CE / CME
Bispecific Antibodies in Relapsed/Refractory Follicular Lymphoma: Efficacy, Toxicities, and Practical Implementation
Physician Assistants/Physician Associates: 0.75 AAPA Category 1 CME credit
Pharmacists: 0.75 contact hour (0.075 CEUs)
Physicians: maximum of 0.75 AMA PRA Category 1 Credit™
Nurse Practitioners/Nurses: 0.75 Nursing contact hour
Released: February 03, 2026
Expiration: August 02, 2026
Activity
What Is a Bispecific Antibody?
Jennifer Crombie, MD:
Bispecific antibodies are expanding treatment options for patients with hematologic malignancies, including FL. Understanding how these agents work and where they fit into the current treatment paradigm allows us to have better insight into the efficacy and safety of each available bispecific antibody therapy and to be prepared to navigate administration considerations and workflows to manage AEs.
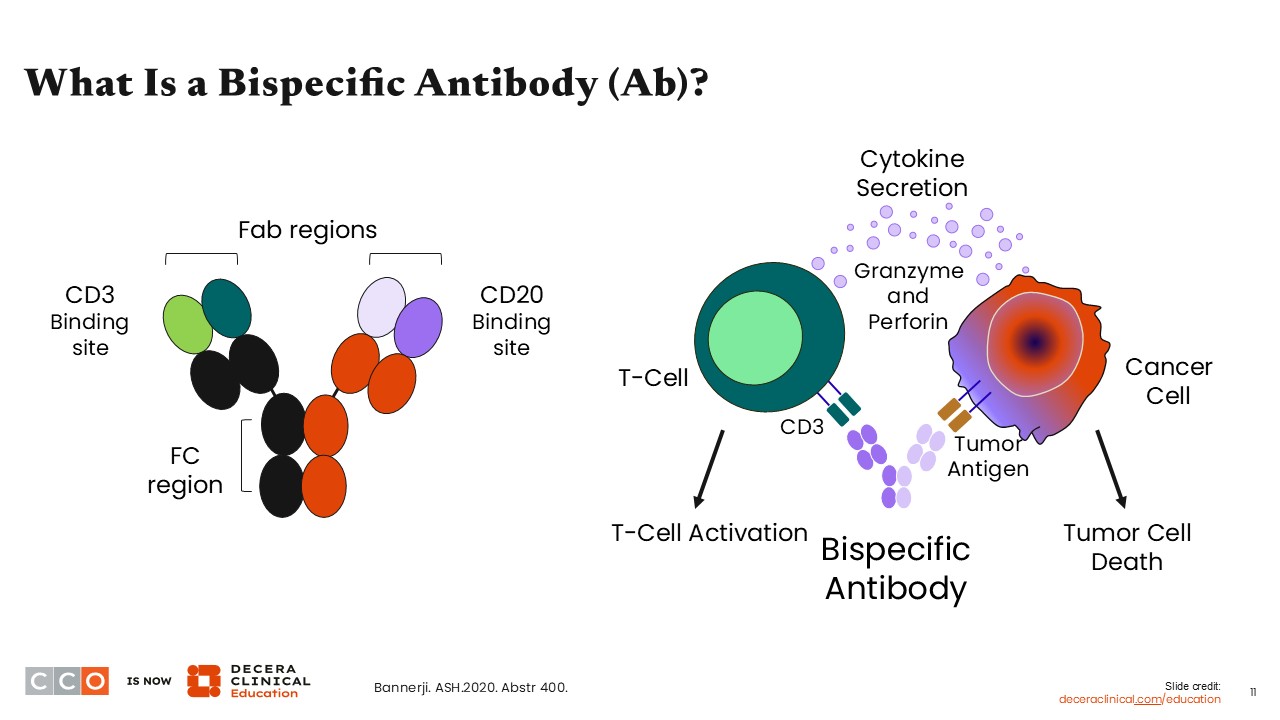
A bispecific antibody is an engineered antibody that is designed to have binding sites for 2 different target antigens. In lymphoma, the most common bispecific antibodies bind CD3, a target on T-cells, and CD20, a marker on mature B-cells, which is highly expressed in many B-cell non-Hodgkin lymphomas, including FL. This dual binding of CD3 x CD20 bispecific antibodies allows a T-cell to be pulled into proximity with a lymphoma tumor cell, resulting in CD3-mediated T-cell activation and targeted tumor cell killing.
Bispecific Antibodies for FL
Jennifer Crombie, MD:
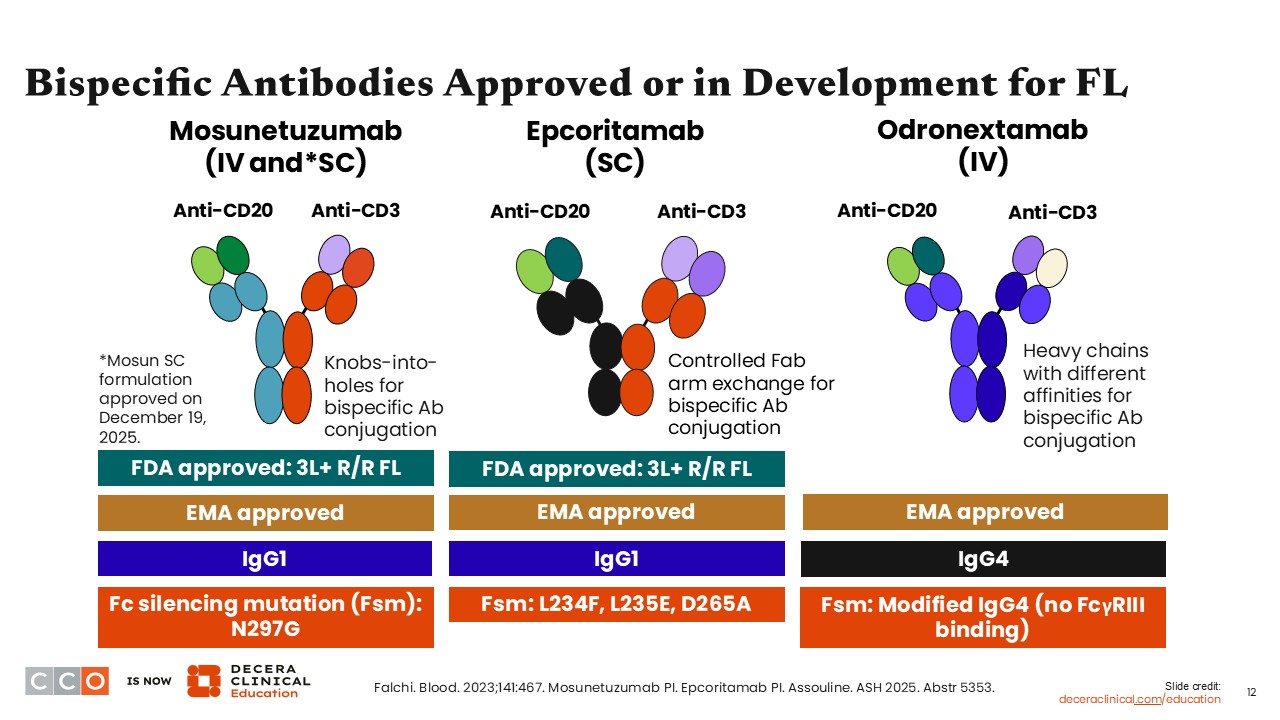
In FL, 3 bispecific antibodies have been integrated as part of the available treatment options for R/R disease and are continuing to be evaluated in additional clinical trials. Mosunetuzumab and epcoritamab are approved by both the FDA and the European Medicines Agency (EMA), and odronextamab is approved by the EMA.
There are some slight differences in administration among these agents. For example, mosunetuzumab can be administered intravenously and was also approved on December 19, 2025, in a subcutaneous form. Epcoritamab is administered subcutaneously, and odronextamab is administered intravenously. There are also differences in administration schedules.1-4
Bispecific Antibodies in B-Cell Lymphomas: Current Indications for FL
Jennifer Crombie, MD:
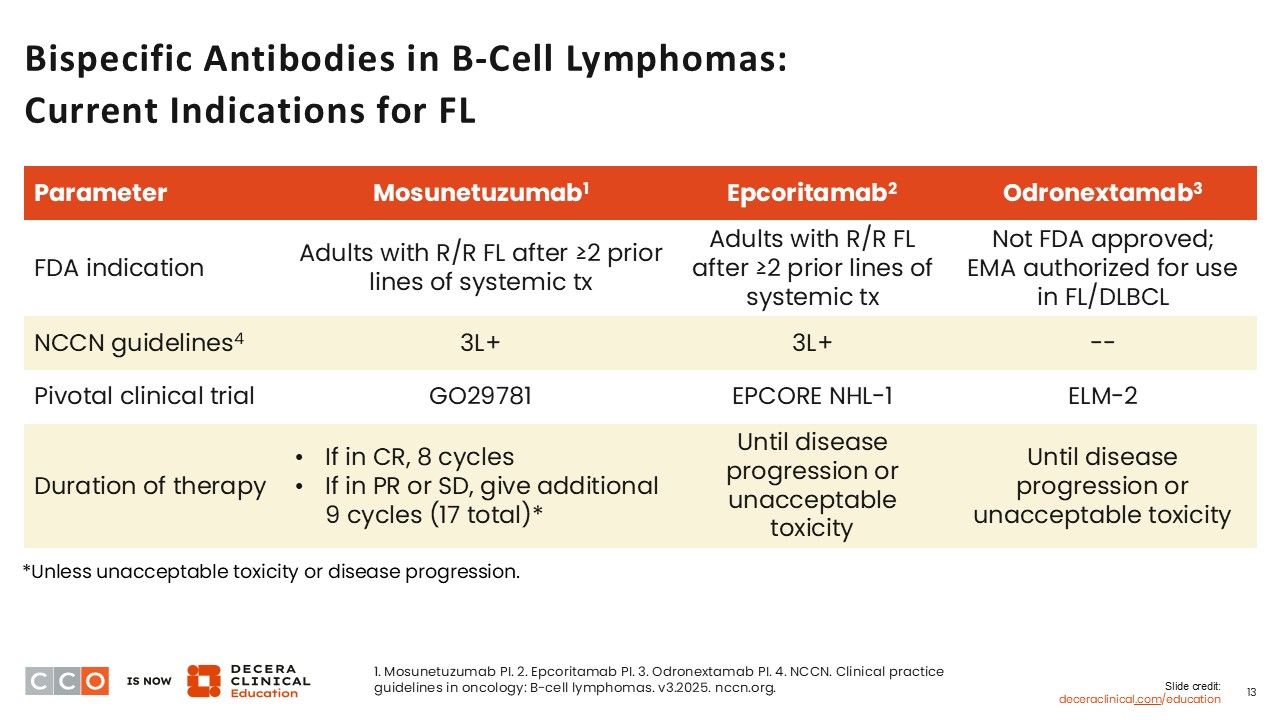
Mosunetuzumab and epcoritamab are approved by the FDA and EMA for use as a single agent in R/R FL after ≥2 prior lines of systemic treatment. Epcoritamab is also approved by the FDA in combination with R2 for patients with R/R FL.
Odronextamab is approved for use by the EMA as a single agent in R/R FL after ≥2 prior lines of systemic treatment and is under consideration with the FDA.
We will summarize the main data that led to these approvals next.
Phase II Dose Expansion Study of Mosunetuzumab in R/R FL After ≥2 Prior LoT
Jennifer Crombie, MD:
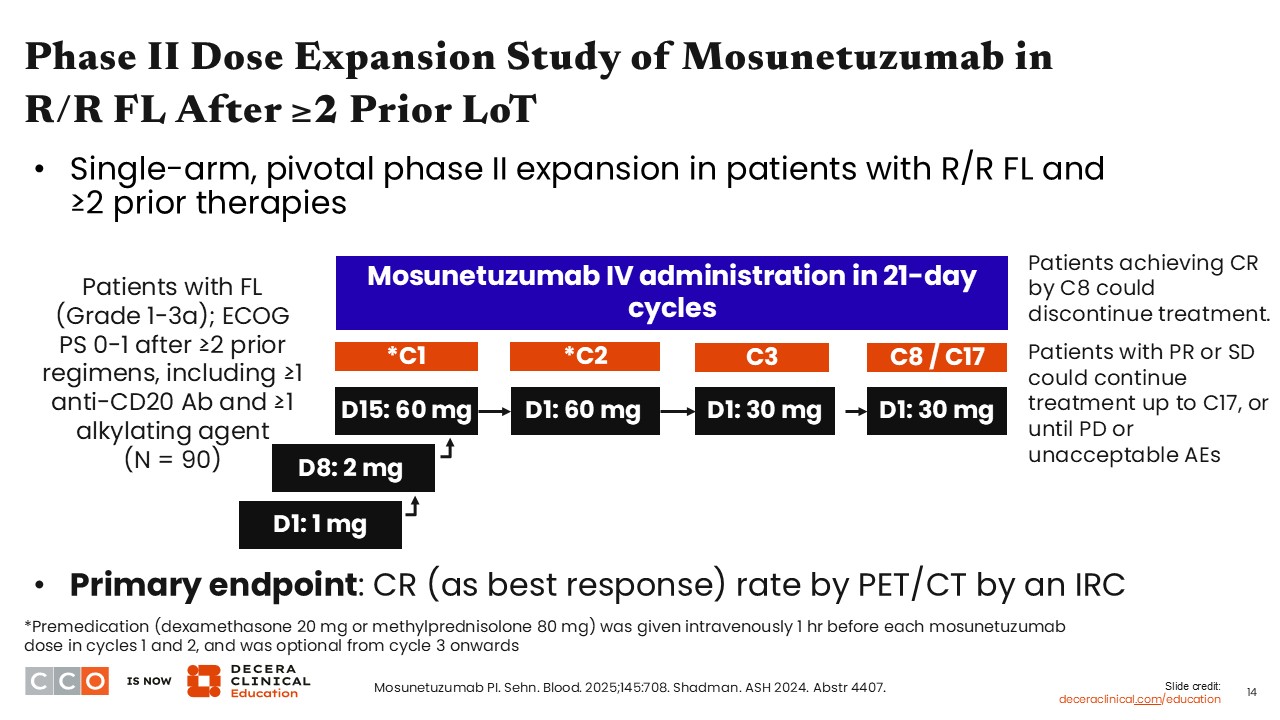
Mosunetuzumab was investigated in a pivotal phase II trial, which included 90 patients with grade 1-3A R/R FL who received ≥2 prior lines of therapy, including a CD20 antibody and an alkylating agent.
Step-up dosing was used during cycle 1, from 1 mg on Day 1 and 2 mg on Day 2 to 60 mg on Day 15. Patients received 60 mg on Day 1 of cycle 2 and then were de-escalated to 30 mg from cycle 3 onwards, which was continued every 21 days for either 8 or 17 cycles. Patients achieving complete response (CR) by cycle 8 could discontinue treatment, whereas those with partial response (PR) or stable disease continued treatment up to cycle 17.2,5,6
Mosunetuzumab in R/R FL: Patient Characteristics
Jennifer Crombie, MD:
Ninety patients were included in the study. The median patient age was 60 years, and the median number of prior lines of therapy was 3, reflecting a heavily pretreated population. Most patients (68.9%) were refractory to their last line of therapy, and more than one half of patients (52.2%) had disease progression within 24 months. Of note, only 3 patients (3.3%) in this study had received prior CAR T-cell therapy.5,6
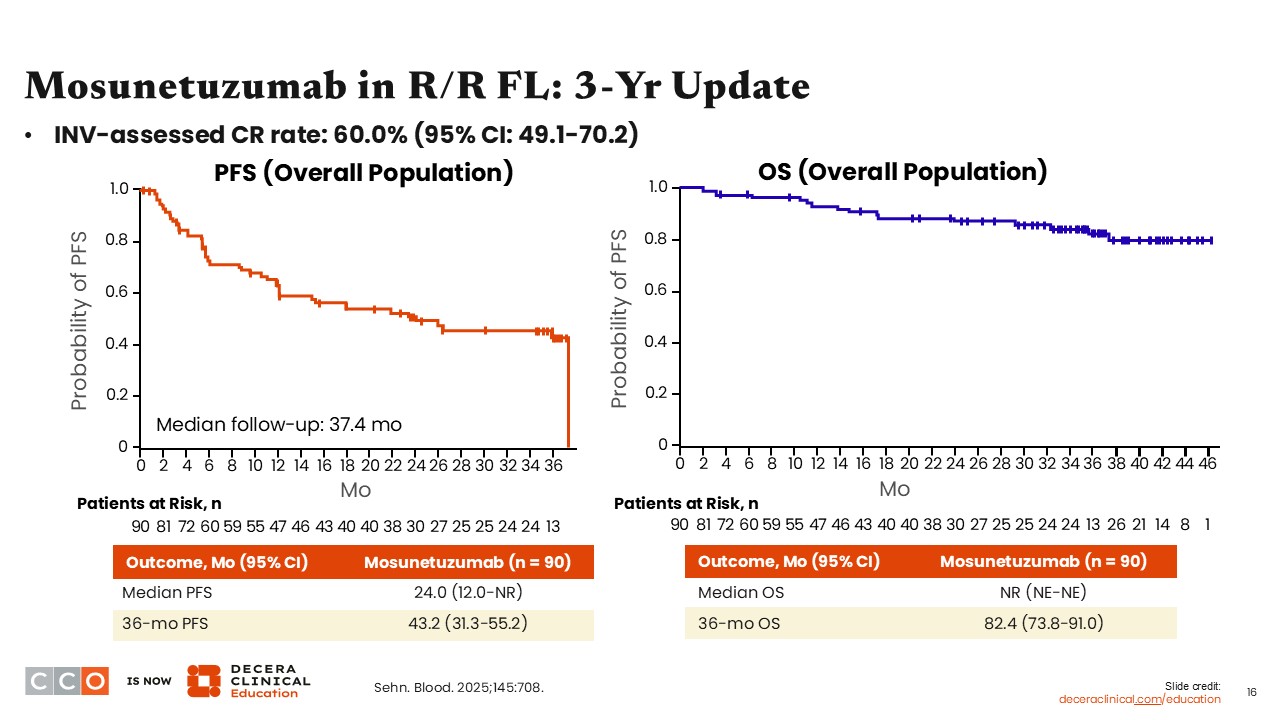
Mosunetuzumab in R/R FL: 3-Year Update
Jennifer Crombie, MD:
A recent 3-year update from the same study reported the efficacy of mosunetuzumab in this population. Investigator-assessed CR rate (primary endpoint) was 60.0% (95% CI: 49.1-70.2). The median progression-free survival (PFS) for the cohort receiving mosunetuzumab was 24 months, and the 36-month PFS rate was 43.2%. Median overall survival (OS) was not reached, and the 36-month OS rate was 82.4%.5
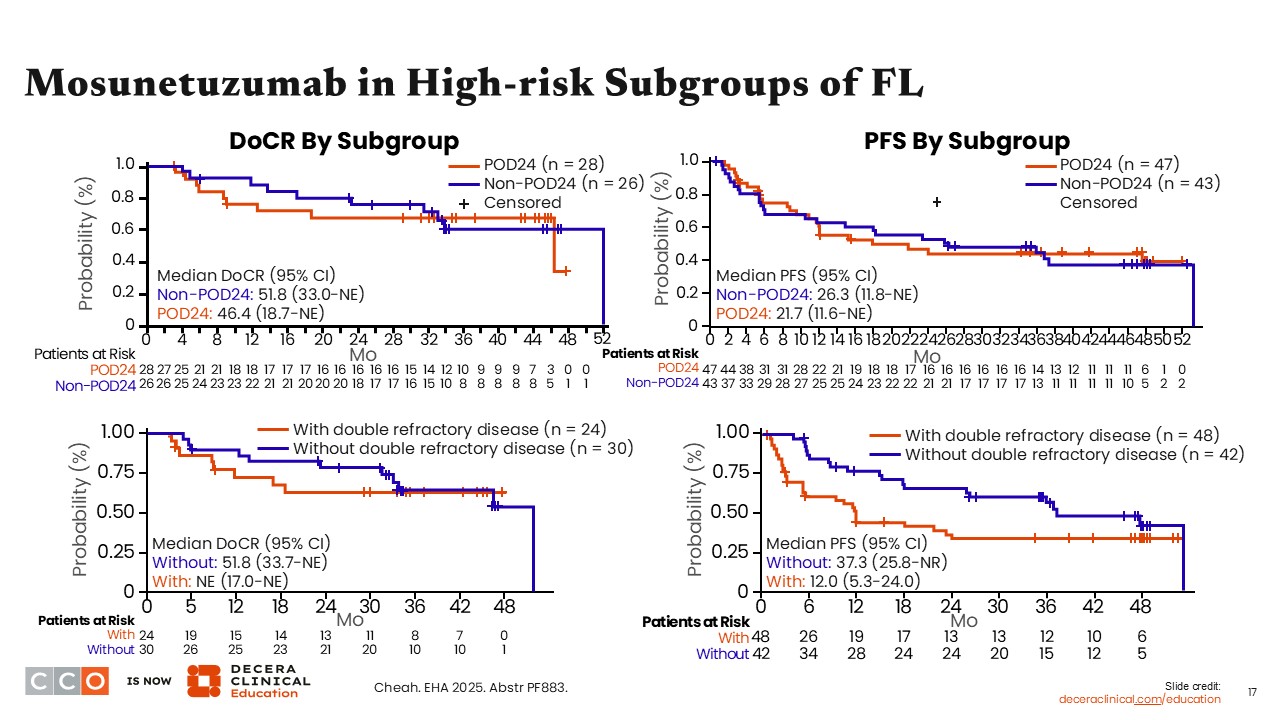
Mosunetuzumab in High-Risk Subgroups of FL
Jennifer Crombie, MD:
In a subset analysis of patients in high-risk subgroups, there was similar duration of complete response and PFS when comparing patients who progressed with disease within 24 months from diagnosis with those who did not. However, patients without double-refractory disease showed better outcomes with duration of complete response and PFS compared with those with double-refractory disease.6
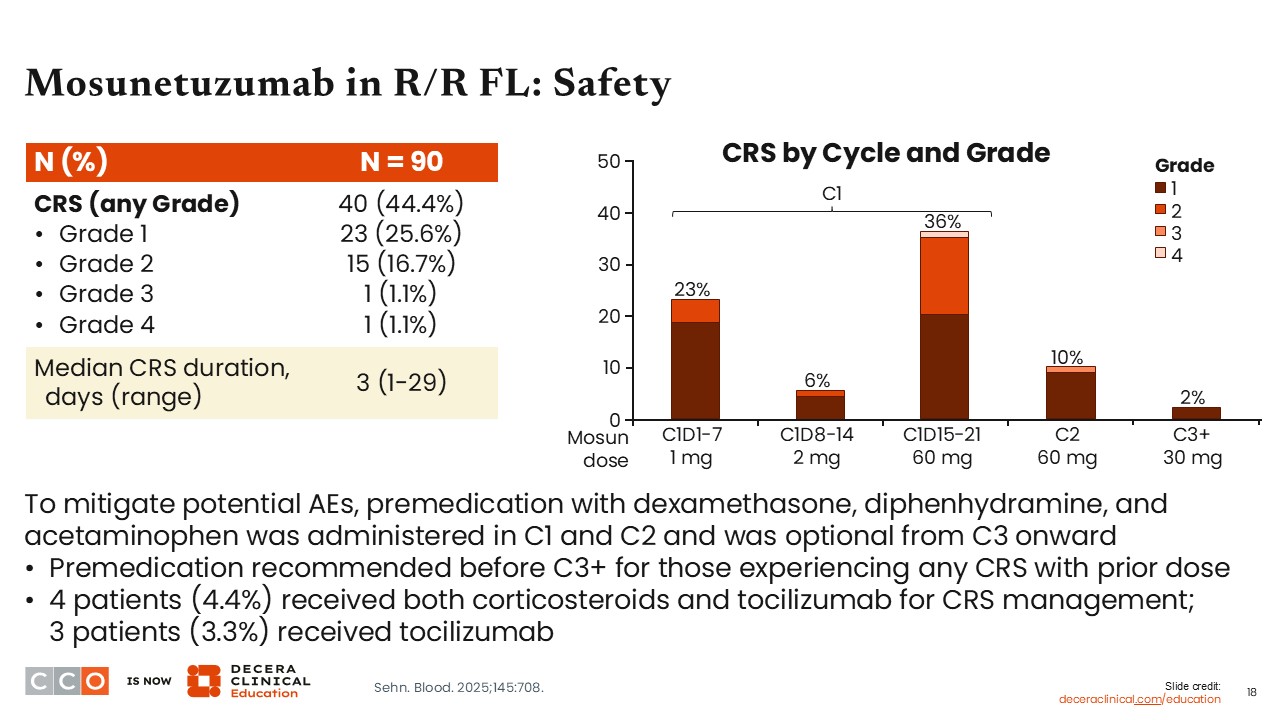
Mosunetuzumab in R/R FL: Safety
Jennifer Crombie, MD:
When considering safety, a key AE of bispecific antibodies is CRS. Among the 90 patients enrolled in the pivotal trial for mosunetuzumab, approximately 44% experienced CRS, but most cases were low grade. Approximately 26% of patients had grade 1 CRS, and approximately 17% had grade 2 CRS. It was rare for patients to experience grade ≥3 CRS.
When assessing CRS by cycle, CRS more commonly occurred during the step-up dosing period, with the highest rates observed with the initial dose, and the first dose up to 60 mg.5
Phase I/II EPCORE NHL-1: Epcoritamab in R/R FL
Jennifer Crombie, MD:
Epcoritamab was also investigated in the phase II EPCORE NHL-1 study in patients with grade 1-3A R/R FL who had previously received ≥2 prior lines of therapy, including a CD20 monoclonal antibody and an alkylating agent or lenalidomide. Epcoritamab was administered subcutaneously, and step-up dosing was used along with CRS prophylaxis. Dose optimization was implemented to reduce the rate of CRS and enable all outpatient dosing of epcoritamab. There was an additional step-up dose and more aggressive hydration and supportive care.8
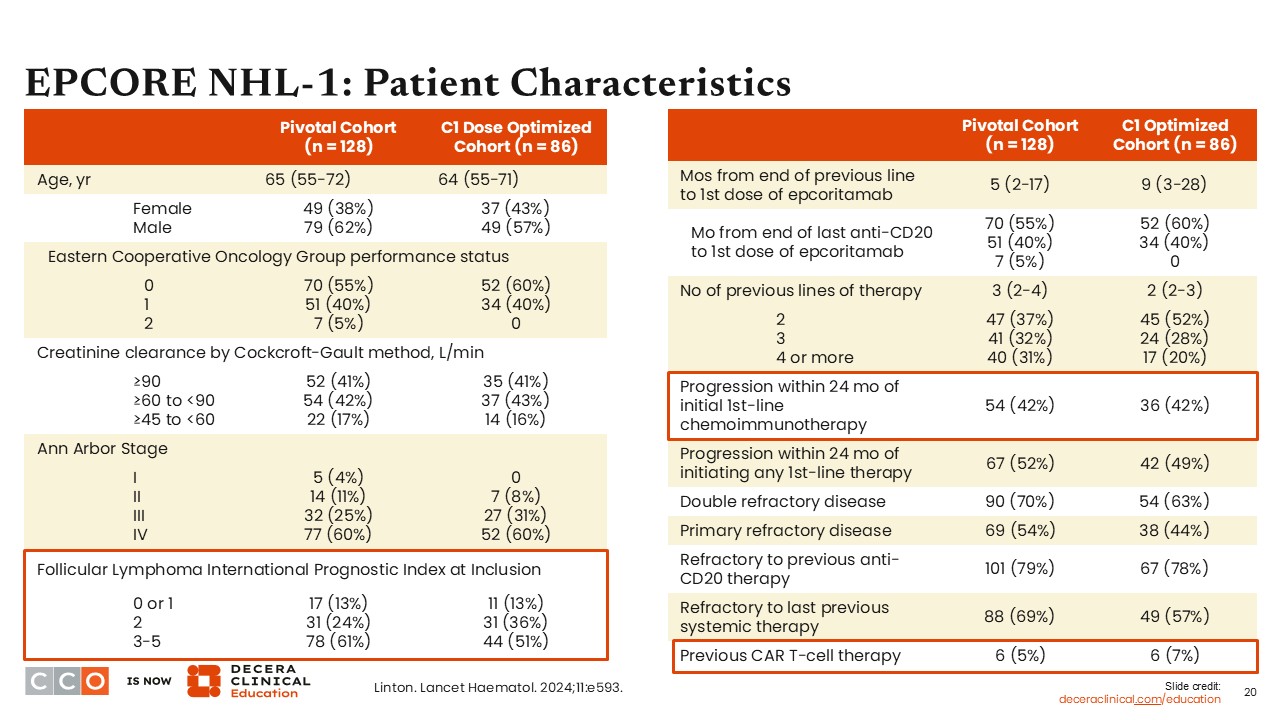
EPCORE NHL-1: Patient Characteristics
Jennifer Crombie, MD:
In the phase I/II EPCORE NHL-1 trial of epcoritamab in patients with R/R B-cell lymphomas, 128 participants were assigned to the pivotal cohort, and 86 were assigned to the dose optimization cohort. Forty-two percent of patients enrolled had previously experienced disease progression within 24 months of initial, first-line chemoimmunotherapy. The median number of prior lines of therapy was 3 in the pivotal cohort and 2 in the dose optimization cohort. It was rare for participants to have received prior CAR T-cell therapy (5% in the pivotal cohort and 7% in the dose optimization cohort).8
EPCORE NHL-1: Efficacy of Epcoritamab in R/R FL
Jennifer Crombie, MD:
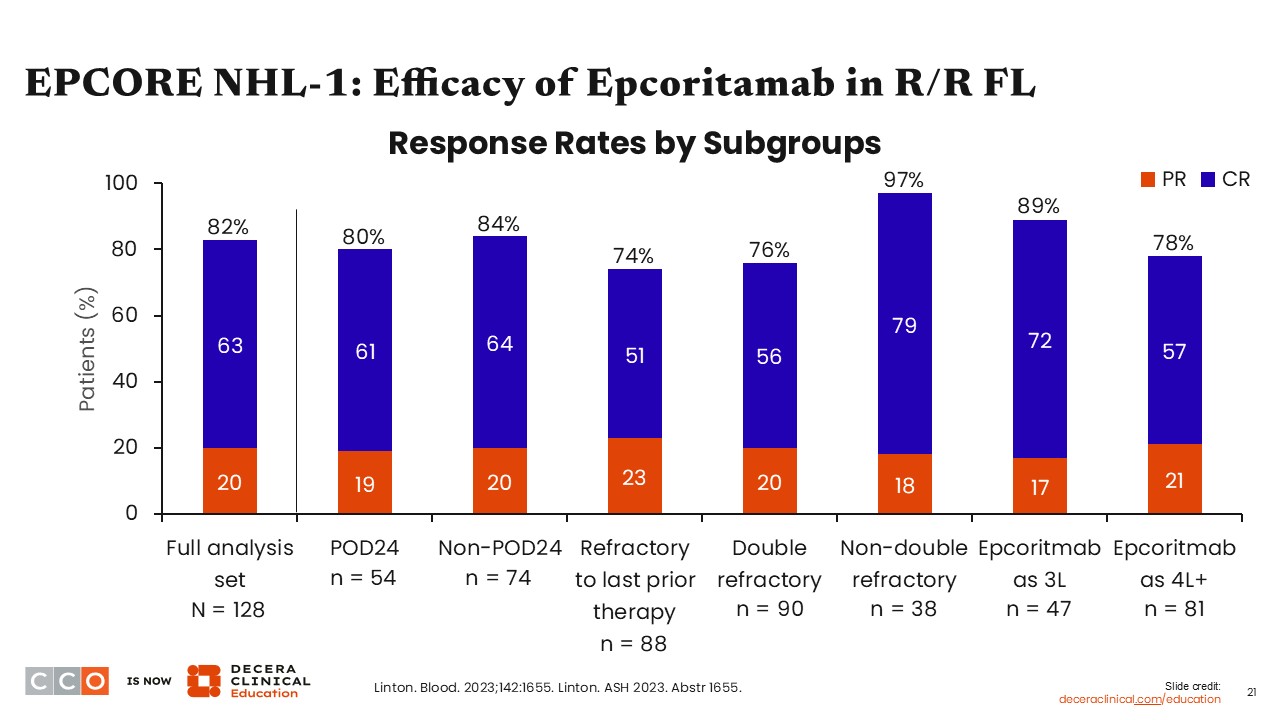
The overall response rate (ORR) for epcoritamab in patients with R/R FL from EPCORE NHL-1 (primary endpoint) was 82%, with a 63% CR rate.
Of note, the response rates were similar across high-risk subgroups, including the POD24 subgroup of participants who experienced disease progression within 24 months of first-line chemoimmunotherapy as well as patients who were refractory to their last line of therapy or received epcoritamab as a later line of therapy.8
EPCORE NHL-1: Progression-Free Survival
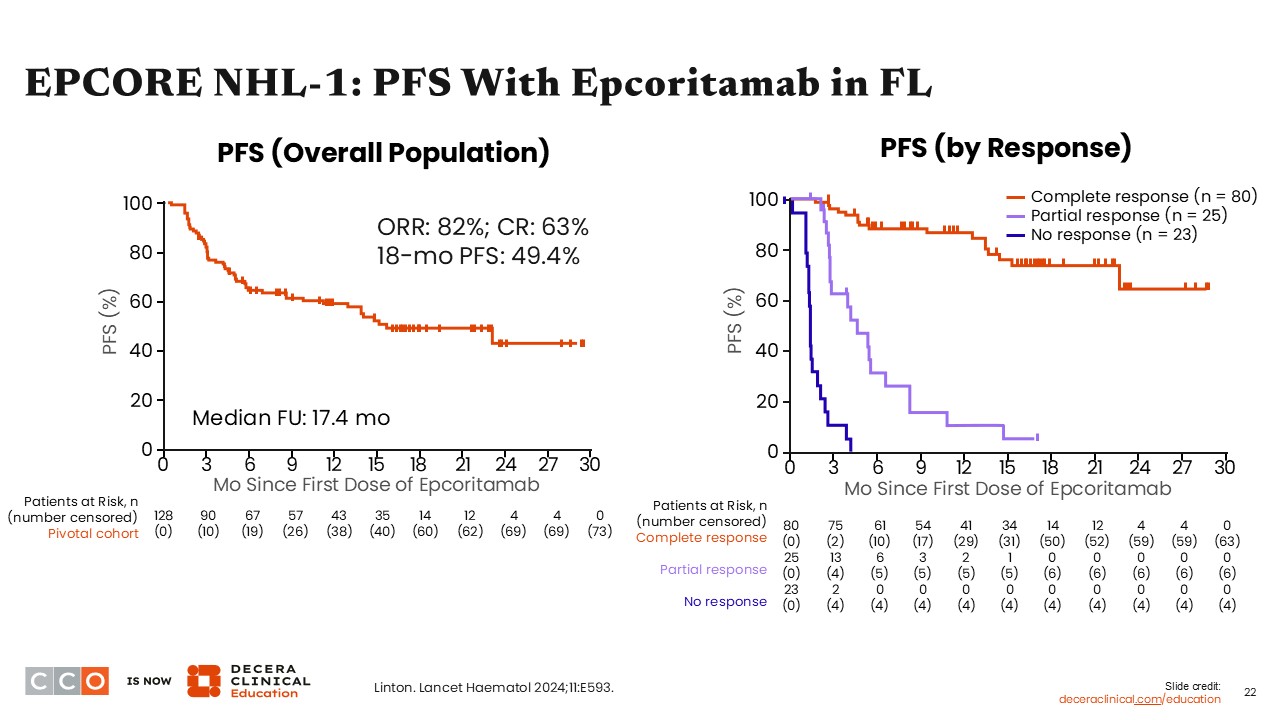
Jennifer Crombie, MD:
The 18-month PFS rate was 49.4% among patients with R/R FL treated with epcoritamab. The ORR was 82%, and the CR rate was 63%. Consistent with data from trials of other bispecific antibodies, PFS seems to be most durable in patients who achieve a CR.8
EPCORE NHL-1: Safety of Epcoritamab in R/R FL
Jennifer Crombie, MD:
This table shows the rates and grades of CRS between cohorts. CRS occurred in 65% of patients in the pivotal cohort. However, in the dose-optimization cohort, CRS occurred in 49% of patients, with no grade ≥3 CRS was observed.8
Phase II ELM-2 FL: Odronextamab in R/R FL
Jennifer Crombie, MD:
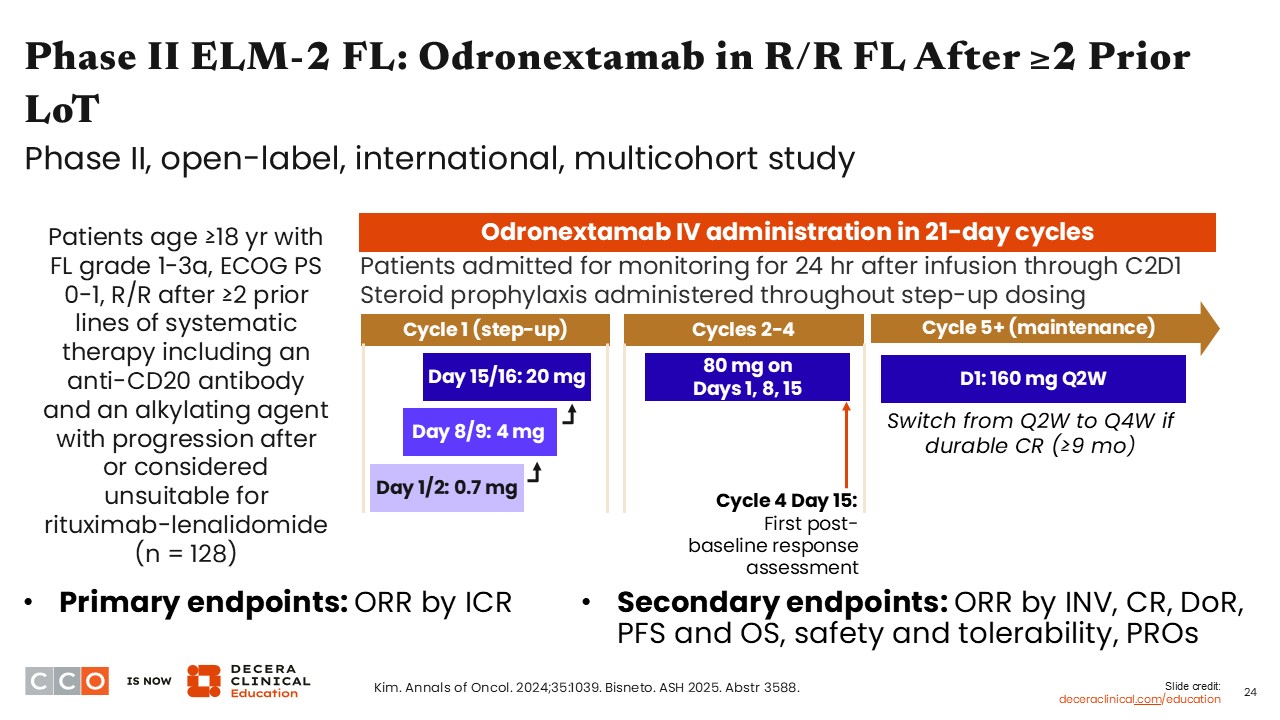
The phase II ELM-2 FL study is evaluating odronextamab in patients with R/R FL after ≥2 prior lines of systemic therapy including an anti-CD20 antibody and an alkylating agent with progression after or considered unsuitable for R2. The step-up dosing differs from that with the other bispecific antibodies because in addition to the step-up dosing, there is also split dosing that occurs on Days 1, 2, 8, 9, 15, and 16 during step-up in cycle 1. Then, in cycles 2-4, odronextamab was given on Days 1, 8, and 15 before transitioning to every-2-week dosing in cycle 5. Patients then are treated with the target dose and continued on a maintenance dose throughout treatment. Patients then are able to switch from every-2-week to every-4-week dosing if they achieve a durable CR (≥9 months). Treatment with odronextamab is continuous, so patients remain on therapy until either disease progression or unacceptable toxicity.9,10
ELM-2 FL: Patient Characteristics
Jennifer Crombie, MD:
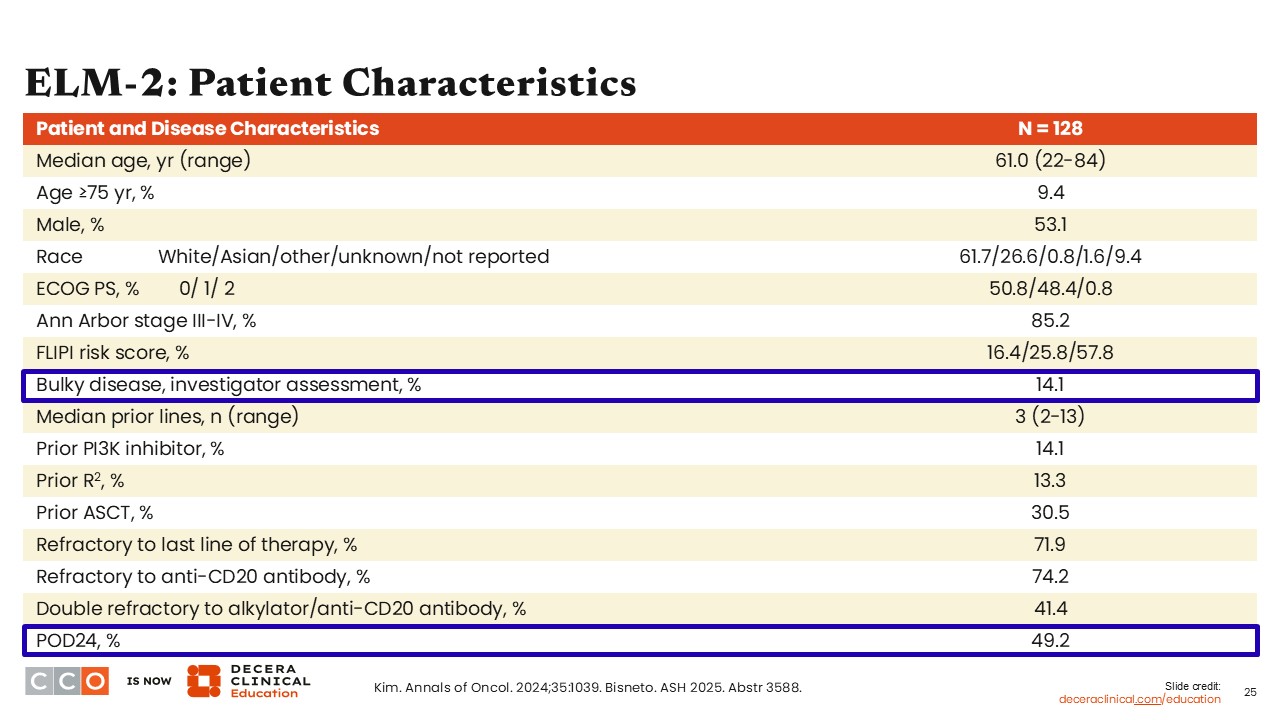
Overall, 128 patients were enrolled on ELM-2. Patients had a median of 3 prior lines of therapy. In addition, approximately one half of patients experienced disease progression within 24 months of first-line chemoimmunotherapy, represented in this table as POD24.9
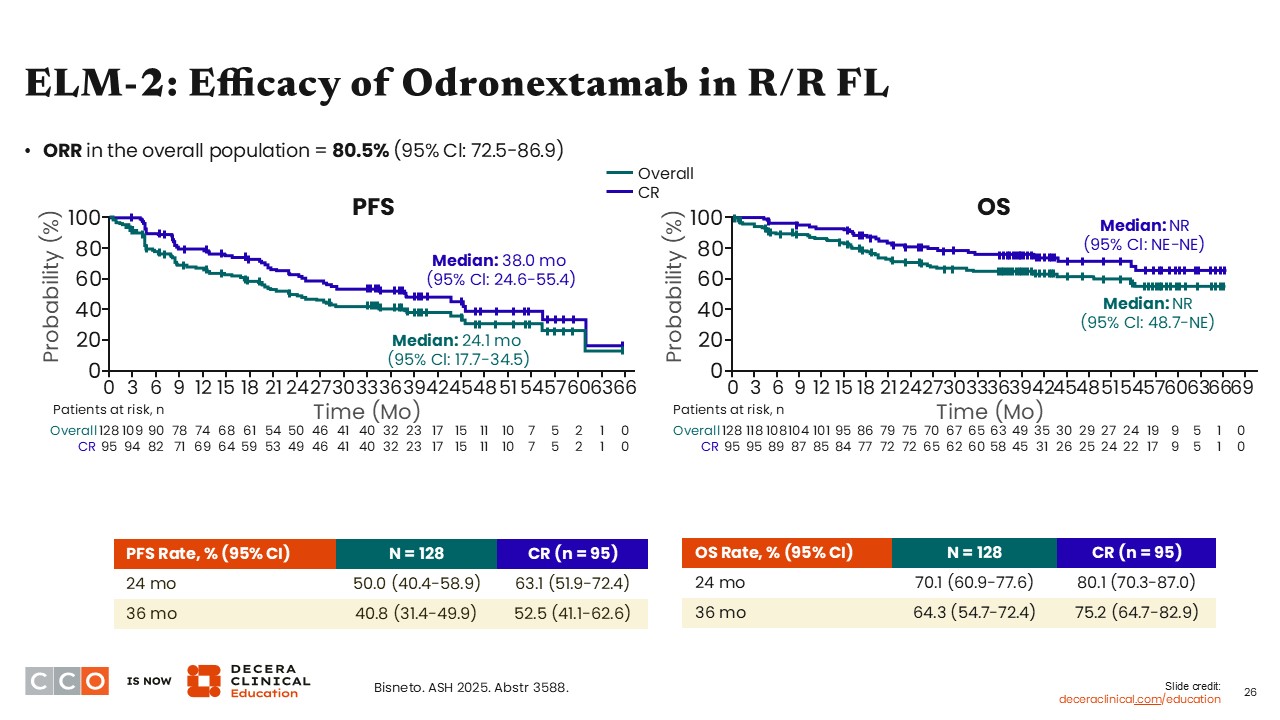
ELM-2: Efficacy of Odronextamab in R/R FL
Jennifer Crombie, MD:
In the ELM-2 trial, ORR in the overall population (primary endpoint) was 80.5% (95% Cl: 72.5%-86.9%) with odronextamab. In the total study population, the median PFS was 24 months. Median PFS was improved in patients who had a CR, reaching 38 months, and median OS among all patients was not reached.10
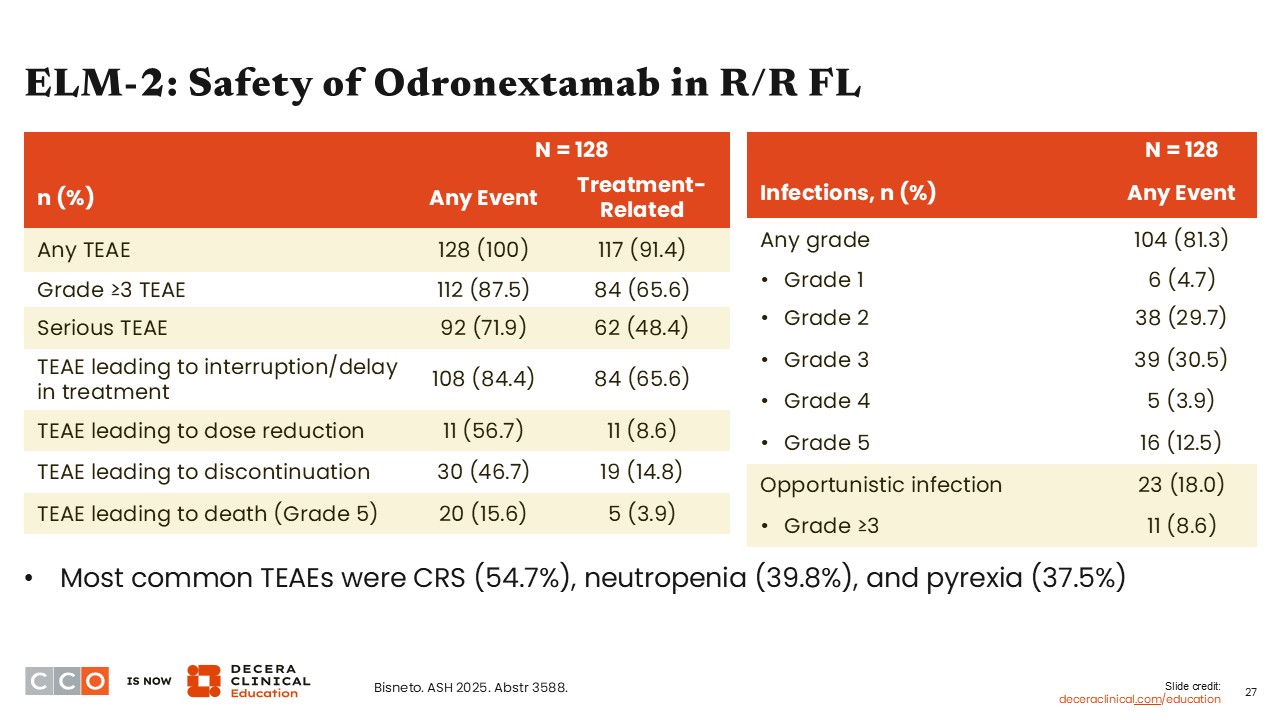
ELM-2: Safety of Odronextamab in R/R FL
Jennifer Crombie, MD:
This table lists some key AEs among patients treated with odronextamab in ELM-2. Infections at any grade occurred in approximately 81% of patients, with most of those being grade 2/3. The most common any-grade treatment-emergent AE was CRS, which occurred in approximately 55% of patients, followed by neutropenia and pyrexia.10
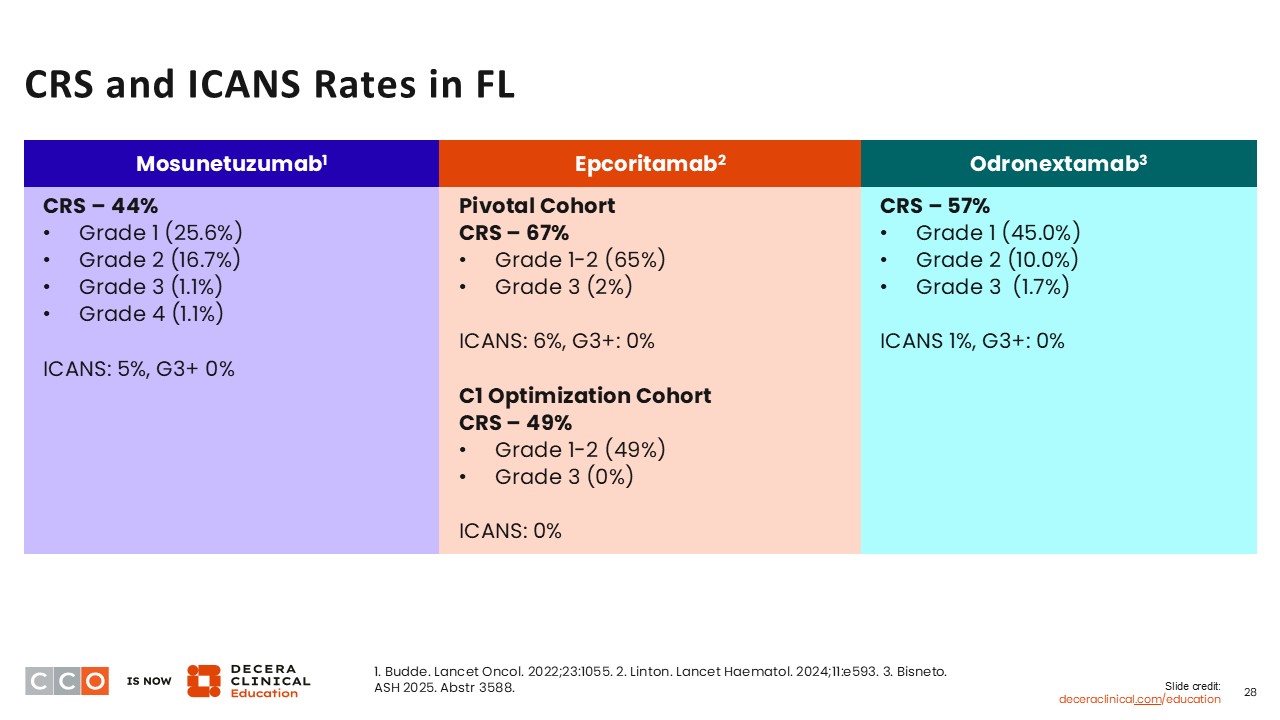
CRS and ICANS Rates in FL
Jennifer Crombie, MD:
As mentioned, CRS is a common AE with most bispecific antibodies. Neurotoxicity is also associated with bispecific antibody therapy but occurs less commonly than CRS.
The rates of any-grade and grade 1/2 CRS are similar with mosunetuzumab, epcoritamab, and odronextamab. Of note, the neurologic toxicity rates were relatively low, ranging between 1% and 6% across the studies. Of importance, there were no cases of grade ≥3 ICANS reported in these studies.8-11
Key Considerations Before Initiating CD3 x CD20 Bispecific Antibodies
Jennifer Crombie, MD:
Oncology care teams face challenges when preparing to integrate bispecific antibodies into treatment algorithms. The studies I just described show that these drugs are active in patients with R/R FL, but they are not routinely used across clinical practices.
One issue that may be contributing to their slow adoption is a lack of education on specific guideline updates for AE management. Although there are some recommendations on AE management, this guidance may not be available or healthcare professionals (HCPs) may not be familiar with these resources.
Another possibility is unfamiliarity with recent data (ie, HCPs may not be aware of updates at recent annual meetings and publications). Use of bispecific antibody therapy does require some specialized knowledge of the optimal dosing and administration considerations, along with the management of their specific AE profiles. There may be an implementation lag whereby these agents will be used across clinical practice soon, but it is taking time to fully adopt them.
Other potential major patient-related issues are cost and reimbursement concerns, especially when therapies may be administered in an inpatient setting. However, as we gain experience with these agents, bispecific antibodies can also be fully administered in the outpatient setting with careful monitoring. Then, if a patient experiences CRS or other specific AEs, they may require hospital admission for management.12
Key Considerations Before Initiating CD3 x CD20 Bispecific Antibodies
Jennifer Crombie, MD:
Another issue to consider includes logistical challenges. Even if healthcare professionals are aware of the data and the guidelines, it still can be difficult to implement these agents, depending on the structure of the clinical practice. Administering bispecific antibodies requires frequent patient visits for step-up dosing and close monitoring for AEs.
Perhaps the most difficult challenge is treating AEs in the outpatient setting. Does the clinic have the infrastructure to see the patient quickly for evaluation, or do they collaborate with an emergency department or inpatient facility that can help manage patients with CRS?
HCPs can consider practical strategies for integrating information into electronic medical records. Could there be a flag in place to identify a specific patient as receiving a bispecific antibody? This approach could be helpful for emergency department or other inpatient HCPs who may be meeting this patient for the first time.12
Farrukh T. Awan, MD, MS, MBA:
Other needs include a patient-specific plan encompassing patient education, caregiver education, and a communication plan to ensure that patients and their care team are aware of the unique risks.
Patients should have access to urgent care evaluations, either in the treatment center or a center that is familiar with the management of patients receiving bispecific antibody therapy. Follow-up care is an important component for overall patient outcomes, especially if patients are receiving treatment at a center at a substantial distance from their residence.
Based on these factors, HCPs can determine whether the patient is a suitable candidate for outpatient or inpatient therapy or at least needs to be admitted for monitoring, especially after the first full dose.
Key Considerations Before Initiating CD3 x CD20 Bispecific Antibodies
Farrukh T. Awan, MD, MS, MBA:
One of the most important things to be vigilant of is patient education: Are patients educated about these AEs, especially if all treatments are done in an outpatient setting? Patients and their caregivers need to be educated about the risks of these AEs, how to monitor for these AEs (eg, monitor their temperature at home), and how to follow-up with the clinical care team to report any fevers, decreases in blood pressure, shortness of breath, or confusion. Patients need to have the resources to monitor their symptoms at home, including a blood pressure monitor and thermometer.
Patients would also benefit from close monitoring of their antihypertensives during these times to prevent further worsening of symptoms. They also need to be vigilant about infectious complications or signs of infections, as these can complicate the diagnosis of these AEs.13
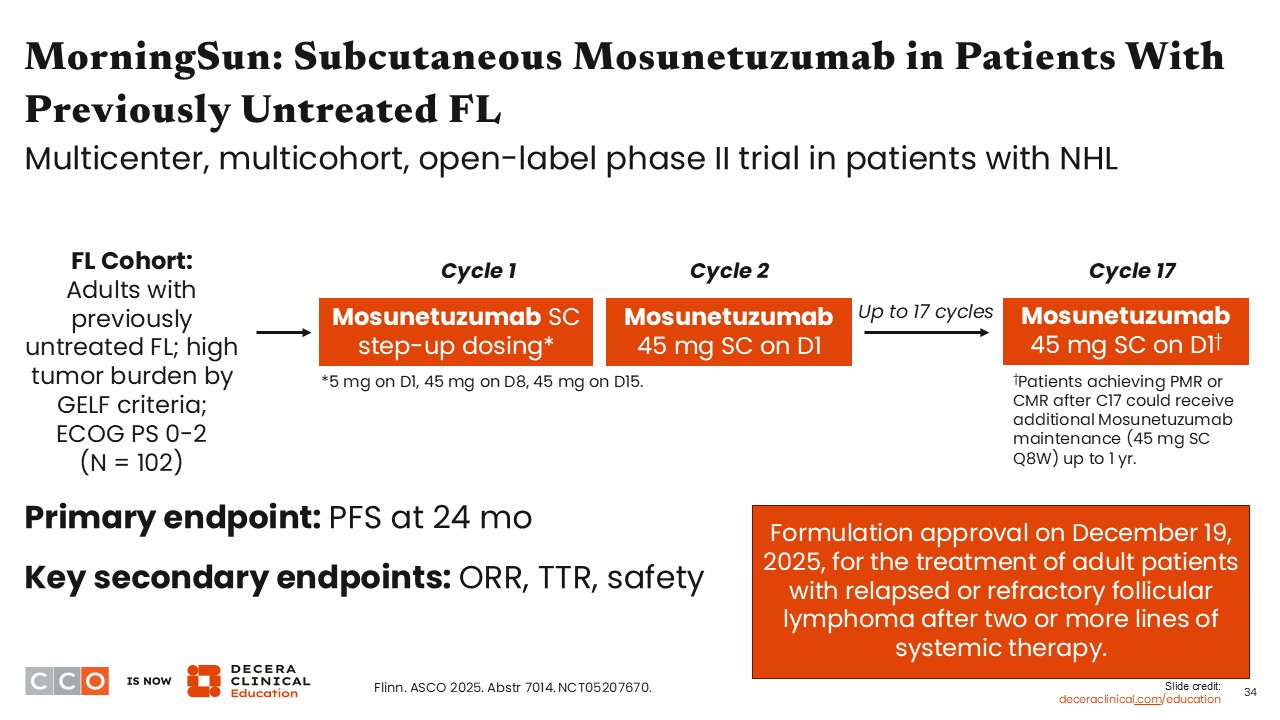
MorningSun: Subcutaneous Mosunetuzumab in Patients With Previously Untreated FL
Jennifer Crombie, MD:
The MorningSun trial evaluated the efficacy of the subcutaneous formulation of mosunetuzumab in adults with untreated FL and high tumor burden by GELF criteria. Similar to the other trials with bispecific antibodies, this trial used step-up dosing in the first cycle to mitigate the risk of CRS. It is important to note that the dosing for subcutaneous mosunetuzumab is different from that for intravenous administration, with step-up dosing from 5 mg on Day 1 of cycle 1 to 45 mg on Days 8 and 15, followed by 45 mg on Day 1 of cycle 2 through the end of treatment at cycle 17. In this trial, if patients achieved either partial molecular response or complete molecular response after cycle 17, they could continue to receive additional mosunetuzumab maintenance (45 mg every 8 weeks for up to 1 year).14
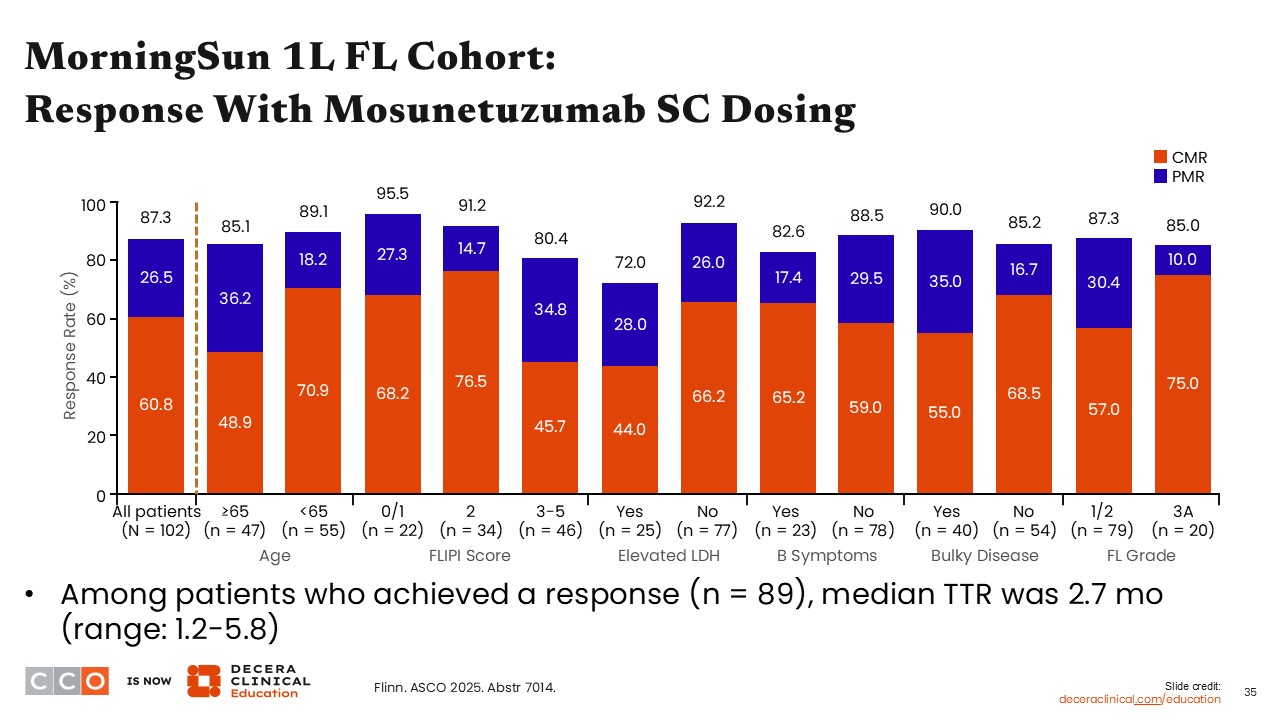
MorningSun 1L FL Cohort: Response With Mosunetuzumab SC Dosing
Jennifer Crombie, MD:
Among all patients (N = 102) treated with subcutaneous mosunetuzumab, 60.8% achieved complete molecular response and 26.5% achieved partial molecular response. Among the patients achieving a response, the median time to response was 2.7 months. Responses were similar across a variety of patient subgroups, including those with bulky disease, B symptoms, or grade 3A disease.14
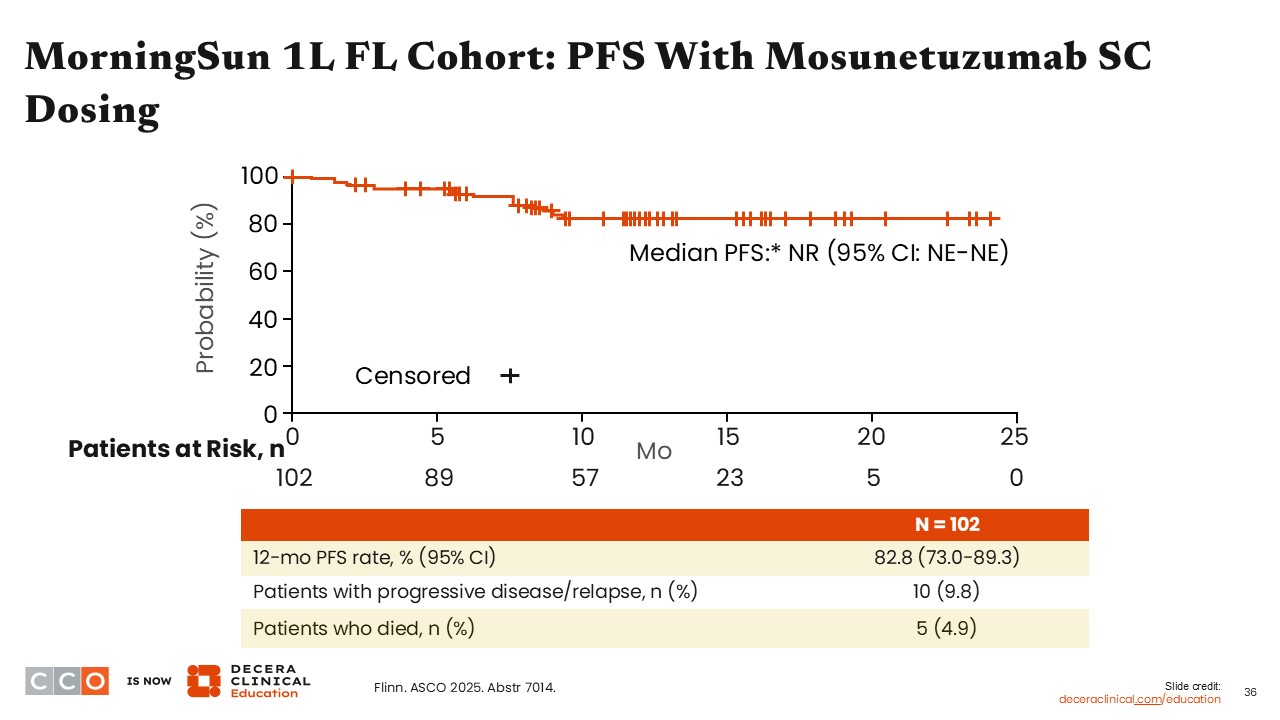
MorningSun 1L FL Cohort: PFS With Mosunetuzumab SC Dosing
Jennifer Crombie, MD:
The 12-month PFS rate among participants treated with subcutaneous mosunetuzumab was 82.8%. Among these patients, 9.8% experienced disease relapse or progression on therapy, and 5 patients (4.9%) died during the study.14
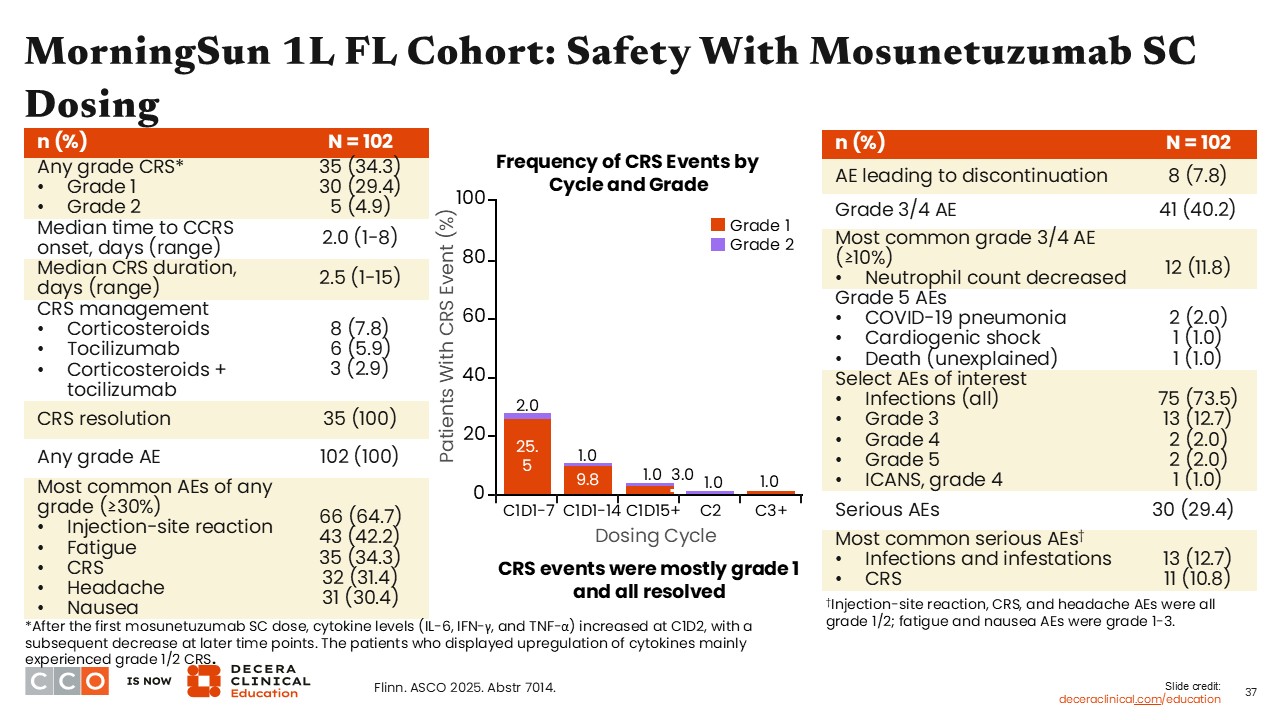
MorningSun 1L FL Cohort: Safety With Mosunetuzumab SC Dosing
Jennifer Crombie, MD:
In patients treated with subcutaneous mosunetuzumab, 34.3% experienced any-grade CRS. Most of these patients (29.4%; n = 30) had grade 1 CRS, and a few (4.9%; n = 5) had grade 2 CRS. Most instances occurred during the first treatment cycle, between Days 1 and 7. All of the CRS events resolved with supportive care and CRS management as needed.14
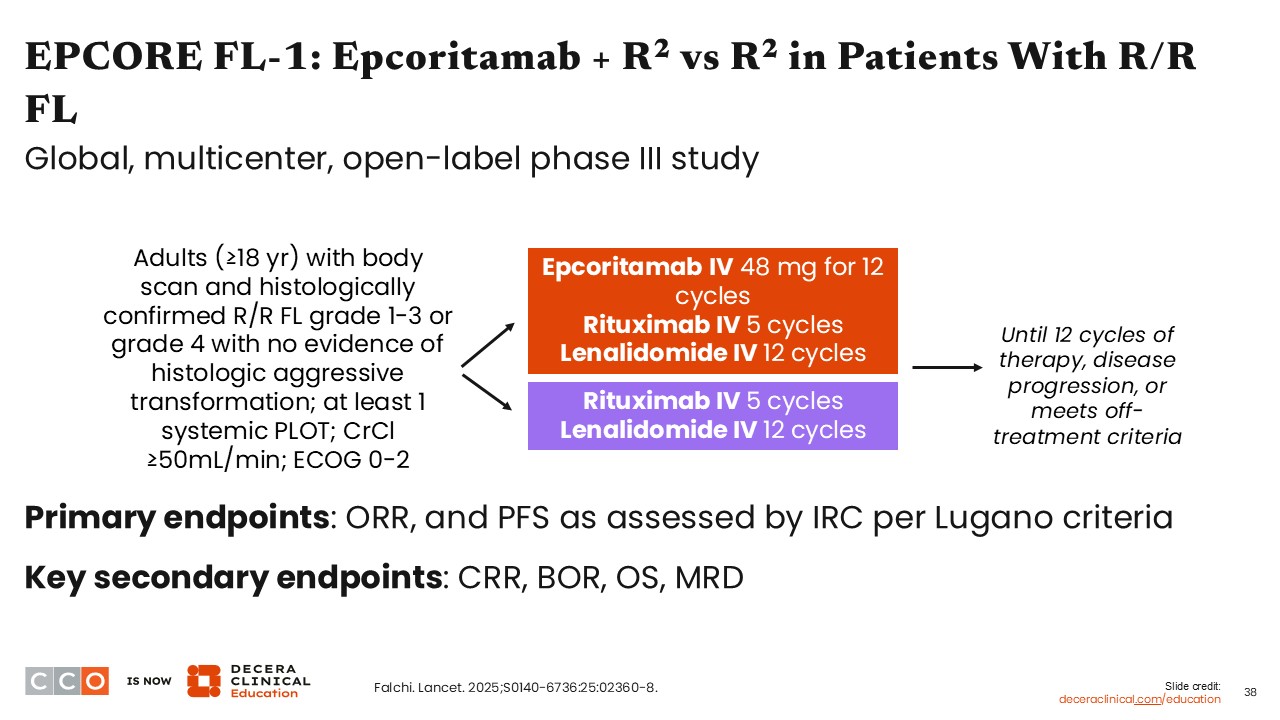
EPCORE FL-1: Epcoritamab + R2 vs R2 in Patients With R/R FL
Jennifer Crombie, MD:
One emerging theme is combining bispecific antibodies with other agents. Bispecific antibodies have shown good activity in R/R FL as single agents, but can patients at high risk achieve even better outcomes if we combine different treatment modalities? A few phase III trials are investigating bispecific antibody combination therapies.
For example, the phase III EPCORE FL1 trial is comparing epcoritamab in combination with R2 vs R2 alone in patients with R/R FL after ≥1 systemic previous line of treatment. The primary endpoints of this trial are ORR and PFS, as assessed by an independent review committee per Lugano criteria.15
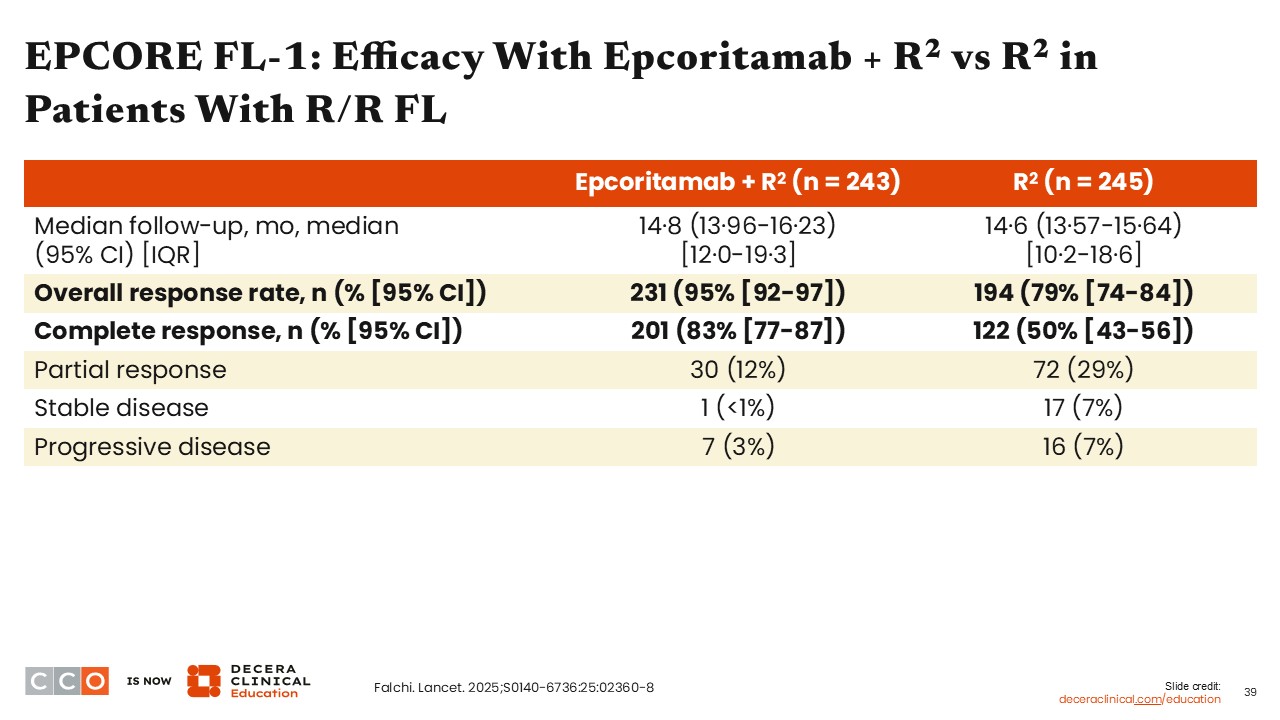
EPCORE FL-1: Efficacy With Epcoritamab + R2 vs R2 in Patients With R/R FL
Jennifer Crombie, MD:
Among patients treated with epcoritamab plus R2, the majority experienced an overall response to therapy (95%) compared with 79% of patients treated with R2. In addition, most patients treated with epcoritamab plus R2 achieved a CR, whereas only one half of the patients treated with R2 alone achieved a CR.15
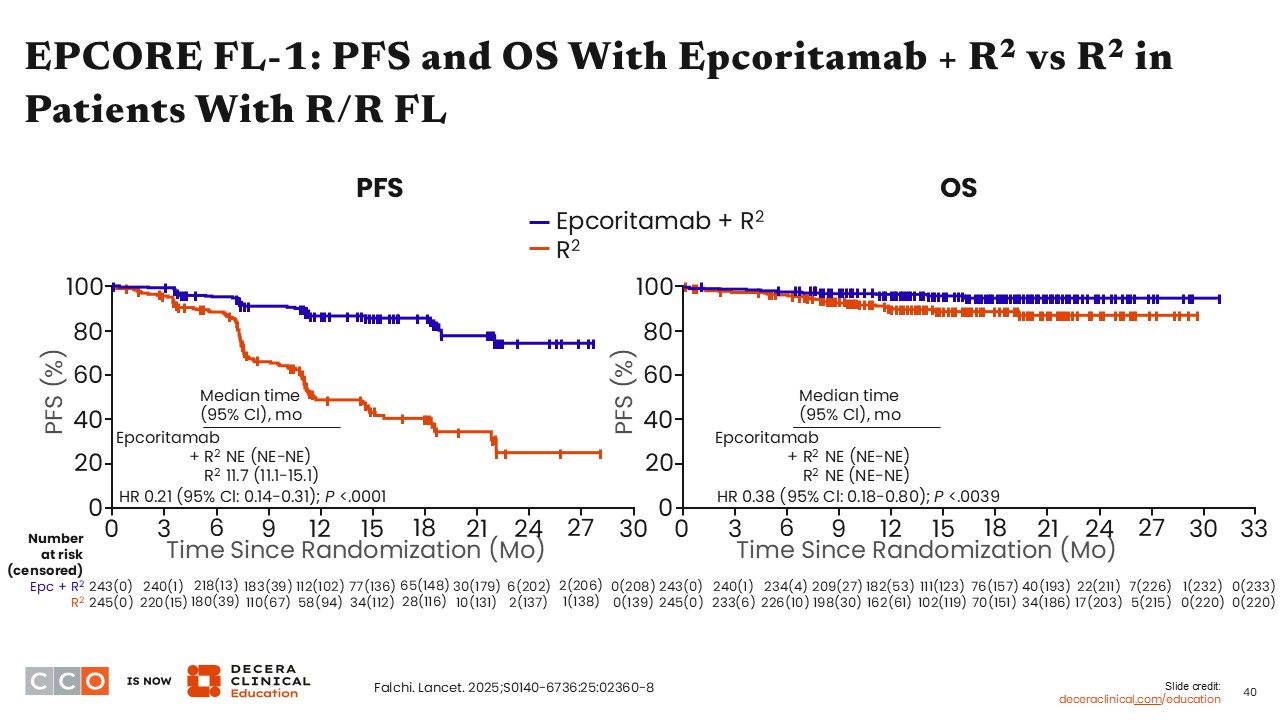
EPCORE FL-1: PFS and OS With Epcoritamab + R2 vs R2 in Patients With R/R FL
Jennifer Crombie, MD:
The median PFS was not reached for patients receiving epcoritamab plus R2 vs 11.7 months for patients treated with R2, resulting in an HR of 0.21 (95% Cl: 0.14-0.31; P <.0001).
Although the OS data are not mature yet, with the median OS note reached with either epcoritamab plus R2 or R2 alone, the 16-month OS rates were 95.8% for epcoritamab plus R2 and 88.8% for R2, with an HR of 0.38 (95% Cl: 0.18-0.80; P <.0039).15
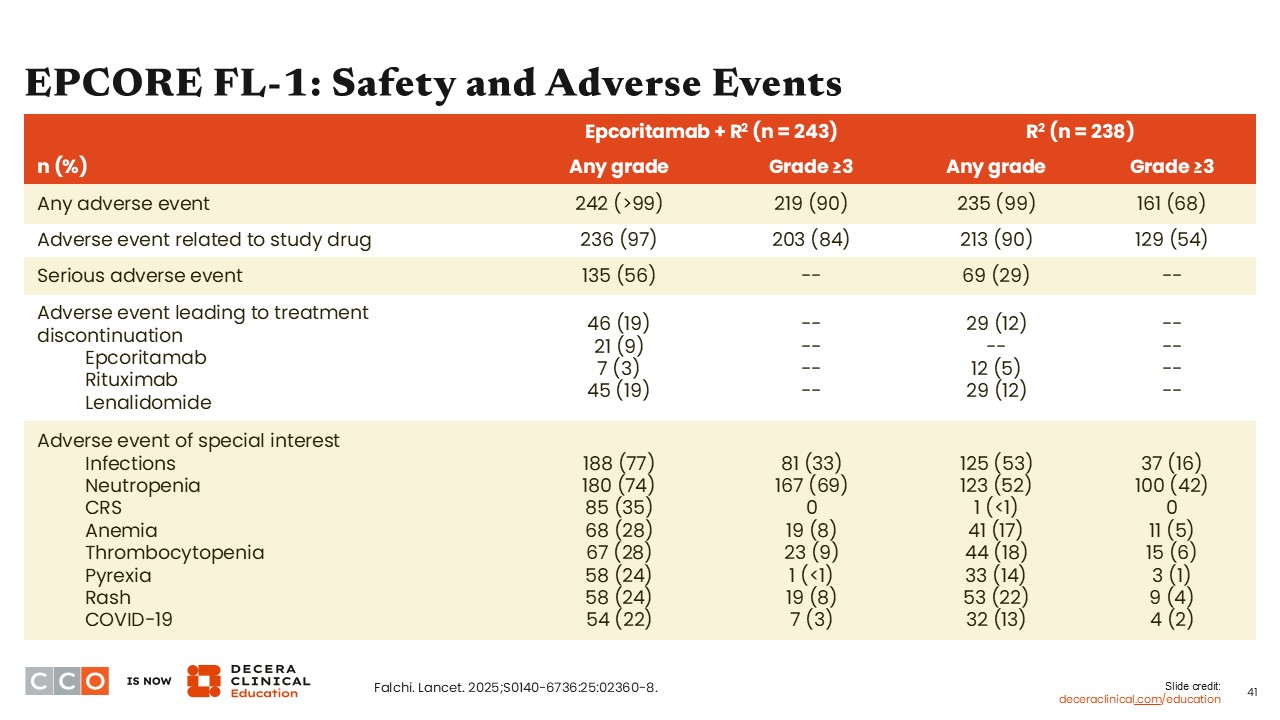
EPCORE FL-1: Safety and Adverse Events
Jennifer Crombie, MD:
Nearly all treated patients experienced an AE with both epcoritamab plus R2 and R2 alone, with somewhat higher percentage of patients epcoritamab plus R2 experiencing grade ≥3 AEs (90% vs 68% with R2). More than one half of the patients treated with epcoritamab plus R2 had serious AEs vs approximately one third of the patients treated with R2 alone.
The most common AE was infections, followed by neutropenia in both treated groups. One third of the patients treated with epcoritamab plus R2 experienced CRS, whereas only 1 patient in the R2 cohort experienced CRS.15
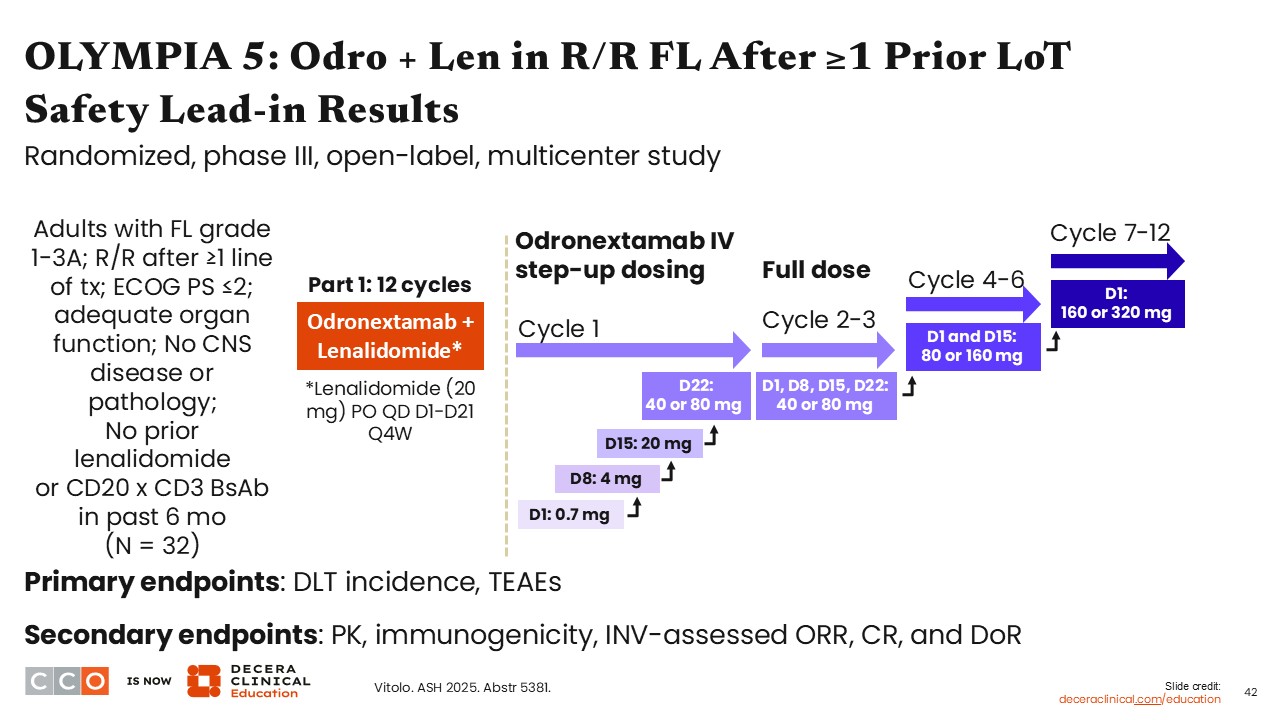
OLYMPIA 5: Odro + Len in R/R FL After ≥1 Prior LoT Safety Lead-in Results
Jennifer Crombie, MD:
The phase III OLYMPIA-5 trial is investigating odronextamab and lenalidomide vs R2 in patients with R/R FL or marginal zone lymphoma after ≥2 cycles of prior systemic therapy that included ≥1 anti-CD20 antibody. In Part 1 of this study, a safety lead-in of odronextamab and lenalidomide was administered for 12 cycles, using step-up dosing of intravenous odronextamab to evaluate dose-limiting toxicity incidence and treatment-emergent AEs.16
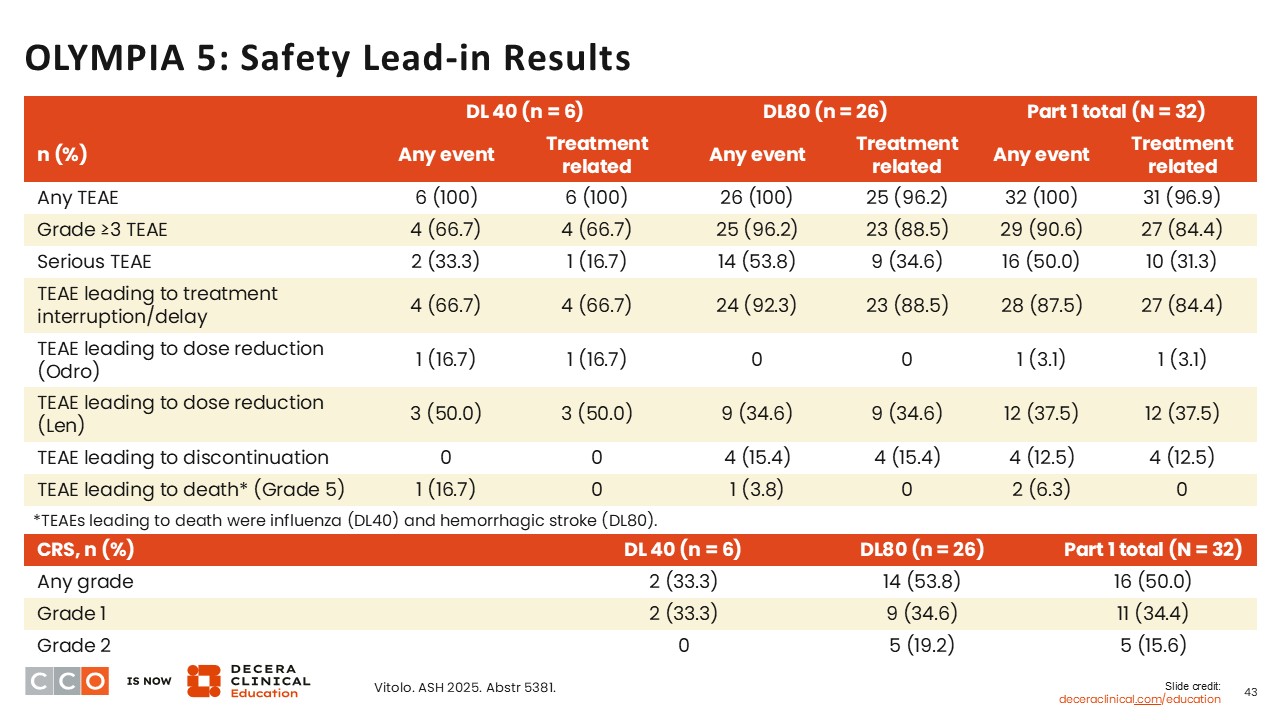
OLYMPIA 5: Safety Lead-in Results
Jennifer Crombie, MD:
In the Part 1 study lead-in, most patients on any dose of intravenous odronextamab experienced a treatment-emergent AE and most of these were grade ≥3. Most treatment-emergent AEs lad to either treatment interruption or treatment delay among patients. Few patients had a dose reduction in odronextamab. For CRS, 50% of patients reported this at any grade, most of which were grade 1 (34.4%).16
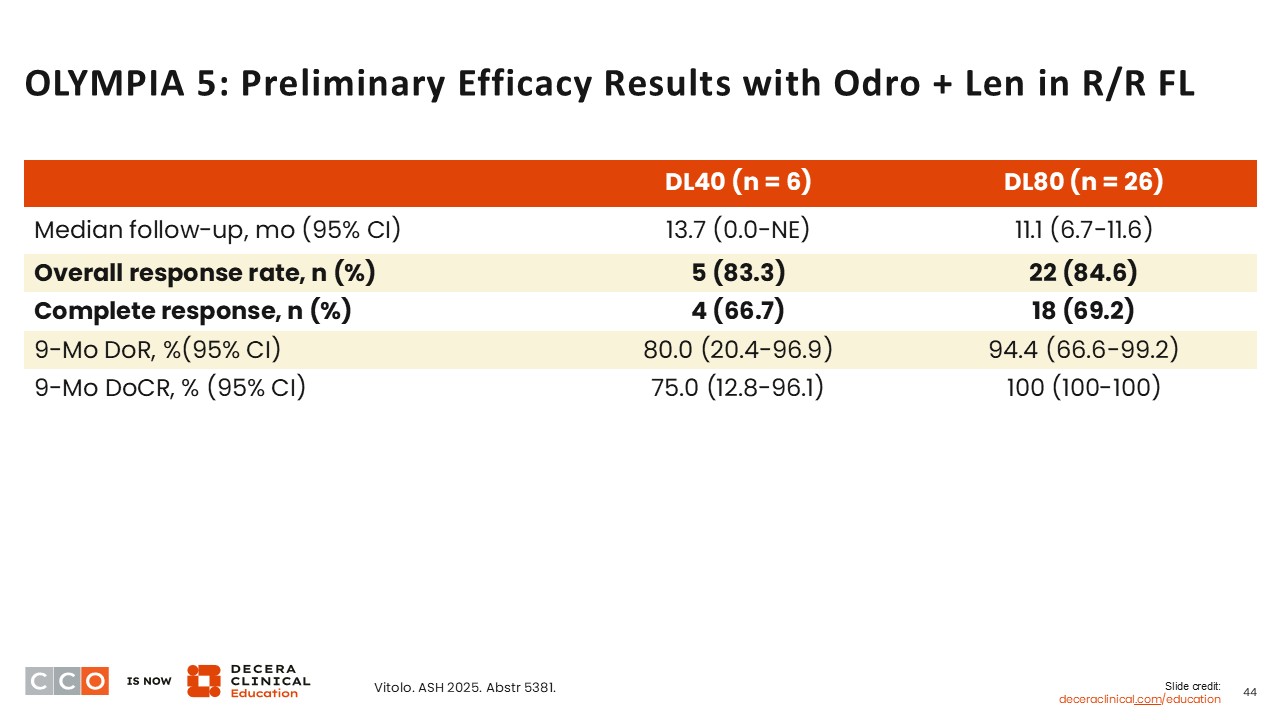
OLYMPIA 5: Preliminary Efficacy Results With Odro + Len in R/R FL
Jennifer Crombie, MD:
Preliminary efficacy results from the phase III OLYMPIA-5 trial showed an ORR of 83.3% among those at the 40-mg dose of odronextamab. For those at 80-mg dose of odronextamab, the ORR was similar at 84.6%. CRs were similar as well at 66.7% and 69.2%, respectively.16
Key Differences Between Investigational and Approved Bispecific Antibodies
Jennifer Crombie, MD:
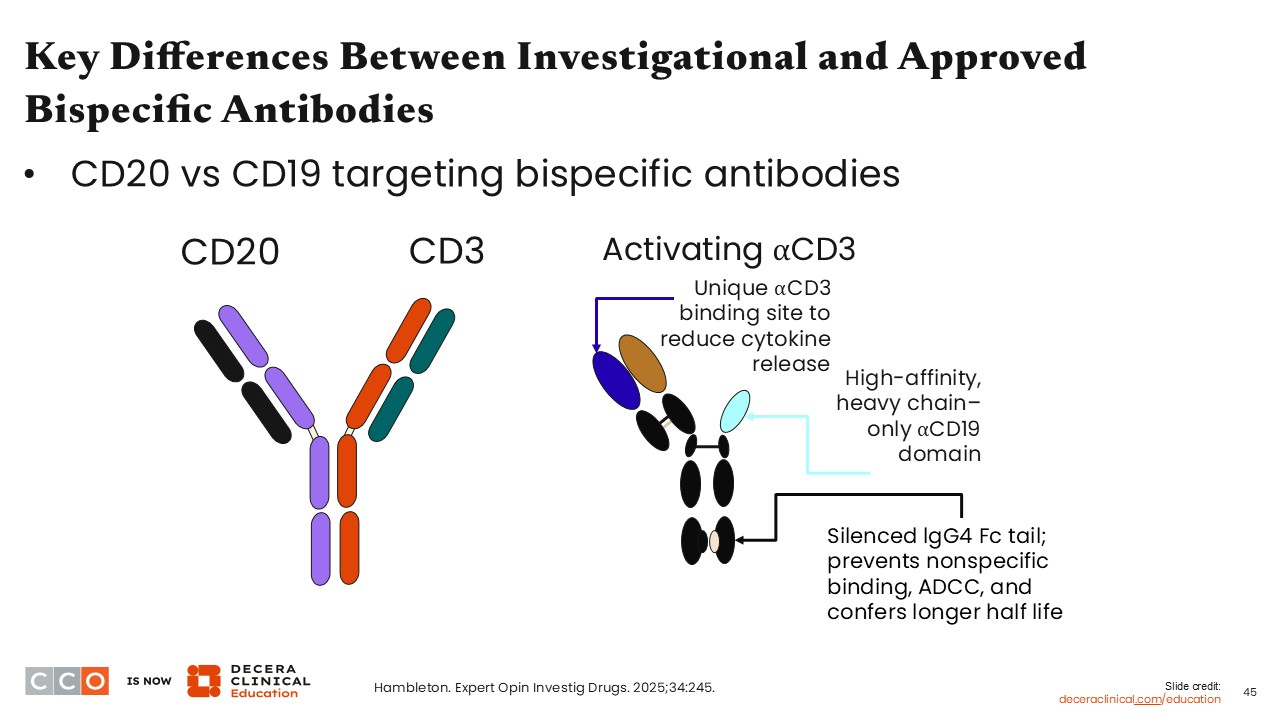
In addition to CD20, CD19 is a possible target for bispecific antibody and also is used as the target for some CAR T-cell therapies for B-cell malignancies.
Surovatamig is a CD19 x CD3–targeted bispecific therapy that is currently being evaluated in clinical trials. This bispecific antibody was designed to limit the rate of CRS by using a unique ⍺CD3 binding site, and a silenced lgG4 Fc tail was developed to prevent nonspecific binding, antibody-dependent cell-mediated cytotoxicity, and improve the drug’s half-life.17
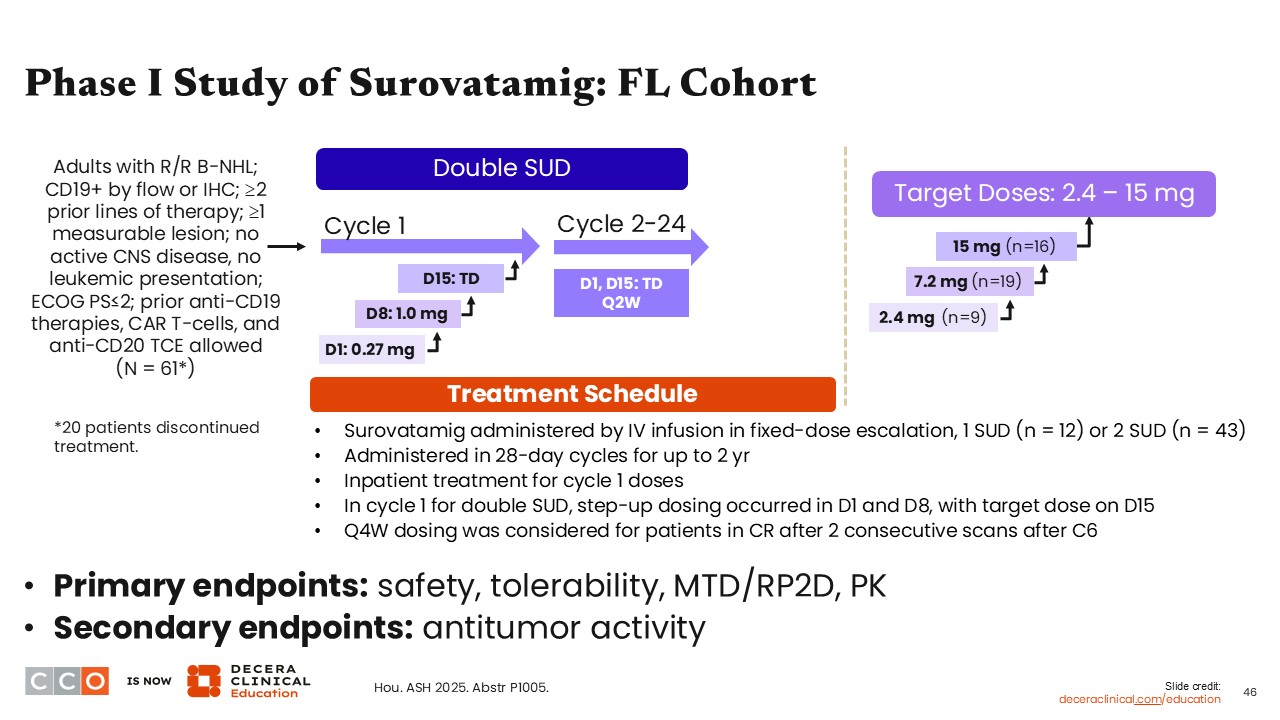
Phase I Study of Surovatamig: FL Cohort
Jennifer Crombie, MD:
Currently, intravenous surovatamig is being evaluated in a phase I dose optimization and expansion study at the recommended phase II dose in patients with CD19+ B-cell non-Hodgkin lymphoma after ≥2 prior lines of therapy. The different doses being evaluated here include the step-up dosing that starts at 0.27 mg and a double step-up dosing schedule. The target dose is 15 mg (NCT04594642).13
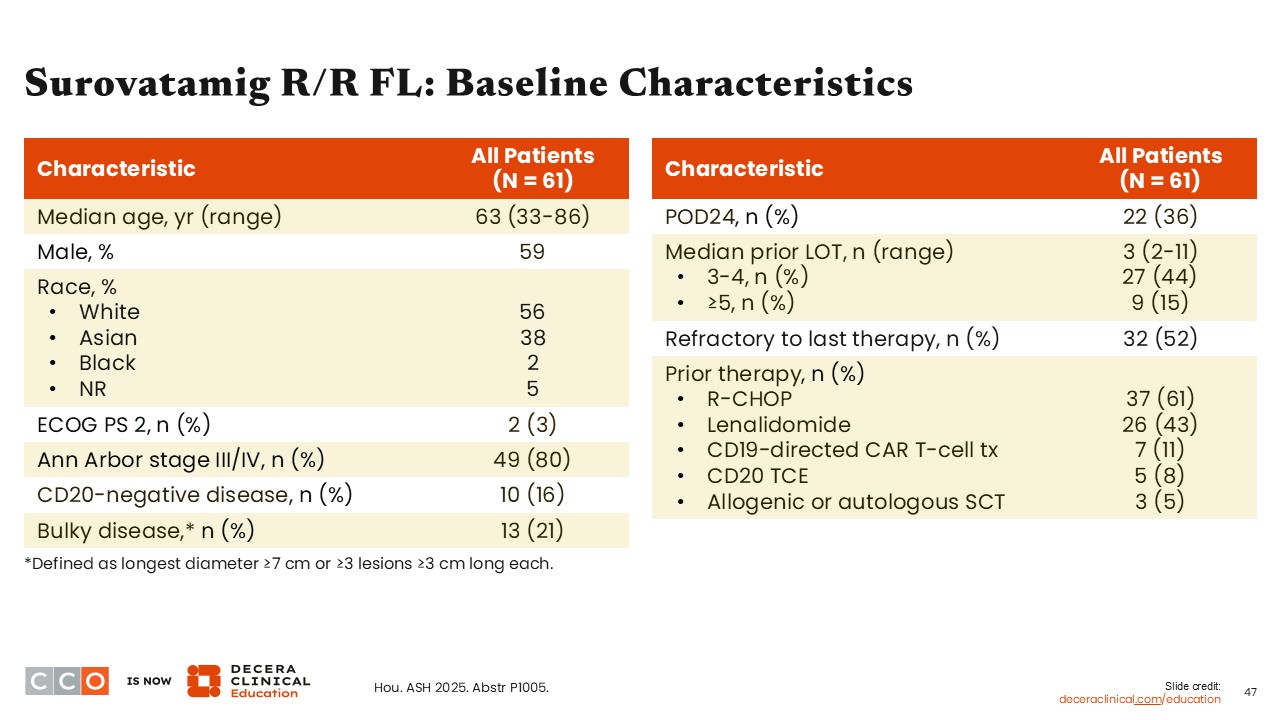
Surovatamig R/R FL: Baseline Characteristics
Jennifer Crombie, MD:
Overall, 61 patients were enrolled in the FL cohort, and 41 received a dose of 2.4 mg or higher. These patients were considered high risk: 36% were POD24, and 59% had received ≥3 prior lines of therapy. Of note, some patients had received prior CD20 bispecific agents as well as CD19-directed CAR T-cell therapy.13
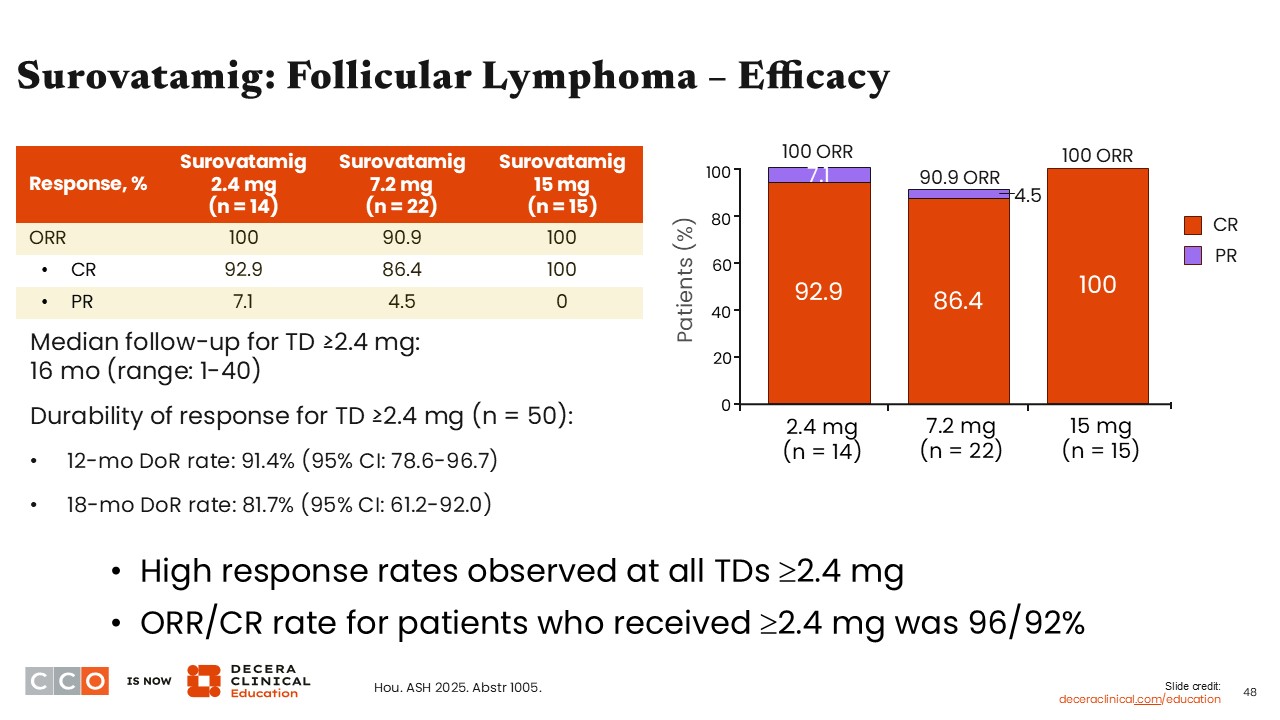
Surovatamig: Follicular Lymphoma—Efficacy
Jennifer Crombie, MD:
In the cohort of patients who received at least 2.4 mg of surovatamig, the ORR was 100% with 2.4 mg or 15 mg surovatamig and 90.9% with 7.2 mg. In the cohort that received 2.4 mg, the CR rate was 92.9% and the PR rate was 7.1%. For patients receiving surovatamig at 7.2 mg, the CR rate was 86.4% and the PR rate was 4.5%. For patients receiving surovatamig at 15 mg, the CR rate was 100% and the PR rate was 0%. Overall in this cohort of patients, the 12-month duration of response rate was 91.4% (95% CI: 78.6%-96.7%) and the 18-month duration of response rate was 81.7% (95% CI: 61.2%-92.0%).13
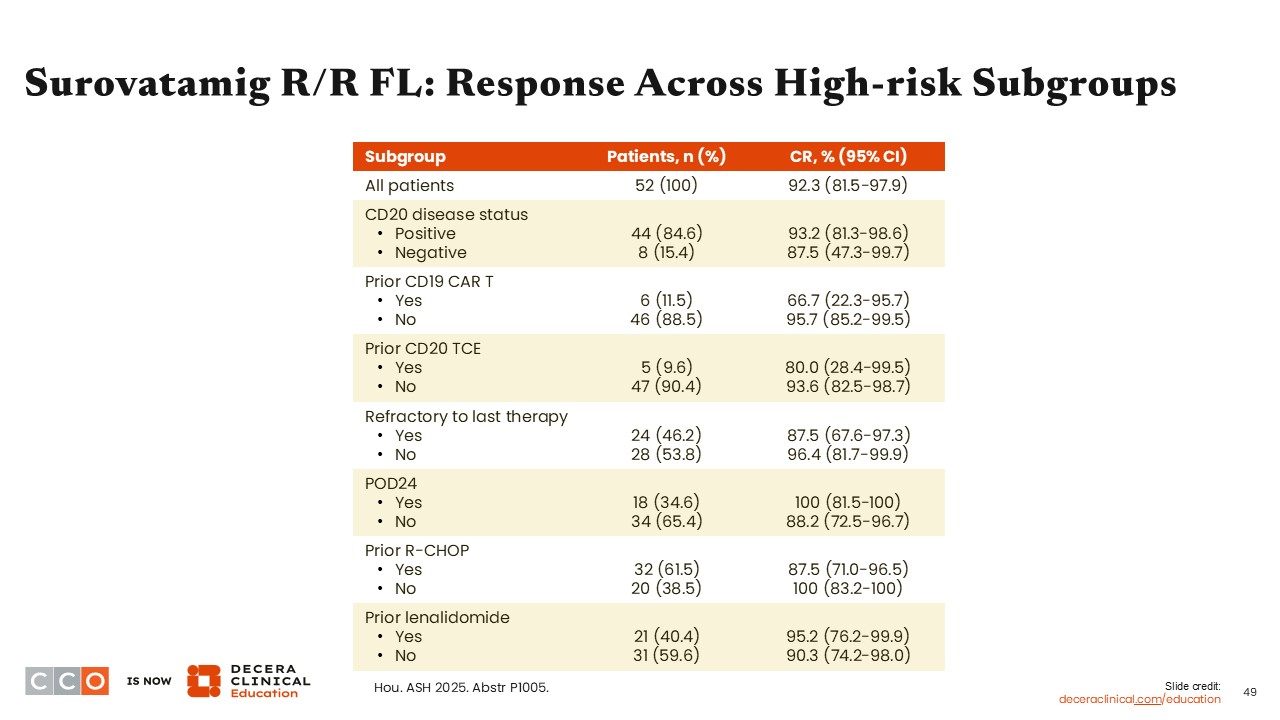
Surovatamig R/R FL: Response Across High-Risk Subgroups
Jennifer Crombie, MD:
This table shows the range in CR among patients with R/R FL who were treated with surovatamig and belong to various subgroups. For example, patients who were refractory to their last therapy had a slightly reduced CR (87.5%) compared with those who were not refractory to their last therapy (CR: 96.4%).13
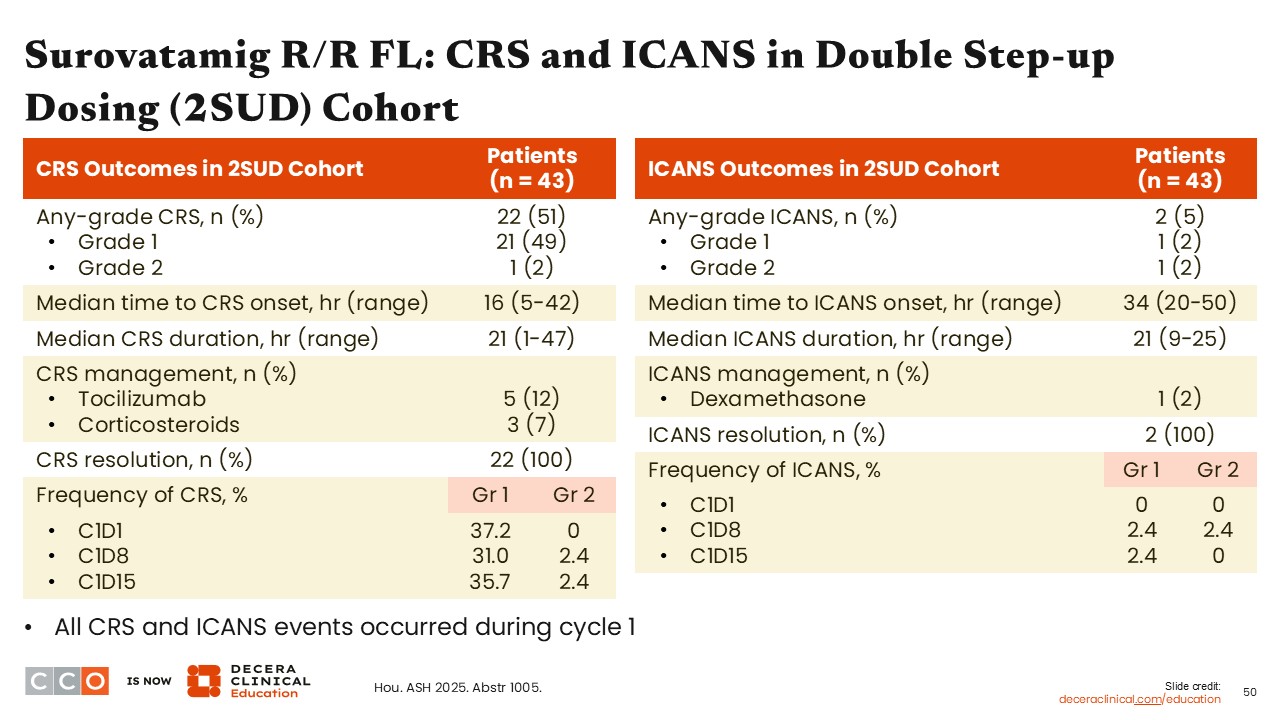
Surovatamig R/R FL: CRS and ICANS in Double Step-up Dosing (2SUD) Cohort
Jennifer Crombie, MD:
When looking at the safety of surovatamig at the recommended phase II dose, approximately one half of all patients developed CRS (49% grade 1 CRS, 2% grade 2, and no grade ≥3 CRS events). Similar to the data with the other bispecific antibodies in R/R FL, the frequency of developing ICANS was low, with only 2 patients developing ICANS.13
Key Differences Between Investigational and Approved Bispecific Antibodies
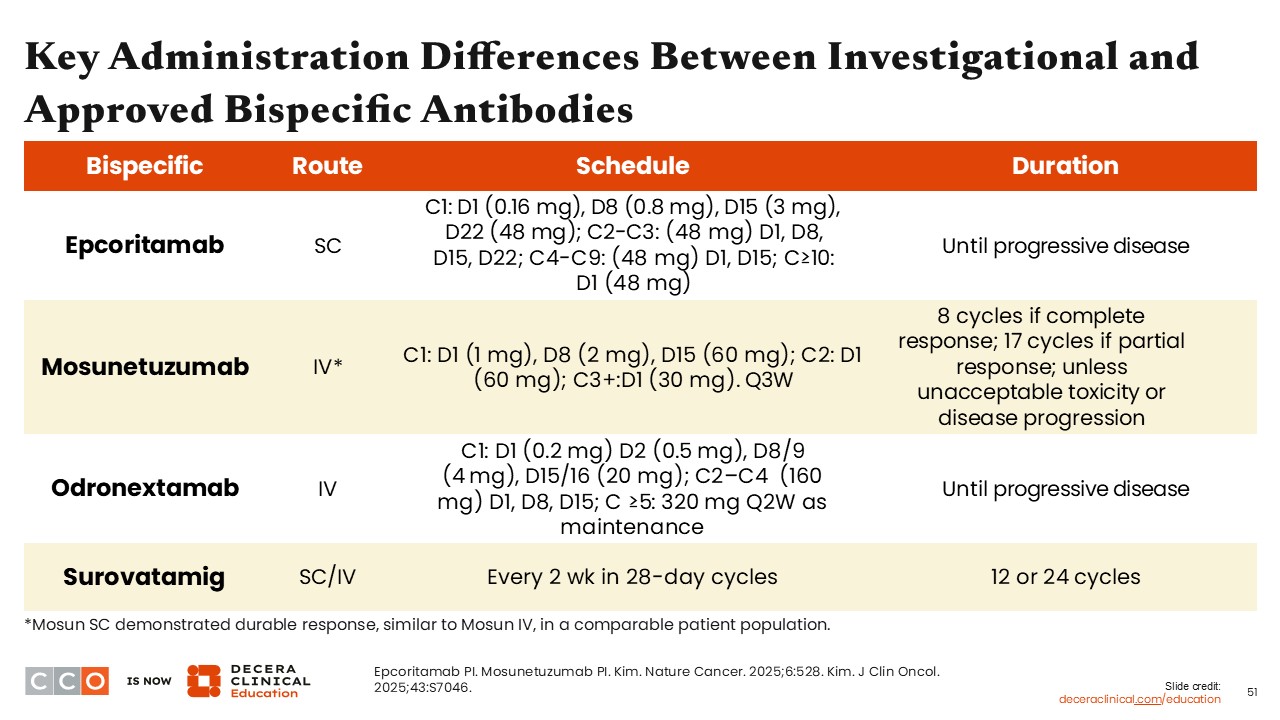
Jennifer Crombie, MD:
As discussed, the table features some key differences among the bispecific antibodies. These agents vary in route of administration, their dosing schedules, and treatment duration. A key difference is that some drugs are time limited, and some are given until disease progression. Patients receive odronextamab and epcoritamab until disease progression, whereas surovatamig and mosunetuzumab are time-limited options.
More specifically, epcoritamab is administered subcutaneously with a step-up dosing strategy during Cycle 1 and a consistent 48 mg dose thereafter, given weekly to monthly until disease progression. In contrast, when mosunetuzumab is administered intravenously, it has a complex step-up dosing schedule in the first two cycles followed by 30 mg every three weeks starting in Cycle 3. Its treatment duration depends on response—8 cycles for complete response and up to 17 for partial response, unless toxicity or progression. For the subcutaneous injection, it is administered for 8 cycles, unless the patient progresses or has unacceptable toxicity. For the subcutaneous dose, it is injected at 5 mg on day 1, 45 mg on day 8 and 45 mg on day 15 in the first cycle. In the second and subsequent cycles, it is injected at 45 mg on day 1 of the cycle.
Odronextamab, given intravenously, uses an accelerated step-up dosing schedule during Cycle 1, followed by fixed dosing in Cycles 2–4 and biweekly maintenance from Cycle 5 onward until disease progression. Surovatamig can be administered either subcutaneous or intravenous and is administered every two weeks in 28-day cycles, with treatment duration capped at either 12 or 24 cycles. Overall, understanding these distinctions is critical for clinicians to optimize therapy selection, manage toxicity, and align treatment logistics with patient preferences.2,3,18
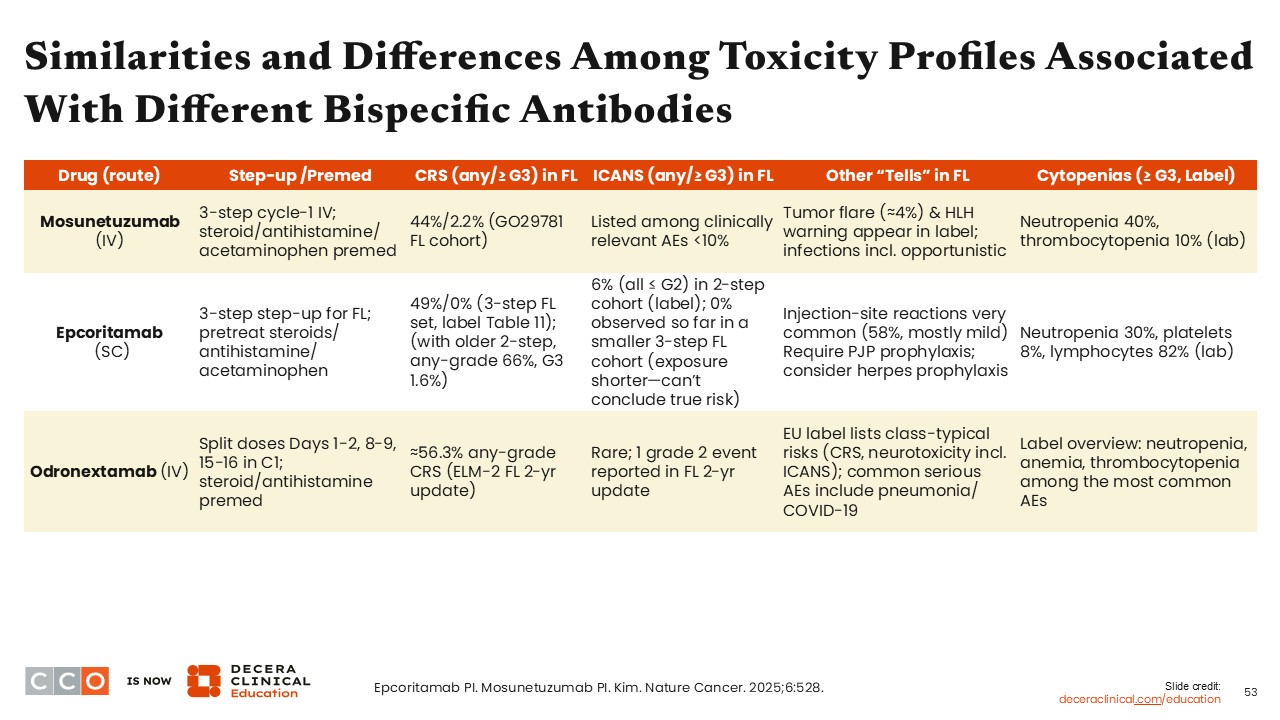
Similarities and Differences Among Toxicity Profiles Associated With Different Bispecific Antibodies
Farrukh T. Awan, MD, MS, MBA:
Managing and mitigating the AEs associated with bispecific antibodies can lead to significant improvement in patient compliance and outcomes. Although there are no head-to-head trials comparing the AEs among agents, we can look to cross-trial comparisons to help us understand each of the different AE profiles. For example, subcutaneous drug administrations are associated with injection-site reactions, which are relatively common and expected.
Overall, bispecific antibodies are associated with a risk of CRS and unique AEs based on the agents’ mode of administration.
Regarding mosunetuzumab, up to 44% of patients in the GO29781 study experienced CRS, and <10% experienced neurotoxicity, another less common but serious AE observed with these immunomodulatory treatments.2 Other AEs that occur with mosunetuzumab are tumor flare and hemophagocytic lymphohistiocytosis (or HLH), which need to be monitored very closely. Hemophagocytic lymphohistiocytosis is a life-threatening syndrome caused by an excessive overactivation of the immune system.2,18
With odronextamab, which is approved in Europe but not currently in the United States, CRS rates reach >56% but ICANS were rare with 1 grade 2 event reported in an update. Patients very rarely experience neurotoxicity with odronextamab.18
For all these bispecific antibodies, CRS can be mitigated by the step-up dosing regimens employed in these trials, along with a strong prophylactic strategy including steroids, antihistamine, and acetaminophen. Neurotoxicity was observed in a small percentage of patients but can be mitigated with various ramp-up dosing schedules.
All of these agents are also associated with a significant risk of cytopenias, specifically neutropenia and thrombocytopenia.
Cytokine-Release Syndrome
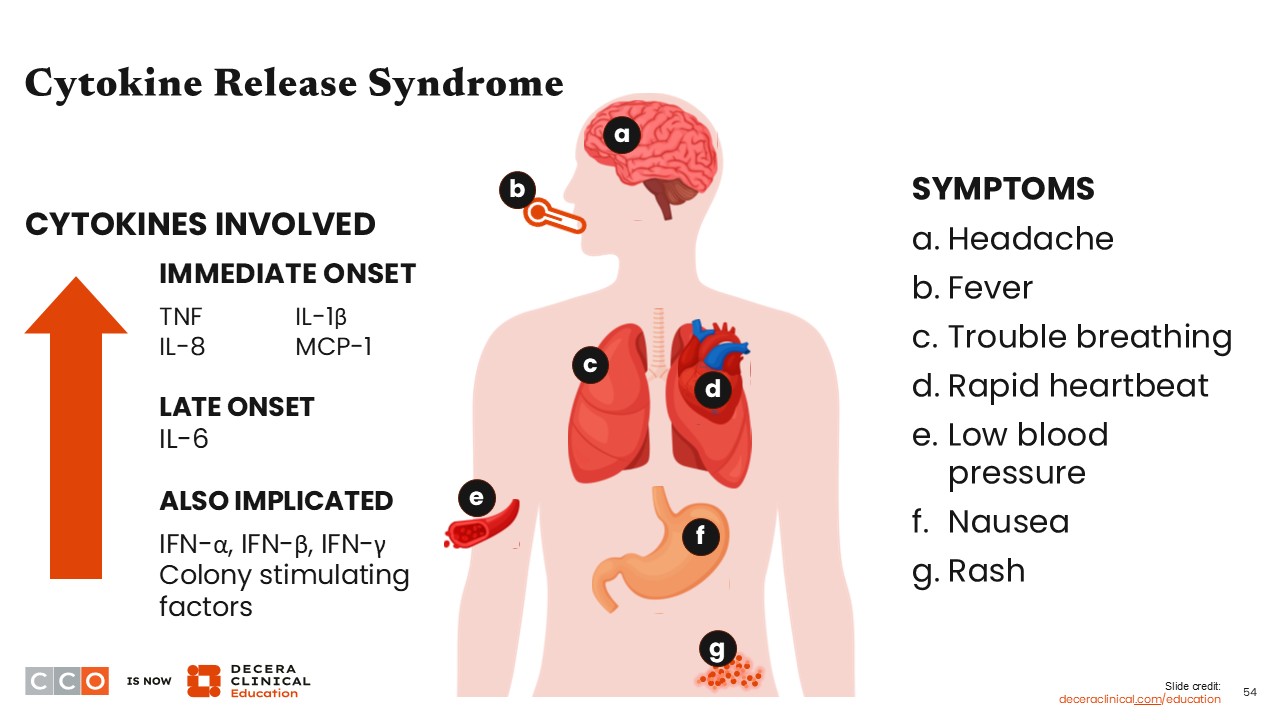
Farrukh T. Awan, MD, MS, MBA:
CRS is one of the more common AEs seen in patients receiving bispecific antibodies and results from nonspecific, broad activation of T-cells and the immune system in the tumor microenvironment. The activation of the immune system results in an elevation of various cytokines that cause the symptoms described during CRS.
One of the most common symptoms resulting from CRS is fever, primarily because of the increased concentration of interleukin-6. The combination of rising cytokines at different rates and concentrations can result in a milieu of symptoms that the patient experiences, ranging from fevers (the most common) to shortness of breath, tachycardia, hypotension, nausea, rash, and headache, among others.
Cytokine Release Syndrome: ASTCT Grading
Farrukh T. Awan, MD, MS, MBA:
The American Society for Transplantation and Cellular Therapy released a consensus guideline for grading CRS based on the presence of fever, hypoxia, and subsequent hypotension. Grade 1 CRS is defined as a temperature ≥38°C (or ≥100.4°F) but not hypoxia or hypotension. Grade 2 CRS includes a fever and hypoxia or hypotension not requiring vasopressors. Grade 3 CRS includes a fever and hypoxia requiring high-flow nasal cannula, nonrebreather mask, or Venturi mask or hypotension requiring 1 vasopressor. Grade 4 CRS includes fever and hypoxia requiring positive pressure or hypotension requiring multiple vasopressors (either 1 or multiple vasopressors, often associated with worsening shortness of breath requiring mechanical ventilation). These cases are typically managed in an intensive care unit.19
Cytokine-Release Syndrome: Management
Farrukh T. Awan, MD, MS, MBA:
There are some strategies that HCPs can employ to help mitigate these effects. CRS is seen in almost 50% of patients who receive a bispecific antibody, and patients can be stratified based on CRS risk.
One of the most predictive risk factors for CRS is tumor burden at the time of treatment, and patients can potentially benefit from early intervention. Close monitoring and ramp-up dosing can help make it fairly predictable when to expect a CRS event—typically during or immediately after the first full dose. These patients can be monitored as inpatients, especially if they are considered high risk for developing CRS.
Because of different severities, the management of CRS is geared to the underlying issue or exact symptom. For example, patients who experience grade 1 CRS (fever with no hypoxia or hypotension) can often be managed with antipyretics. They should also have a workup performed to rule out any infectious complications, since many of these patients have concomitant neutropenia and are at a higher risk of developing secondary infections.
Tocilizumab can also be considered in patients who have fevers that do not respond to antipyretics or in patients who may be considered high risk and have a high disease burden (ie, those who get fevers early in the course of therapy).12
Cytokine-Release Syndrome: Management
Farrukh T. Awan, MD, MS, MBA:
Patients can be premedicated with acetaminophen, antihistamines, and steroids prior to the bispecific antibody, which can mitigate the incidence of CRS in some cases. Patients also should receive prophylactic anti-infective medications, including acyclovir. Pneumocystis jirovecii pneumonia prophylaxis can be considered on a case-by-case basis for these patients.
Similarly, for grade ≥2 CRS, the management is escalated based on the patient. In patients who experience hypotension, hypoxia, and fever, in addition to tocilizumab, steroids and hydration can be used to manage symptoms.
Vasopressors can be employed with aggressive hemodynamic monitoring to manage any hypotension that patients may experience if CRS increases to grade 3/4. High doses of steroids, equivalent to methylprednisolone 1 g/day, also can be used in cases where all other conventional measures of steroids and tocilizumab have not been effective and patients continue to require higher doses of pressor support or additional pressor support for refractory hypotension.
Patients also need to be monitored very closely for organ dysfunction, and they can benefit from aggressive supportive care and frequently require care in the ICU setting. Siltuximab, an interleukin-6 antagonist, can also be considered in select cases in the absence of an adequate response to tocilizumab, especially when patients have refractory fevers and worsening CRS.12
Focus on Neurotoxicity: ICANS
Farrukh T. Awan, MD, MS, MBA:
Similar to CRS, neurotoxicity is a known AE associated with bispecific antibody therapy. ICANS is the predominant neurotoxicity seen with bispecific antibody therapy, and there are consensus criteria for how ICANS is graded to determine severity. Multiple grading criteria have been used, and many schedules have been used to assess patients’ neurologic function. These can differ between outpatient and inpatient settings. Regardless of whether patients start experiencing an altered mental status, findings can be subtle or very pronounced.
Patients who experience ICANS can be treated with steroids and neurologic interventions, including monitoring in the ICU, neurology consults, and additional use of specific interventions like anakinra and antiseizure medications to control worsening or limit the severity of these complications, on a case-by-case basis. Fortunately, for patients receiving bispecific antibodies, the incidence of neurotoxicity is fairly low.12
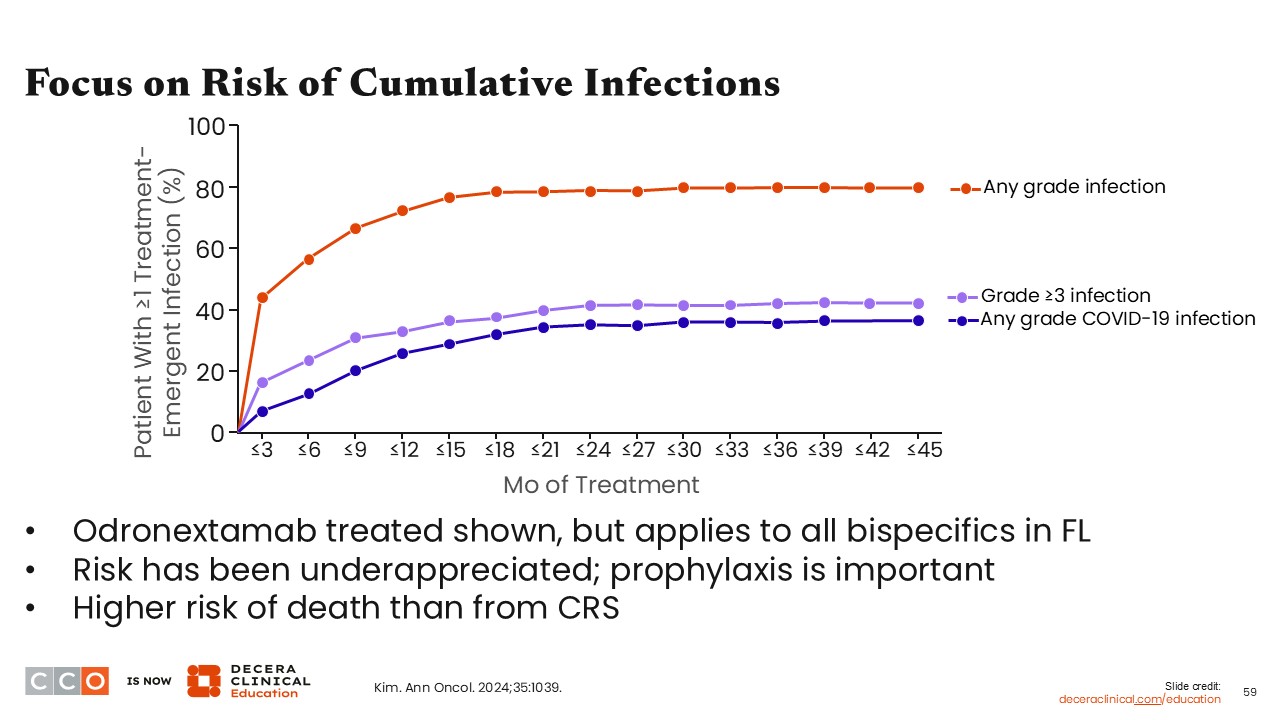
Focus on Risk of Cumulative Infections
Farrukh T. Awan, MD, MS, MBA:
Infectious complications are well recognized with the use of bispecific antibodies. Almost one half of all patients who are treated with bispecific antibodies experience an infectious complication. Fortunately, a significant percentage is minor. However, a substantial percentage may require parenteral antibiotics and aggressive supportive care.9
HCPs should not assume that all fevers during bispecific antibody treatment are because of CRS, since patients can have infectious complications, making diagnosis complex. Approximately 50% of infections associated with bispecific antibody therapy occurs within the first 3-6 months of treatment, with additional infections occurring long term.
Therefore, patients need to be monitored aggressively for secondary infections even when they have fever, which can be difficult to distinguish from CRS. HCPs should be aware of ongoing issues regarding COVID-19 infections and potential treatments for their complications
With close monitoring and support from a collaborative team, these patients can remain compliant and achieve a good outcome for their disease.
Considerations for Managing Infections With Bispecific Antibodies
Farrukh T. Awan, MD, MS, MBA:
Managing infections in patients receiving bispecific antibodies requires a proactive and multidisciplinary approach. One of the primary concerns with bispecific antibodies is B-cell aplasia, which can lead to hypogammaglobulinemia and increased infection risk. Patients with hypogammaglobulinemia may benefit from regular monitoring of IgG levels, with IVIG replacement initiated for levels below 400 mg/dL, depending on clinical need. To manage cytopenias, dose reductions and/or dose modifications, along with growth factor support, can improve the time on treatment.
Antimicrobial prophylaxis, particularly for herpes viruses and Pneumocystis jirovecii pneumonia, is another key consideration. Practices may vary, but prophylaxis is often guided by CD4+ cell counts, especially if they fall <200 cells/μL. Patients should also receive vaccinations on a regular basis or during the early stages of therapy. Doing this can prevent many infectious complications, although these patients typically tend to have poor vaccine responsiveness.
Summary and Final Conclusions
In conclusion, the details outlined in this module provided information and updates on the status of bispecific antibodies for use in R/R FL. Some agents are already approved in the United States or by the EMA, whereas others are in clinical trials. There are substantial other differences in the dosing, administration, and management with these agents for you to consider when incorporating into your practice. Clinical recommendations on the treatment-related AEs and management guidelines are presented for HCPs to make independent and informed treatment decisions. Being up-to-date can facilitate a safe and effective integration of bispecific antibodies into your clinic.