CE / CME
Signs and Symptoms of Cachexia and Optimizing Diagnostic Tools to Assess, Measure, and Document Cancer Cachexia
Physician Assistants/Physician Associates: 0.25 AAPA Category 1 CME credit
Pharmacists: 0.25 contact hour (0.025 CEUs)
Physicians: maximum of 0.25 AMA PRA Category 1 Credit™
Nurse Practitioners/Nurses: 0.25 Nursing contact hour
European Learners: 0.25 EBAC® CE Credit
Released: March 25, 2026
Expiration: September 24, 2026
Activity
What Is Cancer Cachexia?
Patients with cancer often face a complex range of physiologic challenges related to both the disease itself and its treatment. In addition to tumor control, healthcare professionals (HCPs) must address issues such as treatment-related adverse events (TRAEs), metabolic and inflammatory changes, nutritional status, functional decline, and overall quality of life (QoL). Effective management of patients with cancer, therefore, requires a coordinated, multidisciplinary approach that considers the many factors influencing patient health and treatment outcomes. Among these concerns, cancer cachexia represents an important but frequently underrecognized syndrome that can significantly affect prognosis, functional status, and tolerance to anticancer therapy.
Cancer cachexia is not a simple nutritional problem; rather, it is a multifactorial metabolic syndrome driven by complex interactions between the tumor and the host.1,2 In oncology practice, it frequently occurs without being recognized. The syndrome is characterized by a progressive loss of skeletal muscle mass, with or without fat loss. Cancer cachexia is not fully reversible by nutritional support alone. Key biologic drivers of cancer cachexia include tumor‑induced systemic inflammation, altered energy metabolism, and neurohormonal dysregulation.
Signs and Symptoms of Cancer Cachexia
Cachexia occurs most frequently in patients with advanced cancer, and it is associated with reduced treatment tolerance, diminished QoL, and poorer treatment outcomes. Although cancer cachexia is a common complication of cancer, it is often underrecognized and, therefore, not sufficiently addressed in routine oncology practice. Cachexia represents a major unmet clinical need in the management of patients with cancer, and of importance, when recognized early, treatment tolerance and patient outcomes are significantly improved.2
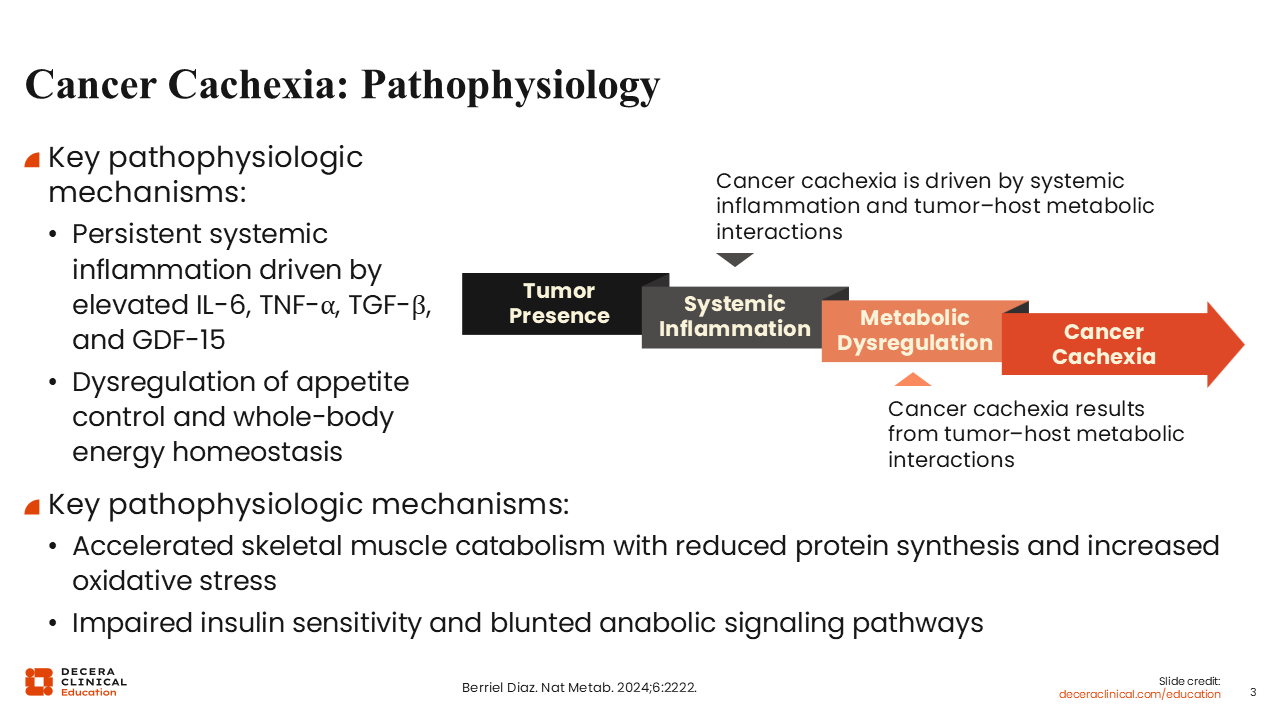
Cancer Cachexia: Pathophysiology
Multiple pathophysiologic mechanisms contribute to the development of cancer cachexia, involving a complex interplay of metabolic dysfunction and systemic inflammation. For instance, the development of cachexia involves persistent systemic inflammation driven by elevated levels of proinflammatory cytokines such as interleukin‑6 (IL‑6), tumor necrosis factor‑alpha, transforming growth factor‑beta, and growth differentiation factor‑15 (GDF‑15). Elevation in the levels of these cytokines is associated with dysregulation of appetite control and whole‑body energy homeostasis. In general, cancer cachexia is associated with accelerated muscle wasting, reduced protein synthesis, increased oxidative stress, and impaired anabolic signaling, particularly reduced insulin sensitivity.
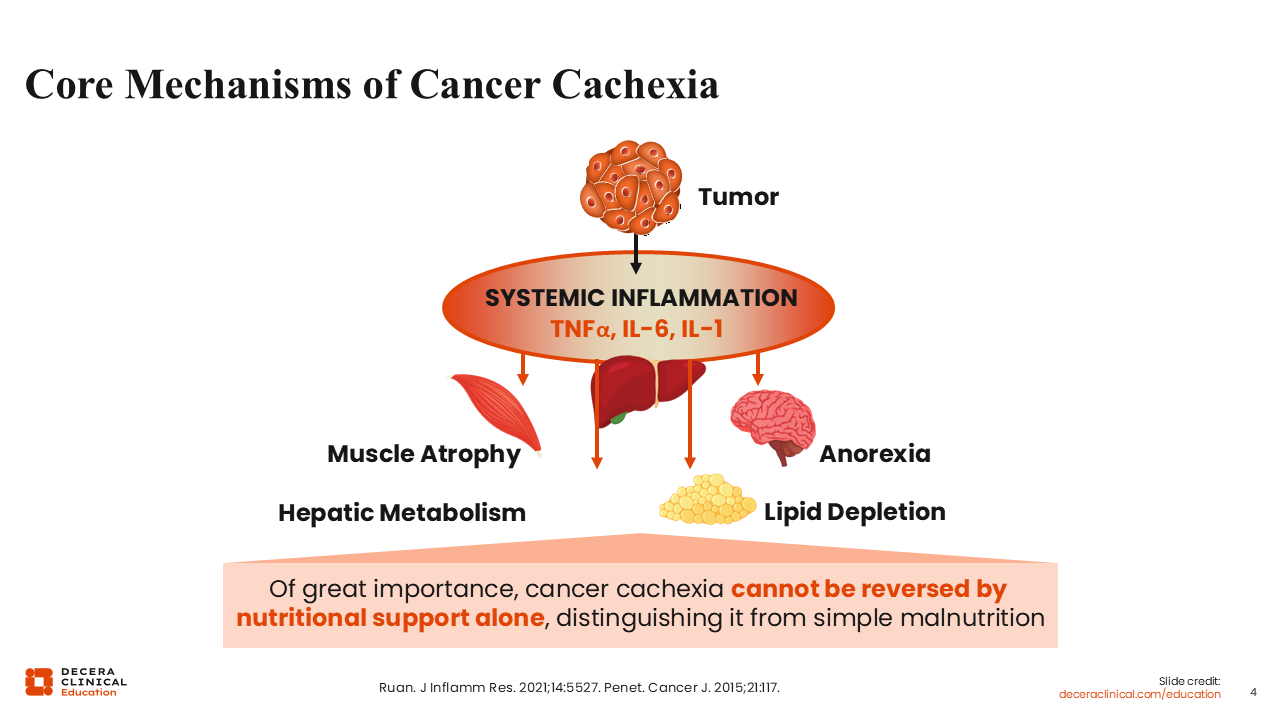
Core Mechanisms of Cancer Cachexia
Tumor‑induced systemic inflammation plays a central role in the development of cancer cachexia, acting through both central and peripheral mechanisms. Cytokine signaling mediates hepatic catabolic pathways, muscle atrophy and loss of adipose tissue. Both tumor and host immune cells produce cytokines, so, elevation of cytokine levels triggers weight loss, anorexia, fat breakdown, reduced insulin levels, and insulin resistance.3-6 Cytokine-driven metabolic changes may promote lipid breakdown, reduce lipid uptake and alter cholesterol metabolism. Of importance, cachexia is not the same as simple malnutrition; it may develop even when caloric intake appears adequate, and it is a major cause of morbidity and mortality among patients with advanced-stage cancer.
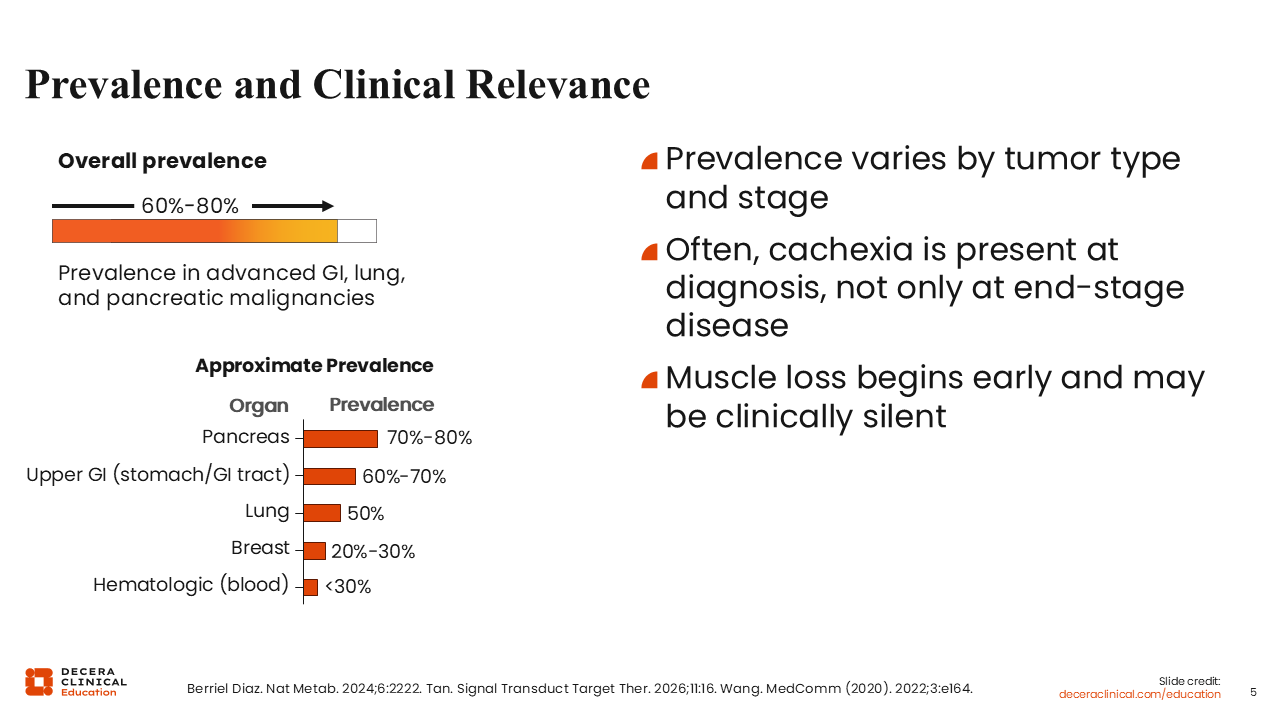
Prevalence and Clinical Relevance
Among patients with advanced gastrointestinal, lung, and/or pancreatic cancer, the incidence of cancer cachexia is approximately 60% to 80%.2,7,8 Its prevalence varies by tumor type and disease stage. Cancer cachexia is less frequently observed in patients with breast cancer or those with hematologic malignancies at diagnosis. Often, cancer cachexia is present at the time of diagnosis of advanced-stage cancer. That muscle loss may be clinically silent in the early stages of cancer cachexia makes its early recognition and diagnosis in patients with advanced cancer challenging.
Clinical Assessment and Diagnostic Criteria
Cancer cachexia develops along a clinical continuum of 3 stages of disease progression: precachexia, cachexia, and refractory cachexia.1,9-11 In the precachexia stage, patients experience early metabolic changes that may develop before mild weight loss of ≤5%. The potential to advance to the next cachexia stage is dependent on disease-related factors such as the type of cancer and stage, biologic factors such as systemic inflammation, and patient-specific factors such as minimal or low food intake and disease response to anticancer therapy.
Patients in the established cachexia stage experience unintentional weight loss of >5% within 6 months, while refractory cachexia develops as a result of advanced cancer or rapidly progressing cancer that is unresponsive to anticancer treatment. In general, patients with refractory cachexia have a low performance status and reduced life expectancy of <3 months.
Diagnostic hallmarks of cancer cachexia include >5% unintentional weight loss within 6 months, anorexia and/or early satiety, profound fatigue and weakness, and systemic inflammation such as increased CRP or decreased albumin.
Screening tools such as the Malnutrition Screening Tool (MST) and the Nutritional Risk Screening 2002 (NRS-2002) can be used at diagnosis and repeated before each anticancer treatment cycle. Also, miniCASCO is a simplified tool that can be used to classify cancer cachexia. It takes into consideration multiple factors including weight trajectory, inflammatory markers, level of physical activity, and overall well-being.
Cancer Cachexia: Symptoms to Look for
Of great importance, looking for weight loss alone as a sign is suboptimal because weight loss is a sign of late-stage cachexia. Early indicators of cancer cachexia include declining handgrip strength, reduced physical performance, and fatigue that is disproportionate to cancer burden. Performing laboratory tests to determine alterations in the levels of biomarkers such as CRP or albumin may also aid in the early diagnosis of cancer cachexia.
The development of cachexia significantly influences treatment outcomes for patients with cancer.11,12 Multiple studies have suggested that in patients with cachexia, there is a higher rate of chemotherapy dose reduction and anticancer treatment discontinuation.13,14 Response to immunotherapy was reduced in these patients, and sarcopenia plays a major role in cancer cachexia–related anorexia across several tumor types.11 Thus, the presence of cachexia contributes to intolerance to anticancer therapies, leads to a higher incidence of TRAEs, and may necessitate reductions in the dose of anticancer therapy being received by patients. Cancer cachexia is also associated with reduced physical activity/function, reduced QoL, and lower overall survival.11-14
Differentiating Cachexia From Overlapping Conditions
Cachexia is often confused with related but distinct conditions such as malnutrition, sarcopenia, and frailty.12,15-17 Malnutrition is primarily food intake driven and is potentially reversible with adequate nutritional support, whereas cachexia cannot be reversed by nutritional support alone. Sarcopenia may occur as a result of aging or inactivity. Although sarcopenia can serve as an assessment criterion for cachexia, it is important to note that cancer cachexia is tumor driven, mediated by inflammatory cytokines, and progressive.
Frailty is particularly prevalent in elderly people, characterized by a decline in physical activity and function due to the loss of physiological reserve, whereas cachexia is a severe wasting condition often caused by an underlying disease such as cancer, and is characterized by weight loss, muscle atrophy, and fat loss due to systemic inflammation.
Although these conditions (malnutrition, sarcopenia, frailty, and sarcopenia) have overlapping characteristics, cachexia requires a distinct diagnostic and therapeutic approach.
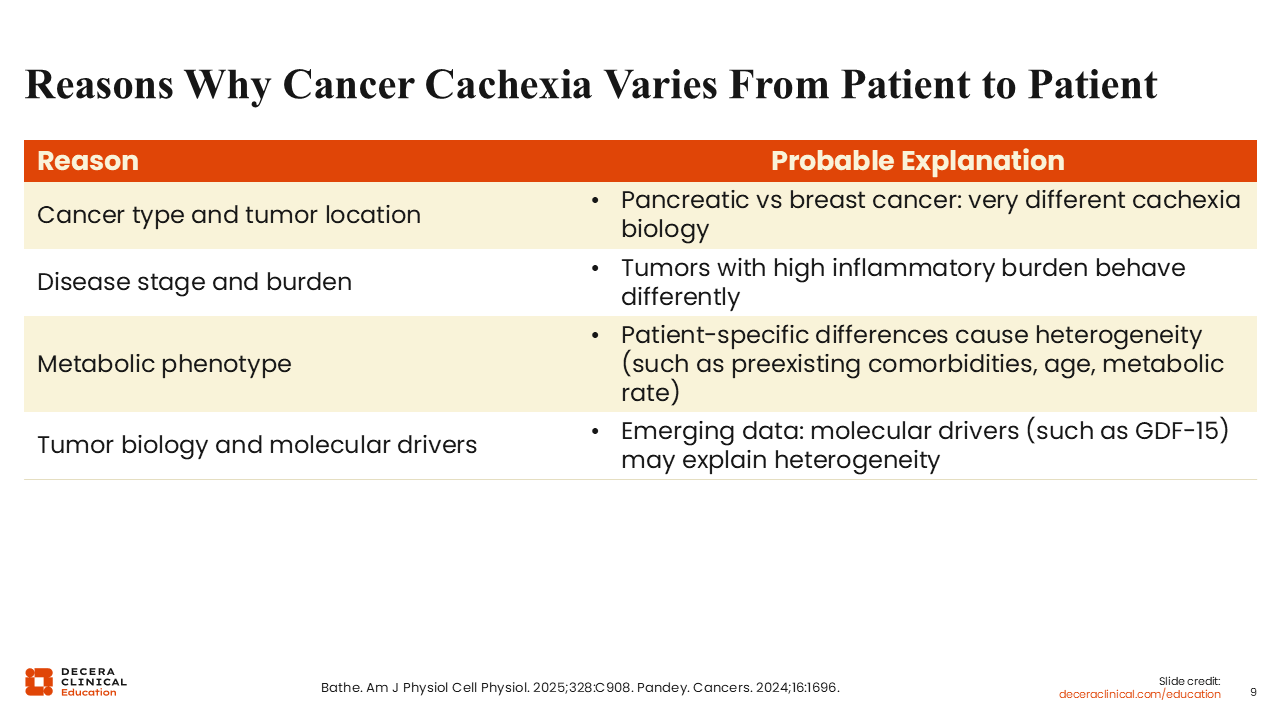
Reasons Why Cancer Cachexia Varies From Patient to Patient
The presentation of cancer cachexia is patient specific as it varies from one patient to the next.18-20 The severity and clinical presentation of cachexia depend on factors such as the type of cancer, the stage of the cancer, tumor burden, sites of cancer metastases, anticancer therapy previously exposed to, tumor biology, and molecular drivers.
Patient-specific differences such as preexisting comorbid conditions, age, and fitness level also factor into the heterogeneity of cancer cachexia. For example, cachexia is more common among patients with pancreatic cancer, and these patients often demonstrate a markedly different profile with more severe, early-onset cachexia than patients with breast cancer, who typically experience cachexia in later stages of the disease.21,22 Emerging data also suggest that molecular drivers such as GDF‑15 may contribute to this heterogeneity.18
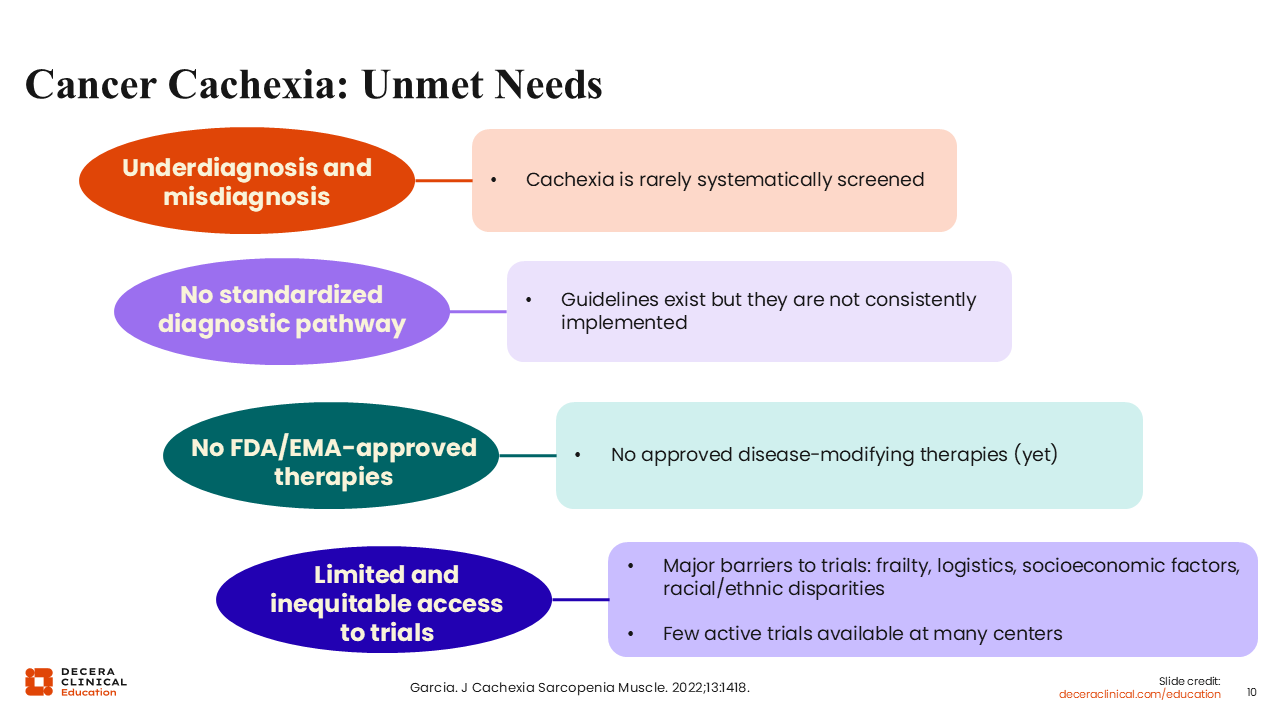
Cancer Cachexia: Unmet Needs
Cachexia remains a major unmet medical need in oncology practice. The condition is frequently underdiagnosed, misclassified, and insufficiently documented in clinical practice.23 Currently, no disease‑modifying therapies are approved by the FDA or the European Medicines Agency specifically for the treatment of cancer cachexia. In addition, participation in clinical trials may be limited due to factors such as patient frailty and socioeconomic or racial disparities. On these bases, cachexia is sometimes viewed as an inevitable consequence of cancer rather than a condition that can be actively diagnosed and appropriately managed, especially when diagnosed early.