CE / CME
MASH Matters: New Evidence and Its Application to Endocrinology and Primary Care Practice
Physician Assistants/Physician Associates: 0.50 AAPA Category 1 CME credit
Nurse Practitioners/Nurses: 0.50 Nursing contact hour
Physicians: maximum of 0.50 AMA PRA Category 1 Credit™
Released: August 01, 2025
Expiration: July 31, 2026
Activity
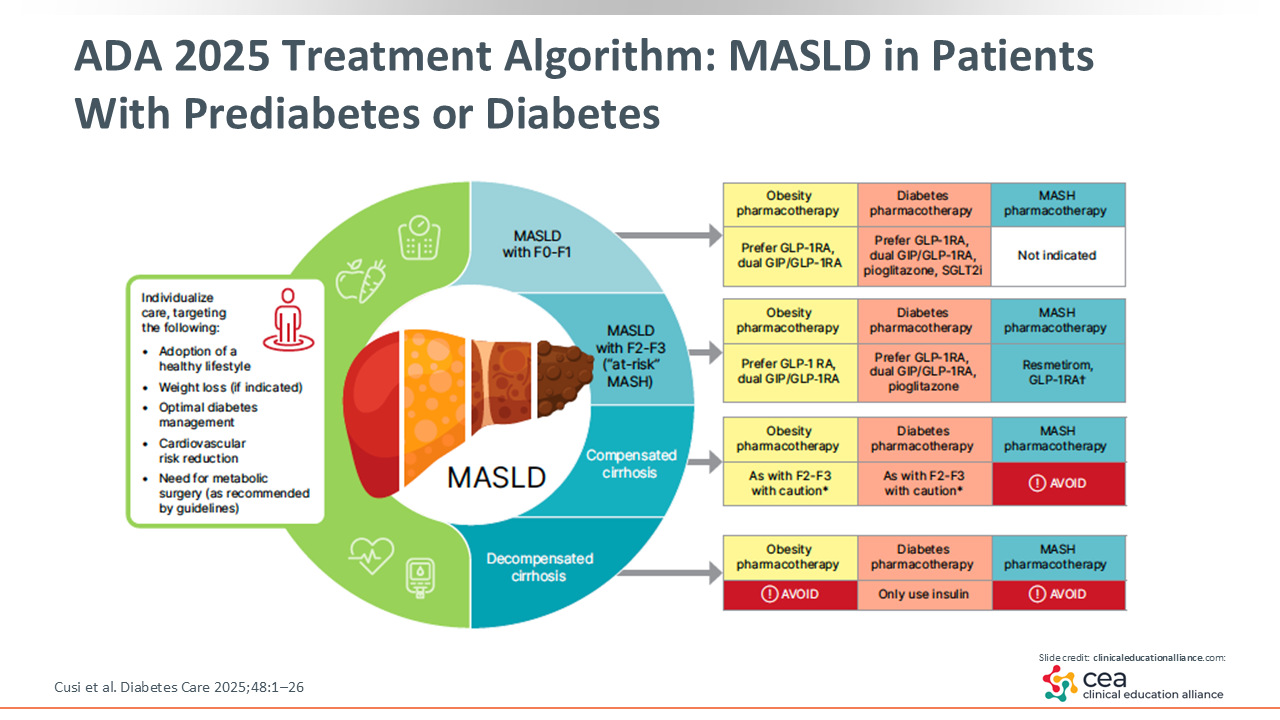
ADA 2025 Treatment Algorithm: MASLD in Patients With Prediabetes or Diabetes
This slide is important. We developed this figure in the 2025 ADA consensus report with a group of experts, including hepatologists, primary care providers, obesity specialists, diabetes educators, nutritionists, and more, to simplify the management of patients with MASLD and prediabetes or diabetes.
Listed on the left are the practical interventions HCPs should use for patients with insulin resistance syndrome, metabolic syndrome, or T2D, including adopting a healthy lifestyle, losing weight when indicated, optimizing diabetes care, reducing CV risk, and referring to metabolic surgery.
The care of patients with prediabetes or diabetes should be managed based on their liver health status. MASLD with no fibrosis is F0 and MASLD with a low degree of fibrosis is F1—there is mild fibrosis that may wax and wane. In these cases, HCPs should consider obesity and diabetes pharmacotherapy to treat or prevent steatosis with accompanying weight loss. HCPs can use a GLP-1 receptor agonist or dual GIP/GLP-1 receptor agonist like tirzepatide. Then there is a PPAR agonist called pioglitazone that has a proven and significant effect on improving fibrosis in clinical trials.15 Of more importance, it is a strong agent that can potentially reverse steatohepatitis. These agents are not strictly indicated for MASH yet because phase III studies are currently underway.
F2 or F3 fibrosis is called at-risk MASH or at-risk for future cirrhosis/MASH. Like F0/1, HCPs should use the same obesity and diabetes pharmacotherapy strategies indicated for F2/3 fibrosis, except for SGLT2 inhibitors. These therapies are not recommended for patients with F2/3 fibrosis because there are no clinical trial data with liver biopsies that show a benefit with SGLT2 inhibitor use. Furthermore, these are the patients for which MASH pharmacotherapy, such as resmetirom, is recommended.
Once patients develop cirrhosis, the management of compensated cirrhosis specifically is quite similar to that of F2/3 fibrosis in terms of obesity or diabetes pharmacotherapy, yet MASH pharmacotherapy with resmetirom or other agents is not recommended. This is because we do not have agents that have completed phase III trials in patients with cirrhosis. And finally, for decompensated cirrhosis, HCPs should concentrate on diabetes pharmacotherapy because there are no data yet on the use of other interventions in this population.2
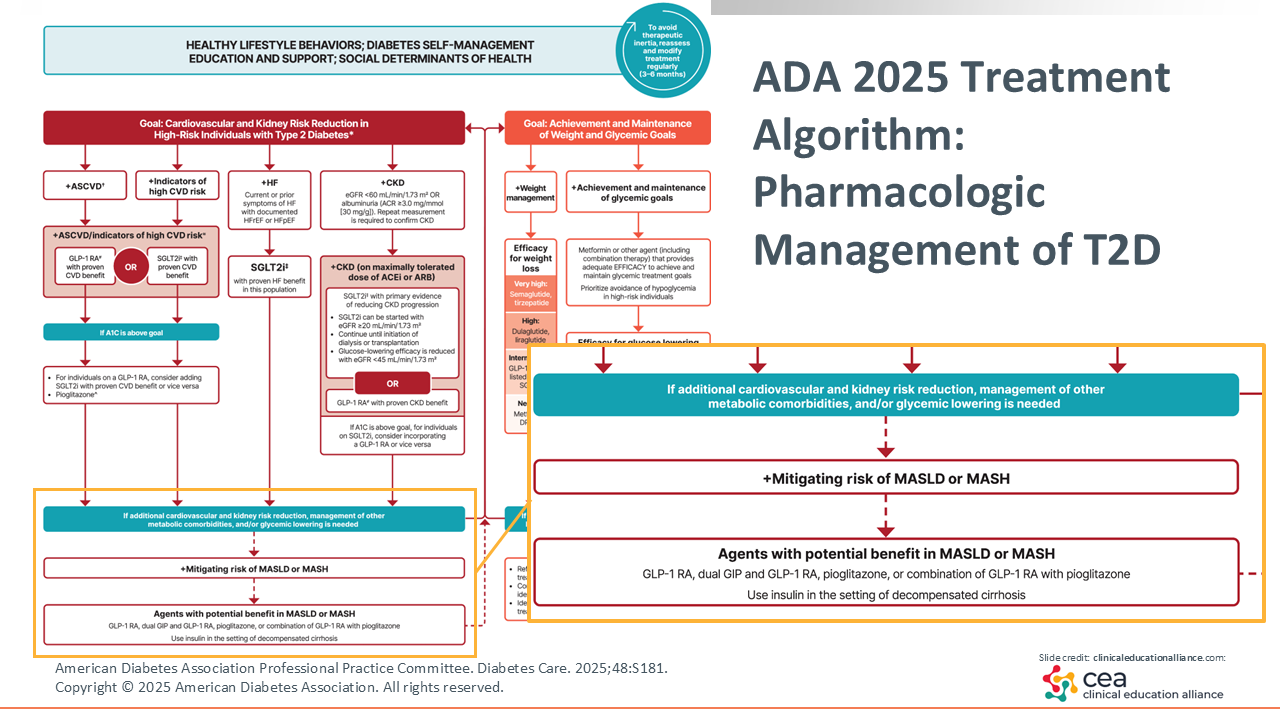
ADA 2025 Treatment Algorithm: Pharmacologic Management of T2D
This year, the 2025 ADA treatment algorithm for the pharmacologic management of T2D added MASLD/MASH as disease states that deserve specific treatment considerations.16
HCPs should be familiar with semaglutide’s ability to reduce CVD, SGLT2 inhibitors’ indications in heart failure, and the recent evidence on GLP-1 receptor agonist efficacy in chronic kidney disease.17,18 MASH is something that HCPs should screen for in patients with T2D because it will inform their diabetes management strategies with semaglutide, tirzepatide, pioglitazone, or a combination of GLP-1 receptor agonism with pioglitazone. This is something that has broad implications.
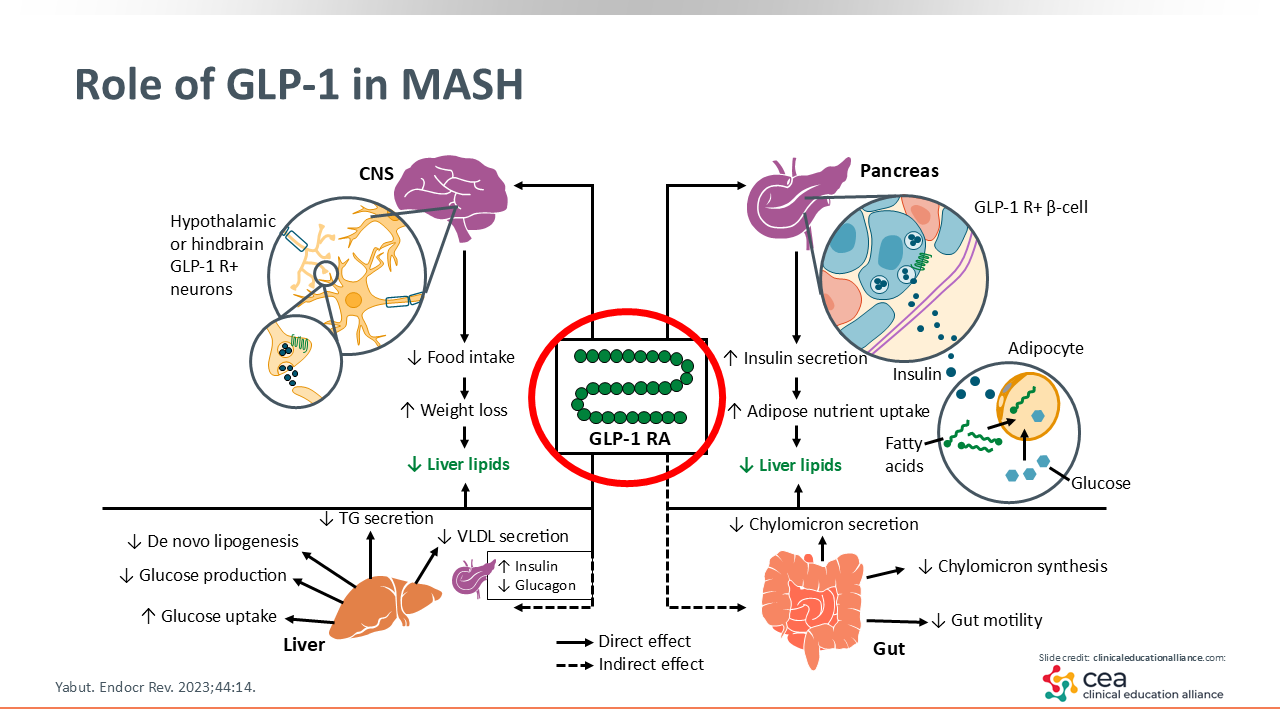
Role of GLP-1 in MASH
You may be familiar with the role of GLP-1 in general and its mechanisms of action. We know it has central nervous system effects, such as decreasing food intake, which directly impacts weight loss. Clearly this class of agents, as well as the dual GIP/GLP-1 receptor agonist tirzepatide, comes with other added benefits. They can enhance insulin secretion, improve a number of lipid and glucose pathways in the liver, and affect the gut and its microbiome, which are not yet completely understood.19
ESSENCE: Semaglutide in Patients With MASH
One study that is extremely important is the ESSENCE trial (NCT04822181). This multicenter, double-blind, placebo-controlled, randomized phase III trial evaluated the use of weekly semaglutide 2.4 mg vs placebo in adults with MASH and F2/3 fibrosis. It enrolled 1205 patients and is ongoing. An interim analysis was presented late last year and is now published in the New England Journal of Medicine. The interim analysis evaluated outcomes at Week 72 in the first 800 patients to complete treatment.
The primary endpoints for the interim analysis are steatohepatitis resolution (no significant inflammation on biopsy) without worsening fibrosis and improvement in fibrosis without worsening steatohepatitis. There also are several secondary endpoints, including safety.20
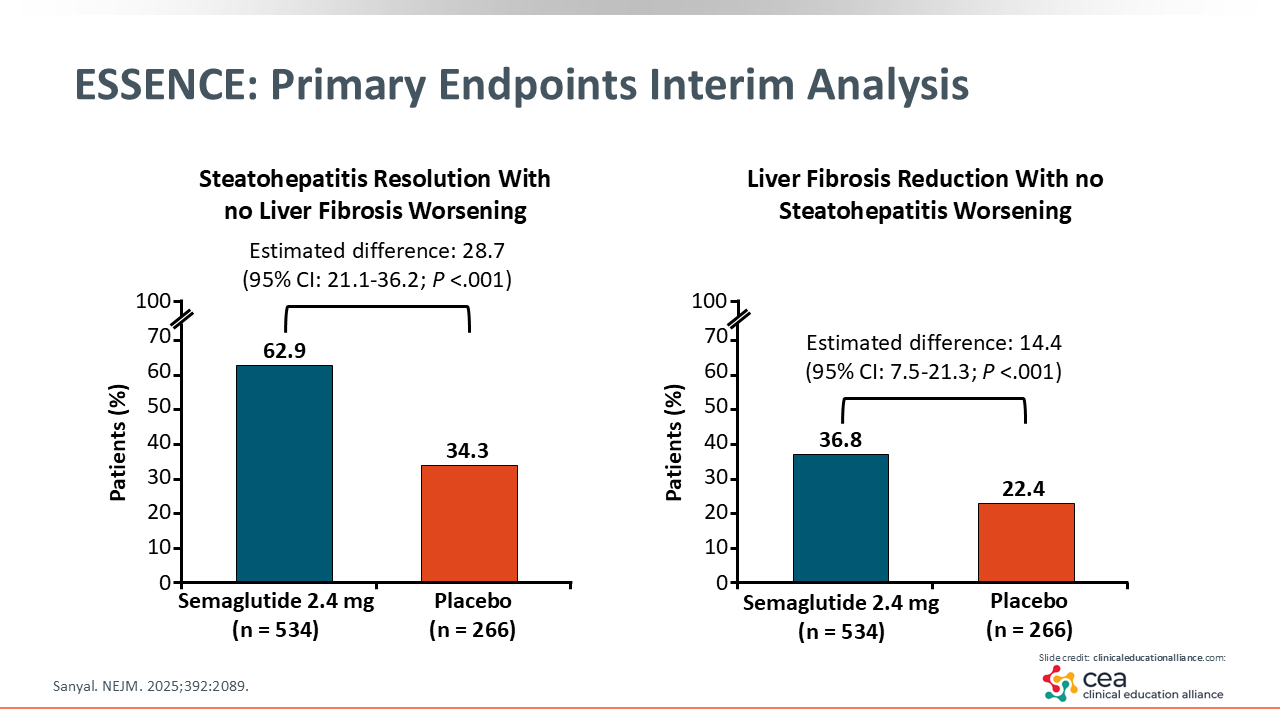
ESSENCE: Primary Endpoints Interim Analysis
This slide summarizes the key message of the ESSENCE trial. On the left is steatohepatitis resolution, which improved in almost two thirds (62.9%) of the patients in the semaglutide arm vs a third (34.3%) of patients in the placebo arm. How can placebo do so well? This tells me there is some importance regarding the interaction between the HCPs and patients in this arm. They likely were told to eat better and increase their physical activity—all aspects that help reverse an unfavorable cardiometabolic profile—which have an impact on the liver.
The figure on the right is critical because historically it has been hard to reverse liver scarring or fibrosis in patients with MASH. Remember, patients recruited in this study had moderate to advanced fibrosis. We can see that approximately one third (36.8%) of patients had improved fibrosis with semaglutide vs 22.4% of patients with placebo. Both outcomes were significant, and this study is being reviewed by the FDA to consider an approval in MASH for semaglutide.20
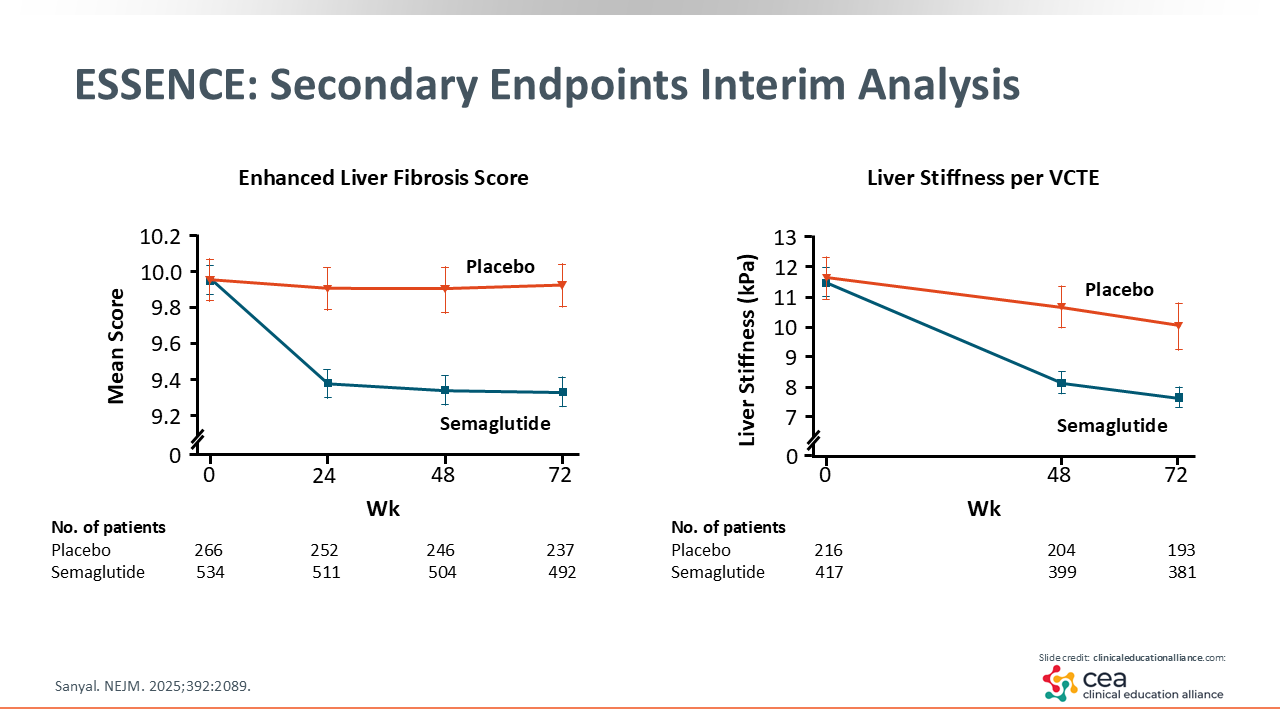
ESSENCE: Secondary Endpoints Interim Analysis
As shown in the figures on this slide, these 2 noninvasive tests are important because HCPs are going to use them in the clinic. ELF scores improved significantly with semaglutide, as baseline scores were initially near 10 and 9.8 is the cutoff for significant liver disease. The figure on the right shows LSM via VCTE, which also improved more with semaglutide vs placebo.20
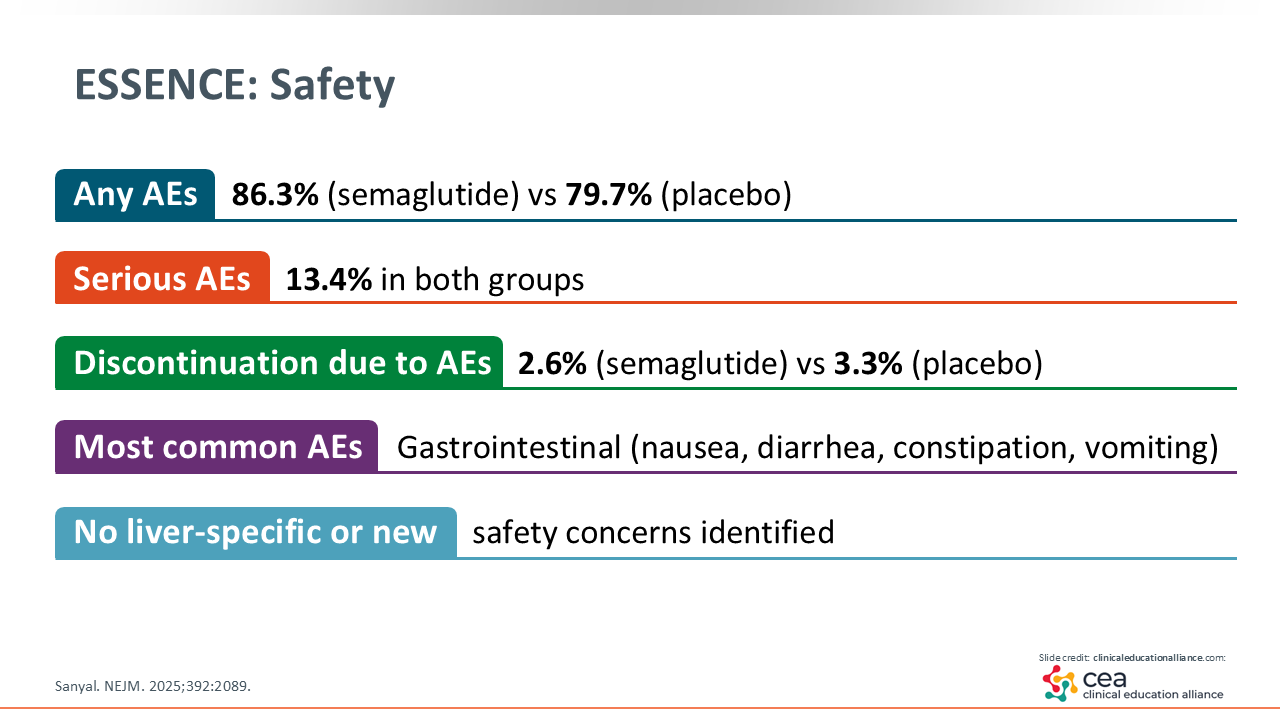
ESSENCE: Safety
The reported adverse events (AEs) are similar to previous studies of semaglutide. The discontinuation rate due to AEs was similar across both arms: 2.6% with semaglutide vs 3.3% with placebo. Most AEs were gastrointestinal related and no new safety concerns were raised.20
ESSENCE: Interim Analysis Conclusions
The bottom line: weekly semaglutide 2.4 mg led to significant histologic improvement in patients with MASH vs placebo. Semaglutide also has indications to reduce the risk of sustained eGFR decline, end-stage kidney disease, and CV death in patients with diabetes and chronic kidney disease and to reduce risk of major adverse CV events in patients with CVD and diabetes, overweight, or obesity.21,22 Furthermore, it is quite safe and a promising treatment option for MASH.20
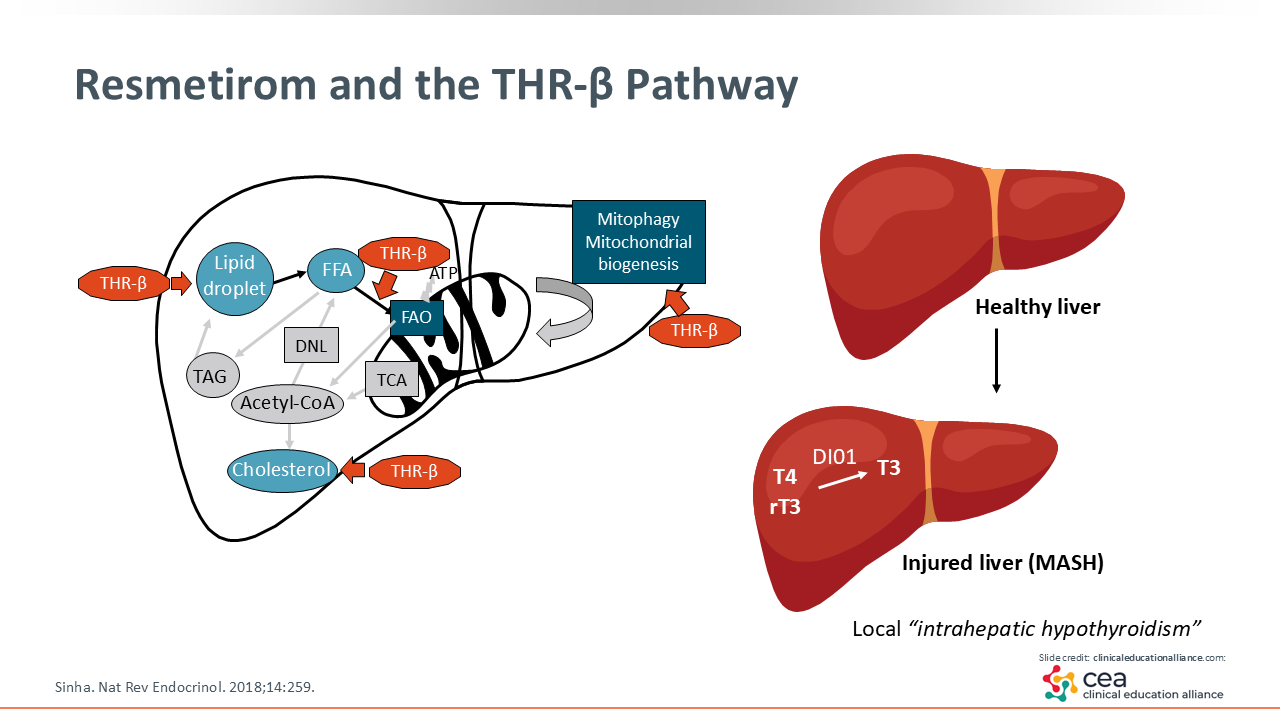
Resmetirom and the THR-β Pathway
Shifting gears, let us discuss resmetirom, which is a thyroid hormone receptor (THR)-β–selective agonist. There are 2 main thyroid hormone receptors in our body: THR-α and THR-β. They are abundant in the heart and bone, with THR-β more specifically present in the liver.23 Furthermore, it might improve mitochondrial function in how it burns fat and it might improve lipid levels.24 In the MAESTRO-NASH trial, resmetirom lowered low-density lipoprotein cholesterol by approximately 20%.25
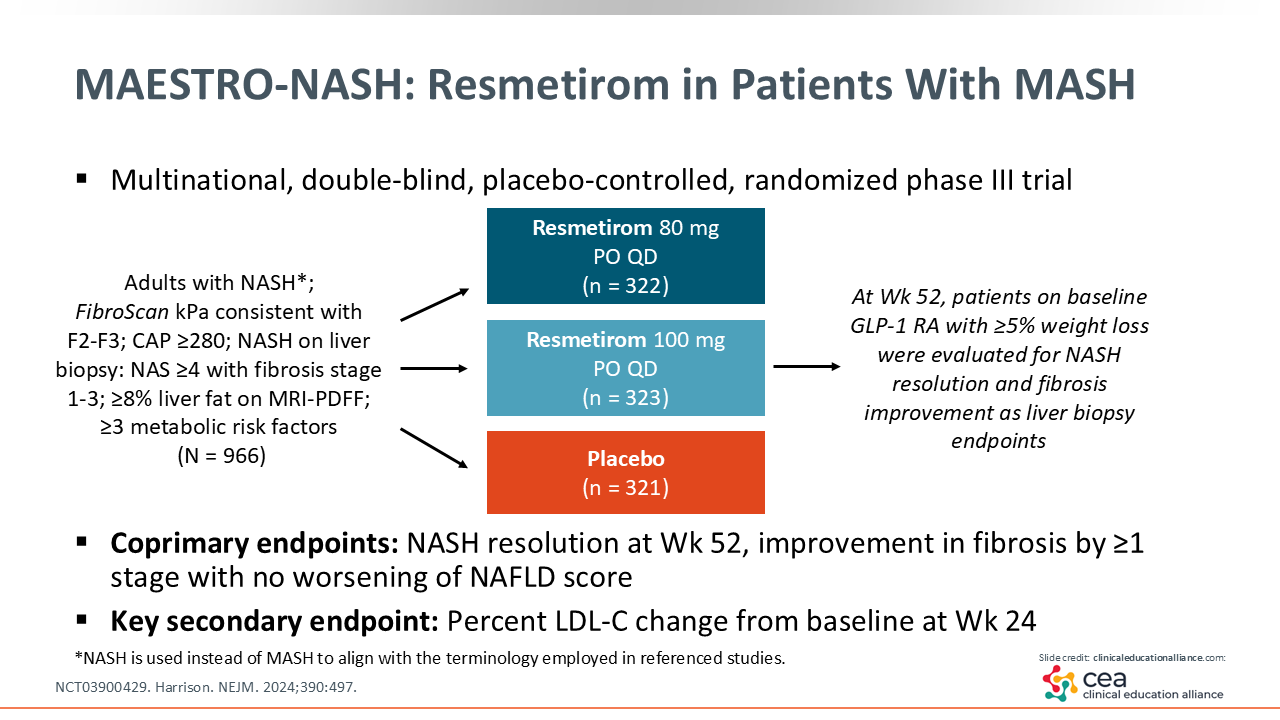
MAESTRO-NASH: Resmetirom in Patients With MASH
The phase III MAESTRO-NASH (NCT03900429) trial enrolled 966 adults with MASH and F2/3 fibrosis who were randomized to receive resmetirom 80 mg or 100 mg vs placebo for 52 weeks. This also is an ongoing 5-year trial like ESSENCE, looking at liver outcomes. Here, the coprimary endpoints are NASH resolution and fibrosis improvement.25
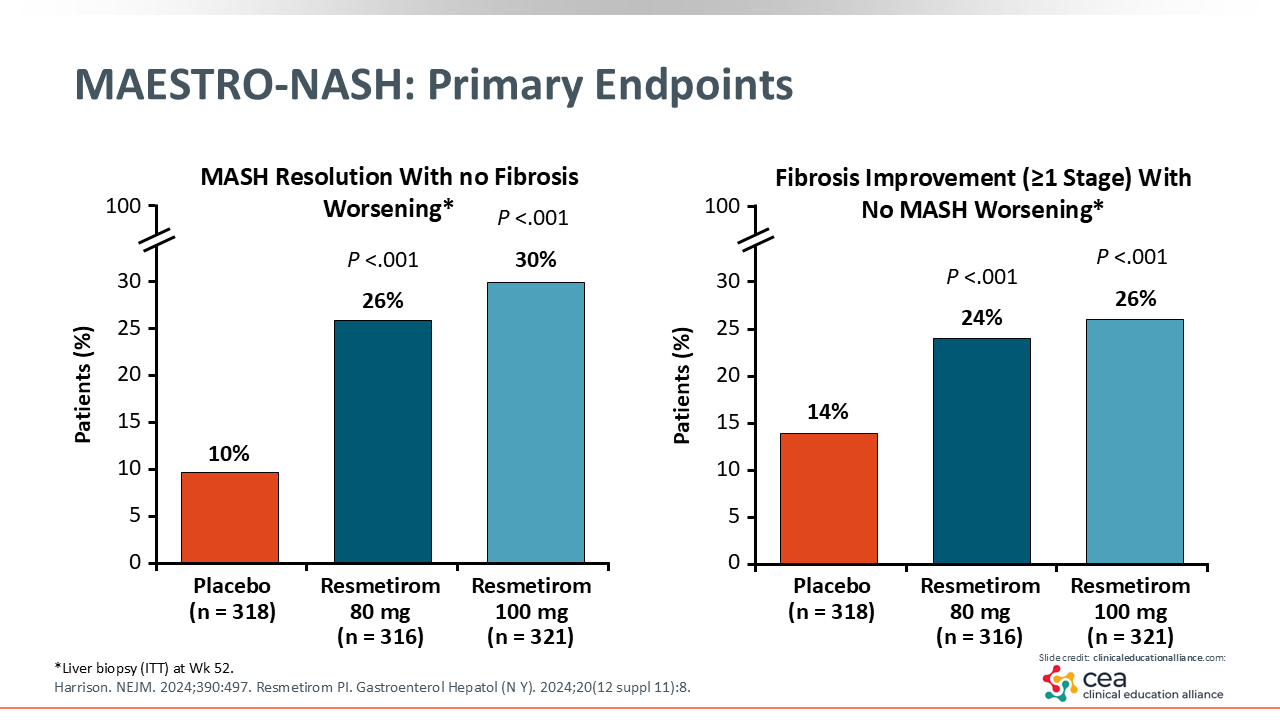
MAESTRO-NASH: Primary Endpoints
The left figure shows the results of MASH resolution: 10% with placebo vs 26% with resmetirom 80 mg and 30% with resmetirom 100 mg. Fibrosis improvement is shown on the right, with 14% in the placebo arm vs 24% to 26% in the resmetirom arms achieving this outcome. The delta effect on MASH resolution was approximately 20%, which is a little bit more of a benefit than with semaglutide. The delta effect on fibrosis improvement was approximately 14%.25
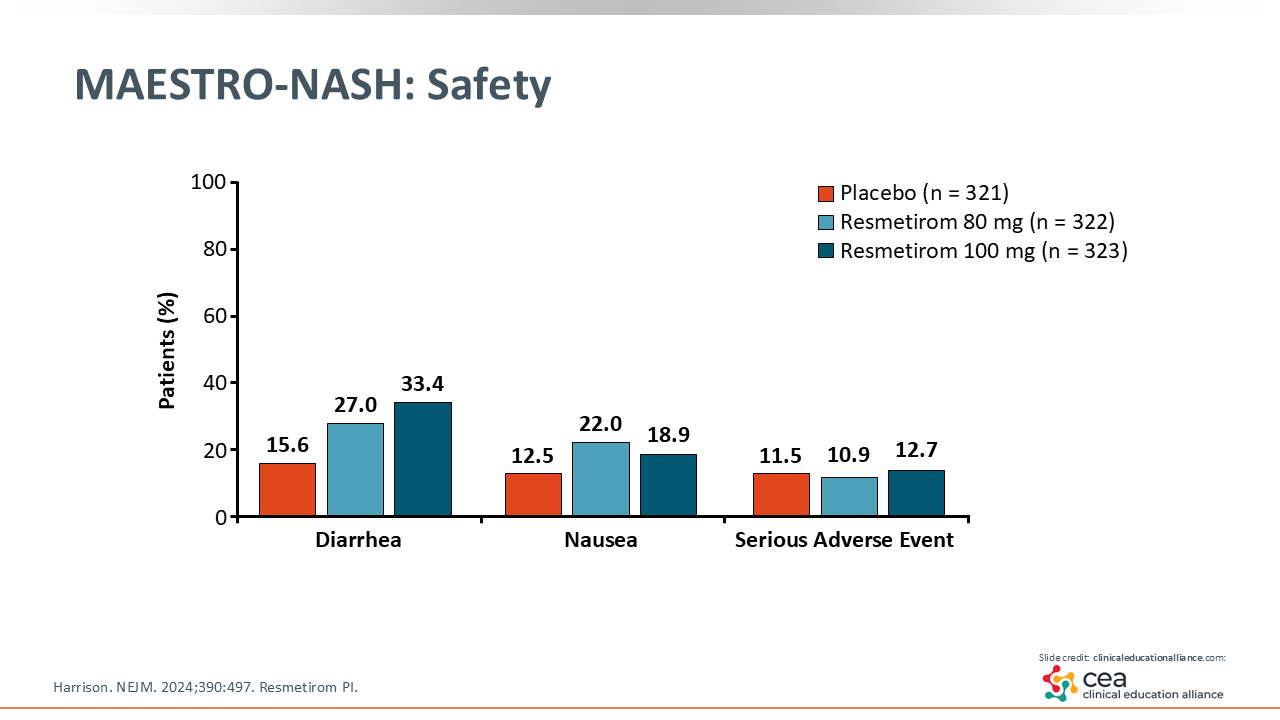
MAESTRO-NASH: Safety
In terms of safety, patients reported milder diarrhea with resmetirom that tends to go away with time.25 Furthermore, the American Association for the Study of Liver Diseases has guidelines on the management of MASH with resmetirom.26 It is vital that HCPs consider the fact that this agent is expensive when prescribing it. To ensure health insurance coverage, patients must have MASH and F2 or F3 fibrosis, which is the approved indication.27
Ensuring coverage of resmetirom can be difficult for endocrinologists to do with noninvasive testing. Therefore, if you have optimized patients’ obesity and/or diabetes management with GLP-1 receptor agonists, pioglitazone, or a combination of the 2, then you should talk to your local gastroenterologist to consider resmetirom therapy if patients’ fibrosis is still moderate to advanced (F2/3). In addition, patients with hypothyroidism or hyperthyroidism were not included in the MAESTRO-NASH trial, so HCPs should ensure patients have normal thyroid function before starting resmetirom.
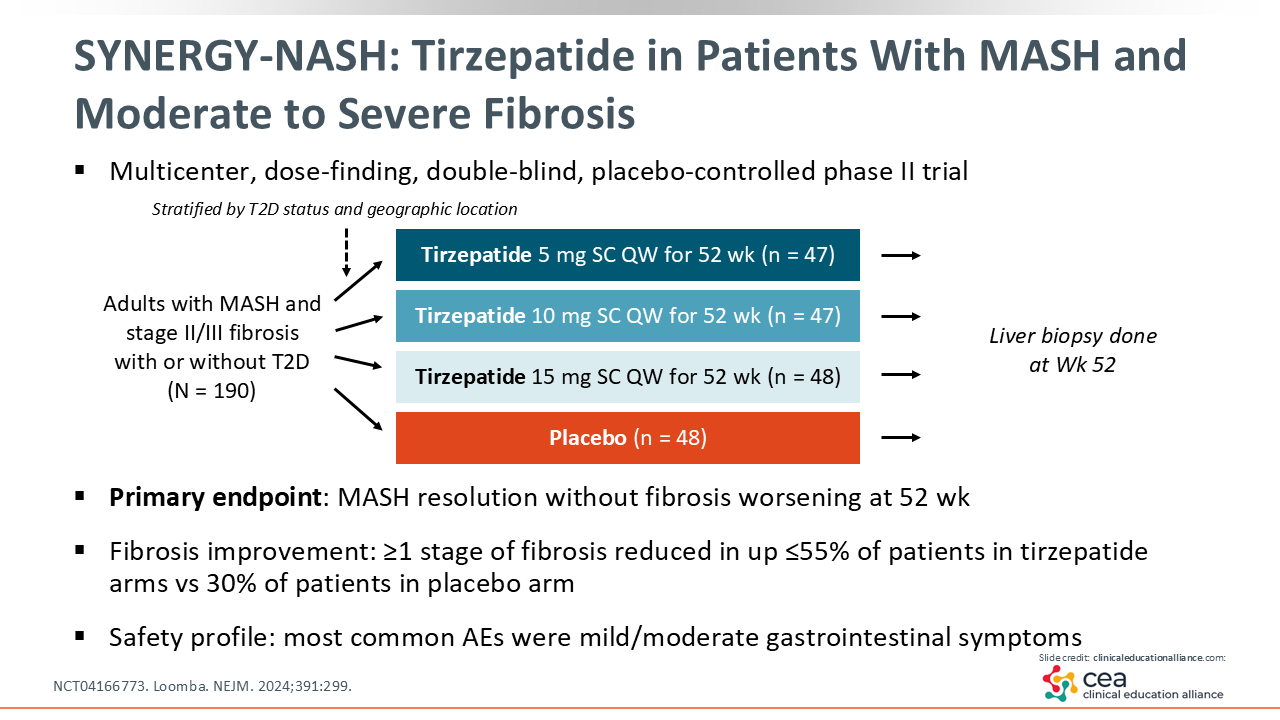
SYNERGY-NASH: Tirzepatide in Patients With MASH and Moderate to Severe Fibrosis
Next is the phase II SYNERGY-NASH (NCT04166773) trial that evaluated tirzepatide vs placebo in adults with MASH and moderate to severe fibrosis (F2/3). The 3 treatment arms were tirzepatide 5 mg, 10 mg, and 15 mg vs placebo. This was a smaller study, only enrolling 190 patients who were treated for 52 weeks.28
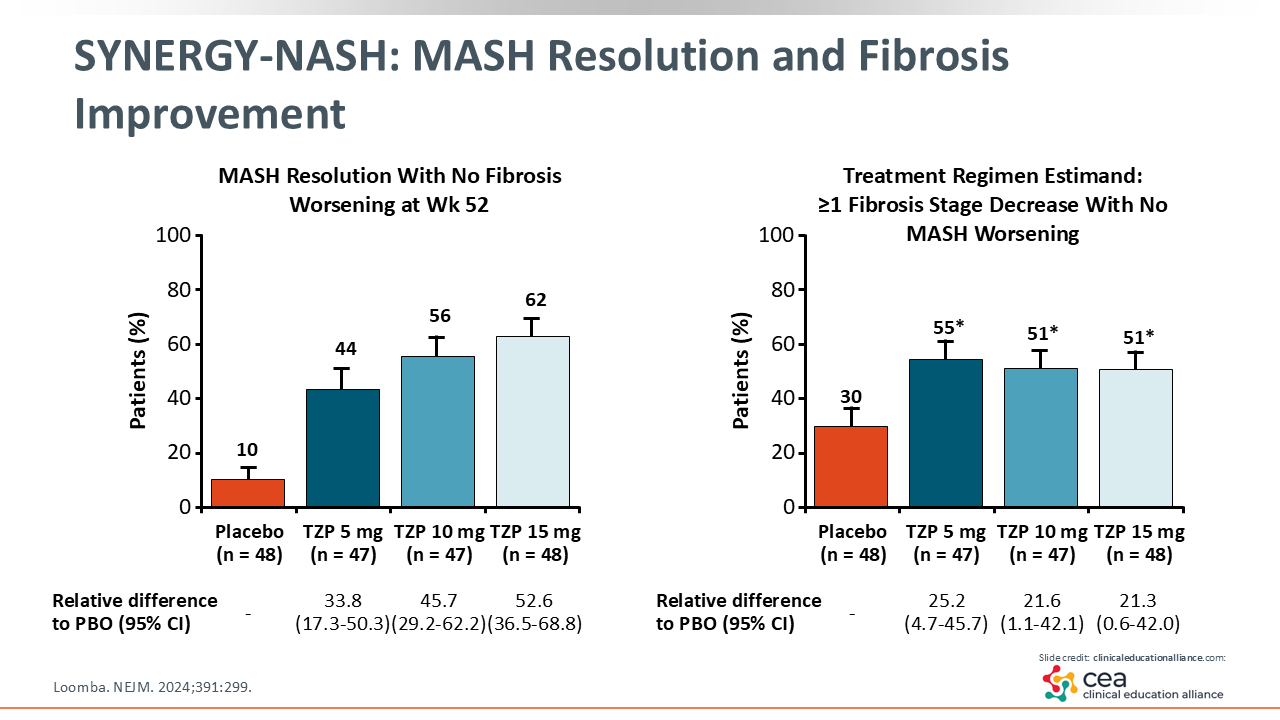
SYNERGY-NASH: MASH Resolution and Fibrosis Improvement
In this trial, MASH resolution improved significantly from 10% with placebo vs 62% with tirzepatide 15 mg. Similar efficacy was seen across all tirzepatide arms in terms of the proportion of patients who had MASH resolution. Furthermore, the lower placebo response rate makes for a greater delta or treatment difference. On the right, you can see there was a 30% improvement in fibrosis with placebo vs 51% to 55% with tirzepatide, resulting in a +20% difference in fibrosis improvement.28
A phase III study for tirzepatide in MASH should start soon, which is great news for the field.
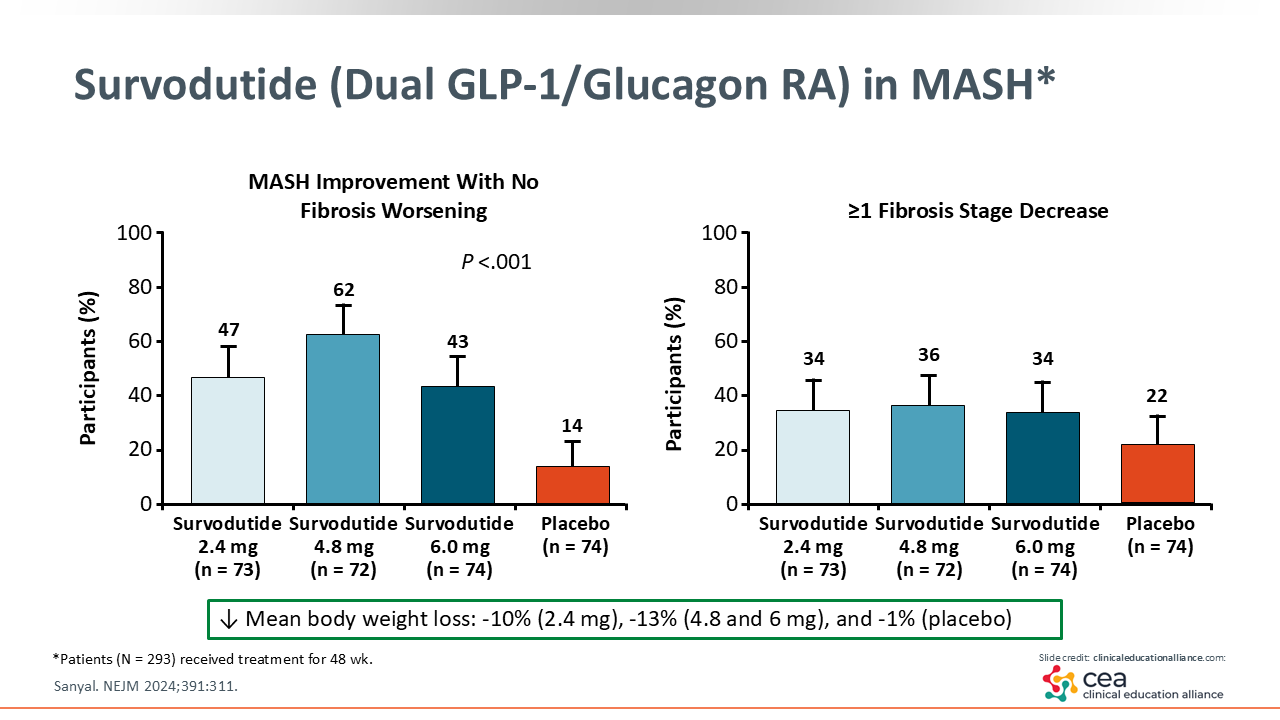
Survodutide (Dual GLP-1/Glucagon RA) in MASH*
Finally, we have the dual GLP-1/glucagon receptor agonist survodutide. This phase II trial reported a mean weight loss between 10% and 13% with survodutide vs 1% with placebo. In addition, 62% of patients saw MASH improvement with survodutide 4.8 mg vs 14% with placebo. These patients were treated for a similar amount of time as in the prior MASH trials. There was some fibrosis improvement in 34% to 36% of patients treated with survodutide compared with 22% treated with placebo.29 These data are very exciting, and a phase III trial is ongoing.
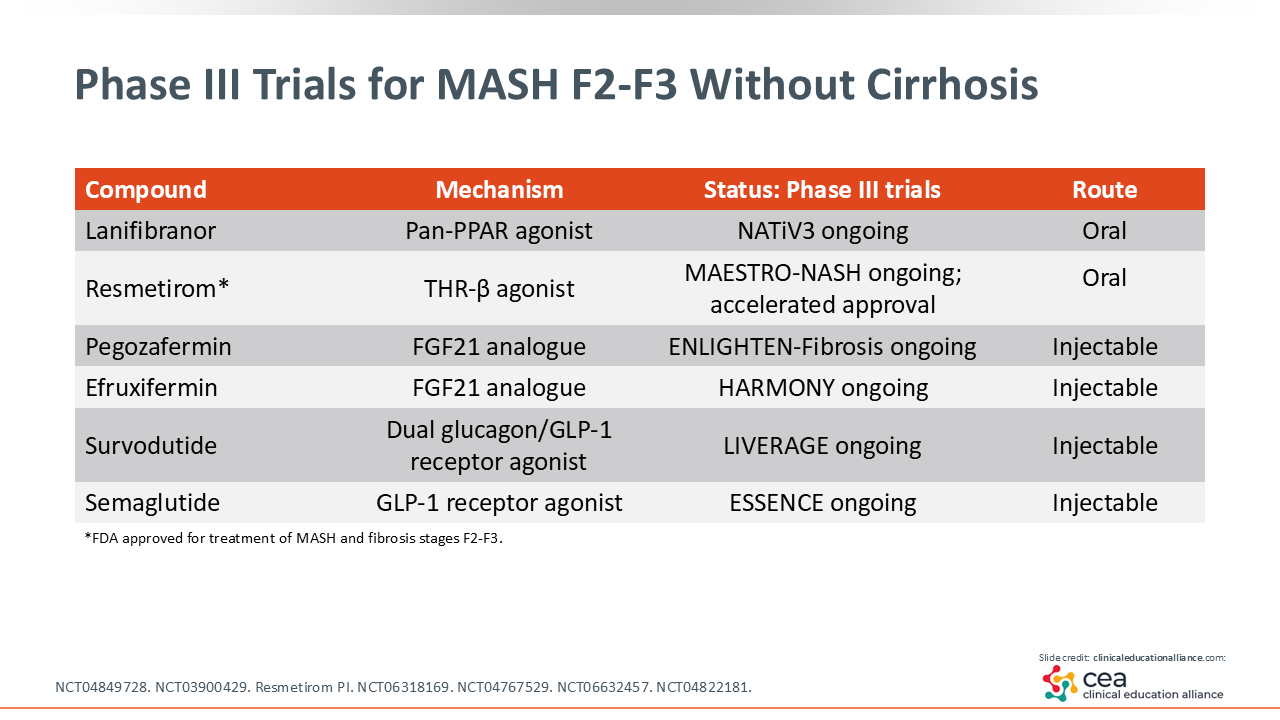
Phase III Trials for MASH F2-F3 Without Cirrhosis
There are many more clinical trials that HCPs should be aware of. I have already mentioned survodutide, semaglutide, and resmetirom. Lanifibranor is a pan-PPAR agonist being evaluated in the phase III NATiV3 study (NCT04849728). This is an agent that can decrease inflammation in the liver.30 Then there are 2 FGF21 analogues (ie, pegozafermin, efruxifermin) being studied, with early data looking promising.31
Phase III Trials for MASH Cirrhosis
There also are several phase III trials for MASH cirrhosis, again, including resmetirom and survodutide. There are some preliminary data with FGF21 analogues for cirrhosis that are exciting.31 I am looking forward to the results of these trials.