CME
Next-Generation CDH6-Targeting Antibody–Drug Conjugates in Ovarian Cancer: The Science, Strategy, and Signals to Inform Clinical Readiness
Physicians: Maximum of 0.75 AMA PRA Category 1 Credit™
Released: January 26, 2026
Expiration: July 25, 2026
Activity
Cadherin-6 (CDH6)
Debra L. Richardson, MD: CDH6 is a protein that is lowly expressed on many healthy, normal tissues, but is overexpressed in several solid tumors including ovarian cancer, renal cell carcinoma (RCC), papillary thyroid cancers, cholangiocarcinoma, and uterine cancer.1-3 The role of this protein is unclear but it appears to mediate cancer progression and is associated with poorer survival outcomes in ovarian and renal cancer.2,4-6 Approximately 65% to 94% of epithelial ovarian cancer tumors express CDH6.4,7,8 Although the role of this protein is unclear, it offers a great opportunity to develop targeted therapies. In this text module, we will discuss the targeting of CDH6 in ovarian cancer.
The first ADC developed to target this protein was HKT288, which had a maytansine payload but was discontinued early in development due to unexpected neurologic toxicities in a phase I study.9-11 The mechanism for these toxicities was not understood and it was unclear if CHD6-targeted ADCs would be feasible, but more recent agents have shown promising results regarding efficacy and safety.
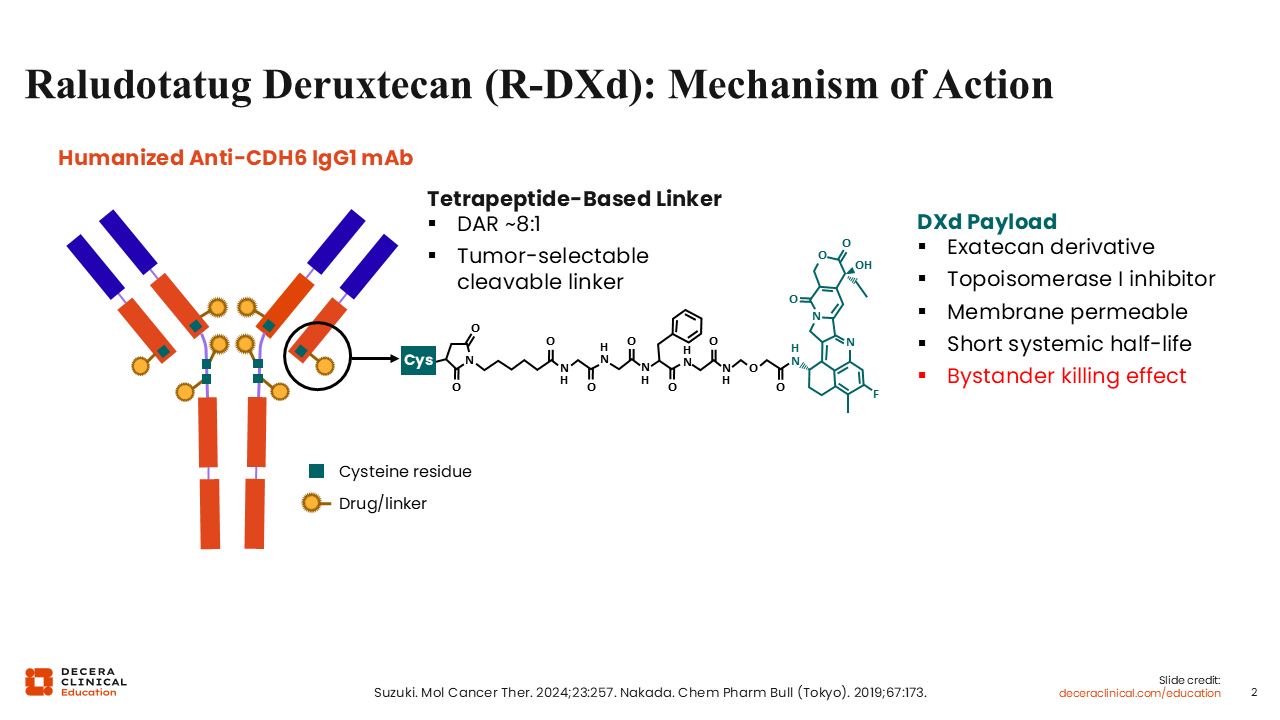
Raludotatug Deruxtecan (R-DXd): Mechanism of Action
Debra L. Richardson, MD: The CDH6-targeting agent with the most evidence in ovarian cancer is R-DXd, which has the deruxtecan topoisomerase I (TOPO-I) inhibitor payload present in other approved ADCs.10,12 R-DXd has a relatively high drug‑to‑antibody ratio of 8, a potent bystander effect, and a tumor-selective cleavable linker that is stable in circulation and decreases off‑target toxicity.
Phase I Dose Escalation/Expansion of R-DXd in Ovarian Cancer
Debra L. Richardson, MD: The dose-escalation stage of the first-in-human phase I study of R-DXd examined 6 different dose levels administered intravenously every 3 weeks in patients with advanced or metastatic ovarian cancer who were not amenable to standard-of-care treatment.13 Patients were previously treated with a taxane and platinum-based chemotherapy but could not have had prior treatment with a TOPO-I-linked ADC. Of importance, patients were not preselected based on CDH6 tumor expression level. The primary objectives were to determine the maximum tolerated dose and the recommended dose level for expansion. The study was originally designed to include patients with RCC, but this component was discontinued.
In the dose-expansion phase, there were 4 ovarian cancer cohorts with dose levels of 4.8, 5.6, 6.4, and 8 mg/kg, with a primary objective of overall response rate (ORR) by RECIST v1.1 criteria. A total of 156 patients were enrolled across dose levels.
Phase I R‑DXd: Baseline Characteristics
Debra L. Richardson, MD: The baseline characteristics were as expected with the majority of patients who received dose levels between 4.8 mg/kg and 8.0 mg/kg (91.7%) having platinum-resistant disease.13 Of note, the median number of prior regimens of 4 was relatively high, ranging from 1-13 prior treatments, so some of these patients were extremely heavily pretreated; 68.3% and 65.0% had prior bevacizumab or PARP inhibitor treatment respectively. Tumor CDH6 H-scores were measured at baseline and ranged from 0-250 with a median of 125.
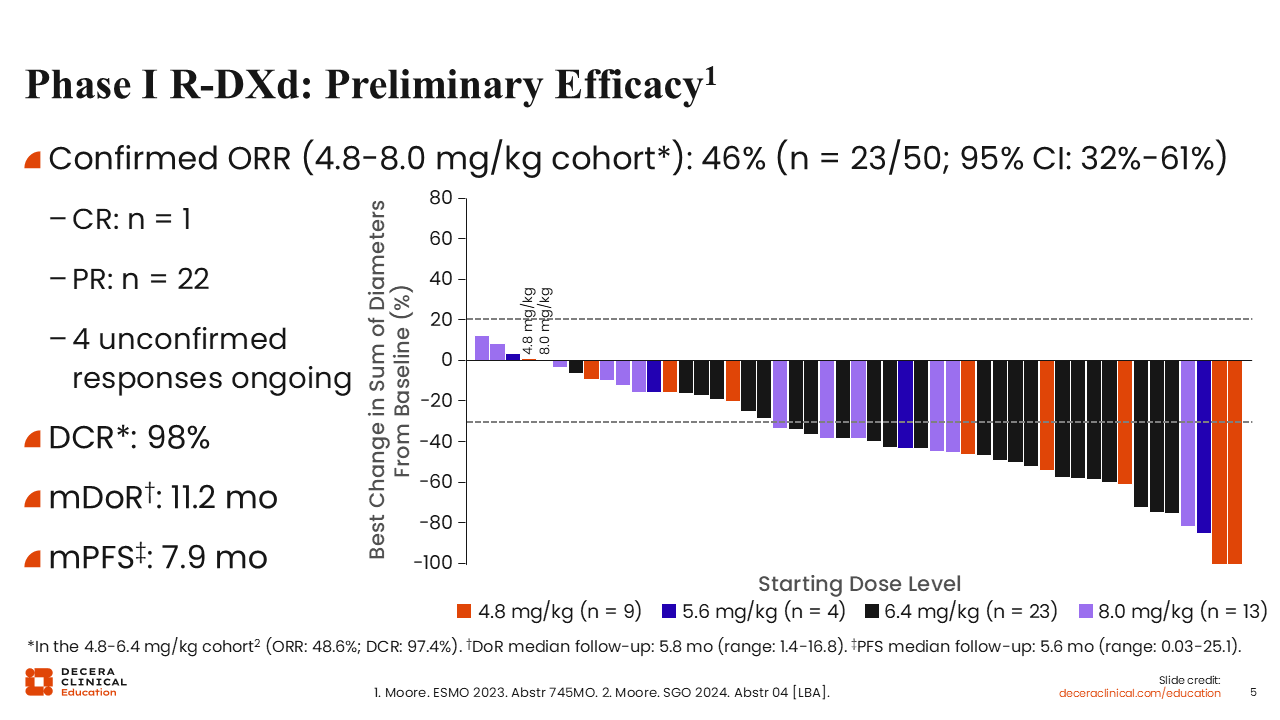
Phase I R‑DXd: Preliminary Efficacy
Debra L. Richardson, MD: The confirmed ORR in the 4.8-8.0 mg/kg cohort was 46%.13 There was 1 CR and 22 PRs, with 4 unconfirmed responses at date of cutoff (July 14, 2023). The disease control rate (DCR) was an impressive 98%. Median duration of response, with a median follow-up of 5.8 months, was 11.2 months and median progression-free survival (PFS), with a median follow-up of 5.6 months, was 7.9 months. Results were similar when analyzing just the 4.8-6.4 mg/kg cohort.14
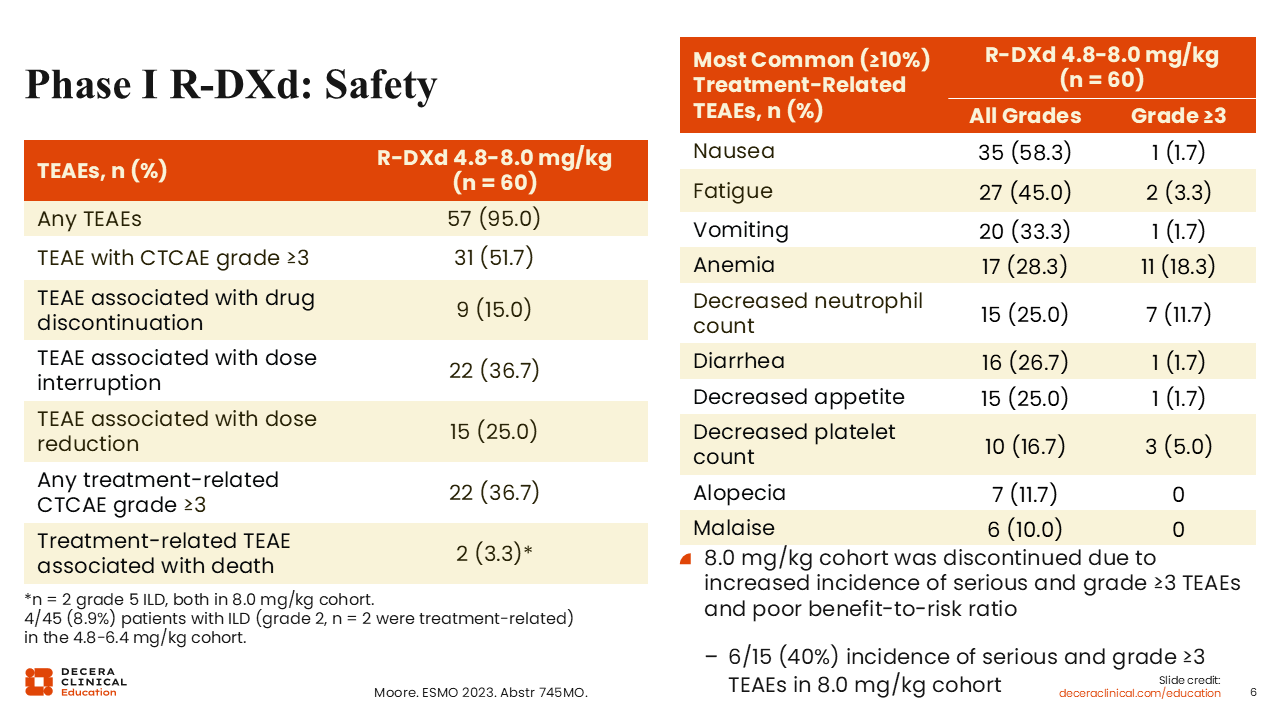
Phase I R‑DXd: Safety
Debra L. Richardson, MD: Regarding the safety results, it is important to remember that antiemetics are often restricted in the first cycle in phase I trials, as was the case for this study. So, although nausea and vomiting were among the most common treatment-related adverse events (TRAEs), they can be mitigated with antiemetics.13,14 Myelosuppression, such as decreased neutrophil and platelet count, was not a surprising effect of R-DXd due to its TOPO-I payload.
The most important takeaways from these data are that only 15.0% of patients discontinued treatment due to a TRAE and 25.0% required dose reduction, but there was a wide range of doses tested in this trial.
Of note, there were 2 treatment-related deaths associated with TEAEs (3.3%) which were both due to grade 5 ILD/pneumonitis and occurred in the 8.0 mg/kg cohort. This dose level was eventually discontinued due to high-grade toxicity without increased benefit relative to the other doses. In the remaining 4.8-6.4 mg/kg cohorts, 4 of 45 patients (8.9%) experienced ILD, all of which were grade 2 and 2 of which were treatment related.
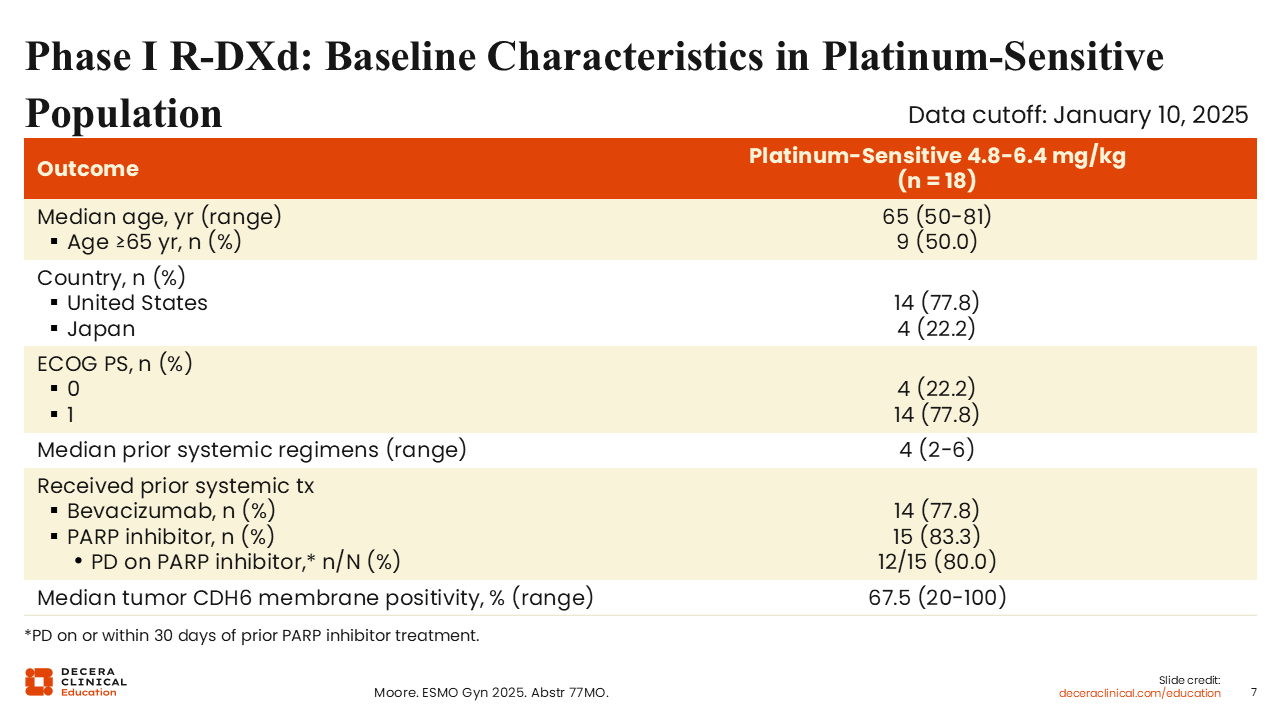
Phase I R‑DXd: Baseline Characteristics in Platinum‑Sensitive Population
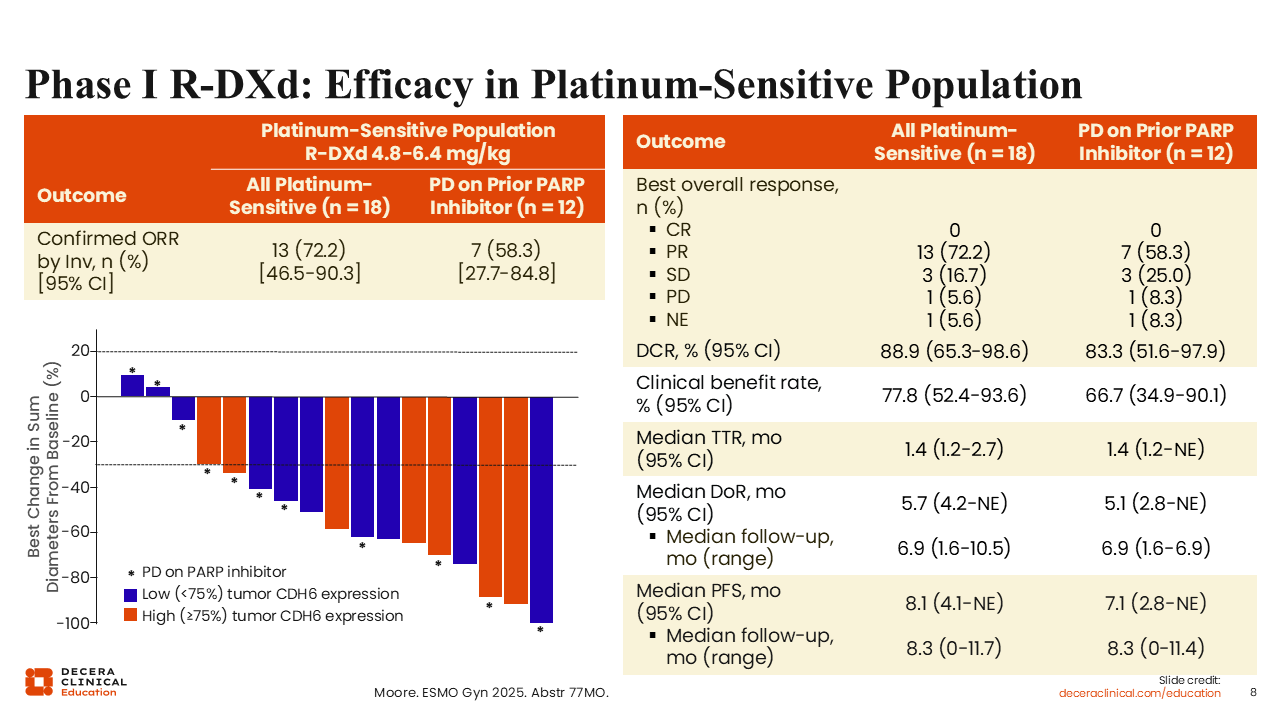
Isabelle Ray-Coquard, MD, PhD: In 2025, results of the phase I study were reported for the small subgroup of 18 platinum-sensitive patients who had experienced at least 6 months of being treatment free after their last dose of platinum chemotherapy. Only those treated with R-DXd 4.8, 5.6, and 6.4 mg/kg were included in the analysis.15 These patients were generally heavily pretreated with a median number of prior systemic treatments of 4. The majority of patients had prior bevacizumab and PARP inhibitor treatment (77.8% and 83.3%, respectively), and 80.0% of those with prior PARP inhibitors experienced progression on that treatment. The mechanisms of resistance for PARP inhibitors and platinum are related, so some healthcare professionals may be resistant to treating with platinum again in that setting. The median tumor CDH6 membrane positivity of 67.5% was relatively low.
Phase I R‑DXd: Efficacy in Platinum‑Sensitive Population
Isabelle Ray-Coquard, MD, PhD: The confirmed ORR was 72.2% (all PRs), which was relatively high and comparable to doublet platinum-based chemotherapy.15,16 The ORR for patients who progressed on a PARP inhibitor was also quite high at 58.3%. Median duration of response was 5.7 months for all platinum-sensitive patients and 5.1 months for those who progressed on a PARP inhibitor. Median PFS was 8.1 months and 7.1 months, respectively. This is a better result than we have seen with platinum-based chemotherapy in patients who had progressed on a PARP inhibitor.Harter 2024
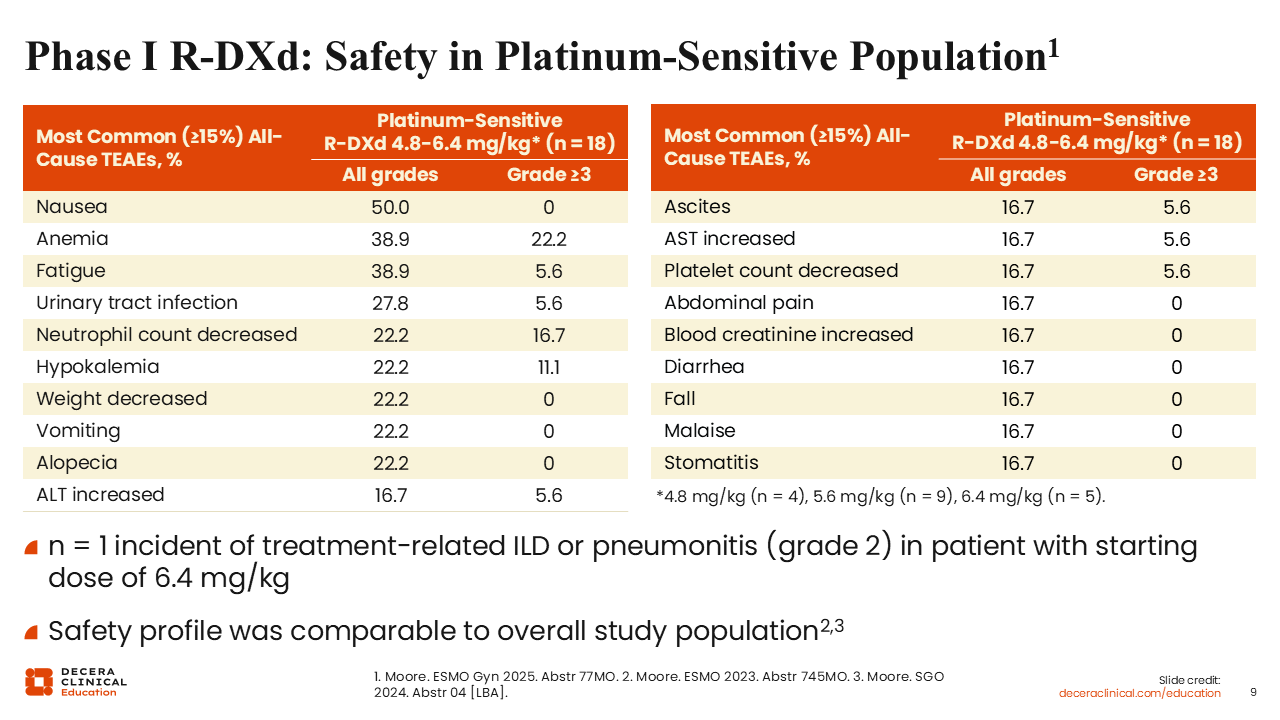
Phase I R‑DXd Safety in Platinum‑Sensitive Population
Isabelle Ray-Coquard, MD, PhD: The safety profile was similar to the overall study population who were primarily platinum resistant.13,14
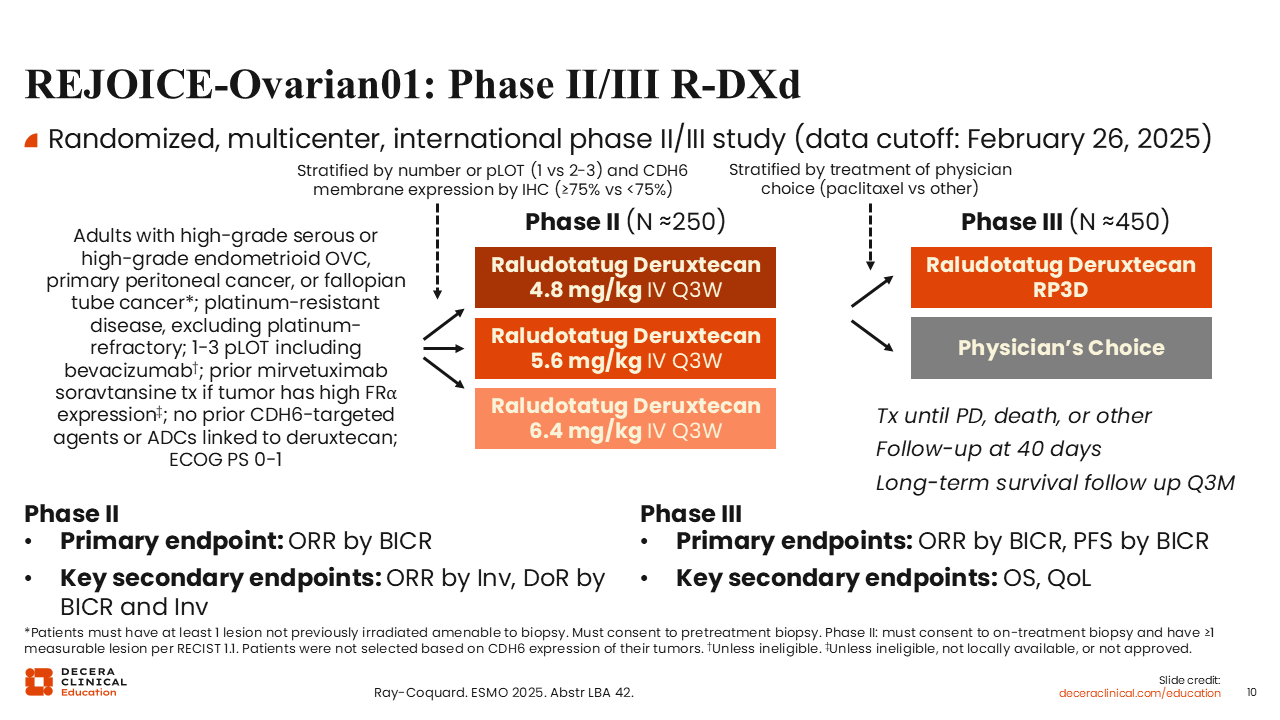
REJOICE‑Ovarian01: Phase II/III R-DXd
Isabelle Ray-Coquard, MD, PhD: The first phase II results of the phase II/III REJOICE-Ovarian01 study were reported at ESMO 2025.8 This study enrolled adult patients with platinum-resistant, high-grade serous or endometrioid ovarian cancer who had received between 1 and 3 prior lines of treatment, including bevacizumab. Prior mirvetuximab soravtansine treatment was allowed and patients were not selected based on the tumor CDH6 expression. Platinum-refractory patients were not eligible.
The dose optimization analysis of R-DXd at doses 4.8, 5.6, and 6.4 mg/kg examined 107 treated patients with primary endpoint of ORR by blinded independent central review. The phase III portion of the study will begin enrolling approximately 450 patients in early 2026 and will evaluate the recommended phase III dose of R-DXd vs physician’s choice of treatment.
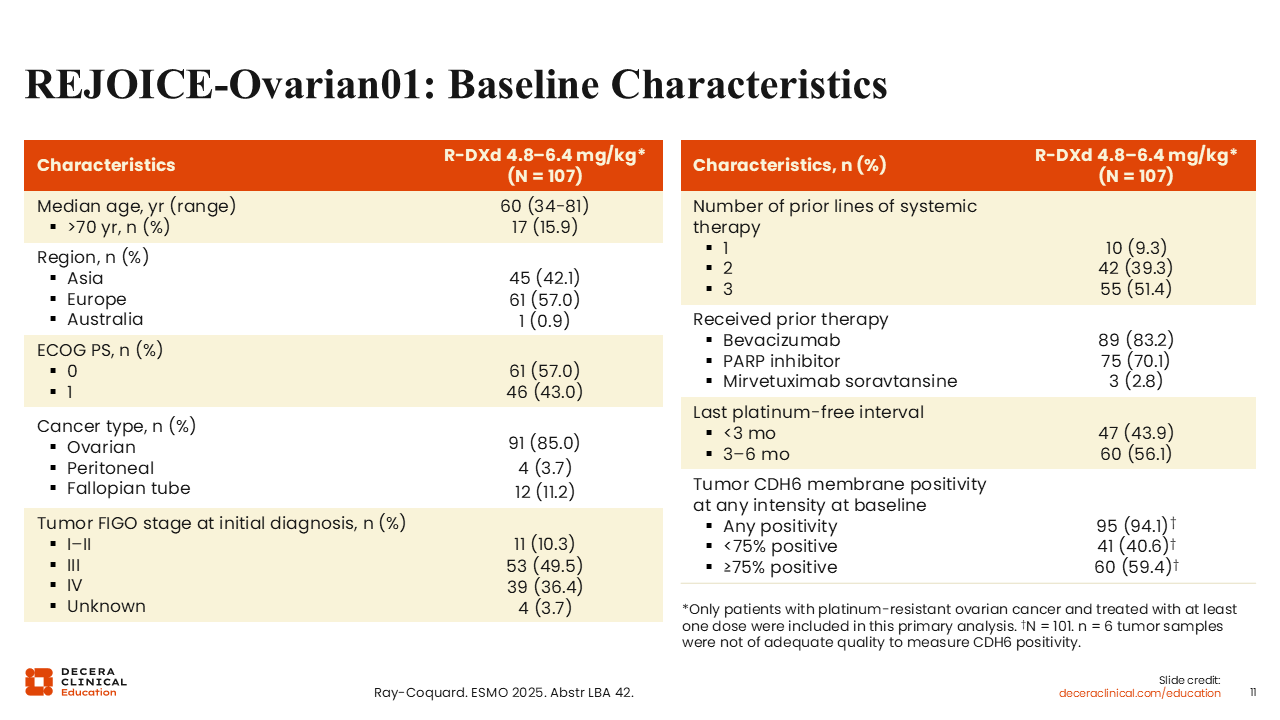
REJOICE‑Ovarian01: Baseline Characteristics
Isabelle Ray-Coquard, MD, PhD: Similar to the phase I study, 83.2% and 70.1% of patients had received prior bevacizumab and a PARP inhibitor, respectively.8 Due to the enrollment criteria, these patients were less pretreated than the phase I patients with approximately half (51.4%) having received 3 lines of prior systemic therapy. Most patients had a last platinum-free interval of 3-6 months (56.1%).
Of interest, 94.1% of evaluable patients had tumors that were positive for CDH6 membrane expression, which was higher than anticipated, and 59.4% of tumors were considered highly positive (≥75% of viable tumor cells positive for CDH6 membrane staining).
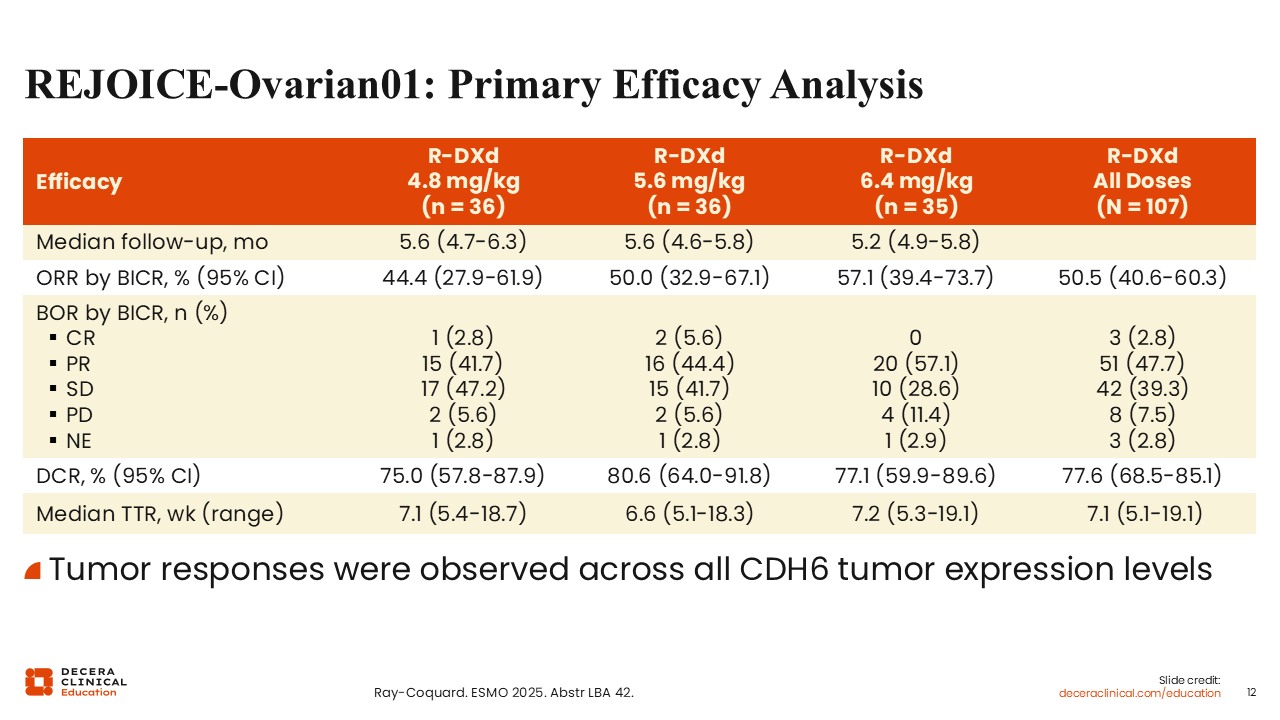
REJOICE‑Ovarian01: Primary Efficacy Analysis
Isabelle Ray-Coquard, MD, PhD: The activity of R-DXd in this phase II trial was impressive, with the vast majority of patients experiencing tumor shrinkage, an ORR across all the doses of 50.5%, and a DCR of 77.6%.8 The median time to response was short (7.1 weeks). ORR increased with increasing dose level with 44.4%, 50.0%, and 57.1% for 4.8, 5.6, and 6.4 mg/kg doses, respectively. DCR was highest for the 5.6 mg/kg dose level (80.6%).
Of interest, tumor responses were observed across a range of baseline CDH6 expression levels from the lowest to the highest positivity, which is important to keep in mind as we continue to study R-DXd.
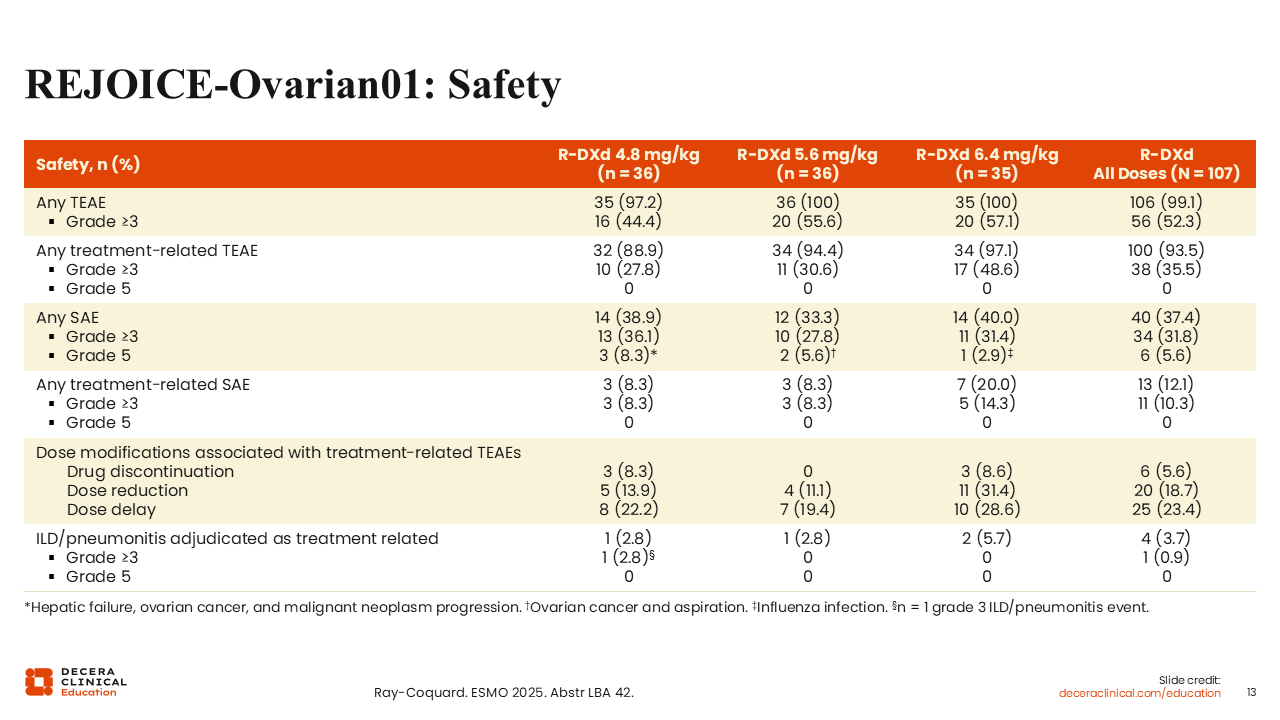
REJOICE‑Ovarian01: Safety
Isabelle Ray-Coquard, MD, PhD: The 6.4 mg/kg cohort experienced more TRAEs and more treatment-related serious AEs compared to the 4.8 and 5.6 mg/kg cohorts.8 Overall, the rates of dose discontinuation, reduction, and delay were low: 5.6%, 18.7%, and 23.4%, respectively. Again, there were more dose modifications in the 6.4 mg/kg cohort.
Treatment-related ILD was also observed in the phase II portion of this trial at a rate of 3.7% in only 4 patients with just 1 grade 3 event at the time of the analysis in the 4.8 mg/kg and 2 lower grade events in the 6.4 mg/kg cohort.
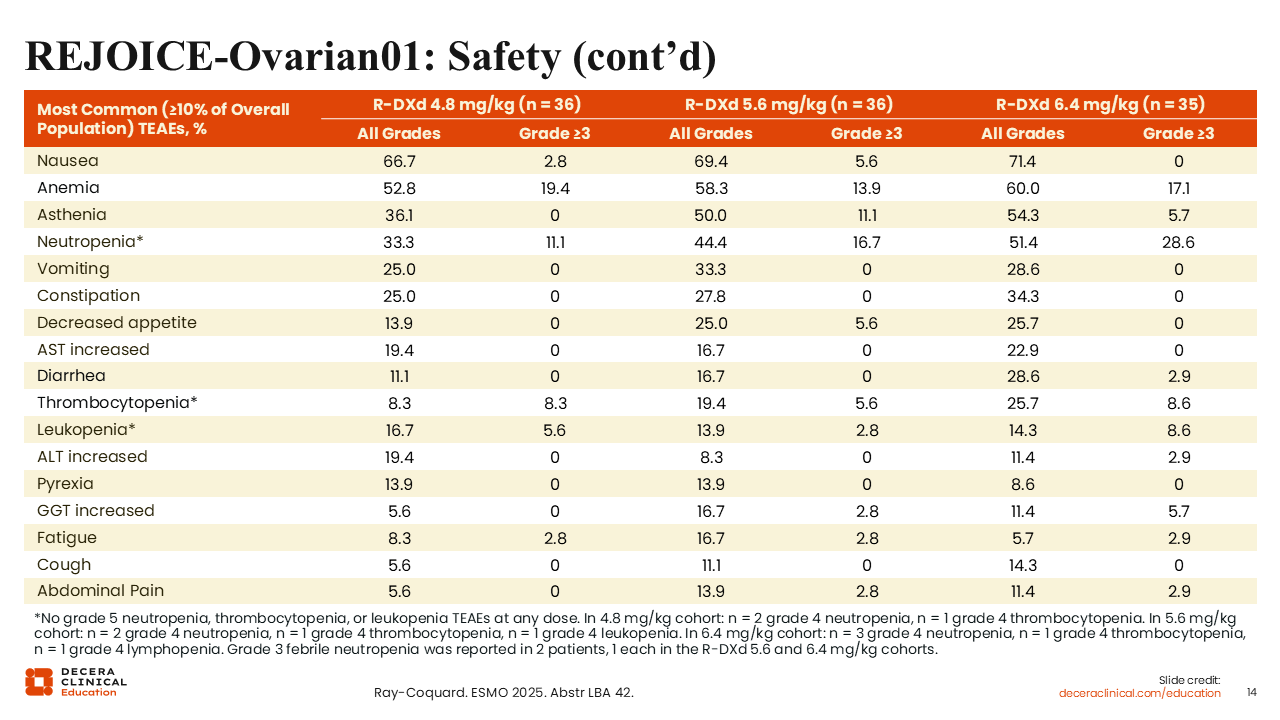
REJOICE‑Ovarian01: Safety (continued)
Isabelle Ray-Coquard, MD, PhD: The safety profile of R-DXd in this phase II analysis was consistent with the phase I data and considered manageable.8,13-15
Overall, these efficacy and safety results are very promising and encourage us to consider using R-DXd in earlier lines of treatment. Finally, 5.6 mg/kg was considered the optimal dose and will be explored in the next phase III step of the study which will begin enrollment soon in the United States and internationally.
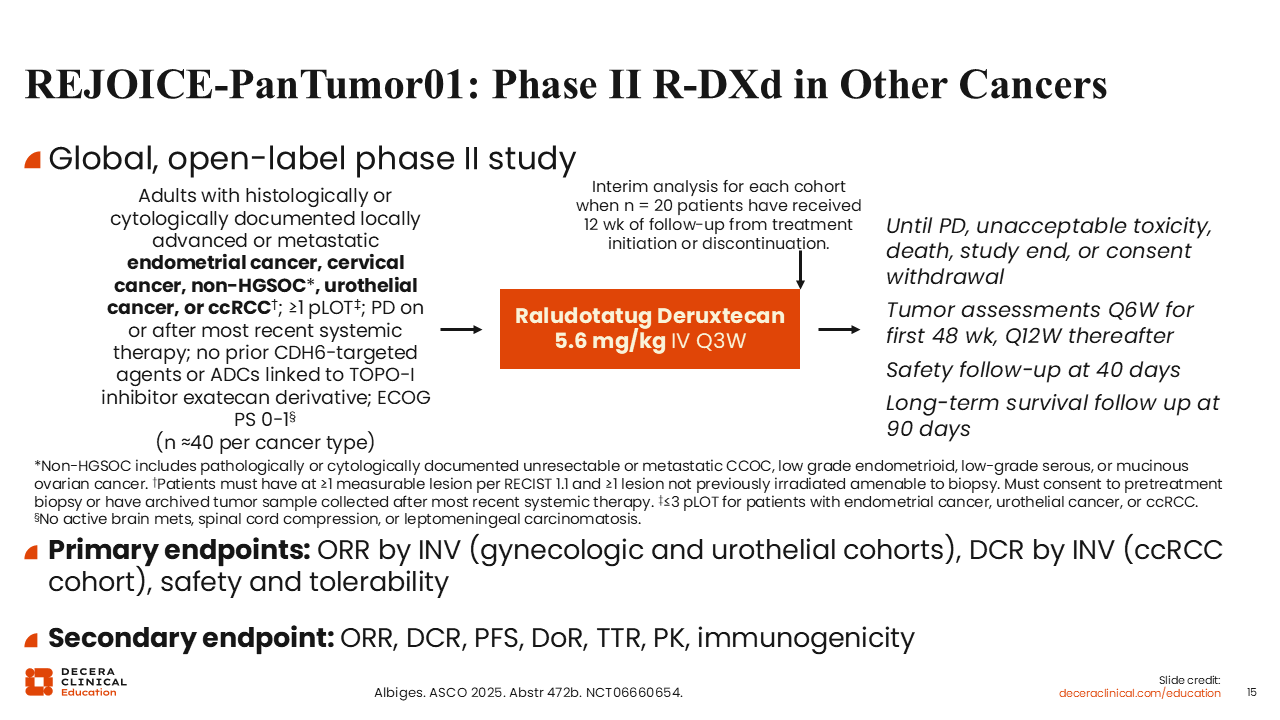
REJOICE‑PanTumor01: Phase II R-DXd in Other Cancers
Isabelle Ray-Coquard, MD, PhD: There are 2 other trials of R-DXd in progress. One is the phase II REJOICE‑PanTumor01 trial, which is investigating whether 5.6 mg/kg of R‑DXd can be used for disease beyond high‑grade serous ovarian cancer including other gynecologic cancers (eg, endometrial, cervical, and ovarian other than high-grade serous), urothelial cancer, and RCC (NCT06660654).18 Enrollment criteria include at least 1 prior line of treatment, progressive disease on or after most recent systemic therapy, and consent to a pretreatment biopsy. Approximately 40 patients will be recruited for each type of cancer. We anticipate seeing preliminary results in 2026.
REJOICE‑Ovarian02: Phase I/II R-DXd Combinations
Isabelle Ray-Coquard, MD, PhD: The second ongoing study is the phase I/II REJOICE-Ovarian02 trial investigating combinations of R‑DXd with other agents (NCT06843447).19,20 The phase Ib dose-escalation portion has 2 arms testing R-DXd with carboplatin and R-DXd with paclitaxel for patients with platinum-sensitive ovarian cancer. The third arm will test R-DXd plus bevacizumab for platinum-resistant ovarian cancer. Approximately 3-10 patients will be enrolled per arm.
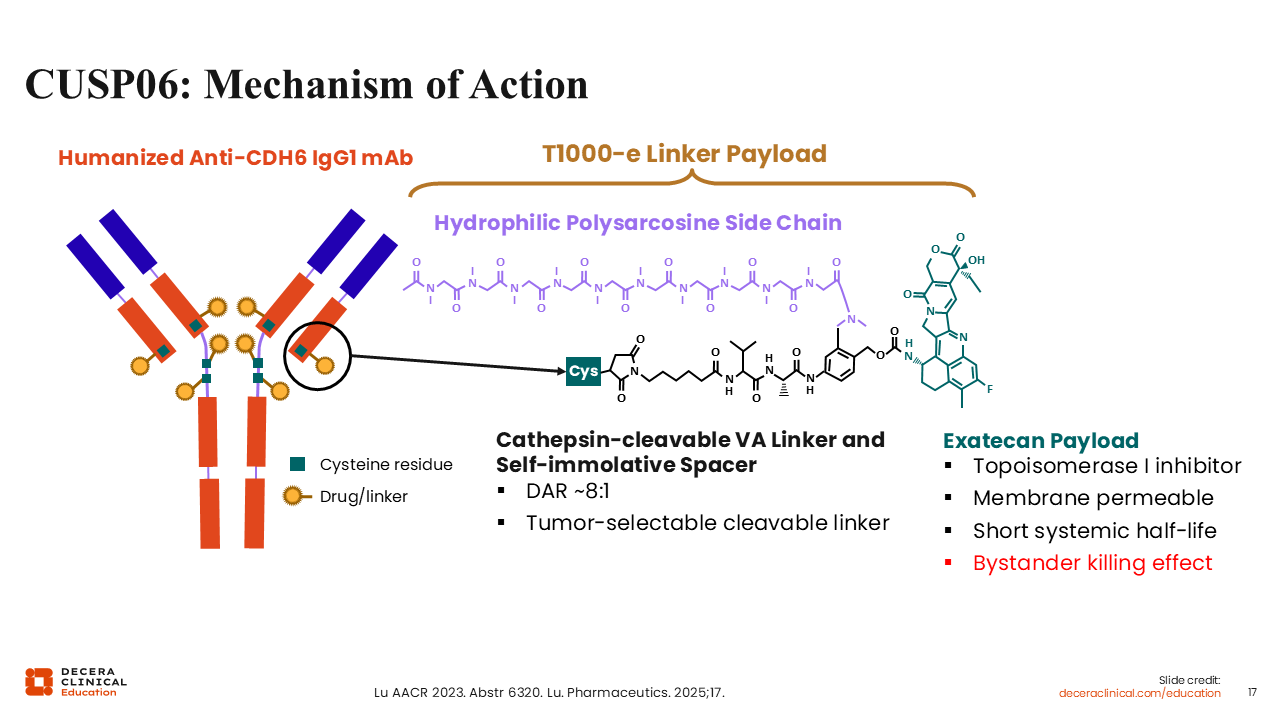
CUSP06: Mechanism of Action
Debra L. Richardson, MD: There is another CDH6-targeted ADC in development called CUSP06 that has an exatecan payload, which is a different TOPO-1 inhibitor than that of R-DXd.21,22 Like R-DXd, it has a drug‑to‑antibody ratio of 8 and bystander antitumor effects.
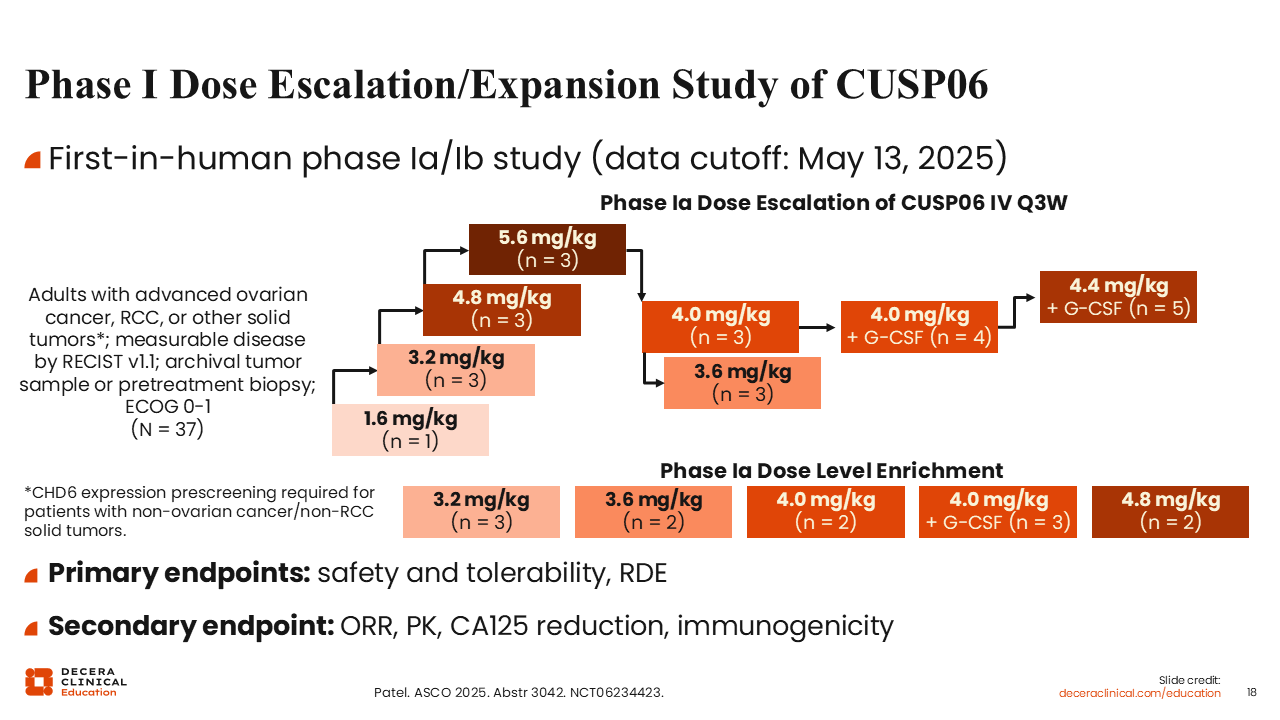
Phase I Dose Escalation/Expansion Study of CUSP06
Debra L. Richardson, MD: The dose escalation phase of the first-in-human phase Ia/Ib study of CUSPO06 is evaluating doses ranging from 1.6 to 5.6 mg/kg in adults with advanced ovarian cancer and RCC.23 Other solid tumors that were prescreened for CDH6 expression were also allowed. There were originally 4 doses of 1.6, 3.2, 4.8, and 5.6 mg/kg, but intermediate doses of 3.6 and 4.0 mg/kg were added as well. Two cohorts of 4.0 and 4.4 mg/kg doses with granulocyte colony-stimulating factor (G-CSF) were also added due to myelosuppression observed in other cohorts. Primary endpoints were safety, tolerability, and determination of the recommended expansion dose. The initial results of the phase Ia dose escalation were reported for 37 patients.
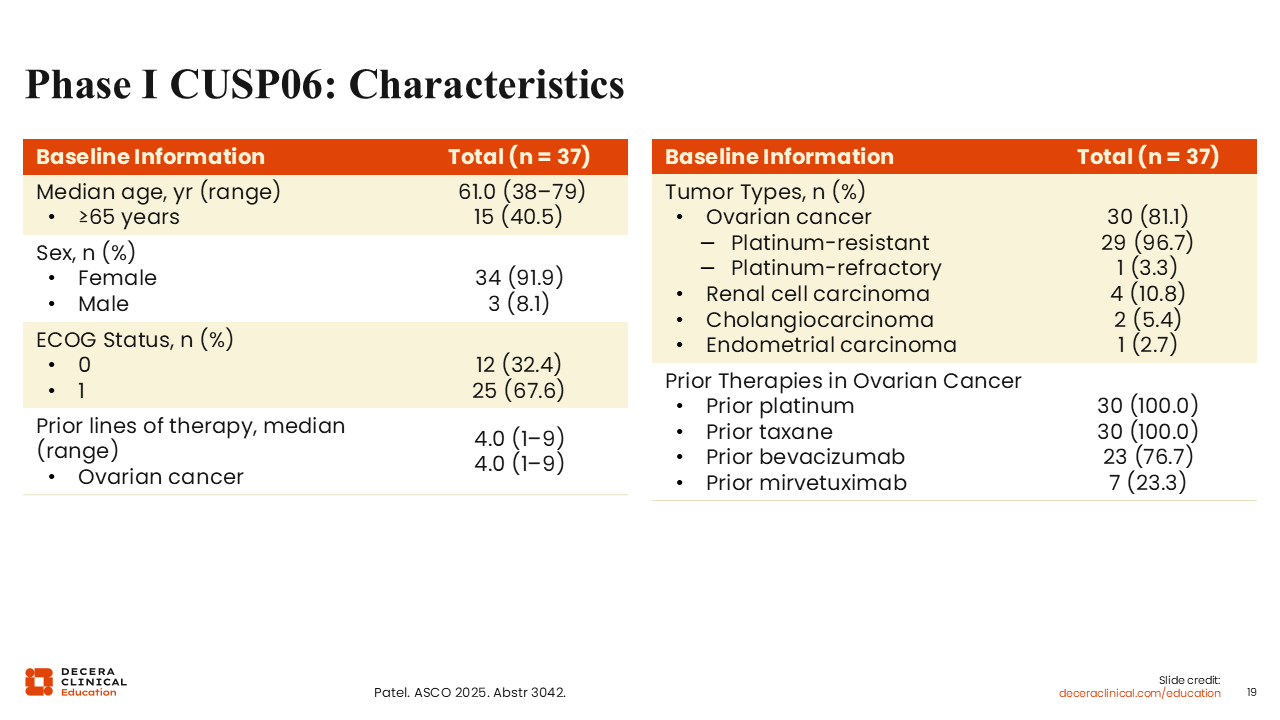
Phase I CUSP06: Characteristics
Debra L. Richardson, MD: Of the patients included in the dose escalation, 34 (91.9%) were female and 30 tumors (81.1%) were ovarian cancer.23 The patients with ovarian cancer were fairly heavily pretreated with a median of 4 prior lines of therapy, ranging from 1-9 prior lines, and were primarily platinum resistant (81.1%). One patient (3.3%) with ovarian cancer was platinum refractory. Other tumor types included were RCC (n = 4; 10.8%), cholangiocarcinoma (n = 2; 5.4%), and endometrial cancer (n = 1; 2.7%).
As expected, all patients with ovarian cancer had received prior platinum and taxane. 76.7% had prior bevacizumab and, of interest, we are starting to see mirvetuximab soravtansine as a prior line in these patients, with 23% having received it previously.
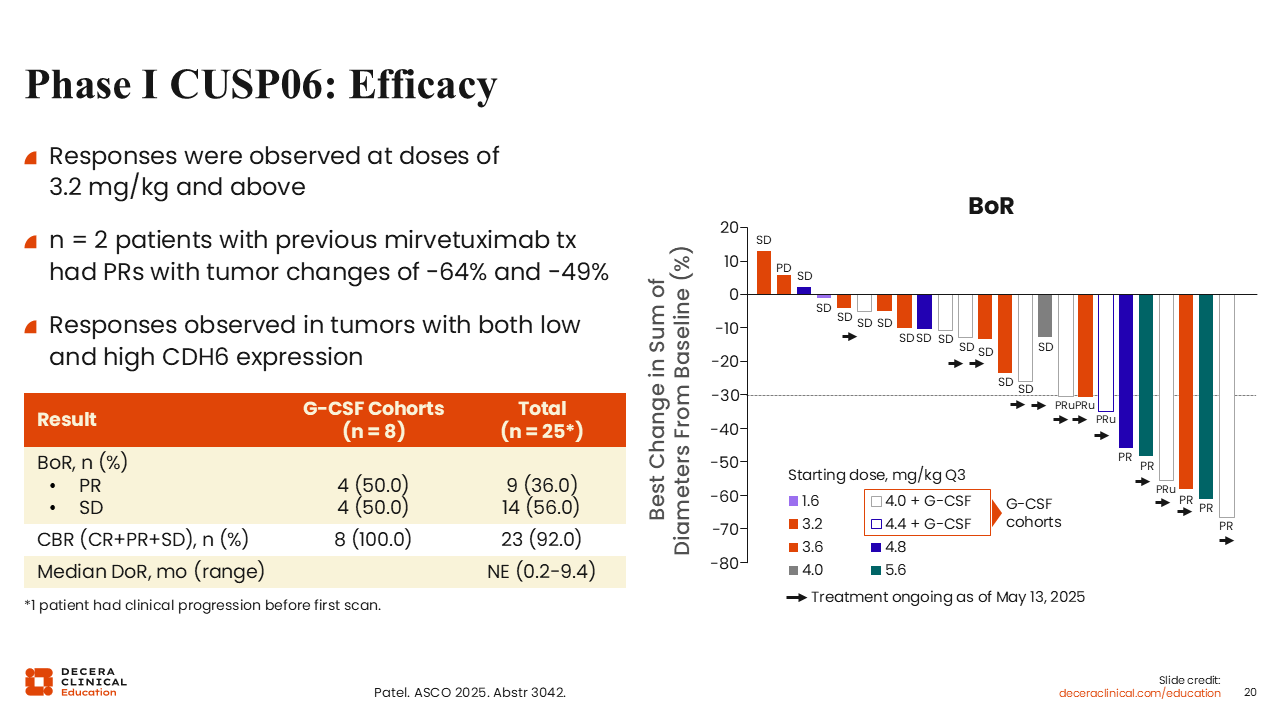
Phase I CUSP06: Efficacy
Debra L. Richardson, MD: Most patients experienced some shrinkage of their target lesion.23 ORR was 36.0%, consisting of all PRs, and clinical benefit rate was very high (92.0%). This indicates CUSP06 has activity in patients who had received mirvetuximab soravtansine. Of interest, the G-CSF cohorts had 50.0% PRs and clinical benefit rate of 100%.
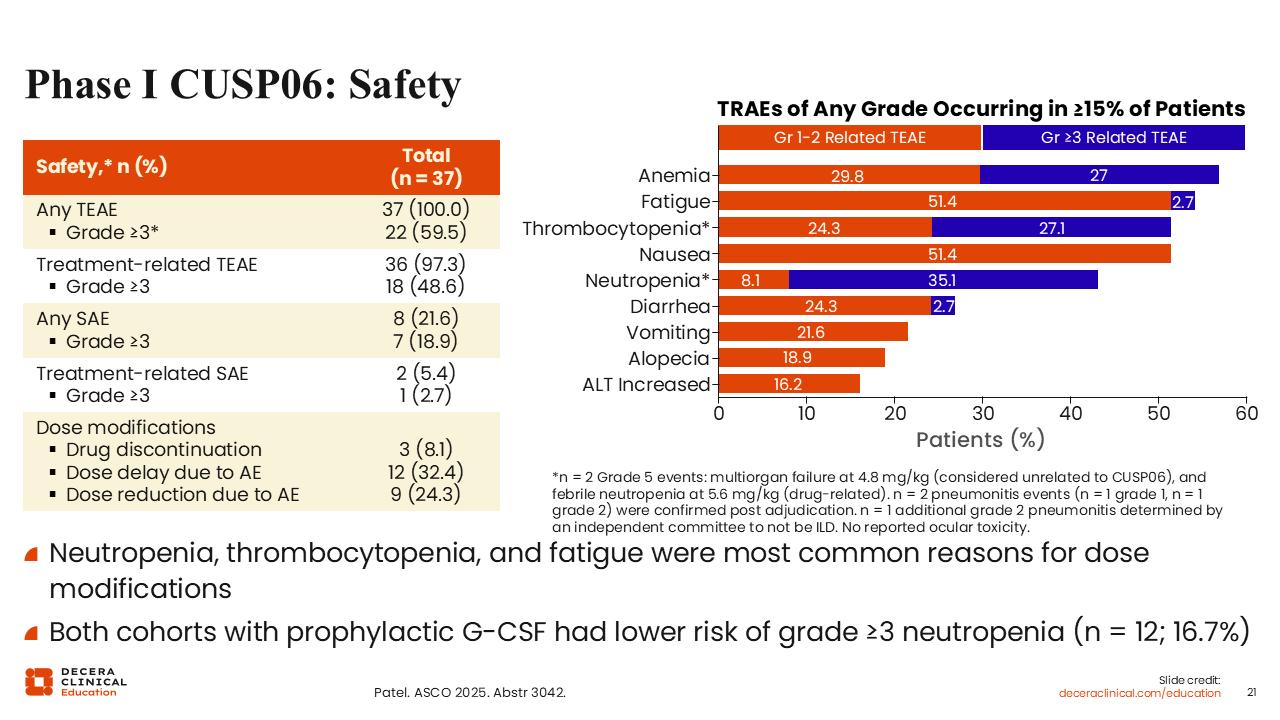
Phase I CUSP06: Safety
Debra L. Richardson, MD: The safety profile was not surprising based on what we know about the exatecan payload with the most common side effects including anemia, fatigue, thrombocytopenia, and nausea.23 Only 5.4% of patients had a treatment‑related serious AE, which is fairly low; 8.1% discontinued treatment, 32.4% required a dose delay due to toxicity, and 24.3% required a dose reduction. Cohorts treated with G-CSF had lower risk of grade 3 or higher neutropenia than the overall study population (16.7% vs 35.1%).
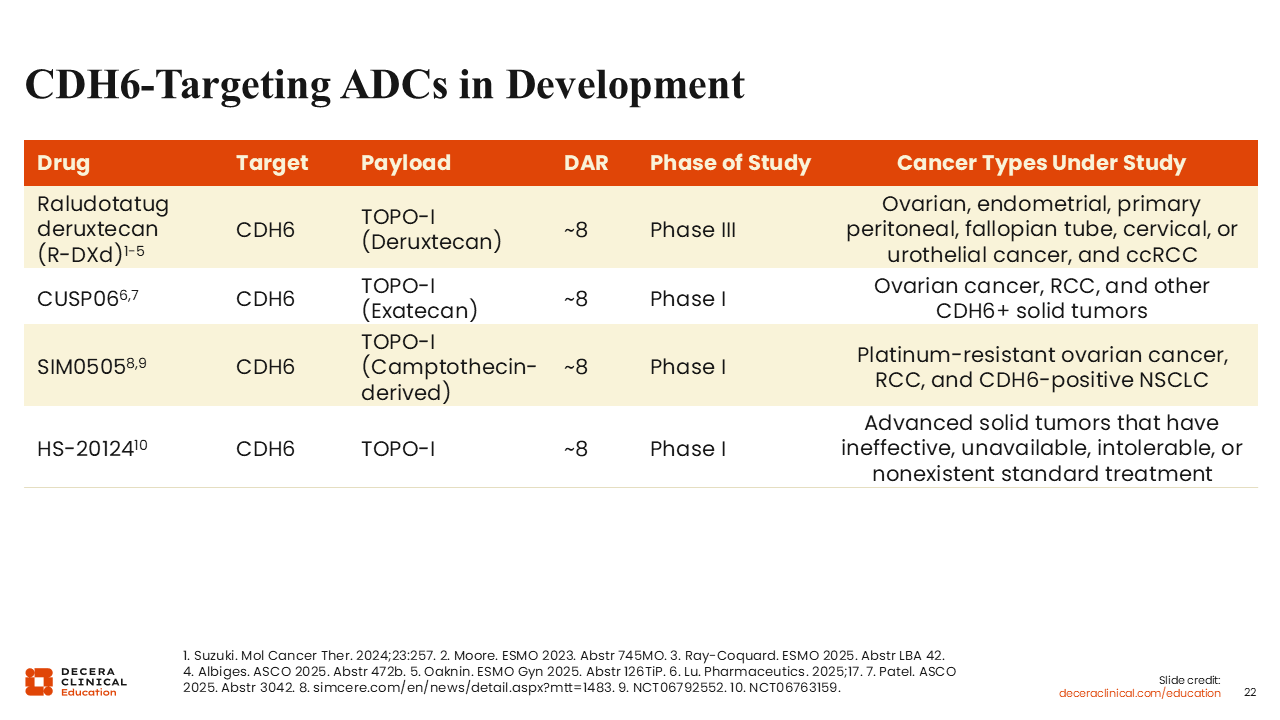
CDH6‑Targeting ADCs in Development
Debra L. Richardson, MD: Two other CDH6-targeting ADCs are currently in phase I trials. One is SIM0505, which uses a camptothecin-derived TOPO-I inhibitor and is in an actively recruiting phase I study in the United States and China for platinum-resistant ovarian cancer, RCC, and CDH6-positive non-small-cell lung cancer (NCT06792552).24 Another phase I study in China for the ADC HS-20124 is recruiting patients with advanced solid tumors that have ineffective, unavailable, intolerable, or nonexistent standard treatment (NCT06763159).
Future Role of CDH6‑Targetting ADCs in Treatment Paradigm
Isabelle Ray-Coquard, MD, PhD: The data for CDH6-targeted ADCs are promising, but we will need to investigate where they should be placed in treatment sequencing and whether we need to select patients based on biomarker expression. For instance, the REJOICE-Ovarian01 study showed tumor responses to R-DXd across CDH6 expression levels.8 However, expression levels may impact other outcomes. We also need to optimize benefit and mitigate toxicity, for instance preventing nausea and vomiting and anticipating and managing ILD with R-DXd, to avoid treatment discontinuation.
Debra L. Richardson, MD: I think the field of ovarian cancer is exciting but also challenging at the moment because we have a number of new drugs in development, including these CDH6-targeted ADCs, and many unanswered questions. For example, how many ADCs can a patient receive? I anticipate that patients with ovarian cancer will likely be treated with up to 2 different ADCs, and they may require different antigen targets or different payloads to optimize efficacy, but we will have to see what the data say. Regarding treatment combinations, I also think it will be difficult to combine ADCs with a TOPO-I payload with carboplatin due to their overlapping toxicity of myelosuppression.
Although I don’t envision these CDH6-targeted ADCs in the frontline treatment for ovarian cancer, we may see them used as maintenance therapy or fairly early in the treatment of the platinum‑resistant or platinum‑ineligible population.
Lower Enrollment of Racial and Ethnic Minorities vs White Patients on Gynecologic Clinical Trials
Debra L. Richardson, MD: One more common theme across emerging investigational therapies, such as the CDH6 ADCs, is that we have historically not been enrolling patients equitably in clinical trials, which is a problem because we need accurate data for all patients we treat in practice.25-29 In ovarian cancer, we are dramatically underenrolling Black and Latinx patients by a factor of 7.4 and 8.1, respectively.27 We should aim to close this disparity because there could be differences in treatment efficacy or tolerability related to race and/or ethnicity.
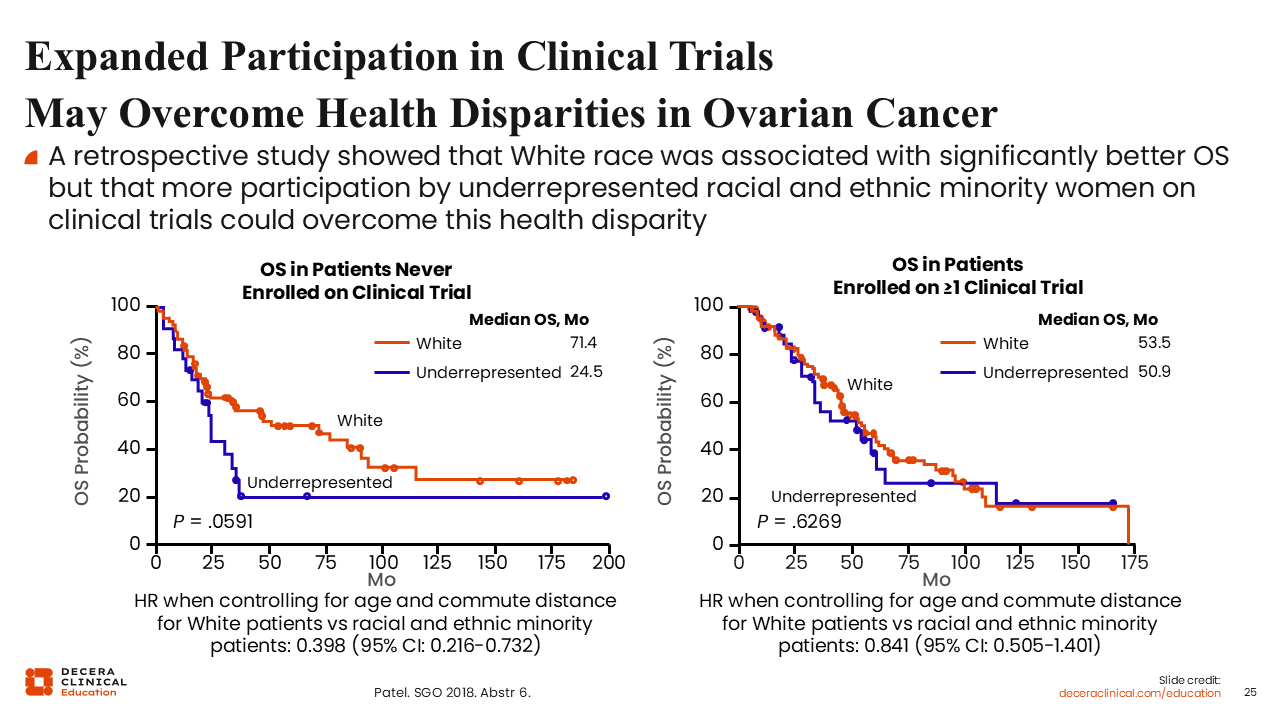
Expanded Participation in Clinical Trials May Overcome Health Disparities in Ovarian Cancer
Debra L. Richardson, MD: We also know that underrepresented racial and ethnic minority patients with ovarian cancer have worse overall survival than White patients, but retrospective data suggest we can nearly eliminate these differences if we enroll patients on trials.30 I think these data should be a highly motivating call to action for healthcare professionals treating these patients to encourage clinical trial enrollment.
Factors Affecting Willingness to Participate in Clinical Trials
Debra L. Richardson, MD: Although there are many factors affecting patient willingness to join clinical trials, it is important to keep in mind that patients cannot participate in a trial if they are not asked to participate. As healthcare professionals, if a patient is potentially eligible for an available trial, it is our job to offer the patient that trial, or at least mention that a trial may be available elsewhere. Sometimes we may be hesitant to discuss clinical trial options with patients due to an assumption of burden or limitations to participation, but it should be up to the patients to decide if they want to participate. I have been amazed by how far my patients are willing to travel and how much of their daily lives they are willing to impact in order to take part in studies.
In addition, we should consider that increased enrollment in trials could rapidly improve survival outcomes of patients overall. We can look at pediatric cancer in the United States as a motivating example, where 5-year survival is now 87%, up from 63% about 50 years ago.31
Participation rates of pediatric patients in clinical trials are several times that of adult patients (20%-30% vs approximately 7%), and this is likely a major driving force for improved real-world outcomes.32,33
Things we can do to improve enrollment include providing testimonials from patients who have experienced clinical trials and offer resources for logistical, financial, and emotional support. Of course, we should also aim to establish trust with our patients and provide thorough and accurate medical information regarding risks and benefits of treatments and trials. We can also discuss biomarker-informed trials to help the study of targeted agents like R-DXd.
Actively recruiting trials are listed on clinicaltrials.gov and the GOG Foundation website for the United States, and the European Network of Gynaecological Oncological Trial groups (ENGOT) website.