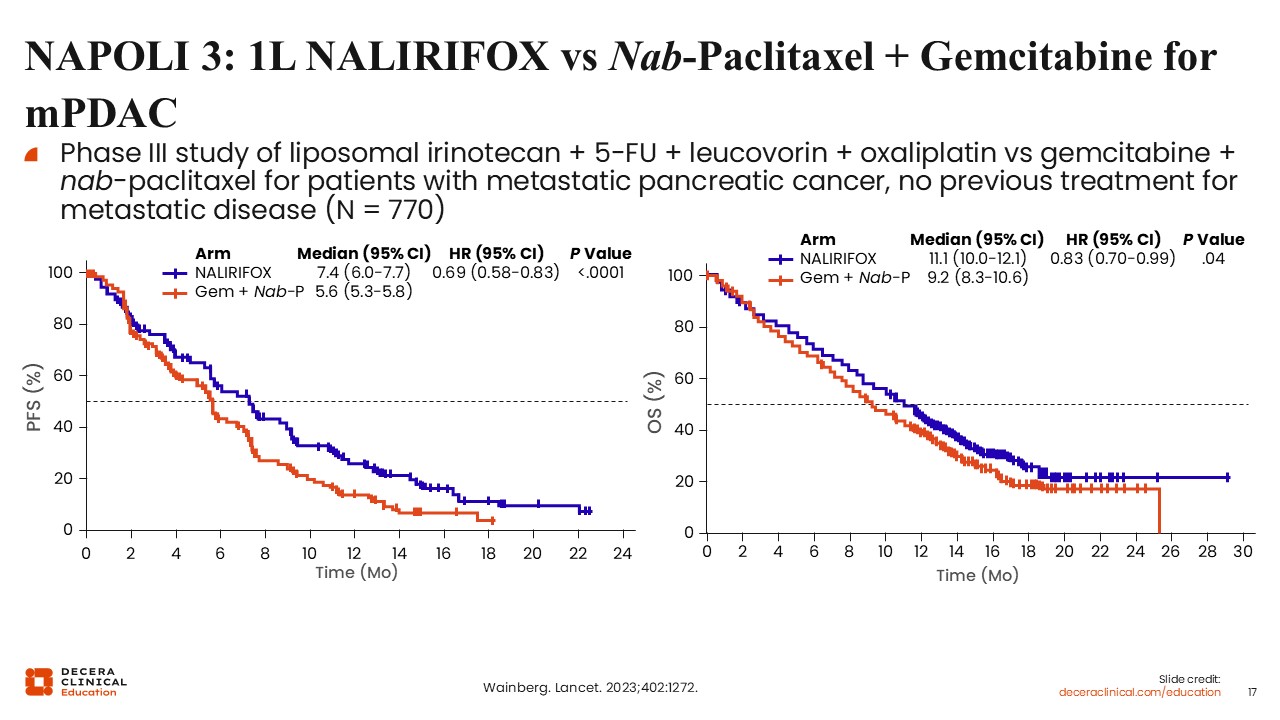
NAPOLI 3: 1L NALIRIFOX vs Nab-Paclitaxel + Gemcitabine for mPDAC
Christopher Lieu, MD, FASCO: More recently, the NAPOLI 3 trial evaluated a regimen consisting of liposomal irinotecan combined with 5-fluorouracil, leucovorin, and oxaliplatin (NALIRIFOX) compared with gemcitabine plus nab-paclitaxel.11
This study demonstrated a statistically significant improvement in OS, with a median OS of 11.1 months in the NALIRIFOX arm compared with 9.2 months in the comparator arm. Although the magnitude of benefit is incremental, this study provided evidence for an additional frontline treatment option.
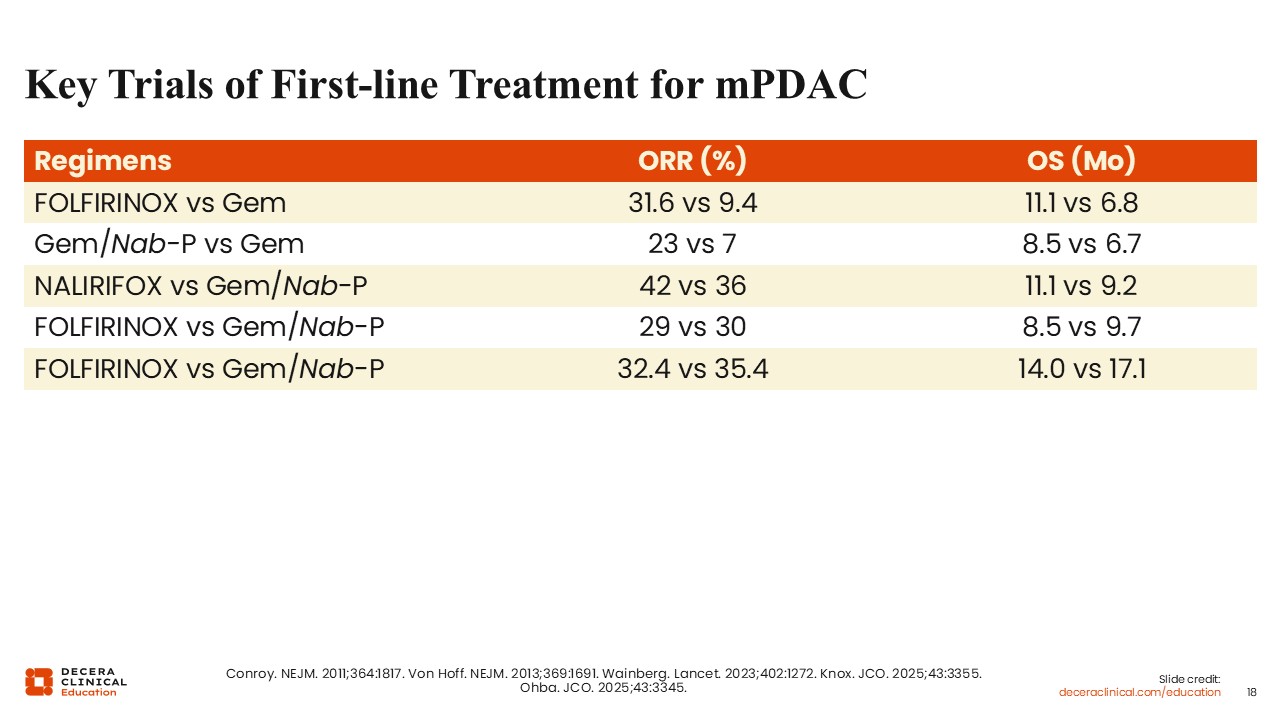
Key Trials of First-line Treatment for mPDAC
Christopher Lieu, MD, FASCO: Frontline trials have assessed FOLFIRINOX vs gemcitabine, gemcitabine plus nab-paclitaxel vs gemcitabine, and NALIRIFOX vs gemcitabine plus nab-paclitaxel. Recent comparative studies of FOLFIRINOX and gemcitabine plus nab-paclitaxel have suggested numerically improved survival outcomes with gemcitabine plus nab-paclitaxel. However, in aggregate, there is probably a fairly good equipoise in terms of selecting either FOLFIRINOX or gemcitabine plus nab-paclitaxel as frontline regimens. Overall, treatment selection should be individualized based on patient characteristics and tolerability.
Dr Park, what is your perspective on how these regimens are selected in clinical practice?
Wungki Park, MD, MS: Historically, the PRODIGE trial established that triplet therapy with FOLFIRINOX was superior to gemcitabine alone, and the NAPOLI 3 study reinforced this benefit. However, there is clear equipoise in many clinical scenarios. Patient selection is critical and should consider factors such as age, performance status, and comorbidities. For example, in older or more frail populations, less intensive regimens may be more appropriate. In addition, genetic background may inform treatment selection. In patients with homologous recombination deficiency, platinum-based therapies may be particularly beneficial.
At the Memorial Sloan Kettering Cancer Center, FOLFIRINOX is frequently used in the first-line setting when feasible, although this may evolve with ongoing clinical trial data.
Christopher Lieu, MD, FASCO: I agree. Many healthcare professionals favor triplet therapy when patients can tolerate it, although gemcitabine plus nab-paclitaxel remains an important option. Overall, outcomes with chemotherapy appear to plateau at approximately 1 year of median OS, suggesting that we may be approaching the limits of what cytotoxic therapy alone can achieve in this disease.
Wungki Park, MD, MS: I agree. Despite incremental improvements over the past several decades, progress with chemotherapy has been limited. This highlights the need for novel therapeutic approaches.
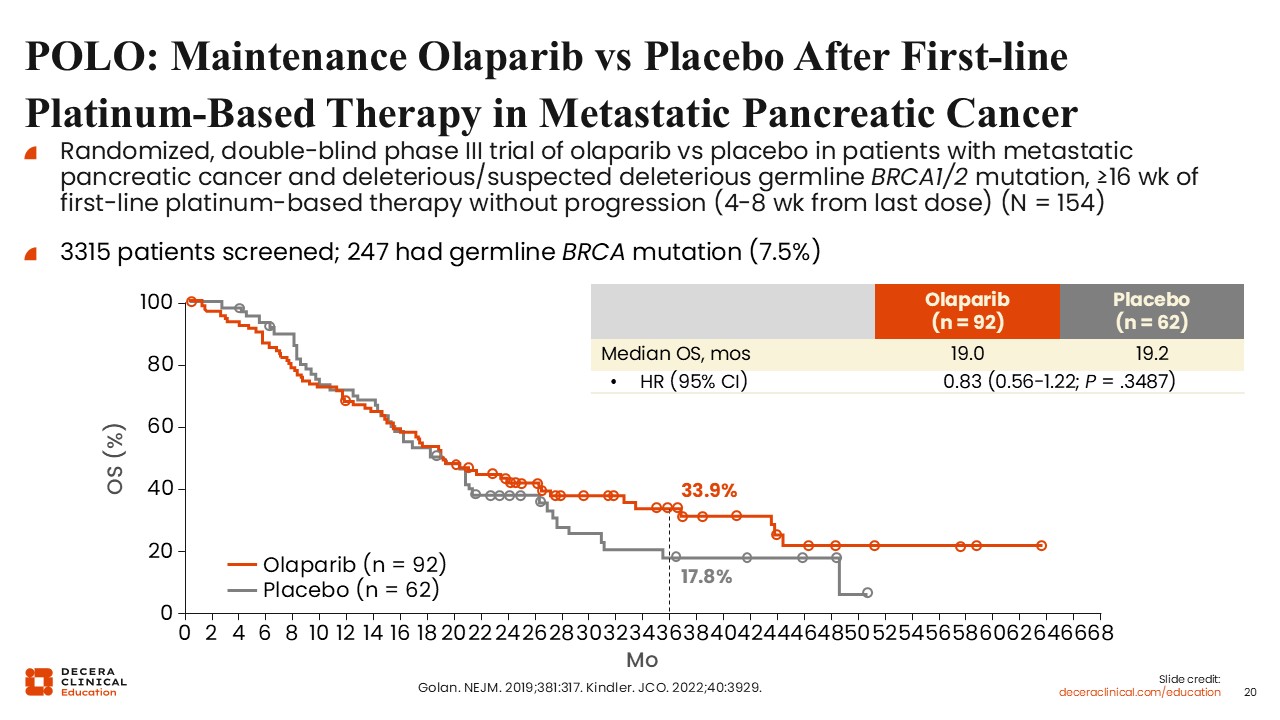
POLO: Maintenance Olaparib vs Placebo After First-line Platinum-Based Therapy in Metastatic Pancreatic Cancer
Christopher Lieu, MD, FASCO: Considering this, let’s discuss targeted therapy approaches in mPDAC.
Approximately 7% to 8% of patients with pancreatic adenocarcinoma harbor germline BRCA mutations. In this population, PARP inhibitors have demonstrated clinical activity through impairment of DNA repair mechanisms.
The phase III POLO trial evaluated maintenance olaparib in patients with germline BRCA1 or BRCA2 mutations whose disease had not progressed after at least 16 weeks of first-line platinum-based chemotherapy. Patients were randomized to receive olaparib 300 mg twice daily or placebo in the maintenance setting.13,14 Of note, more than 3000 patients were screened to identify 247 eligible participants, highlighting the challenges of enrolling this biomarker-defined population.
The primary endpoint of PFS favored olaparib, with a median PFS of 7.4 months compared with 3.8 months for placebo. However, no significant difference in OS was observed between the 2 groups. This may, in part, reflect the use of subsequent therapies, including PARP inhibitors, in the placebo arm.
Although the absence of an OS benefit is notable, olaparib remains a maintenance option for selected patients, with some individuals experiencing durable responses. Identifying those patients most likely to benefit remains an area of ongoing investigation.
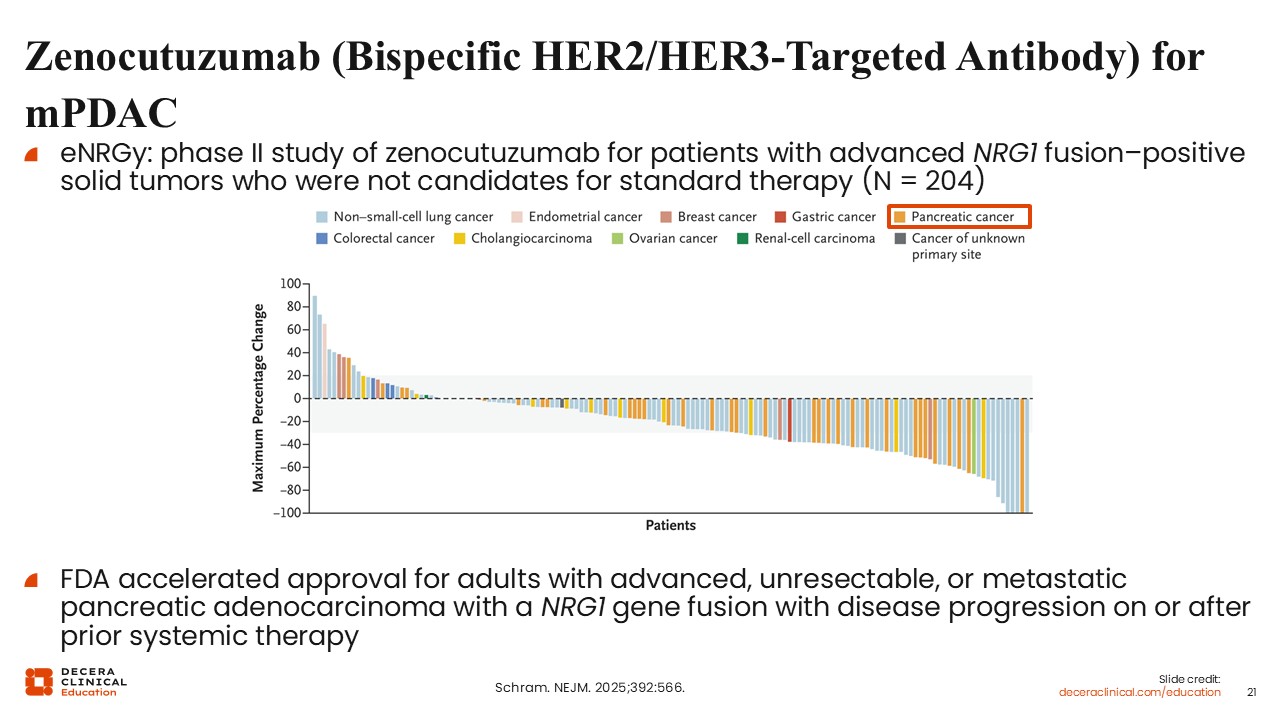
Zenocutuzumab (Bispecific HER2/HER3-Targeted Antibody) for mPDAC
Christopher Lieu, MD, FASCO: Additional targeted therapies are available for select patient populations. Zenocutuzumab is a bispecific antibody targeting HER2 and HER3 and is approved for adults with advanced, unresectable, or metastatic pancreatic adenocarcinoma with a NRG1 gene fusion with disease progression on or after prior systemic therapy.15
NRG1 fusions are rare, occurring in approximately 0.5% to 1% of patients with pancreatic cancer. However, identification of this alteration is clinically important, as targeted therapy can yield meaningful responses in this subset.
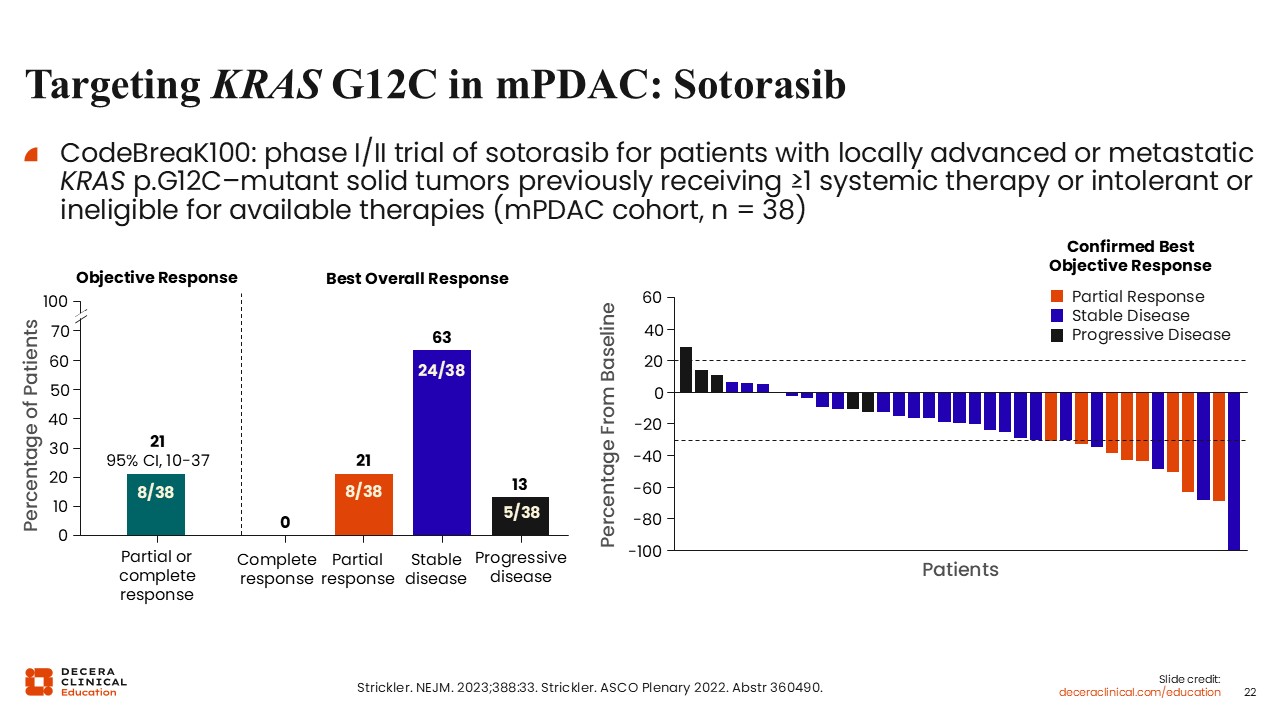
Targeting KRAS G12C in mPDAC: Sotorasib
Christopher Lieu, MD, FASCO: Let’s now turn our attention to KRAS inhibition in PDAC. There is also emerging experience with KRAS G12C inhibitors in pancreatic cancer, although this alteration is found in a small subset of disease, approximately 1% to 2% of cases.
Data from early-phase studies, including CodeBreak 100 and KRYSTAL-1, demonstrated modest activity in PDAC with the KRAS G12C inhibitors sotorasib and adagrasib. In the CodeBreak 100 study of sotorasib for patients with previously treated advanced KRAS p.G12C–mutant PDAC, the median PFS was approximately 4 months, and the median OS was 6.9 months.16
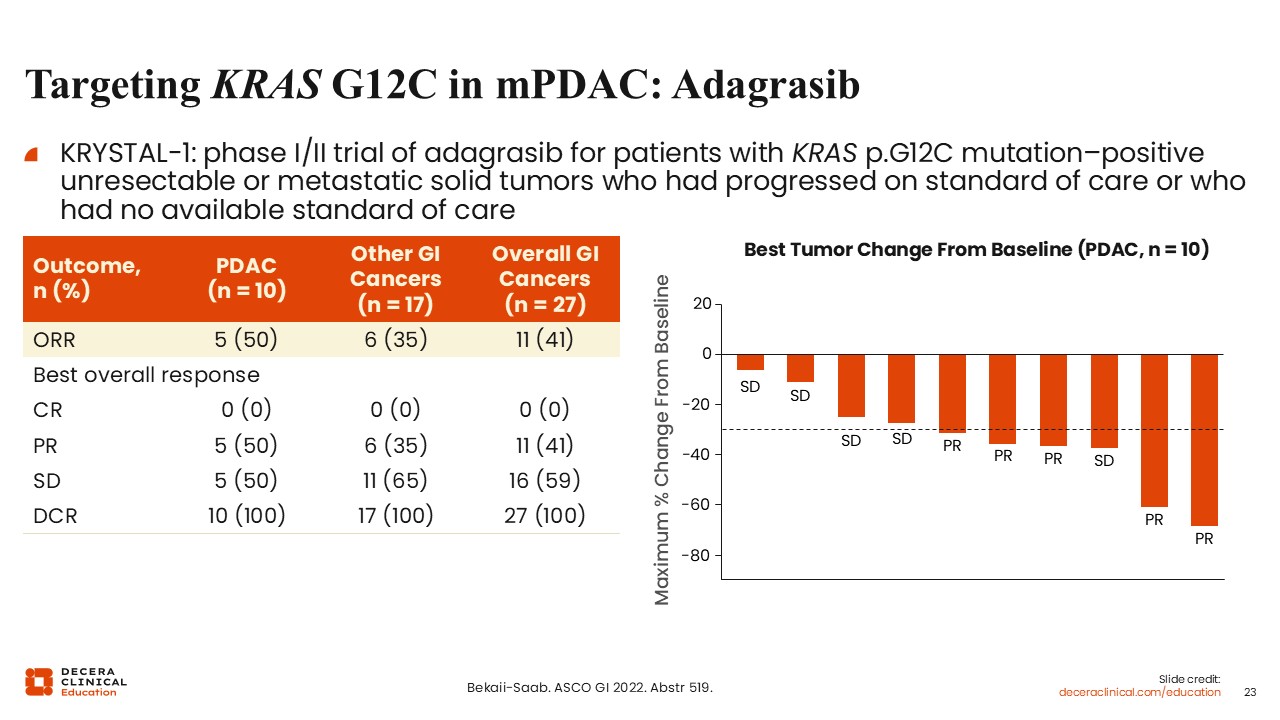
Targeting KRAS G12C in mPDAC: Adagrasib
Christopher Lieu, MD, FASCO: In the KRYSTAL-1 study of adagrasib for patients with KRAS p.G12C mutation–positive solid tumors who had progressed on standard of care or who had no available standard of care, an ORR of 50% was observed in a small cohort of patients with pancreatic cancer.17 Although these data are limited by small sample sizes, they suggest the potential activity of KRAS G12C inhibitors in this setting.
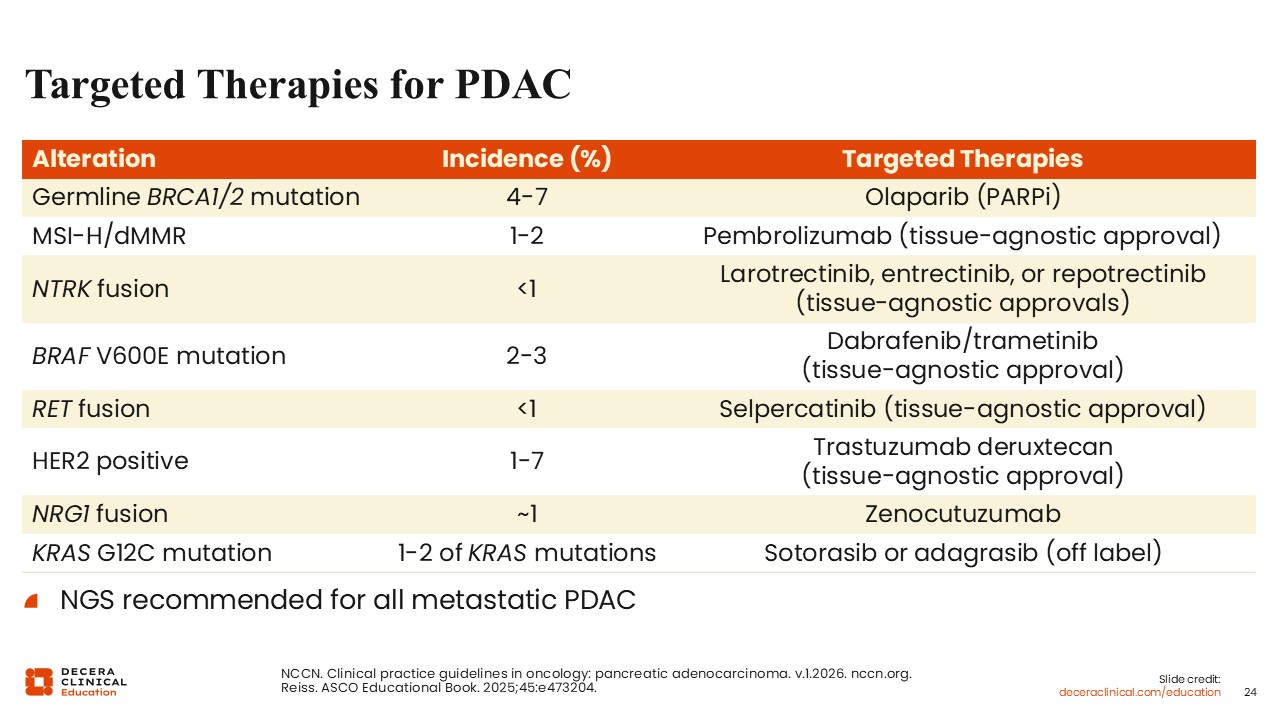
Targeted Therapies for PDAC
Christopher Lieu, MD, FASCO: The targeted therapy landscape in pancreatic cancer highlights 2 key points. First, actionable alterations can be identified through next-generation sequencing, reinforcing the importance of comprehensive biomarker testing. Even though these alterations can be relatively uncommon, they can have significant therapeutic implications.
Second, the majority of patients with pancreatic cancer do not currently have identifiable, targetable alterations with approved therapies. Although subsets such as germline BRCA mutations, microsatellite instability–high tumors, KRAS G12C mutations, and rare gene fusions are clinically important, they represent a minority of cases.
Wungki Park, MD, MS: Targeted therapies can be very meaningful for patients with mPDAC. If an actionable alteration is not identified initially, repeat testing should be considered when feasible. Obtaining tissue in pancreatic cancer can be challenging, particularly in locally advanced disease. Circulating tumor DNA may be useful, although shedding can be limited in this disease. Despite these challenges, repeat testing may identify actionable targets and should be considered as part of routine practice.
Christopher Lieu, MD, FASCO: I agree. Comprehensive genomic profiling is essential. Even though actionable findings may be rare, they can significantly impact treatment decisions when identified. As the field evolves, this will likely become even more important.
From RAS Biology to RAS Therapeutics
Wungki Park, MD, MS: Dr Lieu has outlined the biology of KRAS and its importance in pancreatic cancer. I will now focus on how this biology is translating into therapeutic strategies. We are at an inflection point where KRAS is no longer purely a theoretical target. New therapeutic approaches are emerging and are expected to play a significant role in clinical practice.
As previously mentioned, KRAS mutations are present in approximately up to 95% of pancreatic cancers. In cases where KRAS mutations are not identified, particularly in typical patient populations, repeat testing is often warranted, including cell-free DNA analysis, to confirm mutation status.
Historically, KRAS has been considered undruggable. However, multiple therapeutic classes are now entering clinical development. Although no agents are currently approved in this space for PDAC, this is rapidly evolving.
How to Think About RAS-Targeted Therapy in PDAC in 2026
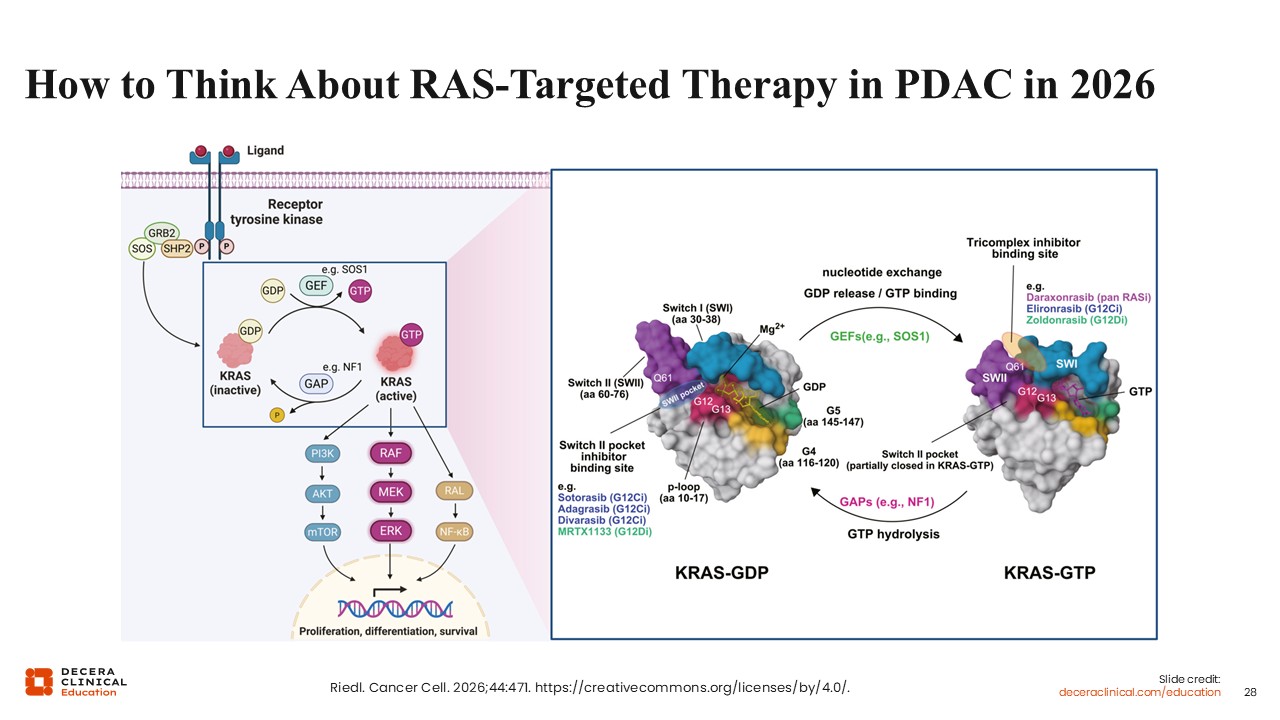
Wungki Park, MD, MS: One approach to KRAS inhibition is multi-allele targeting using pan-RAS inhibitors. Among KRAS mutations, G12D is the most common and particularly relevant in pancreatic and gastrointestinal malignancies. Pan-RAS strategies aim to target multiple KRAS variants simultaneously, potentially broadening applicability across patient populations.
RAS-targeted therapy can be understood within the context of cellular signaling pathways.18 KRAS cycles between an inactive GDP-bound state and an active GTP-bound state. In the active state, KRAS drives downstream signaling through pathways such as RAF, MEK, and ERK, promoting tumor cell proliferation and survival. Many cancers are dependent on this active signaling state, making it a critical therapeutic target.
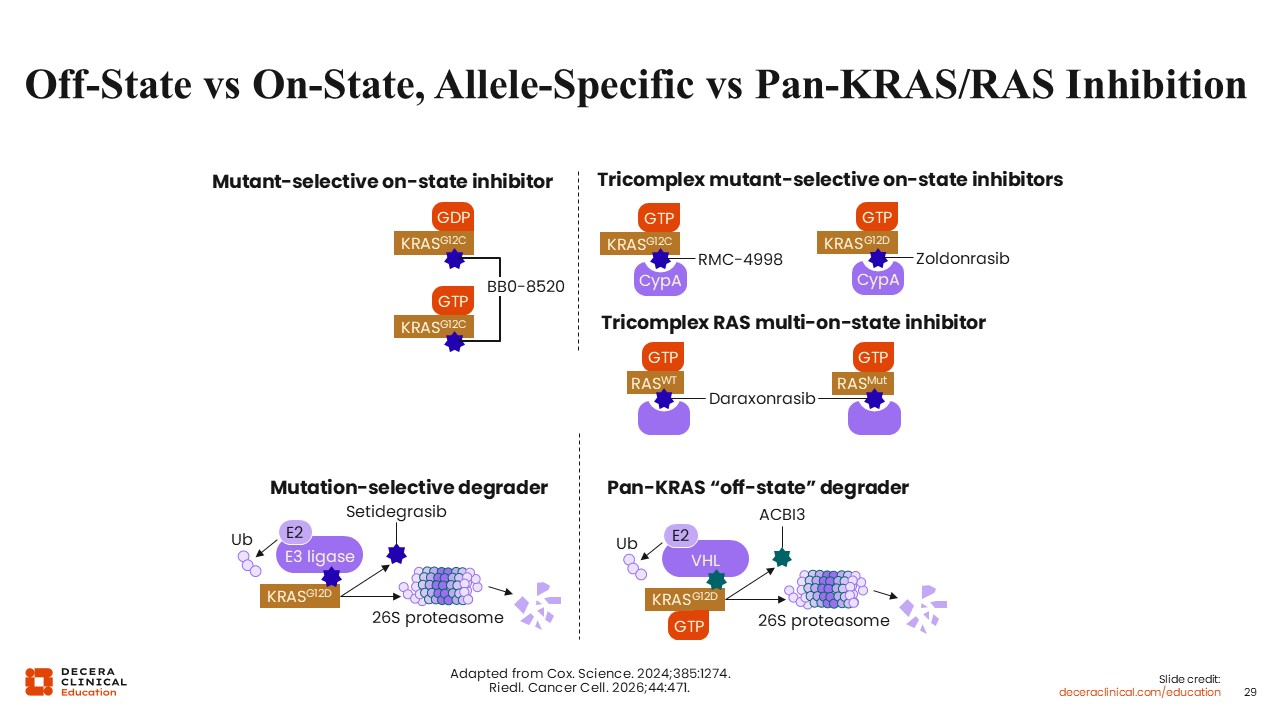
Off-State vs On-State, Allele-Specific vs Pan-KRAS/RAS Inhibition
Wungki Park, MD, MS: Advances in medicinal chemistry have enabled new strategies for targeting KRAS. One approach involves the use of “chemical glue” compounds that facilitate the formation of a tricomplex between KRAS in the “on” formation and other proteins, such as cyclophilin. This interaction stabilizes KRAS in a conformation that prevents downstream signaling, thereby inhibiting tumor growth and proliferation.19 Novel protein degraders against KRAS are also in development, as we will discuss momentarily.
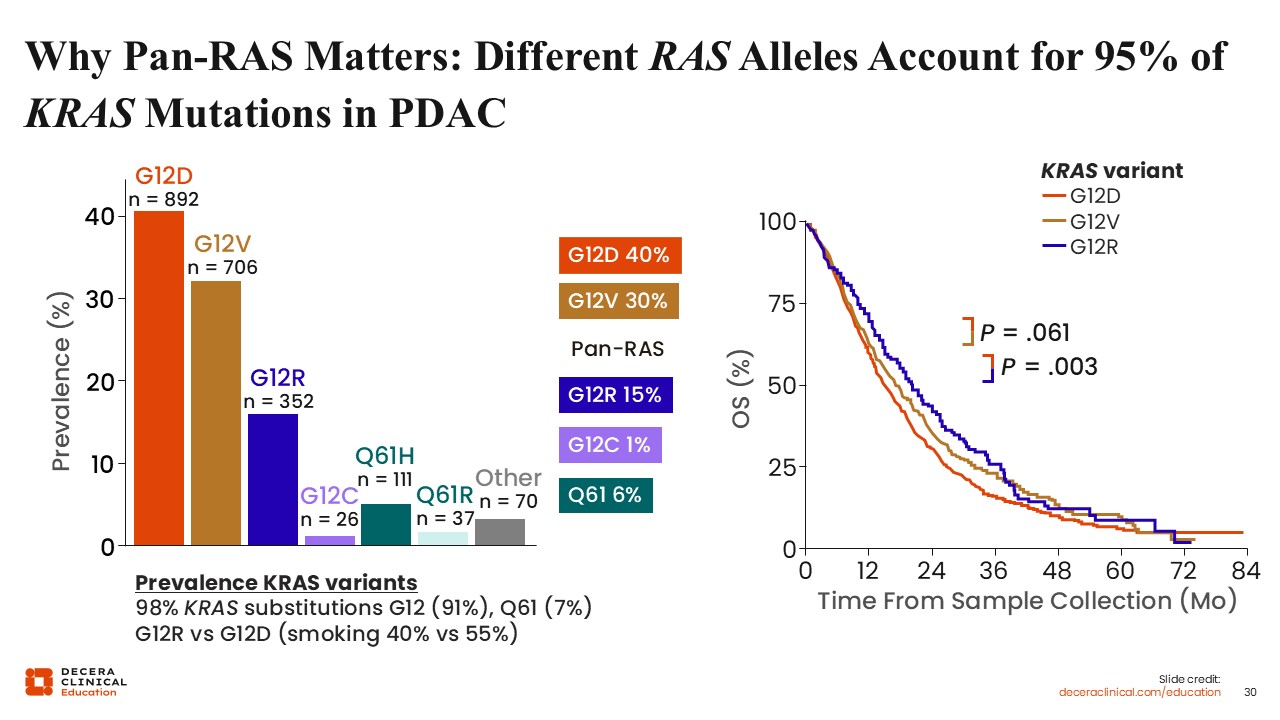
Why Pan-RAS Matters: Different RAS Alleles Account for 95% of KRAS Mutations in PDAC
Wungki Park, MD, MS: Pan-RAS targeting is particularly relevant because multiple KRAS alleles contribute to pancreatic cancer. The most common mutations include G12D (approximately 40%), G12V (approximately 30%), and G12R (approximately 15%).20 Allele-specific differences may influence prognosis and outcomes. However, pan-RAS inhibitors offer the advantage of targeting multiple variants, increasing potential applicability across patients.
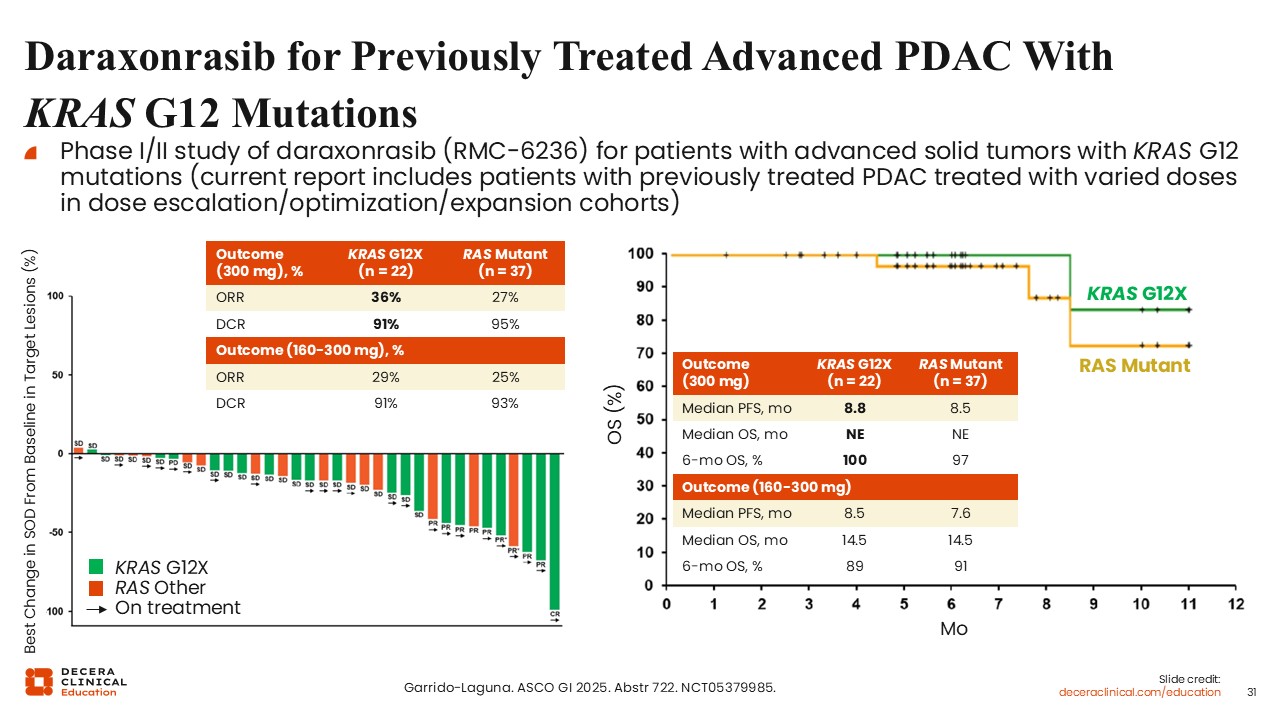
Daraxonrasib for Previously Treated Advanced PDAC With KRAS G12 Mutations
Wungki Park, MD, MS: Early clinical data with pan-RAS inhibitors continue to emerge. Daraxonrasib (RMC-6236) is an oral agent that has been evaluated in a first-in-human study in patients with previously treated metastatic pancreatic cancer with a KRAS G12 mutation.19
The ORR with this agent as second-line therapy for metastatic PDAC with a KRAS G12X mutation was approximately 35%, with a disease control rate of 92%. A median PFS of 8.5 months and a median OS of 13.1 months were observed.
Although this was a single-arm, early-phase study, these results are encouraging, particularly when compared with historical outcomes using chemotherapies in the second-line setting.
Christopher Lieu, MD, FASCO: In the second-line setting, response rates with chemotherapy are typically in the range of 5% to 10%. These results suggest a meaningful increase in response rates, along with a high disease control rate.
However, durability of response remains an important consideration, and randomized data will be necessary to confirm these findings.
Wungki Park, MD, MS: I agree. A phase III trial is ongoing, as we will discuss momentarily.
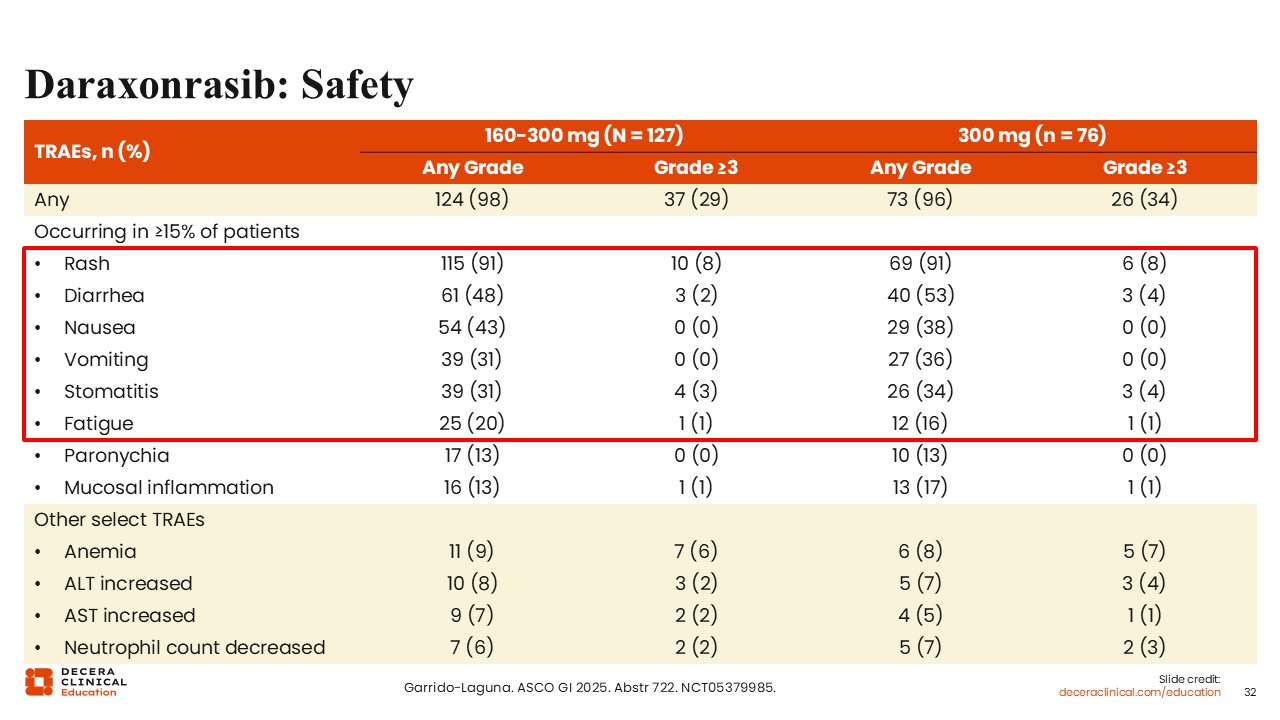
Daraxonrasib: Safety
Wungki Park, MD, MS: As with any targeted therapy, toxicity management is important. Common AEs with daraxonrasib include rash, which occurs in the majority of patients, as well as gastrointestinal toxicities such as diarrhea, nausea, and vomiting. Stomatitis and mucositis may also occur. These toxicities will require clinical familiarity and management strategies as these agents move into broader use. Despite these AEs, as we have shown, patients may experience clinical benefit, including improved disease control and maintenance of functional status.
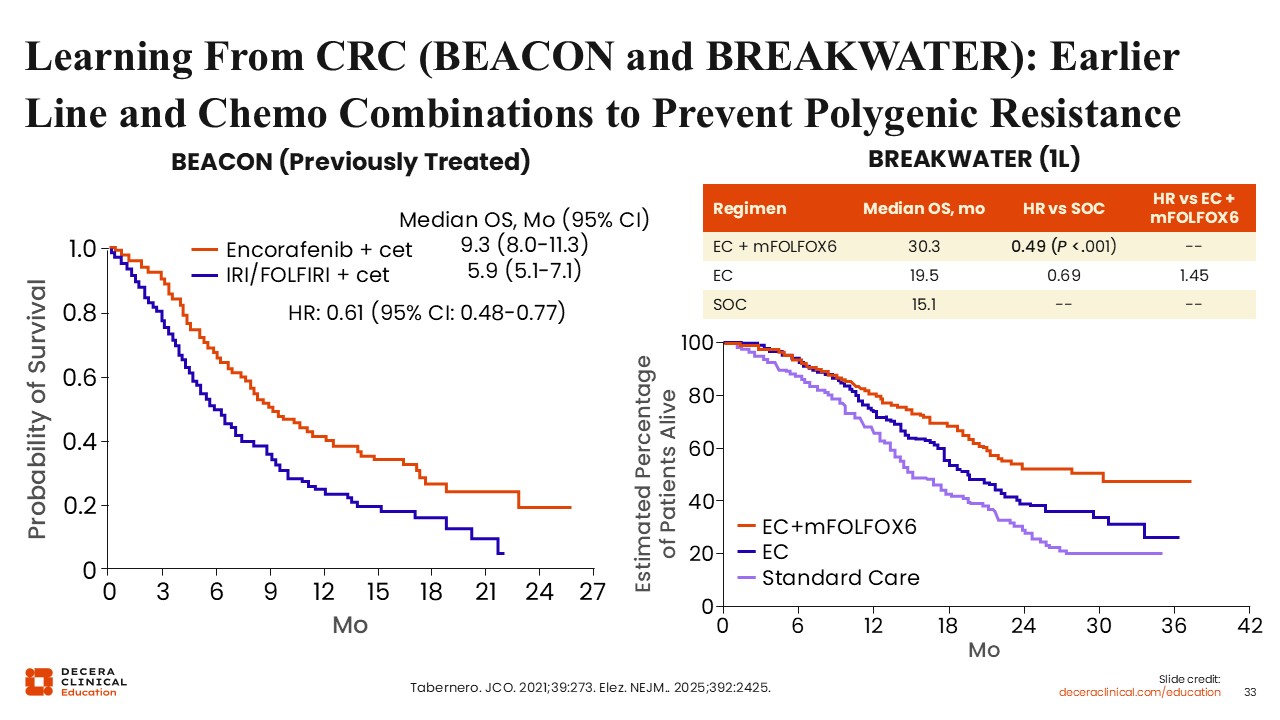
Learning From CRC (BEACON and BREAKWATER): Earlier Line and Chemo Combinations to Prevent Polygenic Resistance
Wungki Park, MD, MS: We can also learn from advances in colorectal cancer with targeted therapy, particularly from studies such as BEACON and BREAKWATER.21,22 In colorectal cancer, BRAF V600E mutations are associated with poor prognosis. Combination strategies incorporating targeted therapies, such as BRAF and EGFR inhibition, demonstrated improved outcomes in later-line settings.
When effective therapies are identified, the approach is often to move them earlier in treatment and to combine them with other agents to reduce the development of resistance. A similar strategy is being explored in pancreatic cancer.
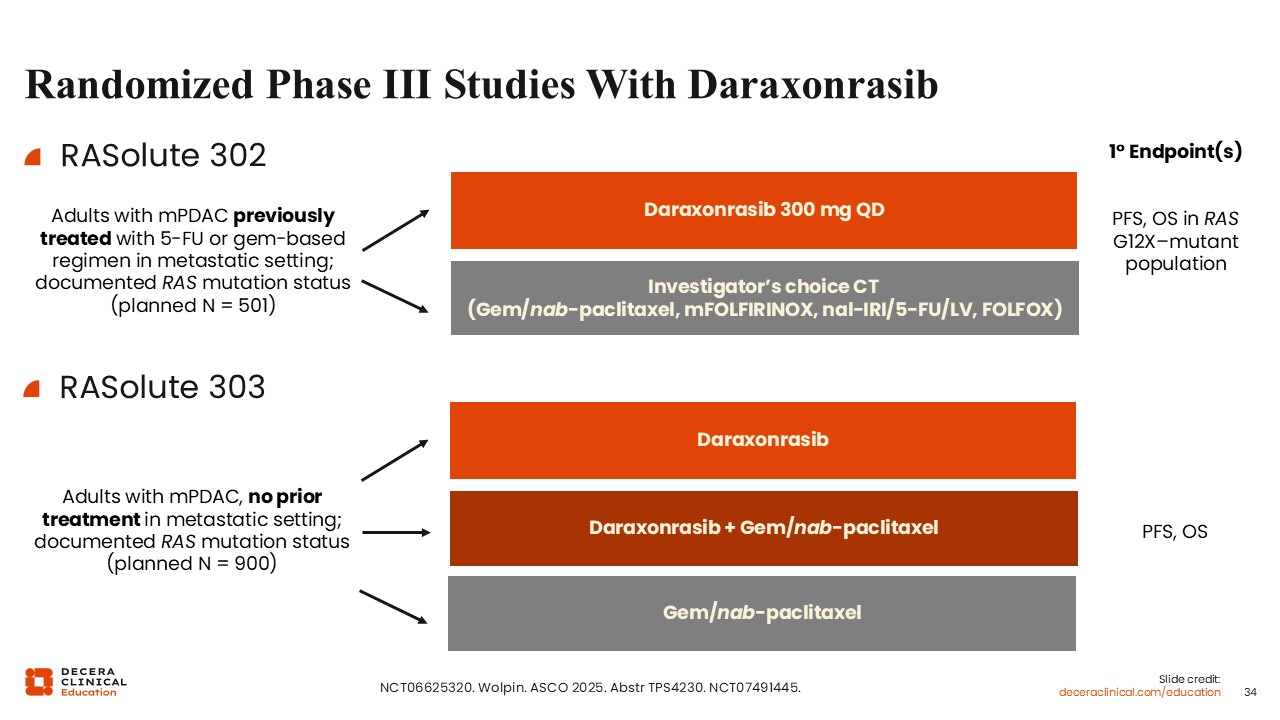
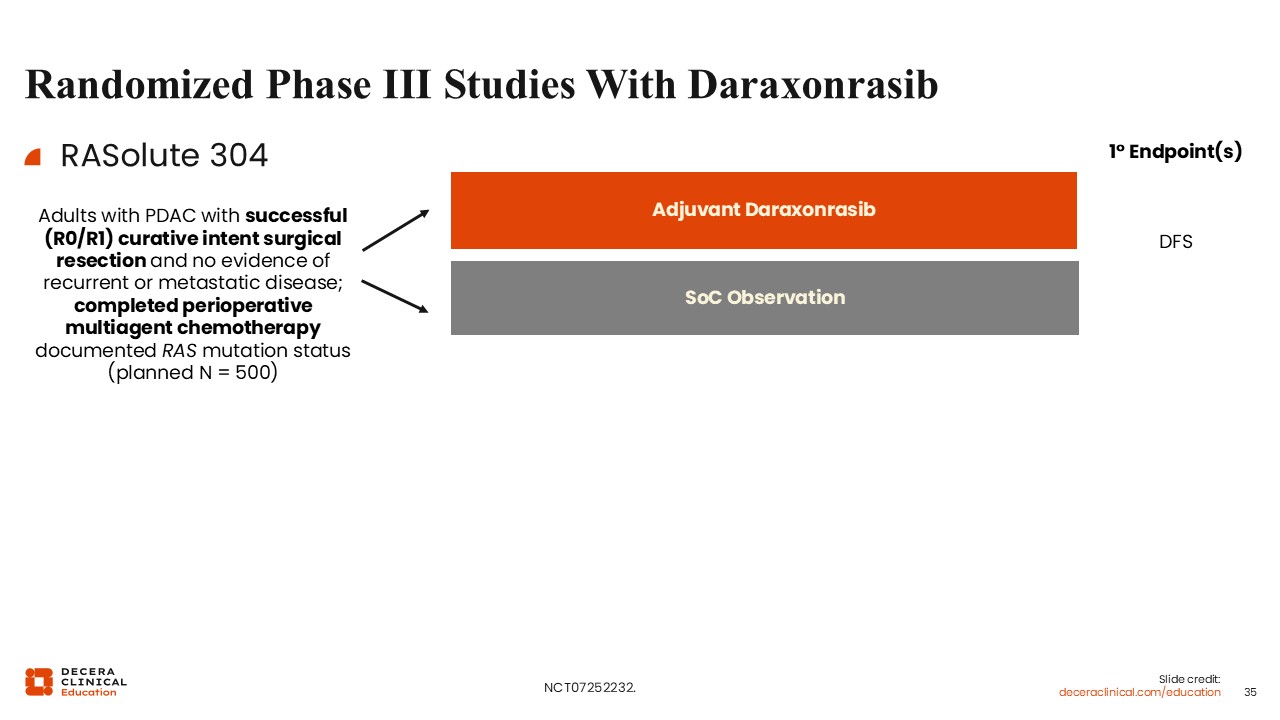
Randomized Phase III Studies With Daraxonrasib
Wungki Park, MD, MS: There are 3 randomized phase III trials evaluating daraxonrasib in PDAC. RASolute 302 (NCT06625320) enrolled previously treated patients with mPDAC (post 5- fluorouracil or gemcitabine-based therapy) and will compare daraxonrasib monotherapy with investigator’s choice of chemotherapy, with PFS and OS in RAS G12X-mutant population as the primary endpoints.
Notably, per an April 2026 press release, this trial met its primary endpoints, with statistically significant improvements in PFS and OS with daraxonrasib. Per the press release, data will be presented at the 2026 ASCO annual meeting.
RASolute 303 (NCT07491445) will evaluate daraxonrasib alone or in combination with gemcitabine/nab-paclitaxel vs standard gemcitabine/nab-paclitaxel for patients with treatment-naive mPDAC, with PFS and OS as primary endpoints.
Randomized Phase III Studies With Daraxonrasib (II)
Wungki Park, MD, MS: Finally, RASolute 304 (NCT07252232) will focus on the adjuvant setting in patients with resected PDAC (R0/R1) who completed perioperative chemotherapy, comparing daraxonrasib with observation, with DFS as the primary endpoint.
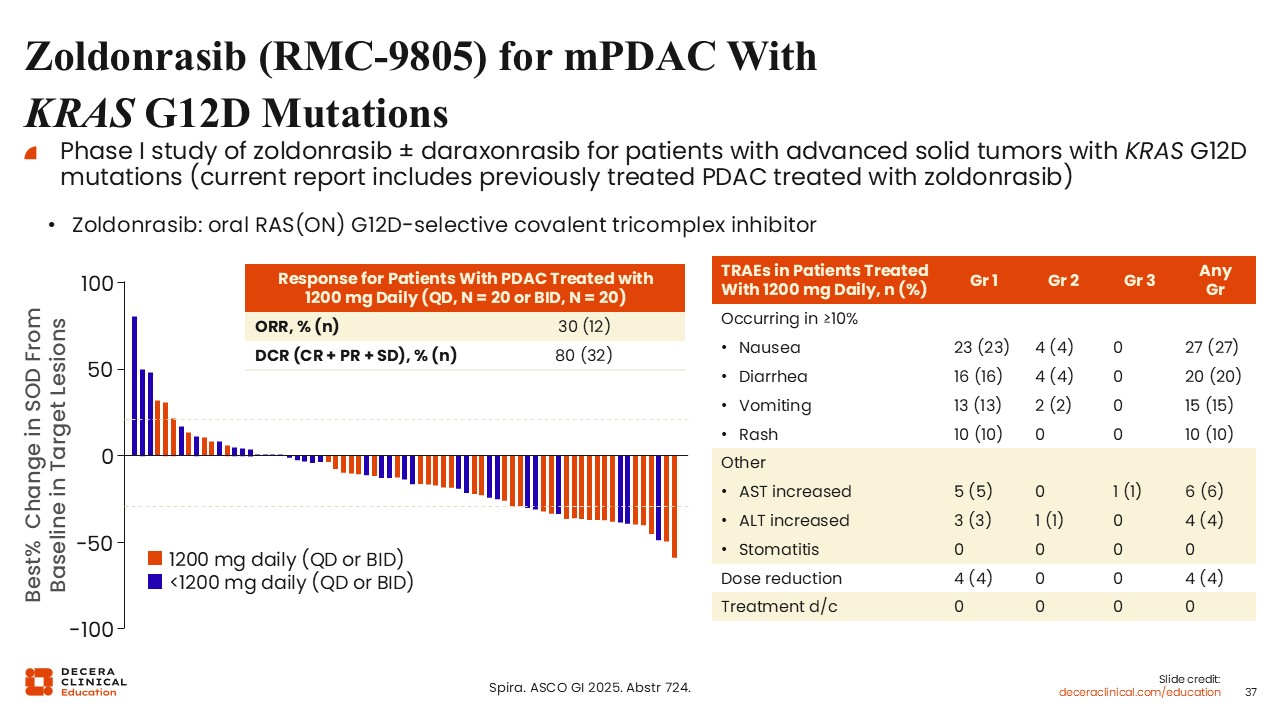
Zoldonrasib (RMC-9805) for mPDAC With KRAS G12D Mutations
Wungki Park, MD, MS: In addition to pan-RAS approaches, allele-specific targeting represents another important strategy. KRAS G12D is the most common mutation in pancreatic cancer and has become a major focus of drug development.
Zoldonrasib is an oral, allele-specific RAS(ON) G12D-selective covalent tricomplex inhibitor that has demonstrated ORRs of 30% and a disease control rate of 80% in an early-phase study in previously treated PDAC with a KRAS G12D mutation.23
Compared with pan-RAS inhibitors, this agent appears to have a more favorable tolerability profile, with predominantly low-grade gastrointestinal AEs and infrequent rash. This highlights a potential trade-off between broader targeting and improved tolerability with more selective agents.
Christopher Lieu, MD, FASCO: These data are compelling. The improved tolerability associated with allele-specific targeting is notable, although it applies to a narrower patient population.
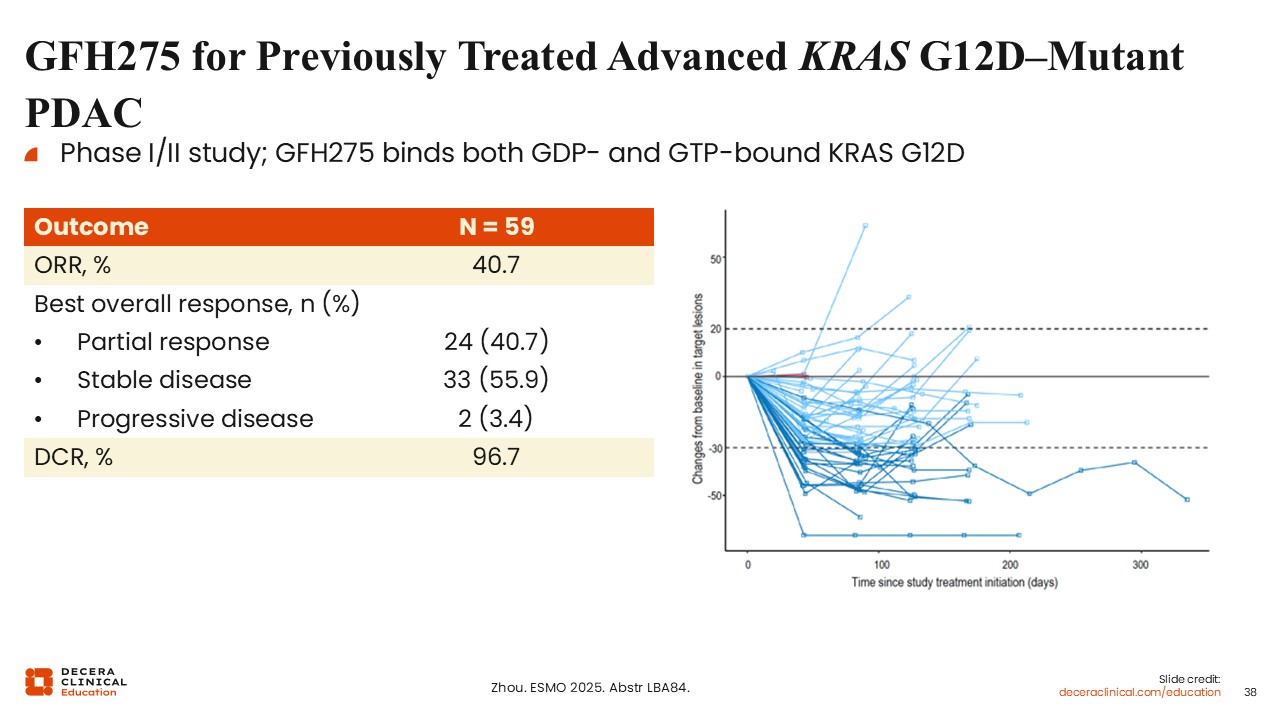
GFH275 for Previously Treated Advanced KRAS G12D-Mutant PDAC
Wungki Park, MD, MS: Additional G12D-targeted agents are in development, including GFH275, an oral agent that binds both GDP- and GTP-bound KRAS G12D.24 Early-phase data suggest response rates in the range of up to 40% for previously treated advanced KRAS G12D-mutant PDAC, although these results should be interpreted cautiously given small sample sizes and variability in patient populations.
Ongoing studies will further define the efficacy and safety of this agent.
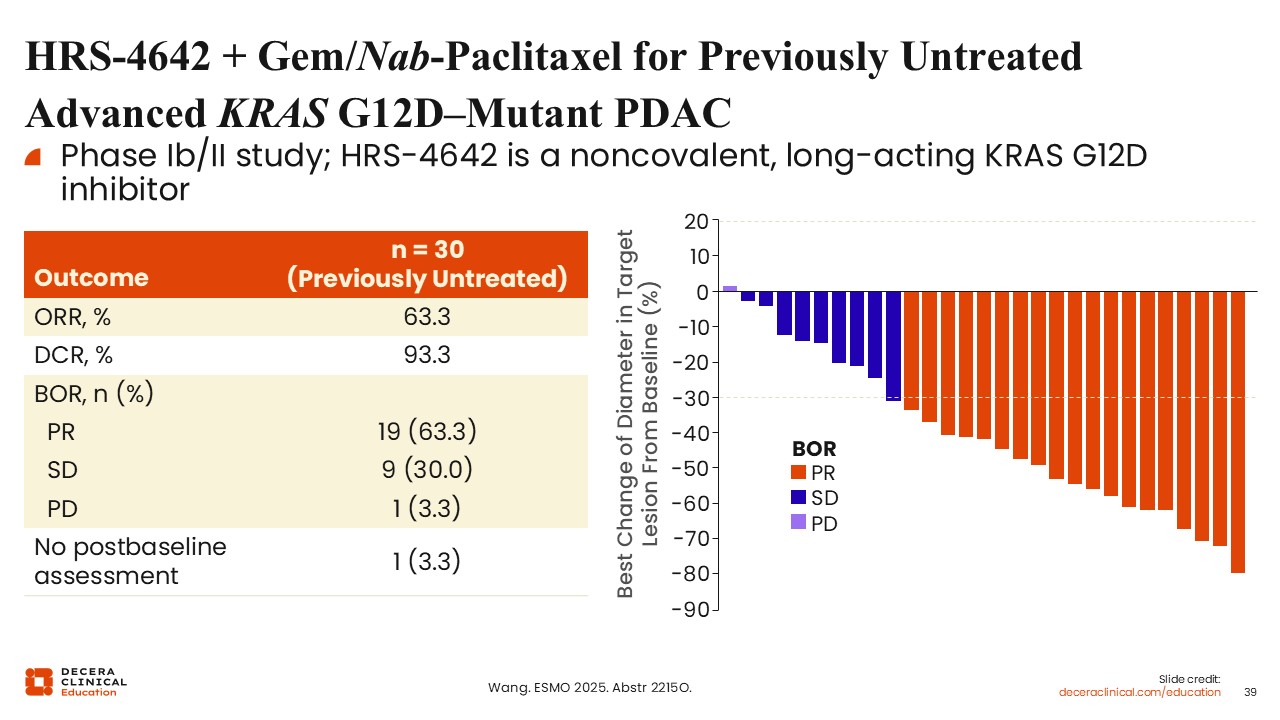
HRS-4642 + Gem/Nab-Paclitaxel for Previously Untreated Advanced KRAS G12D-Mutant PDAC
Wungki Park, MD, MS: HRS-4642 is an intravenous KRAS G12D inhibitor that has been evaluated in combination with gemcitabine and nab-paclitaxel.25 This agent uses a distinct delivery approach and may offer advantages in patients with compromised oral absorption, although further validation is needed.
In early studies, response rates exceeding 60% for previously untreated advanced KRAS-G12D mutant PDAC have been reported. Although these findings are preliminary, they are notable given the historically limited response rates observed in pancreatic cancer.
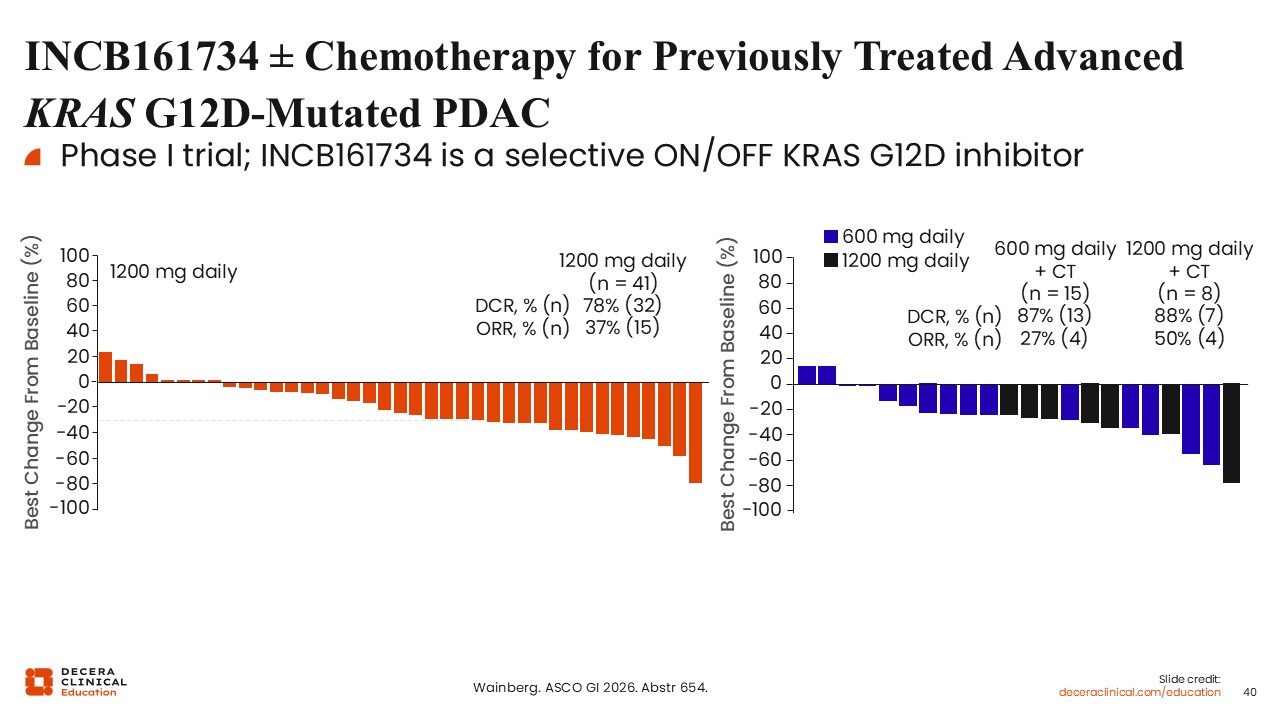
INCB161734 ± Chemotherapy for Previously Treated Advanced KRAS G12D-Mutated PDAC
Wungki Park, MD, MS: INCB16173 is another KRAS G12D inhibitor that has demonstrated activity as monotherapy and in combination with chemotherapy.26
In early-phase studies in previously treated advanced KRAS G12D-mutated PDAC, monotherapy achieved response rates of approximately 37%, increasing to nearly 50% when combined with chemotherapy.
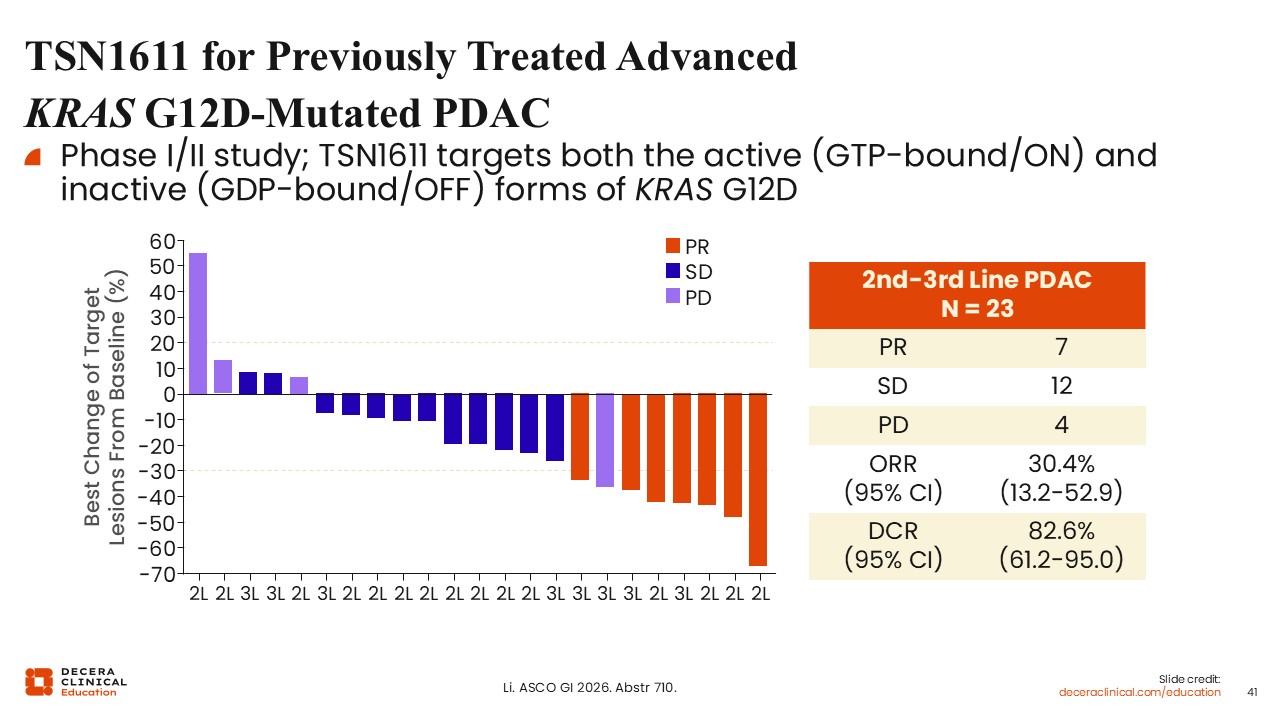
TSN1611 for Previously Treated Advanced KRAS G12D-Mutated PDAC
Wungki Park, MD, MS: TSN1611 is an oral KRAS G12D inhibitor targeting both active and inactive states of the protein.27
Early data in previously treated advanced KRAS G12D–mutated PDAC suggest an ORR of approximately 30%, with ongoing responses observed in some patients. Additional follow-up is needed to assess durability and long-term outcomes.
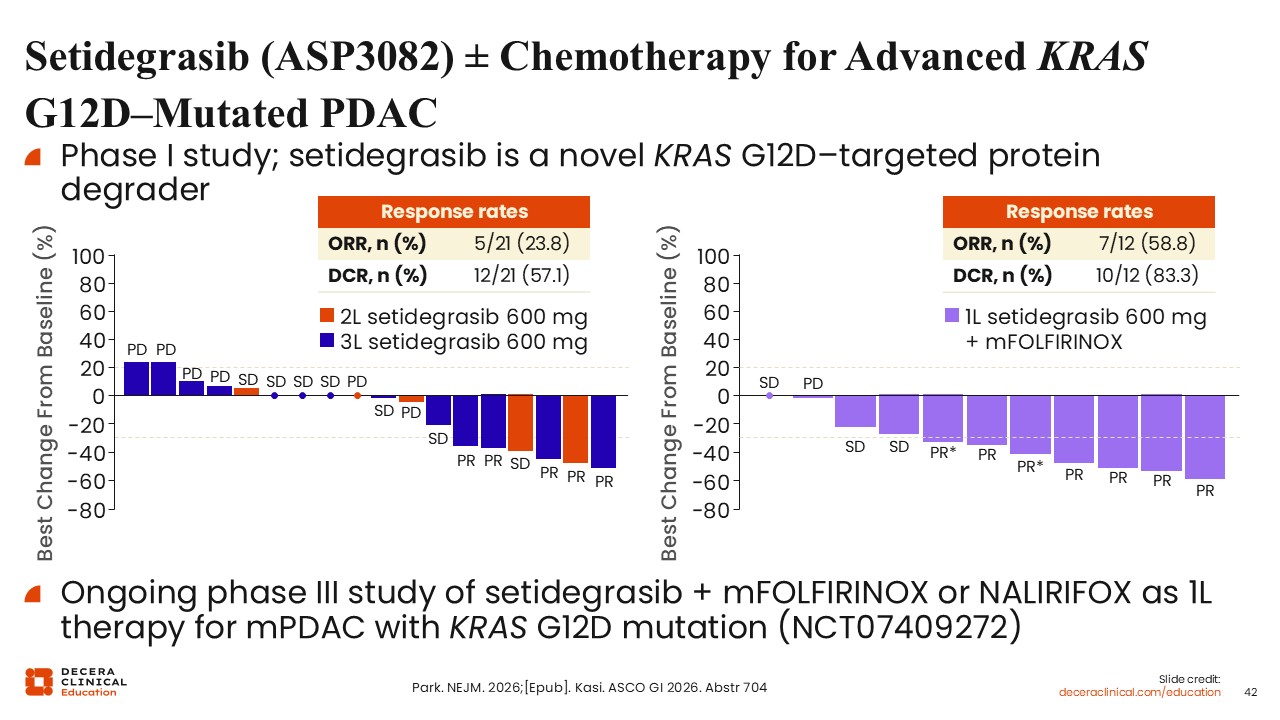
Setidegrasib (ASP3082) ± Chemotherapy for Advanced KRAS G12D-Mutated PDAC
Wungki Park, MD, MS: In addition to direct inhibition, alternative strategies for targeting KRAS are emerging, including approaches that induce degradation of the KRAS protein.
These methods leverage cellular mechanisms to label KRAS as a protein for degradation, thereby reducing its activity and downstream signaling.
Wungki Park, MD, MS: Setidegrasib (ASP3082) is a KRAS G12D–targeted protein degrader that has demonstrated early clinical activity.28 In dose-escalation studies, dose-dependent plasma exposure and corresponding KRAS protein degradation have been observed. This mechanism functions by selectively targeting KRAS for removal, effectively reducing oncogenic signaling.
Single-agent activity has been reported, with response rates of approximately 24% in heavily pretreated populations, including most patients in the third-line setting. This agent has generally been well tolerated, with infusion-related reactions representing the most notable AE.
Combination strategies with chemotherapy have shown increased response rates. Setidegrasib is currently being evaluated in an ongoing phase III trial with mFOLFIRINOX or NALIRIFOX as first-line therapy for mPDAC with a KRAS G12D mutation (NCT07409272).
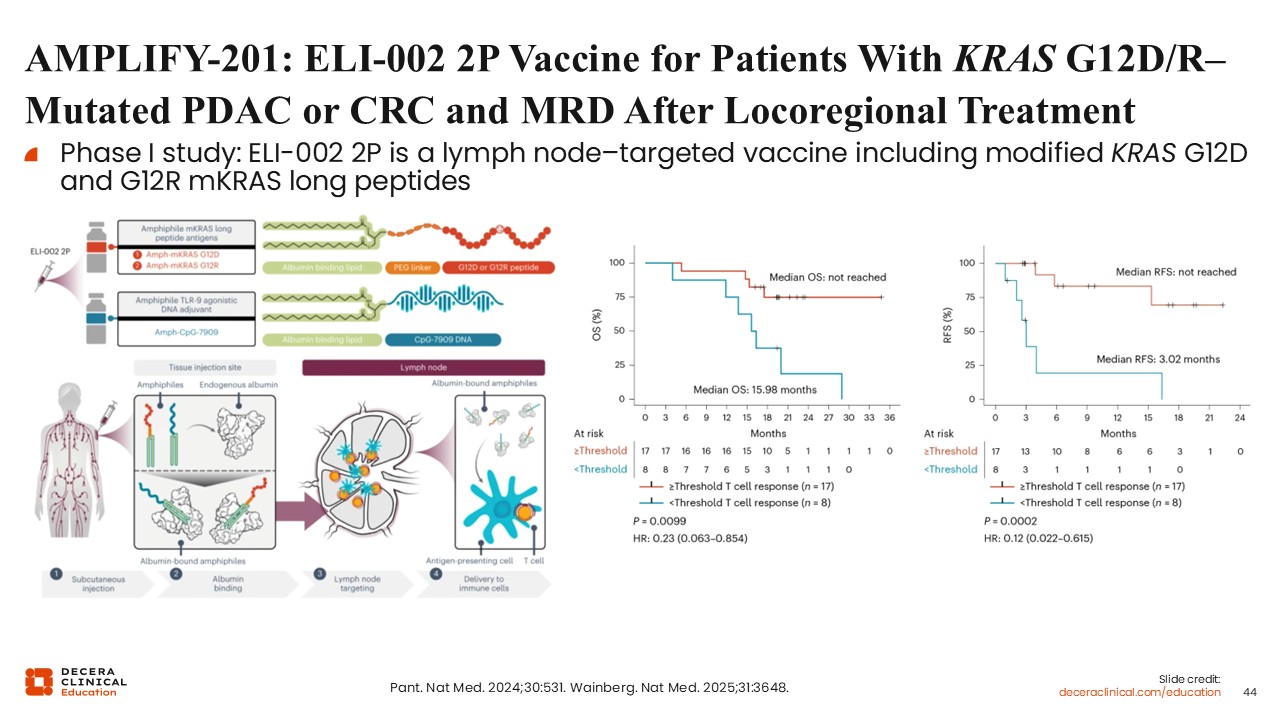
AMPLIFY-201: ELI-002 2P Vaccine for Patients With KRAS G12D/R–mutated PDAC or CRC and MRD After Locoregional Treatment
Wungki Park, MD, MS: Beyond targeted therapies, there is increasing interest in leveraging the immune system to address KRAS-driven cancers.
KRAS-directed peptide vaccines represent 1 such approach. These vaccines are designed to stimulate T-cell responses against specific KRAS mutations, such as G12D and G12R.29,30
Early clinical data from small cohorts suggest that patients who develop measurable T-cell responses may also demonstrate improved recurrence-free survival. Expanded vaccine constructs incorporating multiple KRAS mutations are under development to broaden applicability.
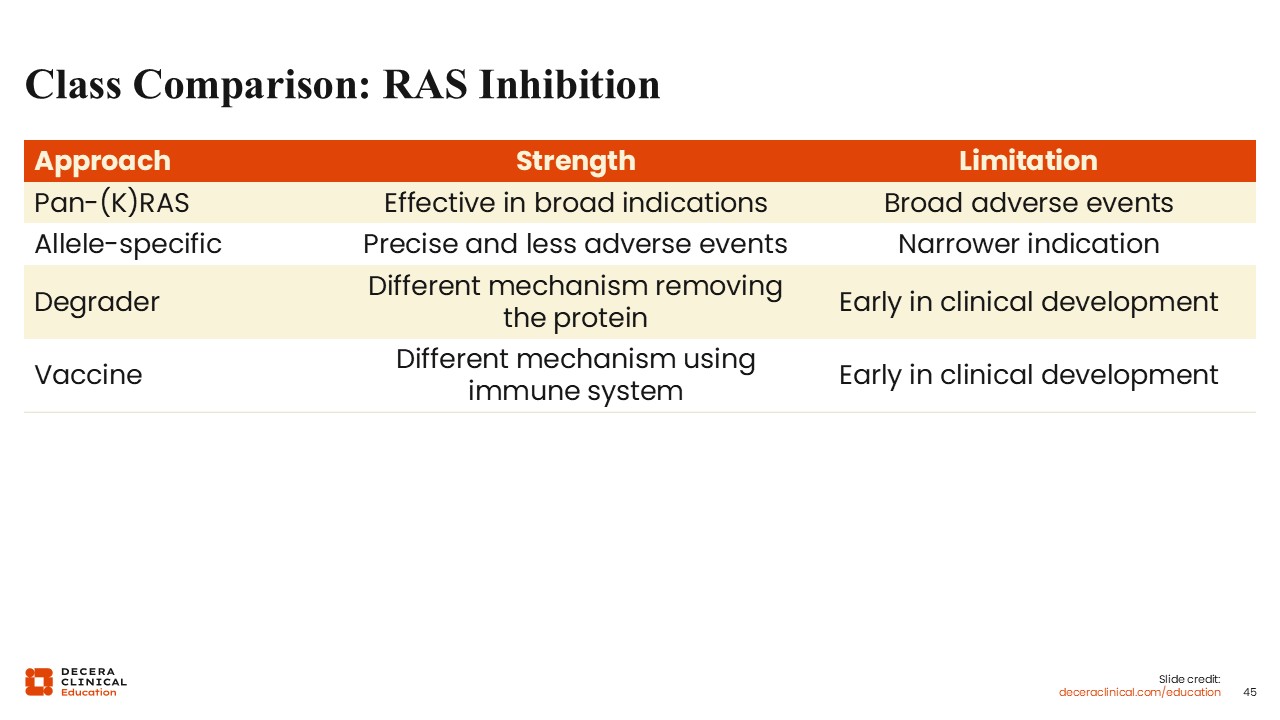
Class Comparison: RAS Inhibition
Wungki Park, MD, MS: In summary, multiple classes of KRAS-targeted therapies are under investigation, each with distinct advantages and limitations.
Pan-RAS inhibitors offer broader applicability across patient populations but may be associated with higher rates of toxicity. In contrast, allele-specific inhibitors provide more selective targeting with potentially improved tolerability but are limited to specific mutation subtypes.
Additional strategies, including protein degraders and immunotherapeutic approaches, further expand the therapeutic landscape.
A combination of these approaches may ultimately be required, with broader targeting strategies potentially used initially, followed by more selective therapies as the field evolves.
Christopher Lieu, MD, FASCO: These data highlight the rapid evolution of KRAS-targeted therapies, with multiple agents advancing through clinical development and several phase III trials expected to report results soon. As these therapies become available, healthcare professionals will need to become familiar with their use, including management of associated toxicities, to ensure patients can remain on treatment and derive maximal benefit. There will also be a need to disseminate knowledge broadly so that patients across different practice settings can access these therapies safely.