CE / CME
Enhancing Outcomes in Limited-Stage Small-Cell Lung Cancer With Recent Therapeutic Advancements
Physician Assistants/Physician Associates: 0.25 AAPA Category 1 CME credit
ABIM MOC: maximum of 0.25 Medical Knowledge MOC point
Pharmacists: 0.25 contact hour (0.025 CEUs)
Nurse Practitioners/Nurses: 0.25 Nursing contact hour
Physicians: maximum of 0.25 AMA PRA Category 1 Credit™
Released: September 18, 2025
Expiration: March 17, 2026
Activity
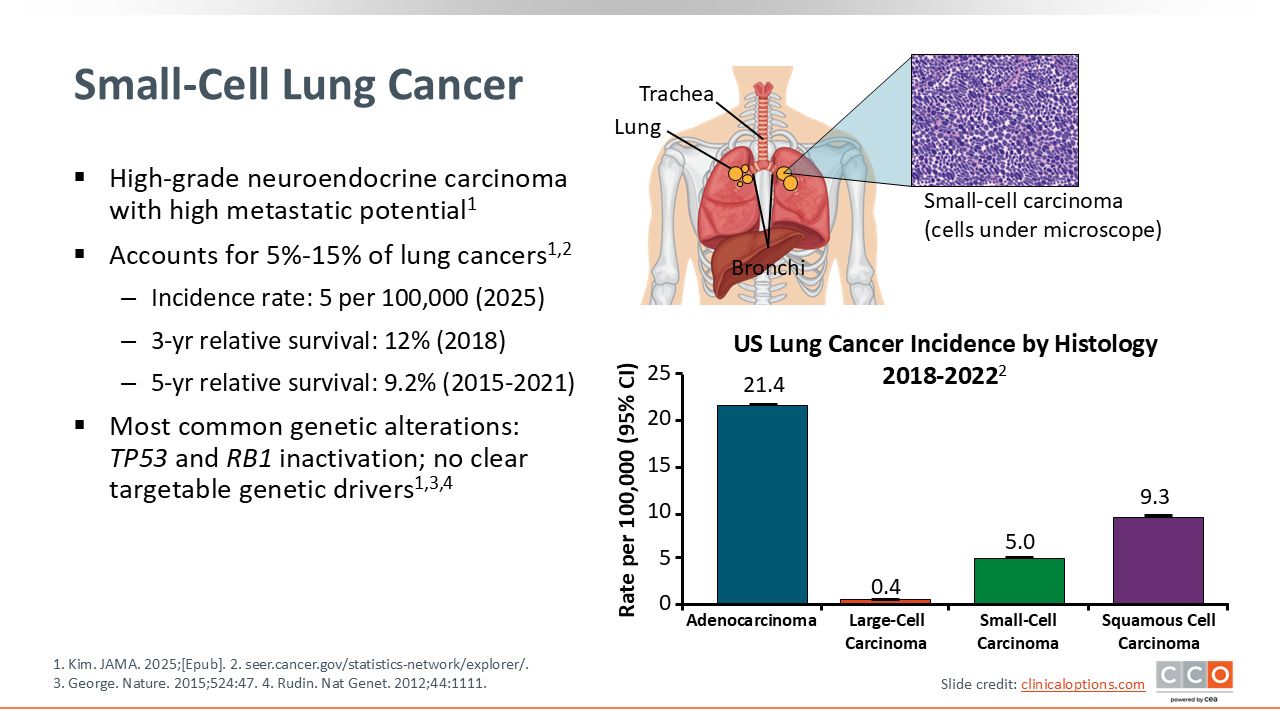
Small-Cell Lung Cancer
SCLC is a high-grade, often metastatic, neuroendocrine carcinoma. It accounts for approximately 13% of all lung cancers, with an incidence rate of approximately 5 per 100,000 cases in the United States.1,2 SCLC most often occurs in the central airways of the lung and is usually aggressive, with a high tumor cell proliferation rate. Because of the aggressive nature of SCLC, symptoms may occur quickly and are usually what drives diagnosis. As such, approximately 70% of patients present with advanced-stage disease at diagnosis.1
SCLC is often associated with smoking or other environmental risk factors, such as asbestos or radon, that cause DNA damage and genetic changes to the tumor cells. The most common genetic alterations seen in SCLC are the inactivation of tumor suppressor genes P53 and RB1. No other clearly targetable genetic drivers have been observed.1,3,4
Staging and General Approach to Treatment
LS-SCLC is diagnosed in only one third of patients with SCLC, but it is treated with definitive chemotherapy and RT and can be considered curable. LS‑SCLC generally corresponds to TNM stage I-III confined to 1 hemithorax such that thoracic RT can encompass all disease. PCI can also be a consideration in this patient population.5,6
Recent studies have shown that median OS with consolidation durvalumab after concurrent CRT has improved, with the ADRIATIC trial reporting a median OS of 56 months vs 33 months with placebo and a 3-year survival of 56.5%.7,8 Despite higher survival rates, more than 50% of patients with LS‑SCLC still experienced disease recurrence.
Extensive-stage SCLC, which represents approximately two thirds of diagnoses, is not a curable disease.1,4 The first-line standard of care in extensive-stage SCLC is chemotherapy with immunotherapy. These therapies can palliate patients and improve quality of life.9 Median survival was between 12 and 13 months in extensive-stage disease.10 There remains a pressing unmet need for better therapies in this setting.
In this text module, we will focus on the current data for patients with LS-SCLC. Additional information on extensive-stage SCLC can be found here.
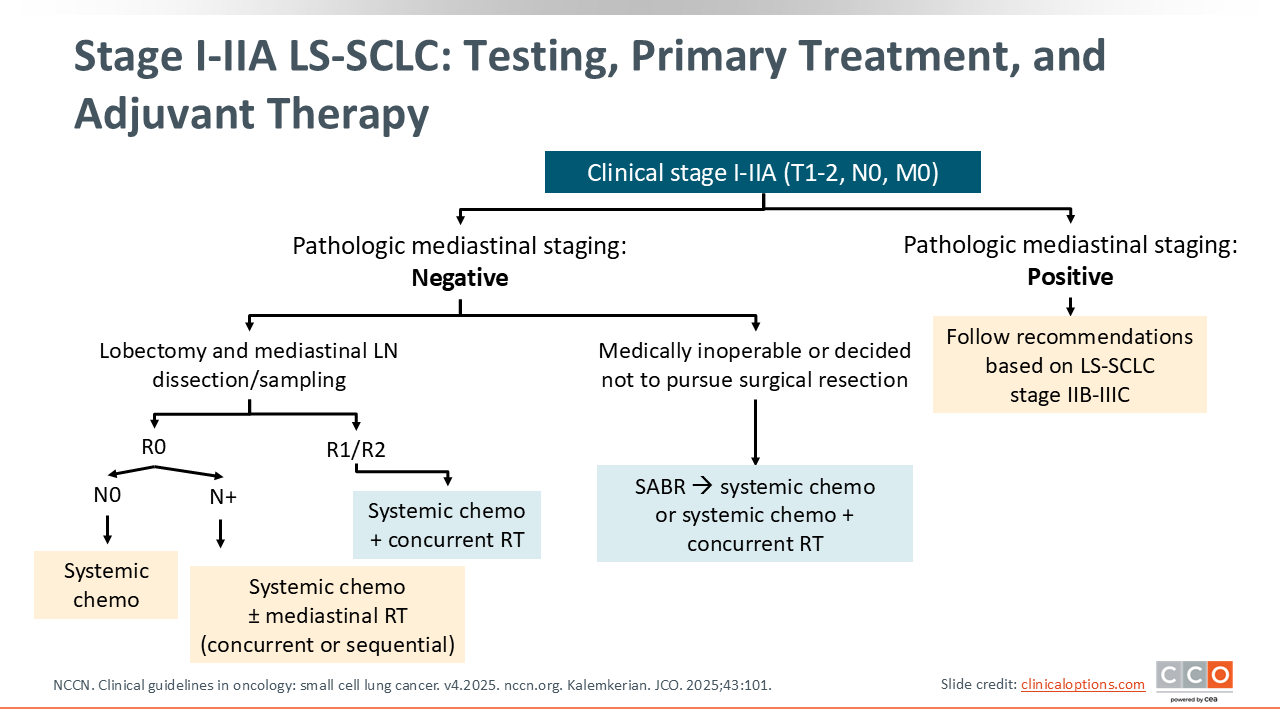
Stage I-IIA LS-SCLC: Testing, Primary Treatment, and Adjuvant Therapy
Management of LS-SCLC, specifically clinical stage I-IIA (T1-2, N0, M0), begins with pathologic mediastinal staging.6,11 If staging is negative, patients may proceed with lobectomy and mediastinal lymph node dissection or sampling. For those with complete resection and no nodal involvement (R0, N0), systemic chemotherapy alone is appropriate. If nodal disease is detected (R0, N+), systemic chemotherapy combined with mediastinal RT—either concurrently or sequentially—is recommended. In cases with incomplete resection (R1/R2), systemic chemotherapy with concurrent RT is the preferred approach.
If patients are medically inoperable or decline surgical resection, stereotactic ablative RT to the primary tumor may be considered, followed by systemic chemotherapy or combined with concurrent CRT. This provides a curative-intent option for patients unable to undergo surgery, ensuring integration of systemic therapy with local control.
If pathologic mediastinal staging is positive, treatment should follow the recommendations for more advanced LS-SCLC (stage IIB-IIIC). This highlights the importance of mediastinal staging in guiding treatment intensity and modality selection, ensuring that patients with more advanced nodal disease receive appropriately escalated therapy.
Current Clinical Guidelines on Primary Treatment for Stage IIB-IIIC LS-SCLC
For patients with stage IIB-IIIC LS-SCLC, current treatment recommendations are guided primarily by PS. Patients with good PS (0-2) should receive 4 cycles of cisplatin/etoposide-based systemic chemotherapy given concurrently with thoracic RT, followed by durvalumab consolidation if there is no progression. For those with poor PS (3-4) attributable to SCLC, systemic chemotherapy remains appropriate, with RT administered either concurrently or sequentially, depending on tolerance.11
Of importance, patients with poor PS unrelated to SCLC should be considered instead for individualized treatment approaches, often prioritizing supportive care rather than intensive systemic therapy.
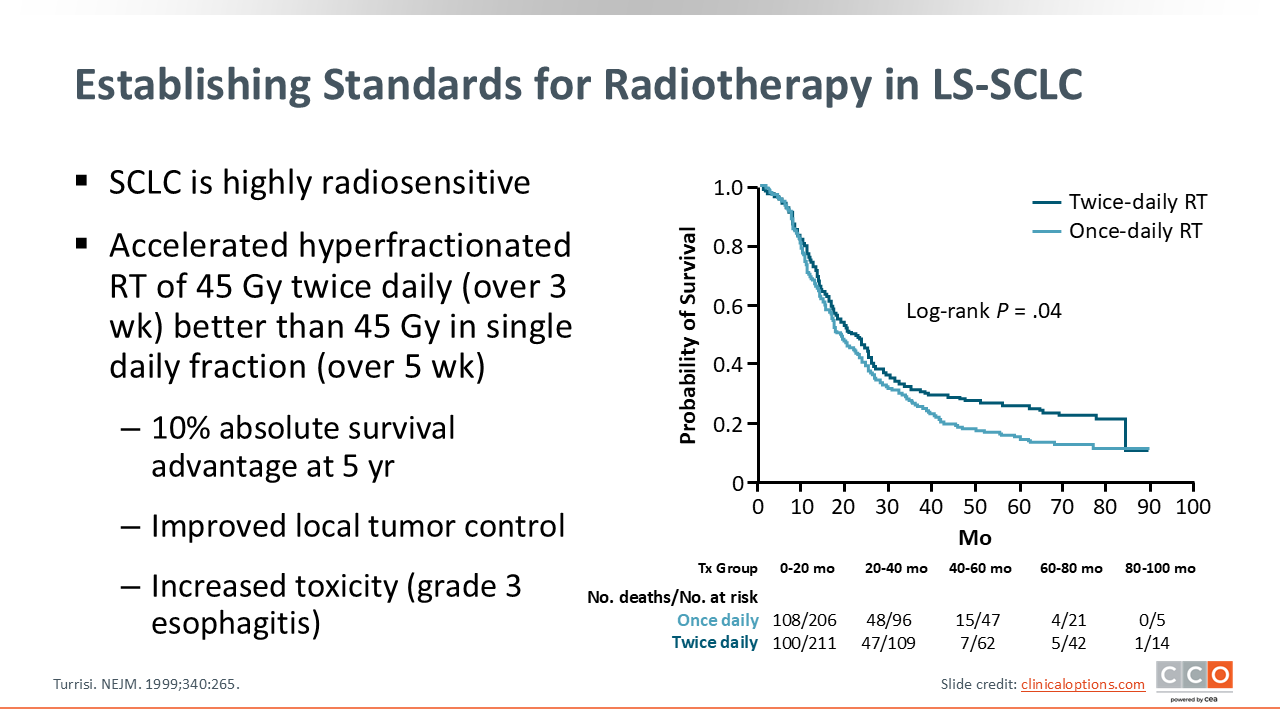
Establishing Standards for Radiotherapy in LS-SCLC
SCLC is highly radiosensitive, and therefore, the standard treatment for LS-SCLC includes RT and concurrent chemotherapy. A trial by the ECOG/RTOG group established the use of chemoimmunotherapy with accelerated (over 3 weeks) hyperfractionated RT of 45 Gy twice daily with etoposide/cisplatin, reporting a 10% absolute survival advantage at 5 years with 45 Gy twice daily over 3 weeks vs once daily over 5 weeks.12 There was also an improvement in local tumor control, but increased incidence of grade 3 esophagitis. This study laid the groundwork for subsequent trials exploring optimal use of RT for LS-SCLC.
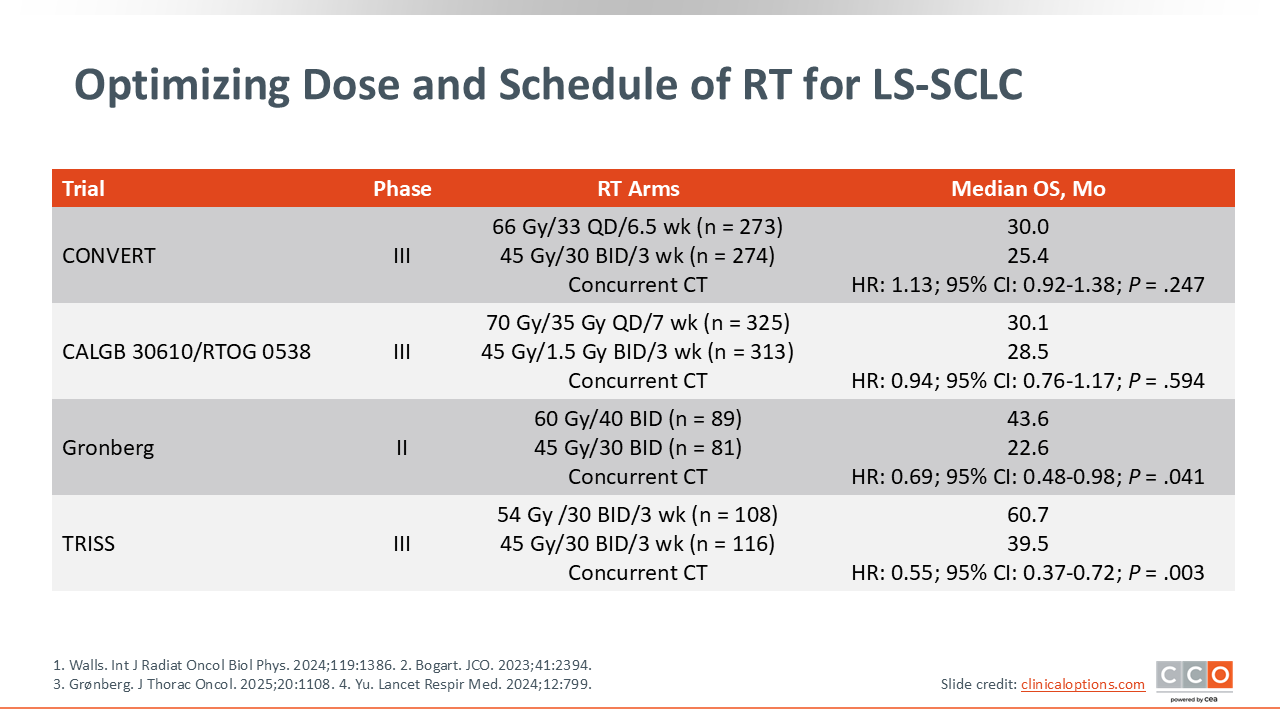
Optimizing Dose and Schedule of RT for LS-SCLC
Multiple trials have subsequently assessed various doses and schedules for RT, but the optimal dose and schedule of RT have not been established. Two randomized phase III trials, CONVERT and CALGB 30610/RTOG 0538, compared 45 Gy twice daily with either 66 Gy or 70 Gy once daily, respectively, in patients with LS-SCLC.13,14 These trials did not demonstrate superiority of higher, once-daily dose compared with the established 45 Gy twice-daily dose, but OS and toxicity were similar.
However, a randomized phase II trial suggests that higher-dose accelerated RT of 60-65 Gy twice daily in 4-5 weeks resulted in improved PFS and OS compared with 45 Gy twice daily in 3-week fractions.15 The randomized phase III TRISS trial demonstrated an OS advantage with high-dose accelerated RT given as 54 Gy simultaneous integrated boost to the gross tumor volume in 30 twice-daily fractions over 3 weeks compared with standard 45 Gy in 30 twice-daily fractions over 3 weeks.16
RT concurrent with systemic therapy is standard and preferred to sequential chemotherapy followed by RT. Most guidelines recommend that RT be started with cycle 1 or 2 of systemic therapy, as a shorter time from the start of any therapy to the end of RT has been associated with improved survival.6
Considerations for Chemotherapy Regimens for LS-SCLC
Currently, the preferred approach for systemic therapy for LS-SCLC includes 4 cycles of platinum-based chemotherapy combined with concurrent RT, followed by durvalumab consolidation therapy.6
Recommended platinum-based regimens include with cisplatin/etoposide or carboplatin/etoposide. Cisplatin regimens include various dosing options—cisplatin 75 mg/m² on Day 1 plus etoposide 100 mg/m² on Days 1-3, cisplatin 60 mg/m² on Day 1 with etoposide 120 mg/m² on Days 1-3, or lower-dose cisplatin 25 mg/m² on Days 1-3 with etoposide 100 mg/m² on Days 1-3. Carboplatin area under the concentration curve of 5-6 on Day 1 with etoposide 100 mg/m² on Days 1-3 can also be considered for patients who are not eligible for cisplatin-based therapy. Based on the phase III ADRIATIC trial, these regimens should be followed by consolidation therapy with durvalumab 1500 mg administered every 28 days.
Treatment cycles are planned every 21-28 days during concurrent RT. Of importance, the use of myeloid growth factors is not recommended during cytotoxic chemotherapy given concurrently with RT because of increased risks of toxicity. This integration of systemic therapy, RT, and immunotherapy reflects a multimodality approach aimed at improving survival outcomes in LS-SCLC.6
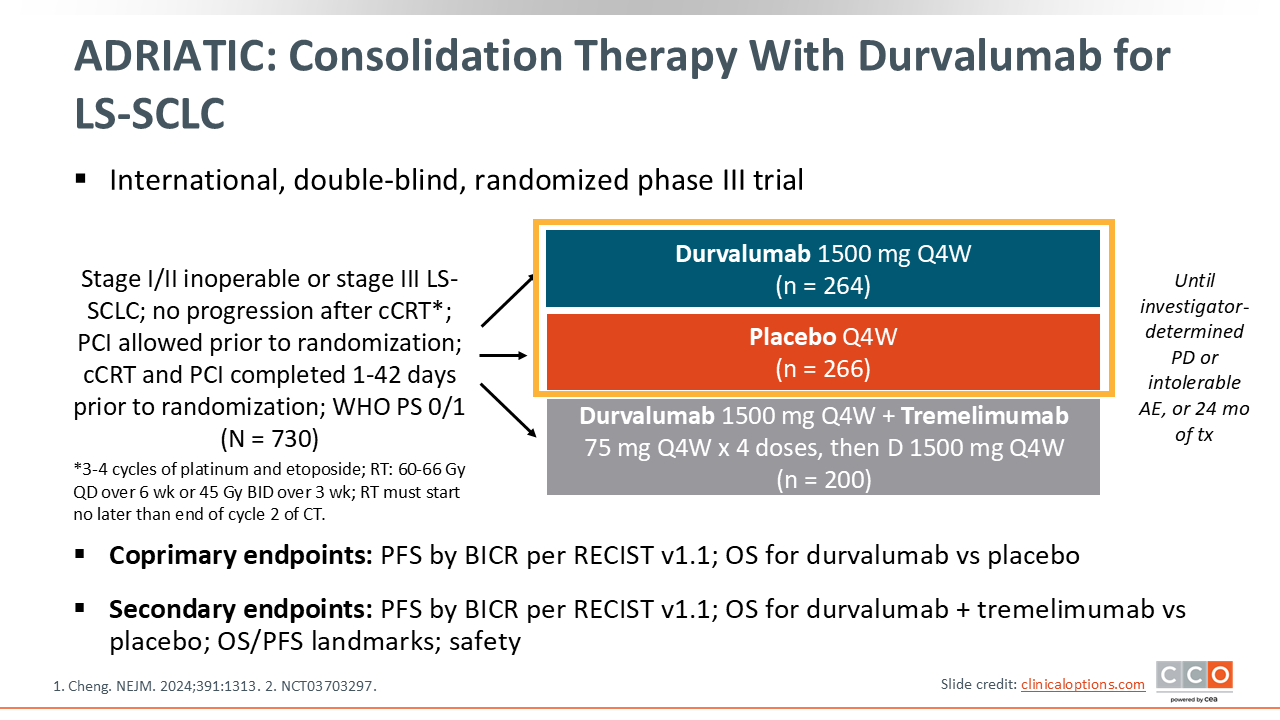
ADRIATIC: Consolidation Therapy With Durvalumab for LS-SCLC
Consolidation therapy is practice-changing breakthrough in LS-SCLC management, and it was established based on the ADRIATIC trial. The ADRIATIC trial was an international, double-blind, randomized phase III trial conducted in patients with stage I/II inoperable or stage III LS-SCLC that had not experienced disease progression following concurrent CRT. Before randomization, patients could undergo PCI after CRT at the investigator’s discretion. The coprimary endpoints of this trial included PFS and OS for durvalumab vs placebo.8
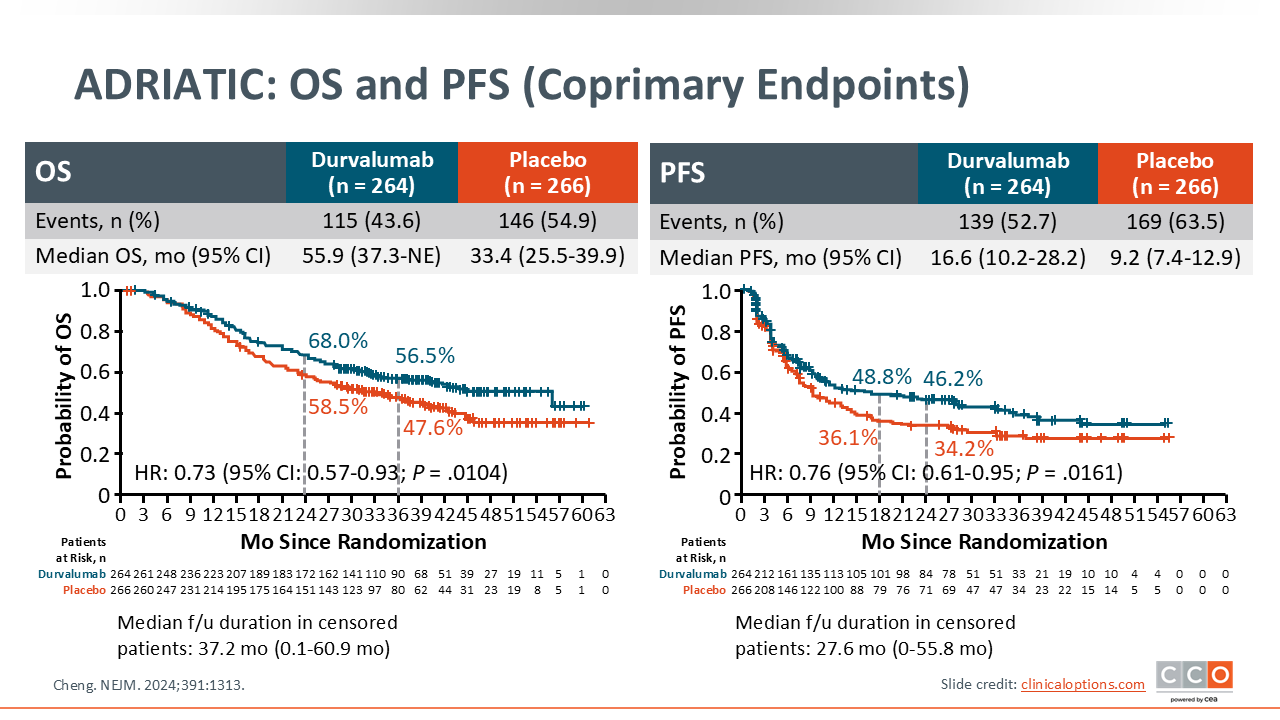
ADRIATIC: OS and PFS (Coprimary Endpoints)
Regarding outcomes, the HR for OS was 0.73 (95% CI: 0.57-0.93; P = .0104), indicating a 27% reduction in the risk of death, with a median OS of 55.9 months with durvalumab vs 33.4 months with placebo. The HR for PFS was 0.76 (95% CI: 0.61-0.95; P = .0161) with a median PFS of 16.6 months with durvalumab vs 9.2 months with placebo.
This trial met its primary endpoints and demonstrated a significant improvement in outcomes with the use of consolidation durvalumab.8
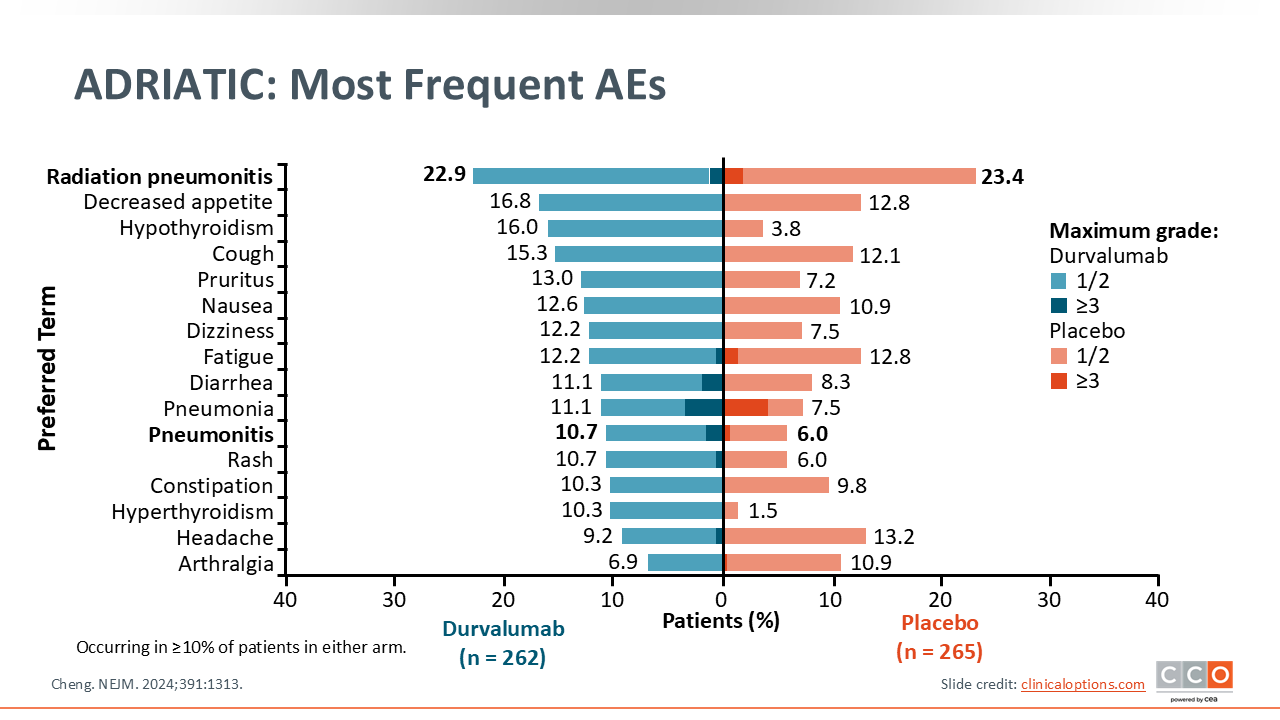
ADRIATIC: Most Frequent Adverse Events
From a clinical perspective, no new or unexpected safety signals were observed with durvalumab in this trial. The adverse effect profile of the placebo and durvalumab cohorts were largely similar. RT pneumonitis, decreased appetite, hypothyroidism, and cough were among the most commonly reported adverse events in patients who received durvalumab.8
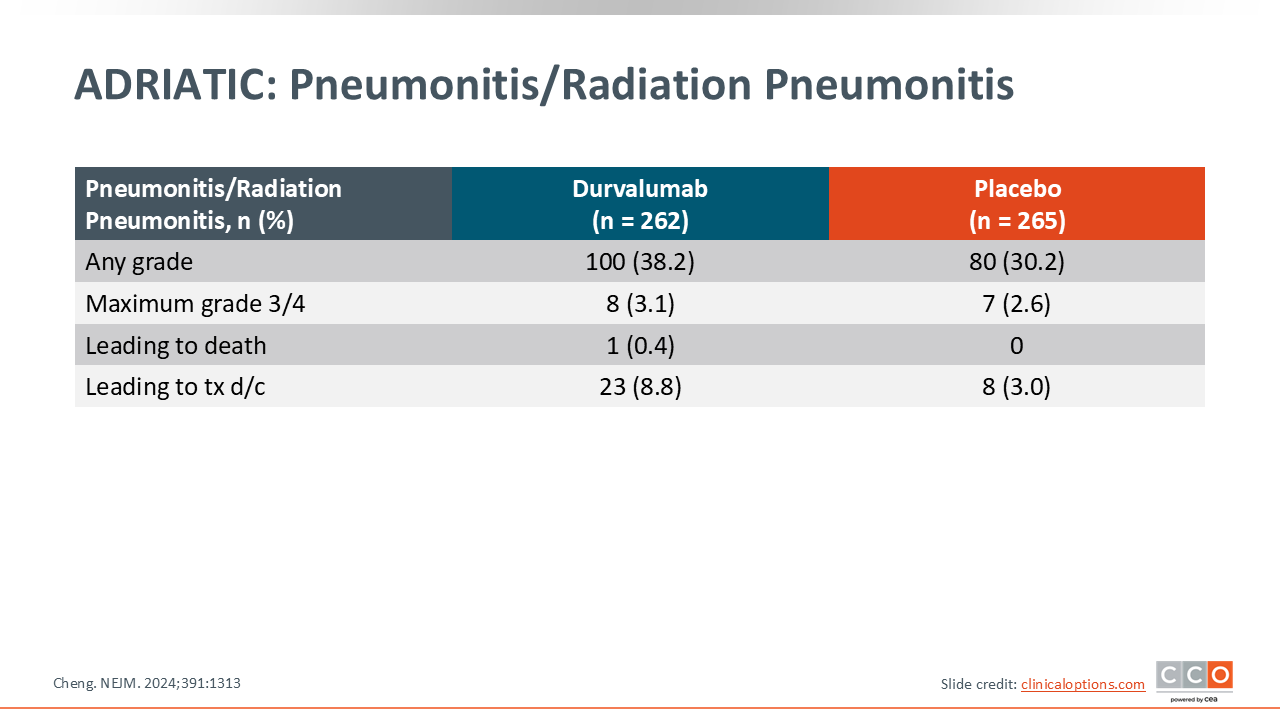
ADRIATIC: Pneumonitis/Radiation Pneumonitis
Stratification of pneumonitis/radiation pneumonitis rates showed a slight increase in all-grade events in patients who received durvalumab compared with those who received placebo. The rate of maximum grade 3/4 pneumonitis was approximately 3% in both arms, and the rate of discontinuation was higher with durvalumab at 8.8% vs placebo at 3.0%.8
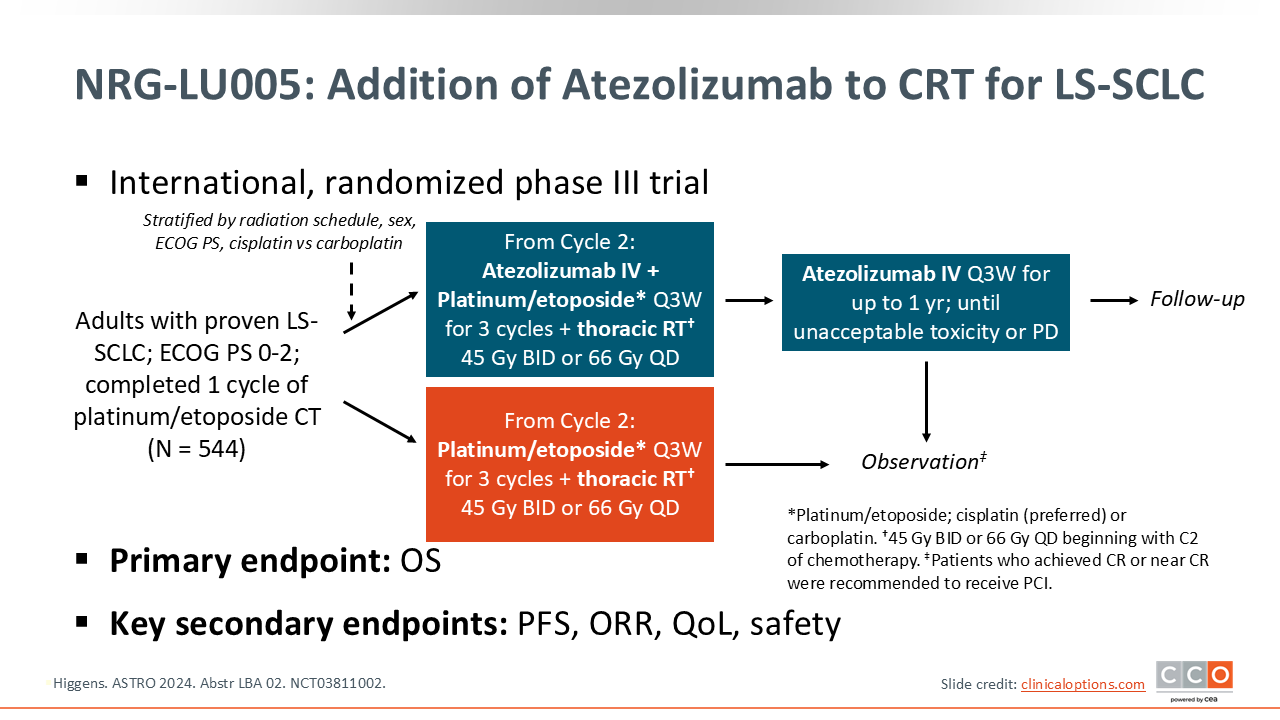
NRG-LU005: Addition of Atezolizumab to CRT for LS-SCLC
The NRG-LU005 trial was an international, randomized phase III trial that evaluated the addition of atezolizumab during the concurrent platinum/etoposide chemotherapy and thoracic radiotherapy in adults with LS-SCLC. Patients were randomized after their first cycle of platinum/etoposide chemotherapy to receive atezolizumab or not. Patients on the experimental arm could continue atezolizumab for up to 1 year or until investigator-assessed progression or intolerable adverse effects. Thoracic RT was administered as either 66 Gy once daily or 45 Gy twice daily.17
NRG-LU005: OS and PFS
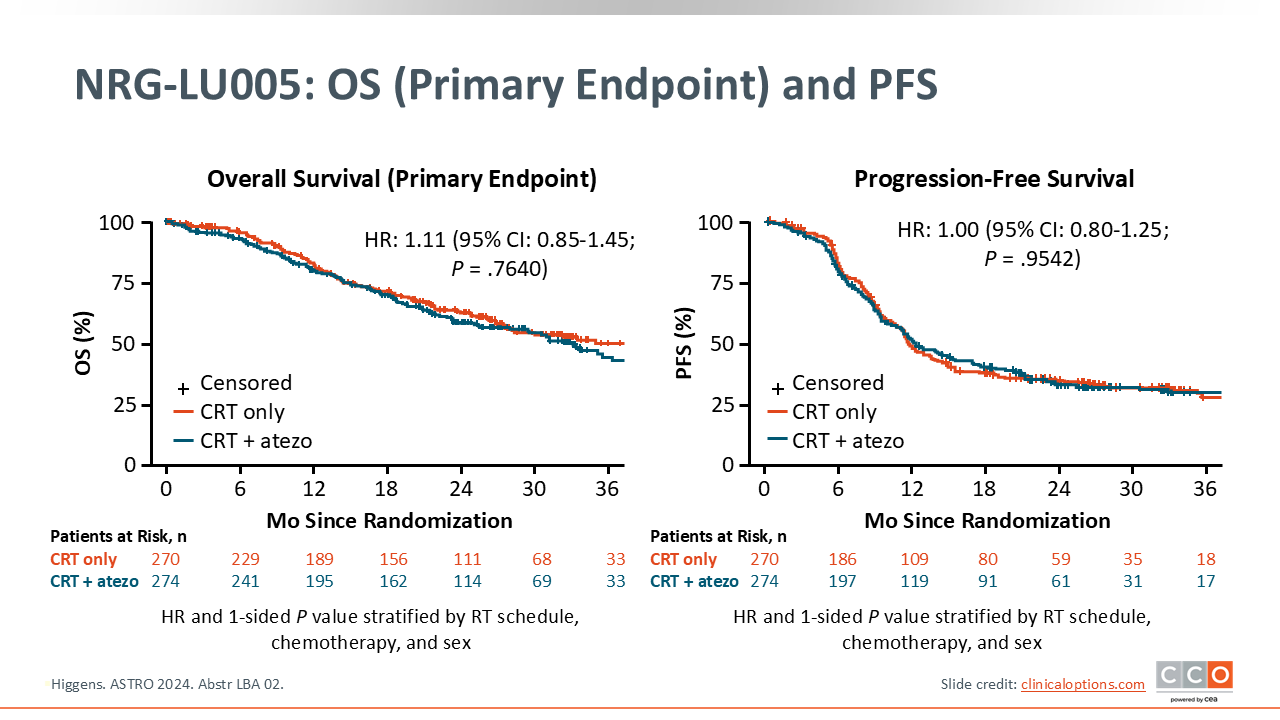
The phase III NRG-LU005 trial tested adding atezolizumab concurrently to standard platinum/etoposide CRT for LS-SCLC. The primary endpoint—OS—was not met: Median OS was 39.5 months with concurrent CRT alone vs 33.1 months with atezolizumab plus concurrent CRT (HR: 1.11; 95% CI: 0.85-1.45). PFS, likewise, showed no benefit: Median PFS was 11.5 months with concurrent CRT alone vs 12.0 months with atezolizumab plus concurrent CRT (HR: 1.00; 95% CI: 0.80-1.25).17
The trial design for NRG-LU005 differed from the ADRIATIC trial, with atezolizumab given concurrently with RT. Based on these results from NRG-LU005, it is not recommended to add an immunotherapy, like atezolizumab, concurrently to CRT in LS-SCLC.17
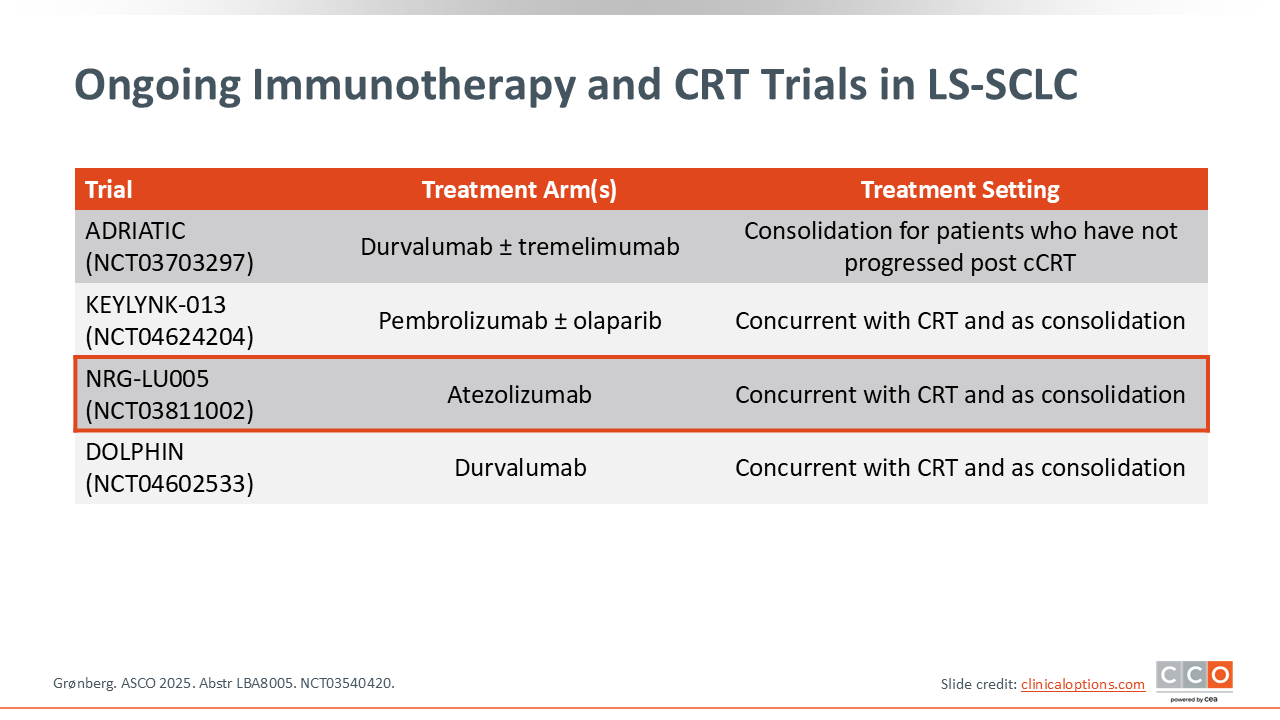
Ongoing Immunotherapy and CRT Trials in LS-SCLC
This table summarizes a few of the other ongoing trials looking at immunotherapy in the LS-SCLC setting. KEYLYNK-013, DOLPHIN, and NRG-LU005 investigated consolidation immunotherapy concurrent with CRT. Recent updates have been made regarding the NRG-LU005 trial.15
Key Takeaways and Future Directions in LS-SCLC
In conclusion, outcomes for LS-SCLC are improving with a coordinated, curative-intent approach that aligns accurate staging with appropriate local and systemic therapy. For stage I-IIA disease, mediastinal staging guides selection of surgery or stereotactic ablative radiotherapy with integration of CRT as indicated. For stage IIB-IIIC disease, concurrent platinum/etoposide CRT remains standard, and the ADRIATIC trial establishes consolidation durvalumab as a practice-changing strategy that meaningfully extends PFS and OS with manageable toxicity. By contrast, adding checkpoint blockade concurrently to concurrent CRT did not improve outcomes in NRG-LU005 and is not recommended. Thoracic RT continues to be foundational, 45 Gy twice daily over 3 weeks is a dependable benchmark, and accelerated and simultaneous integrated boost–based regimens are emerging options in experienced centers, with RT ideally initiated in cycle 1-2. Treatment should be tailored to patients’ PS, with careful attention to supportive care and selective use of PCI. Despite progress, relapse remains common, underscoring the need for biomarker-informed patient selection, optimization of RT dose/fractionation, improved toxicity mitigation, and continued clinical trial participation.