CME
Clinical Advances in Immune-Engager Therapies for the Treatment of Relapsed/Refractory B-ALL
Physicians: Maximum of 0.75 AMA PRA Category 1 Credit™
Released: April 29, 2026
Expiration: October 28, 2026
Activity
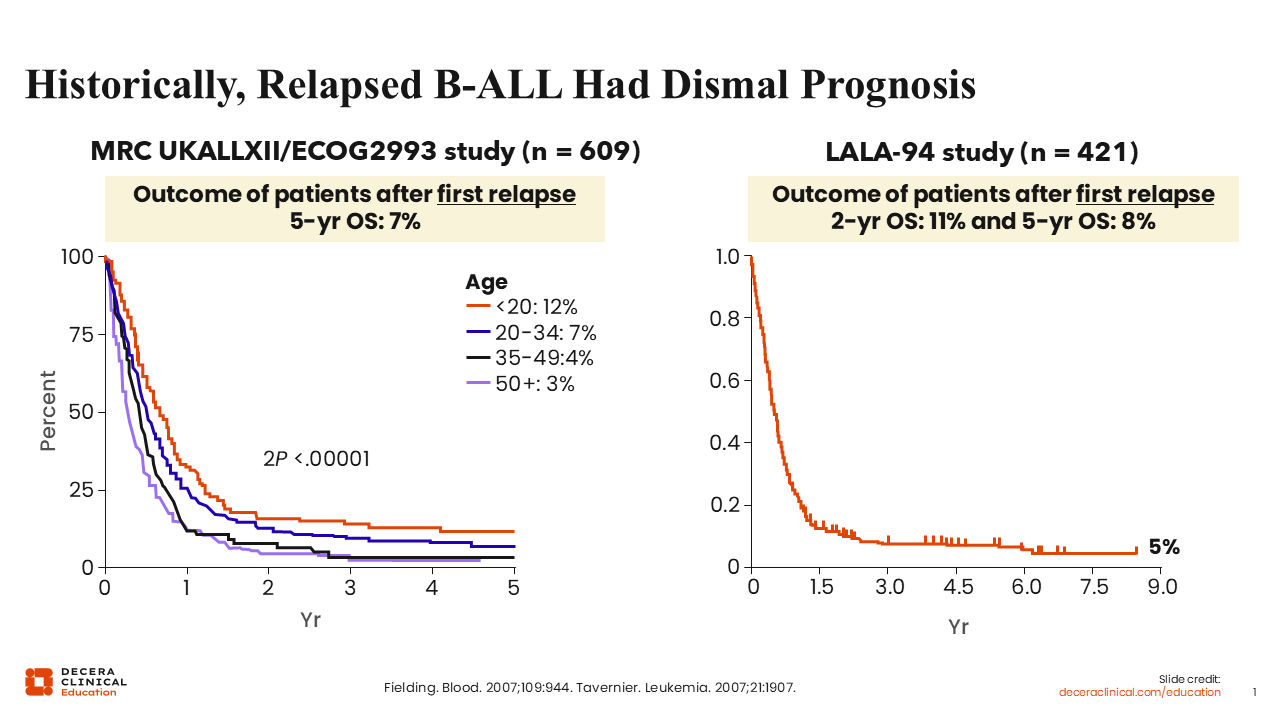
R/R B-ALL Is Historically Associated With a Dismal Prognosis
Historically, R/R B-ALL was associated with a dismal prognosis. These overall survival (OS) curves from the phase III MRC UKALLXII/ECOG2993 trial1 and the multicenter, prospective, randomized LALA-94 study2 show that the 5-year OS rates of patients with ALL after first relapse were <10% when chemotherapy alone was given.
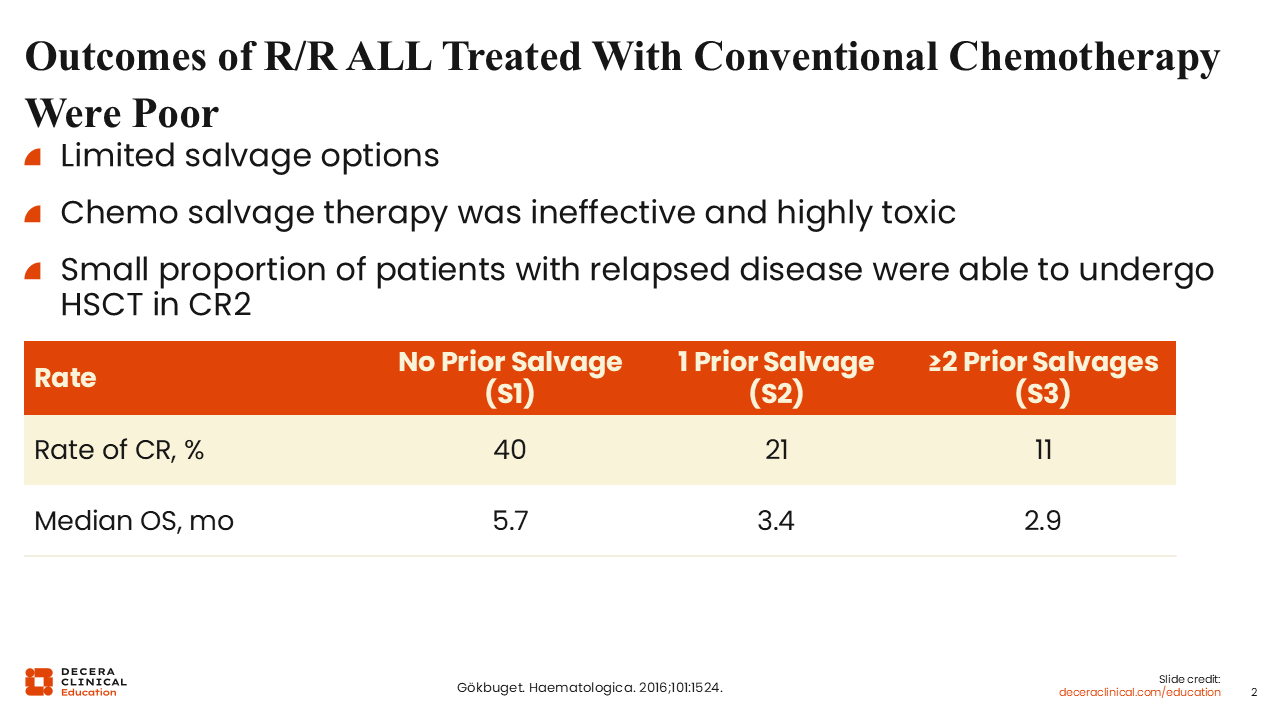
Outcomes of R/R ALL Treated With Conventional Chemotherapy
The outcomes of patients with R/R ALL treated with conventional chemotherapy are poor because salvage chemotherapy is not only largely ineffective but also highly toxic. In a retrospective analysis of 1706 adults with Philadelphia chromosome (Ph)–negative R/R B-ALL, only 40% were able to achieve complete remission (CR) or remain in remission to undergo curative transplant in CR2. These findings show that the outcomes of salvage therapy depend on the line of therapy, as the rate of CR and median OS decreased with each subsequent line of salvage chemotherapy.3
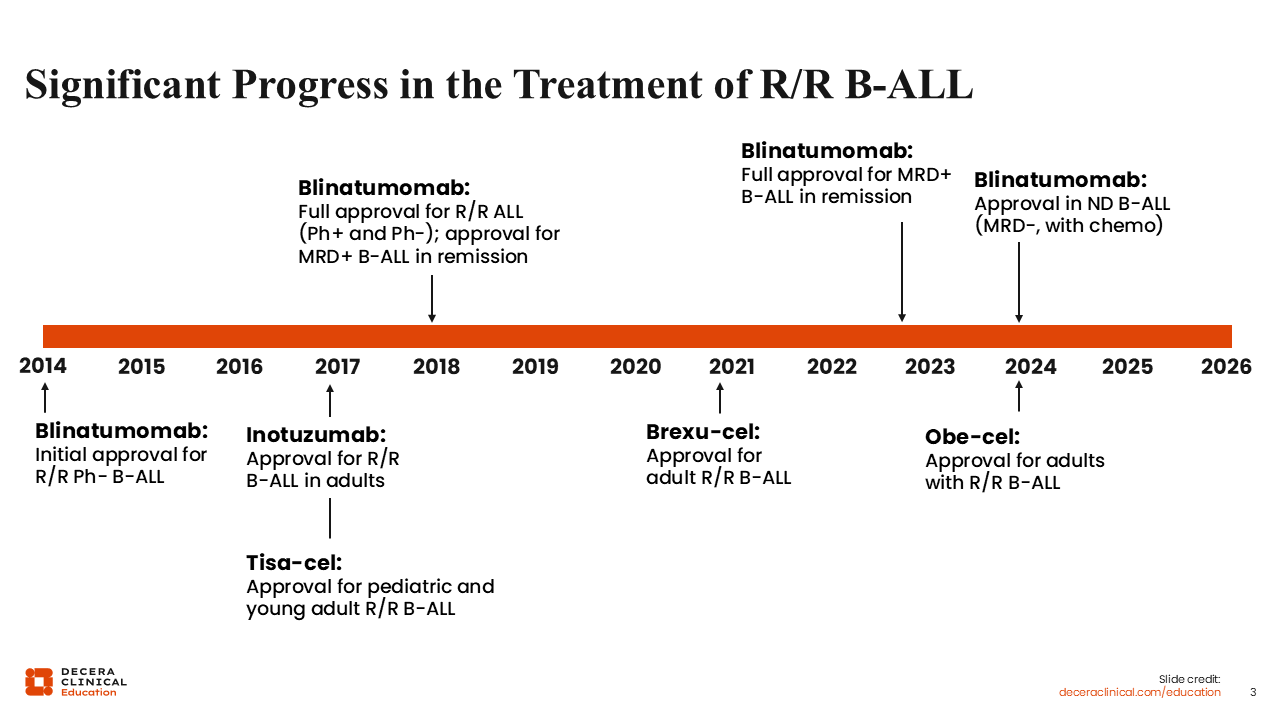
Significant Progress in the Treatment of R/R B-ALL
In the last decade or so, there has been significant progress in the treatment of R/R B-ALL, mainly because of the introduction of various immune therapies such as bispecific T-cell engagers (eg, blinatumomab), monoclonal antibodies (eg, inotuzumab ozogamicin), and CAR T-cell therapies (eg, tisagenlecleucel [tisa-cel], brexucabtagene autoleucel [brexu-cel], and obecabtagene autoleucel [obe-cel]). Blinatumomab was initially approved for R/R disease with Ph-negative B-ALL and subsequently approved for Ph-positive B-ALL as well as MRD-positive B-ALL in patients who achieve CR. Most recently (2024), blinatumomab received approval as a consolidation treatment in Ph-negative B-ALL, regardless of MRD status.4
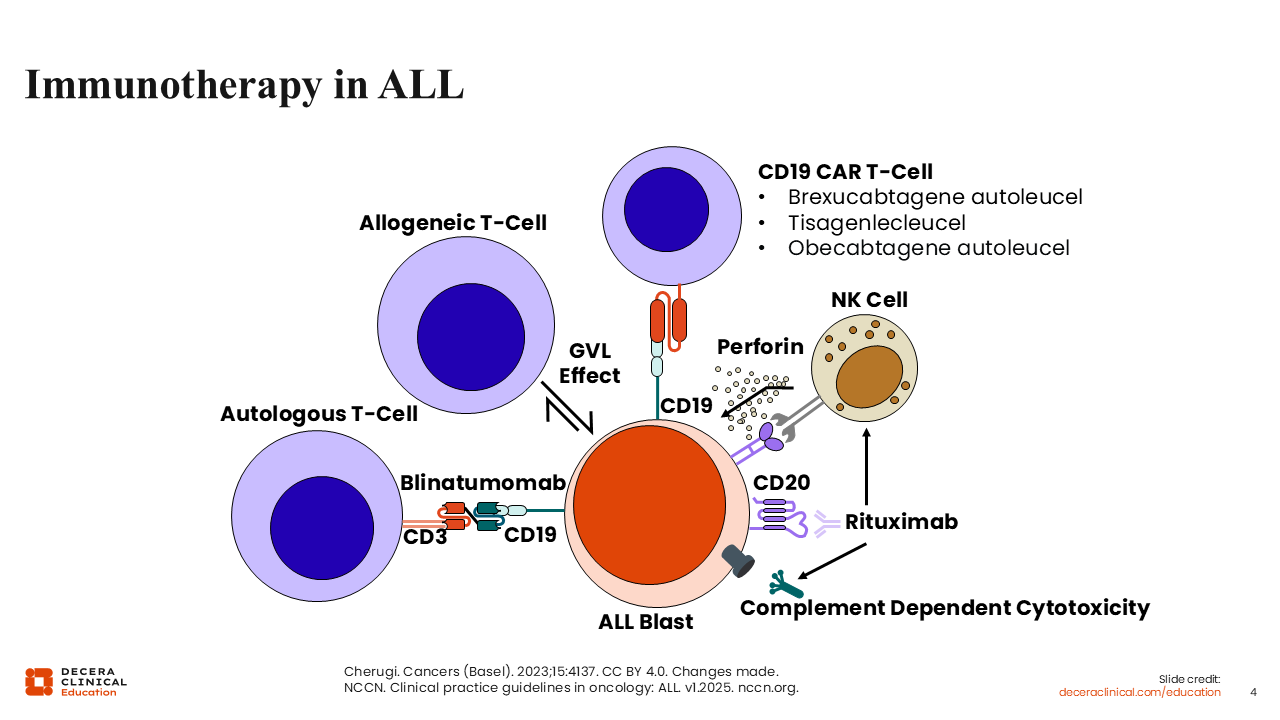
Immunotherapy in ALL
Immunotherapy has been shown to be a very effective treatment for ALL. In B-ALL, immunotherapy works by targeting different cell surface proteins such as CD19, CD20, and CD22. Rituximab is an example of a monoclonal antibody, whereas blinatumomab is an example of a bispecific T-cell engager. CAR T-cell therapies like the ones listed on the slide involve collecting and transducing T-cells from a patient to express CAR. These cells then are expanded and infused back into the patient, exerting a graft-vs-leukemia effect. Unlike allogeneic stem cell transplant, CAR T-cell therapies are highly specific to a particular surface antigen on B-cells.
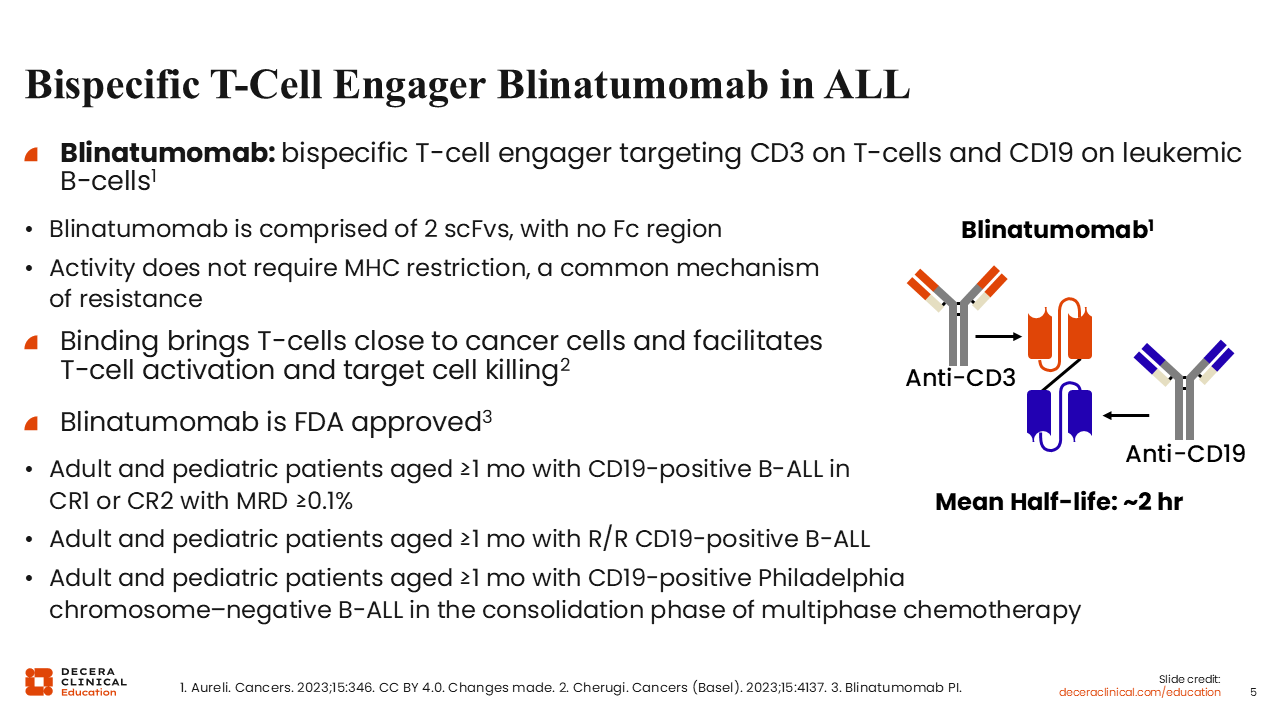
Bispecific T-Cell Engager Blinatumomab in ALL
The bispecific T-cell engager blinatumomab engages CD3-positive T-cells with CD19-positive leukemic B-cells. Its activity does not require major histocompatibility complex restriction, a common mechanism of resistance. Binding brings T-cells close to cancer cells, which facilitates T-cell activation and the direct killing of tumor cells. Blinatumomab was granted FDA approval for adults and pediatric patients with CD19-positive B-ALL in CR1 or CR2 with persistent MRD (≥0.1%), and most recently for CD19-positive Ph-negative B-ALL in the consolidation phase of multiphase chemotherapy.4
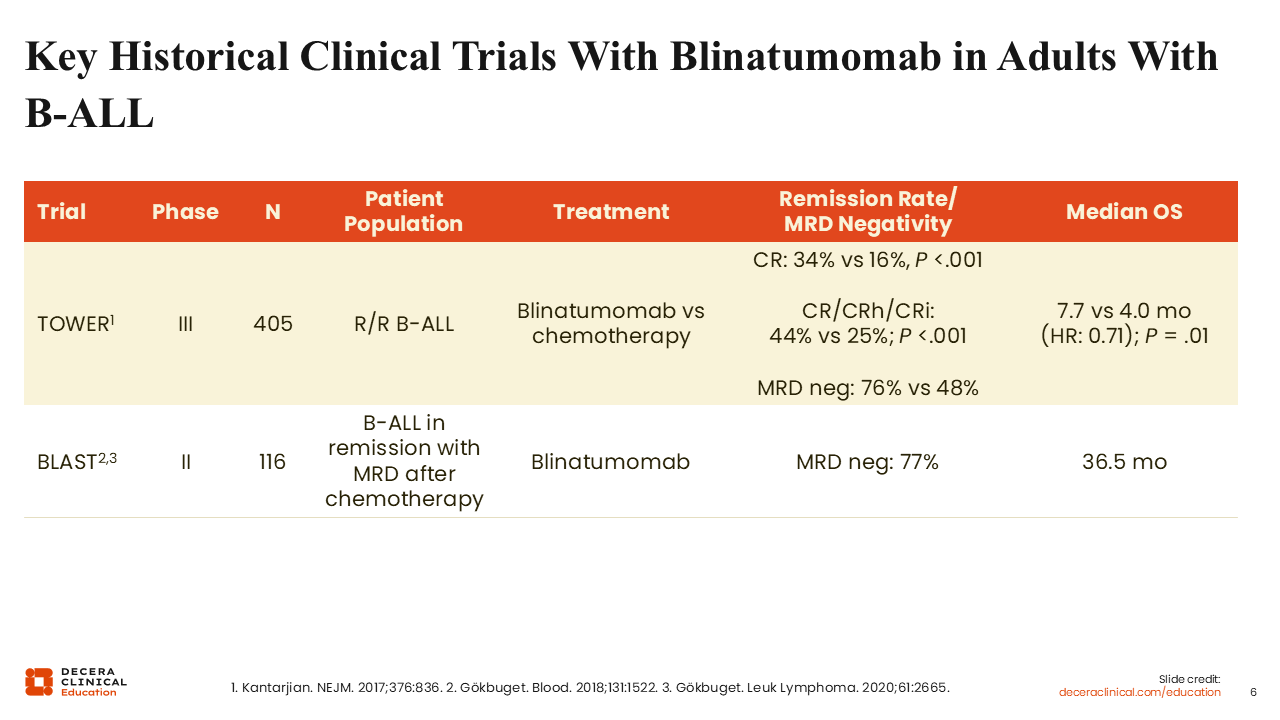
Key Historical Clinical Trials With Blinatumomab in Adults With B-ALL
This table shows the pivotal clinical trials that led to the approval of blinatumomab in adults with B-ALL. The phase III TOWER study enrolled adults with R/R Ph-negative B-ALL. Participants were randomized to blinatumomab or salvage chemotherapy. Blinatumomab was associated with a higher rate of CR with full hematologic recovery (34% vs 16%; P <.001) and prolonged OS (7.7 vs 4.0 months; P = .01),5 which led to its indication for R/R B-ALL. The phase II BLAST study was conducted in adults with B-ALL who had persistent or recurrent MRD ≥10-3 after ≥3 multiagent chemotherapy regimens. Single-agent blinatumomab led to a complete MRD response of 77% after 1 cycle of blinatumomab and a median OS of 36.5 months.6
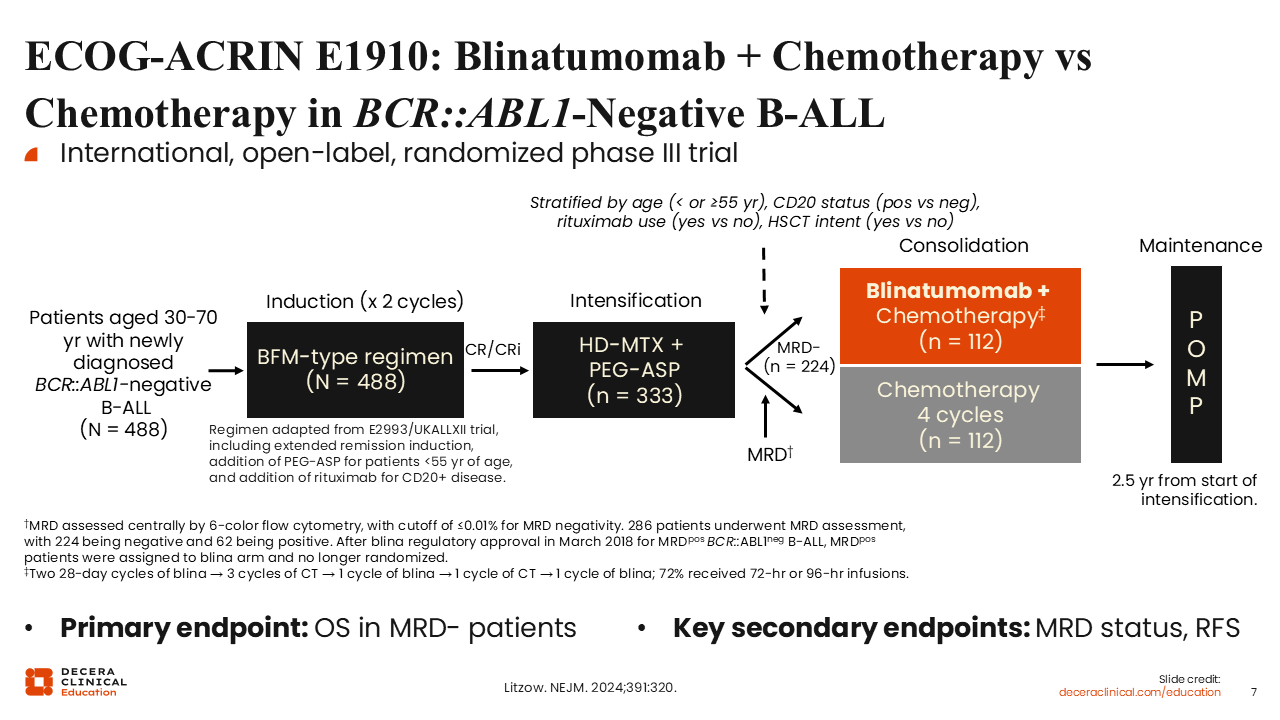
ECOG-ACRIN E1910: Blinatumomab Plus Chemotherapy vs Chemotherapy in BCR::ABL1-Negative B-ALL
Blinatumomab was most recently approved for consolidation therapy in adult and pediatric patients aged 1 month or older with CD19-positive Ph-negative (BCR::ABL1 translocation negative) B-ALL in the consolidation phase, regardless of MRD status, based on the ECOG-ACRIN E1910 study. This international, open-label phase III study randomized adults between the ages of 30 and 70 years with BCR::ABL1-negative B-ALL who achieved MRD-negative remission after 2 cycles of induction therapy and 1 cycle of intensification therapy to either blinatumomab in combination with consolidation chemotherapy or consolidation chemotherapy alone. The primary endpoint was OS, and key secondary endpoints included MRD status and relapse-free survival.7
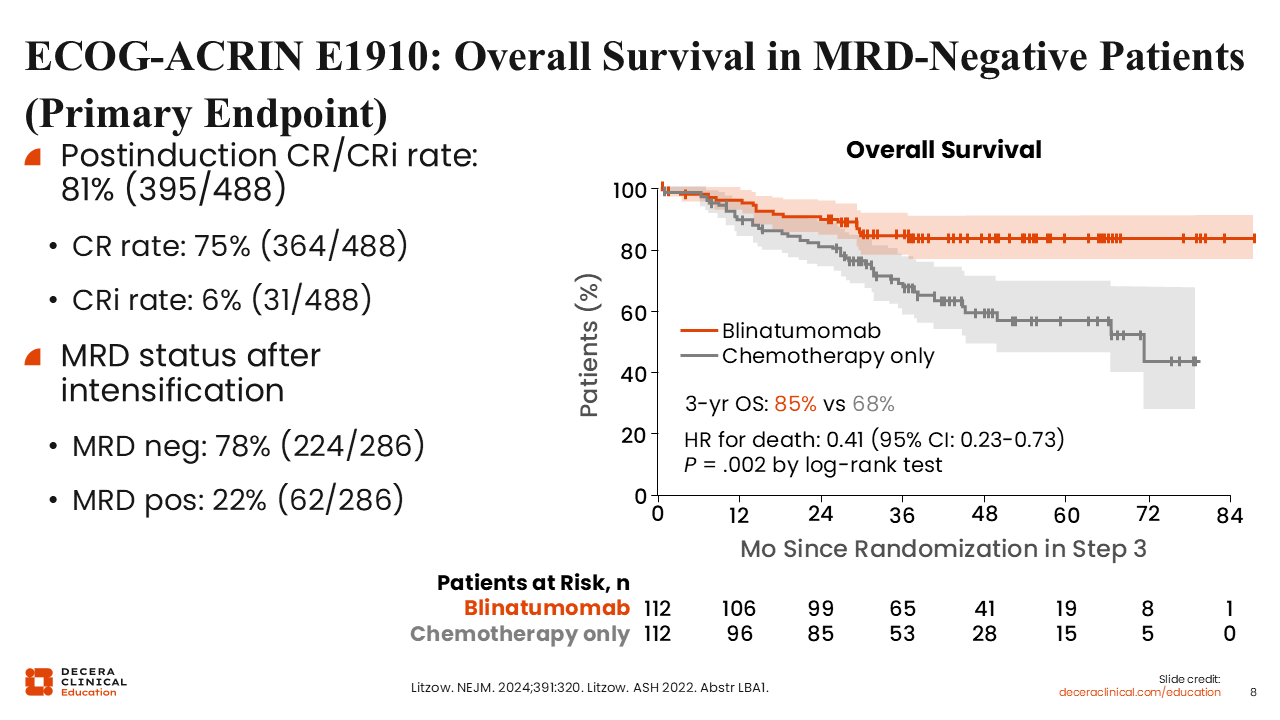
ECOG-ACRIN E1910: OS in MRD-Negative Patients (Primary Endpoint)
CR with or without full count recovery was achieved in 81% of enrolled patients. 286 (78%) of these patients were randomized to either treatment arm after intensification therapy, where 78% achieved MRD negativity. Blinatumomab as part of consolidation led to an OS improvement in patients with MRD-negative disease, as demonstrated by the Kaplan-Meier curve shown here. The 3-year OS rate was 85% for patients who received blinatumomab plus chemotherapy vs 68% for patients who received chemotherapy alone (P = .002).7
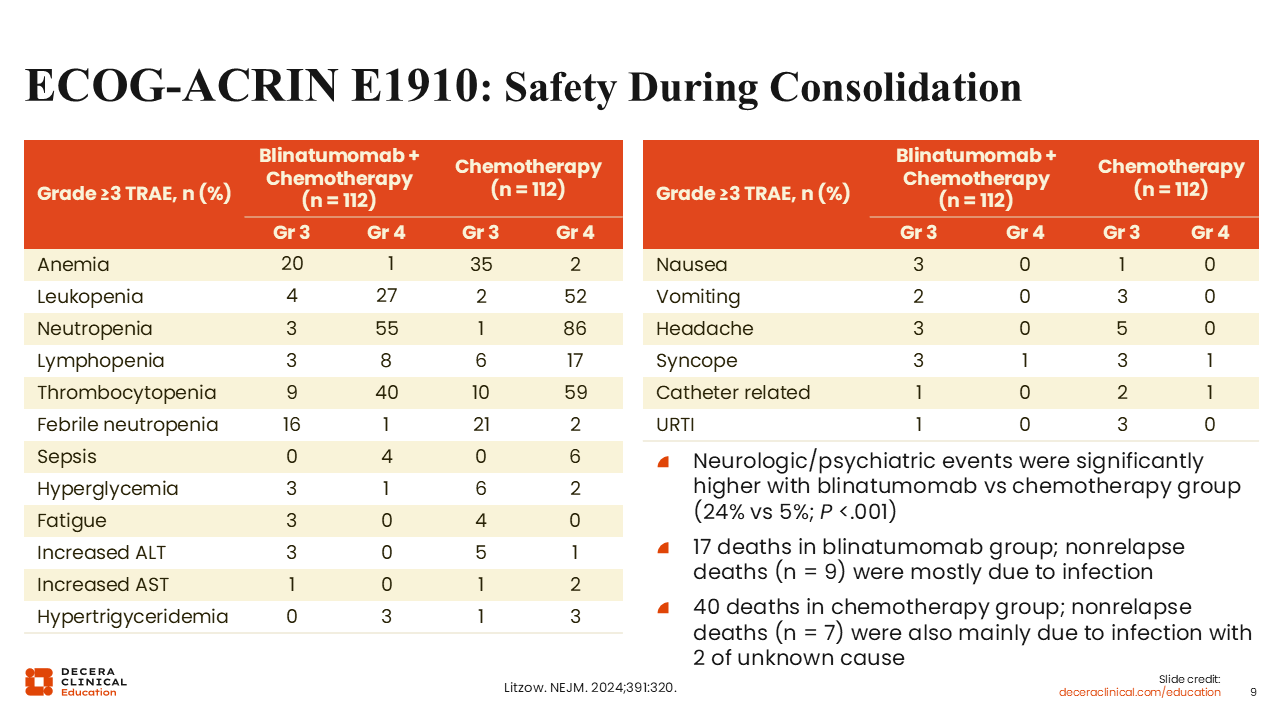
ECOG-ACRIN E1910: Safety During Consolidation
In general, blinatumomab was well tolerated. Shown here are the numbers of patients who experienced grade ≥3 treatment-related adverse events. There were significantly more neurologic and psychiatric events in patients who received blinatumomab vs chemotherapy only (23% vs 5%; P <.001). Overall, there were 17 deaths in the blinatumomab group, including 9 from nonrelapse-related causes (mainly infection), and 40 in the chemotherapy group, including 7 from nonrelapse-related causes (mainly infection). The remainder of deaths were mostly because of disease relapse.7
Medical Need for New and Innovative Immune-Targeted Therapies
There are challenges associated with immune-targeted therapies for B-ALL, and thus, there is room for improvement. Although recent studies incorporating blinatumomab as part of initial therapy have led to improved outcomes for these patients, with a 2-year OS rate of approximately 93%,8 as more patients develop resistance to current immunotherapies, novel therapies will be needed. Since blinatumomab is being integrated as part of frontline therapy for B-ALL, most relapses will involve patients who fail prior blinatumomab.
Blinatumomab Limitations
There are some limitations associated with continuous IV blinatumomab, including a correlation between higher pretreatment disease burden and a lower response with blinatumomab.9 In general, blinatumomab is not highly effective for proliferative leukemia or leukemia with a high disease burden. The continuous infusion of blinatumomab creates some challenges and can be both inconvenient for the patient and pose logistical challenges for the healthcare system as well as the patient.
In addition, extramedullary disease is a common mechanism of relapse and resistance to continuous IV blinatumomab.10,11 As blinatumomab is moved to the frontline setting, the expectation is that most relapses will occur in patients who had prior blinatumomab. This creates a challenging situation with fewer treatment options for patients who relapse after frontline therapy. Thus, new salvage therapies or agents that can treat patients who relapse after prior blinatumomab exposure are warranted.
SC Blinatumomab in R/R B-ALL: Efficacy
SC blinatumomab is under investigation to improve the convenience of administration, but it may also lead to better outcomes and activity. SC blinatumomab is given daily for the first week and 3 times per week thereafter. The dose of SC blinatumomab is significantly higher than the dose of continuous IV blinatumomab. Two doses of SC blinatumomab were tested in a phase I/II study in patients with R/R B-ALL. Both the lower dose (250 µg daily during the first week, followed by 500 µg 3 times per week thereafter) and the higher dose (500 µg daily during the first week, followed by 1000 µg 3 times per week thereafter) were effective, with CR and CR with partial hematologic recovery (CRh) rates of 75% and 79%, respectively. Moreover, the majority of responders achieved MRD negativity despite this being a heavily pretreated population, with many patients having had prior IV blinatumomab, CAR T-cell therapy, hematopoietic stem cell transplant, or inotuzumab ozogamicin.
The CR/CRh rates in patients previously exposed to blinatumomab (65%), CAR T-cell therapy (64%), hematopoietic stem cell transplant (56%), and inotuzumab ozogamicin (69%) were also encouraging. According to previous studies in patients with R/R B-ALL receiving continuous IV blinatumomab, pretreatment disease burden can help predict response. A high disease burden, defined as ≥50% bone marrow blasts, is associated with a lower response to blinatumomab. As this table indicates, SC blinatumomab is effective regardless of disease burden.12
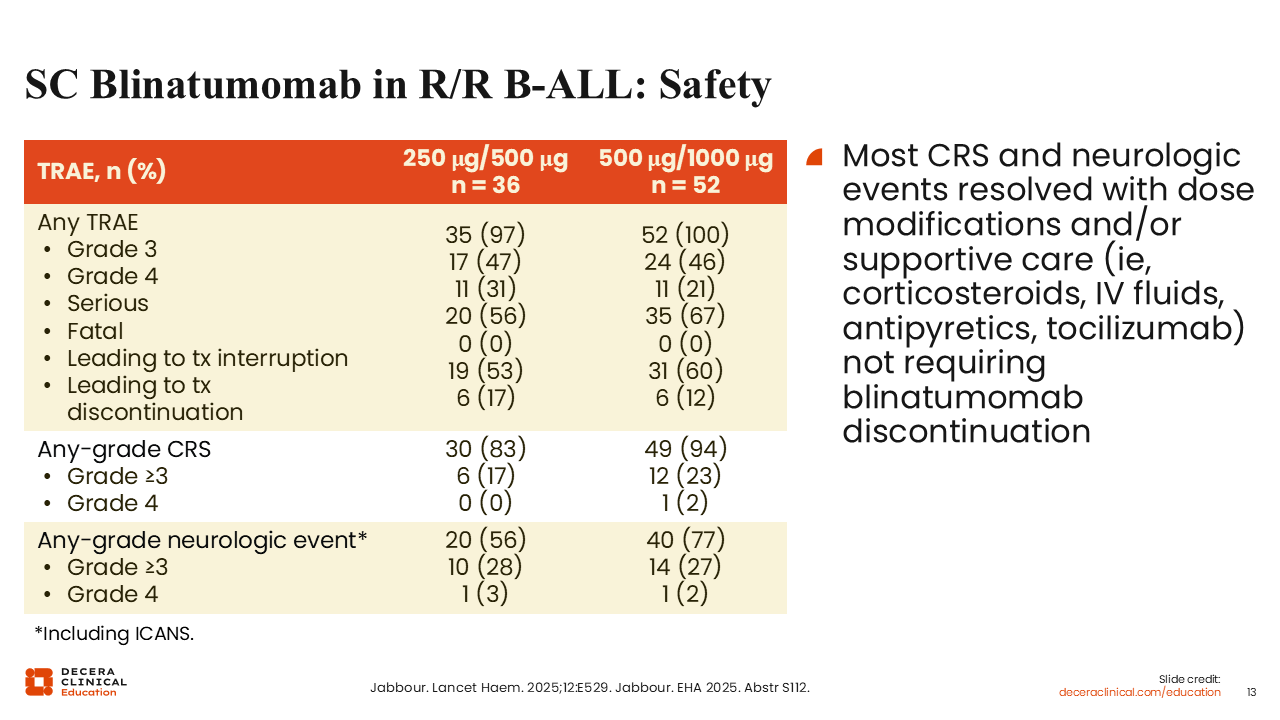
SC Blinatumomab in RR/B-ALL: Safety
In terms of toxicity, the higher dose of SC blinatumomab resulted in more frequent higher-grade CRS and neurotoxicity compared with continuous IV blinatumomab, as expected. Grade ≥3 CRS was reported in 17% of patients on the lower dose and 23% on the higher dose. Grade ≥3 neurologic events, including immune effector cell–associated neurotoxicity syndrome (ICANS), were reported in 28% of patients on the lower dose and 27% on the higher dose. Most of these events occurred early during the first cycle, and all grade ≥3 treatment-related CRS and neurologic events resolved.12
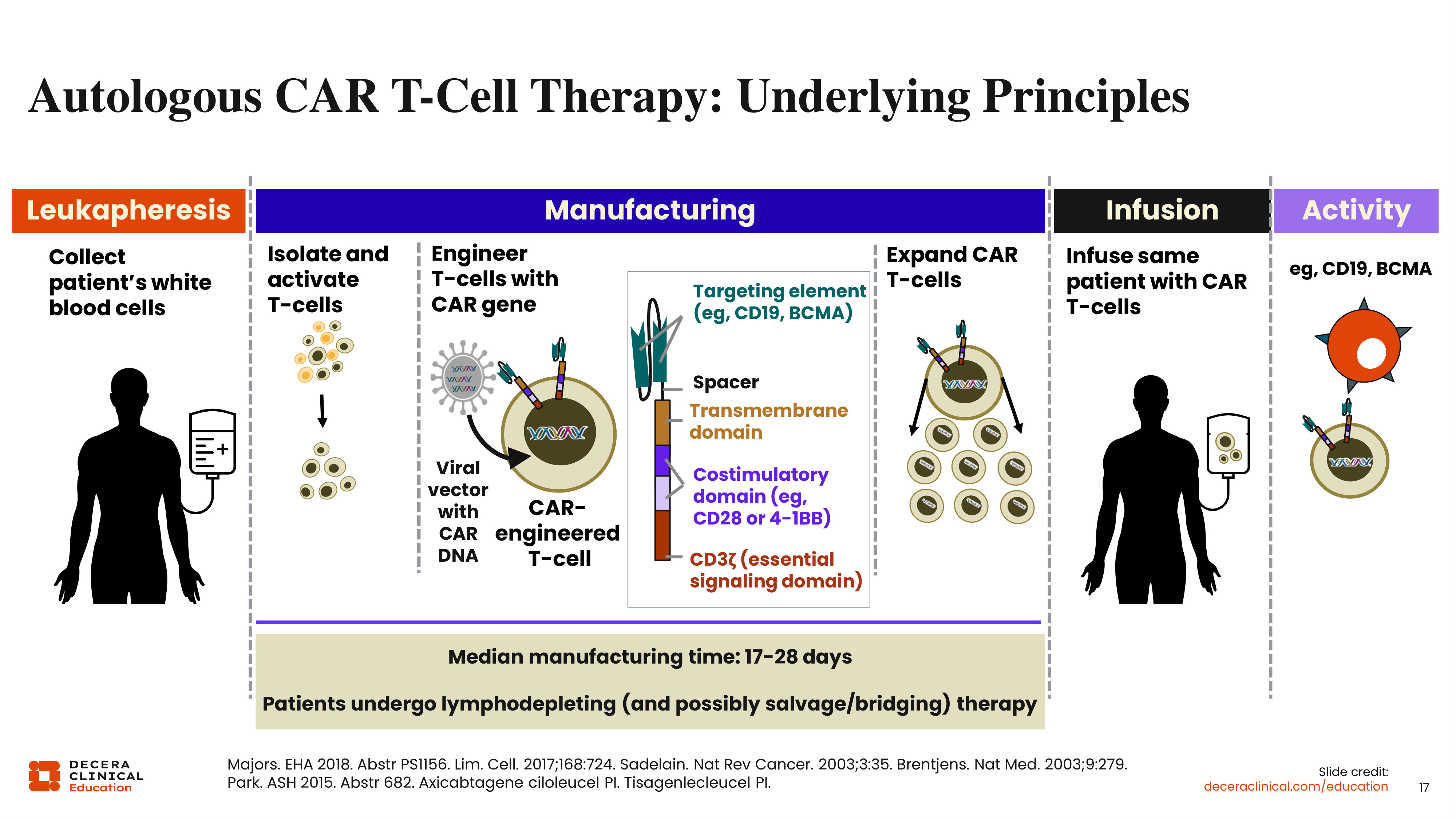
Autologous CAR T-Cell Therapy: Underlying Principles
CAR T-cell therapy has emerged as a very effective therapy in various hematological malignancies, including ALL, which was the first hematologic malignancy to receive FDA approval for a CAR T-cell therapy. All currently approved CAR T-cell therapies in ALL require collecting T-cells from the patient via leukapheresis. Then, approximately 17-28 days are required to manufacture the CAR T-cells. These T-cells are isolated, transduced with a CAR, expanded, and then ready for infusion after the patient has received lymphodepleting therapy to reduce the disease burden and improve T-cell expansion and persistence.13
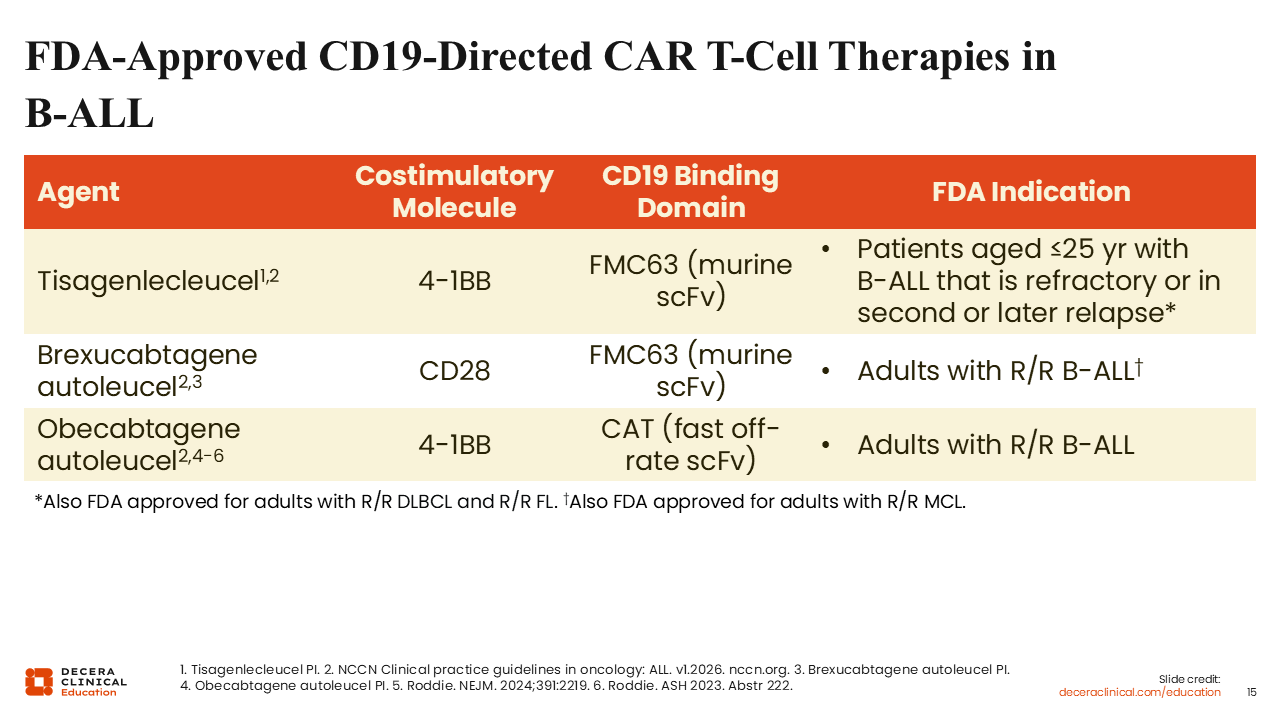
FDA-Approved CD19-Directed CAR T-Cell Therapies in B-ALL
There are currently 3 FDA-approved CD19-directed CAR T-cell therapies for B-ALL: tisa-cel, brexu-cel, and obe-cel. Tisa-cel and obe-cel are costimulated via 4-1BB, whereas brexu-cel is costimulated via CD28. This difference can influence the early expansion as well as persistence of CAR T-cells after infusion. 4-1BB–costimulated CAR T-cells tend to persist longer than CD28-costimulated CAR T-cells, which usually expand early on. These products also differ in terms of their CD19 binding domain. Tisa-cel and brexu-cel contain the FMC63 binding domain, whereas obe-cel contains a low-affinity binding domain that may contribute to its decreased toxicity and potentially alleviate CAR T-cell exhaustion. These agents also have different indications: Brexu-cel and obe-cel are approved for adults with R/R B-ALL regardless of age,14,15 whereas tisa-cel is approved only for children and young adults up to the age of 25 years.13
Real-world Safety and Efficacy of Brexu-cel in R/R B-ALL
Real-world safety and efficacy data for brexu-cel in adults with R/R B-ALL are summarized here. These findings were obtained from a retrospective analysis of 189 adults treated with brexu-cel outside of clinical trials at 31 centers in the United States. These patients were heavily pretreated (median prior lines of therapy: 4). In total, 41% of patients had undergone transplant. Almost 60% of patients had received prior blinatumomab, and almost one half (48%) had received prior inotuzumab ozogamicin. Because these are real-world data, the patient population differed from that in the phase II ZUMA-3 study that led to the approval of brexu-cel, where only patients with active R/R disease were enrolled. Of note, 42% of patients in this real-world analysis would not have met the ZUMA-3 eligibility criteria, as 27% had only MRD-positive disease and 15% were treated for MRD-negative disease.
Brexu-cel was shown to be efficacious. At a median follow-up of 11.4 months, 151 patients achieved CR, and most responders (79%) achieved MRD negativity, similar to ZUMA-3. The median time to relapse was 154 days, median progression-free survival was 9.5 months, and median OS was not reached at the time of the report. Grade ≥3 CRS was noted in 11% of patients, but grade ≥3 ICANS was noted in 31% of patients.16
Nuances With CAR T-Cell Therapies
Some challenges exist with currently available CAR T-cell therapies. First, these therapies are not available off the shelf. In addition, it can take approximately 4-5 weeks from the time patients undergo leukapheresis to the time they receive the therapy, which is a logistical concern. Relapsed leukemia can be challenging to control until the CAR T-cells are ready to administer. However, one concern with bridging therapy is that patients may experience infections that can preclude them from receiving CAR T-cell therapy. There is still a significant risk of CRS and ICANS, which are more common than with bispecific antibodies and can be more severe and last longer.
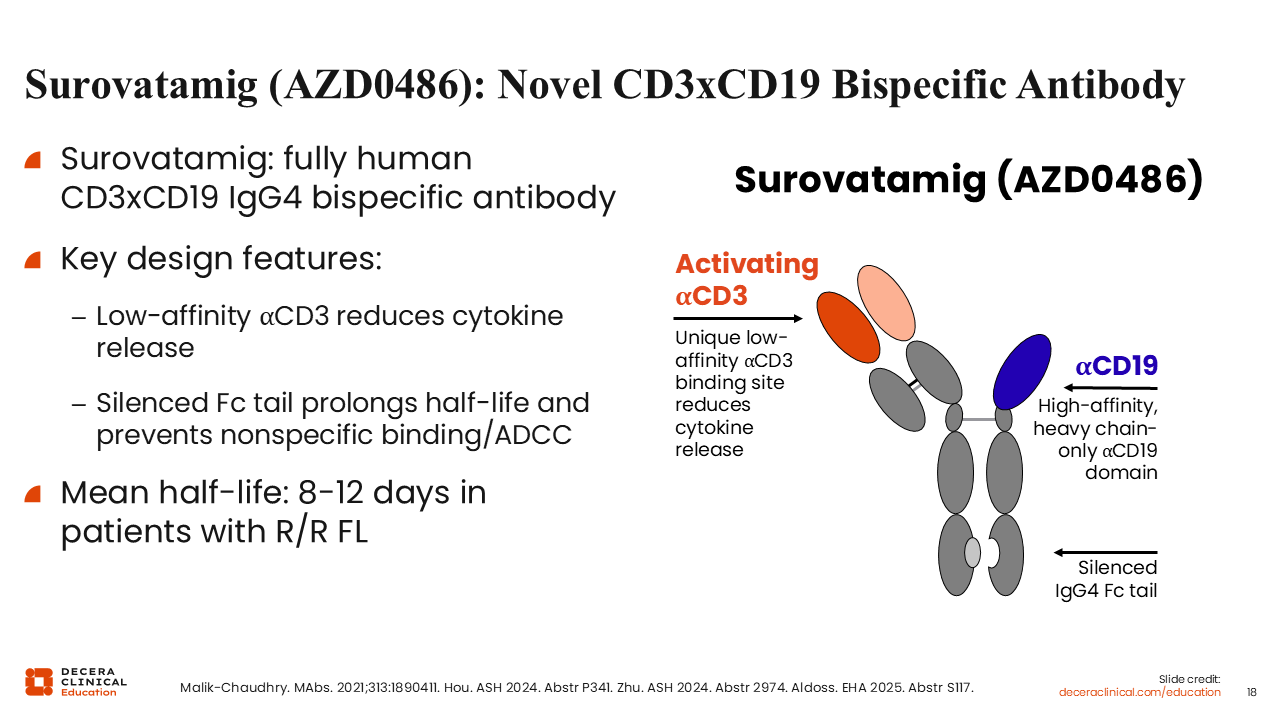
Surovatamig (AZD0486): A Novel CD3xCD19 Bispecific Antibody
New bispecific products may be able to address some of the current challenges with currently approved continuous IV T-cell engager treatment. One such promising drug is surovatamig, previously referred to as AZD0486. Surovatamig is a novel, fully human CD3xCD19 IgG4 bispecific activator of T-cells. It has a unique low-affinity CD3 binding site that leads to reduced cytokine release and reduced toxicity. It also has a silenced IgG4 Fc tail, which prolongs the half-life, with a mean half-life in the range of 8-12 days.17 This allows administration every 2-4 weeks and prevents nonspecific binding and antibody-dependent cellular cytotoxicity. It also contains a high-affinity, heavy chain–only CD19 domain.
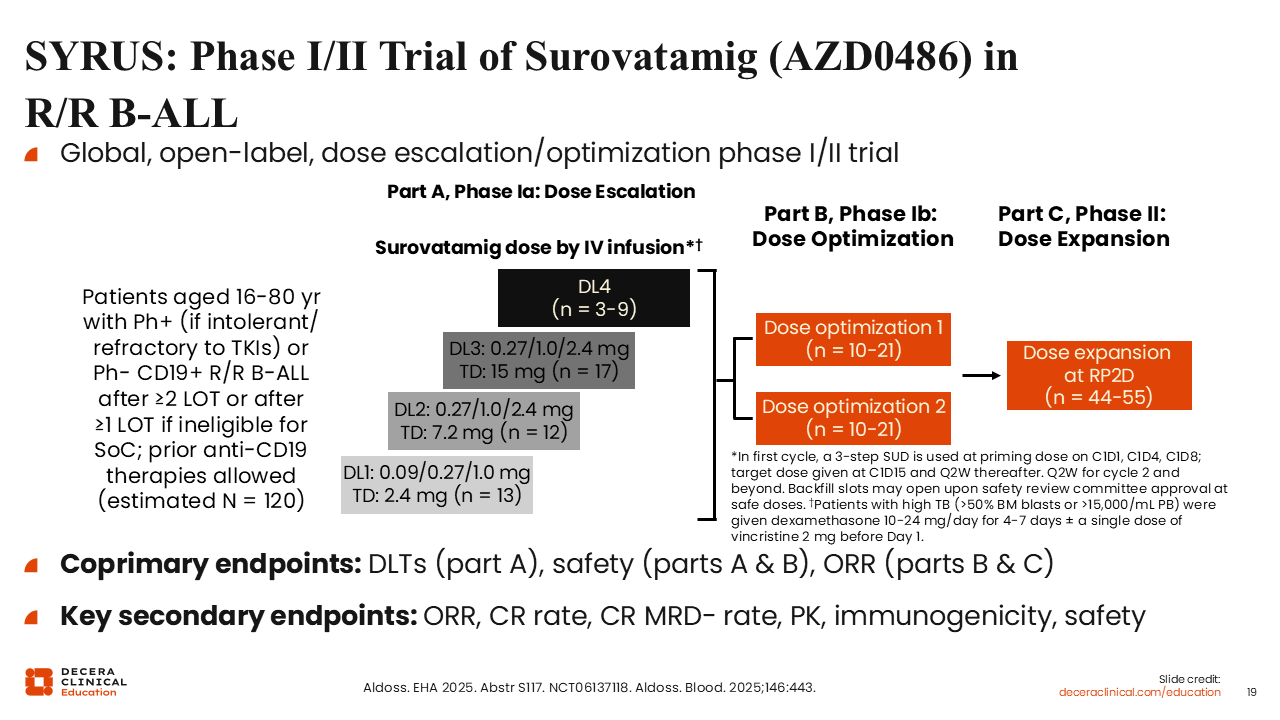
SYRUS: Phase I/II Trial of Surovatamig (AZD0486) in R/R B-ALL
Surovatamig is actively being tested in the phase I/II SYRUS study in patients aged 16-80 years with Ph-negative or Ph-positive (if intolerant or refractory to tyrosine kinase inhibitors) CD19-positive R/R B-ALL who have received ≥2 prior lines of therapy (or ≥1 prior line and who are not eligible for standard of care). Patients who received prior CD19-targeted therapies were eligible. This trial has 3 parts. Part A is the dose escalation phase, part B is the dose optimization phase, and part C is the dose expansion phase. To reduce its toxicity, surovatamig was given via step-up dosing. In the first cycle, the dose was increased on Days 1, 4, and 8, and then the target dose was given on Day 15 and every 2 weeks thereafter.
In part A, 3 target doses were tested: 2.4 mg, 7.2 mg, and 15.0 mg. Coprimary endpoints were dose-limiting toxicities, safety, and ORR. Key secondary endpoints included ORR, CR rate, CR MRD-negative rate, pharmacokinetics (PK), immunogenicity, and safety (NCT06137118).18
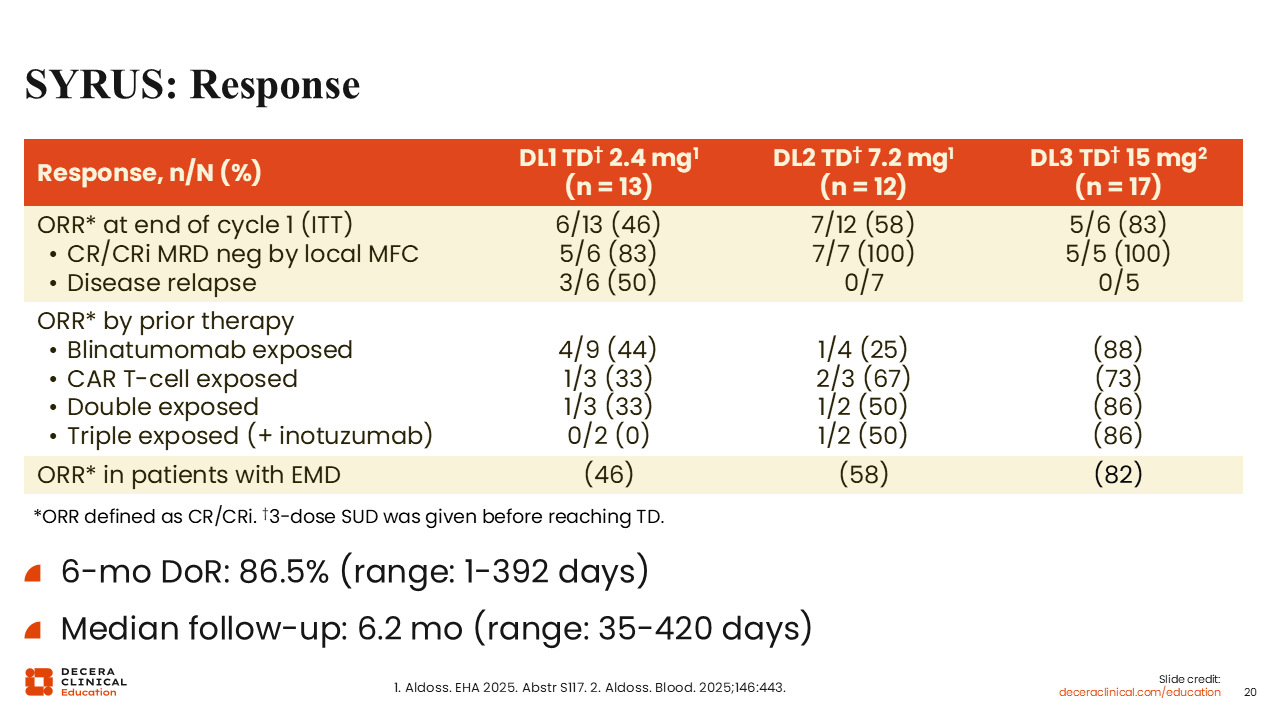
SYRUS: Response
This patient population was heavily pretreated. Regardless of prior therapy, surovatamig demonstrated a dose-dependent improvement in response. The ORR at the end of cycle 1 was 46% at target dose level 1, improved to 58% at dose level 2, and further improved to 83% at dose level 3. Most responders achieved MRD negativity. What was encouraging was the response in patients who received prior CD19-targeted therapy, especially the 80% rate among patients who had been exposed to CAR T-cell therapy at dose level 3. Even the response rates among patients with extramedullary disease were encouraging. The 6-month duration of response was 86.5%, with a median follow-up of 3.2 months.18
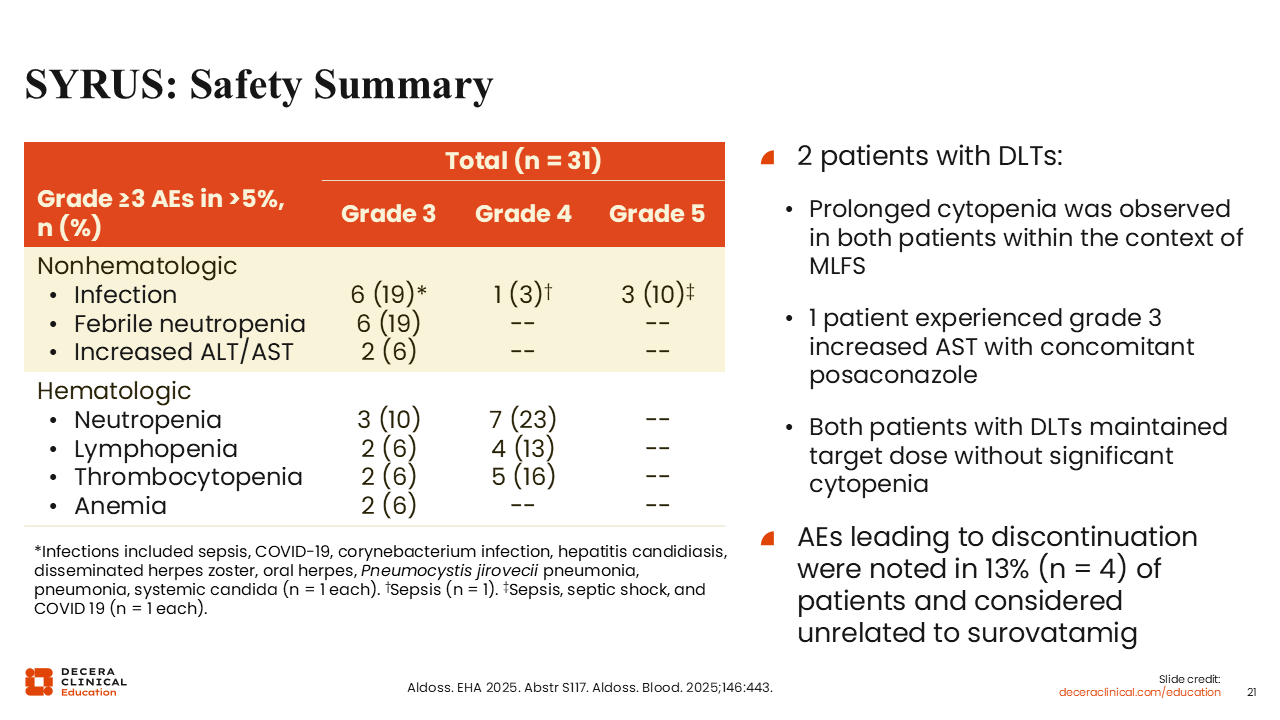
SYRUS: Safety Summary
Overall, surovatamig was well tolerated. Two patients experienced dose-limiting toxicities during the dose escalation phase, both prolonged cytopenia in the context of marrow leukemia–free state. Treatment was continued at the target dose without recurrence. One patient experienced grade 3 elevation in aspartate aminotransferase that eventually resolved with continuous dosing. The rate of adverse events leading to discontinuation was 13%, and the events were considered not related to surovatamig.18
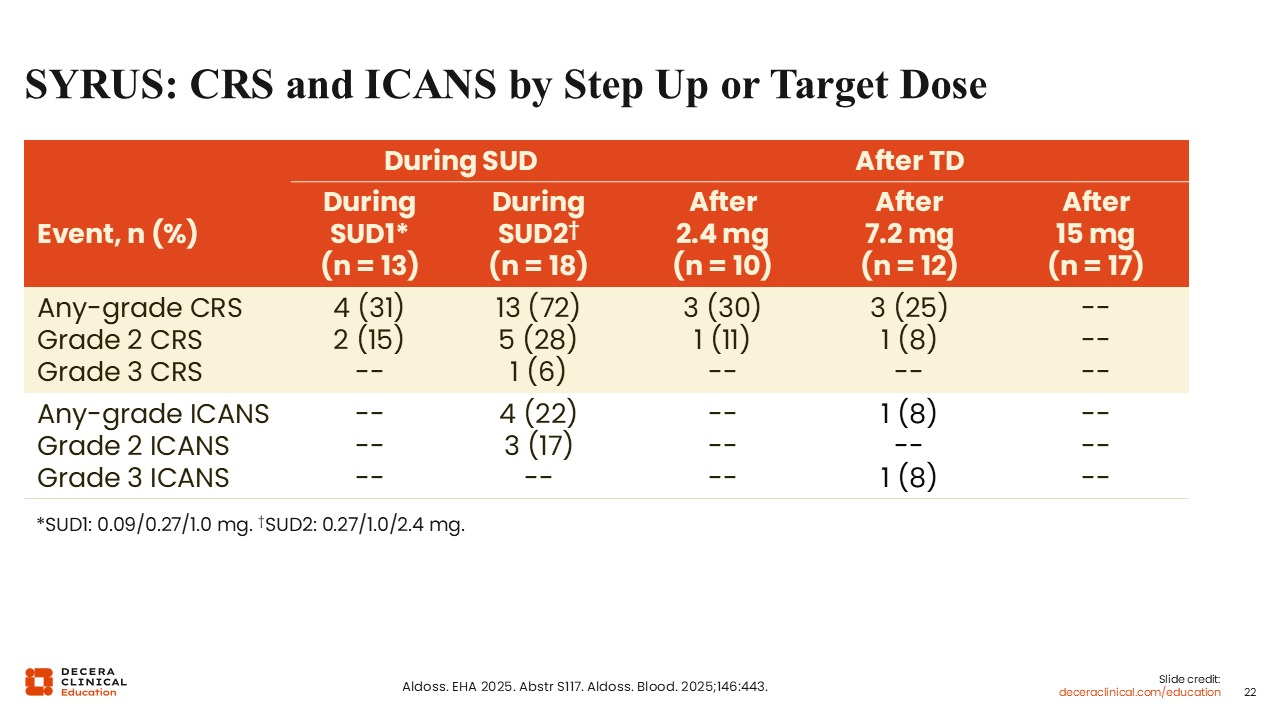
SYRUS: CRS and ICANS by Step-up or Target Dose
Grade 3 CRS and ICANS were rare, occurring in only 1 patient each. The case of grade 3 CRS occurred during step-up dosing and not after the target dose. ICANS was observed only at dose level 2 after the target dose, but most cases of CRS and ICANS were grade 1, with very few grade 2 events.18
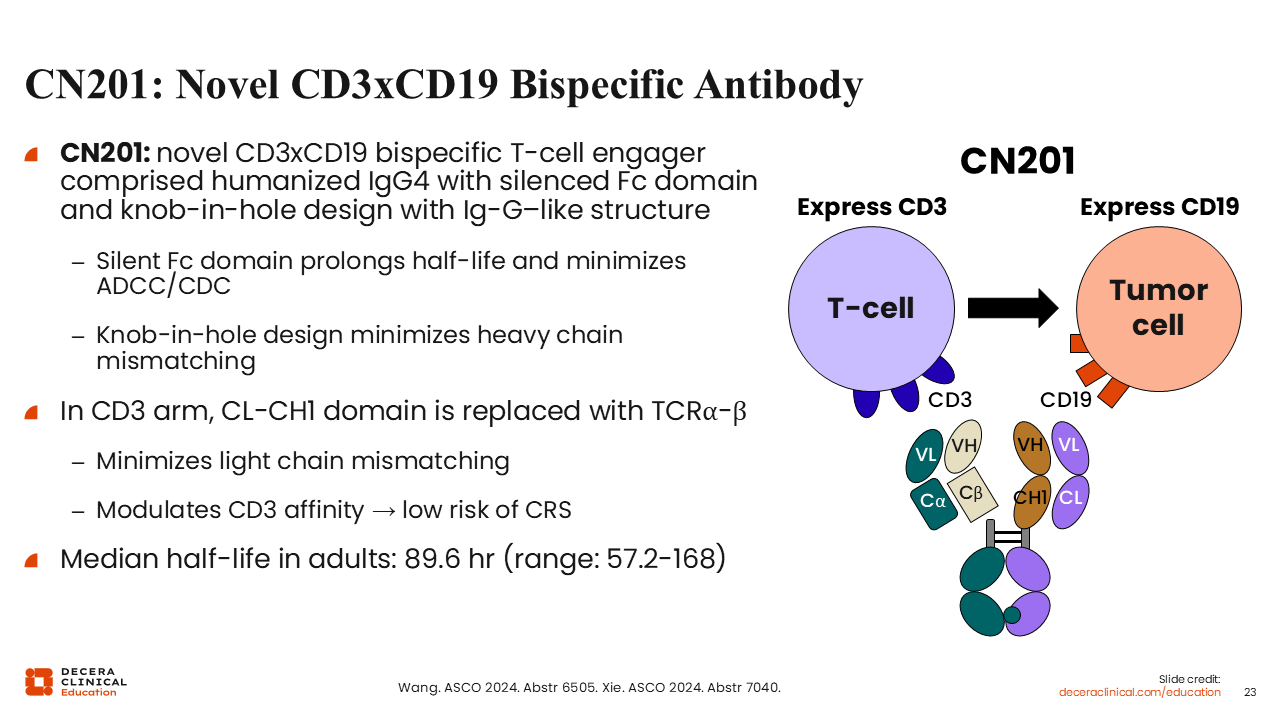
CN201: A Novel CD3xCD19 Bispecific Antibody
CN201 is another novel CD3xCD19 bispecific antibody comprised of human IgG4 with a silenced Fc domain and a knob-in-hole design with an IgG-like structure. This structure leads to a prolonged half-life and minimizes both antibody-dependent cellular cytotoxicity and heavy chain mismatching. CN201 also has low-affinity CD3 binding, which is designed to reduce cytokine release and thus decrease the risk of CRS. Its median half-life in adults is 89.6 hours, which allows once-weekly dosing.19,20
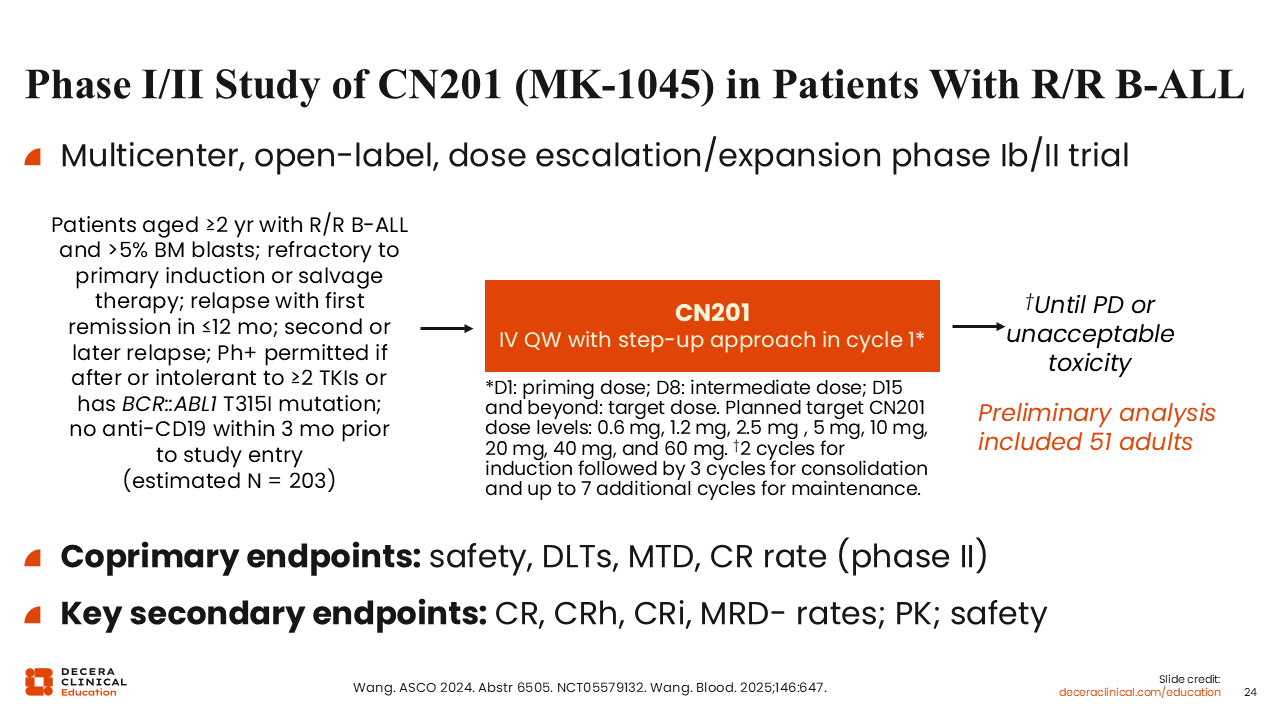
Phase I/II Study of CN201 in Patients With R/R B-ALL
CN201 is being tested in a phase Ib/II study in patients aged 2 years or older with R/R B-ALL. In the preliminary analysis, 42 adults received weekly infusions of escalating doses, with a priming dose on Day 1, followed by an intermediate dose on Day 8. The target dose was given on Day 15 and beyond.19 Coprimary endpoints are safety, dose-limiting toxicities, maximum tolerated dose, and CR rate. Key secondary endpoints include CR/CRh/CR with incomplete count recovery (CRi)/MRD-negative rates, PK, and safety (NCT05579132).
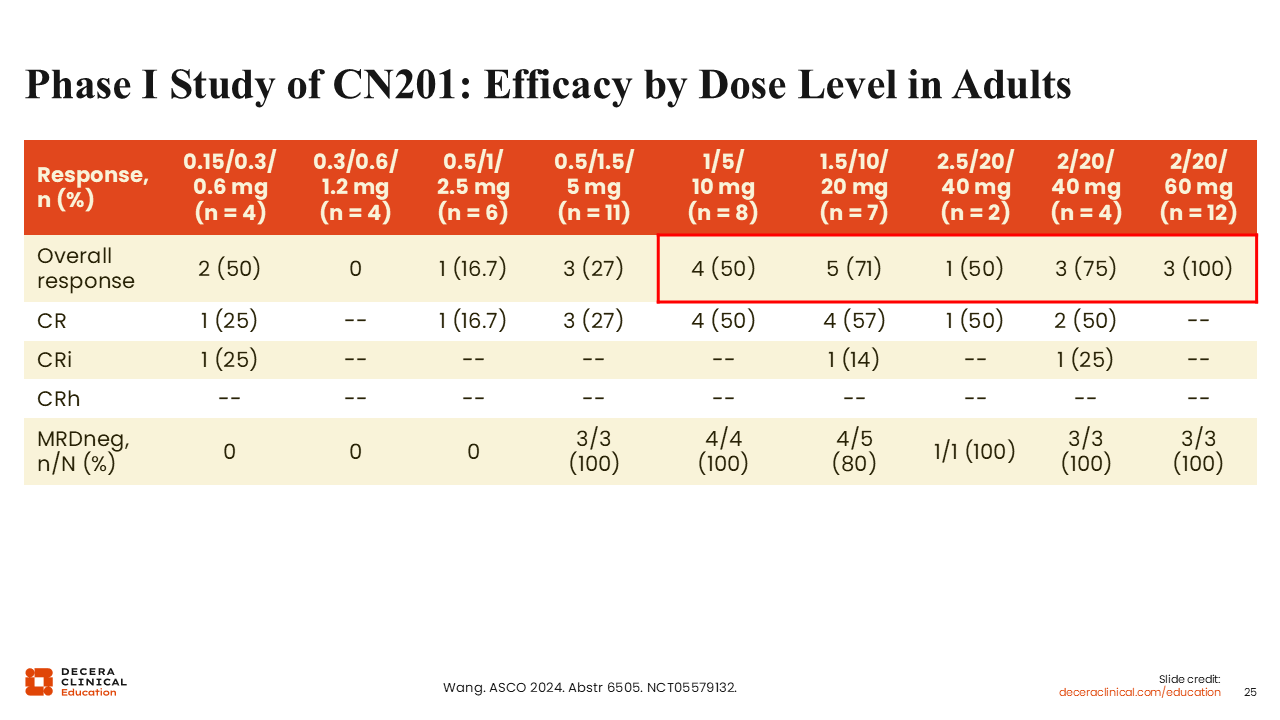
Phase I Study of CN201: Efficacy by Dose Level in Adults
Phase I of this study was performed in China. Patients were not as heavily pretreated as those in the SYRUS study, and there was less exposure to CD19-targeted therapies. Based on updated results from 75 participants, ORRs improved with CN201 in a dose-dependent manner. Among 73 participants evaluable for efficacy, 40 (55%) achieved CR/CRi/CRh. Among participants who received target doses of ≥60 mg, 83% achieved CR/CRi/CRh, with a median duration of complete remission of 11.3 months. In total, 18 of 20 participants who received target doses of ≥60 mg with a CR/CRi/CRh achieved MRD negativity. Among patients previously exposed to blinatumomab or CAR T-cell therapy who had received a target dose ≥60 mg, 100% achieved CR/CRi/CRh.21
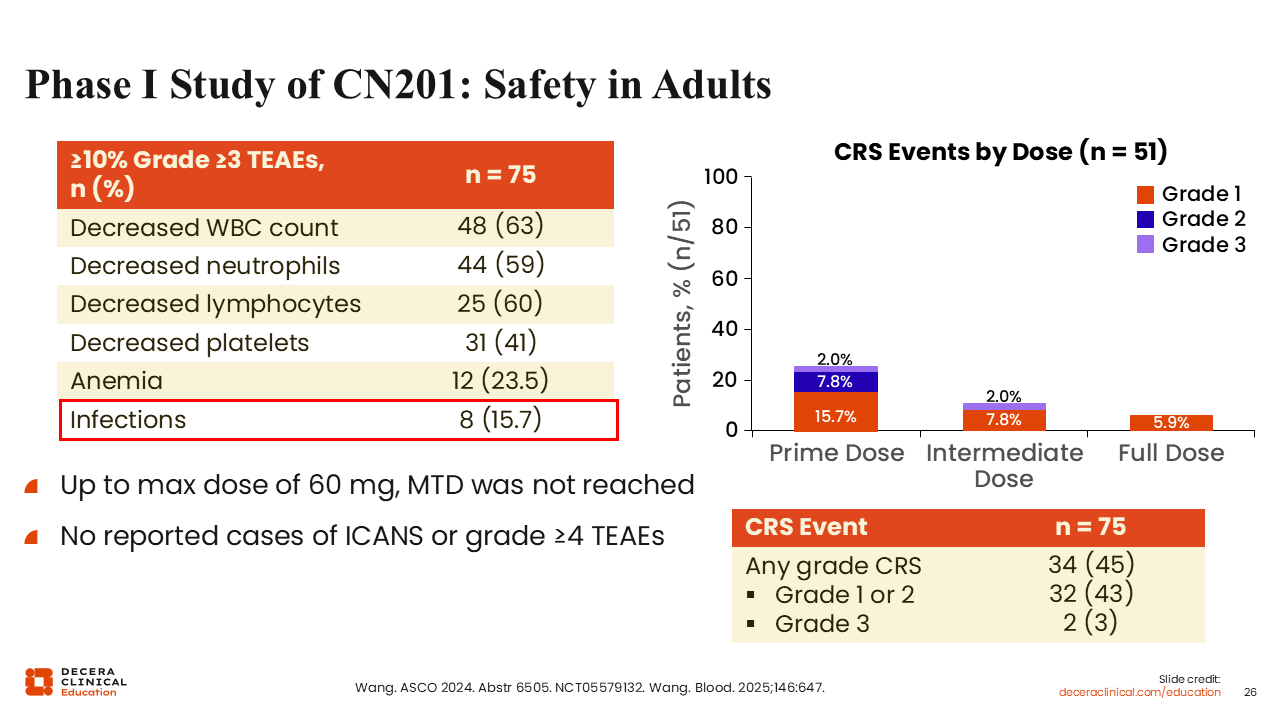
Phase I Study of CN201: Safety in Adults
Overall, CN201 was well tolerated. This table shows grade ≥3 treatment-emergent adverse events occurring in ≥15% of patients. Of importance, there were no reported cases of grade ≥4 ICANS. Most cases of CRS occurred after the primary dosing, and grade 3 CRS events were rare, occurring in only 2 patients (3%). The rate of any-grade CRS was 45%.21
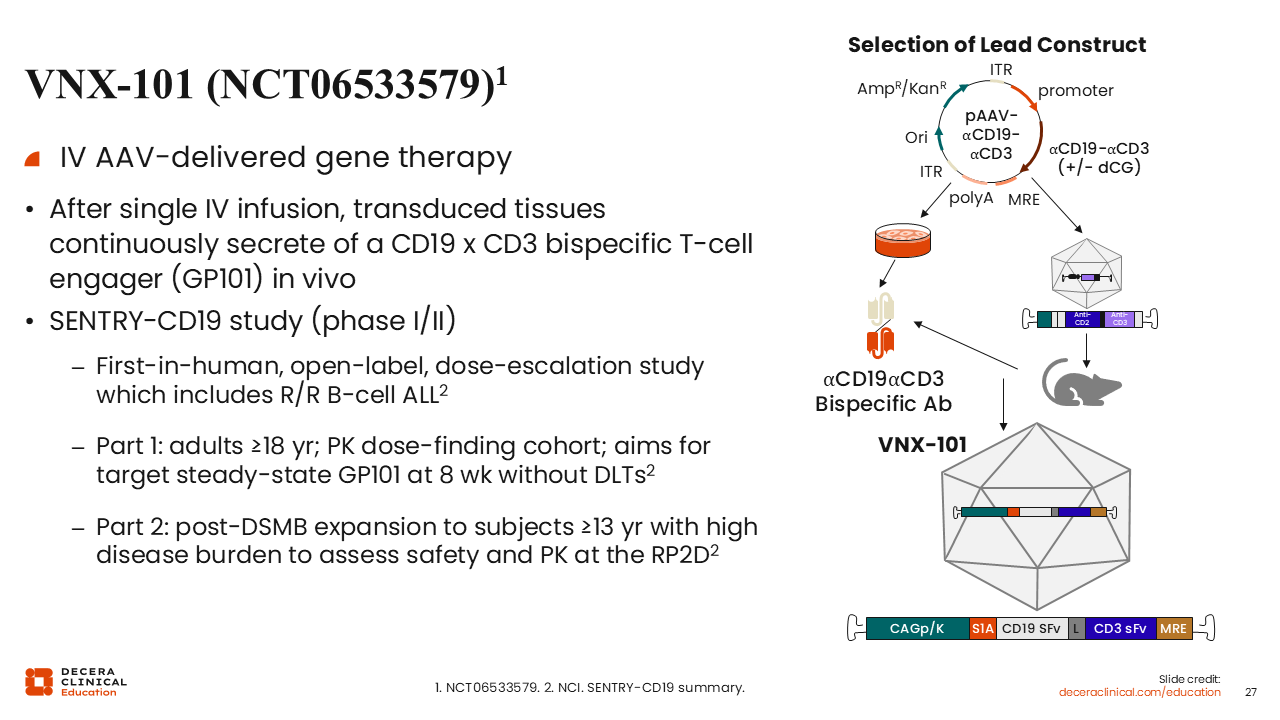
VNX-101 (NCT06533579)
VNX-101 is another promising CD19xCD3 bispecific T-cell engager. This agent is an adeno-associated virus-delivered gene therapy that induces continuous secretion of the T-cell engager from the transduced tissue after a single IV infusion. VNX-101 is currently being tested in SENTRY-CD19, a first-in-human, open-label, dose-escalation phase I/II trial. Eligible candidates for part A include adults aged 18 years of older. This part is aiming to determine the target steady state of serum levels of GP101 at 8 weeks without dose-limiting toxicities. Part B will expand to patients as young as 13 years of age, allowing a high disease burden to assess safety and PK at the recommended phase II dose (NCT06533579).
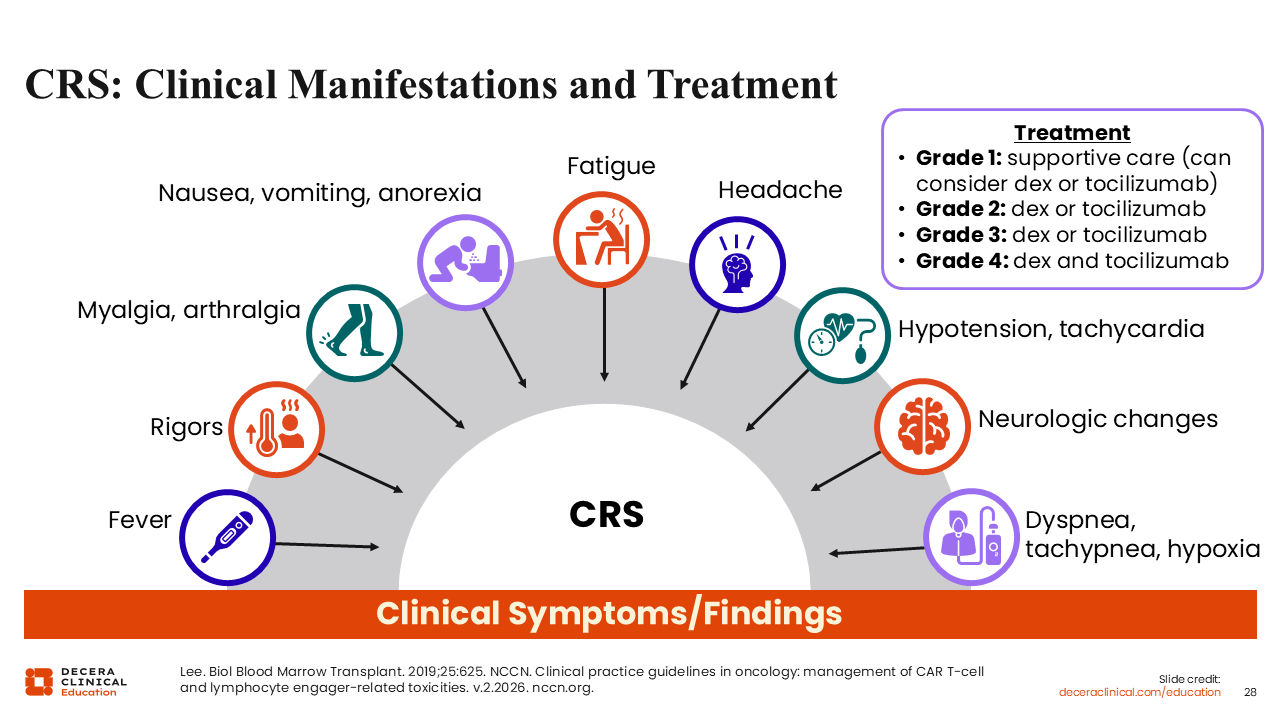
CRS: Clinical Manifestations and Treatment
All T-cell–directed therapies are associated with CRS and ICANS. Early recognition and management of CRS is important for preventing progression and allowing continuous treatment and exposure. CRS usually presents as fever, but it can also present as hypotension, shortness of breath, or decreased oxygenation. CRS is also frequently associated with other symptoms, including arthralgia, myalgia, fatigue, and sometimes headache.
There may be overlap between CRS and ICANS, which can explain some neurologic changes. Depending on the grade of CRS, treatment should be initiated typically with supportive care for grade 1, but tocilizumab with or without dexamethasone can be considered. For grade 4 CRS, both tocilizumab and dexamethasone for neurologic toxicities and ICANS related to immune therapy are recommended. CRS is typically managed with tocilizumab as first-line therapy, with early addition of corticosteroids such as dexamethasone depending on severity and clinical progression. In patients who do not respond to tocilizumab and steroids, escalation to additional immunomodulatory therapies (eg, anakinra) or higher-dose corticosteroids may be considered.22
With an impressive panel of immune-engaging therapies now in practice and on the horizon, we are poised for another decade of substantial progress driving improved outcomes, deeper responses, and a reduced treatment burden through more effective, tolerable, and convenient approaches.